Prickly Pear Seed Oil Extraction, Chemical Characterization and Potential Health Benefits
Abstract
1. Introduction
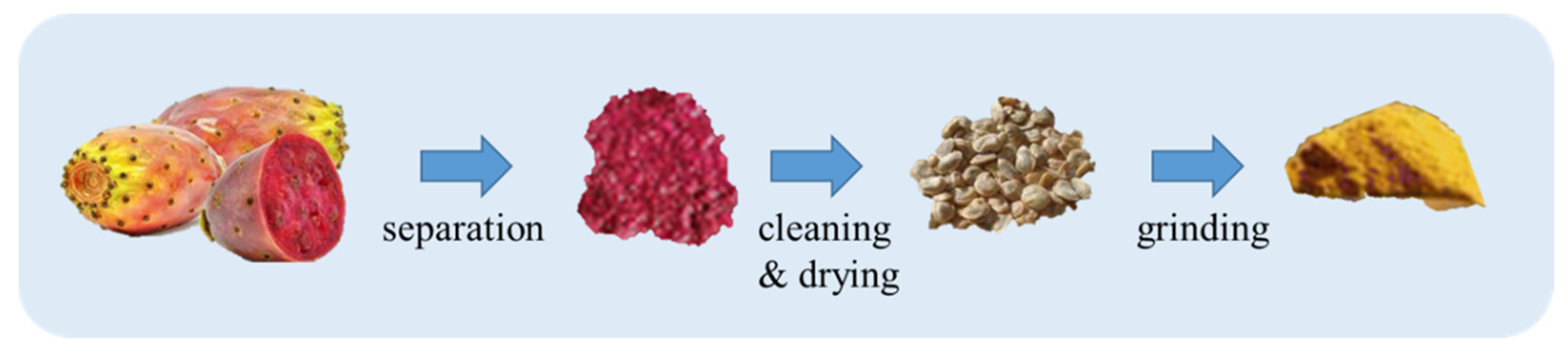
2. Prickly Pear Seed Oil Extraction and Yield
2.1. Conventional Extraction Methods
2.2. Innovative Techniques
3. Characteristics of Prickly Pear Seed Oil
4. Chemical Characterization of the Prickly Pear Seed Oil
4.1. Fatty Acid Composition
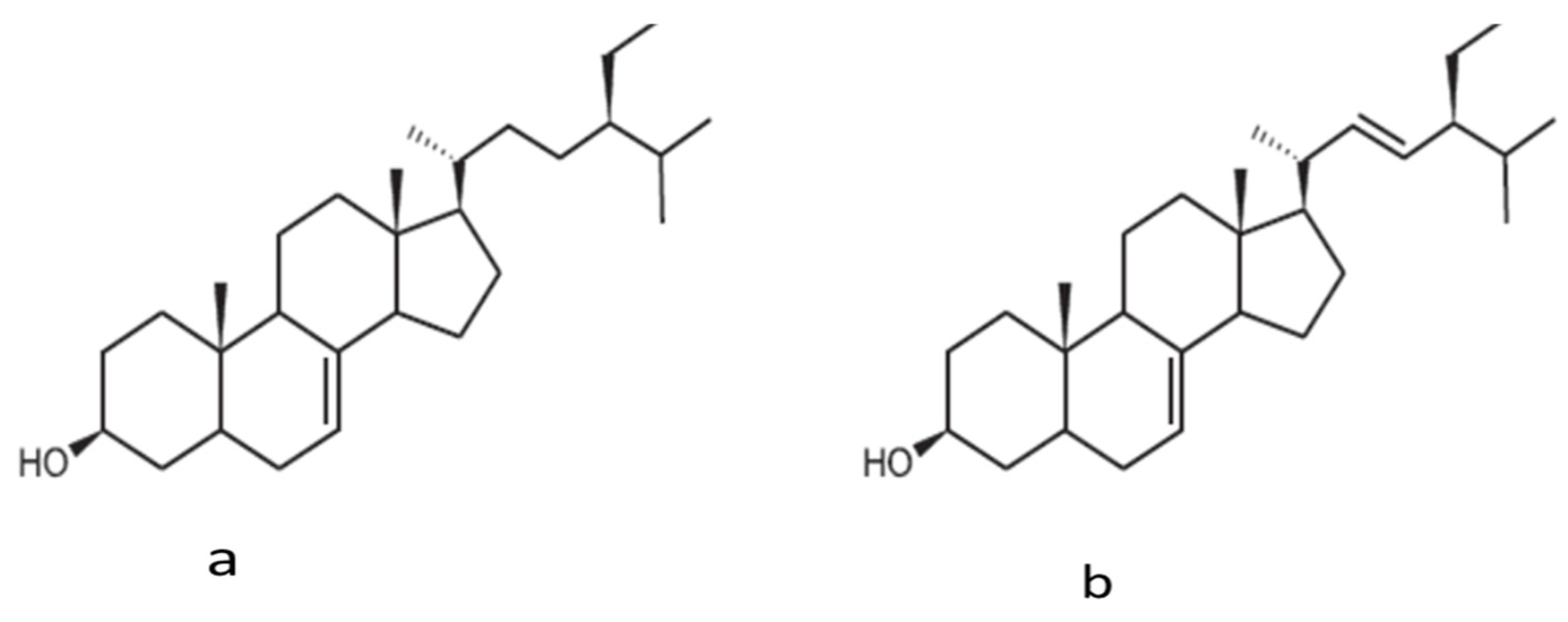
4.2. The Content of the Phytosterols and Tocopherols of Prickly Pear Seed Oil
4.3. Polyphenols, Flavonoid, Carotenoid, and Chlorophyll Contents of Prickly Pear Seed Oil
4.4. Prickly Pear Seed Oil Volatile Compounds
5. Potential Health Benefits of Prickly Pear Seeds Oil
5.1. Antioxidant Activity of Prickly Pear Seeds Oil
5.2. Antimicrobial Activity of Prickly Pear Seeds Oil
5.3. The Antidiabetic Properties of Prickly Pear Seeds Oil
5.4. Effect of Prickly Pear Seed Oil on Lipid Profile and Cholesterol Regulation
5.5. Cytotoxic and Apoptotic Effects of Prickly Pear Seed Oil
5.6. Anti-Inflammatory Effect of Prickly Pear Seed Oil
5.7. Anti-Ulcer Activity of Prickly Pear Seed Oil
5.8. The Effect of Prickly Pear Seed Oil against UV-C Radiation
5.9. Toxicity of Prickly Pear Seed Oil
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- El Mannoubi, I.; Barrek, S.; Skanji, T.; Casabianca, H.; Zarrouk, H. Characterization of Opuntia ficus indica seed oil from Tunisia. Chem. Nat. Compd. 2009, 45, 616. [Google Scholar] [CrossRef]
- Taoufik, F.; Zine, S.; El Hadek, M.; Idrissi Hassani, L.M.; Gharby, S.; Harhar, H.; Matthäus, B. Oil content and main constituents of cactus seed oils opuntia ficus indica of different origin in Morocco. Mediterr. J. Nutr. Metab. 2015, 8, 85–92. [Google Scholar] [CrossRef]
- Berraaouan, A.; Ziyyat, A.; Mekhfi, H.; Legssyer, A.; Sindic, M.; Aziz, M.; Bnouham, M. Evaluation of antidiabetic properties of cactus pear seed oil in rats. Pharm. Biol. 2014, 52, 1286–1290. [Google Scholar] [CrossRef]
- Ramadan, M.F.; Mörsel, J.-T. Oil cactus pear (Opuntia-indica L.). Food Chem. 2003, 82, 339–345. [Google Scholar] [CrossRef]
