Bioaccessibility and Cellular Uptake of Carotenoids Extracted from Bactris gasipaes Fruit: Differences between Conventional and Ionic Liquid-Mediated Extraction
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Chemicals
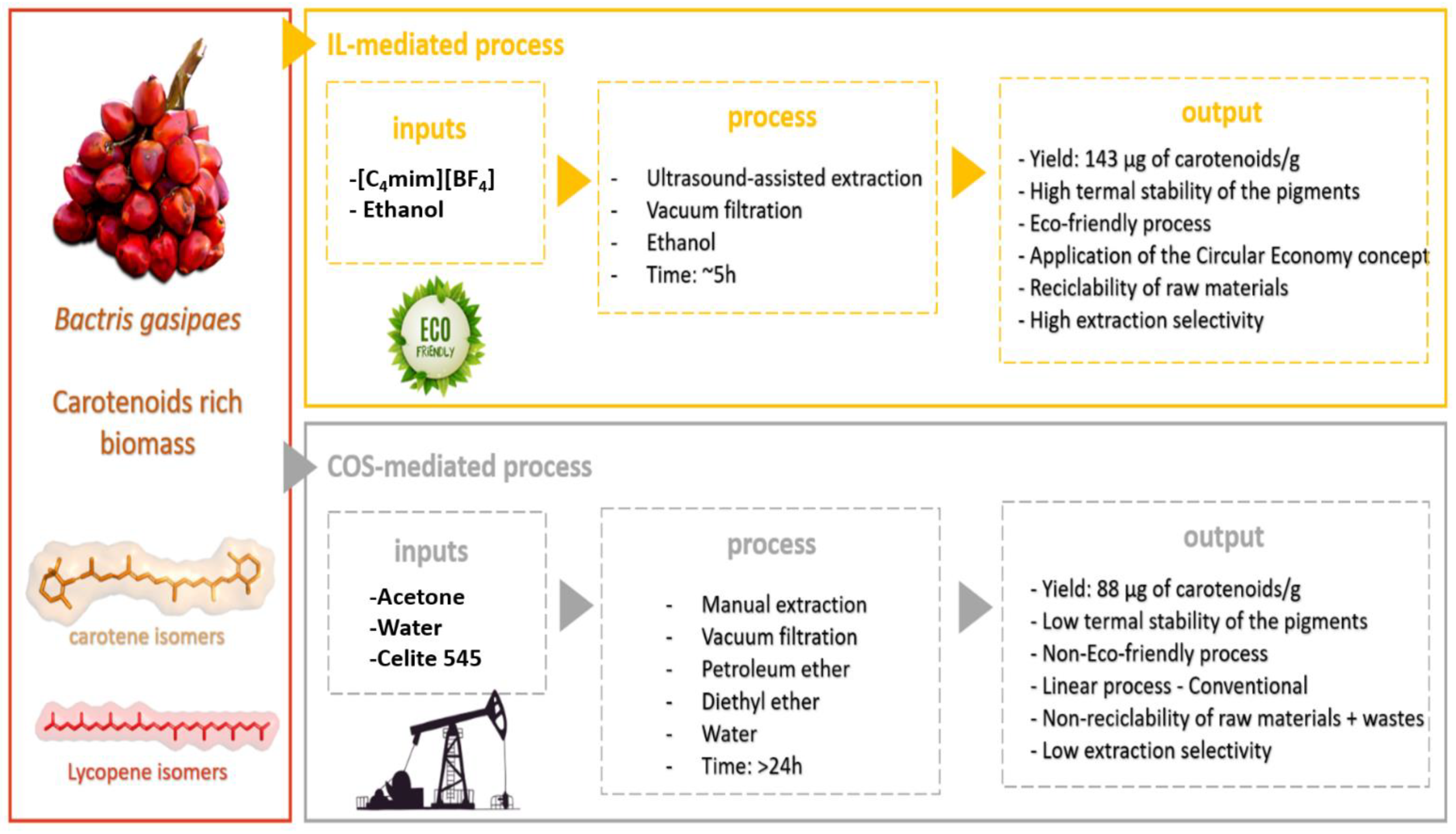
2.3. Solid-Liquid Extraction
2.3.1. Conventional Extraction—Acetone
2.3.2. Alternative Extraction—Ionic Liquids
2.3.3. Quantification of [C4mim][BF4] in Carotenoid Extract
2.4. Bioaccessibility (BCT) and Cellular Uptake Assay
2.4.1. Sample Preparation: Emulsified Carotenoid Extracts
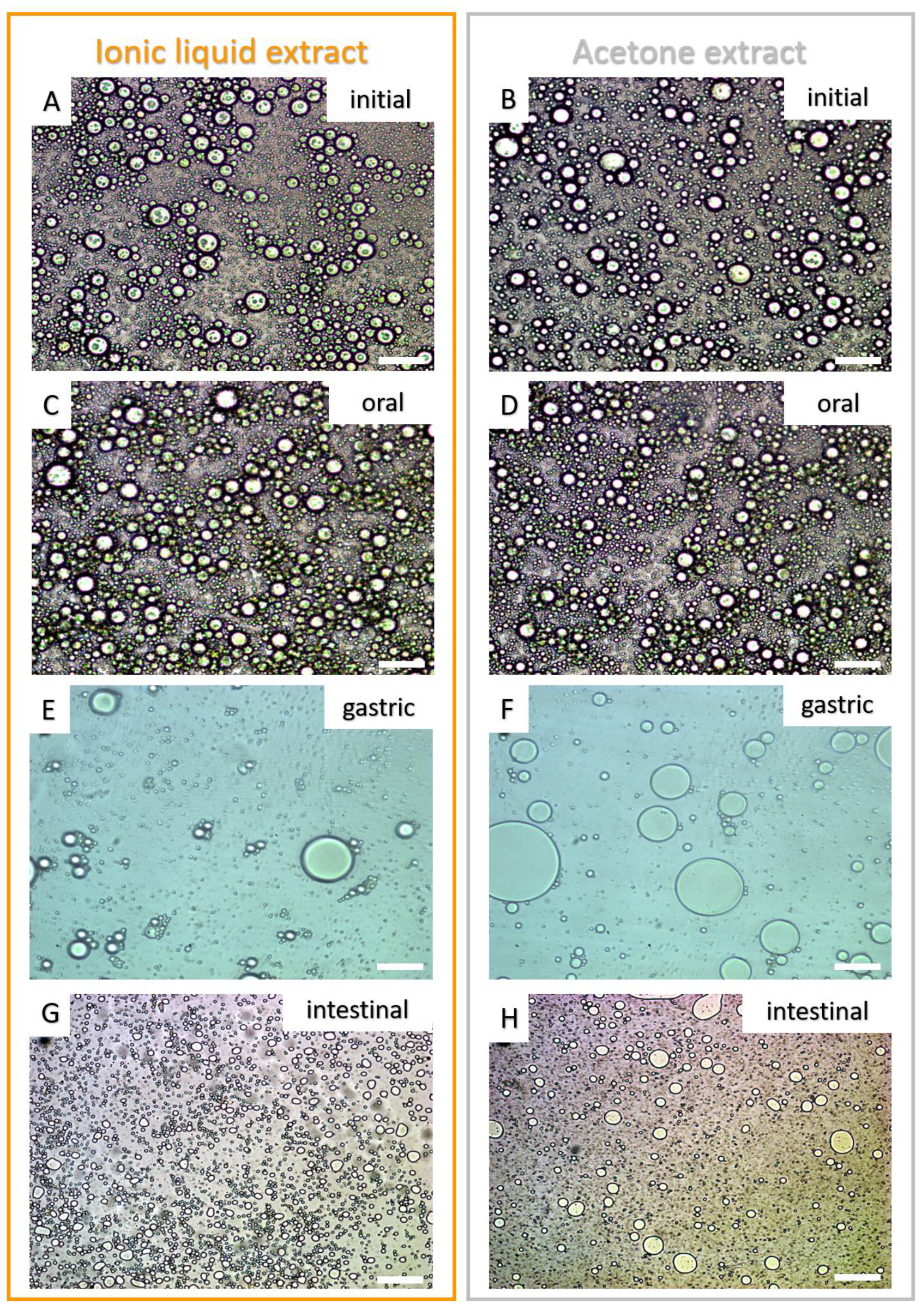
2.4.2. Microstructure of the Emulsion’s Droplets
2.4.3. In Vitro Digestion and Caco-2 Cellular Uptake
2.5. Analysis of the Carotenoids by HPLC-PDA
2.6. Data Presentation and Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Sathasivam:, R.; Ki, J.-S. A review of the biological activities of microalgal carotenoids and their potential use in healthcare and cosmetic industries. Mar. Drugs 2018, 16, 26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagarajan, J.; Ramanan, R.N.; Raghunandan, M.E.; Galanakis, C.M.; Krishnamurthy, N.P. Chapter 8—Carotenoids. In Nutraceutical and Functional Food Components; Galanakis, C.M., Ed.; Academic Press: London, UK, 2017; pp. 259–296. ISBN 9780128052570. [Google Scholar]
- Ngamwonglumlert, L.; Devahastin, S.; Chiewchan, N. Natural colorants: Pigment stability and extraction yield enhancement via utilization of appropriate pretreatment and extraction methods. Crit. Rev. Food Sci. Nutr. 2017, 57, 3243–3259. [Google Scholar] [CrossRef] [PubMed]
- Saini, R.K.; Keum, Y.-S. Carotenoid extraction methods: A review of recent developments. Food Chem. 2018, 240, 90–103. [Google Scholar] [CrossRef] [PubMed]
- Chemat, F.; Abert Vian, M.; Ravi, H.K.; Khadhraoui, B.; Hilali, S.; Perino, S.; Fabiano Tixier, A.-S. Review of alternative solvents for green extraction of food and natural products: Panorama, principles, applications and prospects. Molecules 2019, 24, 3007. [Google Scholar] [CrossRef] [Green Version]
- Ventura, S.P.M.; e Silva, F.A.; Quental, M.V.; Mondal, D.; Freire, M.G.; Coutinho, J.A.P. Ionic-liquid-mediated extraction and separation processes for bioactive compounds: Past, present, and future trends. Chem. Rev. 2017, 117, 6984–7052. [Google Scholar] [CrossRef]
