Acridine Based Small Molecular Hole Transport Type Materials for Phosphorescent OLED Application
Abstract
:1. Introduction
2. Results
3. Materials and Methods
3.1. Materials
3.2. Instrumentation
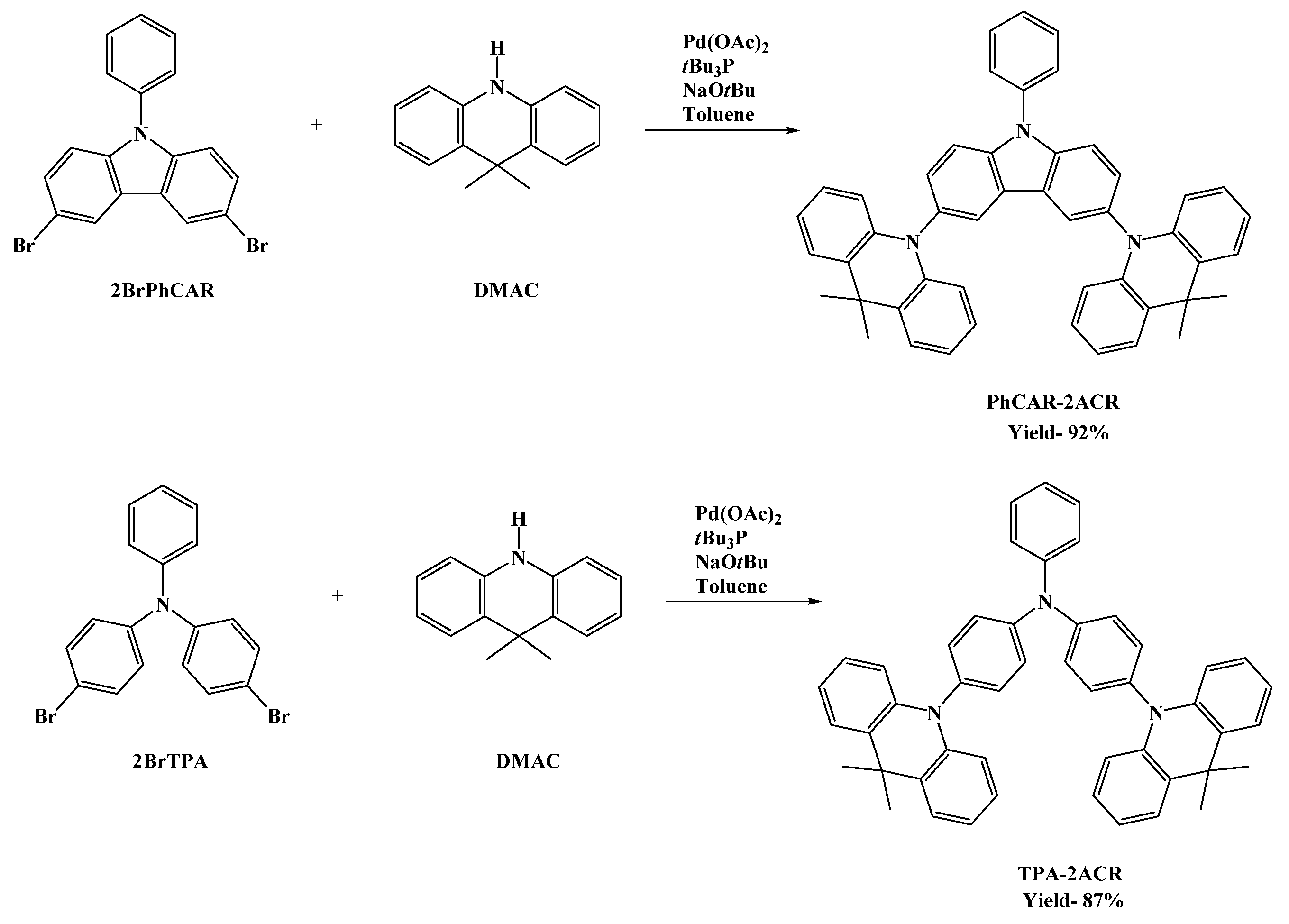
3.3. Synthesis and Characterization of 10,10′-(9-Phenyl-9H-carbazole-3,6-diyl)bis(9,9-dimethyl-9,10-dihydroacridine) PhCAR-2ACR
3.4. Synthesis and Characterization of 4-(9,9-Dimethylacridin-10(9H)-yl)-N-(4-(9,9-dimethylacridin-10(9H)-yl)phenyl)-N-phenylaniline TPA-2ACR
3.5. Device Fabrication
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Long, L.; Zhang, M.; Xu, S.; Zhou, X.; Gao, X.; Shang, Y.; Wei, B. Cyclic arylamines functioning as advanced hole-transporting and emitting materials. Synth. Met. 2012, 162, 448–452. [Google Scholar] [CrossRef]
- D’Andrade, B.W.; Forrest, S.R. White organic light-emitting devices for solid-state lighting. Adv. Mater. 2004, 16, 1585–1595. [Google Scholar] [CrossRef]
