Effect of Systemic Subnormal Deuterium Level on Metabolic Syndrome Related and other Blood Parameters in Humans: A Preliminary Study
Abstract
1. Introduction
2. Results
2.1. DDW Consumption Resulted in Reduced Serum D-concentration
2.2. Change of Body Mass During the Study
2.3. Changes in Blood Count and Blood Chemistry Parameters
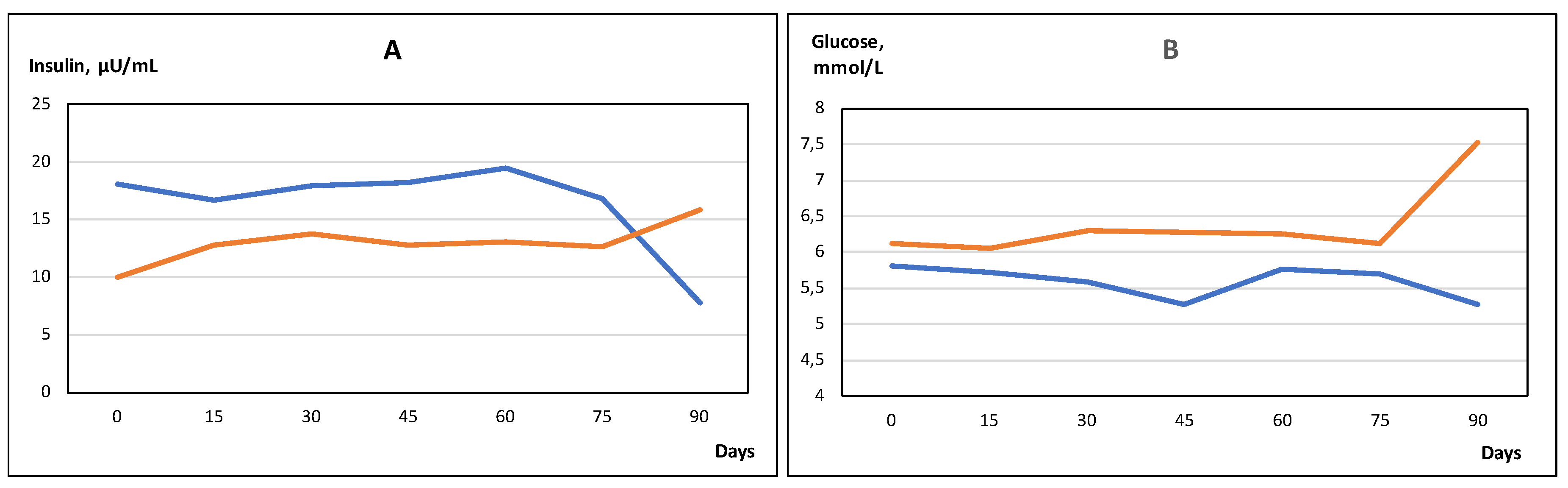
2.4. Effects of DDW Treatment on Fasting and IVGTT-Related Insulin and Glucose Levels, and other Metabolic Parameters
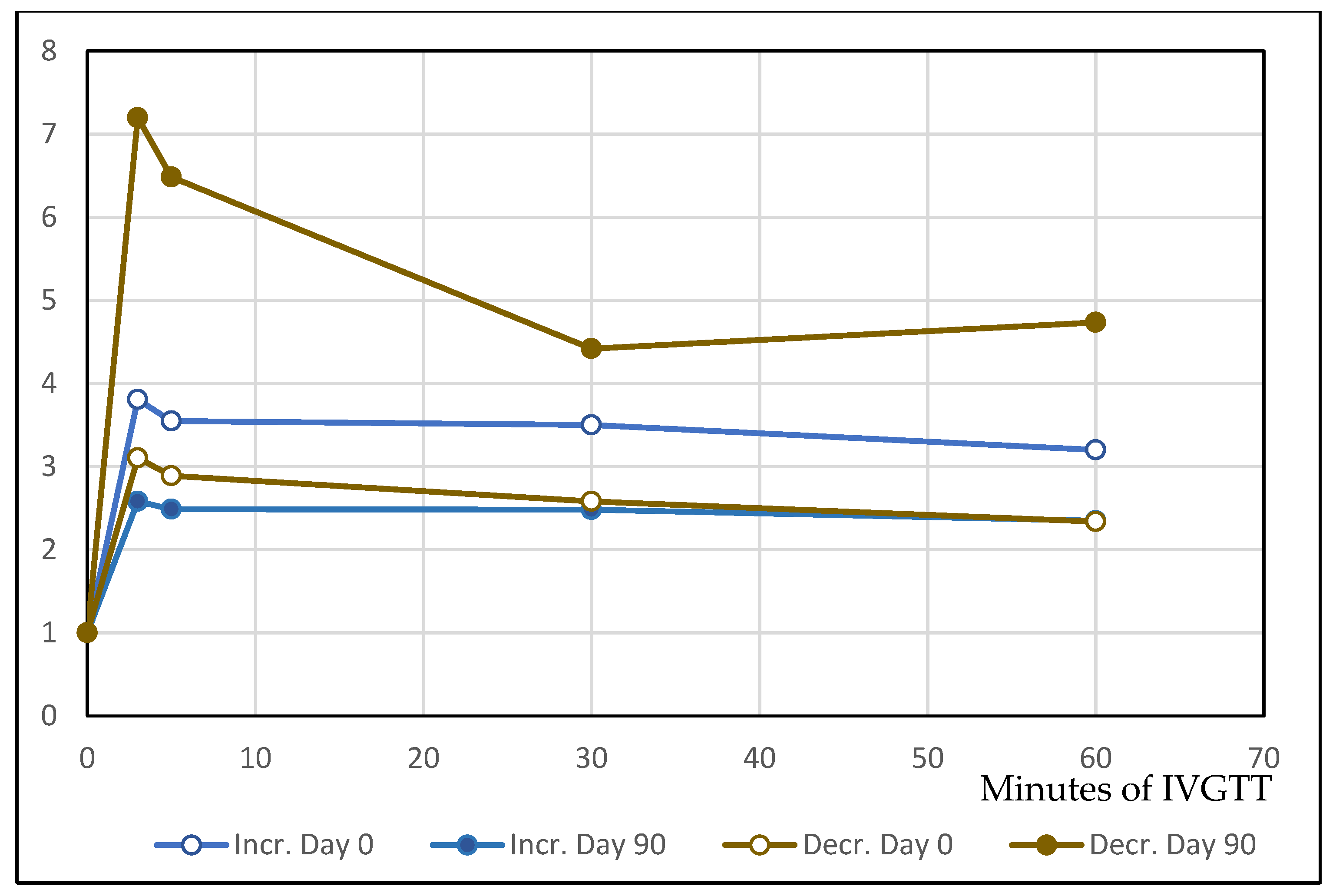
2.5. Effect of DDW Treatment on Peripheral Glucose Disposal
3. Discussion
4. Materials and Methods
4.1. DDW Production
4.2. Patient Selection, Characterization of the Patients Selected
4.3. Determination of Serum D-concentration
4.4. Treatment and Investigations of the Subjects
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Somlyai, G.; Jancsó, G.; Jákli, G.; Vass, K.; Barna, B.; Lakics, V.; Gaál, T. Naturally occurring deuterium is essential for the normal growth rate of cells. FEBS Lett. 1993, 317, 1–4. [Google Scholar] [CrossRef]
- Somlyai, G.; Laskay, G.; Berkényi, T.; Jákly, G.; Jancsó, G. Naturally occuring deuterium may have a central role in cell signalling. In Synthesis and Application of Isotopically Labelled Compounds; Heys, J.R., Mellino, D.G., Eds.; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 1998. [Google Scholar]
- Somlyai, G.; Laskay, G.; Berkényi, T.; Galbács, Z.; Kiss, G.A.; Jákli, G.; Jancsó, G. The biological effects of deuterium-depleted water, a possible new tool in cancer therapy. J. Oncol. 1998, 30, 91–94. [Google Scholar]
- Kovács, A.; Guller, I.; Krempels, K.; Somlyai, I.; Jánosi, I.; Gyöngyi, Z.; Szabó, I.; Ember, I.; Somlyai, G. Deuterium Depletion May Delay the Progression of Prostate Cancer. J. Cancer Ther. 2011, 2, 548–556. [Google Scholar] [CrossRef]
- Gyöngyi, Z.; Somlyai, G. Deuterium depletion can decrease the expression of c-myc, Ha-ras and p53 gene in carcinogen-treated mice. In Vivo 2000, 14, 437–440. [Google Scholar] [PubMed]
