Osteoclast-Mediated Cell Therapy as an Attempt to Treat Elastin Specific Vascular Calcification
Abstract
1. Introduction
2. Materials and Methods
2.1. Microbead Osteoclast Delivery
2.1.1. Collagen/Alginate Bead Preparation and Testing
2.1.2. Osteoclast Labeling and Preparation
2.1.3. Treatment Delivery
2.2. RANKL Injection
RANKL/OPG Injections
2.3. Immediate OC-Encapsulated Collagen Gel Application
2.3.1. Abdominal Aortic Injury Surgery
2.3.2. Histology
2.4. Analysis
2.4.1. Calcium and Desmosine Analysis
2.4.2. RNA Extraction and Gene Analysis
2.4.3. Statistical Analysis
3. Results
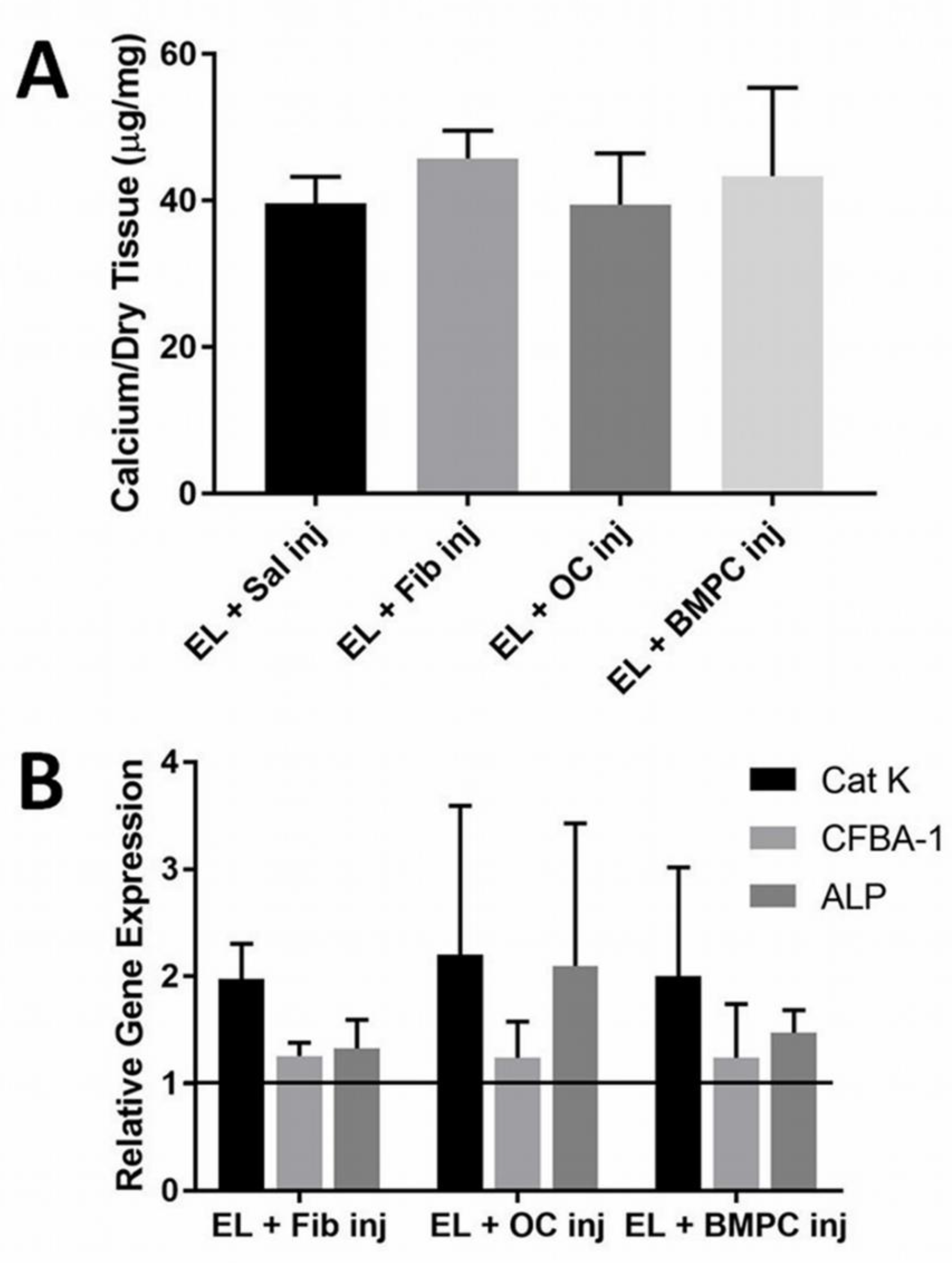
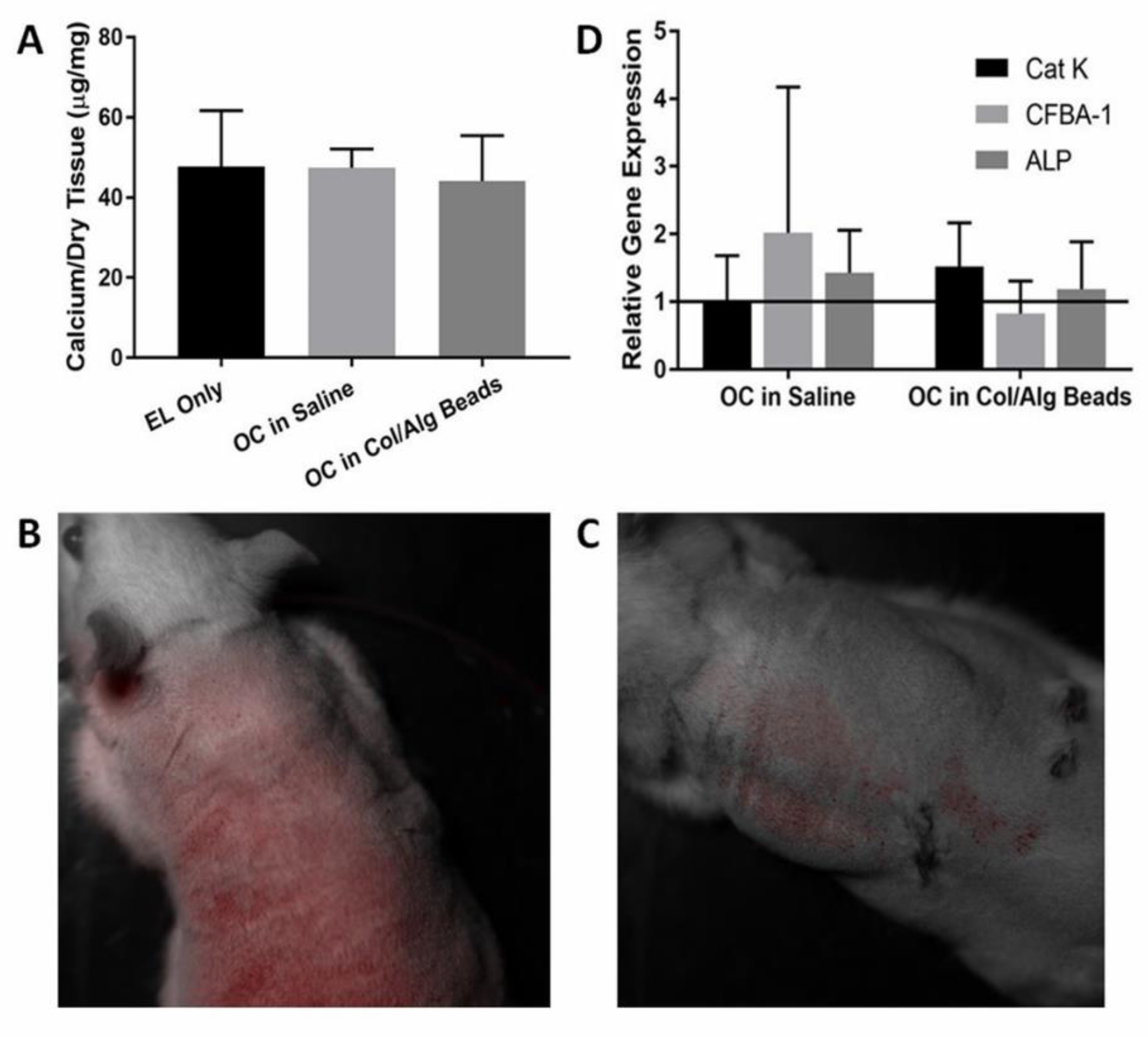
3.1. Microbead Osteoclast Delivery
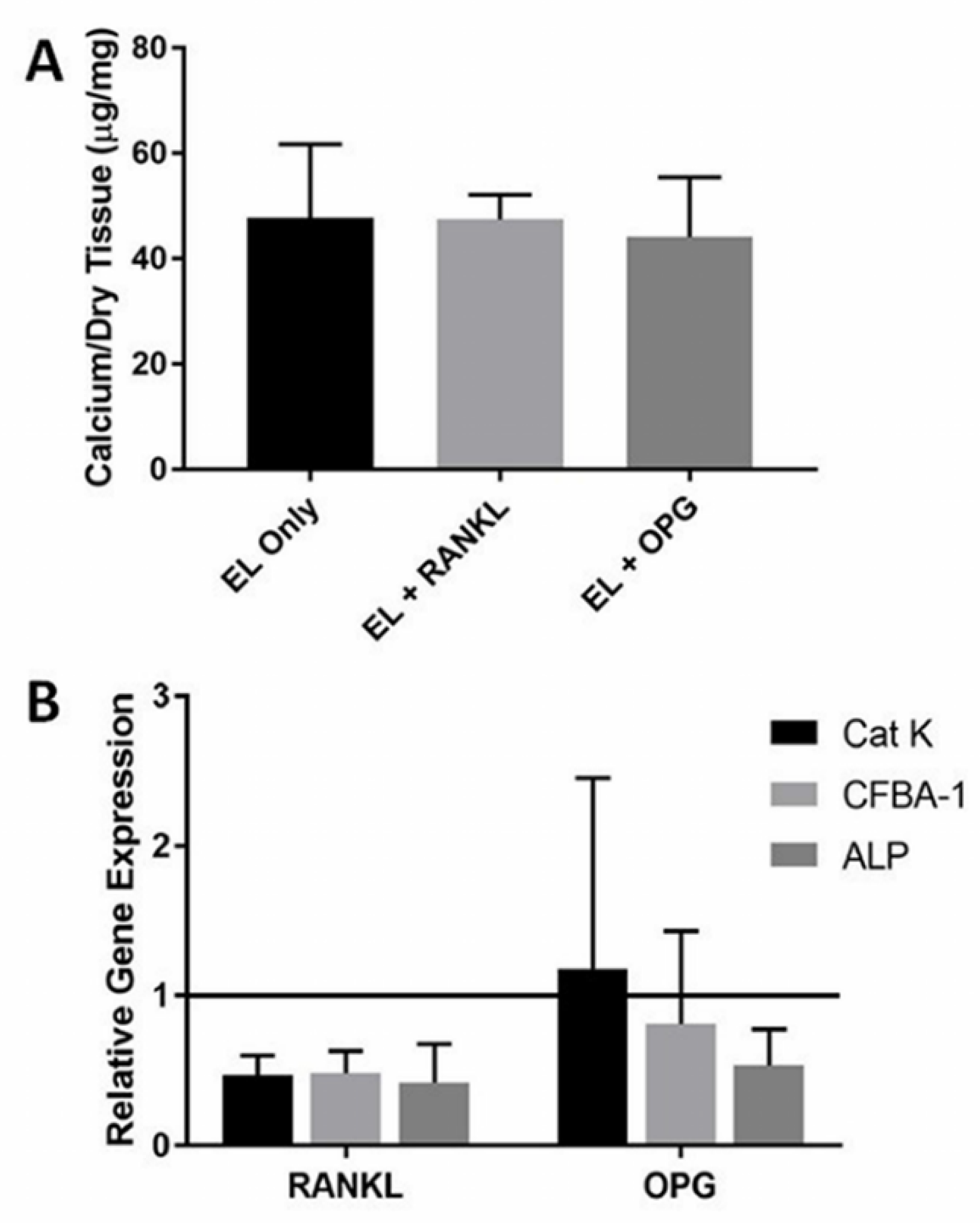
3.2. RANKL Injections
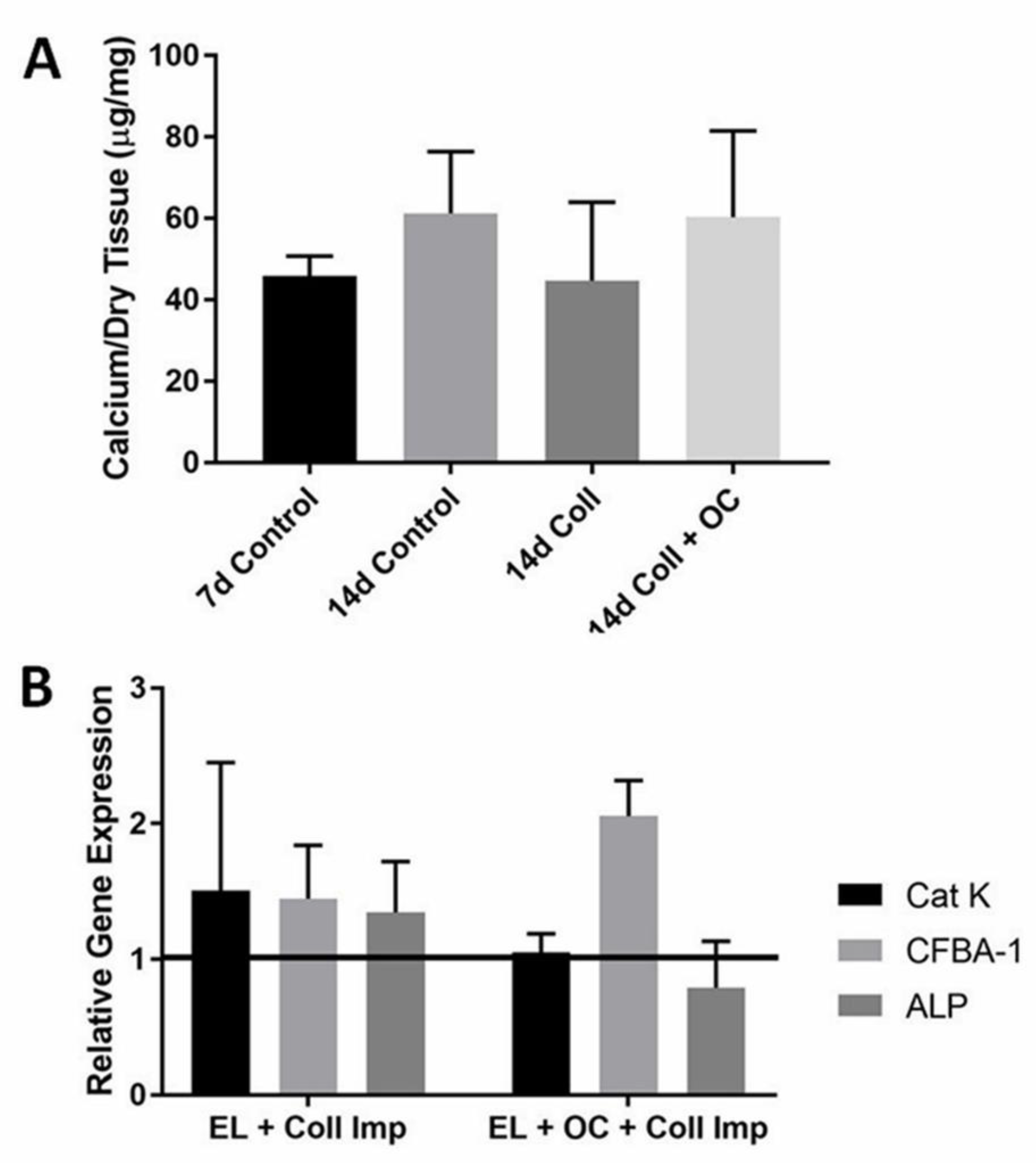
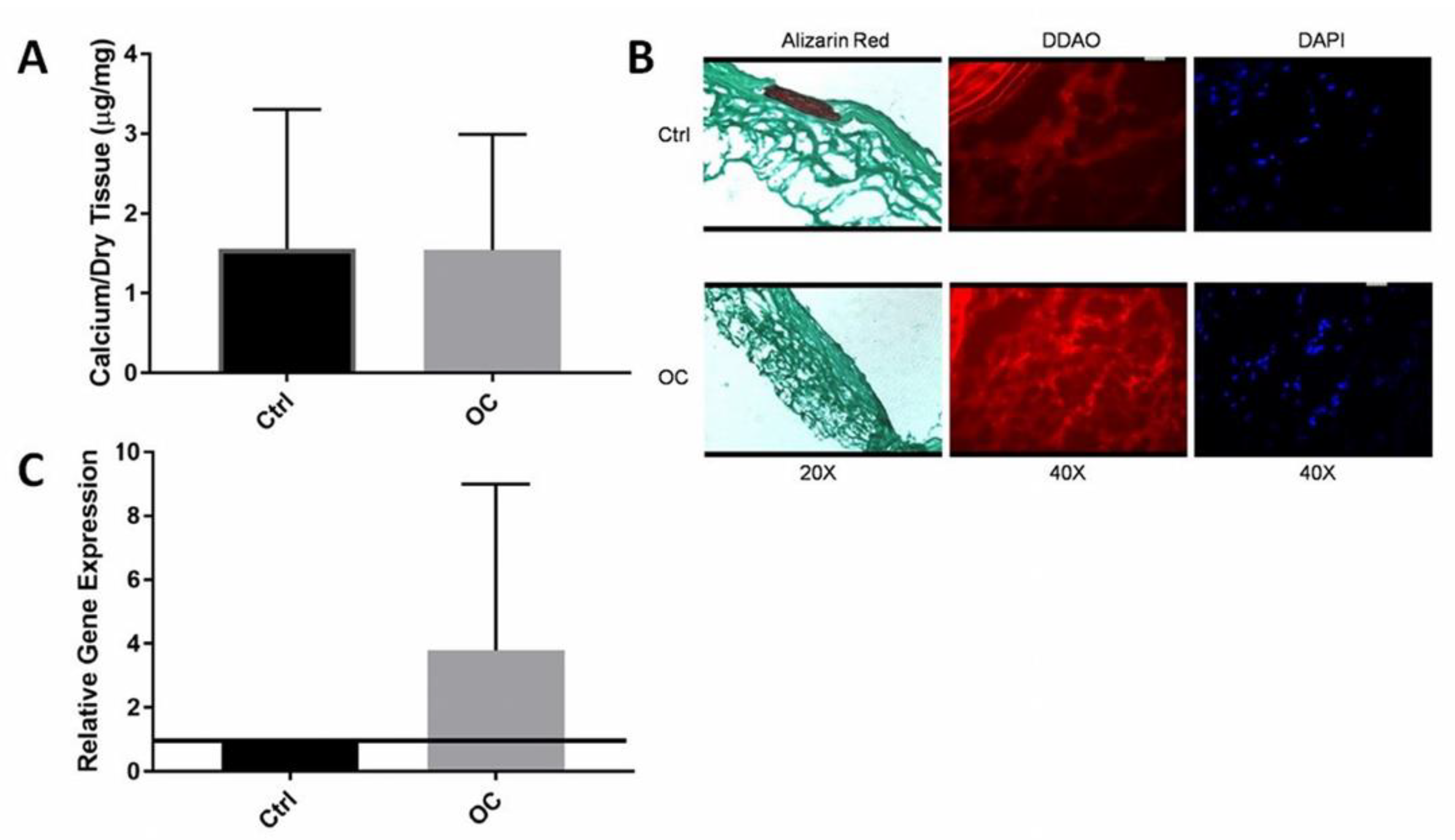
3.3. Immediate OC-Encapsulated Gel Application
4. Discussion
4.1. Microbead Osteoclast Delivery
4.2. RANKL Injections
4.3. Immediate OC-Encapsulated Gel Application
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Yahagi, K.; Kolodgie, F.D.; Lutter, C.; Mori, H.; Romero, M.E.; Finn, A.V.; Virmani, R. Pathology of Human Coronary and Carotid Artery Atherosclerosis and Vascular Calcification in Diabetes Mellitus. Arterioscler. Thromb. Vasc. Biol. 2018, 37, 191–204. [Google Scholar] [CrossRef]
- Paloian, N.J.; Leaf, E.M.; Giachelli, C.M. Osteopontin protects against high phosphate-induced nephrocalcinosis and vascular calcification. Kidney Int. 2016, 89, 1027–1036. [Google Scholar] [CrossRef] [PubMed]
