Chemical Composition, Antioxidant and Anti-Inflammatory Activities of Clary Sage and Coriander Essential Oils Produced on Polluted and Amended Soils-Phytomanagement Approach
Abstract
:1. Introduction
2. Results
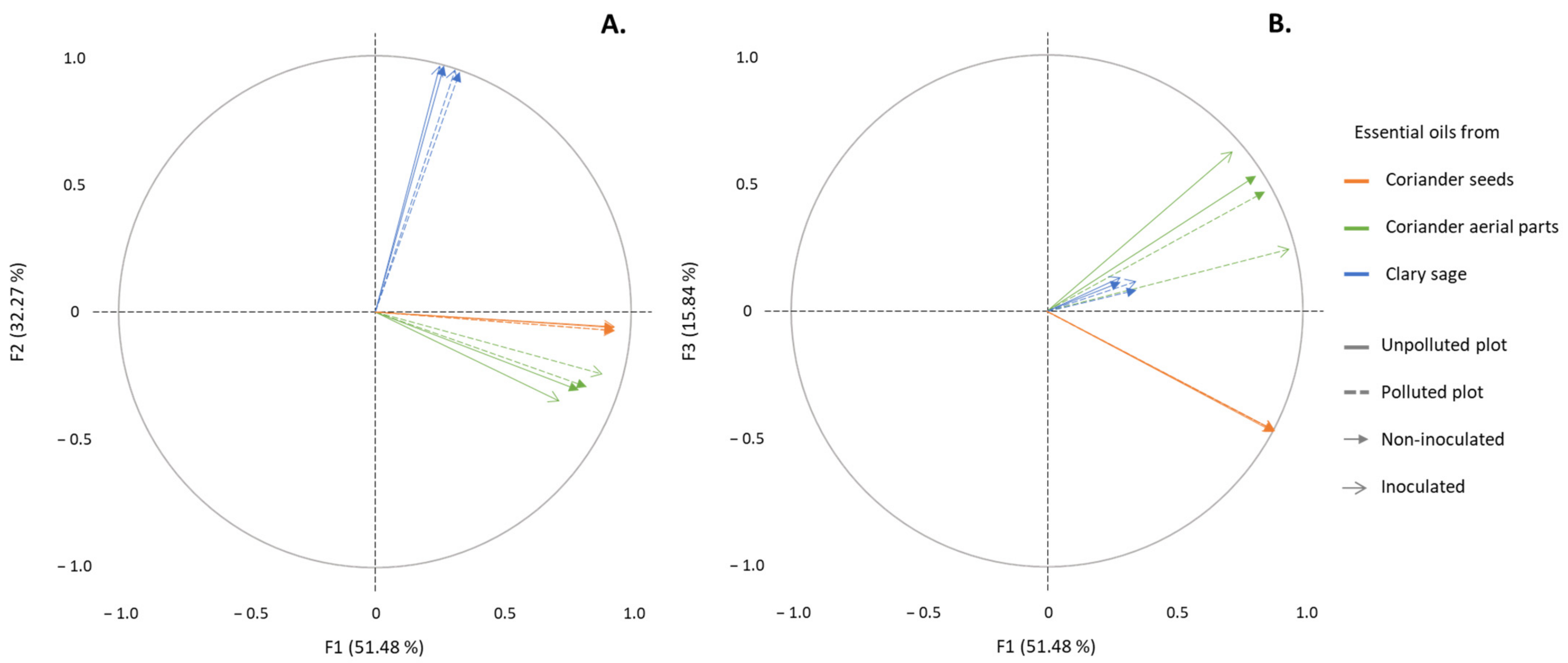
2.1. Determination of the EO Chemical Composition
2.2. Determination of the Retention Capacity of EO by β-CD
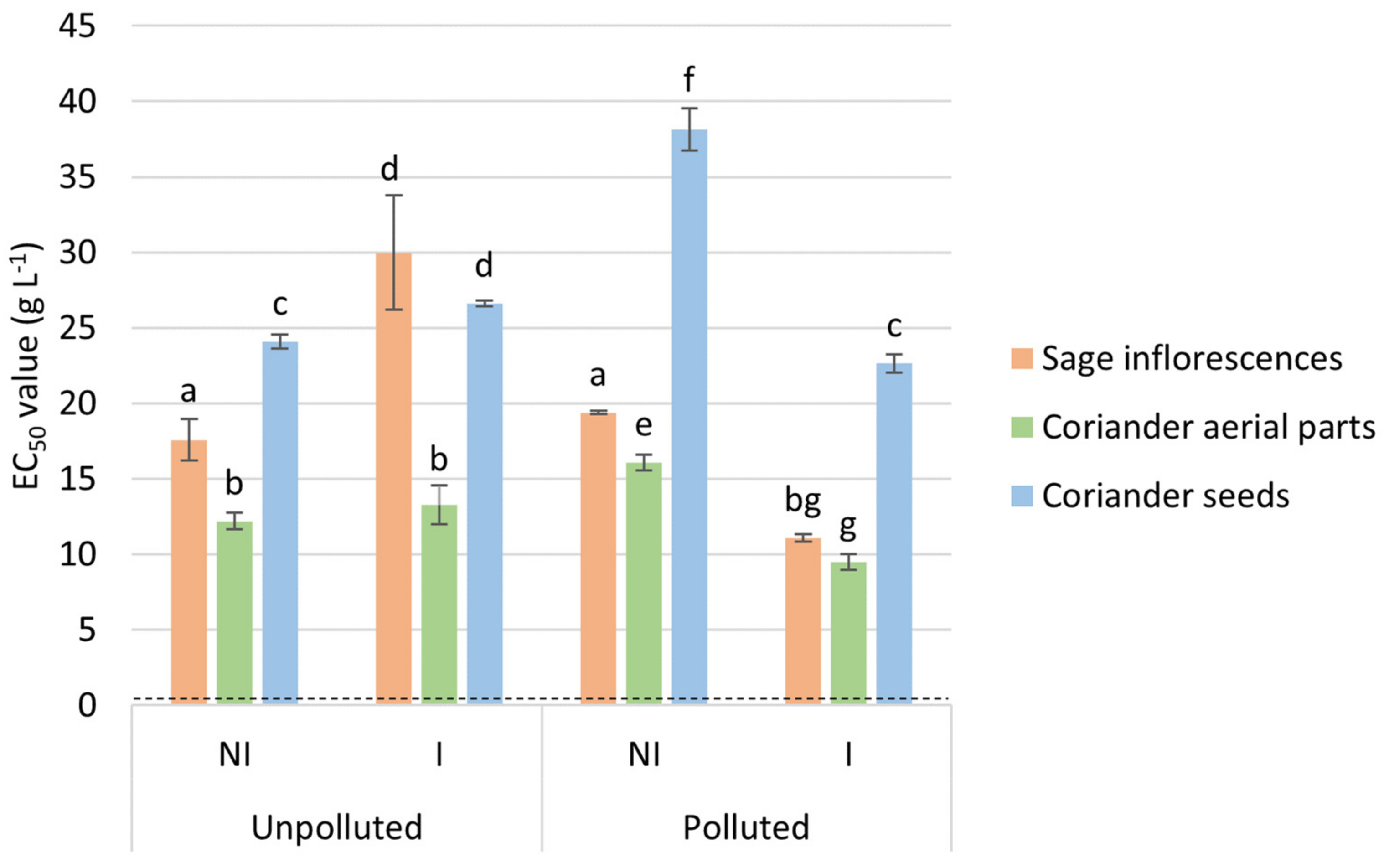
2.3. EO Antioxidant Activity
2.4. EO Anti-Inflammatory Activity
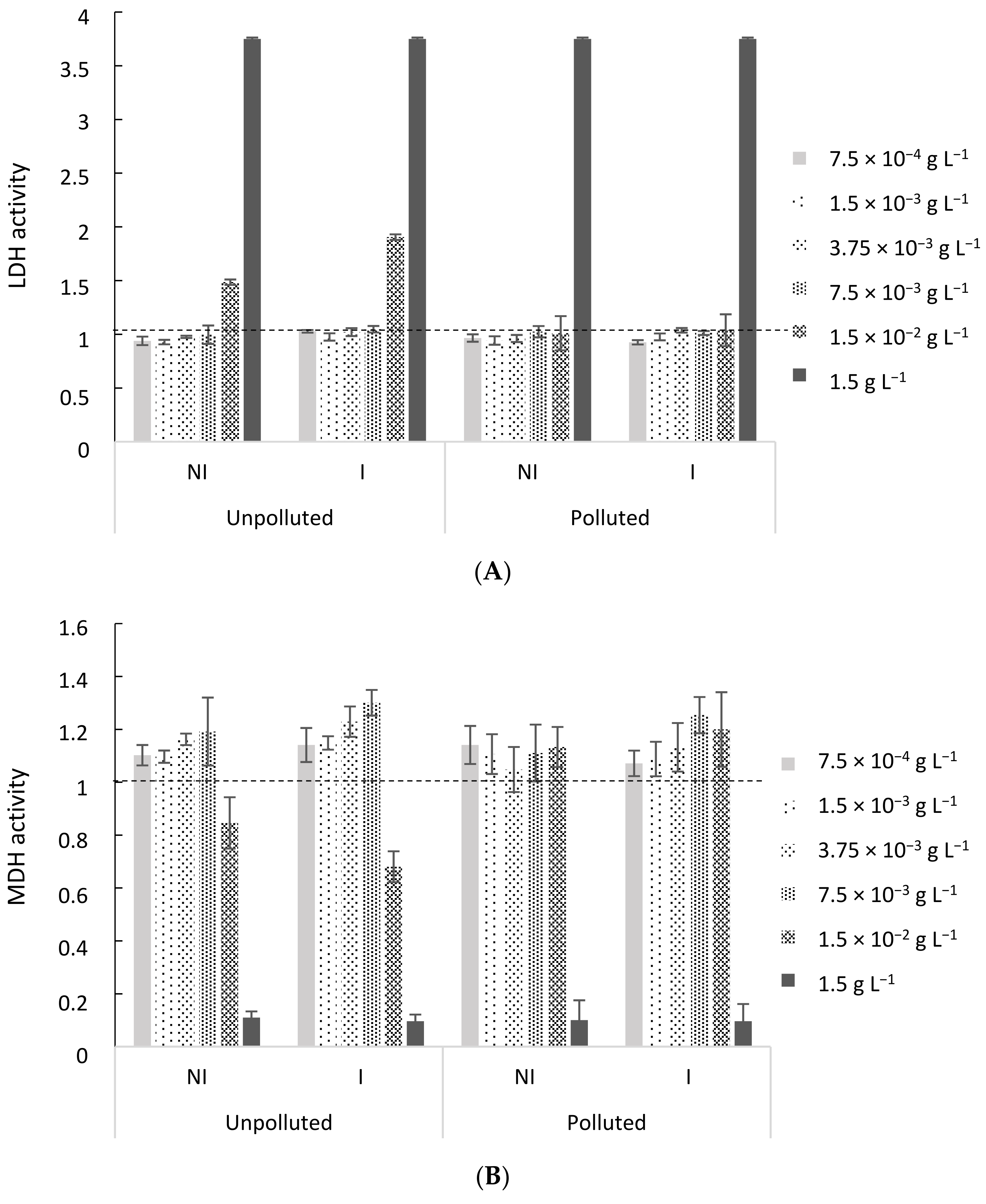
2.4.1. Cytotoxicity Assessment of EO
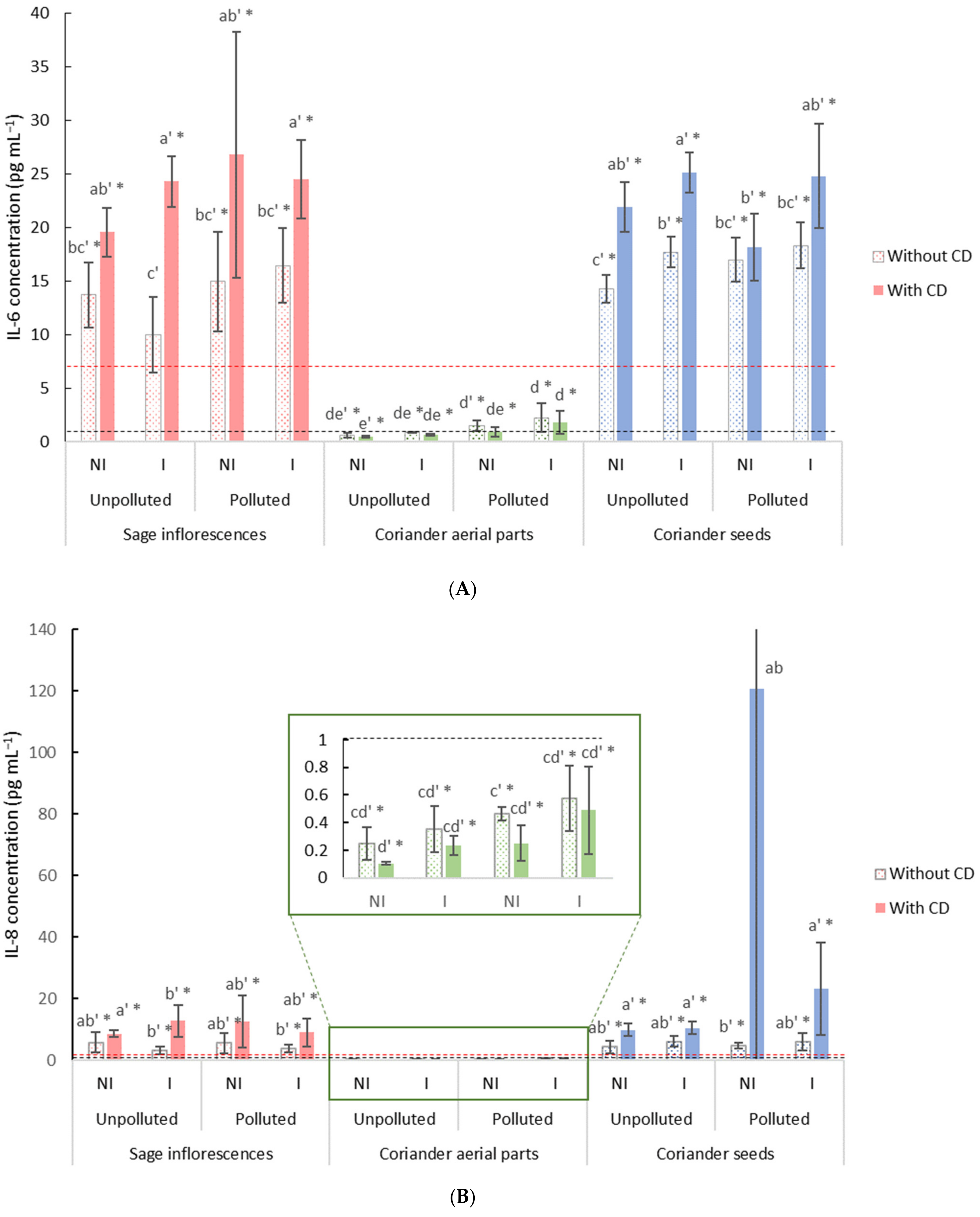
2.4.2. Anti-Inflammatory Properties of Free or Encapsulated EO
3. Discussion
3.1. Effects of Soil Pollution by TE and Mycorrhizal Inoculation on Chemical Compositions of EO Distilled from Clary Sage Inflorescences and Coriander Aerial Parts and Seeds
3.2. EO Potential Use as Natural Therapeutic Products and Insights on the Relationships between EO Composition and Their Biological Effects
4. Materials and Methods
4.1. Essential Oils
4.2. Determination of the EO Chemical Composition
4.3. Determination of the Retention Capacity of EO by β-CD
4.4. Biological Properties of EO
4.4.1. Antioxidant Activity
4.4.2. Anti-Inflammatory Activity
Cell Lines and Culture Conditions
EO Cytotoxicity Assessment
Cell Exposure for the Study of EO Anti-Inflammatory Potential
4.5. Statistical Analyses
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Burges, A.; Alkorta, I.; Epelde, L.; Garbisu, C. From phytoremediation of soil contaminants to phytomanagement of ecosystem services in metal contaminated sites. Int. J. Phytoremediation 2018, 20, 384–397. [Google Scholar] [CrossRef] [PubMed]
- Lydakis-Simantiris, N.; Fabian, M.; Skoula, M. Cultivation of Medicinal and Aromatic Plants in Heavy Metal-Contaminated Soils. Glob. Nest J. 2016, 18, 630–642. [Google Scholar] [CrossRef]
- Cundy, A.B.; Bardos, R.P.; Puschenreiter, M.; Mench, M.; Bert, V.; Friesl-Hanl, W.; Müller, I.; Li, X.N.; Weyens, N.; Witters, N.; et al. Brownfields to green fields: Realising wider benefits from practical contaminant phytomanagement strategies. J. Environ. Manag. 2016, 184, 67–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oustriere, N.; Marchand, L.; Bouchardon, J.L.; Faure, O.; Moutte, J.; Mench, M. Aided phytostabilization of a trace element-contaminated technosol developed on steel mill wastes. J. Hazard. Mater. 2016, 320, 458–468. [Google Scholar] [CrossRef]
- Garaiyurrebaso, O.; Garbisu, C.; Blanco, F.; Lanzén, A.; Martín, I.; Epelde, L.; Becerril, J.M.; Jechalke, S.; Smalla, K.; Grohmann, E.; et al. Long-term effects of aided phytostabilisation on microbial communities of metal-contaminated mine soil. FEMS Microbiol. Ecol. 2017, 93, 1–14. [Google Scholar] [CrossRef]
- Radziemska, M.; Koda, E.; Bilgin, A.; Vaverková, M.D. Concept of Aided Phytostabilization of Contaminated Soils in Postindustrial Areas. Int. J. Environ. Res. Public Health 2018, 15, 24. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Martínez, S.; Zornoza, R.; Gabarrón, M.; Gómez-Garrido, M.; Rosales, R.M.; Muñoz, M.A.; Gómez-López, M.D.; Soriano-Disla, J.M.; Faz, A.; Acosta, J.A. Is aided phytostabilization a suitable technique for the remediation of tailings? Eur. J. Soil Sci. 2019, 70, 862–875. [Google Scholar] [CrossRef]
