Phytochemical Profile, Antioxidant Activity, and Cytotoxicity Assessment of Tagetes erecta L. Flowers
Abstract
1. Introduction
2. Results
2.1. Identification of Compounds Found in the T. erecta Extract
2.2. Identification of Quercetagitrin
2.3. Antioxidant Activity Assays
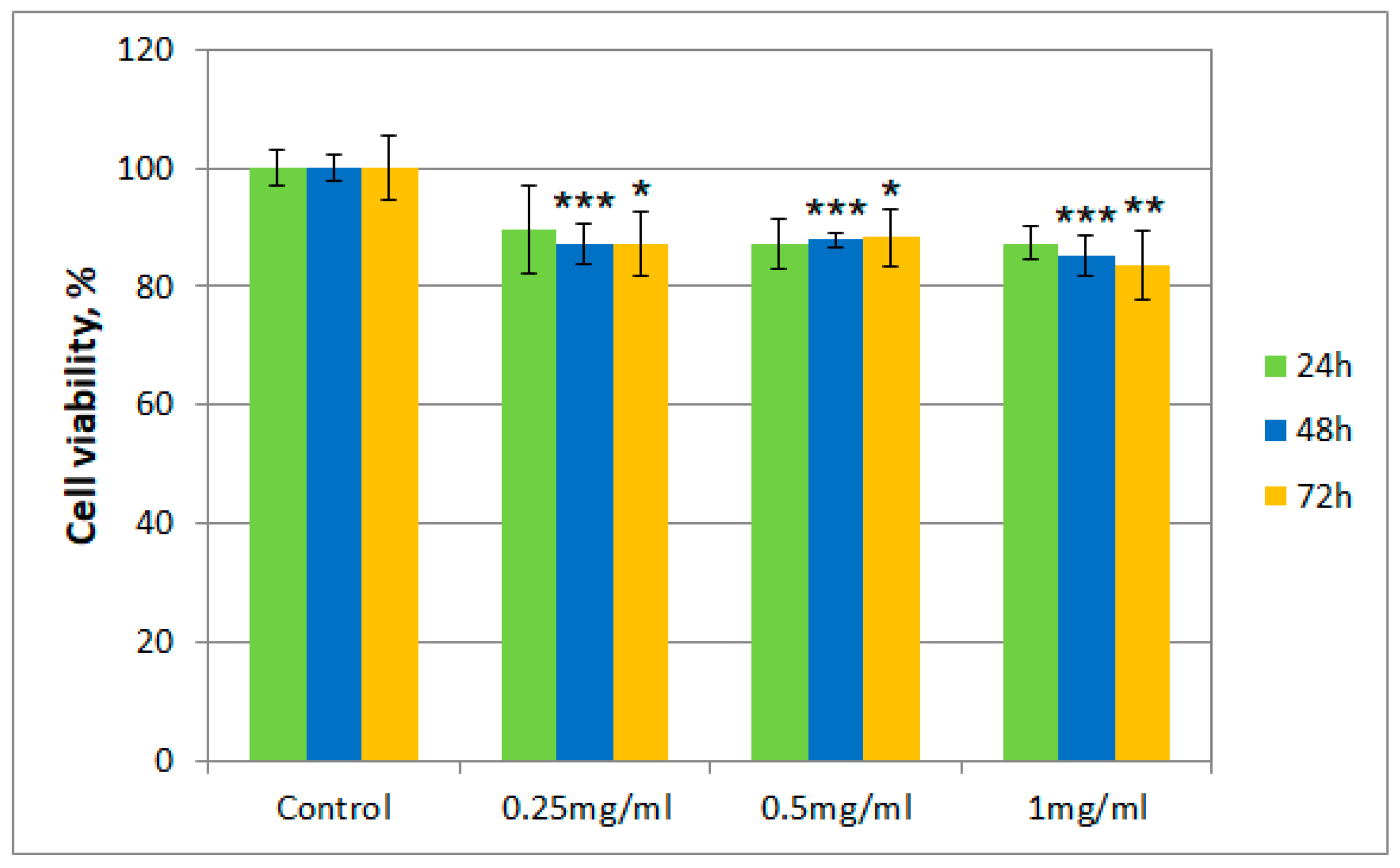
2.4. Cytotoxicity Testing
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagents
4.2. Plant Material and Extract Preparation
4.3. Isolation
4.4. LC-MS and Qualitative Analysis
4.5. NMR Spectroscopy
4.6. Antioxidant Activity Assays
4.6.1. Lipoxygenase Inhibition
4.6.2. Metal Chelation
4.7. Cytotoxicity Testing
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lim, T.K. Tagetes erecta. In Edible Medicinal and Non-Medicinal Plants; Springer International Publishing: Dordrecht, The Netherlands, 2014; pp. 432–447. [Google Scholar]
- Lin, J.-H.; Lee, D.-J.; Chang, J.-S. Lutein production from biomass: Marigold flowers versus microalgae. Bioresour. Technol. 2015, 184, 421–428. [Google Scholar] [CrossRef]
- Jansen, P.C.M.; Cardon, D. Dyes and Tannins—Volume 3 of Plant Resources of Tropical Africa; Backhuys Publisher: Wageningen, The Netherlands, 2005; pp. 157–161. [Google Scholar]
- Royal Horticultural Society. RHS Encyclopedia of Plants and Flowers, 5th ed.; Brickell, C., Ed.; Dorling Kindersley: London, UK, 2010; pp. 712–713. [Google Scholar]
- Coelho, L.C.; Bastos, A.R.R.; Pinho, P.J.; Souza, G.A.; Carvalho, J.G.; Coelho, V.A.T.; Oliveira, L.C.A.; Domingues, R.R.; Faquin, V. Marigold (Tagetes erecta): The potential value in the phytoremediation of chromium. Pedosphere 2017, 27, 559–568. [Google Scholar] [CrossRef]
- Castillo, O.; Dasgupta-Schubert, N.; Alvarado, C.; Zaragoza, E.; Villegas, H. The effect of the symbiosis between Tagetes erecta L. (marigold) and Glomus intraradices in the uptake of Copper(II) and its implications for phytoremediation. New Biotechnol. 2011, 29, 156–164. [Google Scholar] [CrossRef] [PubMed]
- Nakbanpote, W.; Meesungnoen, O.; Prasad, M.; Nakbanpote, W.; Meesungnoen, O.; Prasad, M. Potential of ornamental plants for phytoremediation of heavy metals and income generation. In Bioremediation and Bioeconomy; Elsevier BV: Amsterdam, The Netherlands, 2016; pp. 179–217. [Google Scholar]
- Chitraprabha, K.; Sathyavathi, S. Phytoextraction of chromium from electroplating effluent by Tagetes erecta (L.). Sustain. Environ. Res. 2018, 28, 128–134. [Google Scholar] [CrossRef]
- Eisenhauer, B.; Natoli, S.; Liew, G.; Flood, V.M. Lutein and zeaxanthin—Food sources, bioavailability and dietary variety in age-related macular degeneration protection. Nutrients 2017, 9, 120. [Google Scholar] [CrossRef] [PubMed]
- Evans, W.C. Trease and Evans’ Pharmacognosy, 16th ed.; Saunders Elsevier: Edinburgh, UK, 2009; pp. 474–475. [Google Scholar]
- Bernstein, P.S.; Li, B.; Vachali, P.P.; Gorusupudi, A.; Shyam, R.; Henriksen, B.S.; Nolan, J.M. Lutein, zeaxanthin, and meso-zeaxanthin: The basic and clinical science underlying carotenoid-based nutritional interventions against ocular disease. Prog. Retin. Eye Res. 2016, 50, 34–66. [Google Scholar] [CrossRef] [PubMed]
