Chemical Composition and Cytotoxic Activity of the Fractionated Trunk Bark Essential Oil from Tetraclinis articulata (Vahl) Mast. Growing in Tunisia
Abstract
1. Introduction
2. Results and Discussion
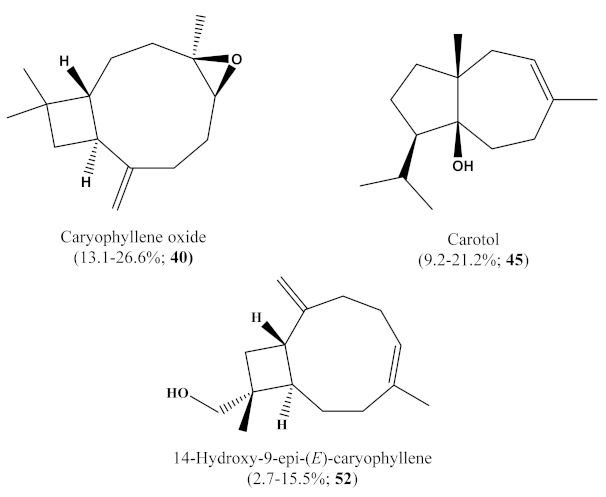
2.1. Chemical Composition of the Trunk Bark Essential Oil (HEE) and Its Fractions (E.1–E.5)
2.2. Cytotoxic Activity
3. Material and Methods
3.1. Plant Material
3.2. Isolation and Fractionation of the Essential Oil
3.3. Gas Chromatography–Mass Spectrometry Analyses and Peak Identification
3.4. Cell Lines
3.5. Determination of Cell Viability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Swamy, M.K.; Sinniah, U.R. A comprehensive review on the phytochemical constituents and pharmacological activities of Pogostemon cablin Benth.: An aromatic medicinal plant of industrial importance. Molecules 2015, 20, 8521–8547. [Google Scholar] [CrossRef]
- El Ayeb-Zakhama, A.; Ben Salem, S.; Sakka-Rouis, L.; Flamini, G.; Ben Jannet, H.; Harzallah-Skhiri, F. Chemical Composition and Phytotoxic Effects of Essential Oils Obtained from Ailanthus altissima (Mill.) Swingle Cultivated in Tunisia. Chem. Biodivers. 2014, 11, 1216–1227. [Google Scholar] [CrossRef]
- Woguem, V.; Fogang, H.P.; Maggi, F.; Tapondjou, L.A.; Womeni, H.M.; Quassinti, L.; Bramucci, M.; Vitali, L.A.; Petrelli, D.; Lupidi, G.; et al. Volatile oil from striped African pepper (Xylopia parviflora, Annonaceae) possesses notable chemopreventive, anti-inflammatory and antimicrobial potential. Food Chem. 2014, 149, 183–189. [Google Scholar] [CrossRef]
- Zardi-Bergaoui, A.; Ben Nejma, A.; Harzallah-Skhiri, F.; Flamini, G.; Ascrizzi, R.; Ben Jannet, H. Chemical Composition and Biological Studies of the Essantial Oil from Aerial Parts of Beta vulgaris subsp. maritima (L.) ARCANG. Growing in Tunisia. Chem. Biodivers. 2017, 14, e1700234. [Google Scholar]
- Baccari, W.; Znati, M.; Zardi-Bergaoui, A.; Chaieb, I.; Flamini, G.; Ascrizzi, R.; Ben Jannet, H. Composition and insecticide potential against Tribolium castaneum of the fractionated essential oil from the flowers of the Tunisian endemic plant Ferula tunetana Pomel ex Batt. Ind. Crops Prod. 2020, 143, 111888. [Google Scholar] [CrossRef]
- Zardi-Bergaoui, A.; Jelizi, S.; Flamini, G.; Ascrizzi, R.; Ben Jannet, H. Comparative study of the chemical composition and bioactivities of essential oils of fresh and dry seeds from Myoporum insulare R. Br. Ind. Crops Prod. 2018, 111, 232–237. [Google Scholar] [CrossRef]
- Wińska, K.; Mączka, W.; Łyczko, J.; Grabarczyk, M.; Czubaszek, A.; Szumny, A. Essential Oils as Antimicrobial Agents—Myth or Real Alternative? Molecules 2019, 24, 2130. [Google Scholar] [CrossRef]
- Rabib, H.; Elagdi, C.; Hsaine, M.; Fougrach, H.; Koussa, T.; Badri, W. Antioxidant and Antibacterial Activities of the Essential Oil of Moroccan Tetraclinis articulata (Vahl) Masters. Biochem. Res. Int. 2020, 2020, 1–6. [Google Scholar] [CrossRef]
