Citrus aurantium L. Active Constituents, Biological Effects and Extraction Methods. An Updated Review
Abstract
1. Introduction
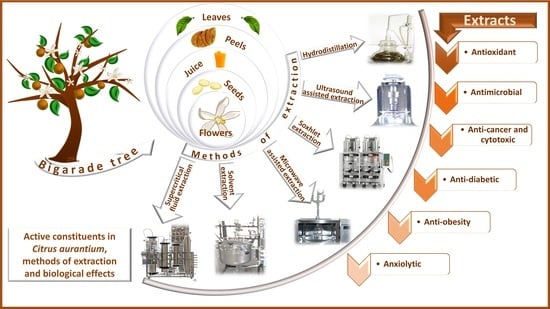
2. C. aurantium Active Constituents
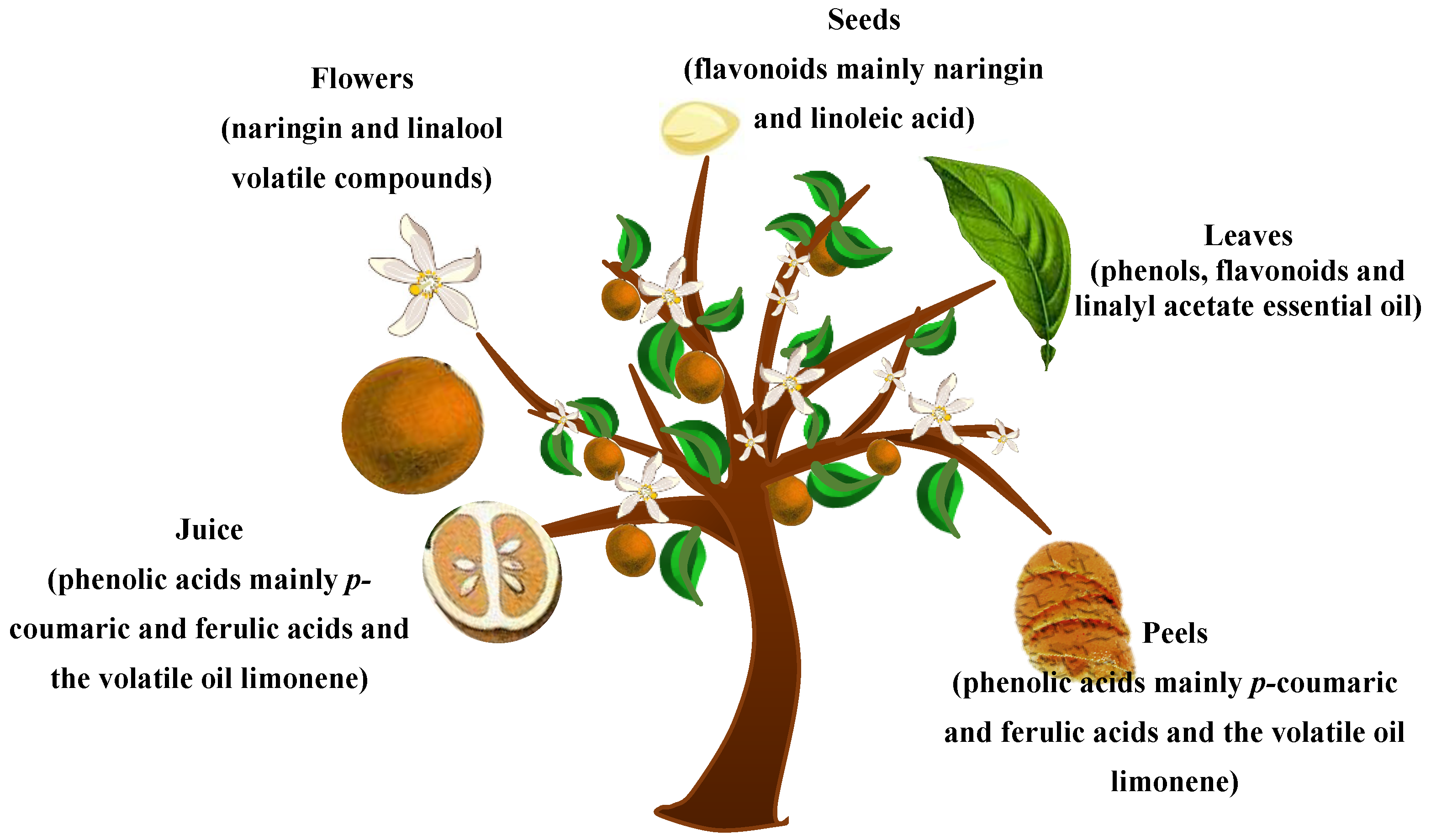
2.1. C. aurantium Juice
2.2. C. aurantium Flowers
2.3. C. aurantium Seeds
2.4. C. aurantium Leaves
2.5. C. aurantium Peels
3. Biological Effects of C. aurantium Active Constituents
3.1. Antioxidant Effect
3.2. Antimicrobial Effect
3.3. Anti-Cancer and Cytotoxic Effect
3.4. Anti-Diabetic Effect
3.5. Anti-Obesity Effect
3.6. Anxiolytic Effect
4. Extraction Methods of Active Constituents from C. aurantium
4.1. Hydrodistillation
4.2. Solvent Extraction
4.3. Soxhlet Extraction
4.4. Ultrasound-Assisted Extraction
4.5. Microwave-Assisted Extraction
4.6. Supercritical Fluid Extraction
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- García-Salas, P.; Gómez-Caravaca, A.M.; Arráez-Román, D.; Segura-Carretero, A.; Guerra-Hernández, E.; García-Villanova, B.; Fernández-Gutiérrez, A. Influence of technological processes on phenolic compounds, organic acids, furanic derivatives, and antioxidant activity of whole-lemon powder. Food Chem. 2013, 141, 869–878. [Google Scholar] [CrossRef]
- Putnik, P.; Bursać Kovačević, D.; Režek Jambrak, A.; Barba, F.J.; Cravotto, G.; Binello, A.; Lorenzo, J.M.; Shpigelman, A. Innovative “green” and novel strategies for the extraction of bioactive added value compounds from citrus wastes—A review. Molecules 2017, 22, 680. [Google Scholar] [CrossRef]
- USDA-Foreign Agricultural Service (USDA-FAS). Citrus: World Markets and Trade; Foreign Agricultural Service: Washington, DC, USA, 2020.
