Abstract
An esterification and amination of benzylic C–H bonds was developed by using 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) under metal- and iodide-free conditions. Both carboxylic acids and amines could be used as ideal coupling partners for the oxidative coupling reactions with various diarylmethanes. A close to equal amount of coupling reagents was enough to afford the product in good to high yields.
1. Introduction
Benzyl esters, which play a key role in many biologically active compounds, can be traditionally prepared by condensation of the corresponding benzyl alcohols with carboxylic acids in the presence of condensation reagents or their activated derivatives such as acyl chlorides in the presence of bases [1,2]. They also serve as the essential protecting groups in amino acids and their derivatives [3]. Therefore, development of efficient methods for rapid construction of benzyl esters has gained much attention and great progress has been made during the past several decades. Recently, the cleavage and functionalization of C–H bonds is of increasing interest for both academia and industry. Generally, the transformation mainly relies on transition metals [4,5,6]. This strategy has also been successfully employed for the direct esterification and amination of C–H bonds including benzylic C–H bonds under oxidative reaction conditions. Various transition-metals efficiently catalyzed the direct esterification [7,8] and amination [9,10,11] of C–H bonds. Due to the toxicity and cost of transition-metal catalysts, increasing interest has been paid on metal-free esterifications of benzylic C–H bonds.
Since Sinha and co-workers reported a 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) promoted acetoxylation of arylalkanes under sonochemical activation conditions in the absence of metal catalyst [12], various efficient esterification methods have been developed under metal-free conditions. The combination of quaternary ammonium iodides with strong oxidants was found to be an effective strategy for direct esterification and amination of benzylic C–H bonds [13,14,15,16]. Recently, Shen and co-workers reported the C–O coupling of C–H with carboxylic acids catalyzed by DDQ and tert-butyl nitrite using oxygen as the terminal oxidant [17]. However, large excess of carboxylic acids or amines are necessary to get satisfied reaction yields [18]. The development of esterification reactions using a near equal amount of reactants under metal-free conditions would be highly desirable. Herein, we report a DDQ promoted esterification and amination of diarylmethanes using only 1.5 equiv. of carboxylic acids and amines without the use of quaternary ammonium iodide or metal catalyst. The chloro-containing solvent plays an important role for this efficient transformation.
2. Results
2.1. Optimization of Reaction Conditions for Synthesis of Benzhydryl Benzoate 3a
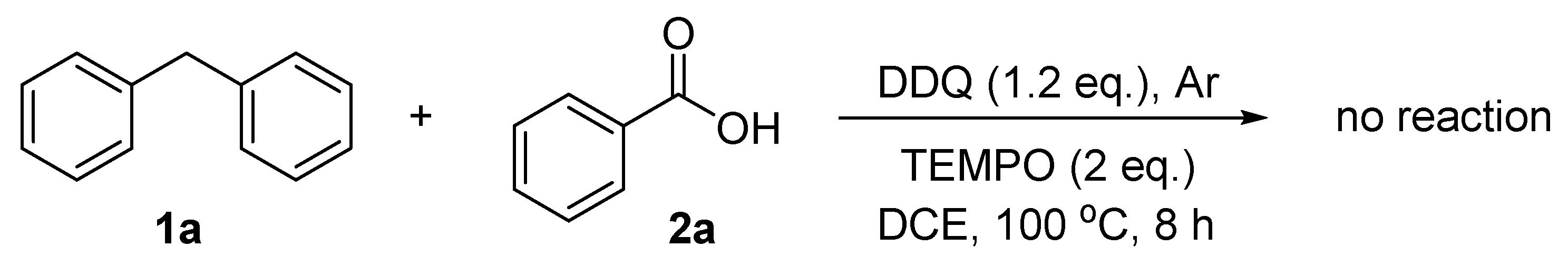
We began our study by examining the reaction of diphenylmethane (1a) and with 1.5 equiv. of benzoic acid (2a) using DDQ as oxidant in 1,2-dichloroethane (DCE) at 100 °C under an atmosphere of argon. DDQ showed very good activity and the desired product benzhydryl benzoate (3a) was obtained in 95% yield as determined by GC (Table 1, entry 1). The oxidant played an important role for this kind of reaction. Other common oxidants, such as benzoquinone, peroxides and hydrogen peroxide, were ineffective and no desired product was observed (entries 2–6). Solvents also played an important role for the direct esterification. The reactions in chloro-containing solvents, such as chloroform, dichloromethane, and tetrachloroethane, all showed very good reactivity (entries 7–9). Other solvents, such as ethyl acetate, acetonitrile, and water, all decreased the reaction yields significantly (entries 10–13). Decreasing the reaction temperature decreased the reaction yield (entry 14). The reaction yields decreased when the reactions were carried out under oxygen and air (entries 15 and 16). Good yield still could be achieved when an equal amount of 2a was used (entry 17).

Table 1.
Optimization of the reaction conditions a.
