Selective Production of Phenol on Bifunctional, Hierarchical ZSM-5 Zeolites
Abstract
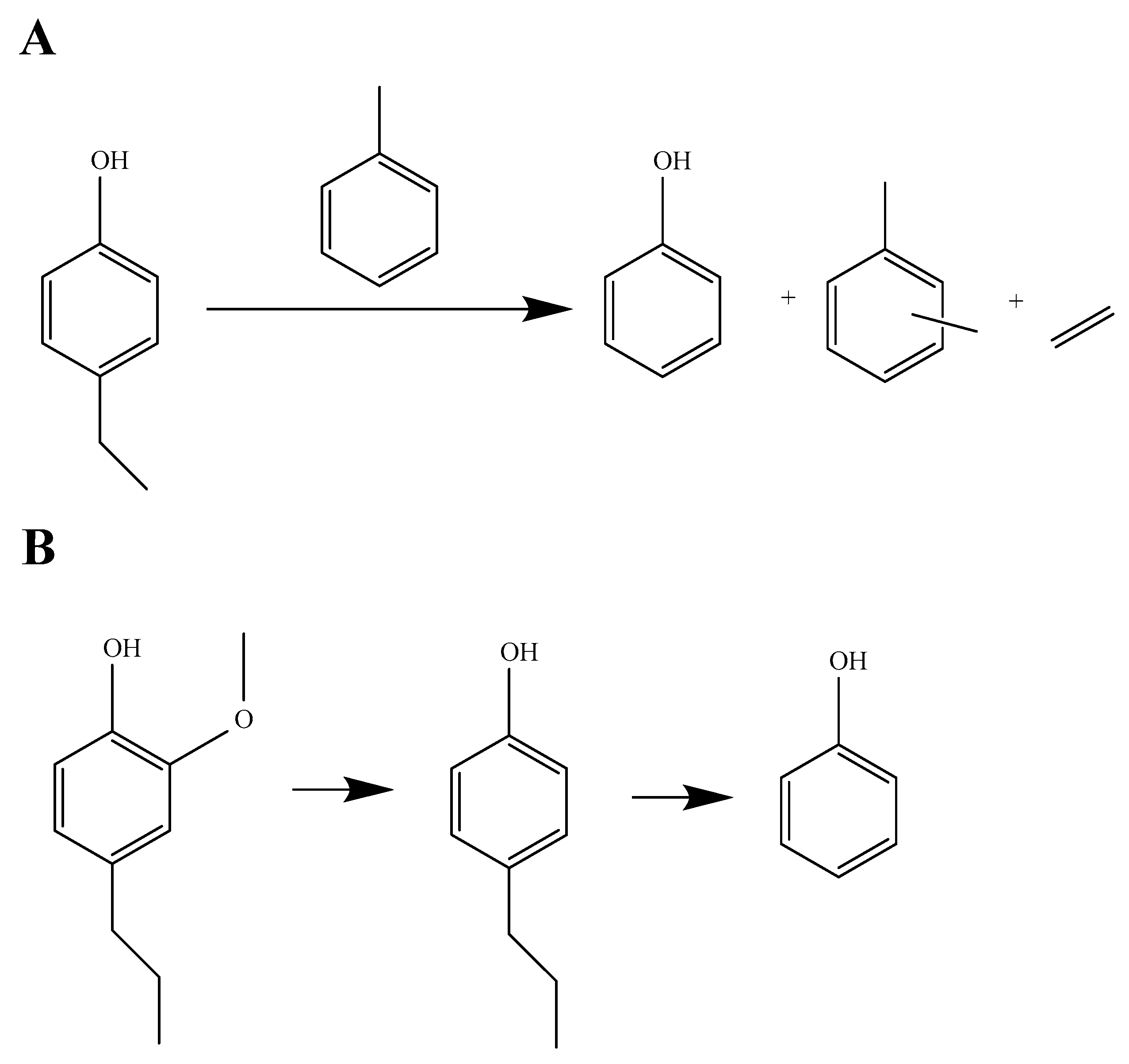
1. Introduction
2. Results and Discussion
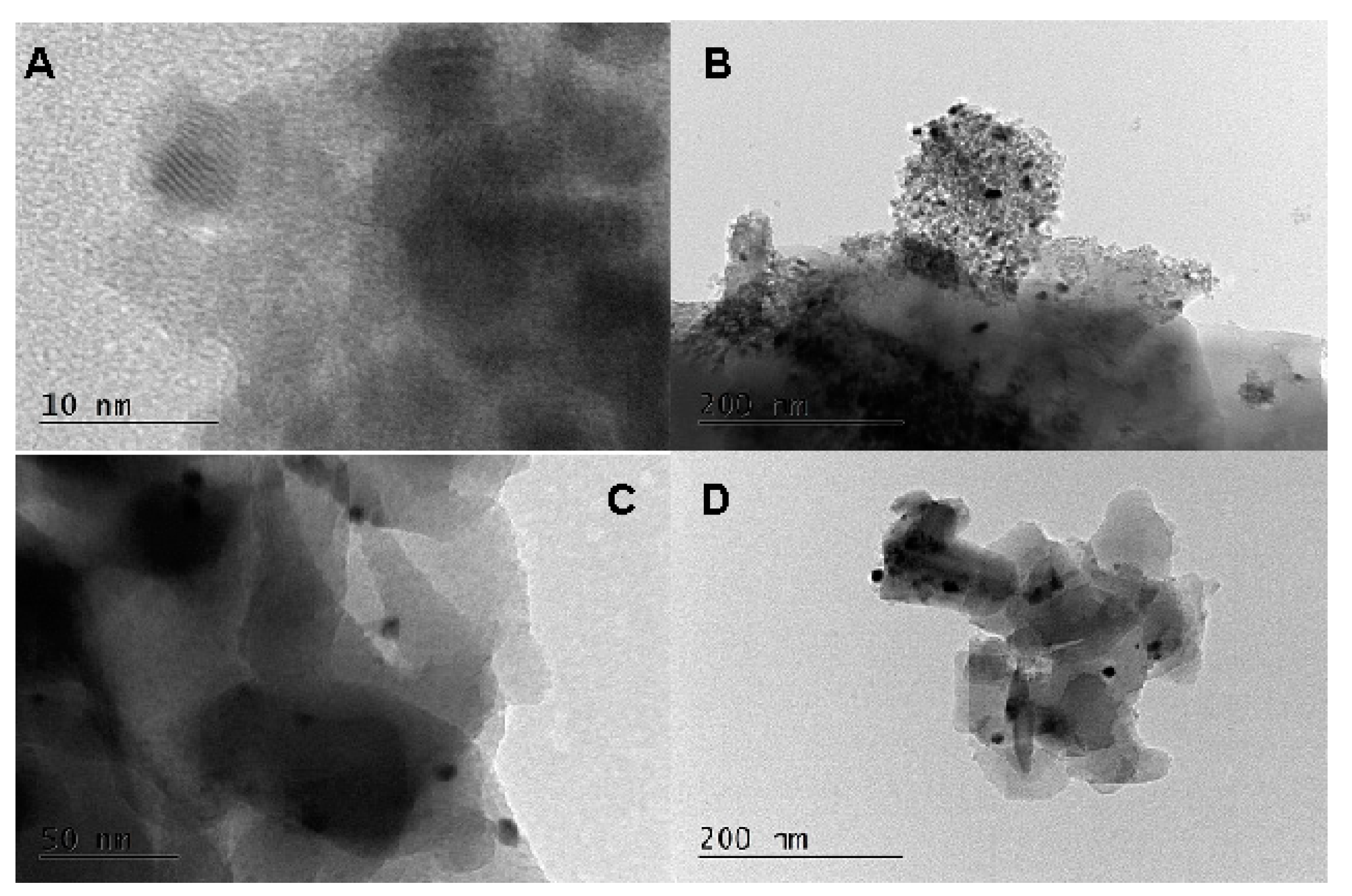
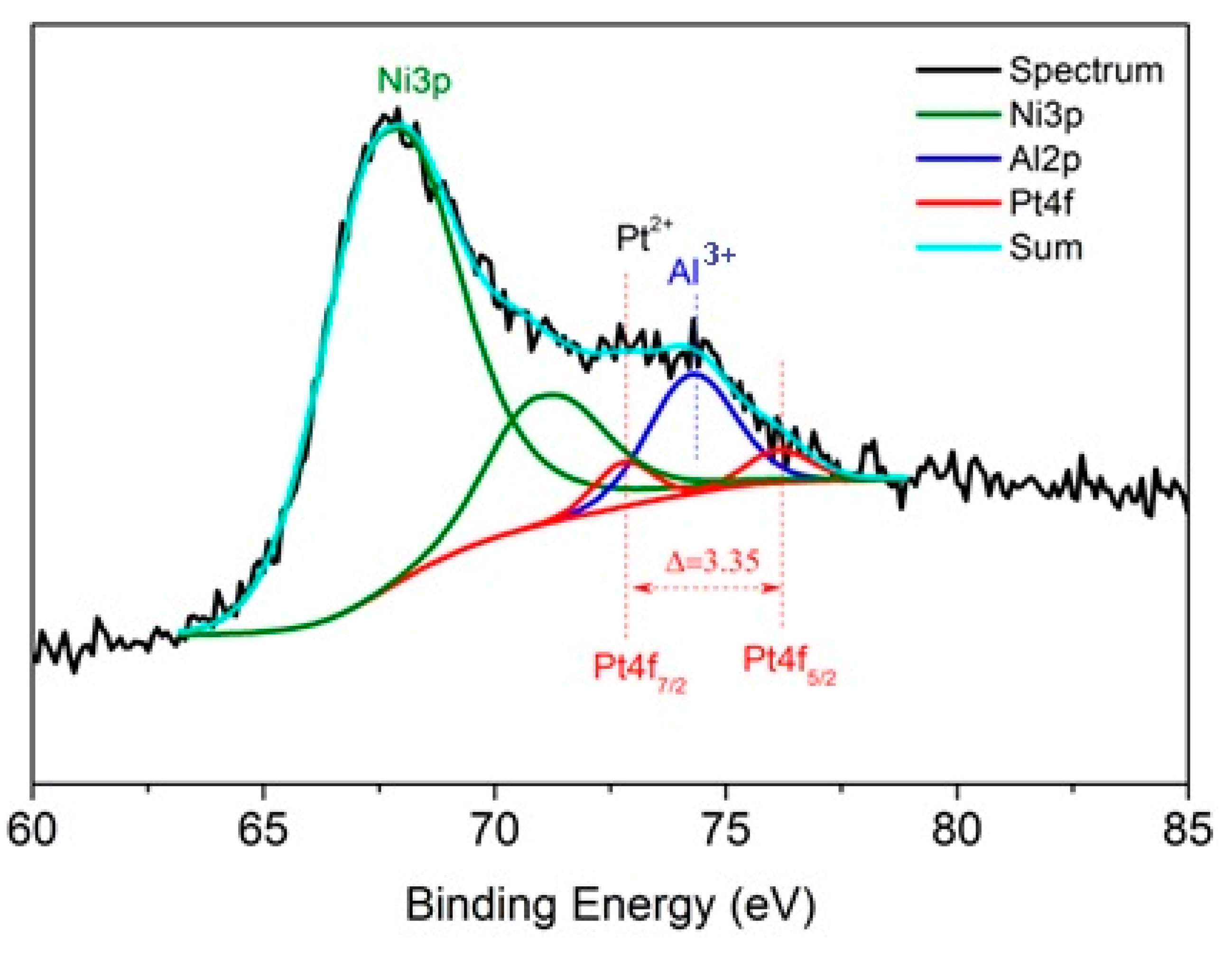
2.1. Physico-Chemical Properties
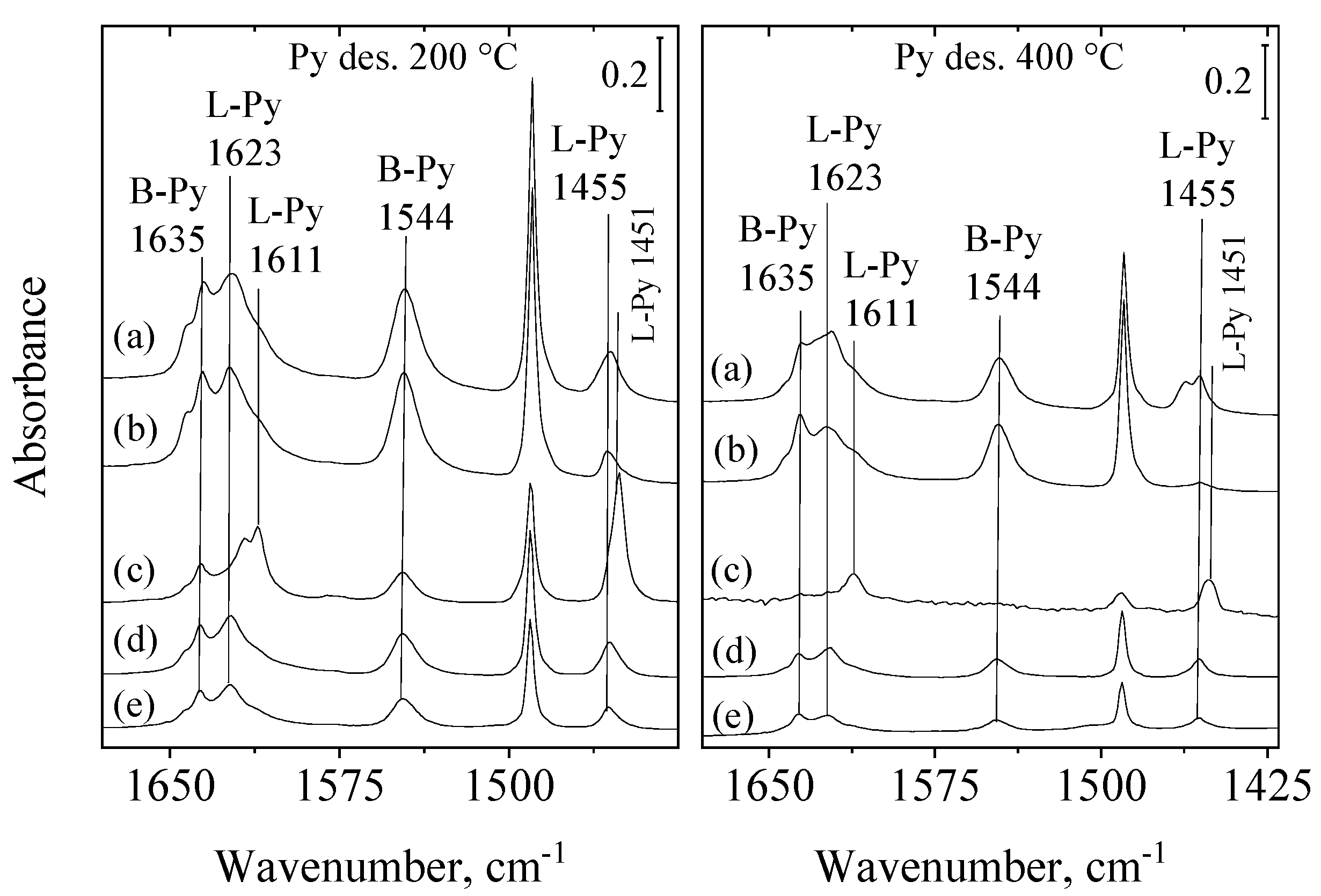
2.1.1. FTIR Spectra of Adsorbed Pyridine
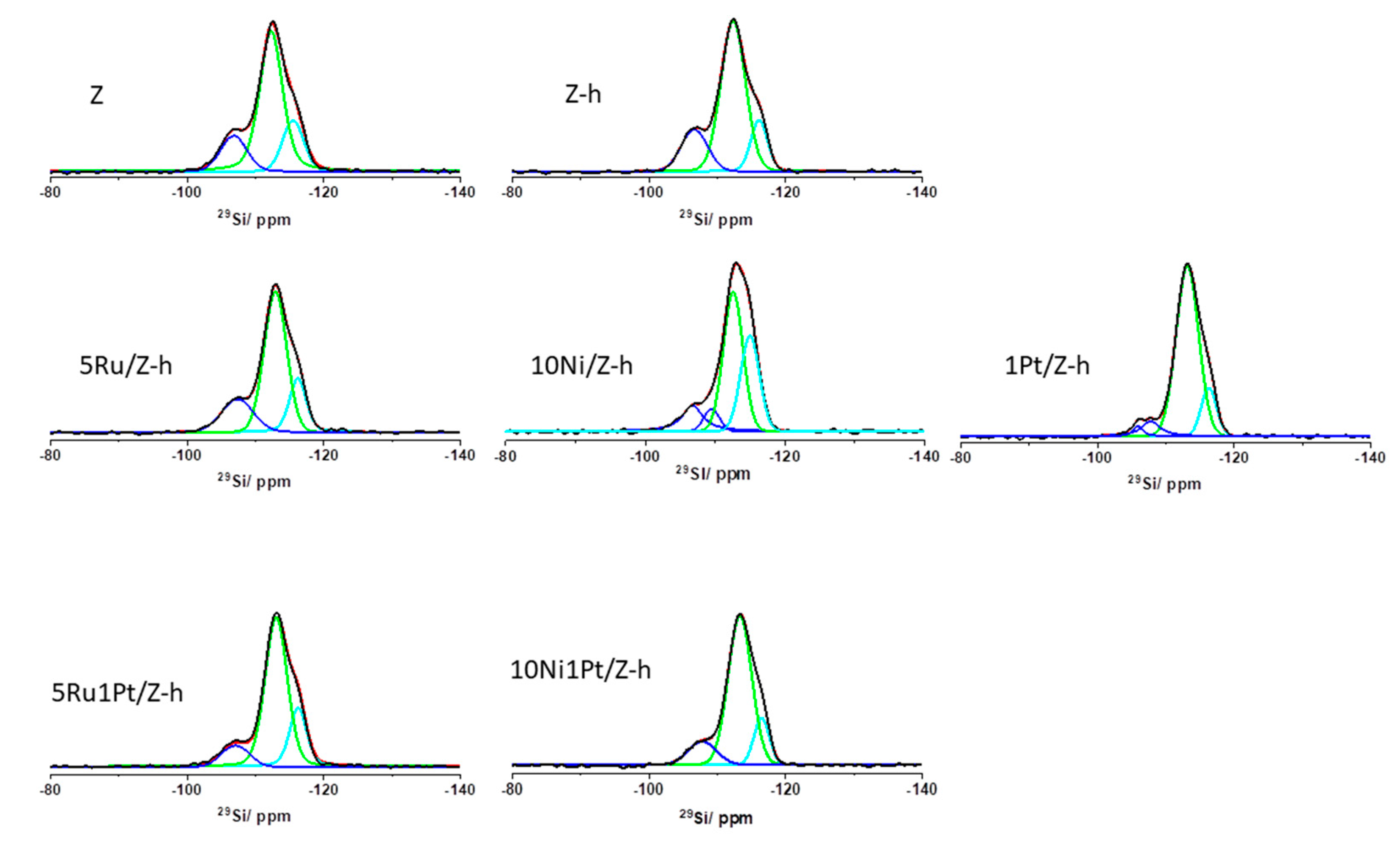
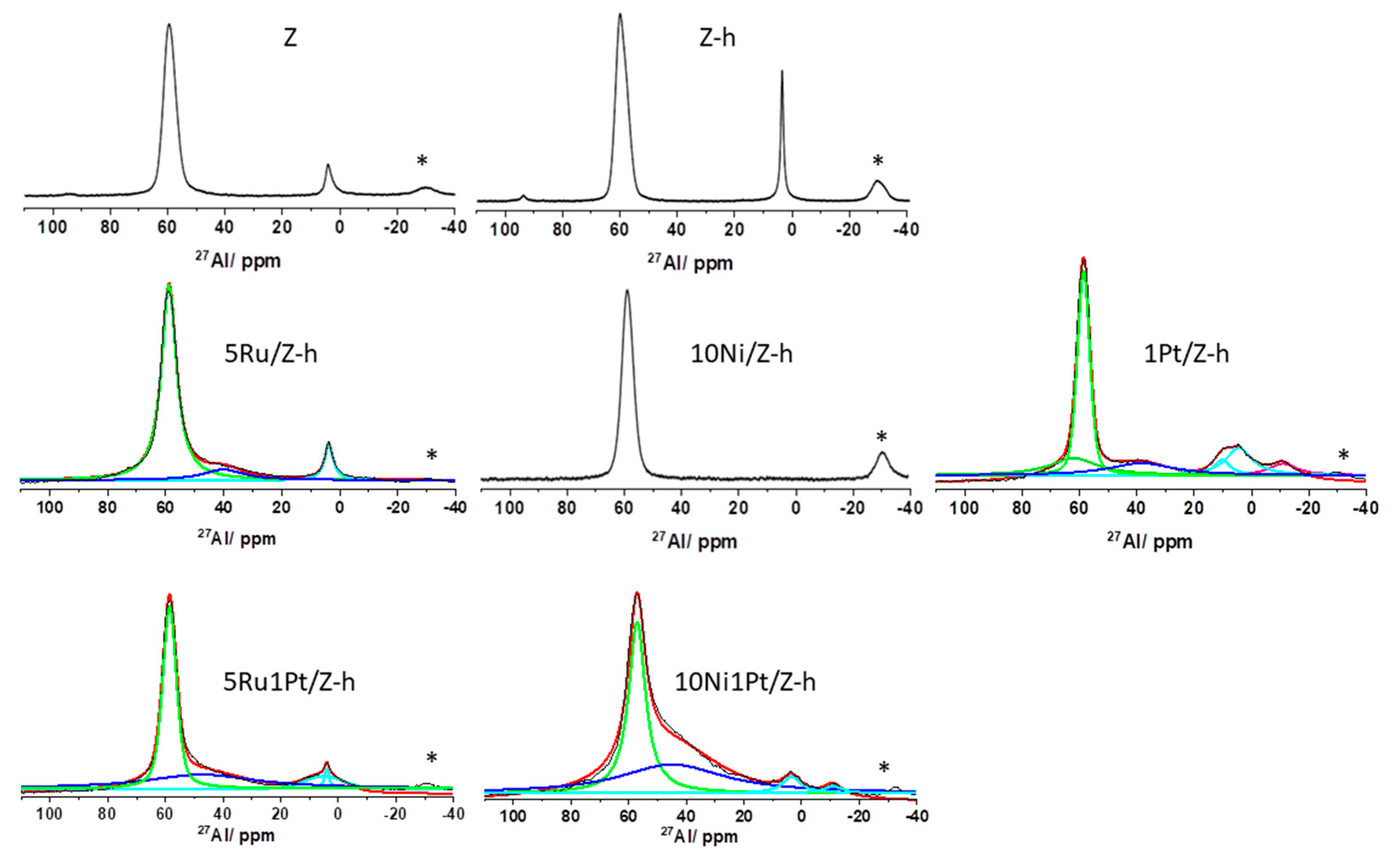
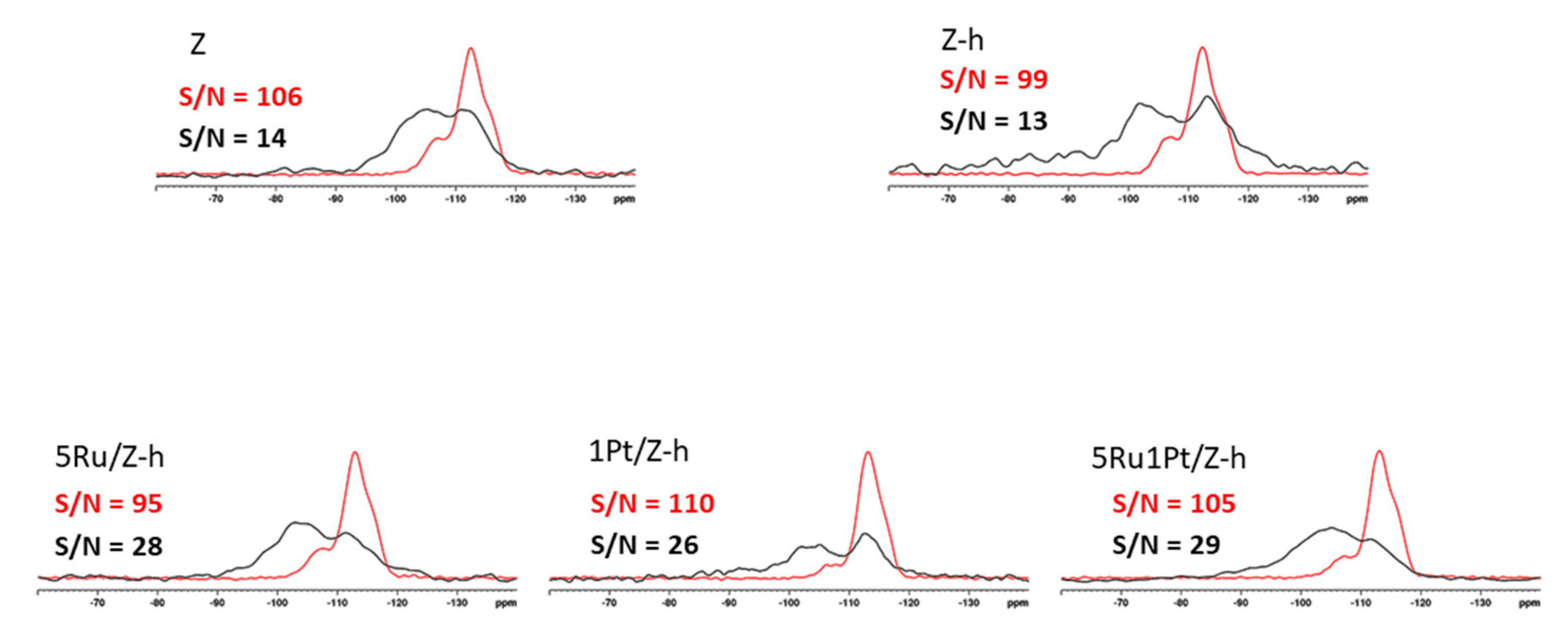
2.1.2. NMR Analysis
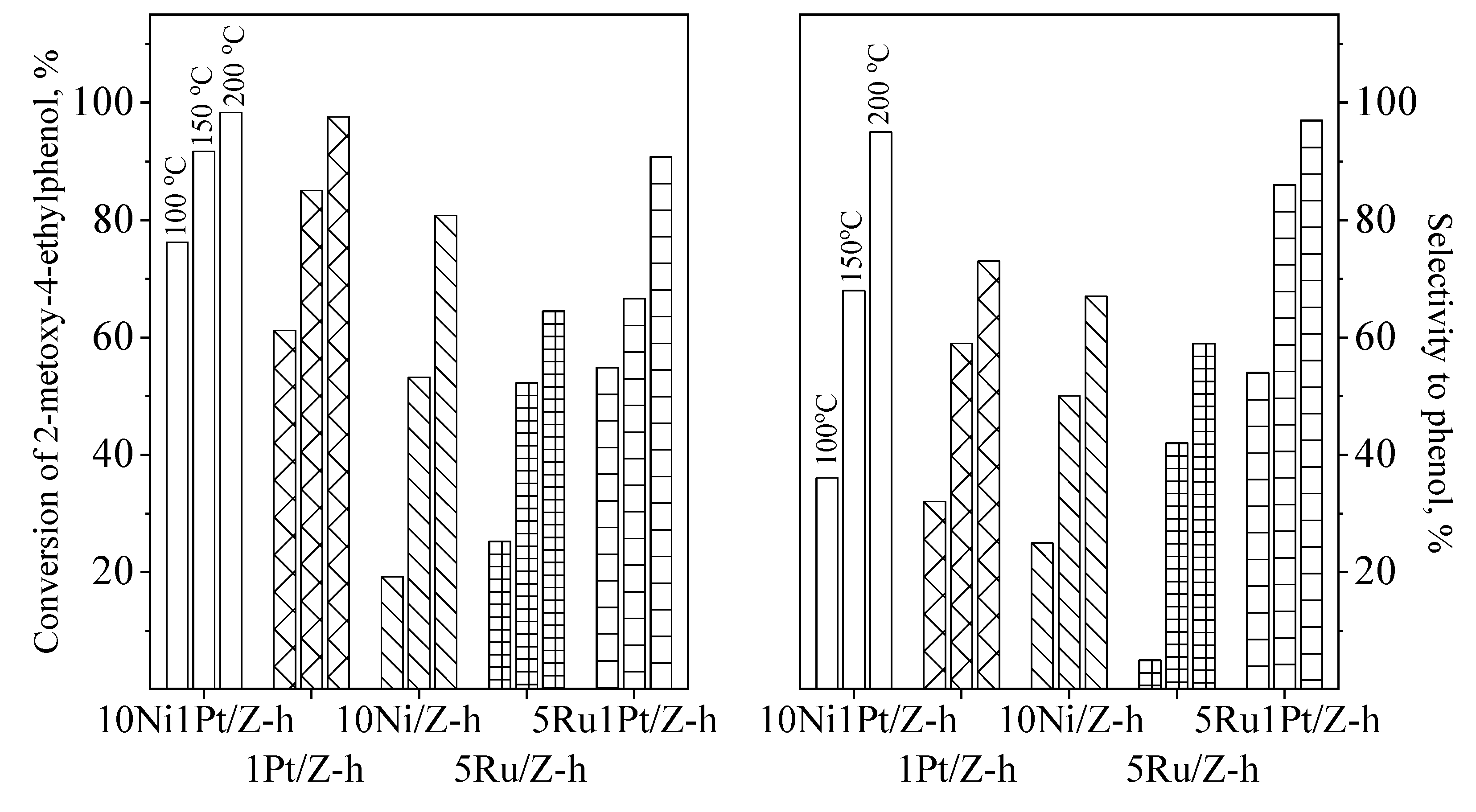
2.2. Catalytic Activity for Phenol Preparation
3. Experimental
3.1. Materials
3.2. Synthesis of ZSM-5 Zeolite
3.3. Preparation of Hierarchical ZSM-5
3.4. Modification with Ni, Ru or/and Pt Nanoparticles of Hierarchical ZSM-5 Zeolite
3.5. Characterization
3.6. Catalytic Activity Measurements
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Sample Availability
References
- Luque, R.; Campelo, J.; Clark, J. Handbook of Biofuels Production: Processes and Technologies; Woodhead Publishing Series in Energy; Elsevier: Amsterdam, The Netherlands, 2013; Volume 15. [Google Scholar]
- Lin, C.S.K.; Pfaltzgraff, L.A.; Herrero-Davila, L.; Mubofu, E.B.; Abderrahim, S.; Clark, J.H.; Koutinas, A.; Kopsahelis, N.; Stamatelatou, K.; Dickson, F.; et al. Food waste as a valuable resource for the production of chemicals, materials and fuels. Current situation and global perspective. Energy Environ. Sci. 2013, 6, 426–464. [Google Scholar] [CrossRef]
- Romero, Y.; Richard, F.; Brunet, S. Hydrodeoxygenation of 2-ethylphenol as a model compound of bio-crude over sulfided Mo-based catalysts: Promoting effect and reaction mechanism. Appl. Catal. B 2010, 98, 213–223. [Google Scholar] [CrossRef]
