Detecting Air Pollutant Molecules Using Tube-Shaped Single Electron Transistor
Abstract
1. Introduction
2. Materials and Methods
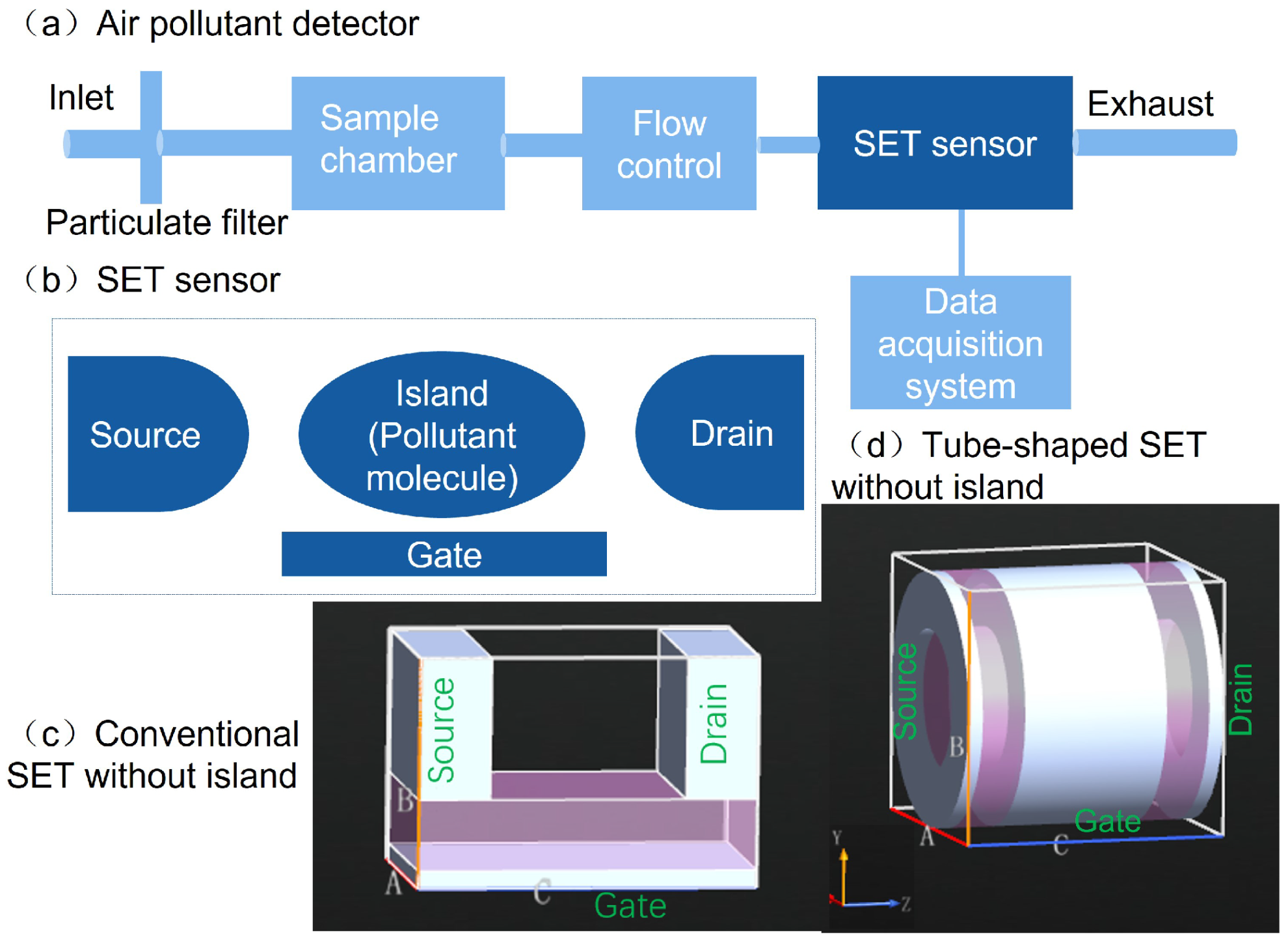
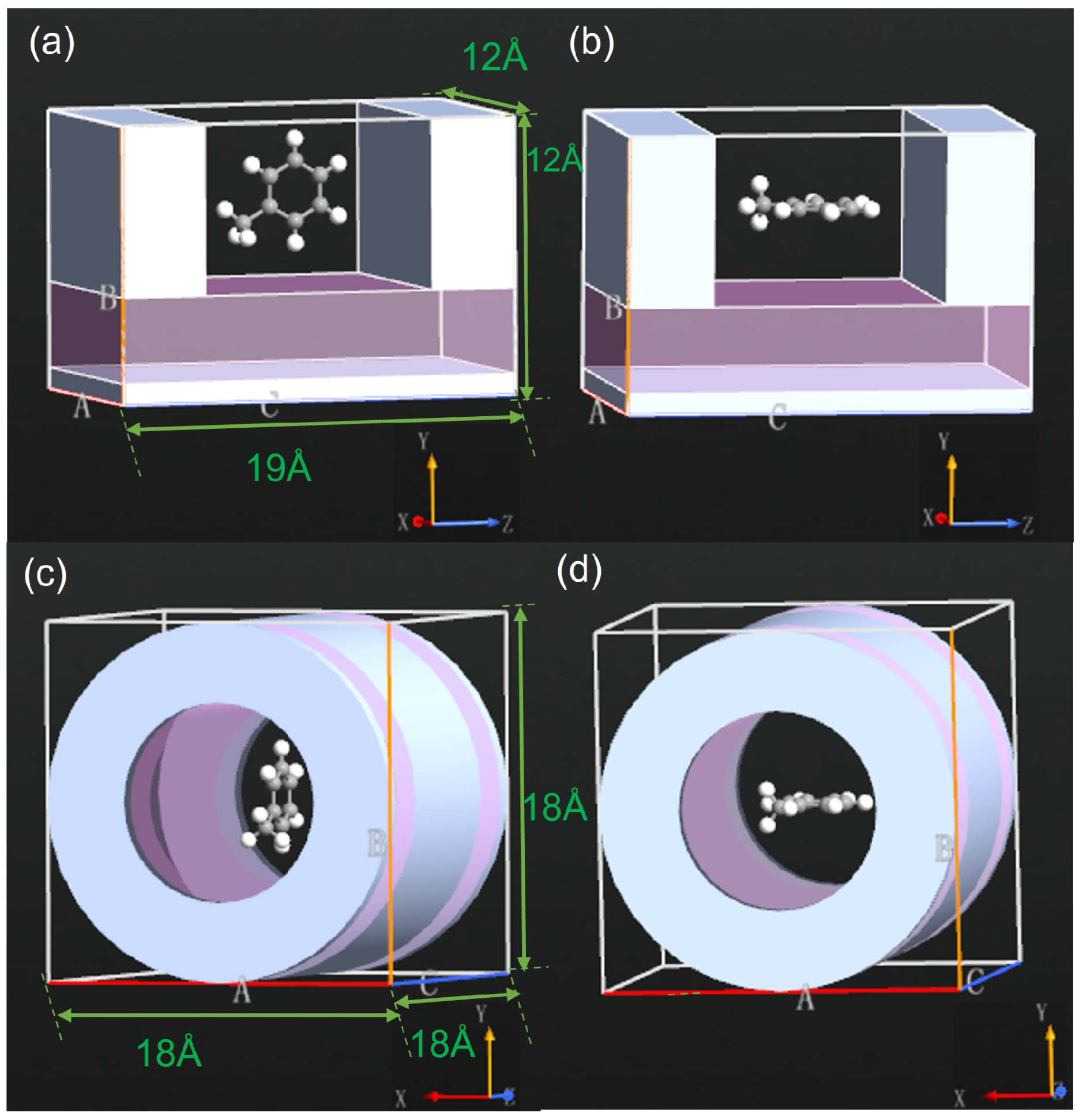
2.1. System Description
2.2. Computational Procedure
3. Results and Discussion
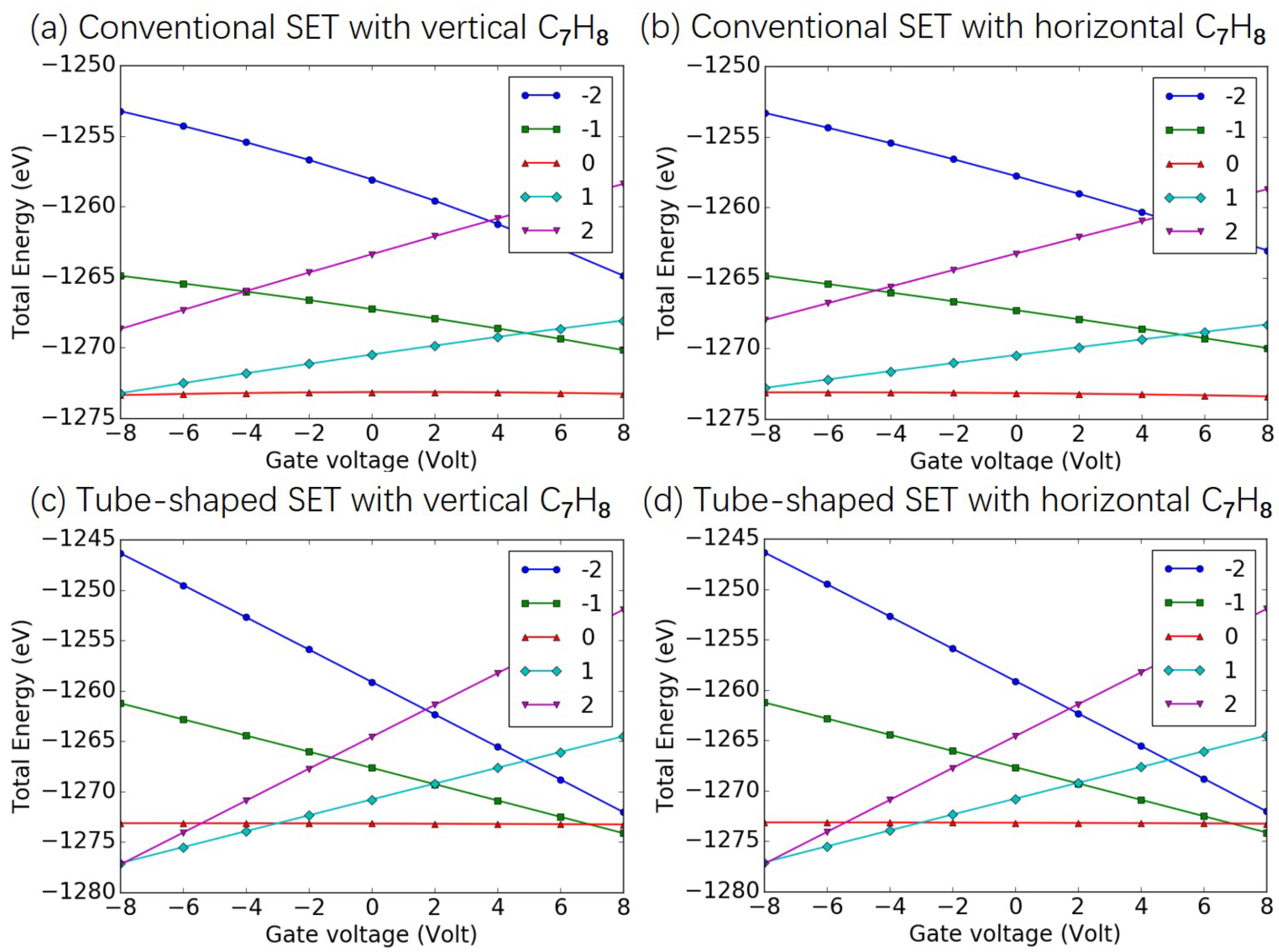
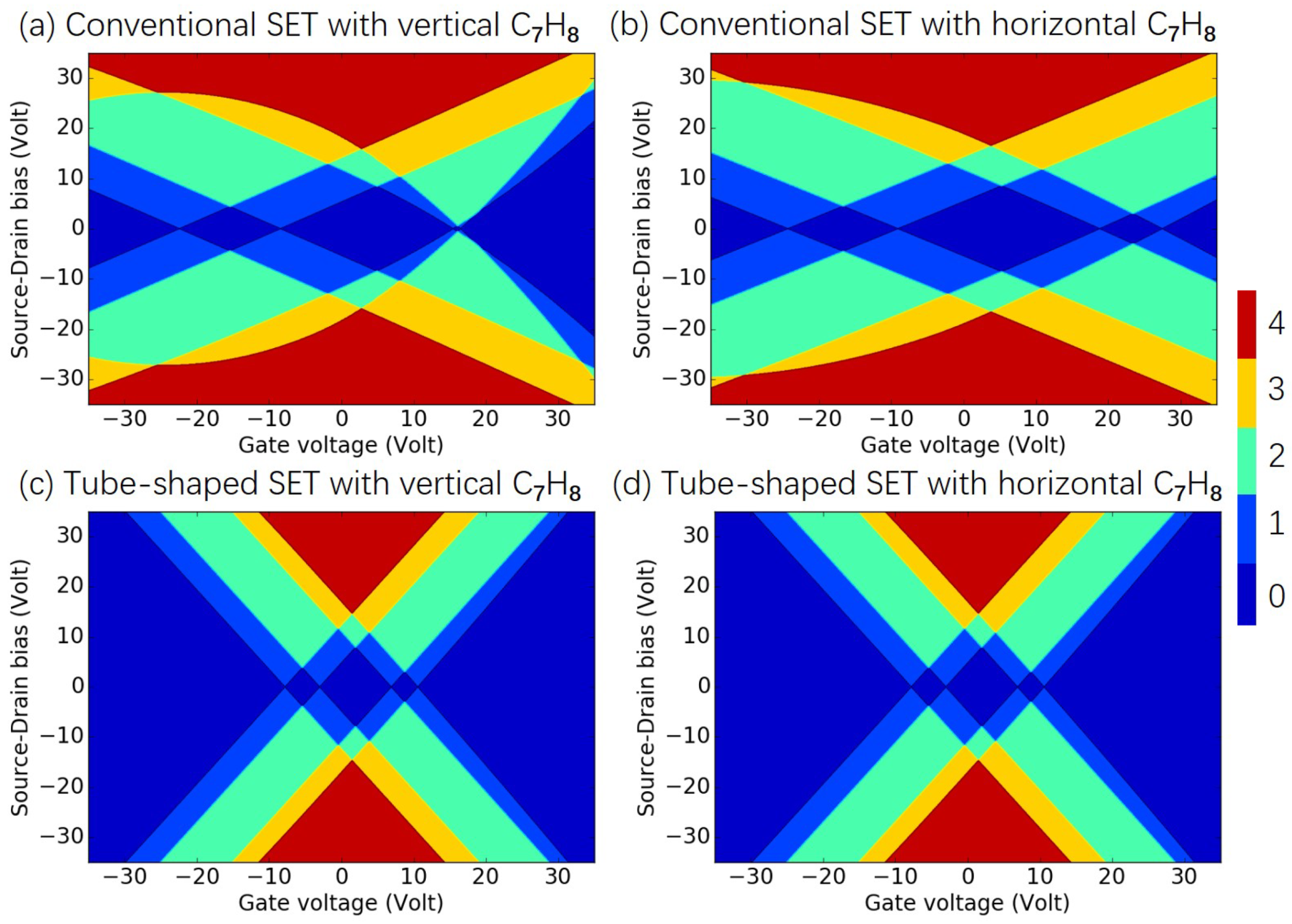
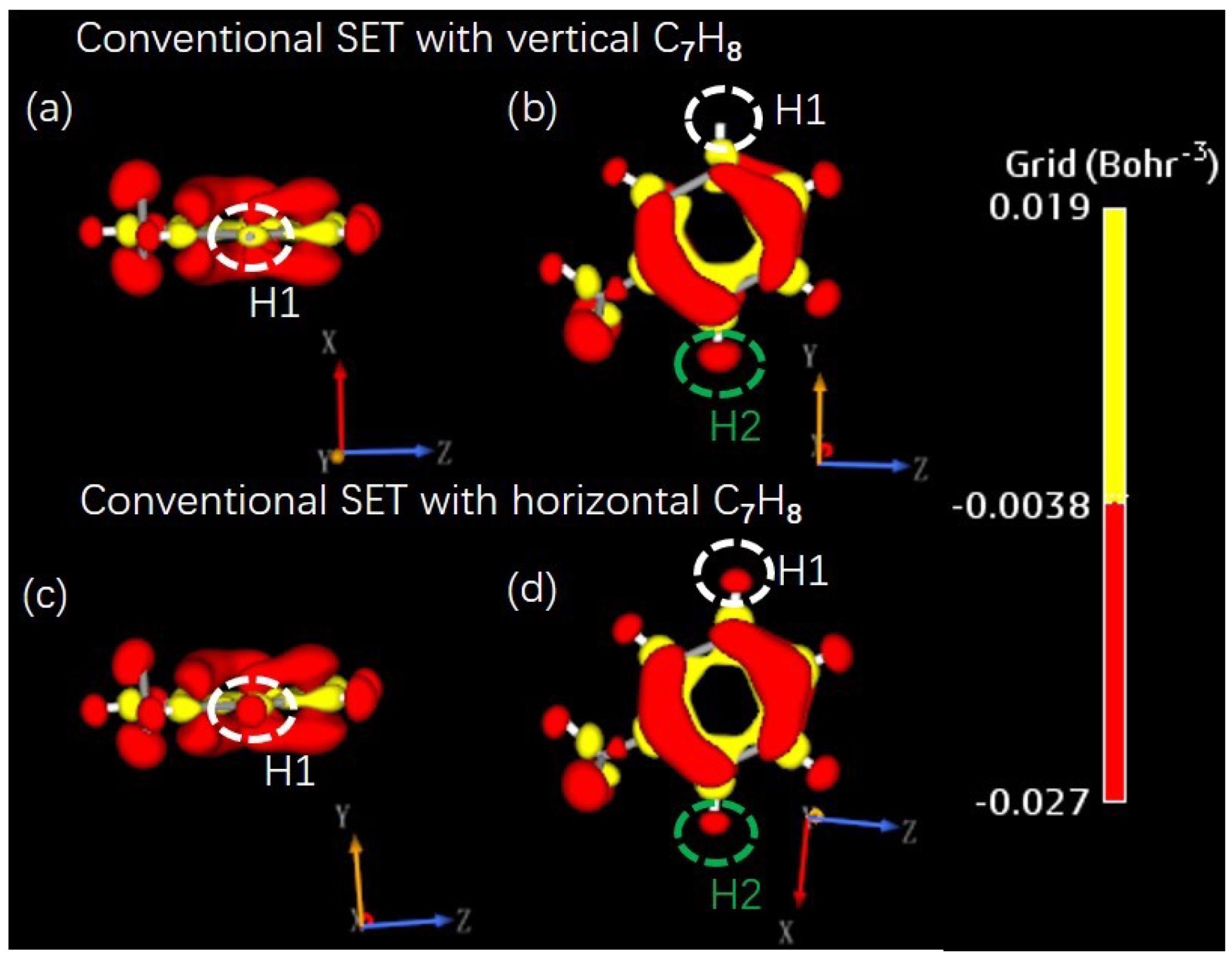
3.1. Effects of SET Structure
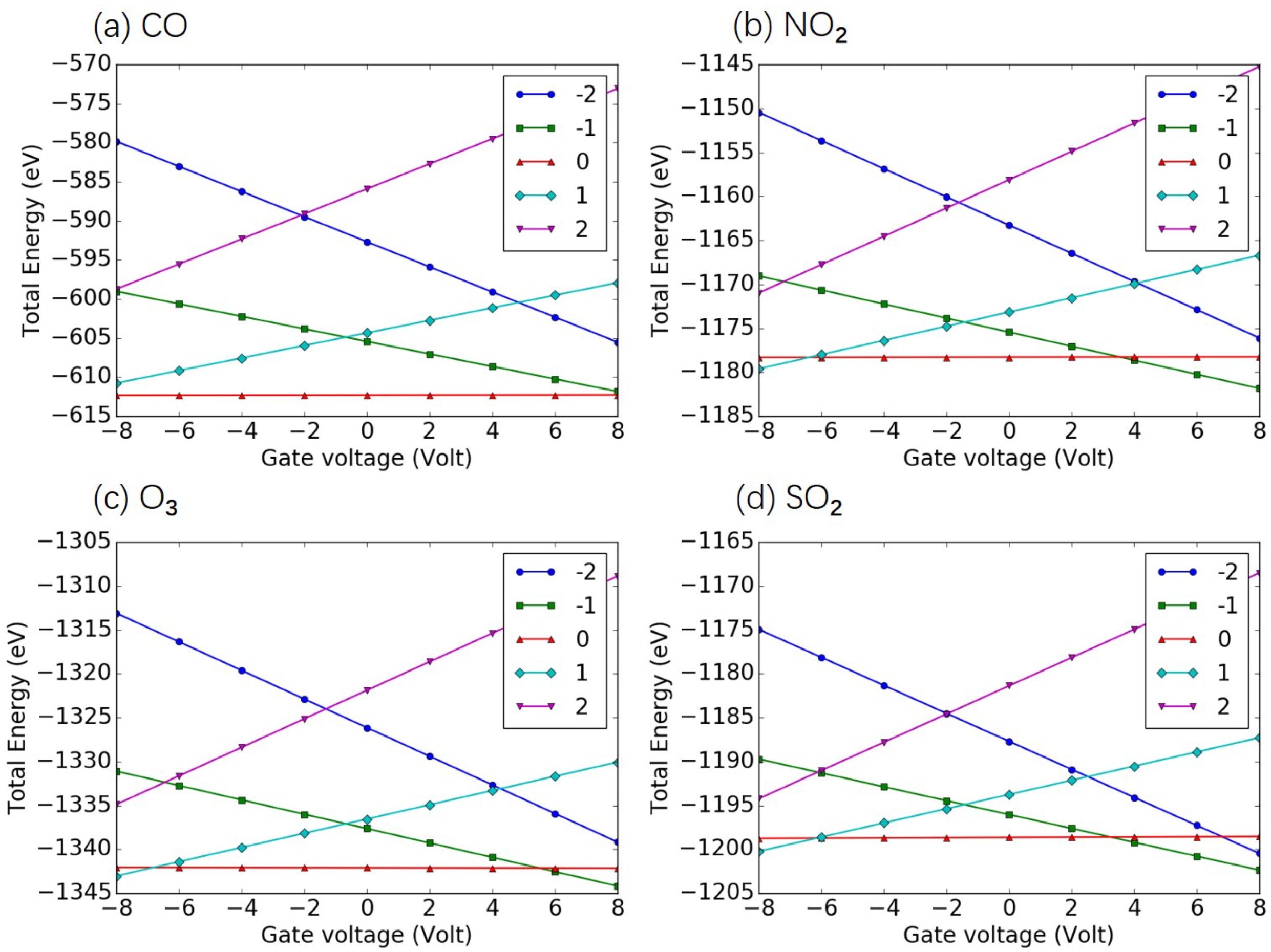
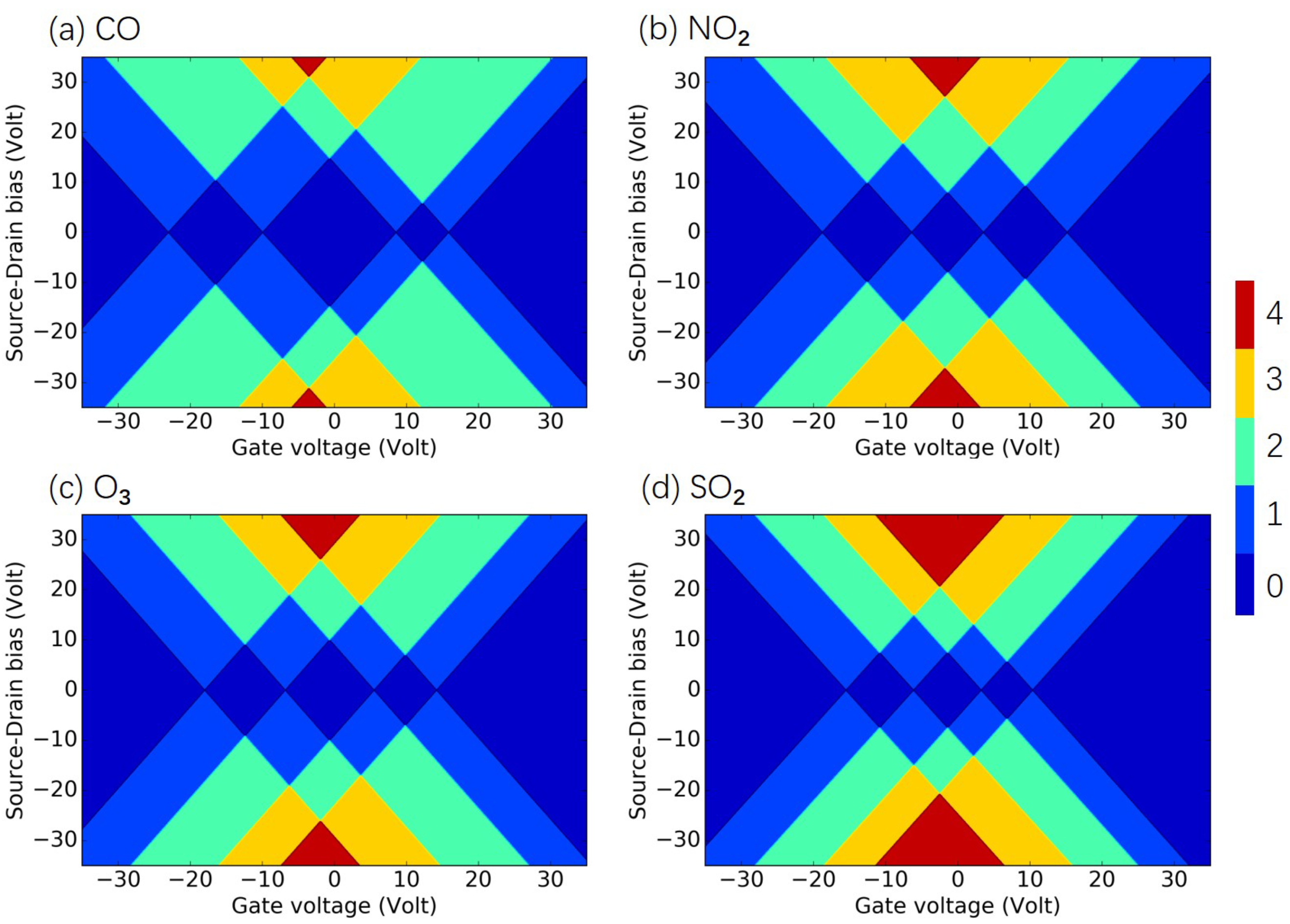
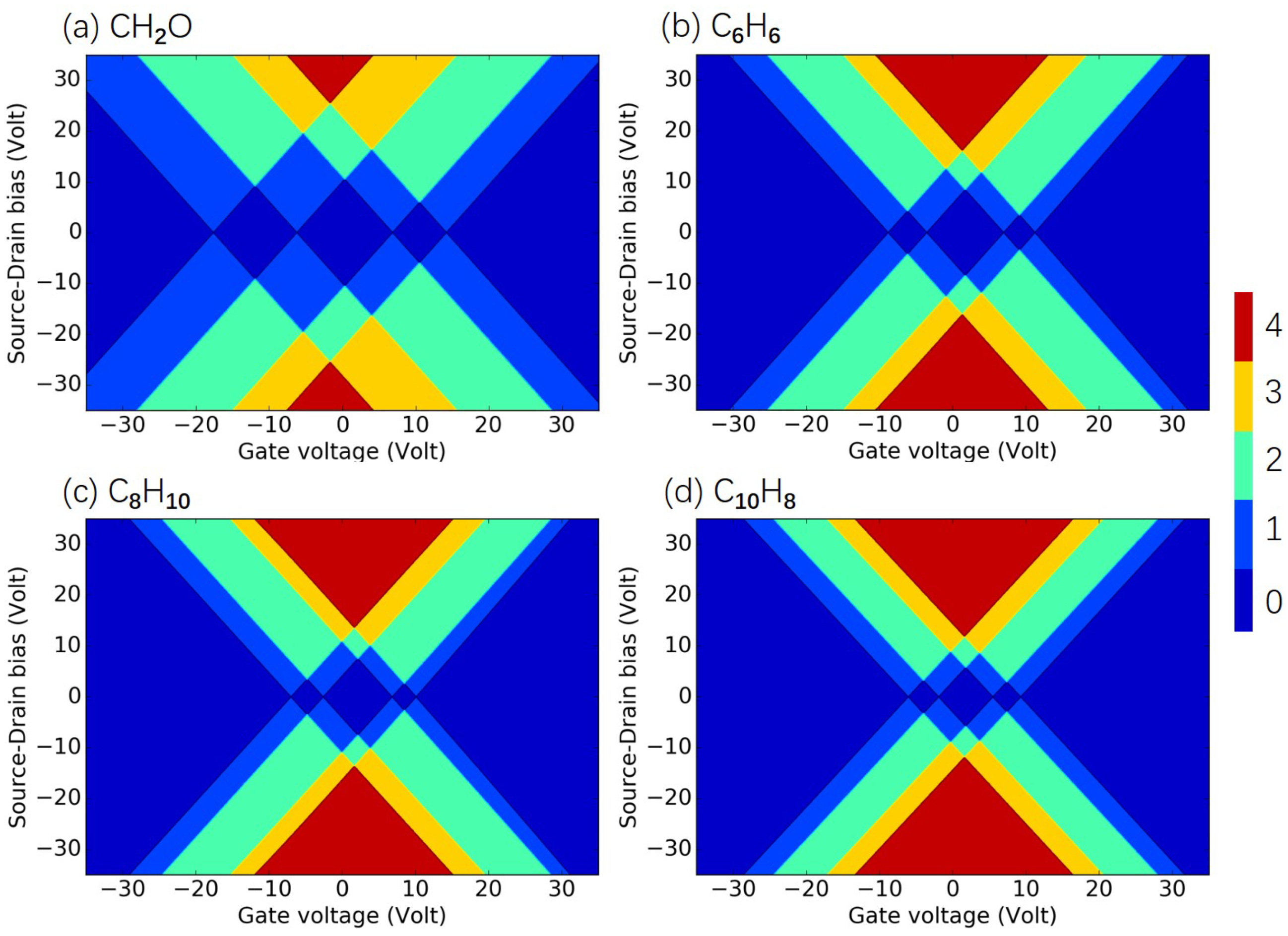
3.2. Identification of Air Pollutants
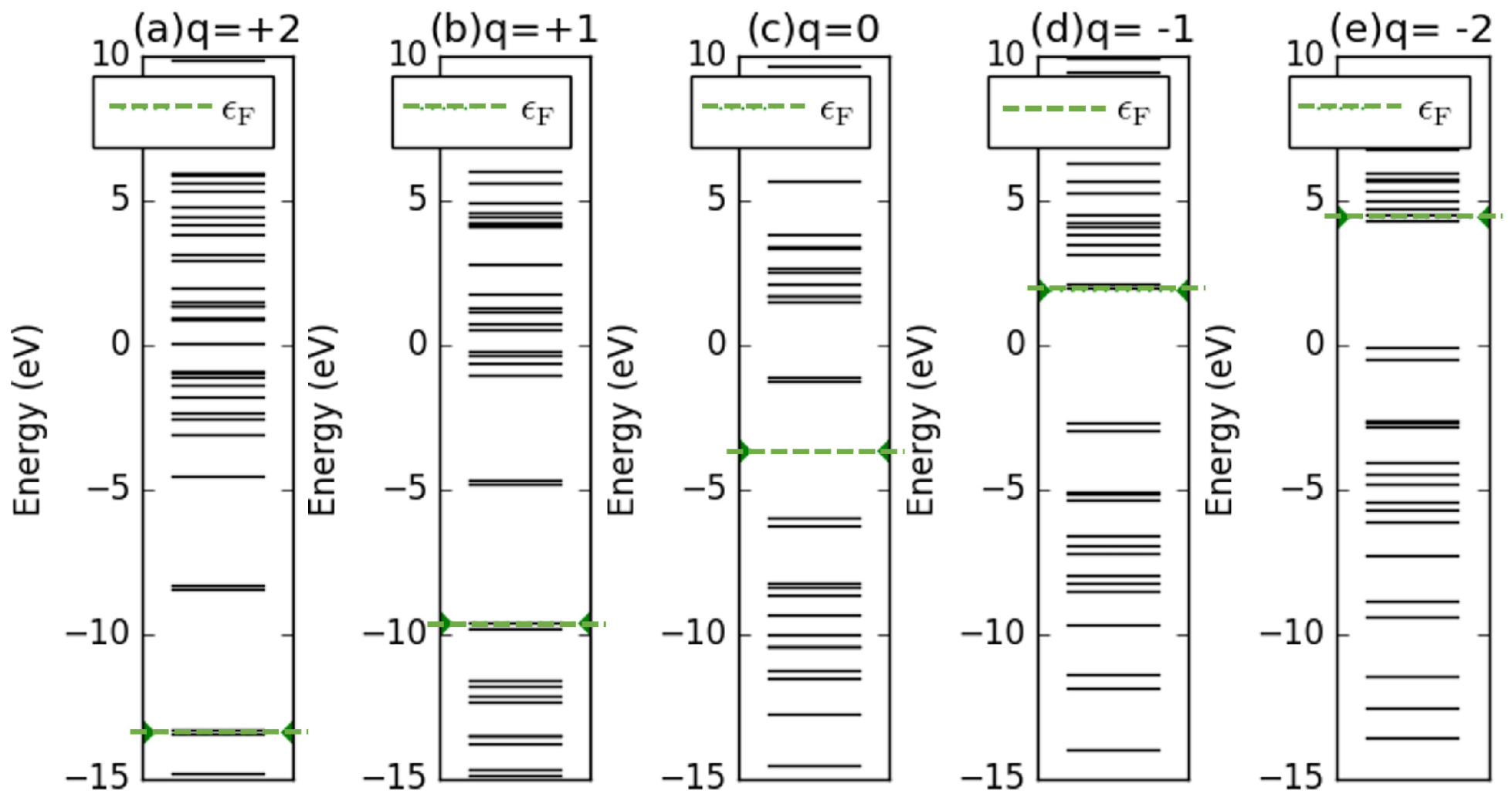
3.3. Quantification of Air Pollutants
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Working Principle of SET for Detection of Air Pollutant Molecules
| Island Molecule | CO | NO | O | SO | CHO | CH | CH | CH | CH |
|---|---|---|---|---|---|---|---|---|---|
| Length of a diagonal line in the central diamond along the gate voltage axis (V) | 18.5 | 9.9 | 12.3 | 9.3 | 13.1 | 10.4 | 9.9 | 9.4 | 7.4 |
| Length of a diagonal line in the central diamond along the source-drain bias (V) | 29.6 | 16.0 | 20.0 | 14.8 | 21 | 16.8 | 15.6 | 14.9 | 11.2 |
Appendix B. SET Configurations with Various Air Pollutant Molecules
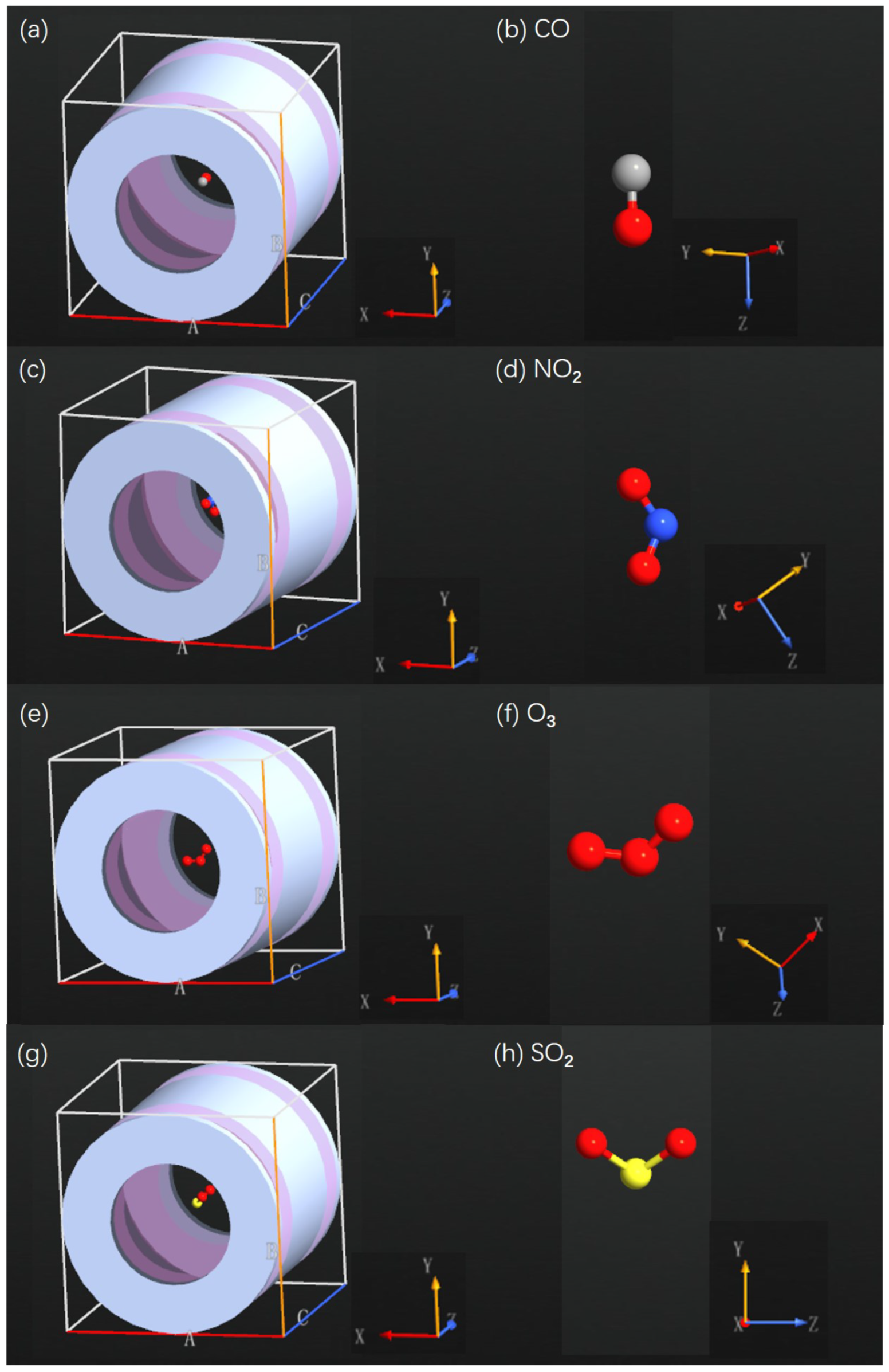
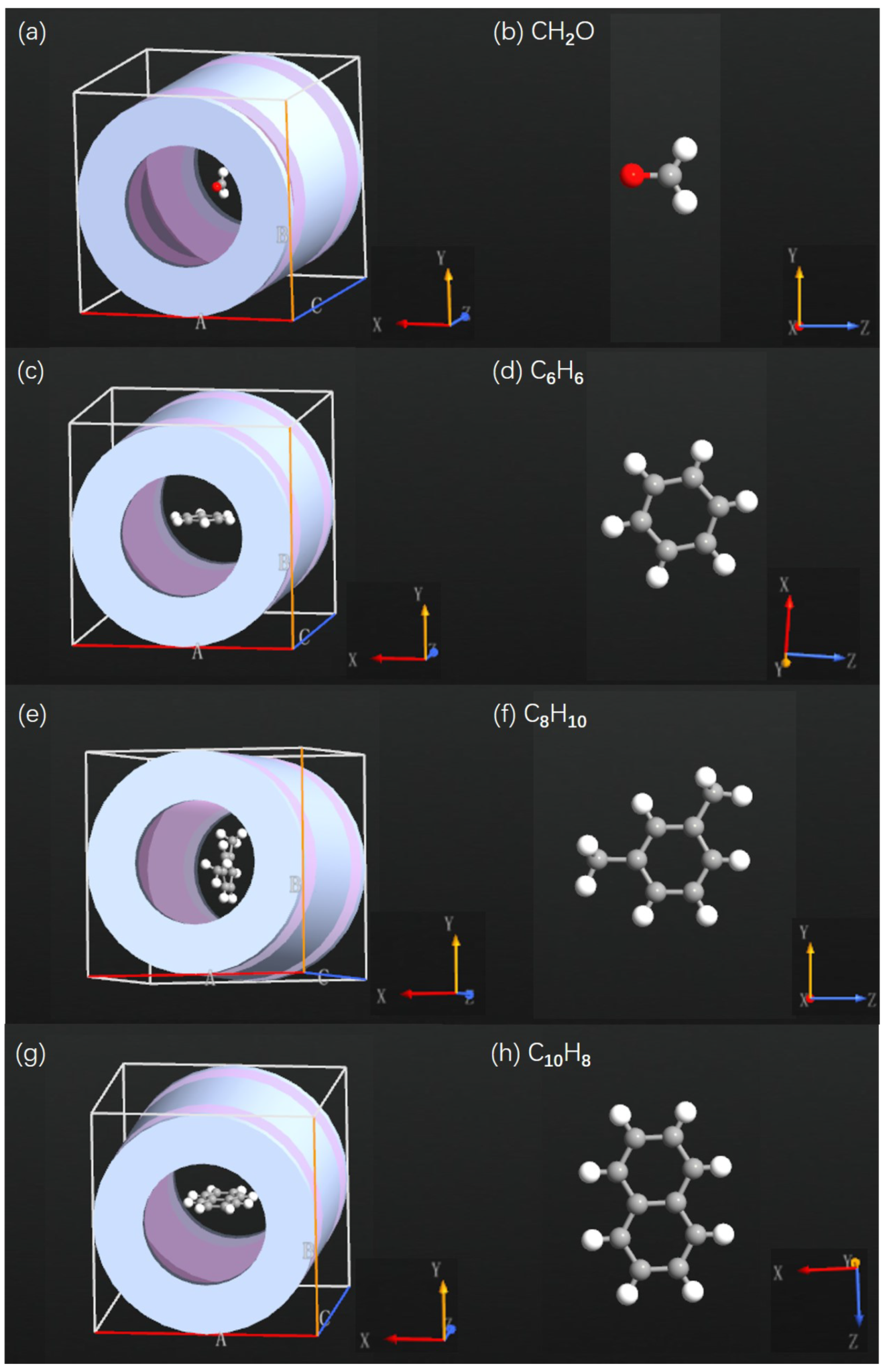
Appendix C. Total Energy as Function of Gate Voltage
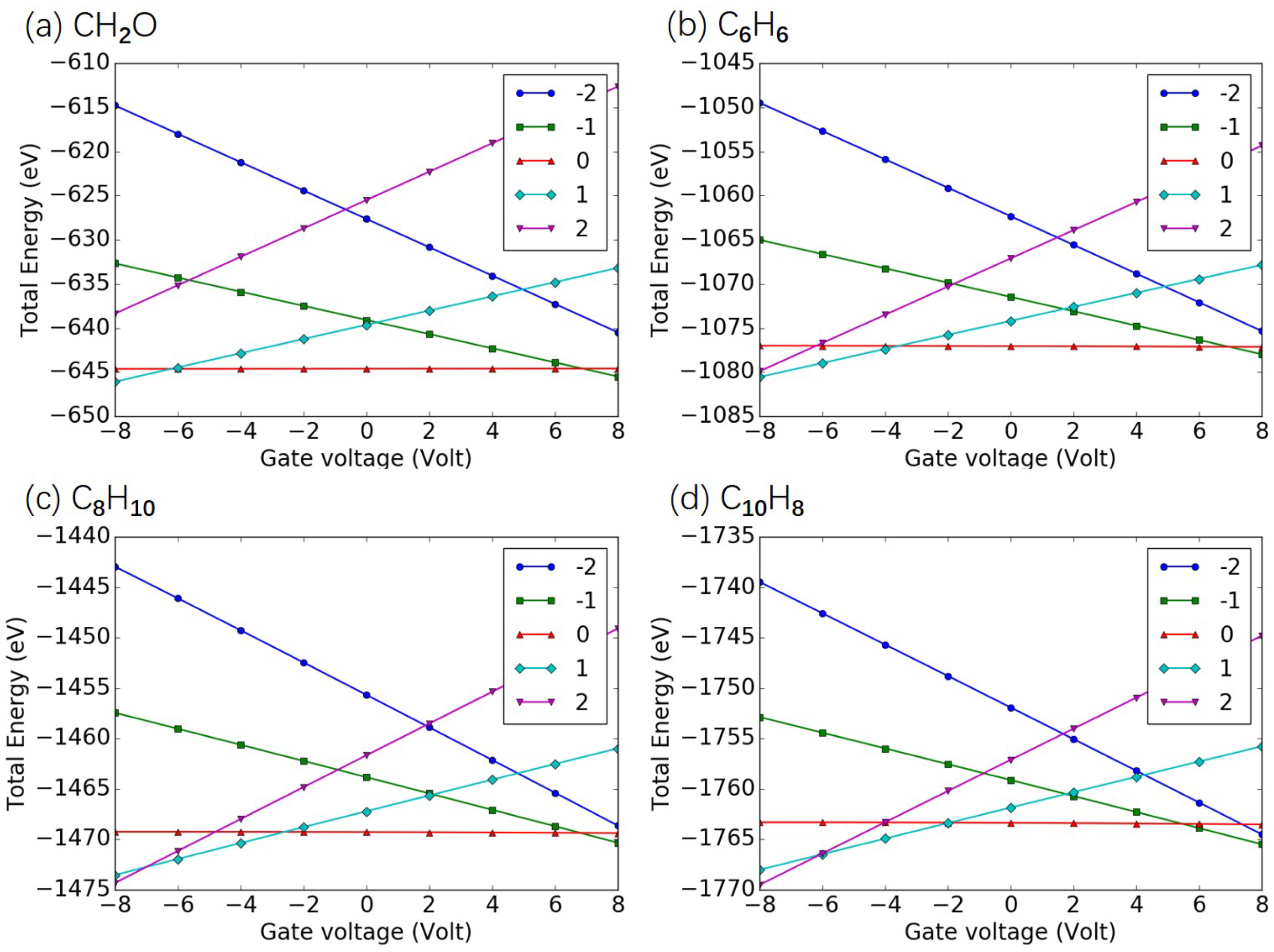
| Island Molecule | CO | NO | O | SO | CHO | CH | CH | CH | CH |
|---|---|---|---|---|---|---|---|---|---|
| (eV) | 23.7019 | 20.3182 | 19.9398 | 17.6599 | 19.4065 | 12.3559 | 11.4820 | 10.8229 | 10.0120 |
