Recent Advances in Biopolymer-Based Dye Removal Technologies
Abstract
:1. Introduction
2. Different Types of Dyes
3. Biopolymer-based Dye Removal Technologies
3.1. Adsorption
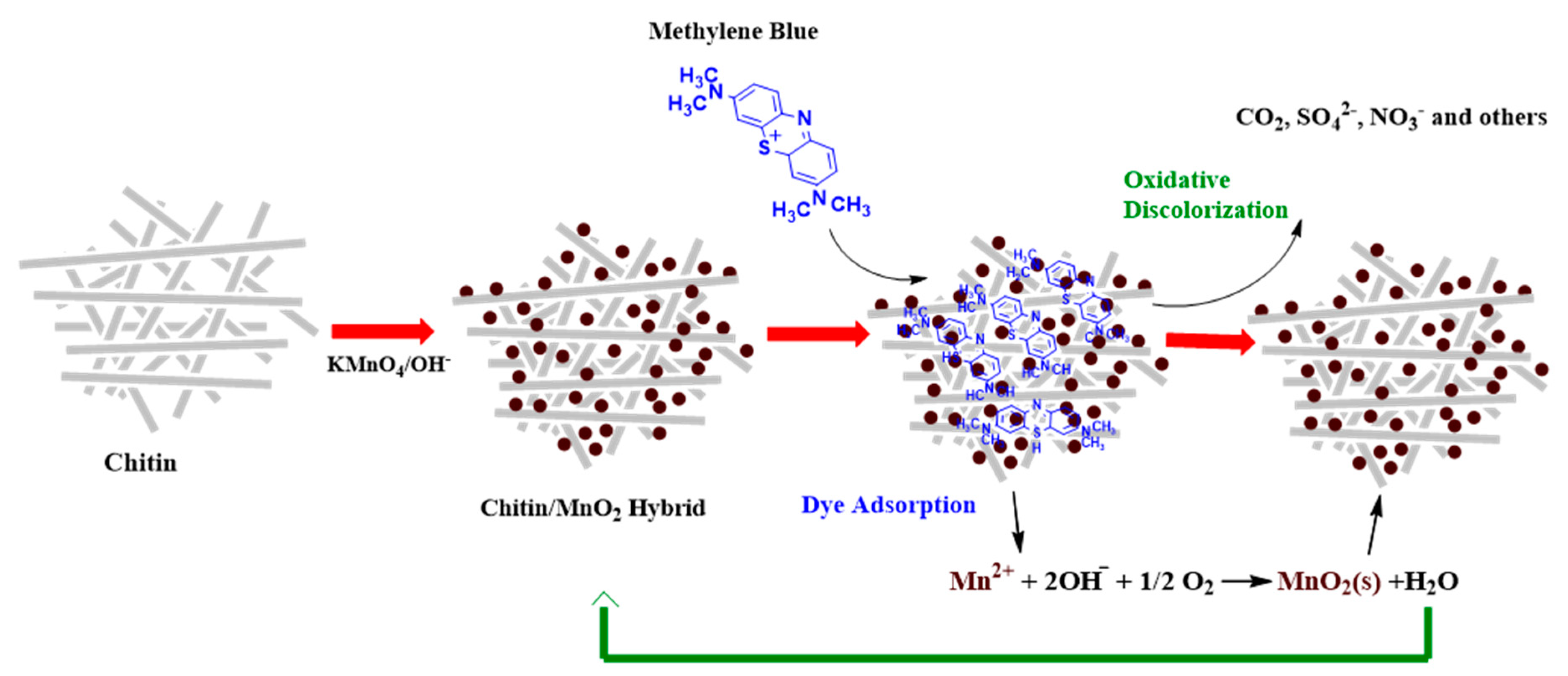
3.2. Advanced Oxidation Processes (AOPs)
3.3. Membrane Filtration
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Hussain, S.; Khan, N.; Gul, S.; Khan, S.; Khan, H. Contamination of Water Resources by Food Dyes and Its Removal Technologies. In Water Chemistry; Eyvaz, M., Yüksel, E., Eds.; Intech Open: London, UK, 2020. [Google Scholar]
- Acharya, S. Cationization of Cotton Fabric for Improved Dye Uptake. Master’s Thesis, Texas Tech University, Lubbock, TX, USA, 2012. [Google Scholar]
- Siva, R. Status of natural dyes and dye-yielding plants in India. Curr. Sci. 2007, 92, 916–925. [Google Scholar]
- Saini, R.D. Textile Organic Dyes: Polluting effects and Elimination Methods from Textile Waste Water. Int. J. Chem. Eng. Res. 2017, 9, 975–6442. [Google Scholar]
- Cañamares, M.V.; Reagan, D.A.; Lombardi, J.R.; Leona, M. TLC-SERS of mauve, the first synthetic dye. J. Raman Spectrosc. 2014, 45, 1147–1152. [Google Scholar] [CrossRef] [Green Version]
- Jamee, R.; Siddique, R. Biodegradation of synthetic dyes of textile effluent by microorganisms: An environmentally and economically sustainable approach. Eur. J. Microbiol. Immunol. 2019, 9, 114–118. [Google Scholar] [CrossRef]
- Rauf, M.A.; Salman Ashraf, S. Survey of recent trends in biochemically assisted degradation of dyes. Chem. Eng. J. 2012, 209, 520–530. [Google Scholar] [CrossRef]
- Katheresan, V.; Kansedo, J.; Lau, S.Y. Efficiency of various recent wastewater dye removal methods: A review. J. Environ. Chem. Eng. 2018, 6, 4676–4697. [Google Scholar] [CrossRef]
- Abdi, J.; Vossoughi, M.; Mahmoodi, N.M.; Alemzadeh, I. Synthesis of amine-modified zeolitic imidazolate framework-8, ultrasound-assisted dye removal and modeling. Ultrason. Sonochem. 2017, 39, 550–564. [Google Scholar] [CrossRef]
- Abdi, J.; Vossoughi, M.; Mahmoodi, N.M.; Alemzadeh, I. Synthesis of metal-organic framework hybrid nanocomposites based on GO and CNT with high adsorption capacity for dye removal. Chem. Eng. J. 2017, 326, 1145–1158. [Google Scholar] [CrossRef]
- Zollinger, H. Color Chemistry: Syntheses, Properties, and Applications of Organic Dyes and Pigments, 3rd ed.; Wiley-VCH: Weinheim, Germany, 2003; ISBN 3906390233. [Google Scholar]
- Wang, Z.; Xue, M.; Huang, K.; Liu, Z. Textile Dyeing Wastewater Treatment. Adv. Treat. Text. Effl. 2011. [Google Scholar] [CrossRef] [Green Version]
- Chequer, F.D.; De Oliveira, G.R.; Ferraz, E.A.; Cardoso, J.C.; Zanoni, M.B.; de Oliveira, D.P. Textile Dyes: Dyeing Process and Environmental Impact. In Eco-Friendly Textile Dyeing and Finishing; Intech Open: London, UK, 2013; pp. 151–176. [Google Scholar]
- Joshi, M.; Bansal, R.; Purwar, R. Colour removal from textile effluents. Indian J. Fibre Text. Res. 2004, 29, 239–259. [Google Scholar]
- Crini, G. Non-conventional low-cost adsorbents for dye removal: A review. Bioresour. Technol. 2006, 97, 1061–1085. [Google Scholar] [CrossRef]
- Forgacs, E.; Cserháti, T.; Oros, G. Removal of synthetic dyes from wastewaters: A review. Environ. Int. 2004, 30, 953–971. [Google Scholar] [CrossRef]
- Isah, U.A.; Abdulraheem, G.; Bala, S.; Muhammad, S.; Abdullahi, M. Kinetics, equilibrium and thermodynamics studies of C.I. Reactive Blue 19 dye adsorption on coconut shell based activated carbon. Int. Biodeterior. Biodegrad. 2015, 102, 265–273. [Google Scholar] [CrossRef]
- Weber, E.J.; Stickney, V.C. Hydrolysis kinetics of Reactive Blue 19-Vinyl Sulfone. Water Res. 1993, 27, 63–67. [Google Scholar] [CrossRef]
- Fatima, M.; Farooq, R.; Lindström, R.W.; Saeed, M. A review on biocatalytic decomposition of azo dyes and electrons recovery. J. Mol. Liq. 2017, 246, 275–281. [Google Scholar] [CrossRef]
- Oblak, R.; Kete, M.; Štangar, U.L.; Tasbihi, M. Alternative support materials for titania photocatalyst towards degradation of organic pollutants. J. Water Process Eng. 2018, 23, 142–150. [Google Scholar] [CrossRef]
- Khan, R.; Patel, V.; Khan, Z. Bioremediation of dyes from textile and dye manufacturing industry effluent. In Abatement of Environmental Pollutants: Trends and Strategies; Elsevier: Amsterdam, The Netherlands, 2020; pp. 107–125. [Google Scholar]