- Zhang, J.; Lombardo, L.; Gözaydın, G.; Dyson, P.J.; Yan, N. Single-step conversion of lignin monomers to phenol: Bridging the gap between lignin and high-value chemicals Chinese. J. Catal. 2018, 39, 1445–1452. [Google Scholar] [CrossRef]
- Serra, J.M.; Guillon, E.; Corma, A. A rational design of alkyl-aromatics dealkylation–transalkylation catalysts using C8 and C9 alkyl-aromatics as reactants. J. Catal. 2004, 227, 459–469. [Google Scholar] [CrossRef]
- Gamliela, D.P.; Karakalos, S.; Valla, J.A. Liquid phase hydrodeoxygenation of anisole, 4-ethylphenol and benzofuran using Ni, Ru and Pd supported on USY zeolite. Appl. Catal. A 2018, 559, 20–29. [Google Scholar] [CrossRef]
- Shao, Y.; Xia, Q.; Dong, L.; Liu, X.; Han, X.; Parker, S.F.; Cheng, Y.; Daemen, L.L.; Ramirez-Cuesta, A.J.; Yang, S.; et al. Selective production of arenes via direct lignin upgrading over a niobium-based catalyst. Nat. Commun. 2017, 8, 16104. [Google Scholar] [CrossRef] [PubMed]
- Shuai, L.; Amiri, M.T.; Questell-Santiago, Y.M.; Héroguel, F.; Li, Y.; Kim, H.; Meilan, R.; Chapple, C.; Ralph, J.; Luterbacher, J.S. Formaldehyde stabilization facilitates lignin monomer production during biomass depolymerisation. Science 2016, 354, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Lancefield, C.S.; Ojo, O.S.; Tran, F.; Westwood, N.J. Isolation of Functionalized Phenolic Monomers through Selective Oxidation and C-O Bond Cleavage of the β-O-4 Linkages in Lignin. Angew. Chem. Int. Ed. 2015, 54, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Deuss, P.J.; Scott, M.; Tran, F.; Westwood, N.J.; de Vries, J.G.; Barta, K. Aromatic monomers by in situ conversion of reactive intermediates in the acid-catalyzed depolymerization of lignin. J. Am. Chem. Soc. 2015, 137, 7456–7467. [Google Scholar] [CrossRef] [PubMed]
