Nanostructured Photothermal Materials for Environmental and Catalytic Applications
Abstract
:1. Introduction
2. Photothermal Conversion
2.1. Solar Energy Utilization
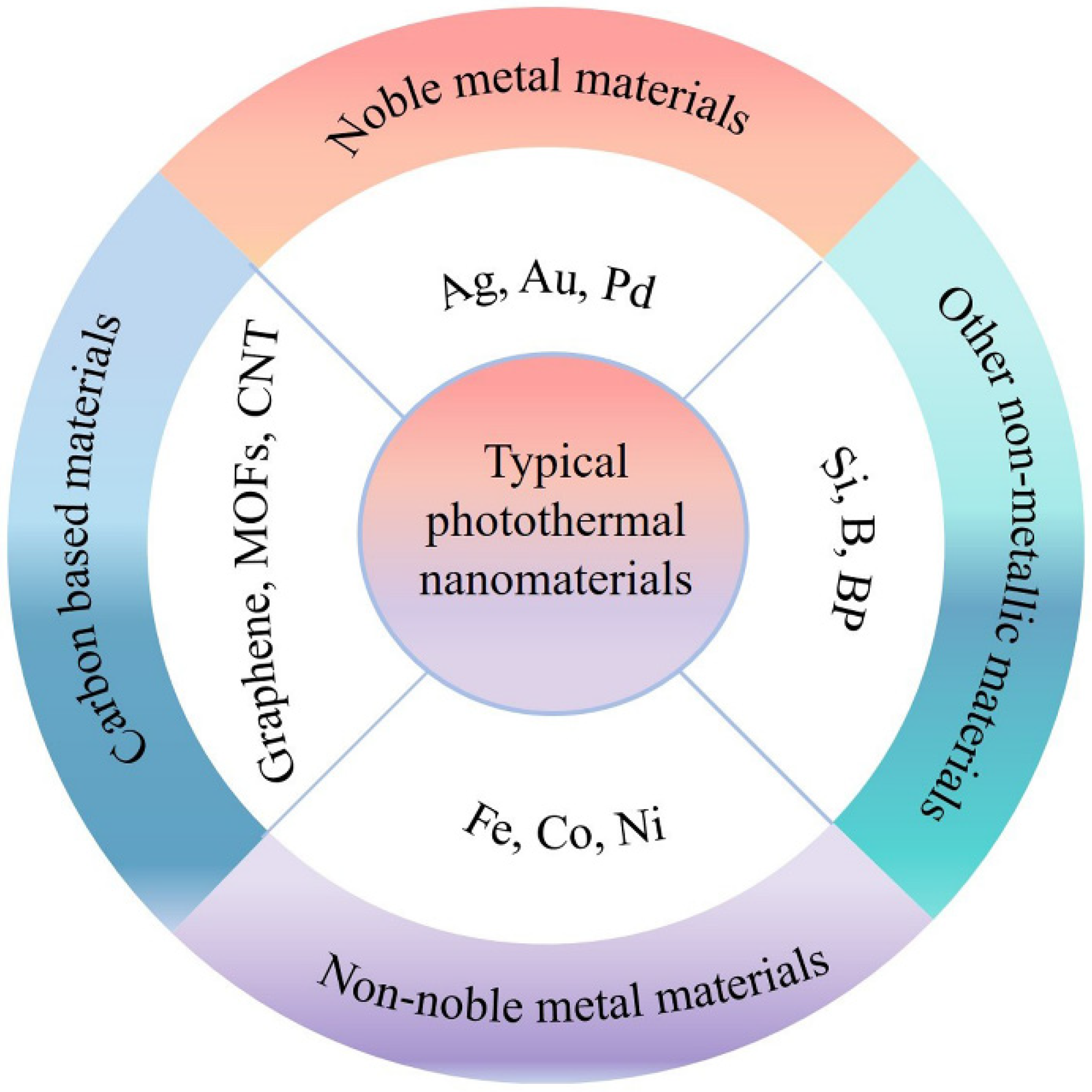
2.2. Photothermal Materials and Regulation Strategies
2.2.1. Metallic Materials
2.2.2. Metal-Free Materials
3. Photothermal Seawater Evaporation
3.1. Research Significance and Status
3.2. Two-Dimensional Nanostructures
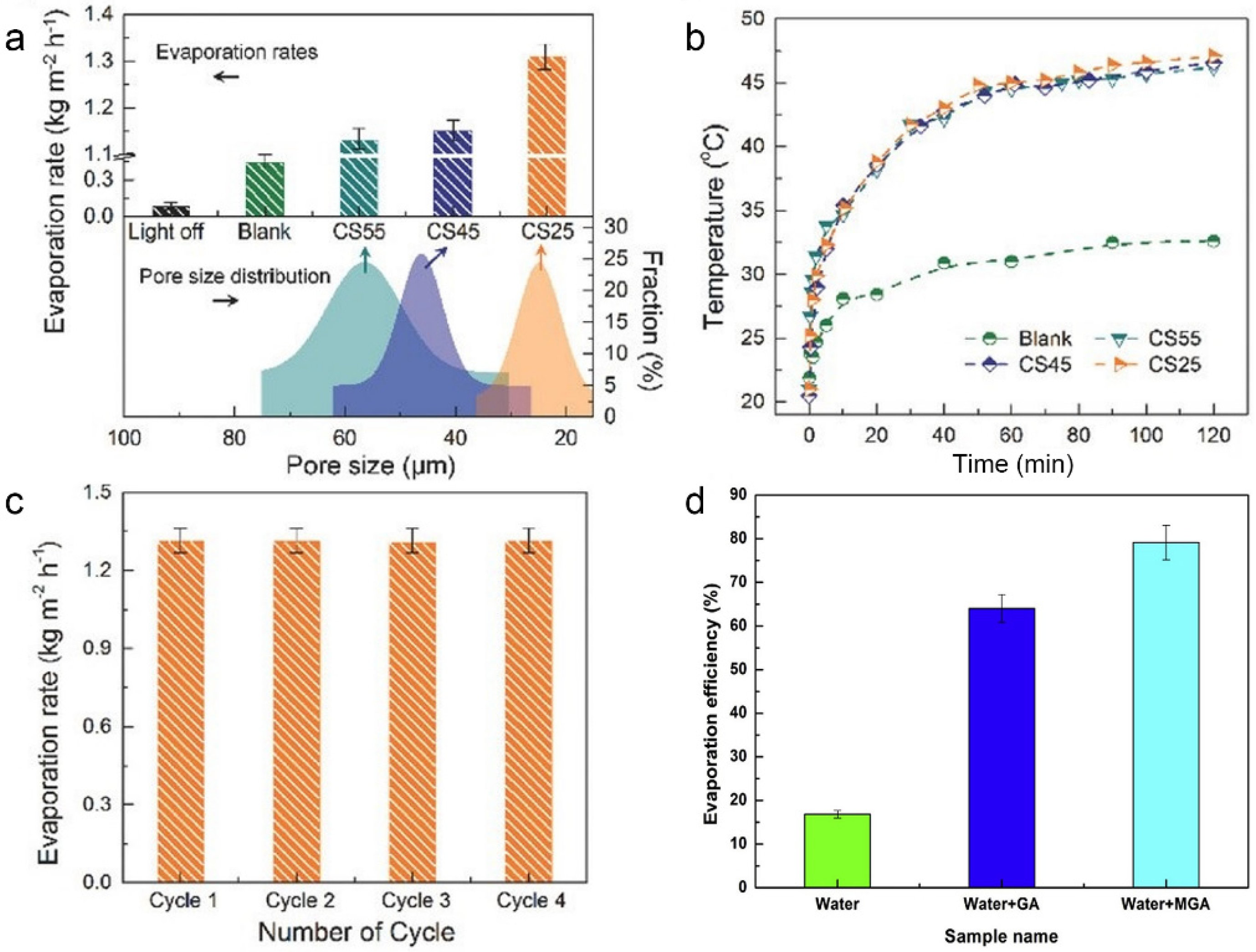
3.3. Three-Dimensional Porous Nanostructures
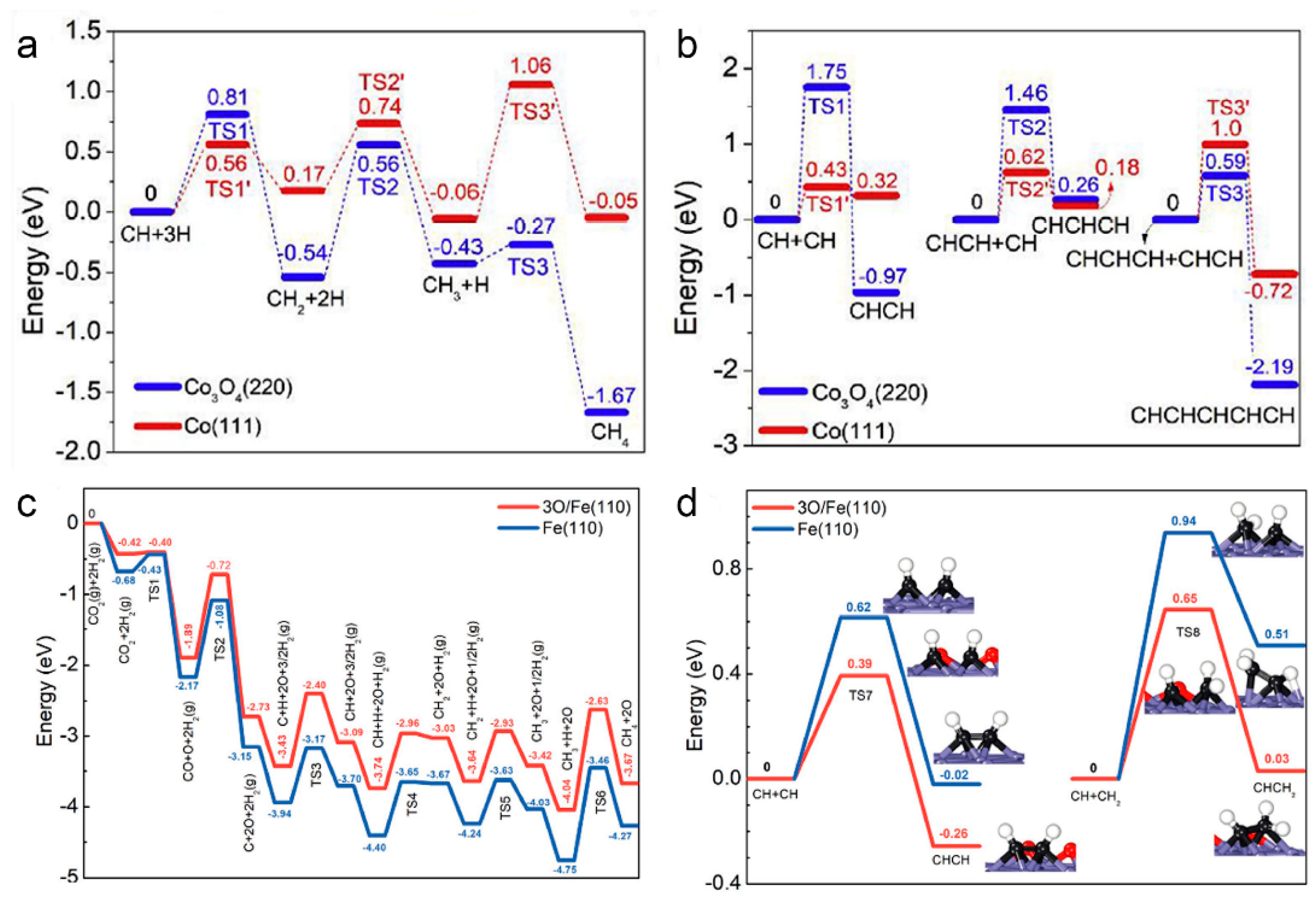
4. Photothermal Catalysis
4.1. Reaction Types
4.2. LDH Topological Transformation Nanostructures
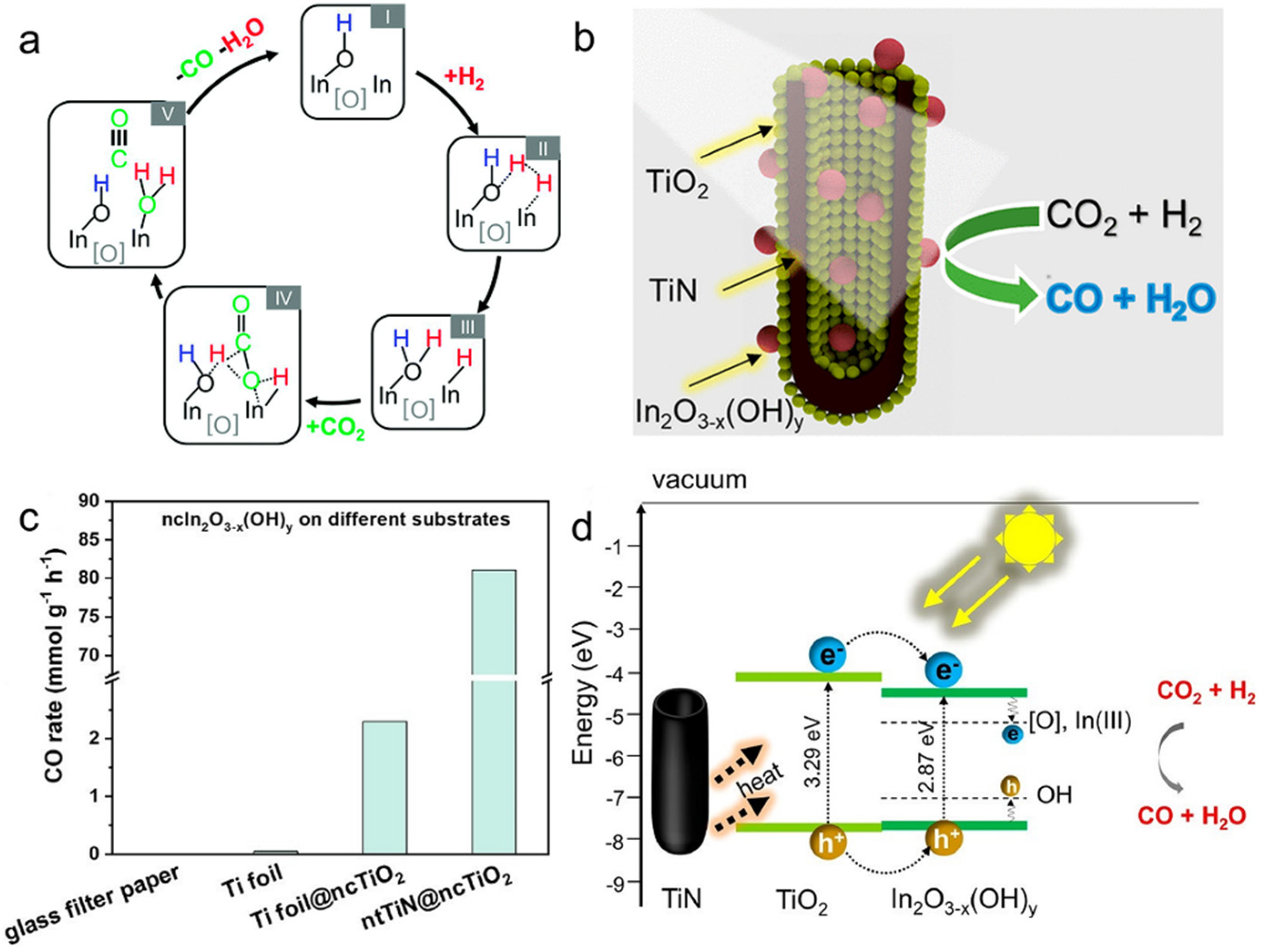
4.3. In2O3−x(OH)y Nanostructures
4.4. Metal Plasmonic Nanostructures
5. Conclusions and Future Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Jiang, X.; Nie, X.; Guo, X.; Song, C.; Chen, J.G. Recent advances in carbon dioxide hydrogenation to methanol via heterogeneous catalysis. Chem. Rev. 2020, 120, 7984–8034. [Google Scholar] [CrossRef] [PubMed]
- Kho, E.T.; Tan, T.H.; Lovell, E.; Wong, R.J.; Scott, J.; Amal, R. A review on photo-thermal catalytic conversion of carbon dioxide. Green Energy Environ. 2017, 2, 204–217. [Google Scholar] [CrossRef]
- Munday, P.L.; Dixson, D.L.; Welch, M.J.; Chivers, D.P.; Domenici, P.; Grosell, M.; Heuer, R.M.; Jones, G.P.; McCormick, M.I.; Meekan, M.; et al. Methods matter in repeating ocean acidification studies. Nature 2020, 586, E20–E24. [Google Scholar] [CrossRef]
- Mekhilef, S.; Saidur, R.; Safari, A. A review on solar energy use in industries. Renew. Sust. Energy Rev. 2011, 15, 1777–1790. [Google Scholar] [CrossRef]
- Alami, A.H.; Aokal, K. Enhancement of spectral absorption of solar thermal collectors by bulk graphene addition via high-pressure graphite blasting. Energy Convers. Manag. 2018, 156, 757–764. [Google Scholar] [CrossRef]
- Campos, C.; Vasco, D.; Angulo, C.; Burdiles, P.A.; Cardemil, J.; Palza, H. About the relevance of particle shape and graphene oxide on the behavior of direct absorption solar collectors using metal based nanofluids under different radiation intensities. Energy Convers. Manag. 2019, 181, 247–257. [Google Scholar] [CrossRef]
- Chandrashekara, M.; Yadav, A. Water desalination system using solar heat: A review. Renew. Sust. Energy Rev. 2017, 67, 1308–1330. [Google Scholar] [CrossRef]
- Shan, X.; Zhao, A.; Lin, Y.; Hu, Y.; Di, Y.; Liu, C.; Gan, Z. Low-cost, scalable, and reusable photothermal layers for highly efficient solar steam generation and versatile energy Conversion. Adv. Sustain. Syst. 2020, 4, 1900153. [Google Scholar] [CrossRef]
- Li, Z.; Liu, J.; Shi, R.; Waterhouse, G.I.N.; Wen, X.D.; Zhang, T. Fe-based catalysts for the direct photohydrogenation of CO2 to value-added hydrocarbons. Adv. Energy Mater. 2021, 11, 2002783. [Google Scholar] [CrossRef]
- Luo, S.; Ren, X.; Lin, H.; Song, H.; Ye, J. Plasmonic photothermal catalysis for solar-to-fuel conversion: Current status and prospects. Chem. Sci. 2021, 12, 5701–5719. [Google Scholar] [CrossRef]
- Du, J.; Du, Z.; Hu, J.-S.; Pan, Z.; Shen, Q.; Sung, J.; Long, D.; Dong, H.; Sun, L.; Zhong, X.; et al. Zn-Cu-In-Se quantum dot solar cells with a certified power conversion efficiency of 11.6%. J. Am. Chem. Soc. 2016, 138, 4201–4209. [Google Scholar] [CrossRef]
- Giordano, F.; Abate, A.; Baena, J.P.C.; Saliba, M.; Matsui, T.; Im, S.H.; Zakeeruddin, S.M.; Nazeeruddin, M.K.; Hagfeldt, A.; Graetzel, M. Enhanced electronic properties in mesoporous TiO2 via lithium doping for high-efficiency perovskite solar cells. Nat. Commun. 2016, 7, 10379. [Google Scholar] [CrossRef]
- Jiang, C.; Moniz, S.J.A.; Wang, A.; Zhang, T.; Tang, J. Photoelectrochemical devices for solar water splitting-materials and challenges. Chem. Soc. Rev. 2017, 46, 4645–4660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, C.; Cao, X.; Wu, X.-J.; He, Q.; Yang, J.; Zhang, X.; Chen, J.; Zhao, W.; Han, S.; Nam, G.-H.; et al. Recent advances in ultrathin two-dimensional nanomaterials. Chem. Rev. 2017, 117, 6225–6331. [Google Scholar] [CrossRef]
- Wang, P.; Guo, S.; Wang, H.J.; Chen, K.K.; Zhang, N.; Zhang, Z.M.; Lu, T.B. A broadband and strong visible-light-absorbing photosensitizer boosts hydrogen evolution. Nat. Commun. 2019, 10, 3155. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Li, C.; Domen, K. Recent developments in heterogeneous photocatalysts for solar-driven overall water splitting. Chem. Soc. Rev. 2019, 48, 2109–2125. [Google Scholar] [CrossRef]
