A Global Model for the Estimation of Speeds of Sound in Deep Eutectic Solvents
Abstract
1. Introduction
2. Methods
2.1. Selected Deep Eutectic Solvents (DESs) and Experimental Data
2.2. The Model
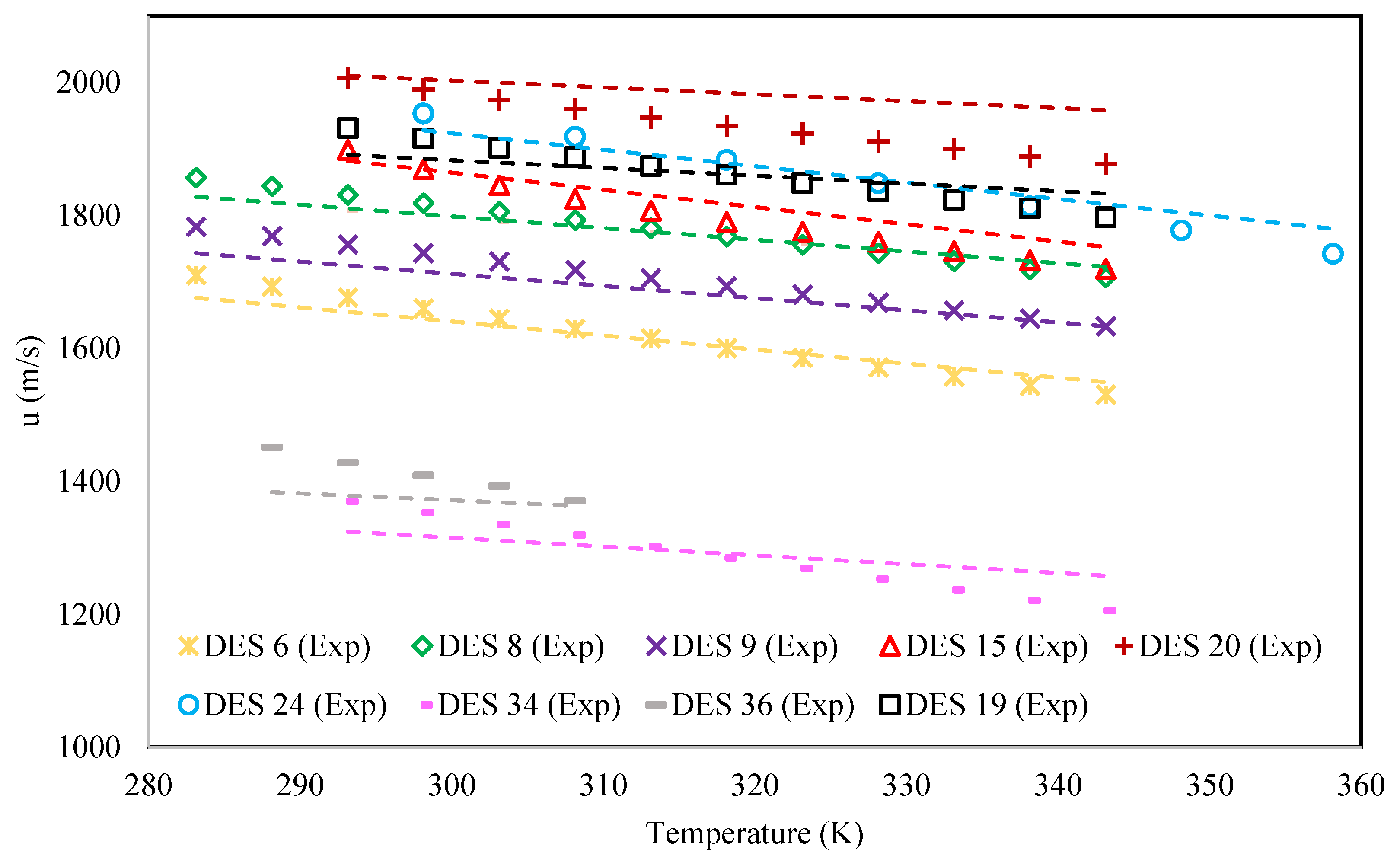
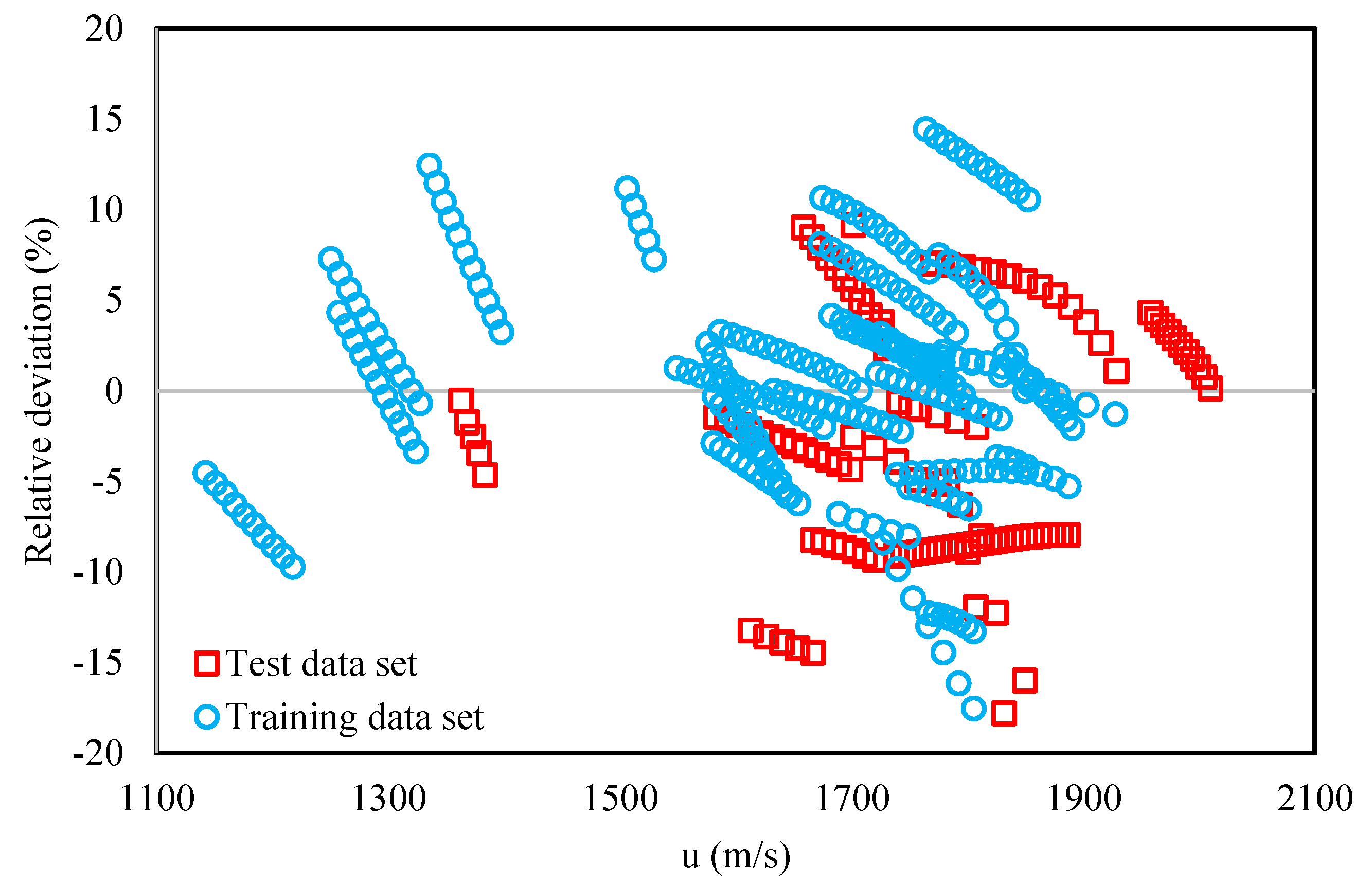
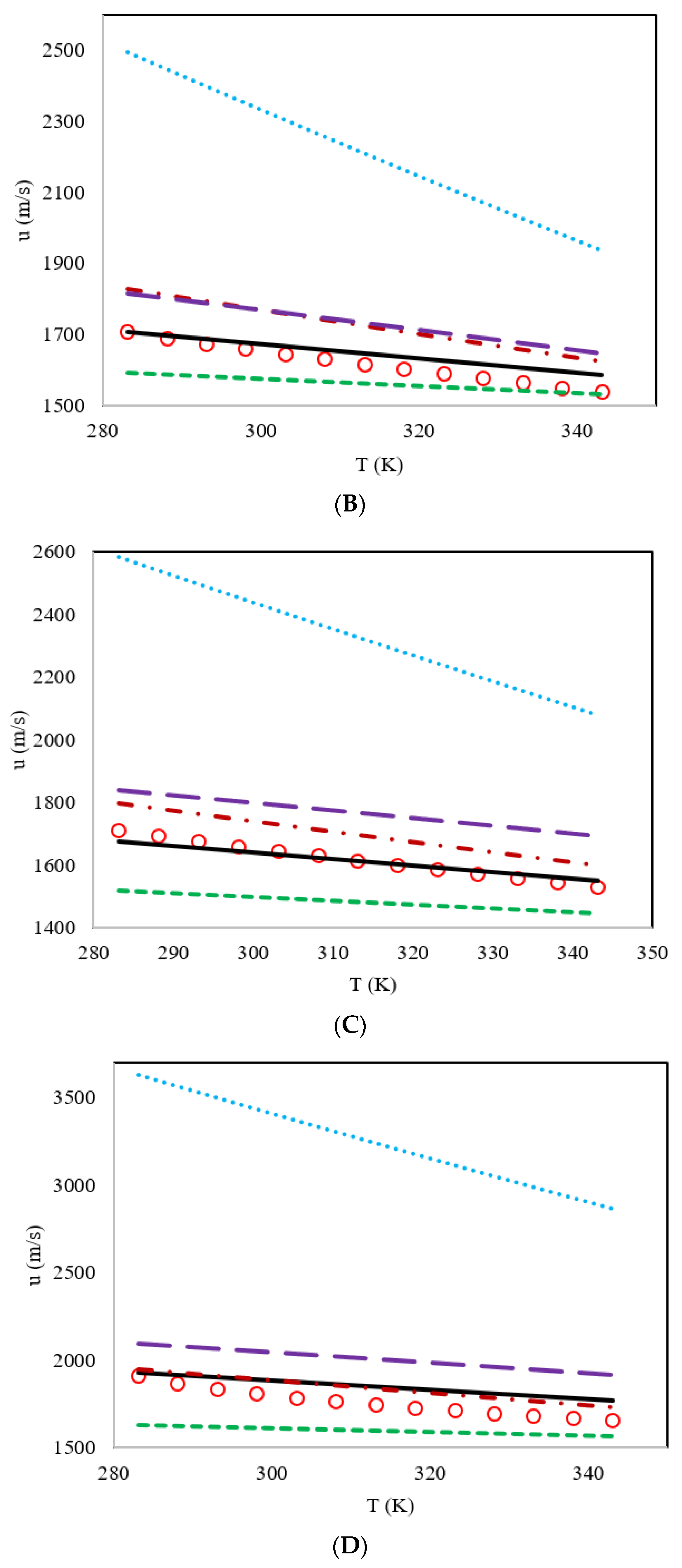
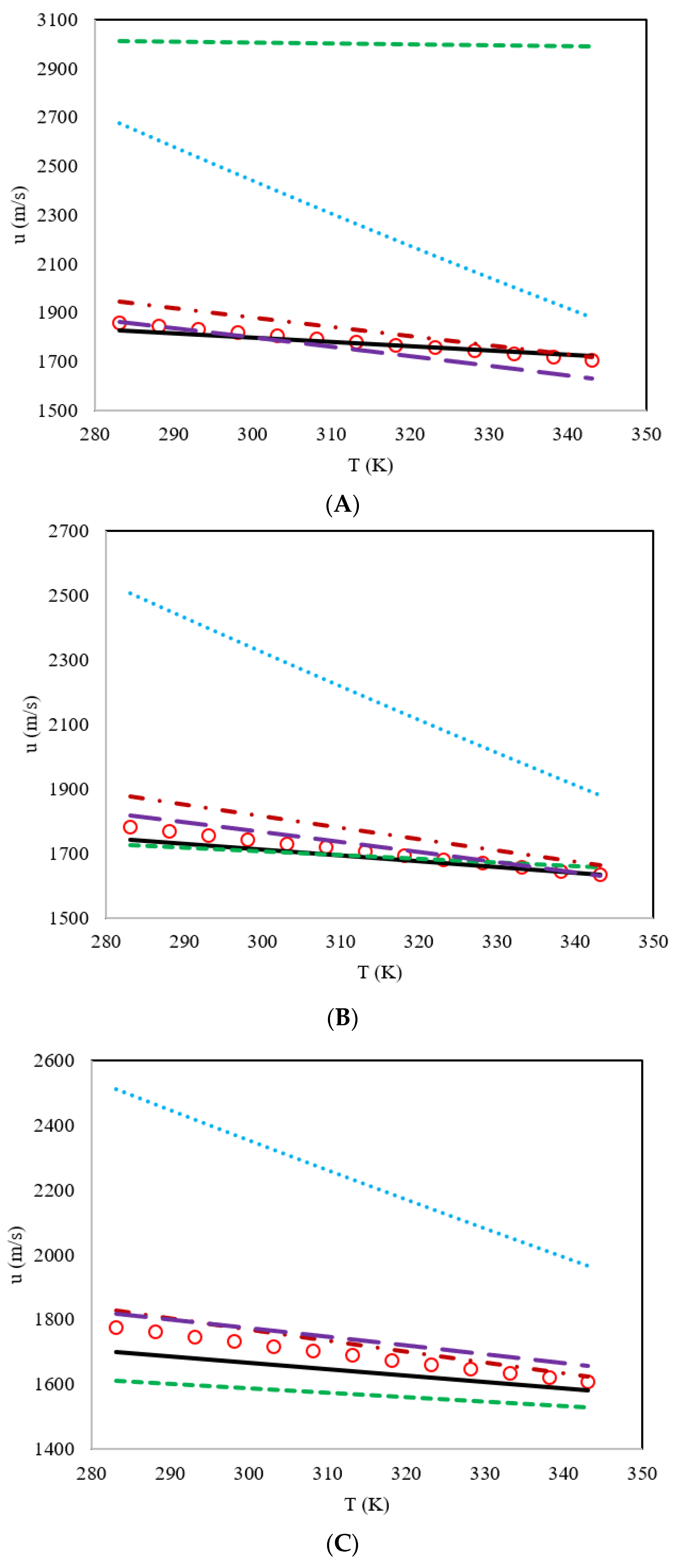
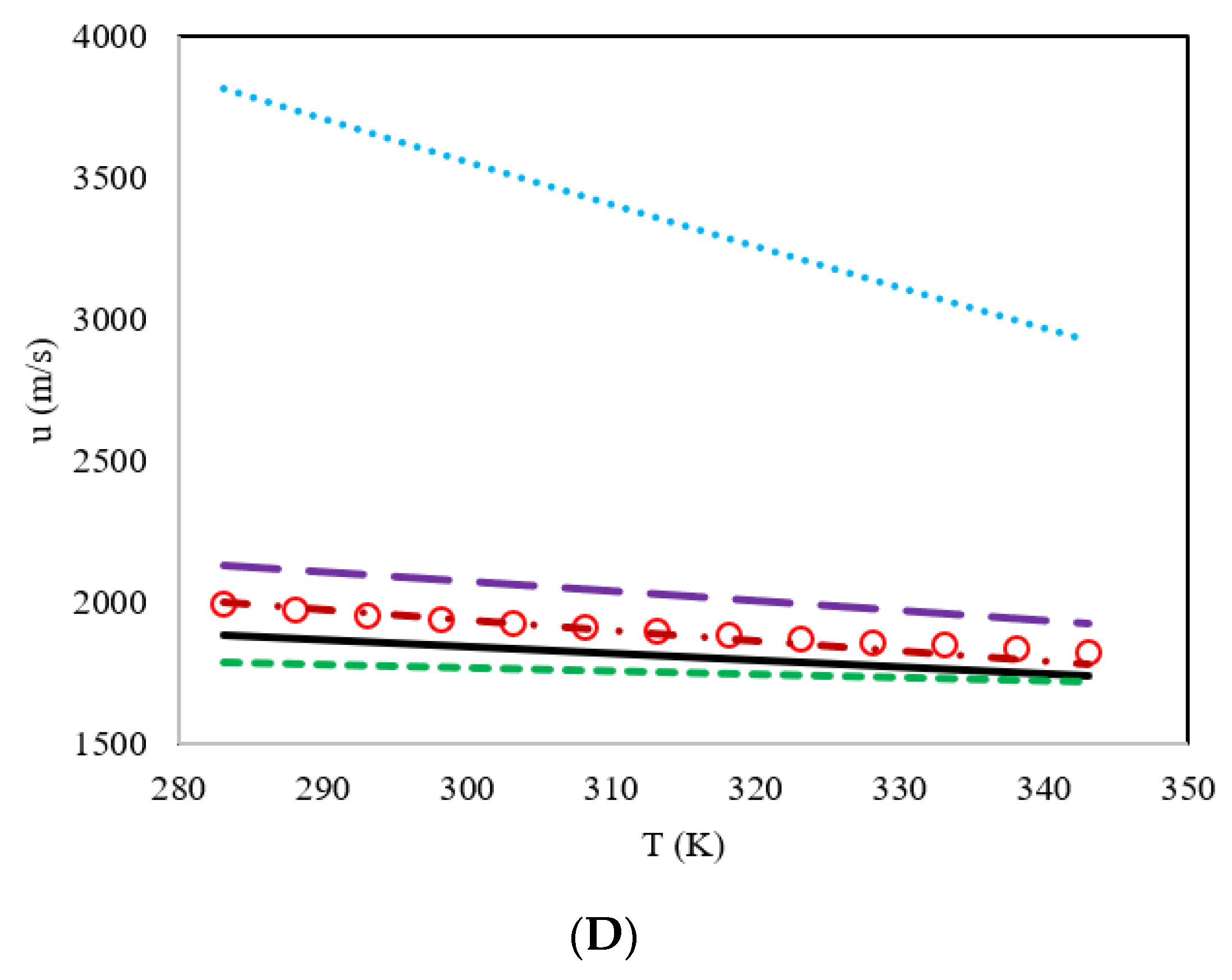
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Häckl, K.; Kunz, W. Some aspects of green solvents. C. R. Chim. 2018, 21, 572–580. [Google Scholar] [CrossRef]
- Pena-Pereira, F.; Tobiszewski, M. The Application of Green Solvents in Separation Processes; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar]
- Singh, S.K.; Savoy, A.W. Ionic liquids synthesis and applications: An overview. J. Mol. Liq. 2020, 297, 112038. [Google Scholar] [CrossRef]
- Irge, D.D. Ionic Liquids: A Review on Greener Chemistry Applications, Quality Ionic Liquid Synthesis and Economical Viability in a Chemical Processes. Am. J. Phys. Chem. 2016, 5, 74–79. [Google Scholar] [CrossRef]
- Chen, Y.; Cao, Y.; Sun, X.; Yan, C.; Mu, T. New criteria combined of efficiency, greenness, and economy for screening ionic liquids for CO2 capture. Int. J. Greenh. Gas. Control. 2013, 16, 13–20. [Google Scholar] [CrossRef]
- Maciel, V.G.; Wales, D.J.; Seferin, M.; Ugaya, C.M.L.; Sans, V. State-of-the-art and limitations in the life cycle assessment of ionic liquids. J. Clean. Prod. 2019, 217, 844–858. [Google Scholar] [CrossRef]
- Vekariya, R.L. A review of ionic liquids: Applications towards catalytic organic transformations. J. Mol. Liq. 2017, 227, 44–60. [Google Scholar] [CrossRef]
- Amiril, S.A.S.; Rahim, E.A.; Syahrullail, S. A review on ionic liquids as sustainable lubricants in manufacturing and engineering: Recent research, performance, and applications. J. J. Clean. Prod. 2017, 168, 1571–1589. [Google Scholar] [CrossRef]
- Troter, D.Z.; Todorović, Z.B.; Đokić-Stojanović, D.R.; Stamenković, O.S.; Veljković, V.B. Application of ionic liquids and deep eutectic solvents in biodiesel production: A review. Renew. Sustain. Energy Rev. 2016, 61, 473–500. [Google Scholar] [CrossRef]
- Heckenbach, M.E.; Romero, F.N.; Green, M.D.; Halden, R.U. Meta-analysis of ionic liquid literature and toxicology. Chemosphere 2016, 150, 266–274. [Google Scholar] [CrossRef]
