Effects of Probiotic Culture Supernatant on Cariogenic Biofilm Formation and RANKL-Induced Osteoclastogenesis in RAW 264.7 Macrophages
Abstract
1. Introduction
2. Results and Discussion
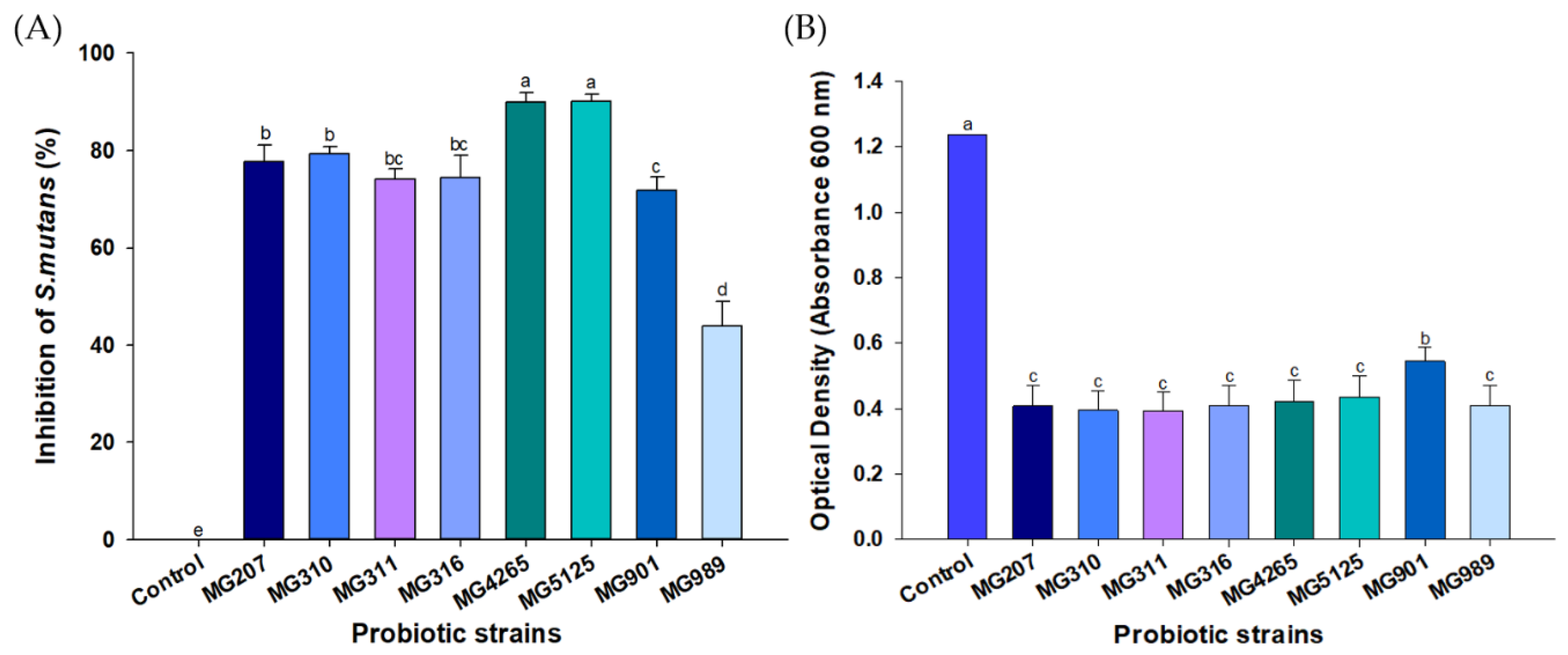
2.1. Effect of Probiotic Strains on the Growth of S. mutans
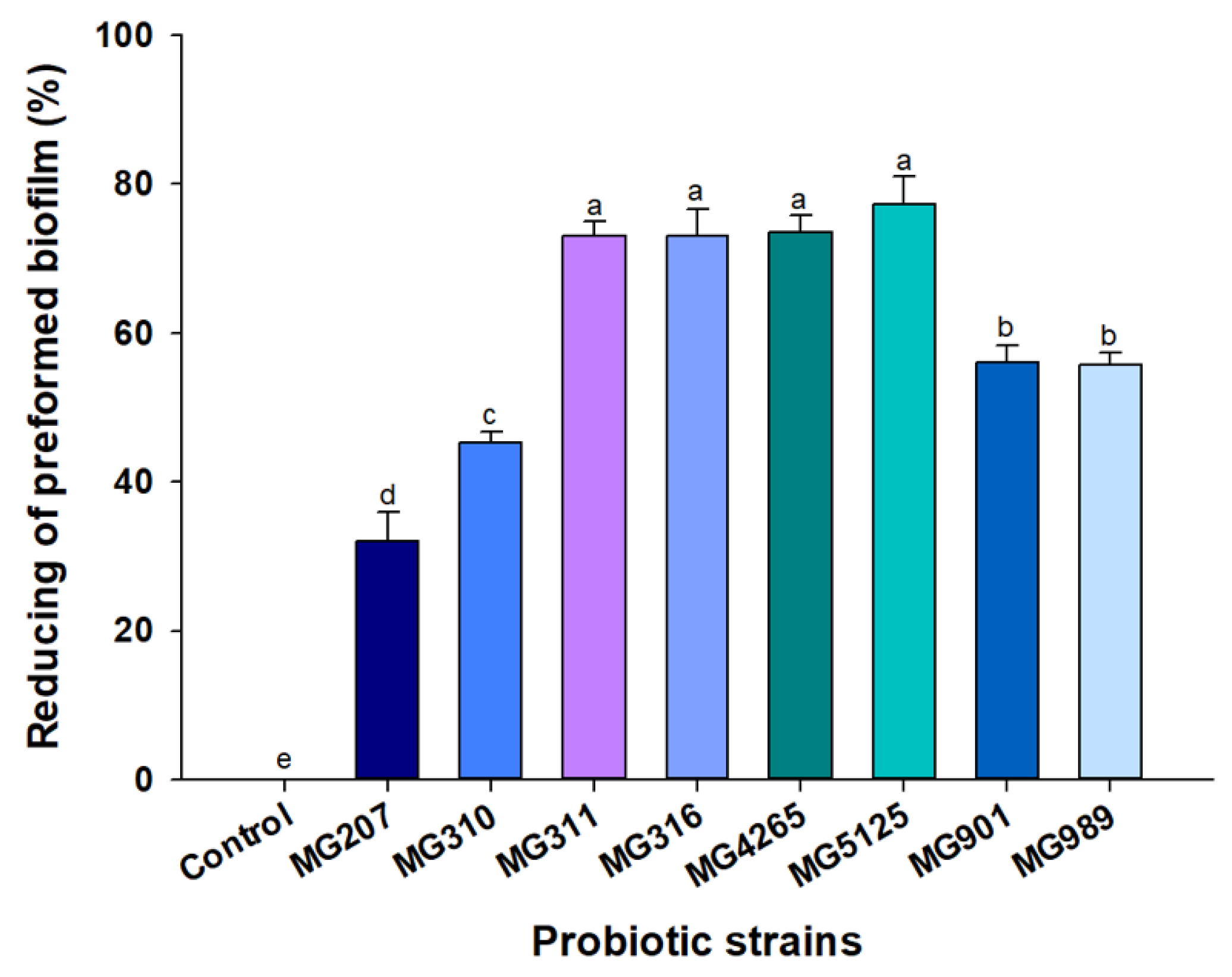
2.2. Effect of SCSs on S. mutans-Induced Preformed Biofilm Formation
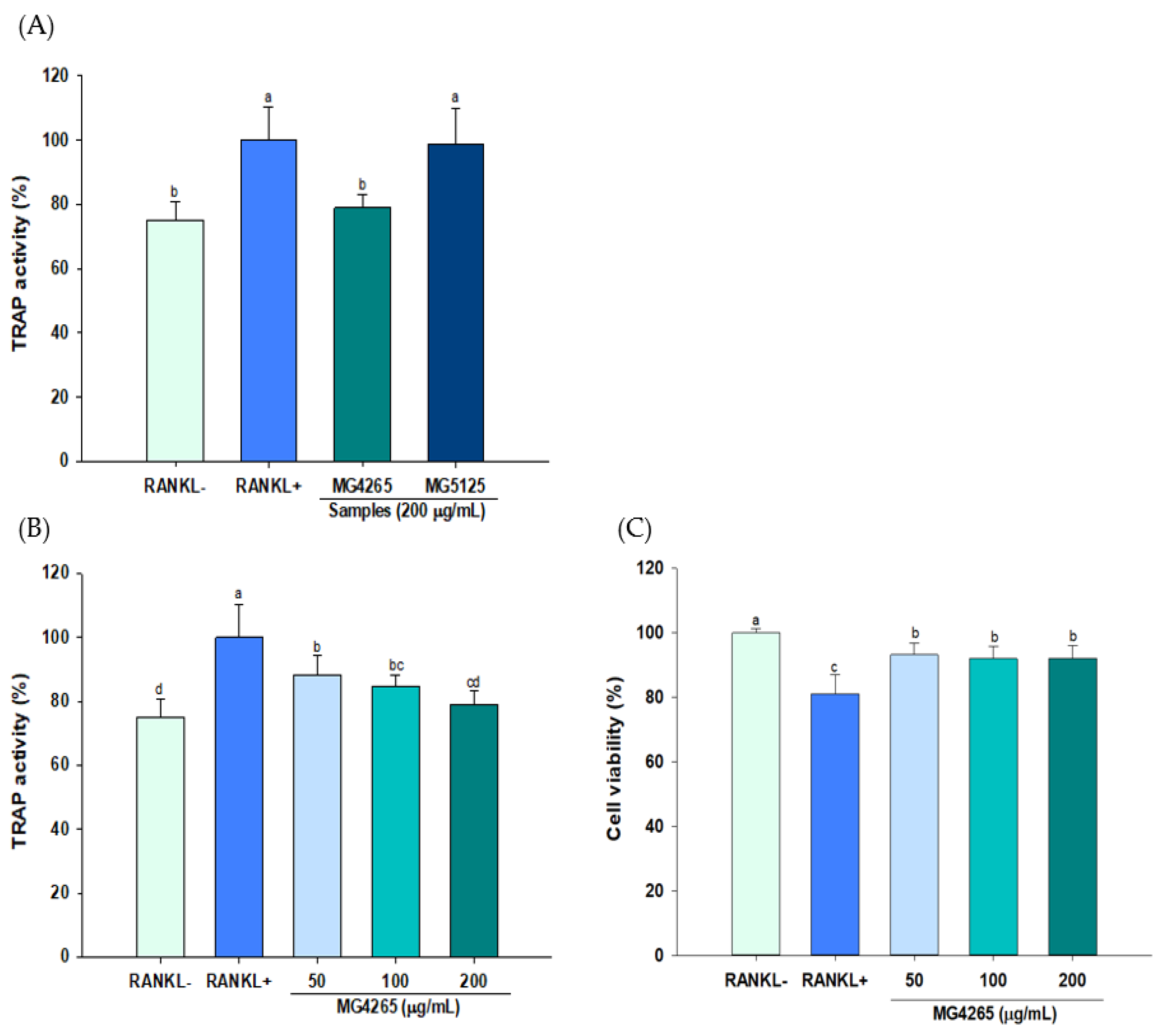
2.3. Effect of SCSs on RANKL-Induced Osteoclast Differentiation
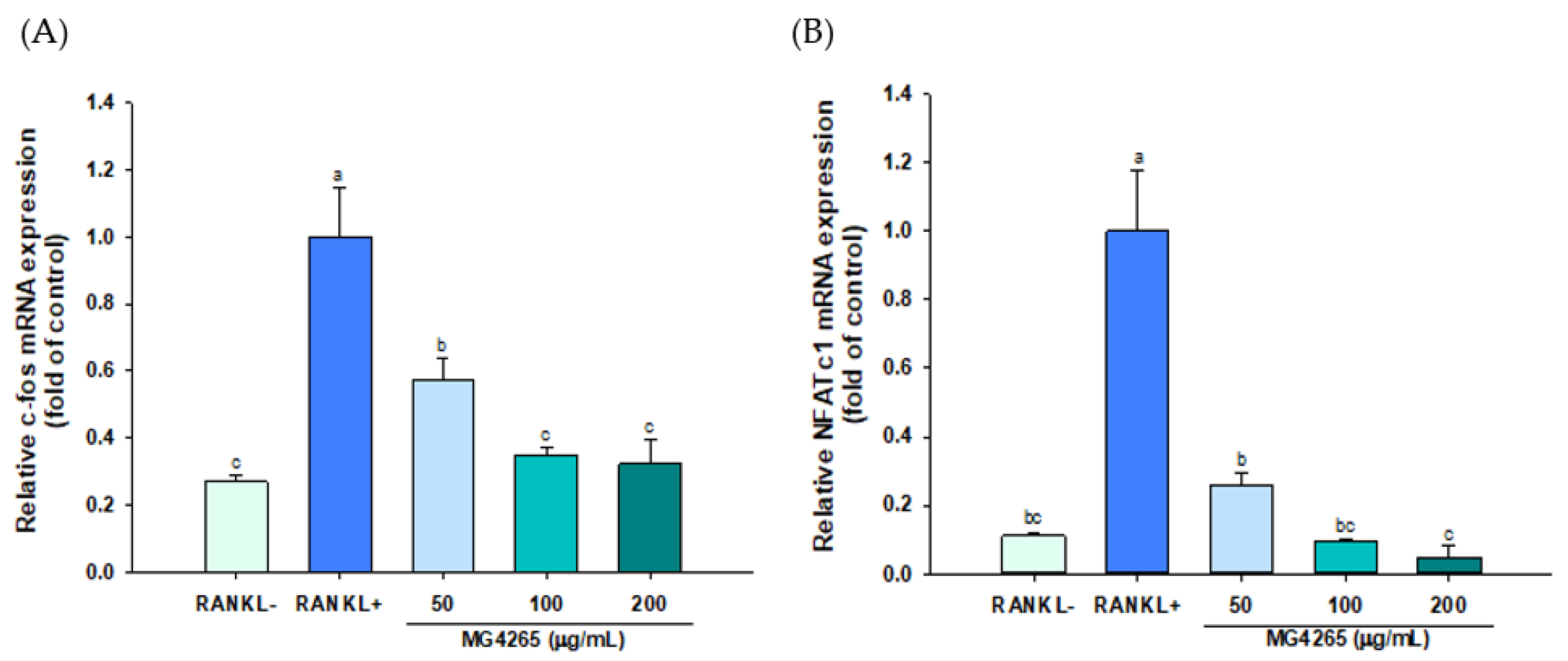
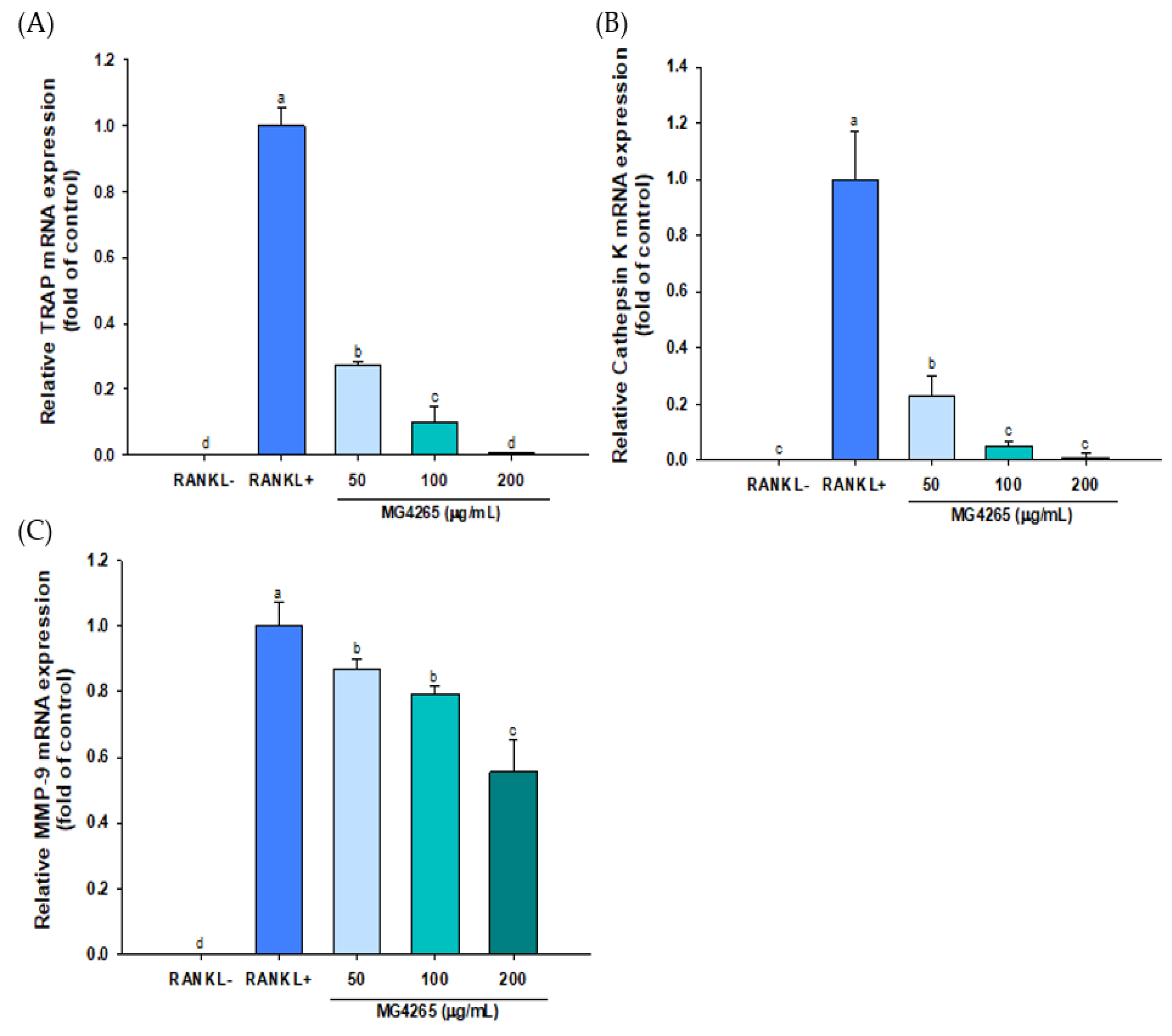
2.4. Effect of the SCS of L. salivarius MG4265 on Osteoclast Specific Transcriptional Factors and Osteoclast-Associated Gene Expression
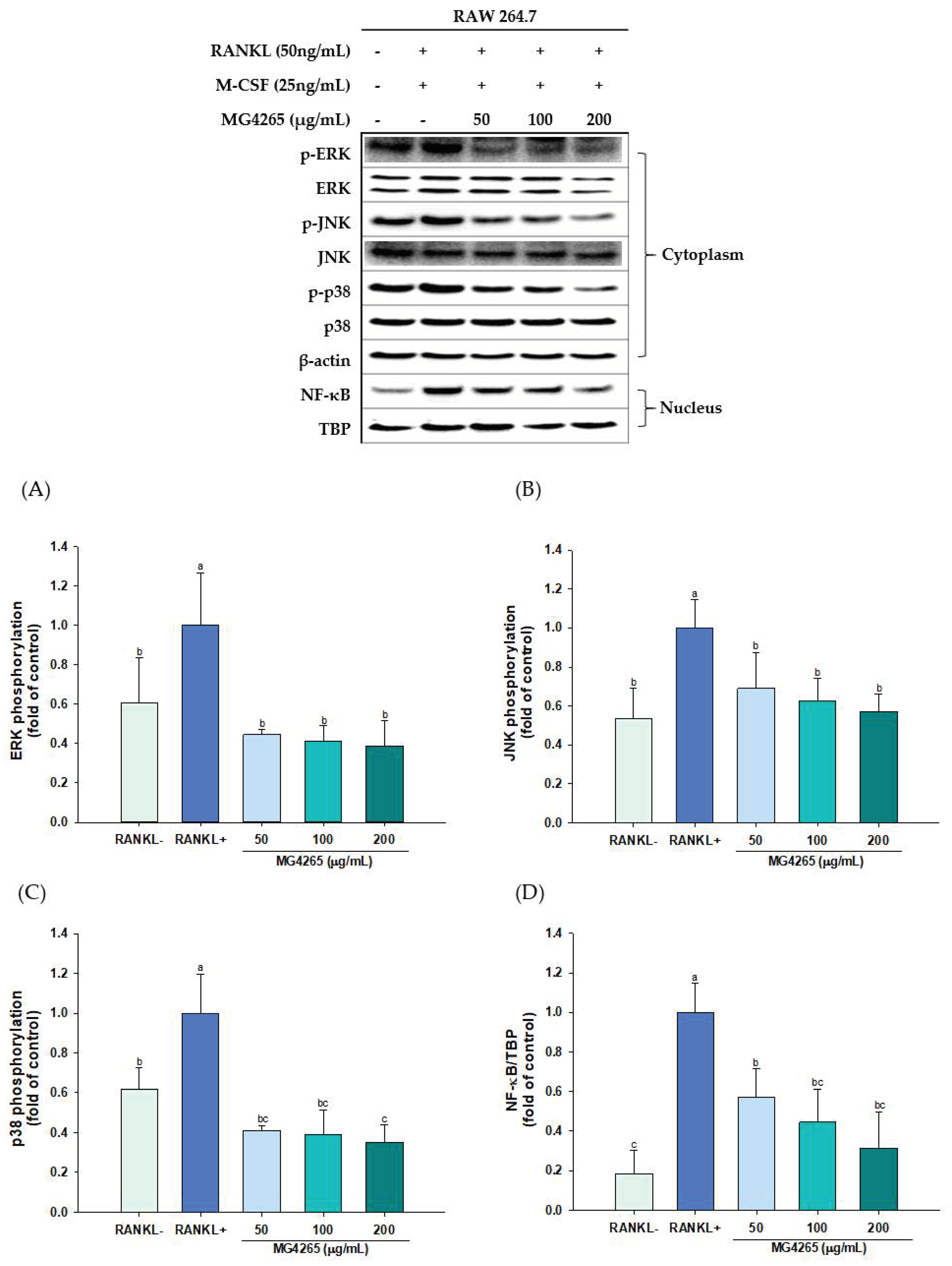
2.5. Effect of the SCS of L. salivarius MG4265 on RANKL-Induced Mitogen Activated Protein Kinase (MAPKs) Activation
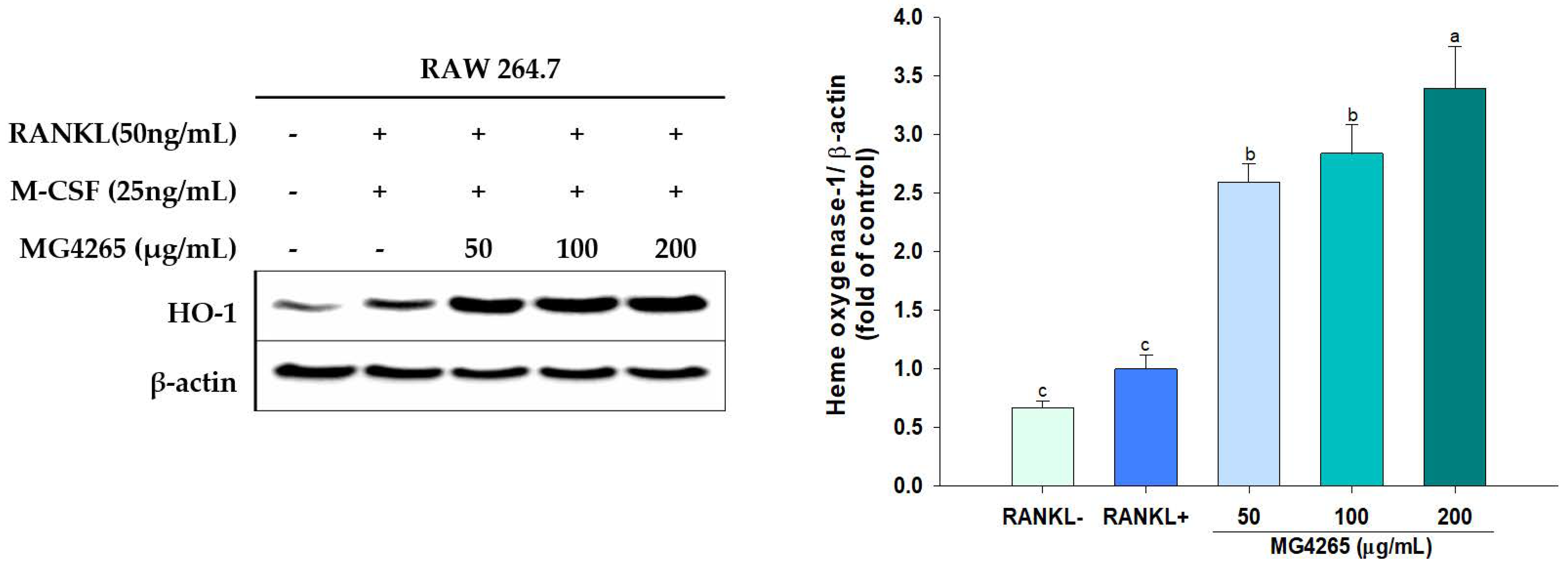
2.6. Effect of the SCS of L. salivarius MG4265 on HO-1 Induction
3. Materials and Methods
3.1. Strains and Cultivation
3.2. Preparation of SCSs
3.3. Effect of Probiotic Strains on the Growth of S. mutans
3.4. Effect of SCSs on the Growth of S. mutans
3.5. Effect of SCSs on S. mutans-Induced Biofilm Formation
3.6. RANKL-Induced Osteoclast Differentiation of Murine Osteoclast Progenitor RAW 264.7 Cells
3.7. Tartrate-Resistant Acid Phosphatase-Positive (TRAP) Activity
3.8. RNA Extraction and Quantitative Real Time PCR (qRT-PCR)
3.9. Western Blotting Analysis
3.10. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- FAO/WHO. Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Acid Bacteria. Report of a Joint FAO/WHO Expert Consultation, Córdoba, Argentina. 2001. Available online: https://www.who.int/foodsafety/publications/fs_management/en/probiotics.pdf?ua=1 (accessed on 17 December 2020).
- Yu, H.-S.; Lee, N.-K.; Choi, A.-J.; Choe, J.-S.; Bae, C.H.; Paik, H.-D. Antagonistic and antioxidant effect of probiotic Weissella cibaria JW15. Food Sci. Biotechnol. 2019, 28, 851–855. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Ji, H.; Wang, S.; Liu, H.; Zhang, W.; Zhang, D.; Wang, Y. Probiotic Lactobacillus plantarum promotes intestinal barrier function by strengthening the epithelium and modulating gut microbiota. Front. Microbiol. 2018, 9, 1953. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-Y.; Bae, W.-Y.; Yu, H.-S.; Chang, K.