- Llorent-Martínez, E.J.; Gouveia, S.C.; Castilho, P.C. Analysis of phenolic compounds in leaves from endemic trees from Madeira Island. A contribution to the chemotaxonomy of Laurisilva forest species. Ind. Crop. Prod. 2015, 64, 135–151. [Google Scholar] [CrossRef]
- Fang, N.; Yu, S.; Prior, R.L. LC/MS/MS characterization of phenolic constituents in dried plums. J. Agric. Food Chem. 2002, 50, 3579–3585. [Google Scholar] [CrossRef] [PubMed]
- Dou, J.; Lee, V.S.Y.; Tzen, J.T.C.; Lee, M.-R. Identification and comparison of phenolic compounds in the preparation of oolong tea manufactured by semifermentation and drying processes. J. Agric. Food Chem. 2007, 55, 7462–7468. [Google Scholar] [CrossRef] [PubMed]
- Garcia, C.J.; García-Villalba, R.; Garrido, Y.; Gil, M.I.; Tomás-Barberán, F.A. Untargeted metabolomics approach using UPLC-ESI-QTOF-MS to explore the metabolome of fresh-cut iceberg lettuce. Metabolomics 2016, 12, 138. [Google Scholar] [CrossRef]
- Spínola, V.; Castilho, P.C. Evaluation of Asteraceae herbal extracts in the management of diabetes and obesity. Contribution of caffeoylquinic acids on the inhibition of digestive enzymes activity and formation of advanced glycation end-products (in vitro). Phytochemistry 2017, 143, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Denzel, K.; Schilling, G.; Gross, G.G. Biosynthesis of gallotannins. Enzymatic conversion of 1,6-digalloylglucose to 1,2,6-trigalloylglucose. Planta 1988, 176, 135–137. [Google Scholar] [CrossRef] [PubMed]
- Molina-García, L.; Martínez-Expósito, R.; Córdova, M.L.F.-D.; Llorent-Martínez, E.J. Determination of the phenolic profile and antioxidant activity of leaves and fruits of Spanish Quercus coccifera. J. Chem. 2018, 2018, 2573270. [Google Scholar] [CrossRef]
- Parejo, I.; Jáuregui, O.; Viladomat, F.; Bastida, J.; Codina, C. Characterization of acylated flavonoid-O-glycosides and methoxylated flavonoids from Tagetes maxima by liquid chromatography coupled to electrospray ionization tandem mass spectrometry. Rapid Commun. Mass Spectrom. 2004, 18, 2801–2810. [Google Scholar] [CrossRef]
- Clifford, M.N.; Johnston, K.L.; Knight, S.; Kuhnert, N. Hierarchical scheme for LC-MSn identification of chlorogenic acids. J. Agric. Food Chem. 2003, 51, 2900–2911. [Google Scholar] [CrossRef] [PubMed]
- Navarro-González, I.; González-Barrio, R.; García-Valverde, V.; Bautista-Ortín, A.B.; Periago, M.J. Nutritional composition and antioxidant capacity in edible flowers: Characterisation of phenolic compounds by HPLC-DAD-ESI/MSn. Int. J. Mol. Sci. 2014, 16, 805–822. [Google Scholar] [CrossRef]
- Meyers, K.J.; Swiecki, T.J.; Mitchell, A.E. Understanding the native californian diet: Identification of condensed and hydrolyzable tannins in tanoak acorns (Lithocarpus densiflorus). J. Agric. Food Chem. 2006, 54, 7686–7691. [Google Scholar] [CrossRef]
- Adnyana, I.K.; Awale, S.; Banskota, A.H.; Tran, K.Q.; Tezuka, Y.; Kadota, S. 1-O-Galloyl-6-O-(4-hydroxy-3,5-dimethoxy)benzoyl-β-D-glucose, a new hepatoprotective constituent from Combretum quadrangulare. Planta Medica 2001, 67, 370–371. [Google Scholar] [CrossRef] [PubMed]
