Mononuclear and Tetranuclear Copper(II) Complexes Bearing Amino Acid Schiff Base Ligands: Structural Characterization and Catalytic Applications
Abstract
:1. Introduction
2. Results and Discussion
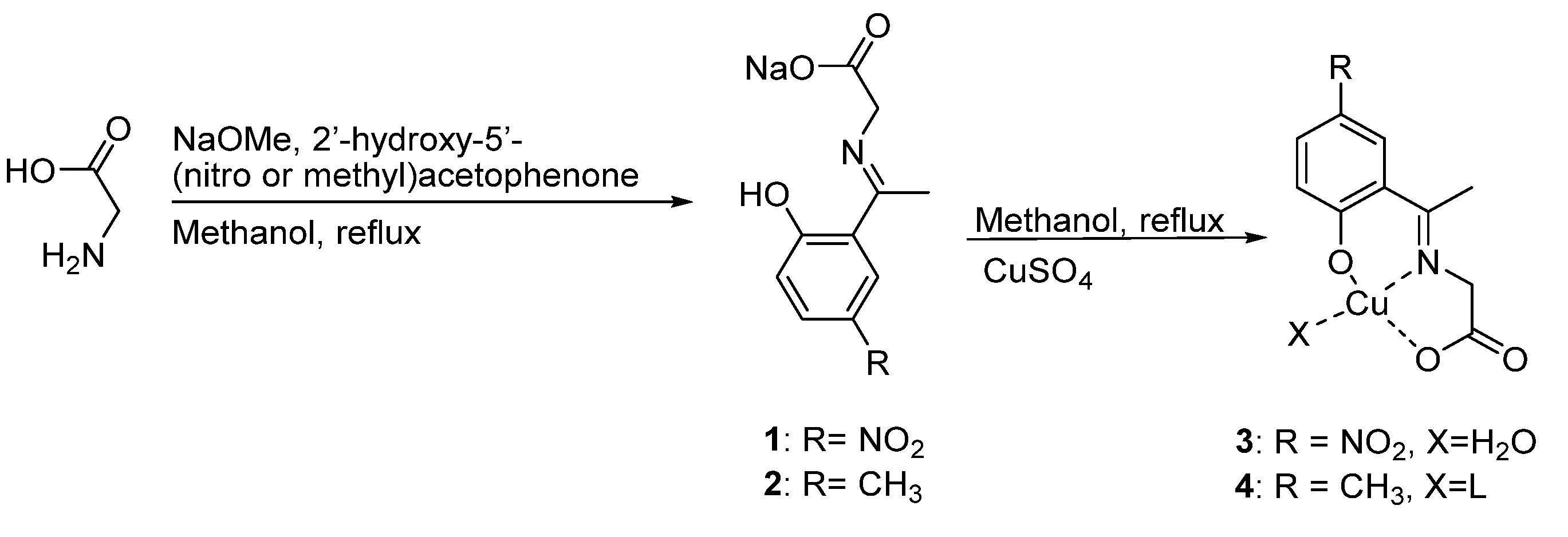
2.1. Synthesis of Schiff Bases Ligands and Copper (II) Complexes
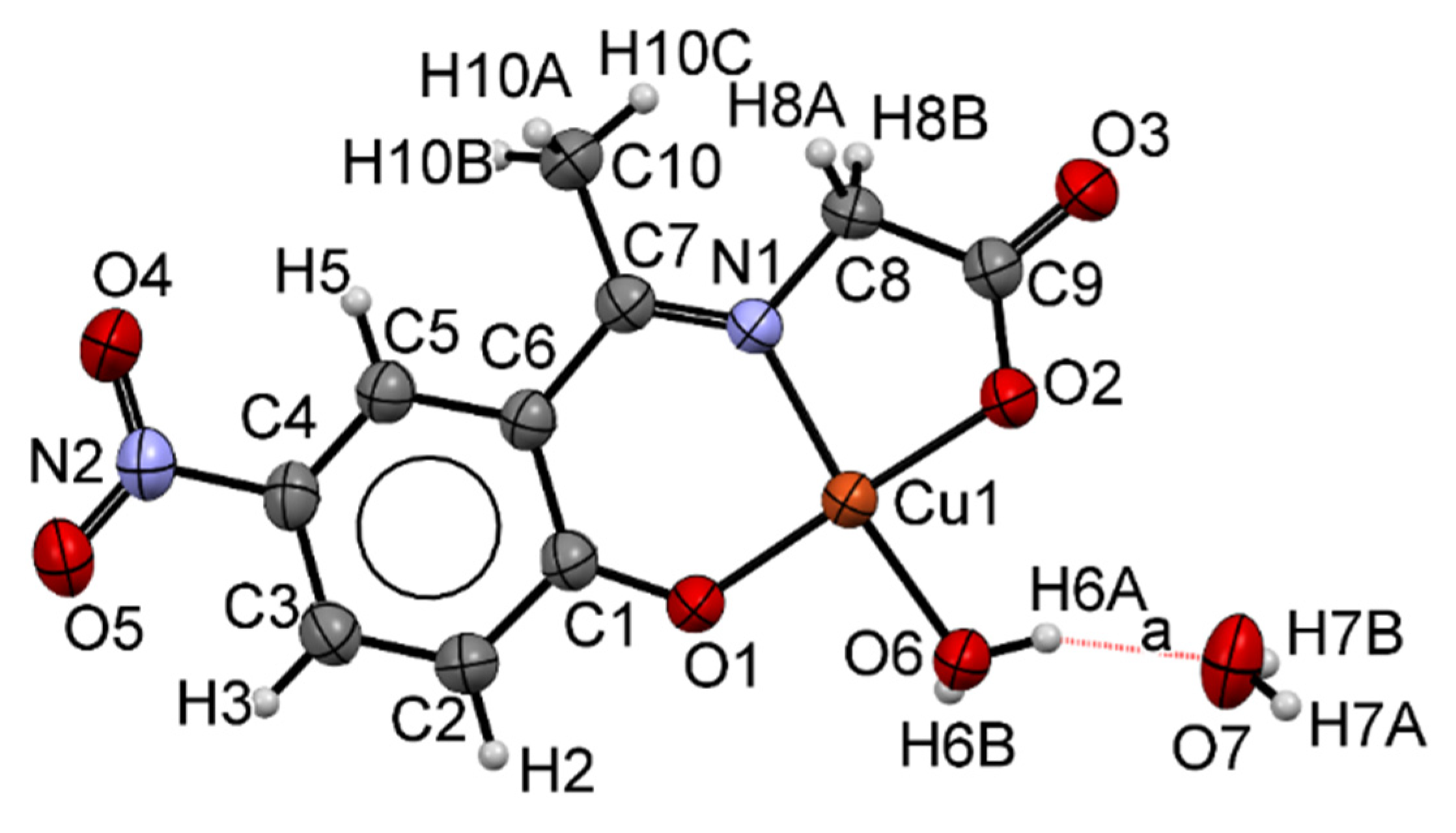
2.2. Single Crystal X-ray Diffraction (SCXRD)
2.3. FTIR Spectra Analysis of Schiff Bases Ligands and Copper (II) Complexes
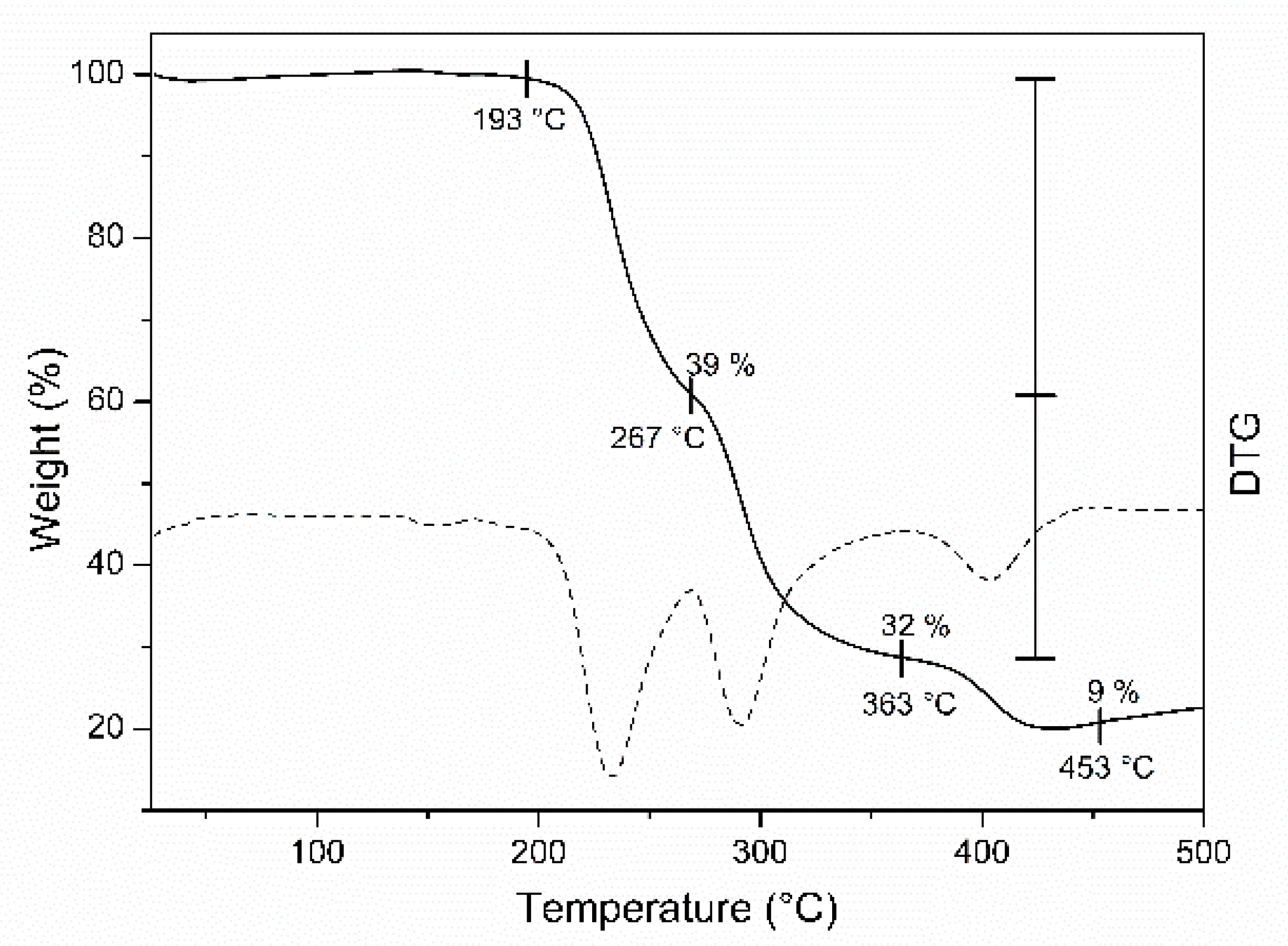
2.4. Thermal Analysis of the Copper (II) Complexes
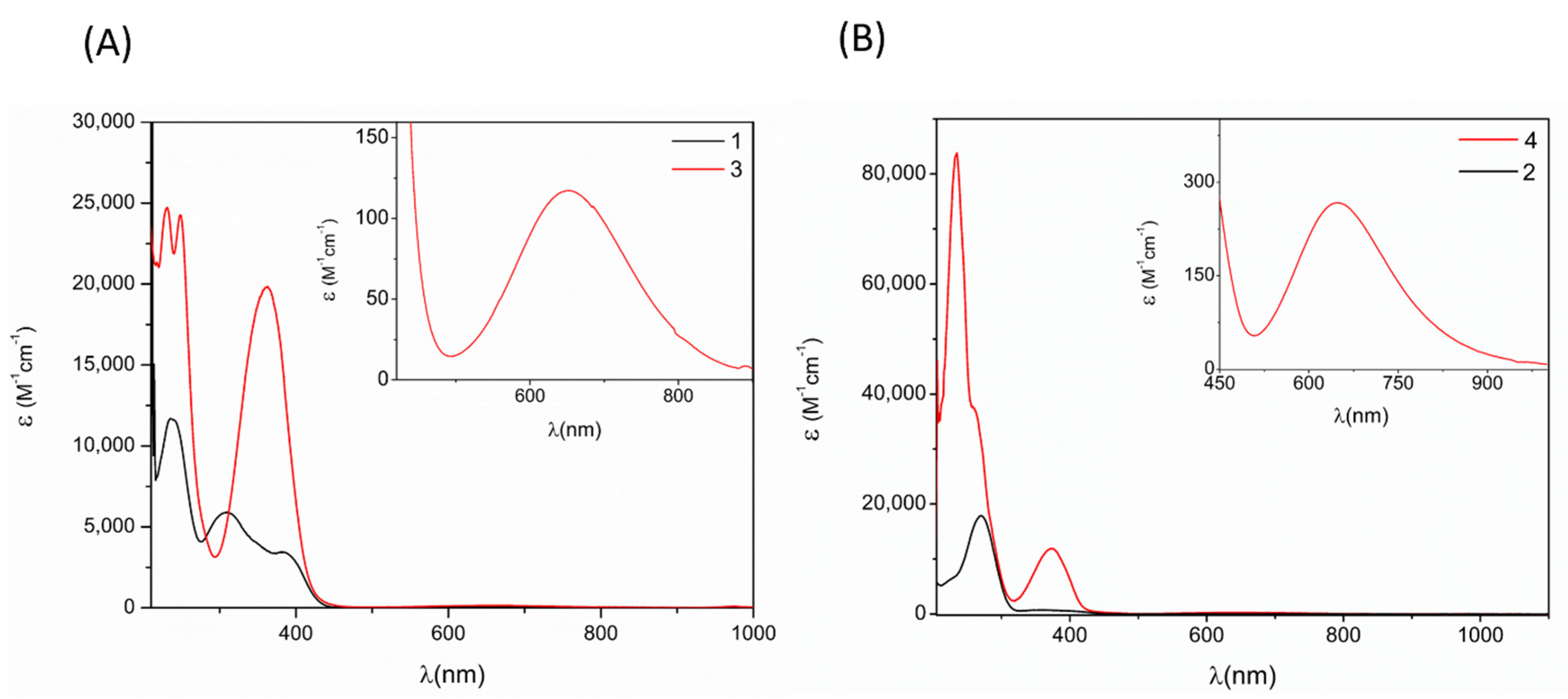
2.5. UV-Vis Spectroscopy of Schiff Bases Ligands and Copper (II) Complexes
2.6. Electron Paramagnetic Resonance of Copper (II) Complexes
2.7. Catalytic Activity of Copper (II) Complexes in Olefin Cyclopropanation
3. Materials and Methods
3.1. General Procedure
3.2. Synthesis of the Copper (II) Complexes
3.3. X-ray Crystallography
3.4. Catalytic Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Subin Kumar, K.; Aravindakshan, K.K. Synthesis, cytotoxic, anticancer and antimicrobial activities of some metal complexes of a novel tetradentate Schiff base ligand, (E)-3-((2-((E)-(1-(2-hydroxyphenyl)ethylidene)amino)ethyl)imino)-N-phenylbutanamide. Results Chem. 2021, 3, 100129. [Google Scholar] [CrossRef]
- Garba, H.W.; Abdullahi, M.S.; Shazwan, M.; Jamil, S.; Endot, N.A. Efficient Catalytic Reduction of 4-Nitrophenol Using Copper ( II ) Complexes wth N,O-Chelating Schiff Base Ligands. Molecules 2021, 26, 5876. [Google Scholar] [CrossRef] [PubMed]
- Dalia, S.A.; Afsan, F.; Hossain, M.S.; Khan, M.N.; Zakaria, C.M.; Zahan, M.K.E.; Ali, M.M. A short review on chemistry of schiff base metal complexes and their catalytic application. Int. J. Chem. Stud. 2018, 6, 2859–2866. [Google Scholar]
- Francesconi, V.; Cichero, E.; Schenone, S.; Naesens, L.; Tonelli, M. Synthesis and Biological Evaluation of Novel (thio)semicarbazone-Based Benzimidazoles as Antiviral Agents against Human Respiratory Viruses. Molecules 2020, 25, 1487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Bindary, A.A.; El-Sonbati, A.Z.; Diab, M.A.; Ghoneim, M.M.; Serag, L.S. Polymeric complexes—LXII. Coordination chemistry of supramolecular Schiff base polymer complexes—A review. J. Mol. Liq. 2016, 216, 318–329. [Google Scholar] [CrossRef]
- Yoon, T.P.; Jacobsen, E.N. Privileged Chiral Catalysts. Science 2003, 299, 1961–1963. [Google Scholar] [CrossRef]