- Forrest, S.R. The path to ubiquitous and low-cost organic electronic appliances on plastic. Nature 2004, 428, 911. [Google Scholar] [CrossRef]
- Baldo, M.A.; O’Brien, D.F.; You, Y.; Shoustikov, A.; Sibley, S.; Thompson, M.E.; Forrest, S.R. Highly efficient phosphorescent emission from organic electroluminescent devices. Nature 1998, 395, 151–154. [Google Scholar] [CrossRef]
- Ito, T.; Sasabe, H.; Nagai, Y.; Watanabe, Y.; Onuma, N.; Kido, J. A series of dibenzofuran-based n-type exciplex host partners realizing high-efficiency and stable deep-red phosphorescent oleds. Chem.—Eur. J. 2019, 25, 7308–7314. [Google Scholar] [CrossRef] [PubMed]
- Hide, F.; DÍaz-GarcÍa, M.A.; Schwartz, B.J.; Heeger, A.J. New developments in the photonic applications of conjugated polymers. Acc. Chem. Res. 1997, 30, 430–436. [Google Scholar] [CrossRef]
- Tang, C.W.; VanSlyke, S.A. Organic electroluminescent diodes. Appl. Phys. Lett. 1987, 51, 913–915. [Google Scholar] [CrossRef]
- Murawski, C.; Leo, K.; Gather, M.C. Efficiency Roll-Off in Organic Light-Emitting Diodes. Adv. Mater. 2013, 25, 6801–6827. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, Z.; Liu, Z.; Yang, C.; Zhong, C.; Qin, J.; Yu, G.; Liu, Y. Multifunctional Fluorene-Based Oligomers with Novel Spiro-Annulated Triarylamine: Efficient, Stable Deep-Blue Electroluminescence, Good Hole Injection, and Transporting Materials with Very High Tg. Adv. Funct. Mater. 2009, 19, 3987–3995. [Google Scholar] [CrossRef]
- Cho, Y.J.; Lee, J.Y. Thermally stable aromatic amine derivative with symmetrically substituted double spirobifluorene core as a hole transport material for green phosphorescent organic light-emitting diodes. Thin Solid Film. 2012, 522, 415–419. [Google Scholar] [CrossRef]
- Zou, Y.; Ye, T.; Ma, D.; Qin, J.; Yang, C. Star-shaped hexakis(9,9-dihexyl-9H-fluoren-2-yl) benzene end-capped with carbazole and diphenylamine units: Solution-processable, high Tg hole-transporting materials for organic light-emitting devices. J. Mater. Chem. 2012, 22, 23485–23491. [Google Scholar] [CrossRef]
