Anticorrosive and Microbial Inhibition Performance of a Coating Loaded with Andrographis paniculata on Stainless Steel in Seawater
Abstract
1. Introduction
2. Results and Discussion
2.1. Optical Studies
2.1.1. Fourier-Transform Infrared (FTIR) Spectroscopy
2.1.2. X-Ray Diffraction (XRD)
2.1.3. High-Performance Liquid Chromatography
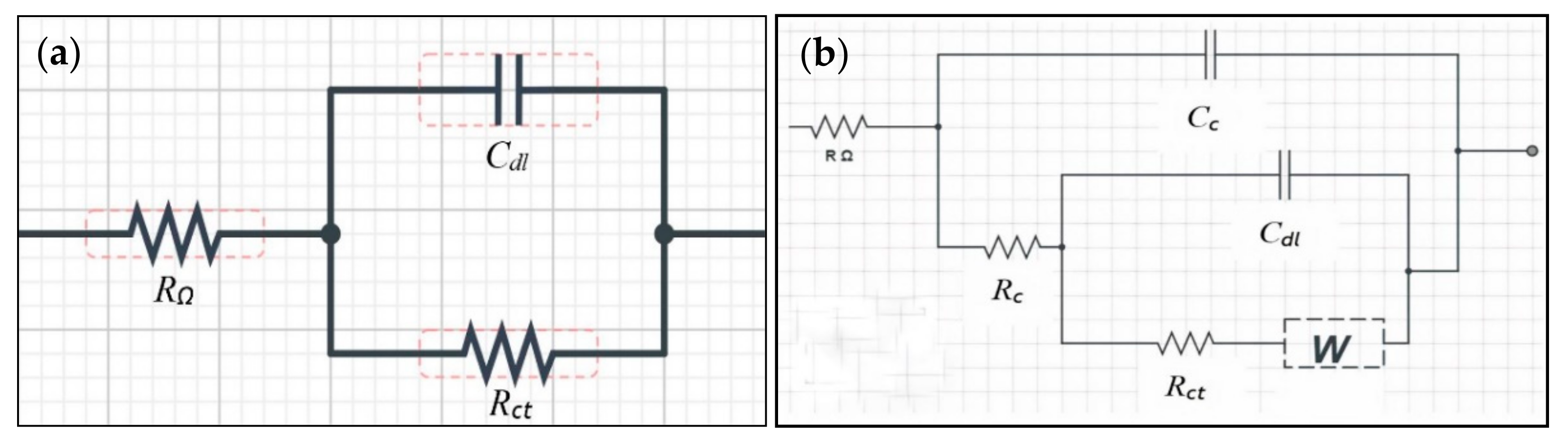
2.2. Electrochemical Impedance Spectroscopy (EIS)
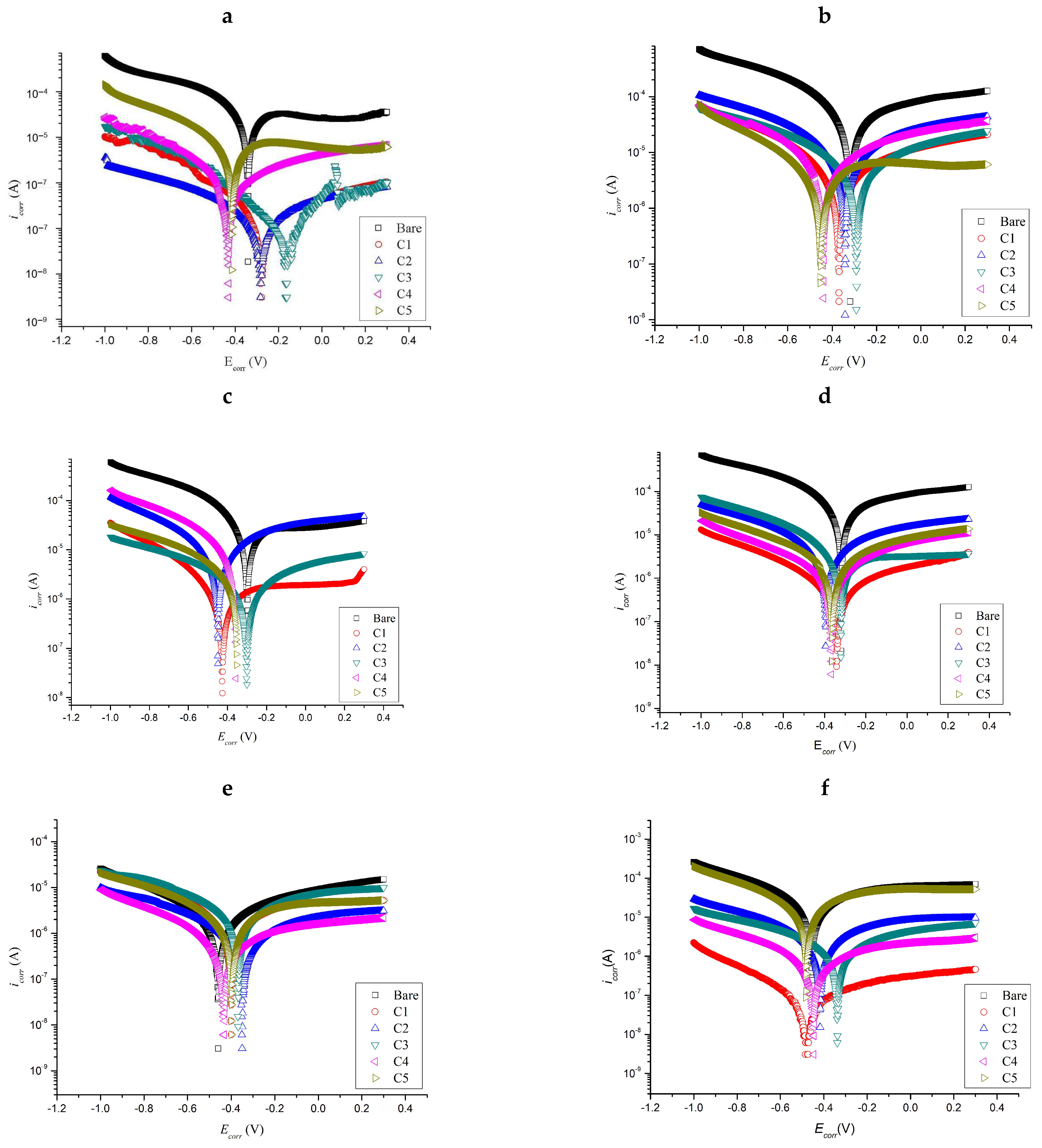
2.3. Tafel Polarization
2.4. Morphology and Element Distribution Studies
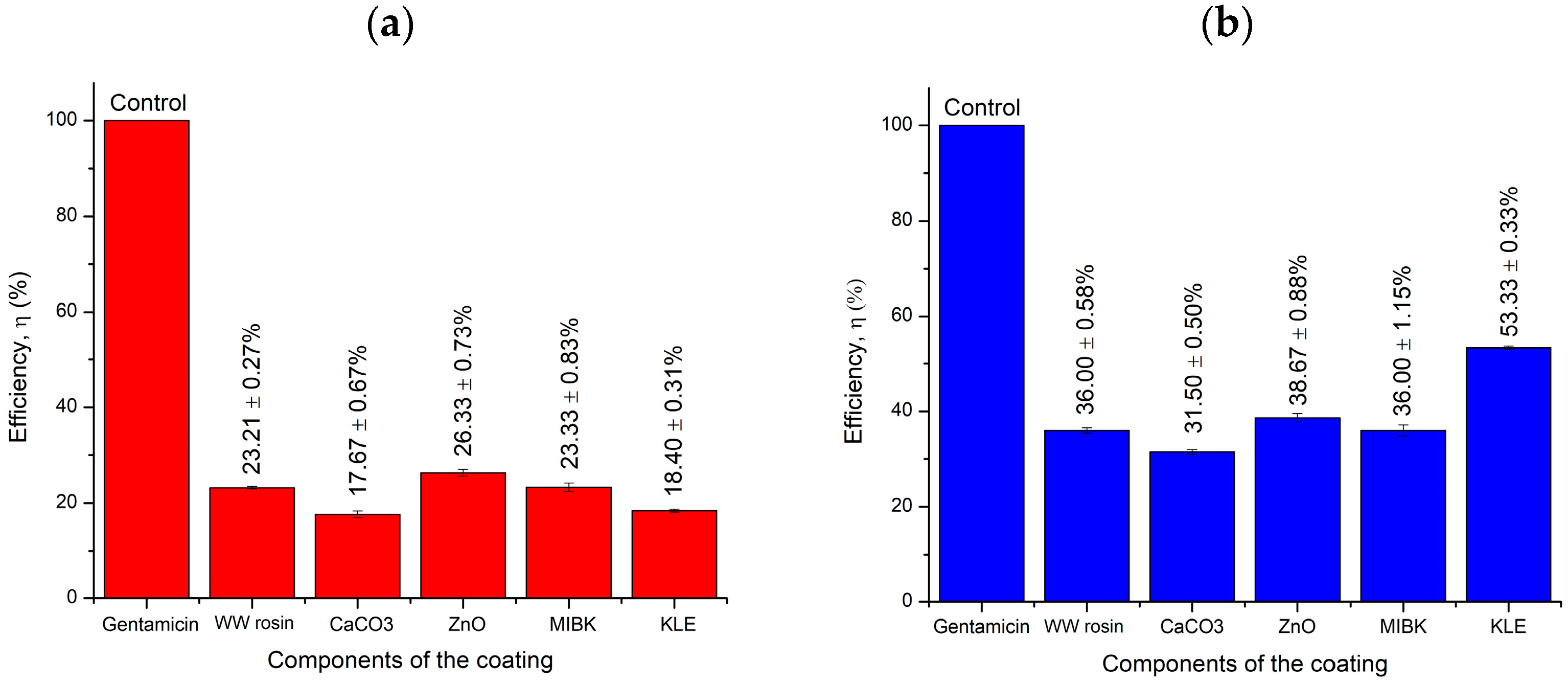
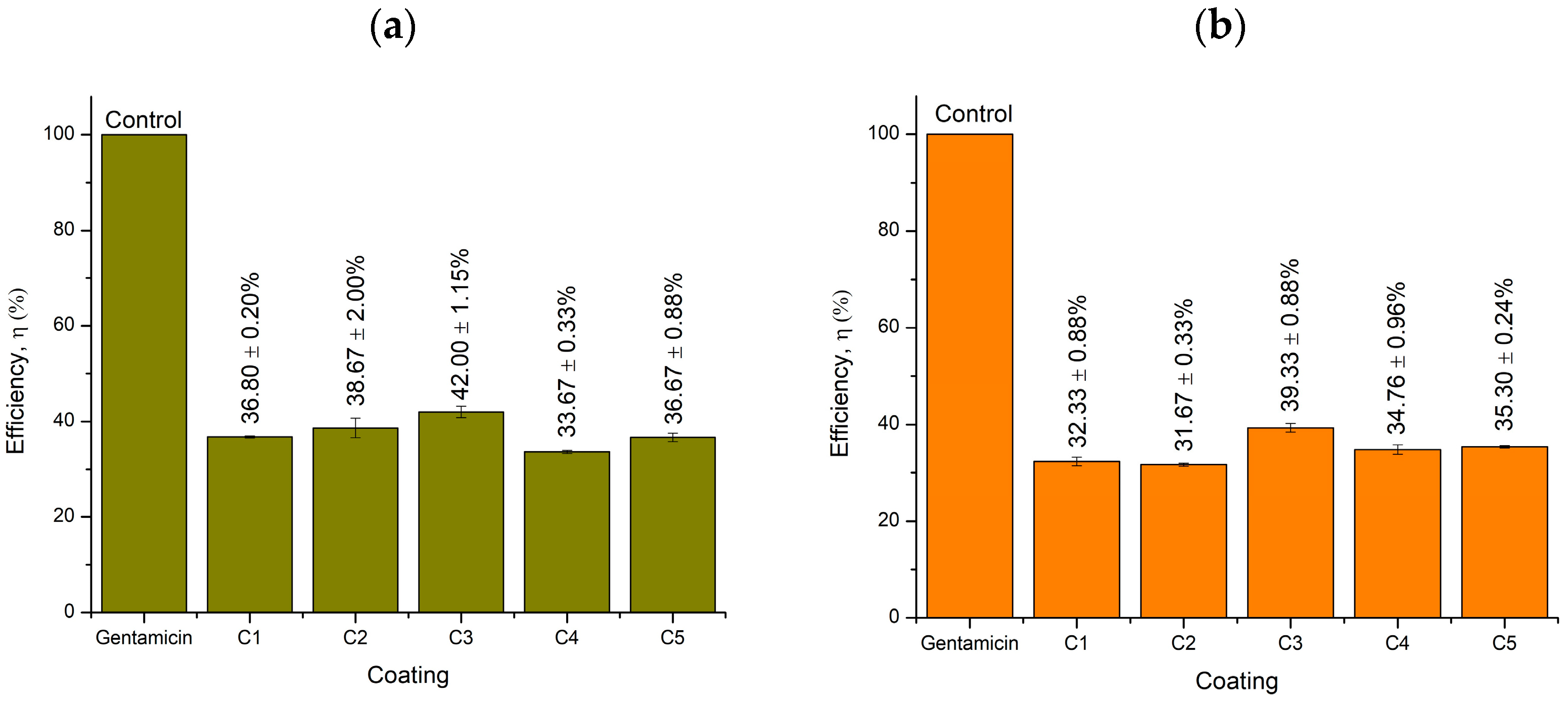
2.5. Well Diffusion Antimicrobial Test
3. Materials and Methods
3.1. Sample Preparation
3.2. Extract Preparation
3.3. Coating Formulation
3.4. Characterization of the Extract
3.4.1. X-Ray Diffraction (XRD)
3.4.2. Fourier-Transform Infrared Spectroscopy (FTIR)
3.4.3. High-Performance Liquid Chromatography (HPLC)
3.5. Immersion Test at the Bay Area of Setiu Terengganu
3.6. Electrochemical Studies
3.6.1. Electrochemical Impedance Spectroscopy (EIS)
3.6.2. Potentiodynamic Polarization (PP)
3.7. Morphology Studies
3.8. Evaluation of Antibacterial Activity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Sharma, S.K.; Peter, A.; Obot, I.B. Potential of Azadirachta indica as a green corrosion inhibitor against mild steel, aluminum, and tin: A review. J. Anal. Sci. Technol. 2015, 6, 1–16. [Google Scholar] [CrossRef]
- Palou, R.M.; Olivares-Xomelt, O.; Likhanova, N.V. Environmentally friendly corrosion inhibitors, in developments in corrosion protection. IntechOpen 2014, 19, 431–465. Available online: https://www.intechopen.com/books/developments-in-corrosion-protection/environmentally-friendly-corrosion-inhibitors (accessed on 2 April 2021).
- Popoola, L.T.; Grema, A.S.; Latinwo, G.K.; Gutti, B.; Balogun, A.S. Corrosion problems during oil and gas production and its mitigation. Int. J. Ind. Chem. 2013, 4, 1–15. [Google Scholar] [CrossRef]
- Fauchais, P.; Vardelle, A. Thermal sprayed coatings used against corrosion and corrosive wear, in advanced plasma spray applications. IntechOpen 2012, 1, 3–38. Available online: https://www.intechopen.com/books/advanced-plasma-spray-applications/thermal-sprayed-coatings-used-against-corrosion-and-corrosive-wear (accessed on 2 April 2021).