- Lv, X.; Zhao, S.; Ning, Z.; Zeng, H.; Shu, Y.; Tao, O.; Xiao, C.; Lu, C.; Liu, Y. Citrus fruits as a treasure trove of active natural metabolites that potentially provide benefits for human health. Chem. Cent. J. 2015, 9, 68. [Google Scholar] [CrossRef]
- Liu, Y.; Heying, E.; Tanumihardjo, S.A. History, Global Distribution, and Nutritional Importance of Citrus Fruits. Compr. Rev. Food Sci. Food Saf. 2012, 11, 530–545. [Google Scholar] [CrossRef]
- Turner, T.; Burri, B.J. Potential nutritional benefits of current citrus consumption. Agriculture 2013, 3, 170–187. [Google Scholar] [CrossRef]
- González-Mas, M.C.; Rambla, J.L.; López-Gresa, M.P.; Amparo Blázquez, M.; Granell, A. Volatile compounds in citrus essential oils: A comprehensive review. Front. Plant Sci. 2019, 10, 1–18. [Google Scholar] [CrossRef]
- Mannucci, C.; Calapai, F.; Cardia, L.; Inferrera, G.; D’Arena, G.; Di Pietro, M.; Navarra, M.; Gangemi, S.; Ventura Spagnolo, E.; Calapai, G. Clinical pharmacology of Citrus aurantium and Citrus sinensis for the treatment of anxiety. Evid. Based Complement. Altern. Med. 2018, 2018, 3624094. [Google Scholar] [CrossRef]
- Suntar, I.; Khan, H.; Patel, S.; Celano, R.; Rastrelli, L. An overview on Citrus aurantium L.: Its functions as food ingredient and therapeutic agent. Oxid. Med. Cell. Longev. 2018, 2018, 7864269. [Google Scholar] [CrossRef]
- Ersus, S.; Cam, M. Determination of organic acids, total phenolic content, and antioxidant capacity of sour Citrus aurantium fruits. Chem. Nat. Compd. 2007, 43, 607–609. [Google Scholar] [CrossRef]
- Tounsi, M.S.; Wannes, W.A.; Ouerghemmi, I.; Jegham, S.; Njima, Y.B.; Hamdaoui, G.; Zemni, H.; Marzouk, B. Juice components and antioxidant capacity of four Tunisian Citrus varieties. J. Sci. Food Agric. 2011, 91, 142–151. [Google Scholar] [CrossRef]
- Jabri Karoui, I.; Marzouk, B. Characterization of bioactive compounds in Tunisian bitter orange (Citrus aurantium L.) peel and juice and determination of their antioxidant activities. BioMed Res. Int. 2013, 2013, 345415. [Google Scholar] [CrossRef]
- Haraoui, N.; Allem, R.; Chaouche, T.M.; Belouazni, A. In-vitro antioxidant and antimicrobial activities of some varieties citrus grown in Algeria. Adv. Tradit. Med. 2020, 20, 23–34. [Google Scholar] [CrossRef]
- Sarrou, E.; Chatzopoulou, P.; Dimassi-Theriou, K.; Therios, I. Volatile constituents and antioxidant activity of peel, flowers and leaf oils of Citrus aurantium L. growing in Greece. Molecules 2013, 18, 10639–10647. [Google Scholar] [CrossRef] [PubMed]
- Labadie, C.; Ginies, C.; Guinebretiere, M.H.; Renard, C.M.G.C.; Cerutti, C.; Carlin, F. Hydrosols of orange blossom (Citrus aurantium), and rose flower (Rosa damascena and Rosa centifolia) support the growth of a heterogeneous spoilage microbiota. Food Res. Int. 2015, 76, 576–586. [Google Scholar] [CrossRef] [PubMed]
- Değirmenci, H.; Erkurt, H. Relationship between volatile components, antimicrobial and antioxidant properties of the essential oil, hydrosol and extracts of Citrus aurantium L. flowers. J. Infect. Public Health 2020, 13, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Karimi, E.; Oskoueian, E.; Hendra, R.; Oskoueian, A.; Jaafar, H.Z.E. Phenolic compounds characterization and biological activities of Citrus aurantium bloom. Molecules 2012, 17, 1203–1218. [Google Scholar] [CrossRef] [PubMed]
- Pasandideh, S.; Arasteh, A. Evaluation of antioxidant and inhibitory properties of Citrus aurantium L. on the acetylcholinesterase activity and the production of amyloid nano-bio fibrils. Int. J. Biol. Macromol. 2021, 182, 366–372. [Google Scholar] [CrossRef]
- Hamedi, A.; Zarshenas, M.; Jamshidzadeh, A.; Ahmadi, S.; Heidari, R.; Pasdran, A. Citrus aurantium (bitter orange) seed oil: Pharmacognostic, anti-inflammatory, and anti-nociceptive properties. Herb. Prod. 2019, 5, 153–164. [Google Scholar] [CrossRef]
