Green Tea Suppresses Brain Aging
Abstract
1. Introduction
2. Catechins
2.1. Lifespan
2.2. Cognitive Function
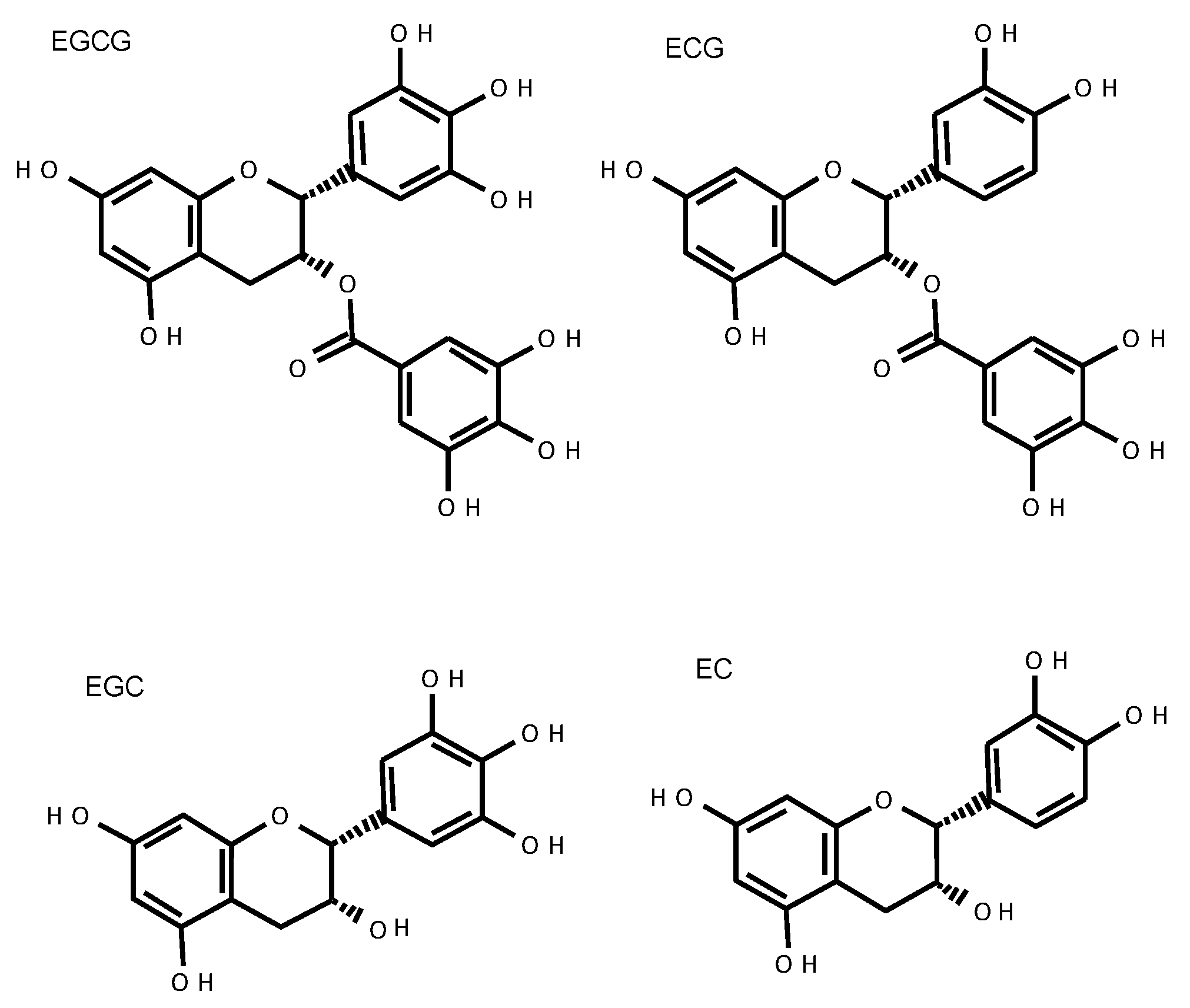
2.3. Absorption and Metabolism of Catechins
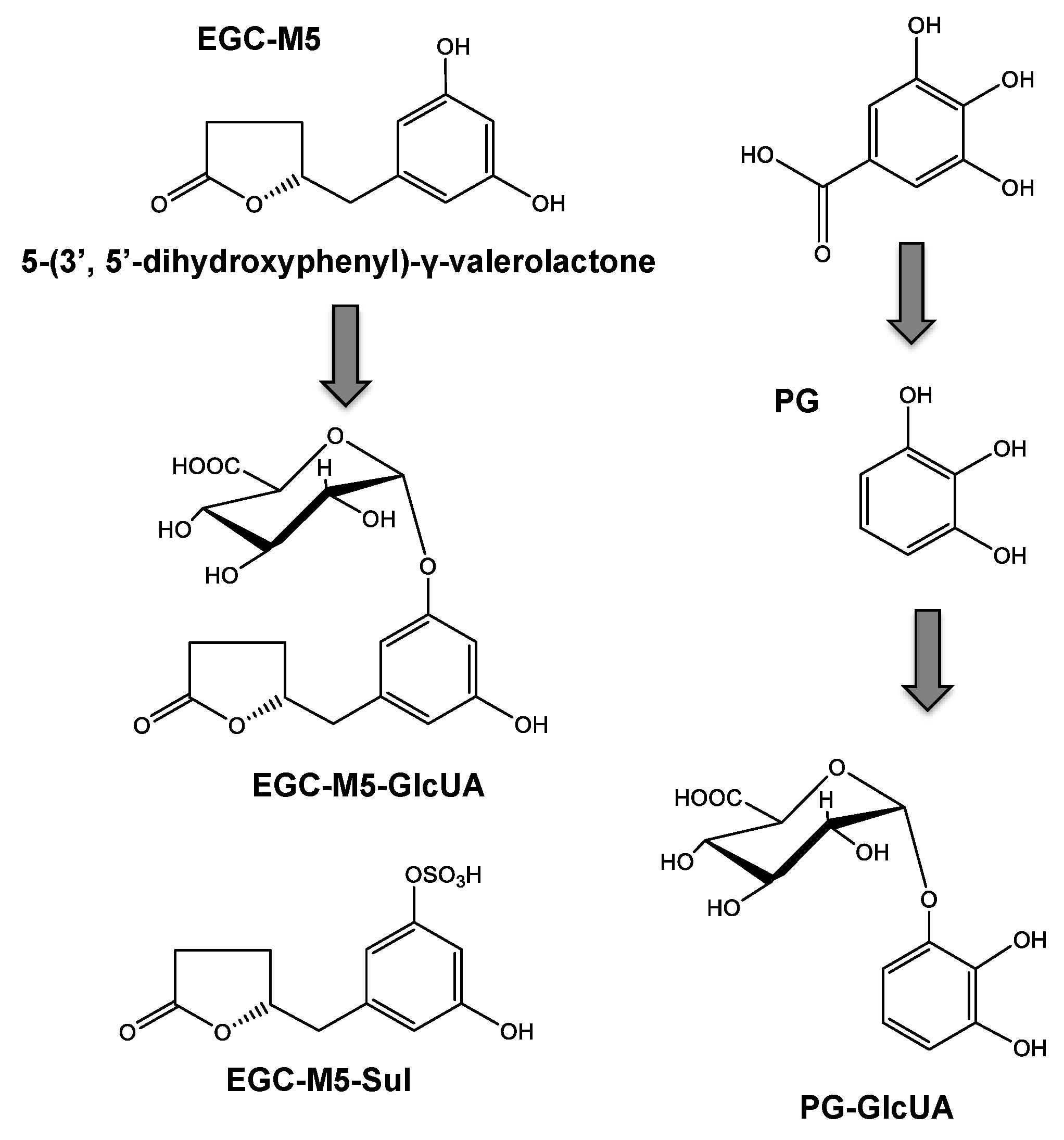
2.4. Blood–Brain Barrier Permeability of Catechins and Their Degradation Products
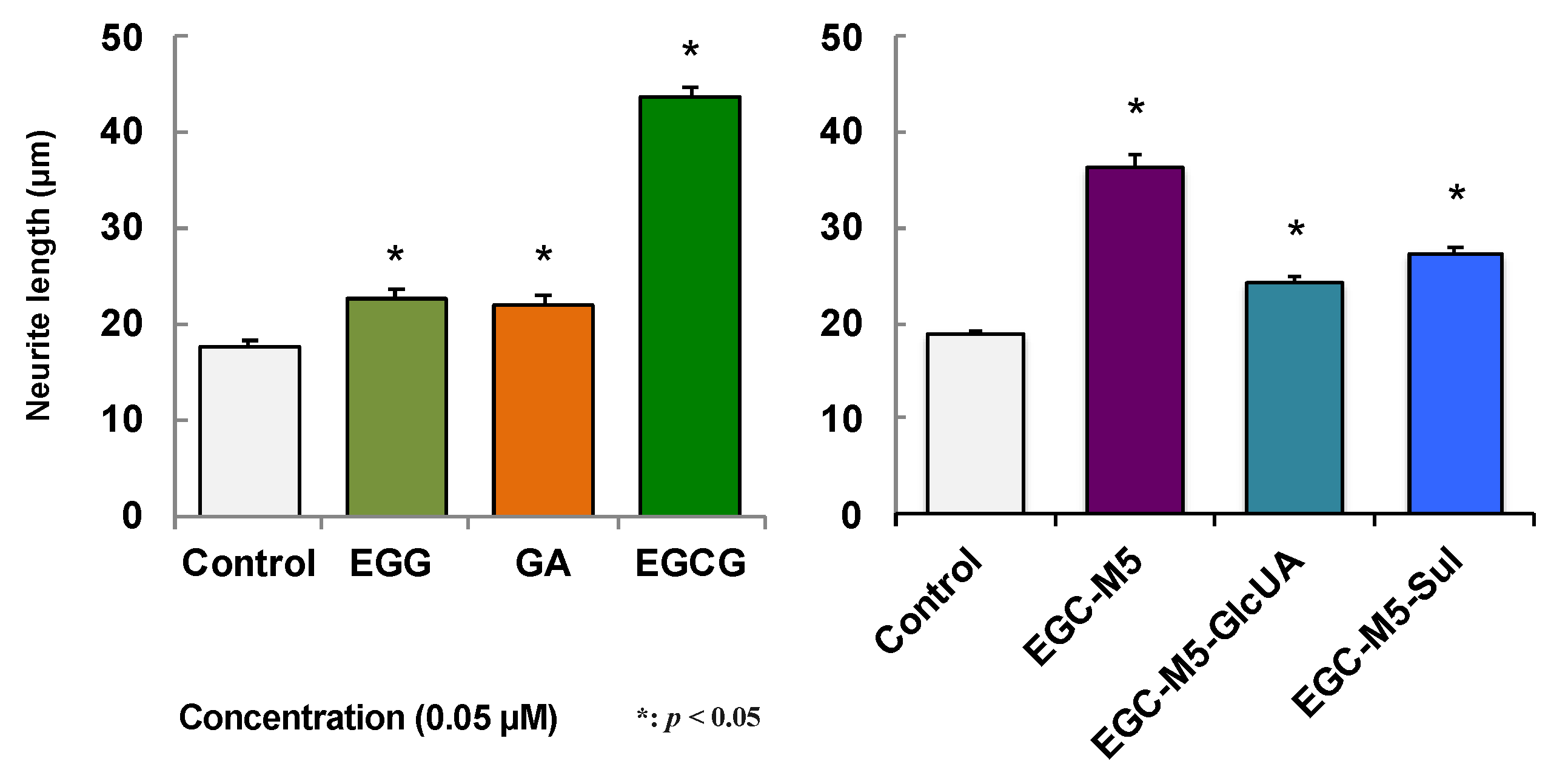
2.5. Effect of Green Tea Catechins on SH-SY5Y Cells
2.6. Altered Gene Expression Due to Green Tea Catechin Ingestion
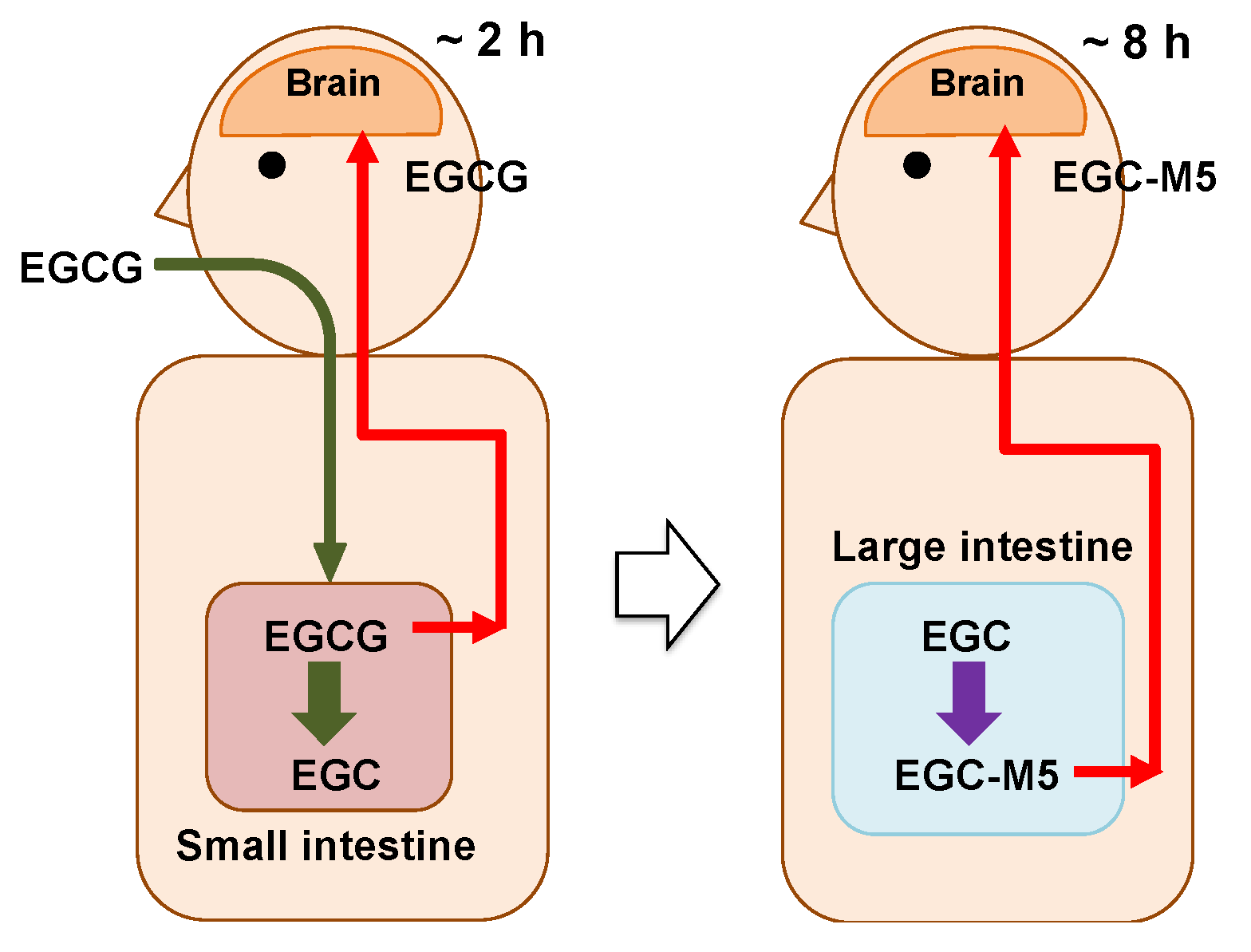
2.7. Actions of EGCG and Its Degradation Products on Brain
- Two to three hours after ingesting green tea, a very small amount of EGCG is taken up from the small intestine and enters the brain parenchyma through the bloodstream.
- The incorporated EGCG activates the nerve cells by enhancing IEG expression and promoting cell differentiation. After this, the EGCG is excreted.
- On the other hand, in the large intestine, most of the EGCG is decomposed from EGC into valerolactones, such as EGC-M5, by the intestinal microbiota over a period of 8 h.
- The resulting EGC-M5 (and other decomposed products) is taken up from the large intestine.
- EGC-M5 and its conjugates are delivered to the brain via systematic circulation.
- Nerve cells are reactivated again.
3. Theanine and Arginine
3.1. Adrenal Hypertrophy by Psychosocial Stress and Its Suppression by Theanine
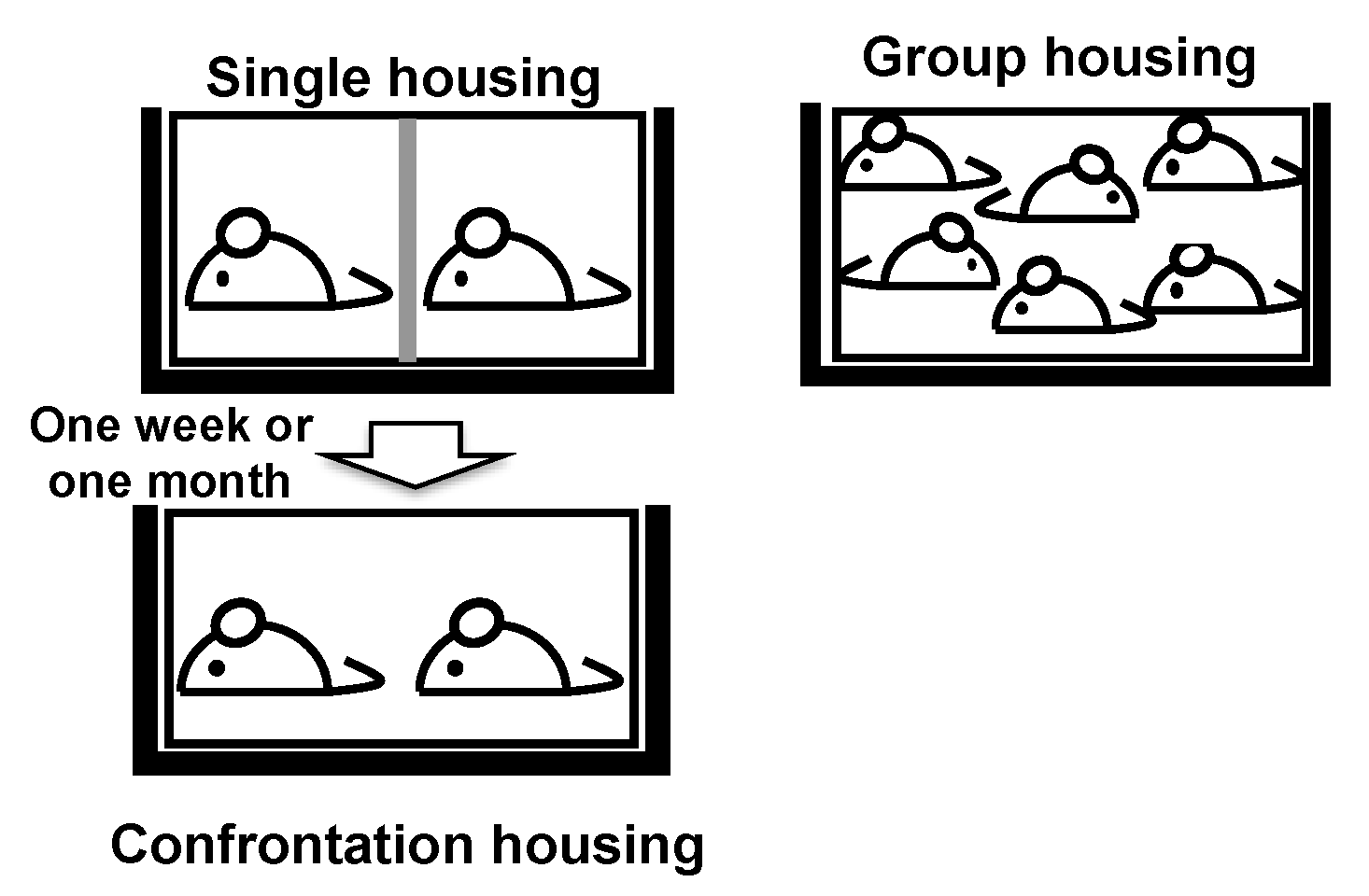
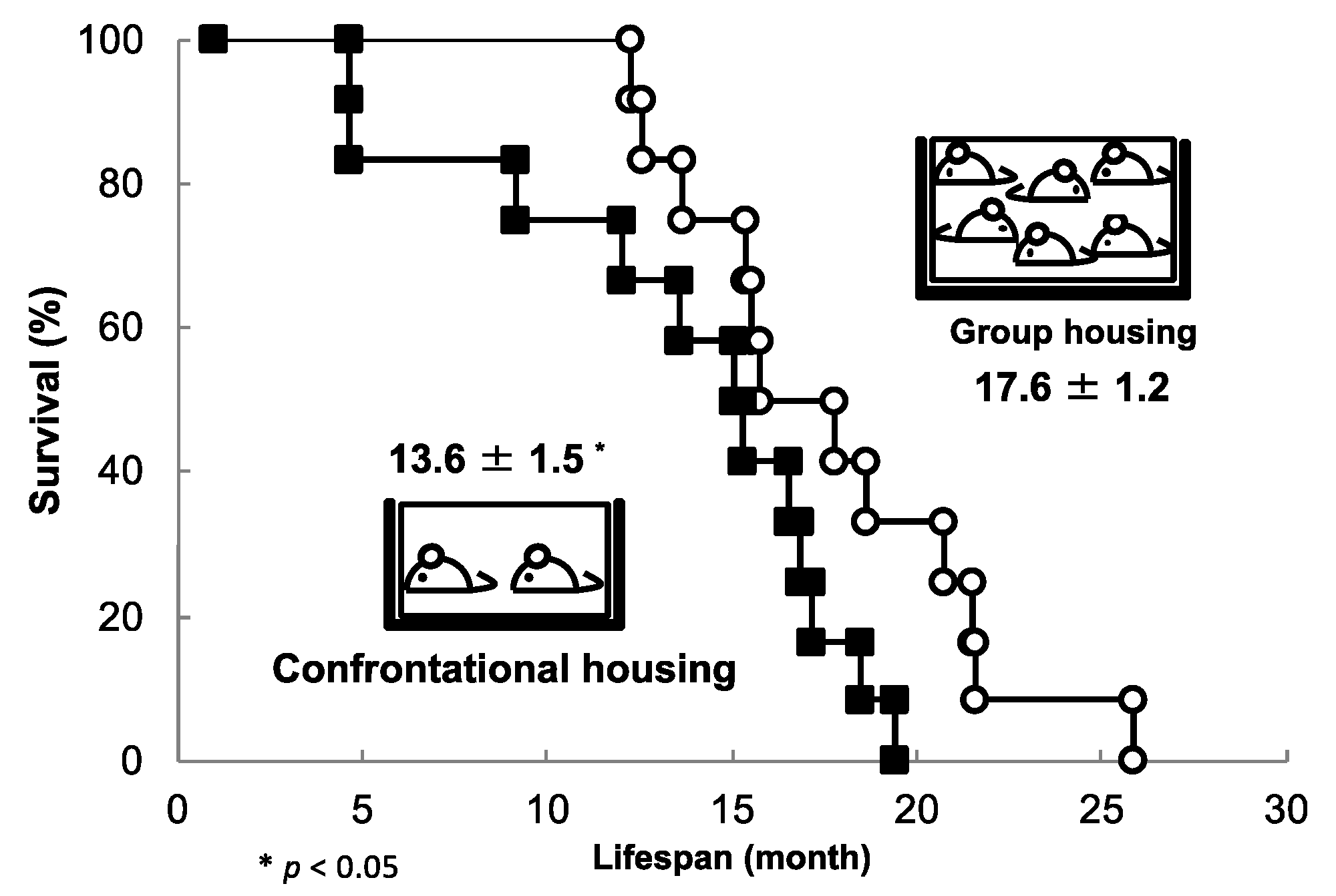
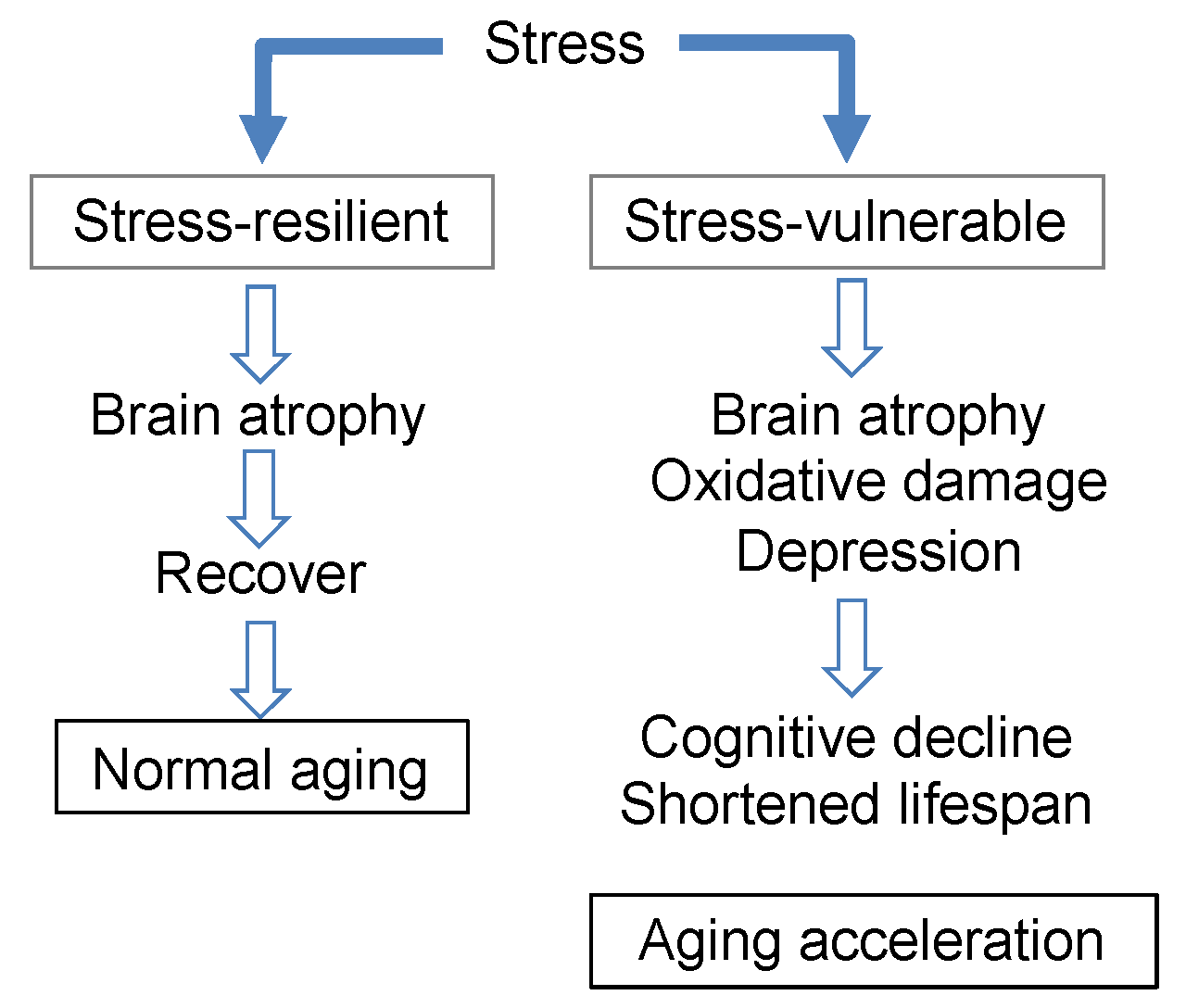
3.2. Longevity Shortened by Stress and Its Suppression by Theanine and Arginine
3.3. Promotion of Cognitive Decline Due to Stress Load, and Accumulation of Oxidative Damage
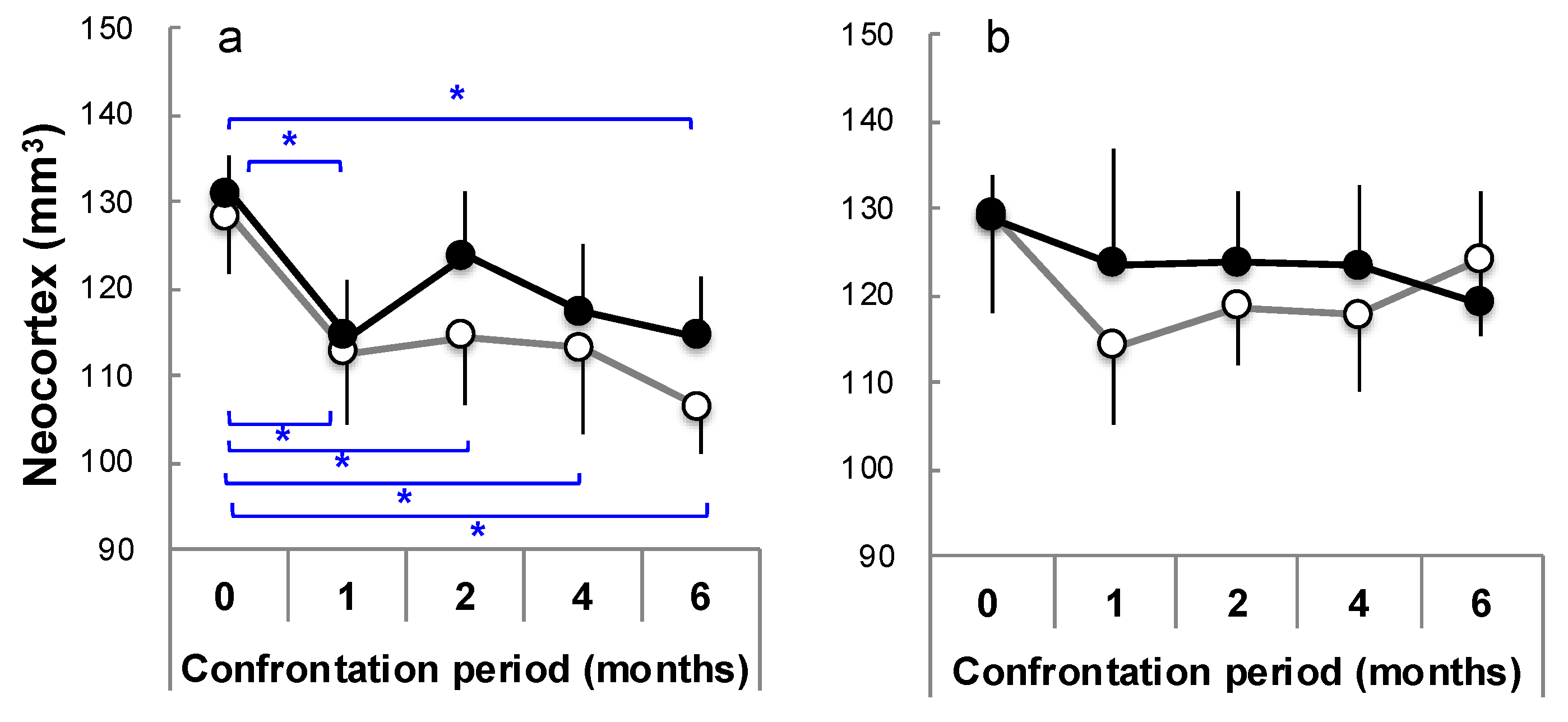
3.4. Brain Atrophy Due to Stress Load and Suppression by Theanine
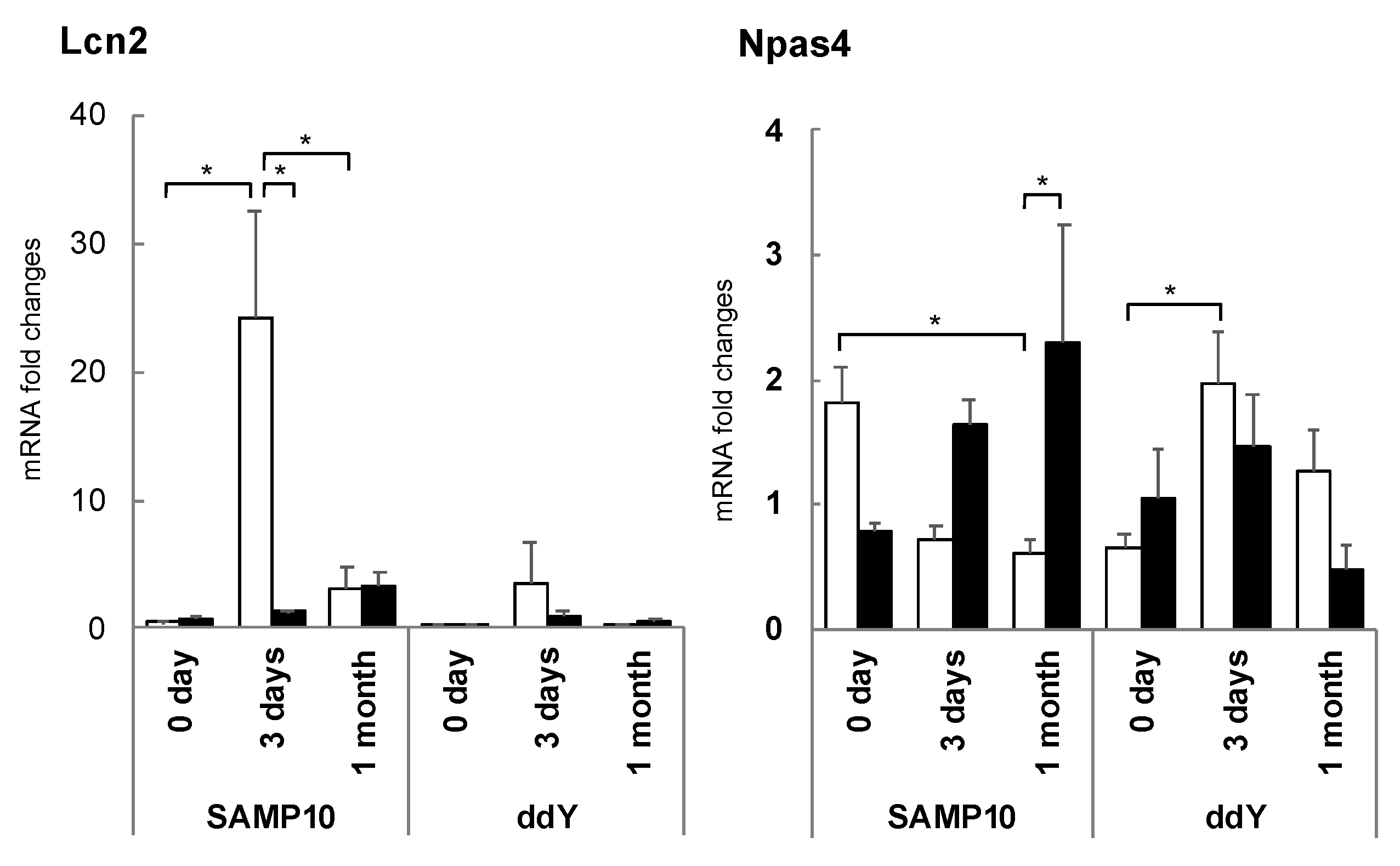
3.5. Changes in Hippocampal Gene Expression Due to Stress Loading and Suppression by Theanine or Arginine
3.6. Changes in Hippocampal Metabolites and Behavior One Month after Stress Loading
3.7. Brain Aging Acceleration Due to Psychosocial Stress
4. Function of Green Tea
4.1. Stress-Reducing Effect of Green Tea with Lowered Caffeine
4.2. Stress Reduction by Matcha
4.3. Anti-Depressant Effect of Shaded White Leaf Tea (SWLT)
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Nishizaki, Y.; Kuwahira, I.; Kawada, H.; Kubo, A.; Kataoka, K.; Tanaka, S.; Sueno, T.; Isozaki, M.; Kobayashi, H.; Nakamura, Y.; et al. Beneficial effects of medical advice provided to elderly persons under the anti-aging health check-up system at Tokai University Tokyo Hospital. Tokai J. Exp. Clin. Med. 2009, 34, 142–151. [Google Scholar] [PubMed]
- Saito, E.; Inoue, M.; Sawada, N.; Shimazu, T.; Yamaji, T.; Iwasaki, M.; Sasazuki, S.; Noda, M.; Iso, H.; Tsugane, S.; et al. Association of green tea consumption with mortality due to all causes and major causes of death in a Japanese population: The Japan Public Health Center-based Prospective Study (JPHC Study). Ann. Epidemiol. 2015, 25, 512–518.e3. [Google Scholar] [CrossRef] [PubMed]
- Noguchi-Shinohara, M.; Yuki, S.; Dohmoto, C.; Ikeda, Y.; Samuraki, M.; Iwasa, K.; Yokogawa, M.; Asai, K.; Komai, K.; Nakamura, H.; et al. Consumption of green tea, but not black tea or coffee, is associated with reduced risk of cognitive decline. PLoS ONE 2014, 9, e96013. [Google Scholar] [CrossRef] [PubMed]
- Kakutani, S.; Watanabe, H.; Murayama, N. Green Tea Intake and Risks for Dementia, Alzheimer’s Disease, Mild Cognitive Impairment, and Cognitive Impairment: A Systematic Review. Nutrients 2019, 11, 1165. [Google Scholar] [CrossRef]
- Ran, L.S.; Liu, W.H.; Fang, Y.Y.; Xu, S.B.; Li, J.; Luo, X.; Pan, D.J.; Wang, M.H.; Wang, W. Alcohol, coffee and tea intake and the risk of cognitive deficits: A dose-response meta-analysis. Epidemiol. Psychiatr. Sci. 2021, 30, e13. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, A.; Zhang, X.; Chen, S.; Wu, S.; Zhao, X.; Zhang, Q. Association between tea consumption and cognitive impairment in middle-aged and older adults. BMC Geriatr. 2020, 20, 447. [Google Scholar] [CrossRef]