- Abbott, A.P.; Capper, G.; Davies, D.L.; Rasheed, R.K.; Tambyrajah, V. Novel solvent properties of choline chloride/urea mixtures. Chem. Commun. 2003, 7, 70–71. [Google Scholar] [CrossRef]
- Abbott, A.P.; Boothby, D.; Capper, G.; Davies, D.L.; Rasheed, R.K. Deep Eutectic Solvents Formed between Choline Chloride and Carboxylic Acids: Versatile Alternatives to Ionic Liquids. J. Am. Chem. Soc. 2004, 126, 9142–9147. [Google Scholar] [CrossRef] [PubMed]
- Carriazo, D.; Serrano, M.C.; Gutiérrez, M.C.; Ferrer, M.L.; del Monte, F. Deep-eutectic solvents playing multiple roles in the synthesis of polymers and related materials. Chem. Soc. Rev. 2012, 41, 4996–5014. [Google Scholar] [CrossRef]
- Maugeri, Z.; Domínguez de María, P. Novel choline-chloride-based deep-eutectic-solvents with renewable hydrogen bond donors: Levulinic acid and sugar-based polyols. RSC Adv. 2012, 2, 421–425. [Google Scholar] [CrossRef]
- Chandran, D.; Khalid, M.; Walvekar, R.; Mubarak, N.M.; Dharaskar, S.; Wong, W.Y.; Gupta, T.C.S.M. Deep eutectic solvents for extraction-desulphurization: A review. J. Mol. Liq. 2019, 275, 312–322. [Google Scholar] [CrossRef]
- Li, X.; Row, K.H. Development of deep eutectic solvents applied in extraction and separation. J. Sep. Sci. 2016, 39, 3505–3520. [Google Scholar] [CrossRef] [PubMed]
- Cai, T.; Qiu, H. Application of deep eutectic solvents in chromatography: A review. TrAC Trends Analyt. Chem. 2019, 120, 115623. [Google Scholar] [CrossRef]
- Pätzold, M.; Siebenhaller, S.; Kara, S.; Liese, A.; Syldatk, C.; Holtmann, D. Deep Eutectic Solvents as Efficient Solvents in Biocatalysis. Trends Biotechnol. 2019, 37, 943–959. [Google Scholar] [CrossRef] [PubMed]
- Zainal-Abidin, M.H.; Hayyan, M.; Hayyan, A.; Jayakumar, N.S. New horizons in the extraction of bioactive compounds using deep eutectic solvents: A review. Anal. Chim. Acta 2017, 979, 1–23. [Google Scholar] [CrossRef]
- Zainal-Abidin, M.H.; Hayyan, M.; Ngoh, G.C.; Wong, W.F.; Looi, C.Y. Emerging frontiers of deep eutectic solvents in drug discovery and drug delivery systems. J. Control. Release 2019, 316, 168–195. [Google Scholar] [CrossRef]
- Sánchez, P.B.; González, B.; Salgado, J.; José Parajó, J.; Domínguez, Á. Physical properties of seven deep eutectic solvents based on l-proline or betaine. J. Chem. Thermodyn. 2019, 131, 517–523. [Google Scholar] [CrossRef]
