Sawdust for the Removal of Heavy Metals from Water: A Review
Abstract
:1. Introduction
2. Methods for Water Purification
3. Adsorbents for Heavy Metals Removal
3.1. Activated Carbon
3.2. Other Related Materials
4. Low-Cost Adsorbents
| Adsorbent | Heavy Metal | Adsorption Capacity (mg/g) | Reference |
|---|---|---|---|
| HCl-treated clay | Zn(II) | 63 | [87] |
| Cu(II) | 83 | [87] | |
| Ni(II) | 81 | [87] | |
| Coffee residues | Zn(II) | 13 | [88] |
| Cu(II) | 31 | [88] | |
| Ni(II) | 11 | [88] | |
| Orange peels | Ni(II) | 158 | [82] |
| Green sands | Zn(II) | 33 | [85] |
| Clarified sludge | Cr(VI) | 26 | [90] |
| Rice husk | Cr(VI) | 26 | [90] |
| Activated alumina | Cr(VI) | 25 | [90] |
| Fly ash | Cr(VI) | 24 | [90] |
| Neem bark | Cr(VI) | 20 | [90] |
| Straw | Cr(III) | 3 | [40] |
| Olive stones | Cd(II) | 49 | [91] |
| Carrot residues | Cr(III) | 45 | [92] |
| Cu(II) | 33 | [92] | |
| Zn(II) | 30 | [92] | |
| Orange wastes | Cd(II) | 48 | [93] |
| Banana peels | Pb(II) | 8 | [94] |
| Ni(II) | 7 | [94] | |
| Zn(II) | 6 | [94] | |
| Cu(II) | 5 | [94] | |
| Orange peels | Pb(II) | 8 | [94] |
| Ni(II) | 6 | [94] | |
| Zn(II) | 5 | [94] | |
| Cu(II) | 4 | [94] | |
| Crab carapace | Cu(II) | 79 | [89] |
| Macroalgae Fucus vesiculosus | Cu(II) | 115 | [89] |
| Peat | Cu(II) | 71 | [89] |
5. Sawdust
5.1. Sawdust Characteristics
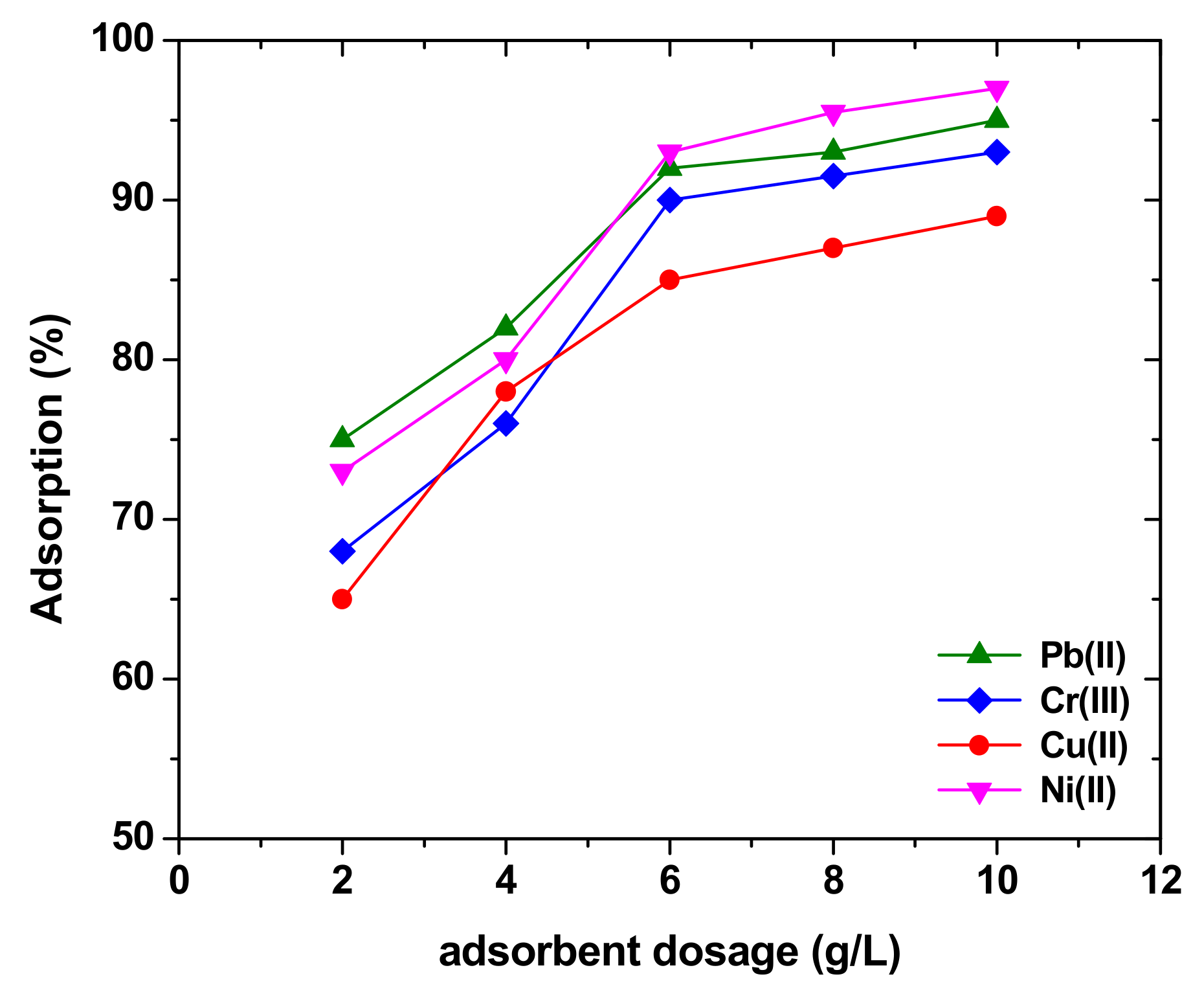
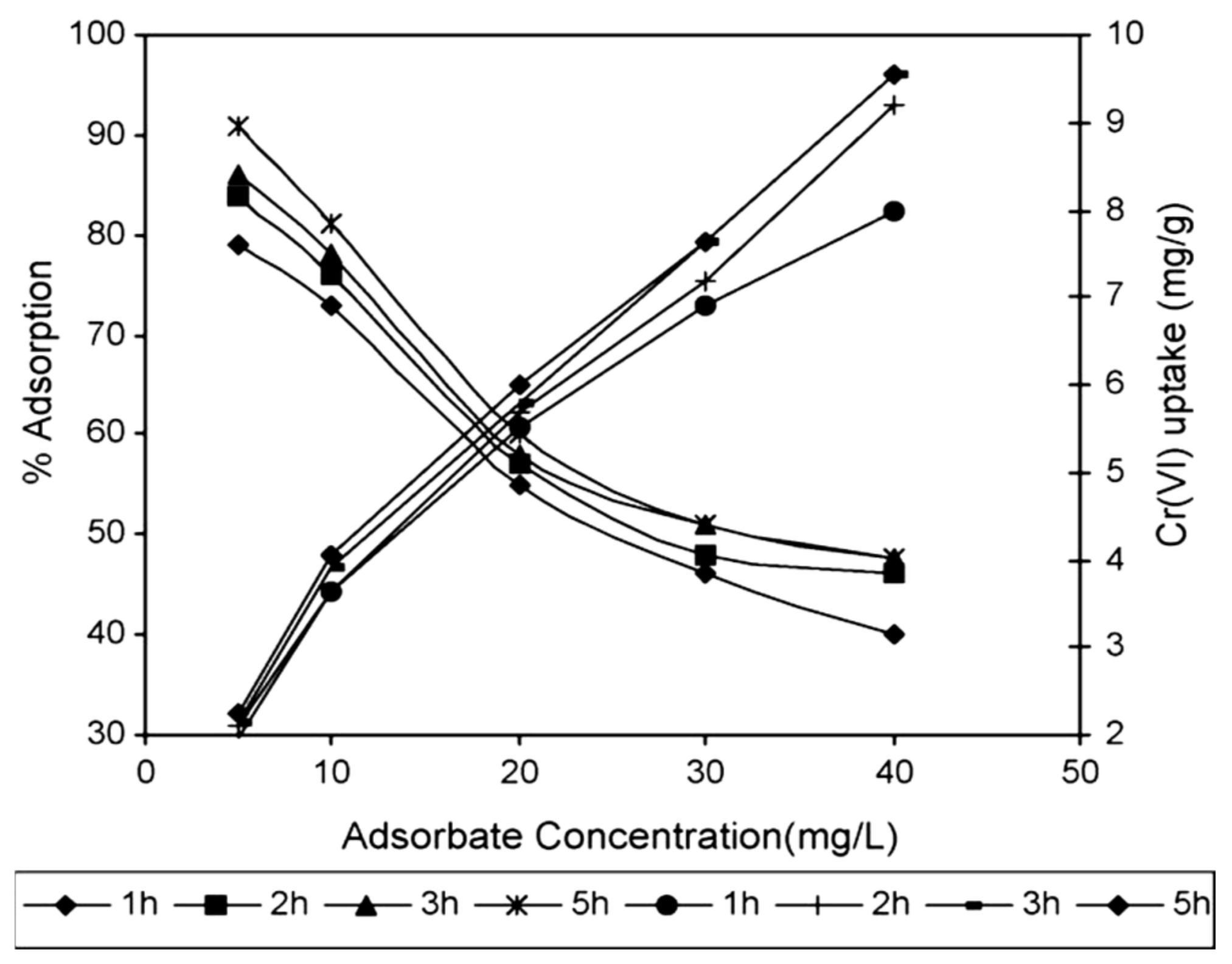
5.2. Parameters Affecting Adsorption onto Sawdust
5.3. Adsorption Isotherms
5.4. Adsorption Kinetics
5.5. Adsorption Thermodynamics
6. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Yang, L.; Tan, W.-F.; Mumford, K.; Ding, L.; Lv, J.-W.; Zhang, X.-W.; Wang, H.-Q. Effects of phosphorus-rich sawdust biochar sorption on heavy metals. Sep. Sci. Technol. 2018, 53, 2704–2716. [Google Scholar] [CrossRef]
- Lee, J.; Rai, P.K.; Jeon, Y.J.; Kim, K.-H.; Kwon, E.E. The role of algae and cyanobacteria in the production and release of odorants in water. Environ. Pollut. 2017, 227, 252–262. [Google Scholar] [CrossRef]
- Shukla, S.; Pai, R.S. Adsorption of Cu(II), Ni(II) and Zn(II) on dye loaded groundnut shells and sawdust. Sep. Purif. Technol. 2005, 43, 1–8. [Google Scholar] [CrossRef]
- Shukla, A.; Zhang, Y.-H.; Dubey, P.; Margrave, J.; Shukla, S.S. The role of sawdust in the removal of unwanted materials from water. J. Hazard. Mater. 2002, 95, 137–152. [Google Scholar] [CrossRef]
- McCann, M. Hazards in cottage industries in developing countries. Am. J. Ind. Med. 1996, 30, 125–129. [Google Scholar] [CrossRef]
- Acar, F.N.; Malkoc, E. The removal of chromium(VI) from aqueous solutions by Fagus orientalis L. Bioresour. Technol. 2004, 94, 13–15. [Google Scholar] [CrossRef] [PubMed]
- Mone, M.; Lambropoulou, D.A.; Bikiaris, D.N.; Kyzas, G.Z. Chitosan grafted with biobased 5-hydroxymethyl-furfural as adsorbent for copper and cadmium ions removal. Polymers 2020, 12, 1173. [Google Scholar] [CrossRef]
- Larous, S.; Meniai, A.-H.; Lehocine, M.B. Experimental study of the removal of copper from aqueous solutions by adsorption using sawdust. Desalination 2005, 185, 483–490. [Google Scholar] [CrossRef]
