Vitamin C Enhances the Antibacterial Activity of Honey against Planktonic and Biofilm-Embedded Bacteria
Abstract
1. Introduction
2. Results
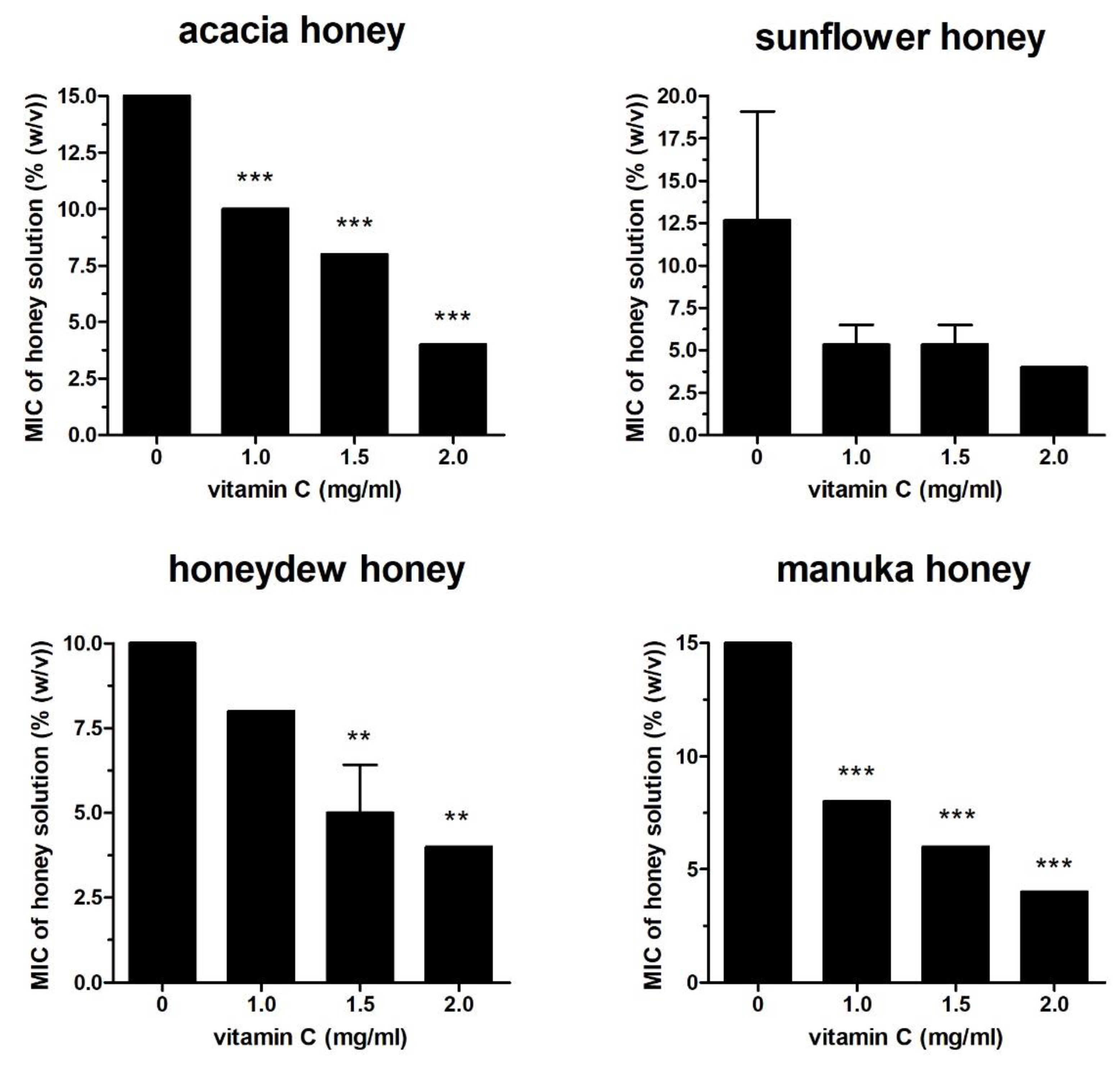
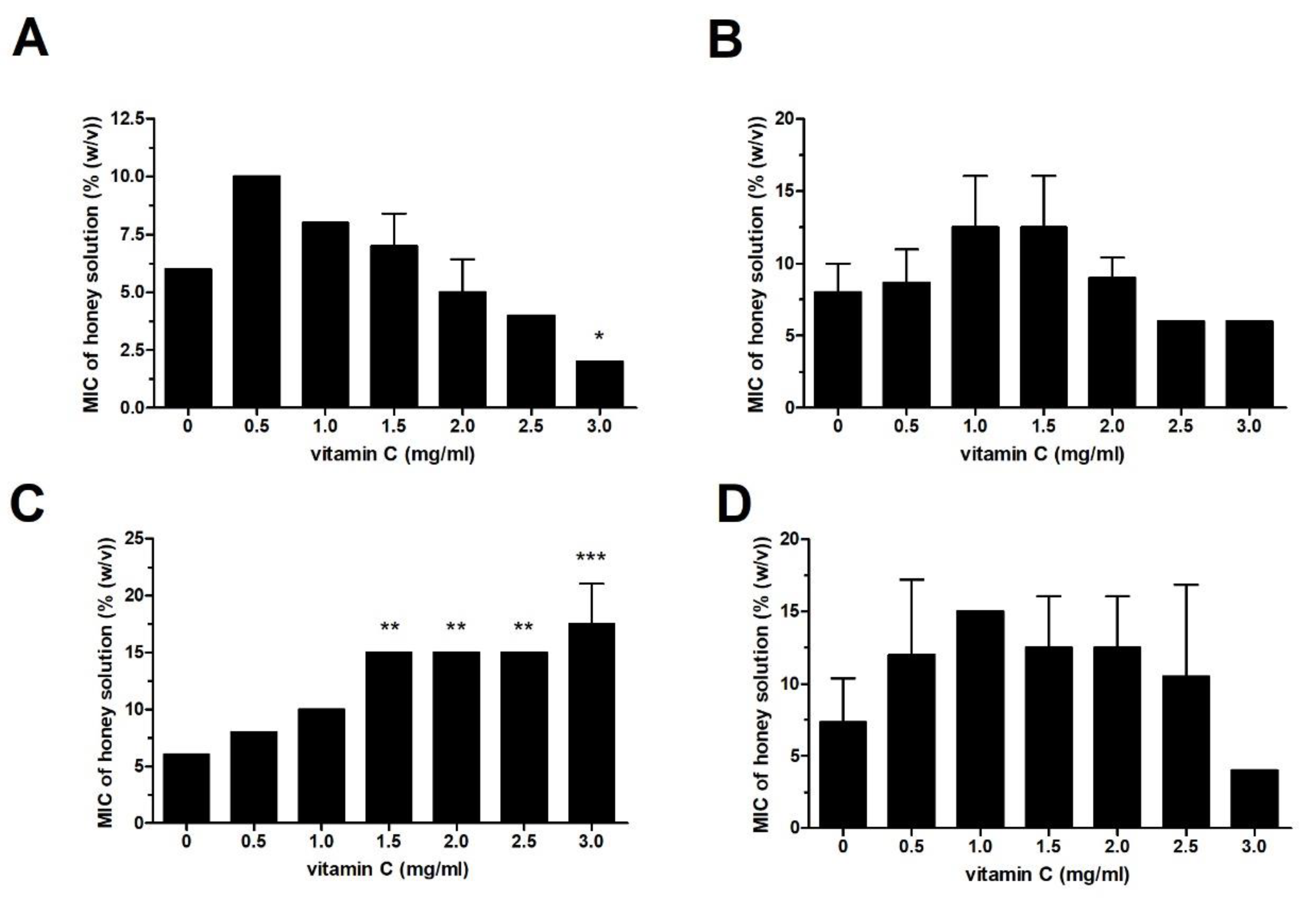
2.1. Effect of Vit C on Honey Antibacterial Activity Against Different Bacterial Isolates
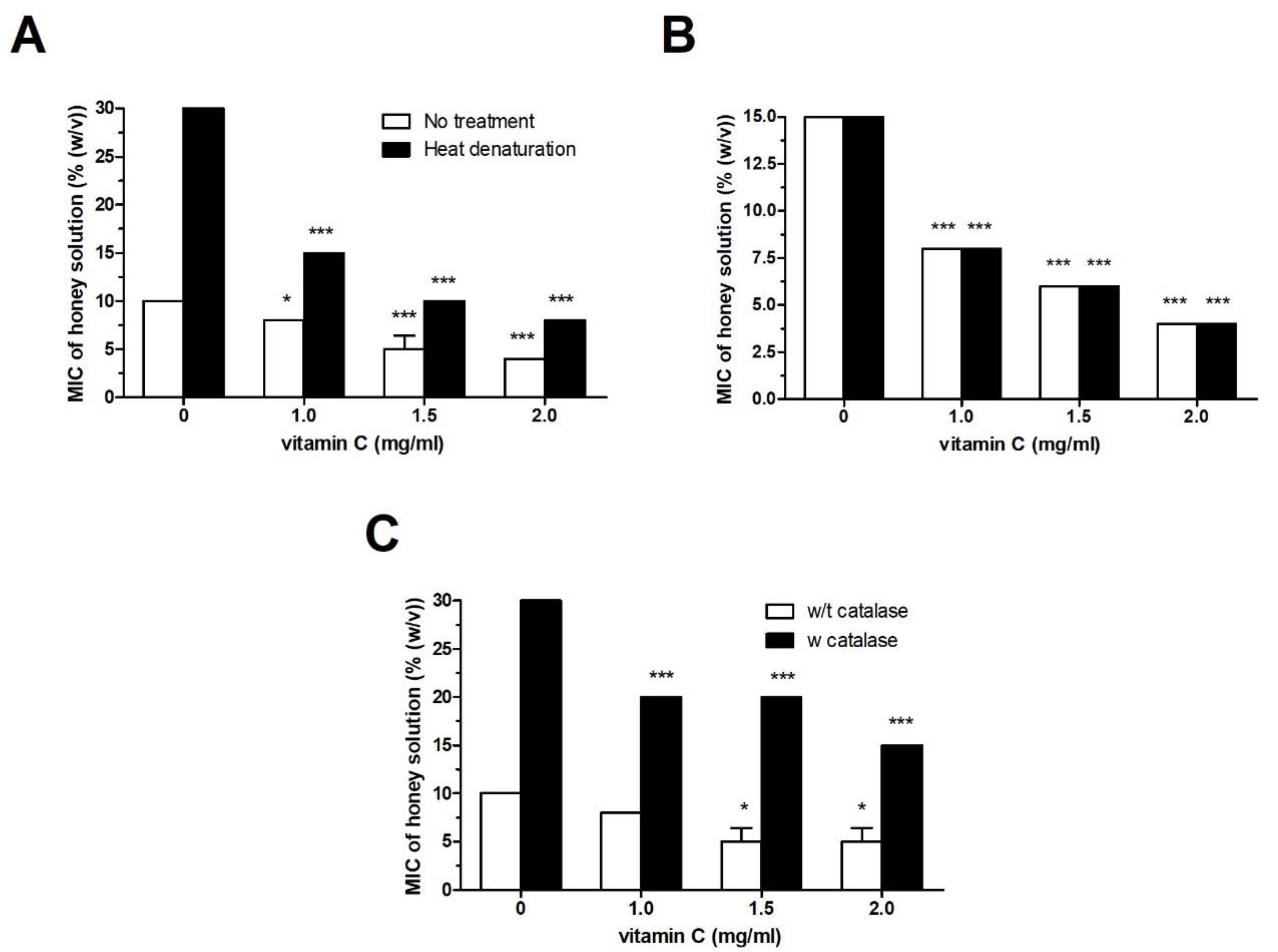
2.2. Effect of Vit C on the Antibacterial Activity of Heat- and Catalase-Treated Honey Samples
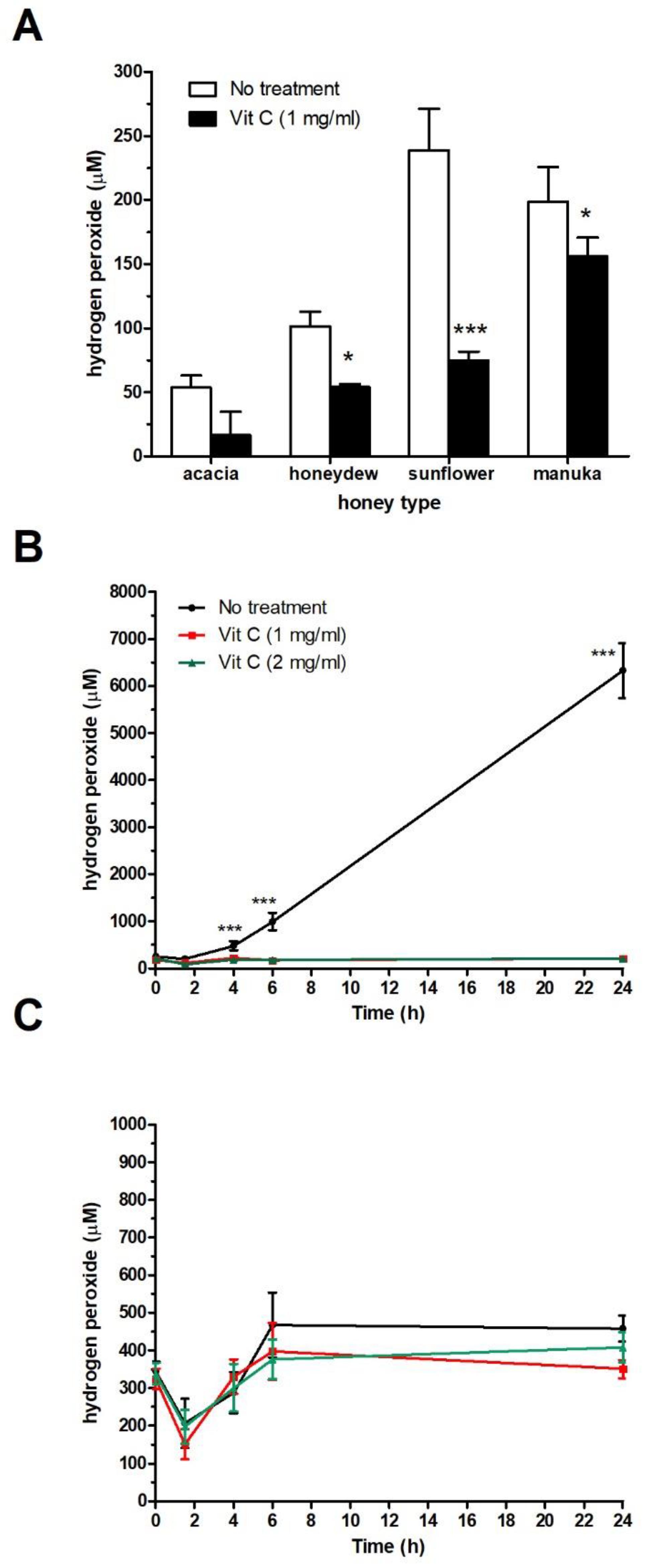
2.3. Effect of Vit C on H2O2 Production in Honey Samples
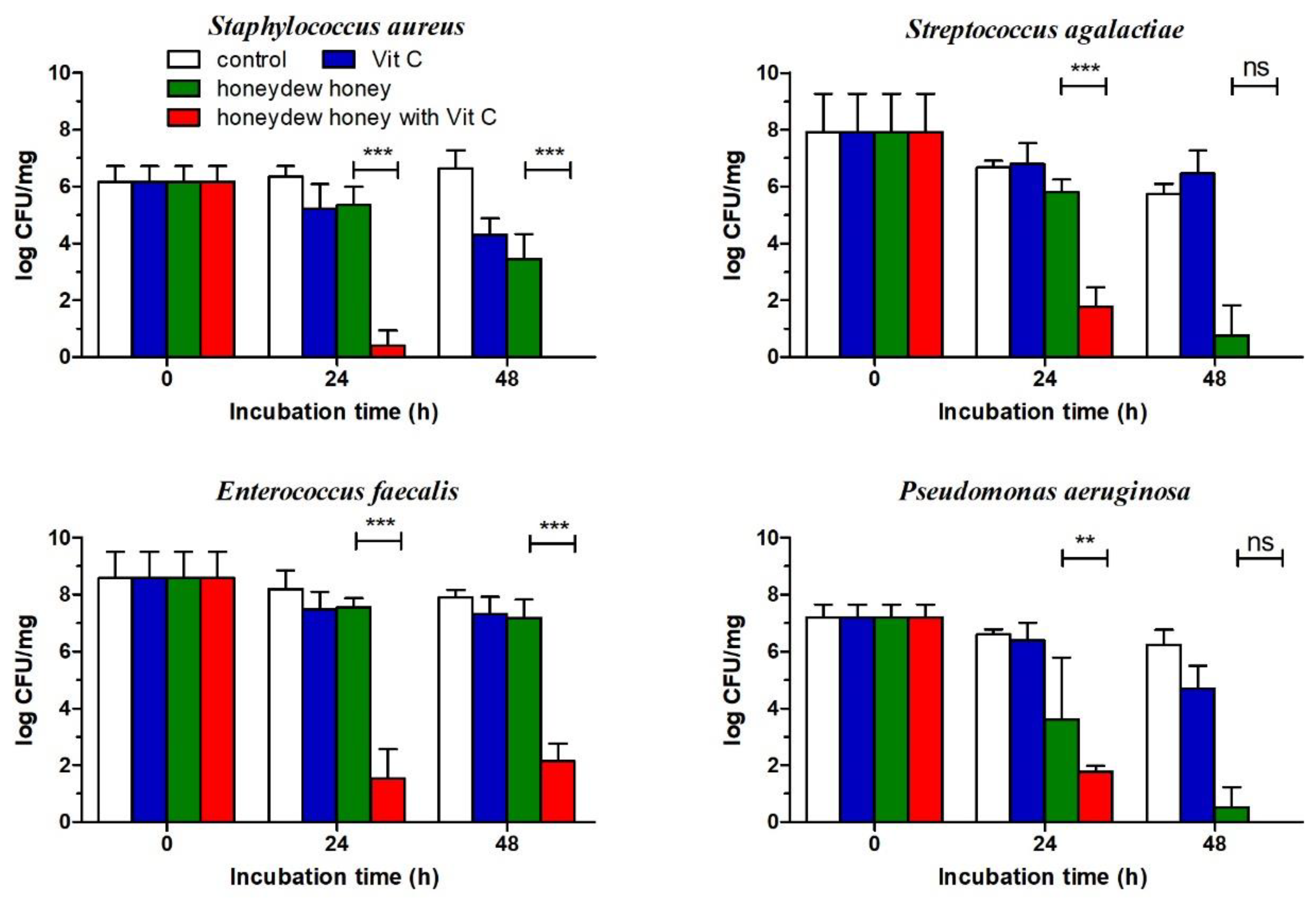
2.4. Effect of Honeydew Honey Supplemented with Vit C Against Bacterial Wound Pathogens Grown in a Multi-Species Biofilm
3. Discussion
4. Materials and Methods
4.1. Honey Samples
4.2. Bacterial Strains for Antibacterial and Antibiofilm Activity Testing
4.3. Determination of the Antibacterial Activities of Honey, Vitamin C (Vit C), and a Mixture of Honey and Vit C
4.4. Antibiofilm Activity Analysis Using a Polybacterial Biofilm
4.5. Determination of Hydrogen Peroxide H2O2 Concentration
4.6. Heat and Catalase Treatment of Honey
4.7. Statistical Analysis
5. Conclusions
6. Patents
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O’Meara, S.; Al-Kurdi, D.; Ologun, Y.; Ovington, L.G.; Martyn-St James, M.; Richardson, R. Antibiotics and antiseptics for venous leg ulcers. Cochrane Databse Syst. Rev. 2014, 2014, CD003557. [Google Scholar] [CrossRef] [PubMed]
