Multicomponent Synthesis of 2-(2,4-Diamino-3-cyano-5H-chromeno[2,3-b]pyridin-5-yl)malonic Acids in DMSO
Abstract
:1. Introduction
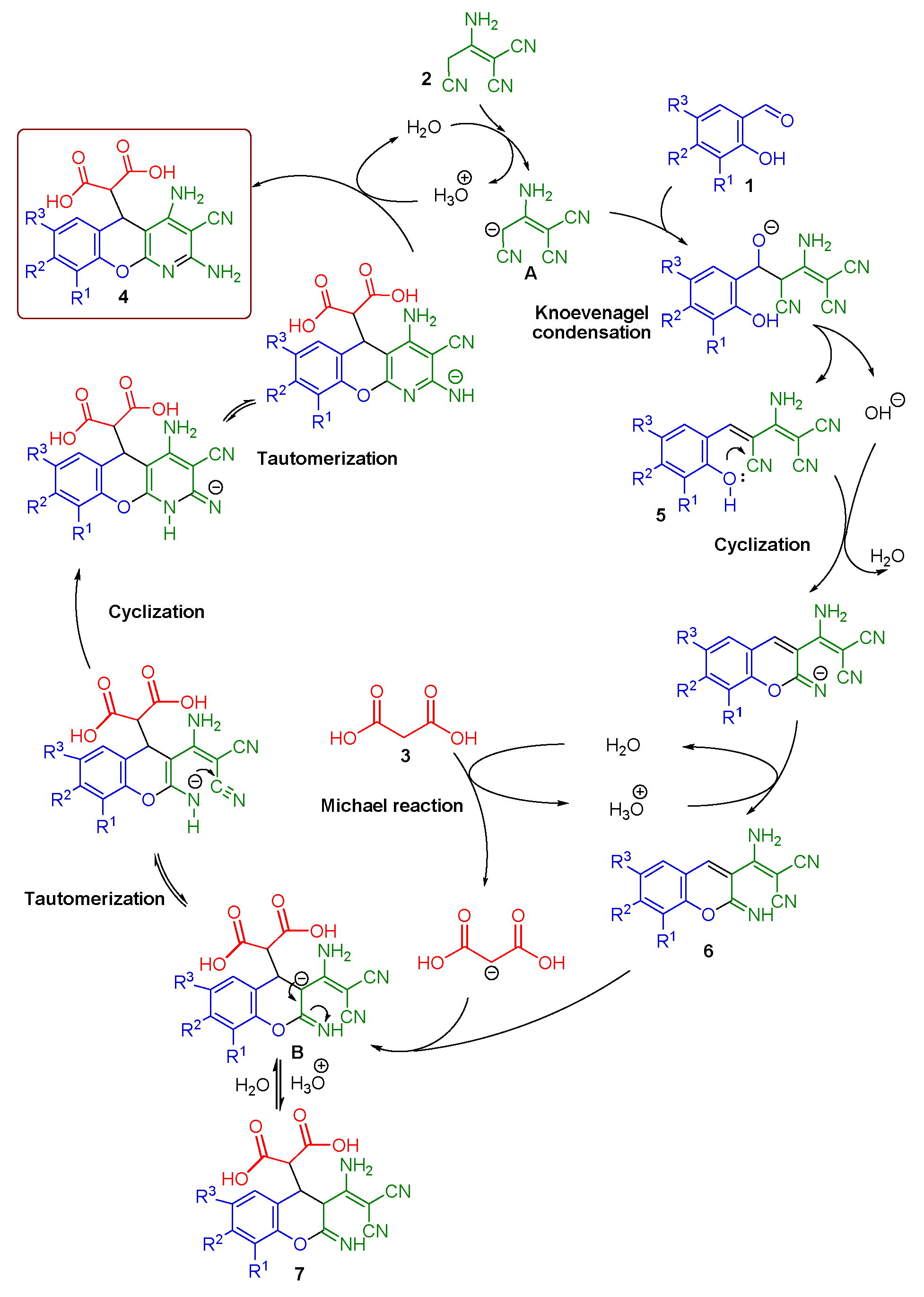
2. Results and Discussion
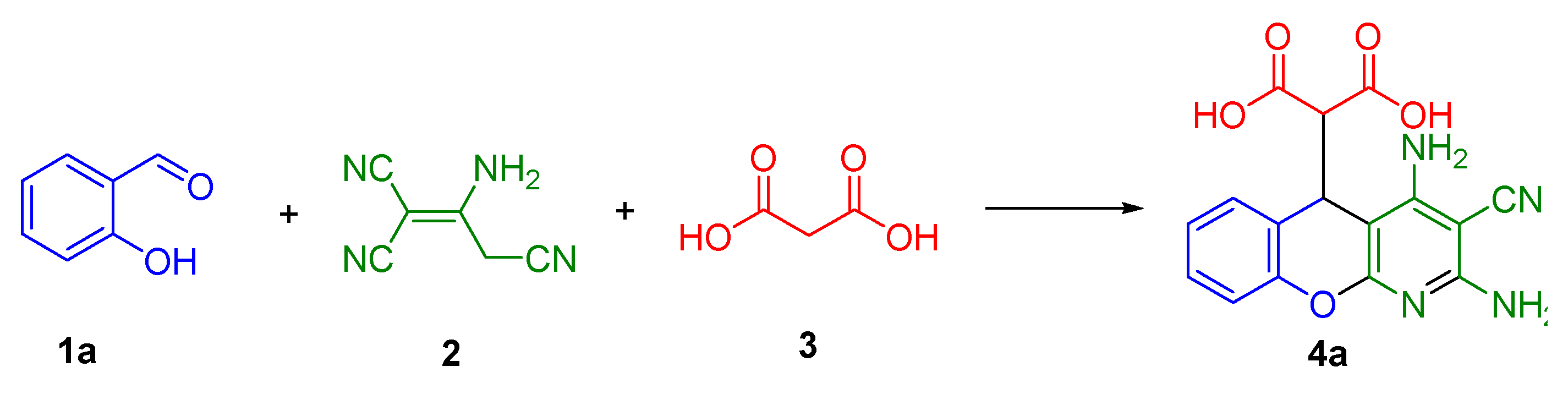
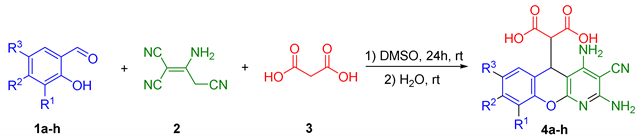
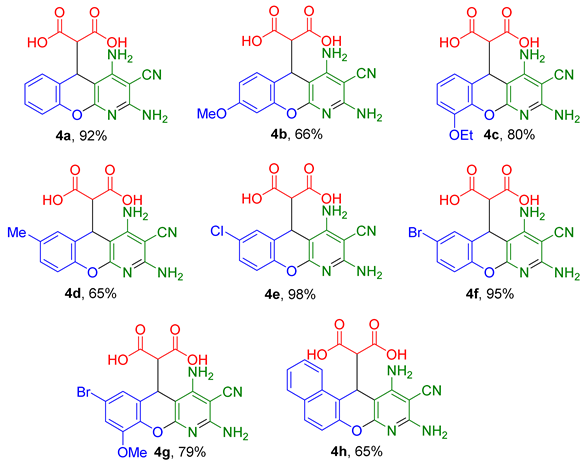
2.1. Multicomponent Synthesis of 2-(2,4-Diamino-3-cyano-5H-chromeno[2,3-b]pyridin-5-yl)malonic Acids 4a–h
2.2. 2D-NMR Study of the Structure of Compound 4f
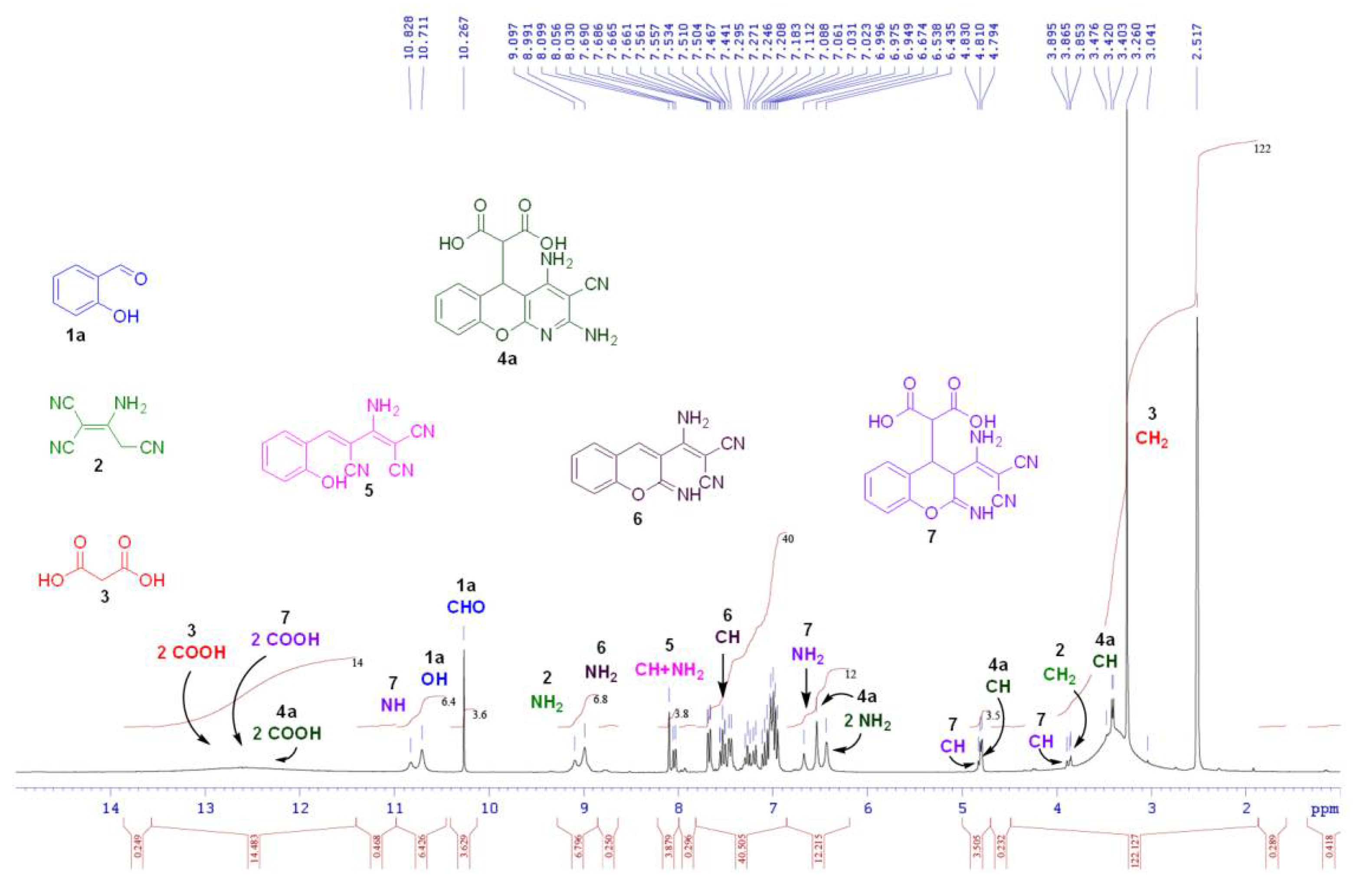
2.3. 1H-NMR Reaction Monitoring
3. Materials and Methods
3.1. General Information
3.2. Synthesis of 2-(2,4-Diamino-3-cyano-5H-chromeno[2,3-b]pyridin-5-yl)malonic Acids 4a–h
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- John, S.E.; Gulatia, S.; Shankaraiah, N. Recent advances in multi-component reactions and their mechanistic insights: A triennium review. Org. Chem. Front. 2021, 8, 4237–4287. [Google Scholar] [CrossRef]
- Younus, H.A.; Al-Rashida, M.; Hameed, A.; Uroos, M.; Salar, U.; Rana, S.; Khan, K.M. Multicomponent reactions (MCR) in medicinal chemistry: A patent review (2010–2020). Expert Opin. Ther. Pat. 2021, 31, 267–289. [Google Scholar] [CrossRef] [PubMed]
- Domling, A.; Wang, W.; Wang, K. Chemistry and biology of multicomponent reactions. Chem. Rev. 2012, 112, 3083–3135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghoneim, A.A.; El-Farargy, A.F.; Abdelaziz, S. Synthesis and Antimicrobial Activities of New S-Nucleosides of Chromeno[2,3-b]Pyridine Derivatives and C-Nucleosides of [1,2,4]Triazolo[1,5-a]Quinoline Derivatives. Nucleosides Nucleotides Nucleic Acids. 2014, 33, 583–596. [Google Scholar] [CrossRef] [PubMed]