- Banoub, J.; Delmas, G.-H.; Joly, N.; MacKenzie, G.; Cachet, N.; Benjelloun-Mlayah, B.; Delmas, M. A critique on the structural analysis of lignins and application of novel tandem mass spectrometric strategies to determine lignin sequencing. J. Mass Spectrom. 2015, 50, 5–48. [Google Scholar] [CrossRef] [PubMed]
- Fracassetti, D.; Costa, C.; Moulay, L.; Tomás-Barberán, F.A. Ellagic acid derivatives, ellagitannins, proanthocyanidins and other phenolics, vitamin C and antioxidant capacity of two powder products from camu-camu fruit (Myrciaria dubia). Food Chem. 2013, 139, 578–588. [Google Scholar] [CrossRef] [PubMed]
- National Center for Biotechnology Information. PubChem Compound Summary for CID 10131194. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/10131194 (accessed on 20 January 2021).
- Jang, G.H.; Kim, H.W.; Lee, M.K.; Jeong, S.Y.; Bak, A.R.; Lee, D.J.; Kim, J.B. Characterization and quantification of flavonoid glycosides in the Prunus genus by UPLC-DAD-QTOF/MS. Saudi J. Biol. Sci. 2018, 25, 1622–1631. [Google Scholar] [CrossRef]
- Alvarado-Sansininea, J.J.; Sánchez-Sánchez, L.; López-Muñoz, H.; Escobar, M.L.; Flores-Guzmán, F.; Tavera-Hernández, R.; Jiménez-Estrada, M. Quercetagetin and patuletin: Antiproliferative, necrotic and apoptotic activity in tumor cell lines. Molecules 2018, 23, 2579. [Google Scholar] [CrossRef]
- Sun, L.; Tao, S.; Zhang, S. Characterization and quantification of polyphenols and triterpenoids in thinned young fruits of ten pear varieties by UPLC-Q TRAP-MS/MS. Molecules 2019, 24, 159. [Google Scholar] [CrossRef] [PubMed]
- Oleszek, M.; Pecio, Ł.; Kozachok, S.; Lachowska-Filipiuk, Ż.; Oszust, K.; Frąc, M. Phytochemicals of apple pomace as prospect bio-fungicide agents against mycotoxigenic fungal species—In vitro experiments. Toxins 2019, 11, 361. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-M.; Ran, X.-K.; Riaz, M.; Yu, M.; Cai, Q.; Dou, D.-Q.; Metwaly, A.M.; Kang, T.-G.; Cai, D.-C. Chemical constituents of stems and leaves of Tagetespatula L. and its fingerprint. Molecules 2019, 24, 3911. [Google Scholar] [CrossRef] [PubMed]
- Yu, B.; Van Ingen, H.; Vivekanandan, S.; Rademacher, C.; Norris, S.E.; Freedberg, D.I. More accurate 1JCH coupling measurement in the presence of 3JHH strong coupling in natural abundance. J. Magn. Reson. 2012, 215, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Castañar, L.; Sistaré, E.; Virgili, A.; Williamson, R.T.; Parella, T. Suppression of phase and amplitude J(HH) modulations in HSQC experiments. Magn. Reson. Chem. 2015, 53, 115–119. [Google Scholar] [CrossRef]
- Żuchowski, J.; Pecio, Ł.; Marciniak, B.; Kontek, R.; Stochmal, A.; Luchowski, J.; Pecio, I. Unusual isovalerylated flavonoids from the fruit of sea buckthorn (Elaeagnus rhamnoides) grown in Sokółka, Poland. Phytochemistry 2019, 163, 178–186. [Google Scholar] [CrossRef]
- Burlec, A.F.; Cioancă, O.; Mircea, C.; Arsene, C.; Tuchiluş, C.; Corciovă, A.; Hăncianu, M. Antioxidant and antimicrobial properties of Chrysanthemum and Tagetes selective extracts. Farmacia 2019, 67, 405–410. [Google Scholar] [CrossRef]