- Anastas, P.T.; Wasserscheid, P.; Stark, A. Green Solvents: Ionic Liquids; John Wiley & Sons: Hoboken, NJ, USA, 2014; ISBN 3527688528. [Google Scholar]
- Poojary, M.; Barba, F.; Aliakbarian, B.; Donsì, F.; Pataro, G.; Dias, D.; Juliano, P. Innovative alternative technologies to extract carotenoids from microalgae and seaweeds. Mar. Drugs 2016, 14, 214. [Google Scholar] [CrossRef]
- Rodriguez-Amaya, D.B. Natural food pigments and colorants. Curr. Opin. Food Sci. 2016, 7, 20–26. [Google Scholar] [CrossRef]
- de Souza Mesquita, L.M.; Ventura, S.P.; Braga, A.R.; Pisani, L.P.; Dias, A.C.; de Rosso, V.V. Ionic liquid-high performance extractive approach to recover carotenoids from Bactris gasipaes fruits. Green Chem. 2019, 21, 2380–2391. [Google Scholar] [CrossRef]
- Garcia Martins, P.L.; de Rosso, V.V.; Martins, P.L.G.; de Rosso, V.V. Thermal and light stabilities and antioxidant activity of carotenoids from tomatoes extracted using an ultrasound-assisted completely solvent-free method. Food Res. Int. 2016, 82, 156–164. [Google Scholar] [CrossRef]
- Murador, D.C.; Braga, A.R.C.; Martins, P.L.G.; Mercadante, A.Z.; de Rosso, V.V. Ionic liquid associated with ultrasonic-assisted extraction: A new approach to obtain carotenoids from orange peel. Food Res. Int. 2019, 126, 108653. [Google Scholar] [CrossRef]
- Clark, J.H.; Farmer, T.J.; Herrero-Davila, L.; Sherwood, J. Circular economy design considerations for research and process development in the chemical sciences. Green Chem. 2016, 18, 3914–3934. [Google Scholar] [CrossRef] [Green Version]
- Oplatowska-Stachowiak, M.; Elliott, C.T. Food colors: Existing and emerging food safety concerns. Crit. Rev. Food Sci. Nutr. 2017, 57, 524–548. [Google Scholar] [CrossRef]
- de Souza Mesquita, L.M.; Martins, M.; Pisani, L.P.; Ventura, S.P.M.; de Rosso, V.V. Insights on the use of alternative solvents and technologies to recover bio-based food pigments. Compr. Rev. Food Sci. Food Saf. 2020, 20, 787–818. [Google Scholar] [CrossRef]
- Comission, E. Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions—A New Circular Economy Action Plan for a Cleaner and more competitive Europe. 2020. Brussels, 11.3.2020. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A52020DC0098 (accessed on 15 May 2021).