- Jull, A.B.; Cullum, N.; Dumville, J.C.; Westby, M.J.; Deshpande, S.; Walker, N. Honey as a topical treatment for wounds. Cochrane Databse Syst. Rev. 2015, 3, CD005083. [Google Scholar] [CrossRef] [PubMed]
- Norman, G.; Dumville, J.C.; Mohapatra, D.P.; Owens, G.L.; Crosbie, E.J. Antibiotics and antiseptics for surgical wounds healing by secondary intention. Cochrane Databse Syst. Rev. 2016, 3, CD011712. [Google Scholar]
- Norman, G.; Dumville, J.C.; Moore, Z.E.; Tanner, J.; Christie, J.; Goto, S. Antibiotics and antiseptics for pressure ulcers. Cochrane Databse Syst. Rev. 2016, 4, CD011586. [Google Scholar] [CrossRef]
- Norman, G.; Christie, J.; Liu, Z.; Westby, M.J.; Jefferies, J.M.; Hudson, T.; Edwards, J.; Mohapatra, D.P.; Hassan, I.A.; Dumville, J.C. Antiseptics for burns. Cochrane Databse Syst. Rev. 2017, 7, CD011821. [Google Scholar] [CrossRef]
- Tian, X.; Yi, L.J.; Ma, L.; Zhang, L.; Song, G.M.; Wang, Y. Effects of honey dressing for the treatment of DFUs: A systematic review. Int. J. Nurs. Sci. 2014, 1, 224–231. [Google Scholar] [CrossRef]
- Lindberg, T.; Andersson, O.; Palm, M.; Fagerström, C. A systematic review and meta-analysis of dressings used for wound healing: The efficiency of honey compared to silver on burns. Contemp. Nurse 2015, 51, 121–123. [Google Scholar] [CrossRef]
