Factors Affecting Synthetic Dye Adsorption; Desorption Studies: A Review of Results from the Last Five Years (2017–2021)
Abstract
:1. Introduction
- Dyes increases the water turbidity;
- Dyes have a major impact on the photosynthetic activity of the aquatic environment because they block the penetration of light into the water, thus inhibiting the growth of algae, which are not only important for oxygen production but are a pillar of the food chain;
- Most of the dyes are carcinogenic (bladder, kidney, liver), mutagenic and toxic to living organisms;
- They can cause allergic reactions: skin, eye, mucous membrane irritation, dermatitis, respiratory problems; and
1.1. Definition of Dyestuff
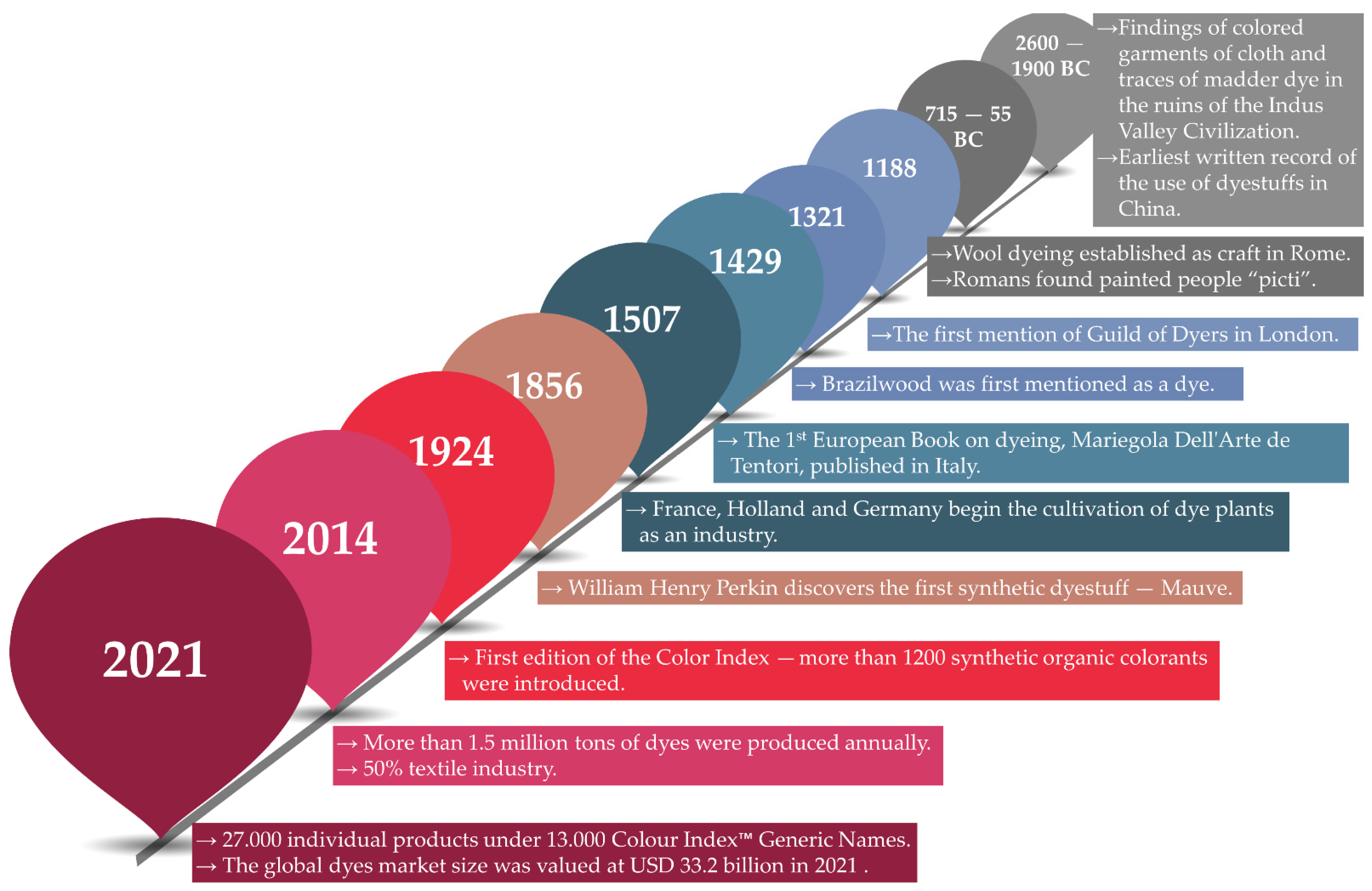
1.2. Brief History of Dye Usage
1.3. Classification of Dyes
1.3.1. Reactive Dyes
1.3.2. Direct Dyes
1.3.3. Acid Dyes
1.3.4. Cationic-Basic Dyes
1.3.5. Disperse Dyes
1.3.6. Vat Dyes
- source of materials/origin (natural–substantive and adjective–synthetic);
- method of application to the substrate (acid, basic, direct, mordant, reactive, disperse, solvent, sulfur);
- their chemical structure (azo, nitro, indigoid, cyanine, xanthene, quinione-imine, acridine, oxazine, anthraquinone, phthalein, triphenylmethane, nitroso, diarylmethane); and
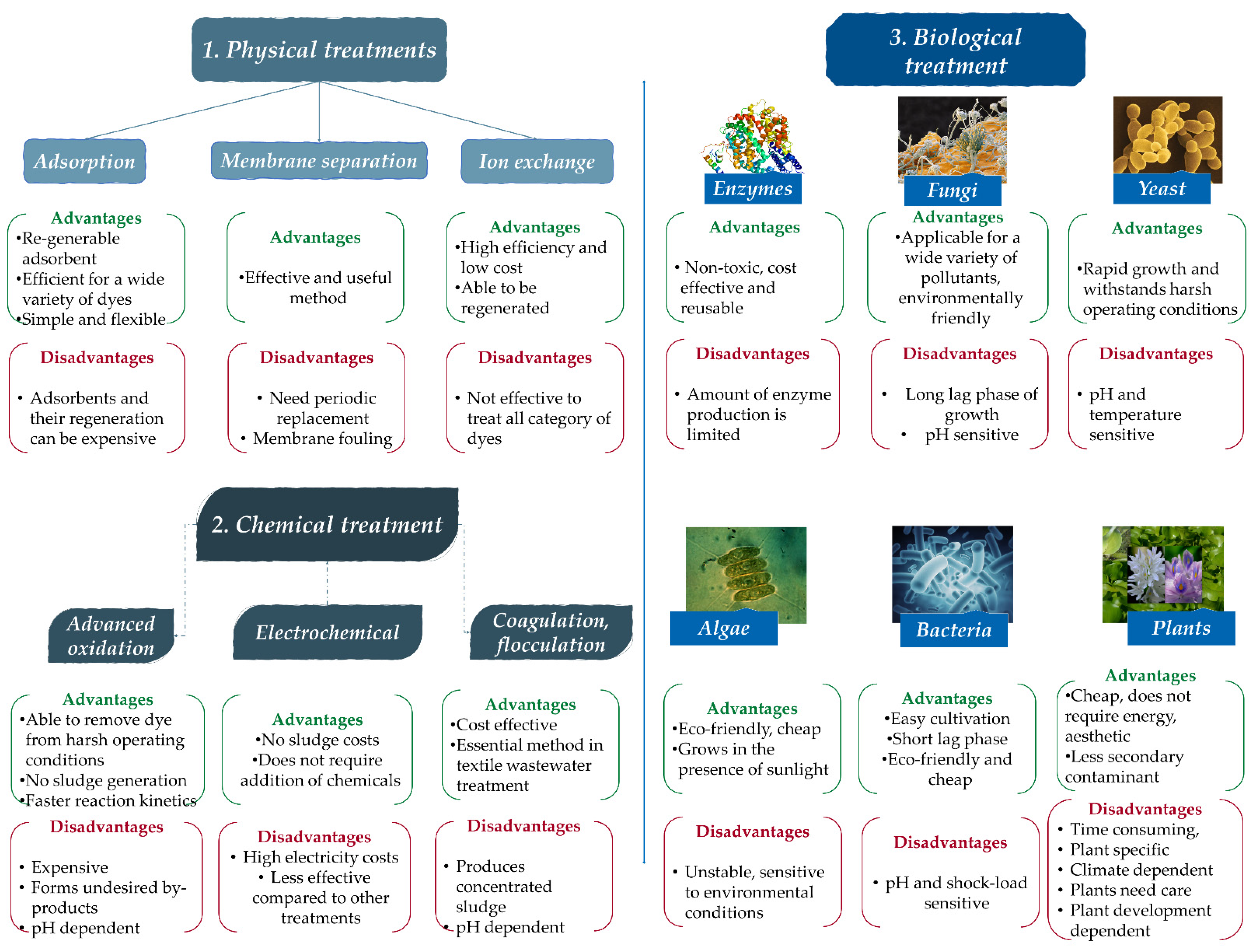
2. Dye Removing Methods, Technologies
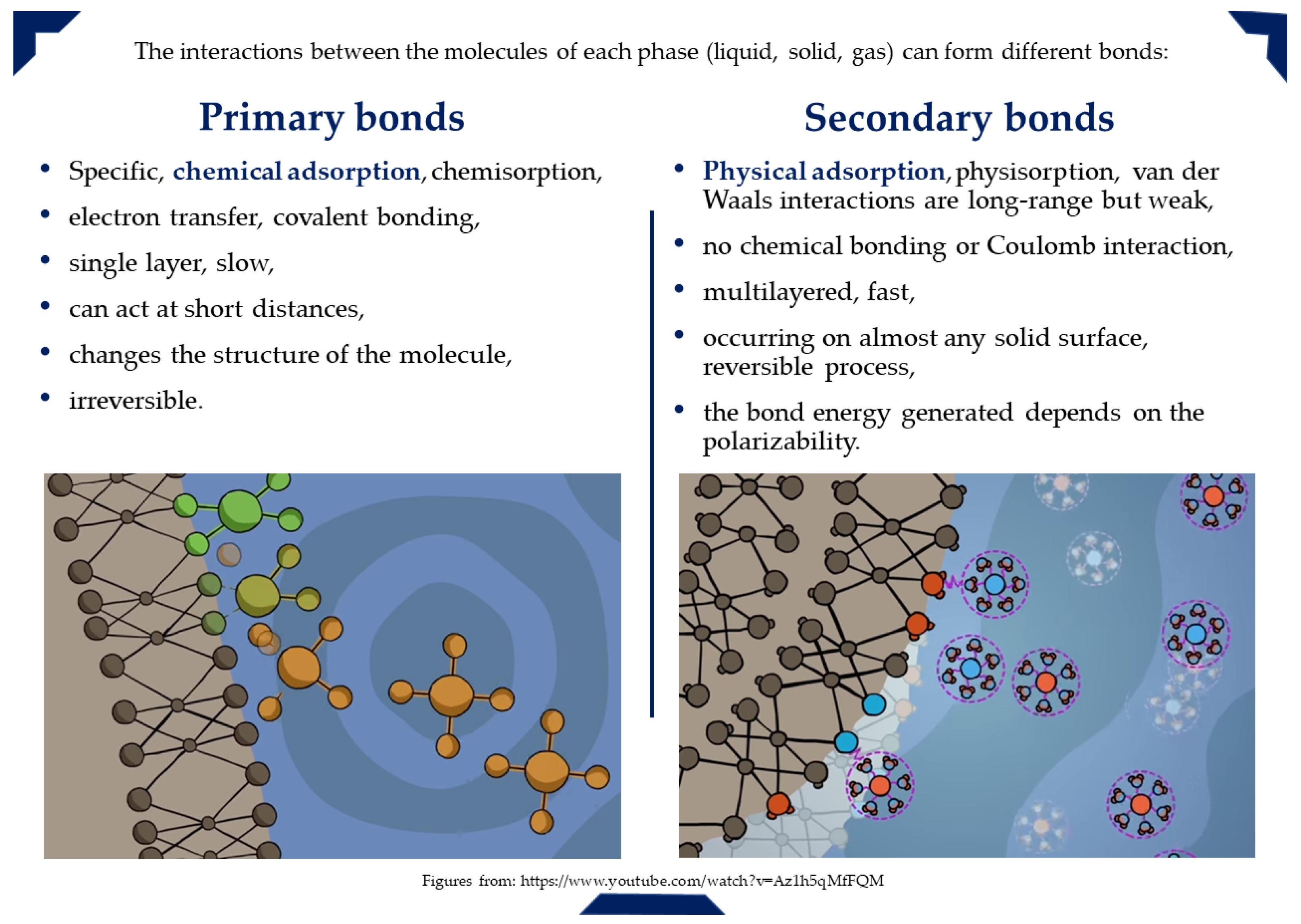
3. General Aspects of Adsorption Process
Possible Adsorbents
- natural materials: sawdust, wood, fuller’s earth or bauxite;
- natural materials treated to develop their structures and properties: activated carbons, activated alumina or silica gel;
- manufactured materials: polymeric resins, zeolites or alumino-silicates;
- agricultural solid wastes and industrial by-products: date pits, fly ash or red mud;
- biosorbents: chitosan, fungi or bacterial biomass.
- Natural adsorbents include carbon, clays, clays minerals, zeolites and ores. These natural materials are often relatively inexpensive, abundant, plentiful and readily available;
- Synthetic adsorbents are adsorbents produced from agricultural products and wastes, household wastes, industrial wastes, sewage sludges and polymer adsorbents.