- Oliveira-Pinto, S.; Pontes, O.; Lopes, D.; Sampaio-Marques, B.; Costa, M.D.; Carvalho, L.; Gonçalves, C.S.; Costa, B.M.; Maciel, P.; Ludovico, P.; et al. Unravelling the anticancer potential of functionalized chromeno[2,3-b]pyridines for breast cancer treatment. Bioorg. Chem. 2020, 100, 103942. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, Y.; Goto, K.; Terasawa, M. Method for Treatment of Rheumatism. Ger. Offen. DE 3010751 19810806, 6 August 1981. [Google Scholar]
- Ukawa, K.; Ishiguro, T.; Kuriki, H.; Nohara, A. Synthesis of the metabolites and degradation products of 2-amino-7-isopropyl-5-oxo-5H-(1)benzopyrano(2,3-b)pyridine-3-carboxylic acid (Amoxanox). Chem. Pharm. Bull. 1985, 33, 4432–4437. [Google Scholar] [CrossRef] [Green Version]
- Oset-Gasque, M.J.; González, M.P.; Pérez-Peña, J.; García-Font, N. Toxicological and pharmacological evaluation, antioxidant, ADMET and molecular modeling of selected racemic chromenotacrines {11-amino-12-aryl-8,9,10,12-tetrahydro-7H-chromeno[2,3-b]quinolin-3-ols} for the potential prevention and treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2014, 74, 491–501. [Google Scholar] [CrossRef] [Green Version]
- Goto, K.; Yaoka, O.; Oe, T. Hypotenseurs. PCT Int. Appl. WO 1984001711A1 19840510, 30 October 1982. [Google Scholar]
- Wu, X.-F.W.; Xiang, J.-C.; Gao, Q.-H.; Wu, A.-X. The Applications of DMSO. In Solvents as Reagents in Organic Synthesis: Reactions and Applications; Wu, X.-F.W., Ed.; Wiley-VCH Verlag GmbH & Co.: Weinheim, Germany, 2017; Chapter 7; pp. 315–353. [Google Scholar] [CrossRef]