- Viera, I.; Pérez-Gálvez, A.; Roca, M. Bioaccessibility of Marine Carotenoids. Mar. Drugs 2018, 16, 397. [Google Scholar] [CrossRef] [Green Version]
- Salvia-Trujillo, L.; Martín-Belloso, O.; McClements, D. Excipient nanoemulsions for improving oral bioavailability of bioactives. Nanomaterials 2016, 6, 17. [Google Scholar] [CrossRef] [Green Version]
- Salvia-Trujillo, L.; Verkempinck, S.H.E.; Sun, L.; Van Loey, A.M.; Grauwet, T.; Hendrickx, M.E. Lipid digestion, micelle formation and carotenoid bioaccessibility kinetics: Influence of emulsion droplet size. Food Chem. 2017, 229, 653–662. [Google Scholar] [CrossRef]
- Failla, M.L.; Chitchumroonchokchai, C.; Ishida, B.K. In vitro micellarization and intestinal cell uptake of cis isomers of lycopene exceed those of all-trans lycopene. J. Nutr. 2008, 138, 482–486. [Google Scholar] [CrossRef]
- Hempel, J.; Amrehn, E.; Quesada, S.; Esquivel, P.; Jiménez, V.M.; Heller, A.; Carle, R.; Schweiggert, R.M. Lipid-dissolved γ-carotene, β-carotene, and lycopene in globular chromoplasts of peach palm (Bactris gasipaes Kunth) fruits. Planta 2014, 240, 1037–1050. [Google Scholar] [CrossRef]
- Bohn, T. Bioavailability of non-provitamin A carotenoids. Curr. Nutr. Food Sci. 2008, 4, 240–258. [Google Scholar] [CrossRef]
- Vargas-Murga, L.; de Rosso, V.V.; Mercadante, A.Z.; Olmedilla-Alonso, B. Fruits and vegetables in the Brazilian Household Budget Survey (2008–2009): Carotenoid content and assessment of individual carotenoid intake. J. Food Compos. Anal. 2016, 50, 88–96. [Google Scholar] [CrossRef] [Green Version]
- Mounien, L.; Tourniaire, F.; Landrier, J.-F. Anti-Obesity Effect of Carotenoids: Direct Impact on Adipose Tissue and Adipose Tissue-Driven Indirect Effects. Nutrients 2019, 11, 1562. [Google Scholar] [CrossRef] [Green Version]
- de Rosso, V.V.; Mercadante, A.Z. Identification and quantification of carotenoids, by HPLC-PDA-MS/MS, from Amazonian fruits. J. Agric. Food Chem. 2007, 55, 5062–5072. [Google Scholar] [CrossRef]
- Passos, H.; Freire, M.G.; Coutinho, J.A.P. Ionic liquid solutions as extractive solvents for value-added compounds from biomass. Green Chem. 2014, 16, 4786–4815. [Google Scholar] [CrossRef] [Green Version]
