A Report on Multi-Target Anti-Inflammatory Properties of Phytoconstituents from Monochoria hastata (Family: Pontederiaceae)
Abstract
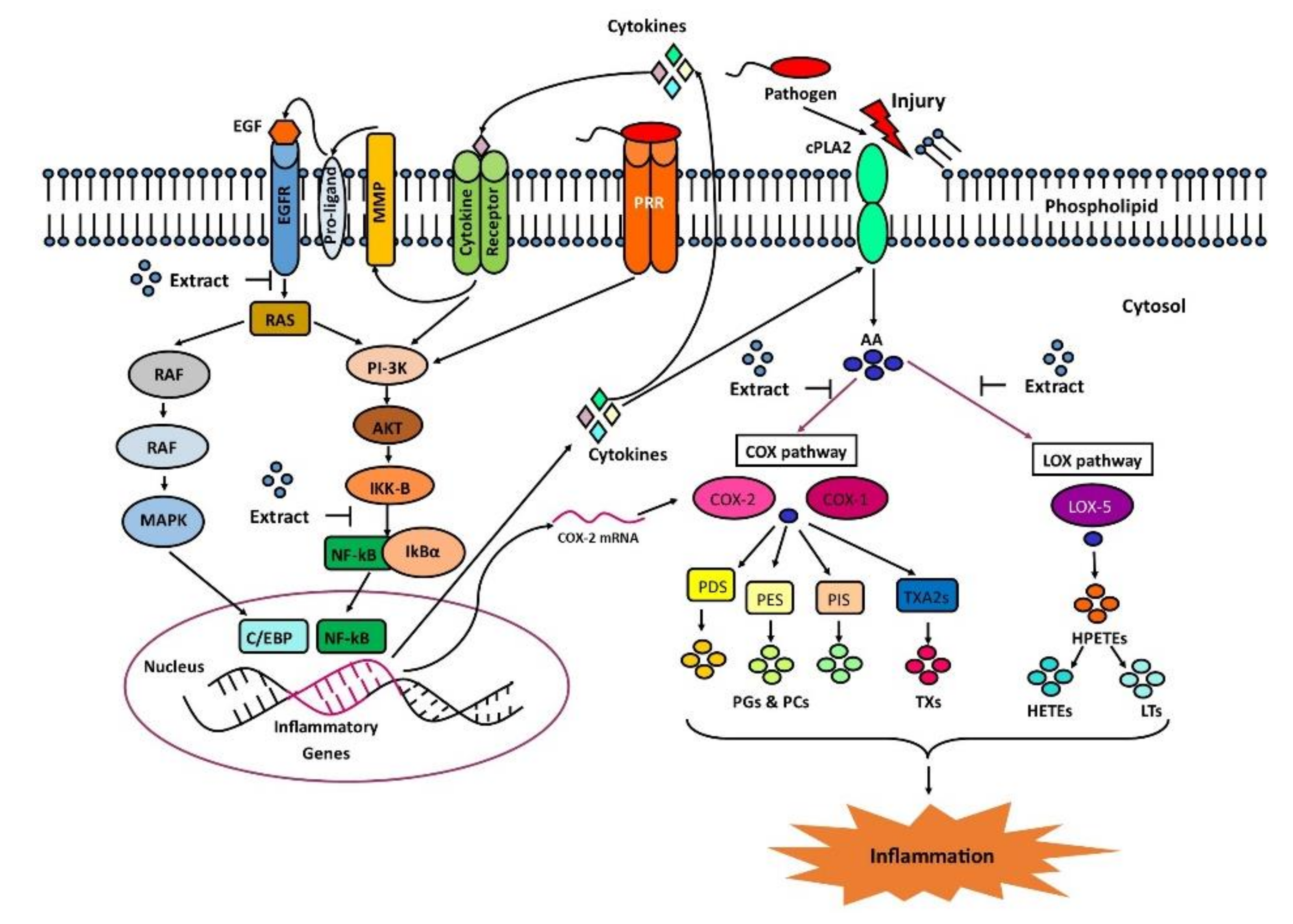
1. Introduction
2. Results
2.1. Analgesic Activity of Monochoria hastata Leaves Extract
2.2. In Silico Analysis
2.2.1. In Silico Prediction of Activity Spectra for Substances (PASS)
2.2.2. Drug-like Properties of Isolated Compounds
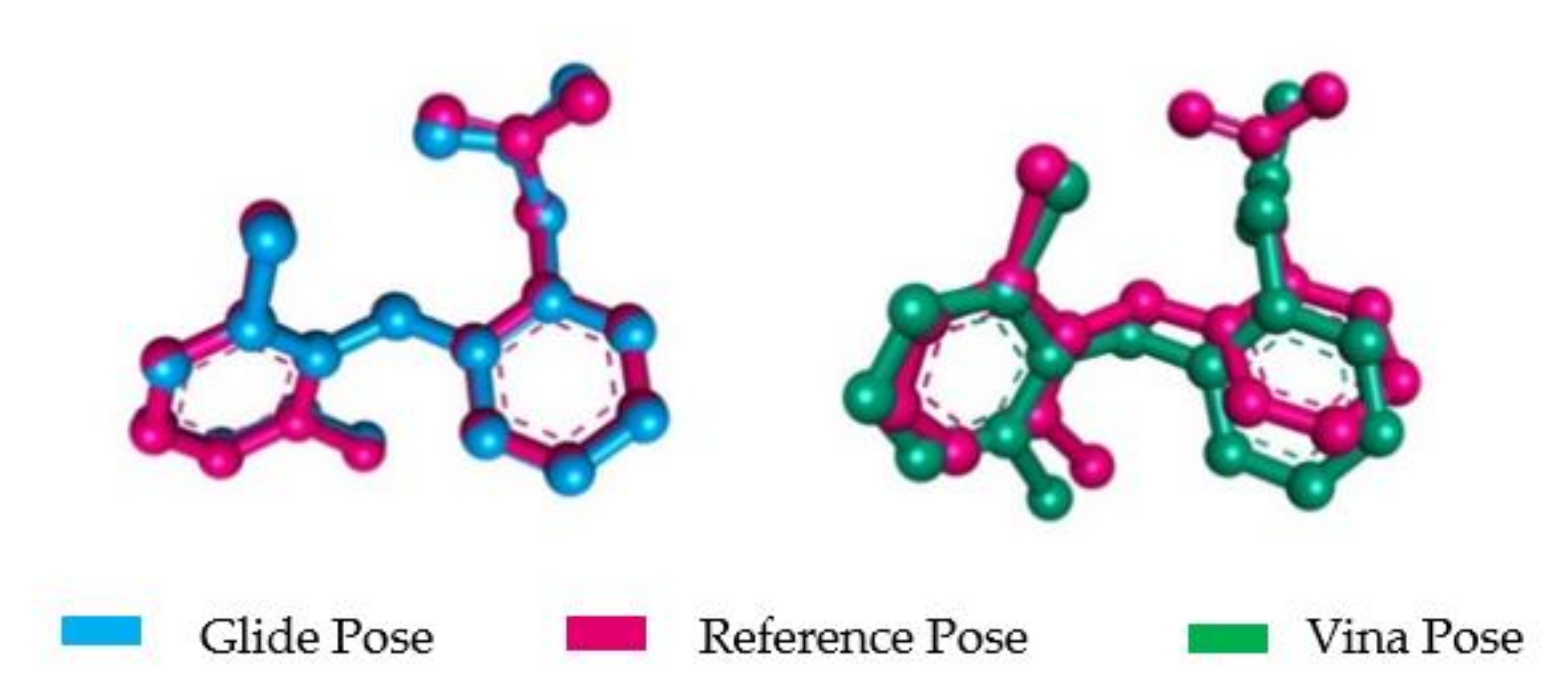
2.2.3. Molecular Docking Validation (MDV)
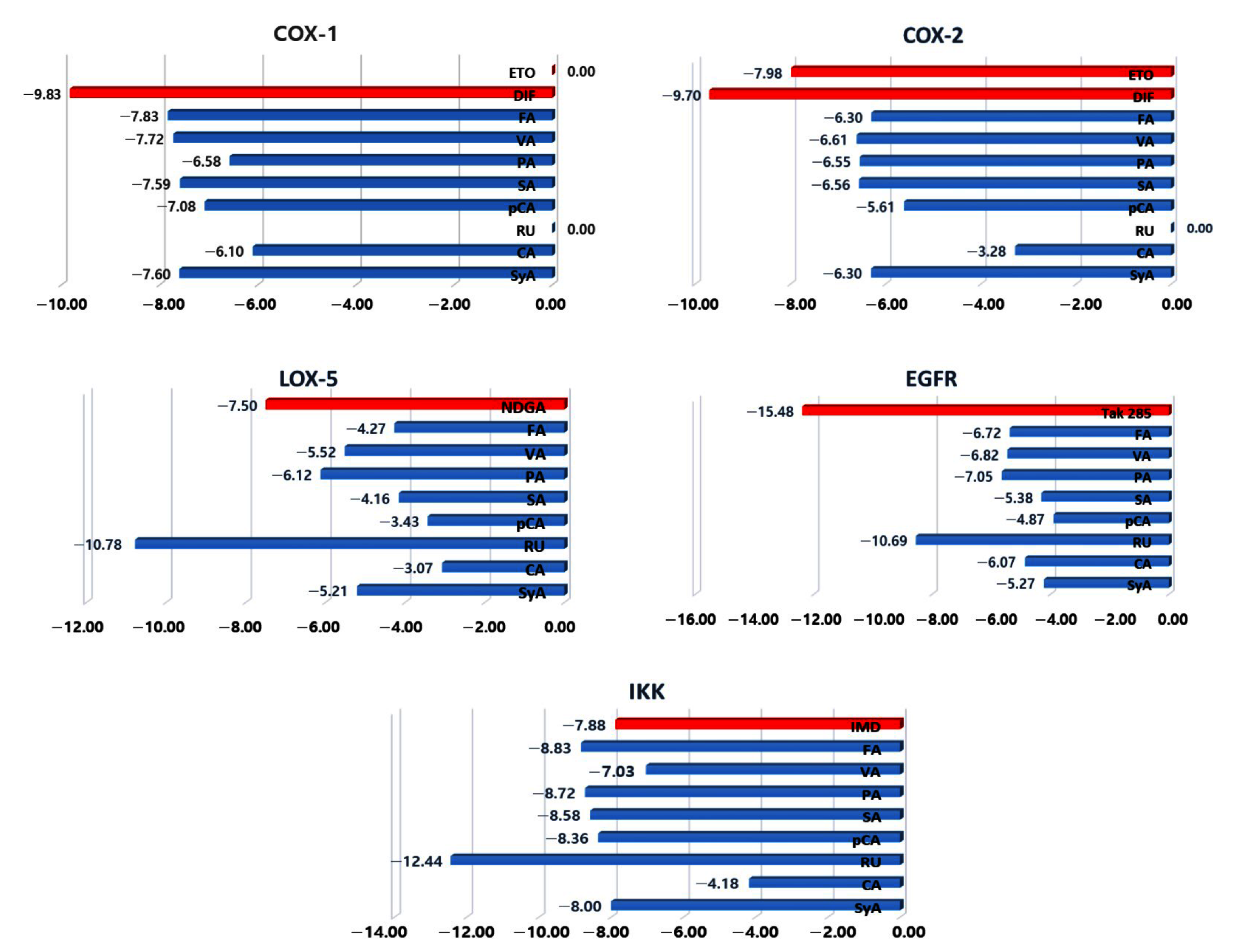
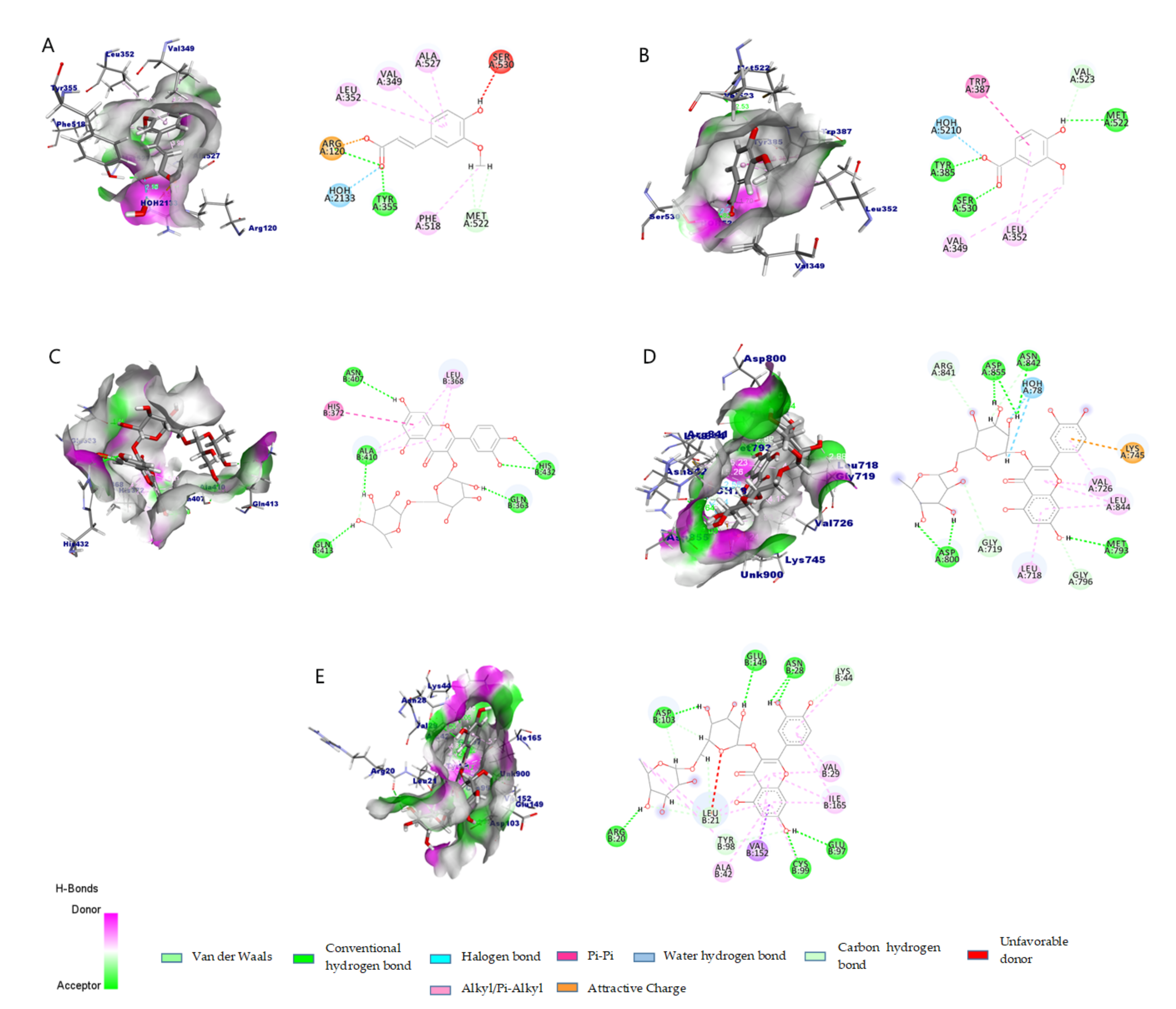
2.2.4. Molecular Docking and Interaction
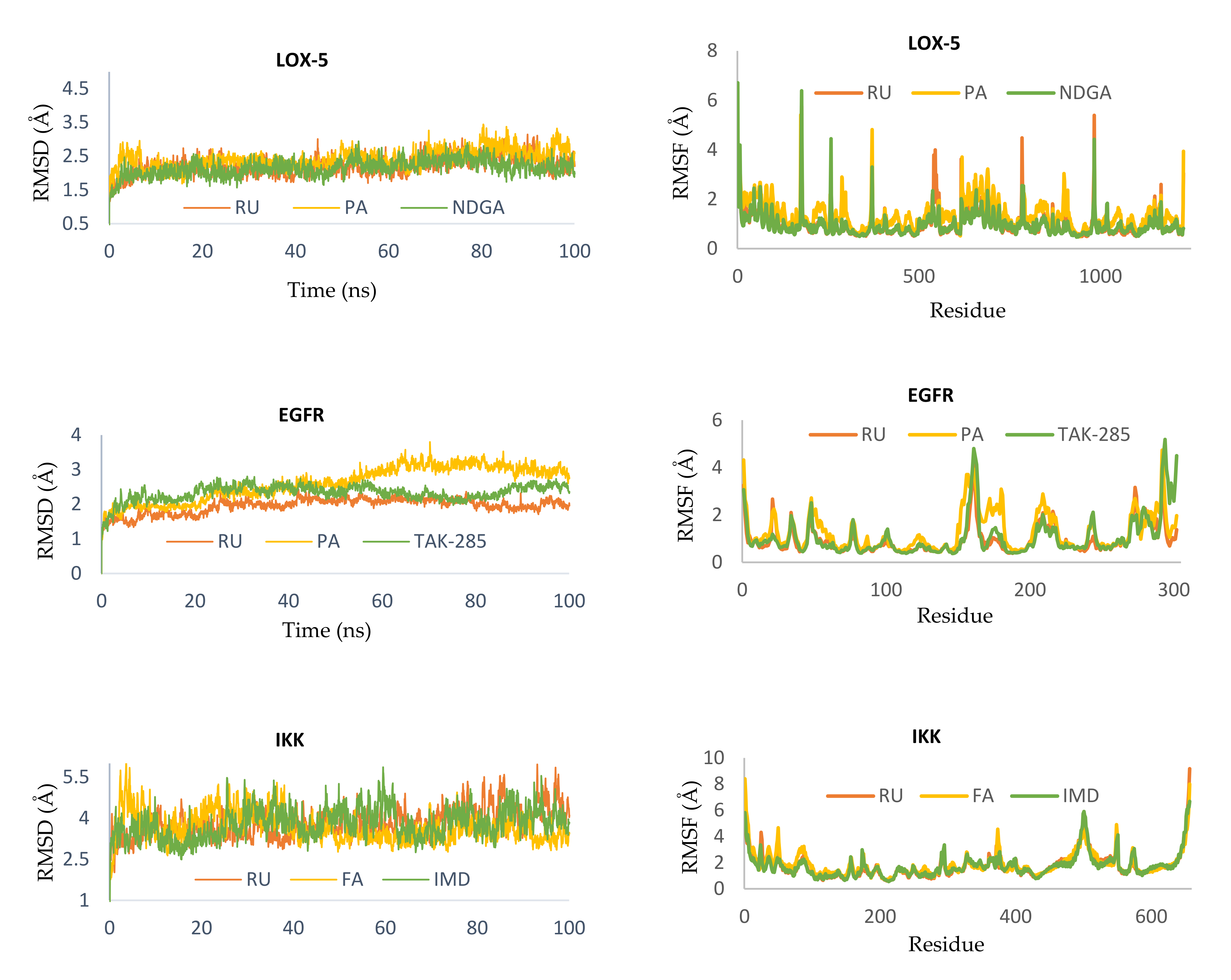
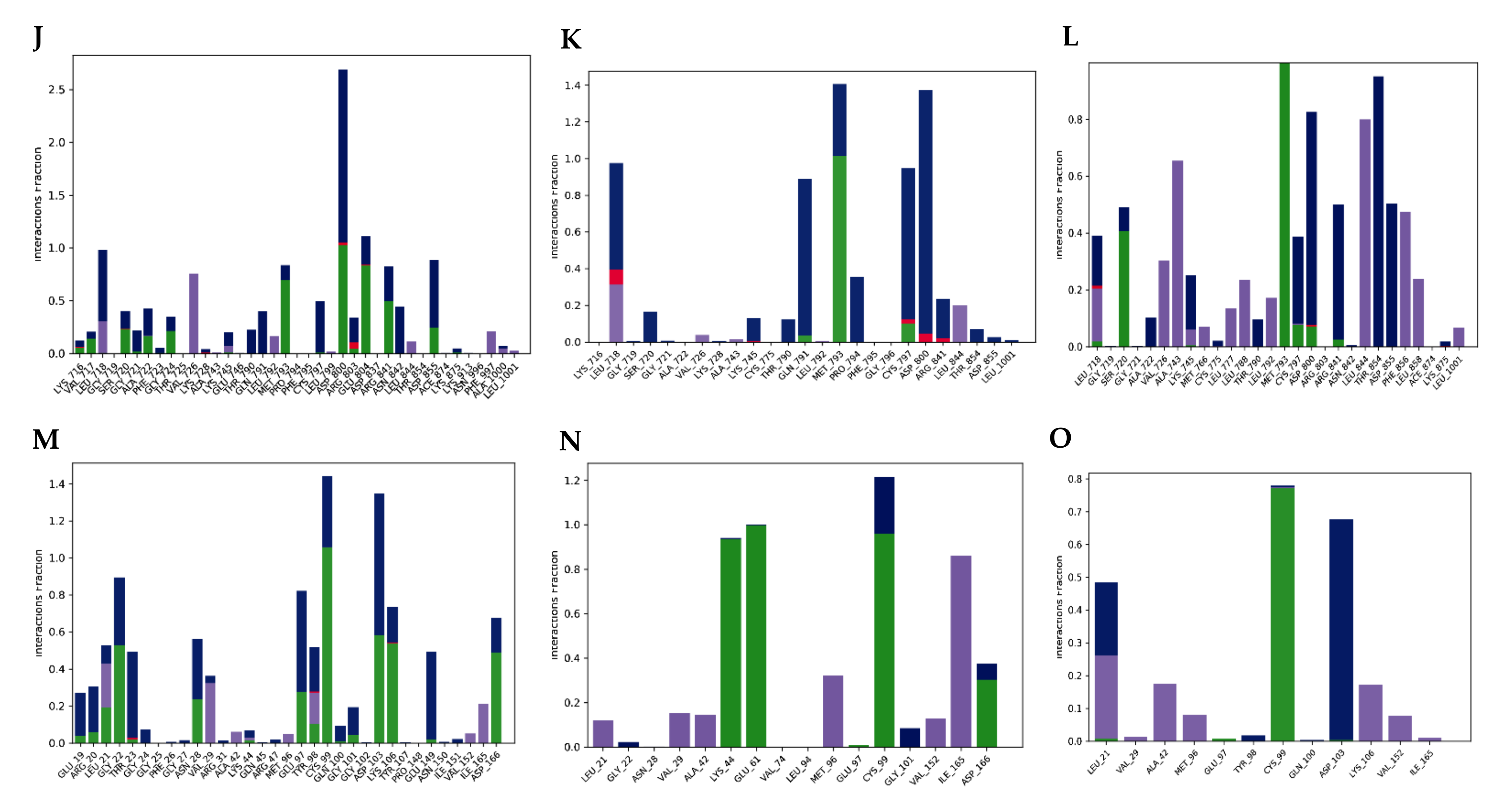
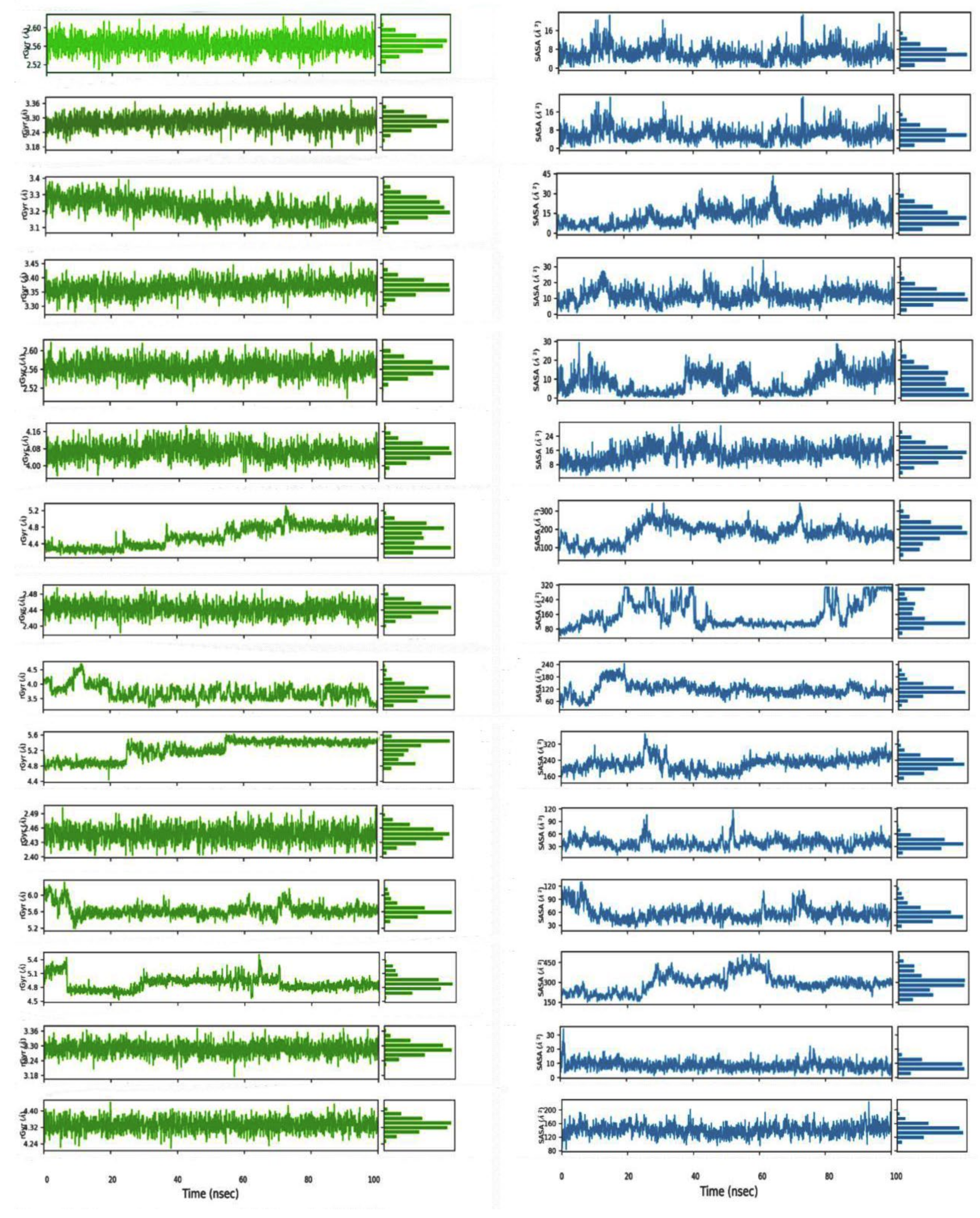
2.2.5. Molecular Dynamics Simulation
2.2.6. ADMET Profile
3. Discussion
4. Materials and Methods
4.1. In Vivo
4.1.1. Collection and Identification of Specimen
4.1.2. Chemicals, Drugs and Solvents
4.1.3. Experimental Animals and Ethical Approval
4.1.4. Preparation of Extract
4.1.5. Analgesic/Anti-Nociceptive Activity by Acetic Acid Induced Writhing Method
4.1.6. Experimental Design for In Vivo Analgesic Activity Assessment
4.1.7. Statistical Analysis
4.2. In Silico Analysis
4.2.1. Receptor and Ligand Structure Acquisition
4.2.2. In Silico Prediction of Activity Spectra for Substances (PASS Prediction)
4.2.3. Lipinski’s Rule of Five Parameters
4.2.4. Molecular Docking
- Protein Preparation
- Ligand Preparation
- Docking Simulation Validation
- Molecular Docking
- Docked Pose Analysis and Visualization
4.2.5. Molecular Dynamics (MD) Simulation
4.2.6. ADMET Calculation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Hawkins, C.; Hanks, G.W. The gastroduodenal toxicity of nonsteroidal anti-inflammatory drugs. A review of the literature. J. Pain Symptom Manage. 2000, 20, 140–151. [Google Scholar] [CrossRef]
- Klein, A.; Eliakim, R. Non Steroidal Anti-Inflammatory Drugs and Inflammatory Bowel Disease. Pharmaceutics 2010, 3, 1084. [Google Scholar] [CrossRef] [PubMed]
- Ramsay, R.R.; Popovic-Nikolic, M.R.; Nikolic, K.; Uliassi, E.; Bolognesi, M.L. A perspective on multi-target drug discovery and design for complex diseases. Clin. Transl. Med. 2018, 7, 3. [Google Scholar] [CrossRef]
- Koeberle, A.; Werz, O. Multi-target approach for natural products in inflammation. Drug Discov. Today 2014, 19, 1871–1882. [Google Scholar] [CrossRef] [PubMed]