-H.; Hong, Y.-H.; Lee, N.-K.; Paik, H.-D. Inula Britannica fermented with probiotic Weissella cibaria D30 exhibited anti-inflammatory effect and increased viability in RAW 264.7 cells. Food Sci. Biotechnol. 2020, 29, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, B.; Delgado, S.; Blanco-Míguez, A.; Lourenço, A.; Gueimonde, M.; Margolles, A. Probiotics, gut microbiota, and their influence on host health and disease. Mol. Nutr. Food Res. 2017, 61, 1600240. [Google Scholar] [CrossRef]
- Wegh, C.A.M.; Geerlings, S.Y.; Knol, J.; Roeselers, G.; Belzer, C. Postbiotics and their potential applications in early life nutrition and beyond. Int. J. Mol. Sci. 2019, 20, 4673. [Google Scholar] [CrossRef]
- Aguilar-Toalá, J.; Garcia-Varela, R.; Garcia, H.; Mata-Haro, V.; González-Córdova, A.; Vallejo-Cordoba, B.; Hernández-Mendoza, A. Postbiotics: An evolving term within the functional foods field. Trends Food Sci. Technol. 2018, 75, 105–114. [Google Scholar] [CrossRef]
- Moradi, M.; Kousheh, S.A.; Almasi, H.; Alizadeh, A.; Guimaraes, G.T.; Yilmaz, N.; Lotfi, A. Postbiotics produced by lactic acid bacteria: The next frontier in food safety. Compr. Rev. Food Sci. Food Saf. 2020, 19, 3390–3415. [Google Scholar] [CrossRef]
- Beristain-Bauza, S.C.; Mani-Lopez, E.; Palou, E.; Lopez-Malo, A. Antimicrobial activity of whey protein films supplemented with Lactobacillus sakei cell-free supernatant on fresh beef. Food Microbiol. 2017, 62, 207–211. [Google Scholar] [CrossRef]
- Moradi, M.; Mardani, K.; Tajik, H. Characterization and application of postbiotics of Lactobacillus spp. on Listeria monocytogenes in vitro and in food models. LWT-Food Sci. Technol. 2019, 111, 457–464. [Google Scholar] [CrossRef]
- Naito, M.; Yuasa, H.; Nomura, Y.; Nakayama, T.; Hamajima, N.; Hanada, N. Oral health status and health-related quality of life: A systematic review. J. Oral Sci. 2006, 48, 1–7. [Google Scholar] [CrossRef]
- Dewhirst, F.E.; Chen, T.; Izard, J.; Paster, B.J.; Tanner, A.C.R.; Yu, W.-H.; Lakshmanan, A.; Wade, W.G. The human oral microbiome. J. Bacteriol. 2010, 192, 5002–5017. [Google Scholar] [CrossRef] [PubMed]
- Saha, S.; Tomaro-Duchesneau, C.; Tabrizian, M.; Prakash, S. Probiotics as oral health biotherapeutics. Expert Opin. Biol. Ther. 2012, 12, 1207–1220. [Google Scholar] [CrossRef] [PubMed]