- Zakowski, K.; Narozny, M.; Szocinski, M.; Darowicki, K. Influence of water salinity on corrosion risk—The case of the southern Baltic Sea coast. Environ. Monit. Assess. 2014, 186, 4871–4879. [Google Scholar] [CrossRef]
- Amand, S.; Musiani, M.; Orazem, M.E.; Pébère, N.; Tribollet, B.; Vivier, V. Constant-phase-element behavior caused by inhomogeneous water uptake in anticorrosion coatings. Electrochim. Acta. 2013, 87, 693–700. [Google Scholar] [CrossRef]
- Deng, S.; Wu, J.; Li, Y.; Wang, G.; Chai, K.; Yu, A.; Liu, F. Effect of Bacillus flexus on the degradation of epoxy resin varnish coating in seawater. Int. J. Electrochem. Sci. 2019, 14, 315–328. [Google Scholar] [CrossRef]
- Li, S.; Li, L.; Qu, Q.; Kang, Y.; Zhu, B.; Yu, D.; Huang, R. Extracellular electron transfer of Bacillus Cereus biofilm and its effect on the corrosion behaviour of 316l stainless steel. Colloids Surf. B 2019, 173, 139–147. [Google Scholar] [CrossRef]
- Enning, D.; Garrelfs, J. Corrosion of iron by sulfate-reducing bacteria: New views of an old problem. Appl. Environ. Microbiol. 2014, 80, 1226–1236. [Google Scholar] [CrossRef]
- Hagenauer, A.; Hilpert, R.; Hack, T. Microbiological investigations of corrosion damages in aircraft. Mater. Corros. 1994, 45, 355–360. [Google Scholar] [CrossRef]
- Hamzah, E.; Hussain, M.; Ibrahim, Z.; Abdolahi, A. Corrosion behaviour of carbon steel in sea water medium in presence of P. Aeruginosa bacteria. Arab. J. Sci. Eng. 2014, 39, 6863–6870. [Google Scholar] [CrossRef]
- Li, H.; Zhang, S.; Qiang, Y. Corrosion retardation effect of a green cauliflower extract on copper in H2SO4 solution: Electrochemical and theoretical explorations. J. Mol. Liq. 2021, 321, 1–13. [Google Scholar] [CrossRef]
- Hajar, H.; Zulkifli, F.; Suriani, M.; Sabri, M.G.M.; Nik, W.W. Lawsonia inermis extract enhances performance of corrosion protection of coated mild steel in seawater. EDP Sci. 2016, 78, 01091. [Google Scholar] [CrossRef]
- Ikhmal, W.M.K.W.M.; Maria, M.F.M.; Rafizah, W.A.W.; Norsani, W.N.W.M.; Sabri, M.G.M. Corrosion inhibition of mild steel in seawater through green approach using Leucaena leucocephala leaves extract. Int. J. Corros. Scale Inhib. 2019, 8, 628–643. [Google Scholar]
