Role of Withaferin A and Its Derivatives in the Management of Alzheimer’s Disease: Recent Trends and Future Perspectives
Abstract
1. Introduction
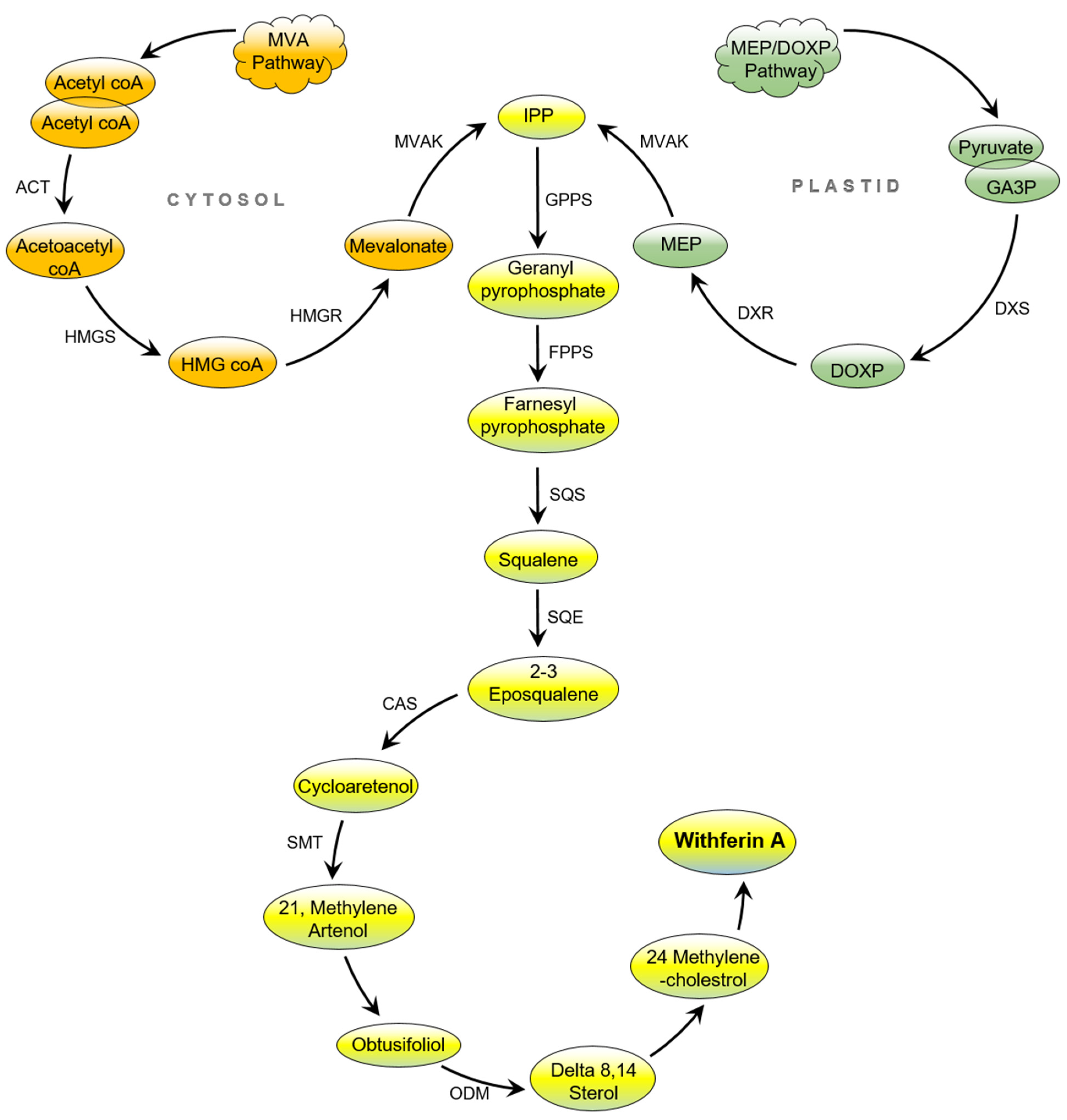
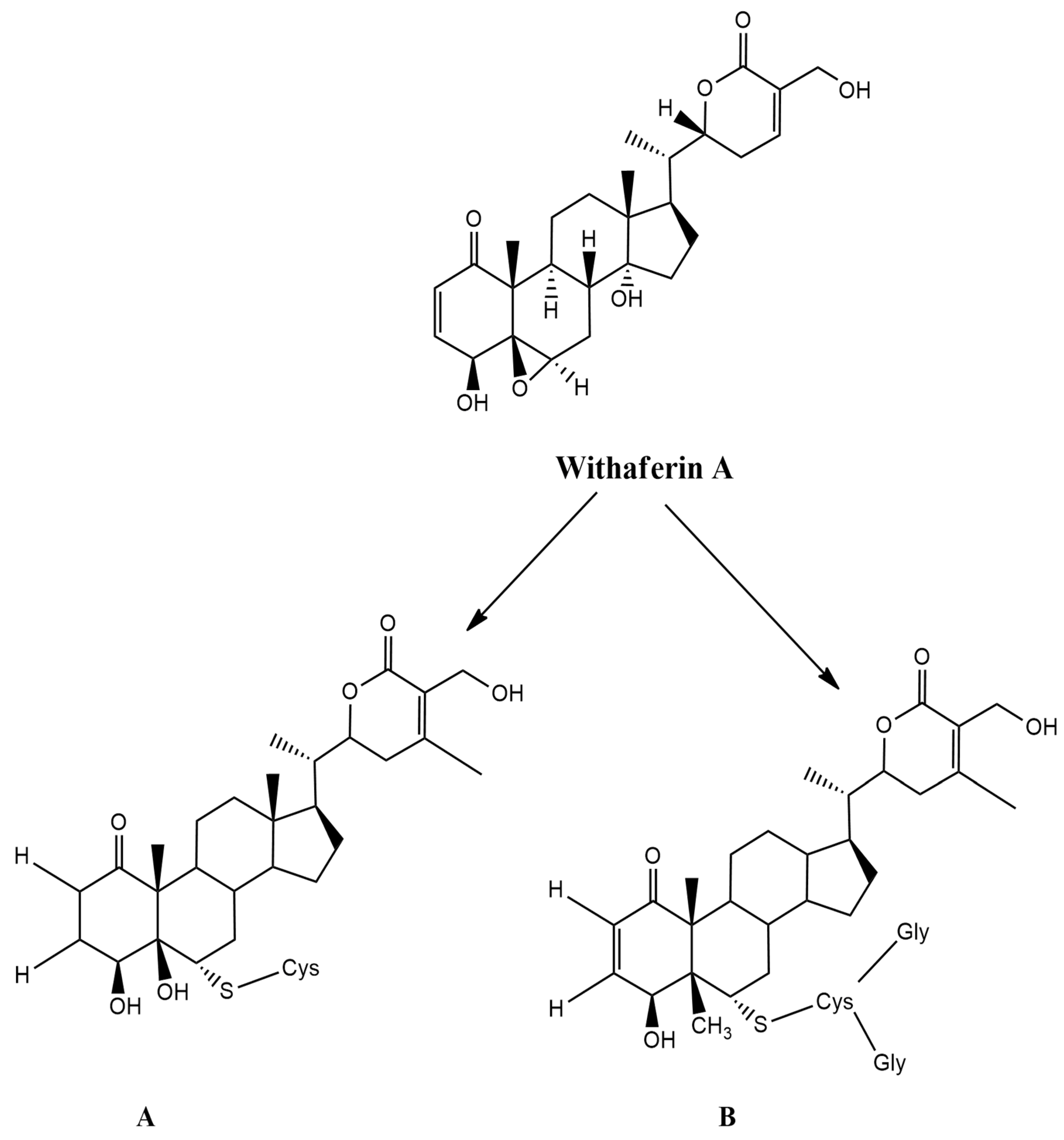
2. Biosynthesis of Withaferin A (WA)
3. Pathogenesis of AD
3.1. The Amyloid Hypothesis
3.1.1. Nonamyloidogenic Pathway
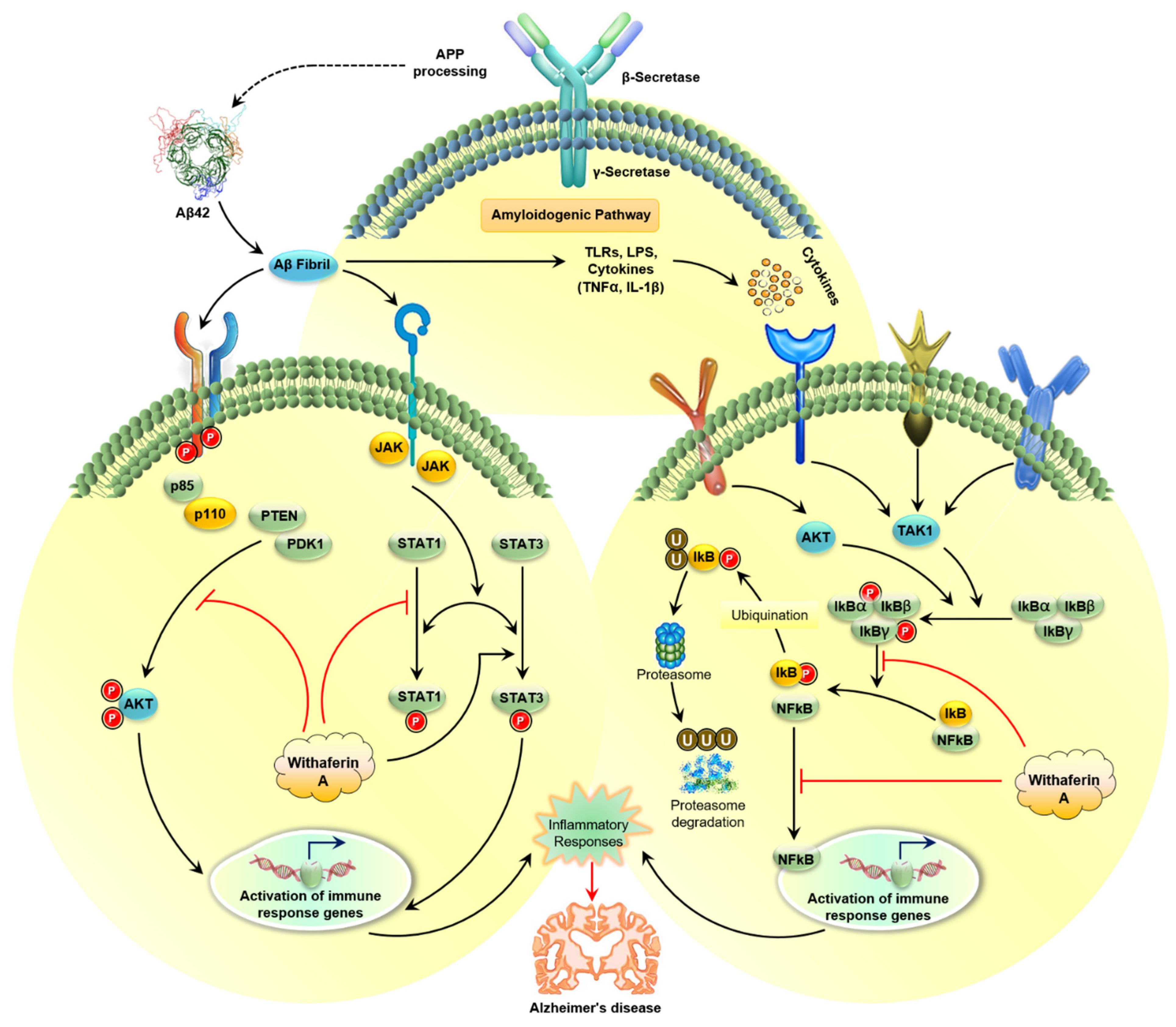
3.1.2. Amyloidogenic Pathway
3.2. The Tau (τ) Hypothesis
3.3. Oxidative Stress Hypothesis
3.4. The Cholinergic Hypothesis
3.5. Inflammatory Hypothesis
4. Epidemiology and Clinical Epidemiology
Clinical Epidemiology
5. Therapeutic Implications of WA in AD
5.1. Role of WA and Its Derivatives
5.1.1. WA and Its Derivatives Aβ Plaque Formation Inhibitors
5.1.2. WA Acts as Antioxidant in AD
5.1.3. WA Inhibits AChE and BuChE Activities
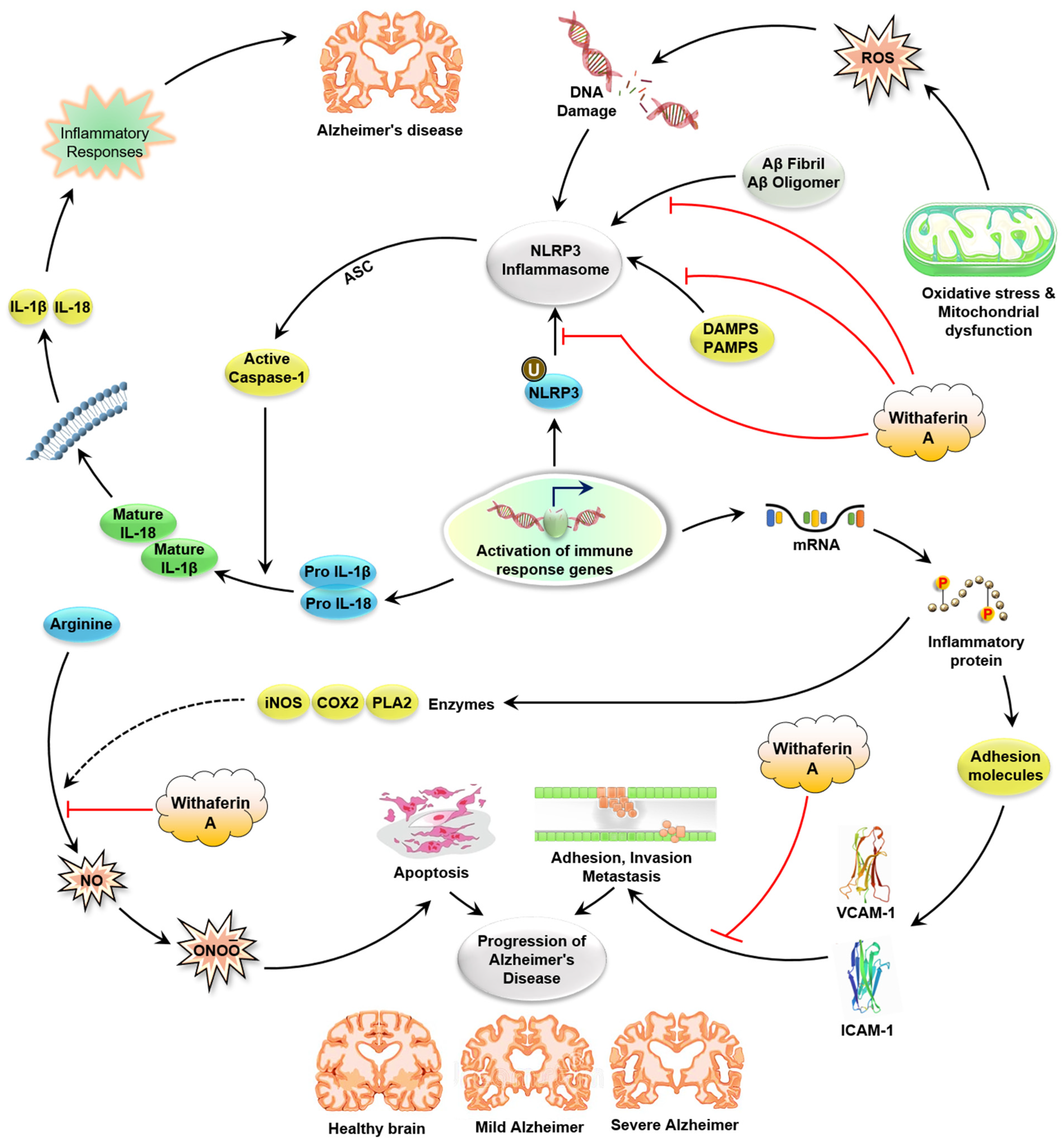
5.1.4. WA Blocks the Neuroinflammation in AD
WA Inhibits the NF-κB Pathway
WA Affects Inflammasome Activation
WA Regulates Heat Shock Proteins
| Plant Extract | Method | Subject | WA Mediated Protective Effect | Possible Mechanism | Up/Down Regulation | References |
|---|---|---|---|---|---|---|
| Aqueous methanol extract of Withania somnifera roots | In vivo | Mice | Reversed anti-AChE activity | Enhance ACh, choline acetyltransferase; ChAT activity in globus pallidus and lateral septum |  | [22] |
| Aqueous chloroform extract of Withania somnifera roots | In vivo | Rat | Anti-cholinergic activity | Reduced cholinergic marker activity | 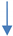 | [23] |
| Withania somnifera root extract | In vitro | SH-SY5Y cells (SHAPP) | Anti-amyloidogenic | Aβ40 | 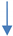 | [24] |
| Withania somnifera root extract | In vitro | SHAPP cells and CHME5 microglial cell line | Anti-inflammatory | NF-κB, JUN and STAT gene, IL-1β | 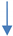 | [24,148] |
| Withania somnifera extract | In vitro | SK-N-SH cells | Anti-oxidant and anti-cholinergic | ROS, Ache, Aβ peptide toxicity | 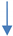 | [25] |
| Aqueous root extract of Withania somnifera | In vitro | Rat pheochromocytoma (PC12) cell line | Anti-Alzheimer activity | H2O2- and Aβ-induced toxicity |  | [26] |
| Plant extract of withania somnifera | In vivo | Male Wistar rats | Anti-Alzheimer activity | Reduced acetyl cholinesterase |  | [27] |
| Plant extract of withania somnifera | In vitro | Amyloid-β marker thioflavin-T | Anti-amyloidogenic | Reduced amyloid beta |  | [184] |
| WA | In vivo | HFD-induced obese mice | Anti-obesity | COX2, NF-κB, TNF-α, inflammation, oxidative stress, and insulin resistance |  | [185] |
| WA | In vitro, in vivo | Human umbilical vein endothelial cells (HUVECs), mouse | Anti-inflammatory | Inhibit phorbol-12-myristate-3-acetate (PMA), TNF-a, (IL)-1β, PMA-stimulated phosphorylation of p38, extracellular regulated kinases (ERK)-1/2, and c-Jun N-terminal kinase (JNK) |  | [186] |
| WA | In vitro | Murine fibrosarcoma | Anti-inflammatory | p38, ERK-1/2, C-Jun (JNK) |  | [136] |
| WA | In vitro | Cellular models of cystic fibrosis inflammation (KKLEB cells) | Anti-inflammatory | NFk-β and IL-8 |  | [187] |
| WA | In vitro | Human melanoma cells (M14, Lu1205, and Sk28) | Anti-cancer | ROS-induced apoptosis increased by lowering the Bax/Bcl2 and Bcl2/Bim ratio |  | [188] |
| WA | In vitro | Breast cancer cells (MDA-MB-231 and MCF-7) | Anti-cancer | Caspase-9 and 3 and PARP |  | [189] |
| WA | In vitro, in vivo | Breast tumor progression in xenograft and transgenic mouse models | Anti-cancer | ERK/RSK axis, death receptor 5 (DR-5), ETS domain containing protein-1 (Elk1), and CAAT/enhancer-binding protein-homologous protein (CHOP) |  | [190] |
| WA | In vitro | Human laryngeal carcinoma Hep2 cells | Anti-cancer | Cell cycle arrest with concomitant blockade of angiogenesis |  | [191] |
| WA | In vitro | Renal cancers (Caki cells) | Anti-cancer | STAT-3 pathway |  | [192] |
| WA | In vitro | Renal cancers (Caki cells) | Anti-cancer | GRP-78 and CHOP |  | [193] |
| Extract of Whitania aristata | In vivo | Male albino Sprague-Dawley rats and male and female albino Swiss mice | Diuretic effect | Diuretic activity, excretion of sodium and potassium ions |  | [194] |
| WA | In vitro | H. pylori-induce bone marrow-derived dendritic cells (BMDCs) | Anti-gastric cancer | NF-κB, IL-1β, NLRP3 |  | [195] |
| Aqueous root extract of Withania somnifera | In vitro | Nicotine induced conditioned place reference in male albino mice | Anti-addictive | Nicotine efficacy |  | [196] |
| WA | In vitro | Microglial cells | Anti-inflammatory | STAT1/3, interferon-gamma activated sequence (GAS)-promoter activity |  | [197] |
| WA | In vitro | Mouse model of FTLD | Neuroprotective | TAR DNA-binding protein-43, NF-κB activity and neuroinflammation |  | [198] |
| WA | In vivo and ex vivo | TNF-stimulated human umbilical vein-endothelial cells | Anti-coagulant | Plasminogen activator inhibitor type 1 (PAI-1/t), tissue-type plasminogen activator (t-PA) |  | [21] |
| WA | In vivo | Swiss albino mice | Anti-diabetic | Hyperglycemia | [199] |
6. Structural Modifications of WA for Further Neuroprotective Activity
7. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Li, C.; Zhao, R.; Gao, K.; Wei, Z.; Yaoyao Yin, M.; Ting Lau, L.; Chui, D.; Cheung Hoi Yu, A. Astrocytes: Implications for Neuroinflammatory Pathogenesis of Alzheimers Disease. Curr. Alzheimer Res. 2011, 8, 67–80. [Google Scholar] [CrossRef]
- Sanabria-Castro, A.; Alvarado-Echeverría, I.; Monge-Bonilla, C. Molecular pathogenesis of alzheimer’s disease: An update. Ann. Neurosci. 2017, 24, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, S.; Venkata, A.; Kaushik, A.; Adriana, Y.; Nair, M. Alzheimer’s Disease Diagnostics and Therapeutics Market. Int. J. Nanomed. 2019, 2019, 5541–5554. [Google Scholar] [CrossRef]
- Fan, L.; Mao, C.; Hu, X.; Zhang, S.; Yang, Z.; Hu, Z.; Sun, H.; Fan, Y.; Dong, Y.; Yang, J.; et al. New Insights Into the Pathogenesis of Alzheimer’s Disease. Front. Neurol. 2020, 10, 1312. [Google Scholar] [CrossRef]
- Guo, T.; Zhang, D.; Zeng, Y.; Huang, T.Y.; Xu, H.; Zhao, Y. Molecular and cellular mechanisms underlying the pathogenesis of Alzheimer’s disease. Mol. Neurodegener. 2020, 15, 40. [Google Scholar] [CrossRef]
- Mohd Sairazi, N.S.; Sirajudeen, K.N.S. Natural Products and Their Bioactive Compounds: Neuroprotective Potentials against Neurodegenerative Diseases. Evid. Based Complement. Altern. Med. 2020, 2020, 6565396. [Google Scholar] [CrossRef]
- Rao, R.V.; Descamps, O.; John, V.; Bredesen, D.E. Ayurvedic medicinal plants for Alzheimer’s disease: A review. Alzheimer’s Res. Ther. 2012, 4, 22. [Google Scholar] [CrossRef]