2.2. Substrate Scope for the Carboxylic Acids and Amines
With the optimized reaction conditions established, the scope of the reaction with respect to diphenylmethane (1a) and various carboxylic acids (2) was investigated (Table 2). The reactions with aromatic carboxylic acids bearing electron-donating groups and electron-withdrawing substituents at the para position proceeded smoothly to give the desired products in good yields (3b–3e). The position of the substituents on the phenyl ring of benzoic acids affected the reaction yield slightly, and the use of ortho-substituted benzoic acids also afforded the desired products in good yields. Under the optimized conditions, halogen substituents were all well tolerated (3e–3h). Good yield was obtained when 2-bromobenzoic acid was used, and the desired product 3h was achieved in 83% yield. More bulky substrates, such as 2,4,6-trimethylbenzoic acid, also efficiently reacted with 1a and gave the product in 85% yield (3j). Notably, high yield was achieved when hetero aromatic carboxylic acids, such as 3-methylthiophene-2-carboxylic acid, reacted with 1a, and the desired product 3k was obtained in 88% yield. To be our delight, aliphatic carboxylic acids, including formic acid, are also good coupling reagents for this kind of transformation, and the desired esters were formed in high yields (3m–3p). In addition to carboxylic acids, amines could also be used as ideal coupling partners for the oxidative coupling reaction. Diphenylmethane smoothly coupled with benzamide and gave the aminated product 4a in 62% yield. Moderate to good yields were obtained when sulfonamides were employed for the coupling reaction (4b–4d).

Table 2.
Reaction of 1a with various carboxylic acids and amines a.
2.3. Substrate Scope for the Diarylmethanes
To further explore the scope of the reaction, various diarylmethanes (1) were employed to react with 2a under the optimized conditions (Table 3). A series of functional groups, including methyl, tert-butyl, fluoro, and chloro, were well tolerated under the optimized conditions (3t–3x). The position of the substituents on the phenyl ring of diphenylmethane only slightly affected the reaction yield (3q–3s). The desired product 3y was obtained in 40% yield when 1-benzyl-4-nitrobenzene was used. Unfortunately, this reaction condition was only suitable for diarylmethane substrates. Other benzylic substrates, such as toluene and ethylbenzene, were not reactive under the optimized reaction conditions.

Table 3.
Esterification of various diarylmethanes a.
2.4. Mechanism
To gather more information about the reaction mechanism, the radical scavenger 2,2,6,6-tetramethyl-piperidine-1-oxyl (TEMPO) was added. No product was obtained from the reaction between diphenylmethane and benzoic acid with the influence of two equivalents of TEMPO (Scheme 1). This indicates that the present reaction process may involve radical intermediates.
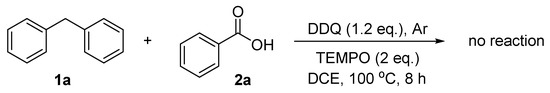
Scheme 1.
Radical trapping experiment.
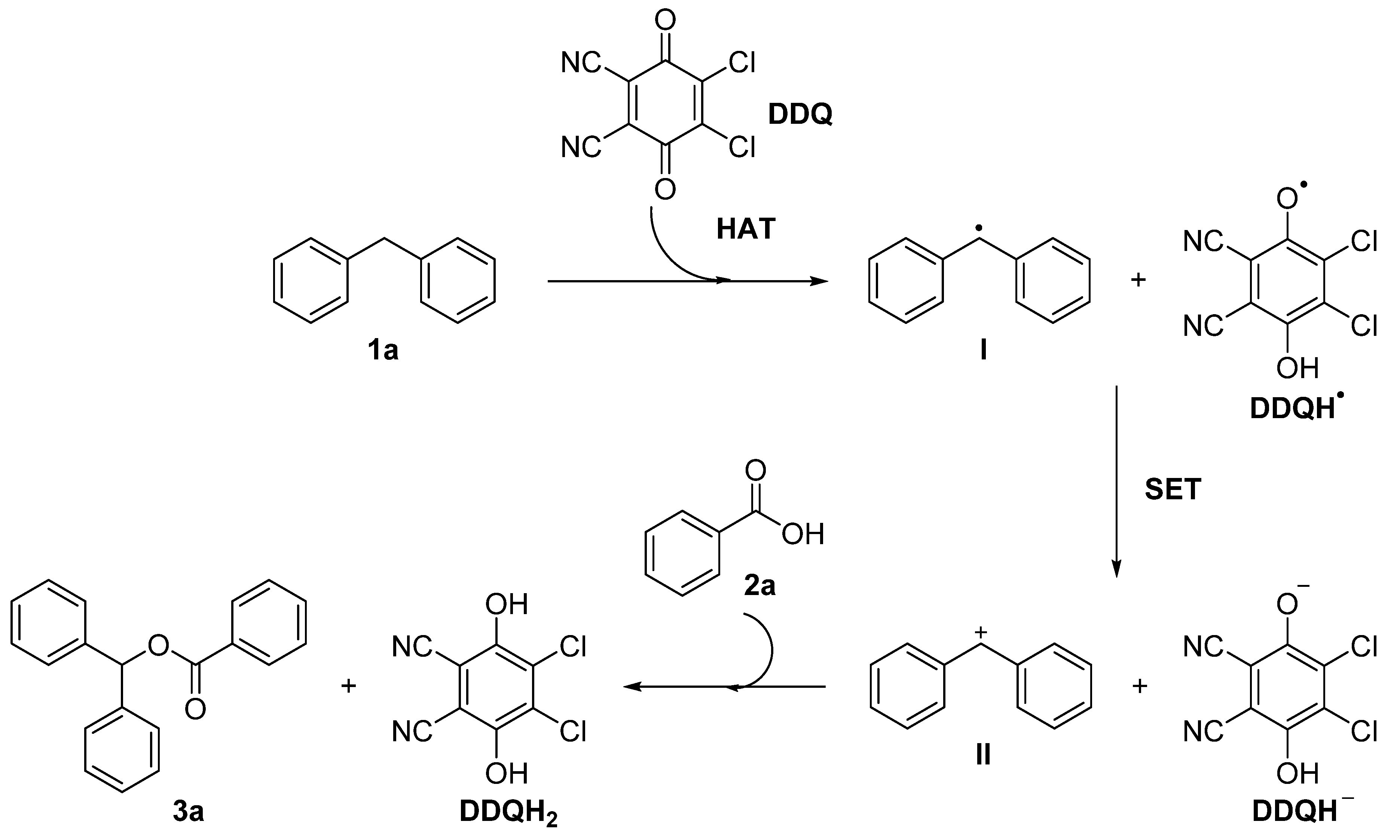
A plausible mechanism to rationalize this transformation is illustrated in Scheme 2. The reaction of DDQ with diphenylmethane 1a generates a benzyl radical I and DDQH· via hydrogen atom transfer (HAT). The radicals continue to be transformed into the corresponding benzyl cation and DDQH anion via single electron transfer (SET) [17,18]. Finally, the benzoic acid 2a of the proton is abstracted by DDQH– and reacts with the cation II to obtain the desired product 3a and reduced hydroquinone DDQH2. However, the reason why the reaction only works well in chloro-containing solvents is not clear at this stage.
