The Neuroprotective Effect of Tea Polyphenols on the Regulation of Intestinal Flora
Abstract
1. Introduction
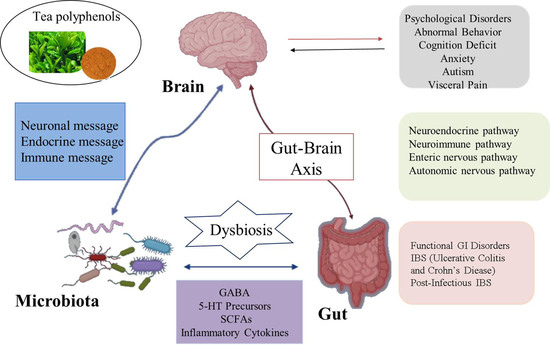
2. The Relationship between Gut Microbiota and Nervous System
2.1. Neural Pathways
2.2. Immune Pathway
2.3. Neuroendocrine Pathway
2.4. Microbial Metabolite Pathway
2.5. Nitric Oxide Pathway
3. Disturbance of Gut Microbiota Leads to Neurodegeneration
4. Mechanisms of the Neuroprotective Effect of TPs
4.1. Antioxidation
4.1.1. Directly Scavenge Active Oxygen Free Radicals and Nitrogen Free Radicals
4.1.2. Inhibition to Oxidase Promotion and Lipid Peroxidation
4.1.3. Activate Intracellular Antioxidant Defense System
4.2. The Activity of the Metal Chelating Agent
4.3. Regulate Signal Pathway
4.3.1. Selectively Activate Protein kinase C (PKC) in Cerebral Neurons
4.3.2. Regulate Other Signal Transduction Pathways
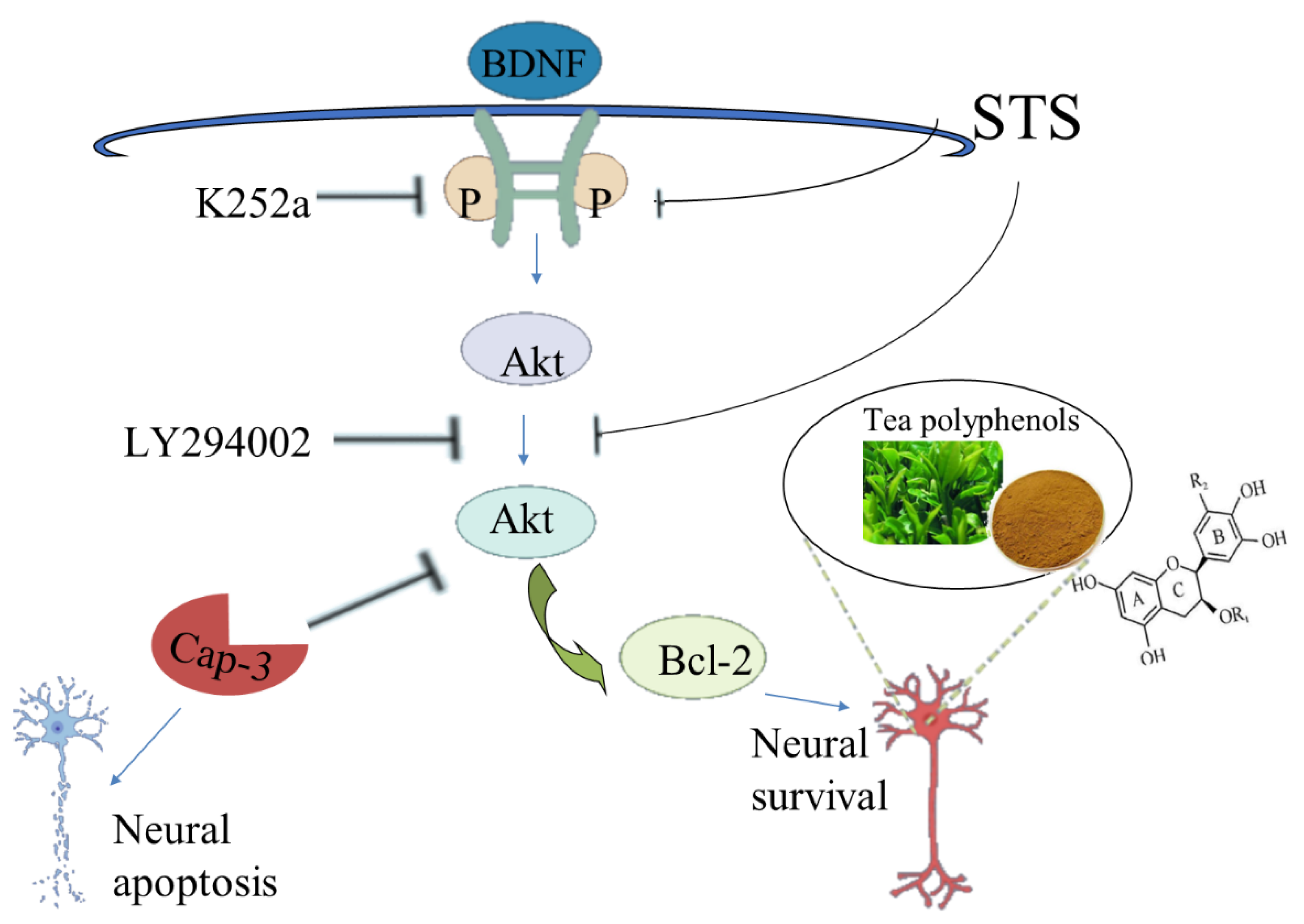
4.3.3. Anti-Apoptosis
4.4. Regulate the Level of Neurotransmitters
4.5. Autophagy
5. The Reciprocal Interactions between TPs and Gut Microbiota Promote Nerve Protection
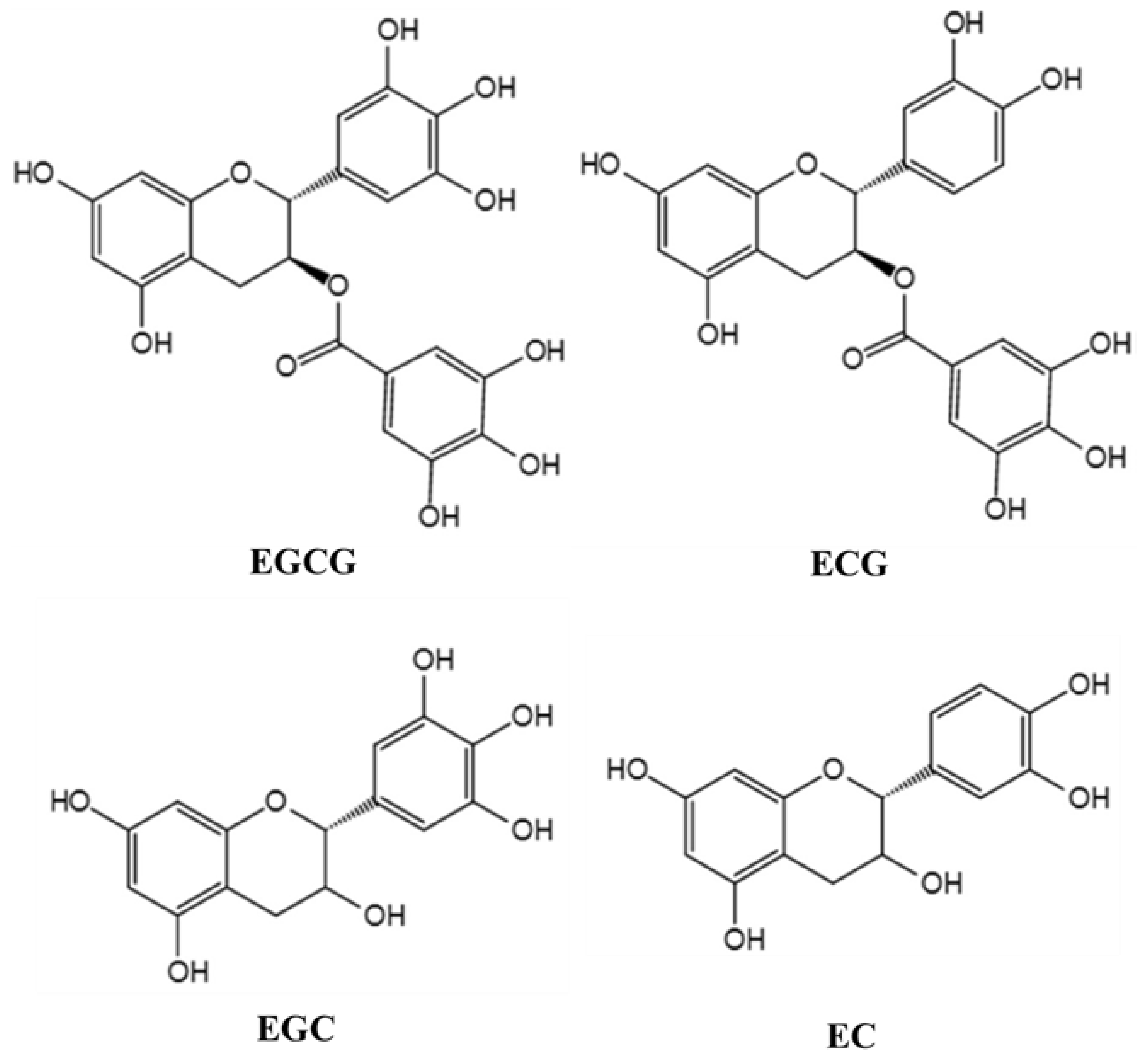
5.1. Biotransformation of TPs by Gut Microbiota
5.2. Regulatory Effect of TPs on Intestinal Flora
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Qin, J.J.; Li, R.Q.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T.; et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59–70. [Google Scholar] [CrossRef]
- Backhed, F.; Ley, R.E.; Sonnenburg, J.L.; Peterson, D.A.; Gordon, J.I. Host-bacterial mutualism in the human intestine. Science 2005, 307, 1915–1920. [Google Scholar] [CrossRef] [PubMed]
- Backhed, F.; Roswall, J.; Peng, Y.Q.; Feng, Q.; Jia, H.J.; Kovatcheva-Datchary, P.; Li, Y.; Xia, Y.; Xie, H.L.; Zhong, H.Z.; et al. Dynamics and stabilization of the human gut microbiome during the first year of life. Cell Host Microbe 2015, 17, 690–703. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Yoo, J.Y.; Dutra, S.V.O.; Morgan, K.H.; Groer, M. The Association between Early-Life Gut Microbiota and Long-Term Health and Diseases. J. Clin. Med. 2021, 10, 459. [Google Scholar] [CrossRef]
- Al-Khafaji, A.H.; Jepsen, S.D.; Christensen, K.R.; Vigsnaes, L.K. The potential of human milk oligosaccharides to impact the microbiota-gut-brain axis through modulation of the gut microbiota. J. Funct. Foods 2020, 74, 104176. [Google Scholar] [CrossRef]
- Saric, S.; Notay, M.; Sivamani, R.K. Green tea and other tea polyphenols: Effects on sebum production and acne vulgaris. Antioxidants 2017, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Qi, R.L.; Mine, Y. The impact of oolong and black tea polyphenols on human health. Food Biosci. 2019, 29, 55–61. [Google Scholar] [CrossRef]
- Molino, S.; Dossena, M.; Buonocore, D.; Ferrari, F.; Venturini, L.; Ricevuti, G.; Verri, M. Polyphenols in dementia: From molecular basis to clinical trials. Life Sci. 2016, 161, 69–77. [Google Scholar] [CrossRef]
- Scholl, C.; Lepper, A.; Steffens, M.; von Mallek, D.; Brockmoller, J.; Stingl, J. Individual variability in the pharmacokinetic of tea polyphenols and gene expression after oral intake of green tea extract. Clin. Ther. 2015, 37, 116. [Google Scholar] [CrossRef]
- Liang, J.; Yan, H.; Puligundla, P.; Gao, X.L.; Zhou, Y.B.; Wan, X.C. Applications of chitosan nanoparticles to enhance absorption and bioavailability of tea polyphenols: A review. Food Hydrocoll. 2017, 69, 286–292. [Google Scholar] [CrossRef]
- Ma, H.; Zhang, B.W.; Hu, Y.Z.; Wang, J.; Liu, J.M.; Qui, R.B.; Lv, S.W.; Wang, S. Correlation analysis of intestinal redox state with the gut microbiota reveals the positive intervention of tea polyphenols on hyperlipidemia in high fat diet fed mice. J. Agric. Food Chem. 2019, 67, 7325–7335. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.D.; Sang, S.M. Biotransformation of tea polyphenols by gut microbiota. J. Funct. Foods 2014, 7, 26–42. [Google Scholar] [CrossRef]
- Ishida, N.; Iizuka, M.; Kataoka, K.; Okazaki, M.; Shiraishi, K.; Yagi, Y.; Jobu, K.; Yokota, J.; Oishi, M.; Moriyama, H.; et al. Improvement of blood lipid profiles by Goishi tea polyphenols in a randomised, double-blind, placebo-controlled clinical study. Int. J. Food Sci. Nutr. 2018, 69, 598–607. [Google Scholar] [CrossRef]
- Li, D.; Zhou, C.R.; Zou, N.; Wu, Y.L.; Zhang, J.B.; An, Q.S.; Li, J.Q.; Pan, C.P. Nanoselenium foliar application enhances biosynthesis of tea leaves in metabolic cycles and associated responsive pathways. Environ. Pollut. 2021, 273, 116503. [Google Scholar] [CrossRef]
- Ronzano, R. Astrocytes and microglia: Active players in synaptic plasticity. Med. Sci. M/S 2017, 33, 1071–1078. [Google Scholar]
- Barres, B.A. The mystery and magic of glia: A perspective on their roles in health and disease. Neuron 2008, 60, 430–440. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Li, X.Y.; Shen, L. Modulation effect of tea consumption on gut microbiota. Appl. Microbiol. Biotechnol. 2020, 104, 981–987. [Google Scholar] [CrossRef]
