Opposite Effects Induced by Cholinium-Based Ionic Liquid Electrolytes in the Formation of Aqueous Biphasic Systems Comprising Polyethylene Glycol and Sodium Polyacrylate
Abstract
1. Introduction
2. Results and Discussion
2.1. Effect of PEG Molecular Weight (MW)
2.2. Effect of [Ch]−Based ILs Concentration in ABS Formation
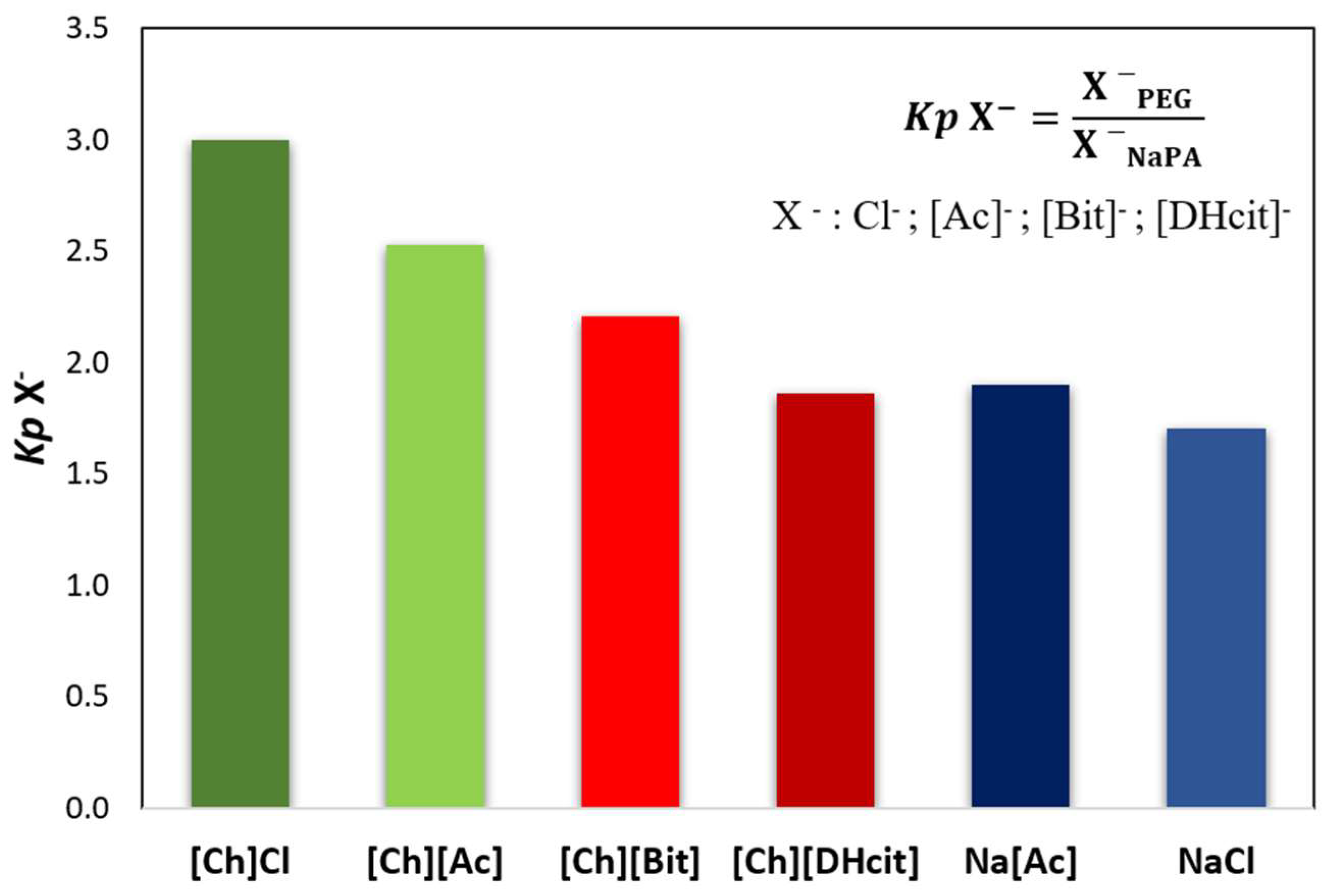
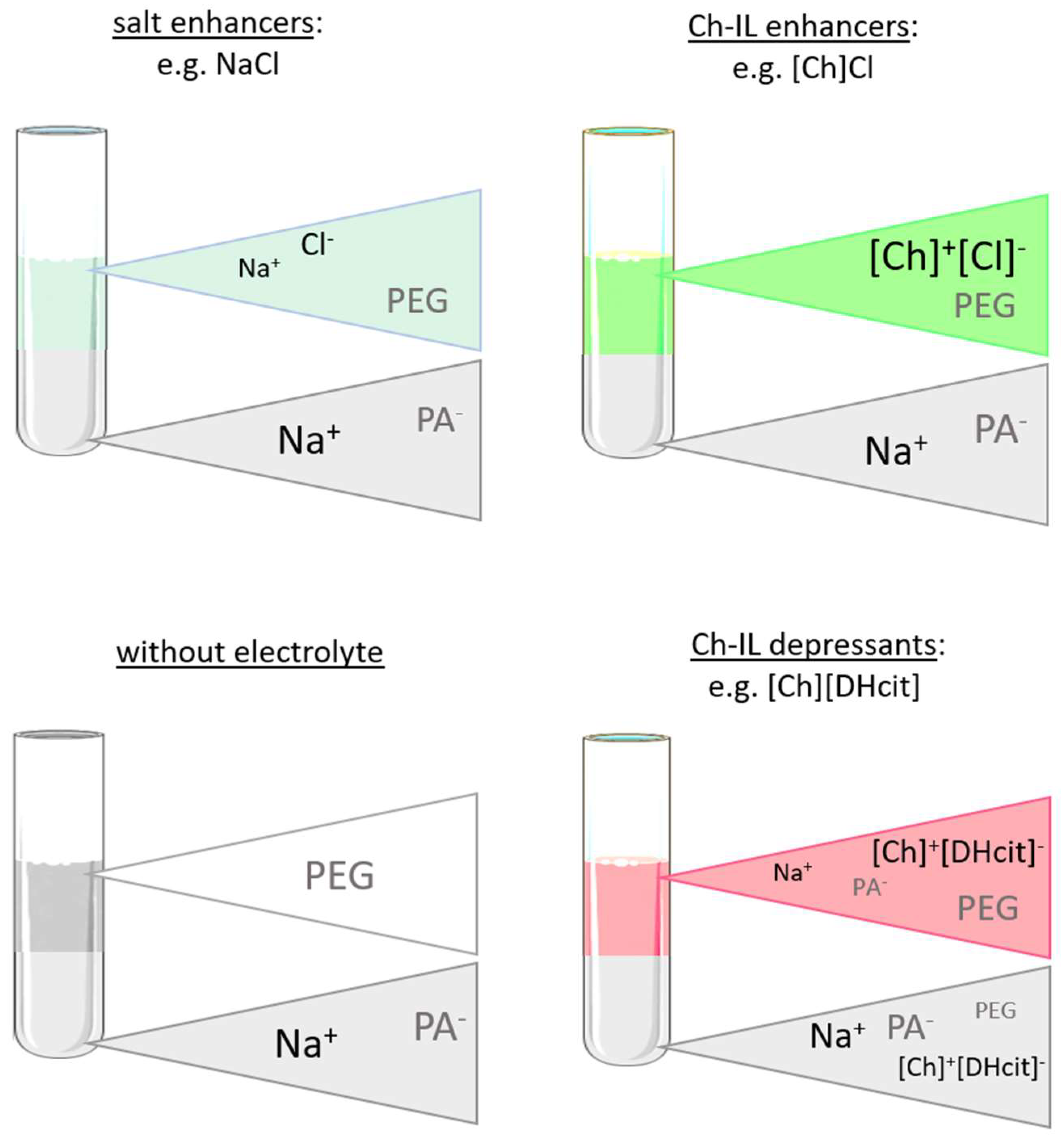
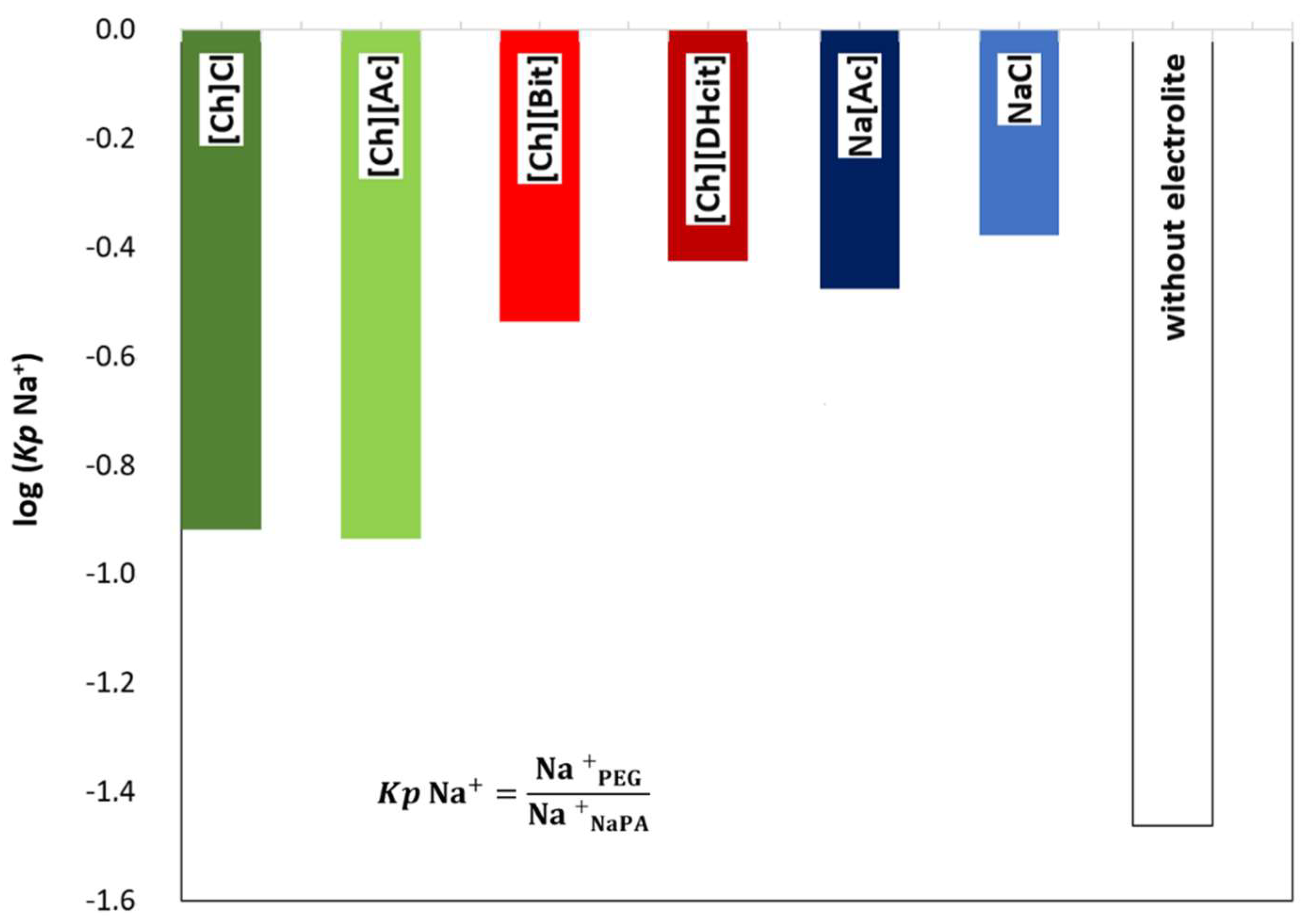
2.3. Opposite Effects Explained: Determination of Electrolyte Partition
3. Materials and Methods
3.1. Materials
3.2. Determination of Phase Diagrams
3.3. Quantification of the Cholinium Cation in the ABS Coexisting Phases
3.4. Quantification of Sodium and Chloride Ions in the ABS Coexisting Phases
3.5. Determination of Partition Coefficients of Electrolytes (Cations and Anions) Present in ABS
3.6. Vibrational Studies of Electrolytes Measured by ATR-FTIR in the ABS Coexisting Phases
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Souza, R.L.; Ventura, S.P.M.; Soares, C.M.F.; Coutinho, J.A.P.; Lima, Á.S. Lipase purification using ionic liquids as adjuvants in aqueous two-phase systems. Green Chem. 2015, 17, 3026–3034. [Google Scholar] [CrossRef]
- Asenjo, J.A.; Andrews, B.A. Aqueous two-phase systems for protein separation: Phase separation and applications. J. Chromatogr. A 2012, 1238, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.F.B.; Freire, M.G.; Coutinho, J.A.P. Aqueous two-phase systems: Towards novel and more disruptive applications. Fluid Phase Equilib. 2020, 505, 112341. [Google Scholar] [CrossRef]
- Gustafsson, Å.; Wennerström, H.; Tjerneld, F. The nature of phase separation in aqueous two-polymer systems. Polymers 1986, 27, 1768–1770. [Google Scholar] [CrossRef]
