First Report on Yield and Chemical Composition of Essential Oil Extracted from Myrcia eximia DC (Myrtaceae) from the Brazilian Amazon
Abstract
1. Introduction
2. Results and Discussion
2.1. Yields
2.2. Chemical Composition of the EO
3. Materials and Methods
3.1. Plant Material
3.2. Preparation and Characterization of the Raw Material
3.3. Hydrodistillation
3.4. Steam Distillation
3.5. Analysis of Volatile Compounds
4. Conclusions
Supplementary Files
Supplementary File 1Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carneiro, N.S.; Alves, C.C.F.; Alves, J.M.; Egea, M.B.; Martins, C.H.G.; Silva, T.S.; Bretanha, L.C.; Balleste, M.P.; Micke, G.A.; Silveira, E.V.; et al. Chemical composition, antioxidant and antibacterial activities of essential oils from leaves and flowers of Eugenia klotzschiana Berg (Myrtaceae). An. Acad. Bras. Cienc. 2017, 89, 1907–1915. [Google Scholar] [CrossRef] [PubMed]
- Dluzniewski, F.D.S.; Vettorato, J.G.; Ghellar Müller, N.T. Abordagem etnobotânica de Myrtaceae no município de Sete de Setembro, Rio Grande do Sul, Brasil. Rev. Interdiscip. Em Ciências Da Saúde E Biológicas – Ricsb 2018, 2, 21–31. [Google Scholar] [CrossRef]
- Santos, C.d.; Galaverna, R.S.; Angolini, C.F.F.; Nunes, V.V.A.; de Almeida, L.F.R.; Ruiz, A.L.T.G.; de Carvalho, J.E.; Duarte, R.M.T.; Duarte, M.C.T.; Eberlin, M.N. Antioxidative, antiproliferative and antimicrobial activities of phenolic compounds from three myrcia species. Molecules 2018, 23, 986. [Google Scholar] [CrossRef] [PubMed]
- da Silva, V.P.; Alves, C.C.F.; Miranda, M.L.D.; Bretanha, L.C.; Balleste, M.P.; Micke, G.A.; Silveira, E.V.; Martins, C.H.G.; Ambrosio, M.A.L.V.; de Souza Silva, T.; et al. Chemical composition and in vitro leishmanicidal, antibacterial and cytotoxic activities of essential oils of the Myrtaceae family occurring in the Cerrado biome. Ind. Crops Prod. 2018, 123, 638–645. [Google Scholar] [CrossRef]
- Stadnik, A.; Oliveira, M.I.U.; de Roque, N. Floristic survey of Myrtaceae in Jacobina municipality, Chapada Diamantina, Bahia State, Brazil. Hoehnea 2016, 43, 87–97. [Google Scholar] [CrossRef][Green Version]
- Cascaes, M.M.; Guilhon, G.M.S.P.; de Aguiar Andrade, E.H.; das Graças Bichara Zoghbi, M.; da Silva Santos, L. Constituents and pharmacological activities of Myrcia (Myrtaceae): A review of an aromatic and medicinal group of plants. Int. J. Mol. Sci. 2015, 16, 23881–23904. [Google Scholar] [CrossRef]