- Li, Z.; Wu, Z.; Fu, W.; Wang, D.; Liu, P.; Jiao, B.; Hao, Y. Stable amorphous bis(diarylamino) biphenyl derivatives as hole-transporting materials in OLEDs. Electron. Mater. Lett. 2013, 9, 655–661. [Google Scholar] [CrossRef]
- Huh, D.H.; Kim, G.W.; Kim, G.H.; Kulshreshtha, C.; Kwon, J.H. High hole mobility hole transport material for organic light-emitting devices. Synth. Met. 2013, 180, 79–84. [Google Scholar] [CrossRef]
- Shirota, Y.; Kageyama, H. Charge carrier transporting molecular materials and their applications in devices. Chem. Rev. 2007, 107, 953–1010. [Google Scholar] [CrossRef]
- He, Q.; Lin, H.; Weng, Y.; Zhang, B.; Wang, Z.; Lei, G.; Bai, F. A Hole-Transporting Material with Controllable Morphology Containing Binaphthyl and Triphenylamine Chromophores. Adv. Funct. Mater. 2006, 16, 1343–1348. [Google Scholar] [CrossRef]
- Baldo, M.A.; Lamansky, S.; Burrows, P.E.; Thompson, M.E.; Forrest, S.R. Very High-Efficiency Green Organic Light-Emitting Devices Based on Electrophosphorescence. Appl. Phys. Lett. 1999, 75, 4–6. [Google Scholar] [CrossRef]