- Berraaouan, A.; Abderrahim, Z.; Hassane, M.; Abdelkhaleq, L.; Mohammed, A.; Mohamed, B. Evaluation of protective effect of cactus pear seed oil (Opuntia ficus-indica L. MILL.) against alloxan-induced diabetes in mice. Asian Pac. J. Trop. Med. 2015, 8, 532–537. [Google Scholar] [CrossRef]
- Al-Naqeb, G. Effect of prickly pear cactus seeds oil on the blood glucose level of streptozotocin-induced diabetic rats and its molecular mechanisms. Int. J. Herb. Med. 2015, 3, 29–34. [Google Scholar]
- Karabagias, V.K.; Karabagias, I.K.; Gatzias, I.; Badeka, A.V. Prickly Pear Seed Oil by Shelf-Grown Cactus Fruits: Waste or Maste? Processes 2020, 8, 132. [Google Scholar] [CrossRef]
- Özcan, M.M.; Al Juhaimi, F.Y. Nutritive value and chemical composition of prickly pear seeds (Opuntia ficus indica L.) growing in Turkey. Int. J. Food Sci. Nutr. 2011, 62, 533–536. [Google Scholar] [CrossRef] [PubMed]
- Bardaa, S.; Turki, M.; Ben Khedir, S.; Mzid, M.; Rebai, T.; Ayadi, F.; Sahnoun, Z. The Effect of Prickly Pear, Pumpkin, and Linseed Oils on Biological Mediators of Acute Inflammation and Oxidative Stress Markers. BioMed Res. Int. 2020, 2020, 5643465. [Google Scholar] [CrossRef]
- Khémiri, I.; Hédi, B.E.; Zouaoui, N.S.; Gdara, N.B.; Bitri, L. The Antimicrobial and Wound Healing Potential of Opuntia ficus indica L. inermis Extracted Oil from Tunisia. Evid. Based Complement. Altern. Med. 2019, 2019, 9148782. [Google Scholar] [CrossRef]
- Ortega-Ortega, M.D.; Cruz-Cansino, N.D.; Alanís-García, E.; Delgado-Olivares, L.; Ariza-Ortega, J.A.; Ramírez-Moreno, E.; Manríquez-Torres, J.D. Optimization of ultrasound extraction of cactus pear (Opuntia ficus indica) seed oil based on antioxidant activity and evaluation of its antimicrobial activity. J. Food Qual. 2017, 2017, 9315360. [Google Scholar] [CrossRef]
- AbdelFattah, M.S.; Badr, S.E.A.; Khalil, E.M.; Elsaid, A.S. Feed efficiency, some blood parameters and In-vitro chemoprevention of prickly pear (Opuntia ficus indica L.) seeds oil growing in Egypt. IBSPR 2020, 8, 20–28. [Google Scholar]