- Wang, C.; Guo, M.; Zhang, N.; Wang, G. Effectiveness of honey dressing in the treatment of diabetic foot ulcers: A systematic review and meta-analysis. Complement. Ther. Clin. Pract. 2019, 34, 123–131. [Google Scholar] [CrossRef]
- Mavric, E.; Wittmann, S.; Barth, G.; Henle, T. Identification and quantification of methylglyoxal as the dominant antibacterial constituent of Manuka (Leptospermum scoparium) honeys from New Zealand. Mol. Nutr. Food Res. 2008, 52, 483–489. [Google Scholar] [CrossRef]
- Adams, C.J.; Boult, C.H.; Deadman, B.J.; Farr, J.M.; Grainger, M.N.C.; Manley-Harris, M.; Snow, M.J. Isolation by HPLC and characterisation of the bioactive fraction of New Zealand manuka (Leptospermum scoparium) honey. Carbohydr. Res. 2009, 343, 651–659. [Google Scholar] [CrossRef]
- Holt, S.; Johnson, K.; Ryan, J.; Catchpole, O.; Zhang, S.; Mitchell, K.A. New Zealand kanuka honey has high levels of methylglyoxal and antimicrobial activity. J. Altern. Complement. Med. 2012, 18, 203–204. [Google Scholar] [CrossRef] [PubMed]
- Bucekova, M.; Buriova, M.; Pekarik, L.; Majtan, V.; Majtan, J. Phytochemicals-mediated production of hydrogen peroxide is crucial for high antibacterial activity of honeydew honey. Sci. Rep. 2018, 8, 9061. [Google Scholar] [CrossRef] [PubMed]
- Bucekova, M.; Jardekova, L.; Juricova, V.; Bugarova, V.; Di Marco, G.; Gismondi, A.; Leonardi, D.; Farkasovska, J.; Godocikova, J.; Laho, M.; et al. Antibacterial activity of different blossom honeys: New findings. Molecules 2019, 24, 1573. [Google Scholar] [CrossRef]