| (eV) | 13.2354 | 10.4107 | 10.8392 | 10.1616 | 10.2545 | 8.1345 | 7.6662 | 7.3421 | 6.7887 |
| (eV) | −1.5923 | 2.4329 | 0.8093 | 2.6771 | −0.2429 | −0.2895 | −0.2405 | −0.1665 | 1.0380 |
| (eV) | −7.4722 | −6.8920 | −6.2228 | −3.0606 | −6.1735 | −3.8604 | −3.2489 | −2.8812 | −1.9152 |
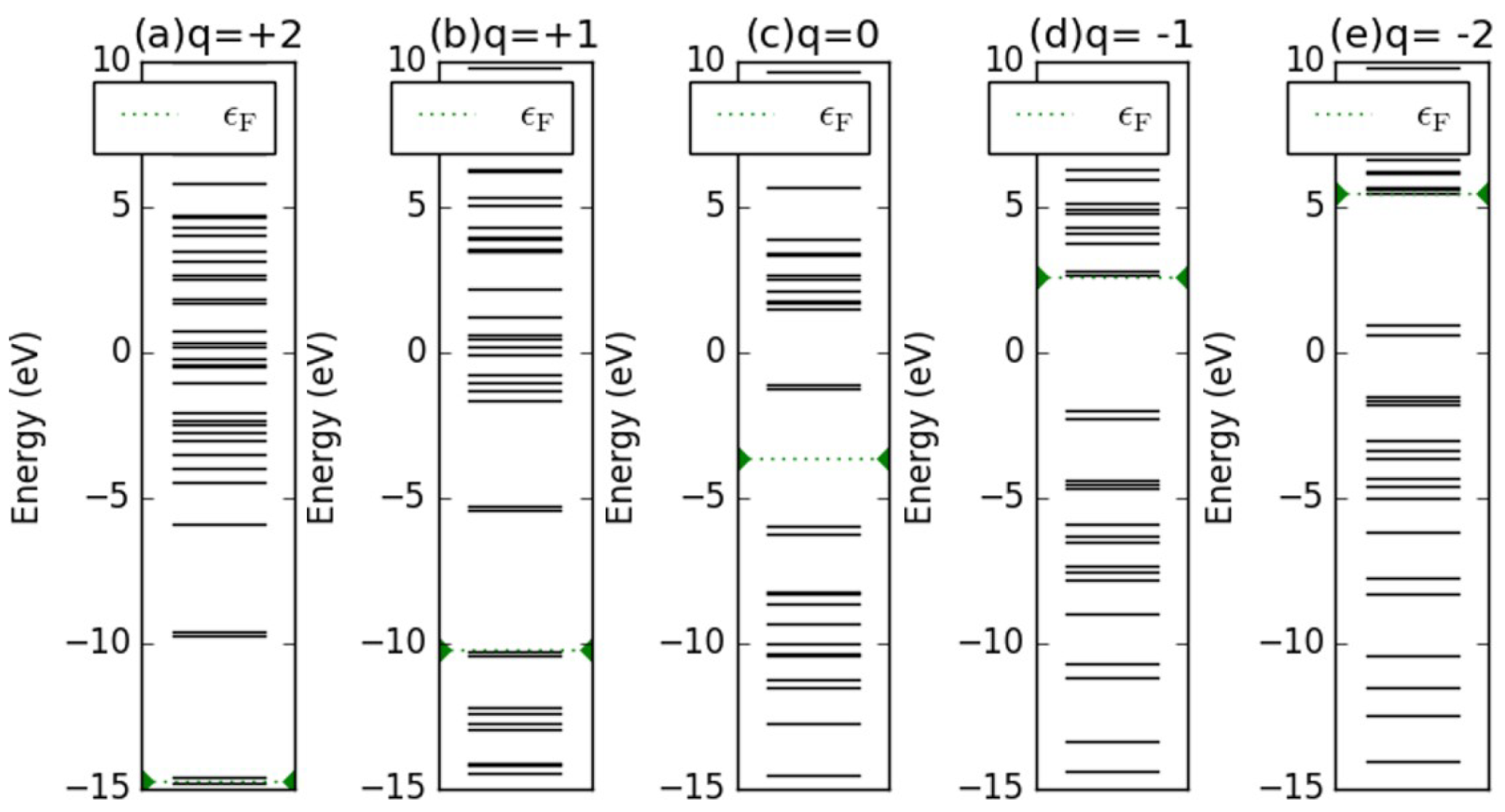
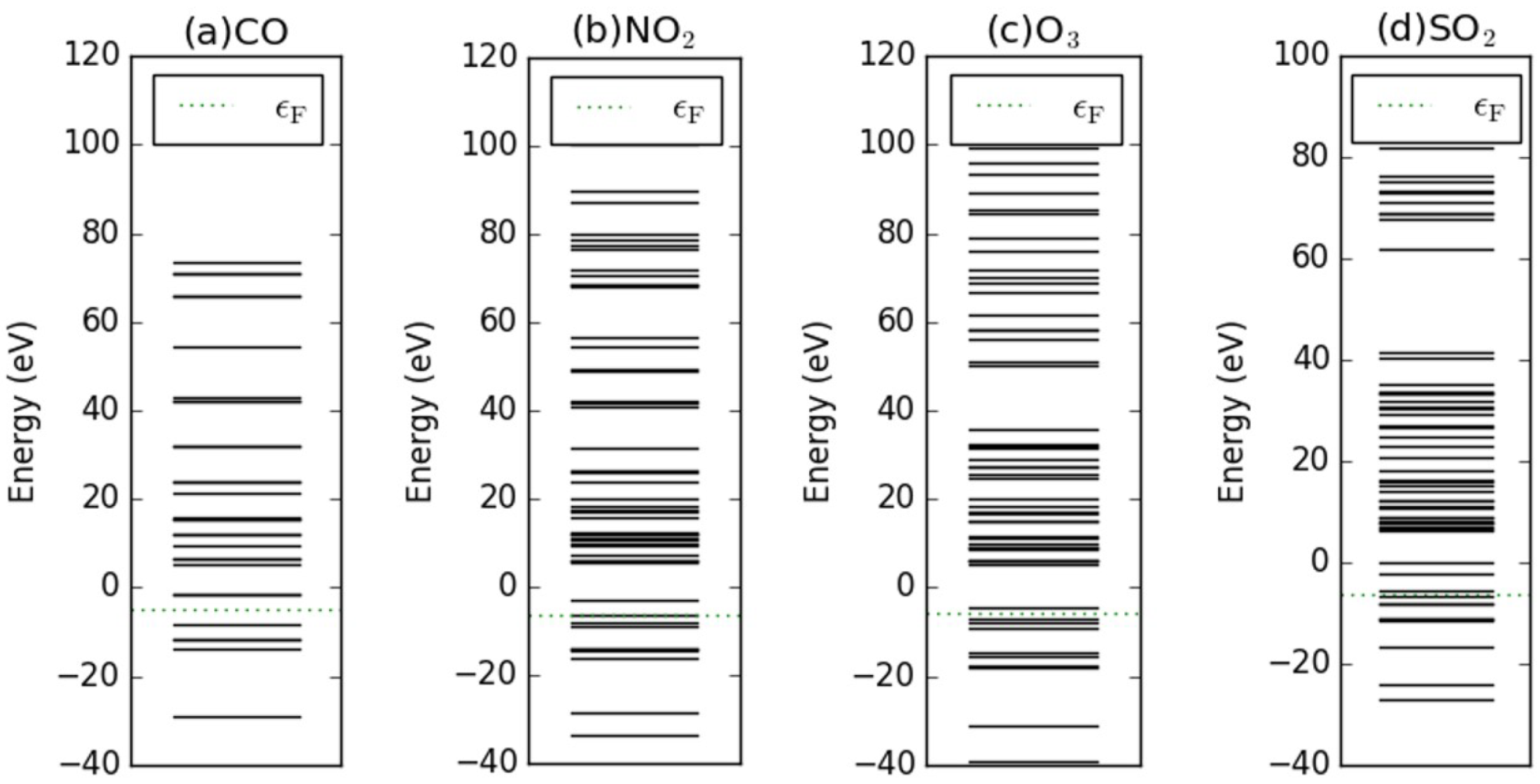
Appendix D. Electronic Structures of the Investigated Molecules
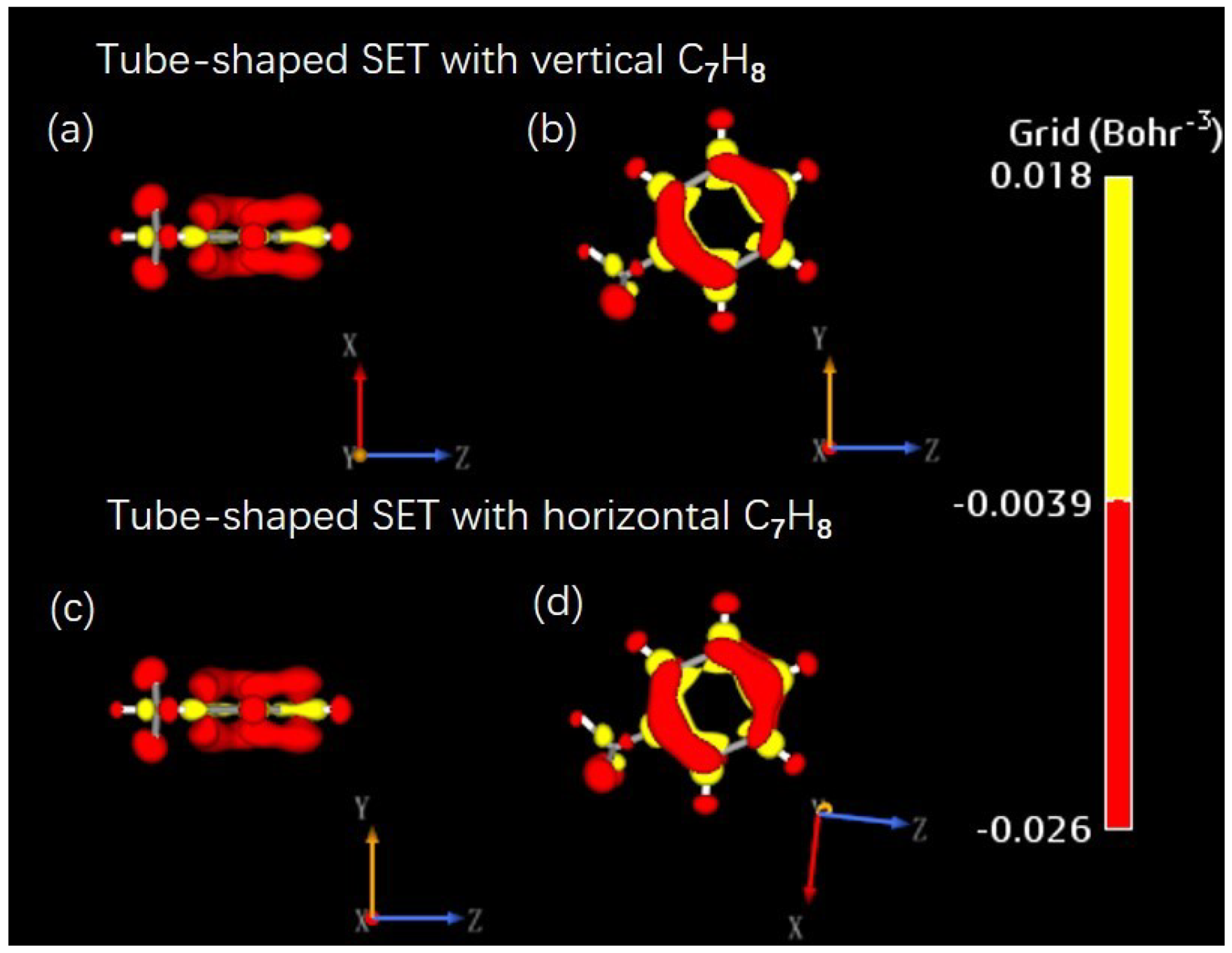
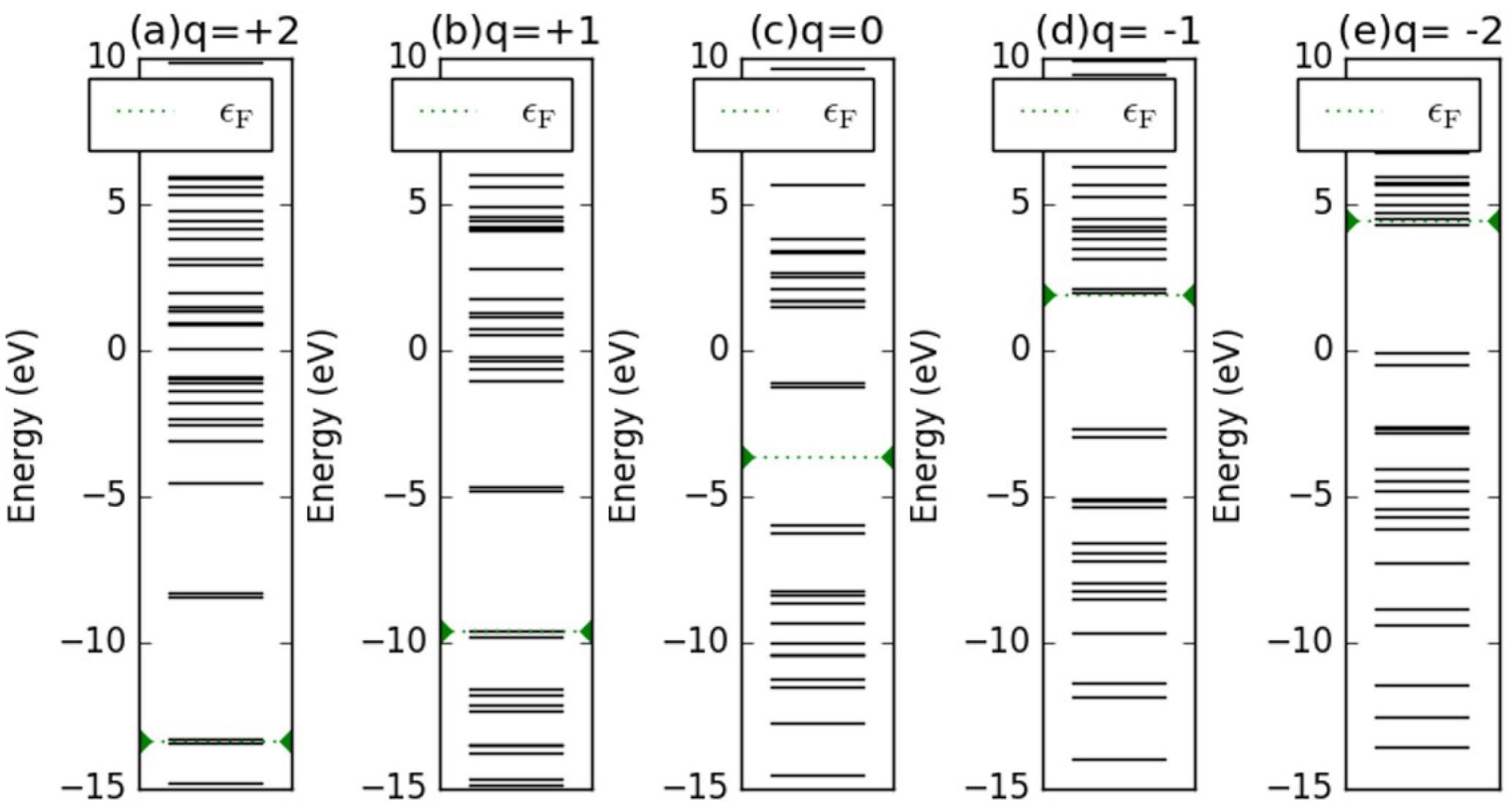
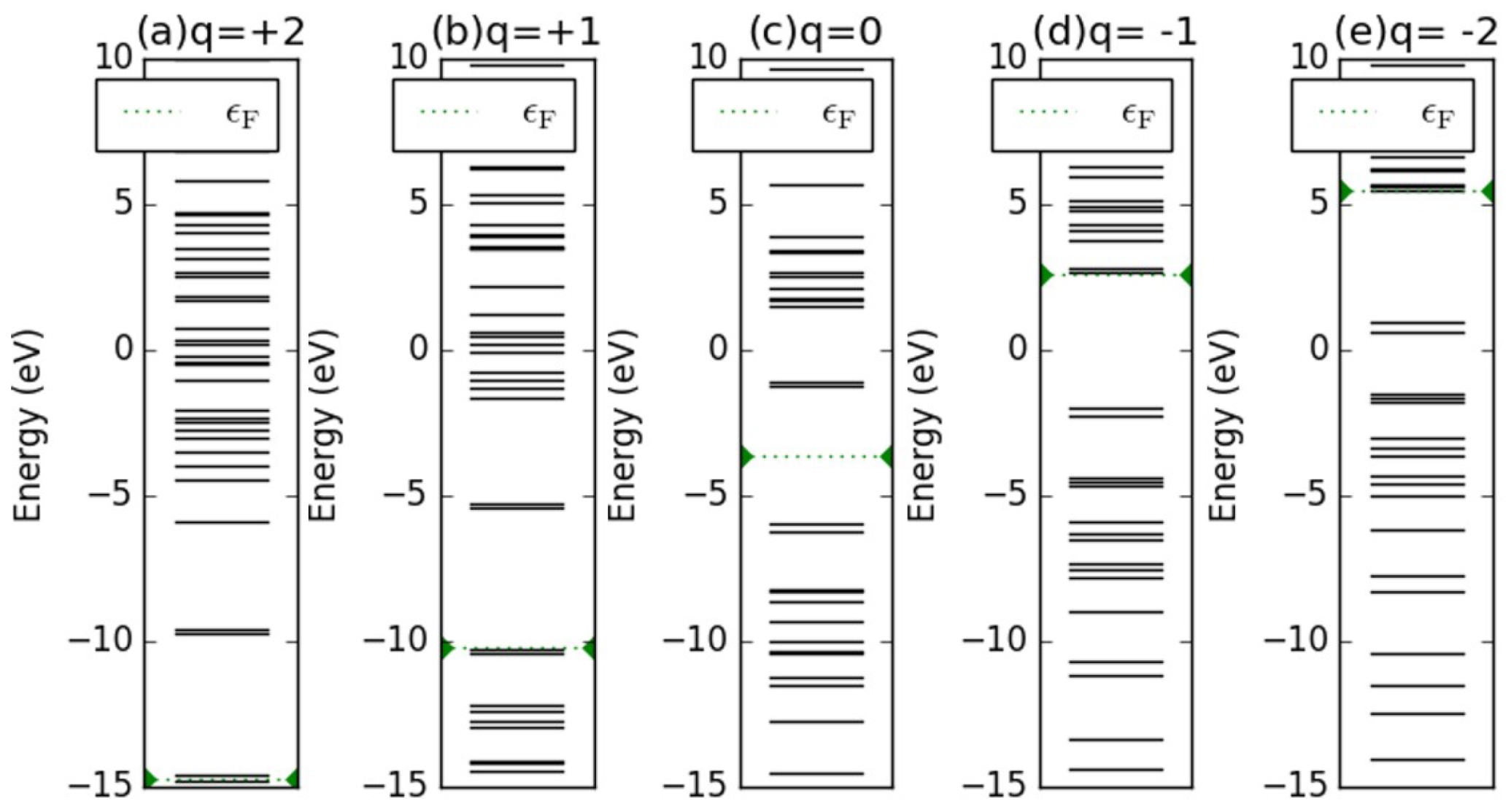

References
- Vallero, D.A. Air Pollution. In Kirkthmer Encyclopedia of Chemical Technology; John Wiley Sons, Inc.: Hoboken, NJ, USA, 2015; pp. 1–48. [Google Scholar] [CrossRef]
- Chau, T.T.; Wang, K.Y. An association between air pollution and daily most frequently visits of eighteen outpatient diseases in an industrial city. Sci. Rep. 2020, 10, 2321. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Guan, D.; Yu, Y.; Westland, S.; Wang, D.; Meng, J.; Wang, X.; He, K.; Tao, S. A psychophysical measurement on subjective well-being and air pollution. Nat. Commun. 2019, 10, 5473. [Google Scholar] [CrossRef] [PubMed]
- Vallero, D. (Ed.) Chapter 14—Air Pollution’s Impact on Ecosystems. In Fundamentals of Air Pollution, 5th ed.; Academic Press: Boston, MA, USA, 2014; pp. 341–368. [Google Scholar] [CrossRef]
- Vallero, D. Chapter 15—Air Pollution’s Impact on Materials and Structures. In Fundamentals of Air Pollution, 5th ed.; Vallero, D., Ed.; Academic Press: Boston, MA, USA, 2014; pp. 369–378. [Google Scholar] [CrossRef]