- Jana, A.; Bhaumick, P.; Panday, A.K.; Mishra, R.; Choudhury, L.H. I2/DMSO mediated multicomponent reaction for the synthesis of 2-arylbenzo[d]imidazo[2,1-b]thiazole derivatives. Org. Biomol. Chem. 2019, 17, 5316–5330. [Google Scholar] [CrossRef] [PubMed]
- Bremner, W.S.; Organ, M.G. Multicomponent reactions to form heterocycles by microwaveassisted continuous flow organic synthesis. J. Comb. Chem. 2007, 9, 14–16. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Liu, S.; Wu, X.; Zhang, J.; Wu, A. Coproduct promoted Povarov reaction: Synthesis of Substituted quinolines from methyl ketones, arylamines, and α-ketoesters. J. Org. Chem. 2015, 80, 5984–5991. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Geng, X.; Zhao, P.; Zhang, J.; Gong, X.; Wu, Y.; Wu, A. I2-promoted Povarov-type reaction using 1,4-dithane-2,5-diol as an ethylene surrogate: Formal [4+2] synthesis of quinolines. Org. Lett. 2017, 19, 1550–1553. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Liu, S.; Wu, X.; Wu, A. Povarov-type reaction using methyl as new input: Direct synthesis of substituted quinolines by I2-mediated formal [3+2+1] cycloaddition. Org. Lett. 2014, 16, 4582–4585. [Google Scholar] [CrossRef]
- Elinson, M.N.; Ryzhkova, Y.E.; Ryzhkov, F.V. Multicomponent design of chromeno[2,3-b]pyridine systems. Russ. Chem. Rev. 2021, 90, 94–115. [Google Scholar] [CrossRef]
- Vereshchagin, A.N.; Elinson, M.N.; Anisina, Y.E.; Ryzhkov, F.V.; Goloveshkin, A.S.; Bushmarinov, I.S.; Zlotin, S.G.; Egorov, M.P. Pot, atom and step economic (PASE) synthesis of 5-isoxazolyl-5H-chromeno[2,3-b]pyridine scaffold. Mendeleev Commun. 2015, 25, 424–426. [Google Scholar] [CrossRef]