- Singh, A.P. The Role of Natural Products in Pharmacotherapy of Alzheimer’s Disease. Ethnobot. Leafl. 2005, 2005, 46. [Google Scholar]
- Reddy, P.H.; Beal, M.F. Are mitochondria critical in the pathogenesis of Alzheimer’s disease? Brain Res. Rev. 2005, 49, 618–632. [Google Scholar] [CrossRef] [PubMed]
- Kumar Thakur, A.; Kamboj, P.; Goswami, K.; Ahuja, K. Pathophysiology and management of alzheimer’s disease: An overview. J. Anal. Pharm. Res. 2018, 7, 226–235. [Google Scholar] [CrossRef]
- Maccioni, R.B.; Rojo, L.E.; Fernández, J.A.; Kuljis, R.O. The role of neuroimmunomodulation in Alzheimer’s disease. Ann. N. Y. Acad. Sci. 2009, 1153, 240–246. [Google Scholar] [CrossRef]
- Brown, T.L. Alzheimer’s Disease Prevention and Treatment Using Herbal Agents. J. Anal. Pharm. Res. 2016, 2, 2–5. [Google Scholar] [CrossRef][Green Version]
- Hamill, R.W.; Pilgrim, D.M. Advances in Alzheimer’s disease. Contemp. Intern. Med. 1995, 7. [Google Scholar] [CrossRef]
- Reddy, C.R.M.; Raju, E.V.N.; Reddy, M.S.; Manjunatha, P.M. Role of medicinal plants in the treatment of Alzheimer’s disease: A review. Sch. Acad. J. Pharm. 2013, 2, 21–26. [Google Scholar]
- Patel, K.C.; Pramanik, S.; Patil, V.C. Ayurvedic approach with a propective to treat and prevent Alzheimer’s and other cognitive diseases: A review. World J. Pharm. Pharm. Sci. 2014, 3, 234–252. [Google Scholar]
- Kalra, J.; Khan, A. Reducing Aβ load and tau phosphorylation: Emerging perspective for treating Alzheimer’s disease. Eur. J. Pharmacol. 2015, 764, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Sahoo, A.K.; Dandapat, J.; Dash, U.C.; Kanhar, S. Features and outcomes of drugs for combination therapy as multi-targets strategy to combat Alzheimer’s disease. J. Ethnopharmacol. 2018, 215, 42–73. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, M.N.; Ansarn, A.A.; Khan, K.Z. Approaches for the Treatment of Alzheimer ’ s disease in Unani Medicine: A Review. J. Integr. Community Health 2017, 2012, 25–30. [Google Scholar]
- Mucke, L. Neuroscience: Alzheimer’s disease. Nature 2009, 461, 895–897. [Google Scholar] [CrossRef] [PubMed]
- Logie, E.; Berghe, W. Vanden Tackling chronic inflammation with withanolide phytochemicals—A withaferin a perspective. Antioxidants 2020, 9, 1107. [Google Scholar] [CrossRef] [PubMed]
- Behl, T.; Sharma, A.; Sharma, L.; Sehgal, A.; Zengin, G.; Brata, R.; Fratila, O.; Bungau, S. Exploring the multifaceted therapeutic potential of withaferin a and its derivatives. Biomedicines 2020, 8, 571. [Google Scholar] [CrossRef]
- Uddin, M.S.; Al Mamun, A.; Kabir, M.T.; Jakaria, M.; Mathew, B.; Barreto, G.E.; Ashraf, G.M. Nootropic and Anti-Alzheimer’s Actions of Medicinal Plants: Molecular Insight into Therapeutic Potential to Alleviate Alzheimer’s Neuropathology. Mol. Neurobiol. 2019, 56, 4925–4944. [Google Scholar] [CrossRef]
- Omar, F.; Tareq, A.M.; Alqahtani, A.M.; Dhama, K.; Sayeed, M.A.; Emran, T.B.; Simal-Gandara, J. Plant-based Indole Alkaloids: A Comprehensive Overview from a Pharmacological Perspective. Molecules 2021, 26, 2297. [Google Scholar] [CrossRef]
- Tiwari, S.; Atluri, V.S.R.; Yndart Arias, A.; Jayant, R.D.; Kaushik, A.; Geiger, J.; Nair, M.N. Withaferin a Suppresses Beta Amyloid in APP Expressing Cells: Studies for Tat and Cocaine Associated Neurological Dysfunctions. Front. Aging Neurosci. 2018, 10, 291. [Google Scholar] [CrossRef]
- Singh, M.; Ramassamy, C. In vitro screening of neuroprotective activity of Indian medicinal plant Withania somnifera. J. Nutr. Sci. 2017, 6. [Google Scholar] [CrossRef]
- Kumar, S.; Seal, C.J.; Howes, M.J.R.; Kite, G.C.; Okello, E.J. In vitro protective effects of Withania somnifera (L.) Dunal root extract against hydrogen peroxide and β-amyloid(1-42)-induced cytotoxicity in differentiated PC12 cells. Phyther. Res. 2010, 24, 1567–1574. [Google Scholar] [CrossRef] [PubMed]
- Vanden Berghe, W.; Sabbe, L.; Kaileh, M.; Haegeman, G.; Heyninck, K. Molecular insight in the multifunctional activities of Withaferin A. Biochem. Pharmacol. 2012, 84, 1282–1291. [Google Scholar] [CrossRef] [PubMed]
- Das, T.; Das, T.; Sloan, M.; Cancer, K.; Shad, K.F. Potential of glycowithanolides from Withania somnifera (ashwagandha) as therapeutic agents for the treatment of Alzheimer’s disease. World J. Pharm. Res. 2015, 4, 16–38. [Google Scholar]
- Patel, S.B.; Rao, N.J.; Hingorani, L.L. Safety assessment of Withania somnifera extract standardized for Withaferin A: Acute and sub-acute toxicity study. J. Ayurveda Integr. Med. 2016, 7, 30–37. [Google Scholar] [CrossRef]
- Rahaman, M.M.; Rakib, A.; Mitra, S.; Tareq, A.T.; Emran, T.B.; Ud-Daula, S.A.F.M.; Amin, M.N.; Simal-Gandara, J. The Genus Curcuma and Inflammation: Overview of the Pharmacological Perspectives. Plants 2021, 10, 63. [Google Scholar] [CrossRef]
- Yang, B.Y.; Xia, Y.G.; Pan, J.; Liu, Y.; Wang, Q.H.; Kuang, H.X. Phytochemistry and biosynthesis of δ-lactone withanolides. Phytochem. Rev. 2016, 15, 771–797. [Google Scholar] [CrossRef]
- Chen, L.X.; He, H.; Qiu, F. Natural withanolides: An overview. Nat. Prod. Rep. 2011, 28, 705–740. [Google Scholar] [CrossRef] [PubMed]
- Monteagudo, E.S.; Veleiro, A.S.; Burton, G.; Gros, E.G. Biosynthesis of Withanolides in Acnistus breviflorus Chemical Degradation of 14C-Labelled Jaborosalactone A and Withaferin A. Z. Naturforsch. Sect. B 1987, 42, 1471–1475. [Google Scholar] [CrossRef]
- Sangwan, R.S.; Das Chaurasiya, N.; Lal, P.; Misra, L.; Tuli, R.; Sangwan, N.S. Withanolide a is inherently de novo biosynthesized in roots of the medicinal plant Ashwagandha (Withania somnifera). Physiol. Plant. 2008, 133, 278–287. [Google Scholar] [CrossRef]
- Gupta, N.; Sharma, P.; Santosh Kumar, R.J.; Vishwakarma, R.K.; Khan, B.M. Functional characterization and differential expression studies of squalene synthase from Withania somnifera. Mol. Biol. Rep. 2012, 39, 8803–8812. [Google Scholar] [CrossRef]
- Razdan, S.; Bhat, W.W.; Rana, S.; Dhar, N.; Lattoo, S.K.; Dhar, R.S.; Vishwakarma, R.A. Molecular characterization and promoter analysis of squalene epoxidase gene from Withania somnifera (L.) Dunal. Mol. Biol. Rep. 2013, 40, 905–916. [Google Scholar] [CrossRef] [PubMed]
- Dhar, N.; Rana, S.; Razdan, S.; Bhat, W.W.; Hussain, A.; Dhar, R.S.; Vaishnavi, S.; Hamid, A.; Vishwakarma, R.; Lattoo, S.K. Cloning and functional characterization of three branch point oxidosqualene cyclases from Withania somnifera (L.) Dunal. J. Biol. Chem. 2014, 289, 17249–17267. [Google Scholar] [CrossRef]
- Patel, N.; Patel, P.; Kendurkar, S.V.; Thulasiram, H.V.; Khan, B.M. Overexpression of squalene synthase in Withania somnifera leads to enhanced withanolide biosynthesis. Plant. Cell. Tissue Organ. Cult. 2015, 122, 409–420. [Google Scholar] [CrossRef]