- Argun, M.E.; Dursun, S.; Ozdemir, C.; Karatas, M. Heavy metal adsorption by modified oak sawdust: Thermodynamics and kinetics. J. Hazard. Mater. 2007, 141, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Memon, S.Q.; Memon, N.; Shah, S.; Khuhawar, M.; Bhanger, M. Sawdust—A green and economical sorbent for the removal of cadmium (II) ions. J. Hazard. Mater. 2007, 139, 116–121. [Google Scholar] [CrossRef]
- Jianfui, C.; Sarjadi, M.S.; Musta, B.; Sarkar, M.S.; Rahman, M.L. Synthesis of Sawdust-based Poly(amidoxime) Ligand for Heavy Metals Removal from Wastewater. ChemistrySelect 2019, 4, 2991–3001. [Google Scholar] [CrossRef]
- Ikem, A.; Adisa, S. Runoff effect on eutrophic lake water quality and heavy metal distribution in recent littoral sediment. Chemosphere 2011, 82, 259–267. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Chen, J.; Ru, X.; Chen, J.; Wu, H.; Wei, C. Heavy metals in river surface sediments affected with multiple pollution sources, South China: Distribution, enrichment and source apportionment. J. Geochem. Explor. 2017, 176, 9–19. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, J.; Xu, Y.; Su, H.; Li, X.; Zhou, J.Z.; Qian, G.; Li, L.; Xu, Z.P. Efficient Selective Catalytic Reduction of NO by Novel Carbon-doped Metal Catalysts Made from Electro-plating Sludge. Environ. Sci. Technol. 2014, 48, 11497–11503. [Google Scholar] [CrossRef]
- Kejellstrom, T.; Shiroishi, K.; Evrin, P.E. Urinary beta/sub 2/-microglobulin excretion among people exposed to cadmium in the general environment. Environ. Res. 1977, 13, 318–344. [Google Scholar] [CrossRef]
- Friberg, C.G.E. Encyclopedia of Occupational Health; International Labor Organization: Geneva, Swizterland, 1985. [Google Scholar]
- Irukayama, K. The pollution of Minamata Bay and Minamata disease. Adv. Water Pollut. Res. 1966, 2, 153–165. [Google Scholar]
- Fu, J.; Zhao, C.; Luo, Y.; Liu, C.; Kyzas, G.Z.; Luo, Y.; Zhao, D.; An, S.; Zhu, H. Heavy metals in surface sediments of the Jialu River, China: Their relations to environmental factors. J. Hazard. Mater. 2014, 270, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Bortoleto, G.G.; Macarovscha, G.T.; Cadore, S. Determination of cadmium by flame-atomic absorption spectrometry after preconcentration on silica gel modified with cupferron. J. Braz. Chem. Soc. 2004, 15, 313–317. [Google Scholar] [CrossRef]
- Galvin, R.M. Occurrence of metals in waters: An overview. Water SA 1996, 22, 7–18. [Google Scholar]
- Selvaraj, K.; Manonmani, S.; Pattabhi, S. Removal of hexavalent chromium using distillery sludge. Bioresour. Technol. 2003, 89, 207–211. [Google Scholar] [CrossRef]
- Kurniawan, T.A.; Chan, G.Y.S.; Lo, W.-H.; Babel, S. Comparisons of low-cost adsorbents for treating wastewaters laden with heavy metals. Sci. Total. Environ. 2006, 366, 409–426. [Google Scholar] [CrossRef] [PubMed]
- Esalah, J.O.; Weber, M.E.; Vera, J.H. Removal of lead, cadmium and zinc from aqueous solutions by precipitation with sodium Di-(n-octyl) phosphinate. Can. J. Chem. Eng. 2000, 78, 948–954. [Google Scholar] [CrossRef]
- Zouboulis, A.; Matis, K.A.; Lanara, B.G.; Loos-Neskovic, C. Removal of Cadmium from Dilute Solutions by Hydroxyapatite. II. Flotation Studies. Sep. Sci. Technol. 1997, 32, 1755–1767. [Google Scholar] [CrossRef]