- Okubo, H.; Inagaki, H.; Gondo, Y.; Kamide, K.; Ikebe, K.; Masui, Y.; Arai, Y.; Ishizaki, T.; Sasaki, S.; Nakagawa, T.; et al. Association between dietary patterns and cognitive function among 70-year-old Japanese elderly: A cross-sectional analysis of the SONIC study. Nutr. J. 2017, 16, 56. [Google Scholar] [CrossRef]
- Pervin, M.; Unno, K.; Ohishi, T.; Tanabe, H.; Miyoshi, N.; Nakamura, Y. Beneficial Effects of Green Tea Catechins on Neurodegenerative Diseases. Molecules 2018, 23, 1297. [Google Scholar] [CrossRef]
- Singh, N.A.; Mandal, A.K.; Khan, Z.A. Potential neuroprotective properties of epigallocatechin-3-gallate (EGCG). Nutr. J. 2016, 15, 60. [Google Scholar] [CrossRef]
- Rezai-Zadeh, K.; Shytle, D.; Sun, N.; Mori, T.; Hou, H.; Jeanniton, D.; Ehrhart, J.; Townsend, K.; Zeng, J.; Morgan, D.; et al. Green tea epigallocatechin-3-gallate (EGCG) modulates amyloid precursor protein cleavage and reduces cerebral amyloidosis in Alzheimer transgenic mice. J. Neurosci. 2005, 25, 8807–8814. [Google Scholar] [CrossRef]
- Mandel, S.A.; Amit, T.; Kalfon, L.; Reznichenko, L.; Weinreb, O.; Youdim, M.B. Cell signaling pathways and iron chelation in the neurorestorative activity of green tea polyphenols: Special reference to epigallocatechin gallate (EGCG). J. Alzheimers Dis. 2008, 15, 211–222. [Google Scholar] [CrossRef]
- Mithul Aravind, S.; Wichienchot, S.; Tsao, R.; Ramakrishnan, S.; Chakkaravarthi, S. Role of dietary polyphenols on gut microbiota, their metabolites and health benefits. Food Res. Int. 2021, 142, 110189. [Google Scholar] [CrossRef]
- Clifford, M.N.; van der Hooft, J.J.; Crozier, A. Human studies on the absorption, distribution, metabolism, and excretion of tea polyphenols. Am. J. Clin. Nutr. 2013, 98 (Suppl. 6), 1619S–1630S. [Google Scholar] [CrossRef]
- Liu, Z.; Bruins, M.E.; Ni, L.; Vincken, J.P. Green and Black Tea Phenolics: Bioavailability, Transformation by Colonic Microbiota, and Modulation of Colonic Microbiota. J. Agric. Food Chem. 2018, 66, 8469–8477. [Google Scholar] [CrossRef]
- Angelino, D.; Carregosa, D.; Domenech-Coca, C.; Savi, M.; Figueira, I.; Brindani, N.; Jang, S.; Lakshman, S.; Molokin, A.; Urban, J.F., Jr.; et al. 5-(Hydroxyphenyl)-γ-Valerolactone-Sulfate, a Key Microbial Metabolite of Flavan-3-ols, Is Able to Reach the Brain: Evidence from Different in Silico, In Vitro and In Vivo Experimental Models. Nutrients 2019, 11, 2678. [Google Scholar] [CrossRef]
- Pervin, M.; Unno, K.; Takagaki, A.; Isemura, M.; Nakamura, Y. Function of Green Tea Catechins in the Brain: Epigallocatechin Gallate and its Metabolites. Int. J. Mol. Sci. 2019, 20, 3630. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Pervin, M.; Nakagawa, A.; Iguchi, K.; Hara, A.; Takagaki, A.; Nanjo, F.; Minami, A.; Nakamura, Y. Blood–brain Barrier Permeability of Green Tea Catechin Metabolites and their Neuritogenic Activity in Human Neuroblastoma SH-SY5Y Cells. Mol. Nutr. Food Res. 2017, 61, e1700294. [Google Scholar] [CrossRef]
- Takagaki, A.; Yoshioka, Y.; Yamashita, Y.; Nagano, T.; Ikeda, M.; Hara-Terawaki, A.; Seto, R.; Ashida, H. Effects of Microbial Metabolites of (-)-Epigallocatechin Gallate on Glucose Uptake in L6 Skeletal Muscle Cell and Glucose Tolerance in ICR Mice. Biol. Pharm. Bull. 2019, 42, 212–221. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.H.; Won, Y.S.; Yang, X.; Kumazoe, M.; Yamashita, S.; Hara, A.; Takagaki, A.; Goto, K.; Nanjo, F.; Tachibana, H. Green Tea Catechin Metabolites Exert Immunoregulatory Effects on CD4(+) T Cell and Natural Killer Cell Activities. J. Agric. Food Chem. 2016, 64, 3591–3597. [Google Scholar] [CrossRef] [PubMed]
- Takagaki, A.; Otani, S.; Nanjo, F. Antioxidative activity of microbial metabolites of (-)-epigallocatechin gallate produced in rat intestines. Biosci. Biotechnol. Biochem. 2011, 75, 582–585. [Google Scholar] [CrossRef]