- Kuwahara, A.; Matsuda, K.; Kuwahara, Y.; Asano, S.; Inui, T.; Marunaka, Y. Microbiota-gut-brain axis: Enteroendocrine cells and the enteric nervous system form an interface between the microbiota and the central nervous system. Biomed Res. 2020, 41, 199–216. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.Q.; Han, Y.; Du, J.; Liu, R.Z.; Jin, K.T.; Yi, W. Microbiota-gut-brain axis and the central nervous system. Oncotarget 2017, 8, 53829–53838. [Google Scholar] [CrossRef] [PubMed]
- Mayer, E.A. Gut feelings: The emerging biology of gut-brain communication. Nat. Rev. Neurosci. 2011, 12, 453–466. [Google Scholar] [CrossRef] [PubMed]
- Mulak, A.; Bonaz, B. Irritable bowel syndrome: A model of the brain-gut interactions. Med. Sci. Monit. 2004, 10, RA55–RA62. [Google Scholar]
- Doifode, T.; Giridharan, V.V.; Generoso, J.S.; Bhatti, G.; Collodel, A.; Schulz, P.E.; Forlenza, O.V.; Barichello, T. The impact of the microbiota-gut-brain axis on Alzheimer’s disease pathophysiology. Pharmacol. Res. 2021, 164, 105314. [Google Scholar] [CrossRef]
- Ohno, M.; Nikaido, M.; Horiuchi, N.; Kawakami, K.; Hatta, K. The enteric nervous system in zebrafish larvae can regenerate via migration into the ablated area and proliferation of neural crest-derived cells. Development 2021, 148, dev195339. [Google Scholar] [PubMed]
- Morais, L.H.; Schreiber, H.L.; Mazmanian, S.K. The gut microbiota-brain axis in behaviour and brain disorders. Nat. Rev. Microbiol. 2021, 19, 241–255. [Google Scholar] [CrossRef] [PubMed]
- Bercik, P.; Park, A.J.; Sinclair, D.; Khoshdel, A.; Lu, J.; Huang, X.; Deng, Y.; Blennerhassett, P.A.; Fahnestock, M.; Moine, D.; et al. The anxiolytic effect of Bifidobacterium longum NCC3001 involves vagal pathways for gut-brain communication. Neurogastroenterol. Motil. 2011, 23, E1132–E1544. [Google Scholar] [CrossRef] [PubMed]
- De Almeida, L.M.V.; Funchal, C.; Gottfried, C.; Wajner, M.; Pessoa-Pureur, R. Propionic acid induces cytoskeletal alterations in cultured astrocytes from rat cerebral cortex. Metab. Brain Dis. 2006, 21, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Lyte, M.; Varcoe, J.J.; Bailey, M.T. Anxiogenic effect of subclinical bacterial infection in mice in the absence of overt immune activation. Physiol. Behav. 1998, 65, 63–68. [Google Scholar] [CrossRef]
- Klarer, M.; Krieger, J.P.; Richetto, J.; Weber-Stadlbauer, U.; Gunther, L.; Winter, C.; Arnold, M.; Langhans, W.; Meyer, U. Abdominal vagal afferents modulate the brain transcriptome and behaviors relevant to Schizophrenia. J. Neurosci. 2018, 38, 1634–1647. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Y.; Yu, Q.Q.; Zhang, X.N.; Wang, X.Y.; Su, Y.S.; He, W.; Li, J.; Wan, H.Y.; Jing, X.H. Electroacupuncture regulates inflammatory cytokines by activating the vagus nerve to enhance antitumor immunity in mice with breast tumors. Life Sci. 2021, 272, 119259. [Google Scholar] [CrossRef]
- Quarta, A.; Meese, T.; Pieters, Z.; Van Breedam, E.; Le Blon, D.; Van Broeckhoven, J.; Hendrix, S.; Goossens, H.; Hens, N.; Berneman, Z.; et al. Murine induced pluripotent stem cell-derived neuroimmune cell culture models emphasize opposite immune-effector functions of interleukin 13-primed microglia and macrophages in terms of neuroimmune toxicity. Glia 2021, 69, 326–345. [Google Scholar] [CrossRef]
- Walter, T.J.; Crews, F.T. Microglial depletion alters the brain neuroimmune response to acute binge ethanol withdrawal. J. Neuroinflamm. 2017, 14, 86. [Google Scholar] [CrossRef] [PubMed]
- Werneburg, S.; Feinberg, P.A.; Johnson, K.M.; Schafer, D.P. A microglia-cytokine axis to modulate synaptic connectivity and function. Curr. Opin. Neurobiol. 2017, 47, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Felix, K.M.; Tahsin, S.; Wu, H.J.J. Host-microbiota interplay in mediating immune disorders. Ann. N. Y. Acad. Sci. 2018, 1417, 57–70. [Google Scholar] [CrossRef] [PubMed]
- Kosiewicz, M.M.; Dryden, G.W.; Chhabra, A.; Alard, P. Relationship between gut microbiota and development of T cell associated disease. FEBS Lett. 2014, 588, 4195–4206. [Google Scholar] [CrossRef] [PubMed]
- Schafer, D.P.; Stevens, B. Microglia function in central nervous system development and plasticity. Cold Spring Harb. Perspect. Biol. 2015, 7, a020545. [Google Scholar] [CrossRef] [PubMed]
- Mossad, O.; Erny, D. The microbiota-microglia axis in central nervous system disorders. Brain Pathol. 2020, 30, 1159–1177. [Google Scholar] [CrossRef]
- Thion, M.S.; Low, D.; Silvin, A.; Chen, J.M.; Grisel, P.; Schulte-Schrepping, J.; Blecher, R.; Ulas, T.; Squarzoni, P.; Hoeffel, G.; et al. Microbiome influences prenatal and adult microglia in a sex-specific manner. Cell 2018, 172, 500–516. [Google Scholar] [CrossRef] [PubMed]
- Rea, K.; Dinan, T.G.; Cryan, J.F. The microbiome: A key regulator of stress and neuroinflammation. Neurobiol. Stress 2016, 4, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Rook, G.A.W.; Raison, C.L.; Lowry, C.A. Microbiota, immunoregulatory old friends and psychiatric disorders. Adv. Exp. Med. Biol. 2014, 817, 319–356. [Google Scholar] [PubMed]
- Miko, E.; Meggyes, M.; Doba, K.; Barakonyi, A.; Szereday, L. Immune checkpoint molecules in reproductive immunology. Front. Immunol. 2019, 10, 846. [Google Scholar] [CrossRef]
- Daulatzai, M.A. Chronic functional bowel syndrome enhances gut-brain axis dysfunction, neuroinflammation, cognitive impairment, and vulnerability to dementia. Neurochem. Res. 2014, 39, 624–644. [Google Scholar] [CrossRef]