- Abou Baker, D.H.; Ibrahim, B.M.M.; Hassan, N.S.; Yousuf, A.F.; Gengaihi, S. El Exploiting Citrus aurantium seeds and their secondary metabolites in the management of Alzheimer disease. Toxicol. Rep. 2020, 7, 723–729. [Google Scholar] [CrossRef]
- Moulehi, I.; Bourgou, S.; Ourghemmi, I.; Tounsi, M.S. Variety and ripening impact on phenolic composition and antioxidant activity of mandarin (Citrus reticulate Blanco) and bitter orange (Citrus aurantium L.) seeds extracts. Ind. Crops Prod. 2012, 39, 74–80. [Google Scholar] [CrossRef]
- Falcinelli, B.; Famiani, F.; Paoletti, A.; D’egidio, S.; Stagnari, F.; Galieni, A.; Benincasa, P. Phenolic compounds and antioxidant activity of sprouts from seeds of Citrus species. Agriculture 2020, 10, 33. [Google Scholar] [CrossRef]
- Dandekar, D.V.; Jayaprakasha, G.K.; Patil, B.S. Hydrotropic extraction of bioactive limonin from sour orange (Citrus aurantium L.) seeds. Food Chem. 2008, 109, 515–520. [Google Scholar] [CrossRef]
- Khettal, B.; Kadri, N.; Tighilet, K.; Adjebli, A.; Dahmoune, F.; Maiza-Benabdeslam, F. Phenolic compounds from Citrus leaves: Antioxidant activity and enzymatic browning inhibition. J. Complement. Integr. Med. 2017, 14, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Suryawanshi, J.A.S. An overview of Citrus aurantium used in treatment of various diseases. Afr. J. Plant Sci. 2011, 5, 390–395. [Google Scholar]
- Isabel, M.; Carvalho, F.; Osta, M. Anxiolytic and Sedative Effects of Extracts and Essential Oil from Citrus aurantium L. Pharm. Soc. Jpn. 2002, 25, 1629–1633. [Google Scholar]
- Gholivand, M.B.; Piryaei, M.; Abolghasemi, M.M. Analysis of volatile oil composition of Citrus aurantium L. by microwave-assisted extraction coupled to headspace solid-phase microextraction with nanoporous based fibers. J. Sep. Sci. 2013, 36, 872–877. [Google Scholar] [CrossRef] [PubMed]
- Periyanayagam, K.; Dhanalakshmi, S.; Karthikeyan, V.; Jagadeesan, M. Phytochemical studies and GC/MS analysis on the isolated essential oil from the leaves of Citrus aurantium Linn. J. Nat. Prod. Plant Resour. 2013, 3, 19–23. [Google Scholar]
- Lagha-Benamrouche, S.; Madani, K. Phenolic contents and antioxidant activity of orange varieties (Citrus sinensis L. and Citrus aurantium L.) cultivated in Algeria: Peels and leaves. Ind. Crops Prod. 2013, 50, 723–730. [Google Scholar] [CrossRef]
- Shan, Y. Functional Components of Citrus Peel. In Comprehensive Utilization of Citrus By-Products; Academic Press: San Diego, CA, USA, 2016; pp. 1–13. ISBN 9780128097854. [Google Scholar]
- Maurya, A.K.; Mohanty, S.; Pal, A.; Chanotiya, C.S.; Bawankule, D.U. The essential oil from Citrus limetta Risso peels alleviates skin inflammation: In-vitro and in-vivo study. J. Ethnopharmacol. 2018, 212, 86–94. [Google Scholar] [CrossRef]
- Singh, B.; Singh, J.P.; Kaur, A.; Yadav, M.P. Insights into the chemical composition and bioactivities of citrus peel essential oils. Food Res. Int. 2021, 143, 110231. [Google Scholar] [CrossRef]
- Karabiyikli, Ş.; Deǧirmenci, H.; Karapinar, M. Inhibitory effect of sour orange (Citrus aurantium) juice on Salmonella Typhimurium and Listeria monocytogenes. LWT Food Sci. Technol. 2014, 55, 421–425. [Google Scholar] [CrossRef]
- Bustamante, J.; van Stempvoort, S.; García-Gallarreta, M.; Houghton, J.A.; Briers, H.K.; Budarin, V.L.; Matharu, A.S.; Clark, J.H. Microwave assisted hydro-distillation of essential oils from wet citrus peel waste. J. Clean. Prod. 2016, 137, 598–605. [Google Scholar] [CrossRef]
- Kademi, H.I.; Garba, U. Citrus peel essential oils: A review on composition and antimicrobial activities. Int. J. Food Saf. 2017, 9, 38–44. [Google Scholar]
- Burnett, C.L.; Fiume, M.M.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.C.; Marks, J.G.; Shank, R.C.; Slaga, T.J.; et al. Safety Assessment of Citrus-Derived Peel Oils as Used in Cosmetics. Int. J. Toxicol. 2019, 38, 33S–59S. [Google Scholar] [CrossRef] [PubMed]
- Hosni, K.; Zahed, N.; Chrif, R.; Abid, I.; Medfei, W.; Kallel, M.; Brahim, N.B.; Sebei, H. Composition of peel essential oils from four selected Tunisian Citrus species: Evidence for the genotypic influence. Food Chem. 2010, 123, 1098–1104. [Google Scholar] [CrossRef]