- Rodríguez-Couto, S.; Osma, J.F.; Toca-Herrera, J.L. Removal of synthetic dyes by an eco-friendly strategy. Eng. Life Sci. 2009, 9, 116–123. [Google Scholar] [CrossRef]
- Zhou, Y.; Lu, J.; Zhou, Y.; Liu, Y. Recent advances for dyes removal using novel adsorbents: A review. Environ. Pollut. 2019, 252, 352–365. [Google Scholar] [CrossRef] [PubMed]
- Zazouli, M.A.; Kalankesh, L.R. Removal of precursors and disinfection by-products (DBPs) by membrane filtration from water; a review. J. Environ. Health Sci. Eng. 2017, 15, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lofrano, G.; Meric, S. A comprehensive approach to winery wastewater treatment: A review of the state-of the-art. Desalin. Water Treat. 2016, 57, 3011–3028. [Google Scholar] [CrossRef]
- Deng, Y.; Zhao, R. Advanced Oxidation Processes (AOPs) in Wastewater Treatment. Curr. Pollut. Rep. 2015, 1, 167–176. [Google Scholar] [CrossRef] [Green Version]
- Zouboulis, A.I.; Moussas, P.A.; Tzoupanos, N.D. Biosorbents based on lignin used in biosrption processes from wastewater treatment: A review. In Lignin; Nova Science Publishers: Hauppauge, NY, USA, 2018; pp. 289–324. [Google Scholar]
- Broadbent, A.D. Basic Principles of Textile Coloration; Society of Dyers and Colorists: Bradford, UK, 2001; ISBN 090195676727. [Google Scholar]
- Pal, P. Chapter 6 - Industry-Specific Water Treatment: Case Studies. In Industrial Water Treatment Process Technology; Pal, P., Ed.; Butterworth-Heinemann: Oxford, UK, 2017; pp. 243–511. ISBN 978-0-12-810391-3. [Google Scholar]
- Cotillas, S.; Llanos, J.; Cañizares, P.; Clematis, D.; Cerisola, G.; Rodrigo, M.A.; Panizza, M. Removal of Procion Red MX-5B dye from wastewater by conductive-diamond electrochemical oxidation. Electrochim. Acta 2018, 263, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Orzel, J.; Daszykowski, M.; Grabowski, I.; Zaleszczyk, G.; Sznajder, M.; Walczak, B. Simultaneous determination of Solvent Yellow 124 and Solvent Red 19 in diesel oil using fluorescence spectroscopy and chemometrics. Talanta 2012, 101, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Steter, J.R.; Barros, W.R.P.; Lanza, M.R.V.; Motheo, A.J. Electrochemical and sonoelectrochemical processes applied to amaranth dye degradation. Chemosphere 2014, 117, 200–207. [Google Scholar] [CrossRef] [Green Version]
- Pedro Silva, J.; Sousa, S.; Rodrigues, J.; Antunes, H.; Porter, J.J.; Gonçalves, I.; Ferreira-Dias, S. Adsorption of acid orange 7 dye in aqueous solutions by spent brewery grains. Sep. Purif. Technol. 2004, 40, 309–315. [Google Scholar] [CrossRef]
- Ahmad, A.; Mohd-Setapar, S.H.; Chuong, C.S.; Khatoon, A.; Wani, W.A.; Kumar, R.; Rafatullah, M. Recent advances in new generation dye removal technologies: Novel search for approaches to reprocess wastewater. RSC Adv. 2015, 5, 30801–30818. [Google Scholar] [CrossRef]
- Kyzioł, A.; Kyzioł, K. Surface Functionalization With Biopolymers via Plasma-Assisted Surface Grafting and Plasma-Induced Graft Polymerization—Materials for Biomedical Applications. In Biopolymer Grafting Applications; Thakur, V.K., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 115–151. ISBN 978-0-12-810462-. [Google Scholar]
- Dąbrowski, A. Adsorption—From theory to practice. Adv. Colloid Interface Sci. 2001, 93, 135–224. [Google Scholar] [CrossRef]
- Yagub, M.T.; Sen, T.K.; Afroze, S.; Ang, H.M. Dye and its removal from aqueous solution by adsorption: A review. Adv. Colloid Interface Sci. 2014, 209, 172–184. [Google Scholar] [CrossRef]
- Zhang, P.; Chen, Y.-P.; Wang, W.; Shen, Y.; Guo, J.-S. Surface plasmon resonance for water pollutant detection and water process analysis. TrAC Trends Anal. Chem. 2016, 85, 153–165. [Google Scholar] [CrossRef]
- Pashin, J.C. Chapter 9—Coal as a Petroleum Source Rock and Reservoir Rock. In Applied Coal Petrology; Suárez-Ruiz, I., Crelling, J.C., Eds.; Elsevier: Burlington, MA, USA, 2008; pp. 227–262. ISBN 978-0-08-045051-3. [Google Scholar]
- Abbasi, Z.; Cseri, L.; Zhang, X.; Ladewig, B.P.; Wang, H. Chapter 7—Metal–Organic Frameworks (MOFs) and MOF-Derived Porous Carbon Materials for Sustainable Adsorptive Wastewater Treatment. In Sustainable Nanoscale Engineering; Szekely, G., Livingston, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 163–194. ISBN 978-0-12-814681-1. [Google Scholar]