- Ramírez-Moreno, E.; Cariño-Cortés, R.; Cruz-Cansino, N.D.; Delgado-Olivares, L.; Ariza-Ortega, J.A.; Montañez-Izquierdo, V.Y.; Hernández-Herrero, M.M.; Filardo-Kerstupp, T. Antioxidant and antimicrobial properties of cactus pear (Opuntia) seed oils. J. Food Qual. 2017, 2017, 3075907. [Google Scholar] [CrossRef]
- Benattia, F.K.; Arrar, Z.; Khabba, Y. Evaluation the Hypo and Antihyperglycemic Activity of Cactus Seeds Extracts (Opuntia ficus-indica L.). PCBS J. 2017, 11. [Google Scholar] [CrossRef]
- Ennouri, M.; Fetoui, H.; Bourret, E.; Zeghal, N.; Attia, H. Evaluation of some biological parameters of Opuntia ficus indica. 1. Influence of a seed oil supplemented diet on rats. Bioresour. Technol. 2006, 97, 1382–1386. [Google Scholar]
- Becer, E.; Kabadayı, H.; Meriçli, F.; Meriçli, A.H.; Kıvançlı, B.; Vatansever, S. Apoptotic Effects of Opuntia ficus indica L. Seed Oils on Colon Adenocarcinoma Cell Lines. Proceedings 2018, 2, 1566. [Google Scholar] [CrossRef]
- Khémiri, I.; Bitri, L. Effectiveness of Opuntia ficus indica L. inermis Seed Oil in the Protection and the Healing of Experimentally Induced Gastric Mucosa Ulcer. Oxid. Med. Cell. Longev. 2019, 2019, 1568720. [Google Scholar] [CrossRef] [PubMed]
- Regalado-Rentería, E.; Aguirre-Rivera, J.R.; González-Chávez, M.M.; Sánchez-Sánchez, R.; Martínez-Gutiérrez, F.; Juárez-Flores, B.I. Assessment of Extraction Methods and Biological Value of Seed Oil from Eight Variants of Prickly Pear Fruit (Opuntia spp.). Waste Biomass Valoriz. 2020, 11, 1181–1189. [Google Scholar] [CrossRef]
- Nounah, I.; Chbani, M.; Matthäus, B.; Charrouf, Z.; Hajib, A.; Willenberg, I. Profile of Volatile Aroma-Active Compounds of Cactus Seed Oil (Opuntia ficus-indica) from Different Locations in Morocco and Their Fate during Seed Roasting. Foods 2020, 9, 1280. [Google Scholar] [CrossRef]
- PNG1ib. Available online: https://www.pnglib.com/prickly-pear-cactus-fruit/ (accessed on 13 June 2021).
- ExportersIndia.com. Natural Product. Available online: https://www.exportersindia.com/natural-product2230260/prickly-pear-seeds-vadodara-india-728257.htm (accessed on 15 June 2021).