- Forssten, S.D.; Bjorklund, M.; Ouwehand, A.C. Streptococcus mutans, acrries and simulation models. Nutrients 2010, 2, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Schwendicke, F.; Korte, F.; Dorfer, C.E.; Kneist, S.; Fawzy El-Sayed, K.; Paris, S. Inhibition of Streptococcus mutans growthand biofilm formation by probiotics in vitro. Caries Res. 2017, 51, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Wasfi, R.; El-Rahman, O.A.A.; Zafer, M.M.; Ashour, H.M. Probiotic Lactobacillus sp. inhibit growth, biofilm formation and gene expression of caries-inducing Streptococcus mutans. J. Cell. Mol. Med. 2018, 22, 1972–1983. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.; Chen, X.; Chen, Y.; Chen, J.H. The effect of five probiotic lactobacilli strains on the growth and biofilm formation of Streptococcus mutans. Oral Dis. 2015, 21, e128–e134. [Google Scholar] [CrossRef]
- Corrêa, M.G.; Pires, P.R.; Ribeiro, F.V.; Pimentel, S.Z.; Casarin, R.C.; Cirano, F.R.; Casati, M.Z. Systemic treatment with resveratrol and/or curcumin reduces the progression of experimental periodontitis in rats. J. Periodontal Res. 2017, 52, 201–209. [Google Scholar] [CrossRef]
- Tóthová, L.U.; Celec, P. Oxidative stress and antioxidants in the diagnosis and therapy of periodontitis. Front. Physiol. 2017, 8, 1055–1068. [Google Scholar] [CrossRef]
- Djordjevic, D.; Wiedmann, M.; Mclandsborough, L. Microtiter plate assay for assessment of Listeria monocytogenes biofilm formation. Appl. Environ. Microbiol. 2002, 68, 2950–2958. [Google Scholar] [CrossRef]
- Liu, T.-H.; Tsai, T.-Y.; Pan, T.-M. Effect of ethanol extract from Lactobacillus paracasei subsp. Paracasei NTU 101 fermented skim milk on lipopolysaccharide-induced periodontal inflammation in rats. Food Funct. 2018, 9, 4916–4925. [Google Scholar] [CrossRef]
- Kim, S.; Kang, S.-S.; Choi, S.-I.; Kim, G.-H.; Imm, J.-Y. Ecklonia cava extract containing dieckol suppresses RANKL-induced osteoclastogenesis via MAPkinase/NF-κB pathway inhibition and heme oxygenase-1 induction. J. Microbiol. Biothechnol. 2019, 29, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Simark-Mattson, C.; Emilson, C.G.; Håkansson, E.G.; Jacobsson, C.; Roos, K.; Holm, S. Lactobacillus-mediated interference of mutans streptococci in caries-free vs. caries-active subjects. Eur. J. Oral Sci. 2007, 115, 308–314. [Google Scholar] [CrossRef] [PubMed]
