Hydroxycinnamoyl Amino Acids Conjugates: A Chiral Pool to Distinguish Commercially Exploited Coffea spp.
Abstract
1. Introduction
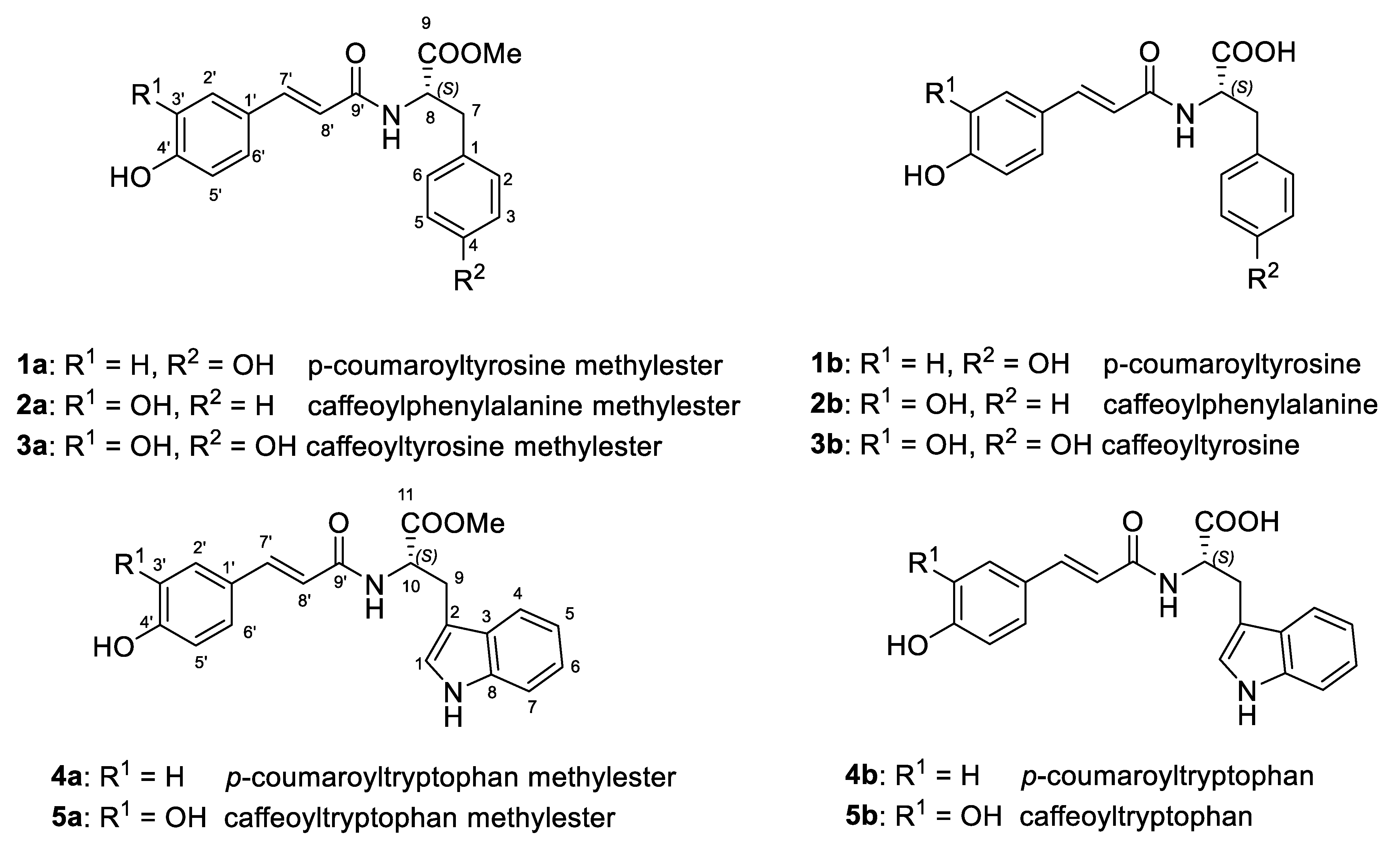
2. Results and Discussion
3. Materials and Methods
3.1. Chemicals
3.2. Instrumentation
3.3. Samples
3.4. General Procedure for the Preparation of the Hydroxycinnamoyl Amides Esters 1a–5a
3.5. General Procedure for the Preparation of the Hydroxycinnamoyl Amides 1b–5b
3.6. Extraction of Hydroxycinnamoyl Amides and Sample Preparation
3.7. Quantitative Analyses
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CD | Circular Dichroism |
| 5-CQA | 5-caffeoylquinic acid |
| DCC | N,N′-Dicyclohexylcarbodiimide |
| DW | Dry weight |
| FA | Formic acid |
| HCA | Hydroxycinnamoyl amide |
References
- Papetti, A.; Daglia, M.; Aceti, C.; Sordelli, B.; Spini, V.; Carazzone, C.; Gazzani, G. Hydroxycinnamic acid derivatives occuring in Cichorium endivia vegetables. J. Pharm. Biomed. Anal. 2008, 48, 472–476. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.Z.; Abbasi, B.H.; Gao, M.; Murch, S.J. Caffeic Acid Derivatives Production By Hairy Root Cultures Of Echinacea purpurea. J. Agric. Food Chem. 2006, 54, 8456–8460. [Google Scholar] [CrossRef] [PubMed]
- Moyo, M.; Amoo, S.O.; Aremu, A.O.; Gruz, J.; Šubrtova, M.; Doležal, K.; Van Staden, J. Plant regeneration and biochemical accumulation of hydroxybenzoic acid and hydroxycinnamic acid derivatives in Hypoxis hemerocallidea organ and callus cultures. Plant Sci. 2014, 227, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Adam, K.P. Caffeic acid derivatives in fronds of the lady fern. Phytochemistry 1995, 40, 1577–1578. [Google Scholar] [CrossRef]
- Clifford, M.N.; Knight, S. The cinnamoyl-ammino acid conjugates of green robusta coffee beans. Food Chem. 2004, 87, 457–463. [Google Scholar] [CrossRef]
- Stark, T.; Justus, H.; Hofmann, T. Quantitative analysis of N-phenylpropenoyl- L-amino acids in roasted coffee and cocoa powder by means of a stable isotope dilution assay. J. Agric. Food Chem. 2006, 54, 2859–2867. [Google Scholar] [CrossRef]
- Hu, X.L.; Lin, J.; Lv, X.-Y.; Feng, J.-H.; Zhang, X.-Q.; Wang, H. Synthesis and biological evaluation of clovamide analogues as potent anti-neuroinflammatory agents in vitro and in vivo. Eur. J. Med. Chem. 2018, 151, 261–271. [Google Scholar] [CrossRef]
- Yuan, X.; Lin, L.; Zhang, X.; Deng, S. Abrusamide A and B, two hepatoprotective isomeric compounds from Abrus mollis Hance. Phytochem. Lett. 2014, 7, 137–142. [Google Scholar] [CrossRef]