- Ho, Y.S.; Ng, J.C.; McKay, G. Removal of lead(II) from effluents by sorption on peat using second-order kinetics. Sep. Sci. Technol. 2001, 36, 241–261. [Google Scholar] [CrossRef]
- Hall, C.; Wales, D.S.; Keane, M.A. Copper removal from aqueous systems: Biosorption by pseudo-monas syringae. Sep. Sci. Technol. 2001, 36, 223–240. [Google Scholar] [CrossRef]
- Sağ, Y.; Akçael, B.; Kutsal, T. Ternary biosorption equilibria of chromium(VI), copper(II), and cadmium(II) onRhizopus arrhizus. Sep. Sci. Technol. 2002, 37, 279–309. [Google Scholar] [CrossRef]
- Canet, L.; Ilpide, M.; Seta, P. Efficient facilitated transport of lead, cadmium, zinc, and silver across a flat-sheet-supported liquid membrane mediated by lasalocid A. Sep. Sci. Technol. 2002, 37, 1851–1860. [Google Scholar] [CrossRef]
- Ahmad, S.; Khalid, N.; Daud, M. Adsorption studies of lead on lateritic minerals from aqueous media. Sep. Sci. Technol. 2002, 37, 343–362. [Google Scholar] [CrossRef]
- Buerge-Weirich, D.; Hari, R.; Xue, H.; Behra, P.; Sigg, L. Adsorption of Cu, Cd, and Ni on Goethite in the Presence of Natural Groundwater Ligands. Environ. Sci. Technol. 2002, 36, 328–336. [Google Scholar] [CrossRef]
- Ravindran, V.; Stevens, M.R.; Badriyha, B.N.; Pirbazari, M. Modeling the sorption of toxic metals on chelant-impregnated adsorbent. AIChE J. 1999, 45, 1135–1146. [Google Scholar] [CrossRef]
- Toles, C.A.; Marshall, W.E. Copper ion removal by almond shell carbons and commercial carbons: Batch and column studies. Sep. Sci. Technol. 2002, 37, 2369–2383. [Google Scholar] [CrossRef]
- Suzuki, M. Role of adsorption in water environment processes. Water Sci. Technol. 1997, 35, 1–11. [Google Scholar] [CrossRef]
- Sekhar, K.; Subramanian, S.; Modak, J.; Natarajan, K. Removal of metal ions using an industrial biomass with reference to environmental control. Int. J. Miner. Process. 1998, 53, 107–120. [Google Scholar] [CrossRef]
- Li, J.; Wang, X.; Zhao, G.; Chen, C.; Chai, Z.; Alsaedi, A.; Hayat, T.; Wang, X. Metal–organic framework-based materials: Superior adsorbents for the capture of toxic and radioactive metal ions. Chem. Soc. Rev. 2018, 47, 2322–2356. [Google Scholar] [CrossRef] [PubMed]
- Rich, G.; Cherry, K.J.C. New York, Hazardons Waste Treatment Technology; Pudvan Publ.: Northbrook, IL, USA, 1987; Volume 20. [Google Scholar]
- Ayyappan, R.; Sophia, C.; Swaminathan, K.; Sandhya, S. Removal of Pb(II) from aqueous solution using carbon derived from agricultural wastes. Process. Biochem. 2005, 40, 1293–1299. [Google Scholar] [CrossRef]
- Shukla, S.S.; Yu, L.J.; Dorris, K.L.; Shukla, A. Removal of nickel from aqueous solutions by sawdust. J. Hazard. Mater. 2005, 121, 243–246. [Google Scholar] [CrossRef]
- Li, Q.; Zhai, J.; Zhang, W.; Wang, M.; Zhou, J. Kinetic studies of adsorption of Pb(II), Cr(III) and Cu(II) from aqueous solution by sawdust and modified peanut husk. J. Hazard. Mater. 2007, 141, 163–167. [Google Scholar] [CrossRef]
- Kumar, A.; Rao, N.; Kaul, S. Alkali-treated straw and insoluble straw xanthate as low cost adsorbents for heavy metal removal–preparation, characterization and application. Bioresour. Technol. 2000, 71, 133–142. [Google Scholar] [CrossRef]