- Yaddanapudi, K.; Hornig, M.; Serge, R.; De Miranda, J.; Baghban, A.; Villar, G.; Lipkin, W.I. Passive transfer of streptococcus-induced antibodies reproduces behavioral disturbances in a mouse model of pediatric autoimmune neuropsychiatric disorders associated with streptococcal infection. Mol. Psychiatr. 2010, 15, 712–726. [Google Scholar] [CrossRef]
- Barrett, E.; Ross, R.P.; O’Toole, P.W.; Fitzgerald, G.F.; Stanton, C. γ-Aminobutyric acid production by culturable bacteria from the human intestine. J. Appl. Microbiol. 2012, 113, 411–417. [Google Scholar] [CrossRef]
- Frye, R.E.; Rose, S.; Slattery, J.; MacFabe, D.F. Gastrointestinal dysfunction in autism spectrum disorder: The role of the mitochondria and the enteric microbiome. Microb. Ecol. Health Dis. 2015, 26, 27458. [Google Scholar] [CrossRef]
- Reigstad, C.S.; Salmonson, C.E.; Rainey, J.F.; Szurszewski, J.H.; Linden, D.R.; Sonnenburg, J.L.; Farrugia, G.; Kashyap, P.C. Gut microbes promote colonic serotonin production through an effect of short-chain fatty acids on enterochromaffin cells. FASEB J. 2015, 29, 1395–1403. [Google Scholar] [CrossRef] [PubMed]
- Clarke, G.; Grenham, S.; Scully, P.; Fitzgerald, P.; Moloney, R.D.; Shanahan, F.; Dinan, T.G.; Cryan, J.F. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol. Psychiatr. 2013, 18, 666–673. [Google Scholar] [CrossRef] [PubMed]
- Tremlett, H.; Bauer, K.C.; Appel-Cresswell, S.; Finlay, B.B.; Waubant, E. The gut microbiome in human neurological disease: A review. Ann. Neurol. 2017, 81, 369–382. [Google Scholar] [CrossRef]
- O’Mahony, S.M.; Clarke, G.; Borre, Y.E.; Dinan, T.G.; Cryan, J.F. Serotonin, tryptophan metabolism and the brain-gut-microbiome axis. Behav. Brain Res. 2015, 277, 32–48. [Google Scholar] [CrossRef] [PubMed]
- Round, J.L.; Lee, S.M.; Li, J.; Tran, G.; Jabri, B.; Chatila, T.A.; Mazmanian, S.K. The toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science 2011, 332, 974–977. [Google Scholar] [CrossRef]
- Round, J.L.; Mazmanian, S.K. Inducible Foxp(3+) regulatory T-cell development by a commensal bacterium of the intestinal microbiota. Proc. Natl. Acad. Sci. USA 2010, 107, 12204–12209. [Google Scholar] [CrossRef] [PubMed]
- Pryde, S.E.; Duncan, S.H.; Hold, G.L.; Stewart, C.S.; Flint, H.J. The microbiology of butyrate formation in the human colon. FEMS Microbiol. Lett. 2002, 217, 133–139. [Google Scholar] [CrossRef]
- Montoya, C.A.; Rutherfurd, S.M.; Moughan, P.J. Kiwifruit fibre level influences the predicted production and absorption of SCFA in the hindgut of growing pigs using a combined in vivo-in vitro digestion methodology. Br. J. Nutr. 2016, 115, 1317–1324. [Google Scholar] [CrossRef]
- Wenzel, T.J.; Gates, E.J.; Ranger, A.L.; Klegeris, A. Short-chain fatty acids (SCFAs) alone or in combination regulate select immune functions of microglia-like cells. Mol. Cell. Neurosci. 2020, 105, 103493. [Google Scholar] [CrossRef]
- Harrison, I.F.; Dexter, D.T. Epigenetic targeting of histone deacetylase: Therapeutic potential in Parkinson’s disease? Pharmacol. Ther. 2013, 140, 34–52. [Google Scholar] [CrossRef]
- Onyszkiewicz, M.; Gawrys-Kopczynska, M.; Konopelski, P.; Aleksandrowicz, M.; Sawicka, A.; Kozniewska, E.; Samborowska, E.; Ufnal, M. Butyric acid, a gut bacteria metabolite, lowers arterial blood pressure via colon-vagus nerve signaling and GPR41/43 receptors. Pflugers Arch. 2019, 471, 1441–1453. [Google Scholar] [CrossRef]
- Tse, J.K. Gut Microbiota, Nitric Oxide, and Microglia as Prerequisites for Neurodegenerative Disorders. ACS Chem. Neurosci. 2017, 8, 1438–1447. [Google Scholar] [CrossRef]
- Pereira, C.; Ferreira, N.R.; Rocha, B.S.; Barbosa, R.M.; Laranjinha, J. The redox interplay between nitrite and nitric oxide: From the gut to the brain. Redox Biol. 2013, 1, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Oleskin, A.V.; Shenderov, B.A. Neuromodulatory effects and targets of the SCFAs and gasotransmitters produced by the human symbiotic microbiota. Microb. Ecol. Health Dis. 2016, 27, 30971. [Google Scholar] [CrossRef]
- Calabrese, V.; Mancuso, C.; Calvani, M.; Rizzarelli, E.; Butterfield, D.A.; Stella, A.M. Nitric oxide in the central nervous system: Neuroprotection versus neurotoxicity. Nat. Rev. Neurosci. 2007, 8, 766–775. [Google Scholar] [CrossRef] [PubMed]
- Asiimwe, N.; Yeo, S.G.; Kim, M.S.; Jung, J.; Jeong, N.Y. Nitric Oxide: Exploring the Contextual Link with Alzheimer’s Disease. Oxid. Med. Cell. Longev. 2016, 2016, 7205747. [Google Scholar] [CrossRef] [PubMed]
- Tilocca, B.; Pieroni, L.; Soggiu, A.; Britti, D.; Bonizzi, L.; Roncada, P.; Greco, V. Gut-brain axis and neurodegeneration: State-of-the-art of meta-omics sciences for microbiota characterization. Int. J. Mol. Sci. 2020, 21, 4045. [Google Scholar] [CrossRef] [PubMed]
- Greco, V.; Longone, P.; Spalloni, A.; Pieroni, L.; Urbani, A. Crosstalk between oxidative stress and mauchondrial damage: Focus on amyotrophic lateral sclerosis. In Mitochondria in Health and in Sickness; Springer: Singapore, 2019; Volume 1158, pp. 71–82. [Google Scholar]