- Xu, L.-W.; Wang, G.-Y.; Shi, Y.-P. Chemical constituents from Tagetes erecta flowers. Chem. Nat. Compd. 2011, 47, 281–283. [Google Scholar] [CrossRef]
- Xu, L.; Chen, J.; Qi, H.; Shi, Y. Phytochemicals and their biological activities of plants in Tagetes L. Chinese Herb. Med. 2012, 4, 103–117. [Google Scholar] [CrossRef]
- Meurer, M.C.; Mees, M.; Mariano, L.N.B.; Boeing, T.; Somensi, L.B.; Mariott, M.; Silva, R.D.C.M.V.D.A.F.D.; Dos Santos, A.C.; Longo, B.; França, T.C.S.; et al. Hydroalcoholic extract of Tagetes erecta L. flowers, rich in the carotenoid lutein, attenuates inflammatory cytokine secretion and improves the oxidative stress in an animal model of ulcerative colitis. Nutr. Res. 2019, 66, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Moliner, C.; Barros, L.; Dias, M.I.; López, V.; Langa, E.; Ferreira, I.C.; Gómez-Rincón, C. Edible flowers of Tagetes erecta L. as functional ingredients: Phenolic composition, antioxidant and protective effects on Caenorhabditis elegans. Nutrients 2018, 10, 2002. [Google Scholar] [CrossRef]
- Cherrak, S.A.; Mokhtari-Soulimane, N.; Berroukeche, F.; Bensenane, B.; Cherbonnel, A.; Merzouk, H.; Elhabiri, M. In vitro antioxidant versus metal ion chelating properties of flavonoids: A structure-activity investigation. PLoS ONE 2016, 11, e0165575. [Google Scholar] [CrossRef] [PubMed]
- Flora, S.J.S. Structural, chemical and biological aspects of antioxidants for strategies against metal and metalloid exposure. Oxidative Med. Cell. Longev. 2009, 2, 191–206. [Google Scholar] [CrossRef] [PubMed]
- Iancu, C.; Cioancă, O.; Mircea, C.; Mocanu, M.; Hăncianu, M. Pelargonium sp.: Characterization of the polyphenols and their biological potential. Farmacia 2016, 64, 333–338. [Google Scholar]
- Leopoldini, M.; Russo, N.; Chiodo, A.S.; Toscano, M. Iron chelation by the powerful antioxidant flavonoid quercetin. J. Agric. Food Chem. 2006, 54, 6343–6351. [Google Scholar] [CrossRef]
- Young, A.J.; Lowe, G.L. Carotenoids—Antioxidant properties. Antioxidants 2018, 7, 28. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.-Y.; Thakur, K.; Wei, C.-K.; Wang, H.; Zhang, J.-G.; Wei, Z.-J. Evaluation of inhibitory activity of natural plant polyphenols on Soybean lipoxygenase by UFLC-mass spectrometry. South. Afr. J. Bot. 2019, 120, 179–185. [Google Scholar] [CrossRef]
- Corciovă, A.; Mircea, C.; Burlec, A.-F.; Cioancă, O.; Tuchiluş, C.; Fifere, A.; Lungoci, A.-L.; Marangoci, N.; Hăncianu, M. Antioxidant, antimicrobial and photocatalytic activities of silver nanoparticles obtained by bee propolis extract assisted biosynthesis. Farmacia 2019, 67, 482–489. [Google Scholar] [CrossRef]
- Lapenna, D.; Ciofani, G.; Pierdomenico, S.D.; Giamberardino, M.A.; Cuccurullo, F. Dihydrolipoic acid inhibits 15-lipoxygenase-dependent lipid peroxidation. Free Radic. Biol. Med. 2003, 35, 1203–1209. [Google Scholar] [CrossRef]
- Burlec, A.F.; Pecio, Ł.; Mircea, C.; Cioancă, O.; Corciovă, A.; Nicolescu, A.; Oleszek, W.; Hăncianu, M. Chemical profile and antioxidant activity of Zinnia elegans Jacq. fractions. Molecules 2019, 24, 2934. [Google Scholar] [CrossRef] [PubMed]