- Rohmer, M. Mevalonate-independent methylerythritol phosphate pathway for isoprenoid biosynthesis. Elucidation and distribution. Pure Appl. Chem. 2003, 75, 375–387. [Google Scholar] [CrossRef]
- Bhat, W.W.; Lattoo, S.K.; Razdan, S.; Dhar, N.; Rana, S.; Dhar, R.S.; Khan, S.; Vishwakarma, R.A. Molecular cloning, bacterial expression and promoter analysis of squalene synthase from Withania somnifera (L.) Dunal. Gene 2012, 499, 25–36. [Google Scholar] [CrossRef]
- Singh, S.; Pal, S.; Shanker, K.; Chanotiya, C.S.; Gupta, M.M.; Dwivedi, U.N.; Shasany, A.K. Sterol partitioning by HMGR and DXR for routing intermediates toward withanolide biosynthesis. Physiol. Plant. 2014, 152, 617–633. [Google Scholar] [CrossRef] [PubMed]
- Thirugnanasambantham, P.; Roy, I.M.; Charles, S.N.; Senthil, K. Ontogenetic assessment of withanolide biogenesis and expression of selected pathway genes in Withania somnifera, a traditional medicinal herb. J. Pharm. Res. 2014, 8, 1344–1351. [Google Scholar]
- Senthil, K.; Wasnik, N.G.; Kim, Y.J.; Yang, D.C. Generation and analysis of expressed sequence tags from leaf and root of Withania somnifera (Ashwgandha). Mol. Biol. Rep. 2010, 37, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.S.; Singh, S.; Pandey, H.; Srivastava, M.; Ray, T.; Soni, S.; Pandey, A.; Shanker, K.; Babu, C.S.V.; Banerjee, S.; et al. Endophytes of Withania somnifera modulate in planta content and the site of withanolide biosynthesis. Sci. Rep. 2018, 8, 5450. [Google Scholar] [CrossRef] [PubMed]
- Mirjalili, M.H.; Moyano, E.; Bonfill, M.; Cusido, R.M.; Palazón, J. Steroidal lactones from Withania somnifera, an ancient plant for novel medicine. Molecules 2009, 14, 2373–2393. [Google Scholar] [CrossRef]
- Yousefian, Z.; Hosseini, B.; Rezadoost, H.; Palazón, J.; Mirjalili, M.H. Production of the anticancer compound withaferin a from genetically transformed hairy root cultures of Withania somnifera. Nat. Prod. Commun. 2018, 13, 943–948. [Google Scholar] [CrossRef]
- Kabir, M.T.; Uddin, M.S.; Jeandet, P.; Emran, T.B.; Mitra, S.; Albadrani, G.M.; Sayed, A.A.; Abdel-Daim, M.M.; Simal-Gandara, J. Anti-Alzheimer’s Molecules Derived from Marine Life: Understanding Molecular Mechanisms and Therapeutic Potential. Mar. Drugs 2021, 19, 251. [Google Scholar] [CrossRef]
- Holmes, C.; Cotterell, D. Role of Infection in the Pathogenesis of Alzheimer’s Disease. CNS Drugs 2009, 23, 993–1002. [Google Scholar] [CrossRef]
- Eikelenboom, P.; Van Exel, E.; Hoozemans, J.J.M.; Veerhuis, R.; Rozemuller, A.J.M.; Van Gool, W.A. Neuroinflammation-An early event in both the history and pathogenesis of Alzheimer’s disease. Neurodegener. Dis. 2010, 7, 38–41. [Google Scholar] [CrossRef]
- Agostinho, P.; A Cunha, R.; Oliveira, C. Neuroinflammation, Oxidative Stress and the Pathogenesis of Alzheimer ’ s Disease. Curr. Pharm. Des. 2010, 16, 2766–2778. [Google Scholar] [CrossRef]
- Holmes, C. Inflammation in Alzheimer’s Disease; Taylor & Francis (CRC Press): Boca Raton, FL, USA, 2017; Volume 21, ISBN 9781498703116. [Google Scholar]
- Chakrabarti, S.; Khemka, V.K.; Banerjee, A.; Chatterjee, G.; Ganguly, A.; Biswas, A. Metabolic risk factors of sporadic Alzheimer’s disease: Implications in the pathology, pathogenesis and treatment. Aging Dis. 2015, 6, 282–299. [Google Scholar] [CrossRef]
- Mannangatti, P.; Naidu, K.N. Indian herbs for the treatment of neurodegenerative disease. Adv. Neurobiol. 2016, 12, 323–336. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.; Selkoe, D.J. The amyloid hypothesis of Alzheimer’s disease: Progress and problems on the road to therapeutics. Science 2002, 297, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.Z.; Rana, M.S.; Hossain, S.; Dutta, E.; Ferdous, S.; Dutta, M.; Emran, T.B. In vivo neuroprotective, antinociceptive, anti-inflammatory potential in Swiss albino mice and in vitro antioxidant and clot lysis activities of fractionated Holigarna longifolia Roxb. bark extract. J. Complement. Integr. Med. 2019, 17, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Scheuner, D.; Eckman, C.; Jensen, M.; Song, X.; Citron, M.; Suzuki, N.; Bird, T.D.; Hardy, J.; Hutton, M.; Kukull, W.; et al. Secreted amyloid β-protein similar to that in the senile plaques of Alzheimer’s disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer’s disease. Nat. Med. 1996, 2, 864–870. [Google Scholar] [CrossRef]
- Di Carlo, M.; Giacomazza, D.; San Biagio, P.L. Alzheimers disease: Biological aspects, therapeutic perspectives and diagnostic tools. J. Phys. Condens. Matter 2012, 24. [Google Scholar] [CrossRef]
- Sajjad, R.; Arif, R.; Shah, A.A.; Manzoor, I.; Mustafa, G. Pathogenesis of Alzheimer’s disease: Role of amyloid-β and hyperphosphorylated tau protein. Indian J. Pharm. Sci. 2018, 80, 581–591. [Google Scholar] [CrossRef]
- Parihar, M.S.; Hemnani, T. Alzheimer’s disease pathogenesis and therapeutic interventions. J. Clin. Neurosci. 2004, 11, 456–467. [Google Scholar] [CrossRef]
- Carr, D.B.; Goate, A.; Phil, D.; Morris, J.C. Current concepts in the pathogenesis of Alzheimer’s disease. Am. J. Med. 1997, 103, 3S–10S. [Google Scholar] [CrossRef]
- Morgan, B.P. Complement in the pathogenesis of Alzheimer’s disease. Semin. Immunopathol. 2018, 40, 113–124. [Google Scholar] [CrossRef]
- Osborn, L.M.; Kamphuis, W.; Wadman, W.J.; Hol, E.M. Astrogliosis: An integral player in the pathogenesis of Alzheimer’s disease. Prog. Neurobiol. 2016, 144, 121–141. [Google Scholar] [CrossRef] [PubMed]
- Suzhen, D.; Yale, D.; Feng, G.; Yinghe, H.; Zheng, Z. Advances in the pathogenesis of Alzheimer’s disease: A re-evaluation of amyloid cascade hypothesis. Transl. Neurodegener. 2012, 1, 18. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Singh, A. Ekavali A review on Alzheimer’s disease pathophysiology and its management: An update. Pharmacol. Rep. 2015, 67, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.W.; Thompson, R.; Zhang, H.; Xu, H. APP processing in Alzheimer’s disease. Mol. Brain 2011, 4, 3. [Google Scholar] [CrossRef]
- Yesmin, S.; Paul, A.; Naz, T.; Rahman, A.B.M.A.; Akhter, S.F.; Wahed, M.I.I.; Emran, T.B.; Siddiqui, S.A. Membrane stabilization as a mechanism of the anti-inflammatory activity of ethanolic root extract of Choi (Piper chaba). Clin. Phytosci. 2020, 6, 59. [Google Scholar] [CrossRef]
- Polis, B.; Samson, A.O. A New Perspective on Alzheimer’s Disease as a Brain Expression of a Complex Metabolic Disorder. Alzheimer’s Dis. 2019, 1–22. [Google Scholar] [CrossRef][Green Version]
- Kim, D.H.; Yeo, S.H.; Park, J.M.; Choi, J.Y.; Lee, T.H.; Park, S.Y.; Ock, M.S.; Eo, J.; Kim, H.S.; Cha, H.J. Genetic markers for diagnosis and pathogenesis of Alzheimer’s disease. Gene 2014, 545, 185–193. [Google Scholar] [CrossRef]