- Unno, T.; Tamemoto, K.; Yayabe, F.; Kakuda, T. Urinary excretion of 5-(3’,4’-dihydroxyphenyl)-gamma-valerolactone, a ring-fission metabolite of (-)-epicatechin, in rats and its in vitro antioxidant activity. J. Agric. Food Chem. 2003, 51, 6893–6898. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.L.; Everett, J.M.; D’Cunha, N.M.; Sergi, D.; Georgousopoulou, E.N.; Keegan, R.J.; McKune, A.J.; Mellor, D.D.; Anstice, N.; Naumovski, N. The Effects of Green Tea Amino Acid L-Theanine Consumption on the Ability to Manage Stress and Anxiety Levels: A Systematic Review. Plant Foods Hum. Nutr. 2020, 75, 12–23. [Google Scholar] [CrossRef]
- Hidese, S.; Ogawa, S.; Ota, M.; Ishida, I.; Yasukawa, Z.; Ozeki, M.; Kunugi, H. Effects of L-Theanine Administration on Stress-Related Symptoms and Cognitive Functions in Healthy Adults: A Randomized Controlled Trial. Nutrients 2019, 11, 2362. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Tanida, N.; Ishii, N.; Yamamoto, H.; Iguchi, K.; Hoshino, M.; Takeda, A.; Ozawa, H.; Ohkubo, T.; Juneja, L.R.; et al. Anti-stress effect of theanine on students during pharmacy practice: Positive correlation among salivary α-amylase activity, trait anxiety and subjective stress. Pharmacol. Biochem. Behav. 2013, 111, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Ozeki, M.; Juneja, L.R.; Ohira, H. L-Theanine reduces psychological and physiological stress responses. Biol. Psychol. 2007, 74, 39–45. [Google Scholar] [CrossRef]
- Kobayashi, K.; Nagato, Y.; Aoi, N.; Juneja, L.R.; Kim, M.; Yamamoto, T.; Sugimoto, S. Effect of L-theanine on the release of α-brain wave in human volunteers. Nippon Nogeikagaku Kaishi 1998, 72, 153–157. [Google Scholar] [CrossRef]
- Unno, K.; Hara, A.; Nakagawa, A.; Iguchi, K.; Ohshio, M.; Morita, A.; Nakamura, Y. Anti-stress effects of drinking green tea with lowered caffeine and enriched theanine, epigallocatechin and arginine on psychosocial stress induced adrenal hypertrophy in mice. Phytomedicine 2016, 23, 1365–1374. [Google Scholar] [CrossRef]
- Pervin, M.; Unno, K.; Konishi, T.; Nakamura, Y. L-Arginine Exerts Excellent Anti-Stress Effects on Stress-Induced Shortened Lifespan, Cognitive Decline and Depression. Int. J. Mol. Sci. 2021, 22, 508. [Google Scholar] [CrossRef]
- Unno, K.; Furushima, D.; Hamamoto, S.; Iguchi, K.; Yamada, H.; Morita, A.; Horie, H.; Nakamura, Y. Stress-Reducing Function of Matcha Green Tea in Animal Experiments and Clinical Trials. Nutrients 2018, 10, 1468. [Google Scholar] [CrossRef]
- Ansell, E.B.; Rando, K.; Tuit, K.; Guarnaccia, J.; Sinha, R. Cumulative adversity and smaller gray matter volume in medial prefrontal, anterior cingulate, and insula regions. Biol. Psychiatry 2012, 72, 57–64. [Google Scholar] [CrossRef]
- Bremner, J.D. Stress and brain atrophy. CNS Neurol. Disord. Drug Targets 2006, 5, 503–512. [Google Scholar] [CrossRef]
- Schoenfeld, T.J.; McCausland, H.C.; Morris, H.D.; Padmanaban, V.; Cameron, H.A. Stress and Loss of Adult Neurogenesis Differentially Reduce Hippocampal Volume. Biol. Psychiatry 2017, 82, 914–923. [Google Scholar] [CrossRef] [PubMed]
- Tomoda, A.; Polcari, A.; Anderson, C.M.; Teicher, M.H. Reduced visual cortex gray matter volume and thickness in young adults who witnessed domestic violence during childhood. PLoS ONE 2012, 7, e52528. [Google Scholar] [CrossRef] [PubMed]
- Tomoda, A.; Sheu, Y.S.; Rabi, K.; Suzuki, H.; Navalta, C.P.; Polcari, A.; Teicher, M.H. Exposure to parental verbal abuse is associated with increased gray matter volume in superior temporal gyrus. Neuroimage 2011, 54 (Suppl. 1), S280–S286. [Google Scholar] [CrossRef]
- Tomoda, A.; Navalta, C.P.; Polcari, A.; Sadato, N.; Teicher, M.H. Childhood sexual abuse is associated with reduced gray matter volume in visual cortex of young women. Biol. Psychiatry 2009, 66, 642–648. [Google Scholar] [CrossRef]
- Guessoum, S.B.; Lachal, J.; Radjack, R.; Carretier, E.; Minassian, S.; Benoit, L.; Moro, M.R. Adolescent psychiatric disorders during the COVID-19 pandemic and lockdown. Psychiatry Res. 2020, 291, 113264. [Google Scholar] [CrossRef]
- Heitzman, J. Impact of COVID-19 pandemic on mental health. Psychiatr. Pol. 2020, 54, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Tsujimura, M. On Tea Catechin Isolated from Green Tea. J. Agric. Chem. Soc. Jpn. 1930, 6, 62–69. [Google Scholar] [CrossRef]