- Hossain, S.M.; Roy, P.K.; Kudrat-E-Zahan, M.; Zakaria, C.M. Selected Schiff base coordination complexes and their microbial application: A review. Int. J. Chem. Stud. 2018, 6, 19–31. [Google Scholar]
- Pramanik, H.A.R.; Das, D.; Paul, P.C.; Mondal, P.; Bhattacharjee, C.R. Newer mixed ligand Schiff base complexes from aquo-N-(2′-hydroxy acetophenone) glycinatocopper(II) as synthon: DFT, antimicrobial activity and molecular docking study. J. Mol. Struct. 2014, 1059, 309–319. [Google Scholar] [CrossRef]
- Wang, C.Y.; Cao, F.; Wang, P.; Wu, X.; Yuan, C.J. Bis{2-[2-(isopropyl-ammonio)ethyliminomethyl]-5-methoxyphenolato}copper(II) bis-(perchlorate). Acta Crystallogr. Sect. E Struct. Reports Online 2010, 66, m669–m670. [Google Scholar] [CrossRef]
- Warda, S.A. A (4-ethylpyridine)(N-salicylideneglycinato)copper(II) tetramer. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 1997, 53, 697–699. [Google Scholar] [CrossRef]
- Pradhan, R.; Thomas, A.M.; Mukherjee, A.; Dhar, S.; Nethaji, M.; Chakravarty, A.R. Synthesis, crystal structure and DNA hydrolysis activity of ternary (N-salicylidene-L-methioninato) copper(II) complexes of heterocyclic bases. Indian J. Chem. Sect. A Inorganic, Phys. Theor. Anal. Chem. 2005, 44, 18–26. [Google Scholar]
- Usman, A.; Fun, H.K.; Baul, T.S.B.; Paul, P.C. Aqua[{[2-(2-hydroxyphenyl)ethylidene]amino}-acetato]copper(II) monohydrate. Acta Crystallogr. Sect. E Struct. Reports Online 2003, 59, 438–440. [Google Scholar] [CrossRef] [Green Version]
- Butcher, R.J.; Mockler, G.M.; McKern, O. (Piperidine-κN)[N-(salicylidene)phenylalaninato-κ 3O′N′O′]copper(II). Acta Crystallogr. Sect. E Struct. Reports Online 2003, 59, 61–63. [Google Scholar] [CrossRef]
- Oohora, K.; Meichin, H.; Zhao, L.; Wolf, M.W.; Nakayama, A.; Hasegawa, J.Y.; Lehnert, N.; Hayashi, T. Catalytic Cyclopropanation by Myoglobin Reconstituted with Iron Porphycene: Acceleration of Catalysis due to Rapid Formation of the Carbene Species. J. Am. Chem. Soc. 2017, 139, 17265–17268. [Google Scholar] [CrossRef]
- Bartoli, G.; Bencivenni, G.; Dalpozzo, R. Asymmetric cyclopropanation reactions. Synthesis 2014, 46, 979–1029. [Google Scholar] [CrossRef]
- Li, Z.; Liu, G.; Zheng, Z.; Chen, H. Asymmetric Cyclopropanation of Styrene Catalyzed by Cu –( Chiral Schiff-Base ) Complexes. Tetrahedron 2000, 56, 7187–7191. [Google Scholar] [CrossRef]