- Ability to work under several wastewater parameters;
- Cost effectiveness;
- Removal capability of diverse contaminants;
- High adsorption capacity;
- High selectivity for various concentrations;
- High porosity and specific surface area;
- High durability;
- Reusability of adsorbent, ease of regeneration;
- Fast kinetics; and
- Being present in large quantities.
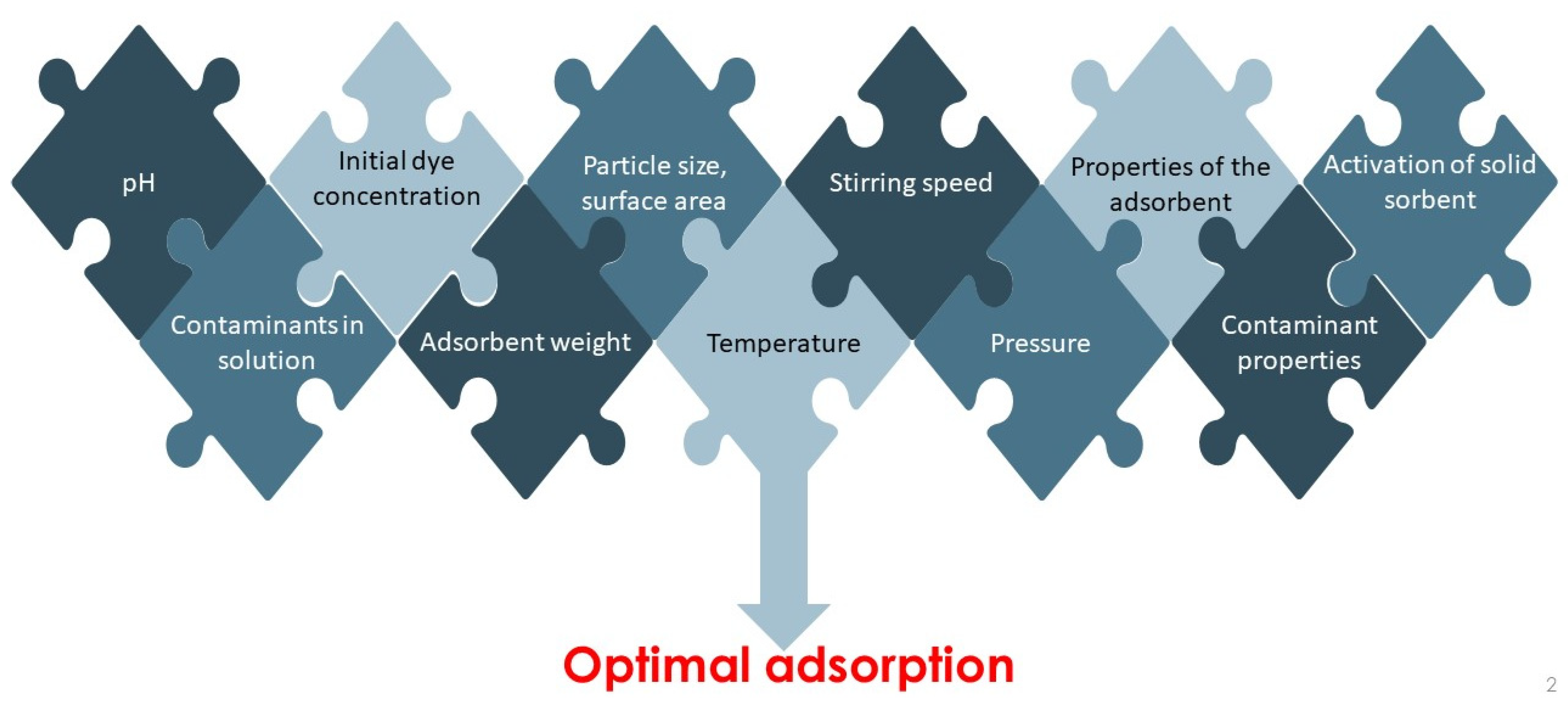
4. Factors Affecting Adsorption Process
4.1. The Effect of Initial Dye Concentration
- the removal efficiency decreases as the initial concentration increases;
- removal efficiency increases as the initial concentration increases; and
- no significant change in removal efficiency.
| Dyestuff | Adsorbent | Concentration (mg/L) | Reaction Time (min) | Efficiency Range (%) | Quantity in Equilibrium Range (qe mg/g) | Reference |
|---|---|---|---|---|---|---|
| Methylene Blue | Algerian palygorskite | 3–30 | 5 | up to 97% | 2.5–10 | [155] |
| Methylene Blue | clinoptilolite | 50–100 | 60 | increased but no significant difference > 95% | - | [156] |
| Brilliant Green | activated carbon derived from medlar nucleus | 110–200 | 60 | - | 100–180 | [157] |
| Methylene Blue | green olive stone | 50–1000 | 24 h | fluctuating values, highest 65.9 at 50 ppm | - | [158] |
| Methylene Blue | black olive stone | 50–1000 | 24 h | fluctuating values, highest 93.5 at 400 ppm | - | [158] |
| Acid Brown | Haloxylon recurvum plant | 10–60 | 180 | - | 2.846–10.011 | [159] |
| Congo Red | cocoa bean shells | 40–120 | 4–36 h | negative linear effect | [160] | |
| Methylene Blue | fava bean peels, utilizing ultrasonic-assisted (US) shaking | 3.6–100 | 70 | 70–90 | - | [161] |
| Methylene Blue | fava bean peels, conventional (CV) shaking | 3.6–100 | 70 | 80–95 | - | [161] |
| Reactive Blue 19 | corn silk | 10–500 | 60 | - | 2.0–71.6 | [162] |
| Reactive Red 218 | corn silk | 10–500 | 60 | - | 2.0–63.3 | [162] |
| Reactive Black 5 | pent tea leaves | 50–100 | 5–200 | 98.7–43.5 | 24.8 –6.7 | [163] |
| Methyl Orange | pent tea leaves | 50–100 | 5–200 | 88.7–32.7 | 22.2 –1.6 | [163] |
| Methylene Blue | Citrus limetta peel | 5–25 | 10–60 | ~100–97 | 0.06–1.62 | [164] |
| Malachite Green | Citrus limetta peel | 5–25 | 10–60 | ~97–95 | 0.17–4.70 | [164] |
| Congo Red | Citrus limetta peel | 5–25 | 10–60 | ~90–75 | 0.17–3.77 | [164] |
| Crystal Violet | mango stone biocomposite | 20–50 | 60 | - | ~25–352.79 | [165] |
| Congo Red | chitosan | 50–2000 | 30 | - | increased to 0.2 | [166] |
| Methylene Blue | chitosan | 25–100 | 30 | ~100–50 | increased to 1457.1 | [166] |
| Rhodamine B | chitosan | 25–100 | 30 | ~55–35 | increased to 990 | [166] |
| Reactive Red 120 | Moringa oleifera seed | 10–100 | 30 | - | 18.54–173.99 | [167] |
| Crystal Violet | olive leaves powder | 10–100 | 5–70 | - | ~5–45 | [168] |
4.2. The Effect of Solution pH
- When HCl is added to the solution, the surface of the adsorbent in the solution is protonated, allowing the anionic dye to bind more efficiently on its surface, due to the electrostatic attraction.
- Conversely, in basic medium, the addition of NaOH deprotonates the biomass surface, resulting in a repulsive force between the anionic dye and the biomass. Thus, the reverse phenomenon is observed for cationic dyes.
| Dyestuff | Adsorbent | Dyes Ionic Nature | pH | Observations: with the Increase (↑) of pH | Reference |
|---|---|---|---|---|---|
| Direct Red 5B | spent mushroom waste | anionic | 2 to 10 | E% ↓; Emax_pH=2 = 95% | [178] |
| Direct Black 22 | anionic | 2 to 10 | E% ↓; Emax_pH=2 = 98% | [178] | |
| Direct Black 71 | anionic | 2 to 10 | E% ↓; Emax_pH=2 = 95% | [178] | |
| Reactive Black 5 | anionic | 2 to 10 | E% ↓; Emax_pH=2 = 96% | [178] | |
| Congo Red | powdered activated carbon: rubber seed | anionic | 4 to 11 | E% ↓ | [179] |
| powdered activated carbon: rubber seed shells | 4 to 11 | E% ↓ | [179] | ||
| Methylene Blue | powdered activated carbon: rubber seed | cationic | 4 to 11 | E% ↑ | [179] |
| powdered activated carbon: rubber seed shells | 4 to 11 | E% ↑ | [179] | ||
| Eriochrome Black T | powdered vegetables wastes | anionic | 2 to 10 | E% ↓; 50.65 to 4.01% | [180] |
| calcined vegetables wastes | 2 to 10 | E% ↓; 68.87 to 31.23% | [180] | ||
| Methyl Orange | natural olive stone | anionic | 2 to 12 | q (mg/g) ↓; 26.4 to 3.3 mg/g | [158] |
| olive stone activated carbons | 2 to 12 | q (mg/g) ↓; 120 to 15 mg/g | [158] | ||
| Methylene Blue | natural olive stone | cationic | 2 to 12 | q (mg/g) ↑; 18 to 120 mg/g | [158] |
| olive stone activated carbons | 2 to 12 | q (mg/g) ↑ | [158] | ||
| Reactive Orange 16 | carbon from Phyllanthus reticulatus | anionic | 2 to 11 | q (mg/g) ↓ | [181] |
| Cationic Red X-5GN | ceramic | cationic | 2 to 10 | E% ↑ | [182] |
| Cationic Blue X-GRRL | cationic | 2 to 10 | E% ↑ | [182] | |
| Methylene Blue | activated carbon/cellulose biocomposite films | cationic | 3 to 11 | q (mg/g) ↑; 50.54 to 60.48 mg/g | [183] |
| Eriochrome Black T | almond shell | anionic | 2 to 11 | q (mg/g) ↓ | [184] |
| Malachite Green | cationic | 2 to 11 | q (mg/g) ↑ | [184] | |
| Basic Yellow 37 | bast fibers: ramie | cationic | 2 to 12 | E% ↑; Emax_pH=12 = 91% | [185] |
| bast fibers: flax | cationic | 2 to 12 | E% ↑; Emax_pH=12 = 88% | [185] | |
| bast fibers: kenaf | cationic | 2 to 12 | E% ↑; Emax_pH=12 = 78% | [185] | |
| Remazol Brilliant Violet | Trichoderma viride | anionic | 4 to 9 | E% ↓; 79.05 to 50.25% | [186] |
| Congo Red | eggshell powder | anionic | 2 to 10 | E% ↓; 98.71 to 93.17% | [133] |
| Bromphenol Blue | anionic | 2 to 10 | E% ↓; 67.61 to 1.2% | [133] | |
| Methylene Blue | cationic | 2 to 10 | E% ↑; 14.8 to 75.1% | [133] | |
| Malachite Green | cationic | 2 to 10 | E% ↑; 89.95 to 97.92% | [133] |
4.3. The Effect of Adsorbent Dosage
- adsorption sites remain unsaturated while the number of sites available for adsorption increases; or
- aggregation or agglomeration of adsorbent particles may occur, reducing the available surface area and increasing the diffusion path length.
4.4. The Effect of Adsorbent Particle Size
- the chemical structure of the dye molecule (its ionic charge) and its chemistry (its ability to form hydrolyzed species); and
- the intrinsic characteristic of the adsorbent (its crystallinity, porosity and rigidity of the polymeric chains).
4.5. The Effect of Solution Temperature
- As the temperature rises, the rate of chemical reaction also increases, so if the sorption process is chemisorption (∆Hchemisorption = −200 kJ/mol), then higher sorption efficiency will be seen at higher temperatures (this would eventually reach equilibrium).