- Vereshchagin, A.N.; Elinson, M.N.; Anisina, Y.E.; Ryzhkov, F.V.; Goloveshkin, A.S.; Novikov, R.A.; Egorov, M.P. Synthesis, structural, spectroscopic and docking studies of new 5C-substituted 2,4-diamino-5H-chromeno[2,3-b]pyridine-3-carbonitriles. J. Mol. Struct. 2017, 1146, 766–772. [Google Scholar] [CrossRef]
- Elinson, M.N.; Vereshchagin, A.N.; Anisina, Y.E.; Fakhrutdinov, A.N.; Goloveshkin, A.S.; Egorov, M.P. Pot-, Atom- and Step-Economic (PASE) Multicomponent approach to the 5-(Dialkylphosphonate)-Substituted 2,4-Diamino-5H-chromeno[2,3-b]pyridine scaffold. Eur. J. Org. Chem. 2019, 2019, 4171–4178. [Google Scholar] [CrossRef]
- Elinson, M.N.; Vereshchagin, A.N.; Anisina, Y.E.; Egorov, M.P. Efficient Multicomponent Approach to the Medicinally Relevant 5-aryl-chromeno[2,3-b]pyridine Scaffold. Polycycl. Aromat. Compd. 2020, 40, 108–115. [Google Scholar] [CrossRef]
- Ryzhkov, F.V.; Ryzhkova, Y.E.; Elinson, M.N.; Vorobyev, S.V.; Fakhrutdinov, A.N.; Vereshchagin, A.N.; Egorov, M.P. Catalyst-Solvent System for PASE Approach to Hydroxyquinolinone-Substituted Chromeno[2,3-b]pyridines Its Quantum Chemical Study and Investigation of Reaction Mechanism. Molecules 2020, 25, 2573. [Google Scholar] [CrossRef] [PubMed]
- Elinson, M.N.; Vereshchagin, A.N.; Anisina, Y.E.; Krymov, S.K.; Fakhrutdinov, A.N.; Egorov, M.P. Potassium fluoride catalysed multicomponent approach to medicinally privileged 5-[3-hydroxy-6-(hydroxymethyl)-4-oxo-4H-pyran-2-yl] substituted chromeno[2,3-b]pyridine scaffold. Arkivoc 2019, 2, 38–49. [Google Scholar] [CrossRef]
- Patai, S.; Israeli, Y. 411. The kinetics and mechanisms of carbonyl–methylene condensations. Part VII. The reaction of malononitrile with aromatic aldehydes in ethanol. J. Chem. Soc. 1960, 2025–2030. [Google Scholar] [CrossRef]