- Mcarthur, K.M.; Kay, A.M.; Mosier, J.A.; Grant, J.N.; Stewart, J.A.; Simpson, C.L. Regulating the Phenotypic Switch of Smooth Muscle Cells in Vascular Calcification Using a Protein-Based Inhibitor. AIMS Cell Tissue Eng. 2017, 1, 165–179. [Google Scholar]
- Timraz, S.B.H.; Farhat, I.A.H.; Alhussein, G.; Christoforou, N.; Teo, J.C.M. In-depth evaluation of commercially available human vascular smooth muscle cells phenotype: Implications for vascular tissue engineering. Exp. Cell Res. 2016, 343, 168–176. [Google Scholar] [CrossRef]
- Yang, H.; Curinga, G.; Giachelli, C.M. Elevated extracellular calcium levels induce smooth muscle cell matrix mineralization in vitro. Kidney Int. 2004, 66, 2293–2299. [Google Scholar] [CrossRef] [PubMed]
- Pai, A.; Leaf, E.M.; El-Abbadi, M.; Giachelli, C.M. Elastin degradation and vascular smooth muscle cell phenotype change precede cell loss and arterial medial calcification in a uremic mouse model of chronic kidney disease. Am. J. Pathol. 2011, 178, 764–773. [Google Scholar] [CrossRef] [PubMed]
- Simionescu, A.; Philips, K.; Vyavahare, N. Elastin-derived peptides and TGF-beta1 induce osteogenic responses in smooth muscle cells. Biochem. Biophys. Res. Comm. 2005, 334, 524–532. [Google Scholar] [CrossRef]