- White, J.W.; Subers, M.H.; Schepartz, A.I. The identification of inhibine, the antibacterial factor in honey, as hydrogenperoxide and its origin in a honey glucose-oxidase system. Biochim. Biophys. Acta 1963, 73, 57–70. [Google Scholar] [CrossRef]
- Kwakman, P.H.; de Boer, L.; Ruyter-Spira, C.P.; Creemers-Molenaar, T.; Helsper, J.P.; Vandenbroucke-Grauls, C.M.; Zaat, S.A.; te Velde, A.A. Medical-grade honey enriched with antimicrobial peptides has enhanced activity against antibiotic-resistant pathogens. Eur. J. Clin. Microbiol. Infect. Dis. 2011, 30, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Swift, S.; Chepulis, L.M.; Uy, B.; Radcliff, F.J. Enhanced antibacterial activity of MGOTM manuka honey complexed with—cyclodextrin (manuka honey with CycloPowerTM). Funct. Food Health Dis. 2014, 4, 172–181. [Google Scholar] [CrossRef]
- Imtara, H.; Elamine, Y.; Lyoussi, B. Honey antibacterial effect boosting using Origanum vulgare L. essential oil. Evid.-Based Complement. Altern. Med. 2018, 2018, 7842583. [Google Scholar] [CrossRef]
- Ciulu, M.; Solinas, S.; Floris, I.; Panzanelli, A.; Pilo, M.I.; Piu, P.C.; Spano, N.; Sanna, G. RP-HPLC determination of water-soluble vitamins in honey. Talanta 2011, 83, 924–929. [Google Scholar] [CrossRef]