- Xiong, X.; Mao, C.; Yang, Z.; Zhang, Q.; Waterhouse, G.I.N.; Gu, L.; Zhang, T. Photocatalytic CO2 reduction to CO over Ni single atoms supported on defect-rich zirconia. Adv. Energy Mater. 2020, 10, 2002928. [Google Scholar] [CrossRef]
- Zhao, Y.; Chen, G.; Bian, T.; Zhou, C.; Waterhouse, G.I.N.; Wu, L.-Z.; Tung, C.-H.; Smith, L.J.; O’Hare, D.; Zhang, T. Defect-rich ultrathin ZnAl-layered double hydroxide nanosheets for efficient photoreduction of CO2 to CO with water. Adv. Mater. 2015, 27, 7824–7831. [Google Scholar] [CrossRef]
- Zhang, S.; Zhao, Y.; Shi, R.; Zhou, C.; Waterhouse, G.I.N.; Wang, Z.; Weng, Y.; Zhang, T. Sub-3 nm ultrafine Cu2O for visible light driven nitrogen fixation. Angew. Chem. Int. Ed. 2021, 60, 2554–2560. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Chen, G.Y.; Owens, G.; Chu, D.; Xu, H. Photothermal materials: A key platform enabling highly efficient water evaporation driven by solar energy. Mater. Today Energy 2019, 12, 277–296. [Google Scholar] [CrossRef]
- Ghoussoub, M.; Xia, M.; Duchesne, P.N.; Segal, D.; Ozin, G. Principles of photothermal gas-phase heterogeneous CO2 catalysis. Energy Environ. Sci. 2019, 12, 1122–1142. [Google Scholar] [CrossRef]
- Li, Y.; Li, R.; Li, Z.; Wei, W.; Ouyang, S.; Yuan, H.; Zhang, T. Effect of support on catalytic performance of photothermal Fischer-Tropsch synthesis to produce lower olefins over Fe5C2-based catalysts. Chem. Res. Chin. Univ. 2020, 36, 1006–1012. [Google Scholar] [CrossRef]
- Marxer, D.; Furler, P.; Takacs, M.; Steinfeld, A. Solar thermochemical splitting of CO2 into separate streams of CO and O2 with high selectivity, stability, conversion, and efficiency. Energy Environ. Sci. 2017, 10, 1142–1149. [Google Scholar] [CrossRef] [Green Version]
- Mateo, D.; Cerrillo, J.L.; Durini, S.; Gascon, J. Fundamentals and applications of photo-thermal catalysis. Chem. Soc. Rev. 2021, 50, 2173–2210. [Google Scholar] [CrossRef]
- Fan, W.K.; Tahir, M. Recent developments in photothermal reactors with understanding on the role of light/heat for CO2 hydrogenation to fuels: A review. Chem. Eng. J. 2022, 427, 131617. [Google Scholar] [CrossRef]
- Liu, H.; Shi, L.; Zhang, Q.; Qi, P.; Zhao, Y.; Meng, Q.; Feng, X.; Wang, H.; Ye, J. Photothermal catalysts for hydrogenation reactions. Chem. Commun. 2021, 57, 1279–1294. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Duchesne, P.N.; Wang, L.; Song, R.; Xia, M.; Ulmer, U.; Sun, W.; Dong, Y.; Loh, J.Y.Y.; Kherani, N.P.; et al. High-performance, scalable, and low-cost copper hydroxyapatite for photothermal CO2 reduction. ACS Catal. 2020, 10, 13668–13681. [Google Scholar] [CrossRef]
- Zhang, F.; Li, Y.-H.; Qi, M.-Y.; Yamada, Y.M.A.; Anpo, M.; Tang, Z.-R.; Xu, Y.-J. Photothermal catalytic CO2 reduction over nanomaterials. Chem Catal. 2021, 1, 272–297. [Google Scholar] [CrossRef]
- Ibrahim, I.; Seo, D.H.; McDonagh, A.M.; Shon, H.K.; Tijing, L. Semiconductor photothermal materials enabling efficient solar steam generation toward desalination and wastewater treatment. Desalination 2021, 500, 114853. [Google Scholar] [CrossRef]
- Zhu, L.; Gao, M.; Peh, C.K.N.; Ho, G.W. Solar-driven photothermal nanostructured materials designs and prerequisites for evaporation and catalysis applications. Mater. Horiz. 2018, 5, 323–343. [Google Scholar] [CrossRef]
- Lewis, N.S. Research opportunities to advance solar energy utilization. Science 2016, 351, aad1920. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vinoth Kanna, I.; Pinky, D. Solar research—A review and recommendations for the most important supplier of energy for the earth with solar systems. Int. J. Ambient Energy 2018, 41, 962–968. [Google Scholar] [CrossRef]
- Crabtree, G.W.; Lewis, N.S. Solar energy conversion. Phys. Today 2007, 60, 37–42. [Google Scholar] [CrossRef]
- Panwar, N.L.; Kaushik, S.C.; Kothari, S. Role of renewable energy sources in environmental protection: A review. Renew. Sust. Energy Rev. 2011, 15, 1513–1524. [Google Scholar] [CrossRef]
- Habisreutinger, S.N.; Schmidt-Mende, L.; Stolarczyk, J.K. Photocatalytic reduction of CO2 on TiO2 and other semiconductors. Angew. Chem. Int. Ed. 2013, 52, 7372–7408. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.I.H.; Qurashi, A.; Nazeeruddin, M.K. Frontiers, opportunities, and challenges in perovskite solar cells: A critical review. J. Photochem. Photobiol. C 2018, 35, 1–24. [Google Scholar] [CrossRef]
- Hou, W.; Xiao, Y.; Han, G.; Lin, J.Y. The applications of polymers in solar cells: A review. Polymers 2019, 11, 143. [Google Scholar] [CrossRef] [Green Version]