- Scalvenzi, L.; Grandini, A.; Spagnoletti, A.; Tacchini, M.; Neill, D.; Ballesteros, J.L.; Sacchetti, G.; Guerrini, A. Myrcia splendens (Sw.) DC. (syn. M. fallax (Rich.) DC.) (myrtaceae) essential oil from amazonian Ecuador: A chemical characterization and bioactivity profile. Molecules 2017, 22, 1163. [Google Scholar] [CrossRef]
- Cândido, C.S.; Portella, C.S.A.; Laranjeira, B.J.; da Silva, S.S.; Arriaga, A.M.C.; Santiago, G.M.P.; Gomes, G.A.; Almeida, P.C.; Carvalho, C.B.M. Effects of Myrcia ovata Cambess. essential oil on planktonic growth of gastrointestinal microorganisms and biofilm formation of Enterococcus faecalis. Brazilian J. Microbiol. 2010, 41, 621–627. [Google Scholar] [CrossRef]
- Andrade, G.S.; Guimarães, A.G.; Santana, M.T.; Siqueira, R.S.; Passos, L.O.; Machado, S.M.F.; Ribeiro, A.D.S.; Sobral, M.; Almeida, J.R.G.S.; Quintans-Júnior, L.J. Phytochemical screening, antinociceptive and anti-inflammatory effects of the essential oil of Myrcia pubiflora in mice. Brazilian J. Pharmacogn. 2011, 22, 181–188. [Google Scholar] [CrossRef]
- Jiménez, D.; Araque, M.; Rojas, L.; Cordero, A.; Briceño, B. Componentes volátiles y actividad antibacteriana del vástago de Myrcia splendens (Sw.) DC. Rev. la Fac. Farm. 2012, 54, 7–11. [Google Scholar]
- Do Silva, A.N.; Uetanabaro, A.P.T.; Lucchese, A.M. Chemical composition and antibacterial activity of essential oils from Myrcia alagoensis (Myrtaceae). Nat. Prod. Commun. 2013, 8, 269–271. [Google Scholar] [CrossRef]
- De Cerqueira, M.D.; Souza-Neta, L.C.; Passos, M.D.G.V.M.; Lima, E.D.O.; Roque, N.F.; Martins, D.; Guedes, M.L.S.; Cruz, F.G. Seasonal variation and antimicrobial activity of Myrcia myrtifolia essential oils. J. Braz. Chem. Soc. 2007, 18, 998–1003. [Google Scholar] [CrossRef]
- Limberger, R.P.; Sobral, M.; Henriques, A.T.; Menut, C.; Bessière, J.M. Óleos voláteis de espécies de Myrcia nativas do Rio Grande do Sul. Quim. Nova 2004, 27, 916–919. [Google Scholar] [CrossRef]
- Amaral, D.D.; Viera, I.C.G.; Salomão, R.P.; de Almeida, S.S.; Jardim, M.A.G. Checklist da Flora Arbórea de Remanescentes Florestais da Região Metropolitana de Belém, Pará, Brasil. Bol. do Mus. Para. Emilio Goeldi Ciências Nat. 2009, 4, 231–289. [Google Scholar]
- Grandtner, M.M.; Chevrette, J. Dictionary of Trees; Academic Press: Cambridge, MA, USA, 2013; Volume 2. [Google Scholar]
- Maia, O.G.S.; Andrade, L.H.A. Database of the amazon aromatic plants and their essential oils. Quim. Nova. 2009, 32, 595–622. [Google Scholar] [CrossRef]