- Combarros-Fuertes, P.; Estevinho, L.M.; Dias, L.G.; Castro, J.M.; Tomás-Barberán, F.A.; Tornadijo, M.E.; Fresno-Baro, J.M. Bioactive components and antioxidant and antibacterial activities of different varieties of honey: A screening prior to clinical application. J. Agric. Food Chem. 2019, 67, 688–698. [Google Scholar] [CrossRef]
- Carr, A.; Frei, B. Does vitamin C act as a pro-oxidant under physiological conditions? FASEB J. 1999, 13, 1007–1024. [Google Scholar] [CrossRef]
- Verghese, R.J.; Ramya, S.R.; Kanungo, R. In vitro antibacterial activity of vitamin C and in combination with ciprofloxacin against uropathogenic Escherichia coli. J. Clin. Diagn. Res. 2017, 11, DC01–DC05. [Google Scholar] [CrossRef]
- Verghese, R.J.; Mathew, S.K.; David, A. Antimicrobial activity of vitamin C demonstrated on uropathogenic Escherichia coli and Klebsiella pneumoniae. J. Curr. Res. Sci. Med. 2017, 3, 88–93. [Google Scholar]
- Kallio, J.; Jaakkola, M.; Mäki, M.; Kilpeläinen, P.; Virtanen, V. Vitamin C inhibits Staphylococcus aureus growth and enhances the inhibitory effect of quercetin on growth of Escherichia coli in vitro. Planta Med. 2012, 78, 1824–1830. [Google Scholar] [CrossRef] [PubMed]
- Kwiecińska-Piróg, J.; Skowron, K.; Bogiel, T.; Białucha, A.; Przekwas, J.; Gospodarek-Komkowska, E. Vitamin C in the presence of sub-Inhibitory concentration of aminoglycosides and fluoroquinolones alters Proteus mirabilis biofilm inhibitory rate. Antibiotics 2019, 8, 116. [Google Scholar] [CrossRef] [PubMed]