- Hutton, M.; Hardy, J. The presenilins and Alzheimer’s disease. Hum. Mol. Genet. 1997, 6, 1639–1646. [Google Scholar] [CrossRef]
- Roberts, G.W.; Gentleman, S.M.; Lynch, A.; Murray, L.; Landon, M.; Graham, D.I. β3 Amyloid protein deposition in the brain after severe head injury: Implications for the pathogenesis of Alzheimer’s disease. J. Neurol. Neurosurg. Psychiatry 1994, 57, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Jarrett, J.T.; Berger, E.P.; Lansbury, P.T. The Carboxy Terminus of the β Amyloid Protein Is Critical for the Seeding of Amyloid Formation: Implications for the Pathogenesis of Alzheimer’s Disease. Biochemistry 1993, 32, 4693–4697. [Google Scholar] [CrossRef]
- Bibl, M.; Mollenhauer, B.; Lewczuk, P.; Esselmann, H.; Wolf, S.; Trenkwalder, C.; Otto, M.; Stiens, G.; Rüther, E.; Kornhuber, J.; et al. Validation of amyloid-β peptides in CSF diagnosis of neurodegenerative dementias. Mol. Psychiatry 2007, 12, 671–680. [Google Scholar] [CrossRef] [PubMed]
- Iwatsubo, T.; Odaka, A.; Suzuki, N.; Mizusawa, H.; Nukina, N.; Ihara, Y. Visualization of Aβ42(43) and Aβ40 in senile plaques with end-specific Aβ monoclonals: Evidence that an initially deposited species is Aβ42(43). Neuron 1994, 13, 45–53. [Google Scholar] [CrossRef]
- Chakraborty, A.J.; Mitra, S.; Tallei, T.E.; Tareq, A.M.; Nainu, F.; Cicia, D.; Dhama, K.; Simal-Gandara, J.; Emran, T.B.; Capasso, R. Bromelain a Potential Bioactive Compound: A Comprehensive Overview from a Pharmacological Perspective. Life 2021, 11, 317. [Google Scholar] [CrossRef]
- Jan, A.; Gokce, O.; Luthi-Carter, R.; Lashuel, H.A. The ratio of monomeric to aggregated forms of Aβ40 and Aβ42 is an important determinant of amyloid-β aggregation, fibrillogenesis, and toxicity. J. Biol. Chem. 2008, 283, 28176–28189. [Google Scholar] [CrossRef]
- Tang, W.; Huang, Q.; Wang, Y.; Wang, Z.Y.; Yao, Y.Y. Assessment of CSF Aβ42 as an aid to discriminating Alzheimer’s disease from other dementias and mild cognitive impairment: A meta-analysis of 50 studies. J. Neurol. Sci. 2014, 345, 26–36. [Google Scholar] [CrossRef]
- Jana, M.; Palencia, C.A.; Pahan, K. Fibrillar Amyloid-β Peptides Activate Microglia via TLR2: Implications for Alzheimer’s Disease. J. Immunol. 2008, 181, 7254–7262. [Google Scholar] [CrossRef]
- Reed-Geaghan, E.G.; Savage, J.C.; Hise, A.G.; Landreth, G.E. CD14 and toll-like receptors 2 and 4 are required for fibrillar Aβ-stimulated microglial activation. J. Neurosci. 2009, 29, 11982–11992. [Google Scholar] [CrossRef]
- Stewart, C.R.; Stuart, L.M.; Wilkinson, K.; Van Gils, J.M.; Deng, J.; Halle, A.; Rayner, K.J.; Boyer, L.; Zhong, R.; Frazier, W.A.; et al. CD36 ligands promote sterile inflammation through assembly of a Toll-like receptor 4 and 6 heterodimer. Nat. Immunol. 2010, 11, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, S.; Baglietto-Vargas, D.; Caballero, C.; Moreno-Gonzalez, I.; Torres, M.; Sanchez-Varo, R.; Ruano, D.; Vizuete, M.; Gutierrez, A.; Vitorica, J. Inflammatory response in the hippocampus of PS1M146L/APP 751SL mouse model of Alzheimer’s disease: Age-dependent switch in the microglial phenotype from alternative to classic. J. Neurosci. 2008, 28, 11650–11661. [Google Scholar] [CrossRef]
- Heneka, M.T.; O’Banion, M.K.; Terwel, D.; Kummer, M.P. Neuroinflammatory processes in Alzheimer’s disease. J. Neural Transm. 2010, 117, 919–947. [Google Scholar] [CrossRef]
- Wang, H.; Lin, S.; Allen, J.P.; Williams, J.C.; Blankert, S.; Laser, C.; Woodbury, N.W. Reducing endogenous tau ameliorates amyloid beta-induced deficits in an Alzheimer’s disease mouse model. Science 2007, 316, 750–754. [Google Scholar]
- Neve, R.L.; Harris, P.; Kosik, K.S.; Kurnit, D.M.; Donlon, T.A. Identification of cDNA clones for the human microtubule-associated protein tau and chromosomal localization of the genes for tau and microtubule-associated protein 2. Mol. Brain Res. 1986, 1, 271–280. [Google Scholar] [CrossRef]
- Rakib, A.; Ahmed, S.; Islam, M.A.; Haye, A.; Uddin, S.N.; Uddin, M.M.N.; Hossain, M.K.; Paul, A.; Emran, T.B. Antipyretic and hepatoprotective potential of Tinospora crispa and investigation of possible lead compounds through in silico approaches. Food Sci. Nutr. 2020, 8, 547–556. [Google Scholar] [CrossRef]
- Wischik, C.M.; Novak, M.; Edwards, P.C.; Klug, A.; Tichelaar, W.; Crowther, R.A. Structural characterization of the core of the paired helical filament of Alzheimer disease. Proc. Natl. Acad. Sci. USA 1988, 85, 4884–4888. [Google Scholar] [CrossRef]
- Duan, Y.; Dong, S.; Gu, F.; Hu, Y.; Zhao, Z. Advances in the Pathogenesis of Alzheimer ’ s Disease: Focusing on Tau-Mediated Neurodegeneration. Transl. Neurodegener. 2012, 1, 24. [Google Scholar] [CrossRef]
- Grundke-Iqbal, I.; Iqbal, K.; Tung, Y.C.; Quinlan, M.; Wisniewski, H.M.; Binder, L.I. Abnormal phosphorylation of the microtubule-associated protein tau (tau) in Alzheimer cytoskeletal pathology. Proc. Natl. Acad. Sci. USA 1986, 83, 4913–4917. [Google Scholar] [CrossRef]
- Jara, C.; Aránguiz, A.; Cerpa, W.; Tapia-Rojas, C.; Quintanilla, R.A. Genetic ablation of tau improves mitochondrial function and cognitive abilities in the hippocampus. Redox Biol. 2018, 18, 279–294. [Google Scholar] [CrossRef]
- Braak, H.; Alafuzoff, I.; Arzberger, T.; Kretzschmar, H.; Tredici, K. Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathol. 2006, 112, 389–404. [Google Scholar] [CrossRef] [PubMed]
- Mocanu, M.M.; Nissen, A.; Eckermann, K.; Khlistunova, I.; Biernat, J.; Drexler, D.; Petrova, O.; Schönig, K.; Bujard, H.; Mandelkow, E.; et al. The potential for β-structure in the repeat domain of tau protein determines aggregation, synaptic decay, neuronal loss, and coassembly with endogenous Tau in inducible mouse models of tauopathy. J. Neurosci. 2008, 28, 737–748. [Google Scholar] [CrossRef]
- Mitra, S.; Rauf, A.; Tareq, A.M.; Jahan, S.; Emran, T.B.; Shahriar, T.G.; Dhama, K.; Alhumaydhi, F.A.; Aljohani, A.S.M.; Rebezov, M.; et al. Potential health benefits of carotenoid lutein: An updated reviews. Food Chem. Toxicol. 2021, 154, 112328. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Su, B.; Zheng, L.; Perry, G.; Smith, M.A.; Zhu, X. The role of abnormal mitochondrial dynamics in the pathogenesis of Alzheimer’s disease. J. Neurochem. 2009, 109, 153–159. [Google Scholar] [CrossRef] [PubMed]
- Axelsen, P.H.; Komatsu, H.; Murray, I.V.J. Oxidative stress and cell membranes in the pathogenesis of Alzheimer’s disease. Physiology 2011, 26, 54–69. [Google Scholar] [CrossRef]
- Bezprozvanny, I.; Mattson, M.P. Neuronal Calcium Mishandling and the Pathogenesis of Alzheimer’s Disease. Trends Neurosci. 2009, 31, 454–463. [Google Scholar] [CrossRef]