- Onwudili, J.A.; Williams, P.T. Catalytic depolymerization of alkali lignin in subcritical water: Influence of formic acid and Pd/C catalyst on the yields of liquid monomeric aromatic products. Green Chem. 2014, 16, 4740–4748. [Google Scholar] [CrossRef]
- Jung, K.B.; Lee, J.; Ha, J.M.; Lee, H.; Suh, D.J.; Jun, C.H.; Jae, J. Effective hydrodeoxygenation of lignin-derived phenols using bimetallic RuRe catalysts: Effect of carbon supports. Catal. Today 2018, 303, 191–199. [Google Scholar] [CrossRef]
- Renders, T.; Schutyser, W.; Van den Bosch, S.; Koelewijn, S.F.; Vangeel, T.; Courtin, C.M.; Sels, B.F. Influence of Acidic (H3PO4) and Alkaline (NaOH) Additives on the Catalytic Reductive Fractionation of Lignocellulose. ACS Catal. 2016, 6, 2055–2066. [Google Scholar] [CrossRef]
- Liao, Y.; d’Halluin, M.; Makshina, E.; Verboekend, D.; Sels, B.F. Shape selectivity vapor-phase conversion of lignin-derived 4-ethylphenol to phenol and ethylene over acidic aluminosilicates: Impact of acid properties and pore constraint. Appl. Catal. B 2018, 234, 117–129. [Google Scholar] [CrossRef]
- Verboekend, D.; Liao, Y.; Schutyser, W.; Sels, B.F. Alkylphenols to phenol and olefins by zeolite catalysis: A pathway to valorize raw and fossilized lignocellulose. Green Chem. 2016, 18, 297–306. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Umezawa, T.; Nakasaka, Y.; Masuda, T. Conversion of alkylphenol to phenol via transalkylation using zeolite catalysts. Catal. Today 2020, 347, 110–114. [Google Scholar] [CrossRef]