- Vallero, D. Chapter 7—Air Pollutant Hazards. In Fundamentals of Air Pollution, 5th ed.; Vallero, D., Ed.; Academic Press: Boston, MA, USA, 2014; pp. 197–214. [Google Scholar] [CrossRef]
- Johannson, K.A.; Balmes, J.R.; Collard, H.R. Air Pollution Exposure: A Novel Environmental Risk Factor for Interstitial Lung Disease? CHEST 2015, 147, 1161–1167. [Google Scholar] [CrossRef]
- Ham, B. Indoor chemical pollution impacts often remain invisible. Science 2019, 366, 1084. [Google Scholar] [CrossRef][Green Version]
- Vallero, D. Chapter 25—Methods for Measuring Air Pollutants. In Fundamentals of Air Pollution, 5th ed.; Vallero, D., Ed.; Academic Press: Boston, MA, USA, 2014; pp. 579–626. [Google Scholar]
- Kong, J.; Franklin, N.R.; Zhou, C.; Chapline, M.G.; Peng, S.; Cho, K.; Dai, H. Nanotube Molecular Wires as Chemical Sensors. Science 2000, 287, 622–625. [Google Scholar] [CrossRef]
- Iijima, S. Helical microtubules of graphitic carbon. Nature 1991, 354, 56–58. [Google Scholar] [CrossRef]
- Li, Y.B.; Bando, Y.; Sato, T.; Kurashima, K. ZnO nanobelts grown on Si substrate. Appl. Phys. Lett. 2002, 81, 144–146. [Google Scholar] [CrossRef]
- Morales, A.M.; Lieber, C.M. A Laser Ablation Method for the Synthesis of Crystalline Semiconductor Nanowires. Science 1998, 279, 208–211. [Google Scholar] [CrossRef]
- Li, C.; Zhang, D.; Liu, X.; Han, S.; Tang, T.; Han, J.; Zhou, C. In2O3 nanowires as chemical sensors. Appl. Phys. Lett. 2003, 82, 1613–1615. [Google Scholar] [CrossRef]