- Gunawardhana, B.P.N.; Gunathilake, C.A.; Dayananda, K.E.D.Y.T.; Dissanayake, D.M.S.N.; Mantilaka, M.M.M.G.P.G.; Kalpage, C.S.; Rathnayake, R.M.L.D.; Rajapakse, R.M.G.; Manchanda, A.S.; Etampawala, T.N.B.; et al. Synthesis of Hematite Nanodiscs from Natural Laterites and Investigating Their Adsorption Capability of Removing Ni2+ and Cd2+ Ions from Aqueous Solutions. J. Compos. Sci. 2020, 4, 57. [Google Scholar] [CrossRef]
- Rezakazemi, M.; Shirazian, S. Lignin-chitosan blend for methylene blue removal: Adsorption modeling. J. Mol. Liq. 2019, 274, 778–791. [Google Scholar] [CrossRef]
- GUPTA, V.K.; MOHAN, D.; SHARMA, S.; SHARMA, M. Removal of Basic Dyes (Rhodamine B and Methylene Blue) from Aqueous Solutions Using Bagasse Fly Ash. Sep. Sci. Technol. 2000, 35, 2097–2113. [Google Scholar] [CrossRef]
- Mondal, S.; Purakait, M.K.; De, S. Advances in Dye Removal Technologies, 1st ed.; Springer: Singapore, 2018. [Google Scholar]
- Malik, P.K. Dye removal from wastewater using activated carbon developed from sawdust: Adsorption equilibrium and kinetics. J. Hazard. Mater. 2004, 113, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Baysal, M.; Bilge, K.; Yılmaz, B.; Papila, M.; Yürüm, Y. Preparation of high surface area activated carbon from waste-biomass of sunflower piths: Kinetics and equilibrium studies on the dye removal. J. Environ. Chem. Eng. 2018, 6, 1702–1713. [Google Scholar] [CrossRef] [Green Version]
- Sabzehmeidani, M.M.; Mahnaee, S.; Ghaedi, M.; Heidari, H.; Roy, V.A.L. Carbon based materials: A review of adsorbents for inorganic and organic compounds. Mater. Adv. 2021, 2, 598–627. [Google Scholar] [CrossRef]
- Gupta, V.K. Suhas Application of low-cost adsorbents for dye removal—A review. J. Environ. Manag. 2009, 90, 2313–2342. [Google Scholar] [CrossRef]
- Kaşgöz, H.; Durmus, A. Dye removal by a novel hydrogel-caly nanocomposite with enhanced swellin properties. Polym. Adv. Technol. 2008, 19, 838–845. [Google Scholar] [CrossRef]
- Shahadat, M.; Isamil, S. Regeneration performance of clay-based adsorbents for the removal of industrial dyes: A review. RSC Adv. 2018, 8, 24571–24587. [Google Scholar] [CrossRef]
- Alver, E.; Metin, A.Ü. Anionic dye removal from aqueous solutions using modified zeolite: Adsorption kinetics and isotherm studies. Chem. Eng. J. 2012, 200–202, 59–67. [Google Scholar] [CrossRef]
- Piri, F.; Mollahosseini, A.; Khadir, A.; Milani Hosseini, M. Enhanced adsorption of dyes on microwave-assisted synthesized magnetic zeolite-hydroxyapatite nanocomposite. J. Environ. Chem. Eng. 2019, 7, 103338. [Google Scholar] [CrossRef]
- Homaeigohar, S. The Nanosized Dye Adsorbents for Water Treatment. Nanomaterials 2020, 10, 295. [Google Scholar] [CrossRef] [Green Version]
- Zare, E.N.; Motahari, A.; Sillanpää, M. Nanoadsorbents based on conducting polymer nanocomposites with main focus on polyaniline and its derivatives for removal of heavy metal ions/dyes: A review. Environ. Res. 2018, 162, 173–195. [Google Scholar] [CrossRef]
- Paradelo, R.; Vecino, X.; Moldes, A.B.; Barral, M.T. Potential use of composts and vermicomposts as low-cost adsorbents for dye removal: An overlooked application. Environ. Sci. Pollut. Res. 2019, 26, 21085–21097. [Google Scholar] [CrossRef]
- Sardar, M.; Manna, M.; Maharana, M.; Sen, S. Remediation of Dyes from Industrial Wastewater Using Low-Cost Adsorbents. In Green Adsorbents to Remove Metals, Dyes and Boron from Polluted Water; Inamuddin, Ahamed, M.I., Lichtfouse, E., Asiri, A.M., Eds.; Springer: Cham, Switzerland, 2021; pp. 377–403. [Google Scholar]
- Tony, M.A. An industrial ecology approach: Green cellulose-based bio-adsorbent from sugar industry residue for treating textile industry wastewater effluent. Int. J. Environ. Anal. Chem. 2021, 101, 167–183. [Google Scholar] [CrossRef]
- Wang, H.; Li, Z.; Yahyaoui, S.; Hanafy, H.; Seliem, M.K.; Bonilla-Petriciolet, A.; Luiz Dotto, G.; Sellaoui, L.; Li, Q. Effective adsorption of dyes on an activated carbon prepared from carboxymethyl cellulose: Experiments, characterization and advanced modelling. Chem. Eng. J. 2021, 417, 128116. [Google Scholar] [CrossRef]