- Murador, D.C.; de Souza Mesquita, L.M.; Vannuchi, N.; Braga, A.R.C.; de Rosso, V.V. Bioavailability and Biological Effects of Bioactive Compounds Extracted with Natural Deep Eutectic Solvents and Ionic Liquids: Advantages Over Conventional Organic Solvents. Curr. Opin. Food Sci. 2019, 25–34. [Google Scholar] [CrossRef]
- Cao, Y.; Chen, Y.; Sun, X.; Mu, T. Quantification of ionic liquids concentration in water and qualification of conjugated and inductive effects of ionic liquids by UV spectroscopy. CLEAN Soil Air Water 2014, 42, 1162–1169. [Google Scholar] [CrossRef]
- Failla, M.L.; Chitchumronchokchai, C. In Vitro Models as Tools for Screening the Relative Bioavailabilities of Provitamin a Carotenoids in Foods; International Food Policy Research Institute: Washington, DC, USA, 2005. [Google Scholar]
- Kruger, N.J. The Bradford method for protein quantitation. In The Protein Protocols Handbook; Springer: New York, NY, USA, 2009; pp. 17–24. [Google Scholar]
- de Souza Mesquita, L.M.; Martins, M.; Maricato, É.; Nunes, C.; Quinteiro, P.S.G.N.; Dias, A.C.R.V.; Coutinho, J.A.P.; Pisani, L.P.; de Rosso, V.V.; Ventura, S.P.M. Ionic liquid-mediated recovery of carotenoids from the Bactris gasipaes fruit waste and their application in food-packaging chitosan films. ACS Sustain. Chem. Eng. 2020, 8, 4085–4095. [Google Scholar] [CrossRef]
- de Souza Mesquita, L.M.; Neves, B.V.; Pisani, L.P.; de Rosso, V.V. Mayonnaise as a model food for improving the bioaccessibility of carotenoids from Bactris gasipaes fruits. LWT 2020, 109022. [Google Scholar] [CrossRef]
- Bogacz-Radomska, L.; Harasym, J. β-Carotene—Properties and production methods. Food Qual. Saf. 2018, 2, 69–74. [Google Scholar] [CrossRef] [Green Version]
- Singh, S.K.; Savoy, A.W. Ionic liquids synthesis and applications: An overview. J. Mol. Liq. 2020, 297, 112038. [Google Scholar] [CrossRef]
- de Souza Mesquita, L.M.; Casagrande, B.P.; Santamarina, A.B.; Sertorio, M.N.; de Souza, D.V.; Mennitti, L.V.; Jucá, A.; Jamar, G.; Estadella, D.; Ribeiro, D.A.; et al. Carotenoids obtained by an ionic liquid mediated process display anti-inflammatory response in adipose tissue-liver axis. Food Funct. 2021, accepted for publication (in press). [Google Scholar]