- On the other hand, if the process is a physical adsorption (∆Hphysisorption ≈ −20 kJ/mol), then the higher temperature will negatively affect the adsorption. Temperature can chemically alter the adsorbent, its adsorption sites and activity [110].
| Dyestuff | Adsorbent | Temperature (K) | Efficiency Range (%) | Type of the Process | Quantity in Equilibrium Range (qe mg/g) | Reference |
|---|---|---|---|---|---|---|
| Basic Orange 2 | alkaline-modified nanoclay | 288–308 | 80–100 | endothermic | - | [219] |
| Congo Red | cross-linked TTU-chitosan | 298, 308 and 328 | - | endothermic | increased | [218] |
| Congo Red | modified Zeolite A | 297–309 | - | exothermic | decreased | [216] |
| Direct Sky Blue | ZnO | Beyond 313 K, the adsorption capacity was decreased, which is an indication of being endothermic up to 313 K, and exothermic beyond this temperature | highest: 40.94 | [220] | ||
| MgO | highest: 46.25 | [220] | ||||
| FeO | highest: 42.86 | [220] | ||||
| Methyl Orange | cationic polymer (Amberlite IRA 402) | 293, 303, 328 and 348 | - | endothermic | increased | [221] |
| Remazol Red | chitosan Schiff base | 293, 303, and 313 | - | endothermic | increased | [222] |
| Reactive Red 120 | activated carbon | The adsorption of RR-120 on activated carbon is of the physisorption type, as confirmed by the adsorbed energy values, and it is exothermic as verified by the internal energy | [223] | |||
| Methylene Blue | hydroxyapatite/gold nanocomposite | 290–305 | - | endothermic | increased | [217] |
| 305–330 | - | exothermic | decreased | [217] | ||
| Reactive Red 35 | multiwalled carbon nanotubes | 298, 308, 318 and 328 | 63.33–9.07 | exothermic | - | [215] |
| poly (acrylonitrile-styrene) impregnated with activated carbon | 298, 308, 318 and 328 | 67.55–97.61 | endothermic | - | [215] | |
| Methylene Blue | Citrullus colocynthis seed | 293–333 | 93.58–98.00 | endothermic | - | [143] |
| Citrullus colocynthis peel | 294–333 | 91.43–82.52 | exothermic | - | [143] | |
| Methylene Blue | magnetic carboxyl functional nanoporous polymer | 298, 308 and 318 | - | endothermic | 52.16–52.58–53.75 | [224] |
4.6. Activation of Solid Sorbent, Surface Modification
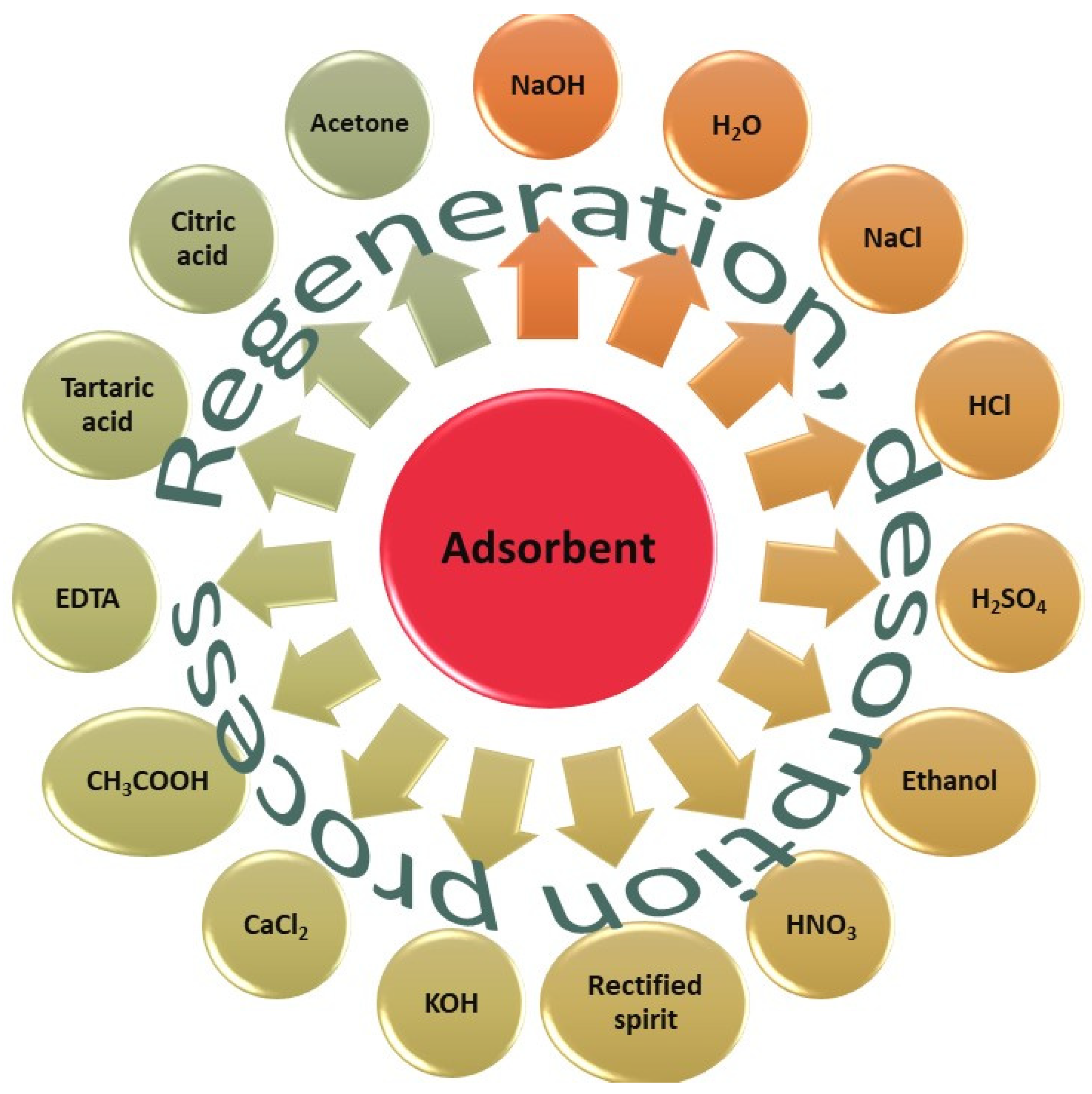
5. Desorption Studies
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tiyasha; Tung, T.M.; Yaseen, Z.M. A Survey on River Water Quality Modelling Using Artificial Intelligence Models: 2000–2020. J. Hydrol. 2020, 585, 124670. [Google Scholar] [CrossRef]
- World Health Organization (WHO) and the United Nations Children’s Fund (UNICEF). Progress on Household Drinking-Water, Sanitation and Hygiene: Five Years into the SDGs; WHO: Geneva, Switzerland, 2021. [Google Scholar]
- Dutta, S.; Gupta, B.; Kumar Srivastava, S.; Kumar Gupta, A. Recent Advances on the Removal of Dyes from Wastewater Using Various Adsorbents: A Critical Review. Mater. Adv. 2021, 2, 4497–4531. [Google Scholar] [CrossRef]
- de Oliveira, M.; Frihling, B.E.F.; Velasques, J.; Filho, F.J.C.M.; Cavalheri, P.S.; Migliolo, L. Pharmaceuticals Residues and Xenobiotics Contaminants: Occurrence, Analytical Techniques and Sustainable Alternatives for Wastewater Treatment. Sci. Total Environ. 2020, 705, 135568. [Google Scholar] [CrossRef] [PubMed]
- Katheresan, V.; Kansedo, J.; Lau, S.Y. Efficiency of Various Recent Wastewater Dye Removal Methods: A Review. J. Environ. Chem. Eng. 2018, 6, 4676–4697. [Google Scholar] [CrossRef]
- De Gisi, S.; Lofrano, G.; Grassi, M.; Notarnicola, M. Characteristics and Adsorption Capacities of Low-Cost Sorbents for Wastewater Treatment: A Review. Sustain. Mater. Technol. 2016, 9, 10–40. [Google Scholar] [CrossRef] [Green Version]
- Bouabidi, Z.B.; El-Naas, M.H.; Cortes, D.; McKay, G. Steel-Making Dust as a Potential Adsorbent for the Removal of Lead (II) from an Aqueous Solution. Chem. Eng. J. 2018, 334, 837–844. [Google Scholar] [CrossRef]
- Kant, R. Textile Dyeing Industry an Environmental Hazard. Nat. Sci. 2011, 4, 22–26. [Google Scholar] [CrossRef] [Green Version]
- Yeow, P.K.; Wong, S.W.; Hadibarata, T. Removal of Azo and Anthraquinone Dye by Plant Biomass as Adsorbent—A Review. Biointerface Res. Appl. Chem. 2020, 11, 8218–8232. [Google Scholar] [CrossRef]
- Ishak, S.A.; Murshed, M.F.; Md Akil, H.; Ismail, N.; Md Rasib, S.Z.; Al-Gheethi, A.A.S. The Application of Modified Natural Polymers in Toxicant Dye Compounds Wastewater: A Review. Water 2020, 12, 2032. [Google Scholar] [CrossRef]
- Jadhav, A.; Jadhav, N. Treatment of textile wastewater using adsorption and adsorbents. In Sustainable Technologies for Textile Wastewater Treatments; Woodhead Publishing: Sawston, UK, 2021; pp. 235–273. ISBN 978-0-323-85829-8. [Google Scholar]
- Sachidhanandham, A.; Periyasamy, A.P. Environmentally Friendly Wastewater Treatment Methods for the Textile Industry. In Handbook of Nanomaterials and Nanocomposites for Energy and Environmental Applications; Springer: Cham, Switzerland, 2020; pp. 1–40. ISBN 978-3-030-11155-7. [Google Scholar]
- ZDHC: Zero Discharge of Hazardous Chemicals Textile Industry Wastewater Discharge Quality Standards. Available online: https://wastewater.sustainabilityconsortium.org/downloads/textile-industry-wastewater-discharge-quality-standards/ (accessed on 22 August 2021).
- Mazharul, I.K. Difference between Dyes and Pigments. Available online: https://textilelearner.net/dyes-and-pigments/ (accessed on 31 July 2021).
- Chequer, F.M.D.; de Oliveira, G.A.R.; Ferraz, E.R.A.; Cardoso, J.C.; Zanoni, M.V.B.; de Oliveira, D.P. Textile Dyes: Dyeing Process and Environmental Impact; IntechOpen: London, UK, 2013; ISBN 978-953-51-0892-4. [Google Scholar]
- Benkhaya, S.; M’rabet, S.; El Harfi, A. A Review on Classifications, Recent Synthesis and Applications of Textile Dyes. Inorg. Chem. Commun. 2020, 115, 107891. [Google Scholar] [CrossRef]
- Chatwal, G.R. Synthetic Dyes; Himalaya Publishing House: Mumbai, India, 2009; ISBN 978-81-8488-220-9. [Google Scholar]
- Society of Dyers and Colourists & AATCC Definitions of a Dye and a Pigment|Colour Index. Available online: https://colour-index.com/definitions-of-a-dye-and-a-pigment (accessed on 31 July 2021).
- IARC Working Group on the Evaluation of Carcinogenic Risk to Humans. General Introduction to the Chemistry of Dyes; International Agency for Research on Cancer: Lyon, France, 2010; ISBN 978-92-832-1299-7. [Google Scholar]
- Gürses, A.; Açıkyıldız, M.; Güneş, K.; Gürses, M.S. Dyes and Pigments; SpringerBriefs in Green Chemistry for Sustainability; Springer International Publishing: Cham, Switzerland, 2016; ISBN 978-3-319-33890-3. [Google Scholar]
- Mahapatra, N.N. Textile Dyes; Woodhead Publishing India PVT. Limited: New Delhi, India, 2016; ISBN 978-93-85059-04-9. [Google Scholar]
- Berradi, M.; Hsissou, R.; Khudhair, M.; Assouag, M.; Cherkaoui, O.; El Bachiri, A.; El Harfi, A. Textile Finishing Dyes and Their Impact on Aquatic Environs. Heliyon 2019, 5, e02711. [Google Scholar] [CrossRef]
- El-Sikaily, A.; Khaled, A.; El Nemr, A. Textile Dyes Xenobiotic and Their Harmful Effect. In Non-Conventional Textile Waste Water Treatment; Nova Science Publishers: New York, NY, USA, 2012; pp. 31–64. ISBN 978-1-62100-079-2. [Google Scholar]
- Letha Malan Oelz Textile Dyeing: Dyeing Fabric… (Textiles—Dyeing Process). Available online: https://www.linkedin.com/pulse/textile-dyeing-fabric-textiles-process-letha-oelz/ (accessed on 26 July 2021).
- Druding, S.C. Dye History from 2600 BC to the 20th Century. Available online: https://www.studypool.com/documents/116420/dye-history-from-2600-bc-to-the-20th-century (accessed on 26 July 2021).
- Nawab, Y.; Ashraf, M.; Hussain, T.; Rasheed, A.; Shaker, K.; Basit, A.; Jabbar, M.; Malik, Z.; Fiaz, H.; Shehzad, K.; et al. Textile Engineering. An Introduction; De Gruyter (Walter de Gruyter): Berlin, Germany, 2016; pp. 143–159. ISBN 978-3-11-041324-3. [Google Scholar]
- Mansour, H. Textile Dyeing: Environmental Friendly Osage Orange Extract on Protein Fabrics. In Eco-Friendly Textile Dyeing and Finishing; Gunay, M., Ed.; IntechOpen Limited: London, UK, 2013; ISBN 978-953-51-0892-4. [Google Scholar]
- Hossain, M.I. Sources and Applications of Natural Dyes. Available online: https://textilefocus.com/sources-applications-natural-dyes/ (accessed on 26 July 2021).
- Kassinger, R. Dyes: From Sea Snails to Synthetics; Lerner Publishing Group, Twenty-First Century Books: Minneapolis, MN, USA, 2003; ISBN 978-0-7613-2112-5. [Google Scholar]
- Hunger, K. (Ed.) Industrial Dyes: Chemistry, Properties, Applications, 1st ed.; Wiley: Weinheim, Germany, 2002; ISBN 978-3-527-30426-4. [Google Scholar]
- Elkady, M.F.; Ibrahim, A.M.; El-Latif, M.M.A. Assessment of the Adsorption Kinetics, Equilibrium and Thermodynamic for the Potential Removal of Reactive Red Dye Using Eggshell Biocomposite Beads. Desalination 2011, 278, 412–423. [Google Scholar] [CrossRef]
- Forgács, E.; Cserháti, T.; Oros, G. Removal of Synthetic Dyes from Wastewaters: A Review. Environ. Int. 2004, 30, 953–971. [Google Scholar] [CrossRef] [PubMed]
- Asif Tahir, M.; Bhatti, H.N.; Iqbal, M. Solar Red and Brittle Blue Direct Dyes Adsorption onto Eucalyptus Angophoroides Bark: Equilibrium, Kinetics and Thermodynamic Studies. J. Environ. Chem. Eng. 2016, 4, 2431–2439. [Google Scholar] [CrossRef]
- Market Data Forecast Ltd. Dyes Market|Size, Share & Trends|2021–2026. Available online: http://www.marketdataforecast.com/ (accessed on 23 July 2021).