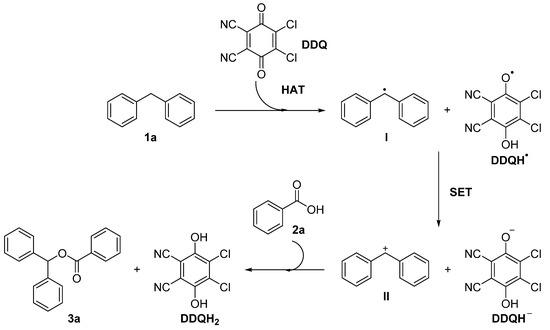
Scheme 2.
Proposed mechanism for the esterification of diarylmethanes.
3. Materials and Methods
3.1. General Information
All experiments were carried out under an atmosphere of argon. Flash column chromatography was performed over silica gel 48–75 μm. 1H-NMR and 13C-NMR spectra were recorded on a Bruker-AV (400 and 100 MHz, respectively) instrument (Billerica, MA, USA) internally referenced to SiMe4 or chloroform signals. MS analyses were performed on an Agilent 5975 GC-MS instrument (EI) (Santa Clara, CA, USA). The new compounds were characterized by 1H NMR, 13C NMR, MS, and HRMS. The structure of known compounds were further corroborated by comparing their 1H-NMR, 13C-NMR, and MS data with those in the literature. All reagents were used as received from commercial sources without further purification. Diarylmethane derivatives 1 were synthesized based on relevant literatures [19].
3.2. General Procedure for the Synthesis of Benzhydryl Benzoate
An oven-dried pressure tube (10 mL) was charged with diphenylmethane (1a, 33.4 μL, 0.2 mmol), benzoic acid (2a, 36.6 mg, 0.3 mmol), DDQ (54.5 mg, 0.24 mmol), and DCE (0.6 mL). The reaction vessel was flushed with argon and sealed. The resulting solution was heated to 100 °C for 8 h. After cooling to room temperature, the volatiles were removed under vacuum and the residue was purified by column chromatography (silica gel, petroleum ether/ethyl acetate = 9:1) to give 3a as white solid; yield: 46.1 mg (80%). (NMR spectra for all compounds shown in the Supplementary Materials).
3.3. Product Characterization
Benzhydryl benzoate (3a) [17]: white solid, 80% yield (46.1 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.15 (d, J = 7.6 Hz, 2H), 7.58 (d, J = 7.0 Hz, 1H), 7.43–7.46 (m, 6H), 7.36 (t, J = 7.4 Hz, 4H), 7.30 (d, J = 7.2 Hz, 2H), 7.12 (s, 1H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.6, 140.4, 133.1, 130.1, 129.8, 128.6, 128.5, 128.0, 127.2, 77.5. MS (EI) m/z (%) 288, 183, 166 (100), 152, 105, 77.
Benzhydryl 4-methylbenzoate (3b) [20]: white solid, 80% yield (48.3 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.03 (d, J = 8.0 Hz, 2H), 7.27–7.44 (m, 12H), 7.10 (s, 1H), 2.42 (s, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.7, 143.9, 140.5, 129.9, 129.2, 128.6, 128.0, 127.6, 127.2, 77.3, 21.7. MS (EI) m/z (%) 302, 166 (100), 152, 119, 91.
Benzhydryl 3-methylbenzoate (3c): White solid, 84% yield (50.7 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.94 (m, 2H), 7.30–7.45 (m, 12H), 7.12 (s, 1H), 2.41 (s, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.8, 140.4, 138.3, 134.0, 132.5, 130.3, 128.6, 128.4, 128.0, 127.2, 127.0, 77.4, 21.4; MS (EI) m/z (%) 302, 166 (100), 152, 119, 91; HRMS calcd. for: C21H18NaO2 [M + Na]+: 325.1201, found m/z 325.1205.