- Kolmakov, A.; Zhang, Y.; Cheng, G.; Moskovits, M. Detection of CO and O2 Using Tin Oxide Nanowire Sensors. Adv. Mater. 2003, 15, 997–1000. [Google Scholar] [CrossRef]
- Schedin, F.; Geim, A.K.; Morozov, S.V.; Hill, E.W.; Blake, P.; Katsnelson, M.I.; Novoselov, K.S. Detection of individual gas molecules adsorbed on graphene. Nat. Mater. 2007, 6, 652–655. [Google Scholar] [CrossRef]
- Li, H.; Yin, Z.; He, Q.; Li, H.; Huang, X.; Lu, G.; Fam, D.W.H.; Tok, A.I.Y.; Zhang, Q.; Zhang, H. Fabrication of Single- and Multilayer MoS2 Film-Based Field-Effect Transistors for Sensing NO at Room Temperature. Small 2012, 8, 63–67. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.C.; Zheng, J.; Liu, J.; Gong, X.T.; Chen, Z.Z.; Guo, R.X.; Huang, X.; Zhang, Y.P.; Zhang, L.; Li, R.; et al. Nonadditive Transport in Multi-Channel Single-Molecule Circuits. Small 2020, 16, 2002808. [Google Scholar] [CrossRef] [PubMed]
- Kastner, M.A. The single-electron transistor. Rev. Mod. Phys. 1992, 64, 849–858. [Google Scholar] [CrossRef]
- Bitton, O.; Gutman, D.B.; Berkovits, R.; Frydman, A. Multiple periodicity in a nanoparticle-based single-electron transistor. Nat. Commun. 2017, 8, 402. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, K.; Ishii, M.; Segawa, K.; Oka, Y.; Vartanian, B.J.; Harris, J.S. Room temperature operation of a single electron transistor made by the scanning tunneling microscope nanooxidation process for the TiOx/Ti system. Appl. Phys. Lett. 1996, 68, 34–36. [Google Scholar] [CrossRef]
- Shin, S.J.; Lee, J.J.; Kang, H.J.; Choi, J.B.; Yang, S.R.E.; Takahashi, Y.; Hasko, D.G. Room-Temperature Charge Stability Modulated by Quantum Effects in a Nanoscale Silicon Island. Nano Lett. 2011, 11, 1591–1597. [Google Scholar] [CrossRef] [PubMed]