- Global Dyes & Pigments Market Size Report, 2021–2028. Available online: https://www.grandviewresearch.com/industry-analysis/dyes-and-pigments-market (accessed on 23 July 2021).
- Statista Research Department Forecast: Industry Revenue of »Manufacture of Dyes and Pigments« in Romania 2011–2023. Available online: https://www.statista.com/forecasts/395518/manufacture-of-dyes-and-pigments-revenue-in-romania (accessed on 23 July 2021).
- Bhardwaj, D.; Bharadvaja, N. Phycoremediation of Effluents Containing Dyes and Its Prospects for Value-Added Products: A Review of Opportunities. J. Water Process Eng. 2021, 41, 102080. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and Its Removal from Aqueous Solution by Adsorption: A Review. Adv. Colloid Interface Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Benkhaya, B.; Harfi, S.E.; Harfi, A.E. Classifications, Properties and Applications of Textile Dyes: A Review. Appl. J. Environ. Eng. Sci. 2017, 3, 311–320. [Google Scholar] [CrossRef]
- Farouk, R.; Gaffer, H.E. Simultaneous Dyeing and Antibacterial Finishing for Cotton Cellulose Using a New Reactive Dye. Carbohydr. Polym. 2013, 97, 138–142. [Google Scholar] [CrossRef]
- Demirbas, A. Agricultural Based Activated Carbons for the Removal of Dyes from Aqueous Solutions: A Review. J. Hazard. Mater. 2009, 167, 1–9. [Google Scholar] [CrossRef]
- Labanda, J.; Sabaté, J.; Llorens, J. Modeling of the Dynamic Adsorption of an Anionic Dye through Ion-Exchange Membrane Adsorber. J. Membr. Sci. 2009, 340, 234–240. [Google Scholar] [CrossRef]
- Prol, A.E.A. Study of Environmental Concerns of Dyes and Recent Textile Effluents Treatment Technology: A Review. Asian J. Fish. Aquat. Res. 2019, 3, 1–18. [Google Scholar] [CrossRef]
- Rachakornkij, M.; Sirawan, R.; Sumate, T. Removal of Reactive Dyes from Aqueous Solution Using Bagasse Fly Ash. Songklanakarin J. Sci. Technol. 2004, 26, 13–24. [Google Scholar]
- Raval, N.P.; Shah, P.U.; Shah, N.K. Malachite Green “a Cationic Dye” and Its Removal from Aqueous Solution by Adsorption. Appl. Water Sci. 2017, 7, 3407–3445. [Google Scholar] [CrossRef] [Green Version]
- Bhate, P.M.; Devi, R.V.; Dugane, R.; Hande, P.R.; Shaikh, L.; Vaidya, S.; Masand, S. A Novel Reactive Dye System Based on Diazonium Salts. Dyes Pigments 2017, 145, 208–215. [Google Scholar] [CrossRef]
- Rys, P.; Zollinger, H. Reactive dye-fibre systems. In Theory of Coloration of Textiles; Dyers Company Publications Trust: Bradford, UK, 1989; p. 564. ISBN 0-901956-48-1. [Google Scholar]
- Li, S.; Zeng, Z.; Xue, W. Kinetic and Equilibrium Study of the Removal of Reactive Dye Using Modified Walnut Shell. Water Sci. Technol. 2019, 80, 874–883. [Google Scholar] [CrossRef] [PubMed]
- Easton, R. The dye maker’s view. In Colour in Dyehouse Effluents; Cooper, P., Ed.; The Aldren Press: Bradford, UK, 1995; pp. 9–21. [Google Scholar]
- Vijayaraghavan, K.; Won, S.W.; Yun, Y.-S. Treatment of Complex Remazol Dye Effluent Using Sawdust- and Coal-Based Activated Carbons. J. Hazard. Mater. 2009, 167, 790–796. [Google Scholar] [CrossRef]
- Rápó, E.; Aradi, L.E.; Szabó, Á.; Posta, K.; Szép, R.; Tonk, S. Adsorption of Remazol Brilliant Violet-5R Textile Dye from Aqueous Solutions by Using Eggshell Waste Biosorbent. Sci. Rep. 2020, 10, 8385. [Google Scholar] [CrossRef]
- Lazaridis, N.K.; Karapantsios, T.D.; Georgantas, D. Kinetic Analysis for the Removal of a Reactive Dye from Aqueous Solution onto Hydrotalcite by Adsorption. Water Res. 2003, 37, 3023–3033. [Google Scholar] [CrossRef]
- Hassaan, M.A.; El Nemr, A.; Madkour, F.F. Testing the Advanced Oxidation Processes on the Degradation of Direct Blue 86 Dye in Wastewater. Egypt. J. Aquat. Res. 2017, 43, 11–19. [Google Scholar] [CrossRef] [Green Version]
- Garg, D.; Majumder, C.B.; Kumar, S.; Sarkar, B. Removal of Direct Blue-86 Dye from Aqueous Solution Using Alginate Encapsulated Activated Carbon (PnsAC-Alginate) Prepared from Waste Peanut Shell. J. Environ. Chem. Eng. 2019, 7, 103365. [Google Scholar] [CrossRef]
- Irshad, S.; Sultana, H.; Usman, M.; Saeed, M.; Akram, N.; Yusaf, A.; Rehman, A. Solubilization of Direct Dyes in Single and Mixed Surfactant System: A Comparative Study. J. Mol. Liq. 2021, 321, 114201. [Google Scholar] [CrossRef]
- Horng, J.Y.; Huang, S.D. Removal of Organic Dye (Direct Blue) from Synthetic Wastewater by Adsorptive Bubble Separation Techniques. Environ. Sci. Technol. 1993, 27, 1169–1175. [Google Scholar] [CrossRef]
- Burkinshaw, S.M.; Salihu, G. The Role of Auxiliaries in the Immersion Dyeing of Textile Fibres: Part 6 Analysis of Conventional Models That Describe the Manner by Which Inorganic Electrolytes Promote Reactive Dye Uptake on Cellulosic Fibres. Dyes Pigments 2019, 161, 595–604. [Google Scholar] [CrossRef]
- Burkinshaw, S.M. Chemical Principles of Synthetic Fibre Dyeing; Springer: Dordrecht, The Netherlands, 1995; ISBN 978-0-7514-0043-4. [Google Scholar]
- Yao, L.; Yang, J.; Zhang, P.; Deng, L. In Situ Surface Decoration of Fe3C/Fe3O4/C Nanosheets: Towards Bi-Functional Activated Carbons with Supercapacitance and Efficient Dye Adsorption. Bioresour. Technol. 2018, 256, 208–215. [Google Scholar] [CrossRef]
- Wu, J.; Li, Q.; Li, W.; Li, Y.; Wang, G.; Li, A.; Li, H. Efficient Removal of Acid Dyes Using Permanent Magnetic Resin and Its Preliminary Investigation for Advanced Treatment of Dyeing Effluents. J. Clean. Prod. 2020, 251, 119694. [Google Scholar] [CrossRef]
- Patil, S.; Renukdas, S.; Patel, N. Removal of Methylene Blue, a Basic Dye from Aqueous Solutions by Adsorption Using Teak Tree (Tectona grandis) Bark Powder. Int. J. Environ. Sci. 2011, 1, 711–726. [Google Scholar]
- Dai, L.; Zhu, W.; He, L.; Tan, F.; Zhu, N.; Zhou, Q.; He, M.; Hu, G. Calcium-Rich Biochar from Crab Shell: An Unexpected Super Adsorbent for Dye Removal. Bioresour. Technol. 2018, 267, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Kyzas, G.Z.; Kostoglou, M.; Lazaridis, N.K.; Bikiaris, D.N. Decolorization of Dyeing Wastewater Using Polymeric Absorbents—An Overview; IntechOpen: London, UK, 2013; ISBN 978-953-51-0892-4. [Google Scholar]
- Morais da Silva, P.M.; Camparotto, N.G.; Grego Lira, K.T.; Franco Picone, C.S.; Prediger, P. Adsorptive Removal of Basic Dye onto Sustainable Chitosan Beads: Equilibrium, Kinetics, Stability, Continuous-Mode Adsorption and Mechanism. Sustain. Chem. Pharm. 2020, 18, 100318. [Google Scholar] [CrossRef]
- Bayram, E.; Ayranci, E. Investigation of Changes in Properties of Activated Carbon Cloth upon Polarization and of Electrosorption of the Dye Basic Blue-7. Carbon 2010, 48, 1718–1730. [Google Scholar] [CrossRef]
- Vikrant, K.; Giri, B.S.; Raza, N.; Roy, K.; Kim, K.-H.; Rai, B.N.; Singh, R.S. Recent Advancements in Bioremediation of Dye: Current Status and Challenges. Bioresour. Technol. 2018, 253, 355–367. [Google Scholar] [CrossRef]
- Benvenuti, J.; Fisch, A.; dos Santos, J.H.Z.; Gutterres, M. Silica-Based Adsorbent Material with Grape Bagasse Encapsulated by the Sol-Gel Method for the Adsorption of Basic Blue 41 Dye. J. Environ. Chem. Eng. 2019, 7, 103342. [Google Scholar] [CrossRef]
- Kausar, A.; Iqbal, M.; Javed, A.; Aftab, K.; Nazli, Z.-H.; Bhatti, H.N.; Nouren, S. Dyes Adsorption Using Clay and Modified Clay: A Review. J. Mol. Liq. 2018, 256, 395–407. [Google Scholar] [CrossRef]
- Liu, J.; Wang, N.; Zhang, H.; Baeyens, J. Adsorption of Congo Red Dye on FexCo3-XO4 Nanoparticles. J. Environ. Manag. 2019, 238, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Chavan, R.B. Environmentally friendly dyes. In Handbook of Textile and Industrial Dyeing; Clark, M., Ed.; Woodhead Publishing Series in Textiles; Woodhead Publishing: Sawaston, UK, 2011; Chapter 16; Volume 1, pp. 515–561. ISBN 978-1-84569-695-5. [Google Scholar]
- Cie, C. (Ed.) Fixing ink jet printed textiles. In Ink Jet Textile Printing; Woodhead Publishing Series in Textiles; Woodhead Publishing: Sawaston, UK, 2015; Chapter 8; pp. 99–110. ISBN 978-0-85709-230-4. [Google Scholar]
- Shamey, R. Improving the colouration/dyeability of polyolefin fibres. In Polyolefin Fibres; Ugbolue, S.C.O., Ed.; Woodhead Publishing Series in Textiles; Woodhead Publishing: Sawaston, UK, 2009; Chapter 12; pp. 363–397. ISBN 978-1-84569-207-0. [Google Scholar]
- Clark, M. Fundamental Principles of Dyeing; Woodhead Publishing: Sawaston, UK, 2011; Volume 1, pp. 3–27. ISBN 978-1-84569-695-5. [Google Scholar]
- Qin, Y.; Yuan, M.; Hu, Y.; Lu, Y.; Lin, W.; Ma, Y.; Lin, X.; Wang, T. Preparation and Interaction Mechanism of Nano Disperse Dye Using Hydroxypropyl Sulfonated Lignin. Int. J. Biol. Macromol. 2020, 152, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Qayyum, S.; Nasir, A.; Mian, A.H.; Rehman, S.; Qayum, S.; Siddiqui, M.F.; Kalsoom, U. Extraction of Peroxidase Enzyme from Different Vegetables for Biodetoxification of Vat Dyes. Appl. Nanosci. 2020, 10, 5191–5199. [Google Scholar] [CrossRef]
- Božič, M.; Kokol, V. Ecological Alternatives to the Reduction and Oxidation Processes in Dyeing with Vat and Sulphur Dyes. Dyes Pigments 2008, 76, 299–309. [Google Scholar] [CrossRef]
- Zhang, T.; Fei, X.; Wang, S.; Zhou, C. Pigmentation of Vat Blue RS by Ball Milling in Solvents. Dyes Pigments 2000, 45, 15–21. [Google Scholar] [CrossRef]
- Balan, D.S.L.; Monteiro, R.T.R. Decolorization of Textile Indigo Dye by Ligninolytic Fungi. J. Biotechnol. 2001, 89, 141–145. [Google Scholar] [CrossRef]
- Chaari, I.; Medhioub, M.; Jamoussi, F.; Hamzaoui, A.H. Acid-Treated Clay Materials (Southwestern Tunisia) for Removing Sodium Leuco-Vat Dye: Characterization, Adsorption Study and Activation Mechanism. J. Mol. Struct. 2021, 1223, 128944. [Google Scholar] [CrossRef]
- Hessel, C.; Allegre, C.; Maisseu, M.; Charbit, F.; Moulin, P. Guidelines and Legislation for Dye House Effluents. J. Environ. Manag. 2007, 83, 171–180. [Google Scholar] [CrossRef]
- Singh, P.K.; Singh, R.L. Bio-Removal of Azo Dyes: A Review. Int. J. Appl. Sci. Biotechnol. 2017, 5, 108–126. [Google Scholar] [CrossRef]
- Bhatia, D.; Sharma, N.R.; Singh, J.; Kanwar, R.S. Biological Methods for Textile Dye Removal from Wastewater: A Review. Crit. Rev. Environ. Sci. Technol. 2017, 47, 1836–1876. [Google Scholar] [CrossRef]