- Talevi, A. Multi-target pharmacology: Possibilities and limitations of the “skeleton key approach” from a medicinal chemist perspective. Front. Pharmacol. 2015, 6, 205. [Google Scholar] [CrossRef]
- Zhou, Y.J.; Xu, X.H.; Qiao, F.Y.; Zhang, J.P.; Yu, L.Q. Isolation and identification of an antioxidant from Monochoria vaginalis. Chin. J. Appl. Ecol. 2007, 18, 509–513. [Google Scholar]
- Chandran, R.; Parimelazhagan, T. Nutritional Assessment of Monochoria vaginalis, a Wild Edible Vegetable Supplement to the Human Diet. Int. J. Veg. Sci. 2012, 18, 199–207. [Google Scholar] [CrossRef]
- Palani, S.; Raja, S.; Kumar, R.P.; Selvaraj, R.; Kumar, B.S. Evaluation of phytoconstituents and anti-nephrotoxic and antioxidant activities of Monochoria vaginalis. Pak. J. Pharm. Sci. 2011, 24, 293–301. [Google Scholar]
- Institute of ayurveda.org. Ayurvedic Plants of Sri Lanka: Plants Details. 2017. Available online: http://www.instituteofayurveda.org/plants/plants_detail.php?i=1080&s=Family_name (accessed on 10 February 2021).
- Astutik, S.; Pretzsch, J.; Ndzifon Kimengsi, J. Asian Medicinal Plants’ Production and Utilization Potentials: A Review. Sustainability 2019, 11, 5483. [Google Scholar] [CrossRef]
- Dennis, E.A.; Cao, J.; Hsu, Y.H.; Magrioti, V.; Kokotos, G. Phospholipase A2 enzymes: Physical structure, biological function, disease implication, chemical inhibition, and therapeutic intervention. Chem. Rev. 2011, 111, 6130–6185. [Google Scholar] [CrossRef]
- Thun, M.J.; Jane Henley, S.; Patrono, C. Nonsteroidal anti-inflammatory drugs as anticancer agents: Mechanistic, pharmacologic, and clinical issues. J. Natl. Cancer Inst. 2002, 94, 252–266. [Google Scholar] [CrossRef] [PubMed]
- Kidd, M.; Schimmack, S.; Lawrence, B.; Alaimo, D.; Modlin, I.M. EGFR/TGFα and TGFβ/CTGF signaling in neuroendocrine neoplasia: Theoretical therapeutic targets. Neuroendocrinology 2013, 97, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Abu-Yousif, A.O.; Moor, A.C.E.; Zheng, X.; Savellano, M.D.; Yu, W.; Selbo, P.K.; Hasan, T. Epidermal growth factor receptor-targeted photosensitizer selectively inhibits EGFR signaling and induces targeted phototoxicity in ovarian cancer cells. Cancer Lett. 2012, 321, 120–127. [Google Scholar] [CrossRef]
- Heuckmann, J.M.; Rauh, D.; Thomas, R.K. Epidermal growth factor receptor (EGFR) signaling and covalent EGFR inhibition in lung cancer. J. Clin. Oncol. 2012, 30, 3417–3420. [Google Scholar] [CrossRef] [PubMed]