- Lopez, T.; Schriner, S.E.; Okoro, M.; Lu, D.; Chiang, B.T.; Huey, J.; Jafari, M. Green tea polyphenols extend the lifespan of male drosophila melanogaster while impairing reproductive fitness. J. Med. Food. 2014, 17, 1314–1321. [Google Scholar] [CrossRef]
- Abbas, S.; Wink, M. Epigallocatechin gallate from green tea (Camellia sinensis) increases lifespan and stress resistance in Caenorhabditis elegans. Planta Med. 2009, 75, 216–221. [Google Scholar] [CrossRef]
- Unno, K.; Pervin, M.; Taguchi, K.; Konishi, T.; Nakamura, Y. Green Tea Catechins Trigger Immediate-Early Genes in the Hippocampus and Prevent Cognitive Decline and Lifespan Shortening. Molecules 2020, 25, 1484. [Google Scholar] [CrossRef]
- Qiu, L.; Sautter, J.; Gu, D. Associations between frequency of tea consumption and health and mortality: Evidence from old Chinese. Br. J. Nutr. 2012, 108, 1686–1697. [Google Scholar] [CrossRef]
- Shirai, Y.; Kuriki, K.; Otsuka, R.; Kato, Y.; Nishita, Y.; Tange, C.; Tomida, M.; Imai, T.; Ando, F.; Shimokata, H. Green tea and coffee intake and risk of cognitive decline in older adults: The National Institute for Longevity Sciences, Longitudinal Study of Aging. Public Health Nutr. 2020, 23, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Kuriyama, S.; Hozawa, A.; Ohmori, K.; Shimazu, T.; Matsui, T.; Ebihara, S.; Awata, S.; Nagatomi, R.; Arai, H.; Tsuji, I. Green tea consumption and cognitive function: A cross-sectional study from the Tsurugaya Project 1. Am. J. Clin. Nutr. 2006, 83, 355–361. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Takabayashi, F.; Kishido, T.; Oku, N. Suppressive effect of green tea catechins on morphologic and functional regression of the brain in aged mice with accelerated senescence (SAMP10). Exp. Gerontol. 2004, 39, 1027–1034. [Google Scholar] [CrossRef]
- Unno, K.; Takabayashi, F.; Yoshida, H.; Choba, D.; Fukutomi, R.; Kikunaga, N.; Kishido, T.; Oku, N.; Hoshino, M. Daily consumption of green tea catechin delays memory regression in aged mice. Biogerontology 2007, 8, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Ishikawa, Y.; Takabayashi, F.; Sasaki, T.; Takamori, N.; Iguchi, K.; Hoshino, M. Daily ingestion of green tea catechins from adulthood suppressed brain dysfunction in aged mice. Biofactors 2008, 34, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Pervin, M.; Unno, K.; Nakagawa, A.; Takahashi, Y.; Iguchi, K.; Yamamoto, H.; Hoshino, M.; Hara, A.; Takagaki, A.; Nanjo, F.; et al. Blood brain barrier permeability of (-)-epigallocatechin gallate, its proliferation-enhancing activity of human neuroblastoma SH-SY5Y cells, and its preventive effect on age-related cognitive dysfunction in mice. Biochem. Biophys. Rep. 2017, 9, 180–186. [Google Scholar] [CrossRef]
- Chen, L.; Lee, M.J.; Li, H.; Yang, C.S. Absorption, distribution, elimination of tea polyphenols in rats. Drug Metab. Dispos. 1997, 25, 1045–1050. [Google Scholar]
- Nakagawa, K.; Okuda, S.; Miyazawa, T. Dose-dependent incorporation of tea catechins, (-)-epigallocatechin-3-gallate and (-)-epigallocatechin, into human plasma. Biosci. Biotechnol. Biochem. 1997, 61, 1981–1985. [Google Scholar] [CrossRef]
- Kohri, T.; Matsumoto, N.; Yamakawa, M.; Suzuki, M.; Nanjo, F.; Hara, Y.; Oku, N. Metabolic fate of (-)-[4-(3)H]epigallocatechin gallate in rats after oral administration. J. Agric. Food Chem. 2001, 49, 4102–4112. [Google Scholar] [CrossRef]
- Mena, P.; Bresciani, L.; Brindani, N.; Ludwig, I.A.; Pereira-Caro, G.; Angelino, D.; Llorach, R.; Calani, L.; Brighenti, F.; Clifford, M.N.; et al. Phenyl-γ-valerolactones and phenylvaleric acids, the main colonic metabolites of flavan-3-ols: Synthesis, analysis, bioavailability, and bioactivity. Nat. Prod. Rep. 2019, 36, 714–752. [Google Scholar] [CrossRef]
- Faria, A.; Pestana, D.; Teixeira, D.; Couraud, P.O.; Romero, I.; Weksler, B.; de Freitas, V.; Mateus, N.; Calhau, C. Insights into the putative catechin and epicatechin transport across blood–brain barrier. Food Funct. 2011, 2, 39–44. [Google Scholar] [CrossRef]