- Njeru, E.M.; Avio, L.; Bocci, G.; Sbrana, C.; Turrini, A.; Bàrberi, P.; Giovannetti, M.; Oehl, F. Contrasting effects of cover crops on “hot spot” arbuscular mycorrhizal fungal communities in organic tomato. Biol. Fertil. Soils 2015, 51, 151–166. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D. Mycorrhizal Symbiosis; Academic Press: Cambridge, MA, USA, 2008; ISBN 9780123705266. [Google Scholar]
- Antoniadis, V.; Levizou, E.; Shaheen, S.M.; Ok, Y.S.; Sebastian, A.; Baum, C.; Prasad, M.N.V.; Wenzel, W.W.; Rinklebe, J. Trace elements in the soil-plant interface: Phytoavailability, translocation, and phytoremediation–A review. Earth Sci. Rev. 2017, 171, 621–645. [Google Scholar] [CrossRef]
- Gong, Y.; Zhao, D.; Wang, Q. An overview of field-scale studies on remediation of soil contaminated with heavy metals and metalloids: Technical progress over the last decade. Water Res. 2018, 147, 440–460. [Google Scholar] [CrossRef]
- Evangelou, M.W.H.; Papazoglou, E.G.; Robinson, B.H.; Schulin, R. Phytomanagement: Phytoremediation and the Production of Biomass for Economic Revenue on Contaminated Land. In Phytoremediation; Springer: Berlin/Heidelberg, Germany, 2015; pp. 115–132. [Google Scholar]
- Pandey, J.; Verma, R.K.; Singh, S. Suitability of aromatic plants for phytoremediation of heavy metal contaminated areas: A review. Int. J. Phytoremediation 2019, 21, 405–418. [Google Scholar] [CrossRef]
- Sim, L.Y.; Rani, N.Z.A.; Husain, K. Lamiaceae: An Insight on Their Anti-Allergic Potential and Its Mechanisms of Action. Front. Pharmacol. 2019, 10. [Google Scholar] [CrossRef]
- Singh, P.; Pandey, A.K. Prospective of Essential Oils of the Genus Mentha as Biopesticides: A Review. Front. Plant Sci. 2018, 9, 1–14. [Google Scholar] [CrossRef]
- Sá, R.A.; Alberton, O.; Gazim, Z.C.; Laverde, A., Jr.; Caetano, J.; Amorin, A.C.; Dragunski, D.C. Phytoaccumulation and effect of lead on yield and chemical composition of Mentha crispa essential oil. Desalination Water Treat. 2015, 53, 3007–3017. [Google Scholar] [CrossRef]
- Aćimović, M.; Kiprovski, B.; Rat, M.; Sikora, V.; Popović, V.; Koren, A.; Brdar-Jokanović, M. Salvia sclarea: Chemical compo-sition and biological activity. J. Agron. Technol. Eng. Manag. 2018, 1, 18–28. [Google Scholar]
- Blaskó, A.; Gazdag, Z.; Gróf, P.; Máté, G.; Sárosi, S.; Krisch, J.; Vágvölgyi, C.; Makszin, L.; Pesti, M. Effects of clary sage oil and its main components, linalool and linalyl acetate, on the plasma membrane of Candida albicans: An in vivo EPR study. Apoptosis 2017, 22, 175–187. [Google Scholar] [CrossRef]
- Bhuiyan, M.N.I.; Begum, J.; Sultana, M. Chemical composition of leaf and seed essential oil of Coriandrum sativum L. from Bangladesh. Bangladesh J. Pharmacol. 2009, 4, 150–153. [Google Scholar] [CrossRef]
- Freires, I.D.A.; Murata, R.M.; Furletti, V.F.; Sartoratto, A.; de Alencar, S.M.; Figueira, G.M.; Rodrigues, J.A.D.O.; Duarte, M.C.T.; Rosalen, P.L. Coriandrum sativum L. (Coriander) Essential Oil: Antifungal Activity and Mode of Action on Candida spp., and Molecular Targets Affected in Human Whole-Genome Expression. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [Green Version]
- Kumar, R.; Sharma, S.; Pathania, V. Effect of shading and plant density on growth, yield and oil composition of clary sage (Salvia sclarea L.) in north western Himalaya. J. Essent. Oil Res. 2013, 25, 23–32. [Google Scholar] [CrossRef]