- Yu, M.; Han, Y.; Li, J.; Wang, L. Magnetic N-doped carbon aerogel from sodium carboxymethyl cellulose/collagen composite aerogel for dye adsorption and electrochemical supercapacitor. Int. J. Biol. Macromol. 2018, 115, 185–193. [Google Scholar] [CrossRef]
- Wang, H.; Gong, Y.; Wang, Y. Cellulose-based hydrophobic carbon aerogels as versatile and superior adsorbents for sewage treatment. RSC Adv. 2014, 4, 45753–45759. [Google Scholar] [CrossRef]
- Harada, N.; Nakamura, J.; Uyama, H. Single-step fabrication and environmental applications of activated carbon-containing porous cellulose beads. React. Funct. Polym. 2021, 160, 104830. [Google Scholar] [CrossRef]
- Wan, Y.; Liu, Z.-Y.; Song, P.; Zhang, X.-Q.; Song, J.-C.; Fu, Y.-J.; Yao, X.-H.; Wang, J.; Chen, T.; Zhang, D.-Y.; et al. Ionic liquid groups modified 3D porous cellulose microspheres for selective adsorption of AO7 dye. J. Clean. Prod. 2019, 240, 118201. [Google Scholar] [CrossRef]
- Jiang, F.; Dinh, D.M.; Hsieh, Y.-L. Adsorption and desorption of cationic malachite green dye on cellulose nanofibril aerogels. Carbohydr. Polym. 2017, 173, 286–294. [Google Scholar] [CrossRef] [Green Version]
- Srasri, K.; Thongroj, M.; Chaijiraaree, P.; Thiangtham, S.; Manuspiya, H.; Pisitsak, P.; Ummartyotin, S. Recovery potential of cellulose fiber from newspaper waste: An approach on magnetic cellulose aerogel for dye adsorption material. Int. J. Biol. Macromol. 2018, 119, 662–668. [Google Scholar] [CrossRef]
- Li, Y.; Xiao, H.; Pan, Y.; Wang, L. Novel Composite Adsorbent Consisting of Dissolved Cellulose Fiber/Microfibrillated Cellulose for Dye Removal from Aqueous Solution. ACS Sustain. Chem. Eng. 2018, 6, 6994–7002. [Google Scholar] [CrossRef]
- Liang, L.; Zhang, S.; Goenaga, G.A.; Meng, X.; Zawodzinski, T.A.; Ragauskas, A.J. Chemically Cross-Linked Cellulose Nanocrystal Aerogels for Effective Removal of Cation Dye. Front. Chem. 2020, 8, 570. [Google Scholar] [CrossRef] [PubMed]
- Peter, S.; Lyczko, N.; Gopakumar, D.; Maria, H.J.; Nzihou, A.; Thomas, S. Chitin and Chitosan Based Composites for Energy and Environmental Applications: A Review. Waste Biomass Valoriz. 2021, 12, 4777–4804. [Google Scholar] [CrossRef]
- Kumar, D.; Gihar, S.; Shrivash, M.K.; Kumar, P.; Kundu, P.P. A review on the synthesis of graft copolymers of chitosan and their potential applications. Int. J. Biol. Macromol. 2020, 163, 2097–2112. [Google Scholar] [CrossRef]
- Gautam, D.; Saya, L.; Hooda, S. Fe3O4 loaded chitin—A promising nano adsorbent for Reactive Blue 13 dye. Environ. Adv. 2020, 2, 100014. [Google Scholar] [CrossRef]
- Kim, S.J.; Hong, B.M.; Park, W.H. The effects of chitin/chitosan nanowhiskers on the thermal, mechanical and dye adsorption properties of electrospun PVA nanofibrous membranes. Cellulose 2020, 27, 5771–5783. [Google Scholar] [CrossRef]
- Xu, X.; Yu, J.; Liu, C.; Yang, G.; Shi, L.; Zhuang, X. Xanthated chitosan/cellulose sponges for the efficient removal of anionic and cationic dyes. React. Funct. Polym. 2021, 160, 104840. [Google Scholar] [CrossRef]
- Karimi-Maleh, H.; Ranjbari, S.; Tanhaei, B.; Ayati, A.; Orooji, Y.; Alizadeh, M.; Karimi, F.; Salmanpour, S.; Rouhi, J.; Sillanpää, M.; et al. Novel 1-butyl-3-methylimidazolium bromide impregnated chitosan hydrogel beads nanostructure as an efficient nanobio-adsorbent for cationic dye removal: Kinetic study. Environ. Res. 2021, 195, 110809. [Google Scholar] [CrossRef]
- Song, Y.; Wang, Q.; Yang, W.; Chen, Q.; Zhou, Y.; Zhou, L. Chitosan-nickel oxide composite as an efficient adsorbent for removal of Congo red from aqueous solution. J. Dispers. Sci. Technol. 2021, 1–16. [Google Scholar] [CrossRef]
- Ramakrishnan, R.K.; Padil, V.V.T.; Wacławek, S.; Černík, M.; Varma, R.S. Eco-Friendly and Economic, Adsorptive Removal of Cationic and Anionic Dyes by Bio-Based Karaya Gum—Chitosan Sponge. Polymers 2021, 13, 251. [Google Scholar] [CrossRef]
- Morais da Silva, P.M.; Camparotto, N.G.; de Figueiredo Neves, T.; Grego Lira, K.T.; Mastelaro, V.R.; Siqueira Franco Picone, C.; Prediger, P. Effective removal of basic dye onto sustainable chitosan beads: Batch and fixed-bed column adsorption, beads stability and mechanism. Sustain. Chem. Pharm. 2020, 18, 100348. [Google Scholar] [CrossRef]