- Ohta, H.; Feng, B.; Kobayashi, H.; Haraa, K.; Fukuoka, A. Selective hydrodeoxygenation of lignin-related 4-propylphenol into n-propylbenzene in water by Pt-Re/ZrO2 catalysts. Catal. Today 2014, 234, 139–144. [Google Scholar] [CrossRef]
- Shin, E.-J.; Keane, M.A. Gas-phase hydrogenation/hydrogenolysis of phenol over supported nickel catalysts. Ind. Eng. Chem. Res. 2000, 39, 883–892. [Google Scholar] [CrossRef]
- Sun, J.; Karim, A.M.; Zhang, H.; Kovarik, L.; Li, X.S.; Hensley, A.J.; McEwen, J.-S.; Wang, Y. Carbon-supported bimetallic Pd–Fe catalysts for vapor-phase hydrodeoxygenation of guaiacol. J. Catal. 2013, 306, 47–57. [Google Scholar] [CrossRef]
- Jin, S.; Xiao, Z.; Li, C.; Chen, X.; Wang, L.; Xing, J.; Li, W.; Liang, C. Catalytic hydrodeoxygenation of anisole as lignin model compound over supported nickel catalysts. Catal. Today 2014, 234, 125–132. [Google Scholar] [CrossRef]
- Echeandia, S.; Pawelec, B.; Barrio, V.L.; Arias, P.L.; Cambra, J.F.; Loricera, C.V.; Fierro, J.L.G. Enhancement of phenol hydrodeoxygenation over Pd catalysts supported on mixed HY zeolite and Al2O3. An approach to O-removal from bio-oils. Fuel 2014, 117, 1061–1073. [Google Scholar] [CrossRef]
- Fakin, T.; Ristic, A.; Mavrodinova, V.; Zabukovec Logar, N. Highly crystalline binder-free ZSM-5 granules preparation. Micropor. Mesopor. Mater. 2015, 213, 108–117. [Google Scholar] [CrossRef]
- Berenguer, A.; Gutiérrez-Rubio, S.; Linares, M.; Ochoa-Hernández, C.; Moreno, I.; García-Fierro, J.L.; Coronado, J.M.; Serrano, D.P.; Pizarro, P. On the Feasibility of Using Hierarchical ZSM-5 and Beta Zeolites as Supports of Metal Phosphides for Catalytic Hydrodeoxygenation of Phenol. Energy Technol. 2019, 7, 1900214. [Google Scholar] [CrossRef]
