The Potential Effects of Phytoestrogens: The Role in Neuroprotection
Abstract
1. Introduction
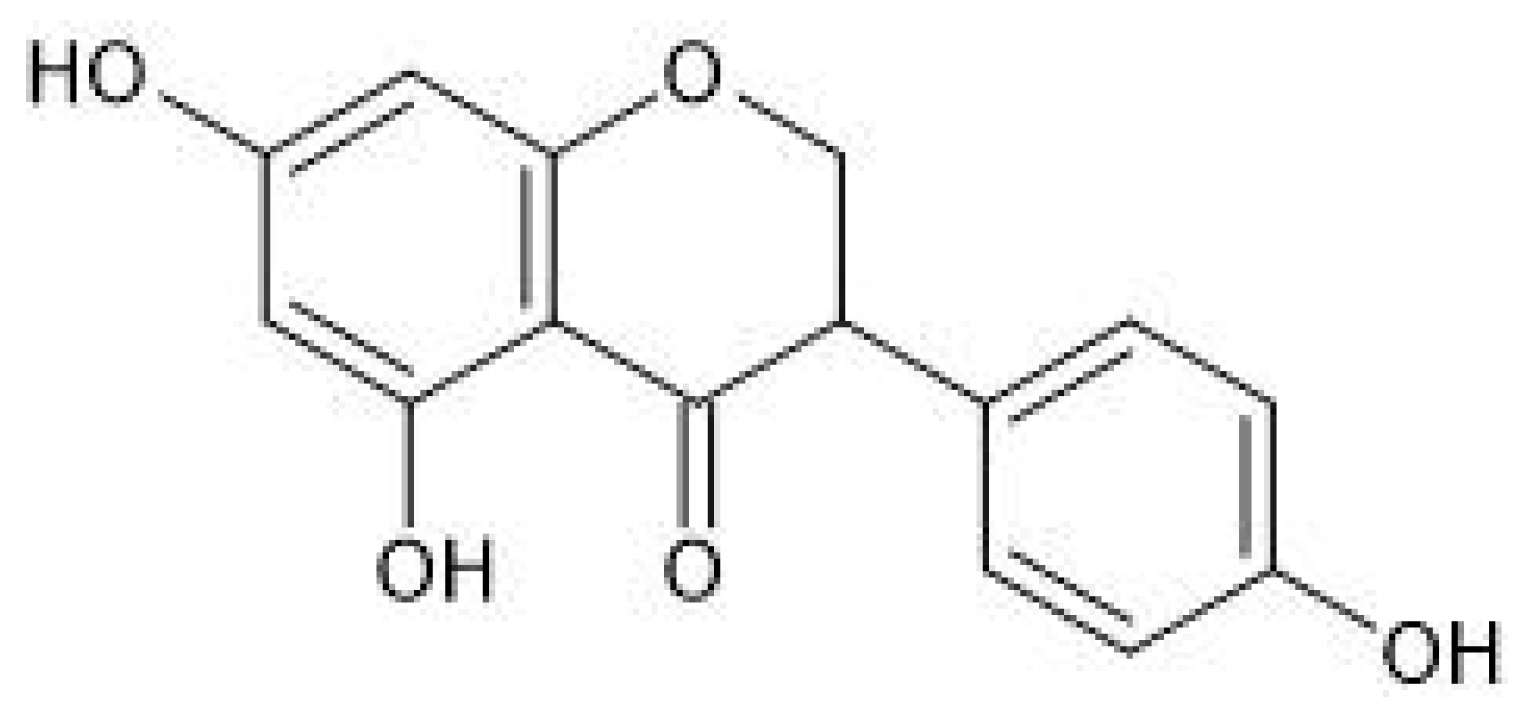
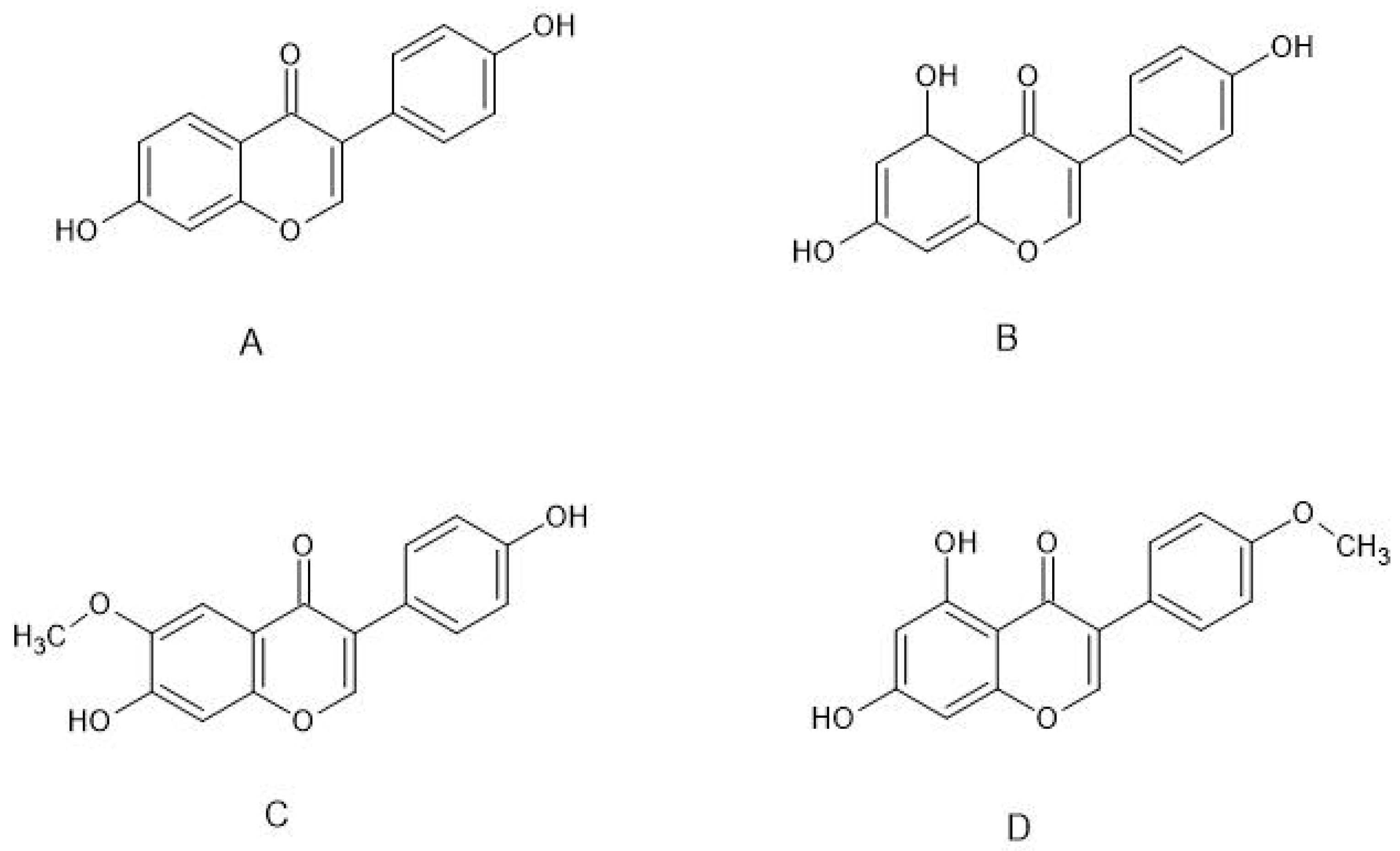
2. Neuroprotective Effects of Selected Phytoestrogens
3. Other selected Applications of Phytoestrogens
3.1. Phytoestrogens in Postmenopausal Indications
3.2. Phytoestrogens and Cardiovascular Health
3.3. Phytoestrogens in Cancer Prevention
3.4. Thyroidal Effects of Phytoestrogens
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hughes, C.L., Jr. Phytochemical mimicry of reproductive hormones and modulation of herbivore fertility by phytoestrogens. Environ. Health Perspect. 1988, 78, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Adlercreutz, H. Phyto-oestrogens and cancer. Lancet Oncol. 2002, 3, 364–373. [Google Scholar] [CrossRef]
- Bhathena, S.J.; Velasquez, M.T. Beneficial role of dietary phytoestrogens in obesity and diabetes. Am. J. Clin. Nutr. 2002, 76, 1191–1201. [Google Scholar] [CrossRef]
- Karahalil, B. Benefits and Risks of Phytoestrogens. Phytoestrogens. In Functional Foods; Yildiz, F., Ed.; CRC Press: Boca Raton, FL, USA, 2005; pp. 210–211. ISBN 978-1-4398-1588-5. [Google Scholar]
- Cederroth, C.R.; Nef, S. Soy, phytoestrogens and metabolism: A review. Mol. Cell. Endocrinol. 2009, 304, 30–42. [Google Scholar] [CrossRef]
- Patisaul, H.B.; Jefferson, W. The pros and cons of phytoestrogens. Front. Neuroendocrinol. 2010, 31, 400–419. [Google Scholar] [CrossRef]
- Zhao, E.; Mu, Q. Phytoestrogen biological actions on Mammalian reproductive system and cancer growth. Sci. Pharm. 2011, 79, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Jungbauer, A.; Medjakovic, S. Phytoestrogens and the metabolic syndrome. J. Steroid Biochem. Mol. Biol. 2014, 139, 277–289. [Google Scholar] [CrossRef]
- Kładna, A.; Berczyński, P.; Kruk, I.; Piechowska, T.; Aboul-Enein, H.Y. Studies on the antioxidant properties of some phytoestrogens. Luminescence 2016, 31, 1201–1206. [Google Scholar] [CrossRef] [PubMed]
- Torrens-Mas, M.; Roca, P. Phytoestrogens for Cancer Prevention and Treatment. Biology 2020, 9, 427. [Google Scholar] [CrossRef]
- Bennetts, H.W.; Underwood, E.J.; Shier, F.L. A specific breeding problem of sheep on subterranean clover pastures in Western Australia. Austral. Vet. J. 1946, 22, 2–12. [Google Scholar] [CrossRef]
- Stafford, H.A. Roles of flavonoids in symbiotic and defense functions in legume roots. Bot. Rev. 1997, 63, 27–39. [Google Scholar] [CrossRef]
- Sirtori, C.R.; Arnoldi, A.; Johnson, S.K. Phytoestrogens: End of a tale? Ann. Med. 2005, 37, 423–438. [Google Scholar] [CrossRef] [PubMed]
- Franke, A.A.; Custer, L.J.; Cerna, C.N.; Narala, K.N. Quantitation of Phytoestrogens in Legumes by HPLC. J. Agric. Food Chem. 1994, 42, 1905–1913. [Google Scholar] [CrossRef]
- Setchell, K.D.; Lawson, A.M.; Borriello, S.P.; Harkness, R.; Gordon, H.; Morgan, D.M.; Kirk, D.N.; Adlercreatz, H.; Anderson, L.C.; Axelson, M. Lignan formation in man--microbial involvement and possible roles in relation to cancer. Lancet 1981, 2, 4–7. [Google Scholar] [CrossRef]
- Chen, Z.; Zheng, W.; Custer, L.J.; Dai, Q.; Shu, X.O.; Jin, F. Usual dietary consumption of soy foods and its correlation with the excretion rate of isoflavonoids in overnight urine samples among Chinese women in Shanghai. Nutr. Cancer 1999, 33, 82–87. [Google Scholar] [CrossRef]
- Van Erp-Baart, M.A.J.; Brants, H.A.M.; Kiely, M. Isoflavone intake in four different European countries: The VENUS approach. Br. J. Nutr. 2003, 89, 25–30. [Google Scholar] [CrossRef]