- Eskici, G.; Axelsen, P.H. Copper and oxidative stress in the pathogenesis of Alzheimer’s disease. Biochemistry 2012, 51, 6289–6311. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kumar, Y. Nature’s Derivative(s) as Alternative Anti-Alzheimer’s Disease Treatments. J. Alzheimer’s Dis. Rep. 2019, 3, 279–297. [Google Scholar] [CrossRef] [PubMed]
- Iova, A.; Micle, O.; Vicaş, L.; Micle, L.; Iova, S.; Mureşan, M.; Ioniţǎ, C.A. Oxidative stress in Alzheimer’s dementia. Farmacia 2014, 62, 538–546. [Google Scholar]
- Houghton, P.J.; Howes, M.J. Natural products and derivatives affecting neurotransmission relevant to Alzheimer’s and Parkinson’s disease. Neurosignals 2005, 14, 6–22. [Google Scholar] [CrossRef]
- Guerreiro, R.; Wojtas, A.; Bras, J.; Carrasquillo, M.; Rogaeva, E.; Majounie, E.; Cruchaga, C.; Sassi, C.; Kauwe, J.S.K.; Younkin, S.; et al. TREM2 Variants in Alzheimer’s Disease. N. Engl. J. Med. 2013, 368, 117–127. [Google Scholar] [CrossRef]
- Heneka, M.T. Neuroinfl ammation in Alzheimer’s disease Michael. Lancet 2015, 14, 388–405. [Google Scholar] [CrossRef]
- Tran, L.; Melquist, S.; Fluder, E.; Zhu, J.; Gudnason, V.; Stone, D.J.; Zhang, B.; Lamb, J.R.; MacDonald, M.E.; Narayanan, M.; et al. Integrated systems approach identifies genetic nodes and networks in late-onset Alzheimer’s disease. Cell 2013, 153, 707–720. [Google Scholar]
- Bradshaw, E.M.; Chibnik, L.B.; Keenan, B.T.; Ottoboni, L.; Raj, T.; Tang, A.; Rosenkrantz, L.L.; Imboywa, S.; Lee, M.; Von Korff, A.; et al. CD33 Alzheimer’s disease locus: Altered monocyte function and amyloid biology. Nat. Neurosci. 2013, 16, 848–850. [Google Scholar] [CrossRef] [PubMed]
- Griciuc, A.; Serrano-Pozo, A.; Parrado, A.R.; Lesinski, A.N.; Asselin, C.N.; Mullin, K.; Hooli, B.; Choi, S.H.; Hyman, B.T.; Tanzi, R.E. Alzheimer’s disease risk gene cd33 inhibits microglial uptake of amyloid beta. Neuron 2013, 78, 631–643. [Google Scholar] [CrossRef]
- Bamberger, M.E.; Harris, M.E.; McDonald, D.R.; Husemann, J.; Landreth, G.E. A cell surface receptor complex for fibrillar β-amyloid mediates microglial activation. J. Neurosci. 2003, 23, 2665–2674. [Google Scholar] [CrossRef] [PubMed]
- Banu, N.; Alam, N.; Islam, M.N.; Islam, S.; Sakib, S.A.; Hanif, N.B.; Chowdhury, M.R.; Tareq, A.M.; Chowdhury, K.H.; Jahan, S.; et al. Insightful Valorization of the Biological Activities of Pani Heloch Leaves through Experimental and Computer-Aided Mechanisms. Molecules 2020, 25, 5153. [Google Scholar] [CrossRef]
- El Khoury, J.B.; Moore, K.J.; Means, T.K.; Leung, J.; Terada, K.; Toft, M.; Freeman, M.W.; Luster, A.D. CD36 mediates the innate host response to β-amyloid. J. Exp. Med. 2003, 197, 1657–1666. [Google Scholar] [CrossRef] [PubMed]
- Jyoti, M.A.; Barua, N.; Hossain, M.S.; Hoque, M.; Bristy, T.A.; Mahmud, S.; Kamruzzaman; Adnan, M.; Chy, M.N.U.; Paul, A.; et al. Unravelling the biological activities of the Byttneria pilosa leaves using experimental and computational approaches. Molecules 2020, 25, 4737. [Google Scholar] [CrossRef] [PubMed]
- Sheedy, F.J.; Grebe, A.; Rayner, K.J.; Kalantari, P.; Ramkhelawon, B.; Carpenter, S.B.; Becker, C.E.; Ediriweera, H.N.; Mullick, A.E.; Golenbock, D.T.; et al. CD36 coordinates NLRP3 inflammasome activation by facilitating intracellular nucleation of soluble ligands into particulate ligands in sterile inflammation. Nat. Immunol. 2013, 14, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, H.; Barger, S.; Barnum, S.; Bradt, B.; Bauer, J.; Cole, G.M.; Cooper, N.R.; Eikelenboom, P.; Emmerling, M.; Fiebich, B.L.; et al. Inflammation and Alzheimer ’s Disease-Neuroinflammation Working Group; Frontiers Media SA: Lausanne, Switzerland, 2000; Volume 21, ISBN 1623876532. [Google Scholar]
- Cunningham, C. Microglia and neurodegeneration: The role of systemic inflammation. Glia 2013, 61, 71–90. [Google Scholar] [CrossRef] [PubMed]
- Gold, M.; El Khoury, J. β-amyloid, microglia, and the inflammasome in Alzheimer’s disease. Semin. Immunopathol. 2015, 37, 607–611. [Google Scholar] [CrossRef]
- Cho, S.; Kim, Y.; Cruz, M.O.; Park, E.M.; Chu, C.K.; Song, G.Y.; Joh, T.H. Repression of proinflammatory cytokine and inducible nitric oxide synthase (NOS2) gene expression in activated microglia by N-acetyl-O-methyldopamine: Protein kinase A-Dependent mechanism. Glia 2001, 33, 324–333. [Google Scholar] [CrossRef]
- Yates, S.L.; Burgess, L.H.; Kocsis-Angle, J.; Antal, J.M.; Dority, M.D.; Embury, P.B.; Piotrkowski, A.M.; Brunden, K.R. Amyloid β and amylin fibrils induce increases in proinflammatory cytokine and chemokine production by THP-1 cells and murine microglia. J. Neurochem. 2000, 74, 1017–1025. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T.; Kummer, M.P.; Stutz, A.; Delekate, A.; Saecker, A.; Griep, A.; Axt, D.; Remus, A.; Tzeng, T.; Gelpi, E.; et al. NLRP3 is activated in Alzheimer’s disease and contributes to pathology in APP/PS1 mice. Nature 2013, 493, 674–678. [Google Scholar] [CrossRef] [PubMed]
- Hebert, L.E.; Scherr, P.A.; Bienias, J.L.; Bennett, D.A.; Evans, D.A. Alzheimer disease in the US population: Prevalence estimates using the 2000 census. Arch. Neurol. 2003, 60, 1119–1122. [Google Scholar] [CrossRef] [PubMed]
- Madmoli, M.; Kord, Z.; Bandani, A.; Sedighi, N.; Shandiz, M.R.; Darabiyan, P.; AfsharNia, A. Epidemiological and clinical study of patients with Alzheimer’s in Five Cities of Khuzestan Province in 2016–2018. Med. Sci. 2019, 23, 1–5. [Google Scholar]
- Nussbaum, R.L.; Ellis, C.E. Alzheimer’s Disease and Parkinson’s Disease. N. Engl. J. Med. 2003, 348, 2588. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.P.; Ding, H.L. Alzheimer’s disease: Epidemiology, genetics, and beyond. Neurosci. Bull. 2008, 24, 105–109. [Google Scholar] [CrossRef]
- Hall, K.; Murrell, J.; Ogunniyi, A.; Deeg, M.; Baiyewu, O.; Gao, S.; Gureje, O.; Dickens, J.; Evans, R.; Smith-Gamble, V.; et al. Cholesterol, APOE genotype, and Alzheimer disease: An epidemiologic study of Nigerian Yoruba. Neurology 2006, 66, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Barua, N.; Aziz, M.A.I.; Tareq, A.M.; Sayeed, M.A.; Alam, N.; ul Alam, N.; Uddin, M.A.; Lyzu, C.; Emran, T.B. In vivo and in vitro evaluation of pharmacological activities of Adenia trilobata (Roxb.). Biochem. Biophys. Rep. 2020, 23, 100772. [Google Scholar] [CrossRef]
- Singh, N.; Bhalla, M.; De Jager, P.; Gilca, M. An Overview on Ashwagandha: A Rasayana (Rejuvenator) of Ayurveda. Afr. J. Tradit. Complement. Altern. Med. 2011, 8. [Google Scholar] [CrossRef]
- Al Mahmud, Z.; Emran, T.B.; Qais, N.; Bachar, S.C.; Sarker, M.; Uddin, M.M.N. Evaluation of analgesic, anti-inflammatory, thrombolytic and hepatoprotective activities of roots of Premna esculenta (Roxb). J Basic Clin. Physiol. Pharmacol. 2016, 27, 63–70. [Google Scholar] [CrossRef]