- Malterud, K.E.; Rydland, K.M. Inhibitors of 15-lipoxygenase from orange peel. J. Agric. Food Chem. 2000, 48, 5576–5580. [Google Scholar] [CrossRef] [PubMed]
- Venditti, E.; Bacchetti, T.; Tiano, L.; Carloni, P.; Greci, L.; Damiani, E. Hot vs. cold water steeping of different teas: Do they affect antioxidant activity? Food Chem. 2010, 119, 1597–1604. [Google Scholar] [CrossRef]
- Dinis, T.; Madeira, V.; Almeida, L. Action of phenolic derivatives (Acetaminophen, salicylate, and 5-aminosalicylate) as inhibitors of membrane lipid peroxidation and as peroxyl radical scavengers. Arch. Biochem. Biophys. 1994, 315, 161–169. [Google Scholar] [CrossRef]
- Mahajan, S.D.; Law, W.-C.; Aalinkeel, R.; Reynolds, J.; Nair, B.B.; Yong, K.-T.; Roy, I.; Prasad, P.N.; Schwartz, S.A. Nanoparticle-mediated targeted delivery of antiretrovirals to the brain. In Methods in Enzymology; Elsevier BV: San Diego, CA, USA, 2012; Volume 509, pp. 41–60. [Google Scholar]
- Gutiérrez, L.; Stepien, G.; Pérez-Hernández, M.; Pardo, J.; Grazú, V.; De La Fuente, J. Nanotechnology in drug discovery and development. In Comprehensive Medicinal Chemistry III; Elsevier BV: Amsterdam, The Netherlands, 2017; pp. 264–295. [Google Scholar]
- Duceac, I.A.; Verestiuc, L.; Dimitriu, C.D.; Maier, V.; Coseri, S. Design and preparation of new multifunctional hydrogels based on chitosan/acrylic polymers for drug delivery and wound dressing applications. Polymers 2020, 12, 1473. [Google Scholar] [CrossRef]
- International Organization for Standardization. ISO 10993-5 Biological Evaluation of Medical Devices—Part 5: Tests for In Vitro Cytotoxicity; ISO: Geneva, Switzerland, 2009. [Google Scholar]
- Vunjak-Novakovic, G.; Freshney, R.I. Culture of Cells for Tissue Engineering; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005. [Google Scholar]
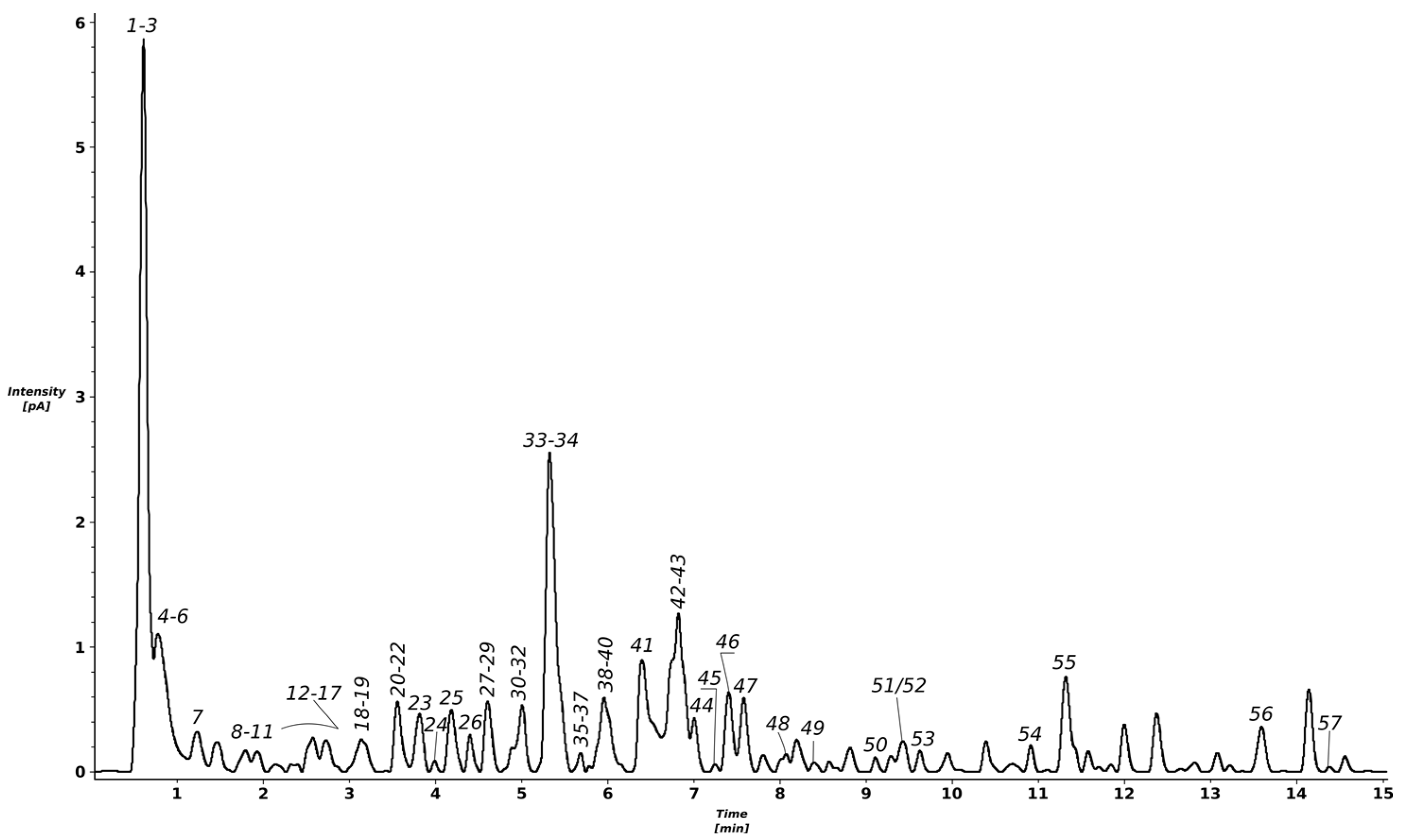

| No. | Compound Name | RT * (min) | Formula | Error (ppm) ** | mσ *** | Observed [M-H]- | Major Fragments (%) | Reference |
|---|---|---|---|---|---|---|---|---|
| 1. | Quinic acid | 0.63 | C7H12O6 | 0.1 | 1.2 | 191.0561 | - | [12] |
| 2. | Quinic acid hexoside | 0.63 | C13H22O11 | 0.5 | 9.4 | 353.1088 | 173.0455 (100), 191.0560 (5) | - |
| 3. | Dihexoside | 0.63 | C12H22O11 | 0.5 | 7.0 | 341.1088 | - | - |
| 4. | Galloyl hexoside | 0.76 | C13H16O10 | 0.5 | 16.0 | 331.0669 | 169.0137 (100), 211.0247 (85), 271.0457 (35), 125.0245 (19), 241.0356 (7) | [13] |
| 5. | Shikimic acid hexoside | 0.76 | C13H20O10 | 0.4 | 15.3 | 335.0983 | 215.0522 (68) | - |
| 6. | Theogallin | 0.79 | C14H16O10 | 0.3 | 9.6 | 343.0670 | 191.0560 (100) | [14] |
| 7. | Phenylalanine | 1.25 | C9H11NO2 | 3.6 | 6.3 | 164.0711 | - | [15] |
| 8. | Syringic acid-hexoside I | 1.76 | C15H20O10 | 0.4 | 8.4 | 359.0982 | 197.0457 (100), 182.0067 (91), 138.0308 (48), 153.0546 (27) | [16] |
| 9. | Methyl-gallic acid | 1.81 | C8H8O5 | −0.8 | 45.6 | 183.0300 | 168.0062 (100), 124.0157 (97) | - |
| 10. | Digalloyl-hexoside I | 1.81 | C20H20O14 | −0.3 | 79.7 | 483.0782 | 271.0457 (100), 211.0245 (87), 169.0128 (75), 313.0572 (15), 423.0574 (10), 124.0191 (9) | [17] |