- Yu, Z.; Liu, H.; Zhu, M.; Li, Y.; Li, W. Interfacial charge transport in 1D TiO2 based photoelectrodes for photoelectrochemical water splitting. Small 2021, 17, 1903378. [Google Scholar] [CrossRef]
- Zarei, E.; Ojani, R. Fundamentals and some applications of photoelectrocatalysis and effective factors on its efficiency: A review. J. Solid State Electrochem. 2016, 21, 305–336. [Google Scholar] [CrossRef]
- Wang, J.; Wei, Y.; Yang, B.; Wang, B.; Chen, J.; Jing, H. In situ grown heterojunction of Bi2WO6/BiOCl for efficient photoelectrocatalytic CO2 reduction. J. Catal. 2019, 377, 209–217. [Google Scholar] [CrossRef]
- Zhao, G.; Huang, X.; Wang, X.; Wang, X. Progress in catalyst exploration for heterogeneous CO2 reduction and utilization: A critical review. J. Mater. Chem. A 2017, 5, 21625–21649. [Google Scholar] [CrossRef]
- Wang, H.; Xia, Y.; Li, H.; Wang, X.; Yu, Y.; Jiao, X.; Chen, D. Highly active deficient ternary sulfide photoanode for photoelectrochemical water splitting. Nat. Commun. 2020, 11, 3078. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef]
- Maeda, K.; Domen, K. Photocatalytic water splitting: Recent progress and future challenges. J. Phys. Chem. Lett. 2010, 1, 2655–2661. [Google Scholar] [CrossRef]
- Yuan, L.; Qi, M.Y.; Tang, Z.R.; Xu, Y.J. Coupling strategy for CO2 valorization integrated with organic synthesis by heterogeneous photocatalysis. Angew. Chem. Int. Ed. 2021, 60, 21150–21172. [Google Scholar] [CrossRef]
- Zhang, W.; Mohamed, A.R.; Ong, W.J. Z-Scheme photocatalytic systems for carbon dioxide reduction: Where are we now? Angew. Chem. Int. Ed. 2020, 59, 22894–22915. [Google Scholar] [CrossRef] [PubMed]
- Shi, R.; Zhang, X.; Waterhouse, G.I.N.; Zhao, Y.; Zhang, T. The journey toward low temperature, low pressure catalytic nitrogen fixation. Adv. Energy Mater. 2020, 10, 2000659. [Google Scholar] [CrossRef] [Green Version]
- Lin, Y.; Xu, H.; Shan, X.; Di, Y.; Zhao, A.; Hu, Y.; Gan, Z. Solar steam generation based on the photothermal effect: From designs to applications, and beyond. J. Mater. Chem. A 2019, 7, 19203–19227. [Google Scholar] [CrossRef]
- Lu, Q.; Yang, Y.; Feng, J.; Wang, X. Oxygen-defected molybdenum oxides hierarchical nanostructure constructed by atomic-level thickness nanosheets as an efficient absorber for solar steam generation. Sol. RRL 2019, 3, 1800277. [Google Scholar] [CrossRef]
- Xie, Z.; Duo, Y.; Lin, Z.; Fan, T.; Xing, C.; Yu, L.; Wang, R.; Qiu, M.; Zhang, Y.; Zhao, Y.; et al. The rise of 2D photothermal materials beyond graphene for clean water production. Adv. Sci. 2020, 7, 1902236. [Google Scholar] [CrossRef] [Green Version]
- Zha, X.J.; Zhao, X.; Pu, J.H.; Tang, L.S.; Ke, K.; Bao, R.Y.; Bai, L.; Liu, Z.Y.; Yang, M.B.; Yang, W. Flexible anti-biofouling MXene/cellulose fibrous membrane for sustainable solar-driven water purification. ACS Appl. Mater. Interfaces 2019, 11, 36589–36597. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Liu, J.; Zhao, Y.; Shi, R.; Waterhouse, G.I.N.; Wang, Y.; Wu, L.-Z.; Tung, C.-H.; Zhang, T. Photothermal hydrocarbon synthesis using alumina-supported cobalt metal nanoparticle catalysts derived from layered-double-hydroxide nanosheets. Nano Energy 2019, 60, 467–475. [Google Scholar] [CrossRef]
- Ni, W.; Kou, X.; Yang, Z.; Wang, J. Tailoring longitudinal surface plasmon wavelengths, scattering and absorption cross sections of gold nanorods. ACS Nano 2008, 2, 677–686. [Google Scholar] [CrossRef] [PubMed]
- Boerigter, C.; Campana, R.; Morabito, M.; Linic, S. Evidence and implications of direct charge excitation as the dominant mechanism in plasmon-mediated photocatalysis. Nat. Commun. 2016, 7, 10545. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, X.; Tang, S.; Mu, X.; Dai, Y.; Chen, G.; Zhou, Z.; Ruan, F.; Yang, Z.; Zheng, N. Freestanding palladium nanosheets with plasmonic and catalytic properties. Nat. Nanotechnol. 2011, 6, 28–32. [Google Scholar] [CrossRef]
- Wang, F.; Li, C.; Chen, H.; Jiang, R.; Sun, L.D.; Li, Q.; Wang, J.; Yu, J.C.; Yan, C.H. Plasmonic harvesting of light energy for Suzuki coupling reactions. J. Am. Chem. Soc. 2013, 135, 5588–5601. [Google Scholar] [CrossRef]
- Chen, G.; Gao, R.; Zhao, Y.; Li, Z.; Waterhouse, G.I.N.; Shi, R.; Zhao, J.; Zhang, M.; Shang, L.; Sheng, G.; et al. Alumina-supported CoFe alloy catalysts derived from layered-double-hydroxide nanosheets for efficient photothermal CO2 hydrogenation to hydrocarbons. Adv. Mater. 2018, 30, 1704663. [Google Scholar] [CrossRef]