- Hajar, H.M.; Zulkifli, F.; Sabri, M.G.M.; Norsani, W.N.W.M. Protection against corrosion of aluminum alloy in marine environment by Lawsonia inermis. Int. J. Corros. 2016, 2016, 1–6. [Google Scholar] [CrossRef]
- Nasr-Esfahani, M.; Pourriahi, M.; Motalebi, A.; Zendehdel, M. Improvement of the corrosion performance of 304l stainless steel by a nanostructure hybrid coating/henna extract. Anti-Corros. Methods M. 2013, 61, 1–9. [Google Scholar] [CrossRef]
- Ibrahim, T.H.; Chehade, Y.; Zour, M.A. Corrosion inhibition of mild steel using potato peel extract in 2m HCl solution. Int. J. Electrochem. Sci. 2011, 6, 6542–6556. [Google Scholar]
- Maria, M.F.F.; Ikhmal, W.M.K.W.M.; Amirah, M.; Manja, S.; Syaizwadi, S.M.; Chan, K.; Sabri, M.G.M.; Adnan, A. Green approach in anticorrosion coating by using Andrographis paniculata leaves extract as additives of stainless steel 316l in seawater. Int. J. Corros. Scale Inhib. 2019, 8, 644–658. [Google Scholar]
- Shirisha, K.; Mastan, M. Andrographis paniculata and its bioactive phytochemical constituents for oxidative damage: A systemic review. Pharmacophore 2013, 4, 212–229. [Google Scholar]
- Mussard, E.; Cesaro, A.; Lespessailles, E.; Legrain, B.; Berteina-Raboin, S.; Toumi, H. Andrographolide, a natural antioxidant: An update. Antioxidants 2019, 8, 571. [Google Scholar] [CrossRef]
- Chakraborty, S.; Biswas, S.; Manna, J.S.; Das, S.; Dey, R. Sol-gel derived silica-gel as a controlled delivery system of Andrographis paniculata extract and its antimicrobial efficacy. Trans. Indian Inst. Met. 2011, 64, 189. [Google Scholar] [CrossRef]
- Sajeeb, B.; Kumar, U.; Halder, S.; Bachar, S.C. Identification and quantification of andrographolide from Andrographis paniculata (Burm. f.) wall. ex nees by RP-HPLC method and standardization of its market preparations. Dhaka Univ. J. Pharm. Sci. 2015, 14, 71–78. [Google Scholar] [CrossRef]
- Suresh, K.; Goud, N.R.; Nangia, A. Andrographolide: Solving chemical instability and poor solubility by means of cocrystals. Chem. Asian J. 2013, 8, 3032–3041. [Google Scholar] [CrossRef]
- Syukri, Y.; Martien, R.; Lukitaningsih, E.; Nugroho, A.E. Quantification of andrographolide isolated from Andrographis paniculata nees obtained from traditional market in Yogyakarta using validated HPLC. Indones. J. Chem. 2016, 16, 190–197. [Google Scholar] [CrossRef]