- Bendaha, H.; Bouchal, B.; El Mounsi, I.; Salhi, A.; Berrabeh, M.; El Bellaoui, M.; Mimouni, M. Chemical composition, antioxidant, antibacterial and antifungal activities of peel essential oils of Citrus aurantium grown in Eastern Morocco. Der Pharm. Lett. 2016, 8, 239–245. [Google Scholar]
- Teneva, D.; Denkova-Kostova, R.; Goranov, B.; Hristova-Ivanova, Y.; Slavchev, A.; Denkova, Z.; Kostov, G. Chemical composition, antioxidant activity and antimicrobial activity of essential oil from Citrus aurantium L zest against some pathogenic microorganisms. Zeitschrift fur Naturforsch. Sect. C J. Biosci. 2019, 74, 105–111. [Google Scholar] [CrossRef]
- Tundis, R.; Loizzo, M.R.; Bonesi, M.; Menichini, F.; Mastellone, V.; Colica, C.; Menichini, F. Comparative Study on the Antioxidant Capacity and Cholinesterase Inhibitory Activity of Citrus aurantifolia Swingle, C. aurantium L., and C. bergamia Risso and Poit. Peel Essential Oils. J. Food Sci. 2012, 77, 40–46. [Google Scholar] [CrossRef]
- Ben Hsouna, A.; Gargouri, M.; Dhifi, W.; Ben Saad, R.; Sayahi, N.; Mnif, W.; Saibi, W. Antioxidant capacity and UPLC–PDA ESI–MS polyphenolic profile of Citrus aurantium extracts obtained by ultrasound assisted extraction. Environ. Toxicol. 2019, 34, 388–400. [Google Scholar] [CrossRef]
- Hosseini, S.S.; Khodaiyan, F.; Kazemi, M.; Najari, Z. Optimization and characterization of pectin extracted from sour orange peel by ultrasound assisted method. Int. J. Biol. Macromol. 2019, 125, 621–629. [Google Scholar] [CrossRef]
- Hosseini, S.S.; Khodaiyan, F.; Yarmand, M.S. Optimization of microwave assisted extraction of pectin from sour orange peel and its physicochemical properties. Carbohydr. Polym. 2016, 140, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Divya, P.J.; Jamuna, P.; Jyothi, L.A. Antioxidant properties of fresh and processed Citrus aurantium fruit. Cogent Food Agric. 2016, 2, 1184119. [Google Scholar] [CrossRef]
- Zeghad, N.; Ahmed, E.; Belkhiri, A.; Vander Heyden, Y.; Demeyer, K. Antioxidant activity of Vitis vinifera, Punica granatum, Citrus aurantium and Opuntia ficus indica fruits cultivated in Algeria. Heliyon 2019, 5, e01575. [Google Scholar] [CrossRef]
- Kadhim Hindi, N.K.; Ghani Chabuck, Z.A. Antimicrobial activity of different aqueous lemon extracts. J. Appl. Pharm. Sci. 2013, 3, 74–78. [Google Scholar] [CrossRef]
- Gopal, P.V. Evaluation of Anti-Microbial Activity of Citrus aurantium Against Some Gram Positive and Negative Bacterial Strains. Pharmacia 2012, 1, 107–109. [Google Scholar]
- Okla, M.K.; Alamri, S.A.; Salem, M.Z.M.; Ali, H.M.; Behiry, S.I.; Nasser, R.A.; Alaraidh, I.A.; Al-Ghtani, S.M.; Soufan, W. Yield, phytochemical constituents, and antibacterial activity of essential oils from the leaves/twigs, branches, branch wood, and branch bark of sour orange (Citrus aurantium L.). Processes 2019, 7, 363. [Google Scholar] [CrossRef]
- Kačániová, M.; Terentjeva, M.; Galovičová, L.; Ivanišová, E.; Štefániková, J.; Valková, V.; Borotová, P.; Łukasz Kowalczewski, P.; Kunová, S.; Felšöciová, S.; et al. Biological Activity and Antibiofilm Molecular Profile of Citrus aurantium Essential Oil and Its Application in a Food Model. Molecules 2020, 25, 3956. [Google Scholar] [CrossRef]
- Benzaid, C.; Belmadani, A.; Tichati, L.; Djeribi, R.; Rouabhia, M. Effect of Citrus aurantium L. Essential oil on streptococcus mutans growth, biofilm formation and virulent genes expression. Antibiotics 2021, 10, 54. [Google Scholar] [CrossRef]
- Park, K.; Park, H.S.; Kim, M.K.; Hong, G.E.; Nagappan, A.; Lee, H.J.; Yumnam, S.; Lee, W.S.; Won, C.K.; Shin, S.C.; et al. Flavonoids identified from Korean Citrus aurantium L. inhibit Non-Small Cell Lung Cancer growth in vivo and in vitro. J. Funct. Foods 2014, 7, 287–297. [Google Scholar] [CrossRef]