- Freire, M.G.; Cláudio, A.F.M.; Araújo, J.M.M.; Coutinho, J.A.P.; Marrucho, I.M.; Lopes, J.N.C.; Rebelo, L.P.N. Aqueous biphasic systems: A boost brought about by using ionic liquids. Chem. Soc. Rev. 2012, 41, 4966–4995. [Google Scholar] [CrossRef]
- Piculell, L.; Lindman, B. Association and Segregation in Aqueous Polymer/Polymer, Polymer Surfactant, and Surfactant Surfactant Mixtures—Similarities and Differences. Adv. Colloid Interface Sci. 1992, 41, 149–178. [Google Scholar] [CrossRef]
- Sturesson, S.; Tjerneld, F.; Johansson, G. Partition of macromolecules and cell particles in aqueous two-phase systems based on hydroxypropyl starch and poly(ethylene glycol). Appl. Biochem. Biotechnol. 1990, 26, 281–295. [Google Scholar] [CrossRef]
- Saravanan, S.; Rao, J.R.; Nair, B.U.; Ramasami, T. Aqueous two-phase poly(ethylene glycol)-poly(acrylic acid) system for protein partitioning: Influence of molecular weight, pH and temperature. Process Biochem. 2008, 43, 905–911. [Google Scholar] [CrossRef]
- Diamond, A.D.; Hsu, J.T. Phase diagrams for dextran-PEG aqueous two-phase systems at 22 °C. Biotechnol. Tech. 1989, 3, 119–124. [Google Scholar] [CrossRef]
- Hughes, P.; Lowe, C.R. Purification of proteins by aqueous two-phase partition in novel acrylic co-polymer systems. Enzyme Microb. Technol. 1988, 10, 115–122. [Google Scholar] [CrossRef]
- Perrau, M.B.; Iliopoulos, I.; Audebert, R. Phase separation of polyelectrolyte/nonionic polymer systems in aqueous solution: Effects of salt and charge density. Polymers 1989, 30, 2112–2117. [Google Scholar] [CrossRef]