- Biswas, S.; Koul, M.; Bhatnagar, A.K. Effect of Salt, Drought and Metal Stress on Essential Oil Yield and Quality in Plants. Nat. Prod. Commun. 2011, 6, 1559–1564. [Google Scholar] [CrossRef] [Green Version]
- Jezler, C.N.; De Almeida, A.F.; De Jesus, R.M.; Oliveira, R.A. De Pb and Cd on growth, leaf ultrastructure and essential oil yield mint (Mentha arvensis L.). Crop production 2015, 45, 392–398. [Google Scholar]
- Prasad, A.; Chand, S.; Kumar, S.; Chattopadhyay, A.; Patra, D.D. Heavy Metals Affect Yield, Essential Oil Compound, and Rhizosphere Microflora of Vetiver (Vetiveria zizanioides Linn. nash) Grass. Commun. Soil Sci. Plant Anal. 2014, 45, 1511–1522. [Google Scholar] [CrossRef]
- Golubkina, N.; Logvinenko, L.; Novitsky, M.; Zamana, S.; Sokolov, S.; Molchanova, A.; Shevchuk, O.; Sekara, A.; Tallarita, A.; Caruso, G. Yield, Essential Oil and Quality Performances of Artemisia dracunculus, Hyssopus officinalis and Lavandula angustifolia as Affected by Arbuscular Mycorrhizal Fungi under Organic Management. Plants 2020, 9, 375. [Google Scholar] [CrossRef] [Green Version]
- Khaosaad, T.; Vierheilig, H.; Nell, M.; Zitterl-Eglseer, K.; Novák, J. Arbuscular mycorrhiza alter the concentration of essential oils in oregano (Origanum sp., Lamiaceae). Mycorrhiza 2006, 16, 443–446. [Google Scholar] [CrossRef]
- Camele, I.; Elshafie, H.S.; Caputo, L.; De Feo, V. Anti-quorum Sensing and Antimicrobial Effect of Mediterranean Plant Essential Oils Against Phytopathogenic Bacteria. Front. Microbiol. 2019, 10, 1–6. [Google Scholar] [CrossRef]
- Michel, J.; Rani, N.Z.A.; Husain, K. A Review on the Potential Use of Medicinal Plants From Asteraceae and Lamiaceae Plant Family in Cardiovascular Diseases. Front. Pharmacol. 2020, 11, 1–26. [Google Scholar] [CrossRef]
- Zaïri, A.; Nouir, S.; Zarrouk, A.; Haddad, H.; Khélifa, A.; Achour, L.; Tangy, F.; Chaouachi, M.; Trabelsi, M. Chemical composition, Fatty acids profile and Biological properties of Thymus capitatus (L.) Hoffmanns, essential Oil. Sci. Rep. 2019, 9, 20134–20138. [Google Scholar] [CrossRef]
- Nascimento, L.D.D.; De Moraes, A.A.B.; Da Costa, K.S.; Galúcio, J.M.P.; Taube, P.S.; Costa, C.M.L.; Cruz, J.N.; Andrade, E.H.D.A.; De Faria, L.J.G. Bioactive Natural Compounds and Antioxidant Activity of Essential Oils from Spice Plants: New Findings and Potential Applications. Biomolecules 2020, 10, 988. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Camele, I. An Overview of the Biological Effects of Some Mediterranean Essential Oils on Human Health. BioMed Res. Int. 2017, 2017, 1–14. [Google Scholar] [CrossRef]
- Kostić, M.; Kitić, D.; Petrović, M.B.; Jevtović-Stoimenov, T.; Jović, M.; Petrović, A.; Živanović, S. Anti-inflammatory effect of the Salvia sclarea L. ethanolic extract on lipopolysaccharide-induced periodontitis in rats. J. Ethnopharmacol. 2017, 199, 52–59. [Google Scholar] [CrossRef]
- Kfoury, M.; Auezova, L.; Greige-Gerges, H.; Fourmentin, S. Promising applications of cyclodextrins in food: Improvement of essential oils retention, controlled release and antiradical activity. Carbohydr. Polym. 2015, 131, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.; Luís, Â.; Oleastro, M.; Domingues, F.C. Antioxidant properties of coriander essential oil and linalool and their potential to control Campylobacter spp. Food Control. 2016, 61, 115–122. [Google Scholar] [CrossRef]