- Haghbakhsh, R.; Bardool, R.; Bakhtyari, A.; Duarte, A.R.C.; Raeissi, S. Simple and global correlations for the densities of deep eutectic solvents. J. Mol. Liq. 2019, 296, 111830. [Google Scholar] [CrossRef]
- Wu, K.-J.; Chen, Q.-L.; He, C.-H. Speed of sound of ionic liquids: Database, estimation, and its application for thermal conductivity prediction. AIChE J. 2014, 60, 1120–1131. [Google Scholar] [CrossRef]
- Haghbakhsh, R.; Keshtkari, S.; Raeissi, S. Simple estimations of the speed of sound in ionic liquids, with and without any physical property data available. Fluid Phase Equilib. 2020, 503, 112291. [Google Scholar] [CrossRef]
- Yebra, F.; Zemánková, K.; Troncoso, J. Speed of sound as a function of temperature and pressure for propane derivatives. J. Chem. Thermodyn. 2017, 109, 117–123. [Google Scholar] [CrossRef]
- Cavuoto, G.; Lago, S.; Giuliano Albo, P.A.; Serazio, D. Speed of sound measurements in liquid methane (CH4) at cryogenic temperatures between (130 and 162) K and at pressures up to 10 MPa. J. Chem. Thermodyn. 2020, 142, 106007. [Google Scholar] [CrossRef]
- Gardas, R.L.; Coutinho, J.A.P. Estimation of speed of sound of ionic liquids using surface tensions and densities: A volume based approach. Fluid Phase Equilib. 2008, 267, 188–192. [Google Scholar] [CrossRef]
- Hekayati, J.; Esmaeilzadeh, F. Predictive correlation between surface tension, density, and speed of sound of ionic liquids: Auerbach model revisited. J. Mol. Liq. 2019, 274, 193–203. [Google Scholar] [CrossRef]
- Singh, M.P.; Singh, R.K. Correlation between ultrasonic velocity, surface tension, density and viscosity of ionic liquids. Fluid Phase Equilib. 2011, 304, 1–6. [Google Scholar] [CrossRef]
- Zhu, J.; Xu, Y.; Feng, X.; Zhu, X. A detailed study of physicochemical properties and microstructure of EmimCl-EG deep eutectic solvents: Their influence on SO2 absorption behavior. J.Ind. Eng. Chem. 2018, 67, 148–155. [Google Scholar] [CrossRef]
- Basaiahgari, A.; Panda, S.; Gardas, R.L. Effect of Ethylene, Diethylene, and Triethylene Glycols and Glycerol on the Physicochemical Properties and Phase Behavior of Benzyltrimethyl and Benzyltributylammonium Chloride Based Deep Eutectic Solvents at 283.15–343.15 K. J. Chem. Eng. Data 2018, 63, 2613–2627. [Google Scholar]
- Basaiahgari, A.; Panda, S.; Gardas, R.L. Acoustic, volumetric, transport, optical and rheological properties of Benzyltripropylammonium based Deep Eutectic Solvents. Fluid Phase Equilib. 2017, 448, 41–49. [Google Scholar] [CrossRef]