- Huang, H.; Mao, M.; Zhang, Q.; Li, Y.; Bai, J.; Yang, Y.; Zeng, M.; Zhao, X. Solar-light-driven CO2 reduction by CH4 on silica-cluster-modified Ni nanocrystals with a high solar-to-fuel efficiency and excellent durability. Adv. Energy Mater. 2018, 8, 1702472. [Google Scholar] [CrossRef]
- Tong, Z.; Yang, D.; Shi, J.; Nan, Y.; Sun, Y.; Jiang, Z. Three-dimensional porous aerogel constructed by g-C3N4 and graphene oxide nanosheets with excellent visible-light photocatalytic performance. ACS Appl. Mater. Interfaces 2015, 7, 25693–25701. [Google Scholar] [CrossRef]
- Zhou, S.; Shang, L.; Zhao, Y.; Shi, R.; Waterhouse, G.I.N.; Huang, Y.C.; Zheng, L.; Zhang, T. Pd single-atom catalysts on nitrogen-doped graphene for the highly selective photothermal hydrogenation of acetylene to ethylene. Adv. Mater. 2019, 31, 1900509. [Google Scholar] [CrossRef]
- Han, C.; Li, J.; Ma, Z.; Xie, H.; Waterhouse, G.I.N.; Ye, L.; Zhang, T. Black phosphorus quantum dot/g-C3N4 composites for enhanced CO2 photoreduction to CO. Sci. China Mater. 2018, 61, 1159–1166. [Google Scholar] [CrossRef] [Green Version]
- Liu, G.; Meng, X.; Zhang, H.; Zhao, G.; Pang, H.; Wang, T.; Li, P.; Kako, T.; Ye, J. Elemental boron for efficient carbon dioxide reduction under light irradiation. Angew. Chem. Int. Ed. 2017, 56, 5570–5574. [Google Scholar] [CrossRef] [PubMed]
- Hoch, L.B.; O’Brien, P.G.; Jelle, A.; Sandhel, A.; Perovic, D.D.; Mims, C.A.; Ozin, G.A. Nanostructured indium oxide coated silicon nanowire arrays: A hybrid photothermal/photochemical approach to solar fuels. ACS Nano 2016, 10, 9017–9025. [Google Scholar] [CrossRef]
- Wu, D.; Deng, K.; Hu, B.; Lu, Q.; Liu, G.; Hong, X. Plasmon-assisted photothermal catalysis of low-pressure CO2 hydrogenation to methanol over Pd/ZnO Catalyst. ChemCatChem 2019, 11, 1598–1601. [Google Scholar] [CrossRef]
- Deng, Z.; Zhou, J.; Miao, L.; Liu, C.; Peng, Y.; Sun, L.; Tanemura, S. The emergence of solar thermal utilization: Solar-driven steam generation. J. Mater. Chem. A 2017, 5, 7691–7709. [Google Scholar] [CrossRef]
- Lalisse, A.; Tessier, G.; Plain, J.; Baffou, G. Quantifying the efficiency of plasmonic materials for near-field enhancement and photothermal conversion. J. Phys. Chem. C 2015, 119, 25518–25528. [Google Scholar] [CrossRef]
- Song, H.; Meng, X.; Dao, T.D.; Zhou, W.; Liu, H.; Shi, L.; Zhang, H.; Nagao, T.; Kako, T.; Ye, J. Light-enhanced carbon dioxide activation and conversion by effective plasmonic coupling effect of Pt and Au nanoparticles. ACS Appl. Mater. Interfaces 2018, 10, 408–416. [Google Scholar] [CrossRef]
- Gao, P.; Li, S.; Bu, X.; Dang, S.; Liu, Z.; Wang, H.; Zhong, L.; Qiu, M.; Yang, C.; Cai, J.; et al. Direct conversion of CO2 into liquid fuels with high selectivity over a bifunctional catalyst. Nat. Chem. 2017, 9, 1019–1024. [Google Scholar] [CrossRef]
- Rodemerck, U.; Holeňa, M.; Wagner, E.; Smejkal, Q.; Barkschat, A.; Baerns, M. Catalyst development for CO2 hydrogenation to fuels. ChemCatChem 2013, 5, 1948–1955. [Google Scholar] [CrossRef]
- Wei, J.; Ge, Q.; Yao, R.; Wen, Z.; Fang, C.; Guo, L.; Xu, H.; Sun, J. Directly converting CO2 into a gasoline fuel. Nat. Commun. 2017, 8, 15174. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; de Tymowski, B.; Vigneron, F.; Florea, I.; Ersen, O.; Meny, C.; Nguyen, P.; Pham, C.; Luck, F.; Pham-Huu, C. Titania-decorated silicon carbide-containing cobalt catalyst for Fischer–Tropsch synthesis. ACS Catal. 2013, 3, 393–404. [Google Scholar] [CrossRef]
- Zhong, L.; Yu, F.; An, Y.; Zhao, Y.; Sun, Y.; Li, Z.; Lin, T.; Lin, Y.; Qi, X.; Dai, Y.; et al. Cobalt carbide nanoprisms for direct production of lower olefins from syngas. Nature 2016, 538, 84–87. [Google Scholar] [CrossRef]
- Xu, M.; Hu, X.; Wang, S.; Yu, J.; Zhu, D.; Wang, J. Photothermal effect promoting CO2 conversion over composite photocatalyst with high graphene content. J. Catal. 2019, 377, 652–661. [Google Scholar] [CrossRef]
- Wang, S.; Shang, L.; Li, L.; Yu, Y.; Chi, C.; Wang, K.; Zhang, J.; Shi, R.; Shen, H.; Waterhouse, G.I.; et al. Metal-organic-framework-derived mesoporous carbon nanospheres containing porphyrin-like metal centers for conformal phototherapy. Adv. Mater. 2016, 28, 8379–8387. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; He, Y.; Cheng, G.; Shi, L.; Liu, X.; Zhu, J. Direct vapor generation through localized solar heating via carbon-nanotube nanofluid. Energy Convers. Manag. 2016, 130, 176–183. [Google Scholar] [CrossRef]
- Gan, Z.; Wu, X.; Meng, M.; Zhu, X.; Yang, L.; Chu, P.K. Photothermal contribution to enhanced photocatalytic performance of graphene-based nanocomposites. ACS Nano 2014, 8, 9304–9310. [Google Scholar] [CrossRef]