- Gharby, S.; Ravi, H.K.; Guillaume, D.; Vian, M.A.; Chemat, F.; Charrouf, Z. 2-methyloxolane as alternative solvent for lipid extraction and its effect on the cactus (Opuntia ficus-indica L.) seed oil fractions. OCL 2020, 27, 1–7. [Google Scholar] [CrossRef]
- Kolniak-Ostek, J.; Kita, A.; Miedzianka, J.; Andreu-Coll, L.; Legua, P.; Hernandez, F. Characterization of Bioactive Compounds of Opuntia ficus-indica (L.) Mill. Seeds from Spanish Cultivars. Molecules 2020, 25, 5734. [Google Scholar] [CrossRef]
- Matthäus, B.; Özcan, M.M. Habitat effects on yield, fatty acid composition and tocopherol contents of prickly pear (Opuntia ficus-indica L.) seed oils. Sci. Hortic. 2011, 131, 95–98. [Google Scholar] [CrossRef]
- Boukeloua, A.; Belkhiri, A.; Djerrou, Z.; Bahri, L.; Boulebda, N.; Hamdi Pacha, Y. Acute toxicity of Opuntia ficus indica and Pistacia lentiscus seed oils in mice. Afr. J. Tradit. Complement. Altern. Med. 2012, 9, 607–611. [Google Scholar] [CrossRef]
- Chougui, N.; Tamendjari, A.; Hamidj, W.; Hallal, S.; Barras, A.; Richard, T.; Larbat, R. Oil composition and characterisation of phenolic compounds of Opuntia ficus-indica seeds. Food Chem. 2013, 139, 796–803. [Google Scholar] [CrossRef]
- Ennouri, M.; Evelyne, B.; Laurence, M.; Hamadi, A. Fatty acid composition and rheological behaviour of prickly pear seed oils. Food Chem. 2005, 93, 431–437. [Google Scholar] [CrossRef]
- Yeddes, N.; Chérif, J.K.; Jrad, A.; Barth, D.; Trabelsi-Ayadi, M. Supercritical SC-CO2 and Soxhlet n-Hexane Extract of Tunisian Opuntia ficus indica Seeds and Fatty Acids Analysis. J. Lipids 2012, 2012, 914693. [Google Scholar] [CrossRef] [PubMed]
- AbdelFattah, M.S.; Badr, S.E.A.; Salah Elsaid, A. Nutritive value and chemical composition of prickly pear seeds (Opuntia ficus indica L.) growing in Egypt. IJAPR 2020, 8, 1–10. [Google Scholar]
- Sawaya, W.N.; Khatchadourian, H.A.; Safi, W.M.; Al-Muhamma, H.M. Chemical characterisation of prickly pear pulp, Opuntia ficus-indica, and the manufacturing of prickly pear jam. J. Food Technol. 1983, 18, 183–193. [Google Scholar] [CrossRef]
- De Wit, M.; Hugo, A.; Shongwe, N. South African Cactus Pear Seed Oil: A Comprehensive Study on 42 Spineless Burbank Opuntia ficus-indica and Opuntia robusta Cultivars. Eur. J. Lipid Sci. Technol. 2017, 120, 1700343. [Google Scholar] [CrossRef]
- De Wit, M.; Hugo, A.; Shongwe, N.; Van der Merwe, R. Effect of cultivar, season and locality on lipid content and fatty acid composition of cactus pear seed oil. S. Afr. J. Plant Soil 2016, 33, 279–288. [Google Scholar] [CrossRef]
- Khaled, S.; Dahmoune, F.; Madani, K.; Urieta, J.; Mainar, A. Supercritical fractionation of antioxidants from Algerian Opuntia ficus-indica (L.) Mill. seeds. J. Food Process. Preserv. 2020, 44, e14343. [Google Scholar] [CrossRef]
- Rgui, L.; Casas, L.; Fernández, M.T.; Mantell, C.; Fadly, Z.; Metni, R.; de la Ossa, E.M. Supercritical fluid extraction of oil obtained from Opuntia ficus indica. In Proceedings of the Chemistry, Chemical Engineering and Biotechnology, Madrid, Spain, 1–4 July 2014. [Google Scholar]
- Zito, P.; Sajeva, M.; Bruno, M.; Rosselli, S.; Maggio, A.; Senatore, F. Essential oils composition of two Sicilian cultivars of Opuntia ficus-indica (L.) Mill. (Cactaceae) fruits (prickly pear). Nat. Prod. Res. 2013, 27, 1305–1314. [Google Scholar] [CrossRef]