- Shen, C.Y.; Yang, L.; Jiang, J.G.; Zheng, C.Y.; Zhu, W. Immune enhancement effects and extraction optimization of polysaccharides from Citrus aurantium L. var. amara Engl. Food Funct. 2017, 8, 796–807. [Google Scholar] [CrossRef]
- Han, M.H.; Lee, W.S.; Lu, J.N.; Kim, G.; Jung, J.M.; Ryu, C.H.; Kim, G.I.Y.; Hwang, H.J.; Kwon, T.K.; Choi, Y.H. Citrus aurantium L. exhibits apoptotic effects on U937 human leukemia cells partly through inhibition of Akt. Int. J. Oncol. 2012, 40, 2090–2096. [Google Scholar] [CrossRef] [PubMed]
- Jayaprakasha, G.K.; Mandadi, K.K.; Poulose, S.M.; Jadegoud, Y.; Nagana Gowda, G.A.; Patil, B.S. Novel triterpenoid from Citrus aurantium L. possesses chemopreventive properties against human colon cancer cells. Bioorg. Med. Chem. 2008, 16, 5939–5951. [Google Scholar] [CrossRef]
- Zhao, H.Y.; Yang, L.; Wei, J.; Huang, M.; Jiang, J.G. Bioactivity evaluations of ingredients extracted from the flowers of Citrus aurantium L. var. amara Engl. Food Chem. 2012, 135, 2175–2181. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.W.; Lee, D.R.; Choi, B.K.; Kim, H.S.; Yang, S.H.; Suh, J.W.; Kim, K.S. Protective effects of a polymethoxy flavonoids-rich Citrus aurantium peel extract on liver fibrosis induced by bile duct ligation in mice. Asian Pac. J. Trop. Med. 2016, 9, 1158–1164. [Google Scholar] [CrossRef] [PubMed]
- Jiao, S.; Huang, C.; Wang, H.; Yu, S. Effects of Citrus aurantium extract on liver antioxidant defense function in experimental diabetic mouse. Wei Sheng Yan Jiu 2007, 36, 689. [Google Scholar] [PubMed]
- Kim, G.S.; Park, H.J.; Woo, J.H.; Kim, M.K.; Koh, P.O.; Min, W.; Ko, Y.G.; Kim, C.H.; Won, C.K.; Cho, J.H. Citrus aurantium flavonoids inhibit adipogenesis through the Akt signaling pathway in 3T3-L1 cells. BMC Complement. Altern. Med. 2012, 12, 31. [Google Scholar] [CrossRef]
- Jia, S.; Hu, Y.; Zhang, W.; Zhao, X.; Chen, Y.; Sun, C.; Li, X.; Chen, K. Hypoglycemic and hypolipidemic effects of neohesperidin derived from Citrus aurantium L. in diabetic KK-Ay mice. Food Funct. 2015, 6, 878–886. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Kim, H.L.; Jung, Y.; Ahn, K.S.; Kwak, H.J.; Um, J.Y. Bitter orange (Citrus aurantium linné) improves obesity by regulating adipogenesis and thermogenesis through AMPK activation. Nutrients 2019, 11, 1988. [Google Scholar] [CrossRef]
- Peixoto, J.S.; Comar, J.F.; Moreira, C.T.; Soares, A.A.; De Oliveira, A.L.; Bracht, A.; Peralta, R.M. Effects of Citrus aurantium (Bitter Orange) Fruit Extracts and p-Synephrine on Metabolic Fluxes in the Rat Liver. Molecules 2012, 17, 5854–5869. [Google Scholar] [CrossRef]
- Stohs, S.J.; Preuss, H.G.; Shara, M. The safety of Citrus aurantium (bitter orange) and its primary protoalkaloid p-synephrine. Phyther. Res. 2011, 25, 1421–1428. [Google Scholar] [CrossRef]
- Stohs, S.J. Safety, Efficacy, and Mechanistic Studies Regarding Citrus aurantium (Bitter Orange) Extract and p-Synephrine. Phyther. Res. 2017, 31, 1463–1474. [Google Scholar] [CrossRef]
- Li, X.Y.; Hao, Y.F.; Hao, Z.X.; Jiang, J.G.; Liu, Q.; Shen, Q.; Liu, L.; Yi, Y.K.; Shen, C.Y. Inhibitory effect of chloroform extracts from Citrus aurantium L. var. amara Engl. on fat accumulation. Phytomedicine 2021, 90, 153634. [Google Scholar] [CrossRef]
- Pimenta, F.C.F.; Alves, M.F.; Pimenta, M.B.F.; Melo, S.A.L.; Almeida, A.A.F.D.; Leite, J.R.; Pordeus, L.C.D.M.; Diniz, M.D.F.F.M.; Almeida, R.N.D. Anxiolytic Effect of Citrus aurantium L. on Patients with Chronic Myeloid Leukemia. Phyther. Res. 2016, 30, 613–617. [Google Scholar] [CrossRef]
- Neto, G.C.; Braga, J.E.F.; Alves, M.F.; de Morais Pordeus, L.C.; Dos Santos, S.G.; Scotti, M.T.; Almeida, R.N.; Diniz, M.F.F.M. Anxiolytic Effect of Citrus aurantium L. in Crack Users. Evid. Based Complement. Altern. Med. 2017, 2017, 7217619. [Google Scholar] [CrossRef]
- Azmir, J.; Zaidul, I.S.M.; Rahman, M.M.; Sharif, K.M.; Mohamed, A.; Sahena, F.; Jahurul, M.H.A.; Ghafoor, K.; Norulaini, N.A.N.; Omar, A.K.M. Techniques for extraction of bioactive compounds from plant materials: A review. J. Food Eng. 2013, 117, 426–436. [Google Scholar] [CrossRef]