- Reynolds, J.L.; Joannides, A.J.; Skepper, J.N.; McNair, R.; Schurgers, L.J.; Proudfoot, D.; Jahnen-Dechent, W.; Weissberg, P.L.; Shanahan, C.M. Human vascular smooth muscle cells undergo vesicle-mediated calcification in response to changes in extracellular calcium and phosphate concentrations: A potential mechanism for accelerated vascular calcification in ESRD. JASN 2004, 15, 2857–2867. [Google Scholar] [CrossRef] [PubMed]
- Byon, C.H.; Javed, A.; Dai, Q.; Kappes, J.C.; Clemens, T.L.; Darley-Usmar, V.M.; McDonald, J.M.; Chen, Y. Oxidative stress induces vascular calcification through modulation of the osteogenic transcription factor Runx2 by AKT signaling. J. Biol. Chem. 2008, 283, 15319–15327. [Google Scholar] [CrossRef] [PubMed]
- Bessueille, L.; Magne, D. Inflammation: A culprit for vascular calcification in atherosclerosis and diabetes. Cell. Mol. Life Sci. 2015, 72, 2475–2489. [Google Scholar] [CrossRef]
- Kang, Y.H.; Jin, J.S.; Son, S.M. Long Term Effect of High Glucose and Phosphate Levels on the OPG/RANK/RANKL/TRAIL System in the Progression of Vascular Calcification in rat Aortic Smooth Muscle Cells. Korean J. Physiol. Pharm. 2015, 19, 111–118. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bessueille, L.; Fakhry, M.; Hamade, E.; Badranb, B.; Magne, D. Glucose stimulates chondrocyte differentiation of vascular smooth muscle cells and calcification: A possible role for IL-1β. FEBS Lett. 2015, 589. [Google Scholar] [CrossRef] [PubMed]