Benzhydryl 2-methylbenzoate (3d) [21]: colorless liquid, 82% yield (49.5 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.08 (d, J = 7.6 Hz, 1H), 7.31–7.46 (m, 13H), 7.01 (s, 1H), 2.61 (s, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 166.4, 140.7, 140.5, 132.2, 131.9, 130.8, 129.5, 128.6, 128.0, 127.2, 125.9, 77.4, 22.0; MS (EI) m/z (%) 302, 166 (100), 152, 119, 91.
Benzhydryl 4-fluorobenzoate (3e) [22]: colorless liquid, 70% yield (42.9 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.10 (t, J = 6.8 Hz, 2H), 7.20–7.37 (m, 10H), 7.04–7.09 (m, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.9 (d, J = 259.2 Hz), 164.7, 140.2, 132.4 (d, J = 9.3 Hz), 130.0, 128.6, 128.0, 127.2, 115.6 (d, J = 21.9 Hz), 77.7; MS (EI) m/z (%) 306, 183, 166 (100), 152, 123, 95.
Benzhydryl 2-fluorobenzoate (3f) [23]: colorless liquid, 82% yield (50.2 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.91 (d, J = 7.2 Hz, 1H), 7.69 (d, J = 7.6 Hz, 1H), 7.47 (d, J = 7.2 Hz, 4H), 7.32–7.40 (m, 8H), 7.14 (s, 1H); 13C NMR (100 MHz, CDCl3, ppm) δ 163.5, 162.2 (d, J = 258.9 Hz), 140.2, 134.7 (d, J = 9.0 Hz), 132.4, 128.6, 128.0, 127.2, 124.1 (d, J = 4.0 Hz), 118.8, 117.1 (d, J = 22.4 Hz), 78.0; MS (EI) m/z (%) 306, 183, 166 (100), 152, 123, 95.
Benzhydryl 2-chlorobenzoate (3g) [23]: colorless liquid, 81% yield (52.3 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.93 (d, J = 7.6 Hz, 1H), 7.30–7.46 (m, 13H), 7.13 (s, 1H); 13C NMR (100 MHz, CDCl3, ppm) δ 164.6, 140.0, 134.1, 132.7, 131.7, 131.2, 130.1, 128.6, 128.0, 127.3, 126.6, 78.4; MS (EI) m/z (%) 322, 183, 166 (100), 152, 139, 77.
Benzhydryl 2-bromobenzoate (3h) [24]: white solid, 83% yield (61.0 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.96 (t, J = 7.2 Hz, 1H), 7.10–7.48 (m, 13H), 7.07 (s, 1H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.1, 139.9, 134.6, 132.8, 132.0, 131.6, 128.6, 128.1, 127.3, 127.2, 122.0, 78.5. MS (EI) m/z (%) 366, 183, 167 (100), 152, 77.
Benzhydryl 2-methoxybenzoate (3i) [24]: colorless liquid, 70% yield (44.6 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.91 (d, J = 7.6 Hz, 1H), 7.28–7.50 (m, 11H), 7.10 (s, 1H), 6.97–7.00 (m, 2H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.1, 159.6, 140.6, 133.8, 132.0, 128.5, 127.8, 127.3, 126.6, 120.2, 112.2, 77.3, 56.0. MS (EI) m/z (%) 318, 183, 167 (100), 152, 135, 77.
Benzhydryl 2,4,6-trimethylbenzoate (3j): colorless liquid, 85% yield (56.2 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.30–7.43 (m, 10H), 7.17 (s, 1H), 6.84 (s, 2H), 2.29 (s, 3H), 2.17 (s, 6H); 13C NMR (100 MHz, CDCl3, ppm) δ 169.3, 140.1, 139.4, 135.2, 130.9, 128.5, 128.4, 128.0, 127.4, 77.5, 21.2, 19.7. MS (EI) m/z (%) 330, 167 (100), 155, 147, 77; HRMS calcd. for: C23H22NaO2 [M + Na]+: 353.1514, found m/z 353.1509.
Benzhydryl 3-methylthiophene-2-carboxylate (3k): colorless liquid, 88% yield (54.3 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.30–7.46 (m, 11H), 7.07 (s, 1H), 6.94 (d, J = 4.8 Hz, 1H), 2.59 (s, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 161.7, 146.7, 140.4, 131.8, 130.3, 128.6, 127.9, 127.1, 126.7, 77.2, 16.0; MS (EI) m/z (%) 308, 167 (100), 152, 125, 77; HRMS calcd. for: C19H16NaO2S [M + Na]+: 331.0765, found m/z 331.0768.
Benzhydryl cinnamate (3l) [25]: white solid, 82% yield (51.6 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.76 (d, J = 16.0 Hz, 1H), 7.54 (m, 2H), 7.30–7.39 (m, 13H), 7.02 (s, 1H), 6.57 (d, J = 16.4 Hz, 1H); 13C NMR (100 MHz, CDCl3, ppm) δ 166.0, 145.4, 140.3, 134.4, 130.4, 128.9, 128.6, 128.2, 127.9, 127.2, 118.1, 77.1; MS (EI) m/z (%) 314, 268, 236, 167 (100), 131, 77.
Benzhydryl formate (3m) [26]: colorless liquid, 80% yield (34.0 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.24 (s, 1H), 7.26–7.36 (m, 10H), 7.01 (s, 1H); 13C NMR (100 MHz, CDCl3, ppm) δ 159.9, 139.6, 128.6, 128.1, 127.2, 76.6. MS (EI) m/z (%) 212, 184, 166 (100), 152, 77.