| 11. | Tryptophan | 1.94 | C11H12N2O2 | 0.3 | 8.4 | 203.0825 | 116.0493 (100), 186.0526 (16) | [15] |
| 12. | Digalloyl-hexoside II | 2.20 | C20H20O14 | −0.3 | 15.0 | 483.0782 | 271.0457 (100), 211.0249 (93), 169.0139 (79), 347.0620 (36), 423.0571 (23), 313.0570 (17) | [18] |
| 13. | Syringic acid-hexoside II | 2.34 | C15H20O10 | −0.7 | 17.3 | 359.0986 | 197.0455 (100), 182.0221 (91), 138.0308 (48), 153.0546 (27) | [19] |
| 14. | Digalloyl-dihexoside | 2.44 | C26H30O19 | −0.4 | 26.0 | 645.1311 | 313.0569 (100), 271.0448 (82), 169.0139 (65), 493.1200 (57), 475.1103 (34) | - |
| 15. | Digalloyl-hexoside III | 2.60 | C20H20O14 | −0.6 | 1.5 | 483.0783 | 271.0460 (100), 211.0249 (51), 169.0140 (43), 313.0567 (27), 125.0237 (8) | [18] |
| 16. | Galloyl-syringic acid-hexoside I | 2.62 | C22H24O14 | 0.1 | 21.5 | 511.1093 | 169.0142 (100), 125.0238 (31), 313.0557 (13), 197.0451 (13), 182.0246 (4), 271.0461 (4) | - |
| 17. | Chlorogenic acid (5-CQA) | 2.74 | C16H18O9 | −0.3 | 7.6 | 353.0879 | 191.0562 (100) | [20] |
| 18. | Galloyl-syringic acid-hexoside II | 3.11 | C22H24O14 | 0.0 | 6.4 | 511.1093 | 169.0140 (100), 125.0237 (29), 197.0455 (21), 211.0245 (15), 271.0464 (13), 313.0566 (12) | - |
| 19. | Trigalloyl-dihexoside | 3.15 | C33H34O23 | 2.2 | 21.4 | 797.1401 | 169.0137 (100), 313.0564 (49), 493.1206 (40), 475.1091 (32), 271.0461 (24) | [21] |
| 20. | Syringic acid | 3.57 | C9H10O5 | −1.5 | 4.4 | 197.0459 | 182.0221 (100), 166.9983 (37), 123.0071 (11) | [19] |
| 21. | Trigalloyl-hexoside I | 3.57 | C27H24O8 | −1.1 | 22.0 | 635.0897 | 169.0147 (100), 271.0460 (32), 211.0247 (26), 313.0572 (25), 125.0242 (18), 423.0579 (12) | [22] |
| 22. | Galloyl-syringic acid-hexoside III | 3.65 | C22H24O14 | −1.1 | 20.6 | 511.1099 | 169.0142(100), 197.0459 (98), 183.0298 (41), 125.0237 (37), 313.0542 (20) | [23] |
| 23. | Syringic acid-(dihydroxydimethoxybenzoic acid)-hexoside I | 3.83 | C24H28O15 | 0.4 | 3.6 | 555.1353 | 197.0457 (100), 182.0224 (31), 153.0549 (23), 121.0283 (14), 138.0308 (7), 166.9987 (6) | [19] |
| 24. | Sinapoyl alcohol | 4.00 | C11H14O4 | 0.4 | 10.0 | 209.0818 | 149.0585 (100) | [24] |
| 25. | Syringic acid-(dihydroxydimethoxybenzoic acid)-hexoside II | 4.19 | C24H28O15 | −0.1 | 0.9 | 555.1356 | 197.0458 (100), 153.0548 (30), 182.0222 (29), 121.0286 (17), 138.0308 (9), 166.9985 (4) | [19] |
| 26. | Ellagic acid-hexoside I | 4.41 | C20H16O13 | −1.1 | 3.6 | 463.0523 | 300.9993 (100), 271.9965 (3) | [25] |
| 27. | Quercetagetin−7-O-(Ac-Pen-Hex)-3-O-hexoside I | 4.59 | C34H40O23 | 0.3 | 15.0 | 815.1885 | 317.0306 (100) | [19] |
| 28. | Galloyl-eudesmic acid-hexoside I | 4.62 | C23H26O14 | −0.3 | 6.9 | 525.1251 | 183.0304 (100), 168.0064 (27), 197.0459 (17), 139.0391 (15), 312.0492 (10), 124.0161 (10) | [19] |
| 29. | Ellagic acid-hexoside II | 4.68 | C20H16O13 | −1.2 | 75.4 | 463.0524 | 299.9915 (100) | [25] |
| 30. | N-Malonylphenylalanine | 4.97 | C12H13NO5 | −1.5 | 10.1 | 250.0725 | 164.0716 (100), 206.0814 (47), 147.0443 (21) | [26] |
| 31. | Quercetagetin-3-O-hexoside | 4.97 | C21H20O13 | −1.3 | 18.1 | 479.0837 | 316.0236 (100), 287.0210 (20), 271.0255 (11), 165.9909 (7), 243.0323 (4) | [19] |