- Heroux, N.A.; Osborne, B.F.; Miller, L.A.; Kawan, M.; Buban, K.N.; Rosen, J.B.; Stanton, M.E. Differential expression of the immediate early genes c-Fos, Arc, Egr-1, and Npas4 during long-term memory formation in the context preexposure facilitation effect (CPFE). Neurobiol. Learn. Mem. 2018, 147, 128–138. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, Y.; Ertürk, A.; Kallop, D.; Jiang, Z.; Weimer, R.M.; Kaminker, J.; Sheng, M. Activity-induced Nr4a1 regulates spine density and distribution pattern of excitatory synapses in pyramidal neurons. Neuron 2014, 83, 431–443. [Google Scholar] [CrossRef]
- Kwapis, J.L.; Alaghband, Y.; LÓpez, A.J.; Long, J.M.; Li, X.; Shu, G.; Bodinayake, K.K.; Matheos, D.P.; Rapp, P.R.; Wood, M.A. HDAC3-mediated repression of the Nr4a family contributes to age-related impairments in long-term memory. J. Neurosci. 2019, 39, 4999–5009. [Google Scholar] [CrossRef]
- Malik, A.R.; Urbanska, M.; Gozdz, A.; Swiech, L.J.; Nagalski, A.; Perycz, M.; Blazejczyk, M.; Jaworski, J. Cyr61, a matricellular protein, is needed for dendritic arborization of hippocampal neurons. J. Biol. Chem. 2013, 288, 8544–8559. [Google Scholar] [CrossRef] [PubMed]
- Miyashita, T.; Kubik, S.; Lewandowski, G.; Guzowski, J.F. Networks of neurons, networks of genes: An integrated view of memory consolidation. Neurobiol. Learn. Mem. 2008, 89, 269–284. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.H.; Cheng, J.; Li, C.R.; Ye, M.; Ma, Z.; Cai, F. Modulation of Ca2+ signals by epigallocatechin-3-gallate (EGCG) in cultured rat hippocampal neurons. Int. J. Mol. Sci. 2011, 12, 742–754. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.H.; Mun, K.C.; Park, W.K.; Lee, S.R.; Suh, S.I.; Baek, W.K.; Yim, M.B.; Kwon, T.K.; Song, D.K. EGCG attenuates AMPA-induced intracellular calcium increase in hippocampal neurons. Biochem. Biophys. Res. Commun. 2002, 290, 1506–1512. [Google Scholar]
- Del Rio, D.; Calani, L.; Cordero, C.; Salvatore, S.; Pellegrini, N.; Brighenti, F. Bioavailability and catabolism of green tea flavan-3-ols in humans. Nutrition 2010, 26, 1110–1116. [Google Scholar] [CrossRef]
- Sakato, Y. Studies on the Chemical Constituents of Tea. Part III. A New Amide Theanine. Nippon Nogeikagaku Kaishi. 1950, 23, 262–267. [Google Scholar] [CrossRef]
- Nagata, T. Study on useful tea ingredients in leaves in Camellia plants. Bull Tea Res Station. Minist. Agric. For. 1986, 21, 59–120. [Google Scholar]
- Horie, H.; Kohata, K. General ingredients of tea. In Health Science of Tea. New Possibility for Physiological Function; Muramatsu, K., Oguni, I., Isemura, M., Yamamoto-Maeda, M., Eds.; Gakkai Shuppan Center: Tokyo, Japan, 2002; pp. 26–31. [Google Scholar]
- Konishi, S. Tea plant biochemistry. In Science of Tea; Muramatsu, K., Ed.; Asakura Shoten: Tokyo, Japan, 1991; pp. 32–42. [Google Scholar]
- Yamada, T.; Terashima, T.; Okubo, T.; Juneja, L.R.; Yokogoshi, H. Effects of theanine, r-glutamylethylamide, on neurotransmitter release and its relationship with glutamic acid neurotransmission. Nutr. Neurosci. 2005, 8, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, N.; Horie, H.; Mukai, T.; Goto, T. Content of Individual Free Amino Acids in the First and Autumn Flushes of Cultivars for Several Tea Types. Tea Res. J. 1993, 78, 67–75. [Google Scholar] [CrossRef][Green Version]
- Yokogoshi, H.; Kobayashi, M.; Mochizuki, M.; Terashima, T. Effect of theanine, r-glutamylethylamide, on brain monoamines and striatal dopamine release in conscious rats. Neurochem. Res. 1998, 23, 667–673. [Google Scholar] [CrossRef] [PubMed]
- Kakuda, T.; Nozawa, A.; Unno, T.; Okamura, N.; Okai, O. Inhibiting effects of theanine on caffeine stimulation evaluated by EEG in the rat. Biosci. Biotechnol. Biochem. 2000, 64, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Kakuda, T. Neuroprotective effects of theanine and its preventive effects on cognitive dysfunction. Pharmacol. Res. 2011, 64, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Yoneda, Y.; Kawada, K.; Kuramoto, N. Selective Upregulation by Theanine of Slc38a1 Expression in Neural Stem Cell for Brain Wellness. Molecules 2020, 25, 347. [Google Scholar] [CrossRef]