- Reed, K.E.; Camargo, J.; Hamilton-Reeves, J.; Kurzer, M.; Messina, M. Neither soy nor isoflavone intake affects male reproductive hormones: An expanded and updated meta-analysis of clinical studies. Reprod. Toxicol. 2021, 100, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Kuiper, G.G.J.M.; Enmark, E.; PeltoHuikko, M.; Nilsson, S.; Gustafsson, J.A. Cloning of a novel estrogen receptor expressed in rat prostate and ovary. Proc. Natl. Acad. Sci. USA 1996, 93, 5925–5930. [Google Scholar] [CrossRef]
- Ogawa, S.; Inoue, S.; Watanabe, T.; Orimo, A.; Hosoi, T.; Ouchi, Y.; Muramatsu, M. Molecular cloning and characterization of human estrogen receptor beta cx: A potential inhibitor of estrogen action in human. Nucleic Acids Res. 1998, 26, 3505–3512. [Google Scholar] [CrossRef]
- Harris, H.A. Estrogen receptor-beta: Recent lessons from in vivo studies. Mol. Endocrinol. 2007, 21, 1–13. [Google Scholar] [CrossRef]
- Paterni, I.; Granchi, C.; Katzenellenbogen, J.A.; Minutolo, F. Estrogen receptors alpha (ERα) and beta (ERβ): Subtype-selective ligands and clinical potential. Steroids 2014, 90, 13–29. [Google Scholar] [CrossRef]
- Sotoca, A.; Ratman, D.; Van Der Saag, P.; Ström, A.; Gustafsson, J.; Vervoort, J.; Rietjens, I.; Murk, A. Phytoestrogen-mediated inhibition of proliferation of the human T47D breast cancer cells depends on the ERalpha/ERbeta ratio. J. Steroid Biochem. Mol. Biol. 2008, 112, 171–178. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.; Edvardsson, K.; Lewandowski, S.A.; Strom, A.; Gustafsson, J.A. A genome-wide study of the repressive effects of estrogen receptor beta on estrogen receptor alpha signaling in breast cancer cells. Oncogene 2008, 27, 1019–1032. [Google Scholar] [CrossRef] [PubMed]
- Thomas, C.; Gustafsson, J.A. The different roles of ER subtypes in cancer biology and therapy. Nat. Rev. Cancer 2011, 11, 597–608. [Google Scholar] [CrossRef] [PubMed]
- Covaleda, A.M.S.; Berg, H.V.D.; Vervoort, J.; Van Der Saag, P.; Ström, A.; Gustafsson, J.-Å.; Rietjens, I.; Murk, A.J. Influence of cellular ERalpha/ERbeta ratio on the ERalpha-agonist induced proliferation of human T47D breast cancer cells. Toxicol. Sci. 2008, 105, 303–311. [Google Scholar] [CrossRef]
- Barnes, S.; Peterson, T.G.; Coward, L. Rationale for the use of genistein-containing soy matrices in chemoprevention trials for breast and prostate-cancer. J. Cell. Biochem. 1995, 22, 181–187. [Google Scholar] [CrossRef]
- Ikeda, K.; Arao, Y.; Otsuka, H.; Nomoto, S.; Horiguchi, H.; Kato, S.; Kayama, F. Terpenoids found in the umbelliferae family act as agonists/antagonists for ER(alpha) and ERbeta: Differential transcription activity between ferutinine-liganded ER(alpha) and ERbeta. Biochem. Biophys. Res. Commun. 2002, 291, 354–360. [Google Scholar] [CrossRef]
- De Angelis, M.; Stossi, F.; Waibel, M.; Katzenellenbogen, B.S.; Katzenellenbogen, J.A. Isocoumarins as estrogen receptor beta selective ligands: Isomers of isoflavone phytoestrogens and their metabolites. Bioorg. Med. Chem. 2005, 13, 6529–6542. [Google Scholar] [CrossRef]
- Park, J.; Kim, D.H.; Ahn, H.N.; Song, Y.S.; Lee, Y.J.; Ryu, J.H. Activation of estrogen receptor by bavachin from Psoralea corylifolia. Biomol. Ther. (Seoul) 2012, 20, 183–188. [Google Scholar] [CrossRef]
- Djiogue, S.; Halabalaki, M.; Njamen, D.; Kretzschmar, G.; Lambrinidis, G.; Hoepping, J.; Raffaelli, F.M.; Mikros, E.; Skaltsounis, A.-L.; Vollmer, G. Erythroidine alkaloids: A novel class of phytoestrogens. Planta Med. 2014, 80, 861–869. [Google Scholar] [CrossRef]
- Liu, X.; Nam, J.-W.; Song, Y.S.; Viswanath, A.N.I.; Pae, A.N.; Kil, Y.-S.; Kim, H.-D.; Park, J.H.; Seo, E.-K.; Chang, M. Psoralidin, a coumestan analogue, as a novel potent estrogen receptor signaling molecule isolated from Psoralea corylifolia. Bioorg. Med. Chem. Lett. 2014, 24, 1403–1406. [Google Scholar] [CrossRef]