- Lakkaboyana, S.K.; Soontarapa, K.; Vinaykumar; Marella, R.K.; Kannan, K. Preparation of novel chitosan polymeric nanocomposite as an efficient material for the removal of Acid Blue 25 from aqueous environment. Int. J. Biol. Macromol. 2021, 168, 760–768. [Google Scholar] [CrossRef]
- Dassanayake, R.S.; Rajakaruna, E.; Abidi, N. Preparation of aerochitin-TiO2 composite for efficient photocatalytic degradation of methylene blue. J. Appl. Polym. Sci. 2018, 135, 1–10. [Google Scholar] [CrossRef]
- Pham, T.H.; Bui, H.M.; Bui, T.X. Chapter 13—Advanced oxidation processes for the removal of pesticides. In Current Developments in Biotechnology and Bioengineering; Varjani, S., Pandey, A., Tyagi, R.D., Ngo, H.H., Larroche, C., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 309–330. ISBN 978-0-12-819594-9. [Google Scholar]
- Wols, B.A.; Hofman-Caris, C.H.M. Review of photochemical reaction constants of organic micropollutants required for UV advanced oxidation processes in water. Water Res. 2012, 46, 2815–2827. [Google Scholar] [CrossRef]
- Danish, M.S.S.; Estrella, L.L.; Alemaida, I.M.A.; Lisin, A.; Moiseev, N.; Ahmadi, M.; Nazari, M.; Wali, M.; Zaheb, H.; Senjyu, T. Photocatalytic Applications of Metal Oxides for Sustainable Environmental Remediation. Metals 2021, 11, 80. [Google Scholar] [CrossRef]
- Javaid, R.; Qazi, U.Y. Catalytic Oxidation Process for the Degradation of Synthetic Dyes: An Overview. Int. J. Environ. Res. Public Health 2019, 16, 2066. [Google Scholar] [CrossRef] [Green Version]
- Dassanayake, R.S.; Rajakaruna, E.; Abidi, N. Borax-Cross-Linked Guar Gum-Manganese Dioxide Composites for Oxidative Decolorization of Methylene Blue. J. Nanomater. 2019, 2019, 7232715. [Google Scholar] [CrossRef]
- Victor Odhiambo, S.; Wilfrida, N.N. Advanced Oxidation Processes for Dye Removal From Wastewater. In Impact of Textile Dyes on Public Health and the Environment; Ahmad, W.K., Kumari, J.N., Rashid, B.A., Eds.; IGI Global: Harshey, PA, USA, 2020; pp. 205–238. [Google Scholar]
- Ogundare, S.A.; van Zyl, W.E. A review of cellulose-based substrates for SERS: Fundamentals, design principles, applications. Cellulose 2019, 26, 6489–6528. [Google Scholar] [CrossRef]
- Rajagopal, S.; Paramasivam, B.; Muniyasamy, K. Photocatalytic removal of cationic and anionic dyes in the textile wastewater by H2O2 assisted TiO2 and micro-cellulose composites. Sep. Purif. Technol. 2020, 252, 117444. [Google Scholar] [CrossRef]
- Rajeswari, A.; Vismaiya, S.; Pius, A. Preparation, characterization of nano ZnO-blended cellulose acetate-polyurethane membrane for photocatalytic degradation of dyes from water. Chem. Eng. J. 2017, 313, 928–937. [Google Scholar] [CrossRef]
- Thomas, M.; Naikoo, G.A.; Sheikh, M.U.D.; Bano, M.; Khan, F. Effective photocatalytic degradation of Congo red dye using alginate/carboxymethyl cellulose/TiO2 nanocomposite hydrogel under direct sunlight irradiation. J. Photochem. Photobiol. A Chem. 2016, 327, 33–43. [Google Scholar] [CrossRef]
- Ren, W.; Gao, J.; Lei, C.; Xie, Y.; Cai, Y.; Ni, Q.; Yao, J. Recyclable metal-organic framework/cellulose aerogels for activating peroxymonosulfate to degrade organic pollutants. Chem. Eng. J. 2018, 349, 766–774. [Google Scholar] [CrossRef]
- Zhu, Z.-S.; Qu, J.; Hao, S.-M.; Han, S.; Jia, K.-L.; Yu, Z.-Z. α-Fe2O3 Nanodisk/Bacterial Cellulose Hybrid Membranes as High-Performance Sulfate-Radical-Based Visible Light Photocatalysts under Stirring/Flowing States. ACS Appl. Mater. Interfaces 2018, 10, 30670–30679. [Google Scholar] [CrossRef]
- Diao, H.; Zhang, Z.; Liu, Y.; Song, Z.; Zhou, L.; Duan, Y.; Zhang, J. Facile fabrication of carboxylated cellulose nanocrystal–MnO2 beads for high-efficiency removal of methylene blue. Cellulose 2020, 27, 7053–7066. [Google Scholar] [CrossRef]
- Dassanayake, R.S.; Rajakaruna, E.; Moussa, H.; Abidi, N. One-pot synthesis of MnO2–chitin hybrids for effective removal of methylene blue. Int. J. Biol. Macromol. 2016, 93, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Drumm, F.C.; de Oliveira, J.S.; Foletto, E.L.; Dotto, G.L.; Moraes Flores, E.M.; Peters Enders, M.S.; Müller, E.I.; Janh, S.L. Response surface methodology approach for the optimization of tartrazine removal by heterogeneous photo-Fenton process using mesostructured Fe2O3-suppoted ZSM-5 prepared by chitin-templating. Chem. Eng. Commun. 2018, 205, 445–455. [Google Scholar] [CrossRef]