- Aramrueang, N.; Asavasanti, S.; Khanunthong, A. Leafy Vegetables. In Integrated Processing Technologies for Food and Agricultural By-Products; Academic Press: San Diego, CA, USA, 2019; pp. 245–272. ISBN 9780128141397. [Google Scholar]
- Sawamura, M.; Chemat, F. Techniques for Oil Extraction. In Citrus Essential Oils: Flavor and Fragrance; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2010; pp. 9–36. ISBN 9780470372180. [Google Scholar]
- Bonaccorsi, I.; Sciarrone, D.; Schipilliti, L.; Trozzi, A.; Fakhry, H.A.; Dugo, G. Composition of Egyptian nerolì oil. Nat. Prod. Commun. 2011, 6, 1009–1014. [Google Scholar] [CrossRef] [PubMed]
- Haj Ammar, A.; Bouajila, J.; Lebrihi, A.; Mathieu, F.; Romdhane, M.; Zagrouba, F. Chemical composition and in vitro antimicrobial and antioxidant activities of Citrus aurantium L. flowers essential oil (Neroli Oil). Pak. J. Biol. Sci. 2012, 15, 1034–1040. [Google Scholar] [CrossRef]
- Hsouna, A.B.; Hamdi, N.; Halima, N.B.; Abdelkafi, S. Characterization of essential oil from Citrus aurantium L. flowers: Antimicrobial and antioxidant activities. J. Oleo Sci. 2013, 62, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Ines, E.; Hajer, D.; Rachid, C. Aromatic Quality of Tunisian Sour Orange Essential Oils: Comparison Between Traditional and Industrial Extraction. Nat. Volatiles Essent. Oils 2014, 1, 66–72. [Google Scholar]
- Rahimi, A.; Hashemi, P.; Talei, G.R.; Borzuei, M.; Ghiasvand, A.R. Comparative Analyses of the Volatile Components of Citrus aurantium L. Flowers Using Ultrasonic-Assisted Headspace SPME and Hydrodistillation Combined with GC-MS and Evaluation of their Antimicrobial Activities. Anal. Bioanal. Chem. Rev. 2014, 1, 83–91. [Google Scholar]
- Kusuma, H.; Putra, A.; Mahfud, M. Comparison of Two Isolation Methods for Essential Oils from Orange Peel (Citrus aurantium L.) as a Growth Promoter for Fish: Microwave Steam Distillation and Conventional Steam Distillation. J. Aquac. Res. Dev. 2016, 7, 409. [Google Scholar] [CrossRef]
- Zhang, Q.W.; Lin, L.G.; Ye, W.C. Techniques for extraction and isolation of natural products: A comprehensive review. Chin. Med. 2018, 13, 20. [Google Scholar] [CrossRef]
- Wen, L.; He, M.; Yin, C.; Jiang, Y.; Luo, D.; Yang, B. Phenolics in Citrus aurantium fruit identified by UHPLC-MS/MS and their bioactivities. LWT 2021, 147, 111671. [Google Scholar] [CrossRef]
- Zygler, A.; Słomińska, M.; Namieśnik, J. Soxhlet extraction and new developments such as soxtec. Compr. Sampl. Sample Prep. 2012, 2, 65–82. [Google Scholar] [CrossRef]
- Rajput, R.; Mishra, J.; Sharma, R.K.; Misra, K. Characterization Techniques for Herbal Products. In Management of High Altitude Pathophysiology; Academic Press: Cambridge, MA, USA, 2018; pp. 171–202. ISBN 9780128140000. [Google Scholar]
- Elmsellem, H.; Bendaha, H.; Aouniti, A.; Chetouani, A.; Mimouni, M.; Bouyanzer, A. Comparative study of the inhibition of extracts from the peel and seeds of Citrus aurantium against the corrosion of steel in molar HCl solution. Moroc. J. Chem. 2014, 2, 1–9. [Google Scholar]
- Anticona, M.; Blesa, J.; Frigola, A.; Esteve, M.J. High biological value compounds extraction from citrus waste with non-conventional methods. Foods 2020, 9, 811. [Google Scholar] [CrossRef]
- Picó, Y. Ultrasound-assisted extraction for food and environmental samples. TrAC Trends Anal. Chem. 2013, 43, 84–99. [Google Scholar] [CrossRef]
- Esclapez, M.D.; García-Pérez, J.V.; Mulet, A.; Cárcel, J.A. Ultrasound-Assisted Extraction of Natural Products. Food Eng. Rev. 2011, 3, 108–120. [Google Scholar] [CrossRef]
- Alissandrakis, E.; Daferera, D.; Tarantilis, P.A.; Polissiou, M.; Harizanis, P.C. Ultrasound-assisted extraction of volatile compounds from citrus flowers and citrus honey. Food Chem. 2003, 82, 575–582. [Google Scholar] [CrossRef]