- Abdel Jabbar, N.M.; Mjalli, F.S. Ultrasonic study of binary aqueous mixtures of three common eutectic solvents. Phys. Chem. Liquids 2019, 57, 1–18. [Google Scholar] [CrossRef]
- Mjalli, F.S.; Abdel Jabbar, N.M. Acoustic investigation of choline chloride based ionic liquids analogs. Fluid Phase Equilib. 2014, 381, 71–76. [Google Scholar] [CrossRef]
- Shekaari, H.; Zafarani-Moattar, M.T.; Mohammadi, B. Thermophysical characterization of aqueous deep eutectic solvent (choline chloride/urea) solutions in full ranges of concentration at T=(293.15–323.15)K. J. Mol. Liq. 2017, 243, 451–461. [Google Scholar] [CrossRef]
- Lapeña, D.; Lomba, L.; Artal, M.; Lafuente, C.; Giner, B. The NADES glyceline as a potential Green Solvent: A comprehensive study of its thermophysical properties and effect of water inclusion. J. Chem. Thermodyn. 2019, 128, 164–172. [Google Scholar] [CrossRef]
- Vuksanović, J.; Kijevčanin, M.L.; Radović, I.R. Effect of water addition on extraction ability of eutectic solvent choline chloride+ 1,2-propanediol for separation of hexane/heptane+ethanol systems. Korean J. Chem. Eng. 2018, 35, 1477–1487. [Google Scholar] [CrossRef]
- Sas, O.G.; Fidalgo, R.; Domínguez, I.; Macedo, E.A.; González, B. Physical Properties of the Pure Deep Eutectic Solvent, [ChCl]:[Lev] (1:2) DES, and Its Binary Mixtures with Alcohols. J. Chem. Eng. Data 2016, 61, 4191–4202. [Google Scholar] [CrossRef]
- Kuddushi, M.; Nangala, G.S.; Rajput, S.; Ijardar, S.P.; Malek, N.I. Understanding the peculiar effect of water on the physicochemical properties of choline chloride based deep eutectic solvents theoretically and experimentally. J. Mol. Liq. 2019, 278, 607–615. [Google Scholar] [CrossRef]
- Shekaari, H.; Zafarani-Moattar, M.T.; Mokhtarpour, M.; Faraji, S. Volumetric and compressibility properties for aqueous solutions of choline chloride based deep eutectic solvents and Prigogine–Flory–Patterson theory to correlate of excess molar volumes at T = (293.15 to 308.15) K. J. Mol. Liq. 2019, 289, 111077. [Google Scholar] [CrossRef]
- Sas, O.G.; Castro, M.; Domínguez, Á.; González, B. Removing phenolic pollutants using Deep Eutectic Solvents. Sep. Purif. Technol. 2019, 227, 115703. [Google Scholar] [CrossRef]
