Effects of Angiotensin II on Erythropoietin Production in the Kidney and Liver
Abstract
1. Introduction
2. Results
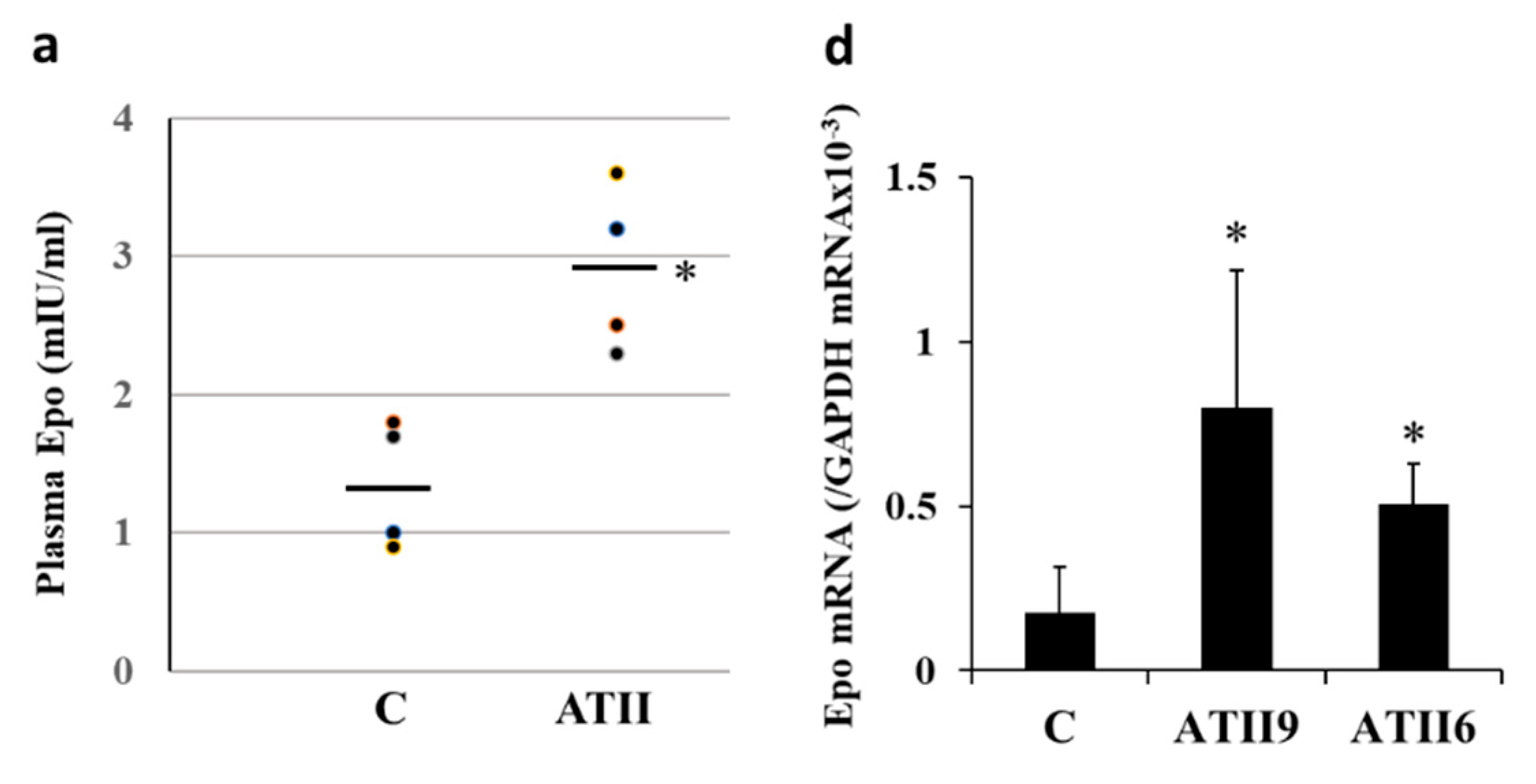
2.1. Plasma Epo Concentration
2.2. Epo mRNA Expression in the Renal Cortex and Microdissected Proximal Convoluted Tubules (PCT) and Medullary Thick Ascending Limbs (MAL)
2.3. In Situ Hybridization of Epoand the Effects of ATII
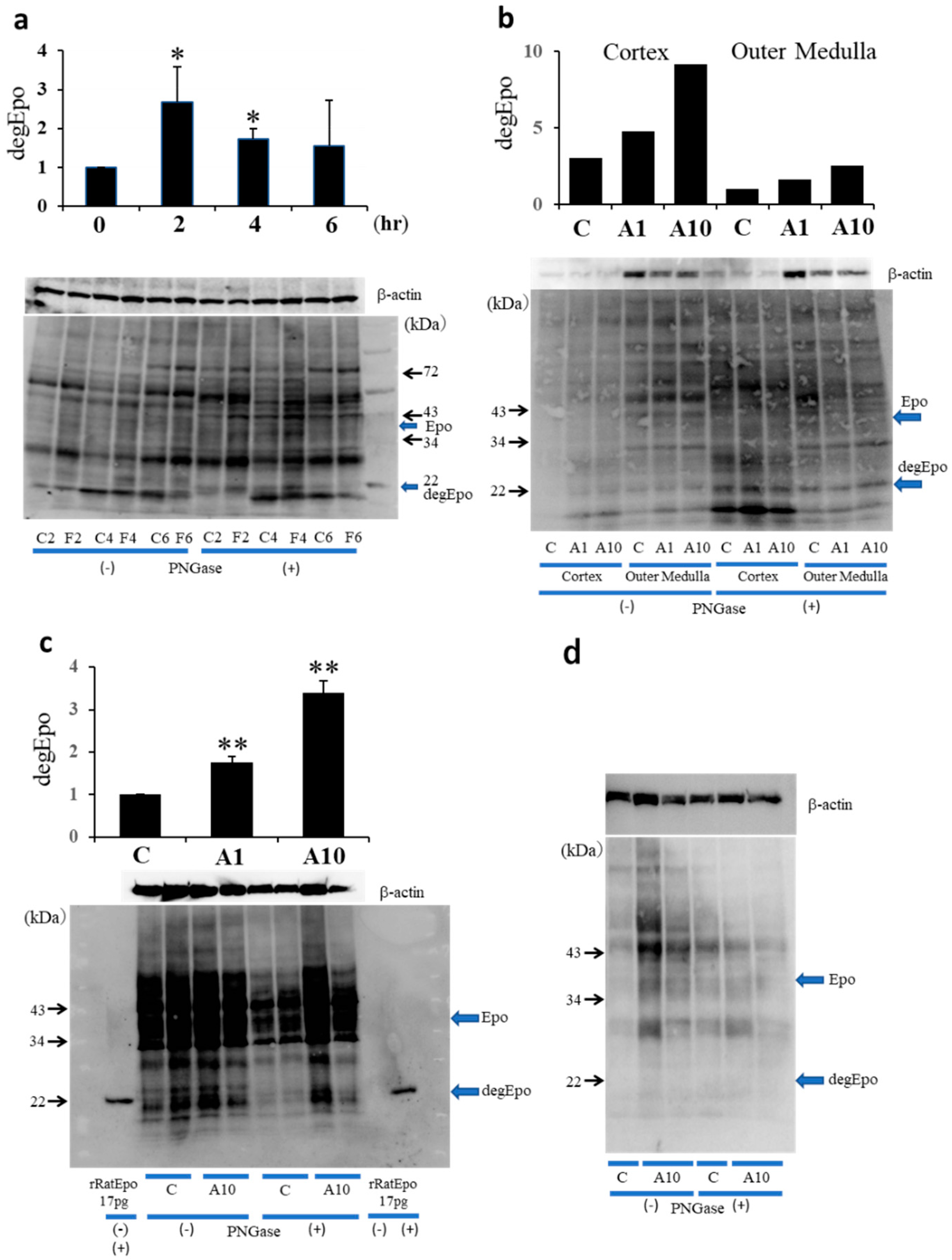
2.4. Western Blotting Analysis of ATII-Induced Epo Production in the Kidney and Liver
2.5. Immunohistochemistry of Epo Production by the Kidney
3. Discussion
4. Materials and Methods
4.1. Materials and Animals
4.2. Real-Time PCR
4.3. In Situ Hybridization
4.4. Immunohistochemistry
4.5. Western Blotting Analysis with Deglycosylation
4.6. Plasma Epo Concentration Measurements
4.7. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Ebert, B.L.; Bunn, H.F. Regulation of the erythropoietin gene. Blood 1999, 94, 1864–1877. [Google Scholar] [CrossRef] [PubMed]
- Haase, V.H. Regulation of erythropoiesis by hypoxia-inducible factors. Blood Rev. 2013, 27, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Koury, M.J.; Haase, V.H. Anaemia in kidney disease: Harnessing hypoxia responses for therapy. Nat. Rev. Nephrol. 2015, 11, 394–410. [Google Scholar] [CrossRef] [PubMed]
- Jelkmann, W. Regulation of erythropoietin production. J. Physiol. 2011, 589, 1251–1258. [Google Scholar] [CrossRef] [PubMed]