- Naz, S.; Hanif, M.A.; Bhatti, H.N.; Ansari, T.M. Impact of Supercritical Fluid Extraction and Traditional Distillation on the Isolation of Aromatic Compounds from Cannabis indica and Cannabis sativa. J. Essent. Oil Bear. Plants 2017, 20, 175–184. [Google Scholar] [CrossRef]
- Abd El-Gaber, A.S.; El Gendy, A.N.G.; Elkhateeb, A.; Saleh, I.A.; El-Seedi, H.R. Microwave Extraction of Essential Oil from Anastatica hierochuntica (L): Comparison with Conventional Hydro-Distillation and Steam Distillation. J. Essent. Oil Bear. Plants 2018, 21, 1003–1010. [Google Scholar] [CrossRef]
- Khanavi, M.; Hadjiakhoondi, A.; Amin, G.; Amanzadeh, Y.; Rustaiyan, A.; Shafiee, A. Comparison of the Volatile Composition of Stachys persica Gmel. and Stachys byzantina C. Koch. Oils Obtained by Hydrodistillation and Steam Distillation. Zeitschrift für Naturforsch. C 2004, 59, 463–467. [Google Scholar] [CrossRef]
- Conde-Hernández, L.A.; Espinosa-Victoria, J.R.; Trejo, A.; Guerrero-Beltrán, J.Á. CO2 -supercritical extraction, hydrodistillation and steam distillation of essential oil of rosemary ( Rosmarinus officinalis ). J. Food Eng. 2017, 200, 81–86. [Google Scholar] [CrossRef]
- Périno-Issartier, S.; Ginies, C.; Cravotto, G.; Chemat, F. A comparison of essential oils obtained from lavandin via different extraction processes: Ultrasound, microwave, turbohydrodistillation, steam and hydrodistillation. J. Chromatogr. A 2013, 1305, 41–47. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectroscopy, 4th ed.; Allured Publishing Corporation: Carol Stream, IL, USA, 2007. [Google Scholar]
- Mondello, L. FFNSC 2: Flavors and Fragrances of Natural and Synthetic Compounds, Mass Spectral Database, 2nd ed.; Wiley Online Library: Hoboken, NJ, USA, 2011. [Google Scholar]
- Silva, S.G.; Figueiredo, P.L.B.; Nascimento, L.D.; da Costa, W.A.; Maia, J.G.S.; Andrade, E.H.A. Planting and seasonal and circadian evaluation of a thymol-type oil from Lippia thymoides Mart. & Schauer. Chem. Cent. J. 2018, 12, 113. [Google Scholar] [PubMed]
- Ribeiro, A.F.; Andrade, E.H.A.; Salimena, F.R.G.; Maia, J.G.S. Circadian and seasonal study of the cinnamate chemotype from Lippia origanoides Kunth. Biochem. Syst. Ecol. 2014, 55, 249–259. [Google Scholar] [CrossRef]
- Bezerra, F.W.F.; de Oliveira, M.S.; Bezerra, P.N.; Cunha, V.M.B.; Silva, M.P.; da Costa, W.A.; Pinto, R.H.H.; Cordeiro, R.M.; da Cruz, J.N.; Chaves Neto, A.M.J.; et al. Extraction of bioactive compounds. In Green Sustainable Process for Chemical and Environmental Engineering and Science; Academic Press: Cambridge, MA, USA, 2020; pp. 149–167. [Google Scholar]