Benzhydryl acetate (3n) [17]: colorless liquid, 93% yield (42.1 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.28–7.35 (m, 10H), 6.89 (s, 1H), 2.16 (s, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 170.0, 140.3, 128.5, 127.9, 127.2, 76.9, 21.3. MS (EI) m/z (%) 226, 184, 165 (100), 152, 105, 77.
Benzhydryl pivalate (3o) [22]: white solid, 83% yield (44.5 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.30–7.36 (m, 10H), 6.84 (s, 1H), 1.27 (s, 9H); 13C NMR (100 MHz, CDCl3, ppm) δ 177.3, 140.7, 128.9, 127.8, 126.9, 76.6, 39.0, 27.2. MS (EI) m/z (%) 268, 211, 183, 167 (100), 152, 57.
Benzhydryl hexanoate (3p) [27]: colorless liquid, 82% yield (46.3 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.30–7.36 (m, 10H), 6.90 (s, 1H), 2.43 (t, J = 7.4 Hz, 2H), 1.66–1.72 (m, 2H), 1.27–1.31 (m, 4H), 0.89 (t, J = 7.4 Hz, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 172.8, 140.5, 128.5, 127.9, 127.1, 76.7, 34.6, 31.3, 24.7, 22.3, 13.9. MS (EI) m/z (%) 282, 184, 166 (100), 152, 77.
Phenyl(p-tolyl)methyl benzoate (3q) [17]: white solid, 82% yield (49.6 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.16 (d, J = 7.2 Hz, 2H), 7.60 (t, J = 7.2 Hz, 1H), 7.31–7.50 (m, 9H), 7.18 (d, J = 7.6 Hz, 2H), 7.11 (s, 1H), 2.35 (s, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.6, 140.5, 137.7, 137.5, 133.1, 130.5, 129.8, 129.3, 128.5, 128.4, 127.9, 127.2, 127.1, 77.4, 21.1. MS (EI) m/z (%) 302, 197, 180 (100), 165 (100), 105, 77.
Phenyl(m-tolyl)methyl benzoate (3r): colorless liquid, 80% yield (48.4 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.17 (d, J = 7.6 Hz, 2H), 7.60 (t, J = 7.2 Hz, 1H), 7.27–7.50 (m, 10H), 7.11–7.13 (m, 2H), 2.36 (s, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.6, 140.5, 140.3, 138.2, 133.1, 130.5, 129.8, 128.8, 128.5, 128.5, 128.4, 127.9, 127.2, 124.2, 77.6, 21.5. MS (EI) m/z (%) 302, 197, 180, 165 (100), 105, 77. HRMS calcd. for: C21H18NaO2 [M]+: m/z 325.11990, found m/z 325.12047.
Phenyl(o-tolyl)methyl benzoate (3s): colorless liquid, 88% yield (53.2 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.15 (d, J = 7.6 Hz, 2H), 7.22–7.61 (m, 13H), 2.39 (s, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.6, 139.6, 138.2, 136.0, 133.1, 130.7, 130.1, 129.8, 128.5, 128.4, 128.0, 127.9, 127.5, 127.2, 126.2, 75.0, 19.5; MS (EI) m/z (%) 302, 179 (100), 165, 105, 77; HRMS calcd. for: C21H18NaO2 [M + Na]+: 325.1199, found 325.1202.
(4-Fluorophenyl)(phenyl)methyl benzoate (3t): colorless liquid, 80% yield (49.0 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.15 (d, J = 7.2 Hz, 2H), 7.61 (d, J = 7.0 Hz, 1H), 7.33–7.51 (m, 9H), 7.13 (s, 1H), 7.06 (t, J = 8.6 Hz, 2H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.5, 162.5 (d, J = 245.4 Hz), 140.1, 136.2, 133.2, 132.7 (d, J = 9.0 Hz), 129.8, 129.1 (d, J = 8.0 Hz), 128.6, 128.5, 128.1, 127.0, 111.5 (d, J = 21.5 Hz), 76.8. MS (EI) m/z (%) 306, 201, 184 (100), 165, 105, 77. HRMS calcd. for: C20H15FNaO2 [M]+: m/z 329.09483, found m/z 329.09537.
(2-Fluorophenyl)(phenyl)methyl benzoate (3u): colorless liquid, 84% yield (51.5 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.15 (d, J = 7.5 Hz, 2H), 7.47–7.60 (m, 6H), 7.28–7.37 (m, 5H), 7.15 (t, J = 7.4 Hz, 1H), 7.07 (t, J = 9.2 Hz, 1H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.3, 160.1 (d, J = 246.8 Hz), 139.4, 133.2, 130.2, 129.8, 129.7 (d, J = 8.2 Hz), 128.6, 128.5, 128.1, 128.1, 127.9 (d, J = 13.2 Hz), 126.9, 124.3 (d, J = 3.6 Hz), 115.8 (d, J = 21.3 Hz), 71.8 (d, J = 2.9 Hz). MS (EI) m/z (%) 306, 184 (100), 165, 105, 77. HRMS calcd. for: C20H15FNaO2 [M]+: m/z 329.09483, found m/z 329.09526.