- Choudhary, N.F.; Connelly, N.G.; Hitchcock, P.B.; Leigh, G.J. New compounds of tetradentate Schiff bases with vanadium(IV) and vanadium(V). J. Chem. Soc. Dalt. Trans. 1999, 4437–4446. [Google Scholar] [CrossRef]
- Chen, D.; Martell, A.E. Dioxygen Affinities of Synthetic Cobalt Schiff Base Complexes. Inorg. Chem. 1987, 26, 1026–1030. [Google Scholar] [CrossRef]
- García-Raso, A.; Fiol, J.J.; López-Zafra, A.; Cabrero, A.; Castro, J.; Mata, I.; Molins, E. Crystal structure of the copper ( II ) ternary complex of N-salicylidene- Toxicity studies against Drosophila melanogaster. Polyhedron. 2003, 22, 403–409. [Google Scholar] [CrossRef]
- Zhao, G.Q.; Tian, D.M.; Han, Y.J.; Xue, L.W.; Peng, Q.L. Aqua-{N-[1-(2-oxidophen-yl)ethyl-idene]-l-serinato}copper(II) monohydrate. Acta Crystallogr. Sect. E Struct. Reports Online 2009, 65, m1505. [Google Scholar] [CrossRef] [Green Version]
- Sun, Y.X.; Xu, L.; Zhao, T.H.; Liu, S.H.; Liu, G.H.; Dong, X.T. Synthesis and crystal structure of a 3D supramolecular copper(II) complex with 1-(3-{[(E)-3-bromo-5-chloro-2-hydroxybenzylidene]amino}phenyl) ethanone oxime. Synth. React. Inorganic, Met. Nano-Metal Chem. 2013, 43, 509–513. [Google Scholar] [CrossRef]
- You, Z.L.; Zhang, L.; Shi, D.H.; Wang, X.L.; Li, X.F.; Ma, Y.P. Synthesis, crystal structures and urease inhibitory activity of copper(II) complexes with Schiff bases. Inorg. Chem. Commun. 2010, 13, 996–998. [Google Scholar] [CrossRef]
- Allen, F.H.; Kennard, O.; Watson, D.G.; Brammer, L.; Orpen, A.G.; Taylor, R. Tables of bond lengths determined by x-ray and neutron diffraction. Part 1. Bond lengths in organic compounds. J. Chem. Soc. Perkin Trans. 1987, 2, 1–19. [Google Scholar] [CrossRef]
- Soberanes, Y.; López-Gastélum, K.A.; Moreno-Urbalejo, J.; Salazar-Medina, A.J.; del Carmen Estrada-Montoya, M.; Sugich-Miranda, R.; Hernandez-Paredes, J.; Gonzalez-Córdova, A.F.; Vallejo-Cordoba, B.; Sotelo-Mundo, R.R.; et al. Tetrameric copper(II) metallocyclic complex bearing an amino acid derived Schiff base ligand: Structure, catalytic and antioxidant activities. Inorg. Chem. Commun. 2018, 94, 139–141. [Google Scholar] [CrossRef]
- Tang, G.; Lin, Y.J.; Jin, G.X. Syntheses, structures and catalytic activity of copper(II) complexes with hydroxyindanimine ligands. J. Organomet. Chem. 2007, 692, 4106–4112. [Google Scholar] [CrossRef]
- Carlini, C.; Giaiacopi, S.; Marchetti, F.; Pinzino, C.; Galletti, A.M.R.; Sbrana, G. Vinyl polymerization of norbornene by bis(salicylaldiminate)copper(H)/methylalumoxane catalysts. Organometallics 2006, 25, 3659–3664. [Google Scholar] [CrossRef]