- Normanno, N.; De Luca, A.; Bianco, C.; Strizzi, L.; Mancino, M.; Maiello, M.R.; Carotenuto, A.; De Feo, G.; Caponigro, F.; Salomon, D.S. Epidermal growth factor receptor (EGFR) signaling in cancer. Gene 2006, 366, 2–16. [Google Scholar] [CrossRef] [PubMed]
- Dangle, P.; Zaharieva, B.; Jia, H.; Pohar, K. Ras-MAPK Pathway as a Therapeutic Target in Cancer—Emphasis on Bladder Cancer. Recent Pat. Anticancer. Drug Discov. 2009, 4, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, M.V. RTK/Ras/MAPK signaling. WormBook 2006, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Moritz, A.; Li, Y.; Guo, A.; Villén, J.; Wang, Y.; MacNeill, J.; Kornhauser, J.; Sprott, K.; Zhou, J.; Possemato, A.; et al. Akt—RSK—S6 kinase signaling networks activated by oncogenic receptor tyrosine kinases. Sci. Signal. 2010, 3, ra64. [Google Scholar] [CrossRef]
- Dallob, A.; Hawkey, C.J.; Greenberg, H.; Wight, N.; De Schepper, P.; Waldman, S.; Wong, P.; DeTora, L.; Gertz, B.; Agrawal, N.; et al. Characterization of etoricoxib, a novel, selective COX-2 inhibitor. J. Clin. Pharmacol. 2003, 43, 573–585. [Google Scholar] [CrossRef]
- Ooh, K.F.; Ong, H.C.; Wong, F.C.; Sit, N.W.; Chai, T.T. High performance liquid chromatography profiling of health-promoting phytochemicals and evaluation of antioxidant, anti-lipoxygenase, iron chelating and anti-glucosidase activities of wetland macrophytes. Pharmacogn. Mag. 2014, 10, S443–S455. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.N.; Baruah, M.M.; Sharma, N. Structure Based docking studies towards exploring potential anti-androgen activity of selected phytochemicals against Prostate Cancer. Sci. Rep. 2017, 7, 1955. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2012, 64, 4–17. [Google Scholar] [CrossRef]
- Choi, S.; Lim, T.G.; Hwang, M.K.; Kim, Y.A.; Kim, J.; Kang, N.J.; Jang, T.S.; Park, J.S.; Yeom, M.H.; Lee, K.W. Rutin inhibits B[a]PDE-induced cyclooxygenase-2 expression by targeting EGFR kinase activity. Biochem. Pharmacol. 2013, 86, 1468–1475. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, N.C.; Gerstmeier, J.; Schexnaydre, E.E.; Börner, F.; Garscha, U.; Neau, D.B.; Werz, O.; Newcomer, M.E. Structural and mechanistic insights into 5-lipoxygenase inhibition by natural products. Nat. Chem. Biol. 2020, 16, 783–790. [Google Scholar] [CrossRef] [PubMed]