- Hidese, S.; Ota, M.; Wakabayashi, C.; Noda, T.; Ozawa, H.; Okubo, T.; Kunugi, H. Effects of chronic l-theanine administration in patients with major depressive disorder: An open-label study. Acta Neuropsychiatr. 2017, 29, 72–79. [Google Scholar] [CrossRef]
- Juruena, M.F.; Eror, F.; Cleare, A.J.; Young, A.H. The Role of Early Life Stress in HPA Axis and Anxiety. Adv. Exp. Med. Biol. 2020, 1191, 141–153. [Google Scholar] [CrossRef] [PubMed]
- Dar, T.; Radfar, A.; Abohashem, S.; Pitman, R.K.; Tawakol, A.; Osborne, M.T. Psychosocial Stress and Cardiovascular Disease. Curr Treat. Options Cardiovasc. Med. 2019, 21, 23. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Iguchi, K.; Tanida, N.; Fujitani, K.; Takamori, N.; Yamamoto, H.; Ishii, N.; Nagano, H.; Nagashima, T.; Hara, A.; et al. Ingestion of theanine, an amino acid in tea, suppresses psychosocial stress in mice. Exp. Physiol. 2013, 98, 290–303. [Google Scholar] [CrossRef]
- Liston, C.; Cichon, J.M.; Jeanneteau, F.; Jia, Z.; Chao, M.V.; Gan, W.B. Circadian glucocorticoid oscillations promote learning-dependent synapse formation and maintenance. Nat. Neurosci. 2013, 16, 698–705. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Fujitani, K.; Takamori, N.; Takabayashi, F.; Maeda, K.; Miyazaki, H.; Tanida, N.; Iguchi, K.; Shimoi, K.; Hoshino, M. Theanine intake improves the shortened lifespan, cognitive dysfunction and behavioural depression that are induced by chronic psychosocial stress in mice. Free Radic. Res. 2011, 45, 966–974. [Google Scholar] [CrossRef]
- Taylor, D.L.; Edwards, A.D.; Mehmet, H. Oxidative metabolism, apoptosis and perinatal brain injury. Brain Pathol. 1999, 9, 93–117. [Google Scholar] [CrossRef]
- Sasaki, T.; Unno, K.; Tahara, S.; Shimada, A.; Chiba, Y.; Hoshino, M.; Kaneko, T. Age-related increase of superoxide generation in the brains of mammals and birds. Aging Cell. 2008, 7, 459–469. [Google Scholar] [CrossRef]
- Kishido, T.; Unno, K.; Yoshida, H.; Choba, D.; Fukutomi, R.; Asahina, S.; Iguchi, K.; Oku, N.; Hoshino, M. Decline in glutathione peroxidase activity is a reason for brain senescence: Consumption of green tea catechin prevents the decline in its activity and protein oxidative damage in ageing mouse brain. Biogerontology 2007, 8, 423–430. [Google Scholar] [CrossRef]
- Unno, K.; Sumiyoshi, A.; Konishi, T.; Hayashi, M.; Taguchi, K.; Muguruma, Y.; Inoue, K.; Iguchi, K.; Nonaka, H.; Kawashima, R.; et al. Theanine, the Main Amino Acid in Tea, Prevents Stress-Induced Brain Atrophy by Modifying Early Stress Responses. Nutrients 2020, 12, 174. [Google Scholar] [CrossRef]
- Suk, K. Lipocalin-2 as a therapeutic target for brain injury: An astrocentric perspective. Prog. Neurobiol. 2016, 144, 158–172. [Google Scholar] [CrossRef]
- Sun, X.; Lin, Y. Npas4: Linking Neuronal Activity to Memory. Trends Neurosci. 2016, 39, 264–275. [Google Scholar] [CrossRef] [PubMed]
- Benatti, C.; Radighieri, G.; Alboni, S.; Blom, J.M.C.; Brunello, N.; Tascedda, F. Modulation of neuroplasticity-related targets following stress-induced acute escape deficit. Behav. Brain Res. 2019, 364, 140–148. [Google Scholar] [CrossRef]
- Evanson, N.K.; Van Hooren, D.C.; Herman, J.P. GluR5-mediated glutamate signaling regulates hypothalamo-pituitary-adrenocortical stress responses at the paraventricular nucleus and median eminence. Psychoneuroendocrinology 2009, 34, 1370–1379. [Google Scholar] [CrossRef]
- Unno, K.; Muguruma, Y.; Inoue, K.; Konishi, T.; Taguchi, K.; Hasegawa-Ishii, S.; Shimada, A.; Nakamura, Y. Theanine, Antistress Amino Acid in Tea Leaves, Causes Hippocampal Metabolic Changes and Antidepressant Effects in Stress-Loaded Mice. Int. J. Mol. Sci. 2020, 22, 193. [Google Scholar] [CrossRef]
- Yun, J.; Koike, H.; Ibi, D.; Toth, E.; Mizoguchi, H.; Nitta, A.; Yoneyama, M.; Ogita, K.; Yoneda, Y.; Nabeshima, T.; et al. Chronic restraint stress impairs neurogenesis and hippocampus-dependent fear memory in mice: Possible involvement of a brain-specific transcription factor Npas4. J. Neurochem. 2010, 114, 1840–1851. [Google Scholar] [CrossRef] [PubMed]