- Khameneh, B.; Bazzaz, B.S.F.; Amani, A.; Rostami, J.; Vahdati-Mashhadian, N. Combination of anti-tuberculosis drugs with vitamin C or NAC against different Staphylococcus aureus and Mycobacterium tuberculosis strains. Microb. Pathog. 2016, 93, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.; Ribeiro, H.G.; Silva, A.C.; Silva, M.D.; Sousa, J.C.; Rodrigues, C.F.; Melo, L.D.R.; Henriques, A.F.; Sillankorva, S. Synergistic antimicrobial interaction between honey and phage against Escherichia coli biofilms. Front. Microbiol. 2017, 8, 2407. [Google Scholar] [CrossRef]
- Oliveira, A.; Sousa, J.C.; Silva, A.C.; Melo, L.D.R.; Sillankorva, S. Chestnut honey and bacteriophage application to control Pseudomonas aeruginosa and Escherichia coli biofilms: Evaluation in an ex vivo wound model. Front. Microbiol. 2018, 9, 1725. [Google Scholar] [CrossRef]
- García-Solache, M.; Rice, L.B. The Enterococcus: A model of adaptability to its environment. Clin. Microbiol. Rev. 2019, 32, e00058-18. [Google Scholar] [CrossRef]
- Sojka, M.; Valachova, I.; Bucekova, M.; Majtan, J. Antibiofilm efficacy of honey and bee-derived defensin-1 on multispecies wound biofilm. J. Med. Microbiol. 2016, 65, 337–344. [Google Scholar] [CrossRef]