- Le, N.T.; Donadu, M.G.; Ho, D.V.; Doan, T.Q.; Le, A.T.; Raal, A.; Usai, D.; Sanna, G.; Marchetti, M.; Usai, M.; et al. Biological activities of essential oil extracted from leaves of Atalantia Sessiflora Guillauminin Vietnam. J. Infect. Dev. Ctries. 2020, 14, 1054–1064. [Google Scholar] [CrossRef]
- Bellakhdar, J.; Honda, G.; Miki, W. Herb–Drugs and Herbalists in the Maghrib; Institute of the Study of Languages and Cultures of Asia and Africa: Tokyo, Japan, 1982. [Google Scholar]
- Ait Igri, M.; Holeman, M.; Ilidrissi, A.; Berrada, M. Contribution to the chemical study of branches and wood essential oils of Tetraclinis articulata (Vahl) Masters. Plant Med. Phytother. 1990, 24, 36–43. [Google Scholar]
- Barrero, A.F.; Herrador, M.M.; Arteaga, P.; Quilez, J.; Akssira, M.; Mellouki, F.; Akkad, S. Chemical Composition of the Essential Oils of Leaves and Wood of Tetraclinis articulata (Vahl) Masters. J. Essent. Oil. Res. 2005, 17, 166–168. [Google Scholar] [CrossRef]
- Bourkhiss, M.; Hnach, M.; Lakhlifi, T.; Bourkhiss, B.; Ouhssine, M.; Satrani, B. Production et Caractérisation de l’huile essntielle de la scuire de bois de Tetraclinis articulata (Vahl) Masters. Bull. Soc. R. Sci. Liège 2010, 79, 4–11. [Google Scholar]
- Pottier-Alapetite, G. Flore de la Tunisie: Angiospermes-Dicotylédones, Apétales Dialypétales; Publications Scientifiques Tunisiennes: Tunis, Tunisia, 1981; pp. 36–37. [Google Scholar]
- Abi-Ayad, F.Z.; Abi-Ayad, M.; Lazzouni, H.A.; Rebiahi, S.A.; Bessiere. Antibacterial activity of essential oil extracted from leaves of Tetraclinis articulata (Vahl) Masters from Algeria flora. J. Microbiol. Biotechnol. Res. 2011, 1, 1–6. [Google Scholar]
- Tekaya-Karoui, A.; Boughalle, N.; Hammami, S.; Ben Jannet, H.; Mighri, Z. Chemical composition and antifungal activity of volatile components from woody terminal branches and roots of Tetraclinis articulata (Vahl) Masters growing in Tunisia. Afr. J. Plant Sci. 2011, 5, 115–122. [Google Scholar]
- Buhagiar, J.A.; Podesta, M.T.; Wilson, A.P.; Micallef, M.J.; Ali, S. The induction of apoptosis in human melanoma, breast and ovarian cancer cell lines using an essential oil extract from the conifer Tetraclinis articulata. Anticancer Res. 1999, 19, 5435–5443. [Google Scholar]
- Djouahri, A.; Boudarene, L. Antioxidant and anti-inflammatory activity of methanolic, chloroform and ethyl acetate extracts of leaves Tetraclinis articulata (Vahl) Masters Algerian. Int. J. Res. Phytochem. Pharm. 2012, 2, 7–11. [Google Scholar]
- Chikhoune, A.; Hazzit, M.; Kerbouche, L.; Baaliouamer, A.; Aissat, K. Tetraclinis articulata (Vahl.) Masters essentials: Chemical composition and biological activities. J. Essent. Oil. Res. 2013, 25, 300–307. [Google Scholar] [CrossRef]