- Song, J.; Wang, F.; Yang, X.; Ning, B.; Harp, M.G.; Culp, S.H.; Hu, S.; Huang, P.; Nie, L.; Chen, J.; et al. Gold nanoparticle coated carbon nanotube ring with enhanced raman scattering and photothermal conversion property for theranostic applications. J. Am. Chem. Soc. 2016, 138, 7005–7015. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Brien, P.G.; Sandhel, A.; Wood, T.E.; Jelle, A.A.; Hoch, L.B.; Perovic, D.D.; Mims, C.A.; Ozin, G.A. Photomethanation of gaseous CO2 over Ru/silicon nanowire catalysts with visible and near-infrared photons. Adv. Sci. 2014, 1, 1400001. [Google Scholar] [CrossRef]
- Zhao, H.; Chen, H.; Guo, Z.; Zhang, W.; Yu, H.; Zhuang, Z.; Zhong, H.; Liu, Z. In situ photothermal activation of necroptosis potentiates black phosphorus-mediated cancer photo-immunotherapy. Chem. Eng. J. 2020, 394, 124314. [Google Scholar] [CrossRef]
- Gude, V.G. Desalination and sustainability—An appraisal and current perspective. Water Res. 2016, 89, 87–106. [Google Scholar] [CrossRef]
- Shao, B.; Wang, Y.; Wu, X.; Lu, Y.; Yang, X.; Chen, G.Y.; Owens, G.; Xu, H. Stackable nickel–cobalt@polydopamine nanosheet based photothermal sponges for highly efficient solar steam generation. J. Mater. Chem. A 2020, 8, 11665–11673. [Google Scholar] [CrossRef]
- Li, R.; Zhang, L.; Shi, L.; Wang, P. MXene Ti3C2: An effective 2D light-to-heat conversion material. ACS Nano 2017, 11, 3752–3759. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghim, D.; Jiang, Q.; Cao, S.; Singamaneni, S.; Jun, Y.-S. Mechanically interlocked 1T/2H phases of MoS2 nanosheets for solar thermal water purification. Nano Energy 2018, 53, 949–957. [Google Scholar] [CrossRef]
- Yao, J.; Yang, G. An efficient solar-enabled 2D layered alloy material evaporator for seawater desalination. J. Mater. Chem. A 2018, 6, 3869–3876. [Google Scholar] [CrossRef]
- Yang, X.; Yang, Y.; Fu, L.; Zou, M.; Li, Z.; Cao, A.; Yuan, Q. An ultrathin flexible 2D membrane based on single-walled nanotube-MoS2 hybrid film for high-performance solar steam generation. Adv. Funct. Mater. 2018, 28, 1704505. [Google Scholar] [CrossRef]
- Li, K.; Chang, T.H.; Li, Z.; Yang, H.; Fu, F.; Li, T.; Ho, J.S.; Chen, P.Y. Biomimetic MXene textures with enhanced light-to-heat conversion for solar steam generation and wearable thermal management. Adv. Energy Mater. 2019, 9, 1901687. [Google Scholar] [CrossRef]
- Ming, X.; Guo, A.; Wang, G.; Wang, X. Two-dimensional defective tungsten oxide nanosheets as high performance photo-absorbers for efficient solar steam generation. Sol. Energy Mater. Sol. Cells 2018, 185, 333–341. [Google Scholar] [CrossRef]
- Yin, J.; You, X.; Zhang, Z.; Guo, Z.; Wang, J.; Wang, X. Boron nanosheets loaded with MoS2 porous sponges for water purification. J. Water Process. Eng. 2021, 41, 102048. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, X.; Fu, L.; Zou, M.; Cao, A.; Du, Y.; Yuan, Q.; Yan, C.-H. Two-dimensional flexible bilayer janus membrane for advanced photothermal water desalination. ACS Energy Lett. 2018, 3, 1165–1171. [Google Scholar] [CrossRef]
- Qu, W.; Zhao, H.; Zhang, Q.; Xia, D.; Tang, Z.; Chen, Q.; He, C.; Shu, D. Multifunctional Au/Ti3C2 photothermal membrane with antibacterial ability for stable and efficient solar water purification under the full spectrum. ACS Sustain. Chem. Eng. 2021, 9, 11372–11387. [Google Scholar] [CrossRef]
- Zhu, L.; Gao, M.; Peh, C.K.N.; Wang, X.; Ho, G.W. Self-contained monolithic carbon sponges for solar-driven interfacial water evaporation distillation and electricity generation. Adv. Energy Mater. 2018, 8, 1702149. [Google Scholar] [CrossRef]
- Liang, H.; Liao, Q.; Chen, N.; Liang, Y.; Lv, G.; Zhang, P.; Lu, B.; Qu, L. Thermal efficiency of solar steam generation approaching 100% through capillary water transport. Angew. Chem. Int. Ed. 2019, 58, 19041–19046. [Google Scholar] [CrossRef]
- Fu, Y.; Wang, G.; Ming, X.; Liu, X.; Hou, B.; Mei, T.; Li, J.; Wang, J.; Wang, X. Oxygen plasma treated graphene aerogel as a solar absorber for rapid and efficient solar steam generation. Carbon 2018, 130, 250–256. [Google Scholar] [CrossRef]
- Zhang, Z.; Mu, P.; He, J.; Zhu, Z.; Sun, H.; Wei, H.; Liang, W.; Li, A. Facile and scalable fabrication of surface-modified sponge for efficient solar steam generation. ChemSusChem 2019, 12, 426–433. [Google Scholar] [CrossRef]
- Hu, X.; Xu, W.; Zhou, L.; Tan, Y.; Wang, Y.; Zhu, S.; Zhu, J. Tailoring graphene oxide-based aerogels for efficient solar steam generation under one sun. Adv. Mater. 2017, 29, 160431. [Google Scholar] [CrossRef] [PubMed]