- Diamond, A.D.; Hsu, J.T. Protein partitioning in PEG/dextran aqueous two-phase systems. AIChE J. 1990, 36, 1017–1024. [Google Scholar] [CrossRef]
- Soares, R.R.G.; Azevedo, A.M.; Van Alstine, J.M.; Raquel Aires-Barros, M. Partitioning in aqueous two-phase systems: Analysis of strengths, weaknesses, opportunities and threats. Biotechnol. J. 2015, 10, 1158–1169. [Google Scholar] [CrossRef]
- Kroner, K.H.; Hustedt, H.; Kula, M.-R. Evaluation of crude dextran as phase-forming polymer for the extraction of enzymes in aqueous two-phase systems in large scale. Biotechnol. Bioeng. 1982, 24, 1015–1045. [Google Scholar] [CrossRef] [PubMed]
- Passos, H.; Ferreira, A.R.; Cláudio, A.F.M.; Coutinho, J.A.P.; Freire, M.G. Characterization of aqueous biphasic systems composed of ionic liquids and a citrate-based biodegradable salt. Biochem. Eng. J. 2012, 67, 68–76. [Google Scholar] [CrossRef]
- Johansson, H.O.; Magaldi, F.M.; Feitosa, E.; Pessoa, A. Protein partitioning in poly(ethylene glycol)/sodium polyacrylate aqueous two-phase systems. J. Chromatogr. A 2008, 1178, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Saravanan, S.; Reena, J.A.; Rao, J.R.; Murugesan, T.; Nair, B.U. Phase equilibrium compositions, densities, and viscosities of aqueous two-phase poly(ethylene glycol) + poly(acrylic acid) system at various temperatures. J. Chem. Eng. Data 2006, 51, 1246–1249. [Google Scholar] [CrossRef]
- Gupta, V.; Nath, S.; Chand, S. Role of water structure on phase separation in polyelectrolyte-polyethyleneglycol based aqueous two-phase systems. Polymers 2002, 43, 3387–3390. [Google Scholar] [CrossRef]
- Johansson, H.-O.; Feitosa, E.; Junior, A.P. Phase Diagrams of the Aqueous Two-Phase Systems of Poly(ethylene glycol)/Sodium Polyacrylate/Salts. Polymers 2011, 3, 587–601. [Google Scholar] [CrossRef]