- Ghnaya, A.B.; Amri, I.; Hanana, M.; Gargouri, S.; Jamoussi, B.; Romane, A.; Hamrouni, L. Tetraclinis articulata (Vahl.) Masters essential oil from Tunisia: Chemical characterization and herbicidal and antifungal activities assessment. Ind. Crops Prod 2016, 83, 113–117. [Google Scholar] [CrossRef]
- Rguez, S.; Ben Slimene, I.; Abid, G.; Hammemi, M.; Kefi, A.; Elkahoui, S.; Ksouri, R.; Hamrouni Sellami, I.; Djebali, N. Tetraclinis articulata essential oil reduces Botrytis cinerea infections on tomato. Sci. Hortic. 2020, 266, 109291. [Google Scholar] [CrossRef]
- Tekaya-Karoui, A.; Ben Jannet, H.; Mighri, Z. Essential oil composition of terminal branches, cones and roots of Tetraclinis articulata from Tunisia. Pak. J. Biol. 2007, 10, 2495–2499. [Google Scholar] [CrossRef]
- Larabi, F.; Benhassaini, H.; Bennaoum, Z. Phytochemical Screening of Non Woody Terminal Branches of Tetraclinis articulata from the North-western Region of Algeria. TEOP 2016, 19, 1816–1820. [Google Scholar]
- Buhagiar, J.; Camilleri Podestà, M.T.; Cioni, P.L.; Flamini, G.; Morelli, I. Essential oil composition of different parts of Tetraclinis articulata. J. Essent. Oil Res. 2000, 12, 29–32. [Google Scholar] [CrossRef]
- Jlizi, S.; Zardi-Bergaoui, A.; Znati, M.; Flamini, G.; Ascrizzi, R.; Ben Jannet, H. Chemical composition and biological evaluation of the resin from Tetraclinis articulata (Vahl.) Masters: A promising source of bioactive secondary metabolites. Ind. Crops Prod 2018, 124, 74–83. [Google Scholar] [CrossRef]
- Fidyt, K.; Fiedorowicz, A.; Strządała, L.; Szumny, A. β-caryophyllene and β-caryophyllene oxide—natural compounds of anticancer and analgesic properties. Cancer Med. 2016, 5, 3007–3017. [Google Scholar] [CrossRef] [PubMed]
- Sieniawska, E.; Swiatek, Ł.; Rajtar, B.; Kozioł, E.; Polz-Dacewicz, M.; Skalicka-Wozniak, K. Carrot seed essential oil—Source of carotol and cytotoxicity study. Ind. Crops Prod. 2016, 92, 109–115. [Google Scholar] [CrossRef]
- Alidadiyani, N.; Salehi, R.; Ghaderi, S.; Samadi, N.; Davaran, S. Synergistic antiproliferative effects of methotrexate-loaded smart silica nanocomposites in MDA-MB-231 breast cancer cells. Artif. Cells Nanomed. Biotechnol. 2016, 44, 603–609. [Google Scholar] [CrossRef]
- Guan, X.; Guan, Y. Artemisinin induces selective and potent anticancer effects in drug-resistant breast cancer cells by inducing cellular apoptosis and autophagy and G2/M cell cycle arrest. JBUON 2020, 25, 1330–1336. [Google Scholar] [PubMed]
- Oliveira, P.F.; Morais Alves, J.; Lopes Damasceno, J.; Oliveira, R.A.M.; Júnior Dias, H.; Eduardo Miller Crotti, A.; Crispim Tavares, D. Cytotoxicity screening of essential oils in cancer cell lines. Rev. Bras. Farm. 2015, 25, 183–188. [Google Scholar] [CrossRef]