(4-Chlorophenyl)(phenyl)methyl benzoate (3v): colorless liquid, 78% yield (50.4 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.13 (d, J = 7.6 Hz, 2H), 7.59 (t, J = 7.2 Hz, 1H), 7.31–7.49 (m, 11H), 7.08 (s, 1H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.5, 139.9, 138.9, 133.9, 133.3, 131.5, 129.8, 128.8, 128.7, 128.6, 128.5, 128.2, 127.1, 76.8. MS (EI) m/z (%) 322, 200, 165 (100), 105, 77.
(4-Tert-butylphenyl)(phenyl)methyl benzoate (3w): colorless liquid, 76% yield (52.4 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.15 (d, J = 7.5 Hz, 2H), 7.57 (t, J = 7.1 Hz, 1H), 7.44–7.47 (m, 4H), 7.29–7.37 (m, 7H), 7.11 (s, 1H), 1.30 (s, 9H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.6, 150.9, 140.5, 137.3, 133.0, 130.5, 129.8, 128.5, 128.4, 127.8, 127.1, 127.0, 125.5, 77.4, 34.6, 31.3; MS (EI) m/z (%) 344, 239, 222 (100), 207, 105, 77; HRMS calcd. for: C24H24NaO2 [M + Na]+: 367.1669, found 367.1669.
(4-Chlorophenyl)(p-tolyl)methyl benzoate (3x): colorless liquid, 72% yield (48.5 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.12 (d, J = 7.5 Hz, 2H), 7.58 (t, J = 7.2 Hz, 1H), 7.46 (t, J = 7.6 Hz, 2H), 7.31–7.37 (m, 6H), 7.17 (d, J = 7.7 Hz, 2H), 7.05 (s, 1H), 2.34 (s, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 165.5, 139.1, 138.0, 136.9, 133.8, 133.2, 130.2, 129.8, 129.4, 128.7, 128.5, 128.5, 127.1, 76.7, 21.1. MS (EI) m/z (%) 336, 214 (100), 179 (100), 165, 105, 77. HRMS calcd. for: C21H17ClNaO2 [M]+: m/z 359.08093, found m/z 359.08145.
(4-Nitrophenyl)(phenyl)methyl benzoate (3y): colorless liquid, 40% yield (26.7 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 8.22 (d, J = 8.5 Hz, 2H), 8.14 (d, J = 7.5 Hz, 2H), 7.61 (d, J = 8.4 Hz, 3H), 7.49 (t, J = 7.6 Hz, 2H), 7.34–7.44 (m, 5H), 7.16 (s, 1H).13C NMR (100 MHz, CDCl3, ppm) δ 165.3, 147.4, 138.9, 133.5, 130.7, 130.1, 129.8, 128.9, 128. 7, 128.6, 127.8, 127.3, 123.9, 76.5. MS (EI) m/z (%) 333, 211, 165, 105 (100), 77.
N-Benzhydrylbenzamide (4a) [28]: white solid, 62% yield (35.6 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.85 (d, J = 7.6 Hz, 2H), 7.54 (t, J = 7.2 Hz, 1H), 7.47 (t, J = 7.4 Hz, 2H), 7.32–7.40 (m, 10H), 6.68 (d, J = 6.8 Hz, 1H), 6.48 (d, J = 7.6 Hz, 1H); 13C NMR (100 MHz, CDCl3, ppm) δ 166.6, 141.5, 134.3, 131.7, 131.0, 128.8, 128.7, 127.6, 127.1, 57.5. MS (EI) m/z (%) 287, 182, 165, 105 (100), 77.
N-Benzhydrylbenzenesulfonamide (4b) [29]: white solid, 76% yield (49.2 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.70 (d, J = 7.6 Hz, 2H), 7.49 (t, J = 7.4 Hz, 1H), 7.36 (t, J = 7.6 Hz, 2H), 7.11–7.23 (m, 10H), 5.63 (d, J = 7.2 Hz, 1H), 5.11 (d, J = 6.8 Hz, 1H); 13C NMR (100 MHz, CDCl3, ppm) δ 140.4, 132.4, 131.0, 128.8, 128.6, 127.7, 127.4, 127.2, 61.4. MS (EI) m/z (%) 322, 246, 182 (100), 167, 104, 77.
N-Benzhydryl-4-methylbenzenesulfonamide (4c) [29]: white solid, 70% yield (47.2 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.50 (d, J = 8.0 Hz, 2H), 7.04–7.20 (m, 12H), 5.51 (d, J = 6.8 Hz, 1H), 4.94 (d, J = 6.4 Hz, 1H), 2.32 (s, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 143.1, 140.7, 137.6, 129.3, 128.5, 127.5, 127.4, 127.2, 61.4, 21.4. MS (EI) m/z (%) 336, 182 (100), 167, 91, 77.
N-Benzhydrylmethanesulfonamide (4d) [30]: white solid, 67% yield (35.0 mg), 1H NMR (400 MHz, CDCl3, ppm) δ 7.36–7.41 (m, 10H), 5.79 (d, J = 6.8 Hz, 1H), 4.99 (d, J = 6.4 Hz, 1H), 2.30 (s, 3H); 13C NMR (100 MHz, CDCl3, ppm) δ 140.7, 128.9, 128.0, 127.5, 61.3, 41.9. MS (EI) m/z (%) 259, 180 (100), 165, 104, 77.