- Gyöngyi, Z.; Budán, F.; Szabó, I.; Ember, I.; Kiss, I.; Krempels, K.; Somlyai, I.; Somlyai, G. Deuterium Depleted Water Effects on Survival of Lung Cancer Patients and Expression of Kras, Bcl2, and Myc Genes in Mouse Lung. Nutr. Cancer 2013, 65, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Siniak, I.; Turusov, V.S.; Grigor’ev, A.I.; Zaridze, D.G.; Gaidadymov, V.B.; Gus’kova, E.I.; Antoshina, E.E.; Gor’kova, T.G.; Trukhanova, L.S. Consideration of the deuterium-free water supply to an expedition to Mars. Aviakosm. Ekolog. Ned. 2003, 37, 60–63. [Google Scholar]
- Cong, F.S.; Zhang, Y.R.; Sheng, H.C.; Ao, Z.H.; Zhang, S.Y. Deuterium-depleted water inhibits human lung carcinoma cell growth by apoptosis. Exp. Ther. Med. 2010, 1, 277–283. [Google Scholar] [CrossRef]
- Somlyai, G.; Javaheri, B.; Davari, H.; Gyöngyi, Z.; Somlyai, I.; Tamaddon, K.A.; Boros, L.G. Pre-Clinical and Clinical Data Confirm the Anticancer Effect of Deuterium Depletion. Biomacromol. J. 2016, 2, 1–7. [Google Scholar] [CrossRef]
- Krempels, K.; Somlyai, I.; Somlyai, G. A retrospective evaluation of the effects of deuterium depleted water consumption on four patients with brain metastases from lung cancer. Integr. Cancer Ther. 2008, 7, 172–181. [Google Scholar] [CrossRef]
- Strekalova, T.; Evans, M.; Chernopiatko, A.; Couch, Y.; Costa-Nunes, J.; Cespuglio, R.; Chesson, L.; Vignisse, J.; Steinbusch, H.W.; Anthony, D.C.; et al. Deuterium content of water increases depression susceptibility: The potential role of a serotonin-related mechanism. Behav. Brain Res. 2015, 277, 237–244. [Google Scholar] [CrossRef]
- Mladin, C.; Ciobica, A.; Lefter, R.; Popescu, A.; Bild, W. Deuterium Depleted Water has stimulating effects on long-term memory in rats. Neurosci. Lett. 2014, 583, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Ávila, D.S.; Somlyai, G.; Somlyai, I.; Aschner, M. Anti-aging effects of deuterium depletion on Mn-induced toxicity in a C.elegans model. Toxicol. Lett. 2012, 211, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Molnár, M.; Horváth, K.; Dankó, T.; Somlyai, G. Effect of deuterium oxide (D2O) content of drinking water on glucose metabolism on STZ-induced diabetic rats. In Proceedings of the 7th International Conference Functional Foods in the Prevention and Management of Metabolic Syndrome, North Charleston, SC, USA, 4–5 December 2010; pp. 154–155. [Google Scholar]
- Huang, P.L. A comprehensive definition for metabolic syndrome. Dis. Model. Mech. 2009, 2, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2005, 365, 1415–1428. [Google Scholar] [CrossRef]