- Pan, Z.; Wang, S.-K.; Cheng, X.-L.; Tian, X.-W.; Wang, J. Caryophyllene oxide exhibits anti-cancer effects in MG-63 human os-teosarcoma cells via the inhibition of cell migration, generation of reactive oxygen species and induction of apoptosis. Bangla-desh J. Pharmacol. 2016, 11, 817–823. [Google Scholar] [CrossRef]
- Di Giacomo, S.; Di Sotto, A.; Mazzanti, G.; Wink, M. Chemosensitizing Properties of β-Caryophyllene and β-Caryophyllene Oxide in Combination with Doxorubicin in Human Cancer Cells. Anticancer Res. 2017, 37, 1191–1196. [Google Scholar]
- Lin, J.; Cai, Q.-Y.; Xu, W.; Lin, J.-M.; Peng, J. Chemical Composition, Anticancer, Anti-neuroinflammatory, and Antioxidant Activities of the Essential Oil of Patrinia scabiosaefolia. Chin. J. Integr. Med. 2018, 24, 207–212. [Google Scholar] [CrossRef]
- Jun, N.J.; Mosaddik, A.; Moon, J.Y.; Jang, K.-C.; Lee, D.-S.; Ahn, K.S.; Cho, S.K. Cytotoxic Activity of β-Caryophyllene Oxide Isolated from Jeju Guava (Psidium cattleianum Sabine) Leaf. Rec. Nat. Prod. 2011, 5, 242–246. [Google Scholar]
- Sylvestre, M.; Legault, J.; Dufour, D.; Pichette, A. Chemical composition and anticancer activity of leaf essential oil of Myrica gale L. Phytomedicine 2005, 12, 299–304. [Google Scholar] [CrossRef] [PubMed]
- Lattaoui, N.; Tantaoui-Elaraki, A. Individual and combined oils. Rivista Italiana EPPOS 1994, 13, 13–19. [Google Scholar]
- Yu, J.Q.; Lei, J.Q.; Yu, H.D.; Cai, X.; Zou, G.L. Chemical composition and antimicrobial activity of essential oil of Scutellaria barbata. Phytochemistry 2004, 65, 881–884. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Standards and Technology. NIST/EPA/NIH Mass Spectral Library; The NIST Mass Spectrometry Data Center: Gaithersburg, MD, USA, 2014. [Google Scholar]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy, 4th ed.; Allured Pub. Corp.: Carol Stream, IL, USA, 2007. [Google Scholar]
- Scagliarini, A.; Mathey, A.; Aires, V.; Delmas, D. Xanthohumol, a Prenylated Flavonoid from Hops, Induces DNA Damages in Colorectal Cancer Cells and Sensitizes SW480 Cells to the SN38 Chemotherapeutic Agent. Cells 2020, 9, 932. [Google Scholar] [CrossRef]

| Compound | LRI a | Composition (%) | m/zc | ||||||
|---|---|---|---|---|---|---|---|---|---|
| HEE | E.1 | E.2 | E.3 | E.4 | E.5 | ||||
| 1 | 2-Nonanol | 1100 | 0.1 | 0.2 | - b | - | - | - | 45, 69, 144 |
| 2 | Fenchol | 1113 | 0.2 | 0.4 | - | - | - | - | 81, 80, 154 |
| 3 | α-Campholenal | 1126 | 0.2 | 0.3 | - | - | - | - | 108, 93, 152 |
| 4 | trans-Pinocarveol | 1141 | 0.3 | 0.9 | - | - | - | - | 92, 70, 152 |
| 5 | cis-Verbenol | 1142 | 0.1 | 0.2 | - | - | - | - | 94, 109, 152 |
| 6 | trans-Verbenol | 1143 | 0.4 | 1.2 | - | - | - | - | 91, 109, 152 |
| 7 | Camphene hydrate | 1150 | 0.1 | 0.4 | - | - | - | - | 71, 43, 154 |
| 8 | Isoborneol | 1156 | 0.1 | 0.4 | - | - | - | - | 95, 41, 154 |