- Wang, Z.; Li, M.; Gan, L.; Cai, X.; Li, B.; Chen, D.; Su, S.-J. Predicting operational stability for organic light-emitting diodes with exciplex cohosts. Adv. Sci. 2019, 6, 1802246. [Google Scholar] [CrossRef]
- Braveenth, R.; Ahn, D.H.; Han, J.-H.; Moon, J.S.; Kim, S.W.; Lee, H.; Qiong, W.; Kwon, J.H.; Chai, K.Y. Utilizing triazine/pyrimidine acceptor and carbazole-triphenylamine donor based bipolar novel host materials for highly luminescent green phosphorescent oleds with lower efficiency roll-off. Dye. Pigment. 2018, 157, 377–384. [Google Scholar] [CrossRef]
- Lee, D.R.; Lee, C.W.; Lee, J.Y. High triplet energy host materials for blue phosphorescent organic light-emitting diodes derived from carbazole modified orthophenylene. J. Mater. Chem. C 2014, 2, 7256–7263. [Google Scholar] [CrossRef]
- Serevičius, T.; Komskis, R.; Adomėnas, P.; Adomėnienė, O.; Kreiza, G.; Jankauskas, V.; Kazlauskas, K.; Miasojedovas, A.; Jankus, V.; Monkman, A.; et al. Triplet–triplet annihilation in 9,10-diphenylanthracene derivatives: The role of intersystem crossing and exciton diffusion. J. Phys. Chem. C 2017, 121, 8515–8524. [Google Scholar] [CrossRef]