- Hao, K.; Hu, W.; Hou, M.; Cao, D.; Wang, Y.; Guan, Q.; Zhang, X.; Wang, A.; Yu, J.; Guo, B. Optimization of ultrasonic-assisted extraction of total phenolics from Citrus aurantium L. Blossoms and evaluation of free radical scavenging, Anti-HMG-CoA Reductase Activities. Molecules 2019, 24, 2368. [Google Scholar] [CrossRef]
- Yang, L.; Cao, Y.L.; Jiang, J.G.; Lin, Q.S.; Chen, J.; Zhu, L. Response surface optimization of ultrasound-assisted flavonoids extraction from the flower of Citrus aurantium L. var. amara Engl. J. Sep. Sci. 2010, 33, 1349–1355. [Google Scholar] [CrossRef] [PubMed]
- Ojito, K.; Herrera, Y.; Vega, N.; Portal, O. Actividad antioxidante in vitro y toxicidad de extractos hidroalcohólicos de hojas de Citrus spp. (Rutaceae). Rev. Cuba. Plantas Med. 2012, 17, 368–379. [Google Scholar]
- Ana, C.C.; Jesús, P.V.; Hugo, E.A.; Teresa, A.T.; Ulises, G.C.; Neith, P. Antioxidant capacity and UPLC–PDA ESI–MS polyphenolic profile of Citrus aurantium extracts obtained by ultrasound assisted extraction. J. Food Sci. Technol. 2018, 55, 5106–5114. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Zhou, H.; Wu, C.; Feng, X.; Han, C.; Chen, H.; Liu, Y.; Li, Y. Ultrasound-assisted aqueous two-phase extraction of synephrine, naringin, and neohesperidin from Citrus aurantium L. fruitlets. Prep. Biochem. Biotechnol. 2020, 51, 780–791. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Tomao, V.; Virot, M. Ultrasound-Assisted Extraction in Food Analysis. In Otles Handbook of Food Analysis Instruments; Taylor & Francis: New York, NY, USA, 2008; ISBN 9781420045666. [Google Scholar]
- Golmakani, M.T.; Moayyedi, M. Comparison of microwave-assisted hydrodistillation and solvent-less microwave extraction of essential oil from dry and fresh Citrus limon (Eureka variety) peel. J. Essent. Oil Res. 2016, 28, 272–282. [Google Scholar] [CrossRef]
- Wang, J.; Sun, B.; Cao, Y.; Tian, Y.; Li, X. Optimisation of ultrasound-assisted extraction of phenolic compounds from wheat bran. Food Chem. 2008, 106, 804–810. [Google Scholar] [CrossRef]
- Delazar, A.; Nahar, L.; Hamedeyazdan, S.; Sarker, S.D. Microwave-assisted extraction in natural products isolation. Methods Mol. Biol. 2012, 864, 89–115. [Google Scholar] [CrossRef]
- Kuchekar, D.M. Spectrophotometric Quantification of Flavonoid Content in Herbal Drugs Extracts and Optimization of Microwave Assisted Extraction Technique By Using Different Solvents. World J. Pharm. Res. 2017, 6, 562–569. [Google Scholar] [CrossRef][Green Version]
- Mohagheghniapour, A.; Saharkhiz, M.J.; Golmakani, M.T.; Niakousari, M. Variations in chemical compositions of essential oil from sour orange (Citrus aurantium L.) blossoms by different isolation methods. Sustain. Chem. Pharm. 2018, 10, 118–124. [Google Scholar] [CrossRef]
- Chemat, F.; Lucchesi, M.E.; Smadia, J. Solvent-Free Microwave Extraction of Volatile Natural Substances-United States Patent Office. U.S. Patent 0187340A1, 30 September 2004. [Google Scholar]
- Ferhat, M.A.; Meklati, B.Y.; Smadja, J.; Chemat, F. An improved microwave Clevenger apparatus for distillation of essential oils from orange peel. J. Chromatogr. A 2006, 1112, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Ferhat, M.; Meklati, B.; Chemat, F. Comparison of different isolation methods of essential oil from Citrus fruits: Cold pressing, hydrodistillation and microwave ‘dry’ distillation. Flavour Fragr. J. 2008, 22, 494–504. [Google Scholar] [CrossRef]
- Rovetto, L.J.; Aieta, N.V. Supercritical carbon dioxide extraction of cannabinoids from Cannabis sativa L. J. Supercrit. Fluids 2017, 129, 16–27. [Google Scholar] [CrossRef]
- De Cássia Rodrigues Batista, C.; De Oliveira, M.S.; Araújo, M.E.; Rodrigues, A.M.C.; Botelho, J.R.S.; Da Silva Souza Filho, A.P.; Machado, N.T.; Carvalho, R.N. Supercritical CO2 extraction of açaí (Euterpe oleracea) berry oil: Global yield, fatty acids, allelopathic activities, and determination of phenolic and anthocyanins total compounds in the residual pulp. J. Supercrit. Fluids 2015, 107, 364–369. [Google Scholar] [CrossRef]
