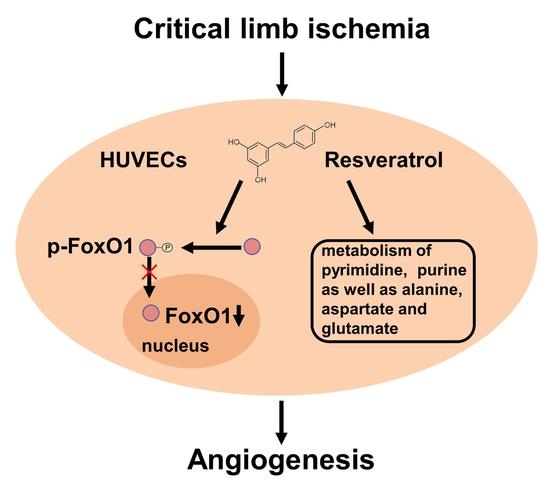
Resveratrol Promotes Angiogenesis in a FoxO1-Dependent Manner in Hind Limb Ischemia in Mice
Abstract
:1. Introduction
2. Results
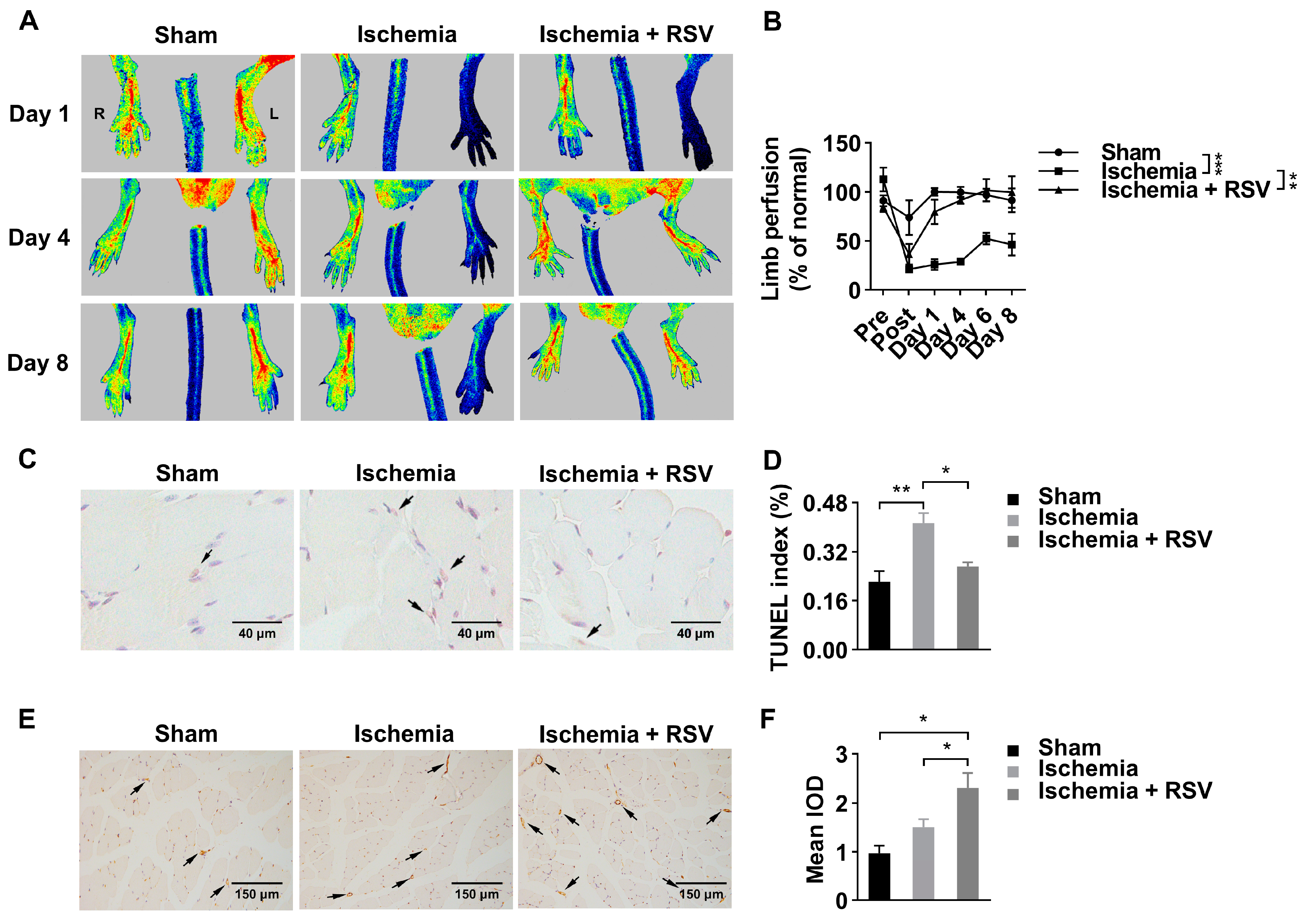
2.1. RSV Treatment Improves Blood Restoration in Hind Limb Ischemia Mouse Model
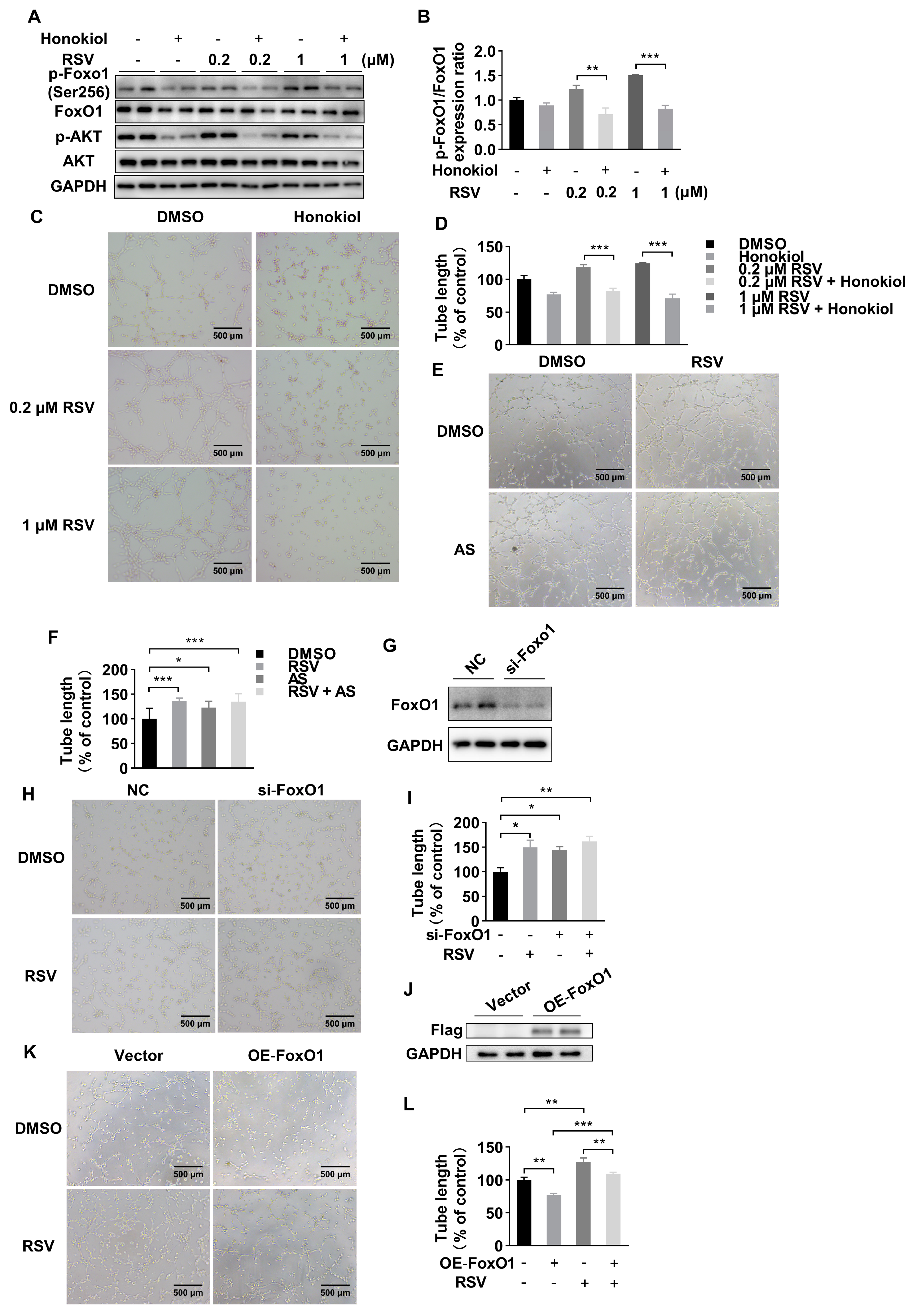
2.2. RSV Inhibits FoxO1 Pathway in Ischemic Muscle and HUVECs
2.3. RSV Promotes Angiogenesis in a FoxO1-Dependent Manner
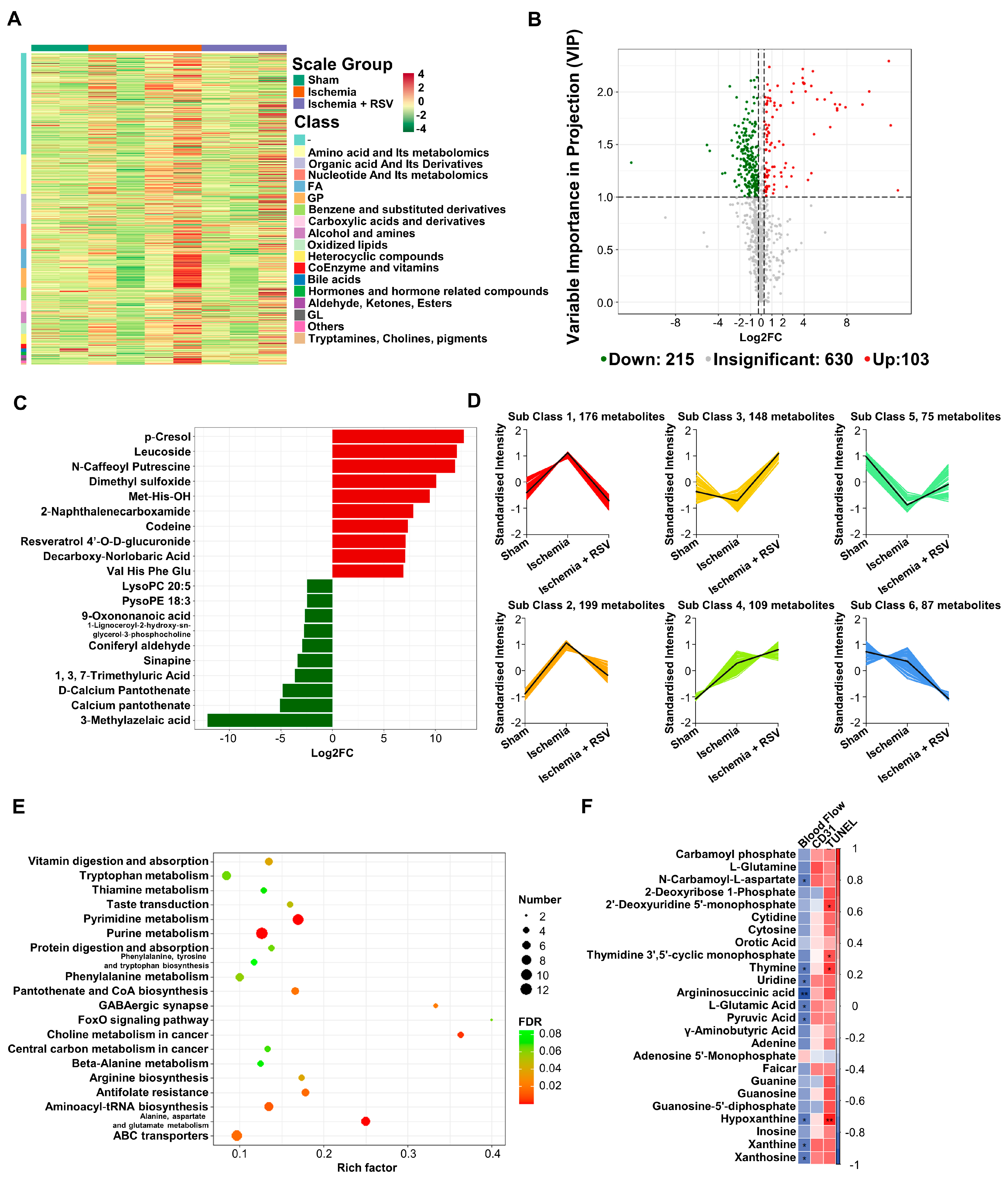
2.4. Differential Metabolites Analysis of Skeletal Muscle Samples
3. Discussion
4. Materials and Methods
4.1. Drug and Animals
4.2. Hind Limb Ischemia Model Establishment and Function Assessment
4.3. Terminal-Deoxynucleotidyl Transferase Mediated Nick End Labeling (TUNEL) Assay
4.4. Immunohistochemical Staining
4.5. Metabolomics Analysis
4.6. Cell Culture and Ischemia Treatments
4.7. Matrigel Tube Formation Assay
4.8. Cell Transfection
4.9. Western Blot
4.10. Statistics
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Murray, C.J.L.; Barber, R.M.; Foreman, K.J.; Ozgoren, A.A.; Abd-Allah, F.; Abera, S.F.; Aboyans, V.; Abraham, J.P.; Abubakar, I.; Abu-Raddad, L.J.; et al. Global, regional, and national disability-adjusted life years (DALYs) for 306 diseases and injuries and healthy life expectancy (HALE) for 188 countries, 1990–2013: Quantifying the epidemiological transition. Lancet 2015, 386, 2145–2191. [Google Scholar] [CrossRef] [Green Version]
- Fowkes, F.G.R.; Rudan, D.; Rudan, I.; Aboyans, V.; Denenberg, J.O.; McDermott, M.M.; Norman, P.E.; Sampson, U.K.; Williams, L.J.; Mensah, G.A.; et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: A systematic review and analysis. Lancet 2013, 382, 1329–1340. [Google Scholar] [CrossRef]