- Yasuoka, Y.; Fukuyama, T.; Izumi, Y.; Nakayama, Y.; Inoue, H.; Yanagita, K.; Oshima, T.; Yamazaki, T.; Uematsu, T.; Kobayashi, N.; et al. Erythropoietin production by the kidney and the liver in response to severe hypoxia evaluated by Western blotting with deglycosylation. Physiol. Rep. 2020, 8, 14485. [Google Scholar] [CrossRef] [PubMed]
- Li, L.H.; Chiu, Y.H.; Meng, C.; Kao, W.F.; How, C.K. Effects of 100-km ultramarathon on erythropoietin variation in runners with hepatitis B virus carrier. Chin. J. Physiol. 2021, 64, 125–128. [Google Scholar]
- Yasuoka, Y.; Izumi, Y.; Nagai, T.; Fukuyama, T.; Nakayama, Y.; Inoue, H.; Horikawa, K.; Kimura, M.; Nanami, M.; Yanagita, K.; et al. Fludrocortisone stimulates erythropoietin production in the intercalated cells of the collecting ducts. Biochem. Biophys. Res. Commun. 2018, 503, 3121–3127. [Google Scholar] [CrossRef]
- Gossmann, J.; Burkhardt, R.; Harder, S.; Lenz, T.; Sedlmeyer, A.; Klinkhardt, U.; Helmut, G.; Scheuermann, E.H. Angiotensin II infusion increases plasma erythropoietin levels via an angiotensin II type 1 receptor-dependent pathway. Kidney Int. 2001, 60, 83–86. [Google Scholar] [CrossRef]
- Yasuoka, Y.; Fukuyama, T.; Izumi, Y.; Yamashita, T.; Nakayama, Y.; Inoue, H.; Yanagita, K.; Oshima, T.; Yamazaki, T.; Uematsu, T.; et al. Differentiation of endogenous erythropoietin and exogenous ESAs by Western blotting. Heliyon 2020, 6, e05389. [Google Scholar] [CrossRef]