- Abri, A.; Babajani, N.; Zonouz, A.M.; Shekaari, H. Spectral and thermophysical properties of some novel deep eutectic solvent based on l-menthol and their mixtures with ethanol. J. Mol. Liq. 2019, 285, 477–487. [Google Scholar] [CrossRef]
- Deb, K. An introduction to genetic algorithms. Sadhana 1999, 24, 293–315. [Google Scholar] [CrossRef]
- Valderrama, J.O.; Robles, P.A. Critical Properties, Normal Boiling Temperatures, and Acentric Factors of Fifty Ionic Liquids. Ind. Eng. Chem. Res. 2007, 46, 1338–1344. [Google Scholar] [CrossRef]
- Valderrama, J.O.; Sanga, W.W.; Lazzús, J.A. Critical Properties, Normal Boiling Temperature, and Acentric Factor of Another 200 Ionic Liquids. Ind. Eng. Chem. Res. 2008, 47, 1318–1330. [Google Scholar] [CrossRef]
- Knapp, H.; Döring, R.; Oellrich, L.; Plöcker, U.; Prausnitz, J.M.; Langhorst, R.; Zeck, S. Vapor-liquid equilibria for mixtures of low boiling substances. In Chemistry Data Series VI; DECHEMA: Frankfurt am Mein, Germany, 1982. [Google Scholar]
- Taherzadeh, M.; Haghbakhsh, R.; Duarte, A.R.C.; Raeissi, S. Estimation of the Heat Capacities of Deep Eutectic Solvents. J. Mol. Liq. 2020. under review. [Google Scholar] [CrossRef]
- Pitzer, K.S. Thermodynamics, 3rd ed.; McGraw-Hill: New York, NY, USA, 1995. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |


| DES # | Data Set | HBA | HBD | HBA:HBD Molar Ratio | Ndp 1 | Ref. |
|---|---|---|---|---|---|---|
| DES1 | Test | 1-Ethyl-3-methylimidazolium chloride | Ethylene glycol | 2:1 | 5 | [30] |
| DES2 | Training | 1-Ethyl-3-methylimidazolium chloride | Ethylene glycol | 1:1 | 5 | [30] |
| DES3 | Test | 1-Ethyl-3-methylimidazolium chloride | Ethylene glycol | 1:2 | 5 | [30] |
| DES4 | Training | Benzyl-tributyl-ammonium-chloride | Ethylene glycol | 1:3 | 13 | [31] |
| DES5 | Training | Benzyl-tributyl-ammonium-chloride | Diethylene glycol | 1:3 | 13 | [31] |
| DES6 | Training | Benzyl-tributyl-ammonium-chloride | Triethylene glycol | 1:3 | 13 | [31] |
| DES7 | Test | Benzyl-tributyl-ammonium-chloride | Glycerol | 1:3 | 13 | [31] |
| DES8 | Training | Benzyl-trimethyl-ammonium-chloride | Ethylene glycol | 1:3 | 13 | [31] |
| DES9 | Training | Benzyl-trimethyl-ammonium-chloride | Diethylene glycol | 1:3 | 13 | [31] |
| DES10 | Test | Benzyl-trimethyl-ammonium-chloride | Triethylene glycol | 1:3 | 13 | [31] |
| DES11 | Training | Benzyl-trimethyl-ammonium-chloride | Glycerol | 1:3 | 13 | [31] |