- Liu, D.; Sun, K.; Zhao, G.; Wei, J.; Duan, J.; Xia, M.; Jiang, W.; Sun, Y. Spatial separation of a tadf sensitizer and fluorescent emitter with a core-dendron system to block the energy loss in deep blue organic light-emitting diodes. J. Mater. Chem. C 2019, 7, 11005–11013. [Google Scholar] [CrossRef]
- Braveenth, R.; Jung, H.; Kim, K.; Kim, B.M.; Bae, I.-J.; Kim, M.; Chai, K.Y. Fluorene–Triphenylamine-Based Bipolar Materials: Fluorescent Emitter and Host for Yellow Phosphorescent OLEDs. Appl. Sci. 2020, 10, 519. [Google Scholar] [CrossRef] [Green Version]
- Sasabe, H.; Toyota, N.; Nakanishi, H.; Ishizaka, T.; Pu, Y.J.; Kido, J. 3,3′-Bicarbazole-Based Host Materials for High-Efficiency Blue Phosphorescent OLEDs with Extremely Low Driving Voltage. Adv. Mater. 2012, 24, 3212–3217. [Google Scholar] [CrossRef]
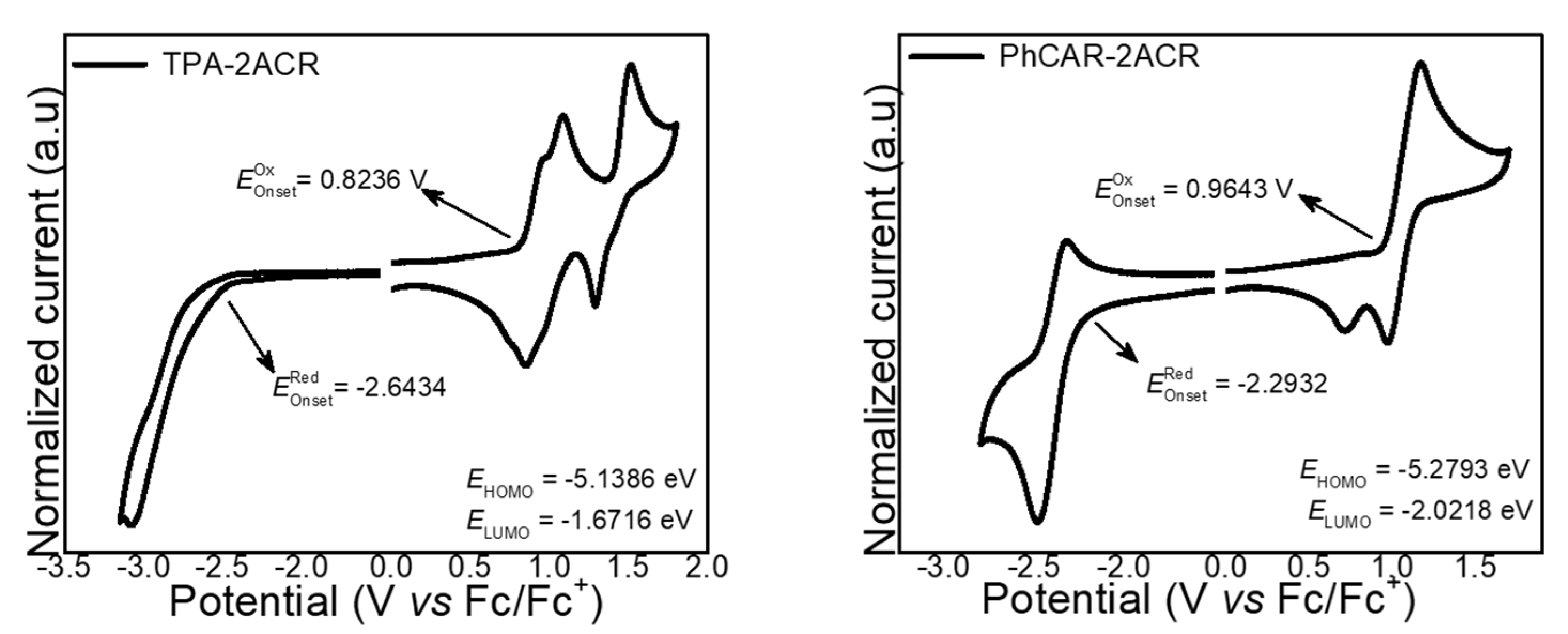
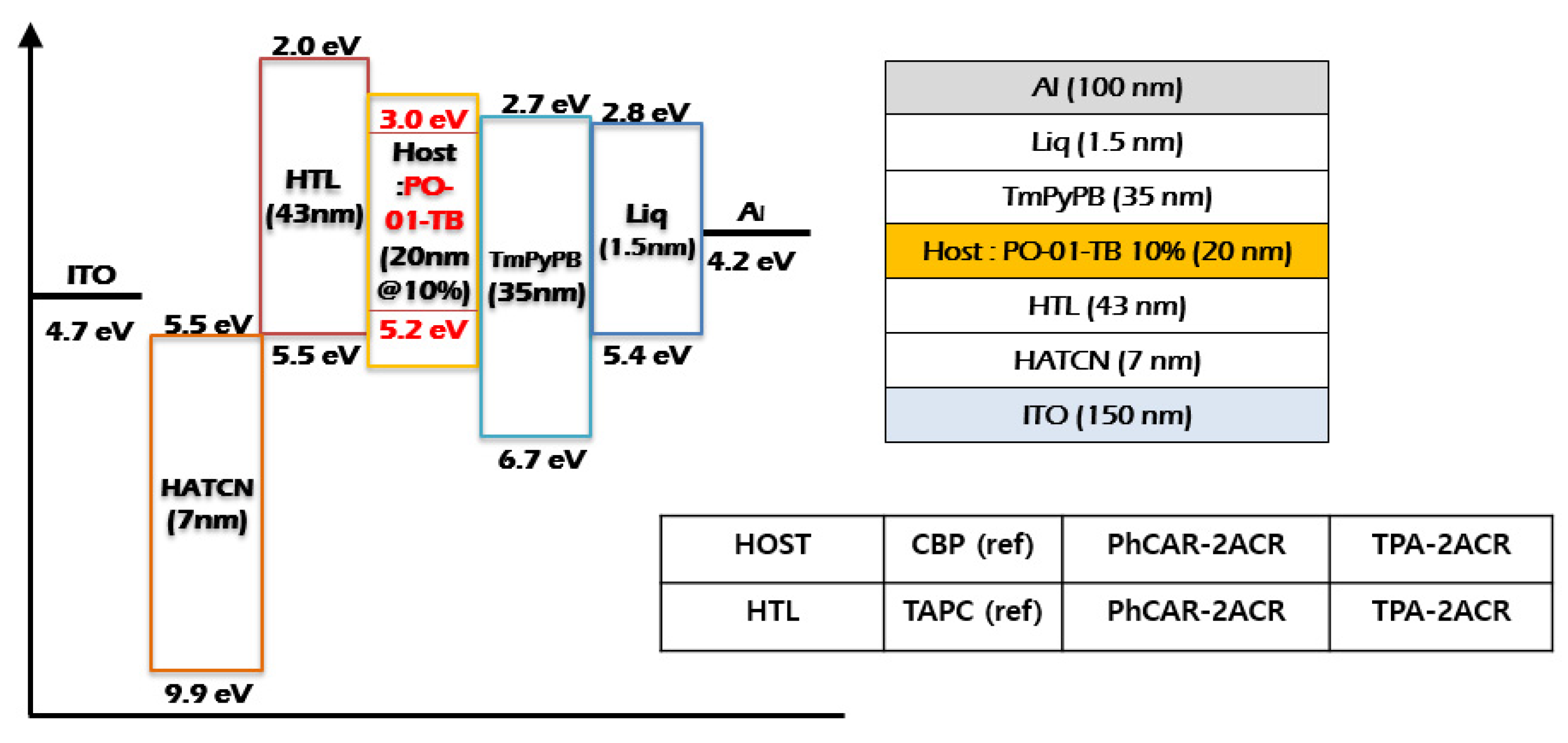