- Abarca, R.L.; Rodríguez, F.J.; Guarda, A.; Galotto, M.J.; Bruna, J.E. Characterization of beta-cyclodextrin inclusion complexes containing an essential oil component. Food Chem. 2016, 196, 968–975. [Google Scholar] [CrossRef] [PubMed]
- Dima, C.; Cotarlet, M.; Tiberius, B.; Bahrim, G.; Alexe, P.; Dima, S. Encapsulation of coriander essential oil in beta-cyclodextrin: Antioxidant and antimicrobial properties evaluation. Rom. Biotechnol. Lett. 2014, 19, 9128–9141. [Google Scholar]
- Fourmentin, S.; Ciobanu, A.; Landy, D.; Wenz, G. Space filling of β-cyclodextrin and β-cyclodextrin derivatives by volatile hydrophobic guests. Beilstein J. Org. Chem. 2013, 9, 1185–1191. [Google Scholar] [CrossRef]
- Matasyoh, J.C.; Maiyo, Z.C.; Ngure, R.M.; Chepkorir, R. Chemical composition and antimicrobial activity of the essential oil of Coriandrum sativum. Food Chem. 2009, 113, 526–529. [Google Scholar] [CrossRef]
- Santoyo, S.; Jaime, L.; García-Risco, M.R.; Ruiz-Rodríguez, A.; Reglero, G. Antiviral Properties of Supercritical CO2 Extracts from Oregano and Sage. Int. J. Food Prop. 2014, 17, 1150–1161. [Google Scholar] [CrossRef] [Green Version]
- El-Zaeddi, H.; Martínez-Tomé, J.; Calín-Sánchez, Á.; Burló, F.; Carbonell-Barrachina, A. Volatile Composition of Essential Oils from Different Aromatic Herbs Grown in Mediterranean Regions of Spain. Foods 2016, 5, 41. [Google Scholar] [CrossRef] [Green Version]
- Raveau, R.; Fontaine, J.; Bert, V.; Perlein, A.; Tisserant, B.; Ferrant, P.; Sahraoui, A.L.H. In Situ cultivation of aromatic plant species for the phytomanagement of an aged-trace element polluted soil: Plant biomass improvement options and techno-economic assessment of the essential oil production channel. Sci. Total. Environ. 2021, 789, 147944. [Google Scholar] [CrossRef]
- Zutic, I.; Nitzan, N.; Chaimovitsh, D.; Schechter, A.; Dudai, N. Geographical location is a key component to effective breeding of clary sage (Salvia sclarea) for essential oil composition. Isr. J. Plant Sci. 2016, 63, 134–141. [Google Scholar] [CrossRef]
- Sharopov, F.S.; Setzer, W.N. The essential oil of Salvia sclarea L. from Tajikistan. Rec. Nat. Prod. 2012, 6, 75–79. [Google Scholar]
- Satyal, P.; Setzer, W.N. Chemical Compositions of Commercial Essential Oils from Coriandrum sativum Fruits and Aerial Parts. Nat. Prod. Commun. 2020, 15. [Google Scholar] [CrossRef]
- Chizzola, R. Essential Oil Composition of Wild Growing Apiaceae from Europe and the Mediterranean. Nat. Prod. Commun. 2010, 5, 1477–1492. [Google Scholar] [CrossRef] [Green Version]
- Mazloomifar, A.; Bigdeli, M.; Saber-Tehrani, M.; Rustaiyan, A.; Masoudi, S.; Ameri, N. Essential Oil of Prangos uloptera DC. from Iran. J. Essent. Oil Res. 2004, 16, 415–416. [Google Scholar] [CrossRef]
- Pavela, R.; Žabka, M.; Bednář, J.; Tříska, J.; Vrchotová, N. New knowledge for yield, composition and insecticidal activity of essential oils obtained from the aerial parts or seeds of fennel (Foeniculum vulgare Mill.). Ind. Crop. Prod. 2016, 83, 275–282. [Google Scholar] [CrossRef]
- Hazrati, S.; Rowshan, V.; Hosseini, S.J.; Sedaghat, M.; Mohammadi, H. Variation of Essential Oil Composition and Antioxidant Activity in Aerial Parts of Stachys Schtschegleevi Sosn at Different Growing Stages. J. Essent. Oil Bear. Plants 2020, 23, 1054–1071. [Google Scholar] [CrossRef]