- Rafatullah, M.; Sulaiman, O.; Hashim, R.; Ahmad, A. Adsorption of copper (II), chromium (III), nickel (II) and lead (II) ions from aqueous solutions by meran-ti sawdust. J. Hazard. Mater. 2009, 170, 969–977. [Google Scholar] [CrossRef] [PubMed]
- Bulut, Y.; Tez, Z. Removal of heavy metals from aqueous solution by sawdust adsorption. J. Environ. Sci. 2007, 19, 160–166. [Google Scholar] [CrossRef]
- Sadeek, S.A.; Negm, N.; Hefni, H.; Wahab, M.M.A. Metal adsorption by agricultural biosorbents: Adsorption isotherm, kinetic and biosorbents chemical structures. Int. J. Biol. Macromol. 2015, 81, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Monser, L.; Adhoum, N. Modified activated carbon for the removal of copper, zinc, chromium and cyanide from wastewater. Sep. Purif. Technol. 2002, 26, 137–146. [Google Scholar] [CrossRef]
- Leyva-Ramos, R. Adsorption of zinc(II) from an aqueous solution onto activated carbon. J. Hazard. Mater. 2002, 90, 27–38. [Google Scholar] [CrossRef]
- Leyva-Ramos, R.; Rangel-Mendez, J.R.; Mendoza-Barron, J.; Fuentes-Rubio, L.; Guerrero-Coronado, R.M. Adsorption of cadmium (II) from aqueous solution onto activated carbon. Water Sci. Technol. 1997, 35, 205–211. [Google Scholar] [CrossRef]
- Shim, J.W.; Lee, S.M.; Rhee, B.S.; Ryu, S.K. Adsorption of Ni (II), Cu (II), Cr (VI) from Multicomponent Aqueous Solution by Pitch-Based ACF; InExtended abstracts; British Carbon Group: Newcastle, UK, 1996. [Google Scholar]
- Yu, B.; Zhang, Y.; Shukla, A.; Shukla, S.S.; Dorris, K.L. The removal of heavy metal from aqueous solutions by sawdust adsorption—Removal of copper. J. Hazard. Mater. 2000, 80, 33–42. [Google Scholar] [CrossRef]
- Ouki, S.K.; Neufeld, R.D. ChemInform Abstract: Use of Activated Carbon for the Recovery of Chromium from Industrial Wastewaters. Chemistry 2010, 28, 3–8. [Google Scholar] [CrossRef]
- Raji, C.; Anirudhan, T.S. Kinetics of Pb (II) Adsorption by Polyacrylamide Grafted Sawdust. Indian J. Chem. Technol. 1997, 4, 157–162. [Google Scholar]
- Summers, B.L.; Gress, L.B.; Philipp, W.H.; Eastep, S.B. Peat-moss pellets for removal of metalion contaminants dissolved in dilute wastewater. US, 11 pp., Cont. Part US Ser. 1995, 733. [Google Scholar]
- Ferrero, F.; Prati, M.P. Coal fly ash and alginate for the removal of heavy metals from aqueous solutions. Ann. Chim. 1996, 86, 125–132. [Google Scholar]
- Mathur, A.; Khare, S.K.; Rupainwar, D.C. Removal of heavy metals from main sewer-water of Varanasi city by ad-sorption on fly ash and blast furnace slag. J. Ind. Pollut. Control 1989, 5, 52–57. [Google Scholar]
- Saravanane, R.; Sundararajan, T.; Reddy, S.S. Efficiency of chemically modified low cost adsorbents for the removal of heavy metals from waste water: A comparative study. Indian J. Environ. Health 2002, 44, 78–87. [Google Scholar]
- Lee, C.-H. Proceedings of the Third Pacific Basin Conference on Adsorption Science and Technology, Kyongju, Korea, 25–29 May 2003.
- Ouki, S.K.; Kavannagh, M. Treatment of metals-contaminated wastewaters by use of natural zeolites. Water Sci. Technol. 1999, 39, 115–122. [Google Scholar] [CrossRef]