- Wang, Y.; Xu, E.; Musich, P.R.; Lin, F. Mitochondrial dysfunction in neurodegenerative diseases and the potential countermeasure. CNS Neurosci. Ther. 2019, 25, 816–824. [Google Scholar] [CrossRef] [PubMed]
- Saint-Georges-Chaumet, Y.; Attaf, D.; Pelletier, E.; Edeas, M. Targeting microbiota-mitochondria inter-talk: Microbiota control mitochondria metabolism. Cell. Mol. Biol. 2015, 61, 121–124. [Google Scholar]
- Claesson, M.J.; Cusack, S.; O’Sullivan, O.; Greene-Diniz, R.; de Weerd, H.; Flannery, E.; Marchesi, J.R.; Falush, D.; Dinan, T.; Fitzgerald, G.; et al. Composition, variability, and temporal stability of the intestinal microbiota of the elderly. Proc. Natl. Acad. Sci. USA 2011, 108, 4586–4591. [Google Scholar] [CrossRef]
- O’Toole, P.W.; Jeffery, I.B. Gut microbiota and aging. Science 2015, 350, 1214–1215. [Google Scholar] [CrossRef]
- Hashimoto, M.; Ho, G.; Takamatsu, Y.; Shimizu, Y.; Sugama, S.; Takenouchi, T.; Waragai, M.; Masliah, E. Evolvability and neurodegenerative disease: Antagonistic pleiotropy phenomena derived from amyloid aggregates. J. Parkinsons Dis. 2018, 8, 405–408. [Google Scholar] [CrossRef] [PubMed]
- Ceppa, F.A.; Izzo, L.; Sardelli, L.; Raimondi, I.; Tunesi, M.; Albani, D.; Giordano, C. Human gut-microbiota interaction in neurodegenerative disorders and current engineered tools for its modeling. Front. Cell. Infect. Microbiol. 2020, 10, 297. [Google Scholar] [CrossRef]
- Go, J.; Sun, I.C.; Ji, E.K.; Lee, Y.J.; Kwak, M.H.; Koh, E.K.; Ji, E.S.; Hwang, D.Y. Effect of reserpine on the behavioral defects, Aβ-42 deposition and NGF metabolism in Tg2576 transgenic mouse model for Alzheimer’s disease. J. Life Sci. 2013, 23, 812–824. [Google Scholar] [CrossRef]
- Choi, S.I.; Go, J.; Kim, J.E.; Lee, Y.J.; Kwak, M.H.; Jung, Y.J.; Hwang, D.Y. Precautionary effects of Red Liriope platyphylla on NGF secretion and Abeta42 deposition under the preclinical stage of Alzheimer’s disease in Tg2576 mice. Lab. Anim. Res. 2013, 29, 212–220. [Google Scholar] [CrossRef]
- Lee, J.K.; Byun, H.G. A novel BACE inhibitor isolated from Eisenia bicyclis exhibits neuroprotective activity against β-amyloid toxicity. Fish. Aquat. Sci. 2018, 21, 1–9. [Google Scholar] [CrossRef]
- Yang, S.H. Cellular and molecular mediators of neuroinflammation in Alzheimer disease. Int. Neurourol. J. 2019, 23, S54–S62. [Google Scholar] [CrossRef]
- Gonzalez, H.; Pacheco, R. T-cell-mediated regulation of neuroinflammation involved in neurodegenerative diseases. J. Neuroinflamm. 2014, 11, 201. [Google Scholar] [CrossRef] [PubMed]
- Markham, A.; Bains, R.; Franklin, P.; Spedding, M. Changes in mitochondrial function are pivotal in neurodegenerative and psychiatric disorders: How important is BDNF? Br. J. Pharmacol. 2014, 171, 2206–2229. [Google Scholar] [CrossRef]
- Belayev, L.; Hong, S.H.; Menghani, H.; Marcell, S.J.; Obenaus, A.; Freitas, R.S.; Khoutorova, L.; Balaszczuk, V.; Jun, B.; Oria, R.B.; et al. Docosanoids promote neurogenesis and angiogenesis, blood-brain barrier integrity, penumbra protection, and neurobehavioral recovery after experimental ischemic stroke. Mol. Neurobiol. 2018, 55, 7090–7106. [Google Scholar] [CrossRef] [PubMed]
- Yahfoufi, N.; Matar, C.; Ismail, N. Adolescence and aging: Impact of adolescence inflammatory stress and microbiota alterations on brain development, aging, and neurodegeneration. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2020, 75, 1251–1257. [Google Scholar] [CrossRef] [PubMed]
- Bhattarai, Y.; Kashyap, P.C. Parkinson’s disease: Are gut microbes involved? Am. J. Physiol. Gastroint. Liver Physiol. 2020, 319, G529–G540. [Google Scholar] [CrossRef]
- Zhu, S.B.; Jiang, Y.F.; Xu, K.L.; Cui, M.; Ye, W.M.; Zhao, G.M.; Jin, L.; Chen, X.D. The progress of gut microbiome research related to brain disorders. J. Neuroinflamm. 2020, 17, 25. [Google Scholar] [CrossRef]
- Sarkar, S.R.; Banerjee, S. Gut microbiota in neurodegenerative disorders. J. Neuroimmunol. 2019, 328, 98–104. [Google Scholar] [CrossRef]
- Fang, X.; Wang, X.; Yang, S.G.; Meng, F.J.; Wang, X.L.; Wei, H.; Chen, T.T. Evaluation of the microbial diversity in amyotrophic lateral sclerosis using high-throughput sequencing. Front. Microbiol. 2016, 7, 1479. [Google Scholar] [CrossRef]
- Brenner, D.; Hiergeist, A.; Adis, C.; Mayer, B.; Gessner, A.; Ludolph, A.C.; Weishaupt, J.H. The fecal microbiome of ALS patients. Neurobiol. Aging 2018, 61, 132–137. [Google Scholar] [CrossRef]
- Leslie, P.; Smithard, D.G. Is dysphagia under diagnosed or is normal swallowing more variable than we think? Reported swallowing problems in people aged 18–65 years. Dysphagia 2020, 2, 1–9. [Google Scholar]
- Vali, L.; Hahn, O.; Kupcsulik, P.; Drahos, A.; Sarvary, E.; Szentmihalyi, K.; Pallai, Z.; Kurucz, T.; Sipos, P.; Blazovics, A. Oxidative stress with altered element content and decreased ATP level of erythrocytes hepatocellular carcinoma and colorectal liver metastases. Eur. J. Gastroenterol. Hepatol. 2008, 20, 393–398. [Google Scholar] [CrossRef]
- Uttara, B.; Singh, A.V.; Zamboni, P.; Mahajan, R.T. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr. Neuropharmacol. 2009, 7, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Tate, P.S.; Marazita, M.C.; Marquioni-Ramella, M.D.; Suburo, A.M. Ilex paraguariensis extracts and its polyphenols prevent oxidative damage and senescence of human retinal pigment epithelium cells. J. Funct. Foods 2020, 67, 103833. [Google Scholar] [CrossRef]