- Poli, J.P.; Guinoiseau, E.; Luciani, A.; Yang, Y.; Battesti, M.J.; Paolini, J.; Costa, J.; Berti, L.; Lorenzi, V. Investigating the antibacterial action of Corsican honeys on nosocomial and foodborne pathogens. J. Apic. Res. 2018, 57, 186–194. [Google Scholar] [CrossRef]
- Fidaleo, M.; Lavecchia, R.; Zuorro, A. Antibacterial and anti-quorum sensing activities of selected Italian honeys against antibiotic-resistant pathogens. Online J. Biol. Sci. 2015, 15, 236–243. [Google Scholar] [CrossRef]
- Shlezinger, M.; Coppenhagen-Glazer, S.; Gelman, D.; Beyth, N.; Hazan, R. Eradication of vancomycin-resistant enterococci by combining phage and vancomycin. Viruses 2019, 11, 954. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, T.; Srivastava, S.K.; Gaurav, A.; Kumar, A.; Kumar, P.; Yadav, A.S.; Pathania, R.; Navani, N.K. A combination of linalool, vitamin C, and copper synergistically triggers reactive oxygen species and DNA damage and inhibits Salmonella enterica subsp. enterica Serovar Typhi and Vibrio fluvialis. Appl. Environ. Microbiol. 2019, 85, e02487-18. [Google Scholar] [CrossRef] [PubMed]
- Schencke, C.; Vasconcellos, A.; Salvo, J.; Veuthey, C.; Del Sol, M. Healing effect of ulmo honey (Eucryphia cordifolia) supplemented with ascorbic acid as treatment in burns. Int. J. Morphol. 2015, 33, 137–143. [Google Scholar] [CrossRef][Green Version]
- Atashbeyk, D.G.; Khameneh, B.; Tafaghodi, M.; Fazly Bazzaz, B.S. Eradication of methicillin-resistant Staphylococcus aureus infection by nanoliposomes loaded with gentamicin and oleic acid. Pharm. Biol. 2014, 52, 1423–1428. [Google Scholar] [CrossRef]