- Maeda, K.; Okabayashi, N.; Kano, S.; Takeshita, S.; Tanaka, D.; Sakamoto, M.; Teranishi, T.; Majima, Y. Logic Operations of Chemically Assembled Single-Electron Transistor. ACS Nano 2012, 6, 2798–2803. [Google Scholar] [CrossRef]
- Tsukanov, A.V. Measurement of a charge qubit using a single-electron transistor based on a triple quantum dot. Phys. Rev. A 2019, 100, 062305. [Google Scholar] [CrossRef]
- Keith, D.; House, M.; Donnelly, M.; Watson, T.; Weber, B.; Simmons, M. Single-Shot Spin Readout in Semiconductors Near the Shot-Noise Sensitivity Limit. Phys. Rev. X 2019, 9, 041003. [Google Scholar] [CrossRef]
- Rani, S.; Ray, S.J. Detection of gas molecule using C3N island single electron transistor. Carbon 2019, 144, 235–240. [Google Scholar] [CrossRef]
- Sharma, A.; Husain, M.; Srivastava, A.; Khan, M.S. Anti-site defected MoS2 sheet-based single electron transistor as a gas sensor. AIP Conf. Proc. 2018, 1953, 140075. [Google Scholar] [CrossRef]
- Sharma, A.; Anu; Khan, M.S.; Husain, M.; Khan, M.S.; Srivastava, A. Sensing of CO and NO on Cu-Doped MoS2 Monolayer-Based Single Electron Transistor: A First Principles Study. IEEE Sens. J. 2018, 18, 2853–2860. [Google Scholar] [CrossRef]
- Ray, S.J. Single molecule transistor based nanopore for the detection of nicotine. J. Appl. Phys. 2014, 116, 244307. [Google Scholar] [CrossRef]
- Ray, S.J. Humidity sensor using a single molecular transistor. J. Appl. Phys. 2015, 118, 044307. [Google Scholar] [CrossRef]