- Rorrer, G.L.; Hsien, T.Y.; Way, J.D. Synthesis of porous-magnetic chitosan beads for removal of cadmium ions from wastewater. Ind. Eng. Chem. Res. 1993, 32, 2170–2178. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Singh, A.K.; Sharma, A. Studies on the uptake of lead and zinc by lignin obtained from black liquor–A paper industry waste material. Environ. Technol. 1994, 15, 353–361. [Google Scholar] [CrossRef]
- Tuin, B.; Tels, M. Removing heavy metals from contaminated clay soils by extraction with hydrochloric acid, edta or hypochlorite solutions. Environ. Technol. 1990, 11, 1039–1052. [Google Scholar] [CrossRef]
- Zhao, Y.; Hao, Y.; Ramelow, G.J. Evaluation of treatment techniques for increasing the uptake of metal ions from solution by nonliving sea-weed algal biomass. Environ. Monit. Assess. 1994, 33, 61–70. [Google Scholar] [CrossRef]
- Hanway, J.E.; Mumford, R.G.; Mishra, P.N. Treatment of industrial effluents for heavy metals removal using the cellulose xan-thate process. InAIChE Symp. Ser. 1979, 75, 306–614. [Google Scholar]
- Dávila-Jiménez, M.; Elizalde-González, M.P.; Geyer, W.; Mattusch, J.; Wennrich, R.; Dávila-Jiménez, M.; Elizalde-González, M.P.; Geyer, W.; Mattusch, J.; Wennrich, R. Adsorption of metal cations from aqueous solution onto a natural and a model biocomposite. Colloids Surfaces A Physicochem. Eng. Asp. 2003, 219, 243–252. [Google Scholar] [CrossRef]
- Klimmek, S.; Stan, H.-J.; Wilke, A.; Bunke, G.; Buchholz, R. Comparative Analysis of the Biosorption of Cadmium, Lead, Nickel, and Zinc by Algae. Environ. Sci. Technol. 2001, 35, 4283–4288. [Google Scholar] [CrossRef]
- Wang, J.; Teng, X.; Wang, H.; Ban, H. Characterizing the Metal Adsorption Capability of a Class F Coal Fly Ash. Environ. Sci. Technol. 2004, 38, 6710–6715. [Google Scholar] [CrossRef] [PubMed]
- Cheung, C.W.; Chan, C.K.; Porter, J.F.; McKay, G. Combined diffusion model for the sorption of cadmium, copper, and zinc ions onto bone char. Environ. Sci. Technol. 2001, 35, 1511–1522. [Google Scholar] [CrossRef] [PubMed]
- Kumar, U.; Bandyopadhyay, M. Sorption of cadmium from aqueous solution using pretreated rice husk. Bioresour. Technol. 2006, 97, 104–109. [Google Scholar] [CrossRef] [PubMed]
- Göksungur, Y.; Üren, S.; Güvenç, U. Biosorption of cadmium and lead ions by ethanol treated waste baker’s yeast biomass. Bioresour. Technol. 2005, 96, 103–109. [Google Scholar] [CrossRef]
- Romero-González, M.E.; Williams, C.J.; Gardiner, P.H.E.; Romero-González, M.E.; Williams, C.J.; Gardiner, P.H.E. Study of the mechanisms of cadmium biosorption by dealginated seaweed waste. Environ. Sci. Technol. 2001, 35, 3025–3030. [Google Scholar] [CrossRef] [PubMed]
- Taty-Costodes, V.; Fauduet, H.; Porte, C.; Delacroix, A. Removal of Cd(II) and Pb(II) ions, from aqueous solutions, by adsorption onto sawdust of Pinus sylvestris. J. Hazard. Mater. 2003, 105, 121–142. [Google Scholar] [CrossRef]
- Cullen, G.V.; Siviour, N.G. Removing metals from waste solutions with low rank coals and related materials. Water Res. 1982, 16, 1357–1366. [Google Scholar] [CrossRef]
- Singh, K.K.; Rastogi, R.; Hasan, S.H. Removal of cadmium from wastewater using agricultural waste ‘rice polish’. J. Hazard. Mater. 2005, 121, 51–58. [Google Scholar] [CrossRef]
- Low, K.; Lee, C.; Liew, S. Sorption of cadmium and lead from aqueous solutions by spent grain. Process. Biochem. 2000, 36, 59–64. [Google Scholar] [CrossRef]