- Essington, M.E. Environmental Soil Chemistry. Soil Sci. 1997, 162, 229–231. [Google Scholar] [CrossRef]
- Sauvant, M.P.; Pepin, D.; Bohatier, J.; Groliere, C.A.; Guillot, J. Toxicity assessment of 16 inorganic environmental pollutants by six bioassays. Ecotoxicol. Environ. Saf. 1997, 37, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Santos, J.H.P.M.; Silva, F.A.E.; Coutinho, J.A.P.; Ventura, S.P.M.; Pessoa, A. Ionic liquids as a novel class of electrolytes in polymeric aqueous biphasic systems. Process Biochem. 2015, 50, 661–668. [Google Scholar] [CrossRef][Green Version]
- Santos, J.H.P.M.; Martins, M.; Silvestre, A.J.D.; Coutinho, J.A.P.; Ventura, S.P.M. Fractionation of phenolic compounds from lignin depolymerisation using polymeric aqueous biphasic systems with ionic surfactants as electrolytes. Green Chem. 2016, 34, 29–41. [Google Scholar] [CrossRef]
- Egorova, K.S.; Ananikov, V.P. Toxicity of ionic liquids: Eco(cyto)activity as complicated, but unavoidable parameter for task-specific optimization. ChemSusChem 2014, 7, 336–360. [Google Scholar] [CrossRef]
- Frade, R.F.M.; Afonso, C.A.M. Impact of ionic liquids in environment and humans: An overview. Hum. Exp. Toxicol. 2010, 29, 1038–1054. [Google Scholar] [CrossRef] [PubMed]
- Ventura, S.P.M.; Gonçalves, A.M.M.; Sintra, T.; Pereira, J.L.; Gonçalves, F.; Coutinho, J.A.P. Designing ionic liquids: The chemical structure role in the toxicity. Ecotoxicology 2013, 22, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Merchuk, J.C.; Andrews, B.A.; Asenjo, J.A. Aqueous two-phase systems for protein separation: Studies on phase inversion. J. Chromatogr. B Biomed. Sci. Appl. 1998, 711, 285–293. [Google Scholar] [CrossRef]