- Sundaravel, K.; Suresh, E.; Palaniandavar, M. Synthesis, structures, spectral and electrochemical properties of copper(II) complexes of sterically hindered Schiff base ligands. Inorganica Chim. Acta 2009, 362, 199–207. [Google Scholar] [CrossRef]
- Garg, B.S.; Kurup, M.P.; Jain, S.K.; Bhoon, Y.K. Manganese (II) complexes of substituted thio- and selenosemicarbazones of 2-acetylpyridine: Esr, magnetic and electronic spectral studies. Transit. Met. Chem. 1988, 13, 92–95. [Google Scholar] [CrossRef]
- Sreekanth, A.; Fun, H.K.; Kurup, M.R.P. Formation of first gold ( III ) complex of an N ( 4 ) -disubstituted thiosemicarbazone derived from 2-benzoylpyridine : Structural and spectral studies. Inorg. Chem. Commun. 2004, 7, 1250–1253. [Google Scholar] [CrossRef]
- Marburg, P. The Chain Polymers (Imidazole)(N-salicylideneglycinato)copper(II) and (2-Ethylimidazole)(N-salicylideneglycinato)copper(II). Acta Crystallogr. Sect. C: Cryst. Struct. Commun. 1997, 334, 1590–1593. [Google Scholar] [CrossRef]
- Tyagi, P.; Chandra, S.; Saraswat, B.S.; Yadav, D. Design, spectral characterization, thermal, DFT studies and anticancer cell line activities of Co(II), Ni(II) and Cu(II) complexes of Schiff bases derived from 4-amino-5-(pyridin-4-yl)-4H-1,2,4-triazole-3-thiol. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 145, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Tyagi, M.; Chandra, S.; Akhtar, J.; Chand, D. Modern spectroscopic technique in the characterization of biosensitive macrocyclic Schiff base ligand and its complexes : Inhibitory activity against plantpathogenic fungi. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 118, 1056–1061. [Google Scholar] [CrossRef] [PubMed]
- Geary, W.J. The use of conductivity measurements in organic solvents. Coord. Chem. Rev. 1971, 7, 81–122. [Google Scholar] [CrossRef]
- Salazar-Medina, A.J.; Gámez-Corrales, R.; Ramírez, J.Z.; González-Aguilar, G.A.; Velázquez-Contreras, E.F. Characterization of metal-bound water in bioactive Fe(III)-cyclophane complexes. J. Mol. Struct. 2018, 1154, 225–231. [Google Scholar] [CrossRef]
- Kavitha, N.; Anantha Lakshmi, P.V. Synthesis, characterization and thermogravimetric analysis of Co(II), Ni(II), Cu(II) and Zn(II) complexes supported by ONNO tetradentate Schiff base ligand derived from hydrazino benzoxazine. J. Saudi Chem. Soc. 2017, 21, S457–S466. [Google Scholar] [CrossRef] [Green Version]