- Jancsó, G. Isotope Effects. In Handbook of Nuclear Chemistry; Vértes, A., Nagy, S., Klencsár, Z., Eds.; Kluwer Academic: Dordrecht, The Netherlands, 2003; Volume 2, pp. 85–116. [Google Scholar]
- Collins, C.J.A.; Bowman, N.S. Isotope Effects in Chemical Reactions; Van Nostrand Reinhold: New York, NY, USA, 1971. [Google Scholar]
- Katz, J.J.; Crespi, H.L. Isotope Effects in Biological Systems. In Isotope Effects in Chemical Reactions; Collins, C.J., Bowman, N.S., Eds.; Van Nostrand Reinhold: New York, NY, USA, 1971; pp. 286–363. [Google Scholar]
- Rundel, P.W.; Ehleringer, J.R.; Nagy, K.A. Stable Isotopes in Ecological Research; Springer: New York, NY, USA, 1988. [Google Scholar]
- Kotyk, A.; Dvorakova, M.; Koryta, J. Deuterons cannot replace protons in active transport processes in yeast. Febs Lett. 1990, 264, 203–205. [Google Scholar] [CrossRef]
- Laskay, G.; Somlyai, G.; Jancsó, G. Reduced deuterium concentration of water stimulates O2-uptake and electrogenic H+-efflux in the aquatic macrophyte Elodea Canadensis. Jpn. J. Deuterium Sci. 2001, 10, 17–23. [Google Scholar]
- Boros, L.G.; D’Agostino, D.P.; Katz, H.E.; Roth, E.J.P.; Meuillet, J.; Somlyai, G. Submolecular regulation of cell transformation by deuterium depleting water exchange reactions in the tricarboxylic acid substrate cycle. Med Hypotheses 2016, 87, 69–74. [Google Scholar] [CrossRef]
- DeFronzo, R.A. The Effect of Insulin on Renal Sodium Metabolism. A Review with Clinical Implications. Diabetologia 1981, 21, 165–171. [Google Scholar] [CrossRef]
- Lei, C.Y.; Shen, C.H.; Lu, Z.M. Effects of deuterium depleted water and liquor on lipid metabolism in rats models of hyperlipidemia. Chin. J. Arterioscler. 2011, 19, 819–824. [Google Scholar]
- Basov, A.; Fedulova, L.; Baryshev, M.; Dzhimak, S. Deuterium-Depleted Water Influence on the Isotope 2H/1H Regulation in Body and Individual Adaptation. Nutrients 2019, 11, 1903. [Google Scholar] [CrossRef]
- Boros, L.G.; Collins, T.Q.; Somlyai, G. What to eat or what not to eat—that is still the question. Neuro Oncol. 2017, 19, 595–596. [Google Scholar] [CrossRef] [PubMed]
- Boros, L.G.; Somlyai, G. Compartmentalized NADPH Synthesis, Intramolecular Deuterium. Disequilibrium and Water Pools of Mammalian Cells. Mol. Cell 2014, 55, 253–263. [Google Scholar]