- Simpson, C.L.; Lindley, S.; Eisenberg, C.; Basalyga, D.M.; Starcher, B.C.; Simionescu, D.T.; Vyavahare, N.R. Toward cell therapy for vascular calcification: Osteoclast-mediated demineralization of calcified elastin. Cardiovasc. Pathol. Pathol. 2007, 16, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Hunt, N.C.; Grover, L.M. Cell encapsulation using biopolymer gels for regenerative medicine. Biotechnol. Lett. 2010, 32, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [PubMed]
- Dufrane, D.; Goebbels, R.-M.; Saliez, A.; Guiot, Y.; Gianello, P. Six-month survival of microencapsulated pig islets and alginate biocompatibility in primates: Proof of concept. Transplantation 2006, 81, 1345–1353. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.R.; Hwang, J.W.; Choi, Y.Y.; Wong, S.F.; Hwang, Y.H.; Lee, D.Y.; Lee, S.-H. In situ formation and collagen-alginate composite encapsulation of pancreatic islet spheroids. Biomaterials 2012, 33, 837–845. [Google Scholar] [CrossRef]
- Mayfield, A.E.; Tilokee, E.L.; Latham, N.; McNeill, B.; Lam, B.K.; Ruel, M.; Suuronen, E.J.; Courtman, D.W.; Stewart, D.J.; Davis, D.R. The effect of encapsulation of cardiac stem cells within matrix-enriched hydrogel capsules on cell survival, post-ischemic cell retention and cardiac function. Biomaterials 2014, 35, 133–142. [Google Scholar] [CrossRef]
- Zhou, H.; Xu, H.H. The fast release of stem cells from alginate-fibrin microbeads in injectable scaffolds for bone tissue engineering. Biomaterials 2011, 32, 7503–7513. [Google Scholar] [CrossRef]
- Hofbauer, L.C.; Brueck, C.C.; Shanahan, C.M.; Schoppet, M.; Dobnig, H. Vascular calcification and osteoporosis—From clinical observation towards molecular understanding. Osteoporos. Int. 2007, 18, 251–259. [Google Scholar] [CrossRef]
