Biflavonoids from Selaginella doederleinii as Potential Antitumor Agents for Intervention of Non-Small Cell Lung Cancer
Abstract
:1. Introduction
2. Results
2.1. Structural Elucidation
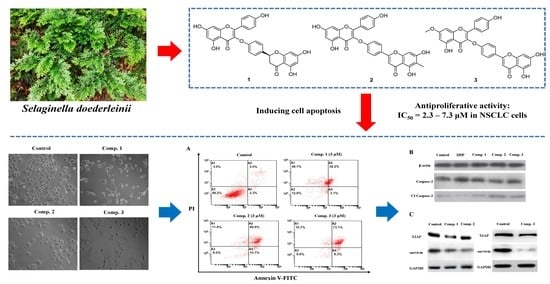
2.2. Assessment of Anti-Proliferative Activity of Compounds 1–5 in NSCLC Cells
2.3. Compound 1–3 Induced Apoptosis in A549 Cells
2.4. Compound 1–3 Promoted Upregulation of Caspase-3/Cleaved-Caspase-3 in A549 Cells
2.5. Compound 1–3 Reduced the of Expression of XIAP and Survivin in A549 Cells
2.6. Compounds 3 Caused Cell Cycle Arrest in A549 Cells
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Plant Material
4.3. Extraction and Isolation
4.4. Cell Culture
4.5. Cytotoxicity Assay
4.6. Morphological Observations
4.7. Apoptosis Assay
4.8. Cell Cycle Analysis
4.9. Western Blotting Assay
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Zhang, N.; Nan, A.R.; Chen, L.J.; Li, X.; Jia, Y.Y.; Qiu, M.Y.; Dai, X.; Zhou, H.Y.; Zhu, J.L.; Zhang, H.; et al. Circular RNA circSATB2 promotes progression of non-small cell lung cancer cells. Mol. Cancer 2020, 19, 101. [Google Scholar] [CrossRef]
- Zheng, H.M.; Zhan, Y.T.; Liu, S.L.; Lu, J.M.; Luo, J.D.; Feng, J.; Fan, S.Q. The roles of tumor-derived exosomes in non-small cell lung cancer and their clinical implications. J. Exp. Clin. Canc. Res. 2018, 37, 226. [Google Scholar] [CrossRef]
- Vrankar, M.; Zwitter, M.; Kern, I.; Stanic, K. PD-L1 expression can be regarded as prognostic factor for survival of non-small cell lung cancer patients after chemoradiotherapy. Neoplasma 2018, 65, 140–146. [Google Scholar] [CrossRef]
- Xu, J.F.; Yang, L.J.; Wang, R.; Zeng, K.; Fan, B.Y.; Zhao, Z.B. The biflavonoids as protein tyrosine phosphatase 1B inhibitors from Selaginella uncinata and their antihyperglycemic action. Fitoterapia 2019, 139, 104255. [Google Scholar] [CrossRef] [PubMed]
- Goossens, J.F.; Goossens, L.; Bailly, C. Hinokiflavone and related C-O-C-type biflavonoids as anti-cancer compounds: Properties and mechanism of action. Nat. Prod. Bioprospect. 2021, 11, 365–377. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.X.; Wang, N.L.; Liu, H.W.; Chen, H.F.; Li, M.M.; Wu, L.Y.; Fan, M.; Yao, X.S. Four new biflavonoids from Selaginella uncinata and their anti-anoxic effect. J. Asian Nat. Prod. Res. 2008, 10, 945–952. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.M.; Wang, S.; Yu, M.; Wu, D.; Lei, J.; Li, W.W.; He, Y.Q.; Gang, W. Ultrasonic-assisted ionic liquid extraction of two biflavonoids from Selaginella tamariscina. ACS Omega 2020, 5, 33113–33124. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Qian, Y.; Tian, Y.J.; Yuan, S.M.; Wei, W.; Wang, G. Optimization of ionic liquid-assisted extraction of biflavonoids from Selaginella doederleinii and evaluation of its antioxidant and antitumor activity. Molecules 2017, 22, 586. [Google Scholar] [CrossRef] [Green Version]