- Martins, P.L.G.; de Rosso, V.V.; da Silva, V.H.P.; de Moura, C.F.G.; Ribeiro, D.A. Genotoxicity, mutagenicity and cytotoxicity of carotenoids extracted from ionic liquid in multiples organs of Wistar rats. Exp. Toxicol. Pathol. 2016, 68, 571–578. [Google Scholar] [CrossRef]
- Kaulmann, A.; André, C.M.; Schneider, Y.-J.; Hoffmann, L.; Bohn, T. Carotenoid and polyphenol bioaccessibility and cellular uptake from plum and cabbage varieties. Food Chem. 2016, 197, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Cláudio, A.F.M.; Ferreira, A.M.; Freire, M.G.; Coutinho, J.A.P. Enhanced extraction of caffeine from guarana seeds using aqueous solutions of ionic liquids. Green Chem. 2013, 15, 2002–2010. [Google Scholar] [CrossRef]
- Xavier, A.A.O.; Mercadante, A.Z. The bioaccessibility of carotenoids impacts the design of functional foods. Curr. Opin. Food Sci. 2019, 26, 1–8. [Google Scholar] [CrossRef]
- Radice, M.; Viafara, D.; Neill, D.; Asanza, M.; Sacchetti, G.; Guerrini, A.; Maietti, S. Chemical characterization and antioxidant activity of Amazonian (Ecuador) Caryodendron orinocense Karst. and Bactris gasipaes Kunth seed oils. J. Oleo Sci. 2014, 63, 1243–1250. [Google Scholar] [CrossRef] [Green Version]
- Nagao, A.; Kotake-Nara, E.; Hase, M. Effects of fats and oils on the bioaccessibility of carotenoids and vitamin E in vegetables. Biosci. Biotechnol. Biochem. 2013, 77, 1055–1060. [Google Scholar] [CrossRef] [Green Version]
- Schweiggert, R.M.; Steingass, C.B.; Mora, E.; Esquivel, P.; Carle, R. Carotenogenesis and physico-chemical characteristics during maturation of red fleshed papaya fruit (Carica papaya L.). Food Res. Int. 2011, 44, 1373–1380. [Google Scholar] [CrossRef]
- Lipkie, T.E.; De Moura, F.F.; Zhao, Z.-Y.; Albertsen, M.C.; Che, P.; Glassman, K.; Ferruzzi, M.G. Bioaccessibility of carotenoids from transgenic provitamin A biofortified sorghum. J. Agric. Food Chem. 2013, 61, 5764–5771. [Google Scholar] [CrossRef]
- Brown, M.J.; Ferruzzi, M.G.; Nguyen, M.L.; Cooper, D.A.; Eldridge, A.L.; Schwartz, S.J.; White, W.S. Carotenoid bioavailability is higher from salads ingested with full-fat than with fat-reduced salad dressings as measured with electrochemical detection. Am. J. Clin. Nutr. 2004, 80, 396–403. [Google Scholar] [CrossRef]