- Norgren, L.; Hiatt, W.R.; Dormandy, J.A.; Nehler, M.R.; Harris, K.A.; Fowkes, F.G.R. Inter-Society Consensus for the Management of Peripheral Arterial Disease (TASC II). J. Vasc. Surg. 2007, 45 (Suppl. S), S5–S67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cerqueira, L.D.O.; Júnior, E.G.D.; Barros, A.L.D.S.; Cerqueira, J.R.; de Araújo, W.J.B. WIfI classification: The Society for Vascular Surgery lower extremity threatened limb classification system, a literature review. J. Vasc. Bras. 2020, 19, e20190070. [Google Scholar] [CrossRef] [PubMed]
- Beckman, J.A.; Schneider, P.A.; Conte, M.S. Advances in Revascularization for Peripheral Artery Disease: Revascularization in PAD. Circ. Res. 2021, 128, 1885–1912. [Google Scholar] [CrossRef] [PubMed]
- Bonaca, M.P.; Hamburg, N.M.; Creager, M.A. Contemporary Medical Management of Peripheral Artery Disease. Circ. Res. 2021, 128, 1868–1884. [Google Scholar] [CrossRef]
- Takeshita, S.; Zheng, L.P.; Brogi, E.; Kearney, M.; Pu, L.Q.; Bunting, S.; Ferrara, N.; Symes, J.F.; Isner, J.M. Therapeutic angiogenesis. A single intraarterial bolus of vascular endothelial growth factor augments revascularization in a rabbit ischemic hind limb model. J. Clin. Investig. 1994, 93, 662–670. [Google Scholar] [CrossRef]
- Atluri, P.; Woo, Y.J. Pro-Angiogenic Cytokines as Cardiovascular Therapeutics: Assessing the potential. BioDrugs: Clinical immunotherapeutics, biopharmaceuticals and gene therapy. BioDrugs 2008, 22, 209–222. [Google Scholar] [CrossRef]
- Zhao, L.; Johnson, T.; Liu, D. Therapeutic angiogenesis of adipose-derived stem cells for ischemic diseases. Stem Cell Res. Ther. 2017, 8, 125. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Chen, X.; Luo, Q.; Liu, X.; Wang, X.; Cui, Z.; He, A.; He, S.; Jiang, Z.; Wu, N.; et al. Retinoblastoma cell-derived exosomes promote angiogenesis of human vesicle endothelial cells through microRNA-92a-3p. Cell Death Dis. 2021, 12, 695. [Google Scholar] [CrossRef]