- Di, X.; Guo, F.; Zhu, Z.; Xu, Z.; Qian, Z.; Zhang, Q. In situ synthesis of ZnO-GO/CGH composites for visible light photocatalytic degradation of methylene blue. RSC Adv. 2019, 9, 41209–41217. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Y.; Kang, S.; Qin, L.; Wang, W.; Zhang, T.; Song, S.; Komarneni, S. Self-assembled gels of Fe-chitosan/montmorillonite nanosheets: Dye degradation by the synergistic effect of adsorption and photo-Fenton reaction. Chem. Eng. J. 2020, 379, 122322. [Google Scholar] [CrossRef]
- Aziz, A.; Ali, N.; Khan, A.; Bilal, M.; Malik, S.; Ali, N.; Khan, H. Chitosan-zinc sulfide nanoparticles, characterization and their photocatalytic degradation efficiency for azo dyes. Int. J. Biol. Macromol. 2020, 153, 502–512. [Google Scholar] [CrossRef]
- Saad, A.M.; Abukhadra, M.R.; Abdel-Kader Ahmed, S.; Elzanaty, A.M.; Mady, A.H.; Betiha, M.A.; Shim, J.-J.; Rabie, A.M. Photocatalytic degradation of malachite green dye using chitosan supported ZnO and Ce–ZnO nano-flowers under visible light. J. Environ. Manag. 2020, 258, 110043. [Google Scholar] [CrossRef]
- Yang, Y.; Ali, N.; Khan, A.; Khan, S.; Khan, S.; Khan, H.; Xiaoqi, S.; Ahmad, W.; Uddin, S.; Ali, N.; et al. Chitosan-capped ternary metal selenide nanocatalysts for efficient degradation of Congo red dye in sunlight irradiation. Int. J. Biol. Macromol. 2021, 167, 169–181. [Google Scholar] [CrossRef]
- Vigneshwaran, S.; Sirajudheen, P.; Nabeena, C.P.; Meenakshi, S. In situ fabrication of ternary TiO2 doped grafted chitosan/hydroxyapatite nanocomposite with improved catalytic performance for the removal of organic dyes: Experimental and systemic studies. Colloids Surf. A Physicochem. Eng. Asp. 2021, 611, 125789. [Google Scholar] [CrossRef]
- Hube, S.; Eskafi, M.; Hrafnkelsdóttir, K.F.; Bjarnadóttir, B.; Bjarnadóttir, M.Á.; Axelsdóttir, S.; Wu, B. Direct membrane filtration for wastewater treatment and resource recovery: A review. Sci. Total Environ. 2020, 710, 136375. [Google Scholar] [CrossRef]
- Speed, D.E. 10—Environmental aspects of planarization processes. In Advances in Chemical Mechanical Planarization (CMP); Babu, S., Ed.; Woodhead Publishing: Sawston, UK, 2016; pp. 229–269. ISBN 978-0-08-100165-3. [Google Scholar]
- Schlosser, Š. Membrane filtration. In Engineering Aspects of Food Biotechnology; Teixeira, J.A., Vicente, A.A., Eds.; CRC Press: Boca Raton, FL, USA, 2014; pp. 144–181. ISBN 9781439895467. [Google Scholar]
- Obotey Ezugbe, E.; Rathilal, S. Membrane Technologies in Wastewater Treatment: A Review. Membranes 2020, 10, 89. [Google Scholar] [CrossRef] [PubMed]
- Aliyu, U.M.; Rathilal, S.; Isa, Y.M. Membrane desalination technologies in water treatment: A review. Water Pract. Technol. 2018, 13, 738–752. [Google Scholar] [CrossRef]
- Song, Y.; Seo, J.Y.; Kim, H.; Beak, K.-Y. Structural control of cellulose nanofibrous composite membrane with metal organic framework (ZIF-8) for highly selective removal of cationic dye. Carbohydr. Polym. 2019, 222, 115018. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Feng, X. Polymer-enhanced ultrafiltration: Fundamentals, applications and recent developments. J. Memb. Sci. 2019, 586, 53–83. [Google Scholar] [CrossRef]
- Weng, R.; Chen, L.; Xiao, H.; Huang, F.; Lin, S.; Cao, S.; Huang, L. Preparation and characterization of cellulose nanofiltration membrane through hydrolysis followed by carboxymethylation. Fibers Polym. 2017, 18, 1235–1242. [Google Scholar] [CrossRef]
- Falca, G.; Musteata, V.-E.; Behzad, A.R.; Chisca, S.; Nunes, S.P. Cellulose hollow fibers for organic resistant nanofiltration. J. Memb. Sci. 2019, 586, 151–161. [Google Scholar] [CrossRef]
- Hu, Y.; Yue, M.; Yuan, F.; Yang, L.; Chen, C.; Sun, D. Bio-inspired fabrication of highly permeable and anti-fouling ultrafiltration membranes based on bacterial cellulose for efficient removal of soluble dyes and insoluble oils. J. Memb. Sci. 2021, 621. [Google Scholar] [CrossRef]
- Song, Y.; Sun, Y.; Chen, M.; Huang, P.; Li, T.; Zhang, X.; Jiang, K. Efficient removal and fouling-resistant of anionic dyes by nanofiltration membrane with phosphorylated chitosan modified graphene oxide nanosheets incorporated selective layer. J. Water Process. Eng. 2020, 34. [Google Scholar] [CrossRef]