- Gardini, F.; Lanciotti, R.; Guerzoni, M.E. Effect of trans-2-hexenal on the growth of Aspergillus flavus in relation to its concentration, temperature and water activity. Lett. Appl. Microbiol. 2001, 33, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Trombetta, D.; Saija, A.; Bisignano, G.; Arena, S.; Caruso, S.; Mazzanti, G.; Uccella, N.; Castelli, F. Study on the mechanisms of the antibacterial action of some plant alpha,beta-unsaturated aldehydes. Lett. Appl. Microbiol. 2002, 35, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Lanciotti, R.; Belletti, N.; Patrignani, F.; Gianotti, A.; Gardini, F.; Guerzoni, M.E. Application of Hexanal, (E)-2-Hexenal, and Hexyl Acetate To Improve the Safety of Fresh-Sliced Apples. J. Agric. Food Chem. 2003, 51, 2958–2963. [Google Scholar] [CrossRef]
- Zhang, J.; Sun, H.; Chen, S.; Zeng, L.; Wang, T. Anti-fungal activity, mechanism studies on α-Phellandrene and Nonanal against Penicillium cyclopium. Bot. Stud. 2017, 58, 1–9. [Google Scholar] [CrossRef]
- Zavala-Sánchez, M.A.; Pérez-Gutiérrez, S.; Pérez-González, C.; Sánchez-Saldivar, D.; Arias-García, L. Antidiarrhoeal activity of nonanal, an aldehyde isolated from Artemisia ludoviciana. Pharm. Biol. 2002, 40, 263–268. [Google Scholar] [CrossRef]
- Kim, M.; Sowndhararajan, K.; Choi, H.J.; Park, S.J.; Kim, S. Olfactory Stimulation Effect of Aldehydes, Nonanal, and Decanal on the Human Electroencephalographic Activity, According to Nostril Variation. Biomedicines 2019, 7, 57. [Google Scholar] [CrossRef]
- Barbosa, R.M.R.; Furtado, A.; Regis, L.; Leal, W.S. Evaluation of an oviposition-stimulating kairomone for the yellow fever mosquito, Aedes aegypti, in Recife, Brazil. J. Vector Ecol. 2010, 35, 204–207. [Google Scholar] [CrossRef]
- Turkez, H.; Togar, B.; Tatar, A.; Geyıkoglu, F.; Hacımuftuoglu, A. Cytotoxic and cytogenetic effects of α-copaene on rat neuron and N2a neuroblastoma cell lines. Biologia 2014, 69, 936–942. [Google Scholar] [CrossRef]
- Shelly, T.E. Exposure to α-Copaene and α-Copaene-Containing Oils Enhances Mating Success of Male Mediterranean Fruit Flies (Diptera: Tephritidae). Ann. Entomol. Soc. Am. 2006, 94, 497–502. [Google Scholar] [CrossRef]
- Pieri, F.A.; de Castro Souza, M.C.; Vermelho, L.L.R.; Vermelho, M.L.R.; Perciano, P.G.; Vargas, F.S.; Borges, A.P.B.; da Veiga-Junior, V.F.; Moreira, M.A.S. Use of β-caryophyllene to combat bacterial dental plaque formation in dogs. BMC Vet. Res. 2016, 12. [Google Scholar] [CrossRef]