- Li, S.G.; Zhao, M.F.; Li, Y.X.; Sui, Y.X.; Yao, H.; Huang, L.Y.; Lin, X.H. Preparative isolation of six anti-tumour biflavonoids from Selaginella Doederleinii Hieron by high-speed counter-current chromatography. Phytochem. Anal. 2014, 25, 127–133. [Google Scholar] [CrossRef]
- Yang, S.F.; Chu, S.C.; Liu, S.J.; Chen, Y.C.; Chang, Y.Z.; Hsieh, Y.S. Antimetastatic activities of Selaginella tamariscina (Beauv.) on lung cancer cells in vitro and in vivo. J. Ethnopharmacol. 2007, 110, 483–489. [Google Scholar] [CrossRef]
- Jung, Y.J.; Lee, E.H.; Lee, C.G.; Rhee, K.J.; Jung, W.S.; Choi, Y.; Pan, C.H.; Kang, K. AKR1B10-inhibitory Selaginella tamariscina extract and amentoflavone decrease the growth of A549 human lung cancer cells in vitro and in vivo. J. Ethnopharmacol. 2017, 202, 78–84. [Google Scholar] [CrossRef]
- Huang, W.Z.; Liu, C.; Liu, F.G.; Liu, Z.Y.; Lai, G.E.; Yi, J. Hinokiflavone induces apoptosis and inhibits migration of breast cancer cells via EMT signalling pathway. Cell Biochem. Funct. 2020, 38, 249–256. [Google Scholar] [CrossRef]
- Zou, Z.X.; Zhang, S.; Tan, J.B.; Chen, D.K.; Xu, Y.R.; Xu, K.P.; Tan, G.S. Two new biflavonoids from Selaginella doederleinii. Phytochem. Lett. 2020, 40, 126–129. [Google Scholar] [CrossRef]
- Zou, Z.X.; Xu, P.S.; Zhang, G.G.; Cheng, F.; Chen, K.; Li, J.; Zhu, W.X.; Cao, D.S.; Xu, K.P.; Tan, G.S. Selagintriflavonoids with BACE1 inhibitory activity from the fern Selaginella doederleinii. Phytochemistry 2017, 134, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.X.; Xu, P.S.; Wu, C.R.; Zhu, W.X.; Zhu, G.Z.; He, X.A.; Zhang, G.G.; Hu, J.Z.; Liu, S.; Zeng, W.; et al. Carboxymethyl flavonoids and a chromone with antimicrobial activity from Selaginella moellendorffii Hieron. Fitoterapia 2016, 111, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.X.; Xu, K.P.; Xu, P.S.; Li, X.M.; Cheng, F.; Li, J.; Yu, X.; Cao, D.S.; Li, D.; Zeng, W.; et al. Seladoeflavones A-F, six novel flavonoids from Selaginella doederleinii. Fitoterapia 2017, 116, 66–71. [Google Scholar] [CrossRef]
- Zou, Z.X.; Tan, G.S.; Zhang, G.G.; Yu, X.; Xu, P.S.; Xu, K.P. New cytotoxic apigenin derivatives from Selaginella doederleinii. Chinese Chem. Lett. 2017, 28, 931–934. [Google Scholar] [CrossRef]