- Amić, D.; Davidović, A.D.; Beslo, D.; Rastija, V.; Lucić, B.; Trinajstić, N. SAR and QSAR of the antioxidant activity of flavonoids. Curr. Med. Chem. 2007, 14, 827–845. [Google Scholar] [CrossRef]
- Sandoval, A.C.; Ferreira, J.; Speisky, H. Polyphenols and mitochondria: An update on their increasingly emerging ROS-scavenging independent actions. Arch. Biochem. Biophys. 2014, 559, 75–90. [Google Scholar] [CrossRef]
- Wei, I.H.; Wu, Y.C.; Wen, C.Y.; Shieh, J.Y. Green tea polyphenol (−)-epigallocatechin gallate attenuates the neuronal NADPH-d/nNOS expression in the nodose ganglion of acute hypoxic rats. Brain Res. 2004, 999, 73–80. [Google Scholar] [CrossRef]
- Maccallini, C.; Amoroso, R. Targeting neuronal nitric oxide synthase as a valuable strategy for the therapy of neurological disorders. Neural Regen. Res. 2016, 11, 1731–1734. [Google Scholar] [CrossRef]
- Wei, I.H.; Tu, H.C.; Huang, C.C.; Tsai, M.H.; Tseng, C.Y.; Shieh, J.Y. (−)-Epigallocatechin gallate attenuates NADPH-d/nNOS expression in motor neurons of rats following peripheral nerve injury. BMC Neurosci. 2011, 12, 52. [Google Scholar] [CrossRef]
- Prasanth, M.I.; Sivamaruthi, B.S.; Chaiyasut, C.; Tencomnao, T. A review of the role of green tea (Camellia sinensis) in antiphotoaging, stress resistance, neuroprotection, and autophagy. Nutrients 2019, 11, 474. [Google Scholar] [CrossRef]
- Yasuda, S.; Sowa, Y.; Hashimoto, H.; Nakagami, T.; Tsuno, T.; Sakai, T. Cycloartenyl ferulate and beta-sitosteryl ferulate—Steryl ferulates of gamma-oryzanol—Suppress intracellular reactive oxygen species in cell-based system. J. Oleo Sci. 2019, 68, 765–768. [Google Scholar] [CrossRef]
- Yan, Z.M.; Zhong, Y.Z.; Duan, Y.H.; Chen, Q.H.; Li, F.N. Antioxidant mechanism of tea polyphenols and its impact on health benefits. Anim. Nutr. 2020, 6, 115–123. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, G.; Li, C.J.; Zhang, M.; Zhao, H.; Sheng, J.; Shi, W. Pu-erh ta reduces nitric oxide levels in rats by inhibiting inducible nitric oxide synthase expression through Toll-like receptor 4. Int. J. Mol. Sci. 2012, 13, 7174–7185. [Google Scholar] [CrossRef] [PubMed]
- Roman, M.J.; Decker, E.A.; Goddard, J.M. Biomimetic polyphenol coatings for antioxidant active packaging applications. Colloid Interface Sci. Commun. 2016, 13, 10–13. [Google Scholar] [CrossRef]
- Perron, N.R.; Hodges, J.N.; Jenkins, M.; Brumaghim, J.L. Predicting how polyphenol antioxidants prevent DNA damage by binding to iron. Inorg. Chem. 2008, 47, 6153–6161. [Google Scholar] [CrossRef] [PubMed]
- Perron, N.R.; Wang, H.C.; DeGuire, S.N.; Jenkins, M.; Lawson, M.; Brumaghim, J.L. Kinetics of iron oxidation upon polyphenol binding. Dalton Trans. 2010, 39, 9982–9987. [Google Scholar] [CrossRef] [PubMed]
- Ward, R.; Zucca, F.A.; Duyn, J.H.; Crichton, R.R.; Zecca, L. The role of iron in brain ageing and neurodegenerative disorders. Lancet Neurol. 2014, 13, 1045–1060. [Google Scholar] [CrossRef]
- Malar, D.S.; Prasanth, M.I.; Brimson, J.M.; Sharika, R.; Sivamaruthi, B.S.; Chaiyasut, C.; Tencomnao, T. Neuroprotective properties of green tea (Camellia sinensis) in Parkinson’s disease: A Review. Molecules 2020, 25, 3926. [Google Scholar] [CrossRef]
- Alu, A.; Han, X.J.; Ma, X.L.; Wu, M.; Wei, Y.Q.; Wei, X.W. The role of lysosome in regulated necrosis. Acta Pharm. Sin. B 2020, 10, 1880–1903. [Google Scholar] [CrossRef]
- Quinn, P.M.J.; Wijnholds, J. Retinogenesis of the human fetal retina: An apical polarity perspective. Genes 2019, 10, 987. [Google Scholar] [CrossRef]
- Mandel, S.; Maor, G.; Youdim, M.B.H. Iron and alpha-synuclein in the substantia nigra of MPTP-treated Mice. J. Mol. Neurosci. 2004, 24, 401–416. [Google Scholar] [CrossRef]
- Levites, Y.; Amit, T.; Mandel, S.; Youdim, M.B.H. Neuroprotection and neurorescue against A beta toxicity and PKC-dependent release of non-amyloidogenic soluble precursor protein by green tea polyphenol (−)-epigallocatechin-3-gallate. FASEB J. 2003, 17, 952–954. [Google Scholar] [CrossRef]
- Siddique, Y.H.; Jyoti, S.; Naz, F. Effect of epicatechin gallate dietary supplementation on transgenic Drosophila model of Parkinson’s disease. J. Diet. Suppl. 2014, 11, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Miao, W.; Yan, Y.; Bao, T.H.; Jia, W.J.; Yang, F.; Wang, Y.; Zhu, Y.H.; Yin, M.; Han, J.H. Ischemic postconditioning exerts neuroprotective effect through negatively regulating PI3K/Akt2 signaling pathway by microRNA-124. Biomed. Pharmacother. 2020, 126, 109786. [Google Scholar] [CrossRef]
- Park, S. Protective effects of green tea catechins against high glucose-induced cell death in neuron cells. J. Korean Tea Soc. 2012, 18, 62–67. [Google Scholar]