- Refat, M.S.; El-Deen, I.M.; Ibrahim, H.K.; El-Ghool, S. Synthesis and spectroscopic studies of some transition metal complexes of a novel Schiff base ligands derived from 5-phenylazo-salicyladehyde and o-amino benzoic acid. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2006, 65, 1208–1220. [Google Scholar] [CrossRef]
- Soliman, A.A.; Mohamed, G.G. Study of the ternary complexes of copper with salicylidene-2- aminothiophenol and some amino acids in the solid state. Thermochim. Acta 2004, 421, 151–159. [Google Scholar] [CrossRef]
- Shobana, S.; Subramaniam, P.; Mitu, L.; Dharmaraja, J.; Arvind Narayan, S. Synthesis, structural elucidation, biological, antioxidant and nuclease activities of some 5-Fluorouracil-amino acid mixed ligand complexes. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2015, 134, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Bagihalli, G.B.; Avaji, P.G.; Patil, S.A.; Badami, P.S. Synthesis, spectral characterization, in vitro antibacterial, antifungal and cytotoxic activities of Co(II), Ni(II) and Cu(II) complexes with 1,2,4-triazole Schiff bases. Eur. J. Med. Chem. 2008, 43, 2639–2649. [Google Scholar] [CrossRef]
- Rahman, L.H.A.; Abu-dief, A.M.; Hamdan, S.K.; Seleem, A.A. Nano Structure Iron ( II ) and Copper ( II ) Schiff Base Complexes of a NNO-Tridentate Ligand as New Antibiotic Agents : Spectral, Thermal Behaviors and DNA Binding Ability 2 Experimental 2. 1 Materials and methods. Int. J. Nanomater. Chem. 2015, 77, 65–77. [Google Scholar] [CrossRef]
- Reena, T.A.; Kurup, M.R.P. Copper(II) complexes derived from di-2-pyridyl ketone-N4-phenyl-3-semicarbazone: Synthesis and spectral studies. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2010, 76, 322–327. [Google Scholar] [CrossRef]
- Ran, X.; Wang, L.; Cao, D.; Lin, Y.; Hao, J. Synthesis, characterization and in vitro biological activity of cobalt(II), copper(II) and zinc(II) Schiff base complexes derived from salicylaldehyde and D,L-selenomethionine. Appl. Organomet. Chem. 2011, 25, 9–15. [Google Scholar] [CrossRef]
- Ünver, H.; Hayvali, Z. Spectrochimica Acta Part A : Molecular and Biomolecular Spectroscopy Synthesis, spectroscopic studies and structures of square-planar nickel ( II ) and copper ( II ) complexes derived from. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 2010, 75, 782–788. [Google Scholar] [CrossRef]