- Xie, Y.; Zhou, X.; Li, J.; Yao, X.C.; Liu, W.L.; Kang, F.H.; Zou, Z.X.; Xu, K.P.; Xu, P.S.; Tan, G.S. Identification of a new natural biflavonoids against breast cancer cells induced ferroptosis via the mitochondrial pathway. Bioorg. Chem. 2021, 109, 104744. [Google Scholar] [CrossRef]
- Liu, L.F.; Sun, H.H.; Tan, J.B.; Huang, Q.; Cheng, F.; Xu, K.P.; Zou, Z.X.; Tan, G.S. New cytotoxic biflavones from Selaginella doederleinii. Nat. Prod. Res. 2021, 35, 930–936. [Google Scholar] [CrossRef]
- Lin, L.C.; Chou, C.J. Three new biflavonoids from selaginella delicatula. Chin. Pharm. J. 2000, 52, 211–218. [Google Scholar]
- Swamy, R.C.; Kunert, O.; Schühly, W.; Bucar, F.; Ferreira, D.; Rani, V.S.; Kumar, B.R.; Rao, A.V.N.A. Structurally unique biflavonoids from Selaginella chrysocaulos and Selaginella bryopteris. Chem. Biodivers. 2006, 3, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Slade, D.; Ferreira, D.; Marais, J.P.J. Circular dichroism, a powerful tool for the assessment of absolute configuration of flavonoids. Phytochemistry 2005, 66, 2177–2215. [Google Scholar] [CrossRef] [PubMed]
- Cheung, C.H.A.; Chang, Y.C.; Lin, T.Y.; Cheng, S.M.; Leung, E. Anti-apoptotic proteins in the autophagic world: An update on functions of XIAP, Survivin, and BRUCE. J. Biomed. Sci. 2020, 27, 31. [Google Scholar] [CrossRef]
- Bedoui, S.; Herold, M.J.; Strasser, A. Emerging connectivity of programmed cell death pathways and its physiological implications. Nat. Rev. Mol. Cell Biol. 2020, 21, 678–695. [Google Scholar] [CrossRef] [PubMed]









| Position | 1 a | 2 a | 3 a | 4 b |
|---|---|---|---|---|
| 6 | 6.24, d (2.0) | 6.05, d (2.0) | 6.26, d (2.0) | 6.24, d (1.6) |
| 8 | 6.54, d (2.0) | 6.32, d (2.0) | 6.55, d (2.0) | 6.54, d (1.6) |
| 7-OCH3 | 3.87, s | |||
| 2′/6′ | 7.87, d (8.5) | 7.78, d (9.0) | 7.84, d (9.0) | 7.87, d (8.8) |
| 3′/5′ | 6.89, d (8.5) | 6.84, d (9.0) | 6.89, d (9.0) | 6.89, d (8.8) |
| 2″ | 5.50, dd (13.0, 3.0) | 5.46, dd (12.8, 2.4) | ||
| 3″ | 3.26, dd (17.0, 3.0) | 6.77, s | 6.95, s | 3.24, dd (16.8, 2.4) |
| 2.71, dd (17.0, 3.0) | 2.72, dd, (16.8, 2.4) | |||
| 6″ | 5.89, d (2.0) | 6.40, d (2.0) | ||
| 8″ | 5.90, d (2.0) | 6.43, s | 6.78, d (2.0) | 5.99, s |
| 6″-CH3 | 1.97, s | 1.88, s | ||
| 2′′′/6′′′ | 7.45, d (8.5) | 7.95, d (8.5) | 8.04, d (9.0) | 7.44, d (8.8) |
| 3′′′/5′′′ | 7.10, d (8.5) | 7.19, d (8.5) | 7.27, d (9.0) | 7.10, d (8.8) |
| 5-OH | 12.30, s | 12.21, br s | 12.24, s | 12.31, br s |
| 7-OH | 10.96, br s | 10.89, br s | ||
| 4′-OH | 10.37, br s | 10.38, br s | 10.39, br s | |
| 5″-OH | 12.14, s | 13.07, br s | 12.88, s | 12.40, s |
| 7″-OH | 10.82, br s | 11.04, br s | 10.89, br s |
| Position | 1 a | 2 a | 3 a | 4 b |