- Li, J.; Wang, L.; Zhang, D.; Qian, J.; Liu, L. One-step synthesis of hierarchical ZSM-5 zeolites and their catalytic performance on the conversion of methanol to aromatics. Reac. Kinet. Mech. Catal. 2020, 130, 519–530. [Google Scholar] [CrossRef]
- Li, W.; Li, F.; Wang, H.; Liao, M.; Li, P.; Zheng, J.; Tu, C.; Li, R. Hierarchical mesoporous ZSM-5 supported nickel catalyst for the catalytic hydrodeoxygenation of anisole to cyclohexane. Mol. Catal. 2020, 480, 1106422. [Google Scholar] [CrossRef]
- Jenkins, R.; Snyder, R.L. Introduction to X-ray Powder Diffractometry; John Wiley & Sons Inc.: Hoboken, NJ, USA, 1996; pp. 89–91. [Google Scholar]
- Holder, C.F.; Schaak, R.E. Tutorial on Powder X-ray Diffraction for Characterizing Nanoscale Materials. ACSNano 2019, 13, 7356–7359. [Google Scholar] [CrossRef]
- Landau, M.V.; Tavor, D.; Regev, O.; Kaliya, M.L.; Herskowitz, M.; Valtchev, V.; Mintova, S. Colloidal nanocrystals of zeolite β stabilized in alumina matrix. Chem. Mater. 1999, 8, 2030–2037. [Google Scholar] [CrossRef]
- Zhang, F.; Guo, X.; Wang, X.; Li, G.; Zhao, Q.; Bao, X.; Han, X.; Lin, L. Preparation of titanium-containing zeolites with MEL structure from B-ZSM-11 and their characterization. Appl. Catal. A 2000, 192, 157–163. [Google Scholar] [CrossRef]
- Brodie-Linder, N.; Besse, R.; Audonnet, F.; LeCaer, S.; Deschamps, J.; Impéror-Clerc, M.; Alba-Simionesco, C. The key to control Cu II loading in silica based mesoporous materials. Micropor. Mesopor. Mater. 2010, 132, 518–525. [Google Scholar] [CrossRef]
- Ma, Y.-K.; Rigolet, S.; Michelin, L.; Paillaud, J.-L.; Mintova, S.; Khoerunnisa, F.; Jean Daou, T.; Ng, E.-P. Facile and fast determination of Si/Al ratio of zeolites using FTIR spectroscopy technique. Appl. Catal. A 2018, 559, 20–29. [Google Scholar] [CrossRef]