- Mittelbach, M. An improved and facile synthesis of 2-amino-1,1,3-tricyanopropene. Monatsh. Chem. 1985, 116, 689–691. [Google Scholar] [CrossRef]

| Entry | Solvent | Time (h) | Temperature (°C) | Water Volume (mL) | Yield (%) |
|---|---|---|---|---|---|
| 1 | DMSO | 24 | 23 (rt) | 15 | 92 2 |
| 2 | DMF | 24 | 23 (rt) | 15 | 75 2 |
| 3 | NMP | 24 | 23 (rt) | 15 | 70 2 |
| 4 | DMSO | 12 | 23 (rt) | 15 | 62 2 |
| 5 | DMSO | 24 | 23 (rt) | 10 | 81 2 |
| 6 | DMSO | 24 | 40 | 15 | 66 |
| 7 | Py | 4 | 115 | - | - |
| 8 | EtOH/Py (3:1) | 4 | 81 | - | 15 |
 |
 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ryzhkova, Y.E.; Elinson, M.N.; Maslov, O.I.; Fakhrutdinov, A.N. Multicomponent Synthesis of 2-(2,4-Diamino-3-cyano-5H-chromeno[2,3-b]pyridin-5-yl)malonic Acids in DMSO. Molecules 2021, 26, 6839. https://doi.org/10.3390/molecules26226839
Ryzhkova YE, Elinson MN, Maslov OI, Fakhrutdinov AN. Multicomponent Synthesis of 2-(2,4-Diamino-3-cyano-5H-chromeno[2,3-b]pyridin-5-yl)malonic Acids in DMSO. Molecules. 2021; 26(22):6839. https://doi.org/10.3390/molecules26226839
Chicago/Turabian StyleRyzhkova, Yuliya E., Michail N. Elinson, Oleg I. Maslov, and Artem N. Fakhrutdinov. 2021. "Multicomponent Synthesis of 2-(2,4-Diamino-3-cyano-5H-chromeno[2,3-b]pyridin-5-yl)malonic Acids in DMSO" Molecules 26, no. 22: 6839. https://doi.org/10.3390/molecules26226839
APA StyleRyzhkova, Y. E., Elinson, M. N., Maslov, O. I., & Fakhrutdinov, A. N. (2021). Multicomponent Synthesis of 2-(2,4-Diamino-3-cyano-5H-chromeno[2,3-b]pyridin-5-yl)malonic Acids in DMSO. Molecules, 26(22), 6839. https://doi.org/10.3390/molecules26226839