- Aertgeerts, K.; Skene, R.; Yano, J.; Sang, B.C.; Zou, H.; Snell, G.; Jennings, A.; Iwamoto, K.; Habuka, N.; Hirokawa, A.; et al. Structural analysis of the mechanism of inhibition and allosteric activation of the kinase domain of HER2 protein. J. Biol. Chem. 2011, 286, 18756–18765. [Google Scholar] [CrossRef] [PubMed]
- Blobaum, A.L.; Marnett, L.J. Structural and functional basis of cyclooxygenase inhibition. J. Med. Chem. 2007, 50, 1425–1441. [Google Scholar] [CrossRef] [PubMed]
- Childers, M.C.; Daggett, V. Insights from molecular dynamics simulations for computational protein design. Mol. Syst. Des. Eng. 2017, 2, 9–33. [Google Scholar] [CrossRef]
- Durrant, J.D.; McCammon, J.A. Molecular dynamics simulations and drug discovery. BMC Biol. 2011, 9, 71. [Google Scholar] [CrossRef]
- Ribeiro, J.M.L.; Filizola, M. Insights From Molecular Dynamics Simulations of a Number of G-Protein Coupled Receptor Targets for the Treatment of Pain and Opioid Use Disorders. Front. Mol. Neurosci. 2019, 12, 207. [Google Scholar] [CrossRef]
- Levita, J.; Nawawi, A.; Mutholib, A.; Ibrahim, S. Andrographolide inhibits COX-2 expression in human fibroblast cells due to its interaction with arginine and histidine in cyclooxygenase site. J. Appl. Sci. 2010, 10, 1481–1484. [Google Scholar] [CrossRef][Green Version]
- Lin, N.; Liu, C.; Xiao, C.; Jia, H.; Imada, K.; Wu, H.; Ito, A. Triptolide, a diterpenoid triepoxide, suppresses inflammation and cartilage destruction in collagen-induced arthritis mice. Biochem. Pharmacol. 2007, 73, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Mancini, J.A.; Riendeau, D.; Falgueyret, J.P.; Vickers, P.J.; O’Neill, G.P. Arginine 120 of prostaglandin G/H synthase-1 is required for the inhibition by nonsteroidal anti-inflammatory drugs containing a carboxylic acid moiety. J. Biol. Chem. 1995, 270, 29372–29377. [Google Scholar] [CrossRef] [PubMed]
- González, Y.; Torres-Mendoza, D.; Jones, G.E.; Fernandez, P.L. Marine Diterpenoids as Potential Anti-Inflammatory Agents. Mediators Inflamm. 2015, 2015, 263543. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, C.; Cagide, F.; Teixeira, J.; Amorim, R.; Sequeira, L.; Mesiti, F.; Silva, T.; Garrido, J.; Remião, F.; Vilar, S.; et al. Hydroxybenzoic Acid Derivatives as Dual-Target Ligands: Mitochondriotropic Antioxidants and Cholinesterase Inhibitors. Front. Chem. 2018, 6, 126. [Google Scholar] [CrossRef] [PubMed]