- Hollands, C.; Tobin, M.K.; Hsu, M.; Musaraca, K.; Yu, T.S.; Mishra, R.; Kernie, S.G.; Lazarov, O. Depletion of adult neurogenesis exacerbates cognitive deficits in Alzheimer’s disease by compromising hippocampal inhibition. Mol. Neurodegener. 2017, 12, 64. [Google Scholar] [CrossRef]
- Anacker, C.; Hen, R. Adult hippocampal neurogenesis and cognitive flexibility—Linking memory and mood. Nat. Rev. Neurosci. 2017, 18, 335–346. [Google Scholar] [CrossRef]
- Gonçalves, J.T.; Schafer, S.T.; Gage, F.H. Adult neurogenesis in the hippocampus: From stem cells to behavior. Cell 2016, 167, 897–914. [Google Scholar] [CrossRef] [PubMed]
- Lieberwirth, C.; Pan, Y.; Liu, Y.; Zhang, Z.; Wang, Z. Hippocampal adult neurogenesis: Its regulation and potential role in spatial learning and memory. Brain Res. 2016, 1644, 127–140. [Google Scholar] [CrossRef]
- Li, C.C.; Jiang, N.; Gan, L.; Zhao, M.J.; Chang, Q.; Liu, X.M.; Pan, R.L. Peripheral and cerebral abnormalities of the tryptophan metabolism in the depression-like rats induced by chronic unpredicted mild stress. Neurochem. Int. 2020, 138, 104771. [Google Scholar] [CrossRef] [PubMed]
- Cattelan Souza, L.; de Brito, M.L.O.; Jesse, C.R.; Boeira, S.P.; de Gomes, M.G.; Goes, A.T.R.; Fabbro, L.D.; Machado, F.R.; Prigol, M.; Nogueira, C.W. Involvement of kynurenine pathway in depressive-like behaviour induced by nandrolone decanoate in mice. Steroids 2020, 164, 108727. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Sun, Y.; He, Z.; Xu, Y.; Li, X.; Ding, J.; Lu, M.; Hu, G. Kynurenine regulates NLRP2 inflammasome in astrocytes and its implications in depression. Brain Behav. Immun. 2020, 88, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.E.; Stevens, D.R.; Haas, H.L. The physiology of brain histamine. Prog. Neurobiol. 2001, 63, 637–672. [Google Scholar] [CrossRef]
- Thakkar, M.M. Histamine in the regulation of wakefulness. Sleep Med. Rev. 2011, 15, 65–74. [Google Scholar] [CrossRef]
- Rao, T.P.; Ozeki, M.; Juneja, L.R. In Search of a Safe Natural Sleep Aid. J. Am. Coll. Nutr. 2015, 34, 436–447. [Google Scholar] [CrossRef]
- Hipkiss, A.R. Possible Benefit of Dietary Carnosine towards Depressive Disorders. Aging Dis. 2015, 6, 300–303. [Google Scholar] [CrossRef]
- Tomonaga, S.; Yamane, H.; Onitsuka, E.; Yamada, S.; Sato, M.; Takahata, Y.; Morimatsu, F.; Furuse, M. Carnosine-induced antidepressant-like activity in rats. Pharmacol. Biochem. Behav. 2008, 89, 627–632. [Google Scholar] [CrossRef]
- Nakamura, Y. Characteristics of Japanese Green Tea. In Health Benefits of Green Tea. An Evidence-Based Approach; Hara, Y., Yang, C.S., Isemura, M., Tomita, I., Eds.; CAB International: Wallingford, UK, 2017; pp. 9–18. [Google Scholar]
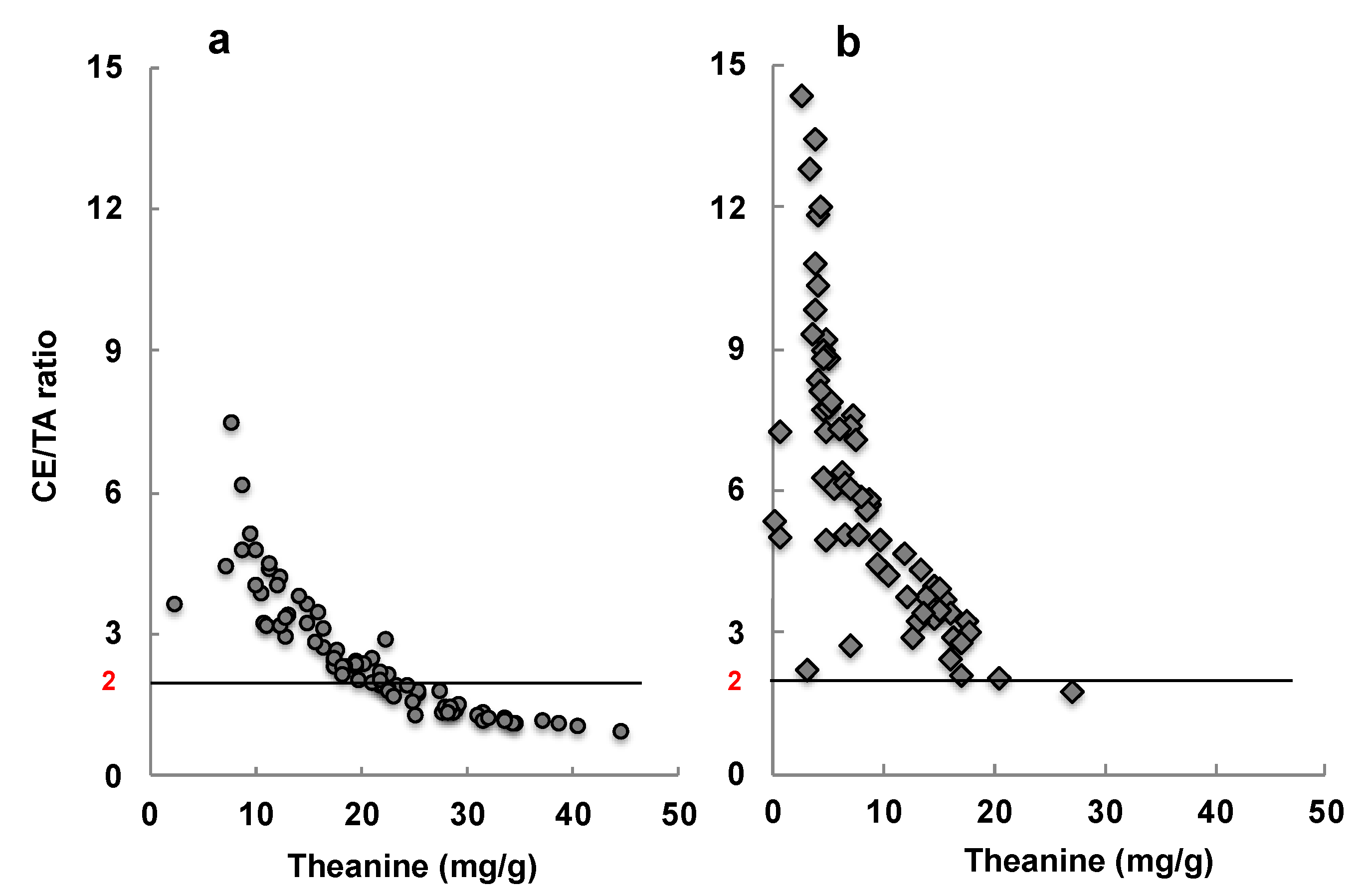
- Monobe, M.; Ema, K.; Tokuda, Y.; Maeda-Yamamoto, M. Effect on the epigallocatechin gallate/epigallocatechin ratio in a green tea (Camellia sinensis L.) extract of different extraction temperatures and its effect on IgA production in mice. Biosci. Biotechnol. Biochem. 2010, 74, 2501–2503. [Google Scholar] [CrossRef] [PubMed]
- Nater, U.M.; Rohleder, N. Salivary alpha-amylase as a non-invasive biomarker for the sympathetic nervous system: Current state of research. Psychoneuroendocrinology 2009, 34, 486–496. [Google Scholar] [CrossRef]
- Unno, K.; Yamada, H.; Iguchi, K.; Ishida, H.; Iwao, Y.; Morita, A.; Nakamura, Y. Anti-stress Effect of Green Tea with Lowered Caffeine on Humans: A Pilot Study. Biol. Pharm. Bull. 2017, 40, 902–909. [Google Scholar] [CrossRef]
- Unno, K.; Noda, S.; Kawasaki, Y.; Yamada, H.; Morita, A.; Iguchi, K.; Nakamura, Y. Reduced Stress and Improved Sleep Quality Caused by Green Tea Are Associated with a Reduced Caffeine Content. Nutrients 2017, 9, 777. [Google Scholar] [CrossRef] [PubMed]
- Unno, K.; Noda, S.; Kawasaki, Y.; Yamada, H.; Morita, A.; Iguchi, K.; Nakamura, Y. Ingestion of green tea with lowered caffeine improves sleep quality of the elderly via suppression of stress. J. Clin. Biochem. Nutr. 2017, 61, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Patel, D.; Steinberg, J.; Patel, P. Insomnia in the Elderly: A Review. J. Clin. Sleep Med. 2018, 14, 1017–1024. [Google Scholar] [CrossRef]