- Ray, S.J. Single molecular transistor as a superior gas sensor. J. Appl. Phys. 2015, 118, 034303. [Google Scholar] [CrossRef]
- Ray, S.J. Gate engineered performance of single molecular transistor. J. Appl. Phys. 2016, 119, 204302. [Google Scholar] [CrossRef]
- Ray, S.J.; Chowdhury, R. Double gated single molecular transistor for charge detection. J. Appl. Phys. 2014, 116, 034307. [Google Scholar] [CrossRef]
- Anu; Srivastava, A.; Khan, M.S. Charge stability diagram and addition energy spectrum for single-electron transistor based on Ni-dithiolene derivatives. Org. Electron. 2018, 59, 125–130. [Google Scholar] [CrossRef]
- Stokbro, K. First-Principles Modeling of Molecular Single-Electron Transistors. J. Phys. Chem. C 2010, 114, 20461–20465. [Google Scholar] [CrossRef]
- Garaj, S.; Hubbard, W.; Reina, A.; Kong, J.; Branton, D.; Golovchenko, J.A. Graphene as a subnanometre trans-electrode membrane. Nature 2010, 467, 190–193. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Stein, D.; McMullan, C.; Branton, D.; Aziz, M.J.; Golovchenko, J.A. Ion-beam sculpting at nanometre length scales. Nature 2001, 412, 166–169. [Google Scholar] [CrossRef]
- Smidstrup, S.; Markussen, T.; Vancraeyveld, P.; Wellendorff, J.; Schneider, J.; Gunst, T.; Verstichel, B.; Stradi, D.; Khomyakov, P.A.; Vej-Hansen, U.G.; et al. QuantumATK: An integrated platform of electronic and atomic-scale modelling tools. J. Phys. Condens. Matter 2019, 32, 015901. [Google Scholar] [CrossRef] [PubMed]
- Brandbyge, M.; Kobayashi, N.; Tsukada, M. Conduction channels at finite bias in single-atom gold contacts. Phys. Rev. B 1999, 60, 17064–17070. [Google Scholar] [CrossRef]