- Mu, P.; Zhang, Z.; Bai, W.; He, J.; Sun, H.; Zhu, Z.; Liang, W.; Li, A. Superwetting monolithic hollow-carbon-nanotubes aerogels with hierarchically nanoporous structure for efficient solar steam generation. Adv. Energy Mater. 2019, 9, 1802158. [Google Scholar] [CrossRef] [Green Version]
- Fan, X.; Yang, Y.; Shi, X.; Liu, Y.; Li, H.; Liang, J.; Chen, Y. A MXene-based hierarchical design enabling highly efficient and stable solar-water desalination with good salt resistance. Adv. Funct. Mater. 2020, 30, 2007110. [Google Scholar] [CrossRef]
- Wang, Z.; Yu, K.; Gong, S.; Mao, H.; Huang, R.; Zhu, Z. Cu3BiS3/MXenes with excellent solar-thermal conversion for continuous and efficient seawater desalination. ACS Appl. Mater. Interfaces 2021, 13, 16246–16258. [Google Scholar] [CrossRef]
- Zhang, J.; Luo, X.; Zhang, X.; Xu, Y.; Xu, H.; Zuo, J.; Liu, D.; Cui, F.; Wang, W. Three-dimensional porous photo-thermal fiber felt with salt-resistant property for high efficient solar distillation. Chin. Chem. Lett. 2021, 32, 1442–1446. [Google Scholar] [CrossRef]
- Yuan, J.; Lei, X.; Yi, C.; Jiang, H.; Liu, F.; Cheng, G.J. 3D-printed hierarchical porous cellulose/alginate/carbon black hydrogel for high-efficiency solar steam generation. Chem. Eng. J. 2022, 430, 132765. [Google Scholar] [CrossRef]
- Storer, D.P.; Phelps, J.L.; Wu, X.; Owens, G.; Khan, N.I.; Xu, H. Graphene and rice-straw-fiber-based 3D photothermal aerogels for highly efficient solar evaporation. ACS Appl. Mater. Interfaces 2020, 12, 15279–15287. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric field effect in atomically thin carbon films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, G.; Jin, W.; Xu, N. Two-dimensional-material membranes: A new family of high-performance separation membranes. Angew. Chem. Int. Ed. 2016, 55, 13384–13397. [Google Scholar] [CrossRef]
- Alhabeb, M.; Maleski, K.; Anasori, B.; Lelyukh, P.; Clark, L.; Sin, S.; Gogotsi, Y. Guidelines for synthesis and processing of two-dimensional titanium carbide (Ti3C2Tx MXene). Chem. Mater. 2017, 29, 7633–7644. [Google Scholar] [CrossRef]
- Xu, D.; Li, Z.; Li, L.; Wang, J. Insights into the photothermal conversion of 2D MXene nanomaterials: Synthesis, mechanism, and applications. Adv. Funct. Mater. 2020, 30, 2000712. [Google Scholar] [CrossRef]
- Wang, Z.; Xu, W.; Yu, K.; Gong, S.; Mao, H.; Huang, R.; Zhu, Z. NiS2 nanocubes coated Ti3C2 nanosheets with enhanced light-to-heat conversion for fast and efficient solar seawater steam generation. Sol. RRL 2021, 5, 2100183. [Google Scholar] [CrossRef]
- Xie, Z.; Peng, Y.-P.; Yu, L.; Xing, C.; Qiu, M.; Hu, J.; Zhang, H. Solar-inspired water purification based on emerging 2D materials: Status and challenges. Sol. RRL 2020, 4, 1900400. [Google Scholar] [CrossRef]
- Gu, Y.; Jiao, Y.; Zhou, X.; Wu, A.; Buhe, B.; Fu, H. Strongly coupled Ag/TiO2 heterojunctions for effective and stable photothermal catalytic reduction of 4-nitrophenol. Nano Res. 2018, 11, 126–141. [Google Scholar] [CrossRef]
- Nikitenko, S.I.; Chave, T.; Cau, C.; Brau, H.-P.; Flaud, V. Photothermal hydrogen production using noble-metal-free Ti@TiO2 core–shell nanoparticles under visible–NIR light irradiation. ACS Catal. 2015, 5, 4790–4795. [Google Scholar] [CrossRef]
- Song, R.; Luo, B.; Geng, J.; Song, D.; Jing, D. Photothermocatalytic hydrogen evolution over Ni2P/TiO2 for full-spectrum solar energy conversion. Ind. Eng. Chem. Res. 2018, 57, 7846–7854. [Google Scholar] [CrossRef]
- Wang, Z.J.; Song, H.; Liu, H.; Ye, J. Coupling of solar energy and thermal energy for carbon dioxide reduction: Status and prospects. Angew. Chem. Int. Ed. 2020, 59, 8016–8035. [Google Scholar] [CrossRef] [PubMed]
- Jia, J.; Wang, H.; Lu, Z.; O’Brien, P.G.; Ghoussoub, M.; Duchesne, P.; Zheng, Z.; Li, P.; Qiao, Q.; Wang, L.; et al. Photothermal catalyst engineering: Hydrogenation of gaseous CO2 with high activity and tailored selectivity. Adv. Sci. 2017, 4, 1700252. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Wang, Y.; Cheng, Y.; Liu, Z.; Guo, Q.; Ha, M.N.; Zhao, Z. Hydrogen-treated mesoporous WO3 as a reducing agent of CO2 to fuels (CH4 and CH3OH) with enhanced photothermal catalytic performance. J. Mater. Chem. A 2016, 4, 5314–5322. [Google Scholar] [CrossRef]
- Wang, L.; Ghoussoub, M.; Wang, H.; Shao, Y.; Sun, W.; Tountas, A.A.; Wood, T.E.; Li, H.; Loh, J.Y.Y.; Dong, Y.; et al. Photocatalytic hydrogenation of carbon dioxide with high selectivity to methanol at atmospheric pressure. Joule 2018, 2, 1369–1381. [Google Scholar] [CrossRef]