- Amirmoradi, S.; Moghaddam, P.R.; Koocheki, A.; Danesh, S.; Fotovat, A. Effect of Cadmium and Lead on Quantitative and Essential Oil Traits of Peppermint (Mentha piperita L.). Not. Sci. Biol. 2012, 4, 101–109. [Google Scholar] [CrossRef] [Green Version]
- Lajayer, B.A.; Ghorbanpour, M.; Nikabadi, S. Heavy metals in contaminated environment: Destiny of secondary metabolite biosynthesis, oxidative status and phytoextraction in medicinal plants. Ecotoxicol. Environ. Saf. 2017, 145, 377–390. [Google Scholar] [CrossRef]
- Gautam, M.; Agrawal, M. Influence of metals on essential oil content and composition of lemongrass (Cymbopogon citratus (D.C.) Stapf.) grown under different levels of red mud in sewage sludge amended soil. Chemosphere 2017, 175, 315–322. [Google Scholar] [CrossRef]
- Angelova, V.R.; Ivanova, R.; Todorov, G.M.; Ivanov, K.I. Potential of Salvia sclarea L. for Phytoremediation of Soils Contaminated with Heavy Metals. Int. J. Agric. Biosyst. Eng. 2016, 10, 780–790. [Google Scholar]
- Ferrol, N.; Tamayo, E.; Vargas, P. The heavy metal paradox in arbuscular mycorrhizas: From mechanisms to biotechnological applications. J. Exp. Bot. 2016, 67, 6253–6265. [Google Scholar] [CrossRef] [Green Version]
- Harrison, M.J. Molecular and Cellular Aspects of the Arbuscular Mycorrhizal Symbiosis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1999, 50, 361–389. [Google Scholar] [CrossRef] [Green Version]
- Hause, B.; Fester, T. Molecular and cell biology of arbuscular mycorrhizal symbiosis. Planta 2005, 221, 184–196. [Google Scholar] [CrossRef]
- Kapoor, R.; Giri, B.; Mukerji, K.G. Mycorrhization of coriander (Coriandrum sativum L.) to enhance the concentration and quality of essential oil. J. Sci. Food Agric. 2002, 82, 339–342. [Google Scholar] [CrossRef]
- Janoušková, M.; Krak, K.; Vosátka, M.; Püschel, D.; Storchova, H. Inoculation effects on root-colonizing arbuscular mycorrhizal fungal communities spread beyond directly inoculated plants. PLoS ONE 2017, 12, e0181525. [Google Scholar] [CrossRef]
- Raveau, R.; Fontaine, J.; Hijri, M.; Sahraoui, A.L.-H. The Aromatic Plant Clary Sage Shaped Bacterial Communities in the Roots and in the Trace Element-Contaminated Soil More Than Mycorrhizal Inoculation—A Two-Year Monitoring Field Trial. Front. Microbiol. 2020, 11, 1–18. [Google Scholar] [CrossRef]
- Berka-Zougali, B.; Ferhat, M.-A.; Hassani, A.; Chemat, F.; Allaf, K.S. Comparative Study of Essential Oils Extracted from Algerian Myrtus communis L. Leaves Using Microwaves and Hydrodistillation. Int. J. Mol. Sci. 2012, 13, 4673–4695. [Google Scholar] [CrossRef] [Green Version]
- Ben Hsouna, A.; Hamdi, N.; Ben Halima, N.; Abdelkafi, S. Characterization of Essential Oil from Citrus aurantium L. Flowers: Antimicrobial and Antioxidant Activities. J. Oleo Sci. 2013, 62, 763–772. [Google Scholar] [CrossRef] [Green Version]
- Ben Marzoug, H.N.; Romdhane, M.; Lebrihi, A.; Mathieu, F.; Couderc, F.; Abderrabba, M.; Khouja, M.L.; Bouajila, J. Eucalyptus oleosa Essential Oils: Chemical Composition and Antimicrobial and Antioxidant Activities of the Oils from Different Plant Parts (Stems, Leaves, Flowers and Fruits). Molecules 2011, 16, 1695–1709. [Google Scholar] [CrossRef]
- Ye, C.-L.; Dai, D.-H.; Hu, W.-L. Antimicrobial and antioxidant activities of the essential oil from onion (Allium cepa L.). Food Control. 2013, 30, 48–53. [Google Scholar] [CrossRef]