- Lee, S.U.; Shin, C.-G.; Lee, C.-K.; Lee, Y.S. Caffeoylglycolic and caffeoylamino acid derivatives, halmers of L-chicoric acid, as new HIV-1 integrase inhibors. Eur. J. Med. Chem. 2007, 42, 1309–1315. [Google Scholar] [CrossRef]
- Adisakwattana, S.; Sookkongwaree, K.; Roengsumran, S.; Petsom, A.; Ngamrojnavanich, N.; Chavasiri, W.; Deesamer, S.; Yibchok-anun, S. Structure-activity relationships of trans-cinnamic acid derivatives on alpha-glucosidase inhibition. Bioorg. Med. Chem. Lett. 2004, 14, 2893–2896. [Google Scholar] [CrossRef]
- Stark, T.; Hofmann, T. Isolation, structure determination, synthesis and sensory activity of N-phenylpropenoyl-L-amino acids from Cocoa (Theobroma cacao). J. Agric. Food Chem. 2005, 53, 5419–5428. [Google Scholar] [CrossRef] [PubMed]
- Patay, E.B.; Bencsik, T.; Papp, N. Phytochemical overview and medicinal importance of Coffea species from the past until now. Asian Pac. J. Trop. Med. 2016, 9, 1127–1135. [Google Scholar] [CrossRef] [PubMed]
- Asamenew, G.; Kim, H.-W.; Lee, M.-K.; Lee, S.-H.; Lee, S.; Cha, Y.-S.; Lee, S.H.; Yoo, S.M.; Kim, J.-B. Comprehensive characterization of hydroxycinnamoyl derivatives in green and roasted beans: A new group of methyl hydroxycinnamoyl quinate. Food Chem. 2019, 2, 100033. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Salces, R.M.; Serra, F.; Reniero, F.; Heberger, K. Botanical and Geographical Characterization of green coffee (Coffea arabica and Coffea canephora): Chemometric evaluation of phenolic and methylxanthine contents. J. Agric. Food Chem. 2009, 57, 4224–4235. [Google Scholar] [CrossRef]
- Garrett, R.; Vaz, B.G.; Hovell, A.M.C.; Eberlin, M.N.; Rezende, C.M. Arabica and Robusta coffees: Identification of major polar compounds and quantification of blends by direct-infusion electrospray ionization-mass spectrometry. J. Agric. Food Chem. 2012, 60, 4253–4258. [Google Scholar] [CrossRef] [PubMed]
- Babova, O.; Occhipinti, A.; Maffei, M.E. Chemical partitioning and antioxidant capacity of green coffee (Coffea arabica and Coffea canephora) of different geographical origin. Phytochemistry 2016, 123, 33–39. [Google Scholar] [CrossRef]
- Murata, M.; Okada, H.; Homma, S. Hydroxycinnamic acid derivatives and p-coumaroyl-N-tryptophan, a novel hydroxycannamic acid derivative from coffee beans. Biosci. Biotechnol. Biochem. 1995, 59, 1887–1890. [Google Scholar] [CrossRef]
- Rodrigues, N.P.; de Jesus Garcia Salva, T.; Bragagnolo, N. Influence of coffee genotype on bioactive compounds and the in vitro capacity to scavenge reactive oxygen and nitrogen species. J. Agric. Food Chem. 2015, 63, 4815–4826. [Google Scholar] [CrossRef]