- Lodeiro, P.; Cordero, B.; Barriada, J.L.; Herrero, R.; de Vicente, M.S. Biosorption of cadmium by biomass of brown marine macroalgae. Bioresour. Technol. 2005, 96, 1796–1803. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sen, A.K.; De, A.K. Adsorption of mercury(II) by coal fly ash. Water Res. 1987, 21, 885–888. [Google Scholar] [CrossRef]
- Pandey, M.P.; Chaudhuri, M. Removal of inorganic mercury from water by bituminous coal. Water Res. 1982, 16, 1113–1118. [Google Scholar] [CrossRef]
- Tiwari, D.P.; Singh, D.K.; Saksena, D.N. Hg (II) adsorption from aqueous solutions using rice-husk ash. J. Environ. Eng. 1995, 121, 479–481. [Google Scholar] [CrossRef]
- Atanassova, I.D. Adsorption and desorption of Cu at high equilibrium concentrations by soil and clay samples from Bulgaria. Environ. Pollut. 1995, 87, 17–21. [Google Scholar] [CrossRef]
- Aksu, Z.; Açıkel, Ü.; Kabasakal, E.; Tezer, S. Equilibrium modelling of individual and simultaneous biosorption of chromium(VI) and nickel(II) onto dried activated sludge. Water Res. 2002, 36, 3063–3073. [Google Scholar] [CrossRef]
- Toprak, R.; Girgin, I. Removal of chromium from leather industry waste water by activated clinoptilolite. Turk. J. Eng. Environ. Sci. 2000, 24, 343–351. [Google Scholar]
- Sharma, D.; Forster, C. Removal of hexavalent chromium using sphagnum moss peat. Water Res. 1993, 27, 1201–1208. [Google Scholar] [CrossRef]
- Panday, K.K.; Prasad, G.; Singh, V.N. Removal of Cr(V1) from aqueous solutions by adsorption on fly ash-wollastonite. J. Chem. Technol. Biotechnol. Chem. Technol. 2007, 34, 367–374. [Google Scholar] [CrossRef]
- Ajmal, M.; Rao, R.A.; Ahmad, R.; Ahmad, J. Adsorption studies on Citrus reticulata (fruit peel of orange): Removal and recovery of Ni (II) from electro-plating wastewater. J. Hazard. Mater. 2000, 79, 117–131. [Google Scholar] [CrossRef]
- Babel, S.; Kurniawan, T.A. Low-cost adsorbents for heavy metals uptake from contaminated water: A review. J. Hazard. Mater. 2003, 97, 219–243. [Google Scholar] [CrossRef]
- Bailey, S.E.; Olin, T.J.; Bricka, R.M.; Adrian, D.D. A review of potentially low-cost sorbents for heavy metals. Water Res. 1999, 33, 2469–2479. [Google Scholar] [CrossRef]
- Lee, T.; Park, J.-W.; Lee, J.-H. Waste green sands as reactive media for the removal of zinc from water. Chemosphere 2004, 56, 571–581. [Google Scholar] [CrossRef]
- Bayat, B. Comparative study of adsorption properties of Turkish fly ashes: I. The case of nickel (II), copper (II) and zinc (II). J. Hazard. Mater. 2002, 95, 251–273. [Google Scholar] [CrossRef]
- Vengris, T.; Binkien, R.; Sveikauskait, A. Nickel, copper and zinc removal from waste water by a modified clay sorbent. Appl. Clay Sci. 2001, 18, 183–190. [Google Scholar] [CrossRef]
- Boonamnuayvitaya, V.; Chaiya, C.; Tanthapanichakoon, W.; Jarudilokkul, S. Removal of heavy metals by adsorbent prepared from pyrolyzed coffee residues and clay. Sep. Purif. Technol. 2004, 35, 11–22. [Google Scholar] [CrossRef]
- Cochrane, E.; Lu, S.; Gibb, S.; Villaescusa, I. A comparison of low-cost biosorbents and commercial sorbents for the removal of copper from aqueous media. J. Hazard. Mater. 2006, 137, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, A.K.; Naiya, T.K.; Mandal, S.N.; Das, S.K. Adsorption, kinetics and equilibrium studies on removal of Cr(VI) from aqueous solutions using different low-cost adsorbents. Chem. Eng. J. 2008, 137, 529–541. [Google Scholar] [CrossRef]