- Cipriano, M.; Neta, S.; Vittorazzi, C.; Guimarães, A.C.; Damasceno, J.; Martins, L.; Fronza, M.; Coutinho Endringer, D.; Scherer, R.; Guimar, A.C.; et al. Pharmaceutical Biology Effects of β-caryophyllene and Murraya paniculata essential oil in the murine hepatoma cells and in the bacteria and fungi 24-h time-kill curve studies. Pharm. Biol. 2017, 55. [Google Scholar] [CrossRef]
- Venturi, C.R.; Danielli, L.J.; Klein, F.; Apel, M.A.; Montanha, J.A.; Bordignon, S.A.L.; Roehe, P.M.; Fuentefria, A.M.; Henriques, A.T. Pharmaceutical Biology Chemical analysis and in vitro antiviral and antifungal activities of essential oils from Glechon spathulata and Glechon marifolia Chemical analysis and in vitro antiviral and antifungal activities of essential oils from Glechon spa. John M. Pezzuto Pharm. Biol. 2015, 53, 682–688. [Google Scholar] [CrossRef]
- Brito, L.F.; Oliveira, H.B.M.; Neves Selis, N.; Souza, C.L.S.; Júnior, M.N.S.; Souza, E.P.; Silva, L.S.C.d.; Souza Nascimento, F.; Amorim, A.T.; Campos, G.B.; et al. Anti-inflammatory activity of β-caryophyllene combined with docosahexaenoic acid in a model of sepsis induced by Staphylococcus aureus in mice. J. Sci. Food Agric. 2019, 99, 5870–5880. [Google Scholar] [CrossRef]
- Dahham, S.S.; Tabana, Y.M.; Iqbal, M.A.; Ahamed, M.B.K.; Ezzat, M.O.; Majid, A.S.A.; Majid, A.M.S.A. The anticancer, antioxidant and antimicrobial properties of the sesquiterpene β-caryophyllene from the essential oil of Aquilaria crassna. Molecules 2015, 20, 11808–11829. [Google Scholar] [CrossRef]
- Fidyt, K.; Fiedorowicz, A.; Strządała, L.; Szumny, A. β -caryophyllene and β -caryophyllene oxide-natural compounds of anticancer and analgesic properties. Cancer Med. 2016, 5, 3007–3017. [Google Scholar] [CrossRef]
- Vuko, E.; Rusak, G.; Dunkic, V.; Kremer, D.; Kosalec, I.; Rada, B.; Bezic, N. Inhibition of satellite RNA associated cucumber mosaic virus infection by essential oil of micromeria croatica (pers.) schott. Molecules 2019, 24, 1342. [Google Scholar] [CrossRef]
- Moreno, É.M.; Leal, S.M.; Stashenko, E.E.; García, L.T. Induction of programmed cell death in Trypanosoma cruzi by Lippia alba essential oils and their major and synergistic terpenes (citral, limonene and caryophyllene oxide). BMC Complement. Altern. Med. 2018, 18, 225. [Google Scholar] [CrossRef]