- Akçay, M. The surface acidity and characterization of Fe-montmorillonite probed by in situ FT-IR spectroscopy of adsorbed pyridine. Appl. Catal. A 2005, 294, 156–160. [Google Scholar] [CrossRef]
- Parry, E.P. An infrared Study of Pyridine Adsorbed on Acidic Solids. Characterization of Surface Acidity. J. Catal. 1963, 2, 371–379. [Google Scholar] [CrossRef]
- Engelhardt, G.; Lohse, U.; Lippmaa, E.; Tarmak, M.; Mägi, M.Z. 29Si NMR -Untersuchungen zur Verteilung der Silicium-und Aluminiumatome im Alumosilicatgitter von Zeolithen mit Faujasit-Struktur. Anorg. Allg. Chem. 1981, 482, 49–64. [Google Scholar] [CrossRef]
- Massiot, D.; Fayon, F.; Capron, M.; King, I.; Le Calve, S.; Alonso, B.; Durand, J.O.; Bujoli, B.; Gan, Z.; Hoatson, G. Modelling one- and two-dimensional solid-state NMR spectra. Magn. Reson. Chem. 2000, 40, 70–76. [Google Scholar] [CrossRef]
- Hunger, M. Solid-State NMR Spectroscopy, Chapter 2. In Zeolite Characterization and Catalysis; Chester, A.W., Derouane, E.G., Eds.; Springer: Dordrecht, The Netherlands, 2009; pp. 65–105. [Google Scholar]
- Brunner, E.; Ernst, H.; Freude, D.; Frohlich, T.; Hunger, M.; Pfeifer, H. Magic Angle Spinning NMR Studies of Acid Sites in Zeolite H-ZSM-5. J. Catal. 1991, 127, 34–41. [Google Scholar] [CrossRef]
- van Bokhoven, J.A.; Koningsberger, D.C.; Kunkeler, P.; van Bekkum, H.; Kentgens, A.P.M. Stepwise Dealumination of Zeolite Beta at Specific T-Sites Observed with 27Al MAS and 27Al MQ MAS NMR. J. Am. Chem. Soc. 2000, 122, 12842–12847. [Google Scholar] [CrossRef]
- Bourgeat-Lami, E.; Massiani, P.; Di Renzo, F.; Espiau, P.; Fajula, F.; Des Courières, T. Study of the state of aluminium in zeolite-β. Appl. Catal. 1991, 72, 139–152. [Google Scholar] [CrossRef]