- Kassim, D.M.; Hameed, M.S. Direct Extraction-Separation of Essential Oils from Citrus Peels by Supercritical Carbon Dioxide. Sep. Sci. Technol. 1989, 24, 1427–1435. [Google Scholar] [CrossRef]
- Trabelsi, D.; Aydi, A.; Zibetti, A.W.; Della Porta, G.; Scognamiglio, M.; Cricchio, V.; Langa, E.; Abderrabba, M.; Mainar, A.M. Supercritical extraction from Citrus aurantium amara peels using CO2 with ethanol as co-solvent. J. Supercrit. Fluids 2016, 117, 33–39. [Google Scholar] [CrossRef]
- Jerkovic, I.; Druzic, J.; Marijanovic, Z.; Gugic, M.; Jokić, S.D.; Roje, M. GC-FID/MS profiling of supercritical CO2 extracts of peels from Citrus aurantium, C. sinensis cv. Washington navel, C. sinensis cv. Tarocco and C. sinensis cv. Doppio Sanguigno from Dubrovnik Area (Croatia). Nat. Prod. Commun. 2015, 10, 1315–1318. [Google Scholar] [CrossRef]
| Plant Part | Extraction Method | Solvent | Active Constituents | References |
|---|---|---|---|---|
| Juice | Solvent extraction | Ether-pentane (1:1) |
| [12] |
| Solvent extraction | 80% methanol |
| [13] | |
| Solvent extraction | 80% methanol |
| [11] | |
| Flowers | UAE | n-pentane:diethylether |
| [84] |
| UAE | Ethanol |
| [86] | |
| Solvent extraction | Ethanol, methanol, and hot water |
| [17] | |
| HD | Cyclohexane |
| [73] | |
| UA-HS-SPME | Distilled water |
| [74] | |
| HD | n-hexane |
| [15] | |
| MAHD, SLME, SFME | Water |
| [95] | |
| UAE | Ethanol |
| [85] | |
| UAE | Distilled water and citric acid |
| [42] | |
| UAE | Distilled water |
| [52] | |
| Soxhlet extraction | Ethanol |
| [16] | |
| Solvent extraction | 80% ethanol |
| [18] | |
| Seeds | Solvent extraction | Na-CuS and Na-Sal |
| [23] |
| Solvent extraction | Pure methanol |
| [21] | |
| Soxhlet extraction | Acetone and petroleum ether |
| [20] | |
| Soxhlet extraction | n-hexane |
| [19] | |
| Solvent extraction | Methanol |
| [22] | |
| Leaves | UAE | Ethanol and methanol |
| [87] |
| MAE followed by HS-SPME | Water |
| [27] | |
| HD | Not specified |
| [28] | |
| Solvent extraction | Methanol:water (80:20) |
| [29] | |
| Solvent extraction | Absolute methanol |
| [24] | |
| Solvent extraction | Methanol:water (80:20) |
| [13] | |
| Peels | SFE CO2 | CO2 with ethanol as co-solvent |
| [101] |
| HD | Not specified |
| [40] | |
| HD | Diethyl ether |
| [12] | |
| SFE CO2 | CO2 with diethyl ether |
| [103] | |
| MAE | Citric acid aqueous solution |
| [43] | |
| Soxhlet and SD | Hexane |
| [38] | |
| SD | Water |
| [75] | |
| MSD | Water |
| [75] | |
| SFE CO2 | CO2 with ethanol as co-solvent |
| [102] | |
| MAE | 70% aqueous ethanol |
| [94] | |
| UAE |
|
| [88] | |
| Peel rinds squeezing | n-hexane |
| [41] | |
| HD | Not specified |
| [36] | |
| Fruit | UA-ATPE | Ethanol |
| [89] |
| Solvent extraction | 80% acetone |
| [77] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maksoud, S.; Abdel-Massih, R.M.; Rajha, H.N.; Louka, N.; Chemat, F.; Barba, F.J.; Debs, E. Citrus aurantium L. Active Constituents, Biological Effects and Extraction Methods. An Updated Review. Molecules 2021, 26, 5832. https://doi.org/10.3390/molecules26195832
Maksoud S, Abdel-Massih RM, Rajha HN, Louka N, Chemat F, Barba FJ, Debs E. Citrus aurantium L. Active Constituents, Biological Effects and Extraction Methods. An Updated Review. Molecules. 2021; 26(19):5832. https://doi.org/10.3390/molecules26195832
Chicago/Turabian StyleMaksoud, Sawssan, Roula M. Abdel-Massih, Hiba N. Rajha, Nicolas Louka, Farid Chemat, Francisco J. Barba, and Espérance Debs. 2021. "Citrus aurantium L. Active Constituents, Biological Effects and Extraction Methods. An Updated Review" Molecules 26, no. 19: 5832. https://doi.org/10.3390/molecules26195832
APA StyleMaksoud, S., Abdel-Massih, R. M., Rajha, H. N., Louka, N., Chemat, F., Barba, F. J., & Debs, E. (2021). Citrus aurantium L. Active Constituents, Biological Effects and Extraction Methods. An Updated Review. Molecules, 26(19), 5832. https://doi.org/10.3390/molecules26195832