- McGeer, E.G.; McGeer, P.L. Innate Immunity in Alzheimer’s Disease. Mol. Interv. 2001, 1, 22–29. [Google Scholar] [PubMed]
- Hoshino, T.; Murao, N.; Namba, T.; Takehara, M.; Adachi, H.; Katsuno, M.; Sobue, G.; Matsushima, T.; Suzuki, T.; Mizushima, T. Suppression of Alzheimer’s disease-related phenotypes by expression of heat shock protein 70 in mice. J. Neurosci. 2011, 31, 5225–5234. [Google Scholar] [CrossRef] [PubMed]
- Martorana, F.; Guidotti, G.; Brambilla, L.; Rossi, D. Withaferin A Inhibits Nuclear Factor- B-Dependent Pro-Inflammatory and Stress Response Pathways in the Astrocytes. Neural Plast. 2015, 2015. [Google Scholar] [CrossRef]
- Blair, L.J.; Nordhues, B.A.; Hill, S.E.; Scaglione, K.M.; O’Leary, J.C.; Fontaine, S.N.; Breydo, L.; Zhang, B.; Li, P.; Wang, L.; et al. Accelerated neurodegeneration through chaperone-mediated oligomerization of tau. J. Clin. Investig. 2013, 123, 4158–4169. [Google Scholar] [CrossRef] [PubMed]
- Ansar, S.; Burlison, J.A.; Hadden, M.K.; Yu, X.M.; Desino, K.E.; Bean, J.; Neckers, L.; Audus, K.L.; Michaelis, M.L.; Blagg, B.S.J. A non-toxic Hsp90 inhibitor protects neurons from Aβ-induced toxicity. Bioorg. Med. Chem. Lett. 2007, 17, 1984–1990. [Google Scholar] [CrossRef]
- Sinadinos, C.; Quraishe, S.; Sealey, M.; Samson, P.B.; Mudher, A.; Wyttenbach, A. Low endogenous and chemical induced heat shock protein induction in a 0N3Rtau-Expressing drosophila larval model of Alzheimer’s disease. J. Alzheimer’s Dis. 2013, 33, 1117–1133. [Google Scholar] [CrossRef]
- Blair, L.J.; Sabbagh, J.J.; Dickey, C.A. Targeting Hsp90 and its co-chaperones to treat Alzheimer’s disease. Expert Opin. Ther. Targets 2014, 18, 1219–1232. [Google Scholar] [CrossRef]
- Nile, S.H.; Nile, A.; Gansukh, E.; Baskar, V.; Kai, G. Subcritical water extraction of withanosides and withanolides from ashwagandha (Withania somnifera L.) and their biological activities. Food Chem. Toxicol. 2019, 132. [Google Scholar] [CrossRef] [PubMed]
- Sandhir, R.; Sood, A. Neuroprotective potential of Withania somnifera (ashwagandha) in neurological conditions. Sci. Ashwagandha Prev. Ther. Potentials 2017, 373–387. [Google Scholar] [CrossRef]
- Sun, G.Y.; Li, R.; Cui, J.; Hannink, M.; Gu, Z.; Fritsche, K.L.; Lubahn, D.B.; Simonyi, A. Withania somnifera and Its Withanolides Attenuate Oxidative and Inflammatory Responses and Up-Regulate Antioxidant Responses in BV-2 Microglial Cells. Neuromol. Med. 2016, 18, 241–252. [Google Scholar] [CrossRef]
- Ahmer, S.; Khan, S.A. Alzheimer’s disease in perspective of Unani system of medicine. Int. Hum. Resour. J. 2015, 3, 2347–7067. [Google Scholar]
- Tareq, A.M.; Farhad, S.; Uddin, A.N.; Hoque, M.; Nasrin, M.S.; Uddin, M.M.R.; Hasan, M.; Sultana, A.; Munira, M.S.; Lyzu, C. Chemical profiles, pharmacological properties, and in silico studies provide new insights on Cycas pectinata. Heliyon 2020, 6, e04061. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Rakib, A.; Islam, M.A.; Khanam, B.H.; Faiz, F.B.; Paul, A.; Chy, M.N.U.; Bhuiya, N.M.A.; Uddin, M.M.N.; Ullah, S.A. In vivo and in vitro pharmacological activities of Tacca integrifolia rhizome and investigation of possible lead compounds against breast cancer through in silico approaches. Clin. Phytosci. 2019, 5, 36. [Google Scholar] [CrossRef]
- Heyninck, K.; Lahtela-Kakkonen, M.; Van Der Veken, P.; Haegeman, G.; Berghe, W. Vanden Withaferin A inhibits NF-kappaB activation by targeting cysteine 179 in IKKβ. Biochem. Pharmacol. 2014, 91, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Wallings, R.L.; Tansey, M.G. LRRK2 regulation of immune-pathways and inflammatory disease. Biochem. Soc. Trans. 2019, 47, 1581–1595. [Google Scholar] [CrossRef]
- Narayan, M.; Zhang, J.; Braswell, K.; Gibson, C.; Zitnyar, A.; Lee, D.; Varghese-Gupta, S.; Jinwal, U. Withaferin A Regulates LRRK2 Levels by Interfering with the Hsp90- Cdc37 Chaperone Complex. Curr. Aging Sci. 2015, 8, 259–265. [Google Scholar] [CrossRef]
- Al Mahmud, Z.; Qais, N.; Bachar, S.C.; Hasan, C.M.; Emran, T.B.; Uddin, M.M.N. Phytochemical investigations and antioxidant potential of leaf of Leea macrophylla (Roxb.). BMC Res. Notes 2017, 10, 245. [Google Scholar] [CrossRef]
- Dembitsky, V.M.; Dzhemileva, L.; Gloriozova, T.; D’yakonov, V. Natural and synthetic drugs used for the treatment of the dementia. Biochem. Biophys. Res. Commun. 2020, 524, 772–783. [Google Scholar] [CrossRef]
- Farooqui, A.A.; Farooqui, T.; Madan, A.; Ong, J.H.J.; Ong, W.Y. Ayurvedic Medicine for the Treatment of Dementia: Mechanistic Aspects. Evid. Based Complement. Altern. Med. 2018, 2018. [Google Scholar] [CrossRef]
- Dutta, T.; Paul, A.; Majumder, M.; Sultan, R.A.; Emran, T.B. Pharmacological evidence for the use of Cissus assamica as a medicinal plant in the management of pain and pyrexia. Biochem. Biophys. Rep. 2020, 21, 100715. [Google Scholar] [CrossRef]
- Batumalaie, K.; Amin, M.A.; Murugan, D.D.; Sattar, M.Z.A.; Abdullah, N.A. Withaferin A protects against palmitic acid-induced endothelial insulin resistance and dysfunction through suppression of oxidative stress and inflammation. Sci. Rep. 2016, 6, 27236. [Google Scholar] [CrossRef]
- Palliyaguru, D.L.; Chartoumpekis, D.V.; Wakabayashi, N.; Skoko, J.J.; Yagishita, Y.; Singh, S.V.; Kensler, T.W. Withaferin A induces Nrf2-dependent protection against liver injury: Role of Keap1-independent mechanisms. Free Radic. Biol. Med. 2016, 101, 116–128. [Google Scholar] [CrossRef] [PubMed]
- Emran, T.B.; Rahman, M.A.; Uddin, M.M.N.; Rahman, M.M.; Uddin, M.Z.; Dash, R.; Layzu, C. Effects of organic extracts and their different fractions of five Bangladeshi plants on in vitro thrombolysis. BMC Complement. Altern. Med. 2015, 15, 128. [Google Scholar] [CrossRef] [PubMed]
- Jang, M.H.; Piao, X.L.; Kim, J.M.; Kwon, S.W.; Park, J.H. Inhibition of cholinesterase and amyloid-&bgr; aggregation by resveratrol oligomers from Vitis amurensis. Phyther. Res. 2008, 22, 544–549. [Google Scholar] [CrossRef]
- Panza, F.; Lozupone, M.; Logroscino, G.; Imbimbo, B.P. A critical appraisal of amyloid-β-targeting therapies for Alzheimer disease. Nat. Rev. Neurol. 2019, 15, 73–88. [Google Scholar] [CrossRef] [PubMed]
- Atluri, V.S.R.; Tiwari, S.; Rodriguez, M.; Kaushik, A.; Yndart, A.; Kolishetti, N.; Yatham, M.; Nair, M. Inhibition of Amyloid-Beta Production, Associated Neuroinflammation, and Histone Deacetylase 2-Mediated Epigenetic Modifications Prevent Neuropathology in Alzheimer’s Disease in vitro Model. Front. Aging Neurosci. 2020, 11, 342. [Google Scholar] [CrossRef] [PubMed]
- Kuboyama, T.; Tohda, C.; Komatsu, K. Effects of Ashwagandha (Roots of Withania somnifera) on neurodegenerative diseases. Biol. Pharm. Bull. 2014, 37, 892–897. [Google Scholar] [CrossRef]