- Taylor, J.; Guo, H.; Wang, J. Ab initio modeling of open systems: Charge transfer, electron conduction, and molecular switching of a C60 device. Phys. Rev. B 2001, 63, 121104. [Google Scholar] [CrossRef]
- Brandbyge, M.; Mozos, J.L.; Ordejón, P.; Taylor, J.; Stokbro, K. Density-functional method for nonequilibrium electron transport. Phys. Rev. B 2002, 65, 165401. [Google Scholar] [CrossRef]
- Kaasbjerg, K.; Flensberg, K. Strong Polarization-Induced Reduction of Addition Energies in Single-Molecule Nanojunctions. Nano Lett. 2008, 8, 3809–3814. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 1996, 77, 3865–3868. [Google Scholar] [CrossRef]
- Ray, S.J. Single atom impurity in a single molecular transistor. J. Appl. Phys. 2014, 116, 154302. [Google Scholar] [CrossRef]
- Guo, Y.D.; Yan, X.H.; Xiao, Y. Computational Investigation of DNA Detection Using Single-Electron Transistor-Based Nanopore. J. Phys. Chem. C 2012, 116, 21609–21614. [Google Scholar] [CrossRef]
- Zhang, Y.; Heath, G.; Carpenter, A.; Fisher, N. Air pollutant emissions inventory of large-scale production of selected biofuels feedstocks in 2022. Biofuels Bioprod. Biorefining 2016, 10, 56–69. [Google Scholar] [CrossRef]
- Aaroe, W.H.; Light, E. Indoor air pollution. Science 1984, 223, 6. [Google Scholar] [CrossRef]
- Zharinov, V.S.; Picot, T.; Scheerder, J.E.; Janssens, E.; Van de Vondel, J. Room temperature single electron transistor based on a size-selected aluminium cluster. Nanoscale 2020, 12, 1164–1170. [Google Scholar] [CrossRef] [PubMed]
| SET Configuration | Gate-Island Coupling Strength | |||