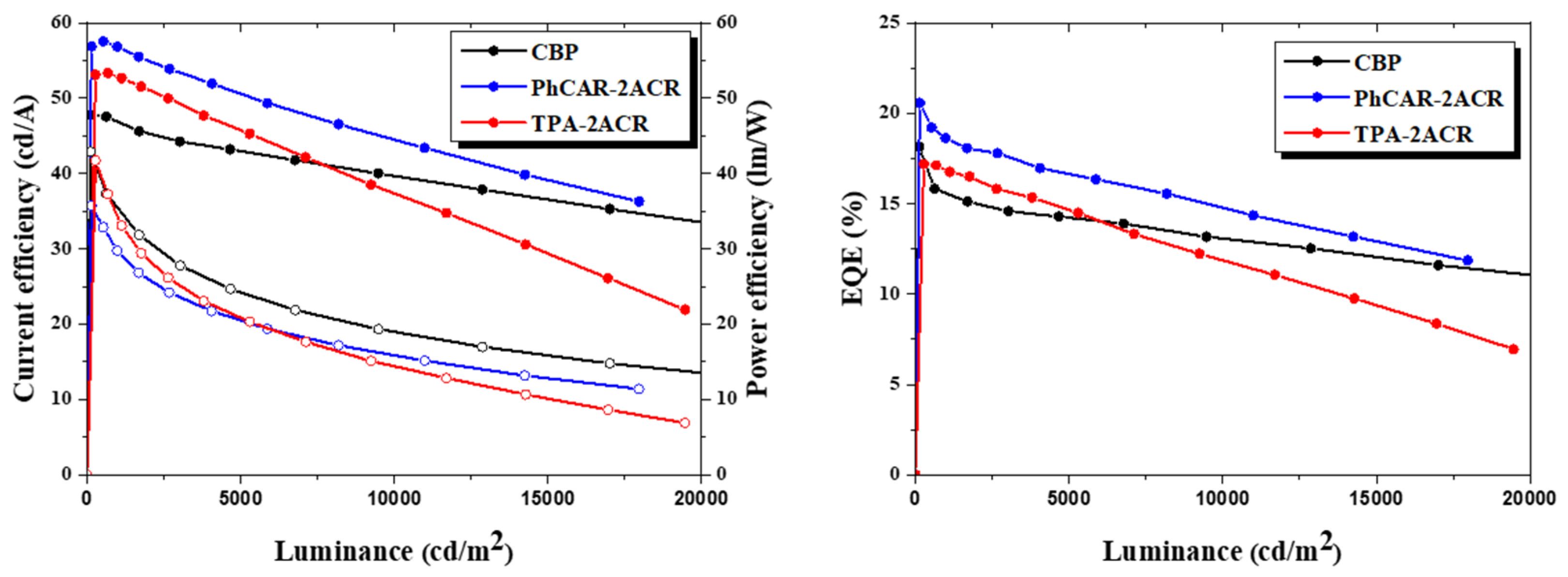
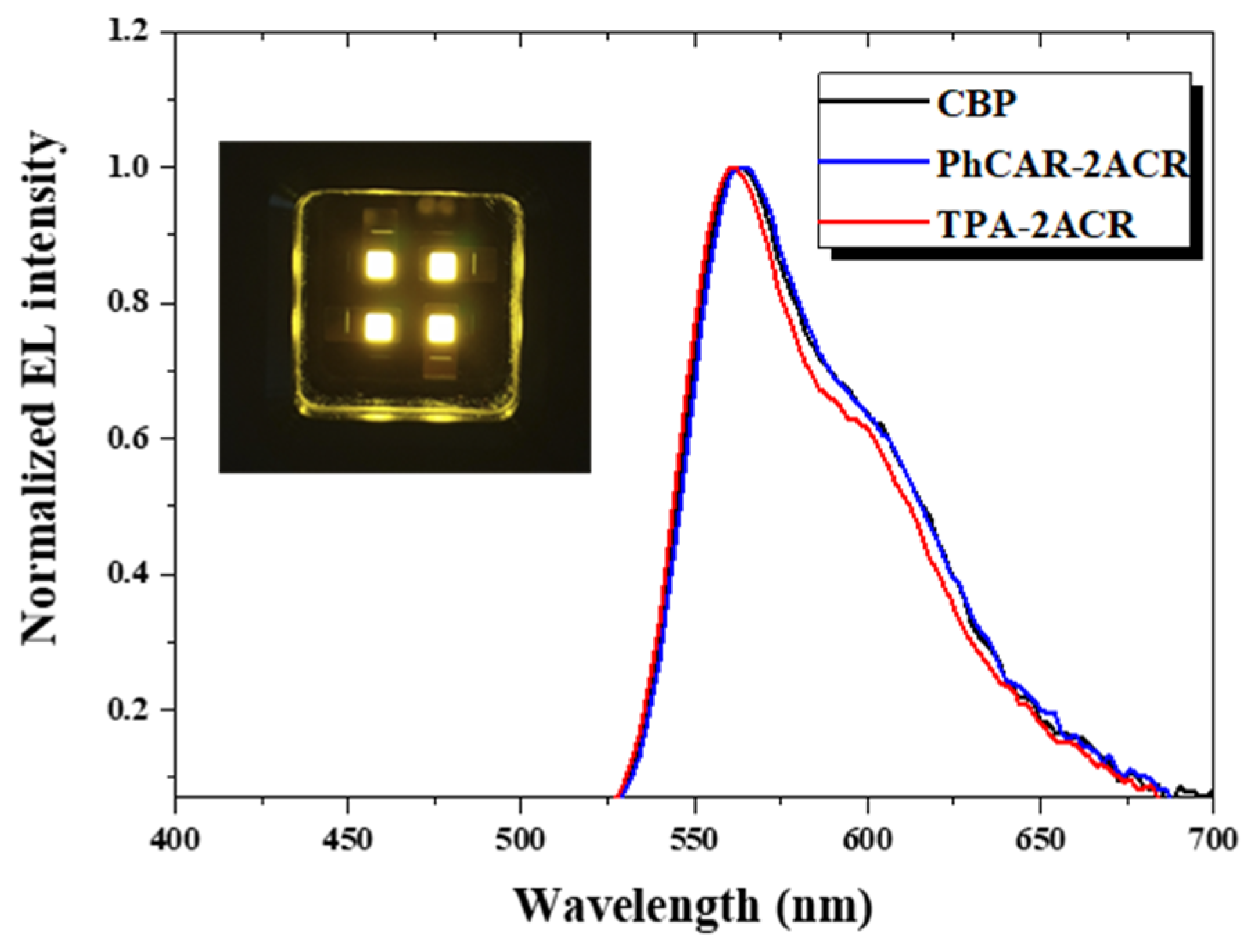
| Materials | PhCAR-2ACR | TPA-2ACR |
|---|---|---|
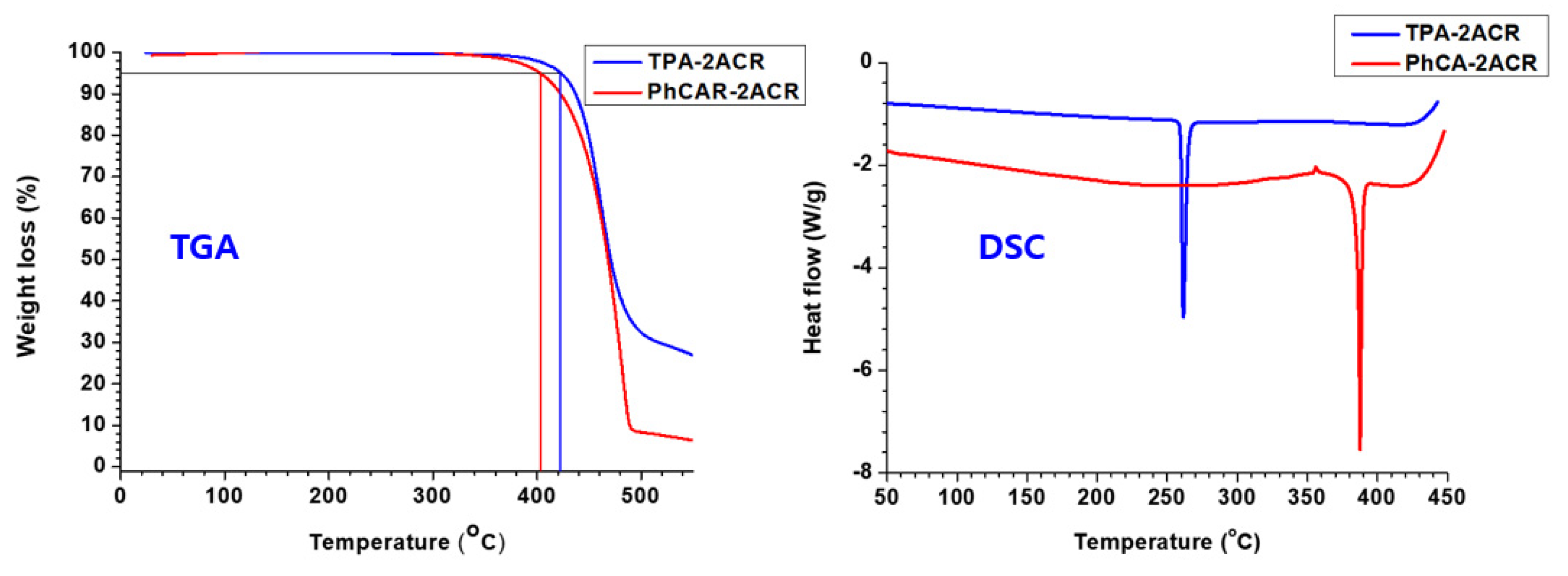
| Td (°C) | 402 | 422 |
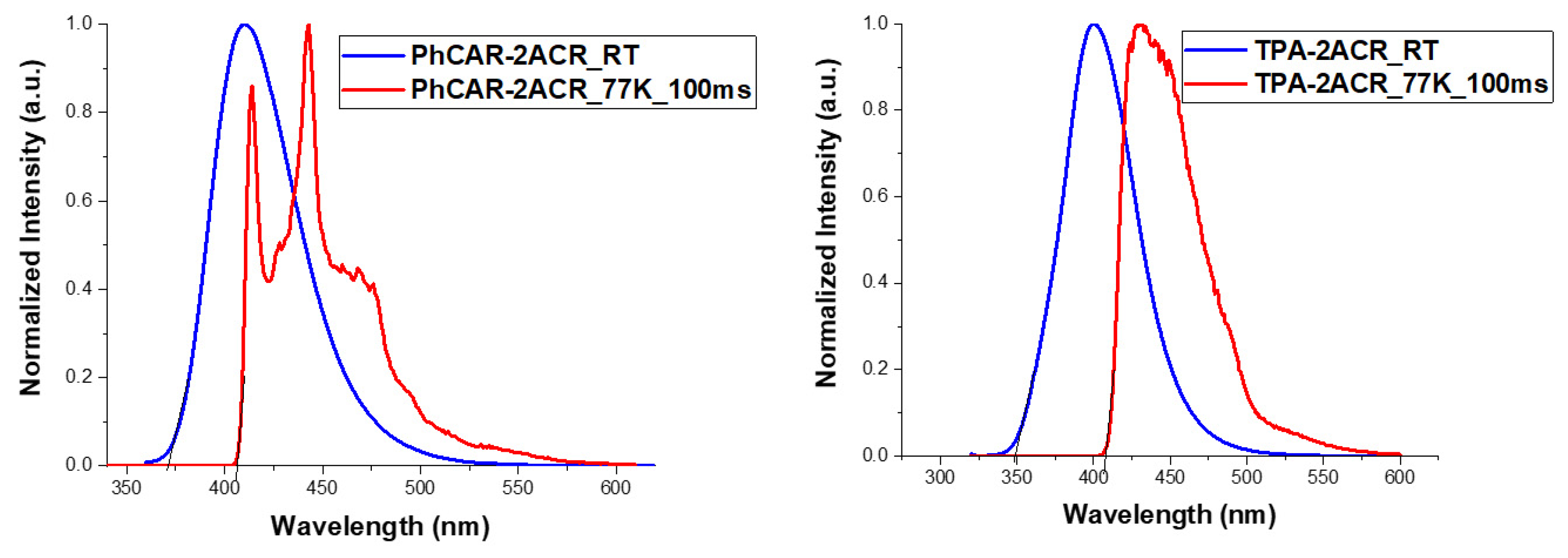
| PL (nm) | 403 | 401 |
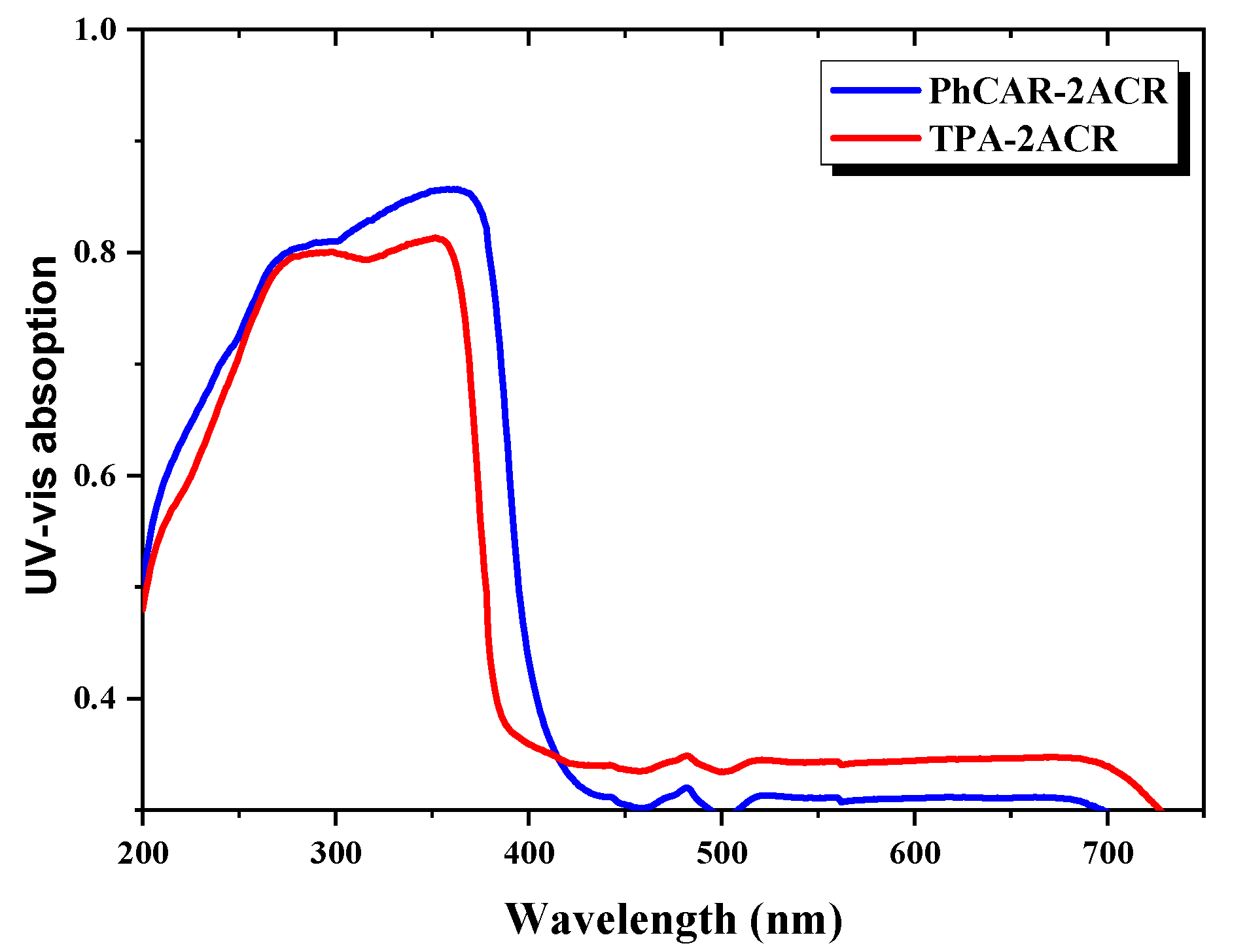
| Band gap (eV) | 2.89 | 2.96 |
| Triplet energy (eV) | 3.05 | 3.04 |
| HOMO (eV) | 5.27 | 5.13 |
| LUMO (eV) | 2.02 | 1.67 |
| Device Characteristics | PhCAR- 2ACR (HTL) | TPA- 2ACR (HTL) | TAPC (Reference) | PhCAR- 2ACR (Host) | TPA- 2ACR (Host) | CBP (Reference) |
|---|---|---|---|---|---|---|
| Current Efficiency (cd/A) | 23.13 | 55.74 | 32.53 | 56.90 | 53.15 | 47.83 |
| Power Efficiency (lm/W) | 6.05 | 29.28 | 18.58 | 35.75 | 41.74 | 42.94 |
| EQE (%) | 7.65 | 21.59 | 10.6 | 20.57 | 17.20 | 18.16 |
| CIE (x, y) | 0.49, 0.49 | 0.49, 0.49 | 0.49, 0.49 | 0.50, 0.50 | 0.49, 0.51 | 0.50, 0.50 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Braveenth, R.; Kim, K.; Bae, I.