- Campbell, S.M.; Bibby, D.M.; Coddington, J.M.; Howe, R.F.; Meinhold, R.H. Dealumination of HZSM-5 Zeolites, I. Calcination and Hydrothermal Treatment. J. Catal. 1996, 161, 338–349. [Google Scholar] [CrossRef]
- Yoshida, K.; Okumura, K.; Miyao, T.; Naito, S.; Ito, S.I.; Kunimori, K.; Tomishige, K. Oxidative steam reforming of methane over Ni/a-Al2O3 modified with trace noble metals. Appl. Catal. A 2009, 358, 186–192. [Google Scholar] [CrossRef]
- Abu Bakar, N.H.H.; Bettahar, M.M.; Abu Bakar, M.; Monteverdi, S.; Ismail, J. Low temperature activation of Pt/Ni supported MCM-41 catalysts for hydrogenation of benzene. J. Mol. Catal. A 2010, 333, 11–19. [Google Scholar] [CrossRef]
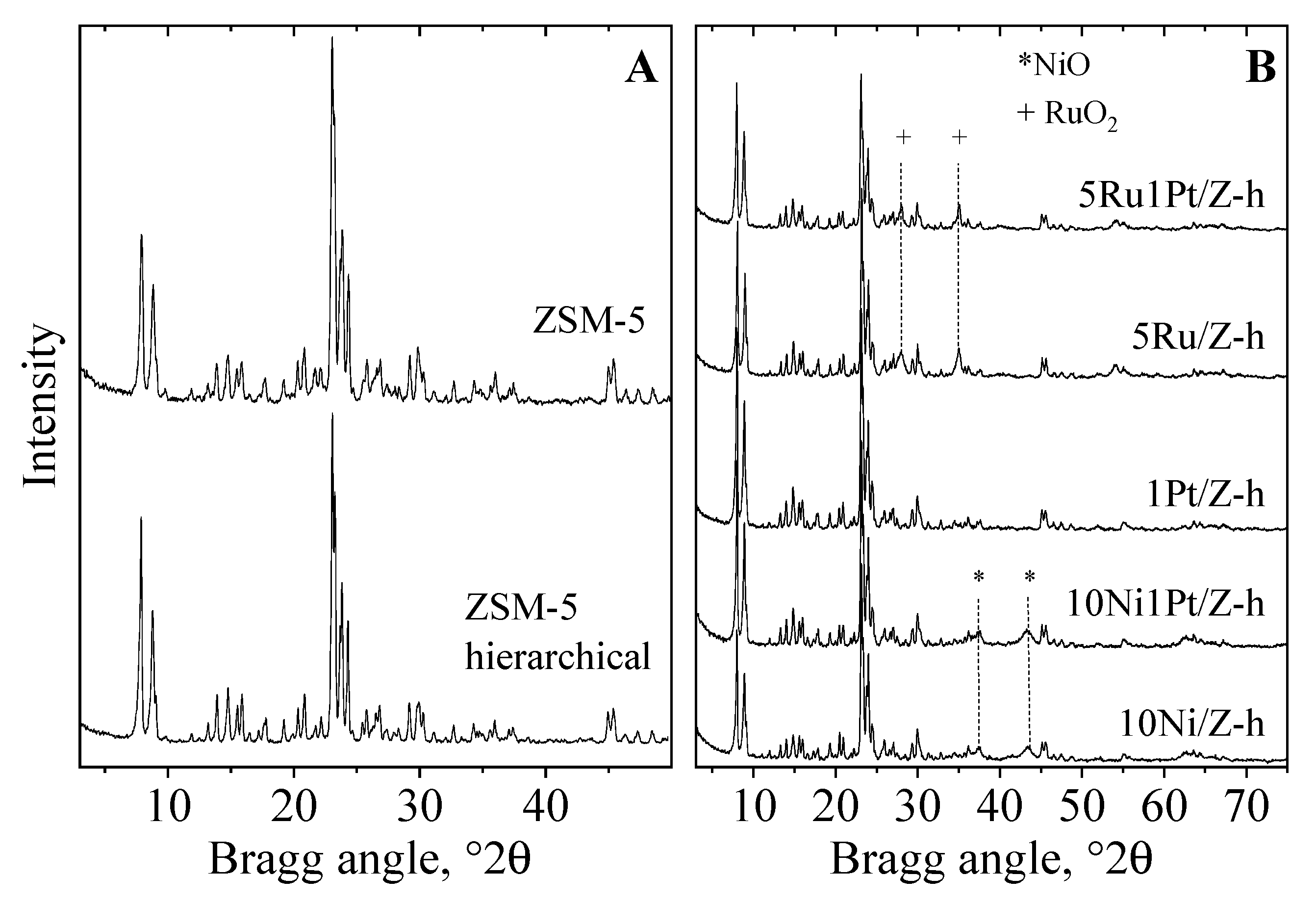
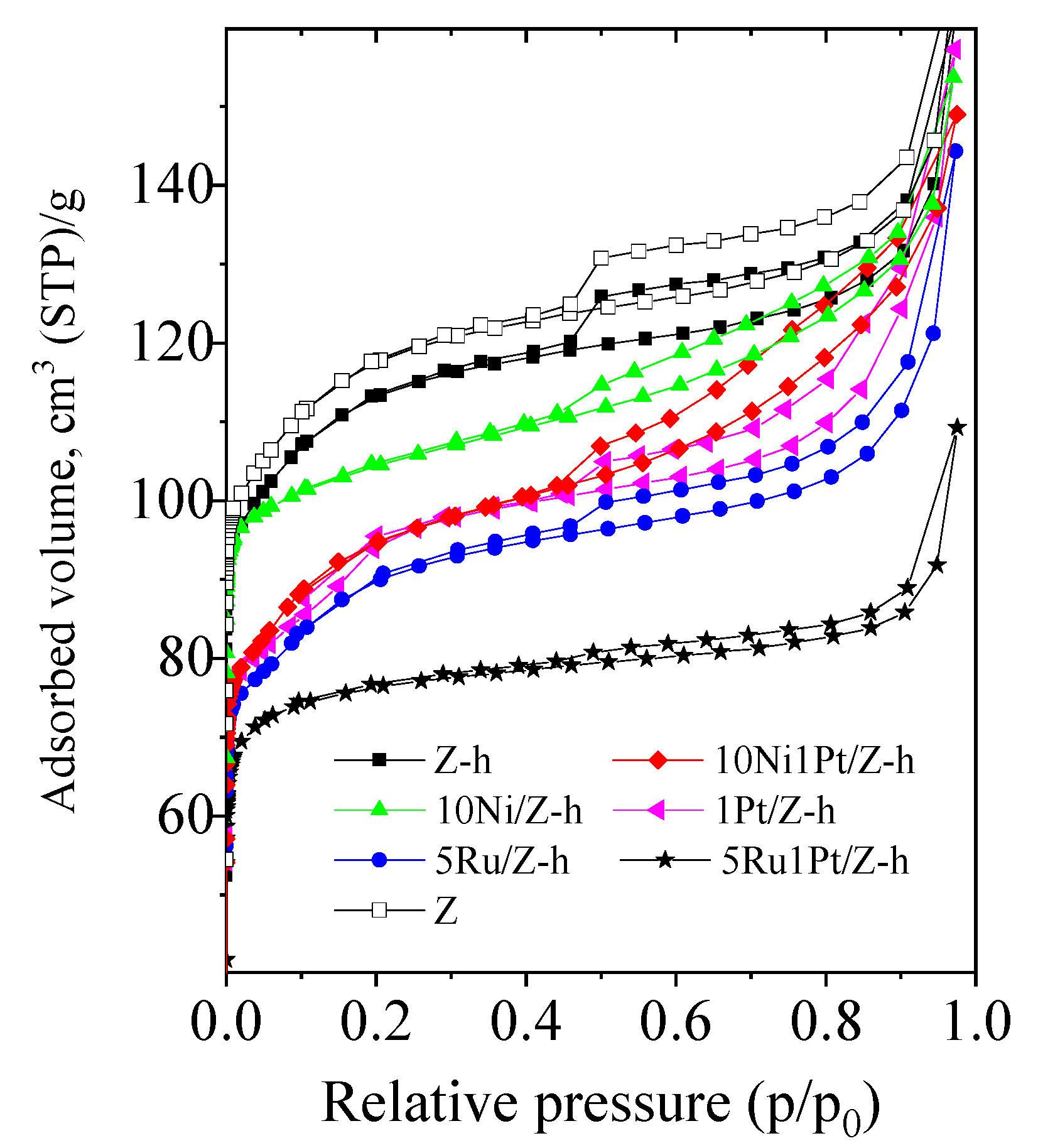
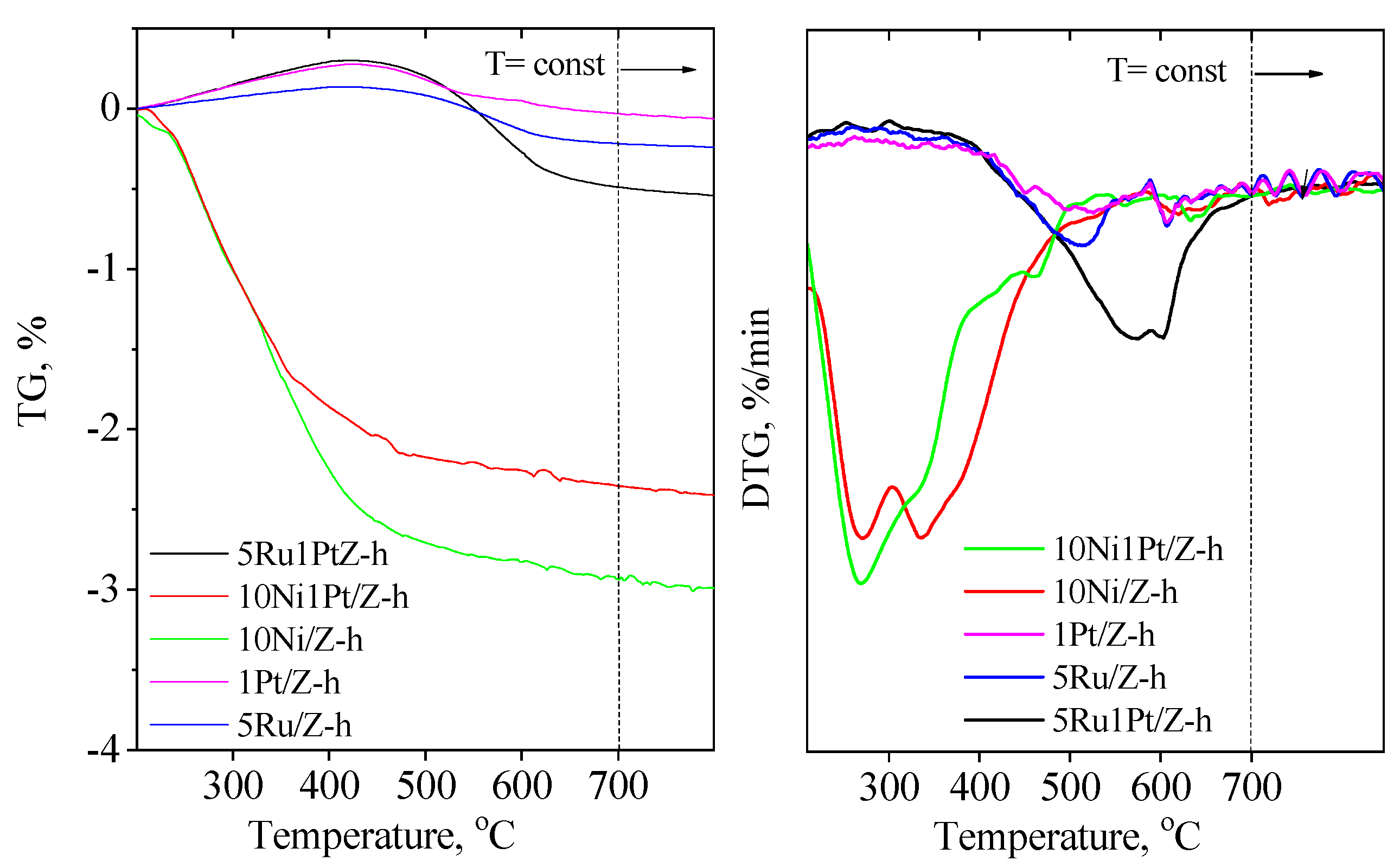
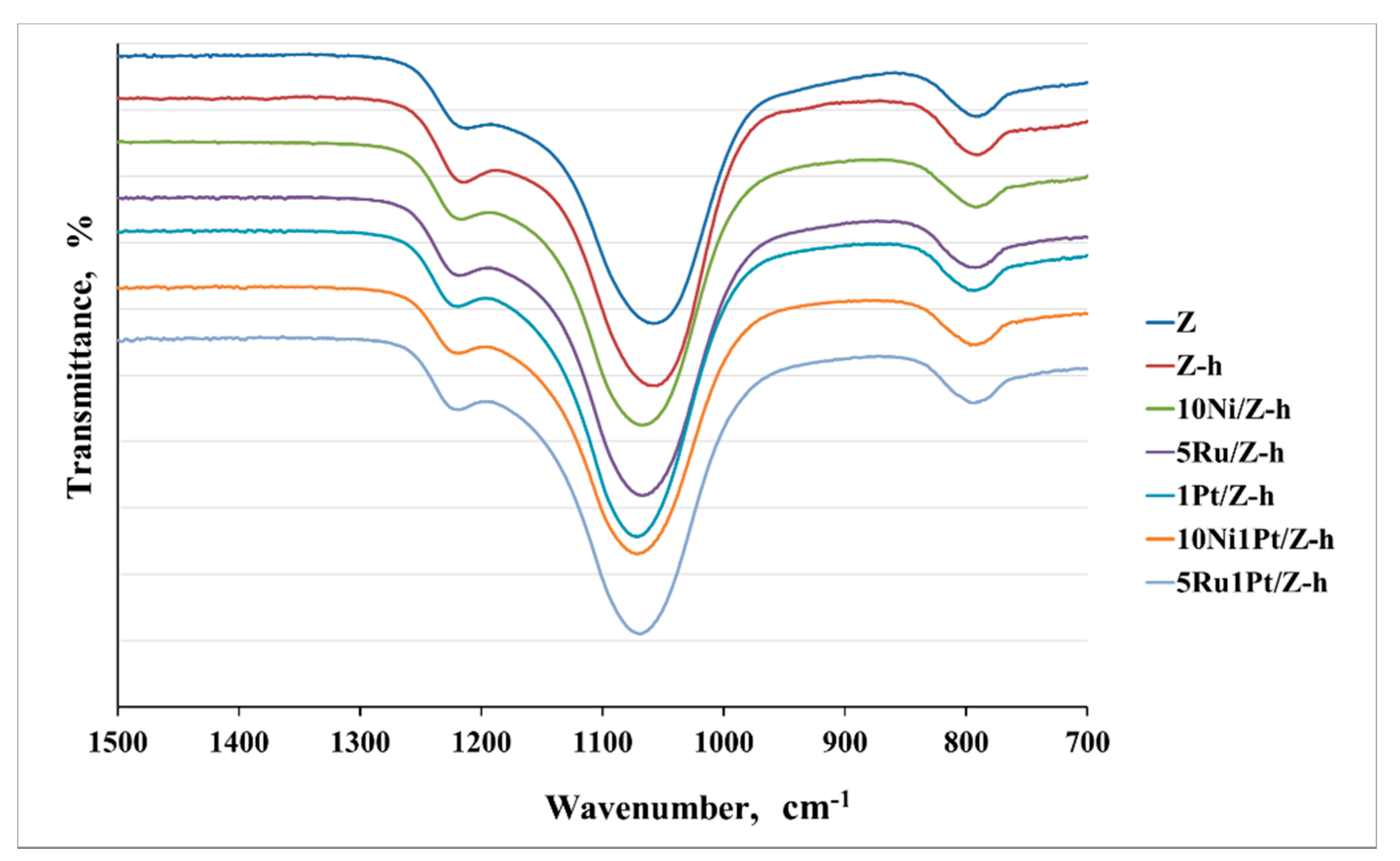
| Samples | BET Surface Area, (m2/g) | Pore Volume, (cm3/g) | Mesopore Volume, (cm3/g) | Crystallite Size, (nm) | Reducibility a, (%) |
|---|---|---|---|---|---|
| Z | 525 | 0.35 | 0.05 | - | - |
| Z-h | 410 | 0.27 | 0.17 | - | - |
| 10Ni/Z-h | 351 | 0.26 | 0.18 | 8 (NiO) | 80 (NiO) |
| 10Ni1Pt/Z-h | 335 | 0.32 | 0.19 | 8 (NiO) | 100 (NiO) |
| 1Pt/Z-h | 404 | 0.32 | 0.20 | - | 100 |
| 5Ru/Z-h | 330 | 0.33 | 0.16 | 23 (RuO2) | 60 (RuO2) |
| 5Ru1Pt/Z-h | 282 | 0.26 | 0.15 | 35 (RuO2) | 77 (RuO2) |
| Samples | Q4(1Al) −107 ppm | Q4(0Al) −113 ppm | Q4(0Al) −115 ppm | Si/Al | FAl 62–58 ppm % | FAl distorted 40–38 ppm, % | EFAl 7–0 ppm % |
|---|---|---|---|---|---|---|---|
| Z | 17 | 64 | 19 | 24 | 91 | -/- | 9 |
| Z-h | 19 | 65 | 16 | 21 | 72 | -/- | 18 |
| 5Ru/Z-h | 20 | 60 | 20 | 20 | 80 | 11 | 9 |