| DES12 | Training | Benzyl-tripropyl-ammonium-chloride | Phenol | 1:3 | 11 | [32] |
| DES13 | Training | Benzyl-tripropyl-ammonium-chloride | Ethylene glycol | 1:3 | 11 | [32] |
| DES14 | Training | Benzyl-tripropyl-ammonium-chloride | Lactic acid | 1:3 | 11 | [32] |
| DES15 | Training | Benzyl-tripropyl-ammonium-chloride | Glycerol | 1:3 | 11 | [32] |
| DES16 | Training | Betaine | Lactic acid | 1:2 | 10 | [21] |
| DES17 | Training | Betaine | Lactic acid | 1:5 | 11 | [21] |
| DES18 | Training | Betaine | Levulinic acid | 1:2 | 11 | [21] |
| DES19 | Training | Betaine | Lactic acid/water | 1:1:1 | 11 | [21] |
| DES20 | Test | Betaine | Citric acid/water | 2:1:6 | 11 | [21] |
| DES21 | Training | Choline-Chloride | Urea | 1:2 | 20 | [33,34,35] |
| DES22 | Test | Choline-Chloride | Ethylene glycol | 1:2 | 13 | [33,34] |
| DES23 | Test | Choline-Chloride | Glycerol | 1:2 | 38 | [33,34,36] |
| DES24 | Training | Choline-Chloride | Fructose | 2:1 | 7 | [34] |
| DES25 | Test | Choline-Chloride | Glucose | 2:1 | 7 | [34] |
| DES26 | Training | Choline-Chloride | 1,2propanediol | 1:3 | 10 | [37] |
| DES27 | Training | Choline-Chloride | Levulinic acid | 1:2 | 11 | [38] |
| DES28 | Training | Choline-Chloride | Malonic acid | 1:1 | 7 | [39] |
| DES29 | Test | Choline-Chloride | Glutaric acid | 1:1 | 7 | [39] |
| DES30 | Training | Choline-Chloride | Oxalic acid | 1:1 | 4 | [40] |
| DES31 | Training | Dodecanoic acid | Octanoic acid | 1:3 | 11 | [41] |
| DES32 | Training | Dodecanoic acid | Decanoic acid | 1:2 | 10 | [41] |
| DES33 | Training | Menthol | Octanoic acid | 1:1 | 11 | [41] |
| DES34 | Training | Menthol | Decanoic acid | 1:1 | 11 | [41] |
| DES35 | Training | Menthol | Salicylic acid | 4:1 | 5 | [42] |
| DES36 | Test | Menthol | Camphor-10-sulfonic acid | 5:1 | 5 | [42] |
| DES37 | Training | Menthol | Ethylene glycol | 1:1 | 5 | [42] |
| DES38 | Test | Proline | Levulinic acid | 1:2 | 11 | [21] |
| DES39 | Training | Proline | Lactic acid | 1:1 | 10 | [21] |
| Total | 420 |
| DES # | Tc (K) | Pc (bar) | Vc (cm3/mol) | ω | Mw (g/mol) |
|---|---|---|---|---|---|
| DES1 | 670.98 | 36.65 | 355.99 | 0.6660 | 118.44 |
| DES2 | 651.23 | 39.77 | 308.96 | 0.7476 | 104.34 |
| DES3 | 632.35 | 43.77 | 264.25 | 0.8293 | 90.25 |
| DES4 | 657.28 | 31.24 | 364.48 | 0.9659 | 124.53 |
| DES5 | 720.58 | 25.62 | 480.62 | 0.9994 | 157.57 |