- Ginwala, R.; Bhavsar, R.; Chigbu, D.I.; Jain, P.; Khan, Z.K. Potential Role of Flavonoids in Treating Chronic Inflammatory Diseases with a Special Focus on the Anti-Inflammatory Activity of Apigenin. Antioxidants 2019, 8, 35. [Google Scholar] [CrossRef]
- Nagasaka, R.; Chotimarkorn, C.; Shafiqul, I.M.; Hori, M.; Ozaki, H.; Ushio, H. Anti-inflammatory effects of hydroxycinnamic acid derivatives. Biochem. Biophys. Res. Commun. 2007, 358, 615–619. [Google Scholar] [CrossRef]
- Cui, Y.; Desevaux, C.; Truebenbach, I.; Sieger, P.; Klinder, K.; Long, A.; Sauer, A. A Bidirectional Permeability Assay for beyond Rule of 5 Compounds. Pharmaceutics 2021, 13, 1146. [Google Scholar] [CrossRef]
- Choi, S.H.; Aid, S.; Bosetti, F. The distinct roles of cyclooxygenase-1 and -2 in neuroinflammation: Implications for translational research. Trends Pharmacol. Sci. 2009, 30, 174–181. [Google Scholar] [CrossRef]
- Rouzer, C.A.; Marnett, L.J. Cyclooxygenases: Structural and functional insights. J. Lipid Res. 2009, 50, S29–S34. [Google Scholar] [CrossRef]
- Williams, C.S.; Mann, M.; DuBois, R.N. The role of cyclooxygenases in inflammation, cancer, and development. Oncogene 1999, 18, 7908–7916. [Google Scholar] [CrossRef] [PubMed]
- Fiorucci, S.; Meli, R.; Bucci, M.; Cirino, G. Dual inhibitors of cyclooxygenase and 5-lipoxygenase. A new avenue in anti-inflammatory therapy? Biochem. Pharmacol. 2001, 62, 1433–1438. [Google Scholar] [CrossRef]
- Charlier, C.; Michaux, C. Dual inhibition of cyclooxygenase-2 (COX-2) and 5-lipoxygenase (5-LOX) as a new strategy to provide safer non-steroidal anti-inflammatory drugs. Eur. J. Med. Chem. 2003, 38, 645–659. [Google Scholar] [CrossRef]
- O’Byrne, P.M. Leukotrienes in the pathogenesis of asthma. Chest 1997, 111, 27S–34S. [Google Scholar] [CrossRef] [PubMed]
- Miossec, P. Understanding the cytokine storm during COVID-19: Contribution of preexisting chronic inflammation. Eur. J. Rheumatol. 2020, 7, S97–S98. [Google Scholar] [CrossRef]
- Di Leo, G.; Sardanelli, F. Statistical significance: P value, 0.05 threshold, and applications to radiomics—reasons for a conservative approach. Eur. Radiol. Exp. 2020, 4, 18. [Google Scholar] [CrossRef]
- Misra, D.; Mandal, M.; Ghosh, N.; Mandal, V. Extraction and volatile compounds profiling of the bioactive fraction of Monochoria hastata (L.) solms. Pharmacogn. Mag. 2020, 16, 517–523. [Google Scholar] [CrossRef]