- Tong, Z.; Zhou, L.; Li, J.; Kuang, R.; Lin, Y.; Ni, L. An in vitro investigation of Lactococus lactis antagonizing carriogenic bacterium Streptococcus mutans. Arch. Oral Biol. 2012, 57, 376–382. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.J.; Lee, S.H. Inhibitory effect of Lactococcus lactis HY 449 on cariogenic biofilm. J. Microbiol. Biotechnol. 2016, 26, 1829–1835. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-C.; Lin, C.-T.; Wu, C.-Y.; Peng, W.-S.; Lee, M.-J.; Tsai, Y.C. Inhibitory effect of Lactobacillus salivarius on Streptococus mutans biofilm formation. Mol. Oral Microbiol. 2015, 30, 16–26. [Google Scholar] [CrossRef]
- Hasslöf, P.; Hedberg, M.; Twetman, S.; Stecksén-Blicks, C. Growth inhibition of oral mutans streptococci and candida by commercial probiotic lactobacilli—An in vitro study. BMC Oral Health 2010, 10, 18. [Google Scholar] [CrossRef]
- Lin, X.; Chen, X.; Tu, Y.; Wang, S.; Chen, H. Effect of probiotic Lactobacilli on the growth of Streptococcus mutans and multispecies biofilms isolated from children with active caries. Med Sci. Monit. 2017, 23, 4175–4181. [Google Scholar] [CrossRef]
- Saha, S.; Tomaro-Duchesneau, C.; Rodes, L.; Malhotra, M.; Tabrizian, M.; Prakash, S. Investigation of probiotic bacteria as dental caries and periodontal disease biotherapeutics. Benef. Microb. 2014, 5, 447–460. [Google Scholar] [CrossRef]
- Caufield, P.W.; Schon, C.N.; Saraithong, P.; Li, Y.; Argimon, S. Oral Lactobacilli and dental caries: A model for niche adaptation in humans. J. Dent. Res. 2015, 94, 110S–118S. [Google Scholar] [CrossRef]
- Marsh, P.D.; Head, D.A.; Devine, D.A. Dental plaque as a biofilm and a microbial community—Implications for treatment. J. Oral Biosci. 2015, 57, 185–191. [Google Scholar] [CrossRef]
- Ansari, J.M.; Abraham, N.M.; Massaro, J.; Murphy, K.; Smith-Carpenter, J.; Fikrig, E. Anti-biofilm activity of a self-aggregating peptide against Streptococcus mutans. Front. Microbiol. 2017, 8, 488. [Google Scholar] [CrossRef] [PubMed]
- Endo, K.; Mine, Y.; Shuto, T.; Taji, T.; Murayama, T.; Nikawa, H. Comprehensive analysis of transcriptional profiles in oral epithelial-like cells stimulated with oral probiotic Lactobacillus spp. Arch. Oral Biol. 2020, 118, 104832. [Google Scholar] [CrossRef] [PubMed]