- Tabbì, G.; Giuffrida, A.; Bonomo, R.P. Determination of formal redox potentials in aqueous solution of copper(II) complexes with ligands having nitrogen and oxygen donor atoms and comparison with their EPR and UV-Vis spectral features. J. Inorg. Biochem. 2013, 128, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Maki, A.H.; McGarvey, B.R. Electron spin resonance in transition metal chelates. I. Copper (II) bis-acetylacetonate. J. Chem. Phys. 1958, 29, 31–34. [Google Scholar] [CrossRef]
- Hathaway, B.J.; Billing, D.E. The electronic properties and stereochemistry of mono-nuclear complexes of the copper(II) ion. Coord. Chem. Rev. 1970, 5, 143–207. [Google Scholar] [CrossRef]
- Jain, S.K.; Garg, B.S.; Bhoon, Y.K. Characterization of copper(II) complexes of N4,N4-disubstituted thiosemicarbazones of 2-acetylpyridine by combined evaluation of electronic and ESR parameters. Spectrochim Acta A Mol Spectrosc. 1986, 42, 959–968. [Google Scholar] [CrossRef]
- Aiswarya, N.; Sithambaresan, M.; Sreejith, S.S.; Ng, S.W.; Kurup, M.R.P. Polymeric polymorphs and a monomer of pseudohalide incorporated Cu(II) complexes of 2,4-dichlorido-6-((2-(dimethylamino)ethylimino)methyl)phenol]: Crystal structures and spectroscopic behavior. Inorganica Chim. Acta 2016, 443, 251–266. [Google Scholar] [CrossRef]
- Chelucci, G.; Cabras, M.A.; Saba, A. Enantioselective cyclopropanation of styrene with optically active nitrogen ligands based on the pyridine framework. J. Mol. Catal. A: Chem. 1995, 95, 7–10. [Google Scholar] [CrossRef]
- Iglesias, A.L.; Aguirre, G.; Somanathan, R.; Parra-hake, M. New chiral Schiff base—Cu ( II ) complexes as cyclopropanation catalysts. Polyhedron 2004, 23, 3051–3062. [Google Scholar] [CrossRef]
- Masterson, D.S.; Shirley, C.; Glatzhofer, D.T. N-(4-[2.2] paracyclophanyl)-2′-hydroxyacetophenone imine: An effective paracyclophane Schiff-base ligand for use in catalytic asymmetric cyclopropanation reactions. J. Mol. Catal. A Chem. 2012, 361–362, 111–115. [Google Scholar] [CrossRef]
- Youssef, N.S.; El-seidy, A.M.A.; Schiavoni, M.; Castano, B.; Ragaini, F.; Gallo, E.; Caselli, A. Thiosemicarbazone copper complexes as competent catalysts for ole fi n cyclopropanations. J. Organomet. Chem. 2012, 714, 94–103. [Google Scholar] [CrossRef] [Green Version]
- Youssef, N.S.; El-zahany, E.; El-seidy, A.M.A.; Caselli, A.; Cenini, S. Synthesis and characterization of some transition metal complexes with a novel Schiff base ligand and their use as catalysts for olefin cyclopropanation. J. Mol. Catal. A Chem. 2009, 308, 159–168. [Google Scholar] [CrossRef]