4. Conclusions
In summary, we have developed a DDQ-promoted esterification and amination of benzylic C–H bonds under metal- and iodide-free conditions. A close to equal amount of coupling reagents is enough to afford the product in good to high yields. Functional groups, such as methyl, methoxy, fluoro, chloro, and bromo, were all well tolerated under the optimized reaction conditions. This method affords an efficient alternative route for the synthesis of esters and amides. The scope, mechanism, and synthetic applications of this reaction are under investigation.
Supplementary Materials
The supplementary materials are available online.
Author Contributions
Conceptualization, S.L. and J.Z.; methodology, R.C.; writing—original draft preparation, S.L. and G.H.; writing—review and editing, S.L.
Funding
We gratefully thank the Normal Project Foundation of Hunan Provincial Education Department (18C0845), the Key Project Foundation of Hunan Provincial Education Department (18A396, 19A085), and the Natural Science Foundation of Hunan Province (2017JJ2018).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Otera, J.; Nishikido, J. Esterification. Methods, Reactions, and Applications, 2nd ed.; WILEY-VCH: Weinheim, Germany, 2010. [Google Scholar]
- Aliboni, A.; D’Andrea, A.; Massanisso, P. Propolis Specimens from Different Locations of Central Italy: Chemical Profiling and Gas Chromatography-mass Spectrometry (GC-MS) Quantitative Analysis of the Allergenic Esters Benzyl Cinnamate and Benzyl Salicylate. J. Agric. Food Chem. 2011, 59, 282–288. [Google Scholar] [CrossRef] [PubMed]
- Isidro-Llobet, A.; Alvarez, M.; Albericio, F. Amino Acid-Protecting Groups. Chem. Rev. 2009, 109, 2455–2504. [Google Scholar] [CrossRef] [PubMed]
- Tzouras, N.V.; Stamatopoulos, I.K.; Papastavrou, A.T.; Liori, A.A.; Vougioukalakis, G.C. Sustainable Metal Catalysis in C–H Activation. Coord. Chem. Rev. 2017, 343, 25–138. [Google Scholar] [CrossRef]
- Gandeepan, P.; Müller, T.; Zell, D.; Gianpiero, C.; Warratz, S.; Ackermann, L. 3d Transition Metals for C-H Activation. Chem. Rev. 2019, 119, 2192–2452. [Google Scholar] [CrossRef]
- Kantam, M.L.; Gadipelly, C.; Deshmukh, G.; Reddy, K.R.; Bhargava, S. Copper Catalyzed C−H Activation. Chem. Rec. 2019, 19, 1302–1318. [Google Scholar] [CrossRef]
- Kumar, S.; Pradhan, S.; Roy, S.; De, P.B. Iron-Catalyzed Regioselective Remote C(sp2)-H Carboxylation of Naphthyl and Quinoline Amides. J. Org. Chem. 2019, 84, 10481–10489. [Google Scholar] [CrossRef]
- Li, Z.Y.; Jing, K.; Li, Q.L.; Wang, G.W. Palladium-Catalyzed Decarboxylative Coupling of Potassium Oxalate Monoester with 2-Aryloxypyridines. Acta Chim. Sinica 2019, 77, 729–734. [Google Scholar] [CrossRef]
- Long, J.G.; Le, L.Y.; Iwasaki, T.; Qiu, R.H.; Kambe, N. Copper-Catalyzed Amination of C(sp3)-H bonds: From Anilides to Indolines. J. Org. Chem. 2020, 85, 482–492. [Google Scholar] [CrossRef]
- Tong, H.R.; Zheng, W.R.; Lv, X.Y.; He, G.; Liu, P.; Chen, G. Asymmetric Synthesis of β-Lactam via Palladium-Catalyzed Enantioselective Intramolecular C(sp3)-H Amidation. ACS Catal. 2020, 10, 114–120. [Google Scholar] [CrossRef]
- Ye, L.; Tian, Y.; Meng, X.; Gu, Q.S.; Liu, X.Y. Enantioselective Copper(I)/Chiral Phosphoric Acid Catalyzed Intramolecular Amination of Allylic and Benzylic C-H Bonds. Angew. Chem. Int. Ed. 2020, 59, 1129–1133. [Google Scholar] [CrossRef]
- Kumar, V.; Sharma, A.; Sharma, M.; Sharma, U.K.; Sinha, A.K. DDQ Catalyzed Benzylic Acetoxylation of Arylalkanes: A Case of Exquisitely Controlled Oxidation under Sonochemical Activation. Tetrahedron 2007, 63, 9718–9723. [Google Scholar] [CrossRef]
- Chen, L.; Shi, E.B.; Wan, X.B. Bu4NI-Catalyzed C-O Bond Formation by Using a Cross-Dehydrogenative Coupling (CDC) Reaction. Chem. Eur. J. 2011, 17, 4085–4089. [Google Scholar] [CrossRef]