- Lubbers, L.S.; Zafian, P.T.; Gautreaux, C.; Gordon, M.; Alves, S.E.; Correa, L.; Lorrain, D.S.; Hickey, G.J.; Luine, V. Estrogen receptor (ER) subtype agonists alter monoamine levels in the female rat brain. J. Steroid Biochem. Mol. Biol. 2010, 122, 310–317. [Google Scholar] [CrossRef]
- Brinton, R.D. Impact of estrogen therapy on Alzheimer’s disease: A fork in the road? CNS Drugs 2004, 18, 405–422. [Google Scholar] [CrossRef] [PubMed]
- Soni, M.; Rahardjo, T.B.W.; Soekardi, R.; Sulistyowati, Y.; Lestariningsih; Yesufu-Udechuku, A.; Irsan, A.; Hogervorst, E. Phytoestrogens and cognitive function: A review. Maturitas 2014, 77, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Chen, Q.F.; Diaz Brinton, R. Neuroprotective and neurotrophic efficacy of phytoestrogens in cultured hippocampal neurons. Exp. Biol. Med. 2002, 227, 509–519. [Google Scholar] [CrossRef]
- Zeng, H.; Chen, Q.; Zhao, B. Genistein ameliorates beta-amyloid peptide (25-35)-induced hippocampal neuronal apoptosis. Free Radic. Biol. Med. 2004, 36, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Azcoitia, I.; Moreno, A.; Carrero, P.; Palacios, S.; Garcia-Segura, L.M. Neuroprotective effects of soy phytoestrogens in the rat brain. Gynecol. Endocrinol. 2006, 22, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, M.; Roghani, M.; Joghataei, M.T.; Mohseni, S. Genistein inhibits aggregation of exogenous amyloid-beta(1)(−)(4)(0) and alleviates astrogliosis in the hippocampus of rats. Brain Res. 2012, 1429, 145–154. [Google Scholar] [CrossRef]
- Yao, J.; Zhao, L.; Mao, Z.; Chen, S.; Wong, K.C.; To, J.; Brinton, R.D. Potentiation of brain mitochondrial function by S-equol and R/S-equol estrogen receptor beta-selective phytoSERM treatments. Brain Res. 2013, 1514, 128–141. [Google Scholar] [CrossRef]
- Choi, E.J.; Lee, B.H. Evidence for genistein mediated cytotoxicity and apoptosis in rat brain. Life Sci. 2004, 75, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Gamba, P.; Leonarduzzi, G.; Tamagno, E.; Guglielmotto, M.; Testa, G.; Sottero, B.; Gargiulo, S.; Biasi, F.; Mauro, A.; Viña, J.; et al. Interaction between 24-hydroxycholesterol, oxidative stress, and amyloid-β in amplifying neuronal damage in Alzheimer’s disease: Three partners in crime. Aging Cell 2011, 10, 403–417. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Fu, J.; Li, S.; Li, Z. Neuroprotective Effects of Genistein in a SOD1-G93A Transgenic Mouse Model of Amyotrophic Lateral Sclerosis. J. Neuroimmune Pharmacol. 2019, 14, 688–696. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.L.; Bi, C.W.; Choi, R.C.; Zhu, K.Y.; Miernisha, A.; Dong, T.T.; Tsim, K.W. Flavonoids induce the synthesis and secretion of neurotrophic factors in cultured rat astrocytes: A signaling response mediated by estrogen receptor. Evid. Based Complement. Alternat. Med. 2013, 2013, 127075. [Google Scholar] [CrossRef] [PubMed]
- Pan, M.; Han, H.; Zhong, C.; Geng, Q. Effects of genistein and daidzein on hippocampus neuronal cell proliferation and BDNF expression in H19-7 neural cell line. J. Nutr. Health Aging 2012, 16, 389–394. [Google Scholar] [CrossRef]
- Gao, Q.G.; Xie, J.X.; Wong, M.S.; Chen, W.F. IGF-I receptor signaling pathway is involved in the neuroprotective effect of genistein in the neuroblastoma SK-N-SH cells. Eur. J. Pharmacol. 2012, 677, 39–46. [Google Scholar] [CrossRef]
- Cai, B.; Ye, S.; Wang, Y.; Hua, R.P.; Wang, T.T.; Lix, L.J.; Jiang, A.J.; Shen, G.M. [Protective effects of genistein on Aβ25–35-induced PC12 cell injury via regulating CaM-CaMKIV signaling pathway]. Zhongguo Zhong Yao Za Zhi 2018, 43, 571–576. [Google Scholar] [CrossRef]