- Tahmourespour, A.; Kasra-Kermanshahi, R.; Salehi, R. Lactobacillus rhamnosus biosurfactant inhibits biofilm formation and gene expression of caries-inducing Streptococcus mutans. Dent. Res. J. 2019, 18, 87–94. [Google Scholar] [CrossRef]
- Brzozowski, B.; Bednarski, W.; Gołek, P. The adhesive capability of two Lactobacillus strains and physicochemical properties of their synthesized biosurfactants. Food Technol. Biotechnol. 2011, 49, 177–186. [Google Scholar]
- Ahn, K.B.; Baik, J.E.; Park, O.J.; Yun, C.H.; Han, S.H. Lactobacillus plantarum lipoteichoic acid inhibits biofilm formation of Streptococcus mutans. PLoS ONE 2018, 13, e0192694. [Google Scholar] [CrossRef]
- Roodman, G. Advances in bone biology: The osteoclast. Endocr. Rev. 1996, 17, 308–332. [Google Scholar]
- Collin-Osdoby, P.; Osdoby, P. RANKL-mediated osteoclast formation from murine RAW 264.7 cells. Methods Mol. Biol. 2012, 816, 187–202. [Google Scholar]
- Grigoriadis, A.E.; Wang, Z.Q.; Cecchini, M.G.; Hofstetter, W.; Felix, R.; Fleisch, H.A.; Wagner, E.F. c-Fos: A key regulator of osteoclast-macrophage lineage determination and bone remodeling. Science 1994, 266, 443–448. [Google Scholar] [CrossRef]
- Zhao, Q.; Wang, X.; Liu, Y.; He, A.; Jia, R. NFATc1: Functions in osteoclasts. Int. J. Biochem. Cell. Biol. 2010, 42, 576–579. [Google Scholar] [CrossRef]
- Fujisaki, K.; Tanabe, N.; Suzuki, N.; Kawato, T.; Takeichi, O.; Tsuzukibashi, O.; Makimura, M.; Ito, K.; Maeno, M. Receptor activator of NF-kappaB ligand induces the expression of carbonic anhydrase II, cathepsin K, and matrix metalloproteinase-9 in osteoclast precursor RAW264.7 cells. Life Sci. 2007, 80, 1311–1318. [Google Scholar] [CrossRef]
- Huang, H.; Chang, E.-J.; Ryu, J.; Lee, Z.H.; Lee, Y.; Kim, H.-H. Induction of c-Fos and NFATc1 during RANKL-stimulated osteoclast differentiation is mediated by the p38 signaling pathway. Biochem. Biophys. Res. Commun. 2006, 351, 99–105. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, K.; Galson, D.L.; Zhao, C.; Peng, L.; Laplace, C.; Wang, K.Z.; Bachler, M.A.; Amano, H.; Aburatani, H.; Ishikawa, H.; et al. Nuclear factor of activated T-cells (NFAT) rescues osteoclastogenesis in precursors lacking c-Fos. J. Biol. Chem. 2004, 279, 26475–26480. [Google Scholar] [CrossRef] [PubMed]
- Henriksen, K.; Tanko, L.B.; Qvist, P.; Delmas, P.D.; Christiansen, C.; Karsdal, M.A. Assessment of osteoclast number and function: Application in the development of new and improved treatment modalities for bone diseases. Osteoporos. Int. 2007, 18, 681–685. [Google Scholar] [CrossRef] [PubMed]