- Morishita, H.; Takai, Y.; Yamada, H.; Fukuda, F.; Sawada, M.; Iwahashi, H.; Kido, R. Caffeoyltryptophan from green robusta coffee beans. Phytochemistry 1987, 26, 1195–1196. [Google Scholar] [CrossRef]
- Clifford, M.N.; Kellard, B.; Ah-Sing, E. Caffeoyltyrosine from green robusta coffee beans. Phytochemistry 1989, 28, 1989–1990. [Google Scholar] [CrossRef]
- Gutiérrez Ortiz, A.L.; Berti, F.; Solano Sánchez, W.; Navarini, L.; Colomban, S.; Crisafulli, P.; Forzato, C. Distribution of p-coumaroylquinic acids in commercial Coffea spp. of different geographical origin and in other wild coffee species. Food Chem. 2019, 286, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez Ortiz, A.L.; Berti, F.; Navarini, L.; Crisafulli, P.; Colomban, S.; Forzato, C. Aqueous extracts of walnut (Juglans regia L.) leaves: Quantitative analyses of hydroxycinnamic and chlorogenic acids. J. Chromatogr. Sci. 2018, 56, 753–760. [Google Scholar] [CrossRef] [PubMed]
- Anthony, F.; Clifford, M.N.; Noirot, M. Biochemical diversity in the genus Coffea L.: Chlorogenic acids, caffeine and mozambioside contents. Genet. Resour. Crop. Evol. 1993, 40, 61–70. [Google Scholar] [CrossRef]
- Correia, A.M.N.G.; Leittio, M.C.A.; Clifford, M.N. Caffeoyl-tyrosine and Angola II as characteristic markers for Angolan robusta coffees. Food Chem. 1995, 53, 309–313. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |








| Compound | tR (min) | [M + H] (m/z) Exp. | Dimer + Na (m/z) Exp. | Specific Rotation |
|---|---|---|---|---|
| p-coumaroyl-N-tyrosine 1b | 26.9 | 328.1 | 677.1 (positive mode) | −39.9 (c 0.1 MeOH) (lit. −26.9 (MeOH) [11] and −17.7 (c 0.1 MeOH) [7]) |
| caffeoyl-N-phenylalanine 2b | 30.6 | 328.0 | 677.2 (positive mode) | −48.8 (c 0.1 MeOH) |
| caffeoyl-N-tyrosine 3b | 24.6 | 344.1 | 709.2 (positive mode) | −41.7 (c 0.1, MeOH) (lit. −35.6 [11]) |
| p-coumaroyl-N-tryptophan 4b | 33.0 | 351.0 | 723.2 (positive mode) | −27.8 (c 0.1, MeOH) |
| caffeoyl-N-tryptophan 5b | 31.1 | 367.1 | 731.1 (negative mode) | −33.2 (c 1.0, MeOH) |
| Sample | Species | 1b | 2b | 3b | 4b | 5b | Total |
|---|---|---|---|---|---|---|---|
| 1 | C. arabica Nicaragua | <LOD | <LOD | <LOD | <LOD | 117 (2) | 117 (2) |
| 2 | C. arabica Peru | <LOD | <LOD | <LOD | <LOD | <LOD | - |
| 3 | C. arabica Rwanda | <LOD | <LOD | <LOD | <LOD | 51 (1) | 51 (1) |
| 4 | C. arabica El Salvador | <LOD | <LOD | <LOD | <LOD | <LOD | - |
| 5 | C. arabica India | <LOD | <LOD | <LOD | <LOD | 125 (3) | 125 (3) |
| 6 | C. arabica Brazil | <LOD | <LOD | <LOD | <LOD | 37 (1) | 37 (1) |