- Abbruzzese, G.; Morón-Oset, J.; Díaz-Castroverde, S.; García-Font, N.; Roncero, C.; López-Muñoz, F.; Marco Contelles, J.L.; Oset-Gasque, M.J. Neuroprotection by Phytoestrogens in the Model of Deprivation and Resupply of Oxygen and Glucose In Vitro: The Contribution of Autophagy and Related Signaling Mechanisms. Antioxidants 2020, 9, 545. [Google Scholar] [CrossRef] [PubMed]
- Elsayed, A.A.; Menze, E.T.; Tadros, M.G.; Ibrahim, B.M.M.; Sabri, N.A.; Khalifa, A.E. Effects of genistein on pentylenetetrazole-induced behavioral and neurochemical deficits in ovariectomized rats. Naunyn Schmiedebergs Arch. Pharmacol. 2018, 391, 27–36. [Google Scholar] [CrossRef]
- Jiang, T.; Wang, X.Q.; Ding, C.; Du, X.L. Genistein attenuates isoflurane-induced neurotoxicity and improves impaired spatial learning and memory by regulating cAMP/CREB and BDNF-TrkB-PI3K/Akt signaling. Korean J. Physiol. Pharmacol. 2017, 21, 579–589. [Google Scholar] [CrossRef]
- Xu, H.N.; Li, L.X.; Wang, Y.X.; Wang, H.G.; An, D.; Heng, B.; Liu, Y.Q. Genistein inhibits Aβ25–35-induced SH-SY5Y cell damage by modulating the expression of apoptosis-related proteins and Ca2+ influx through ionotropic glutamate receptors. Phytother. Res. 2019, 33, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Petry, F.D.S.; Coelho, B.P.; Gaelzer, M.M.; Kreutz, F.; Guma, F.T.C.R.; Salbego, C.G.; Trindade, V.M.T. Genistein protects against amyloid-beta-induced toxicity in SH-SY5Y cells by regulation of Akt and Tau phosphorylation. Phytother. Res. 2020, 34, 796–807. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Yang, F.; Gong, C.; Shi, X.; Wang, G. Protective effect of daidzein against streptozotocin-induced Alzheimer’s disease via improving cognitive dysfunction and oxidative stress in rat model. J. Biochem. Mol. Toxicol. 2019, 33, e22319. [Google Scholar] [CrossRef]
- Subedi, L.; Ji, E.; Shin, D.; Jin, J.; Yeo, J.H.; Kim, S.Y. Equol, a Dietary Daidzein Gut Metabolite Attenuates Microglial Activation and Potentiates Neuroprotection In Vitro. Nutrients 2017, 9, 207. [Google Scholar] [CrossRef]
- Çalışkan, G.; Raza, S.A.; Demiray, Y.E.; Kul, E.; Sandhu, K.V.; Stork, O. Depletion of dietary phytoestrogens reduces hippocampal plasticity and contextual fear memory stability in adult male mouse. Nutr. Neurosci. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Anekonda, T.S.; Reddy, P.H. Neuronal protection by sirtuins in Alzheimer’s disease. J. Neurochem. 2006, 2, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Saiko, P.; Szakmary, A.; Jaeger, W.; Szekeres, T. Resveratrol and its analogs: Defense against cancer, coronary disease and neurodegenerative maladies or just a fad? Mutat. Res. 2008, 658, 68–94. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, J.; Emgard, M.; Brundin, P.; Burkitt, M.J. Trans-resveratrol protects embryonic mesencephalic cells from tert-butyl hydroperoxide: Electron paramagnetic resonance spin trapping evidence for a radical scavenging mechanism. J. Neurochem. 2000, 75, 141–150. [Google Scholar] [CrossRef]
- Hunter, R.L.; Dragicevic, N.; Seifert, K.; Choi, D.Y.; Liu, M.; Kim, H.C.; Cass, W.A.; Sullivan, P.G.; Bing, G. Inflammation induces mitochondrial dysfunction and dopaminergic neurodegeneration in the nigrostriatal system. J. Neurochem. 2007, 100, 1375–1386. [Google Scholar] [CrossRef]
- Sarfraz, A.; Javeed, M.; Shah, M.A.; Hussain, G.; Shafiq, N.; Sarfraz, I.; Riaz, A.; Sadiqa, A.; Zara, R.; Zafar, S.; et al. Biochanin A: A novel bioactive multifunctional compound from nature. Sci. Total Environ. 2020, 722, 137907. [Google Scholar] [CrossRef] [PubMed]