- Madhav, S.; Ahamad, A.; Singh, P.; Mishra, P.K. A Review of Textile Industry: Wet Processing, Environmental Impacts, and Effluent Treatment Methods. Environ. Qual. Manag. 2018, 27, 31–41. [Google Scholar] [CrossRef]
- Yaseen, D.A.; Scholz, M. Textile Dye Wastewater Characteristics and Constituents of Synthetic Effluents: A Critical Review. Int. J. Environ. Sci. Technol. 2019, 16, 1193–1226. [Google Scholar] [CrossRef] [Green Version]
- Pavithra, K.G.; Jaikumar, V. Removal of Colorants from Wastewater: A Review on Sources and Treatment Strategies. J. Ind. Eng. Chem. 2019, 75, 1–19. [Google Scholar] [CrossRef]
- Zhou, Y.; Lu, J.; Zhou, Y.; Liu, Y. Recent Advances for Dyes Removal Using Novel Adsorbents: A Review. Environ. Pollut. 2019, 252, 352–365. [Google Scholar] [CrossRef]
- Beulah, S.S.; Muthukumaran, K. Methodologies of Removal of Dyes from Wastewater: A Review. Int. Res. J. Pure Appl. Chem. 2020, 21, 68–78. [Google Scholar] [CrossRef]
- Donkadokula, N.Y.; Kola, A.K.; Naz, I.; Saroj, D. A Review on Advanced Physico-Chemical and Biological Textile Dye Wastewater Treatment Techniques. Rev. Environ. Sci. Biotechnol. 2020, 19, 543–560. [Google Scholar] [CrossRef]
- Samsami, S.; Mohamadizaniani, M.; Sarrafzadeh, M.-H.; Rene, E.R.; Firoozbahr, M. Recent Advances in the Treatment of Dye-Containing Wastewater from Textile Industries: Overview and Perspectives. Process Saf. Environ. Prot. 2020, 143, 138–163. [Google Scholar] [CrossRef]
- Ihsanullah, I.; Jamal, A.; Ilyas, M.; Zubair, M.; Khan, G.; Atieh, M.A. Bioremediation of Dyes: Current Status and Prospects. J. Water Process Eng. 2020, 38, 101680. [Google Scholar] [CrossRef]
- Selvaraj, V.; Swarna Karthika, T.; Mansiya, C.; Alagar, M. An over Review on Recently Developed Techniques, Mechanisms and Intermediate Involved in the Advanced Azo Dye Degradation for Industrial Applications. J. Mol. Struct. 2021, 1224, 129195. [Google Scholar] [CrossRef]
- Slama, H.B.; Chenari Bouket, A.; Pourhassan, Z.; Alenezi, F.N.; Silini, A.; Cherif-Silini, H.; Oszako, T.; Luptakova, L.; Golińska, P.; Belbahri, L. Diversity of Synthetic Dyes from Textile Industries, Discharge Impacts and Treatment Methods. Appl. Sci. 2021, 11, 6255. [Google Scholar] [CrossRef]
- Mashkoor, F.; Nasar, A. Environmental Application of Agro-Waste Derived Materials for the Treatment of Dye-Polluted Water: A Review. Curr. Anal. Chem. 2021, 17, 904–916. [Google Scholar] [CrossRef]
- Crini, G. Non-Conventional Low-Cost Adsorbents for Dye Removal: A Review. Bioresour. Technol. 2006, 97, 1061–1085. [Google Scholar] [CrossRef]
- Chikri, R.; Elhadiri, N.; Benchanaa, M.; El Maguana, Y. Efficiency of Sawdust as Low-Cost Adsorbent for Dyes Removal. J. Chem. 2020, 2020, e8813420. [Google Scholar] [CrossRef]
- Semeraro, P.; Gabaldón, J.A.; Fini, P.; Núňez, E.; Pellicer, J.A.; Rizzi, V.; Cosma, P. Removal of an Azo Textile Dye from Wastewater by Cyclodextrin-Epichlorohydrin Polymers; IntechOpen: London, UK, 2017; ISBN 978-1-78923-069-7. [Google Scholar]
- Choudhary, A. Removal of Oil from Seawater Using Charcoal and Rice Hull. IOP Conf. Ser. Mater. Sci. Eng. 2017, 263, 032007. [Google Scholar] [CrossRef]
- Tan, K.L.; Hameed, B.H. Insight into the Adsorption Kinetics Models for the Removal of Contaminants from Aqueous Solutions. J. Taiwan Inst. Chem. Eng. 2017, 74, 25–48. [Google Scholar] [CrossRef]
- Abebe, B.; Murthy, H.C.A.; Amare, E. Summary on Adsorption and Photocatalysis for Pollutant Remediation: Mini Review. J. Encapsul. Adsorpt. Sci. 2018, 8, 225–255. [Google Scholar] [CrossRef] [Green Version]
- Qi, Y.; Zhu, J.; Fu, Q.; Hu, H.; Huang, Q. Sorption of Cu by Humic Acid from the Decomposition of Rice Straw in the Absence and Presence of Clay Minerals. J. Environ. Manag. 2017, 200, 304–311. [Google Scholar] [CrossRef] [PubMed]
- Sims, R.A.; Harmer, S.L.; Quinton, J.S. The Role of Physisorption and Chemisorption in the Oscillatory Adsorption of Organosilanes on Aluminium Oxide. Polymers 2019, 11, 410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Ghouti, M.A.; Da’ana, D.A. Guidelines for the Use and Interpretation of Adsorption Isotherm Models: A Review. J. Hazard. Mater. 2020, 393, 122383. [Google Scholar] [CrossRef]
- Ruthven, D.M. Principles of Adsorption and Adsorption Processes; John Wiley & Sons: New York, NY, USA, 1984; ISBN 978-0-471-86606-0. [Google Scholar]
- Kamaya Parashar Adsorption, Presentation at University of Johannesburg. 2015. Available online: https://www.slideshare.net/Kamyaparashar/adsorption-presentation-44669901 (accessed on 3 September 2021).
- Terry, P.A.; Noble, R.D. (Eds.) Adsorption. In Principles of Chemical Separations with Environmental Applications; Cambridge Series in Chemical Engineering; Cambridge University Press: Cambridge, UK, 2004; pp. 182–213. ISBN 978-0-511-61659-4. [Google Scholar]
- Guo, X.; Wang, J. A General Kinetic Model for Adsorption: Theoretical Analysis and Modeling. J. Mol. Liq. 2019, 288, 111100. [Google Scholar] [CrossRef]
- Adsorption Techniques|EMIS. Available online: https://emis.vito.be/en/bat/tools-overview/sheets/adsorption-techniques (accessed on 28 July 2021).
- Factors Affecting Adsorption: Temperature, Pressure, Nature of Adsorbent. The Fact Factor, 3 April 2020.
- Factors Affecting Adsorption. Available online: http://www.brainkart.com/article/Factors-Affecting-Adsorption_35881/ (accessed on 28 July 2021).
- Szende, T.; Eszter, R. Környezeti Szennyezők, Környezeti Problémák, Környezeti Remediáció, 1st ed.; EXIT Kiadó: Cluj Napoca, Romania, 2020; ISBN 978-606-9091-23-4. [Google Scholar]
- Lai, K.C.; Lee, L.Y.; Hiew, B.Y.Z.; Thangalazhy-Gopakumar, S.; Gan, S. Environmental Application of Three-Dimensional Graphene Materials as Adsorbents for Dyes and Heavy Metals: Review on Ice-Templating Method and Adsorption Mechanisms. J. Environ. Sci. 2019, 79, 174–199. [Google Scholar] [CrossRef]
- Mok, C.F.; Ching, Y.C.; Muhamad, F.; Abu Osman, N.A.; Hai, N.D.; Che Hassan, C.R. Adsorption of Dyes Using Poly(Vinyl Alcohol) (PVA) and PVA-Based Polymer Composite Adsorbents: A Review. J. Polym. Environ. 2020, 28, 775–793. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Bikiaris, D.N.; Mitropoulos, A.C. Chitosan Adsorbents for Dye Removal: A Review. Polym. Int. 2017, 66, 1800–1811. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Deliyanni, E.A.; Bikiaris, D.N.; Mitropoulos, A.C. Graphene Composites as Dye Adsorbents: Review. Chem. Eng. Res. Des. 2018, 129, 75–88. [Google Scholar] [CrossRef]
- Afroze, S.; Sen, T.K. A Review on Heavy Metal Ions and Dye Adsorption from Water by Agricultural Solid Waste Adsorbents. Water Air Soil Pollut. 2018, 229, 225. [Google Scholar] [CrossRef]
- Hynes, N.R.J.; Kumar, J.S.; Kamyab, H.; Sujana, J.A.J.; Al-Khashman, O.A.; Kuslu, Y.; Ene, A.; Suresh Kumar, B. Modern Enabling Techniques and Adsorbents Based Dye Removal with Sustainability Concerns in Textile Industrial Sector—A Comprehensive Review. J. Clean. Prod. 2020, 272, 122636. [Google Scholar] [CrossRef]
- Nasar, A.; Mashkoor, F. Application of Polyaniline-Based Adsorbents for Dye Removal from Water and Wastewater—A Review. Environ. Sci. Pollut. Res. 2019, 26, 5333–5356. [Google Scholar] [CrossRef]
- Homaeigohar, S. The Nanosized Dye Adsorbents for Water Treatment. Nanomaterials 2020, 10, 295. [Google Scholar] [CrossRef] [Green Version]
- Nayeri, D.; Mousavi, S.A. Dye Removal from Water and Wastewater by Nanosized Metal Oxides—Modified Activated Carbon: A Review on Recent Researches. J. Environ. Health Sci. Eng. 2020, 18, 1671–1689. [Google Scholar] [CrossRef]
- Aruna; Bagotia, N.; Sharma, A.K.; Kumar, S. A Review on Modified Sugarcane Bagasse Biosorbent for Removal of Dyes. Chemosphere 2021, 268, 129309. [Google Scholar] [CrossRef] [PubMed]
- Pai, S.; Kini, M.S.; Selvaraj, R. A Review on Adsorptive Removal of Dyes from Wastewater by Hydroxyapatite Nanocomposites. Environ. Sci. Pollut. Res. 2021, 28, 11835–11849. [Google Scholar] [CrossRef]
- Bulgariu, L.; Escudero, L.B.; Bello, O.S.; Iqbal, M.; Nisar, J.; Adegoke, K.A.; Alakhras, F.; Kornaros, M.; Anastopoulos, I. The Utilization of Leaf-Based Adsorbents for Dyes Removal: A Review. J. Mol. Liq. 2019, 276, 728–747. [Google Scholar] [CrossRef] [Green Version]
- Chakraborty, R.; Asthana, A.; Singh, A.K.; Jain, B.; Susan, A.B.H. Adsorption of Heavy Metal Ions by Various Low-Cost Adsorbents: A Review. Int. J. Environ. Anal. Chem. 2020, 1–38. [Google Scholar] [CrossRef]
- Pourhakkak, P.; Taghizadeh, A.; Taghizadeh, M.; Ghaedi, M.; Haghdoust, S. Fundamentals of adsorption technology. In Interface Science and Technology; Ghaedi, M., Ed.; Adsorption: Fundamental Processes and Applications; Elsevier: London, UK, 2021; Chapter 1; Volume 33, pp. 1–70. [Google Scholar]
- Wang, Q.; Wang, Y.; Chen, L. A Green Composite Hydrogel Based on Cellulose and Clay as Efficient Absorbent of Colored Organic Effluent. Carbohydr. Polym. 2019, 210, 314–321. [Google Scholar] [CrossRef]
- Ngulube, T.; Gumbo, J.R.; Masindi, V.; Maity, A. An Update on Synthetic Dyes Adsorption onto Clay Based Minerals: A State-of-Art Review. J. Environ. Manag. 2017, 191, 35–57. [Google Scholar] [CrossRef] [PubMed]
- Brião, G.V.; Jahn, S.L.; Foletto, E.L.; Dotto, G.L. Highly Efficient and Reusable Mesoporous Zeolite Synthetized from a Biopolymer for Cationic Dyes Adsorption. Colloids Surf. A Physicochem. Eng. Asp. 2018, 556, 43–50. [Google Scholar] [CrossRef]
- Lipatova, I.M.; Makarova, L.I.; Yusova, A.A. Adsorption Removal of Anionic Dyes from Aqueous Solutions by Chitosan Nanoparticles Deposited on the Fibrous Carrier. Chemosphere 2018, 212, 1155–1162. [Google Scholar] [CrossRef]
- Murcia-Salvador, A.; Pellicer, J.A.; Fortea, M.I.; Gómez-López, V.M.; Rodríguez-López, M.I.; Núñez-Delicado, E.; Gabaldón, J.A. Adsorption of Direct Blue 78 Using Chitosan and Cyclodextrins as Adsorbents. Polymers 2019, 11, 1003. [Google Scholar] [CrossRef] [Green Version]
- Vahedi, S.; Tavakoli, O.; Khoobi, M.; Ansari, A.; Ali Faramarzi, M. Application of Novel Magnetic β-Cyclodextrin-Anhydride Polymer Nano-Adsorbent in Cationic Dye Removal from Aqueous Solution. J. Taiwan Inst. Chem. Eng. 2017, 80, 452–463. [Google Scholar] [CrossRef]