- de Souza Araújo, C.; Paula de Oliveira, A.; Nascimento Lima, R.; Barreto Alves, P.; Coimbra Diniz, T.; Roberto Guedes da Silva Almeida, J. Chemical constituents and antioxidant activity of the essential oil from leaves of Annona vepretorum Mart. (Annonaceae). Pharmacogn. Mag. 2015, 11, 615–618. [Google Scholar] [CrossRef]
- Guerrini, A.; Sacchetti, G.; Grandini, A.; Spagnoletti, A.; Asanza, M.; Scalvenzi, L. Cytotoxic Effect and TLC Bioautography-Guided Approach to Detect Health Properties of Amazonian Hedyosmum sprucei Essential Oil. Evidence-based Complement. Altern. Med. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Zardi-Bergaoui, A.; Jelizi, S.; Flamini, G.; Ascrizzi, R.; Ben Jannet, H. Comparative study of the chemical composition and bioactivities of essential oils of fresh and dry seeds from Myoporum insulare R. Br. Ind. Crops Prod. 2018, 111, 232–237. [Google Scholar] [CrossRef]
- Ali, N.; Chhetri, B.; Dosoky, N.; Shari, K.; Al-Fahad, A.; Wessjohann, L.; Setzer, W. Antimicrobial, Antioxidant, and Cytotoxic Activities of Ocimum forskolei and Teucrium yemense (Lamiaceae) Essential Oils. Medicines 2017, 4, 17. [Google Scholar] [CrossRef]
- de Oliveira, M.S.; da Cruz, J.N.; Gomes Silva, S.; da Costa, W.A.; de Sousa, S.H.B.; Bezerra, F.W.F.; Teixeira, E.; da Silva, N.J.N.; de Aguiar Andrade, E.H.; de Jesus Chaves Neto, A.M.; et al. Phytochemical profile, antioxidant activity, inhibition of acetylcholinesterase and interaction mechanism of the major components of the Piper divaricatum essential oil obtained by supercritical CO. J. Supercrit. Fluids 2019, 145, 74–84. [Google Scholar] [CrossRef]
- Silva, S.G.; da Costa, R.A.; de Oliveira, M.S.; da Cruz, J.N.; Figueiredo, P.L.B.; Brasil, D.d.S.B.; Nascimento, L.D.; Chaves Neto, A.M.d.J.; de Carvalho Junior, R.N.; Andrade, E.H.d.A. Chemical profile of Lippia thymoides, evaluation of the acetylcholinesterase inhibitory activity of its essential oil, and molecular docking and molecular dynamics simulations. PLoS ONE 2019, 14, e0213393. [Google Scholar] [CrossRef]
- Lopes, N.P.; Kato, M.J.; de Aguiar Andrade, E.H.; Soares Maia, J.G.; Yoshida, M. Circadian and seasonal variation in the essential oil from Virola surinamensis leaves. Phytochemistry 1997, 46, 689–693. [Google Scholar] [CrossRef]
- Gurgel, E.S.C.; de Oliveira, M.S.; Souza, M.C.; da Silva, S.G.; de Mendonça, M.S.; Souza Filho, A.P.d.S. Chemical compositions and herbicidal (phytotoxic) activity of essential oils of three Copaifera species (Leguminosae-Caesalpinoideae) from Amazon-Brazil. Ind. Crops Prod. 2019, 142, 111850. [Google Scholar] [CrossRef]