- Pham, T.D.; Verlander, J.W.; Wang, Y.; Romero, C.A.; Yue, Q.; Chen, C.; Thumova, M.; Eaton, D.C.; Lazo-Fernandez, Y.; Wall, S.M. Aldosterone regulates pendrin and epithelial sodium channel activity through intercalated cell mineralocorticoid receptor-dependent and -independent mechanisms over a wide range in serum potassium. J. Am. Soc. Nephrol. 2020, 31, 483–499. [Google Scholar] [CrossRef] [PubMed]
- Yamazaki, O.; Ishizawa, K.; Hirohama, D.; Fujita, T.; Shibata, S. Electrolyte transport in the renal collecting duct and its regulation by the renin-angiotensin-aldosterone system. Clin. Sci. (Lond.) 2019, 133, 75–82. [Google Scholar] [CrossRef]
- Kim, Y.C.; Mungunsukh, O.; McCart, E.A.; Roehrich, P.J.; Yee, D.K.; Day, R.M. Mechanism of erythropoietin regulation by angiotensin II. Mol. Pharmacol. 2014, 85, 898–908. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.C.; Mungunsukh, O.; Day, R.M. Erythropoietin regulation by angiotensin II. Vitam. Horm. 2017, 105, 57–77. [Google Scholar] [PubMed]
- Kato, H.; Ishida, J.; Imagawa, S.; Saito, T.; Suzuki, N.; Matsuoka, T.; Sugaya, T.; Tanimoto, K.; Yokoo, T.; Ohneda, O.; et al. Enhanced erythropoiesis mediated by activation of the renin-angiotensin system via angiotensin II type 1a receptor. Faseb. J. 2005, 19, 2023–2025. [Google Scholar] [CrossRef] [PubMed]
- Kato, H.; Ishida, J.; Matsusaka, T.; Ishimaru, T.; Tanimoto, K.; Sugiyama, F.; Yagami, K.; Nangaku, M.; Fukamizu, A. Erythropoiesis and blood pressure are regulated via at1 receptor by distinctive pathways. PLoS ONE 2015, 10, e0129484. [Google Scholar] [CrossRef] [PubMed]