- McClung, M. Role of RANKL inhibition in osteoporosis. Arthritis Res. Ther. 2007, 9, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Basalyga, D.M.; Simionescu, D.T.; Xiong, W.; Baxter, B.T.; Starcher, B.C.; Vyavahare, N.R. Elastin degradation and calcification in an abdominal aorta injury model: Role of matrix metalloproteinases. Circulation 2004, 110, 3480–3487. [Google Scholar] [CrossRef] [PubMed]
- Jeziorska, M.; McCollum, C.; Wooley, D.E. Observations on bone formation and remodeling in advanced atherosclerotic lesions of human carotid arteries. Virchows Arch. 1998, 433, 559–565. [Google Scholar] [CrossRef]
- Zhu, D.; Mackenzie, N.; Farquharson, C.; MacRae, V. Mechanisms and clinical consequences of vascular calcification. Front. Endocrinol. 2012, 3, 95. [Google Scholar] [CrossRef] [PubMed]
- Evrard, S.; Delanaye, P.; Kamel, S.; Cristol, J.P.; Cavalier, E. Vascular calcification: From pathophysiology to biomarkers. Clin. Chim. Acta 2015, 438, 401–414. [Google Scholar] [CrossRef] [PubMed]
- Bailey, M.T.; Pillarisetti, S.; Xiao, H.; Vyavahare, N.R. Role of elastin in pathologic calcification of xenograft heart valves. J. Biomed. Mater. Res. A 2003, 66, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Basalyga, D.M.; Simionescu, A.; Isenburg, J.C.; Simionescu, D.T.; Vyavahare, N.R. Elastin calcification in the rat subdermal model is accompanied by up-regulation of degradative and osteogenic cellular responses. Am. J. Pathol. 2006, 168, 490–498. [Google Scholar] [CrossRef]