- Schoeller, D.A.; van Santen, E.; Peterson, D.W.; Dietz, W.; Jaspan, J.; Klein, P.D. Total body water measurement in humans with 18O and 2H labeled water. Am. J. Clin. Nutr. 1980, 33, 2686–2693. [Google Scholar] [CrossRef] [PubMed]
- Coleman, M.L.; Shepherd, T.J.; Durham, J.J.; Rouse, J.E.; Moore, G.R. Reduction of water with zinc for hydrogen isotope analysis. Anal. Chem. 1982, 54, 993–995. [Google Scholar] [CrossRef]
- Czuppon, G.; Demény, A.; Leél-Őssy, S.; Óvári, M.; Molnár, M.; Stieber, J.; Kiss, K.; Kármán, K.; Surányi, G.; Haszpra, L. Cave monitoring in the Béke and Baradla Caves (Northeastern Hungary): implications for the conditions for the formation cave carbonates. Int. J. Speleol. 2018, 47, 13–28. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds (DDW) are available from HYD LLC for Cancer Research and Drug Development. |
| Parameter | Day 0 | Day 90 | p Value | Normal Range |
|---|---|---|---|---|
| WBC (109/L) | 6.48 ± 1.36 | 6.96 ± 1.33 | 0.01 | 4.3–10.0 |
| RBC (1012/L) | 4.52 ± 0.37 | 4.66 ± 0.36 | 0.0064 | 4.0–5.9 |
| Hgb (mmol/L) | 8.36 ± 0.71 | 8.61 ± 0.83 | 0.011 | 7.5–11.2 |
| PLT (109/L) | 251 ± 60.6 | 269.9 ± 68.6 | 0.007 | 150–390 |
| Parameter | Day 0 | Day 90 | p Value |
|---|---|---|---|
| triglycerides | 2.07 ± 1.16 | 2.071 ± 1.30 | 0.4937 |
| total cholesterol | 5.27 ± 1.13 | 5.00 ± 1.25 | 0.451 |
| LDL cholesterol | 2.98 ± 0.81 | 3.24 ± 0.96 | 0.108 |
| HDL cholesterol | 1.05 ± 0.45 | 1.34 ± 0.40 | 0.0008 |
| HDL cholesterol, increased (n = 22) | 0.90 ± 0.36 | 1.38 ± 0.39 | 0.0000 |
| HDL cholesterol, decreased (n = 8) | 1.44 ± 0.43 | 1.22 ± 0.41 | 0.005 |
| HDL cholesterol, men (n = 9) | 1.11 ± 0.46 | 1.41 ± 0.43 | 0.006 |
| HDL cholesterol, women (n = 21) | 0.89 ± 0.39 | 1.16 ± 0.24 | 0.032 |
| Ions in the Serum | Day 0 | Day 90 | p Value |
|---|---|---|---|
| Na+ | 141.1 ± 2.42 | 139.0 ± 2.01 | 0.000024 |
| K+ | 4.3 ± 0.31 | 4.3 ± 0.40 | 0.134 |
| Ca2+ | 2.3 ± 0.09 | 2.3 ± 0.14 | 0.249 |
| Insulin Decreased | Sign. Decr vs. Incr | Insulin Increased | |
|---|---|---|---|
| Insulin, day 0 | 18.05 ± 13.38 | p = 0.011 | 9.96 ± 3.43 |
| Insulin, day 90 | 7.75 ± 4.39 | p = 0.002 | 15.89 ± 10.94 |
| Sign. day 90 vs. day 0 | p = 0.007 | p = 0.056 | |
| Glucose, day 0 | 5.58 ± 0.80 | p = 0.319 | 5.83 ± 0.56 |