- Ding, M.L.; Ma, H.; Man, Y.G.; Lv, H.Y. Protective effects of a green tea polyphenol, epigallocatechin-3-gallate, against sevoflurane-induced neuronal apoptosis involve regulation of CREB/BDNF/TrkB and PI3K/Akt/mTOR signalling pathways in neonatal mice. Can. J. Physiol. Pharmacol. 2017, 95, 1396–1405. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.Y.; Ye, L.F.; Xu, X.J.; Huang, B.X.; Zhang, X.D.; Zhu, Y.G.; Chen, X.C. Epigallocatechin-3-gallate suppresses 1-methyl-4-phenyl-pyridine-induced oxidative stress in PC12 cells via the SIRT1/PGC-1 alpha signaling pathway. BMC Complement. Altern. Med. 2012, 12, 82. [Google Scholar] [CrossRef]
- Koh, S.H.; Kim, S.H.; Kwon, H.; Park, Y.; Kim, K.S.; Song, C.W.; Kim, J.; Kim, M.H.; Yu, H.J.; Henkel, J.S.; et al. Epigallocatechin gallate protects nerve growth factor differentiated PC12 cells from oxidative-radical-stress-induced apoptosis through its effect on phosphoinositide 3-kinase/Akt and glycogen synthase kinase-3. Mol. Brain Res. 2003, 118, 72–81. [Google Scholar] [CrossRef]
- Nie, G.J.; Cao, Y.L.; Zhao, B.L. Protective effects of green tea polyphenols and their major component, (-)-epigallocatechin-3-gallate (EGCG), on 6-hydroxydopamine-induced apoptosis in PC12 cells. Redox Rep. 2002, 7, 171–177. [Google Scholar] [CrossRef]
- Levites, Y.; Amit, T.; Youdim, M.B.H.; Mandel, S. Involvement of protein kinase C activation and cell survival/cell cycle genes in green tea polyphenol (−)-epigallocatechin 3-gallate neuroprotective action. J. Biol. Chem. 2002, 277, 30574–30580. [Google Scholar] [CrossRef]
- Shabani, S.; Rabiei, Z.; Amini-Khoei, H. Exploring the multifaceted neuroprotective actions of gallic acid: A review. Int. J. Food Prop. 2020, 23, 736–752. [Google Scholar] [CrossRef]
- Farzaei, M.H.; Bahramsoltani, R.; Abbasabadi, Z.; Braidy, N.; Nabavi, S.M. Role of green tea catechins in prevention of age-related cognitive decline: Pharmacological targets and clinical perspective. J. Cell. Physiol. 2019, 234, 2447–2459. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.R.; Ren, T.T.; Lan, R.; Qin, X.Y. Tea polyphenols attenuate staurosporine-induced cytotoxicity and apoptosis by modulating BDNF-TrkB/Akt and Erk1/2 signaling axis in hippocampal neurons. IBRO Rep. 2020, 8, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Park, S.Y.; Lee, S.H.; Kim, Y.; Kim, Y.J.; Jun, W.; Yoon, H.G. Ameliorative effects of dendropanax morbifera on cognitive impairment via enhancing cholinergic functions and brain-derived neurotrophic factor expression in beta-amyloid-induced mice. J. Med. Food 2019, 22, 587–593. [Google Scholar] [CrossRef]
- Yang, Y. Improvement of cognitive function in early stage of vascular cognition impairment (VCI) after treatment with the acetylcholine precursor choline alfoscerate. Cerebrovasc. Dis. 2017, 43, 1. [Google Scholar]
- Piccardi, M.; Congiu, D.; Squassina, A.; Manconi, F.; Putzu, P.F.; Mereu, R.M.; Chillotti, C.; Del Zompo, M. Alzheimer’s disease: Case-control association study of polymorphisms in ACHE, CHAT, and BCHE genes in a Sardinian sample. Am. J. Med. Genet. B 2007, 144B, 895–899. [Google Scholar] [CrossRef]
- Assuncao, M.; Santos-Marques, M.J.; Carvalho, F.; Lukoyanov, N.V.; Andrade, J.P. Chronic green tea consumption prevents age-related changes in rat hippocampal formation. Neurobiol. Aging 2011, 32, 707–717. [Google Scholar] [CrossRef]
- Tseng, H.C.; Wang, M.H.; Soung, H.S.; Chang, Y.; Chang, K.C. (−)Epigallocatechin-3-gallate prevents the reserpine-induced impairment of short-term social memory in rats. Behav. Pharmacol. 2015, 26, 741–747. [Google Scholar] [CrossRef]
- Magrinelli, F.; Picelli, A.; Tocco, P.; Federico, A.; Roncari, L.; Smania, N.; Zanette, G.; Tamburin, S. Pathophysiology of motor dysfunction in Parkinson’s disease as the rationale for drug treatment and rehabilitation. Parkinsons Dis. 2016, 2016, 9832839. [Google Scholar] [CrossRef]
- Akinyemi, R.O. Epidemiology of Parkinsonism and Parkinson’s disease in Sub-Saharan Africa: Nigerian profile. J. Neurosci. Rural Pract. 2012, 3, 233–234. [Google Scholar] [CrossRef]
- Katunina, E.A.; Bezdolny, Y.N. Epidemiology of Parkinson’s disease. Zhurnal Nevrol. Psikhiatrii Im. SS Korsakova 2013, 113, 81–88. [Google Scholar]
- Salari, S.; Bagheri, M. In Vivo, in Vitro and pharmacologic models of Parkinson’s disease. Physiol. Res. 2019, 68, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Wang, T.; Yue, F.; Li, X.; Wang, P.; Li, Y.; Chan, P.; Yu, S. Tea polyphenols alleviate motor impairments, dopaminergic neuronal injury, and cerebral α-synuclein aggregation in MPTP-intoxicated parkinsonian monkeys. Neuroscience 2015, 286, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Gelpi, E.; Navarro-Otano, J.; Tolosa, E.; Gaig, C.; Compta, Y.; Rey, M.J.; Marti, M.J.; Hernandez, I.; Valldeoriola, F.; Rene, R.; et al. Multiple organ involvement by alpha-synuclein pathology in lewy body disorders. Mov. Disord. 2014, 29, 1010–1018. [Google Scholar] [CrossRef]
- Yin, T.; Yao, W.; Lemenze, A.D.; D’Adamio, L. Danish and British dementia ITM2b/BRI2 mutations reduce BRI2 protein stability and impair glutamatergic synaptic transmission. J. Biol. Chem. 2021, 296, 100054. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Xu, Z.F.; Deng, Y.; Xu, B.; Yang, H.B.; Wei, Y.G.; Feng, S. Excitotoxicity and oxidative damages induced by methylmercury in rat cerebral cortex and the protective effects of tea polyphenols. Environ. Toxicol. 2014, 29, 269–283. [Google Scholar] [CrossRef] [PubMed]