|---|---|---|---|---|
| SET Structure | ISLAND Molecule | Molecule Posture | Linear α | Quadratic β (eV−1) |
| Conventional | CH | Vertical | 0.3417 | −0.0056 |
| Conventional | CH | Horizontal | 0.3004 | −0.0023 |
| Tube−shaped | CH | Vertical | 0.7962 | −0.0006 |
| Tube−shaped | CH | Horizontal | 0.7962 | −0.0006 |
| Island Molecule | Gate–Island Coupling Strength | |
|---|---|---|
| Linear α | Quadratic β (eV−1) | |
| CO | 0.8020 | 0.0000 |
| NO | 0.8029 | 0.0000 |
| O | 0.8138 | 0.0000 |
| SO | 0.8005 | −0.0001 |
| CHO | 0.8035 | 0.0000 |
| CH | 0.8031 | −0.0004 |
| CH | 0.7954 | −0.0009 |
| CH | 0.7768 | −0.0009 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, Z.; Peng, X.; Peng, C.; Huang, J.; Bo, M.; Yao, C.; Li, J. Detecting Air Pollutant Molecules Using Tube-Shaped Single Electron Transistor. Molecules 2021, 26, 7098. https://doi.org/10.3390/molecules26237098
Huang Z, Peng X, Peng C, Huang J, Bo M, Yao C, Li J. Detecting Air Pollutant Molecules Using Tube-Shaped Single Electron Transistor. Molecules. 2021; 26(23):7098. https://doi.org/10.3390/molecules26237098
Chicago/Turabian StyleHuang, Zhongkai, Xiangyang Peng, Cheng Peng, Jin Huang, Maolin Bo, Chuang Yao, and Jibiao Li. 2021. "Detecting Air Pollutant Molecules Using Tube-Shaped Single Electron Transistor" Molecules 26, no. 23: 7098. https://doi.org/10.3390/molecules26237098
APA StyleHuang, Z., Peng, X., Peng, C., Huang, J., Bo, M., Yao, C., & Li, J. (2021). Detecting Air Pollutant Molecules Using Tube-Shaped Single Electron Transistor. Molecules, 26(23), 7098. https://doi.org/10.3390/molecules26237098