- Jamkhande, P.G.; Pathan, S.K.; Wadher, S.J. In silico PASS analysis and determination of antimycobacterial, antifungal, and antioxidant efficacies of maslinic acid in an extract rich in pentacyclic triterpenoids. Int. J. Mycobacteriol. 2016, 5, 417–425. [Google Scholar] [CrossRef]
- Goel, R.K.; Singh, D.; Lagunin, A.; Poroikov, V. PASS-assisted exploration of new therapeutic potential of natural products. Proc. Med. Chem. Res. 2011, 20, 1509–1514. [Google Scholar]
- Huggins, D.J.; Tidor, B. Systematic placement of structural water molecules for improved scoring of protein–ligand interactions. Protein Eng. Des. Sel. 2011, 24, 777–789. [Google Scholar] [CrossRef]
- Harder, E.; Damm, W.; Maple, J.; Wu, C.; Reboul, M.; Xiang, J.Y.; Wang, L.; Lupyan, D.; Dahlgren, M.K.; Knight, J.L.; et al. OPLS3: A Force Field Providing Broad Coverage of Drug-like Small Molecules and Proteins. J. Chem. Theory Comput. 2016, 12, 281–296. [Google Scholar] [CrossRef] [PubMed]
- Friesner, R.A.; Banks, J.L.; Murphy, R.B.; Halgren, T.A.; Klicic, J.J.; Mainz, D.T.; Repasky, M.P.; Knoll, E.H.; Shelley, M.; Perry, J.K.; et al. Glide: A New Approach for Rapid, Accurate Docking and Scoring. 1. Method and Assessment of Docking Accuracy. J. Med. Chem. 2004, 47, 1739–1749. [Google Scholar] [CrossRef] [PubMed]
- Friesner, R.A.; Murphy, R.B.; Repasky, M.P.; Frye, L.L.; Greenwood, J.R.; Halgren, T.A.; Sanschagrin, P.C.; Mainz, D.T. Extra precision glide: Docking and scoring incorporating a model of hydrophobic enclosure for protein-ligand complexes. J. Med. Chem. 2006, 49, 6177–6196. [Google Scholar] [CrossRef] [PubMed]
- Van de Waterbeemd, H.; Gifford, E. ADMET in silico modelling: Towards prediction paradise? Nat. Rev. Drug Discov. 2003, 2, 192–204. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Cheng, F.; Xu, Y.; Li, W.; Tang, Y. Estimation of ADME properties with substructure pattern recognition. J. Chem. Inf. Model. 2010, 50, 1034–1041. [Google Scholar] [CrossRef] [PubMed]
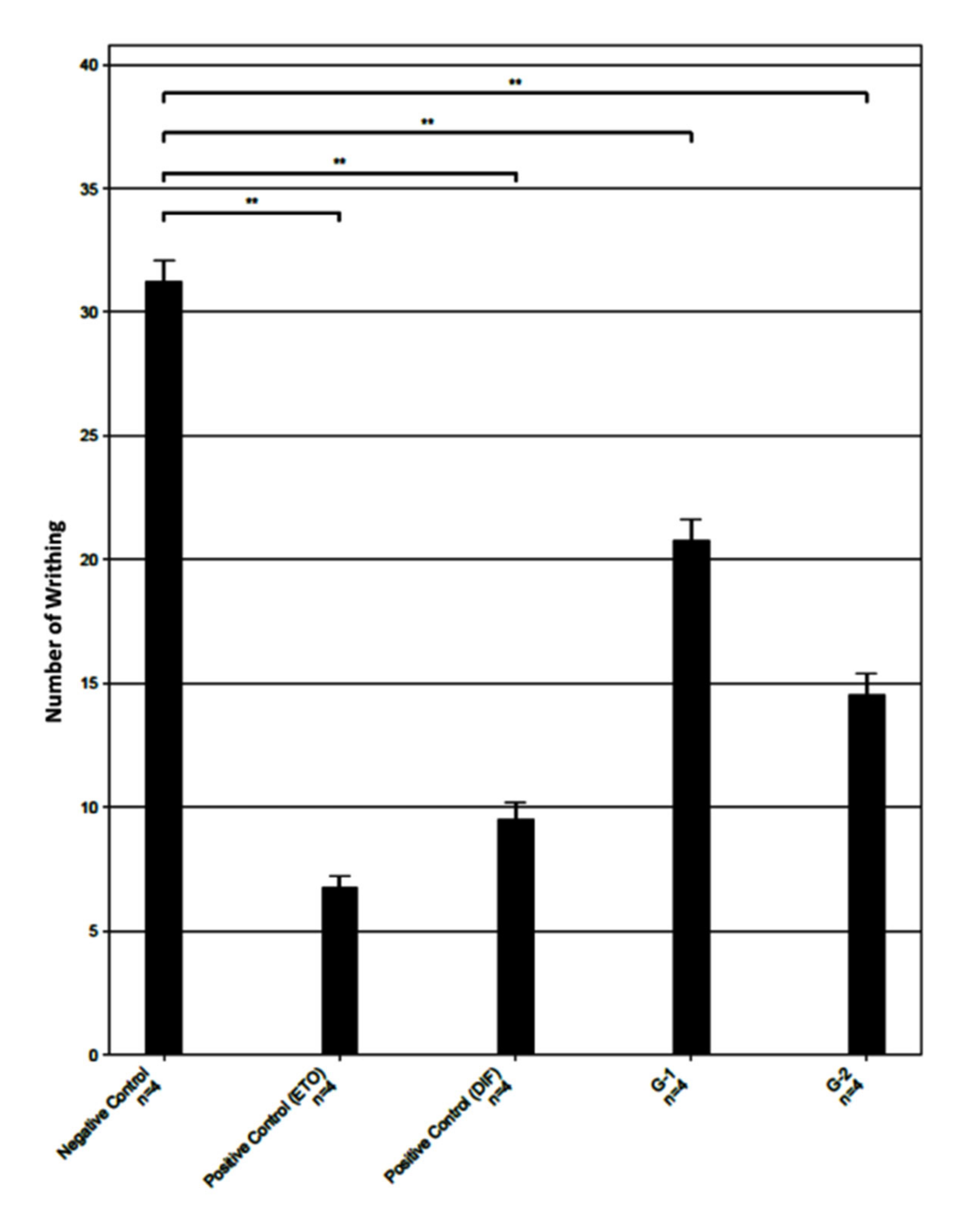


| Group | Number of Writhing 1 | Mean | SD | SEM | Mean ± SEM | Inhibition (%) | |||
|---|---|---|---|---|---|---|---|---|---|
| m1 | m2 | m3 | m4 | ||||||
| Negative Control | 32 | 31 | 29 | 33 | 31.25 | 1.71 | 0.85 | 31.25 ± 0.85 | 0 |