- Nakagami, H.; Maeda, K.; Morishita, R.; Iguchi, S.; Nishikawa, T.; Takami, Y.; Kikuchi, Y.; Saito, Y.; Tamai, K.; Ogihara, T.; et al. Novel Autologous Cell Therapy in Ischemic Limb Disease Through Growth Factor Secretion by Cultured Adipose Tissue–Derived Stromal Cells. Arter. Thromb. Vasc. Biol. 2005, 25, 2542–2547. [Google Scholar] [CrossRef]
- Johnson, T.K.; Zhao, L.; Zhu, D.; Wang, Y.; Xiao, Y.; Oguljahan, B.; Zhao, X.; Kirlin, W.G.; Yin, L.; Chilian, W.M.; et al. Exosomes derived from induced vascular progenitor cells promote angiogenesis in vitro and in an in vivo rat hindlimb ischemia model. Am. J. Physiol. Heart Circ. Physiol. 2019, 317, H765–H776. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Duan, J.; Guo, C.; Wei, G.; Wang, Y.; Guan, Y.; Mu, F.; Yao, M.; Xi, M.; Wen, A. Danshensu accelerates angiogenesis after myocardial infarction in rats and promotes the functions of endothelial progenitor cells through SDF-1α/CXCR4 axis. Eur. J. Pharmacol. 2017, 814, 274–282. [Google Scholar] [CrossRef]
- Zhang, K.; Lu, J.; Mori, T.; Smith-Powell, L.; Synold, T.W.; Chen, S.; Wen, W. Baicalin increases VEGF expression and angiogenesis by activating the ERR/PGC-1 pathway. Cardiovasc. Res. 2010, 89, 426–435. [Google Scholar] [CrossRef] [Green Version]
- Weiskirchen, S.; Weiskirchen, R. Resveratrol: How Much Wine Do You Have to Drink to Stay Healthy? Adv. Nutr. 2016, 7, 706–718. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Sun, J.; Chen, G.; Niu, C.; Wang, Y.; Zhao, C.; Sun, J.; Huang, H.; Huang, S.; Liang, Y.; et al. Resveratrol Promotes Diabetic Wound Healing via SIRT1-FOXO1-c-Myc Signaling Pathway-Mediated Angiogenesis. Front. Pharmacol. 2019, 10, 421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rong, Y.; Ren, J.; Song, W.; Xiang, R.; Ge, Y.; Lu, W.; Fu, T. Resveratrol Suppresses Severe Acute Pancreatitis-Induced Microcirculation Disturbance through Targeting SIRT1-FOXO1 Axis. Oxidative Med. Cell. Longev. 2021, 2021, 8891544. [Google Scholar] [CrossRef] [PubMed]