| DES6 | 778.21 | 22.07 | 589.83 | 1.0507 | 190.61 |
| DES7 | 749.11 | 25.67 | 433.69 | 1.3146 | 147.05 |
| DES8 | 618.43 | 41.08 | 270.56 | 0.8745 | 92.97 |
| DES9 | 678.15 | 31.88 | 377.22 | 0.9080 | 126.01 |
| DES10 | 733.31 | 26.60 | 478.88 | 0.9593 | 159.05 |
| DES11 | 708.07 | 32.89 | 333.89 | 1.2232 | 115.49 |
| DES12 | 701.16 | 37.82 | 380.25 | 0.5152 | 138.05 |
| DES13 | 644.10 | 33.78 | 334.18 | 0.9375 | 114.02 |
| DES14 | 721.27 | 33.15 | 384.56 | 0.9166 | 135.02 |
| DES15 | 735.27 | 27.58 | 401.61 | 1.2862 | 136.53 |
| DES16 | 668.50 | 44.09 | 281.96 | 0.7863 | 99.10 |
| DES17 | 683.07 | 47.23 | 259.82 | 0.8755 | 94.59 |
| DES18 | 701.24 | 38.94 | 356.12 | 0.6195 | 116.46 |
| DES19 | 637.98 | 61.84 | 206.94 | 0.5794 | 75.08 |
| DES20 | 659.71 | 92.43 | 146.46 | 0.5139 | 59.39 |
| DES21 | 644.44 | 49.54 | 254.37 | 0.6509 | 86.58 |
| DES22 | 602.00 | 40.99 | 259.67 | 0.9155 | 87.92 |
| DES23 | 680.67 | 33.46 | 315.17 | 1.2254 | 107.94 |
| DES24 | 742.22 | 27.03 | 424.87 | 1.2278 | 153.13 |
| DES25 | 738.99 | 27.23 | 422.14 | 1.2163 | 153.13 |
| DES26 | 620.93 | 38.44 | 284.11 | 0.9290 | 91.98 |
| DES27 | 702.19 | 35.40 | 376.78 | 0.7301 | 123.95 |
| DES28 | 689.82 | 37.16 | 335.84 | 0.8577 | 121.84 |
| DES29 | 713.43 | 32.24 | 397.17 | 0.8782 | 135.87 |
| DES30 | 676.24 | 40.44 | 303.06 | 0.8531 | 114.83 |
| DES31 | 737.07 | 24.71 | 559.27 | 0.7649 | 158.24 |
| DES32 | 773.88 | 21.55 | 656.40 | 0.8307 | 181.61 |
| DES33 | 717.72 | 28.79 | 493.39 | 0.6173 | 150.24 |
| DES34 | 739.17 | 26.26 | 549.11 | 0.6568 | 164.27 |
| DES35 | 744.23 | 33.56 | 445.77 | 0.5733 | 152.64 |
| DES36 | 777.87 | 31.66 | 504.89 | 0.5094 | 168.94 |
| DES37 | 654.33 | 38.54 | 319.91 | 0.7510 | 109.17 |
| DES38 | 745.61 | 42.88 | 333.41 | 0.7044 | 115.78 |
| DES39 | 721.95 | 48.54 | 272.60 | 0.8243 | 102.61 |
| Data Set | Number of Investigated Data | AARD% |
|---|---|---|
| Training | 292 | 4.8 |
| Test | 128 | 6.8 |
| Overall | 420 | 5.4 |
| DES | Proposed Model | Haghbakhsh et al.’s Model [24] | Hekayati and Esmaeilzadeh’s Model [28] | Gardas and Coutinho’s Model [27] | Singh and Singh’s Model [29] |
|---|---|---|---|---|---|
| DES1 | 13.9 | 14.5 | 8.6 | 15.0 | 4.9 |
| DES2 | 7.5 | 16.6 | 3.6 | 9.0 | 11.8 |
| DES3 | 1.3 | 17.6 | 2.6 | 1.3 | 31.6 |
| DES4 | 5.8 | 2.4 | 8.1 | 5.2 | 33.0 |
| DES5 | 1.8 | 3.3 | 6.6 | 7.1 | 36.5 |
| DES6 | 0.9 | 8.3 | 5.0 | 9.3 | 43.8 |