| 7 | C. arabica Guatemala | <LOD | <LOD | <LOD | <LOD | 97 (2) | 97 (2) |
| 8 | C. arabica – lot 1 Ethiopia | <LOD | <LOD | <LOD | <LOD | 59 (1) | 59 (1) |
| 9 | C. arabica – lot 2 Ethiopia | <LOD | <LOD | <LOD | <LOD | 75 (2) | 75 (2) |
| 10 | C. arabica Burundi | <LOD | <LOD | <LOD | <LOD | 40 (1) | 40 (1) |
| 11 | C. canephora Cameroon | n.q. a | n.q. b | <LOD | 119 (4) | 878 (26) | 997 (30) |
| 12 | C. canephora Ivory Coast | <LOD | n.q. b | <LOD | 116 (4) | 712 (21) | 828 (25) |
| 13 | C. canephora Tanzania | n.q. a | n.q. b | 348 (5) | 319 (3) | 1042 (14) | 1709 (22) |
| 14 | C. canephora Indonesia | <LOD | <LOD | <LOD | 113 (2) | 590 (12) | 703 (14) |
| 15 | C. canephora – lot 1 India | <LOD | n.q. b | <LOD | 103 (3) | 790 (24) | 893 (27) |
| 16 | C. canephora – lot 2 India | <LOD | n.q. b | <LOD | 212 (6) | 1205 (48) | 1417 (54) |
| 17 | C. canephora – lot 3 India | <LOD | n.q. b | <LOD | 128 (4) | 780 (23) | 908 (27) |
| 18 | C. canephora Brazil var. Conilon | n.q. a | 47 (1) | 182 (4) | <LOD | 470 (9) | 699 (14) |
| 19 | C. canephora Uganda | n.q. a | n.q. b | 140 (3) | 195 (5) | 717 (21) | 1052 (29) |
| 20 | C. canephora Vietnam | <LOD | n.q. b | <LOD | 136 (3) | 702 (20) | 838 (23) |
| 21 | C. liberica – lot 1 | <LOD | <LOD | <LOD | <LOD | <LOD | - |
| 22 | C. liberica – lot 2 | <LOD | <LOD | <LOD | <LOD | <LOD | - |
| 23 | C. liberica – lot 3 | <LOD | <LOD | <LOD | <LOD | <LOD | - |
| % A (H2O + 0.1% FA) | % B (CH3CN + 0.1% FA) | Time (min) |
|---|---|---|
| 97 | 3 | 0 |
| 85 | 15 | 10 |
| 60 | 40 | 30 |
| 60 | 40 | 40 |
| 97 | 3 | 45 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berti, F.; Navarini, L.; Colomban, S.; Forzato, C. Hydroxycinnamoyl Amino Acids Conjugates: A Chiral Pool to Distinguish Commercially Exploited Coffea spp. Molecules 2020, 25, 1704. https://doi.org/10.3390/molecules25071704
Berti F, Navarini L, Colomban S, Forzato C. Hydroxycinnamoyl Amino Acids Conjugates: A Chiral Pool to Distinguish Commercially Exploited Coffea spp. Molecules. 2020; 25(7):1704. https://doi.org/10.3390/molecules25071704
Chicago/Turabian StyleBerti, Federico, Luciano Navarini, Silvia Colomban, and Cristina Forzato. 2020. "Hydroxycinnamoyl Amino Acids Conjugates: A Chiral Pool to Distinguish Commercially Exploited Coffea spp." Molecules 25, no. 7: 1704. https://doi.org/10.3390/molecules25071704
APA StyleBerti, F., Navarini, L., Colomban, S., & Forzato, C. (2020). Hydroxycinnamoyl Amino Acids Conjugates: A Chiral Pool to Distinguish Commercially Exploited Coffea spp. Molecules, 25(7), 1704. https://doi.org/10.3390/molecules25071704