- Delmas, D.; Cornebise, C.; Courtaut, F.; Xiao, J.; Aires, V. New Highlights of Resveratrol: A Review of Properties against Ocular Diseases. Int. J. Mol. Sci. 2021, 22, 1295. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, P.; Fnu, G.; Bhatia, D.; Shahid, A.; Sutariya, V. Nanodelivery of Resveratrol-Loaded PLGA Nanoparticles for Age-Related Macular Degeneration. AAPS PharmSciTech 2020, 21, 291. [Google Scholar] [CrossRef]
- Böhmdorfer, M.; Szakmary, A.; Schiestl, R.H.; Vaquero, J.; Riha, J.; Brenner, S.; Thalhammer, T.; Szekeres, T.; Jäger, W. Involvement of UDP-Glucuronosyltransferases and Sulfotransferases in the Excretion and Tissue Distribution of Resveratrol in Mice. Nutrients 2017, 9, 1347. [Google Scholar] [CrossRef] [Green Version]
- Springer, M.; Moco, S. Resveratrol and Its Human Metabolites—Effects on Metabolic Health and Obesity. Nutrients 2019, 11, 143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wen, D.; Huang, X.; Zhang, M.; Zhang, L.; Chen, J.; Gu, Y.; Hao, C.-M. Resveratrol Attenuates Diabetic Nephropathy via Modulating Angiogenesis. PLoS ONE 2013, 8, e82336. [Google Scholar] [CrossRef] [PubMed]
- Hu, W.-H.; Duan, R.; Xia, Y.-T.; Xiong, Q.-P.; Wang, H.-Y.; Chan, G.K.-L.; Liu, S.-Y.; Dong, T.T.-X.; Qin, Q.-W.; Tsim, K.W.-K. Binding of Resveratrol to Vascular Endothelial Growth Factor Suppresses Angiogenesis by Inhibiting the Receptor Signaling. J. Agric. Food Chem. 2018, 67, 1127–1137. [Google Scholar] [CrossRef]
- Liu, D.; Song, G.; Ma, Z.; Geng, X.; Dai, Y.; Yang, T.; Meng, H.; Gong, J.; Zhou, B.; Song, Z. Resveratrol improves the therapeutic efficacy of bone marrow-derived mesenchymal stem cells in rats with severe acute pancreatitis. Int. Immunopharmacol. 2020, 80, 106128. [Google Scholar] [CrossRef] [PubMed]