- Miguel, M.G. Antioxidant and Anti-Inflammatory Activities of Essential Oils: A Short Review. Molecules 2010, 15, 9252–9287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dergham, M.; Lepers, C.; Verdin, A.; Cazier, F.; Billet, S.; Courcot, D.; Shirali, P.; Garçon, G. Temporal–spatial variations of the physicochemical characteristics of air pollution Particulate Matter (PM2.5–0.3) and toxicological effects in human bronchial epithelial cells (BEAS-2B). Environ. Res. 2015, 137, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Kfoury, M.; Borgie, M.; Verdin, A.; Ledoux, F.; Courcot, D.; Auezova, L.; Fourmentin, S. Essential oil components decrease pulmonary and hepatic cells inflammation induced by air pollution particulate matter. Environ. Chem. Lett. 2016, 14, 345–351. [Google Scholar] [CrossRef]
- Choi, H.-J.; Sowndhararajan, K.; Cho, N.-G.; Hwang, K.-H.; Koo, S.-J.; Kim, S. Evaluation of Herbicidal Potential of Essential Oils and their Components under In vitro and Greenhouse Experiments. Weed Turfgrass Sci. 2015, 4, 321–329. [Google Scholar] [CrossRef] [Green Version]
- Bakkali, F.; Averbeck, S.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef]
- Campos, E.; Proença, P.L.F.; Oliveira, J.L.; Pereira, A.; Ribeiro, L.N.D.M.; Fernandes, F.O.; Gonçalves, K.C.; Polanczyk, R.A.; Pasquoto-Stigliani, T.; Lima, R.; et al. Carvacrol and linalool co-loaded in β-cyclodextrin-grafted chitosan nanoparticles as sustainable biopesticide aiming pest control. Sci. Rep. 2018, 8, 7623. [Google Scholar] [CrossRef]
- Del Toro-Sánchez, C.L.; Zavala, J.F.A.; Machi, L.; Santacruz, H.; Villegas-Ochoa, M.A.; Alvarez-Parrilla, E.; González-Aguilar, G.A. Controlled release of antifungal volatiles of thyme essential oil from β-cyclodextrin capsules. J. Incl. Phenom. Macrocycl. Chem. 2010, 67, 431–441. [Google Scholar] [CrossRef]
- Kfoury, M.; Sahraoui, A.L.-H.; Bourdon, N.; Laruelle, F.; Fontaine, J.; Auezova, L.; Greige-Gerges, H.; Fourmentin, S. Solubility, photostability and antifungal activity of phenylpropanoids encapsulated in cyclodextrins. Food Chem. 2016, 196, 518–525. [Google Scholar] [CrossRef]
- Ciobanu, A.; Mallard, I.; Landy, D.; Brabie, G.; Nistor, D.; Fourmentin, S. Retention of aroma compounds from Mentha piperita essential oil by cyclodextrins and crosslinked cyclodextrin polymers. Food Chem. 2013, 138, 291–297. [Google Scholar] [CrossRef]
- Alipour, M.; Saharkhiz, M.J.; Niakousari, M.; Damyeh, M.S. Phytotoxicity of encapsulated essential oil of rosemary on germination and morphophysiological features of amaranth and radish seedlings. Sci. Hortic. 2018, 243, 131–139. [Google Scholar] [CrossRef]
- El-Alam, I.; Raveau, R.; Fontaine, J.; Verdin, A.; Laruelle, F.; Fourmentin, S.; Chahine, R.; Makhlouf, H.; Sahraoui, A.L.-H. Antifungal and Phytotoxic Activities of Essential Oils: In Vitro Assays and Their Potential Use in Crop Protection. Agronomy 2020, 10, 825. [Google Scholar] [CrossRef]
- De Figueiredo, R.; Marques, M.; Nakagawa, J.; Ming, L. Composition of Coriander Essential Oil From Brazil. Acta Hortic. 2004, 629, 135–137. [Google Scholar] [CrossRef]
- Kfoury, M.; Geagea, C.; Ruellan, S.; Greige-Gerges, H.; Fourmentin, S. Effect of cyclodextrin and cosolvent on the solubility and antioxidant activity of caffeic acid. Food Chem. 2019, 278, 163–169. [Google Scholar] [CrossRef]