-J.; Raagulan, K.; Kim, B.M.; Kim, M.; Chai, K.Y. Acridine Based Small Molecular Hole Transport Type Materials for Phosphorescent OLED Application. Molecules 2021, 26, 7680. https://doi.org/10.3390/molecules26247680
Braveenth R, Kim K, Bae I-J, Raagulan K, Kim BM, Kim M, Chai KY. Acridine Based Small Molecular Hole Transport Type Materials for Phosphorescent OLED Application. Molecules. 2021; 26(24):7680. https://doi.org/10.3390/molecules26247680
Chicago/Turabian StyleBraveenth, Ramanaskanda, Keunhwa Kim, Il-Ji Bae, Kanthasamy Raagulan, Bo Mi Kim, Miyoung Kim, and Kyu Yun Chai. 2021. "Acridine Based Small Molecular Hole Transport Type Materials for Phosphorescent OLED Application" Molecules 26, no. 24: 7680. https://doi.org/10.3390/molecules26247680
APA StyleBraveenth, R., Kim, K., Bae, I.-J., Raagulan, K., Kim, B. M., Kim, M., & Chai, K. Y. (2021). Acridine Based Small Molecular Hole Transport Type Materials for Phosphorescent OLED Application. Molecules, 26(24), 7680. https://doi.org/10.3390/molecules26247680