| 9 | Borneol | 1168 | 2.4 | 4.1 | 0.3 | - | - | 0.6 | 95, 110, 154 |
| 10 | 4-Terpineol | 1179 | 0.2 | 0.6 | - | - | - | - | 71, 111, 154 |
| 11 | p-Cymen-8-ol | 1185 | 1.2 | 1.4 | 0.7 | 0.4 | 0.4 | 0.8 | 43, 135, 150 |
| 12 | α-Terpineol | 1191 | 7.5 | 10.4 | 1.3 | 0.4 | 0.3 | 0.9 | 59, 93, 154 |
| 13 | Myrtenol | 1193 | 1.2 | 2.5 | 0.3 | - | - | 0.2 | 79, 91, 152 |
| 14 | Verbenone | 1205 | 1.3 | 1.3 | 0.7 | 0.6 | 0.4 | 1.1 | 107, 135, 150 |
| 15 | trans-Carveol | 1220 | 0.9 | 1.2 | 0.4 | - | - | 0.3 | 109, 84, 152 |
| 16 | cis-Myrtanol | 1251 | 0.4 | 0.7 | - | - | - | - | 41, 69, 154 |
| 17 | trans-Myrtanol | 1258 | 0.7 | 0.9 | 0.4 | - | - | - | 42, 69, 154 |
| 18 | Bornyl acetate | 1286 | 0.5 | 1.1 | - | - | - | - | 95, 43, 196 |
| 19 | Carvacrol | 1298 | 0.1 | 0.4 | - | - | - | - | 135, 91, 150 |
| 20 | Cyclosativene | 1369 | - | - | 0.3 | - | - | 0.3 | 161, 105, 204 |
| 21 | Longicyclene | 1371 | - | - | 0.3 | - | - | 0.5 | 94, 105, 204 |
| 22 | α-Copaene | 1377 | 0.2 | 0.6 | 0.7 | 0.8 | 0.5 | 0.7 | 161, 119, 204 |
| 23 | Longifolene | 1404 | 1.2 | 1.4 | 1.7 | 1.8 | 1.1 | 1.2 | 161, 94, 204 |
| 24 | β-Caryophyllene | 1419 | 5.5 | 5.2 | 2.6 | 2.9 | 0.6 | - | 93, 133, 204 |
| 25 | α-Humulene | 1455 | 1.4 | 1.6 | 1.0 | 1.0 | 0.3 | - | 98, 80, 204 |
| 26 | γ-Muurolene | 1478 | 0.2 | 0.5 | 0.9 | 0.9 | 1.1 | 1.3 | 161, 105, 204 |
| 27 | 8,9-Dehydrothymol isobutyrate | 1480 | 0.6 | - | - | - | - | - | 148, 133, 218 |
| 28 | Thymylisobutyrate | 1481 | 3.4 | 0.1 | - | 0.3 | - | - | 135, 150, 220 |
| 29 | Nerylisobutyrate | 1489 | 1.8 | 0.1 | - | - | - | - | 69, 93, 208 |
| 30 | α-Muurolene | 1499 | 5.5 | 6.7 | 9.1 | 11.6 | 11.2 | 12.5 | 105, 161, 204 |
| 31 | Modhephen-8-β-ol | 1508 | 6.6 | 0.2 | - | - | - | - | 189, 119, 204 |
| 32 | trans-γ-Cadinene | 1514 | - | - | - | 0.3 | 0.3 | 0.4 | 161, 105, 204 |
| 33 | cis-Calamenene | 1523 | - | - | 0.7 | - | 0.7 | 0.8 | 159, 160, 202 |
| 34 | δ-Cadinene | 1524 | 0.9 | 1.0 | - | 1.1 | - | - | 161, 134, 204 |
| 35 | α-Calacorene | 1543 | - | - | - | 0.3 | - | - | 157, 142, 200 |
| 36 | Elemol | 1550 | 0.8 | 1.4 | 1.3 | 0.8 | 0.6 | 0.5 | 59, 93, 222 |
| 37 | Dodecanoicacid | 1567 | - | - | - | - | - | 2.7 | 73, 60, 200 |
| 38 | Palustrol | 1569 | - | - | 0.8 | 0.7 | - | 0.5 | 41, 55, 222 |
| 39 | Neryl 2-methylbutyrate | 1575 | 2.5 | 0.1 | - | - | - | - | 69, 41, 238 |
| 40 | Caryophyllene oxide | 1581 | 13.1 | 25.2 | 26.6 | 20.5 | 20.2 | 21.9 | 43, 41, 220 |
| 41 | Geranyl 2-methylbutyrate | 1586 | 4.9 | 0.2 | - | - | - | - | 69, 41, 238 |
| 42 | 1-Hexadecene | 1592 | 1.8 | - | - | - | - | - | 43, 55, 224 |
| 43 | cis-Arteannuicacid | 1593 | 4.1 | - | 1.2 | - | - | - | 121, 119, 234 |
| 44 | n-Hexadecane | 1600 | - | - | - | - | 0.6 | - | 57, 43, 226 |
| 45 | Carotol | 1602 | 9.2 | 12.0 | 20.3 | 20.7 | 21.2 | 15.1 | 161, 204, 222 |