- Brahmi, F.; Haddad, S.; Bouamara, K.; Yalaoui-Guellal, D.; Prost-Camus, E.; Barros, J.P.; Prost, M.; Atanasov, A.; Madani, K.; Boulekbache-Makhlouf, L.; et al. Comparison of chemical composition and biological activities of Algerian seed oils of Pistacia lentiscus L., Opuntia ficus indica (L.) mill. and Argania spinosa L. Skeels. Ind. Crops Prod. 2020, 151, 112456. [Google Scholar] [CrossRef]
- Harhar, H.; Gharby, S.; Guillaume, D.; Charrouf, Z. Effect of argan kernel storage conditions on argan oil quality. Eur. J. Lipid Sci. Technol. 2010, 112, 915–920. [Google Scholar] [CrossRef]
- Ciriminna, R.; Bongiorno, D.; Scurria, A.; Danzì, C.; Timpanaro, G.; Delisi, R.; Avellone, G.; Pagliaro, M. Sicilian Opuntia ficus-indica Seed Oil: Fatty Acid Composition and Bio-Economical Aspects. Eur. J. Lipid Sci. Technol. 2017, 119, 1700232. [Google Scholar] [CrossRef]
- Shahidi, F.; de Camargo, A.C. Tocopherols and Tocotrienols in Common and Emerging Dietary Sources: Occurrence, Applications, and Health Benefits. Int. J. Mol. Sci. 2016, 17, 1745. [Google Scholar] [CrossRef]
- Azadmard-Damirchi, S. Review of the use of phytosterols as a detection tool for adulteration of olive oil with hazelnut oil. Food Addit. Contam. 2010, 27, 1–10. [Google Scholar] [CrossRef] [PubMed]
- El Kharrassi, Y.; El Maaidan, E.; Chakhchar, A.; Lamaoui, M.; Bourhim, T.; Mabrouk, A.; Essamadi, A.K.; Moustaid, K.; Nasser, B. Physicochemical, phytochemical and antioxidant properties of juice and seed oil of cactus pear Opuntia aequatorialis and Opuntia leucotricha. Vegetos 2020, 33, 682–689. [Google Scholar] [CrossRef]
- El Kharrassi, Y.; Samadi, M.; Lopez, T.; Nury, T.; El Kebbaj, R.; Andreoletti, P.; El Hajj, H.I.; Vamecq, J.; Moustaid, K.; Latruffe, N.; et al. Biological activities of Schottenol and Spinasterol, two natural phytosterols present in argan oil and in cactus pear seed oil, on murine miroglial BV2 cells. Biochem. Biophys. Res. Commun. 2014, 446, 798–804. [Google Scholar] [CrossRef] [PubMed]
- Chaalal, M.; Touati, N.; Louaileche, H. Extraction of phenolic compounds and in vitro antioxidant capacity of prickly pear seeds. Acta Bot. Gall. 2012, 159, 467–475. [Google Scholar] [CrossRef]
- Chbani, M.; Matthäus, B.; Charrouf, Z.; El Monfalouti, H.; Kartah, B.; Gharby, S.; Willenberg, I. Characterization of Phenolic Compounds Extracted from Cold Pressed Cactus (Opuntia ficus-indica L.) Seed Oil and the Effect of Roasting on Their Composition. Foods 2020, 9, 1098. [Google Scholar] [CrossRef]
- Nazzaro, F.; Fratianni, F.; d’Acierno, A.; Caputo, L.; De Feo, V.; Coppola, R. Antibiofilm properties exhibited by the prickly pear (Opuntia ficus-indica) seed oil. Proceedings 2020, 66, 29. [Google Scholar] [CrossRef]
- González Barra, I.; Escobar, M.; Villarroel Guerra, M. Influence of intake of vegetables oils on the in vitro intestinal absorption of glucose in golden hamster. Arch. Latinoam. Nutr. 1995, 45, 290–294. [Google Scholar]
- Ennouri, M.; Fetoui, H.; Hammami, M.; Bourret, E.; Hamadi Attia, H.; Zeghal, N. Effects of diet supplementation with cactus pear seeds and oil on serum and liver lipid parameters in rats. Food Chem. 2007, 101, 248–253. [Google Scholar] [CrossRef]