| Positive control ETO (10 mg/kg bw) | 6 | 8 | 6 | 7 | 6.75 | 0.95 | 0.48 | 6.75 ± 0.48 | 78.4 |
| Positive control DIF (50 mg/kg bw) | 11 | 8 | 10 | 9 | 9.5 | 1.29 | 0.65 | 9.5 ± 0.65 | 69.6 |
| G-1 (MH) 200 mg/kg | 19 | 23 | 20 | 21 | 20.75 | 1.71 | 0.85 | 20.75 ± 0.85 | 33.6 |
| G-2 (MH) 400 mg/kg | 13 | 14 | 14 | 17 | 14.5 | 1.73 | 0.87 | 14.50 ± 0.87 | 53.6 |
| Isolated Compound | PubChem ID | Canonical SMILES |
|---|---|---|
| Ferulic Acid (FA) 1 | CID 445858 | COC1=C(C=CC(=C1)C=CC(=O)O)O |
| Sinapic Acid (SA) 1 | CID 637775 | COC1=CC(=CC(=C1O)OC)C=CC(=O)O |
| Chlorogenic Acid (CA) 1 | CID 1794427 | C1C(C(C(CC1(C(=O)O)O)OC(=O)C=CC2=CC(=C(C=C2)O)O)O)O |
| p-Coumaric Acid (pCA) 1 | CID 637542 | C1=CC(=CC=C1C=CC(=O)O)O |
| Rutin (RU) 2 | CID 5280805 | CC1C(C(C(C(O1)OCC2C(C(C(C(O2)OC3=C(OC4=CC(=CC(=C4C3=O)O)O)C5=CC(=C(C=C5)O)O)O)O)O)O)O)O |
| Syringic Acid (SyA) 2 | CID 10742 | COC1=CC(=CC(=C1O)OC)C(=O)O |
| Vanillic Acid (VA) 3 | CID 8468 | COC1=C(C=CC(=C1)C(=O)O)O |
| Protocatechuic Acid (PA) 3 | CID 72 | C1=CC(=C(C=C1C(=O)O)O)O |
| Compound | TPSA (Å2) | MW ˂500 | miLogP ˂5 | HBD ˂5 | HBA ˂10 | n-ROTB ˂10 | Lipinski’s Violation ˂1 |
|---|---|---|---|---|---|---|---|
| FA | 66.76 | 194.19 | 1.25 | 4 | 2 | 3 | 0 |
| SA | 76 | 224.21 | 1.26 | 2 | 5 | 4 | 0 |
| CA | 164.74 | 354.31 | −0.45 | 6 | 9 | 5 | 1 |
| pCA | 57.53 | 164.16 | 1.43 | 2 | 3 | 2 | 0 |
| RU | 269.43 | 610.52 | −1.06 | 10 | 16 | 6 | 3 |
| SyA | 76 | 198.17 | 1.2 | 2 | 5 | 3 | 0 |
| VA | 66.76 | 168.15 | 1.19 | 2 | 4 | 2 | 0 |
| PA | 77.75 | 154.12 | 0.88 | 3 | 4 | 1 | 0 |
| Parameters | FA | SA | CA | pCA | Rutin | SyA | VA | PA |
|---|---|---|---|---|---|---|---|---|
| BBB | −(53.1) | +(57.9) | +(56.6) | +(52.4) | −(85.4) | +(58.6) | −(51.5) | −(63.8) |
| HIA | +(96.1) | +(95.8) | +(74.3) | +(99.4) | +(80.4) | +(91.7) | +(92.3) | +(88.1) |
| Caco-2 permeability | +(71.8) | +(73.2) | −(80.1) | +(88.4) | −(91.7) | +(71.2) | +(70.6) | +(55.5) |
| CYP450 2C9 Substrate | No (74.6) | No (80.0) | No (79.0) | No (78.9) | No (76.4) | No (82.1) | No (77.2) | No (82.3) |
| CYP450 2D6 Substrate | No (89.2) | No (89.2) | No (89.8) | No (93.6) | No (89.6) | No (89.0) | No (89.1) | No (91.5) |
| CYP450 3A4 Substrate | No (62.9) | No (60.5) | No (54.9) | No (74.6) | No (53.7) | No (62.6) | No (64.8) | No (72.3) |
| CYP450 1A2 Inhibitor | No (75.1) | No (84.5) | No (90.5) | No (94.6) | No (86.7) | No (90.5) | No (88.6) | No (95.5) |