| 46 | Humulene epoxide II | 1607 | 2.6 | 4.0 | 7.2 | 6.1 | 6.2 | 5.4 | 109, 67, 220 |
| 47 | 1-epi-Cubenol | 1629 | - | - | - | 0.8 | 0.6 | - | 119, 41, 222 |
| 48 | γ-Eudesmol | 1631 | - | - | - | 0.7 | 0.8 | - | 189, 204, 222 |
| 49 | Caryophylla-4(14),8(15)-dien-5-ol | 1637 | 1.6 | 1.2 | 2.0 | 2.0 | 1.8 | 1.4 | 136, 91, 220 |
| 50 | T-Cadinol | 1641 | 1.5 | 1.1 | 2.1 | 1.9 | 2.2 | 1.3 | 161, 43, 222 |
| 51 | T-Muurolol | 1642 | 1.7 | 0.9 | 1.9 | 2.3 | 2.7 | 1.8 | 95, 121, 222 |
| 52 | 14-Hydroxy-9-epi-(E)-caryophyllene | 1665 | 3.2 | 2.7 | 6.8 | 9.6 | 13.1 | 15.5 | 91, 93, 220 |
| 53 | Cadalene | 1673 | 1.3 | 1.2 | 2.5 | 3.4 | 3.9 | 3.2 | 183, 168, 198 |
| 54 | 1-Octadecene | 1793 | 1.7 | - | - | - | - | - | 43, 41, 252 |
| Oxygenated monoterpenes | 31.0 | 28.9 | 4.1 | 1.7 | 1.1 | 3.9 | |||
| Sesquiterpene hydrocarbons | 16.2 | 18.2 | 19.8 | 24.1 | 19.7 | 20.9 | |||
| Oxygenated sesquiterpenes | 44.4 | 48.7 | 70.2 | 66.1 | 69.4 | 63.4 | |||
| Nonterpene derivatives | 3.6 | 0.2 | 0.0 | 0.0 | 0.6 | 2.7 | |||
| Total identified | 95.2 | 96.0 | 94.1 | 91.9 | 90.8 | 90.9 | |||
| IC50 | 5-FU | HEE | E.1 | E.2 | E.3 | E.4 | E.5 |
|---|---|---|---|---|---|---|---|
| MDA-MB-231 | 2.2 | 83.0 | 96.5 | 90.7 | 85.9 | 91.8 | 100.0 |
| SW620 | 0.3 | 25.7 | 37.8 | 26.2 | 43.1 | 57.2 | 96.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jlizi, S.; Lahmar, A.; Zardi-Bergaoui, A.; Ascrizzi, R.; Flamini, G.; Harrath, A.H.; Chekir-Ghedira, L.; Ben Jannet, H. Chemical Composition and Cytotoxic Activity of the Fractionated Trunk Bark Essential Oil from Tetraclinis articulata (Vahl) Mast. Growing in Tunisia. Molecules 2021, 26, 1110. https://doi.org/10.3390/molecules26041110
Jlizi S, Lahmar A, Zardi-Bergaoui A, Ascrizzi R, Flamini G, Harrath AH, Chekir-Ghedira L, Ben Jannet H. Chemical Composition and Cytotoxic Activity of the Fractionated Trunk Bark Essential Oil from Tetraclinis articulata (Vahl) Mast. Growing in Tunisia. Molecules. 2021; 26(4):1110. https://doi.org/10.3390/molecules26041110
Chicago/Turabian StyleJlizi, Salma, Aida Lahmar, Afifa Zardi-Bergaoui, Roberta Ascrizzi, Guido Flamini, Abdel Halim Harrath, Leila Chekir-Ghedira, and Hichem Ben Jannet. 2021. "Chemical Composition and Cytotoxic Activity of the Fractionated Trunk Bark Essential Oil from Tetraclinis articulata (Vahl) Mast. Growing in Tunisia" Molecules 26, no. 4: 1110. https://doi.org/10.3390/molecules26041110
APA StyleJlizi, S., Lahmar, A., Zardi-Bergaoui, A., Ascrizzi, R., Flamini, G., Harrath, A. H., Chekir-Ghedira, L., & Ben Jannet, H. (2021). Chemical Composition and Cytotoxic Activity of the Fractionated Trunk Bark Essential Oil from Tetraclinis articulata (Vahl) Mast. Growing in Tunisia. Molecules, 26(4), 1110. https://doi.org/10.3390/molecules26041110