| DES7 | 5.4 | 9.0 | 4.7 | 14.2 | 84.5 |
| DES8 | 0.7 | 68.8 | 2.8 | 1.9 | 27.2 |
| DES9 | 1.1 | 1.4 | 3.6 | 1.1 | 28.0 |
| DES10 | 2.8 | 7.0 | 2.1 | 2.9 | 32.1 |
| DES11 | 4.6 | 7.6 | 0.9 | 6.8 | 76.5 |
| DES12 | 1.9 | 2.7 | 3.6 | 3.6 | 4.4 |
| DES13 | 2.5 | 1.8 | 4.8 | 1.2 | 26.1 |
| DES14 | 8.9 | 22.0 | 11.9 | 13.7 | 54.0 |
| DES15 | 1.3 | 9.0 | 2.1 | 10.9 | 77.4 |
| DES16 | 1.6 | 1.3 | 4.6 | 2.8 | 37.4 |
| DES17 | 12.5 | 7.6 | 16.7 | 20.1 | 82.7 |
| DES18 | 2.8 | 2.0 | 4.9 | 1.0 | 15.8 |
| DES19 | 1.1 | 1.4 | 0.8 | 3.8 | 29.6 |
| DES20 | 2.4 | 1.5 | 1.8 | 6.2 | 79.5 |
| DES21 | 12.6 | 25.2 | 10.4 | 15.5 | 7.8 |
| DES22 | 4.7 | 10.4 | 2.6 | 7.9 | 13.8 |
| DES23 | 8.1 | 5.2 | 4.5 | 4.1 | 66.2 |
| DES24 | 1.0 | 4.7 | 4.6 | 3.6 | 55.2 |
| DES25 | 21.4 | 25.5 | 25.7 | 19.5 | 19.1 |
| DES26 | 2.5 | 11.6 | 8.8 | 4.0 | 36.6 |
| DES27 | 4.5 | 4.6 | 2.7 | 1.3 | 18.9 |
| DES28 | 5.9 | 3.6 | 3.7 | 4.2 | 30.9 |
| DES29 | 8.7 | 1.3 | 4.0 | 4.5 | 26.4 |
| DES30 | 3.9 | 6.8 | 2.6 | 2.7 | 39.3 |
| DES31 | 3.3 | 15.4 | 31.7 | 25.5 | 36.0 |
| DES32 | 7.1 | 6.6 | 27.3 | 25.3 | 39.1 |
| DES33 | 7.7 | 13.1 | 29.0 | 19.5 | 22.7 |
| DES34 | 2.1 | 11.6 | 26.1 | 18.9 | 23.5 |
| DES35 | 9.2 | 3.1 | 24.3 | 17.8 | 36.8 |
| DES36 | 2.6 | 5.7 | 19.7 | 13.3 | 26.7 |
| DES37 | 21.2 | 2.6 | 28.0 | 20.8 | 53.8 |
| DES38 | 6.0 | 2.2 | 12.8 | 13.7 | 56.8 |
| DES39 | 4.8 | 2.6 | 7.8 | 13.1 | 75.8 |
| Total | 5.4 | 9.7 | 8.8 | 9.1 | 40.8 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Peyrovedin, H.; Haghbakhsh, R.; Duarte, A.R.C.; Raeissi, S. A Global Model for the Estimation of Speeds of Sound in Deep Eutectic Solvents. Molecules 2020, 25, 1626. https://doi.org/10.3390/molecules25071626
Peyrovedin H, Haghbakhsh R, Duarte ARC, Raeissi S. A Global Model for the Estimation of Speeds of Sound in Deep Eutectic Solvents. Molecules. 2020; 25(7):1626. https://doi.org/10.3390/molecules25071626
Chicago/Turabian StylePeyrovedin, Hamed, Reza Haghbakhsh, Ana Rita C. Duarte, and Sona Raeissi. 2020. "A Global Model for the Estimation of Speeds of Sound in Deep Eutectic Solvents" Molecules 25, no. 7: 1626. https://doi.org/10.3390/molecules25071626
APA StylePeyrovedin, H., Haghbakhsh, R., Duarte, A. R. C., & Raeissi, S. (2020). A Global Model for the Estimation of Speeds of Sound in Deep Eutectic Solvents. Molecules, 25(7), 1626. https://doi.org/10.3390/molecules25071626