| 32. | Galloyl-eudesmic acid-hexoside II | 5.02 | C23H26O14 | −0.9 | 4.1 | 525.1254 | 183.0303 (100), 168.0061 (34), 197.0459 (31), 225.0406 (15), 139.0387 (13), 124.0162 (10) | [19] |
| 33. | Trigalloyl-hexoside II | 5.26 | C27H24O8 | −0.7 | 27.5 | 635.0895 | 313.0577 (100), 169.0142 (91), 271.0461 (31), 295.0466 (19), 465.0641 (18), 211.0266 (18) | [22] |
| 34. | Quercetagitrin (Quercetagetin-7-O-β-glucopyranoside) | 5.33 | C21H20O13 | −0.9 | 1.5 | 479.0836 | 317.0304 (100) | [19] |
| 35. | Syringic acid-(dihydroxydimethoxybenzoic acid)-hexoside III | 5.68 | C24H28O15 | −1.1 | 25.2 | 555.1361 | 197.0452 (100), 313.0927 (64), 182.0242 (45), 153.0549 (40), 138.0315 (22), 223.0616 (20) | - |
| 36. | Quercetagetin-7-O-dihexoside | 5.69 | C27H30O18 | −0.5 | 6.8 | 641.1363 | 317.0303 (100) | [19] |
| 37. | Quercetagetin-7-O-(Ac-Pen-Hex)-3-O-hexoside II | 5.71 | C34H40O23 | 0.0 | 36.5 | 815.1887 | 317.0309 (100) | [19] |
| 38. | Quercetagetin-7-O-(galloyl-hexoside) | 5.96 | C28H24O17 | 0.4 | 8.9 | 631.0938 | 317.0307 (100) | [19] |
| 39. | Ellagic acid | 5.96 | C14H6O8 | −2.2 | 12.2 | 300.9997 | 300.9996 (100), 283.9964 (5), 229.0145 (5), 257.0097 (3), 245.0099 (3) | [25] |
| 40. | Quercetagetin-3-O-hexoside-7-O-(galloyl-hexoside) | 6.03 | C34H34O22 | −0.1 | 9.1 | 793.1470 | 317.0309 (100), 479.0829 (3), 631.0950 (1) | [19] |
| 41. | Di-syringic acid hexoside I | 6.41 | C24H28O14 | −0.7 | 3.4 | 539.1411 | 197.0460 (100), 182.0227 (71), 153.0549 (17), 166.9988 (12), 326.0645 (11), 239.0565 (9) | [19] |
| 42. | Di-syringic acid hexoside II | 6.74 | C24H28O14 | −1.9 | 11.7 | 539.1411 | 197.0458 (100), 182.0226 (77), 326.0644 (17), 153.0550 (14), 166.9987 (13), 239.0565 (11) | [19] |
| 43. | Quercetin 3-O-hexoside | 6.83 | C21H20O12 | −1.4 | 0.4 | 463.0888 | 301.0359 (100) | [19] |
| 44. | Syringic acid-(hydroxytrimethoxybenzoic acid)-hexoside | 7.01 | C25H30O15 | −1.6 | 5.0 | 569.1521 | 197.0458 (100), 182.0226 (30), 153.0550 (19), 269.0672 (14), 254.0438 (12), 121.0290 (10) | - |
| 45. | Kaempferol 3-O-hexoside I | 7.26 | C21H20O11 | −2.5 | 10.3 | 447.0944 | 285.0404 (100) | [27] |
| 46. | Quercetagetin | 7.41 | C15H10O8 | −2.6 | 4.2 | 317.0311 | 317.0310 (100), 271.0258 (10), 166.9991 (7), 243.0311 (5), 261.0410 (5), 299.0205 (5) | [28] |
| 47. | Patulitrin | 7.58 | C22H22O13 | −1.5 | 4.9 | 493.0995 | 315.0169 (100), 330.0406 (58), 287.0211 (49), 271.0253 (13) | [19] |
| 48. | 3,5 or 4,5 di-CQA | 8.08 | C25H24O12 | −2.5 | 10.7 | 515.1208 | 191.0566 (100), 179.0352 (9), 173.0463 (2), 161.0241 (2) | [20] |
| 49. | Kaempferol 3-O-hexoside II | 8.40 | C21H20O11 | −4.2 | 23.2 | 447.0951 | 285.0408 (100), 257.0460 (35), 151.0029 (9) | [29] |
| 50. | Isorhamnetin 3-O-hexoside | 9.12 | C22H22O12 | −1.4 | 5.7 | 477.1045 | 299.0207 (100), 314.0444 (40), 271.0263 (30) | [19] |
| 51. | 8-Hydroxyquercetagetin | 9.43 | C15H10O9 | −3.6 | 8.7 | 333.0264 | 165.9915 (100), 193.9862 (52), 243.0306 (49), 271.0254 (34), 287.0205 (31), 259.0250 (13) | [19] |
| 52. | Quercetin | 9.43 | C15H10O7 | −3.2 | 6.4 | 301.0363 | 301.0363 (100), 245.0465 (7), 255.0309 (5), 283.0270 (2) | [30] |