- Teitelbaum, S.L. Bone resorption by osteoclasts. Science 2000, 289, 1504–1508. [Google Scholar] [CrossRef] [PubMed]
- Aranguren, X.L.; Verfaillie, C.M.; Luttun, A. Emerging hurdles in stem cell therapy for peripheral vascular disease. J. Mol. Med. 2009, 87, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Melo, L.G.; Pachori, A.S.; Kong, D.; Gnecchi, M.; Wang, K.; Pratt, R.E.; Dzau, V.J. Gene and cell-based therapies for heart disease. FASEB J. 2004, 18, 648–663. [Google Scholar] [CrossRef]
- Yagar, H.; Ertan, F.; Balkan, B. Comparison of some properties of free and immobilized alpha-amylase by Aspergillus sclerotiorum in calcium alginate gel beads. Prep. Biochem. Biotechnol. 2008, 38, 13–23. [Google Scholar] [CrossRef]
- Collin-Osdoby, P. Regulation of vascular calcification by osteoclast regulatory factors RANKL and osteoprotegerin. Circ. Res. 2004, 95, 1046–1057. [Google Scholar] [CrossRef] [PubMed]
- Schoppet, M.; Al-Fakhri, N.; Franke, F.E.; Katz, N.; Barth, P.J.; Maisch, B.; Preissner, K.T.; Hofbauer, L.C. Localization of osteoprotegerin, tumor necrosis factor-related apoptosis-inducing ligand, and receptor activator of nuclear factor-kappaB ligand in Mönckeberg’s sclerosis and atherosclerosis. J. Clin. Endocrinol. Metab. 2004, 89, 4104–4112. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sarosi, I.; Yan, X.Q.; Morony, S.; Capparelli, C.; Tan, H.; McCabe, S.; Elliott, R.; Scully, S.; Van, G.; et al. RANK is the intrinsic hematopoietic cell surface receptor that controls osteoclastogenesis and regulation of bone mass and calcium metabolism. PNAS 2000, 97, 1566–1567. [Google Scholar] [CrossRef]
- Boyce, B.F.; Yamashita, T.; Yao, Z.; Zhang, Q.; Li, F.; Xing, L. Roles for NF-kappaB and c-Fos in osteoclasts. J. Bone Min. Metab. 2005, 23, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Lacey, D.L.; Timms, E.; Tan, H.L.; Kelley, M.J.; Dunstan, C.R.; Burgess, T.; Elliott, R.; Colombero, A.; Elliott, G.; Scully, S.; et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell 1998, 93, 165–176. [Google Scholar] [CrossRef]
- Kong, Y.Y.; Yoshida, H.; Sarosi, I.; Tan, H.L.; Timms, E.; Capparelli, C.; Morony, S.; Oliveira-dos-Santos, A.J.; Van, G.; Annick, I.; et al. OPGL is a key regulator of osteoclastogenesis, lymphocyte development and lymph-node organogenesis. Nature 1999, 397, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.Y.; Feige, U.; Sarosi, I.; Bolon, B.; Tafuri, A.; Morony, S.; Capparelli, C.; Li, J.; Elliott, R.; McCabe, S.; et al. Activated T cells regulate bone loss and joint destruction in adjuvant arthritis through osteoprotegerin ligand. Nature 1999, 402, 304–309. [Google Scholar] [CrossRef]
- Persy, V.; D’Haese, P. Vascular calcification and bone disease: The calcification paradox. Trends Mol. Med. 2009, 15, 405–416. [Google Scholar] [CrossRef] [PubMed]