- Hayman, A.R. Tartrate-resistant acid phosphatase (TRAP) and the osteoclast/immune cell dichotomy. Autoimmunity 2008, 41, 218–223. [Google Scholar] [CrossRef] [PubMed]
- Wittrant, Y.; Theoleyre, S.; Couillaud, S.; Dunstan, C.; Heymann, D.; Redini, F. Regulation of osteoclast protease expression by RANKL. Biochem. Biophys. Res. Commun. 2003, 310, 774–778. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Kim, E.-Y.; Lee, B.; Min, J.-H.; Song, D.-U.; Lim, J.-M.; Eom, J.W.; Yeom, M.; Jung, H.-S.; Sohn, Y. The effects of Lycii Radicis Cortex on RANKL-induced osteoclast differentiation and activation in RAW 264.7 cells. Int. J. Mol. Med. 2016, 37, 649–658. [Google Scholar] [CrossRef]
- Lee, K.; Seo, I.; Choi, M.H.; Jeong, D. Roles of mitogen-activated protein kinases in osteoclast biology. Int. J. Mol. Sci. 2018, 19, 3004. [Google Scholar] [CrossRef]
- Jimi, E.; Aoki, K.; Saito, H.; D’Acquisto, F.; May, M.J.; Nakamura, I.; Sudo, T.; Kojima, T.; Okamoto, F.; Fukushima, H.; et al. Selective inhibition of NF-κB blocks osteoclastogenesis and prevents inflammatory bone destruction in vivo. Nat. Med. 2004, 10, 617–624. [Google Scholar] [CrossRef]
- Chung, Y.H.; Choi, B.; Song, D.H.; Song, Y.; Kang, S.W.; Yoon, S.Y.; Kim, S.W.; Lee, H.K.; Chang, E.J. Interleukin-1beta promotes the lc3-mediated secretory function of osteoclast precursors by stimulating the Ca2+-dependent activation of erk. Int. J. Biochem. Cell Biol. 2014, 54, 198–207. [Google Scholar] [CrossRef]
- Han, Z.; Boyle, D.L.; Chang, L.; Bennett, B.; Karin, M.; Yang, L.; Manning, A.M.; Firestein, G.S. c-Jun N-terminal kinase is required for metalloproteinase expression and joint destruction in inflammatory arthritis. J. Clin. Invest. 2001, 108, 73–81. [Google Scholar] [CrossRef]
- Matsumoto, M.; Kogawa, M.; Wada, S.; Takayanagi, H.; Tsujimoto, M.; Katayama, S.; Hisatake, K.; Nogi, Y. Essential role of p38 mitogen-activated protein kinase in cathepsin k gene expression during osteoclastogenesis through association of nfatc1 and pu.1. J. Biol. Chem. 2004, 279, 45969–45979. [Google Scholar] [CrossRef] [PubMed]
- Lee, Z.H.; Kim, H.-H. Signal transduction by receptor activator of nuclear factor kappa B in osteoclasts. Biochem. Biophys. Res. Commun. 2003, 305, 211–214. [Google Scholar] [CrossRef]