- Balakrishnan, V.V.; Abidin, Z.; Nasir, K.; Jamari, S.; Saidon, N.; Lee, S.; Halim, N.; Yahya, R. The characteristics of a coating system containing lawsone dye colorant and pmma-acrylic polyol blended resin. Pigment. Resin Technol. 2013, 42, 128–136. [Google Scholar]
- Guo, S.; Pan, F.; Zhang, H.; Zhang, D.; Wang, J.; Miao, J.; Su, C.; Zhang, C. Fe-based amorphous coating for corrosion protection of magnesium alloy. Materials Design 2016, 108, 624–631. [Google Scholar] [CrossRef]
- Chen, X.; Shen, K.; Zhang, J. Preparation and anticorrosion properties of polyaniline-sio2-containing coating on Mg-Li alloy. Pigment. Resin Technol. 2010, 39, 322–326. [Google Scholar] [CrossRef]
- Rajani, M.; Shrivastava, N.; Ravishankara, M. A rapid method for isolation of andrographolide from Andrographis paniculata Nees (Kalmegh). Pharm. Biol. 2000, 38, 204–209. [Google Scholar] [CrossRef]
- Ammar, S.; Ramesh, K.; Vengadaesvaran, B.; Ramesh, S.; Arof, A.K. A novel coating material that uses nano-sized SiO2 particles to intensify hydrophobicity and corrosion protection properties. Electrochim. Acta 2016, 220, 417–426. [Google Scholar] [CrossRef]
- Taylor, S.; Moongkhamklang, P. the delineation of local water interaction with epoxy coatings using fluorescence microscopy. Prog. Org. Coat. 2005, 54, 205–210. [Google Scholar] [CrossRef]
- Alam, M.; Rahman, M.; Haque, M. Extraction of henna leaf dye and its dyeing effects on textile fibre. Bangladesh J. Sci. Ind. Res. 2007, 42, 217–222. [Google Scholar] [CrossRef]
- Vasylkiv, O.Y.; Kubrak, O.I.; Storey, K.B.; Lushchak, V.I. Cytotoxicity of chromium ions may be connected with induction of oxidative stress. Chemosphere 2010, 80, 1044–1049. [Google Scholar] [CrossRef] [PubMed]
- Emami-Karvani, Z.; Chehrazi, P. Antibacterial activity of ZnO nanoparticle on Gram-positive and Gram-negative bacteria. Afr. J. Microbiol. Res. 2011, 5, 1368–1373. [Google Scholar]
- Xu, Y.; Marshall, R.L.; Mukkur, T.K. An investigation on the antimicrobial activity of Andrographis paniculata extracts and andrographolide in Vitro. Asian J. Plant Sci. 2006, 5, 527–530. [Google Scholar]
- Astm, Standard Guide for Preparation of Metallographic Specimens. 2011. Available online: https://www.astm.org/Standards/E3 (accessed on 2 April 2021).