- Camarena-Ordonez, D.R.; Gutierrez-Uribe, J.; Hernandez-Brenes, C.; Mertens-Talcott, S. Prickly pear seed extracts with antioxidant and cancer-preventing capacity. FASEB J. 2010, 24, 928-17. [Google Scholar] [CrossRef]
- Ait El Cadi, M.; Makram, S.; Ansar, M.; Khabbal, Y.; Alaoui, K.; Faouzi, M.A.; Cherrah, Y.; Taoufik, J. Anti-inflammatory activity of aqueous and ethanolic extracts of Zygophyllum gaetulum. Ann. Pharm. Fr. 2012, 70, 113–116. [Google Scholar] [CrossRef]
- Morimoto, A.; Nakamori, T.; Watanabe, T.; Ono, T.; Murakami, N. Pattern differences in experimental fevers induced by endotoxin, endogenous pyrogen, and prostaglandins. Am. J. Physiol. 1988, 254, 633–640. [Google Scholar] [CrossRef]

| Extraction Technique | Method | Yield (%) | Reference |
|---|---|---|---|
| Solvent Extraction | |||
| 2-Methyloxolane and n-hexane | Soxhlet system for 8 h | 9.55 ± 0.12 2-methyloxolane 8.86 ± 0.25 n-hexane | [22] |
| Diethyl ether | Soxhlet system for 3 h | 2.61–7.69 | [23] |
| Methanol | Soxhlet system for several h | 16.20 | [29] |
| n-Hexane | Soxhlet apparatus for 4–6 h | 5.40 ± 0.5 | [7] |
| n-Hexane | Soxhlet system | NR | [11] |
| n-Hexane | Soxhlet system for 8 h | 5.40–9.9 | [2] |
| n-Hexane | Soxhlet system for 18 h | 10.45 ± 0.10 | [25] |
| n-Hexane | Soxhlet system | 8.91–10.32 | [28] |
| n-Hexane | Soxhlet system for 9 h at 120 °C | 7.30–9.3 | [26] |
| n-Hexane | Soxhlet system for 10 h | 11.75 | [1] |
| n-Hexane | Soxhlet system for 9 h | 11.00 | [27] |
| Petroleum ether | Soxhlet system for 6 h at 50 °C | 5.00–14.4 | [8] |
| Petroleum ether | Soxhlet system | 17.20 as total fat | [30] |
| Maceration Extraction with Solvents | |||
| n-Hexane | Macerating for 24 h | 6.2 ± 0.3–15.5 ± 0.5 | [18] |
| Different solvents, n-hexane, ethanol, and ethyl acetate | Mixing the powdered seeds with solvents | hexane yield 11.8 ethanol and ethyl acetate 10.00 | [13] |
| n-Hexane | Powdered seeds were immersed in hexane at 25 °C in a dark place for 24 h | NR | [11] |
| Chloroform and methanol 2:1 | Powdered seeds were immersed in chloroform and methanol 2:1 while adding BHT at a concentration of 0.001% | 4.1–8.8 | [31] |
| Chloroform and methanol 2:1 | Powdered seeds were immersed in chloroform and methanol 2:1 with adding BHT at a concentration of 0.001% | 5.4–6.7 | [32] |
| n-Hexane, petroleum ether and chloroform-methanol (2:1 v/v) | Maceration with solvents 1:5 (w/v): at room temperature for 24 h | Chloroform: methanol produced a yield of 7.8 ± 0.4 whereas, the yield extracted using hexane and petroleum ether were 5.0 ± 0.4 and 6.1 ± 0.4, respectively | [6] |
| Chloroform and methanol 2:1 | Powdered seeds were immersed in chloroform and methanol 2:1 | 9.9 | [4] |
| Mechanical Extraction | |||
| Electric powered screw press | The extraction process was followed by centrifugation of the oil for 15 min at 3000 rpm to separate from the sediment | NR | [19] |
| Cold pressure | first cold pressure using a mechanical oil press | NR | [9] |
| Cold press methods | Compression of 20 t was applied on the piston by means of a hydraulic press | 0.5–6.1 | [18] |
| Cold pressing | first cold pressing using a mechanical machine | NR | [19] |
| Supercritical Fluid Extraction Method | |||