|---|---|---|---|---|
| 2 | 160.7 | 161.5 | 160.8 | 161.1 |
| 3 | 132.1 | 131.8 | 131.8 | 132.4 |
| 4 | 176.4 | 175.6 | 176.1 | 176.8 |
| 5 | 161.3 | 161.5 | 161.3 | 161.6 |
| 6 | 99.0 | 100.7 | 99.1 | 99.3 |
| 7 | 164.6 | 163.2 | 164.7 | 165.0 |
| 8 | 94.2 | 95.3 | 93.0 | 94.6 |
| 9 | 156.9 | 156.6 | 156.9 | 157.2 |
| 10 | 104.3 | 104.4 | 104.3 | 104.6 |
| 7-OCH3 | 56.2 | |||
| 1′ | 120.0 | 120.0 | 119.9 | 120.3 |
| 2′/6′ | 130.3 | 130.4 | 130.3 | 130.6 |
| 3′/5′ | 115.9 | 116.3 | 115.9 | 116.2 |
| 4′ | 157.1 | 155.8 | 157.2 | 157.5 |
| 2″ | 78.2 | 162.6 | 163.5 | 78.5 |
| 3″ | 42.1 | 104.4 | 104.5 | 42.5 |
| 4″ | 196.2 | 181.7 | 182.2 | 196.7 |
| 5″ | 163.6 | 158.8 | 161.3 | 161.2 |
| 6″ | 96.0 | 107.8 | 98.2 | 103.8 |
| 7″ | 166.8 | 165.4 | 165.4 | 165.1 |
| 8″ | 95.1 | 93.8 | 94.4 | 94.7 |
| 9″ | 163.0 | 157.6 | 157.5 | 160.7 |
| 10″ | 101.9 | 103.3 | 104.9 | 101.9 |
| 6″-CH3 | 8.0 | 7.4 | ||
| 1′′′ | 132.7 | 125.5 | 125.0 | 133.2 |
| 2′′′/6′′′ | 128.6 | 128.7 | 128.8 | 128.9 |
| 3′′′/5′′′ | 115.1 | 116.1 | 115.9 | 115.4 |
| 4′′′ | 157.1 | 160.0 | 159.8 | 157.2 |
| Compounds | IC50 (μM) a | ||
|---|---|---|---|
| A549 | H1299 | MRC-5 | |
| 1 | 3.1 c,d | 7.3 c,d | >100 |
| 2 | 2.8 c,d | 7.3 c,d | >100 |
| 3 | 2.3 c,d | 4.0 c,d | >100 |
| 4 | 3.1 c,d | 6.8 c,d | 58.2 |
| 5 | 7.9 c,d | 8.4 c,d | 36.8 |
| DDP b | 13.3 | 24.5 | 49.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, F.; Zhang, S.; Chen, D.; Tan, J.; Kuang, M.; Zhang, J.; Zeng, G.; Xu, K.; Zou, Z.; Tan, G. Biflavonoids from Selaginella doederleinii as Potential Antitumor Agents for Intervention of Non-Small Cell Lung Cancer. Molecules 2021, 26, 5401. https://doi.org/10.3390/molecules26175401
Kang F, Zhang S, Chen D, Tan J, Kuang M, Zhang J, Zeng G, Xu K, Zou Z, Tan G. Biflavonoids from Selaginella doederleinii as Potential Antitumor Agents for Intervention of Non-Small Cell Lung Cancer. Molecules. 2021; 26(17):5401. https://doi.org/10.3390/molecules26175401
Chicago/Turabian StyleKang, Fenghua, Sha Zhang, Dekun Chen, Jianbing Tan, Min Kuang, Jinlin Zhang, Guangyuan Zeng, Kangping Xu, Zhenxing Zou, and Guishan Tan. 2021. "Biflavonoids from Selaginella doederleinii as Potential Antitumor Agents for Intervention of Non-Small Cell Lung Cancer" Molecules 26, no. 17: 5401. https://doi.org/10.3390/molecules26175401
APA StyleKang, F., Zhang, S., Chen, D., Tan, J., Kuang, M., Zhang, J., Zeng, G., Xu, K., Zou, Z., & Tan, G. (2021). Biflavonoids from Selaginella doederleinii as Potential Antitumor Agents for Intervention of Non-Small Cell Lung Cancer. Molecules, 26(17), 5401. https://doi.org/10.3390/molecules26175401