- Simão, F.; Pagnussat, A.; Seo, J.H.; Navaratna, D.; Leung, W.; Lok, J.; Guo, S.; Waeber, C.; Salbego, C.G.; Lo, E.H. Pro-Angiogenic Effects of Resveratrol in Brain Endothelial Cells: Nitric Oxide-Mediated Regulation of Vascular Endothelial Growth Factor and Metalloproteinases. Br. J. Pharmacol. 2012, 32, 884–895. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roudier, E.; Milkiewicz, M.; Birot, O.; Slopack, D.; Montelius, A.; Gustafsson, T.; Paik, J.H.; DePinho, R.; Casale, G.P.; Pipinos, I.I.; et al. Endothelial FoxO1 is an intrinsic regulator of thrombospondin 1 expression that restrains angiogenesis in ischemic muscle. Angiogenesis 2013, 16, 759–772. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Gan, L.; Pan, H.; Kan, D.; Majeski, M.; Adam, S.; Unterman, T.G. Multiple elements regulate nuclear/cytoplasmic shuttling of FOXO1: Characterization of phosphorylation- and 14-3-3-dependent and -independent mechanisms. Biochem. J. 2004, 378, 839–849. [Google Scholar] [CrossRef] [Green Version]
- Zhu, L.; Hu, B.; Guo, Y.; Yang, H.; Zheng, J.; Yao, X.; Hu, H.; Liu, H. Effect of Chitosan oligosaccharides on ischemic symptom and gut microbiota disbalance in mice with hindlimb ischemia. Carbohydr. Polym. 2020, 240, 116271. [Google Scholar] [CrossRef]
- Senesi, P.; Ferrulli, A.; Luzi, L.; Terruzzi, I. Diabetes Mellitus and Cardiovascular Diseases: Nutraceutical Interventions Related to Caloric Restriction. Int. J. Mol. Sci. 2021, 22, 7772. [Google Scholar] [CrossRef]
- Liang, L.; Liu, X.; Wang, Q.; Cheng, S.; Zhang, S.; Zhang, M. Pharmacokinetics, tissue distribution and excretion study of resveratrol and its prodrug 3,5,4′-tri-O-acetylresveratrol in rats. Phytomedicine 2013, 20, 558–563. [Google Scholar] [CrossRef]
- Burkon, A.; Somoza, V. Quantification of free and protein-boundtrans-resveratrol metabolites and identification oftrans-resveratrol-C/O-conjugated diglucuronides—Two novel resveratrol metabolites in human plasma. Mol. Nutr. Food Res. 2008, 52, 549–557. [Google Scholar] [CrossRef]