- Kucera, J.; Sojka, M.; Pavlik, V.; Szuszkiewicz, K.; Velebny, V.; Klein, P. Multispecies biofilm in an artificial wound bed--A novel model for in vitro assessment of solid antimicrobial dressings. J. Microbiol. Methods 2014, 103, 18–24. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Majtan, J.; Sojka, M.; Palenikova, H.; Bucekova, M.; Majtan, V. Vitamin C Enhances the Antibacterial Activity of Honey against Planktonic and Biofilm-Embedded Bacteria. Molecules 2020, 25, 992. https://doi.org/10.3390/molecules25040992
Majtan J, Sojka M, Palenikova H, Bucekova M, Majtan V. Vitamin C Enhances the Antibacterial Activity of Honey against Planktonic and Biofilm-Embedded Bacteria. Molecules. 2020; 25(4):992. https://doi.org/10.3390/molecules25040992
Chicago/Turabian StyleMajtan, Juraj, Martin Sojka, Helena Palenikova, Marcela Bucekova, and Viktor Majtan. 2020. "Vitamin C Enhances the Antibacterial Activity of Honey against Planktonic and Biofilm-Embedded Bacteria" Molecules 25, no. 4: 992. https://doi.org/10.3390/molecules25040992
APA StyleMajtan, J., Sojka, M., Palenikova, H., Bucekova, M., & Majtan, V. (2020). Vitamin C Enhances the Antibacterial Activity of Honey against Planktonic and Biofilm-Embedded Bacteria. Molecules, 25(4), 992. https://doi.org/10.3390/molecules25040992