- Aziz, A.; Ouali, M.S.; Elandaloussi, E.H.; De Menorval, L.C.; Lindheimer, M. Chemically modified olive stone: A low-cost sorbent for heavy metals and basic dyes removal from aqueous solutions. J. Hazard. Mater. 2009, 163, 441–447. [Google Scholar] [CrossRef]
- Nasernejad, B.; Zadeh, T.E.; Pour, B.B.; Bygi, M.E.; Zamani, A. Camparison for biosorption modeling of heavy metals (Cr (III), Cu (II), Zn (II)) adsorption from wastewater by carrot residues. Process. Biochem. 2005, 40, 1319–1322. [Google Scholar] [CrossRef]
- Marín, A.B.P.; Zapata, V.M.; Ortuño, J.F.; Aguilar, M.I.; Sáez, J.; Llorens, M. Removal of cadmium from aqueous solutions by adsorption onto orange waste. J. Hazard. Mater. 2007, 139, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Annadurai, G.; Juang, R.-S.; Lee, D. Adsorption of heavy metals from water using banana and orange peels. Water Sci. Technol. 2003, 47, 185–190. [Google Scholar] [CrossRef]
- Chikri, R.; Elhadiri, N.; Benchanaa, M.; El Maguana, Y. Efficiency of Sawdust as Low-Cost Adsorbent for Dyes Removal. J. Chem. 2020, 2020, 1–17. [Google Scholar] [CrossRef]
- Ikenyiri, P.; Ukpaka, C. Overview on the Effect of Particle Size on the Performance of Wood Based Adsorbent. J. Chem. Eng. Process. Technol. 2016, 7, 1000315. [Google Scholar]
- Semerjian, L. Removal of heavy metals (Cu, Pb) from aqueous solutions using pine (Pinus halepensis) sawdust: Equilibrium, kinetic, and thermodynamic studies. Environ. Technol. Innov. 2018, 12, 91–103. [Google Scholar] [CrossRef]
- Baral, S.S.; Das, S.N.; Rath, P. Hexavalent chromium removal from aqueous solution by adsorption on treated sawdust. Biochem. Eng. J. 2006, 31, 216–222. [Google Scholar] [CrossRef]
- Yu, L.J.; Shukla, S.S.; Dorris, K.L.; Shukla, A.; Margrave, J. Adsorption of chromium from aqueous solutions by maple sawdust. J. Hazard. Mater. 2003, 100, 53–63. [Google Scholar] [CrossRef]
- Jeon, C.; Kim, J.H. Removal of lead ions using phosphorylated sawdust. J. Ind. Eng. Chem. 2009, 15, 910–913. [Google Scholar] [CrossRef]
- Zhang, R.; Zhou, Y.; Gu, X.; Lu, J. Competitive Adsorption of Methylene Blue and Cu2+ onto Citric Acid Modified Pine Sawdust. CLEAN–Soil Air Water 2015, 43, 96–103. [Google Scholar] [CrossRef]
- Djeribi, R.; Hamdaoui, O. Sorption of copper(II) from aqueous solutions by cedar sawdust and crushed brick. Desalination 2008, 225, 95–112. [Google Scholar] [CrossRef]
- Pereira, F.V.; Gurgel, L.; Gil, L.F. Removal of Zn2+ from aqueous single metal solutions and electroplating wastewater with wood sawdust and sugarcane bagasse modified with EDTA dianhydride (EDTAD). J. Hazard. Mater. 2010, 176, 856–863. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Božić, D.; Gorgievski, M.; Stanković, V.; Štrbac, N.; Šerbula, S.; Petrović, N. Adsorption of heavy metal ions by beech sawdust–Kinetics, mechanism and equilibrium of the process. Ecol. Eng. 2013, 58, 202–206. [Google Scholar] [CrossRef]
- Dhir, B.; Srivastava, S. Heavy metal removal from a multi-metal solution and wastewater by Salvinia natans. Ecol. Eng. 2011, 37, 893–896. [Google Scholar] [CrossRef]