- Polycarpou, E.; Meira, L.B.; Carrington, S.; Tyrrell, E.; Modjtahedi, H.; Carew, M.A. Resveratrol 3-O -d -glucuronide and resveratrol 4′-O -d -glucuronide inhibit colon cancer cell growth: Evidence for a role of A3 adenosine receptors, cyclin D1 depletion, and G1 cell cycle arrest. Mol. Nutr. Food Res. 2013, 57, 1708–1717. [Google Scholar] [CrossRef] [PubMed]
- Han, K.; Rong, W.; Wang, Q.; Qu, J.; Li, Q.; Bi, K.; Liu, R. Time-dependent metabolomics study of cerebral ischemia–reperfusion and its treatment: Focus on the combination of traditional Chinese medicine and Western medicine. Anal. Bioanal. Chem. 2020, 412, 7195–7209. [Google Scholar] [CrossRef]
- Lee, J.; Jung, Y.; Park, J.Y.; Lee, S.-H.; Ryu, D.H.; Hwang, G.-S. LC/MS-based polar metabolite profiling reveals gender differences in serum from patients with myocardial infarction. J. Pharm. Biomed. Anal. 2015, 115, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.; Chen, R.; Wang, Q.; Li, Z.; Yang, S.; Feng, Y. Transcriptomic and Metabolomic Profiling Reveals the Protective Effect of Acanthopanax senticosus (Rupr. & Maxim.) Harms Combined With Gastrodia elata Blume on Cerebral Ischemia-Reperfusion Injury. Front. Pharmacol. 2021, 12. [Google Scholar] [CrossRef]
- Rashad, S.; Saigusa, D.; Yamazaki, T.; Matsumoto, Y.; Tomioka, Y.; Saito, R.; Uruno, A.; Niizuma, K.; Yamamoto, M.; Tominaga, T. Metabolic basis of neuronal vulnerability to ischemia; an in vivo untargeted metabolomics approach. Sci. Rep. 2020, 10, 6507. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Zou, Z.; Li, Y.-K.; Yuan, H.-B.; Shi, X.-Y. Glutamine-induced heat shock protein protects against renal ischaemia-reperfusion injury in rats. Nephrology 2009, 14, 573–580. [Google Scholar] [CrossRef]
- Krylova, I.B.; Selina, E.N.; Bulion, V.V.; Rodionova, O.M.; Evdokimova, N.R.; Belosludtseva, N.V.; Shigaeva, M.I.; Mironova, G.D. Uridine treatment prevents myocardial injury in rat models of acute ischemia and ischemia/reperfusion by activating the mitochondrial ATP-dependent potassium channel. Sci. Rep. 2021, 11, 16999. [Google Scholar] [CrossRef] [PubMed]