| CYP450 2C9 Inhibitor | No (57.9) | No (83.8) | No (90.7) | No (93.6) | No (90.7) | No (93.2) | No (81.3) | No (95.7) |
| CYP450 2D6 Inhibitor | No (95.9) | No (92.9) | No (93.9) | No (97.7) | No (95.5) | No (94.5) | No (97.0) | No (96.4) |
| CYP450 2C19 Inhibitor | No (62.8) | No (71.8) | No (90.7) | No (91.2) | No (90.3) | No (85.8) | No (82.5) | No (97.1) |
| CYP450 3A4 Inhibitor | No (92.4) | No (87.5) | No (87.4) | No (86.9) | No (92.5) | No (95.4) | No (97.1) | No (95.4) |
| CYP Inhibitory Promiscuity | Low (77.5) | Low (76.1) | Low (96.9) | Low (89.1) | Low (67.9) | Low (87.7) | Low (89.0) | Low (95.6) |
| AMES Toxicity | No (91.3) | No (90.2) | No (91.3) | No (95.2) | No (51.2) | No (93.4) | No (94.2) | No (93.3) |
| Carcinogens | No (90.8) | No (88.5) | No (93.4) | No (82.5) | No (96.1) | No (88.1) | No (90.5) | No (91.5) |
| Acute Oral Toxicity | IV (62.7) | III (45.0) | III (77.8) | III (49.0) | III (59.7) | II (47.7) | III (49.2) | III (50.6) |
| Carcinogenicity (Three-class) | No (59.0) | No (67.0) | No (61.3) | No (60.3) | No (67.4) | No (71.6) | No (62.9) | No (62.2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haq, M.M.; Chowdhury, M.A.R.; Tayara, H.; Abdelbaky, I.; Islam, M.S.; Chong, K.T.; Jeong, S. A Report on Multi-Target Anti-Inflammatory Properties of Phytoconstituents from Monochoria hastata (Family: Pontederiaceae). Molecules 2021, 26, 7397. https://doi.org/10.3390/molecules26237397
Haq MM, Chowdhury MAR, Tayara H, Abdelbaky I, Islam MS, Chong KT, Jeong S. A Report on Multi-Target Anti-Inflammatory Properties of Phytoconstituents from Monochoria hastata (Family: Pontederiaceae). Molecules. 2021; 26(23):7397. https://doi.org/10.3390/molecules26237397
Chicago/Turabian StyleHaq, Md Mazedul, Md Arifur Rahman Chowdhury, Hilal Tayara, Ibrahim Abdelbaky, Md Shariful Islam, Kil To Chong, and Sangyun Jeong. 2021. "A Report on Multi-Target Anti-Inflammatory Properties of Phytoconstituents from Monochoria hastata (Family: Pontederiaceae)" Molecules 26, no. 23: 7397. https://doi.org/10.3390/molecules26237397
APA StyleHaq, M. M., Chowdhury, M. A. R., Tayara, H., Abdelbaky, I., Islam, M. S., Chong, K. T., & Jeong, S. (2021). A Report on Multi-Target Anti-Inflammatory Properties of Phytoconstituents from Monochoria hastata (Family: Pontederiaceae). Molecules, 26(23), 7397. https://doi.org/10.3390/molecules26237397