| Supercritical fluid extraction method | Optimal supercritical antisolvent fractionation conditions were carried out at best conditions of 15 MPa pressure and 30 g min−1 CO2 flow rate | The recovery extract was rich in catechin, epicatechin, and ferulic acid | [34] |
| Supercritical fluid extraction method | Best conditions at pressure of 500 bar, temperature at 35 °C, extraction time 80 min and CO2 flow rate of 20 g/min | 6.5 | [33] |
| Supercritical fluid extraction method | Extraction was carried out at different temperatures and fixed pressure. | 1.9 (Cultivated) 3.4 (wild) | [28] |
| Ultrasound extraction methods | Ultrasound at 1500 W with a constant frequency of 20 kHz and a probe of 25 mm for 5–15 min with a fixed temperature of 25 °C | 3.8–6.0 | [11] |
| Microextraction | |||
| Microwave | Extraction process of PPSO was carried out by mixing about 20 mg of powder seeds with 1 mL of isooctane, then the mixture was put in a microwave oven at 100 °C, 150 w during 13 min, then filtered, then the oil further processed by transesterification and salinization | NR | [18] |
| Hydrodistillation using Clevenger-type apparatus | Ground seeds were extracted with n-hexane as a solvent and a Clevenger-type apparatus | 0.6–3.0 | [35] |
| Reported Parameters | References | ||||
|---|---|---|---|---|---|
| [22] | [36] | [17] | [25] | [26] | |
| Physical state at room temperature | NR | NR | Liquid | Liquid | NR |
| Color | NR | NR | Greenish yellow | Yellow brown | NR |
| Odor | NR | NR | Slightly fruity | NR | NR |
| Property | NR | NR | Dry oil | NR | NR |
| Density at 20 °C (mass/volume) | NR | 0.92 ± 0.01 | 0.91 ± 0.001 | 0.91 ± 0.01 | 0.90 ± 0.0 |
| Refractive index at 20 °C | NR | 1.47 ± 0.010 | 1.5 ± 0.001 | 1.48 ± 0.01 | 1.5 ± 0.0 |
| Acid index | 1.26 ± 0.5 3.02 ± 0.5 | 21.2 ± 0.5 | 1.95 ± 0.03 | 1.82 ± 0.01 | NR |
| Peroxide index (meq O2/kg of oil) | 3.5 ± 1.5 8.6 ± 2.5 | 12.0 ± 0.4 | 2.23 ± 0.06 | NR | NR |
| Iodine index (g I2/100 g of oil) | 131.5 ± 0.5 32 ± 0.3 | NR | 108.52 ± 0.25 | 93.45 ± 0.22 | 101.5 ± 1.0 |
| Saponification index (mg of KOH/g oil) | NR | NR | 171.40 ± 0.43 | 177.10 ± 0.05 | 169.0 ± 0.1 |
| Extinction coefficient (K232) | 2.8 ± 0.5 3.25 ± 0.5 | 0.08 ± 0.010 | NR | NR | NR |
| K270 | 0.51 ± 0.5 2.11 ± 0.5 | 0.13 ± 0.020 | NR | NR | NR |
| Source of PPSO | Main Fatty Acid | References | |||||
|---|---|---|---|---|---|---|---|
| Unsaturated fatty acid % | Saturated fatty acid % | ||||||
| Linoleic | Oleic | Linolenic | Vaccenic | Palmitic | Stearic | ||
| Morocco | 62.0 | 21.0 | 0.30 | NR | 12.0 | 3.0 | [22] |
| Spanish | 57.7–63.1 | 19.0–21.8 | 0.23–1.10 | NR | 12.5–15.1 | 2.6–4.1 | [23] |
| Mexico |
66.5–76.1 60.5–78.8 | 9.3–19.9 10.7–19.9 | NR NR | NR NR | 5.6–56.2 2.9–6.1 | 15.5–36.1 2.9–5.2 | [18] |
| Algeria | 49.7–56.1 | 15.6–19.3 | 0.24 | 4.30 | 10.1 ± 0.2 | 2.8 ± 0.2 | [36] |
| Egypt | 54.03 | 22.41 | 0.63 | NR | 17.11 | 3.49 | [29] |
| Tunisia | 61.6 ± 0.1 | 21.2 ± 0.16 | 0.20 | NR | 12.2 ± 0.03 | 3.3 ± 0.03 | [17] |
| Cyprus | 55.9–60.1 | 15.6–17.6 | NR | NR | 12.4 | 4.0 | [16] |
| Sicilia/Italy | 58.0 | 18.0 | NR | 6.3 | 12.0 | 4.0 | [38] |
| South Africa | 56.9–65.2 | 16.4–22.5 | NR | NR | 12.7–16.1 | 2.2–3.4 | [31] |
| Yemen | 57.0 | 22.30 | 0.23 | NR | 14.0 | 3.0 | [6] |