- Seminotti, B.; Da Silva, J.C.; Ribeiro, R.T.; Leipnitz, G.; Wajner, M. Free Radical Scavengers Prevent Argininosuccinic Acid-Induced Oxidative Stress in the Brain of Developing Rats: A New Adjuvant Therapy for Argininosuccinate Lyase Deficiency? Mol. Neurobiol. 2019, 57, 1233–1244. [Google Scholar] [CrossRef]
- Li, X.; Liang, S.; Tan, C.H.; Cao, S.; Xu, X.; Saw, P.E.; Tao, W. Nanocarriers in the Enhancement of Therapeutic Efficacy of Natural Drugs. BIO Integr. 2021, 2, 40–49. [Google Scholar] [CrossRef]
- Liu, C.; Peng, M.; Zheng, L.; Zhao, Y.; Wang, R.; Su, Q.; Chen, S.; Li, Z. Enhanced autophagy alleviates injury during hindlimb ischemia/reperfusion in mice. Exp. Ther. Med. 2019, 18, 1669–1676. [Google Scholar] [CrossRef] [PubMed] [Green Version]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fan, D.; Liu, C.; Guo, Z.; Huang, K.; Peng, M.; Li, N.; Luo, H.; Wang, T.; Cen, Z.; Cai, W.; et al. Resveratrol Promotes Angiogenesis in a FoxO1-Dependent Manner in Hind Limb Ischemia in Mice. Molecules 2021, 26, 7528. https://doi.org/10.3390/molecules26247528
Fan D, Liu C, Guo Z, Huang K, Peng M, Li N, Luo H, Wang T, Cen Z, Cai W, et al. Resveratrol Promotes Angiogenesis in a FoxO1-Dependent Manner in Hind Limb Ischemia in Mice. Molecules. 2021; 26(24):7528. https://doi.org/10.3390/molecules26247528
Chicago/Turabian StyleFan, Dongxiao, Chenshu Liu, Zeling Guo, Kan Huang, Meixiu Peng, Na Li, Hengli Luo, Tengyao Wang, Zhipeng Cen, Weikang Cai, and et al. 2021. "Resveratrol Promotes Angiogenesis in a FoxO1-Dependent Manner in Hind Limb Ischemia in Mice" Molecules 26, no. 24: 7528. https://doi.org/10.3390/molecules26247528
APA StyleFan, D., Liu, C., Guo, Z., Huang, K., Peng, M., Li, N., Luo, H., Wang, T., Cen, Z., Cai, W., Gu, L., Chen, S., & Li, Z. (2021). Resveratrol Promotes Angiogenesis in a FoxO1-Dependent Manner in Hind Limb Ischemia in Mice. Molecules, 26(24), 7528. https://doi.org/10.3390/molecules26247528