- Wang, Z.-J.; Song, H.; Pang, H.; Ning, Y.; Dao, T.D.; Wang, Z.; Chen, H.; Weng, Y.; Fu, Q.; Nagao, T.; et al. Photo-assisted methanol synthesis via CO2 reduction under ambient pressure over plasmonic Cu/ZnO catalysts. Appl. Catal. B 2019, 250, 10–16. [Google Scholar] [CrossRef]
- Liu, L.; Puga, A.V.; Cored, J.; Concepción, P.; Pérez-Dieste, V.; García, H.; Corma, A. Sunlight-assisted hydrogenation of CO2 into ethanol and C2+ hydrocarbons by sodium-promoted Co@C nanocomposites. Appl. Catal. B 2018, 235, 186–196. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, L.; Wang, K.; Khan, M.U.; Wang, M.; Li, H.; Zeng, J. Integration of photothermal effect and heat insulation to efficiently reduce reaction temperature of CO2 hydrogenation. Small 2017, 13, 1602583. [Google Scholar] [CrossRef]
- Han, B.; Wei, W.; Chang, L.; Cheng, P.; Hu, Y.H. Efficient visible light photocatalytic CO2 reforming of CH4. ACS Catal. 2016, 6, 494–497. [Google Scholar] [CrossRef]
- Liu, H.; Meng, X.; Dao, T.D.; Zhang, H.; Li, P.; Chang, K.; Wang, T.; Li, M.; Nagao, T.; Ye, J. Conversion of carbon dioxide by methane reforming under visible-light irradiation: Surface-plasmon-mediated nonpolar molecule activation. Angew. Chem. Int. Ed. 2015, 54, 11545–11549. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhao, B.; Liu, J.; Chen, G.; Gao, R.; Yao, S.; Li, M.; Zhang, Q.; Gu, L.; Xie, J.; et al. Oxide-modified nickel photocatalysts for the production of hydrocarbons in visible light. Angew. Chem. Int. Ed. 2016, 55, 4215–4219. [Google Scholar] [CrossRef]
- Li, Z.; Liu, J.; Zhao, Y.; Waterhouse, G.I.N.; Chen, G.; Shi, R.; Zhang, X.; Liu, X.; Wei, Y.; Wen, X.-D.; et al. Co-based catalysts derived from layered-double-hydroxide nanosheets for the photothermal production of light olefins. Adv. Mater. 2018, 30, 1800527. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Li, Z.; Li, M.; Liu, J.; Liu, X.; Waterhouse, G.I.N.; Wang, Y.; Zhao, J.; Gao, W.; Zhang, Z.; et al. Reductive transformation of layered-double-hydroxide nanosheets to Fe-based heterostructures for efficient visible-light photocatalytic hydrogenation of CO. Adv. Mater. 2018, 30, 1803127. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Su, X.; Li, L.; Qi, H.; Yang, C.; Liu, W.; Pan, X.; Liu, X.; Yang, X.; Huang, Y.; et al. Ru/TiO2 catalysts with size-dependent metal/support interaction for tunable reactivity in Fischer–Tropsch synthesis. ACS Catal. 2020, 10, 12967–12975. [Google Scholar] [CrossRef]
- Sastre, F.; Oteri, M.; Corma, A.; García, H. Photocatalytic water gas shift using visible or simulated solar light for the efficient, room-temperature hydrogen generation. Energy Environ. Sci. 2013, 6, 2211–2215. [Google Scholar] [CrossRef]
- Zhao, L.; Qi, Y.; Song, L.; Ning, S.; Ouyang, S.; Xu, H.; Ye, J. Solar-driven water–gas shift reaction over CuOx/Al2O3 with 1.1 % of light-to-energy storage. Angew. Chem. Int. Ed. 2019, 58, 7708–7712. [Google Scholar] [CrossRef]
- Cavani, F.; Trifirò, F.; Vaccari, A. Hydrotalcite-type anionic clays: Preparation, properties and applications. Catal. Today 1991, 11, 173–301. [Google Scholar] [CrossRef]
- Gursky, J.A.; Blough, S.D.; Luna, C.; Gomez, C.; Luevano, A.N.; Gardner, E.A. Particle-particle interactions between layered double hydroxide nanoparticles. J. Am. Chem. Soc. 2006, 128, 8376–8377. [Google Scholar] [CrossRef]
- Williams, G.R.; O’Hare, D. Towards understanding, control and application of layered double hydroxide chemistry. J. Mater. Chem. 2006, 16, 3065–3074. [Google Scholar] [CrossRef]
- Zhao, Y.; Jia, X.; Waterhouse, G.I.N.; Wu, L.-Z.; Tung, C.-H.; O’Hare, D.; Zhang, T. Layered double hydroxide nanostructured photocatalysts for renewable energy production. Adv. Energy Mater. 2016, 6, 1501974. [Google Scholar] [CrossRef]
- Ren, J.; Ouyang, S.; Xu, H.; Meng, X.; Wang, T.; Wang, D.; Ye, J. Targeting activation of CO2 and H2 over Ru-loaded ultrathin layered double hydroxides to achieve efficient photothermal CO2 methanation in flow-type system. Adv. Energy Mater. 2017, 7, 1601657. [Google Scholar] [CrossRef]
- Li, Z.; Shi, R.; Zhao, J.; Zhang, T. Ni-based catalysts derived from layered-double-hydroxide nanosheets for efficient photothermal CO2 reduction under flow-type system. Nano Res. 2021, 14, 4828–4832. [Google Scholar] [CrossRef]
- Hoch, L.B.; Wood, T.E.; O’Brien, P.G.; Liao, K.; Reyes, L.M.; Mims, C.A.; Ozin, G.A. The rational design of a single-component photocatalyst for gas-phase CO2 reduction using both UV and visible Light. Adv. Sci. 2014, 1, 1400013. [Google Scholar] [CrossRef]