- Pérez, M.; García, M.; Ruiz, D.; Autino, J.C.; Romanelli, G.; Blustein, G. Antifouling activity of green-synthesized 7-hydroxy-4-methylcoumarin. Mar. Environ. Res. 2016, 113, 134–140. [Google Scholar] [CrossRef]
- Ikhmal, W.M.K.W.M.; Mohd-Fekeri, M.F.; Shaifudin, M.S.; Wan-Abdullah, W.R.; Wan-Nik, W.M.N.; Zulkifli, M.F.R.; Sabri, M.G.M. Enhancement of corrosion resistance and microbial protection analysis of a rosin coating with the incorporation of Leucaena leucocephala. Coatings 2020, 10, 1–20. [Google Scholar]
- Katz, D.S. The streak plate protocol, ASM. ASMscience 2008, 1–14. Available online: https://www.asmscience.org/content/education/protocol/protocol.3160 (accessed on 2 April 2021).
- Koçoğlu, M.E.; Davarci, İ.; Güney, R.; Taşçilar, M.; Zengïn, F.; Samasti, M. Comparison of conventional methods and automated systems for determining antibiotic susceptibility of bacteria isolated from urine culture. Bangladesh J. Med. Sci. 2019, 18, 519–526. [Google Scholar] [CrossRef]
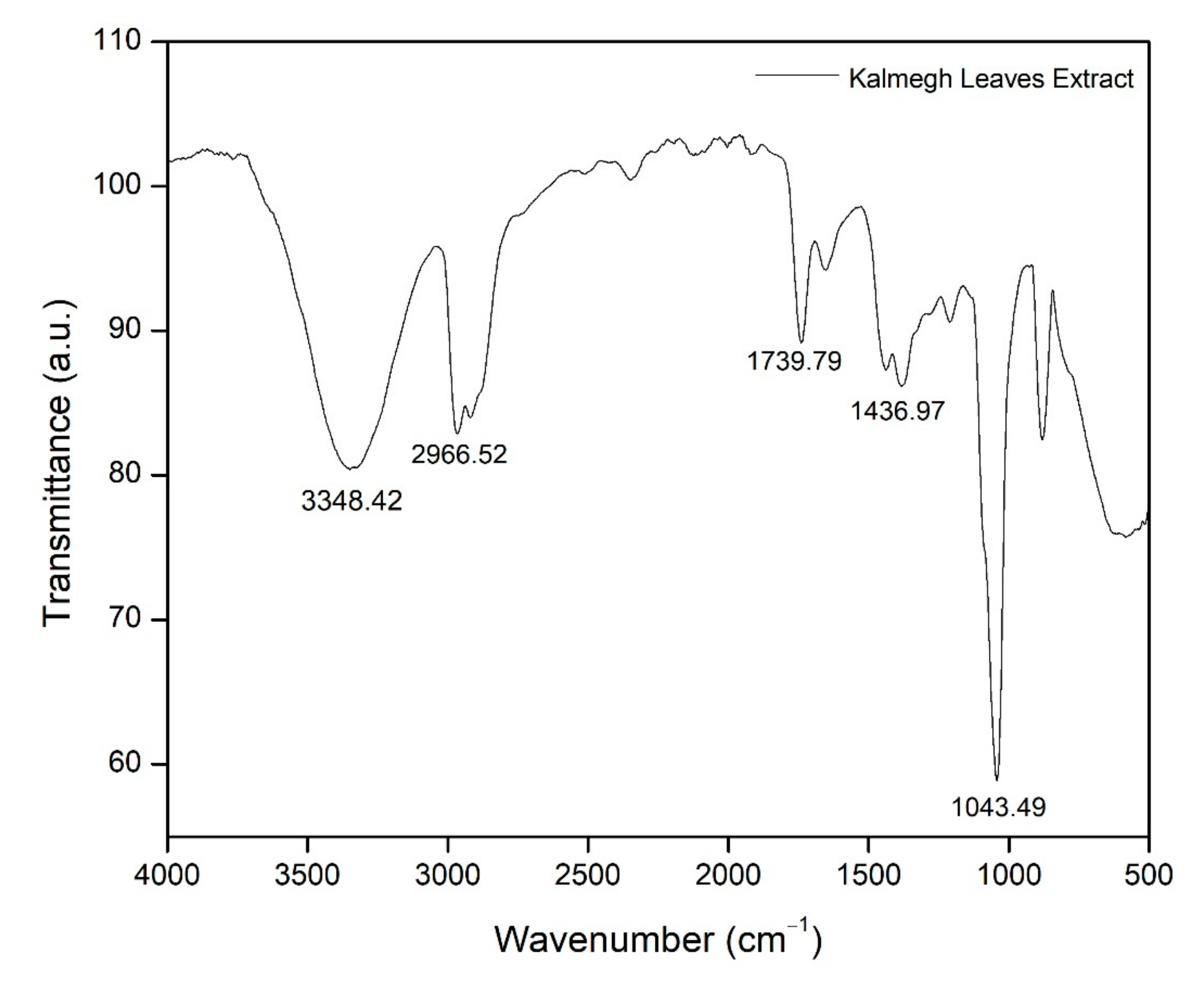
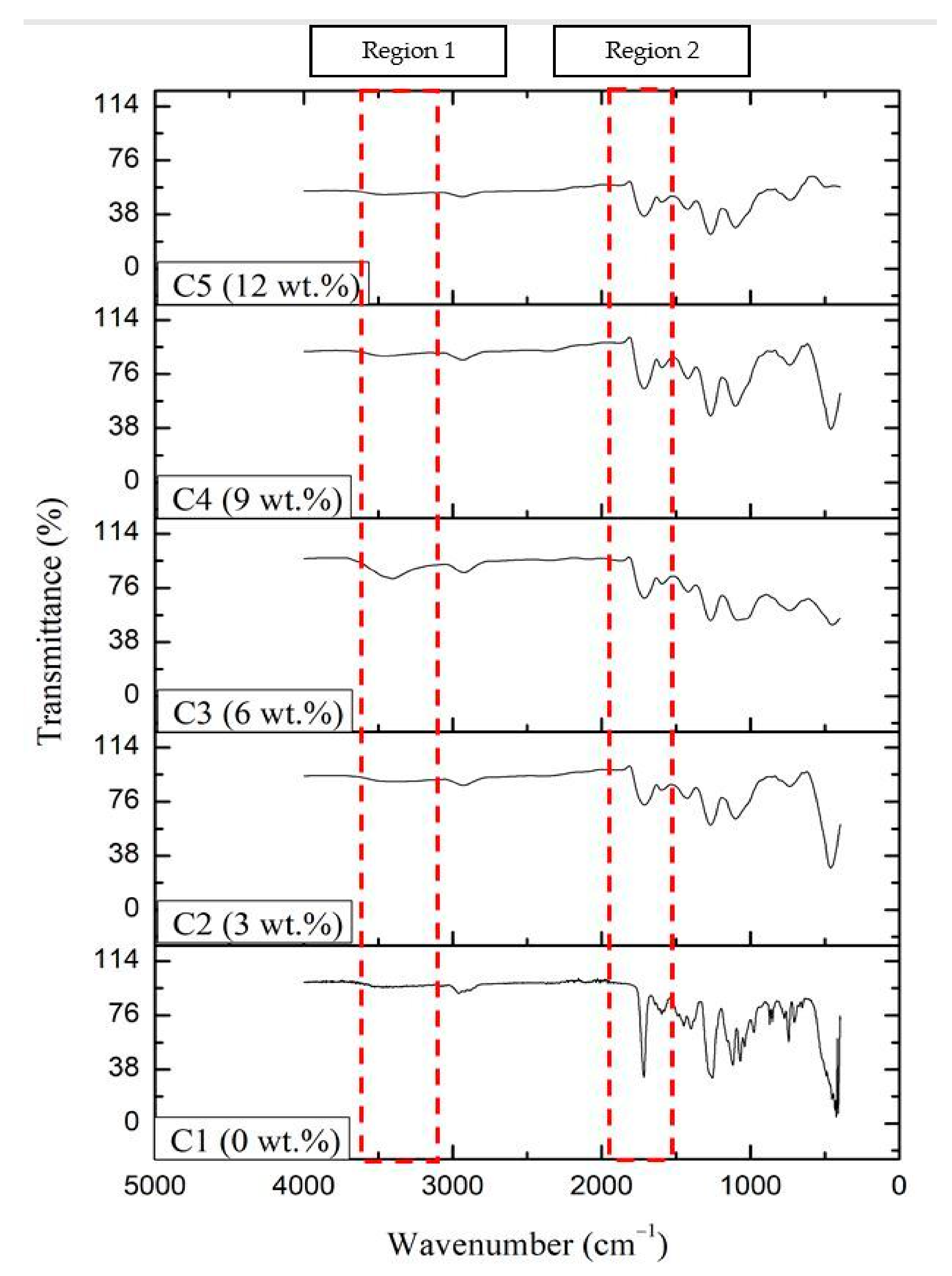




| Wavenumber (cm−1) | Functional Group | Bond |
|---|---|---|
| 3348.42 | Primary alcohol | O−H |
| 2966.52 | Carboxylic acid | R−COOH |
| 1739.79 | Amide | C=O |
| 1436.97 | Alkane | C−H |
| 1043.49 | Ethers | R−O−R |
| Name | Peak | Retention Time (min) | Area (mV*min) | Height (mV) |
|---|---|---|---|---|
| A. paniculata leaves extract | 1 | 4.650 | 4,589,710 | 819,072 |
| Days | Sample | Rc (Ω) | Cc (F) | Rct (Ω) | Cdl (F) | Warburg Impedance, W (µΩ−1) |
|---|---|---|---|---|---|---|
| Before immersion | Bare | − | − | 17.70 × 103 | 152.00 × 10−6 | − |
| C1 | 26.40 × 103 | 12.60 × 10−9 | 124.00 × 103 | 0.50 × 10−6 | 8.51 | |
| C2 | 113.00 × 103 | 1.07 × 10−9 | 129.00 × 103 | 0.02 × 10−6 | 16.30 | |
| C3 | 203.00 × 103 | 159.00 × 10−9 | 202.00 × 103 | 0.77 × 10−6 | 27.70 | |
| C4 | 4.75 × 103 | 208.00 × 10−9 | 29.60 × 103 | 1.06 × 10−6 | 66.00 | |
| C5 | 6.32 × 103 | 37.70 × 10−9 | 38.20 × 103 | 316.00 × 10−9 | 42.10 | |
| 10 | Bare | − | − | 1.65 × 103 | 37.60 × 10−6 | − |
| C1 | 1.79 × 103 | 27.90 × 10−9 | 6.32 × 103 | 3.35 × 10−6 | 257.00 | |
| C2 | 1.69 × 103 | 28.20 × 10−9 | 1.75 × 103 | 2.38 × 10−6 | 617.00 | |
| C3 | 2.44 × 103 | 48.40 × 10−9 | 48.20 × 103 | 4.32 × 10−6 | 366.00 | |