- El-Sherbeeny, N.A.; Soliman, N.; Youssef, A.M.; Abd El-Fadeal, N.M.; El-Abaseri, T.B.; Hashish, A.A.; Abdelbasset, W.K.; El-Saber Batiha, G.; Zaitone, S.A. The protective effect of biochanin A against rotenone-induced neurotoxicity in mice involves enhancing of PI3K/Akt/mTOR signaling and beclin-1 production. Ecotoxicol. Environ. Saf. 2020, 205, 111344. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Lu, H.; Qin, J.; Qu, S.; Wang, W.; Guo, Y.; Liao, W.; Song, M.; Chen, J.; Wang, Y. Biochanin A Provides Neuroprotection Against Cerebral Ischemia/Reperfusion Injury by Nrf2-Mediated Inhibition of Oxidative Stress and Inflammation Signaling Pathway in Rats. Med. Sci. Monit. 2019, 25, 8975–8983. [Google Scholar] [CrossRef] [PubMed]
- Khanna, S.; Stewart, R.; Gnyawali, S.; Harris, H.; Balch, M.; Spieldenner, J.; Sen, C.K.; Rink, C. Phytoestrogen isoflavone intervention to engage the neuroprotective effect of glutamate oxaloacetate transaminase against stroke. FASEB J. 2017, 31, 4533–4544. [Google Scholar] [CrossRef] [PubMed]
- Schreihofer, D.A.; Redmond, L. Soy phytoestrogens are neuroprotective against stroke-like injury in vitro. Neuroscience 2009, 158, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Um, M.Y.; Ahn, J.Y.; Kim, S.; Kim, M.K.; Ha, T.Y. Sesaminol glucosides protect beta-amyloid peptide-induced cognitive deficits in mice. Biol. Pharm. Bull. 2009, 32, 1516–1520. [Google Scholar] [CrossRef]
- Giridharan, V.V.; Thandavarayan, R.A.; Sato, S.; Ko, K.M.; Konishi, T. Prevention of scopolamine-induced memory deficits by schisandrin B, an antioxidant lignan from Schisandra chinensis in mice. Free Radic. Res. 2011, 45, 950–958. [Google Scholar] [CrossRef] [PubMed]
- Jeong, E.J.; Lee, H.K.; Lee, K.Y.; Jeon, B.J.; Kim, D.H.; Park, J.-H.; Song, J.-H.; Huh, J.; Lee, J.-H.; Sung, S.H. The effects of lignan-riched extract of Shisandra chinensis on amyloid-beta-induced cognitive impairment and neurotoxicity in the cortex and hippocampus of mouse. J. Ethnopharmacol. 2013, 146, 347–354. [Google Scholar] [CrossRef]
- Li, X.; Zhao, X.; Xu, X.; Mao, X.; Liu, Z.; Li, H.; Guo, L.; Bi, K.; Jia, Y. Schisantherin A recovers Abeta-induced neurodegeneration with cognitive decline in mice. Physiol. Behav. 2014, 132, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Mao, X.; Liao, Z.; Guo, L.; Xu, X.; Wu, B.; Xu, M.; Zhao, X.; Bi, K.; Jia, Y. Schisandrin C ameliorates learning and memory deficits by abeta-induced oxidative stress and neurotoxicity in mice. Phytother Res. 2015, 29, 1373–1380. [Google Scholar] [CrossRef]
- Franco, O.H.; Burger, H.; Lebrun, C.E.I.; Peeters, P.H.M.; Lamberts, S.W.J.; Grobbee, D.E.; Van Der Schouw, Y.T. Higher dietary intake of lignans is associated with better cognitive performance in postmesopausal women. J. Nutr. 2005, 135, 1190–1195. [Google Scholar] [CrossRef]
- Nooyens, A.C.; Milder, I.E.; van Gelder, B.M.; Bueno-de-Mesquita, H.B.; van Boxtel, M.P.; Verschuren, W.M. Diet and cognitive decline at middle age: The role of antioxidants. Br. J. Nutr. 2015, 113, 1410–1417. [Google Scholar] [CrossRef]
- Greendale, G.A.; Tseng, C.H.; Han, W.; Huang, M.H.; Leung, K.; Crawford, S. Dietary isoflavones and bone mineral density during midlife and the menopausal transition: Cross-sectional and longitudinal results from the Study of Women’s Health Across the Nation Phytoestrogen Study. Menopause 2015, 22, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.P.; Li, P.; Zhang, X.H.; Liu, N.; Wang, J.; Chen, D.D.; Sun, W.J.; Zhang, W. Complementary and alternative medicine for natural and treatment-induced vasomotor symptoms: An overview of systematic reviews and meta-analyses. Complement. Ther. Clin. Pract. 2019, 36, 181–194. [Google Scholar] [CrossRef]
- Colditz, G.A.; Willett, W.C.; Stampfer, M.J.; Rosner, B.; Speizer, F.E.; Hennekens, C.H. Menopause and the risk of coronary heart disease in women. N. Engl. J. Med. 1987, 316, 1105–1110. [Google Scholar] [CrossRef]