- Bai, L.; Liu, Y.; Ding, A.; Ren, N.; Li, G.; Liang, H. Fabrication and characterization of thin-film composite (TFC) nanofiltration membranes incorporated with cellulose nanocrystals (CNCs) for enhanced desalination performance and dye removal. Chem. Eng. J. 2019, 358, 1519–1528. [Google Scholar] [CrossRef]
- Fradj, A.B.; Boubakri, A.; Hafiane, A.; Hamouda, S. Ben Removal of azoic dyes from aqueous solutions by chitosan enhanced ultrafiltration. Results Chem. 2020, 2, 100017. [Google Scholar] [CrossRef]
- Mokhena, T.C.; Luyt, A.S. Development of multifunctional nano/ultrafiltration membrane based on a chitosan thin film on alginate electrospun nanofibres. J. Clean. Prod. 2017, 156, 470–479. [Google Scholar] [CrossRef]
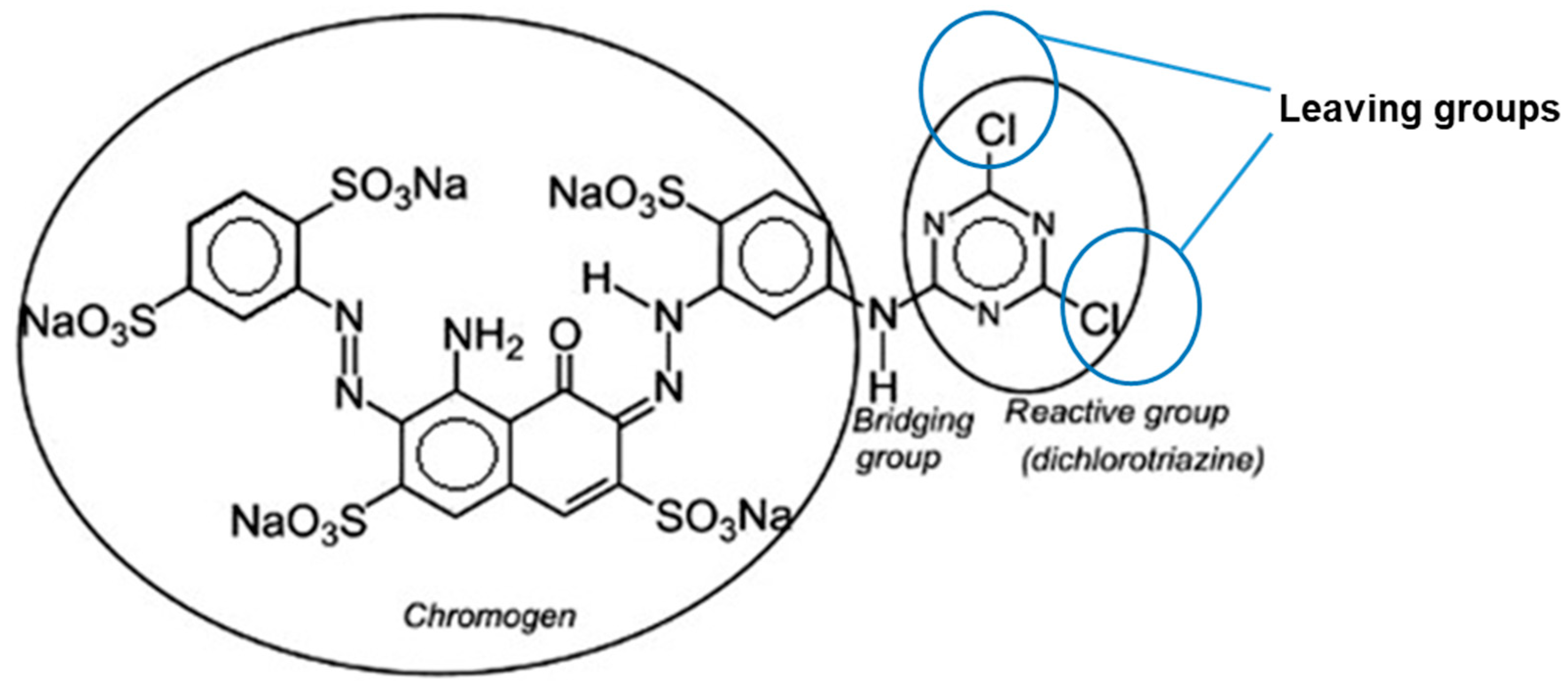
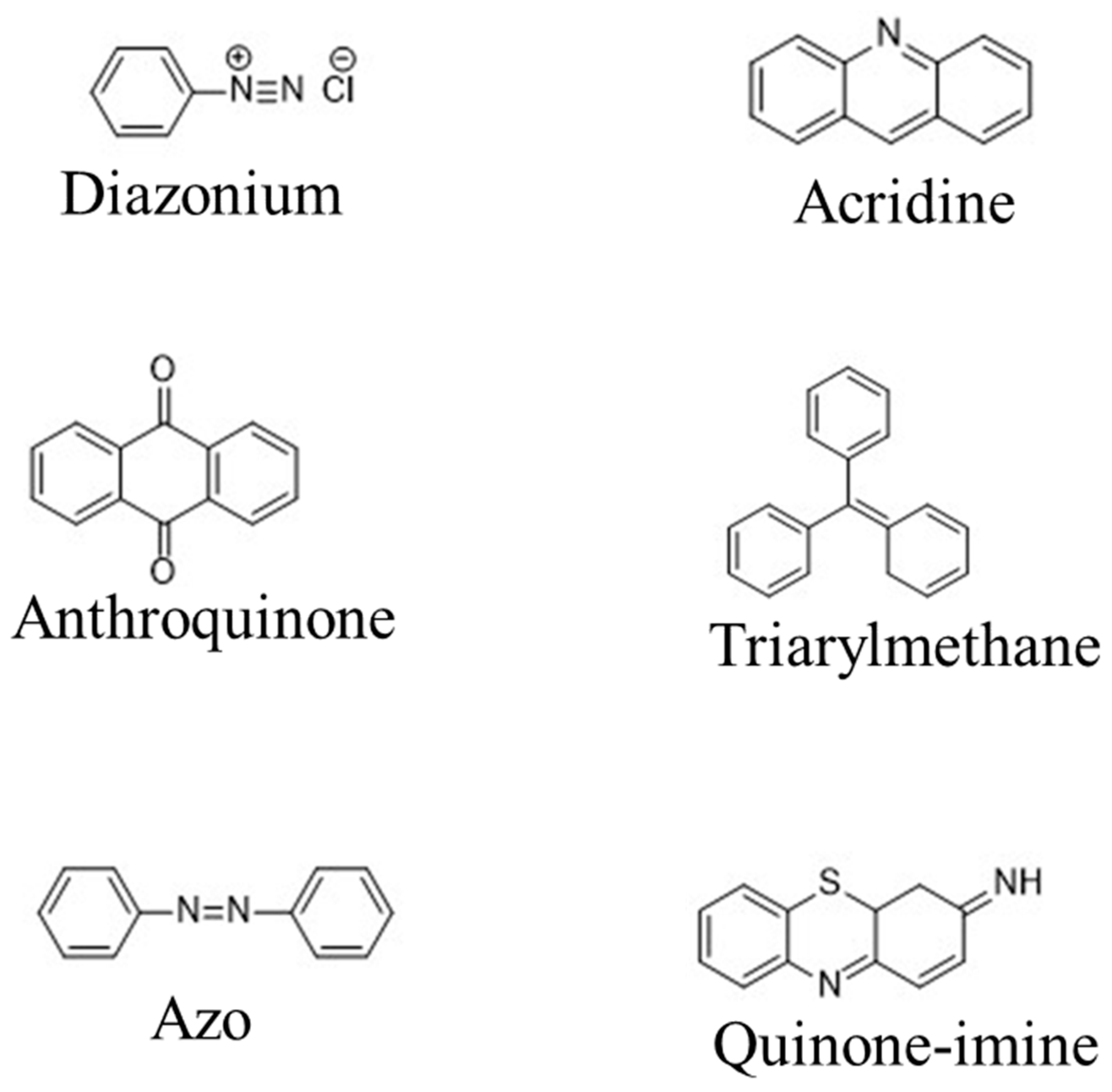
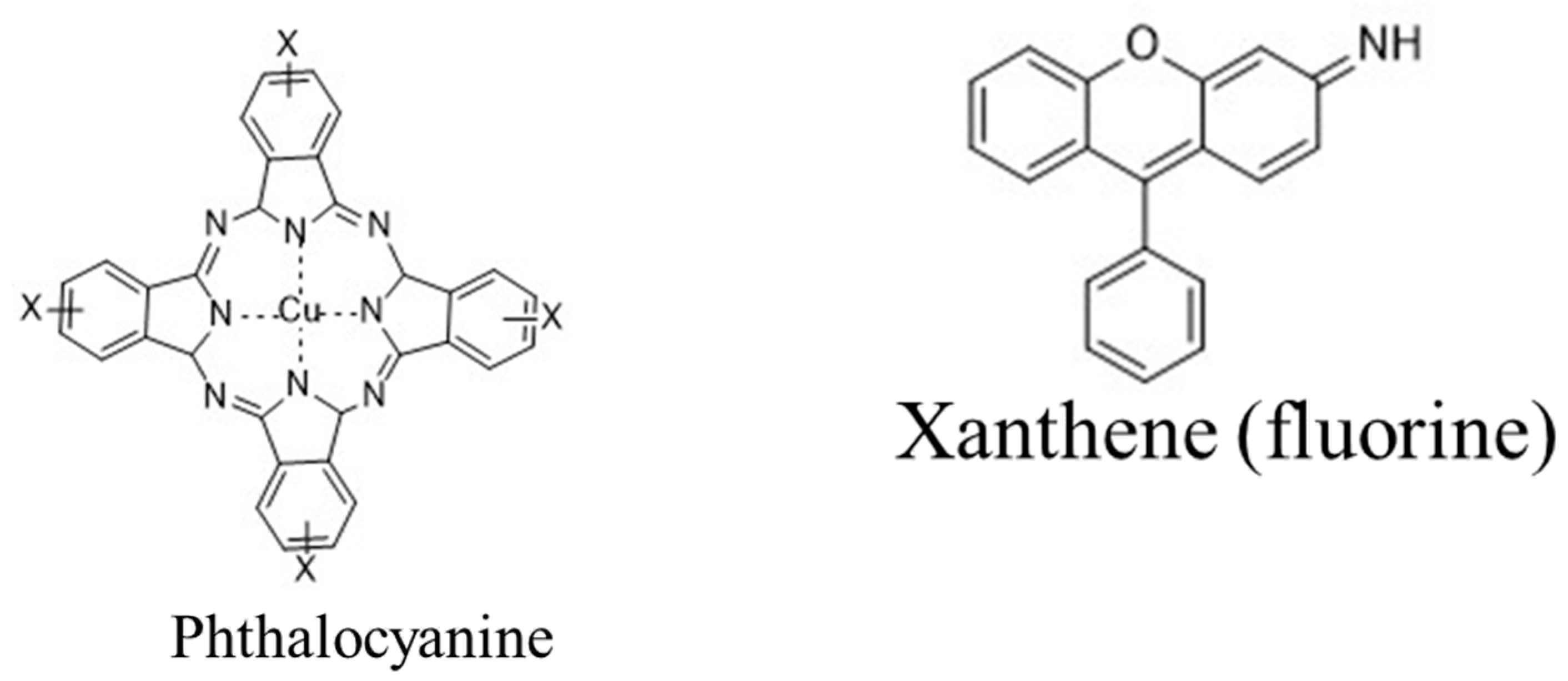
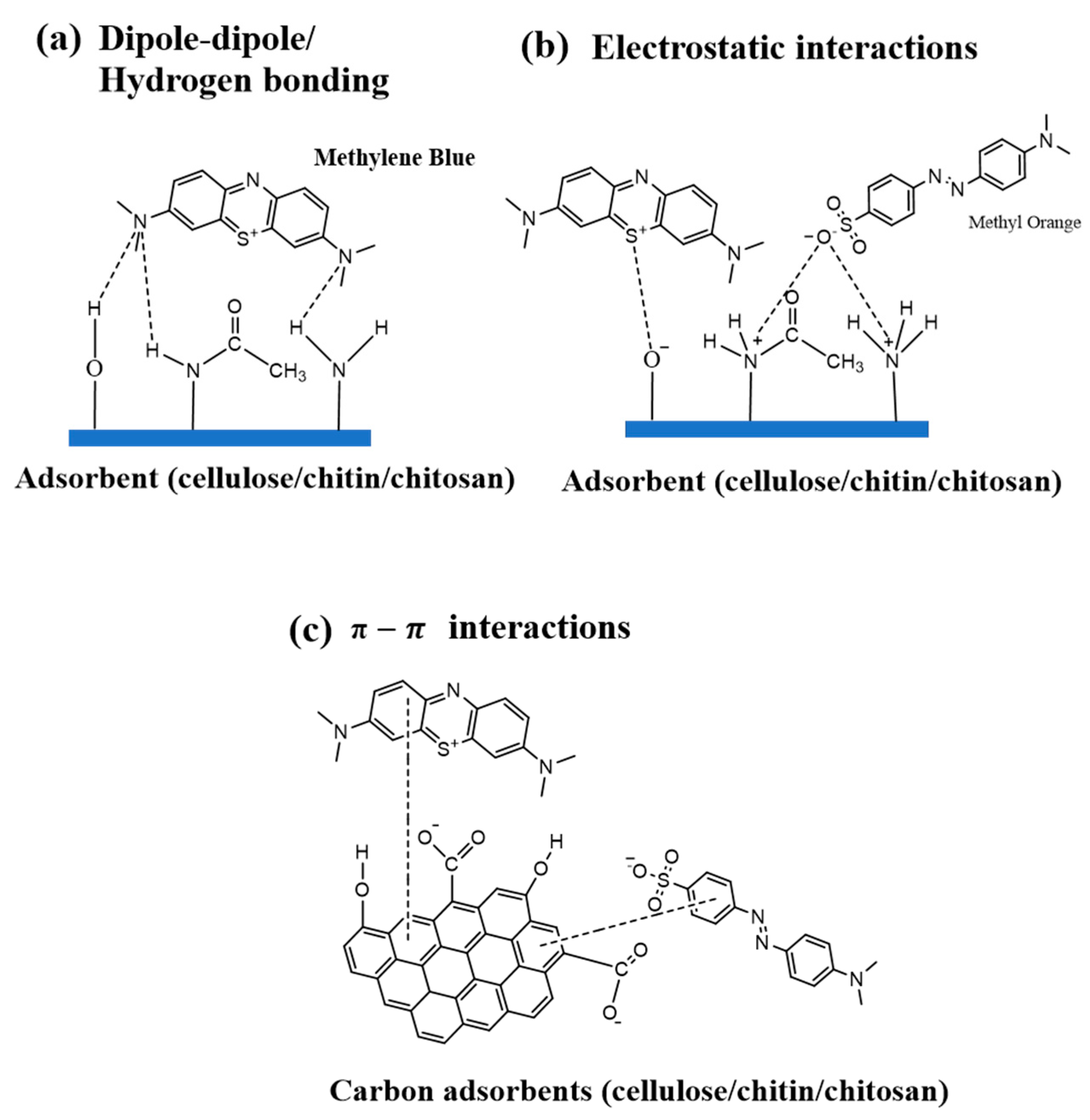
| Type | Water Solubility | Applications | Common Application Method | Example |
|---|---|---|---|---|
| Acid | Soluble | Cosmetics, food, leather, modified acrylics, nylon, paper, printing ink, silk and wool | In dye baths with neutral to acidic conditions | Acid Yellow 36 |
| Azo | Soluble/insoluble | Acetate, cellulose, cotton, rayon and polyester | Coupling component used to impregnate fiber and a solution of stabilized diazonium salt is used for treatment | Bluish Red azo dye |
| Basic | Soluble | Inks, medicine, modified nylon, modified polyester, paper, polyacrylonitrile, polyester, silk, tannin, mordanted cotton and wool | In dye baths with acidic conditions | Methylene Blue |
| Direct | Soluble | Cotton, leather, nylon, rayon, silk and paper | In dye baths with neutral or slightly alkaline conditions with additional electrolyte. | Direct Orange 26 |
| Disperse | Insoluble | Acetate, acrylic fibers, cellulose, cellulose acetate, nylon, polyamide, polyester, polyester–cotton and plastic | Padded on cloth and either baked or thermo-fixed at high pressure and temperature or low temperature carrier methods | Disperse Blue 27, Disperse Red 4, Disperse Yellow 3 |
| Fluorescent brighteners | All fibers, oils, paints, plastics and soaps as well as detergents | Mass dispersion, solution or suspension | 4,4′-bis (ethoxycarbonylvinyl) stilbene | |
| Food, drug, and cosmetics | Food, drug, and cosmetics | Mixing | Food Yellow 4 and tartrazine | |
| Mordant | Anodized aluminum, natural fibers, leather and wool | Along with chromium salts | Mordant Red 11 | |
| Oxidation bases | Cotton, fur and hair | The substrate is oxidized with aromatic amines and phenols | Direct Blue | |
| Reactive | Soluble | Cellulosic, cotton, nylon, silk and wool | Reaction between functional group on fiber and reactive group on dye. Covalently bonding under heat and alkaline pH | Reactive Blue 5 |
| Solvent | Insoluble | Fats, gasoline, inks, lacquers, lubricants, oils, plastics, stains, varnishes and waxes | Substrate dissolution | Solvent Red 26, Solvent Blue 35 |
| Sulfur | Cotton, leather, paper, polyamide fibers, rayon, silk and wood | Aromatic substrate vatted with sodium sulfide and reoxidized to insoluble sulfur-containing products on fiber | Sulfur Black 1 | |
| Vat | Insoluble | Cotton, cellulosic, polyester–cotton, rayon and wool | Water-insoluble dyes solubilized by reducing with sodium hydrogen sulfite, then exhausted on fiber and reoxidized | Vat Blue 4 (Indathrene) |
| Adsorbent (s) | Dye(s) | Maximum Adsorption Capacity, Qm (mg/g) | Ref. |
|---|---|---|---|
| Cellulose-based adsorbent derived from sugarcane bagasse | PB | 9.6 | [56] |
| Carboxymethyl cellulose-derived activated Carbon | MV AR CR | 1351.4 223.2 1779.5 | [57] |
| N-doped carboxymethyl cellulose-based carbon aerogels | MG MB CR TRB G133 | 238.2 230.4 85.2 73.3 | [58] |
| Cellulose-based carbon aerogels | MG MB | 1947 1192 | [59] |
| Activated carbon-containing cellulose beads | TB | 123.5 | [60] |
| 3D cellulose-based microspheres | AO7 | 218.6 | [61] |
| Cellulose nanofibril aerogels | MG | 212.7 | [62] |
| MCMFCs composite beads | MB | 303 | [64] |
| CNC/PMVEMA/PEG | MB | 116.2 | [65] |
| Fe3O4 loaded MCH NM | RB13 | 199.02 | [68] |
| Xanthated chitosan/cellulose sponges | MB CR | 213.220 289.855 | [70] |
| CS-NiO | CR | 188.68 | [72] |
| Kg-Ch sponge | MO MB | 32.81 32.62 | [73] |
| Chitosan-based beads | BB7 | 1410 | [74] |
| CS-PVA@CuO | AB 25 | 171.4 | [75] |