- Cole, J.; Ertoy, D.; Lin, H.; Sutliff, R.L.; Ezan, E.; Guyene, T.T.; Capecchi, M.; Corvol, P.; Bernstein, K.E. Lack of angiotensin II-facilitated erythropoiesis causes anemia in angiotensin-converting enzyme-deficient mice. J. Clin. Investig. 2000, 106, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Kamper, A.L.; Nielsen, O.J. Effect of enalapril on haemoglobin and serum erythropoietin in patients with chronic nephropathy. Scand. J. Clin. Lab. Investig. 1990, 50, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Pratt, M.C.; Lewis-Barned, N.J.; Walker, R.J.; Bailey, R.R.; Shand, B.I.; Livesey, J. Effect of angiotensin converting enzyme inhibitors on erythropoietin concentrations in healthy volunteers. Br. J. Clin. Pharmacol. 1992, 34, 363–365. [Google Scholar] [CrossRef] [PubMed]
- Nagai, T.; Yasuoka, Y.; Izumi, Y.; Horikawa, K.; Kimura, M.; Nakayama, Y.; Uematsu, T.; Fukuyama, T.; Yamazaki, T.; Kohda, Y.; et al. Reevaluation of erythropoietin production by the nephron. Biochem. Biophys. Res. Commun. 2014, 449, 222–228. [Google Scholar] [CrossRef] [PubMed]
- WADA Science/EPO Working Group. Harmonization of Analysis and Reporting of Erythropoietin (EPO) and Other Epo-Receptor Agonists (ESAs) by Polyacrylamaide Gel Electrophoresis (PAGE) Analytical Methods. (TD2021EPO). 2021. Available online: https://www.wada-ama.org/sites/default/files/resources/files/td2021epo_final_eng_v_2.0.pdf (accessed on 1 September 2021).