- Youssef, N.S.; El-zahany, E.; El-seidy, A.M.A.; Caselli, A.; Fantauzzi, S.; Cenini, S. Inorganica Chimica Acta Synthesis and characterisation of new Schiff base metal complexes and their use as catalysts for olefin cyclopropanation. Inorganica Chim. Acta 2014, 362, 2006–2014. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. Sect. A Found. Crystallogr. 2015, 71, 3–8. [Google Scholar] [CrossRef] [Green Version]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. OLEX2: A complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. [Google Scholar] [CrossRef]
- ABSPACK, S. CrysAlisPro, version 1.171. 36.32 2013; Agilent Technologies: Oxfordshire, UK, 2013. [Google Scholar]
- Clark, R.C.; Reid, J.S. The analytical calculation of absorption in multifaceted crystals. Acta Crystallogr. Sect. A 1995, 51, 887–897. [Google Scholar] [CrossRef]
- Farrugia, L.J. WinGX and ORTEP for Windows: An update. J. Appl. Crystallogr. 2012, 45, 849–854. [Google Scholar] [CrossRef]
- Spek, A.L. PLATON SQUEEZE: A tool for the calculation of the disordered solvent contribution to the calculated structure factors. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 9–18. [Google Scholar] [CrossRef] [Green Version]



| 3 | 4 | ||
|---|---|---|---|
| Cu (1)-O (1) | 1.851 (4) | Cu (1)-O (3) # 1 | 1.8560 (17) |
| Cu (1)-N (1) | 1.929 (3) | Cu (1)-N (1) # 1 | 1.9288 (18) |
| Cu (1)-O (2) | 1.902 (2) | Cu (1)-O (1) # 1 | 1.9467 (17) |
| Cu (1)-O (6) | 1.927 (2) | Cu (1)-O (2) | 1.9572 (15) |
| O (1)-Cu (1)-N (1) | 95.28 (11) | O (3) # 1-Cu (1)-N (1) # 1 | 95.19 (7) |
| O (1)-Cu (1)-O (2) | 176.10 (12) | O (3) # 1-Cu (1)-O (1) # 1 | 178.97 (7) |
| N (1)-Cu (1)-O (2) | 86.30 (11) | N (1) # 1-Cu (1)-O (1) # 1 | 85.82 (7) |
| O (1)-Cu (1)-O (6) | 89.40 (10) | O (3) # 1-Cu (1)-O (2) | 91.42 (7) |
| N (1)-Cu (1)-O (6) | 169.41 (12) | N (1) # 1-Cu (1)-O (2) | 173.12 (7) |
| O (2)-Cu (1)-O (6) | 89.64 (11) | O (1) # 1-Cu (1)-O (2) | 87.56 (7) |
| Compound | 1 | 3 | 2 | 4 |
|---|---|---|---|---|
| V (OH) | 3400 | 3065 | 3239 | - |
| v (C=O) | 1616 | 1601 | 1618 | 1605 |
| v (C=N) | 1616 | 1601 | 1618 | 1605 |
| vasym (COO−) | 1398 | 1433 | 1406 | 1452 |
| vsym (COO−) | 1342 | 1382 | 1350 | 1305 |
| v (N-O) | 1348 | 1484, 1324 | - | - |
| Complex | Molecular Formula | Stages | Temperature of TGA (°C) | Temperature of DTG (°C) | Mass Loss (%) | Assignment | |