- Pellicer, J.A.; Rodríguez-López, M.I.; Fortea, M.I.; Lucas-Abellán, C.; Mercader-Ros, M.T.; López-Miranda, S.; Gómez-López, V.M.; Semeraro, P.; Cosma, P.; Fini, P.; et al. Adsorption Properties of β- and Hydroxypropyl-β-Cyclodextrins Cross-Linked with Epichlorohydrin in Aqueous Solution. A Sustainable Recycling Strategy in Textile Dyeing Process. Polymers 2019, 11, 252. [Google Scholar] [CrossRef] [Green Version]
- Pellicer, J.; Rodriguez López, M.I.; Fortea, M.; Gabaldon, J.; Lucas-Abellán, C.; Mercader Ros, M.; Serrano Martínez, A.; Núñez-Delicado, E.; Cosma, P.; Fini, P.; et al. Removing of Direct Red 83:1 Using α- and HP-α-CDs Polymerized with Epichlorohydrin: Kinetic and Equilibrium Studies. Dyes Pigments 2017, 149, 736–746. [Google Scholar] [CrossRef]
- Rápó, E.; Szép, R.; Keresztesi, Á.; Suciu, M.; Tonk, S. Adsorptive Removal of Cationic and Anionic Dyes from Aqueous Solutions by Using Eggshell Household Waste as Biosorbent. Acta Chim. Slov. 2018, 65, 709–717. [Google Scholar] [CrossRef]
- Rápó, E.; Posta, K.; Suciu, M.; Szép, R.; Tonk, S. Adsorptive Removal of Remazol Brilliant Violet-5R Dye from Aqueous Solutions Using Calcined Eggshell as Biosorbent. Acta Chim. Slov. 2019, 66, 648–658. [Google Scholar] [CrossRef] [PubMed]
- Rápó, E.; Jakab, K.; Posta, K.; Suciu, M.; Tonk, S. A Comparative Study on the Adsorption of Two Remazol Dyes on Green Adsorbent. Rev. Chim. 2020, 71, 248–257. [Google Scholar] [CrossRef]
- Ahmed, M.; Mashkoor, F.; Nasar, A. Development, Characterization, and Utilization of Magnetized Orange Peel Waste as a Novel Adsorbent for the Confiscation of Crystal Violet Dye from Aqueous Solution. Groundw. Sustain. Dev. 2020, 10, 100322. [Google Scholar] [CrossRef]
- Mokhtari, N.; Afshari, M.; Dinari, M. Synthesis and Characterization of a Novel Fluorene-Based Covalent Triazine Framework as a Chemical Adsorbent for Highly Efficient Dye Removal. Polymer 2020, 195, 122430. [Google Scholar] [CrossRef]
- Maleš, L.; Fakin, D.; Bračič, M.; Gorgieva, S. Efficiency of Differently Processed Membranes Based on Cellulose as Cationic Dye Adsorbents. Nanomaterials 2020, 10, 642. [Google Scholar] [CrossRef] [Green Version]
- Khamis, M.I.; Ibrahim, T.H.; Jumean, F.H.; Sara, Z.A.; Atallah, B.A. Cyclic Sequential Removal of Alizarin Red S Dye and Cr(VI) Ions Using Wool as a Low-Cost Adsorbent. Processes 2020, 8, 556. [Google Scholar] [CrossRef]
- Doan, C.T.; Tran, T.N.; Wang, C.-L.; Wang, S.-L. Microbial Conversion of Shrimp Heads to Proteases and Chitin as an Effective Dye Adsorbent. Polymers 2020, 12, 2228. [Google Scholar] [CrossRef] [PubMed]
- Hong, G.-B.; Yu, T.-J.; Lee, H.-C.; Ma, C.-M. Using Rice Bran Hydrogel Beads to Remove Dye from Aqueous Solutions. Sustainability 2021, 13, 5640. [Google Scholar] [CrossRef]
- Pham, T.D.; Bui, V.P.; Pham, T.N.; Le, T.M.D.; Nguyen, K.T.; Bui, V.H.; Nguyen, T.D. Adsorptive Removal of Anionic Azo Dye New Coccine Using Silica and Silica-Gel with Surface Modification by Polycation. Polymers 2021, 13, 1536. [Google Scholar] [CrossRef]
- Alghamdi, W.M.; El Mannoubi, I. Investigation of Seeds and Peels of Citrullus Colocynthis as Efficient Natural Adsorbent for Methylene Blue Dye. Processes 2021, 9, 1279. [Google Scholar] [CrossRef]
- Mansouri, F.E.; Farissi, H.E.; Zerrouk, M.H.; Cacciola, F.; Bakkali, C.; Brigui, J.; Lovillo, M.P.; Esteves da Silva, J.C.G. Dye Removal from Colored Textile Wastewater Using Seeds and Biochar of Barley (Hordeum vulgare L.). Appl. Sci. 2021, 11, 5125. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E.; Wilson, L.D.; Morin-Crini, N. Conventional and Non-Conventional Adsorbents for Wastewater Treatment. Environ. Chem. Lett. 2019, 17, 195–213. [Google Scholar] [CrossRef]
- Razi, M.A.M.; Hishammudin, M.N.A.M.; Hamdan, R. Factor Affecting Textile Dye Removal Using Adsorbent From Activated Carbon: A Review. MATEC Web Conf. 2017, 103, 06015. [Google Scholar] [CrossRef] [Green Version]
- Yu, J.; Zhang, X.; Wang, D.; Li, P. Adsorption of Methyl Orange Dye onto Biochar Adsorbent Prepared from Chicken Manure. Water Sci. Technol. 2018, 77, 1303–1312. [Google Scholar] [CrossRef]
- Harrache, Z.; Abbas, M.; Aksil, T.; Trari, M. Thermodynamic and Kinetics Studies on Adsorption of Indigo Carmine from Aqueous Solution by Activated Carbon. Microchem. J. 2019, 144, 180–189. [Google Scholar] [CrossRef]
- Muinde, V.; Onyari, J.M.; Wamalwa, B.M.; Wabomba, J. Adsorption of Malachite Green From Aqueous Solutions Onto Rice Husks: Kinetic and Equilibrium Studies. J. Environ. Prot. 2017, 8, 215–230. [Google Scholar] [CrossRef] [Green Version]
- Idan, I.J.; Malaysia, U.P.; Malaysia, U.P.; Malaysia, U.P. Adsorption of Anionic Dye Using Cationic Surfactant-Modified Kenaf Core Fibers. Open Access Libr. J. 2017, 4, 1. [Google Scholar] [CrossRef]
- Hii, H.T. Adsorption Isotherm And Kinetic Models For Removal Of Methyl Orange And Remazol Brilliant Blue R By Coconut Shell Activated Carbon. Trop. Aquat. Soil Pollut. 2021, 1, 1–10. [Google Scholar] [CrossRef]
- Munagapati, V.S.; Wen, J.-C.; Pan, C.-L.; Gutha, Y.; Wen, J.-H.; Reddy, G.M. Adsorptive Removal of Anionic Dye (Reactive Black 5) from Aqueous Solution Using Chemically Modified Banana Peel Powder: Kinetic, Isotherm, Thermodynamic, and Reusability Studies. Int. J. Phytoremed. 2020, 22, 267–278. [Google Scholar] [CrossRef]
- Legorreta-Castañeda, A.J.; Lucho-Constantino, C.A.; Beltrán-Hernández, R.I.; Coronel-Olivares, C.; Vázquez-Rodríguez, G.A. Biosorption of Water Pollutants by Fungal Pellets. Water 2020, 12, 1155. [Google Scholar] [CrossRef] [Green Version]
- Terangpi, P.; Chakraborty, S. Adsorption Kinetics and Equilibrium Studies for Removal of Acid Azo Dyes by Aniline Formaldehyde Condensate. Appl. Water Sci. 2017, 7, 3661–3671. [Google Scholar] [CrossRef] [Green Version]
- Dali Youcef, L.; Belaroui, L.S.; López-Galindo, A. Adsorption of a Cationic Methylene Blue Dye on an Algerian Palygorskite. Appl. Clay Sci. 2019, 179, 105145. [Google Scholar] [CrossRef]
- Mahmoudi, M.M.; Nadali, A.; Arezoomand, H.R.S.; Mahvi, A.H. Adsorption of Cationic Dye Textile Wastewater Using Clinoptilolite: Isotherm and Kinetic Study. J. Text. Inst. 2019, 110, 74–80. [Google Scholar] [CrossRef]
- Abbas, M. Removal of Brilliant Green (BG) by Activated Carbon Derived from Medlar Nucleus (ACMN)—Kinetic, Isotherms and Thermodynamic Aspects of Adsorption. Adsorpt. Sci. Technol. 2020, 38, 464–482. [Google Scholar] [CrossRef]
- Al-Ghouti, M.A.; Al-Absi, R.S. Mechanistic Understanding of the Adsorption and Thermodynamic Aspects of Cationic Methylene Blue Dye onto Cellulosic Olive Stones Biomass from Wastewater. Sci. Rep. 2020, 10, 15928. [Google Scholar] [CrossRef]
- Hassan, W.; Noureen, S.; Mustaqeem, M.; Saleh, T.A.; Zafar, S. Efficient Adsorbent Derived from Haloxylon Recurvum Plant for the Adsorption of Acid Brown Dye: Kinetics, Isotherm and Thermodynamic Optimization. Surf. Interfaces 2020, 20, 100510. [Google Scholar] [CrossRef]
- Rodríguez-Arellano, G.; Barajas-Fernández, J.; García-Alamilla, R.; Lagunes-Gálvez, L.M.; Lara-Rivera, A.H.; García-Alamilla, P. Evaluation of Cocoa Beans Shell Powder as a Bioadsorbent of Congo Red Dye Aqueous Solutions. Materials 2021, 14, 2763. [Google Scholar] [CrossRef]
- Bayomie, O.S.; Kandeel, H.; Shoeib, T.; Yang, H.; Youssef, N.; El-Sayed, M.M.H. Novel Approach for Effective Removal of Methylene Blue Dye from Water Using Fava Bean Peel Waste. Sci. Rep. 2020, 10, 7824. [Google Scholar] [CrossRef]
- Değermenci, G.D.; Değermenci, N.; Ayvaoğlu, V.; Durmaz, E.; Çakır, D.; Akan, E. Adsorption of Reactive Dyes on Lignocellulosic Waste; Characterization, Equilibrium, Kinetic and Thermodynamic Studies. J. Clean. Prod. 2019, 225, 1220–1229. [Google Scholar] [CrossRef]
- Wong, S.; Tumari, H.H.; Ngadi, N.; Mohamed, N.B.; Hassan, O.; Mat, R.; Saidina Amin, N.A. Adsorption of Anionic Dyes on Spent Tea Leaves Modified with Polyethyleneimine (PEI-STL). J. Clean. Prod. 2019, 206, 394–406. [Google Scholar] [CrossRef]
- Singh, H.; Chauhan, G.; Jain, A.K.; Sharma, S.K. Adsorptive Potential of Agricultural Wastes for Removal of Dyes from Aqueous Solutions. J. Environ. Chem. Eng. 2017, 5, 122–135. [Google Scholar] [CrossRef]
- Shoukat, S.; Bhatti, H.N.; Iqbal, M.; Noreen, S. Mango Stone Biocomposite Preparation and Application for Crystal Violet Adsorption: A Mechanistic Study. Microporous Mesoporous Mater. 2017, 239, 180–189. [Google Scholar] [CrossRef]
- Ma, H.; Kong, A.; Ji, Y.; He, B.; Song, Y.; Li, J. Ultrahigh Adsorption Capacities for Anionic and Cationic Dyes from Wastewater Using Only Chitosan. J. Clean. Prod. 2019, 214, 89–94. [Google Scholar] [CrossRef]
- Çelekli, A.; Al-Nuaimi, A.I.; Bozkurt, H. Adsorption Kinetic and Isotherms of Reactive Red 120 on Moringa Oleifera Seed as an Eco-Friendly Process. J. Mol. Struct. 2019, 1195, 168–178. [Google Scholar] [CrossRef]
- Elsherif, K.; El-Dali, A.; Alkarewi, A.; Ewlad-Ahmed, A.; Treban, A. Adsorption of Crystal Violet Dye Onto Olive Leaves Powder: Equilibrium and Kinetic Studies. Chem. Int. 2021, 7, 79–89. [Google Scholar] [CrossRef]
- Seow, T.W.; Lim, C.K. Removal of Dye by Adsorption: A Review. Int. J. Appl. Eng. Res. 2016, 11, 2675–2679. [Google Scholar]
- Tejada-Tovar, C.; Villabona-Ortíz, Á.; Gonzalez-Delgado, Á.D. Adsorption of Azo-Anionic Dyes in a Solution Using Modified Coconut (Cocos nucifera) Mesocarp: Kinetic and Equilibrium Study. Water 2021, 13, 1382. [Google Scholar] [CrossRef]
- Mondal, N.K.; Kar, S. Potentiality of Banana Peel for Removal of Congo Red Dye from Aqueous Solution: Isotherm, Kinetics and Thermodynamics Studies. Appl. Water Sci. 2018, 8, 157. [Google Scholar] [CrossRef] [Green Version]
- de Farias Silva, C.E.; da Gama, B.M.; da Silva Gonçalves, A.H.; Medeiros, J.A.; de Souza Abud, A.K. Basic-Dye Adsorption in Albedo Residue: Effect of PH, Contact Time, Temperature, Dye Concentration, Biomass Dosage, Rotation and Ionic Strength. J. King Saud Univ.-Eng. Sci. 2020, 32, 351–359. [Google Scholar] [CrossRef]