- Maness, D.L.; Khan, M. Nonpharmacologic Management of Chronic Insomnia. Am. Fam. Physician 2015, 92, 1058–1064. [Google Scholar] [PubMed]
- Unno, K.; Furushima, D.; Hamamoto, S.; Iguchi, K.; Yamada, H.; Morita, A.; Pervin, M.; Nakamura, Y. Stress-reducing effect of cookies containing matcha green tea: Essential ratio among theanine, arginine, caffeine and epigallocatechin gallate. Heliyon 2019, 5, e01653. [Google Scholar] [CrossRef]
- Morita, A.; Ikka, T.; Kunihiro, A. Chemical composition of new shoots in the first crop season of “White Leaf Tea” cultivated in Japan. Tea Res. J. 2011, 111, 63–72. [Google Scholar]
- Unno, K.; Furushima, D.; Nomura, Y.; Yamada, H.; Iguchi, K.; Taguchi, K.; Suzuki, T.; Ozeki, M.; Nakamura, Y. Antidepressant Effect of Shaded White Leaf Tea Containing High Levels of Caffeine and Amino Acids. Molecules 2020, 25, 3550. [Google Scholar] [CrossRef] [PubMed]
- Rothenberg, D.O.; Zhang, L. Mechanisms Underlying the anti-depressive effects of regular tea consumption. Nutrients 2019, 11, 1361. [Google Scholar] [CrossRef]
- John, J.; Kodama, T.; Siegel, J.M. Caffeine promotes glutamate and histamine release in the posterior hypothalamus. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2014, 307, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Chou, C.W.; Huang, W.J.; Tien, L.T.; Wang, S.J. (−)-Epigallocatechin gallate, the most active polyphenolic catechin in green tea, presynaptically facilitates Ca2+-dependent glutamate release via activation of protein kinase C in rat cerebral cortex. Synapse 2007, 61, 889–902. [Google Scholar] [CrossRef]
| Sample | Coexistence Sample | Permeability Coefficient (10−6 cm/s) | BBB Permeability (%) (0.5 h) |
|---|---|---|---|
| EGCG | 13.45 ± 0.57 | 4.00 ± 0.17 | |
| EGCG | EGC | 10.53 ± 0.63 | 3.13 ± 0.19 |
| EGC | 16.70 ± 1.86 | 4.96 ± 0.55 | |
| EGC | EGCG | 12.39 ± 1.56 | 3.68 ± 0.47 |
| EGC | GA | 6.01 ± 1.57 | 1.79 ± 0.47 |
| GA | 31.73 ± 3.39 | 9.42 ± 1.01 | |
| GA | EGC | 26.98 ± 2.77 | 8.01 ± 0.82 |
| EGC-M5 | 17.99 ± 0.79 | 5.34 ± 0.23 | |
| EGC-M5-Sul | 14.61 ± 1.35 | 4.34 ± 0.40 | |
| EGC-M5-GlcUA | 12.53 ± 0.02 | 3.72 ± 0.01 | |
| Pyrogallol | 13.79 ± 1.62 | 4.10 ± 0.48 | |
| Pyro-GlcUA | 9.28 ± 1.41 | 2.76 ± 0.42 | |
| Caffeine | 45.22 ± 4.41 | 13.43 ± 1.31 |
| Green Tea | Caffeine (mg/L) | Catechins (mg/L) | ||||||||
| EGCG | EGC | ECG | EC | CG | (+) C | Total | ||||
| Standard | 120 | 117 | 73.2 | 26.5 | 43.9 | 0.41 | 5.47 | 266 | ||
| Low -caffeine | 37.2 | 64.7 | 145 | 10.7 | 53.8 | 0.26 | 4.13 | 278 | ||
| Green tea | Free amino acids (mg/L) | |||||||||
| Theanine | Glu | Arg | Asp | Gln | Ser | Ala | Asn | GABA | Total | |
| Standard | 36.0 | 9.12 | 7.28 | 7.44 | 5.34 | 2.99 | 0.90 | 0.76 | 0.47 | 70.4 |
| Low -caffeine | 85.2 | 18.2 | 21.6 | 12.9 | 15.8 | 5.73 | 2.16 | 1.21 | 1.51 | 164 |
| Tea | Caffeine (mg/L) | Catechins (mg/L) | |||||||
| EGCG | EGC | ECG | EC | GC | CG | (+) C | |||
| SWLT | 209.8 | 150.4 | 135.2 | 24.6 | 41.0 | 5.0 | 2.8 | 3.4 | |
| Sencha | 112.0 | 134.2 | 229.0 | 21.0 | 46.6 | 13.6 | 4.6 | 2.0 | |
| Tea | Free amino acids (mg/L) | ||||||||
| Theanine | Arg | Gln | Asn | Asp | Glu | Ser | GABA | Total | |
| SWLT | 140.2 | 69.9 | 51.7 | 33.8 | 33.5 | 19.3 | 12.6 | 0 | 361.0 |
| Sencha | 28.8 | 5.4 | 3.9 | 0.7 | 5.5 | 6.9 | 2.2 | 0 | 53.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Unno, K.; Nakamura, Y. Green Tea Suppresses Brain Aging. Molecules 2021, 26, 4897. https://doi.org/10.3390/molecules26164897
Unno K, Nakamura Y. Green Tea Suppresses Brain Aging. Molecules. 2021; 26(16):4897. https://doi.org/10.3390/molecules26164897
Chicago/Turabian StyleUnno, Keiko, and Yoriyuki Nakamura. 2021. "Green Tea Suppresses Brain Aging" Molecules 26, no. 16: 4897. https://doi.org/10.3390/molecules26164897
APA StyleUnno, K., & Nakamura, Y. (2021). Green Tea Suppresses Brain Aging. Molecules, 26(16), 4897. https://doi.org/10.3390/molecules26164897