| 10Ni/Z-h | 13 + 7 | 49 | 31 | 20 | 100 | -/- | -/- |
| 1Pt/Z-h | 3 + 8 | 75 | 14 | 36 | 53 | 20 | 5 + 13 + 9 * |
| 5Ru1Pt/Z-h | 10 | 68 | 22 | 40 | 41 | 50 | 1 + 8 |
| 10Ni1Pt/Z-h | 14 | 71 | 15 | 29 | 32 | 62 | 4 + 3* |
| Samples | 4-Ethylphenol Dealkylation, Conversion, % | Transalkylation of 4-Ethylphenol with Toluene at 300 °C | ||||
|---|---|---|---|---|---|---|
| 170 °C | 200 °C | 250 °C | Conversion, % | Selectivity to Phenol, % | Selectivity to Xylenes, % | |
| Z | 11.6 | 84.0 | 87.7 (85.7) * | 75.5 (65.4) * | 54.7 (53.6) * | 45.3 (46.4) * |
| Z-h | 38.2 | 87.4 | 90.1 (89.8) * | 86.5 (85.4) * | 35.4 (34.8) * | 64.6 (65.2) * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Popova, M.; Szegedi, Á.; Oykova, M.; Lazarova, H.; Koseva, N.; Mihályi, M.R.; Shestakova, P. Selective Production of Phenol on Bifunctional, Hierarchical ZSM-5 Zeolites. Molecules 2021, 26, 3576. https://doi.org/10.3390/molecules26123576
Popova M, Szegedi Á, Oykova M, Lazarova H, Koseva N, Mihályi MR, Shestakova P. Selective Production of Phenol on Bifunctional, Hierarchical ZSM-5 Zeolites. Molecules. 2021; 26(12):3576. https://doi.org/10.3390/molecules26123576
Chicago/Turabian StylePopova, Margarita, Ágnes Szegedi, Manuela Oykova, Hristina Lazarova, Neli Koseva, Magdolna R. Mihályi, and Pavletta Shestakova. 2021. "Selective Production of Phenol on Bifunctional, Hierarchical ZSM-5 Zeolites" Molecules 26, no. 12: 3576. https://doi.org/10.3390/molecules26123576
APA StylePopova, M., Szegedi, Á., Oykova, M., Lazarova, H., Koseva, N., Mihályi, M. R., & Shestakova, P. (2021). Selective Production of Phenol on Bifunctional, Hierarchical ZSM-5 Zeolites. Molecules, 26(12), 3576. https://doi.org/10.3390/molecules26123576