- Uyanik, M.; Suzuki, D.; Yasui, T.; Ishihara, K. In Situ Generated (Hypo)Iodite Catalysts for the Direct α-Oxyacylation of Carbonyl Compounds with Carboxylic Acids. Angew. Chem. Int. Ed. 2011, 50, 5331–5334. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Liang, S.; Chen, S.Y.; Zhang, J.; Fu, S.S.; Yu, X.Q. A Metal-Free Oxidative Esterification of the Benzyl CH Bond. Adv. Synth. Catal. 2012, 354, 1287–1292. [Google Scholar] [CrossRef]
- Baba, H.; Moriyama, K.; Togo, H. Benzylic-Acetoxylation of Alkylbenzenes with PhI(OAc)2 in the Presence of Catalytic Amounts of TsNH2 and I2. Tetrahedron Lett. 2011, 52, 4303–4307. [Google Scholar] [CrossRef]
- Pan, D.C.; Pan, Z.L.; Hu, Z.M.; Li, M.C.; Hu, X.Q.; Jin, L.Q.; Sun, N.; Hu, B.X.; Shen, Z.L. Metal-Free Aerobic Oxidative C-O Coupling of C(sp3)-H with Carboxylic Acids Catalyzed by DDQ and tert-Butyl Nitrite. Eur. J. Org. Chem. 2019, 5650–5655. [Google Scholar] [CrossRef]
- Yi, H.; Liu, Q.; Liu, J.; Zeng, Z.; Yang, Y.H.; Lei, A.W. DDQ-Catalyzed Oxidative C-O Coupling Of sp3 C-H Bonds with Carboxylic Acids. ChemSusChem 2012, 5, 2143–2146. [Google Scholar] [CrossRef]
- Nobre, S.M.; Monteiro, A.L. Synthesis of Diarylmethane Derivatives from Pd-Catalyzed Cross-Coupling Reactions of Benzylic Halides with Arylboronic Acids. Tetrahedron Lett. 2004, 45, 8225–8228. [Google Scholar] [CrossRef]
- Perusquía-Hernández, C.; Lara-Issasi, G.R.; Frontana-Uribe, B.A.; Cuevas-Yañez, E. Synthesis and Esterification Reactions of Aryl Diazomethanes Derived from Hydrazone Oxidations Catalyzed by TEMPO. Tetrahedron Lett. 2013, 54, 3302–3305. [Google Scholar] [CrossRef]
- Tran, V.H.; La, M.T.; Kim, H.K. Iron(III)-Catalyzed Direct Synthesis of Diphenylmethyl Esters from 2-Diphenylmethoxypyridine. Synthetic Commun. 2019, 49, 2379–2387. [Google Scholar] [CrossRef]
- La, M.T.; Kim, H.K. Facile Synthesis of Diphenylmethyl Esters from 2-Diphenylmethoxypyridine Using Catalytic Boron Trifluoridediethyl Etherate. Tetrahedron Lett. 2018, 59, 1855–1859. [Google Scholar] [CrossRef]
- Noto, R.; Buscemi, S.; Consiglio, G.; Spinelli, D. Linear Free Energy ortho-Correlations in the Thiophene Series. Part IX. Kinetics of Esterification with Diazodiphenylmethane of Some 3-, 4-, and 5-Substituted Thiophene-2-Carboxylic Acids in Methanol. J. Heterocyclic Chem. 1981, 18, 735–738. [Google Scholar] [CrossRef]
- Adhikari, A.A.; Shah, J.P.; Howard, K.T.; Russo, C.M.; Wallach, D.R.; Linaburg, M.R.; Chisholm, J.D. Convenient Formation of Diphenylmethyl Esters Using Diphenylmethyl Trichloroacetimidate. Synlett 2014, 25, 283–287. [Google Scholar]
- Mahajani, N.S.; Meador, R.I.L.; Smith, T.J.; Canarelli, S.E.; Adhikari, A.A.; Shah, J.P.; Russo, C.M.; Wallach, D.R.; Howard, K.T.; Millimaci, A.M.; et al. Ester Formation via Symbiotic Activation Utilizing Trichloroacetimidate Electrophiles. J. Org. Chem. 2019, 84, 7871–7882. [Google Scholar] [CrossRef]
- Richter, S.C.; Oestreich, M. Bioinspired Metal-Free Formal Decarbonylation of α-Branched Aliphatic Aldehydes at Ambient Temperature. Chem. Eur. J. 2019, 25, 8508–8512. [Google Scholar] [CrossRef]
- Peterson, P.E.; Stepanian, M. Hydroboration of Vinyl Ethers with Diisopinocampheylborane. J. Org. Chem. 1988, 53, 1903–1907. [Google Scholar] [CrossRef]
- Ju, T.; Fu, Q.; Ye, J.H.; Zhang, Z.; Liao, L.L.; Yan, S.S.; Tian, X.Y.; Luo, S.P.; Li, J.; Yu, D.G. Selective and Catalytic Hydrocarboxylation of Enamides and Imines with CO2 to Generate α, α-Disubstituted α-Amino Acids. Angew. Chem. Int. Ed. 2018, 57, 13897–13901. [Google Scholar] [CrossRef]
- Xu, Q.; Xie, H.M.; Zhang, E.L.; Ma, X.T.; Chen, J.H.; Yu, X.C.; Li, H. Selective Catalytic Hofmann N-alkylation of Poor Nucleophilic Amines and Amides with Catalytic Amounts of Alkyl Halides. Green Chem. 2016, 18, 3940–3944. [Google Scholar] [CrossRef]
- Yang, Z.Y.; Ni, Y.X.; Liu, R.; Song, K.X.; Lin, S.H.; Pan, Q.M. In Situ Generated Cationic Pd(II)/Bipyridine-Catalyzed Addition of Arylboronic Acids to N-Sulfonyl-Arylaldimines. Tetrahedron Lett. 2017, 58, 2034–2037. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).