|---|---|---|---|---|---|---|---|
| found | Calc. | ||||||
| 3 | C10H12CuN2O7 | I II III | 79–192 192–320 320–484 | 138 243 392 | 12 30 37 | 11 | 2 H2O |
| 4 | C44H44Cu4N4O12 | I II III | 193–267 267–363 363–453 | 233 291 403 | 39 32 9 | - | - |
| Compound | π–π * | n–π * | d–d |
|---|---|---|---|
| 1 | 241 | 382 | |
| 3 | 249 | 364 | 652 |
| 2 | 254 | 338 | |
| 4 | 267 | 377 | 638 |
| Compound | Polycrystalline State (298K) | Methanol Solution (77 K) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| giso | g|| | g┴ | G | g|| | g┴ | gav | A|| a | A┴ a | Aav a | |
| 3 | - | 2.203 | 2.047 | 4.4899 | 2.271 | 2.065 | 2.1336 | 164 | 14 | 64 |
| 4 | - | 2.231 | 2.086 | 2.7323 | 2.266 | 2.053 | 2.1240 | 166 | 13 | 64 |
| Complex | Conversion (%) b | Yield (%) c | trans/cisc |
|---|---|---|---|
| 3 | >99.9 | 96 | 98/2 |
| 4 | >99.9 | 90 | >99.9/<0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
López-Gastélum, K.-A.; Velázquez-Contreras, E.F.; García, J.J.; Flores-Alamo, M.; Aguirre, G.; Chávez-Velasco, D.; Narayanan, J.; Rocha-Alonzo, F. Mononuclear and Tetranuclear Copper(II) Complexes Bearing Amino Acid Schiff Base Ligands: Structural Characterization and Catalytic Applications. Molecules 2021, 26, 7301. https://doi.org/10.3390/molecules26237301
López-Gastélum K-A, Velázquez-Contreras EF, García JJ, Flores-Alamo M, Aguirre G, Chávez-Velasco D, Narayanan J, Rocha-Alonzo F. Mononuclear and Tetranuclear Copper(II) Complexes Bearing Amino Acid Schiff Base Ligands: Structural Characterization and Catalytic Applications. Molecules. 2021; 26(23):7301. https://doi.org/10.3390/molecules26237301
Chicago/Turabian StyleLópez-Gastélum, Karla-Alejandra, Enrique F. Velázquez-Contreras, Juventino J. García, Marcos Flores-Alamo, Gerardo Aguirre, Daniel Chávez-Velasco, Jayanthi Narayanan, and Fernando Rocha-Alonzo. 2021. "Mononuclear and Tetranuclear Copper(II) Complexes Bearing Amino Acid Schiff Base Ligands: Structural Characterization and Catalytic Applications" Molecules 26, no. 23: 7301. https://doi.org/10.3390/molecules26237301
APA StyleLópez-Gastélum, K.-A., Velázquez-Contreras, E. F., García, J. J., Flores-Alamo, M., Aguirre, G., Chávez-Velasco, D., Narayanan, J., & Rocha-Alonzo, F. (2021). Mononuclear and Tetranuclear Copper(II) Complexes Bearing Amino Acid Schiff Base Ligands: Structural Characterization and Catalytic Applications. Molecules, 26(23), 7301. https://doi.org/10.3390/molecules26237301