- Parker, W.H.; Broder, M.S.; Chang, E.; Feskanich, D.; Farquhar, C.; Liu, Z.; Shoupe, D.; Berek, J.S.; Hankinson, S.; Manson, J.E. Ovarian conservation at the time of hysterectomy and long-term health outcomes in the nurses’ health study. Obstet. Gynecol. 2009, 113, 1027–1037. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.M.; Lampe, J.W.; Newton, K.M.; Gundersen, G.; Fuller, S.; Reed, S.D.; Frankenfeld, C.L. Being overweight or obese is associated with harboring a gut microbial community not capable of metabolizing the soy isoflavone daidzein to O-desmethylangolensin in peri- and post-menopausal women. Maturitas 2017, 99, 37–42. [Google Scholar] [CrossRef]
- Ribeiro, A.E.; Monteiro, N.E.S.; Moraes, A.V.G.; Costa-Paiva, L.H.; Pedro, A.O. Can the use of probiotics in association with isoflavone improve the symptoms of genitourinary syndrome of menopause? Results from a randomized controlled trial. Menopause 2018, 26, 643–652. [Google Scholar] [CrossRef]
- Felix, F.B.; Araújo, J.M.D.; de Souza, E.V.; Pinho, V.; Camargo, E.A.; Corrêa, C.B.; Grespan, R. Biochanin A attenuates zymosan-induced arthritis in mice similarly to 17-β estradiol: An alternative to hormone replacement therapy? Inflamm. Res. 2020, 69, 1245–1256. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.A.; Ahmed, M.M.; Gomaa, M.; Ebraheim, L.L.M. Bone health consequence of adjuvant Anastrozole in monotherapy or associated with biochanin-A in ovariectomized rat model. Life Sci. 2018, 212, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez Canete, N.; Duran Aguero, S. [Soya isoflavones and evidences on cardiovascular protection]. Nutr. Hosp. 2014, 29, 1271–1282. [Google Scholar] [CrossRef] [PubMed]
- Van der Schouw, Y.T.; Kreijkamp-Kaspers, S.; Peeters, P.H.; Keinan-Boker, L.; Rimm, E.B.; Grobbee, D.E. Prospective study on usual dietary phytoestrogen intake and cardiovascular disease risk in Western women. Circulation 2005, 111, 465–471. [Google Scholar] [CrossRef]
- Kokubo, Y.; Iso, H.; Ishihara, J.; Okada, K.; Inoue, M.; Tsugane, S. Association of dietary intake of soy, beans, and isoflavones with risk of cerebral and myocardial infarctions in Japanese populations: The Japan Public Health Center-based (JPHC) study cohort I. Circulation 2007, 116, 2553–2562. [Google Scholar] [CrossRef] [PubMed]
- Eisenbrand, G. Senate Commission on Food Safety of the German Research F. Isoflavones as phytoestrogens in food supplements and dietary foods for special medical purposes. Opinion of the Senate Commission on Food Safety (SKLM) of the German Research Foundation (DFG)-(shortened version). Mol. Nutr. Food Res. 2007, 51, 1305–1312. [Google Scholar] [CrossRef] [PubMed]
- Petrakis, N.L.; Barnes, S.; King, E.B.; Lowenstein, J.; Wiencke, J.; Lee, M.M.; Miike, R.; Kirk, M.; Coward, L. Stimulatory influence of soy protein isolate on breast secretion in pre- and postmenopausal women. Canc. Epidemiol. Biomark. Prev. 1996, 5, 785–794. [Google Scholar] [PubMed]
- Hargreaves, D.F.; Potten, C.S.; Harding, C.; Shaw, L.E.; Morton, M.S.; Roberts, S.A.; Howell, A.; Bundred, N.J. Two-week dietary soy supplementation has an estrogenic effect on normal premenopausal breast. J. Clin. Endocr. Metab. 1999, 84, 4017–4024. [Google Scholar] [CrossRef] [PubMed]
- Trock, B.J.; Hilakivi-Clarke, L.; Clarke, R. Meta-analysis of soy intake and breast cancer risk. J. Natl. Cancer Inst. 2006, 98, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.H.; Yu, M.C.; Tseng, C.C.; Pike, M.C. Epidemiology of soy exposures and breast cancer risk. Br. J. Cancer 2008, 98, 9–14. [Google Scholar] [CrossRef]
- Dong, J.Y.; Qin, L.Q. Soy isoflavones consumption and risk of breast cancer incidence or recurrence: A meta-analysis of prospective studies. Breast Cancer Res. Treat. 2011, 125, 315–323. [Google Scholar] [CrossRef]