| C4 | 0.70 × 103 | 1170.00 × 10−9 | 3.15 × 103 | 8.11 × 10−6 | 600.00 | |
| C5 | 0.53 × 103 | 402.00 × 10−9 | 2.02 × 103 | 1.18 × 10−6 | 60.00 | |
| 20 | Bare | − | − | 6.23 × 103 | 79.20 × 10−6 | − |
| C1 | 5.23 × 103 | 125.00 × 10−9 | 12.30 × 103 | 0.58 × 10−6 | 1.33.00 | |
| C2 | 1.02 × 103 | 155.00 × 10−9 | 1.82 × 103 | 2.57 × 10−6 | 403.00 | |
| C3 | 7.43 × 103 | 10.70 × 10−9 | 16.30 × 103 | 0.93 × 10−6 | 103.00 | |
| C4 | 0.68 × 103 | 989.00 × 10−9 | 3.15 × 103 | 4.31 × 10−6 | 198.00 | |
| C5 | 0.82 × 103 | 1250.00×10−9 | 6.36 × 103 | 5.16 × 10−6 | 120.00 | |
| 30 | Bare | − | − | 3.40 × 103 | 85.50 × 10−6 | − |
| C1 | 2.07 × 103 | 131.00 × 10−9 | 8.17 × 103 | 1.55 × 10−6 | 42.00 | |
| C2 | 2.12 × 103 | 131.00 × 10−9 | 4.57 × 103 | 4.20 × 10−6 | 218.00 | |
| C3 | 3.36 × 103 | 34.30 × 10−9 | 14.00 × 103 | 0.51 × 10−6 | 25.00 | |
| C4 | 3.30 × 103 | 41.70 × 10−9 | 11.00 × 103 | 1.34 × 10−6 | 114.00 | |
| C5 | 1.77 × 103 | 401.00 × 10−9 | 13.40 × 103 | 5.08 × 10−6 | 146.00 | |
| 40 | Bare | − | − | 14.20 × 103 | 0.27 × 10−6 | − |
| C1 | 2.99 × 103 | 22.90 × 10−9 | 8.58 × 103 | 2.29 × 10−6 | 171.00 | |
| C2 | 1.73 × 103 | 126.00 × 10−9 | 7.57 × 103 | 0.44 × 10−6 | 29.50 | |
| C3 | 3.10 × 103 | 93.40 × 10−9 | 8.22 × 103 | 107.00 × 10−6 | 95.60 | |
| C4 | 2.07 × 103 | 104.00 × 10−9 | 8.09 × 103 | 0.40 × 10−6 | 18.70 | |
| C5 | 0.30 × 103 | 171.00 × 10−9 | 0.68 × 103 | 0.65 × 10−6 | 31.60 | |
| 50 | Bare | − | − | 2.04 × 103 | 14.60 × 10−6 | − |
| C1 | 3.89 × 103 | 2.70 × 10−9 | 2.26 × 103 | 0.04 × 10−6 | 7.54 | |
| C2 | 1.46 × 103 | 209.00 × 10−9 | 2.05 × 103 | 1.78 × 10−6 | 89.10 | |
| C3 | 4.37 × 103 | 275.00 × 10−9 | 15.70 × 103 | 600.00 × 10−6 | 64.20 | |
| C4 | 2.78 × 103 | 43.90 × 10−9 | 7.07 × 103 | 606.00 × 10−6 | 20.90 | |
| C5 | 0.34 × 103 | 1340.00×10−9 | 1.90 × 103 | 10.50 × 10−6 | 294.00 |
| Days | Sample | Ecorr (V) | icorr (A/cm2) | CR (mm/year) |
|---|---|---|---|---|
| Before immersion | Bare | −0.340 | 17.30 × 10−5 | 28.99 × 10−2 |
| C1 | −0.279 | 0.01 × 10−5 | 2.10 × 10−4 | |
| C2 | −0.282 | 0.08 × 10−5 | 13.10 × 10−4 | |
| C3 | −0.163 | 0.04 × 10−6 | 0.70 × 10−4 | |
| C4 | −0.430 | 0.32 × 10−5 | 5.40 × 10−4 | |
| C5 | −0.415 | 2.98 × 10−5 | 4.99 × 10−2 | |
| 10 | Bare | −0.318 | 4.20 × 10−5 | 78.88 × 10−2 |
| C1 | −0.396 | 5.20 × 10−5 | 8.69 × 10−2 | |