- Failla, M.L.; Chitchumronchokchai, C.; Ferruzzi, M.G.; Goltz, S.R.; Campbell, W.W. Unsaturated fatty acids promote bioaccessibility and basolateral secretion of carotenoids and α-tocopherol by Caco-2 cells. Food Funct. 2014, 5, 1101–1112. [Google Scholar] [CrossRef] [Green Version]
- Hornero-Méndez, D.; Mínguez-Mosquera, M.I. Bioaccessibility of carotenes from carrots: Effect of cooking and addition of oil. Innov. Food Sci. Emerg. Technol. 2007, 8, 407–412. [Google Scholar] [CrossRef]
- Palmero, P.; Panozzo, A.; Colle, I.; Chigwedere, C.; Hendrickx, M.; Van Loey, A. Role of structural barriers for carotenoid bioaccessibility upon high pressure homogenization. Food Chem. 2016, 199, 423–432. [Google Scholar] [CrossRef]
- Trancoso-Reyes, N.; Ochoa-Martínez, L.A.; Bello-Pérez, L.A.; Morales-Castro, J.; Estévez-Santiago, R.; Olmedilla-Alonso, B. Effect of pre-treatment on physicochemical and structural properties, and the bioaccessibility of β-carotene in sweet potato flour. Food Chem. 2016, 200, 199–205. [Google Scholar] [CrossRef] [Green Version]
- Carrillo, C.; Buvé, C.; Panozzo, A.; Grauwet, T.; Hendrickx, M. Role of structural barriers in the in vitro bioaccessibility of anthocyanins in comparison with carotenoids. Food Chem. 2017, 227, 271–279. [Google Scholar] [CrossRef]
- Petry, F.C.; Mercadante, A.Z. Impact of in vitro digestion phases on the stability and bioaccessibility of carotenoids and their esters in mandarin pulps. Food Funct. 2017, 8, 3951–3963. [Google Scholar] [CrossRef]
- Reboul, E. Absorption of vitamin A and carotenoids by the enterocyte: Focus on transport proteins. Nutrients 2013, 5, 3563–3581. [Google Scholar] [CrossRef] [Green Version]
- Sy, C.; Gleize, B.; Dangles, O.; Landrier, J.; Veyrat, C.C.; Borel, P. Effects of physicochemical properties of carotenoids on their bioaccessibility, intestinal cell uptake, and blood and tissue concentrations. Mol. Nutr. Food Res. 2012, 56, 1385–1397. [Google Scholar] [CrossRef]
- Thakkar, S.K.; Maziya-Dixon, B.; Dixon, A.G.O.; Failla, M.L. β-Carotene micellarization during in vitro digestion and uptake by Caco-2 cells is directly proportional to β-carotene content in different genotypes of cassava. J. Nutr. 2007, 137, 2229–2233. [Google Scholar] [CrossRef] [Green Version]