- White, P.T.; Subramanian, C.; Motiwala, H.F.; Cohen, M.S. Natural Withanolides in the Treatment of Chronic Diseases; Springer Nature: Gewerbestrasse, Switzerland, 2016; Volume 928, ISBN 9783319413341. [Google Scholar]
- Roy, A. Role of medicinal plants against Alzheimer’s disease. Int. J. Complement. Altern. Med. 2018, 11. [Google Scholar] [CrossRef]
- Jain, R.; Mathur, K. An insight to curative effects of Ashwagandha (Withania somnifera), an Ayurveda herb. J. Med. Plants 2020, 8, 227–235. [Google Scholar]
- Singh, P.N.; Rao, C. V Ethnopharmacological review of native traditional medicinal plants for brain disorders Kalmegh Project View project Ayurvedic Phytopharmacology View project. Pharmacog. Rev. 2007, 1, 20–28. [Google Scholar]
- Summers, W.K.; Majovski, L.V.; Marsh, G.M.; Tachiki, K.; Kling, A. Oral Tetrahydroaminoacridine in Long-Term Treatment of Senile Dementia, Alzheimer Type. N. Engl. J. Med. 1986, 315, 1241–1245. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J. A Hundred Years of Alzheimer’s Disease Research. Neuron 2006, 52, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Lanctôt, K.L.; Rajaram, R.D.; Herrmann, N. Therapy for Alzheimer’s disease: How effective are current treatments? Ther. Adv. Neurol. Disord. 2009, 2, 163–180. [Google Scholar] [CrossRef] [PubMed]
- Islam, M.N.; Rauf, A.; Fahad, F.I.; Emran, T.B.; Mitra, S.; Olatunde, A.; Shariati, M.A.; Rebezov, M.; Mubarak, M.S. Superoxide dismutase: An updated review on its pharmaceutical and industrial applications. Crit. Rev. Food Sci. Nutr. 2021, 2021, 1–19. [Google Scholar] [CrossRef]
- Bhattacharya, S.K.; Kumar, A.; Ghosal, S. Effects of glycowithanolides from Withania somnifera on an animal model of Alzheimer’s disease and perturbed central cholinergic markers of cognition in rats. Phyther. Res. 1995, 9, 110–113. [Google Scholar] [CrossRef]
- Schliebs, R.; Liebmann, A.; Bhattacharya, S.K.; Kumar, A.; Ghosal, S.; Bigl, V. Systemic administration of defined extracts from Withania somnifera (Indian Ginseng) and Shilajit differentially affects cholinergic but not glutamatergic and gabaergic markers in rat brain. Neurochem. Int. 1997, 30, 181–190. [Google Scholar] [CrossRef]
- Roy, A. A Review on Medicinal Plants for Alzheimers Disease. SciFed J. Herb. Med. 2017, 1, 1–6. [Google Scholar]
- Howes, M.R.; Perry, N.S.L.; Houghton, P.J. Plants with Traditional Uses and Activities, Relevant to the Management of Alzheimer’s Disease and Other Cognitive Disorders. Phytother. Res. Int. J. Devoted Pharmacol. Toxicol. Eval. Nat. Prod. Deriv. 2003, 18, 1–18. [Google Scholar] [CrossRef]
- Rakib, A.; Ahmed, S.; Islam, M.A.; Uddin, M.M.N.; Paul, A.; Chy, M.N.U.; Emran, T.B.; Seidel, V. Pharmacological studies on the antinociceptive, anxiolytic and antidepressant activity of Tinospora crispa. Phytother. Res. 2020, 34, 2978–2984. [Google Scholar] [CrossRef]
- Kalra, R.; Kaushik, N. Withania somnifera (Linn.) Dunal: A Review of Chemical and Pharmacological Diversity; Springer: Dordrecht, The Netherlands, 2017; Volume 16, ISBN 1110101795046. [Google Scholar]
- Bristy, T.A.; Barua, N.; Tareq, A.M.; Sakib, S.A.; Etu, S.T.; Chowdhury, K.H.; Jyoti, M.A.; Aziz, M.; Ibn, A.; Reza, A. Deciphering the pharmacological properties of methanol extract of Psychotria calocarpa leaves by in vivo, in vitro and in silico approaches. Pharmaceuticals 2020, 13, 183. [Google Scholar] [CrossRef]
- Rahman, J.; Tareq, A.M.; Hossain, M.M.; Sakib, S.A.; Islam, M.N.; Uddin, A.B.M.N.; Hoque, M.; Nasrin, M.S.; Ali, M.H.; Caiazzo, E.; et al. Biological evaluation, DFT calculations and molecular docking studies on the antidepressant and cytotoxicity activities of Cycas pectinata Buch.-Ham. Compounds. Pharmaceuticals 2020, 13, 232. [Google Scholar] [CrossRef] [PubMed]
- Sudhir, S.; Budhiraja, R.D.; Miglani, G.P.; Arora, B.; Gupta, L.C.; Garg, K.N. Pharmacological studies on leaves of Withania somnifera. Planta Med. 1986, 1, 61–63. [Google Scholar] [CrossRef]
- Zhou, Z.; Xiang, W.; Jiang, Y.; Tian, N.; Wei, Z.; Wen, X.; Wang, W.; Liao, W.; Xia, X.; Li, Q.; et al. Withaferin A alleviates traumatic brain injury induced secondary brain injury via suppressing apoptosis in endothelia cells and modulating activation in the microglia. Eur. J. Pharmacol. 2020, 874, 172988. [Google Scholar] [CrossRef]
- Kaileh, M.; Vanden Berghe, W.; Heyerick, A.; Horion, J.; Piette, J.; Libert, C.; De Keukeleire, D.; Essawi, T.; Haegeman, G. Withaferin A strongly elicits IκB kinase β hyperphosphorylation concomitant with potent inhibition of its kinase activity. J. Biol. Chem. 2007, 282, 4253–4264. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.H.; Kwon, T.K. Withaferin A inhibits tumor necrosis factor-α-induced expression of cell adhesion molecules by inactivation of Akt and NF-κB in human pulmonary epithelial cells. Int. Immunopharmacol. 2009, 9, 614–619. [Google Scholar] [CrossRef]
- Min, K.J.; Choi, K.; Kwon, T.K. Withaferin A down-regulates lipopolysaccharide-induced cyclooxygenase-2 expression and PGE2 production through the inhibition of STAT1/3 activation in microglial cells. Int. Immunopharmacol. 2011, 11, 1137–1142. [Google Scholar] [CrossRef] [PubMed]
- Grover, A.; Shandilya, A.; Punetha, A.; Bisaria, V.S.; Sundar, D. Inhibition of the NEMO/IKKβ association complex formation, a novel mechanism associated with the NF-κB activation suppression by Withania somnifera’s key metabolite withaferin A. BMC Genom. 2010, 11, S25. [Google Scholar] [CrossRef]
- Purushotham, P.M.; Kim, J.M.; Jo, E.K.; Senthil, K. Withanolides against TLR4-Activated Innate Inflammatory Signalling Pathways: A Comparative Computational and Experimental Study. Phyther. Res. 2017, 31, 152–163. [Google Scholar] [CrossRef]
- Christopher, A.M.L.S.M. Inflammasomes: Mechanism of Action, Role in Disease, and Therapeutics. Physiol. Behav. 2016, 176, 100–106. [Google Scholar] [CrossRef]
- Place, D.E.; Kanneganti, T.-D. Recent Advances In Inflammasome Biology. Physiol. Behav. 2016, 176, 100–106. [Google Scholar] [CrossRef]
- Lamkanfi, M.; Dixit, V.M. Mechanisms and functions of inflammasomes. Cell 2014, 157, 1013–1022. [Google Scholar] [CrossRef]
- Tiruveedi, V.L.; Bale, S.; Khurana, A.; Godugu, C. Withaferin A, a novel compound of Indian ginseng (Withania somnifera), ameliorates Cerulein-induced acute pancreatitis: Possible role of oxidative stress and inflammation. Phyther. Res. 2018, 32, 2586–2596. [Google Scholar] [CrossRef] [PubMed]
- Dubey, S.; Yoon, H.; Cohen, M.S.; Nagarkatti, P.; Nagarkatti, M.; Karan, D. Withaferin a associated differential regulation of inflammatory cytokines. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhang, S.; Xiao, Y.; Zhang, W.; Wu, S.; Qin, T.; Yue, Y.; Qian, W.; Li, L. NLRP3 Inflammasome and Inflammatory Diseases. Oxid. Med. Cell. Longev. 2020, 2020. [Google Scholar] [CrossRef]