- Cachon, B.F.; Firmin, S.; Verdin, A.; Ayi-Fanou, L.; Billet, S.; Cazier, F.; Martin, P.; Aissi, F.; Courcot, D.; Sanni, A.; et al. Proinflammatory effects and oxidative stress within human bronchial epithelial cells exposed to atmospheric particulate matter (PM2.5 and PM>2.5) collected from Cotonou, Benin. Environ. Pollut. 2014, 185, 340–351. [Google Scholar] [CrossRef]
- Piao, M.J.; Ahn, M.J.; Kang, K.A.; Ryu, Y.S.; Hyun, Y.J.; Shilnikova, K.; Zhen, A.X.; Jeong, J.W.; Choi, Y.H.; Kang, H.K.; et al. Particulate matter 2.5 damages skin cells by inducing oxidative stress, subcellular organelle dysfunction, and apoptosis. Arch. Toxicol. 2018, 92, 2077–2091. [Google Scholar] [CrossRef] [Green Version]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2019. [Google Scholar]
| Retention Indexes | Distilled Plant | Aerial Parts of Coriander | Seeds of Coriander | Sage Inflorescences | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Biomass Origin | Unpolluted | Polluted | Unpolluted | Polluted | Unpolluted | Polluted | ||||||||
| Exp. | Lit. | EO Compounds | NI | I | NI | I | NI | I | NI | I | NI | I | NI | I |
| 908 | 909 | α-pinene | 0.9 | 1.3 | 1.0 | 1.3 | 5.4 | 4.5 | 3.3 | 2.7 | - | - | - | - |
| 944 | 945 | Camphene | - | - | - | - | 0.6 | 0.5 | 0.4 | 0.3 | - | - | - | - |
| 991 | 991 | β-myrcene | - | - | - | - | - | - | - | - | 2.1 | 1.5 | 0.9 | 1.2 |
| 1005 | 1004 | 4-carene | - | - | - | - | 0.3 | - | - | 0.8 | 0.2 | 0.1 | 0.1 | 0.2 |
| 1027 | 1031 | Limonene | - | - | - | - | 1.7 | 1.6 | 1.3 | 1.7 | 0.8 | - | 0.5 | 0.1 |
| 1034 | 1034 | p-cymene | 0.3 | 0.3 | 0.2 | 1.1 | 1.6 | 1.6 | 1.3 | 1.2 | - | - | - | - |
| 1040 | 1036 | β-phellandrene | 0.7 | - | - | - | - | - | - | - | 0.2 | 0.1 | 0.1 | 0.1 |
| 1049 | 1051 | Ocimene | - | - | - | - | - | - | - | - | 0.7 | 0.6 | 0.4 | 0.7 |
| 1065 | 1066 | γ-terpinene | 1.6 | 1.9 | 1.4 | 1.8 | 8.9 | 8.6 | 6.6 | 8.4 | - | - | - | - |
| 1100 | 1103 | Linalool | 29.3 | 21.8 | 32.1 | 39.2 | 75.0 | 76.8 | 83.2 | 79.3 | 10.0 | 10.4 | 16.5 | 14.8 |
| 1133 | 1140 | Camphor | 1.3 | 1.0 | 1.4 | 1.7 | 3.7 | 3.5 | 3.6 | 3.7 | - | - | - | - |
| 1193 | 1200 | α-terpineol | - | - | - | - | - | - | - | - | 1.9 | 1.4 | 1.8 | 2.4 |
| 1205 | 1207 | Decanal | 6.6 | 7.9 | 7.2 | 8.0 | - | - | - | - | - | - | - | - |
| 1250 | 1253 | Linalyl acetate | - | - | - | - | - | - | - | - | 53.0 | 50.6 | 63.0 | 62.6 |
| 1274 | 1278 | (Z)-2-decenal | 48.0 | 51.4 | 47.1 | 37.9 | - | - | - | - | - | - | - | - |
| 1308 | 1310 | Undecanal | - | - | 3.5 | 0.9 | - | - | - | - | - | - | - | - |
| 1371 | 1371 | 2-undecenal | 1.2 | 1.7 | - | 1.2 | - | - | - | - | - | - | - | - |
| 1375 | 1377 | α-copaene | - | - | - | - | - | - | - | - | 3.6 | 4.5 | 1.9 | 2.0 |
| 1383 | 1383 | Geranyl acetate (cis) | - | - | - | - | - | - | - | - | 1.3 | 0.9 | 0.7 | 1.0 |
| 1386 | 1386 | Geranyl acetate (trans) | - | - | - | - | 1.6 | 3.0 | 0.3 | 1.8 | 2.5 | 2.1 | 1.5 | 2.5 |
| 1389 | 1390 | β-cubebene | - | - | - | - | - | - | - | - | 0.2 | - | 0.1 | 0.1 |
| 1414 | 1418 | β-caryophyllene | - | - | - | - | - | - | - | - | 3.2 | 3.9 | 2.6 | 2.0 |
| 1420 | 1420 | Dodecanal | 0.5 | 0.6 | 0.8 | 0.8 | - | - | - | - | - | - | - | - |
| 1427 | 1430 | β-copaene | - | - | - | - | - | - | - | - | 1.1 | 1.4 | 0.7 | 0.5 |
| 1447 | 1450 | β-farnesene | 1.5 | 1.4 | 0.3 | 0.0 | 0.8 | - | - | - | - | 0.1 | 0.1 | - |
| 1467 | 1468 | 2-dodecenal | 4.6 | 5.8 | 3.0 | 3.7 | - | - | - | - | - | - | - | - |