- Grünfelder, T.; Filho, P.A.P.; Maurer, G. Liquid−Liquid Equilibrium of Aqueous Two-Phase Systems Containing Some Synthetic Polyelectrolytes and Polyethylene Glycol. J. Chem. Eng. Data 2009, 54, 198–207. [Google Scholar] [CrossRef]
- Forciniti, D.; Hall, C.K.; Kula, M.R. Influence of polymer molecular weight and temperature on phase composition in aqueous two-phase systems. Fluid Phase Equilib. 1991, 61, 243–262. [Google Scholar] [CrossRef]
- Glyk, A.; Scheper, T.; Beutel, S. Influence of different phase-forming parameters on the phase diagram of several PEG-salt aqueous two-phase systems. J. Chem. Eng. Data 2014, 59, 850–859. [Google Scholar] [CrossRef]
- Wysoczanska, K.; Macedo, E.A. Influence of the Molecular Weight of PEG on the Polymer/Salt Phase Diagrams of Aqueous Two-Phase Systems. J. Chem. Eng. Data 2016, 61, 4229–4235. [Google Scholar] [CrossRef]
- Freire, M.G.; Pereira, J.F.B.; Francisco, M.; Rodríguez, H.; Rebelo, L.P.N.; Rogers, R.D.; Coutinho, J.A.P. Insight into the interactions that control the phase behaviour of new aqueous biphasic systems composed of polyethylene glycol polymers and ionic liquids. Chem. A Eur. J. 2012, 18, 1831–1839. [Google Scholar] [CrossRef] [PubMed]
- Pereira, J.F.B.; Kurnia, K.A.; Cojocaru, O.A.; Gurau, G.; Rebelo, L.P.N.; Rogers, R.D.; Freire, M.G.; Coutinho, J.A.P. Molecular interactions in aqueous biphasic systems composed of polyethylene glycol and crystalline vs. liquid cholinium-based salts. Phys. Chem. Chem. Phys. 2014, 16, 5723–5731. [Google Scholar] [CrossRef] [PubMed]