| Morocco | 60.2–64.6 | 18.2–22.3 | 0.3 | NR | 11.6–12.4 | 3.0–3.4 | [2] |
| Tunisia | 57.6–57. 5 | 22.3–25.3 | 0.2–0.5 | NR | 14.3–11.3 | 3.12–4.3 | [28] |
| Turkey | 49.3–62.1 | 13.0–23.5 | 0.3 | 5.0–6.3 | 10.6–12.8 | 3.3–5.4 | [24] |
| Tunisia | 70.3 ± 0.6 | 16.8 ± 0.5 | NR | NR | 9.3 ± 0.2 | 3.1 ± 0.1 | [27] |
| Local market, Berlin, Germany | 53.5 ± 4.9 | 18.3 ± 1.6 | NR | 2.6 ± 0.2 | 20.1 ± 2.3 | 2.7 ± 0.1 | [4] |
| Composition | Composition of Total Sterol and Total Vitamin E (%) References | ||||||
|---|---|---|---|---|---|---|---|
| [22] | [40] | [2] | [23] | [1] | [4] | ||
| Hexane | 2-MeO | ||||||
| Phytosterol | NR | NR | |||||
| β-Sitosterol | 75.6 ± 0.7 | 82.8 ± 0.7 | 74.7 ± 1.2 | 75.9–81.8 | 81.9 | 71.60 | 72.0 |
| Campesterol | 11.6 ± 0.1 | 12.3 ± 0.1 | 10.3 ± 0.1 | 8.9–13.1 | 6.4 | 20.2 ± 0.1 | 16.6 ± 0.2 |
| Δ5-Avenasterol | 4.4 ± 0.1 | 4.1 ± 0.1 | 5.1 ± 0.2 | 3.6–6.7 | NR | 5.1 | NR |
| Stigmasterol | 3.3 ± 0.03 | 3.4 ± 0.5 | 1.5 ± 0.04 | 1.8 –3.0 | NR | 4.7 | 3.0 ± 0.04 |
| Δ7-Avenasterol | 2.0 ± 0.03 | 2.2 ± 0.02 | 2.3 ± 0.1 | 0.1–0.9 | NR | 2.3 ± 0.01 | 2.9 ± 0.03 |
| Δ7-Stigmasterol | 1.8 ± 0.02 | 2.2 ± 0.02 | 1.1 ± 0.02 | 0.3–1.5 | NR | - | 0.5 ± 0.01 |
| Cholesterol | 1.8 ± 0.4 | 1.5 ± 0.4 | 0.97 ± 0.03 | 0.9–1.3 | NR | 0.1 ± 0.0 | ND |
| Schottenol | 1.29 | ||||||
| Spinasterol | 1.60 | ||||||
| Campestanol | 0.82 | ||||||
| Sitostanol | 7.94 | 5.7 ± 0.02 | |||||
| vitamin E | |||||||
| α-tocopherol | 1.8± 0.5 | 1.95 ± 0.5 | 1.1 ± 0.1 | 1.3–2.5 | NR | 0.11 ± 0.0 | 0.56 |
| β-tocopherol | ND | ND | ND | 0.0–0.3 | NR | 0.00 | 0.12 |
| γ-tocopherol | 68.4 ± 0.5 | 67.5 ± 0.5 | 98.5 ± 0.1 | 86.5–91.7 | NR | 94.1 | 80.0 |
| δ-tocopherol | 7.5 ± 0.5 | 8.26 ± 0.5 | 0.46 ± 0.1 | 6.5–10.9 | NR | 3.4 | 5.0 |
| Volatile Compound | Amount | ||
|---|---|---|---|
| References | |||
| [19] | [7] | [35] | |
| Hexanal | 57.4 mg/kg | ||
| 2-methyl propanal | 38.9 mg/kg | ||
| Acetaldehyde | 16.2 mg/kg | ||
| Acetic acid | 10.9 mg/kg | ||
| Acetoin | 10.2 mg/kg | ||
| 2,3-butanedione | 6.3 mg/kg | ||
| Acids | 2.70% | ||
| Alcohols | 9.13% | ||
| Aldehydes | 62.72% | ||
| Esters | 2.82% | ||
| Hydrocarbons | 5.06% | ||
| Ketones | 4.38% | ||
| Hydrocarbons | 38.5% in red variety | ||
| Fatty acids and derivatives | 31.9% in red variety 68% in yellow variety | ||
| Terpenes | 12.4% red and 10.9% yellow | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Naqeb, G.; Fiori, L.; Ciolli, M.; Aprea, E. Prickly Pear Seed Oil Extraction, Chemical Characterization and Potential Health Benefits. Molecules 2021, 26, 5018. https://doi.org/10.3390/molecules26165018
Al-Naqeb G, Fiori L, Ciolli M, Aprea E. Prickly Pear Seed Oil Extraction, Chemical Characterization and Potential Health Benefits. Molecules. 2021; 26(16):5018. https://doi.org/10.3390/molecules26165018
Chicago/Turabian StyleAl-Naqeb, Ghanya, Luca Fiori, Marco Ciolli, and Eugenio Aprea. 2021. "Prickly Pear Seed Oil Extraction, Chemical Characterization and Potential Health Benefits" Molecules 26, no. 16: 5018. https://doi.org/10.3390/molecules26165018
APA StyleAl-Naqeb, G., Fiori, L., Ciolli, M., & Aprea, E. (2021). Prickly Pear Seed Oil Extraction, Chemical Characterization and Potential Health Benefits. Molecules, 26(16), 5018. https://doi.org/10.3390/molecules26165018