- Chen, M.; Rao, Y.; Zheng, Y.; Wei, S.; Li, Y.; Guo, T.; Yin, P. Association between soy isoflavone intake and breast cancer risk for pre- and post-menopausal women: A meta-analysis of epidemiological studies. PLoS ONE 2014, 9, e89288. [Google Scholar] [CrossRef]
- Fritz, H.; Seely, D.; Flower, G.; Skidmore, B.; Fernandes, R.; Vadeboncoeur, S.; Kennedy, D.; Cooley, K.; Wong, R.; Sagar, S.; et al. Soy, red clover, and isoflavones and breast cancer: A systematic review. PLoS ONE 2013, 8, e81968. [Google Scholar] [CrossRef]
- Ingram, D.; Sanders, K.; Kolybaba, M.; Lopez, D. Case–control study of phyto-oestrogens and breast cancer. Lancet 1997, 350, 990–994. [Google Scholar] [CrossRef]
- Goodman, M.T.; Shvetsov, Y.B.; Wilkens, L.R.; Franke, A.A.; Le Marchand, L.; Kakazu, K.K.; Nomura, A.M.Y.; Henderson, B.E.; Kolonel, L.N. Urinary phytoestrogen excretion and postmenopausal breast cancer risk: The multiethnic cohort study. Cancer Prev. Res. 2009, 2, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Magee, P.J. Is equol production beneficial to health? Proc. Nutr. Soc. 2011, 70, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Mueller, S.O.; Simon, S.; Chae, K.; Metzler, M.; Korach, K.S. Phytoestrogens and their human metabolites show distinct agonistic and antagonistic properties on estrogen receptor alpha (ERalpha) and ERbeta in human cells. Toxicol. Sci. 2004, 80, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Adlercreutz, H. Lignans and human health. Crit. Rev. Clin. Lab. Sci. 2007, 44, 483–525. [Google Scholar] [CrossRef] [PubMed]
- Penttinen, P.; Jaehrling, J.; Damdimopoulos, A.E.; Inzunza, J.; Lemmen, J.G.; Van Der Saag, P.; Pettersson, K.; Gauglitz, G.; Mäkelä, S.; Pongratz, I. Diet-derived polyphenol metabolite enterolactone is a tissue-specific estrogen receptor activator. Endocrinology 2007, 148, 4875–4886. [Google Scholar] [CrossRef]
- Mense, S.M.; Hei, T.K.; Ganju, R.K.; Bhat, H.K. Phytoestrogens and breast cancer prevention: Possible mechanisms of action. Environ. Health Perspect. 2008, 116, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Saarinen, N.M.; Abrahamsson, A.; Dabrosin, C. Estrogen-induced angiogenic factors derived from stromal and cancer cells are differently regulated by enterolactone and genistein in human breast cancer in vivo. Int. J. Cancer 2010, 127, 737–745. [Google Scholar] [CrossRef]
- Buck, K.R.; Zaineddin, A.K.; Vrieling, A.; Heinz, J.; Linseisen, J.; Fleschjanys, D.; ChangClaude, J. Estimated enterolignans, lignan-rich foods, and fibre in relation to survival after postmenopausal breast cancer. Br. J. Cancer 2011, 105, 1151–1157. [Google Scholar] [CrossRef]
- Shimazu, T.; Inoue, M.; Sasazuki, S.; Iwasaki, M.; Sawada, N.; Yamaji, T.; Tsugane, S. Plasma isoflavones and the risk of lung cancer in women: A nested case–control study in Japan. Canc. Epidemiol. Biomark. Prev. 2011, 20, 419–427. [Google Scholar] [CrossRef]
- Yang, W.S.; Va, P.; Wong, M.Y.; Zhang, H.L.; Xiang, Y.B. Soy intake is associated with lower lung cancer risk: Results from a meta-analysis of epidemiologic studies. Am. J. Clin. Nutr. 2011, 94, 1575–1583. [Google Scholar] [CrossRef]
- Ko, K.-P.; Park, S.K.; Park, B.; Yang, J.J.; Cho, L.Y.; Kang, C.; Kim, C.S.; Gwack, J.; Shin, A.; Kim, Y.; et al. Isoflavones from phytoestrogens and gastric cancer risk: A nested case–control study within the Korean Multicenter Cancer Cohort. Canc. Epidemiol. Biomark. Prev. 2010, 19, 1292–1300. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.W.; Kim, S.Y.; Jee, S.H.; Kim, Y.N.; Nam, C.M. Soy food consumption and risk of prostate cancer: A meta-analysis of observational studies. Nutr. Cancer 2009, 61, 598–606. [Google Scholar] [CrossRef]