| C2 | −0.342 | 5.53 × 10−5 | 9.23 × 10−2 | |
| C3 | −0.290 | 3.15 × 10−5 | 5.27 × 10−2 | |
| C4 | −0.438 | 5.60 × 10−5 | 9.36 × 10−2 | |
| C5 | −0.454 | 6.17 × 10−5 | 10.32 × 10−2 | |
| 20 | Bare | −0.298 | 19.10 × 10−5 | 0.10 × 10−2 |
| C1 | −0.426 | 0.62 × 10−5 | 56.93 × 10−2 | |
| C2 | −0.449 | 3.26 × 10−5 | 1.02 | |
| C3 | −0.301 | 0.16 × 10−5 | 0.36 | |
| C4 | −0.356 | 7.44 × 10−5 | 1.12 | |
| C5 | −0.352 | 0.69 × 10−5 | 0.75 | |
| 30 | Bare | −0.310 | 47.00 × 10−5 | 0.79 |
| C1 | −0.374 | 0.49 × 10−5 | 5.70 × 10−2 | |
| C2 | −0.394 | 4.64 × 10−5 | 7.75 × 10−2 | |
| C3 | −0.321 | 0.13 × 10−5 | 21.10 × 10−4 | |
| C4 | −0.367 | 0.66 × 10−5 | 1.10 × 10−2 | |
| C5 | −0.366 | 2.29 × 10−5 | 3.80 × 10−2 | |
| 40 | Bare | −0.459 | 4.55 × 10−6 | 0.76 |
| C1 | −0.405 | 3.44 × 10−6 | 0.64 × 10−2 | |
| C2 | −0.349 | 1.20 × 10−6 | 1.40 × 10−2 | |
| C3 | −0.368 | 2.67 × 10−6 | 0.45 × 10−2 | |
| C4 | −0.432 | 4.39 × 10−6 | 0.73 × 10−2 | |
| C5 | −0.483 | 8.46 × 10−6 | 0.14 | |
| 50 | Bare | −0.457 | 18.80 × 10−5 | 0.31 |
| C1 | −0.483 | 0.18 × 10−6 | 1.39 × 10−2 | |
| C2 | −0.417 | 2.04 × 10−5 | 3.41 × 10−2 | |
| C3 | −0.337 | 4.34 × 10−6 | 0.73 × 10−2 | |
| C4 | −0.446 | 1.10 × 10−5 | 1.84 × 10−2 | |
| C5 | −0.482 | 8.88 × 10−5 | 0.15 |
| Element | Mass (%) | |
|---|---|---|
| Bare | C3 (6%) | |
| Na | 0.84 | 43.63 |
| Cl | 0.47 | 38.69 |
| Cr | 62.31 | 9.71 |
| Ni | 36.39 | 7.97 |
| Total | 100.00 | 100.00 |
| Sample | S. Aureus (+) | P. Aeruginosa (−) | ||||
|---|---|---|---|---|---|---|
| Average Inhibition Zone (mm) | Efficiency (%) | Standard Deviation | Average Inhibition Zone (mm) | Efficiency (%) | Standard Deviation | |
| Antibiotics | 38 | − | 0 | 28 | − | 0 |
| ZnO | 10 | 26 | 1.26 | 11 | 39 | 1.53 |
| CaCO3 | 7 | 18 | 1.15 | 9 | 32 | 0.87 |
| MIBK | 9 | 23 | 1.44 | 10 | 36 | 2.00 |
| WW rosin | 9 | 23 | 0.47 | 10 | 36 | 1.00 |
| KLE | 7 | 18 | 0.53 | 15 | 53 | 0.33 |
| Sample | S. Aureus (+) | P. Aeruginosa (−) | ||||
|---|---|---|---|---|---|---|
| Inhibition Zone (mm) | Efficiency (%) | Standard Deviation | Inhibition Zone (mm) | Efficiency (%) | Standard Deviation | |
| Antibiotics | 38 | − | 0 | 28 | − | 0 |
| C1 (0 wt%) | 14 | 37 | 0.35 | 9 | 32 | 1.53 |
| C2 (3 wt%) | 15 | 39 | 3.50 | 9 | 32 | 0.58 |
| C3 (6 wt%) | 16 | 42 | 2.00 | 11 | 39 | 1.53 |
| C4 (9 wt%) | 13 | 34 | 0.58 | 10 | 35 | 1.66 |