| 1479 | 1480 | Germacrene D | - | - | - | - | - | - | - | - | 14.8 | 17.1 | 6.7 | 7.3 |
| 1484 | 1484 | α-Humulene | - | - | - | - | - | - | - | - | 0.4 | 0.4 | 0.1 | 0.2 |
| 1515 | 1518 | Tridecanal | - | 0.9 | - | - | - | - | - | - | - | - | - | - |
| 1523 | 1523 | β-cadinene | - | - | - | - | - | - | - | - | 1.2 | 1.6 | 0.4 | 0.6 |
| 1551 | 1556 | Germacrene B | - | - | - | - | - | - | - | - | 1.7 | 2.0 | 0.9 | 0.9 |
| 1570 | 1571 | 2-tridecenal | 3.6 | 4.0 | 2.0 | 2.4 | - | - | - | - | - | - | - | - |
| 1580 | 1581 | Caryophyllene oxide | - | - | - | - | 0.2 | - | - | - | 0.3 | 0.4 | 0.2 | 0.2 |
| 1900 | 1900 | Sclareol oxide | - | - | - | - | - | - | - | - | 0.4 | 0.6 | 0.3 | 0.4 |
| 2220 | 2220 | Sclareol | - | - | - | - | - | - | - | - | 0.3 | 0.3 | 0.4 | 0.3 |
| Monoterpene hydrocarbons | 3.2 | 3.2 | 2.4 | 3.1 | 16.8 | 15.2 | 11.6 | 14.0 | 3.9 | 2.3 | 2.0 | 2.2 | ||
| Oxygenated monoterpenes | 30.6 | 22.8 | 33.5 | 40.9 | 80.5 | 83.2 | 87.1 | 84.8 | 69.0 | 65.3 | 83.5 | 83.2 | ||
| Oxygenated diterpene | - | - | - | - | - | - | - | - | 0.3 | 0.3 | 0.4 | 0.3 | ||
| Sesquiterpene hydrocarbons | 1.5 | 1.4 | 0.3 | - | 0.8 | - | - | - | 26.2 | 31.0 | 13.6 | 13.7 | ||
| Oxygenated sequiterpenes | - | - | - | - | - | - | - | - | - | - | - | - | ||
| Aromatic hydrocarbons | 0.3 | 0.3 | 0.2 | 1.1 | 1.6 | 1.6 | 1.3 | 1.2 | - | - | - | - | ||
| Aliphatic aldehydes | 64.4 | 72.3 | 63.5 | 54.9 | - | - | - | - | - | - | - | - | ||
| Terpenic oxide | - | - | - | - | 0.2 | - | - | - | 0.6 | 1.0 | 0.5 | 0.6 | ||
| Biomass Origin | Distilled Plant Part | Aerial Parts of Coriander | Seeds of Coriander | Sage Inflorescences |
|---|---|---|---|---|
| Unpolluted plot | NI | 74 ± 1.4 | 73 ± 1.0 | 63 ± 7.1 |
| I | 72 ± 1.4 | 74 ± 2.1 | 63 ± 5.7 | |
| Polluted plot | NI | 75 ± 2.8 | 76 ± 2.1 | 81 ± 3.5 * |
| I | 74 ± 1.4 | 73 ± 1.0 | 63 ± 7.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Raveau, R.; Fontaine, J.; Verdin, A.; Mistrulli, L.; Laruelle, F.; Fourmentin, S.; Lounès-Hadj Sahraoui, A. Chemical Composition, Antioxidant and Anti-Inflammatory Activities of Clary Sage and Coriander Essential Oils Produced on Polluted and Amended Soils-Phytomanagement Approach. Molecules 2021, 26, 5321. https://doi.org/10.3390/molecules26175321
Raveau R, Fontaine J, Verdin A, Mistrulli L, Laruelle F, Fourmentin S, Lounès-Hadj Sahraoui A. Chemical Composition, Antioxidant and Anti-Inflammatory Activities of Clary Sage and Coriander Essential Oils Produced on Polluted and Amended Soils-Phytomanagement Approach. Molecules. 2021; 26(17):5321. https://doi.org/10.3390/molecules26175321
Chicago/Turabian StyleRaveau, Robin, Joël Fontaine, Anthony Verdin, Loris Mistrulli, Frédéric Laruelle, Sophie Fourmentin, and Anissa Lounès-Hadj Sahraoui. 2021. "Chemical Composition, Antioxidant and Anti-Inflammatory Activities of Clary Sage and Coriander Essential Oils Produced on Polluted and Amended Soils-Phytomanagement Approach" Molecules 26, no. 17: 5321. https://doi.org/10.3390/molecules26175321
APA StyleRaveau, R., Fontaine, J., Verdin, A., Mistrulli, L., Laruelle, F., Fourmentin, S., & Lounès-Hadj Sahraoui, A. (2021). Chemical Composition, Antioxidant and Anti-Inflammatory Activities of Clary Sage and Coriander Essential Oils Produced on Polluted and Amended Soils-Phytomanagement Approach. Molecules, 26(17), 5321. https://doi.org/10.3390/molecules26175321