- Ghuman, K.K.; Wood, T.E.; Hoch, L.B.; Mims, C.A.; Ozin, G.A.; Singh, C.V. Illuminating CO2 reduction on frustrated lewis pair surfaces: Investigating the role of surface hydroxides and oxygen vacancies on nanocrystalline In2O3−x(OH)y. Phys. Chem. Chem. Phys. 2015, 17, 14623–14635. [Google Scholar] [CrossRef]
- Nguyen, N.T.; Xia, M.; Duchesne, P.N.; Wang, L.; Mao, C.; Jelle, A.A.; Yan, T.; Li, P.; Lu, Z.H.; Ozin, G.A. Enhanced CO2 photocatalysis by indium oxide hydroxide supported on TiN@TiO2 nanotubes. Nano Lett. 2021, 21, 1311–1319. [Google Scholar] [CrossRef]
- Liu, H.; Song, H.; Zhou, W.; Meng, X.; Ye, J. A promising application of optical hexagonal TaN in photocatalytic reactions. Angew. Chem. Int. Ed. 2018, 57, 16781–16784. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Dao, T.D.; Liu, L.; Meng, X.; Nagao, T.; Ye, J. Light assisted CO2 reduction with methane over group VIII metals: Universality of metal localized surface plasmon resonance in reactant activation. Appl. Catal. B 2017, 209, 183–189. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, T.; Wang, J.; Liu, H.; Dao, T.D.; Li, M.; Liu, G.; Meng, X.; Chang, K.; Shi, L.; et al. Surface-plasmon-enhanced photodriven CO2 reduction catalyzed by metal–organic-framework-derived iron nanoparticles encapsulated by ultrathin carbon layers. Adv. Mater. 2016, 28, 3703–3710. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Meng, X.; Wang, Z.-J.; Wang, Z.; Chen, H.; Weng, Y.; Ichihara, F.; Oshikiri, M.; Kako, T.; Ye, J. Visible-light-mediated methane activation for steam methane reforming under mild conditions: A case study of Rh/TiO2 catalysts. ACS Catal. 2018, 8, 7556–7565. [Google Scholar] [CrossRef]


| Types of Nanostructures | Photothermal Materials | Solar Illumination (kW m−2) | Evaporation Rate/Efficiency | Reference |
|---|---|---|---|---|
| 2D nanostructure | Ti3C2/PVDF membrane | 1 | 84% | [82] |
| MoS2 nanosheets | 0.76 | 76% | [83] | |
| BiInSe3@CF | 1 | 1.1 kg m−2 h−1 | [84] | |
| SWNT−MoS2 film | 5 | 6.6 kg m−2 h−1 | [85] | |
| MoOx HNS | 1 | 1.05 kg m−2 h−1 | [49] | |
| Ti3C2Tx/cellulose membrane | 1 | 1.44 kg m−2 h−1 | [51] | |
| biomimetic Mxene | 1 | 1.33 kg m−2 h−1 | [86] | |
| 2D defective WOx nanosheets | 1 | 78.6% | [87] | |
| boron nanosheets modified with MoS2 | 1 | 1.538 kg m−2 h−1 | [88] | |
| SWNT/AuNR Janus film | 5 | 94% | [89] | |
| Au/Ti3C2 membrane | 2 | 2.66 kg m−2 h−1 | [90] | |
| 3D nanostructure | cellular nitrogen-enriched CS | 1 | 1.31 kg m−2 h−1 | [91] |
| 3D graphene foam | 1 | 2.40 kg m−2 h−1 | [92] | |
| modified graphene aerogel | 1 | 76.9% | [93] | |
| graphite-modified sponge | 1 | 73.3% | [94] | |
| RGO−SA−CNT aerogel | 1 | 1.622 kg m−2 h−1 | [95] | |
| hollow carbon nanotubes aerogels | 1 | 86.8% | [96] | |
| Co−CNS/MXenes foam | 1 | 93.39% | [97] | |
| Cu3BiS3/MXenes | 1 | 1.32 kg m−2 h−1 | [98] | |
| photo-thermal fiber felt | 1 | 1.48 kg m−2 h−1 | [99] | |
| cellulose/alginate/carbon black hydrogel | 1 | 1.33 kg m−2 h−1 | [100] | |
| RGO−SA−cellulose aerogel | 1 | 2.25 kg m−2 h−1 | [101] |
| Reaction Type | Catalyst | Reaction Temperature (°C) | Rate/Selectivity | Reference | ||
|---|---|---|---|---|---|---|
| CO | CH4 | C2+ | ||||
| CO conversion | Fe−500 | 230 | - | 28.6% | 60% | [122] |
| Co−700 | 210 | - | 35% | 65% | [52] | |
| Co−450 | 195 | - | 48% | 52% | [121] | |
| CO2 conversion | CoFe-650 | 310 | 4.97% | 59.77% | 35.26% | [57] |
| Ru@FL-LDHs | 350 | - | 277 mmol g−1 h−1 | - | [130] | |
| Fe−500 | 275 | - | 47.1% | 52.9% | [9] | |
| Ni−600 | 290 | - | 278.8 mmol·g−1 h−1 | - | [131] | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, H.; Shi, R.; Zhang, T. Nanostructured Photothermal Materials for Environmental and Catalytic Applications. Molecules 2021, 26, 7552. https://doi.org/10.3390/molecules26247552
Chen H, Shi R, Zhang T. Nanostructured Photothermal Materials for Environmental and Catalytic Applications. Molecules. 2021; 26(24):7552. https://doi.org/10.3390/molecules26247552
Chicago/Turabian StyleChen, Huige, Run Shi, and Tierui Zhang. 2021. "Nanostructured Photothermal Materials for Environmental and Catalytic Applications" Molecules 26, no. 24: 7552. https://doi.org/10.3390/molecules26247552
APA StyleChen, H., Shi, R., & Zhang, T. (2021). Nanostructured Photothermal Materials for Environmental and Catalytic Applications. Molecules, 26(24), 7552. https://doi.org/10.3390/molecules26247552