- Brito, M.J.; Veloso, C.M.; Santos, L.S.; Bonomo, R.C.; Fontan, R.D. Adsorption of the Textile Dye Dianix® Royal Blue CC onto Carbons Obtained from Yellow Mombin Fruit Stones and Activated with KOH and H3PO4: Kinetics, Adsorption Equilibrium and Thermodynamic Studies. Powder Technol. 2018, 339, 334–343. [Google Scholar] [CrossRef]
- Gamoudi, S.; Srasra, E. Adsorption of Organic Dyes by HDPy+-Modified Clay: Effect of Molecular Structure on the Adsorption. J. Mol. Struct. 2019, 1193, 522–531. [Google Scholar] [CrossRef]
- Kanwal, A.; Bhatti, H.N.; Iqbal, M.; Noreen, S. Basic Dye Adsorption onto Clay/MnFe2O4 Composite: A Mechanistic Study. Water Environ. Res. 2017, 89, 301–311. [Google Scholar] [CrossRef]
- Yildirim, A. Removal of the Anionic Dye Reactive Orange 16 by Chitosan/Tripolyphosphate/Mushroom. Chem. Eng. Technol. 2021, 44, 1371–1381. [Google Scholar] [CrossRef]
- Khasri, A.; Jamir, M.R.M.; Ahmad, A.A.; Ahmad, M.A. Adsorption of Remazol Brilliant Violet 5R Dye from Aqueous Solution onto Melunak and Rubberwood Sawdust Based Activated Carbon: Interaction Mechanism, Isotherm, Kinetic and Thermodynamic Properties. DWT 2021, 216, 401–411. [Google Scholar] [CrossRef]
- Alhujaily, A.; Yu, H.; Zhang, X.; Ma, F. Adsorptive Removal of Anionic Dyes from Aqueous Solutions Using Spent Mushroom Waste. Appl. Water Sci. 2020, 10, 183. [Google Scholar] [CrossRef]
- Nizam, N.U.M.; Hanafiah, M.M.; Mahmoudi, E.; Halim, A.A.; Mohammad, A.W. The Removal of Anionic and Cationic Dyes from an Aqueous Solution Using Biomass-Based Activated Carbon. Sci. Rep. 2021, 11, 8623. [Google Scholar] [CrossRef]
- Aziz, E.K.; Abdelmajid, R.; Rachid, L.M.; Mohammadine, E.H. Adsorptive Removal of Anionic Dye from Aqueous Solutions Using Powdered and Calcined Vegetables Wastes as Low-Cost Adsorbent. Arab J. Basic Appl. Sci. 2018, 25, 93–102. [Google Scholar] [CrossRef]
- Kavitha, G.; Subhapriya, P.; Dhanapal, V.; Dineshkumar, G.; Venkateswaran, V. Dye Removal Kinetics and Adsorption Studies of Activated Carbon Derived from the Stems of Phyllanthus reticulatus. Mater. Today Proc. 2021, 45, 7934–7938. [Google Scholar] [CrossRef]
- Zhou, L.; Zhou, H.; Hu, Y.; Yan, S.; Yang, J. Adsorption Removal of Cationic Dyes from Aqueous Solutions Using Ceramic Adsorbents Prepared from Industrial Waste Coal Gangue. J. Environ. Manag. 2019, 234, 245–252. [Google Scholar] [CrossRef]
- Somsesta, N.; Sricharoenchaikul, V.; Aht-Ong, D. Adsorption Removal of Methylene Blue onto Activated Carbon/Cellulose Biocomposite Films: Equilibrium and Kinetic Studies. Mater. Chem. Phys. 2020, 240, 122221. [Google Scholar] [CrossRef]
- Arfi, R.B.; Karoui, S.; Mougin, K.; Ghorbal, A. Adsorptive Removal of Cationic and Anionic Dyes from Aqueous Solution by Utilizing Almond Shell as Bioadsorbent. Euro-Mediterr. J. Environ. Integr. 2017, 2, 20. [Google Scholar] [CrossRef] [Green Version]
- Kyzas, G.Z.; Christodoulou, E.; Bikiaris, D.N. Basic Dye Removal with Sorption onto Low-Cost Natural Textile Fibers. Processes 2018, 6, 166. [Google Scholar] [CrossRef] [Green Version]
- Safitri, A.; Febrianti, W.D.; Rahmaniah, G. Effectiveness of Using Trichoderma Viride as Biosorbent for Remazol Brilliant Purple in Batik Wastewater Treatment. JSMARTech J. Smart Bioprospect. Technol. 2020, 1, 41–45. [Google Scholar] [CrossRef]
- Şentürk, İ.; Alzein, M. Adsorption of Acid Violet 17 Onto Acid-Activated Pistachio Shell: Isotherm, Kinetic and Thermodynamic Studies. Acta Chim. Slov. 2020, 67, 55–69. [Google Scholar] [CrossRef]
- Pernyeszi, T.; Farkas, R.; Kovács, J. Methylene Blue Adsorption Study on Microcline Particles in the Function of Particle Size Range and Temperature. Minerals 2019, 9, 555. [Google Scholar] [CrossRef] [Green Version]
- Ma, C.M.; Hong, G.B.; Wang, Y.K. Performance Evaluation and Optimization of Dyes Removal Using Rice Bran-Based Magnetic Composite Adsorbent. Materials 2020, 13, 2764. [Google Scholar] [CrossRef]
- Neag, E.; Moldovan, A.; Băbălău-Fuss, V.; Török, A.; Cadar, O.; Roman, C. Kinetic, Equilibrium and Phytotoxicity Studies for Dyes Removal by Low Cost Natural Activated Plant-Based Carbon. Acta Chim. Slov. 2019, 66, 850–858. [Google Scholar] [CrossRef]
- Esmaeili, H.; Foroutan, R.; Jafari, D.; Aghil Rezaei, M. Effect of Interfering Ions on Phosphate Removal from Aqueous Media Using Magnesium Oxide@ferric Molybdate Nanocomposite. Korean J. Chem. Eng. 2020, 37, 804–814. [Google Scholar] [CrossRef]
- Salahshour, R.; Shanbedi, M.; Esmaeili, H. Methylene Blue Dye Removal from Aqueous Media Using Activated Carbon Prepared by Lotus Leaves: Kinetic, Equilibrium and Thermodynamic Study. Acta Chim. Slov. 2021, 68, 363–373. [Google Scholar] [CrossRef]
- Dehghani, M.H.; Salari, M.; Karri, R.R.; Hamidi, F.; Bahadori, R. Process Modeling of Municipal Solid Waste Compost Ash for Reactive Red 198 Dye Adsorption from Wastewater Using Data Driven Approaches. Sci. Rep. 2021, 11, 11613. [Google Scholar] [CrossRef] [PubMed]
- Popa, S.; Radulescu-Grad, M.E.; Perdivara, A.; Mosoarca, G. Aspects Regarding Colour Fastness and Adsorption Studies of a New Azo-Stilbene Dye for Acrylic Resins. Sci. Rep. 2021, 11, 5889. [Google Scholar] [CrossRef] [PubMed]
- Miyah, Y.; Lahrichi, A.; Idrissi, M.; Khalil, A.; Zerrouq, F. Adsorption of Methylene Blue Dye from Aqueous Solutions onto Walnut Shells Powder: Equilibrium and Kinetic Studies. Surf. Interfaces 2018, 11, 74–81. [Google Scholar] [CrossRef]
- Alver, E.; Metin, A.Ü.; Brouers, F. Methylene Blue Adsorption on Magnetic Alginate/Rice Husk Bio-Composite. Int. J. Biol. Macromol. 2020, 154, 104–113. [Google Scholar] [CrossRef]
- Hamza, W.; Dammak, N.; Hadjltaief, H.B.; Eloussaief, M.; Benzina, M. Sono-Assisted Adsorption of Cristal Violet Dye onto Tunisian Smectite Clay: Characterization, Kinetics and Adsorption Isotherms. Ecotoxicol. Environ. Saf. 2018, 163, 365–371. [Google Scholar] [CrossRef]
- Naushad, M.; Alqadami, A.A.; Al-Kahtani, A.A.; Ahamad, T.; Awual, M.R.; Tatarchuk, T. Adsorption of Textile Dye Using Para-Aminobenzoic Acid Modified Activated Carbon: Kinetic and Equilibrium Studies. J. Mol. Liq. 2019, 296, 112075. [Google Scholar] [CrossRef]
- Márquez, C.O.; García, V.J.; Guaypatin, J.R.; Fernández-Martínez, F.; Ríos, A.C. Cationic and Anionic Dye Adsorption on a Natural Clayey Composite. Appl. Sci. 2021, 11, 5127. [Google Scholar] [CrossRef]
- Maleki, A.; Mohammad, M.; Emdadi, Z.; Asim, N.; Azizi, M.; Safaei, J. Adsorbent Materials Based on a Geopolymer Paste for Dye Removal from Aqueous Solutions. Arab. J. Chem. 2020, 13, 3017–3025. [Google Scholar] [CrossRef]
- Hamzezadeh, A.; Rashtbari, Y.; Afshin, S.; Morovati, M.; Vosoughi, M. Application of Low-Cost Material for Adsorption of Dye from Aqueous Solution. Int. J. Environ. Anal. Chem. 2020, 1–16. [Google Scholar] [CrossRef]
- Raj, R.A.; Manimozhi, V.; Saravanathamizhan, R. Adsorption Studies on Removal of Congo Red Dye from Aqueous Solution Using Petroleum Coke. Pet. Sci. Technol. 2019, 37, 913–924. [Google Scholar] [CrossRef]
- Stjepanović, M.; Velić, N.; Galić, A.; Kosović, I.; Jakovljević, T.; Habuda-Stanić, M. From Waste to Biosorbent: Removal of Congo Red from Water by Waste Wood Biomass. Water 2021, 13, 279. [Google Scholar] [CrossRef]
- Šljivić-Ivanović, M.; Smičiklas, I. Utilization of C&D waste in radioactive waste treatment—Current knowledge and perspectives. In Advances in Construction and Demolition Waste Recycling; Pacheco-Torgal, F., Ding, Y., Colangelo, F., Tuladhar, R., Koutamanis, A., Eds.; Woodhead Publishing Series in Civil and Structural Engineering; Woodhead Publishing: Sawaston, UK, 2020; Chapter 23; pp. 475–500. ISBN 978-0-12-819055-5. [Google Scholar]
- Aljeboree, A.M.; Alshirifi, A.N.; Alkaim, A.F. Kinetics and Equilibrium Study for the Adsorption of Textile Dyes on Coconut Shell Activated Carbon. Arab. J. Chem. 2017, 10, S3381–S3393. [Google Scholar] [CrossRef] [Green Version]
- Iqbal, J.; Wattoo, F.H.; Wattoo, M.H.S.; Malik, R.; Tirmizi, S.A.; Imran, M.; Ghangro, A.B. Adsorption of Acid Yellow Dye on Flakes of Chitosan Prepared from Fishery Wastes. Arab. J. Chem. 2011, 4, 389–395. [Google Scholar] [CrossRef] [Green Version]
- Nikam, S.; Mandal, D. Experimental Study of the Effect of Different Parameters on the Adsorption and Desorption of Trichloroethylene Vapor on Activated Carbon Particles. Acs Omega 2020, 5, 28080–28087. [Google Scholar] [CrossRef]
- Wekoye, J.N.; Wanyonyi, W.C.; Wangila, P.T.; Tonui, M.K. Kinetic and Equilibrium Studies of Congo Red Dye Adsorption on Cabbage Waste Powder. Environ. Chem. Ecotoxicol. 2020, 2, 24–31. [Google Scholar] [CrossRef]
- Felista, M.M.; Wanyonyi, W.C.; Ongera, G. Adsorption of Anionic Dye (Reactive Black 5) Using Macadamia Seed Husks: Kinetics and Equilibrium Studies. Sci. Afr. 2020, 7, e00283. [Google Scholar] [CrossRef]
- Shakoor, S.; Nasar, A. Adsorptive Treatment of Hazardous Methylene Blue Dye from Artificially Contaminated Water Using Cucumis Sativus Peel Waste as a Low-Cost Adsorbent. Groundw. Sustain. Dev. 2017, 5, 152–159. [Google Scholar] [CrossRef]
- Cheruiyot, G.K.; Wanyonyi, W.C.; Kiplimo, J.J.; Maina, E.N. Adsorption of Toxic Crystal Violet Dye Using Coffee Husks: Equilibrium, Kinetics and Thermodynamics Study. Sci. Afr. 2019, 5, e00116. [Google Scholar] [CrossRef]
- Mahdavinia, G.; Baghban, A.; Zorofi, S.; Massoudi, A. Kappa-Carrageenan Biopolymer-Based Nanocomposite Hydrogel and Adsorption of Methylene Blue Cationic Dye from Water. J. Mater. Environ. Sci. 2014, 5, 330–337. [Google Scholar]
- Shahul Hameed, K.; Muthirulan, P.; Meenakshi Sundaram, M. Adsorption of Chromotrope Dye onto Activated Carbons Obtained from the Seeds of Various Plants: Equilibrium and Kinetics Studies. Arab. J. Chem. 2017, 10, S2225–S2233. [Google Scholar] [CrossRef]
- Badawy, A.A.; Ibrahim, S.M.; Essawy, H.A. Enhancing the Textile Dye Removal from Aqueous Solution Using Cobalt Ferrite Nanoparticles Prepared in Presence of Fulvic Acid. J. Inorg. Organomet. Polym. 2020, 30, 1798–1813. [Google Scholar] [CrossRef]
- Abualnaja, K.M.; Alprol, A.E.; Abu-Saied, M.A.; Ashour, M.; Mansour, A.T. Removing of Anionic Dye from Aqueous Solutions by Adsorption Using of Multiwalled Carbon Nanotubes and Poly(Acrylonitrile-Styrene) Impregnated with Activated Carbon. Sustainability 2021, 13, 7077. [Google Scholar] [CrossRef]