- Ngoungoure, F.P.; Owona, B.A. Withaferin A modulates AIM2 inflammasome and caspase-1 expression in THP-1 polarized macrophages. Exp. Cell Res. 2019, 383, 111564. [Google Scholar] [CrossRef]
- Karnitz, L.M.; Felts, S.J. Cdc37 regulation of the kinome: When to hold ’em and when to fold ’em. Sci. STKE 2007, 2007, pe22. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Rammeloo, A.W.; Heikkila, J.J. Withaferin A Induces Proteasome Inhibition, Endoplasmic Reticulum Stress, the Heat Shock Response and Acquisition of Thermotolerance. PLoS ONE 2012, 7, e50547. [Google Scholar] [CrossRef] [PubMed]
- Grover, A.; Shandilya, A.; Agrawal, V.; Pratik, P.; Bhasme, D.; Bisaria, V.S.; Sundar, D. Withaferin A Targets Heat Shock Protein 90 in Pancreatic Cancer Cells. BMC Bioinform. 2011, 12, S30. [Google Scholar] [CrossRef]
- Kumar, S.; Phaneuf, D.; Julien, J.P. Withaferin-A Treatment Alleviates TAR DNA-Binding Protein-43 Pathology and Improves Cognitive Function in a Mouse Model of FTLD. Neurotherapeutics 2020, 15–17. [Google Scholar] [CrossRef]
- Witter, S.; Samoson, A.; Vilu, R.; Witter, R. Screening of Nutraceuticals and Plant Extracts for Inhibition of Amyloid-β Fibrillation. J. Alzheimer’s Dis. 2020, 73, 1003–1012. [Google Scholar] [CrossRef]
- Abu Bakar, M.H.; Azmi, M.N.; Shariff, K.A.; Tan, J.S. Withaferin A Protects Against High-Fat Diet–Induced Obesity Via Attenuation of Oxidative Stress, Inflammation, and Insulin Resistance. Appl. Biochem. Biotechnol. 2019, 188, 241–259. [Google Scholar] [CrossRef] [PubMed]
- Ku, S.K.; Han, M.S.; Bae, J.S. Withaferin A is an inhibitor of endothelial protein C receptor shedding in vitro and in vivo. Food Chem. Toxicol. 2014, 68, 23–29. [Google Scholar] [CrossRef]
- Maitra, R.; Porter, M.A.; Huang, S.; Gilmour, B.P. Inhibition of NFB by the natural product withaferin a in cellular models of cystic fibrosis inflammation. J. Inflamm. 2009, 6. [Google Scholar] [CrossRef] [PubMed]
- Mayola, E.; Gallerne, C.; Esposti, D.D.; Martel, C.; Pervaiz, S.; Larue, L.; Debuire, B.; Lemoine, A.; Brenner, C.; Lemaire, C. Withaferin A induces apoptosis in human melanoma cells through generation of reactive oxygen species and down-regulation of Bcl-2. Apoptosis 2011, 16, 1014–1027. [Google Scholar] [CrossRef]
- Stan, S.D.; Zeng, Y.; Singh, S.V. Ayurvedic medicine constituent withaferin a causes G2 and M phase cell cycle arrest in human breast cancer cells. Nutr. Cancer 2008, 60, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Nagalingam, A.; Kuppusamy, P.; Singh, S.V.; Sharma, D.; Saxena, N.K. Mechanistic elucidation of the antitumor properties of withaferin a in breast cancer. Cancer Res. 2014, 74, 2617–2629. [Google Scholar] [CrossRef]
- Mathur, R.; Gupta, S.K.; Singh, N.; Mathur, S.; Kochupillai, V.; Velpandian, T. Evaluation of the effect of Withania somnifera root extracts on cell cycle and angiogenesis. J. Ethnopharmacol. 2006, 105, 336–341. [Google Scholar] [CrossRef]
- Choi, M.J.; Park, E.J.; Min, K.J.; Park, J.W.; Kwon, T.K. Endoplasmic reticulum stress mediates withaferin A-induced apoptosis in human renal carcinoma cells. Toxicol. In Vitro 2011, 25, 692–698. [Google Scholar] [CrossRef]
- Um, H.J.; Min, K.J.; Kim, D.E.; Kwon, T.K. Withaferin A inhibits JAK/STAT3 signaling and induces apoptosis of human renal carcinoma Caki cells. Biochem. Biophys. Res. Commun. 2012, 427, 24–29. [Google Scholar] [CrossRef]
- Benjumea, D.; Martín-Herrera, D.; Abdala, S.; Gutiérrez-Luis, J.; Quiñones, W.; Cardona, D.; Torres, F.; Echeverri, F. Withanolides from Whitania aristata and their diuretic activity. J. Ethnopharmacol. 2009, 123, 351–355. [Google Scholar] [CrossRef]
- Kim, J.-E.; Jeong, Y.-J.; Choi, J.-A.; Oh, S.-M.; Lee, K.-B.; Lee, J.-Y.; Kang, M.-J.; Park, J.-H. Withaferin A Inhibits Helicobacter pylori-induced Production of IL-1β in Dendritic Cells by Regulating NF-κB and NLRP3 Inflammasome Activation. Immune Netw. 2015, 15, 269–277. [Google Scholar] [CrossRef]
- Dumore, N.G.; Umekar, M.J.; Taksande, B.G.; Aglawe, M.M.; Kotagale, N.R. Effects of Withania somnifera nicotine induced conditioned place preference in mice. Pharmacogn. J. 2019, 11, 43–47. [Google Scholar] [CrossRef]
- Bahbah, E.I.; Ghozy, S.; Attia, M.S.; Negida, A.; Emran, T.B.; Mitra, S.; Albadrani, G.M.; Abdel-Daim, M.M.; Uddin, M.S.; Simal-Gandara, J. Molecular Mechanisms of Astaxanthin as a Potential Neurotherapeutic Agent. Mar. Drugs 2021, 19, 201. [Google Scholar] [CrossRef] [PubMed]
- Jahan, I.; Tona, M.R.; Sharmin, S.; Sayeed, M.A.; Tania, F.Z.; Paul, A.; Chy, M.; Uddin, N.; Rakib, A.; Emran, T.B. GC-MS phytochemical profiling, pharmacological properties, and in silico studies of Chukrasia velutina leaves: A novel source for bioactive agents. Molecules 2020, 25, 3536. [Google Scholar] [CrossRef] [PubMed]
- Tekula, S.; Khurana, A.; Anchi, P.; Godugu, C. Withaferin-A attenuates multiple low doses of Streptozotocin (MLD-STZ) induced type 1 diabetes. Biomed. Pharmacother. 2018, 106, 1428–1440. [Google Scholar] [CrossRef]
- Hassannia, B.; Wiernicki, B.; Ingold, I.; Qu, F.; Van Herck, S.; Tyurina, Y.Y.; Bayir, H.; Abhari, B.A.; Angeli, J.P.F.; Choi, S.M.; et al. Nano-targeted induction of dual ferroptotic mechanisms eradicates high-risk neuroblastoma. J. Clin. Investig. 2018, 128, 3341–3355. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, R.; Rauf, A.; Akhter, S.; Islam, M.N.; Emran, T.B.; Mitra, S.; Khan, I.N.; Mubarak, M.S. Role of Withaferin A and Its Derivatives in the Management of Alzheimer’s Disease: Recent Trends and Future Perspectives. Molecules 2021, 26, 3696. https://doi.org/10.3390/molecules26123696
Das R, Rauf A, Akhter S, Islam MN, Emran TB, Mitra S, Khan IN, Mubarak MS. Role of Withaferin A and Its Derivatives in the Management of Alzheimer’s Disease: Recent Trends and Future Perspectives. Molecules. 2021; 26(12):3696. https://doi.org/10.3390/molecules26123696
Chicago/Turabian StyleDas, Rajib, Abdur Rauf, Saima Akhter, Mohammad Nazmul Islam, Talha Bin Emran, Saikat Mitra, Ishaq N. Khan, and Mohammad S. Mubarak. 2021. "Role of Withaferin A and Its Derivatives in the Management of Alzheimer’s Disease: Recent Trends and Future Perspectives" Molecules 26, no. 12: 3696. https://doi.org/10.3390/molecules26123696
APA StyleDas, R., Rauf, A., Akhter, S., Islam, M. N., Emran, T. B., Mitra, S., Khan, I. N., & Mubarak, M. S. (2021). Role of Withaferin A and Its Derivatives in the Management of Alzheimer’s Disease: Recent Trends and Future Perspectives. Molecules, 26(12), 3696. https://doi.org/10.3390/molecules26123696