- Gupta, S.; Babu, B. Removal of toxic metal Cr(VI) from aqueous solutions using sawdust as adsorbent: Equilibrium, kinetics and regeneration studies. Chem. Eng. J. 2009, 150, 352–365. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Nanaki, S.G.; Koltsakidou, A.; Papageorgiou, M.; Kechagia, M.; Bikiaris, D.N.; Lambropoulou, D.A. Effectively designed molecularly imprinted polymers for selective isolation of the antidiabetic drug metformin and its transformation product guanylurea from aqueous media. Anal. Chim. Acta 2015, 866, 27–40. [Google Scholar] [CrossRef]
- Kaczala, F.; Marques, M.; Hogland, W. Lead and vanadium removal from a real industrial wastewater by gravitational settling/sedimentation and sorption onto Pinus sylvestris sawdust. Bioresour. Technol. 2009, 100, 235–243. [Google Scholar] [CrossRef] [PubMed]
- Vadivelan, V.; Kumar, K.V. Equilibrium, kinetics, mechanism, and process design for the sorption of methylene blue onto rice husk. J. Colloid Interface Sci. 2005, 286, 90–100. [Google Scholar] [CrossRef] [PubMed]
- Al-Asheh, S.; Banat, F.; Al-Omari, R.; Duvnjak, Z. Predictions of binary sorption isotherms for the sorption of heavy metals by pine bark using single iso-therm data. Chemosphere 2000, 41, 659–665. [Google Scholar] [CrossRef]
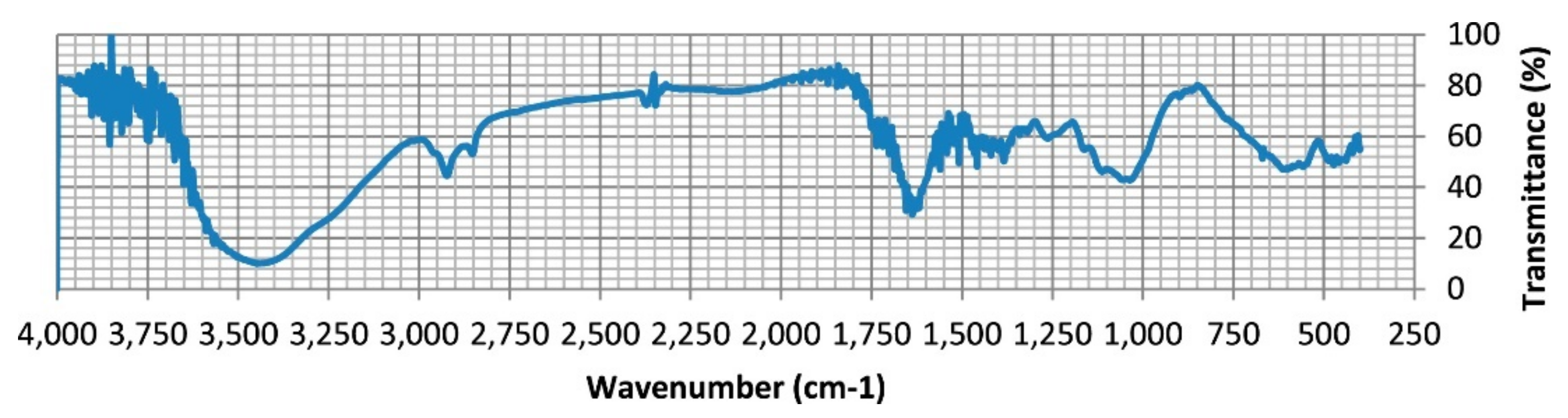
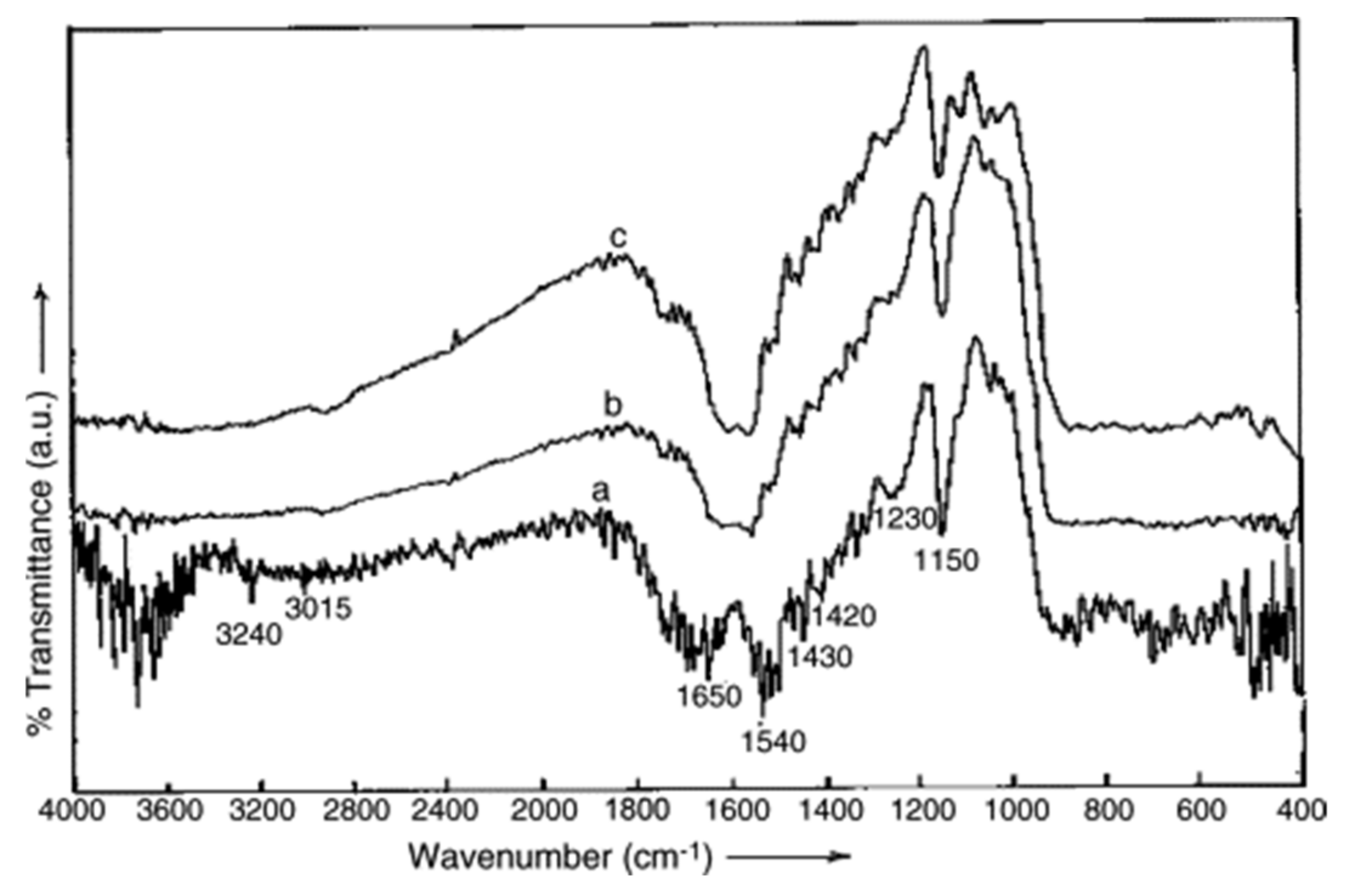
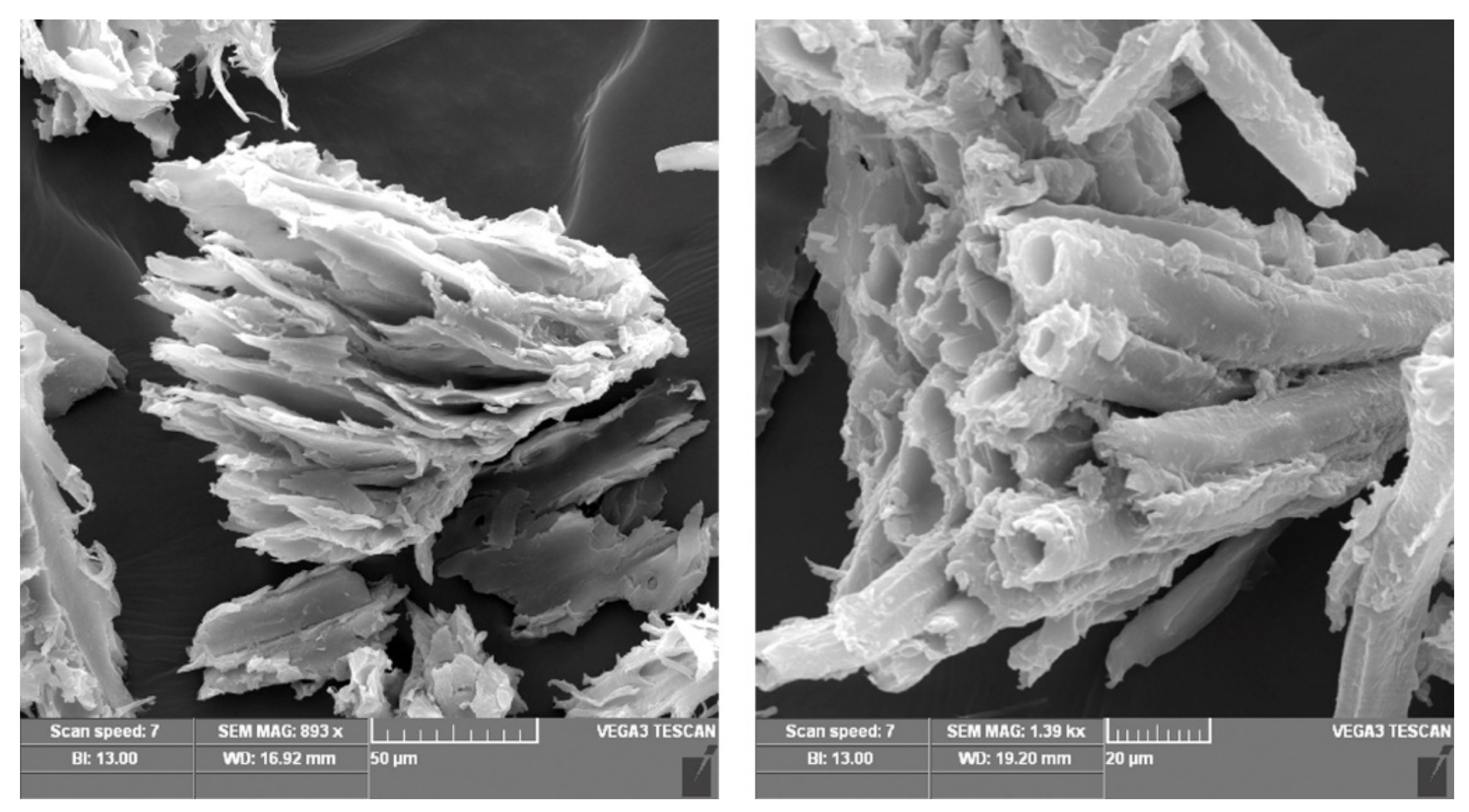


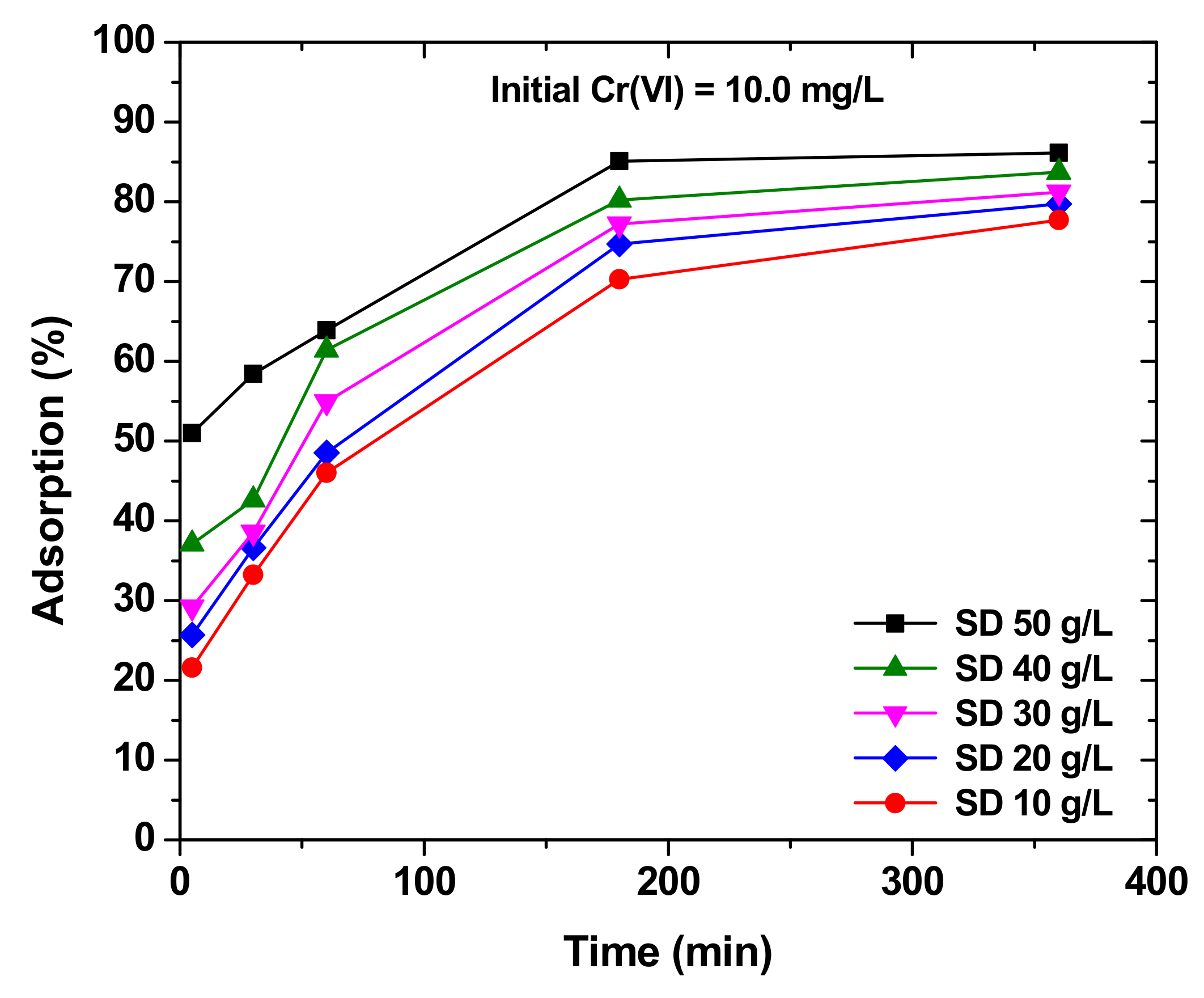

| Sawdust | Elemental Compositions (%) | |||
|---|---|---|---|---|
| C | H | O | N | |
| Pine sawdust | 46.41 | 6.27 | 47.23 | 0.06 |
| Meranti sawdust | 42.38 | 5.27 | 42.41 | 0.14 |
| Chinese fir sawdust | 48.95 | 6.54 | 53.74 | 0.11 |
| Rubber-wood sawdust | 44.01 | 8.04 | 47.50 | 0.45 |
| Sawdust (Canada) | 45.20 | 6.70 | 48.00 | 0.10 |
| Sawdust (Colombia) | 48.50 | 6.60 | 44.90 | 0.00 |
| Red wood sawdust | 27.1 | Not given | 72.5 | Not given |
| Parkia biglobosa sawdust | 57.62 | Not given | 35.93 | Not given |
| Element | Percentage (%) |
|---|---|
| Ash | 1.0–1.8 |
| Crude protein | 0.9–1.2 |
| Acid detergent fiber | 26.4–34.4 |
| Crude fiber | 62.6–68.4 |
| Dry matter | 95.0–99.1 |
| Freundlich | Langmuir | ||||||
|---|---|---|---|---|---|---|---|
| Metal | Kf | nF | R2 | qm | b | R2 | RL |
| Cu | 1.59 | 0.83 | 0.992 | 9.59 | 0.23 | 0.553 | 0.08–0.813 |
| Pb | 4.1 | 0.64 | 0.722 | 13.48 | 0.52 | 0.492 | 0.037–0.658 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meez, E.; Rahdar, A.; Kyzas, G.Z. Sawdust for the Removal of Heavy Metals from Water: A Review. Molecules 2021, 26, 4318. https://doi.org/10.3390/molecules26144318
Meez E, Rahdar A, Kyzas GZ. Sawdust for the Removal of Heavy Metals from Water: A Review. Molecules. 2021; 26(14):4318. https://doi.org/10.3390/molecules26144318
Chicago/Turabian StyleMeez, Elie, Abbas Rahdar, and George Z. Kyzas. 2021. "Sawdust for the Removal of Heavy Metals from Water: A Review" Molecules 26, no. 14: 4318. https://doi.org/10.3390/molecules26144318
APA StyleMeez, E., Rahdar, A., & Kyzas, G. Z. (2021). Sawdust for the Removal of Heavy Metals from Water: A Review. Molecules, 26(14), 4318. https://doi.org/10.3390/molecules26144318