| Samples | Initial Concentration—Input of IL | Concentration Recovered after Extraction Procedure (µg/mL) | Recovery (%) |
|---|---|---|---|
| Carotenoid extract submitted to polishing step (used in the simulated digestion process) | 500 µg/mL | - | - |
| - | |||
| - | |||
| Carotenoid extract without polishing step (control) | 500 µg/mL | 491.22 | 98.37 ± 0.13 |
| 492.35 | |||
| 492.14 |
| Samples | [C4mim][BF4] Carotenoid Extract (µm) | Acetone Carotenoid Extract (µm) |
|---|---|---|
| Initial (before in vitro digestion) | 12.46 ± 2.96 a | 11.98 ± 1.96 a |
| Oral | 11.46 ± 1.63 a | 11.13 ± 1.63 a |
| Gastric | 10.8 ± 5.89 a | 14.14 ± 6.55 a |
| Intestinal | 5.01 ± 1.39 a | 5.62 ± 1.77 a |
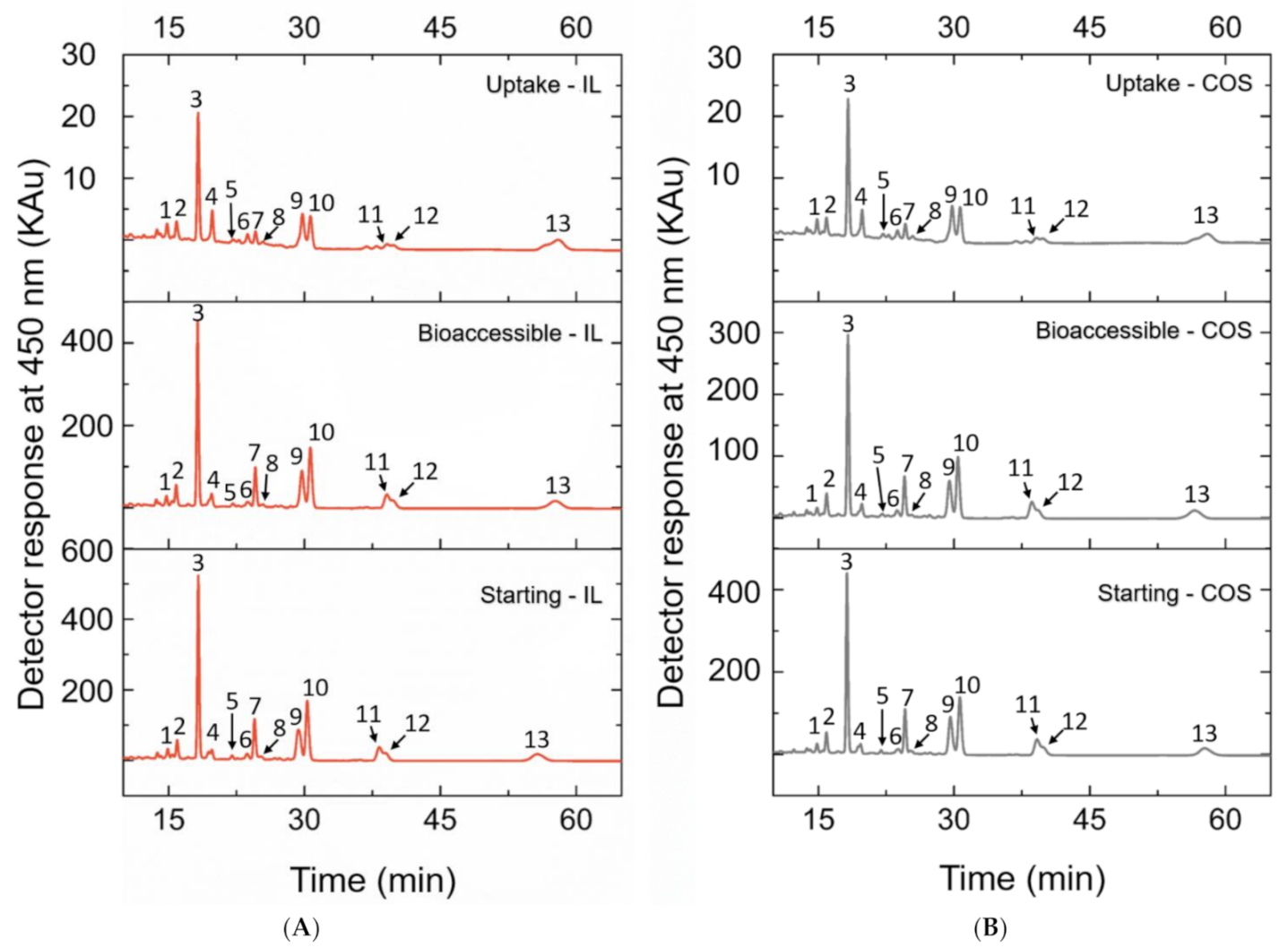
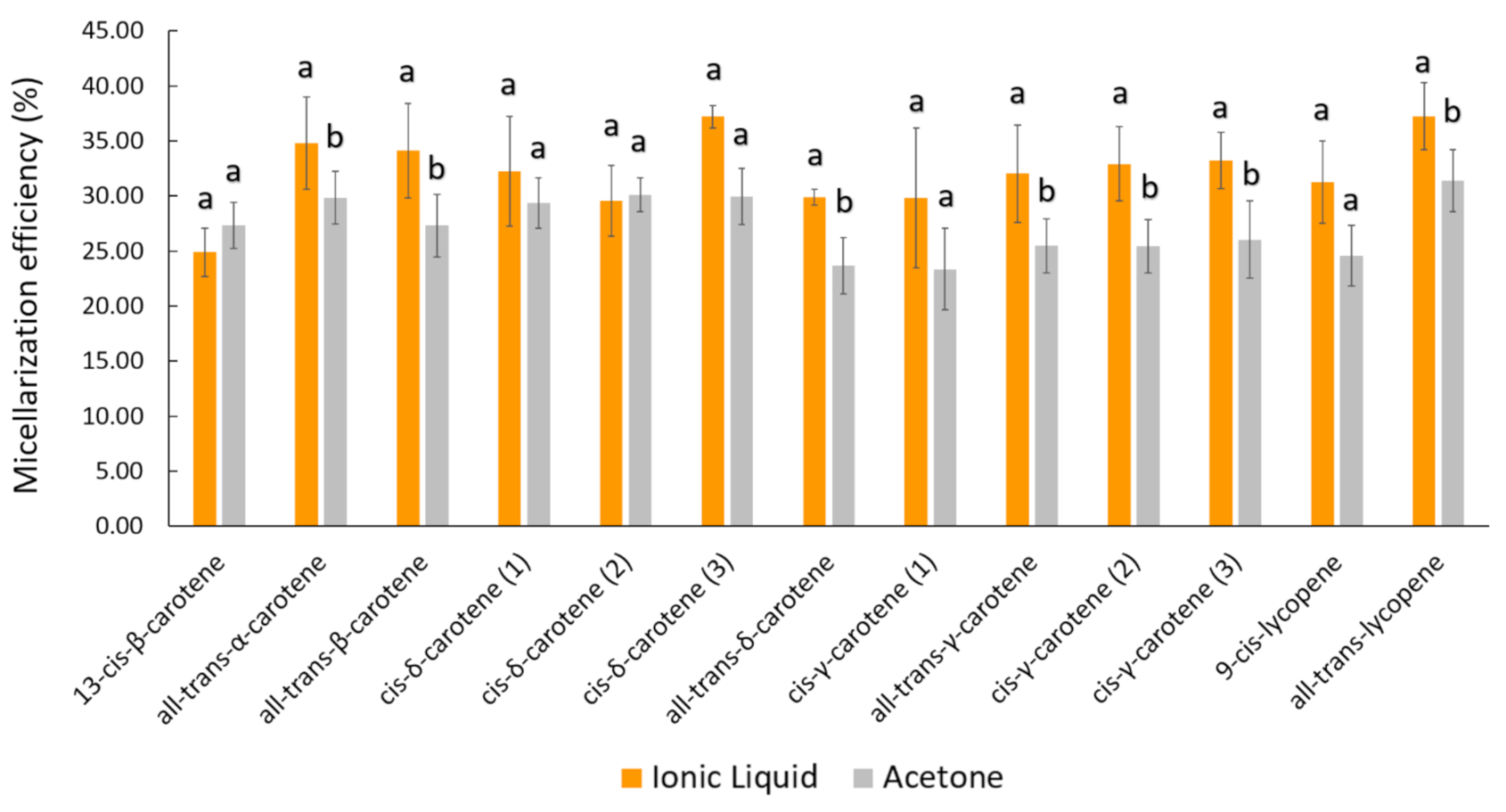
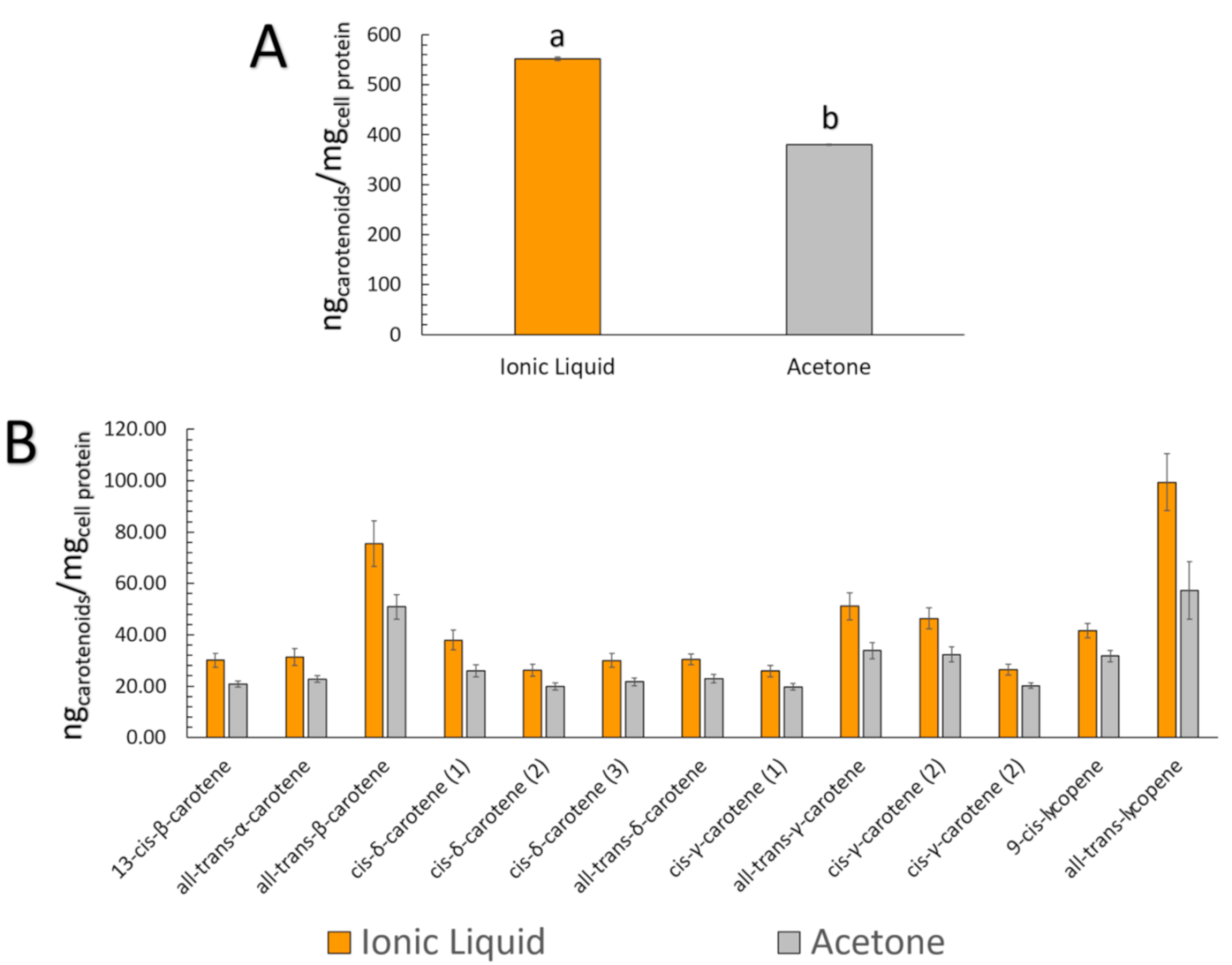
| Peak | Carotenoid | [C4mim][BF4] Carotenoid Extract | Acetone Carotenoid Extract | ||||
|---|---|---|---|---|---|---|---|
| Starting (µg) * | Bioaccessible—BCT (µg) *a | Uptake (ngcarotenoids/mgcell protein) * | Starting (µg) * | Bioaccessible—BCT (µg) *a | Uptake (ngcarotenoids/mgcell protein) * | ||
| 1 | 13-cis-β-carotene | 3.71 ± 0.52 | 0.93 ± 0.21 (24.90%) | 30.07 ± 2.60 | 3.07 ± 0.25 | 0.84 ± 0.05 (27.35%) | 20.84 ± 1.22 |
| 2 | all-trans-α-carotene | 6.11 ± 0.17 | 2.12 ± 0.25 (34.80%) | 31.32 ± 3.29 | 5.48 ± 0.66 | 1.63 ± 0.16 (29.85%) | 22.78 ± 1.24 |
| 3 | all-trans-β-carotene | 48.50 ± 1.69 | 16.52 ± 1.80 (34.13%) | 75.53 ± 8.92 | 36.98 ± 5.62 | 10.02 ± 1.20 (27.31%) | 50.86 ± 4.88 |
| 4 | cis-δ-carotene (1) | 6.67 ± 0.52 | 2.13 ± 0.18 (32.24%) | 37.97 ± 3.89 | 4.83 ± 0.39 | 1.41 ± 0.14 (29.33%) | 26.07 ± 2.33 |
| 5 | cis-δ-carotene (2) | 2.51 ± 0.13 | 0.74 ± 0.04 (29.56%) | 26.12 ± 2.31 | 2.00 ± 0.12 | 0.60 ± 0.03 (30.10%) | 19.91 ± 1.34 |
| 6 | cis-δ-carotene (3) | 3.21 ± 0.07 | 1.19 ± 0.01 (37.21%) | 29.99 ± 2.69 | 3.15 ± 0.31 | 0.94 ± 0.11 (29.96%) | 21.70 ± 1.52 |
| 7 | all-trans-δ-carotene | 14.48 ± 0.74 | 4.33 ± 0.25 (29.88%) | 30.44 ± 2.06 | 12.14 ± 1.70 | 2.86 ± 0.36 (23.67%) | 22.94 ± 1.60 |
| 8 | cis-γ-carotene (1) | 2.26 ± 0.11 | 0.67 ± 0.11 (29.80%) | 25.85 ± 2.12 | 2.39 ± 0.19 | 0.56 ± 0.08 (23.36%) | 19.70 ± 1.30 |