- Calgarotto, A.K.; Maso, V.; Franchi, G.C.; Nowill, A.E.; Latuf, P.; Vassallo, J.; Saad, S.T.O. Antitumor activities of quercetin and green tea in xenografts of human leukemia HL60 cells. Sci. Rep. 2018, 8, 3459. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; He, W.Y.; Zeng, Y.Z.; Hossain, A.; Gou, X. Inhibiting autophagy overcomes docetaxel resistance in castration-resistant prostate cancer cells. Int. Urol. Nephrol. 2018, 50, 675–686. [Google Scholar] [CrossRef]
- Holczer, M.; Besze, B.; Zambo, V.; Csala, M.; Banhegyi, G.; Kapuy, O. Epigallocatechin-3-Gallate (EGCG) promotes autophagy-dependent survival via influencing the balance of mTOR-AMPK pathways upon endoplasmic reticulum stress. Oxidative Med. Cell. Longev. 2018, 2018, 6721530. [Google Scholar] [CrossRef] [PubMed]
- Wei, R.; Mao, L.M.; Xu, P.; Zheng, X.H.; Hackman, R.M.; Mackenzie, G.G.; Wang, Y.F. Suppressing glucose metabolism with epigallocatechin-3-gallate (EGCG) reduces breast cancer cell growth in preclinical models. Food Funct. 2018, 9, 5682–5696. [Google Scholar] [CrossRef]
- Grube, S.; Ewald, C.; Kogler, C.; McLean, A.L.; Kalff, R.; Walter, J. Achievable central nervous system concentrations of the green tea catechin EGCG induce stress in glioblastoma cells in vitro. Nutr. Cancer 2018, 70, 1145–1158. [Google Scholar] [CrossRef]
- Selma, M.V.; Espin, J.C.; Tomas-Barberan, F.A. Interaction between phenolics and gut microbiota: Role in human health. J. Agric. Food Chem. 2009, 57, 6485–6501. [Google Scholar] [CrossRef]
- Liu, Z.B.; de Bruijn, W.J.C.; Bruins, M.E.; Vincken, J.P. Reciprocal interactions between epigallocatechin-3-gallate (EGCG) and human gut microbiota in vitro. J. Agric. Food Chem. 2020, 68, 9804–9815. [Google Scholar] [CrossRef]
- Edwards, C.A.; Havlik, J.; Cong, W.; Mullen, W.; Preston, T.; Morrison, D.J.; Combet, E. Polyphenols and health: Interactions between fibre, plant polyphenols and the gut microbiota. Nutr. Bull. 2017, 42, 356–360. [Google Scholar] [CrossRef]
- Di Meo, F.; Valentino, A.; Petillo, O.; Peluso, G.; Filosa, S.; Crispi, S. Bioactive polyphenols and neuromodulation: Molecular mechanisms in neurodegeneration. Int. J. Mol. Sci. 2020, 21, 2564. [Google Scholar] [CrossRef]
- Van Velzen, E.J.J.; Westerhuis, J.A.; van Duynhoven, J.P.M.; van Dorsten, F.A.; Grun, C.H.; Jacobs, D.M.; Duchateau, G.; Vis, D.J.; Smilde, A.K. Phenotyping tea consumers by nutrikinetic analysis of polyphenolic end-metabolites. J. Proteome Res. 2009, 8, 3317–3330. [Google Scholar] [CrossRef]
- Takagaki, A.; Nanjo, F. Metabolism of (−)-epigallocatechin gallate by rat intestinal flora. J. Agric. Food Chem. 2010, 58, 1313–1321. [Google Scholar] [CrossRef] [PubMed]
- Marin, L.; Miguelez, E.M.; Villar, C.J.; Lombo, F. Bioavailability of dietary polyphenols and gut microbiota metabolism: Antimicrobial properties. BioMed Res. Int. 2015, 2015, 905215. [Google Scholar] [CrossRef]
- Wang, X.; Ye, T.; Chen, W.J.; Lv, Y.; Hao, Z.; Chen, J.; Zhao, J.Y.; Wang, H.P.; Cai, Y.K. Structural shift of gut microbiota during chemo-preventive effects of epigallocatechin gallate on colorectal carcinogenesis in mice. World J. Gastroenterol. 2017, 23, 8128–8139. [Google Scholar] [CrossRef] [PubMed]
- Kohri, T.; Nanjo, F.; Suziki, M.; Seto, R.; Matsumoto, N.; Yamakawa, M.; Hojo, H.; Hara, Y.; Desai, D.; Amin, S.; et al. Synthesis of (−)-[4-3H] epigallocatechin gallate and its metabolic fate in rats after intravenous administration. J. Agric. Food Chem. 2001, 49, 1042–1048. [Google Scholar] [CrossRef]
- Liu, A.B.; Tao, S.Y.; Lee, M.J.; Hu, Q.; Meng, X.F.; Lin, Y.; Yang, C.S. Effects of gut microbiota and time of treatment on tissue levels of green tea polyphenols in mice. BioFactors 2018, 44, 348–360. [Google Scholar] [CrossRef]
- Clifford, M.N.; van der Hooft, J.J.J.; Crozier, A. Human studies on the absorption, distribution, metabolism, and excretion of tea polyphenols. Am. J. Clin. Nutr. 2013, 98, 1619S–1630S. [Google Scholar] [CrossRef]
- Noh, K.; Kang, Y.R.; Nepal, M.R.; Shakya, R.; Kang, M.J.; Kang, W.; Lee, S.; Jeong, H.G.; Jeong, T.C. Impact of gut microbiota on drug metabolism: An update for safe and effective use of drugs. Arch. Pharm. Res. 2017, 40, 1345–1355. [Google Scholar] [CrossRef]
- Wolf, V.G.; Bonacorsi, C.; Raddi, M.S.G.; da Fonseca, L.M.; Ximenes, V.F. Octyl gallate, a food additive with potential beneficial properties to treat Helicobacter pylori infection. Food Funct. 2017, 8, 2500–2511. [Google Scholar] [CrossRef]
- Hara-Terawaki, A.; Takagaki, A.; Kobayashi, H.; Nanjo, F. Inhibitory activity of catechin metabolites produced by intestinal microbiota on proliferation of HeLa cells. Biol. Pharm. Bull. 2017, 40, 1331–1335. [Google Scholar] [CrossRef]
- Lee, C.C.; Kim, J.H.; Kim, J.S.; Oh, Y.S.; Han, S.M.; Park, J.H.Y.; Lee, K.W.; Lee, C.Y. 5-(3′, 4′-Dihydroxyphenyl-gamma-valerolactone), a major microbial metabolite of proanthocyanidin, attenuates THP-1 monocyte-endothelial adhesion. Int. J. Mol. Sci. 2017, 18, 1363. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.W.; Chang, S.C.; Liang, C.J. Persistent organic pollutant lindane degradation by alkaline cold-brew green tea. Chemosphere 2019, 232, 281–286. [Google Scholar] [CrossRef] [PubMed]