- Doherty, T.M.; Fitzpatrick, L.A.; Inoue, D.; Qiao, J.H.; Fishbein, M.C.; Detrano, R.C.; Shah, P.K.; Rajavashisth, T.B. Molecular, endocrine, and genetic mechanisms of arterial calcification. Endocr. Rev. 2004, 25, 629–672. [Google Scholar] [CrossRef]
- Chinetti-Gbaguidi, G.; Daoudi, M.; Rosa, M.; Vinod, M.; Louvet, L.; Copin, C.; Fanchon, M.; Vanhoutte, J.; Derudas, B.; Belloy, L.; et al. Human Alternative Macrophages Populate Calcified Areas of Atherosclerotic Lesions and Display Impaired RANKL-Induced Osteoclastic Bone Resorption Activity. Circ. Res. 2017, 121, 19–30. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Simpson, C.L.; Mosier, J.A.; Vyavahare, N.R. Osteoclast-Mediated Cell Therapy as an Attempt to Treat Elastin Specific Vascular Calcification. Molecules 2021, 26, 3643. https://doi.org/10.3390/molecules26123643
Simpson CL, Mosier JA, Vyavahare NR. Osteoclast-Mediated Cell Therapy as an Attempt to Treat Elastin Specific Vascular Calcification. Molecules. 2021; 26(12):3643. https://doi.org/10.3390/molecules26123643
Chicago/Turabian StyleSimpson, Chartrisa LaShan, Jenna A. Mosier, and Narendra R. Vyavahare. 2021. "Osteoclast-Mediated Cell Therapy as an Attempt to Treat Elastin Specific Vascular Calcification" Molecules 26, no. 12: 3643. https://doi.org/10.3390/molecules26123643
APA StyleSimpson, C. L., Mosier, J. A., & Vyavahare, N. R. (2021). Osteoclast-Mediated Cell Therapy as an Attempt to Treat Elastin Specific Vascular Calcification. Molecules, 26(12), 3643. https://doi.org/10.3390/molecules26123643