- Yan, L.; Spitznagel, E.L. Soy consumption and prostate cancer risk in men: A revisit of a meta-analysis. Am. J. Clin. Nutr. 2009, 89, 1155–1163. [Google Scholar] [CrossRef]
- He, J.; Wang, S.; Zhou, M.; Yu, W.; Zhang, Y.; He, X. Phytoestrogens and risk of prostate cancer: A meta-analysis of observational studies. World J. Surg. Oncol. 2015, 13, 231. [Google Scholar] [CrossRef]
- Horn-Ross, P.L.; Hoggatt, K.J.; Lee, M.M. Phytoestrogens and thyroid cancer risk: The San Francisco Bay area thyroid cancer study. Canc. Epidemiol. Biomark. Prev. 2002, 11, 43–49. [Google Scholar]
- Haselkorn, T.; Stewart, S.L.; Horn-Ross, P.L. Why are thyroid cancer rates so high in southeast asian women living in the United States? The bay area thyroid cancer study. Canc. Epidemiol. Biomark. Prev. 2003, 12, 144–150. [Google Scholar] [PubMed]
- Eden, J.A. Phytoestrogens for menopausal symptoms: A review. Maturitas 2012, 72, 157–159. [Google Scholar] [CrossRef] [PubMed]
- Qu, X.L.; Fang, Y.; Zhang, M.; Zhang, Y.Z. Phytoestrogen intake and risk of ovarian cancer: A meta- analysis of 10 observational studies. Asian Pac. J. Cancer Prev. 2014, 15, 9085–9091. [Google Scholar] [CrossRef]
- Ko, K.-P.; Yeo, Y.; Yoon, J.-H.; Kim, C.-S.; Tokudome, S.; Ngoan, L.T.; Koriyama, C.; Lim, Y.-K.; Chang, S.-H.; Shin, H.-R.; et al. Plasma phytoestrogens concentration and risk of colorectal cancer in two different Asian populations. Clin. Nutr. 2018, 37, 1675–1682. [Google Scholar] [CrossRef]
- Iwasaki, M.; Tsugane, S. Risk factors for breast cancer: Epidemiological evidence from Japanese studies. Cancer Sci. 2011, 102, 1607–1614. [Google Scholar] [CrossRef]
- Divi, R.L.; Chang, H.C.; Doerge, D.R. Anti-thyroid isoflavones from soybean: Isolation, characterization, and mechanisms of action. Biochem. Pharmacol. 1997, 54, 1087–1096. [Google Scholar] [CrossRef]
- Chang, H.C.; Doerge, D.R. Dietary genistein inactivates rat thyroid peroxidase in vivo without an apparent hypothyroid effect. Toxicol. Appl. Pharmacol. 2000, 168, 244–252. [Google Scholar] [CrossRef] [PubMed]
- EFSA ANS Panel. Risk assessment for peri- and post-menopausal women taking food supplements containing isolated isoflavones. EFSA J. 2015, 13, 4246. [Google Scholar] [CrossRef]
- Doerge, D.R.; Sheehan, D.M. Goitrogenic and estrogenic activity of soy isoflavones. Environ. Health Perspect. 2002, 110 (Suppl. S3), 349–353. [Google Scholar] [CrossRef] [PubMed]
- Sathyapalan, T.; Manuchehri, A.M.; Thatcher, N.J.; Rigby, A.S.; Chapman, T.; Kilpatrick, E.S.; Atkin, S.L. The effect of soy phytoestrogen supplementation on thyroid status and cardiovascular risk markers in patients with subclinical hypothyroidism: A randomized, double-blind, crossover study. J. Clin. Endocrinol. Metab. 2011, 96, 1442–1449. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gorzkiewicz, J.; Bartosz, G.; Sadowska-Bartosz, I. The Potential Effects of Phytoestrogens: The Role in Neuroprotection. Molecules 2021, 26, 2954. https://doi.org/10.3390/molecules26102954
Gorzkiewicz J, Bartosz G, Sadowska-Bartosz I. The Potential Effects of Phytoestrogens: The Role in Neuroprotection. Molecules. 2021; 26(10):2954. https://doi.org/10.3390/molecules26102954
Chicago/Turabian StyleGorzkiewicz, Justyna, Grzegorz Bartosz, and Izabela Sadowska-Bartosz. 2021. "The Potential Effects of Phytoestrogens: The Role in Neuroprotection" Molecules 26, no. 10: 2954. https://doi.org/10.3390/molecules26102954
APA StyleGorzkiewicz, J., Bartosz, G., & Sadowska-Bartosz, I. (2021). The Potential Effects of Phytoestrogens: The Role in Neuroprotection. Molecules, 26(10), 2954. https://doi.org/10.3390/molecules26102954