- Scheperjans, F.; Aho, V.; Pereira, P.A.B.; Koskinen, K.; Paulin, L.; Pekkonen, E.; Haapaniemi, E.; Kaakkola, S.; Eerola-Rautio, J.; Pohja, M.; et al. Gut microbiota are related to Parkinson’s disease and clinical phenotype. Mov. Disord. 2015, 30, 350–358. [Google Scholar] [CrossRef]
- Ley, R.E.; Peterson, D.A.; Gordon, J.I. Ecological and evolutionary forces shaping microbial diversity in the human intestine. Cell 2006, 124, 837–848. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.C.; Jenner, A.M.; Low, C.S.; Lee, Y.K. Effect of tea phenolics and their aromatic fecal bacterial metabolites on intestinal microbiota. Res. Microbiol. 2006, 157, 876–884. [Google Scholar] [CrossRef]
- Liao, Z.L.; Zeng, B.H.; Wang, W.; Li, G.H.; Wu, F.; Wang, L.; Zhong, Q.P.; Wei, H.; Fang, X. Impact of the consumption of tea polyphenols on early atherosclerotic lesion formation and intestinal Bifidobacteria in high-fat-fed ApoE−/− mice. Front. Nutr. 2016, 3, 42. [Google Scholar] [CrossRef]
- Cheng, M.; Zhang, X.; Miao, Y.J.; Cao, J.X.; Wu, Z.F.; Weng, P.F. The modulatory effect of (−)-epigallocatechin 3-O-(3-O-methyl) gallate (EGCG3″Me) on intestinal microbiota of high fat diet-induced obesity mice model. Food Res. Int. 2017, 92, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Chen, Y.H.; Zhu, J.Y.; Zhang, M.; Ho, C.T.; Huang, Q.R.; Cao, J.X. Metagenomics analysis of gut microbiota in a high fat diet-induced obesity mouse model fed with (−)-epigallocatechin 3-O-(3-O-methyl) gallate (EGCG3″Me). Mol. Nutr. Food Res. 2018, 62, e1800274. [Google Scholar] [CrossRef]
- Ikarashi, N.; Ogawa, S.; Hirobe, R.; Kon, R.; Kusunoki, Y.; Yamashita, M.; Mizukami, N.; Kaneko, M.; Wakui, N.; Machida, Y.; et al. Epigallocatechin gallate induces a hepatospecific decrease in the CYP3A expression level by altering intestinal flora. Eur. J. Pharm. Sci. 2017, 100, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zeng, B.H.; Liu, Z.W.; Liao, Z.L.; Zhong, Q.P.; Gu, L.H.; Wei, H.; Fang, X. Green tea polyphenols modulate colonic microbiota diversity and lipid metabolism in high-fat diet treated HFA mice. J. Food Sci. 2018, 83, 864–873. [Google Scholar] [CrossRef]
- Cheng, M.; Zhang, X.; Zhu, J.Y.; Cheng, L.; Cao, J.X.; Wu, Z.F.; Weng, P.F.; Zheng, X.J. A metagenomics approach to the intestinal microbiome structure and function in high fat diet-induced obesity mice fed with oolong tea polyphenols. Food Funct. 2018, 9, 1079–1087. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.Y.; Chen, Y.H.; Cheng, M.; Zhang, X.; Zheng, X.J.; Zhang, Z.C. The modulatory effect of polyphenols from green tea, oolong tea and black tea on human intestinal microbiota in vitro. J. Food Sci. Technol. 2018, 55, 399–407. [Google Scholar] [CrossRef]
- Henning, S.M.; Yang, J.P.; Hsu, M.; Lee, R.P.; Grojean, E.M.; Ly, A.; Tseng, C.H.; Heber, D.; Li, Z.P. Decaffeinated green and black tea polyphenols decrease weight gain and alter microbiome populations and function in diet-induced obese mice. Eur. J. Nutr. 2018, 57, 2759–2769. [Google Scholar] [CrossRef]
- Duda-Chodak, A.; Tarko, T.; Satora, P.; Sroka, P. Interaction of dietary compounds, especially polyphenols, with the intestinal microbiota: A review. Eur. J. Nutr. 2015, 54, 325–341. [Google Scholar] [CrossRef]
- Hollman, P.C.H.; Arts, I.C.W. Flavonols, flavones and flavanols—Nature, occurrence and dietary burden. J. Sci. Food Agric. 2000, 80, 1081–1093. [Google Scholar] [CrossRef]
- Parkar, S.G.; Stevenson, D.E.; Skinner, M.A. The potential influence of fruit polyphenols on colonic microflora and human gut health. Int. J. Food Microbiol. 2008, 124, 295–298. [Google Scholar] [CrossRef]
- Duda-Chodak, A. THe inhibitory effect of polyphenols on human gut microbiota. J. Physiol. Pharmacol. 2012, 63, 497–503. [Google Scholar] [PubMed]
- Aranda Cazon, C.; Campos Munoz, L.; Conde Taboada, A.; Lopez Bran, E. Viral wart treatment with green tea sinecatechins. Anales De Pediatria 2016, 84, 236–237. [Google Scholar] [CrossRef] [PubMed]
- Sourabh, A.; Kanwar, S.S.; Sud, R.G.; Ghabru, A.; Sharma, O.P. Influence of phenolic compounds of Kangra tea [Camellia sinensis (L) O Kuntze] on bacterial pathogens and indigenous bacterial probiotics of Western Himalayas. Braz. J. Microbiol. 2013, 44, 709–715. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; Zhang, Y.; Li, J.; Fu, C.; Zhang, X. The Neuroprotective Effect of Tea Polyphenols on the Regulation of Intestinal Flora. Molecules 2021, 26, 3692. https://doi.org/10.3390/molecules26123692
Zhang Z, Zhang Y, Li J, Fu C, Zhang X. The Neuroprotective Effect of Tea Polyphenols on the Regulation of Intestinal Flora. Molecules. 2021; 26(12):3692. https://doi.org/10.3390/molecules26123692
Chicago/Turabian StyleZhang, Zhicheng, Yuting Zhang, Junmin Li, Chengxin Fu, and Xin Zhang. 2021. "The Neuroprotective Effect of Tea Polyphenols on the Regulation of Intestinal Flora" Molecules 26, no. 12: 3692. https://doi.org/10.3390/molecules26123692
APA StyleZhang, Z., Zhang, Y., Li, J., Fu, C., & Zhang, X. (2021). The Neuroprotective Effect of Tea Polyphenols on the Regulation of Intestinal Flora. Molecules, 26(12), 3692. https://doi.org/10.3390/molecules26123692