- Pfennig, A.; Schwerin, A.; Gaube, J. Consistent view of electrolytes in aqueous two-phase systems. J. Chromatogr. B Biomed. Appl. 1998, 711, 45–52. [Google Scholar] [CrossRef]
- Johansson, H.O.; Karlström, G.; Tjerneld, F.; Haynes, C.A. Driving forces for phase separation and partitioning in aqueous two-phase systems. J. Chromatogr. B Biomed. Sci. Appl. 1998, 711, 3–17. [Google Scholar] [CrossRef]
- Shameli, K.; Ahmad, M.A.; Jazayeri, S.D.; Sedaghat, S.; Shabanzadeh, P.; Jahangirian, H.; Mahdavi, M.; Abdollahi, Y. Syntheis and Characterization of Polyethylene Glycol Mediated Silver Nanoparticles by the Green Method. Int. J. Mol. Sci. 2012, 13, 6639–6650. [Google Scholar] [CrossRef]
- Grabowska, B.; Holtzer, M. Structural Examination of the Cross-linking Reaction Mechanism of Polyacrylate Binding Agents. Arch. Metall. Mater. 2009, 54, 427–437. [Google Scholar]
- Lee, D.H.; Condrate, R.A.; Reed, J.S. Infrared spectral investigation of polyacrylate adsorption on alumina. J. Mater. Sci. 1996, 31, 471–478. [Google Scholar] [CrossRef]
- Neves, C.M.S.S.; Ventura, S.P.M.; Freire, M.G.; Marrucho, I.M.; Coutinho, J.A.P. Evaluation of Cation Influence on the Formation and Extraction Capacity of Ionic-Liquid-Based Aqueous Biphasic Systems. J. Phys. Chem. B 2009, 113, 5194–5199. [Google Scholar] [CrossRef] [PubMed]
 ) PEG 600; (
) PEG 600; ( ) PEG 2000; (
) PEG 2000; ( ) PEG 4000; (
) PEG 4000; ( ) PEG 6000; and (✕) PEG 8000. Data corresponding to the latter system were taken from the literature [22].
) PEG 6000; and (✕) PEG 8000. Data corresponding to the latter system were taken from the literature [22].
 ) PEG 600; (
) PEG 600; ( ) PEG 2000; (
) PEG 2000; ( ) PEG 4000; (