- Quach, D.; Parameswaran, N.; McCabe, L.; Britton, R.A. Characterizing how probiotic Lactobacillus reuteri 6475 and lactobacillic acid mediate suppression of osteoclast differentiation. Bone Rep. 2019, 11, 100227. [Google Scholar] [CrossRef] [PubMed]
- Britton, R.A.; Irwin, R.; Quach, D.; Schaefer, L.; Zhang, J.; Lee, T.; Parameswaran, N.; Mccabe, L.R. Probiotic L. reuteri treatment prevents bone loss in a menopausal ovariectomized mouse model. J. Cell. Physiol. 2014, 229, 1822–1830. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.-H.; Chen, T.-H.; Chou, T.-C. Magnolol inhibits RANKL-induced osteoclast differentiation of RAW 264.7 cells through heme oxygenase-1-dependent inhibition of NFATC1 expression. J. Nat. Prod. 2015, 78, 61–68. [Google Scholar] [CrossRef]
- Vijayan, V.; Wagener, F.A.D.T.G.; Immenschuh, S. The macrophage heme-heme oxygenase-1 system and its role in inflammation. Biochem. Pharmacol. 2018, 153, 159–167. [Google Scholar] [CrossRef]
- Waddington, R.J.; Moseley, R.; Embery, G. Reactive oxygen species: A potential role in the pathogenesis of periodontal diseases. Oral Dis. 2000, 6, 138–151. [Google Scholar] [CrossRef]
- Kwon, H.-J.; Kim, J.M.; Han, K.-I.; Jung, E.-G.; Kim, Y.H.; Patnaik, B.B.; Yoon, M.S.; Chung, S.K.; Kim, W.J.; Han, M.-D. Mutan: A mixed linkage α-[(1,3)- and (1,6)]-D-glucan from Streptococcus mutans, that induces osteoclast differentiation and promotes alveolar bone loss. Carb. Polym. 2016, 137, 561–569. [Google Scholar] [CrossRef]
- Shimauchi, H.; Mayanagi, G.; Nakaya, S.; Minamibuchi, M.; Ito, Y.; Yamaki, K.; Hirata, H. Improvement of periodontal condition by probiotics with Lactobacillus salivarius WB21: A randomized, double-blind, placebo-controlled study. J. Clin. Periodontol. 2008, 35, 897–905. [Google Scholar] [CrossRef]
- Ginsburg, I. Role of lipoteichoic acid in infection and inflammation. Lancet Infect. Dis. 2002, 2, 171–179. [Google Scholar] [CrossRef]
- Teame, T.; Wang, A.; Xie, M.; Zhang, Z.; Yang, Y.; Ding, Q.; Gao, C.; Olsen, R.E.; Ran, C.; Zhou, Z. Paraprobiotics and postbiotics of probiotic Lactobacilli, their positive effects on the host and action mechanisms: A review. Front. Nutr. 2020, 7, 570344. [Google Scholar] [CrossRef] [PubMed]