| Glucose, day 90 | 5.24 ± 0.48 | p = 0.005 | 6.21 ± 1.03 |
| Sign. day 90 vs. day 0 | p = 0.005 | p = 0.01 | |
| Insulin/glucose change correlation | p = 0.008 | p = 0.988 |
| Insulin Decreased | Sign. Decr vs. Incr | Insulin Increased | |
|---|---|---|---|
| HDL, day 0 | 1.14 ± 0.5 | p = 0.006 | 0.95 ± 0.38 |
| HDL, day 90 | 1.33 ± 0.44 | p= 0.58 | 1.35 ± 0.37 |
| sign. day 90. vs. day 0 | p = 0.24 | p = 0.000038 |
| Whole-Body M Increased (n = 11) | Sign. Incr vs. Decr | Whole-Body M Decreased (n = 18) | |
|---|---|---|---|
| M, day 0 (mg/kg b.w./min) | 6.9 ± 2.4 | p = 0.172 | 8.4 ± 2.5 |
| M, day 90 | 8.6 ± 2.5 | p = 0.034 | 6.5 ± 2.2 |
| sign. day 90 vs.day 0 | p = 0.0014 | p = 0.00004 | |
| Serum Na, day 0 (mmol/L) | 139.95 ± 2.14 | p = 0.066 | 141.4 ± 2.03 |
| Serum Na, day 90 | 139.09 ± 2.3 | p = 0.915 | 138.9 ± 1.8 |
| sign. day 90 vs. day 0 | p = 0.23 | p = 0.00003 | |
| HDL, day 0 (mmol/L) | 1.20 ± 0.45 | p = 0.147 | 0.95 ± 0.43 |
| HDL, day 90 | 1.17 ± 0.35 | p = 0.095 | 1.43 ± 0.40 |
| sign. day 90 vs. day 0 | p = 0.72 | p = 0.0002 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Somlyai, G.; Somlyai, I.; Fórizs, I.; Czuppon, G.; Papp, A.; Molnár, M. Effect of Systemic Subnormal Deuterium Level on Metabolic Syndrome Related and other Blood Parameters in Humans: A Preliminary Study. Molecules 2020, 25, 1376. https://doi.org/10.3390/molecules25061376
Somlyai G, Somlyai I, Fórizs I, Czuppon G, Papp A, Molnár M. Effect of Systemic Subnormal Deuterium Level on Metabolic Syndrome Related and other Blood Parameters in Humans: A Preliminary Study. Molecules. 2020; 25(6):1376. https://doi.org/10.3390/molecules25061376
Chicago/Turabian StyleSomlyai, Gábor, Ildikó Somlyai, István Fórizs, György Czuppon, András Papp, and Miklós Molnár. 2020. "Effect of Systemic Subnormal Deuterium Level on Metabolic Syndrome Related and other Blood Parameters in Humans: A Preliminary Study" Molecules 25, no. 6: 1376. https://doi.org/10.3390/molecules25061376
APA StyleSomlyai, G., Somlyai, I., Fórizs, I., Czuppon, G., Papp, A., & Molnár, M. (2020). Effect of Systemic Subnormal Deuterium Level on Metabolic Syndrome Related and other Blood Parameters in Humans: A Preliminary Study. Molecules, 25(6), 1376. https://doi.org/10.3390/molecules25061376