- Higuchi, M.; Oh-eda, M.; Kuboniwa, H.; Tomonoh, K.; Shimonaka, Y.; Ochi, N. Role of sugar chains in the expression of the biological activity of human erythropoietin. J. Biol. Chem. 1992, 267, 7703–7709. [Google Scholar] [CrossRef]
- Kodama, D.; Nishimiya, D.; Iwata, K.; Yamaguchi, K.; Yoshida, K.; Kawabe, Y.; Motono, M.; Watanabe, H.; Yamashita, T.; Nishijima, K.; et al. Production of human erythropoietin by chimeric chickens. Biochem. Biophys. Res. Commun. 2008, 367, 834–839. [Google Scholar] [CrossRef] [PubMed]
- Lasne, F.; de Ceaurriz, J. Recombinant erythropoietin in urine. Nature 2000, 405, 635. [Google Scholar] [CrossRef] [PubMed]
- Lasne, F.; Thioulouse, J.; Martin, L.; de Ceaurriz, J. Detection of recombinant human erythropoietin in urine for doping analysis: Interpretation of isoelectric profiles by discriminant analysis. Electrophoresis 2007, 28, 1875–1881. [Google Scholar] [CrossRef] [PubMed]
- Reichel, C. SARCOSYL-PAGE: A new electrophoretic method for the separation and immunological detection of PEGylated proteins. Methods Mol. Biol. 2012, 869, 65–79. [Google Scholar]
- Yamaguchi, K.; Akai, K.; Kawanishi, G.; Ueda, M.; Masuda, S.; Sasaki, R. Effects of site-directed removal of N-glycosylation sites in human erythropoietin on its production and biological properties. J. Biol. Chem. 1991, 266, 20434–20439. [Google Scholar] [CrossRef]
- Yasuoka, Y.; Sato, Y.; Healy, J.M.; Nonoguchi, H.; Kawahara, K. pH-sensitive expression of calcium-sensing receptor (CaSR) in type-B intercalated cells of the cortical collecting ducts (CCD) in mouse kidney. Clin. Exp. Nephrol. 2015, 19, 771–782. [Google Scholar] [CrossRef]
- Kobayashi, M.; Yasuoka, Y.; Sato, Y.; Zhou, M.; Abe, H.; Kawahara, K.; Okamoto, H. Upregulation of calbindin D28k in the late distal tubules in the potassium-loaded adrenalectomized mouse kidney. Clin. Exp. Nephrol. 2011, 15, 355–362. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yasuoka, Y.; Izumi, Y.; Fukuyama, T.; Inoue, H.; Oshima, T.; Yamazaki, T.; Uematsu, T.; Kobayashi, N.; Shimada, Y.; Nagaba, Y.; et al. Effects of Angiotensin II on Erythropoietin Production in the Kidney and Liver. Molecules 2021, 26, 5399. https://doi.org/10.3390/molecules26175399
Yasuoka Y, Izumi Y, Fukuyama T, Inoue H, Oshima T, Yamazaki T, Uematsu T, Kobayashi N, Shimada Y, Nagaba Y, et al. Effects of Angiotensin II on Erythropoietin Production in the Kidney and Liver. Molecules. 2021; 26(17):5399. https://doi.org/10.3390/molecules26175399
Chicago/Turabian StyleYasuoka, Yukiko, Yuichiro Izumi, Takashi Fukuyama, Hideki Inoue, Tomomi Oshima, Taiga Yamazaki, Takayuki Uematsu, Noritada Kobayashi, Yoshitaka Shimada, Yasushi Nagaba, and et al. 2021. "Effects of Angiotensin II on Erythropoietin Production in the Kidney and Liver" Molecules 26, no. 17: 5399. https://doi.org/10.3390/molecules26175399
APA StyleYasuoka, Y., Izumi, Y., Fukuyama, T., Inoue, H., Oshima, T., Yamazaki, T., Uematsu, T., Kobayashi, N., Shimada, Y., Nagaba, Y., Mukoyama, M., Sato, Y., Sands, J. M., Kawahara, K., & Nonoguchi, H. (2021). Effects of Angiotensin II on Erythropoietin Production in the Kidney and Liver. Molecules, 26(17), 5399. https://doi.org/10.3390/molecules26175399