| Advantages | Disadvantages |
|---|---|
| Technologically simple, easy adaptability and handling | Economically non-viable due to the relatively high cost of adsorbents |
| High dye adsorption | Non-selective method and non-efficient with certain types of dyestuff |
| Highly effective process with fast adsorption kinetics | Surface modification is required to improve their adsorption capacity on certain dyes |
| Excellent ability to remove a wide range of dyes and other industrial effluents | Adsorption efficiency depends on the type of material |
| Non-destructive process | Elimination of the adsorbates requires generation, replenishment or incineration of the adsorbent |
| Produce high quality treated effluents | Regeneration of the adsorbent is difficult and expensive |
| Advantages | Disadvantages |
|---|---|
| No pH adjustments | High chemical and energy consumption |
| High discoloration efficiency | Low performances for COD removal |
| Biodegradability improvement | UV radiation requirement for some AOPs |
| Toxicity reduction | Technology complexity |
| Free of sludge production | Inorganic sludge production |
| Catalyst (s) | Dye(s) | Degradation Efficiency, % | Time, min | Reusability, #of Cycles | Ref. |
|---|---|---|---|---|---|
| TiO2+MC composites | MB MV AV | 99 100 100 | 150 420 360 | 4 | [84] |
| CA-PU-ZnO membrane | RR 11 RO 84 | <95 <90 | 40 | - | [85] |
| Ba/Alg/CMC/TiO2 | CR | <90 | 240 | 4 | [86] |
| ZIF/cellulose aerogels | RhB | 99 | 10 | 5 | [87] |
| α-Fe2O3 nanodisk/bacterial cellulose membranes | RhB, MB, CV, MG, MO, OII | 100 | 5040 | - | [88] |
| CNC/MnO2/ALG | MB | 99.8 | 5 | 10 | [89] |
| Aerochitin–TiO2 composite | MB | 98 | 200 | - | [76] |
| Chitin-MnO2 | MB | 99 | 2.5 | 10 | [90] |
| ZSM-5 | Tartrazine dye | 95 | 30 | - | [91] |
| Chitin graphene hydrogel (CGH) supported ZnO–GO | MB | 99 | 150 | 5 | [92] |
| FeCS/MMTNS | MB | 100 | 120 | 5 | [93] |
| CS-ZnS-NPs | AB 98 AB 234 | 92.6 96.7 | 165 100 | 4 | [94] |
| CH/ZnO CH/Ce–ZnO | MG | 100 | 90 60 | 5 | [95] |
| FeNiSe-CHM | CR | 99 | 140 | 5 | [96] |
| TiO2@CS-Hpt | RhB MB | 96.7 98.6 | 120 | 7 | [97] |
| Membrane Process | Molecular Weight Cut-Off, kDa | Retained Diameters, μm | TMP Required, kPa | SystemRecovery |
|---|---|---|---|---|
| MF | 100–500 | 10−1–10 | 10–100 | 90–99+ |
| UF | 20–150 | 10−3–1 | 50–300 | 85–95+ |
| NF | 2–20 | 10−3–10−2 | 200–1500 | 75–90+ |
| RO | 0.2–2 | 10−4–10−3 | 500–8000 | 60–90 |
| Advantages | Disadvantages |
|---|---|
| Technologically simple | Economically non-viable due to high operational and maintenance costs |
| Rapid and highly efficient | High energy requirements |
| Eliminates all types of dyestuffs | Membrane fouling |
| No chemicals used | Limited flow rate and low throughput |
| Non-destructive separation technique | Sludge formation and high cleaning and membrane regeneration costs |
| A wide range of membranes available | Choice membrane system depends on the type of the dye |
| Membrane | Membrane Pressure, kPa | Dye(s) | Rejection Efficiency, % | Permeate Flux, L/h/m2 | Ref. |
|---|---|---|---|---|---|
| UF/TOCN/(ZIF-8) | 100–300 | JG B | 99 | 84 | [103] |
| BC-NFM | 500 | MO MB | 93.0 98.9 | 12.31 10.12 | [107] |
| NF cellulose hollow fibers | 20 | CR | 100 | 48 | [108] |
| NF- CNC-TFC-Ms | 60 | RB CR MO CV MB | 99 | - | [109] |
| PEUF/cellulose/chitosan (CHI) | 200 | MO DB71 | 86 89 | 37.85 25.72 | [110] |
| PET-CaA-AgNPs | 2.07 | CR | 95 | - | [111] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dassanayake, R.S.; Acharya, S.; Abidi, N. Recent Advances in Biopolymer-Based Dye Removal Technologies. Molecules 2021, 26, 4697. https://doi.org/10.3390/molecules26154697
Dassanayake RS, Acharya S, Abidi N. Recent Advances in Biopolymer-Based Dye Removal Technologies. Molecules. 2021; 26(15):4697. https://doi.org/10.3390/molecules26154697
Chicago/Turabian StyleDassanayake, Rohan S., Sanjit Acharya, and Noureddine Abidi. 2021. "Recent Advances in Biopolymer-Based Dye Removal Technologies" Molecules 26, no. 15: 4697. https://doi.org/10.3390/molecules26154697
APA StyleDassanayake, R. S., Acharya, S., & Abidi, N. (2021). Recent Advances in Biopolymer-Based Dye Removal Technologies. Molecules, 26(15), 4697. https://doi.org/10.3390/molecules26154697