| C5 (12 wt%) | 14 | 37 | 1.53 | 10 | 35 | 0.42 |
| Component/Coating | C1 | C2 | C3 | C4 | C5 |
|---|---|---|---|---|---|
| WW rosin | 49.1 | 49.1 | 49.1 | 49.1 | 49.1 |
| MIBK | 20.0 | 20.0 | 20.0 | 20.0 | 20.0 |
| CaCO3 | 10.9 | 10.9 | 10.9 | 10.9 | 10.9 |
| ZnO | 20.0 | 17.0 | 14.0 | 11.0 | 8.0 |
| KLE | 0.0 | 3.0 | 6.0 | 9.0 | 12.0 |
| Parameter | Average Reading | Standard Deviation |
|---|---|---|
| Dissolved oxygen, DO (mg/L) | 5.06 | 0.23 |
| Conductivity (mS) | 43.6 | 0.31 |
| Salinity (ppt) | 26.4 | 0.25 |
| pH | 8.1 | 0.22 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kamaruzzaman, W.M.I.W.M.; Fekeri, M.F.M.; Nasir, N.A.M.; Hamidi, N.A.S.M.; Baharom, M.Z.; Adnan, A.; Shaifudin, M.S.; Abdullah, W.R.W.; Wan Nik, W.M.N.; Suhailin, F.H.; et al. Anticorrosive and Microbial Inhibition Performance of a Coating Loaded with Andrographis paniculata on Stainless Steel in Seawater. Molecules 2021, 26, 3379. https://doi.org/10.3390/molecules26113379
Kamaruzzaman WMIWM, Fekeri MFM, Nasir NAM, Hamidi NASM, Baharom MZ, Adnan A, Shaifudin MS, Abdullah WRW, Wan Nik WMN, Suhailin FH, et al. Anticorrosive and Microbial Inhibition Performance of a Coating Loaded with Andrographis paniculata on Stainless Steel in Seawater. Molecules. 2021; 26(11):3379. https://doi.org/10.3390/molecules26113379
Chicago/Turabian StyleKamaruzzaman, Wan Mohamad Ikhmal Wan Mohamad, Maria Fazira Mohd Fekeri, Nursabrina Amirah Mohd Nasir, Nur Aiman Syafiq Mohd Hamidi, Mohamad Zahid Baharom, Azila Adnan, Muhamad Syaizwadi Shaifudin, Wan Rafizah Wan Abdullah, Wan Mohd Norsani Wan Nik, Fariza Hanim Suhailin, and et al. 2021. "Anticorrosive and Microbial Inhibition Performance of a Coating Loaded with Andrographis paniculata on Stainless Steel in Seawater" Molecules 26, no. 11: 3379. https://doi.org/10.3390/molecules26113379
APA StyleKamaruzzaman, W. M. I. W. M., Fekeri, M. F. M., Nasir, N. A. M., Hamidi, N. A. S. M., Baharom, M. Z., Adnan, A., Shaifudin, M. S., Abdullah, W. R. W., Wan Nik, W. M. N., Suhailin, F. H., Matori, K. A., Kien, C. S., Zaid, M. H. M., & Ghazali, M. S. M. (2021). Anticorrosive and Microbial Inhibition Performance of a Coating Loaded with Andrographis paniculata on Stainless Steel in Seawater. Molecules, 26(11), 3379. https://doi.org/10.3390/molecules26113379