) PEG 4000; ( ) PEG 6000; and (✕) PEG 8000. Data corresponding to the latter system were taken from the literature [22].
) PEG 6000; and (✕) PEG 8000. Data corresponding to the latter system were taken from the literature [22].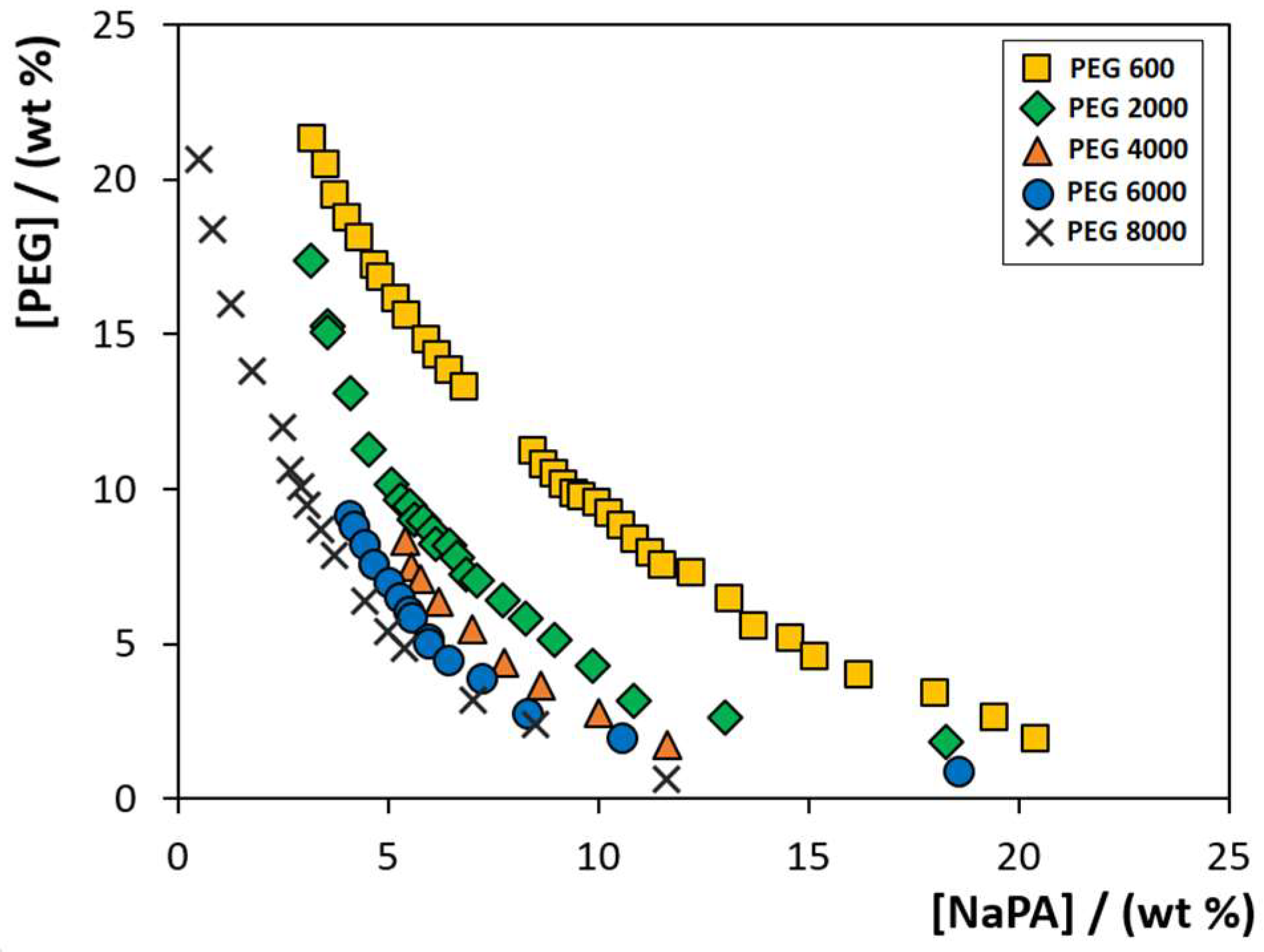


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bernardo, S.C.; Capela, E.V.; Pereira, J.F.B.; Ventura, S.P.M.; Freire, M.G.; Coutinho, J.A.P. Opposite Effects Induced by Cholinium-Based Ionic Liquid Electrolytes in the Formation of Aqueous Biphasic Systems Comprising Polyethylene Glycol and Sodium Polyacrylate. Molecules 2021, 26, 6612. https://doi.org/10.3390/molecules26216612
Bernardo SC, Capela EV, Pereira JFB, Ventura SPM, Freire MG, Coutinho JAP. Opposite Effects Induced by Cholinium-Based Ionic Liquid Electrolytes in the Formation of Aqueous Biphasic Systems Comprising Polyethylene Glycol and Sodium Polyacrylate. Molecules. 2021; 26(21):6612. https://doi.org/10.3390/molecules26216612
Chicago/Turabian StyleBernardo, Sandra C., Emanuel V. Capela, Jorge F. B. Pereira, Sónia P. M. Ventura, Mara G. Freire, and João A. P. Coutinho. 2021. "Opposite Effects Induced by Cholinium-Based Ionic Liquid Electrolytes in the Formation of Aqueous Biphasic Systems Comprising Polyethylene Glycol and Sodium Polyacrylate" Molecules 26, no. 21: 6612. https://doi.org/10.3390/molecules26216612
APA StyleBernardo, S. C., Capela, E. V., Pereira, J. F. B., Ventura, S. P. M., Freire, M. G., & Coutinho, J. A. P. (2021). Opposite Effects Induced by Cholinium-Based Ionic Liquid Electrolytes in the Formation of Aqueous Biphasic Systems Comprising Polyethylene Glycol and Sodium Polyacrylate. Molecules, 26(21), 6612. https://doi.org/10.3390/molecules26216612