| 9 | all-trans-γ-carotene | 21.07 ± 0.67 | 6.73 ± 0.80 (32.02%) | 51.10 ± 5.27 | 16.95 ± 2.37 | 4.29 ± 0.46 (25.46%) | 33.82 ± 3.12 |
| 10 | cis-γ-carotene (2) | 27.52 ± 0.98 | 9.06 ± 0.92 (32.92%) | 46.40 ± 4.13 | 22.25 ± 3.34 | 5.62 ± 0.59 (25.42%) | 32.32 ± 2.93 |
| 11 | cis-γ-carotene (3) | 11.03 ± 0.41 | 3.66 ± 0.31 (33.21%) | 26.36 ± 2.08 | 9.98 ± 1.69 | 2.56 ± 0.14 (26.04%) | 20.23 ± 1.11 |
| 12 | 9-cis-lycopene | 12.35 ± 0.43 | 3.85 ± 0.42 (31.24%) | 41.64 ± 2.84 | 10.81 ± 1.47 | 2.66 ± 0.45 (24.57%) | 31.69 ± 2.32 |
| 13 | all-trans-lycopene | 26.90 ± 1.13 | 10.01 ± 0.86 (37.22%) | 99.37 ± 11.15 | 21.23 ± 3.26 | 6.62 ± 0.76 (31.38%) | 57.29 ± 11.13 |
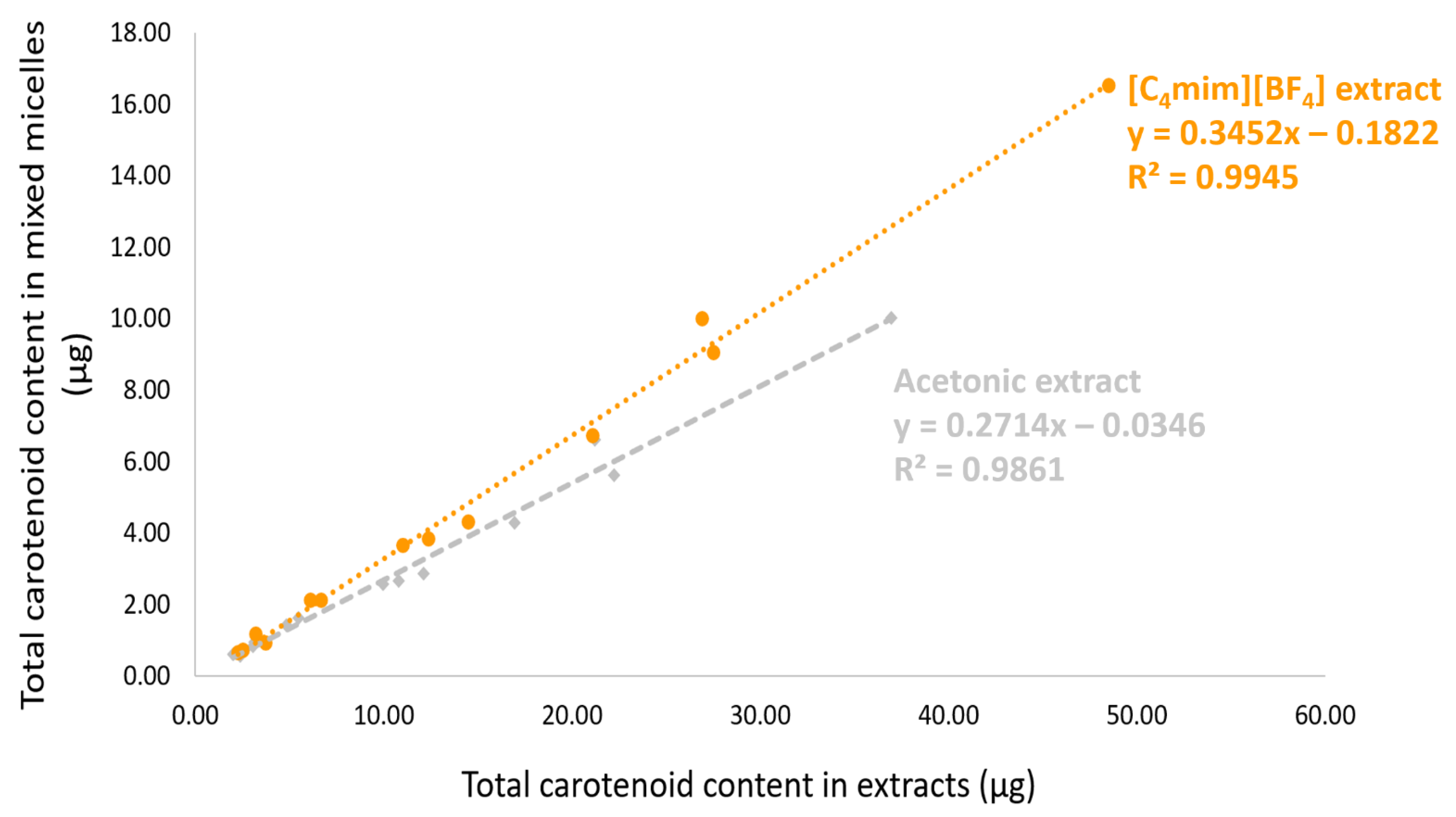
| Total | 186.30 ± 5.39 | 61.95 ± 5.50 (33.25%) # | 552.17 ± 3.65## | 151.25 ± 21.15 | 40.59 ± 4.42 (26.84%) | 380.16 ± 1.71 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Souza Mesquita, L.M.; Murador, D.C.; Neves, B.V.; Braga, A.R.C.; Pisani, L.P.; de Rosso, V.V. Bioaccessibility and Cellular Uptake of Carotenoids Extracted from Bactris gasipaes Fruit: Differences between Conventional and Ionic Liquid-Mediated Extraction. Molecules 2021, 26, 3989. https://doi.org/10.3390/molecules26133989
de Souza Mesquita LM, Murador DC, Neves BV, Braga ARC, Pisani LP, de Rosso VV. Bioaccessibility and Cellular Uptake of Carotenoids Extracted from Bactris gasipaes Fruit: Differences between Conventional and Ionic Liquid-Mediated Extraction. Molecules. 2021; 26(13):3989. https://doi.org/10.3390/molecules26133989
Chicago/Turabian Stylede Souza Mesquita, Leonardo M., Daniella Carisa Murador, Bruna Vitória Neves, Anna Rafaela Cavalcante Braga, Luciana Pellegrini Pisani, and Veridiana Vera de Rosso. 2021. "Bioaccessibility and Cellular Uptake of Carotenoids Extracted from Bactris gasipaes Fruit: Differences between Conventional and Ionic Liquid-Mediated Extraction" Molecules 26, no. 13: 3989. https://doi.org/10.3390/molecules26133989
APA Stylede Souza Mesquita, L. M., Murador, D. C., Neves, B. V., Braga, A. R. C., Pisani, L. P., & de Rosso, V. V. (2021). Bioaccessibility and Cellular Uptake of Carotenoids Extracted from Bactris gasipaes Fruit: Differences between Conventional and Ionic Liquid-Mediated Extraction. Molecules, 26(13), 3989. https://doi.org/10.3390/molecules26133989