| 53. | 2Hβ,3-dihydro-euparin-14-O-β-D-glucoside | 9.63 | C19H24O9 | −2.7 | 20.6 | 395.1358 | 233.0826 (100), 215.0720 (34), 217.0875 (25), 205.0880 (21), 175.0414 (9) | [31] |
| 54. | Kaempferol | 10.91 | C15H10O6 | −0.7 | 4.1 | 285.0407 | - | [19] |
| 55. | Patuletin | 11.32 | C16H12O8 | −0.8 | 5.7 | 331.0462 | 316.0228 (100), 165.9912 (14), 287.0214 (8), 271.0258 (7) | [19] |
| 56. | Isorhamnetin | 13.59 | C16H12O7 | 1.0 | 6.7 | 315.0507 | 271.0247 (100), 300.0277 (96), 243.0298 (70), 255.0299 (21), 165.9907 (12) | [19] |
| 57. | Axillarin | 14.38 | C17H14O8 | 1.7 | 2.2 | 345.0610 | 287.0197 (100), 302.0428 (32), 259.0245 (25), 330.0383 (24) | [19] |
| Position | δH (J in Hz) | δC, Type |
|---|---|---|
| 2 | 147.5, C | |
| 3 | 135.6, C | |
| 4 | 176.1, C | |
| 5 | 145.3, C | |
| 6 | 129.6, C | |
| 7 | 151.5, C | |
| 8 | 6.92 s | 93.6, CH |
| 9 | 148.1, C | |
| 10 | 105.1, C | |
| 1′ | 122.0, C | |
| 2′ | 7.70 d (2.2) | 115.4, CH |
| 3′ | 145.0, C | |
| 4′ | 147.8, C | |
| 5′ | 6.89 d (8.5) | 115.5, CH |
| 6′ | 7.53 dd (8.5, 2.2) | 119.9, CH |
| 3-OH | 9.30 s | |
| 5-OH | 12.21 s | |
| 6-OH | 8.39 s | |
| 3′-OH | 9.24 s | |
| 4′-OH | 8.59 s | |
| β-Glcp | ||
| 1 | 5.01 d (7.3) | 101.0, CH |
| 2 | 3.37 dd (9.2, 7.3) | 73.2, CH |
| 3 | 3.33 t (8.7) | 75.8, CH |
| 4 | 3.21 t (8.7) | 69.7, CH |
| 5 | 3.47 ddd (7.7, 6.2, 1.7) | 77.3, CH |
| 6a | 3.75 dd (11.0, 1.7) | 60.6, CH2 |
| 6b | 3.50 dd (11.0, 6.2) | |
| 2-OH | 5.38 brd (2.9) | |
| 3-OH | 5.09 brs | |
| 4-OH | 5.06 d (5.1) | |
| 6-OH | 4.64 t (4.6) | |
| Lipoxygenase Inhibition | Iron-Chelating Activity | |
|---|---|---|
| Sample | IC50 (μg/mL Final Solution) | EC50 (mg/mL Final Solution) |
| Total extract | 25.85 ± 0.67 * | 0.390 ± 0.001 |
| Fr 4 | 16.49 ± 0.19 | 0.529 ± 0.001 |
| Quercetin | 17.45 ± 0.33 * | 0.417 ± 0.011 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Burlec, A.F.; Pecio, Ł.; Kozachok, S.; Mircea, C.; Corciovă, A.; Vereștiuc, L.; Cioancă, O.; Oleszek, W.; Hăncianu, M. Phytochemical Profile, Antioxidant Activity, and Cytotoxicity Assessment of Tagetes erecta L. Flowers. Molecules 2021, 26, 1201. https://doi.org/10.3390/molecules26051201
Burlec AF, Pecio Ł, Kozachok S, Mircea C, Corciovă A, Vereștiuc L, Cioancă O, Oleszek W, Hăncianu M. Phytochemical Profile, Antioxidant Activity, and Cytotoxicity Assessment of Tagetes erecta L. Flowers. Molecules. 2021; 26(5):1201. https://doi.org/10.3390/molecules26051201
Chicago/Turabian StyleBurlec, Ana Flavia, Łukasz Pecio, Solomiia Kozachok, Cornelia Mircea, Andreia Corciovă, Liliana Vereștiuc, Oana Cioancă, Wiesław Oleszek, and Monica Hăncianu. 2021. "Phytochemical Profile, Antioxidant Activity, and Cytotoxicity Assessment of Tagetes erecta L. Flowers" Molecules 26, no. 5: 1201. https://doi.org/10.3390/molecules26051201
APA StyleBurlec, A. F., Pecio, Ł., Kozachok, S., Mircea, C., Corciovă, A., Vereștiuc, L., Cioancă, O., Oleszek, W., & Hăncianu, M. (2021). Phytochemical Profile, Antioxidant Activity, and Cytotoxicity Assessment of Tagetes erecta L. Flowers. Molecules, 26(5), 1201. https://doi.org/10.3390/molecules26051201