- Lebeer, S.; Bron, P.A.; Marco, M.L.; Van Pijkeren, J.P.; Motherway, M.O.C.; Hill, C.; Pot, B.; Roos, S.; Klaenhammer, T. Identification of probiotic effector molecules: Present state and future perspectives. Curr. Opin. Biotechnol. 2018, 49, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Zaiss, M.M.; Jones, R.M.; Schett, G.; Pacifici, R. The gut-bone axis: How bacterial metabolites bridge the distance. J. Clin. Invest. 2019, 129, 3018–3028. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.-H.; Tsai, T.-Y.; Pan, T.-M. Isolation and identification of anti-periodontitis ingredients in Lactobacillus paracasei subsp. paracasei NTU 101-fermented skim milk in vitro. J. Funct. Food 2019, 60, 103449. [Google Scholar] [CrossRef]
- Krzyściak, W.; Kościelniak, D.; Papież, M.; Vyhouskaya, P.; Zagórska-Świeży, K.; Kołodziej, I.; Bystrowska, B.; Jurczak, A. Effect of a Lactobacillus salivarius probiotic on a double-species Streptococcus mutans and Candida albicans caries biofilm. Nutrients 2017, 9, 1242. [Google Scholar] [CrossRef]
- Lee, D.; Imm, J.-Y. AMP kinase activation and inhibition of nuclear factor-kappa B (NF-κB) translocation contribute to the anti-inflammatory effect of tricin. J. Food Biochem. 2017, 41, e12293. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jung, J.-I.; Baek, S.-M.; Nguyen, T.H.; Kim, J.W.; Kang, C.-H.; Kim, S.; Imm, J.-Y. Effects of Probiotic Culture Supernatant on Cariogenic Biofilm Formation and RANKL-Induced Osteoclastogenesis in RAW 264.7 Macrophages. Molecules 2021, 26, 733. https://doi.org/10.3390/molecules26030733
Jung J-I, Baek S-M, Nguyen TH, Kim JW, Kang C-H, Kim S, Imm J-Y. Effects of Probiotic Culture Supernatant on Cariogenic Biofilm Formation and RANKL-Induced Osteoclastogenesis in RAW 264.7 Macrophages. Molecules. 2021; 26(3):733. https://doi.org/10.3390/molecules26030733
Chicago/Turabian StyleJung, Jae-In, Seung-Min Baek, Trung Hau Nguyen, Jin Woo Kim, Chang-Ho Kang, Seonyoung Kim, and Jee-Young Imm. 2021. "Effects of Probiotic Culture Supernatant on Cariogenic Biofilm Formation and RANKL-Induced Osteoclastogenesis in RAW 264.7 Macrophages" Molecules 26, no. 3: 733. https://doi.org/10.3390/molecules26030733
APA StyleJung, J.-I., Baek, S.-M., Nguyen, T. H., Kim, J. W., Kang, C.-H., Kim, S., & Imm, J.-Y. (2021). Effects of Probiotic Culture Supernatant on Cariogenic Biofilm Formation and RANKL-Induced Osteoclastogenesis in RAW 264.7 Macrophages. Molecules, 26(3), 733. https://doi.org/10.3390/molecules26030733