- Khalaf, I.H.; Al-Sudani, F.T.; AbdulRazak, A.A.; Aldahri, T.; Rohani, S. Optimization of Congo Red Dye Adsorption from Wastewater by a Modified Commercial Zeolite Catalyst Using Response Surface Modeling Approach. Water Sci. Technol. 2021, 83, 1369–1383. [Google Scholar] [CrossRef]
- Sharma, K.; Sharma, S.; Sharma, V.; Mishra, P.K.; Ekielski, A.; Sharma, V.; Kumar, V. Methylene Blue Dye Adsorption from Wastewater Using Hydroxyapatite/Gold Nanocomposite: Kinetic and Thermodynamics Studies. Nanomaterials 2021, 11, 1403. [Google Scholar] [CrossRef]
- El-Harby, N.F.; Ibrahim, S.M.A.; Mohamed, N.A. Adsorption of Congo Red Dye onto Antimicrobial Terephthaloyl Thiourea Cross-Linked Chitosan Hydrogels. Water Sci. Technol. 2017, 76, 2719–2732. [Google Scholar] [CrossRef]
- Geroeeyan, A.; Niazi, A.; Konoz, E. Removal of Basic Orange 2 Dye and Ni2+ from Aqueous Solutions Using Alkaline-Modified Nanoclay. Water Sci. Technol. 2021, 83, 2271–2286. [Google Scholar] [CrossRef] [PubMed]
- Noreen, S.; Khalid, U.; Ibrahim, S.M.; Javed, T.; Ghani, A.; Naz, S.; Iqbal, M. ZnO, MgO and FeO Adsorption Efficiencies for Direct Sky Blue Dye: Equilibrium, Kinetics and Thermodynamics Studies. J. Mater. Res. Technol. 2020, 9, 5881–5893. [Google Scholar] [CrossRef]
- Santander, P.; Oyarce, E.; Sánchez, J. New Insights in the Use of a Strong Cationic Resin in Dye Adsorption. Water Sci. Technol. 2020, 81, 773–780. [Google Scholar] [CrossRef] [Green Version]
- Ibrahim, S.M.; Hassanin, H.M.; Abdelrazek, M.M. Synthesis, and Characterization of Chitosan Bearing Pyranoquinolinone Moiety for Textile Dye Adsorption from Wastewater. Water Sci. Technol. 2020, 81, 421–435. [Google Scholar] [CrossRef] [PubMed]
- Oueslati, K.; Lima, E.C.; Ayachi, F.; Cunha, M.R.; Ben Lamine, A. Modeling the Removal of Reactive Red 120 Dye from Aqueous Effluents by Activated Carbon. Water Sci. Technol. 2020, 82, 651–662. [Google Scholar] [CrossRef]
- Su, H.; Li, W.; Han, Y.; Liu, N. Magnetic Carboxyl Functional Nanoporous Polymer: Synthesis, Characterization and Its Application for Methylene Blue Adsorption. Sci. Rep. 2018, 8, 6506. [Google Scholar] [CrossRef] [Green Version]
- Tran, H.V.; Hoang, L.T.; Huynh, C.D. An Investigation on Kinetic and Thermodynamic Parameters of Methylene Blue Adsorption onto Graphene-Based Nanocomposite. Chem. Phys. 2020, 535, 110793. [Google Scholar] [CrossRef]
- Chen, T.; Da, T.; Ma, Y. Reasonable Calculation of the Thermodynamic Parameters from Adsorption Equilibrium Constant. J. Mol. Liq. 2021, 322, 114980. [Google Scholar] [CrossRef]
- Ali, F.; Ali, N.; Bibi, I.; Said, A.; Nawaz, S.; Ali, Z.; Salman, S.M.; Iqbal, H.M.N.; Bilal, M. Adsorption Isotherm, Kinetics and Thermodynamic of Acid Blue and Basic Blue Dyes onto Activated Charcoal. Case Stud. Chem. Environ. Eng. 2020, 2, 100040. [Google Scholar] [CrossRef]
- Rios-Donato, N.; Peña-Flores, A.M.; Katime, I.; Leyva-Ramos, R.; Mendizábal, E. Kinetics and Thermodynamics of Adsorption of Red Dye 40 from Acidic Aqueous Solutions onto a Novel Chitosan Sulfate. Afinidad 2017, 74, 214–220. [Google Scholar]
- Miraboutalebi, S.M.; Peydayesh, M.; Bagheri, M.; Mohammadi, T. Polyacrylonitrile/α-Fe2O3 Hybrid Photocatalytic Composite Adsorbents for Enhanced Dye Removal. Chem. Eng. Technol. 2020, 43, 1214–1223. [Google Scholar] [CrossRef]
- Pradhan, P.; Bajpai, A. Preparation and Characterization of Films from Chicken Feathers for Dye Adsorption. Mater. Today Proc. 2020, 29, 1204–1212. [Google Scholar] [CrossRef]
- Abdel-Aziz, M.H.; El-Ashtoukhy, E.Z.; Bassyouni, M.; Al-Hossainy, A.F.; Fawzy, E.M.; Abdel-Hamid, S.M.S.; Zoromba, M.S. DFT and Experimental Study on Adsorption of Dyes on Activated Carbon Prepared from Apple Leaves. Carbon Lett. 2020. [Google Scholar] [CrossRef]
- Abegunde, S.M.; Idowu, K.S.; Adejuwon, O.M.; Adeyemi-Adejolu, T. A Review on the Influence of Chemical Modification on the Performance of Adsorbents. Resour. Environ. Sustain. 2020, 1, 100001. [Google Scholar] [CrossRef]
- Girish, C.R. Various Impregnation Methods Used for the Surface Modification of the Adsorbent: A Review. Int. J. Eng. Technol. 2018, 7, 330–334. [Google Scholar] [CrossRef] [Green Version]
- Rehman, A.; Park, M.; Park, S.-J. Current Progress on the Surface Chemical Modification of Carbonaceous Materials. Coatings 2019, 9, 103. [Google Scholar] [CrossRef] [Green Version]
- Abdulrasheed, A.A.; Jalil, A.A.; Triwahyono, S.; Zaini, M.A.A.; Gambo, Y.; Ibrahim, M. Surface Modification of Activated Carbon for Adsorption of SO2 and NOX: A Review of Existing and Emerging Technologies. Renew. Sustain. Energy Rev. 2018, 94, 1067–1085. [Google Scholar] [CrossRef]
- Zheng, Z.; Cox, M.; Li, B. Surface Modification of Hexagonal Boron Nitride Nanomaterials: A Review. J. Mater. Sci. 2018, 53, 66–99. [Google Scholar] [CrossRef]
- Cashin, V.B.; Eldridge, D.S.; Yu, A.; Zhao, D. Surface Functionalization and Manipulation of Mesoporous Silica Adsorbents for Improved Removal of Pollutants: A Review. Environ. Sci. Water Res. Technol. 2018, 4, 110–128. [Google Scholar] [CrossRef]
- Shojaei, S.; Shojaei, S.; Band, S.S. Application of Chemometrics into Removal of Dyes by NaX Nanozeolites: Simultaneous Model; Social Science Research Network: Rochester, NY, USA, 2021. [Google Scholar]
- Jiang, F.; Dinh, D.M.; Hsieh, Y.-L. Adsorption and Desorption of Cationic Malachite Green Dye on Cellulose Nanofibril Aerogels. Carbohydr. Polym. 2017, 173, 286–294. [Google Scholar] [CrossRef] [Green Version]
- Bhatti, H.N.; Safa, Y.; Yakout, S.M.; Shair, O.H.; Iqbal, M.; Nazir, A. Efficient Removal of Dyes Using Carboxymethyl Cellulose/Alginate/Polyvinyl Alcohol/Rice Husk Composite: Adsorption/Desorption, Kinetics and Recycling Studies. Int. J. Biol. Macromol. 2020, 150, 861–870. [Google Scholar] [CrossRef]
- Momina; Mohammad, S.; Suzylawati, I. Study of the Adsorption/Desorption of MB Dye Solution Using Bentonite Adsorbent Coating. J. Water Process Eng. 2020, 34, 101155. [Google Scholar] [CrossRef]
- Murcia-Salvador, A.; Pellicer, J.A.; Rodríguez-López, M.I.; Gómez-López, V.M.; Núñez-Delicado, E.; Gabaldón, J.A. Egg By-Products as a Tool to Remove Direct Blue 78 Dye from Wastewater: Kinetic, Equilibrium Modeling, Thermodynamics and Desorption Properties. Materials 2020, 13, 1262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, H. Review on Solvent Desorption Study from Exhausted Adsorbent. J. Saudi Chem. Soc. 2021, 25, 101302. [Google Scholar] [CrossRef]


| Adsorbent | Dyestuff | Adsorbent Dosage | Efficiency Range (%) | Quantity in Equilibrium Range (qe mg/g) | Reference |
|---|---|---|---|---|---|
| walnut shell | Methylene Blue | 0.5–2 g/L | - | 178.93–47.51 | [195] |
| magnetic alginate/rice husk bio-composite | Methylene Blue | 0.1–1 g | 15–89 | 338–145 | [196] |
| Tunisian smectite clay | Cristal Violet | 0.05–0.3 g/L | 10–100 | - | [197] |
| modified activated carbon (PABA@AC) | Malachite Green | 10–50 mg | 31.3–86.6 | 11.67–6.5 | [198] |
| commercial natural activated plant-based carbon (CNAC) | Methylene Blue | 0.5–1.5 g | 46–78 | - | [190] |
| commercial natural activated plant-based carbon (CNAC) | Eosin Yellow | 0.5–1.5 g | 51–70 | - | [190] |
| commercial natural activated plant-based carbon (CNAC) | Rhodamine B | 0.5–1.5 g | 52–60 | - | [190] |
| calcined eggshell | Remazol Brilliant Violet-5R | 0.5–2 g | 89.83–96.3 | 3.59–0.96 | [134] |
| calcined eggshell | Remazol Red F3B | 0.5–2 g | 92–93.67 | 3.68–0.94 | [135] |
| calcined eggshell | Remazol Blue RR | 0.5–2 g | 92–93.33 | 3.68–0.94 | [135] |
| eggshell | Remazol Brilliant Violet-5R | 0.5–2.5 g | 74.67–93.85 | 2.96–0.75 | [51] |
| activated carbon from lotus leaves | Methylene Blue | 0.5–10 g/L | 82.84–98.032 | 16.57–0.98 | [192] |
| municipial solid waste compost ash | Reactive Red 198 | 0.5–2 g/L | 79.25–92.92 | - | [193] |
| natural clayey composite | Basic Navy Blue 2RN | 0.2–1.2 g/50 mL | 78–97 | - | [199] |
| natural clayey composite | Drimaren Yellow CL-2R | 0.2–1.2 g/50 mL | 87–97 | - | [199] |
| geopolymer | Methylene Blue (10−5 M) | 0.05–0.1 g | 79.8–85.6 | - | [200] |
| mucilage of Salvia seeds | Cationic Blue 41 | 0.5–4 g/L | 34.2–53.9 | 34.2–6.74 | [201] |
| raw petroleum coke | Congo Red | 4–24 g/L | ~10–60 | - | [202] |
| activated petroleum coke | Congo Red | 4–24 g/L | ~15–70 | - | [202] |
| Dyestuff | Adsorbent | Particle Size (μm) | Efficiency Range (%) | Quantity in Equilibrium Range (qe mg/g) | Reference |
|---|---|---|---|---|---|
| Congo Red | cabbage waste powder | 150–300 to 360–4750 | 75.95–8.03 | - | [208] |
| Reactive Black 5 | macadamia seed husks | 150–300 to 2360–4750 | 98.9–33.2 | - | [209] |
| Maxilon Blue GRL | coconut shell activated carbon | 50, 75, and 106 | - | ~27.5–22.5–17.5 | [205] |
| Direct Yellow DY 12 | coconut shell activated carbon | 50, 75, and 107 | - | ~5.5–4.5–3.5 | [205] |
| Methylene Blue | Cucumis sativus peel waste | 80–150, 150–200, and >200 BSS mesh | 80.25–84.15–85.23 | - | [210] |
| Crystal Violet | coffee husks | 0.15–0.3 to 2.36–4.75 mm | 96.082–89.854 | - | [211] |
| Methylene Blue | clay3 | 177–250 to 400–840 | 99–86.4 | - | [212] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rápó, E.; Tonk, S. Factors Affecting Synthetic Dye Adsorption; Desorption Studies: A Review of Results from the Last Five Years (2017–2021). Molecules 2021, 26, 5419. https://doi.org/10.3390/molecules26175419
Rápó E, Tonk S. Factors Affecting Synthetic Dye Adsorption; Desorption Studies: A Review of Results from the Last Five Years (2017–2021). Molecules. 2021; 26(17):5419. https://doi.org/10.3390/molecules26175419
Chicago/Turabian StyleRápó, Eszter, and Szende Tonk. 2021. "Factors Affecting Synthetic Dye Adsorption; Desorption Studies: A Review of Results from the Last Five Years (2017–2021)" Molecules 26, no. 17: 5419. https://doi.org/10.3390/molecules26175419
APA StyleRápó, E., & Tonk, S. (2021). Factors Affecting Synthetic Dye Adsorption; Desorption Studies: A Review of Results from the Last Five Years (2017–2021). Molecules, 26(17), 5419. https://doi.org/10.3390/molecules26175419