- Stein, S.; Mirokhin, D.; Tchekhovskoi, D.; Mallard, G.; Mikaia, A.; Zaikin, V.; Sparkmanm, D. The NIST Mass Spectral Search Program for the Nist/Epa/Nih Mass Spectra Library; Standard Reference Data Program of the National Institute of Standards and Technology: Gaithers-burg, MD, USA, 2011. [Google Scholar]
Sample Availability: Samples of the compounds (essential oil extracted from Myrcia eximia DC) are available from the authors. |
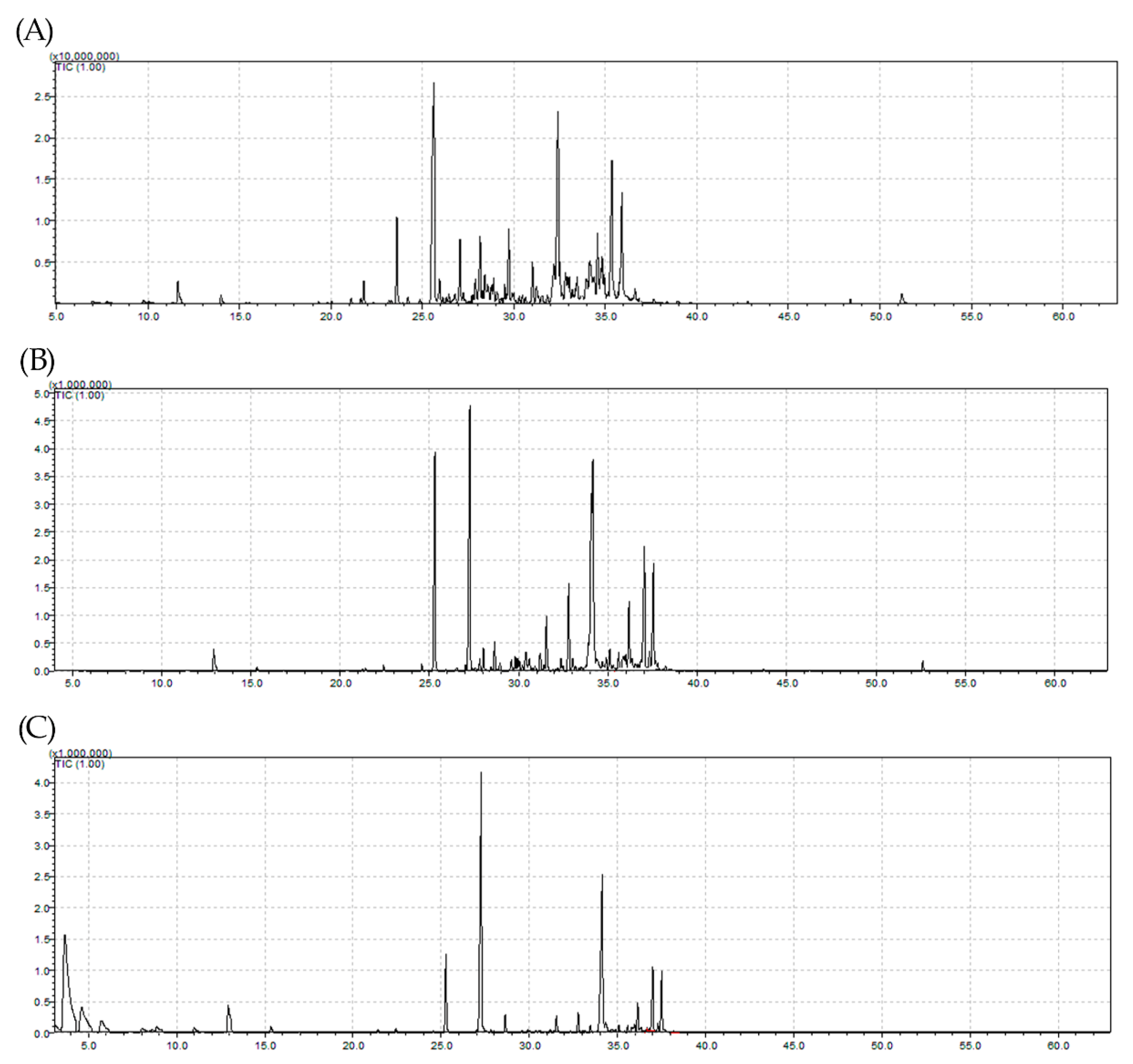
| RI (C) | RI (L) | Constituents | 2017 | 2018 | |
|---|---|---|---|---|---|
| HD | HD | SD | |||
| 798 | 801 | Hexanal | 26.1 | ||
| 845 | 846 | (2E)-Hexenal | 6.63 | ||
| 901 | 901 | Heptanal | 1.78 | ||
| 1003 | 998 | Octanal | 0.59 | ||
| 1009 | 1005 | (2E,4E)-Heptadienal | 0.24 | ||
| 1054 | 1049 | (2E)-Octen-1-al | 0.2 | 0.69 | |
| 1062 | 1060 | (2E)-Octen-1-ol | 0.05 | ||
| 1104 | 1100 | Nonanal | 1.28 | 3.24 | 1.42 |
| 1158 | 1157 | (2E)-Nonen-1-al | 0.41 | 0.53 | 0.2 |
| 1190 | 1186 | α-Terpineol | 0.05 | ||
| 1194 | 1190 | Methyl salicylate | 0.05 | ||
| 1294 | 1292 | (2E,4Z)-Decadienal | 0.12 | 0.18 | 0.1 |
| 1289 | 1299 | 10-Undecenal | 0.06 | 0.04 | |
| 1318 | 1315 | (2E,4E)-Decadienal | 0.2 | 0.23 | 0.22 |
| 1333 | 1335 | δ-Elemene | 0.96 | ||
| 1345 | 1345 | α-Cubebene | 0.05 | ||
| 1362 | 1357 | Undec-(2E)-enal | 0.11 | 0.1 | 0.27 |
| 1369 | 1373 | α-Ylangene | 0.16 | ||
| 1374 | 1374 | α-Copaene | 3.25 | 4.84 | 10.98 |
| 1387 | 1389 | β-Elemene | 0.24 | ||
| 1402 | 1400 | β-Longipinene | 0.19 | ||
| 1404 | 1403 | Eugenol methyl | |||
| 1418 | 1415 | (2E,4E)-Undecadienal | 0.14 | 0.25 | |
| 1420 | 1417 | (E)-Caryophyllene | 15.71 | 20.3 | 15 |
| 1426 | 1419 | β-Ylangene | 0.19 | ||
| 1429 | 1428 | (E)-α-Ionone | 0.12 | ||
| 1431 | 1432 | trans-α-Bergamotene | 0.25 | 0.15 | 0.49 |
| 1435 | 1434 | ϒ-Elemene | 1.08 | 0.07 | |
| 1438 | 1439 | Aromadendrene | 0.25 | 0.97 | |
| 1441 | 1440 | (Z)-β-Farnesene | 0.08 | ||
| 1442 | 1442 | Guaia-6,9-diene | 0.31 | ||
| 1445 | 1447 | Isogermacrene D | 0.65 | ||
| 1453 | 1451 | trans-Muurola-3,5-diene | 0.13 | ||
| 1454 | 1452 | α-Humulene | 2.41 | 1.03 | 1.37 |
| 1455 | 1453 | Geranyl acetone | 0.02 | 0.19 | |
| 1457 | 1458 | Alloaromadendrene | 0.4 | 0.45 | |
| 1465 | 1464 | 9-epi-(E)-Caryophyllene | 0.04 | ||
| 1466 | 1465 | Thujopsadiene | 0.05 | ||
| 1470 | 1471 | Dauca-5,8-diene | 0.35 | ||
| 1475 | 1478 | ϒ-Muurolene | 1.1 | ||
| 1480 | 1479 | α-Curcumene | 0.65 | ||
| 1484 | 1483 | α-Amorphene | 0.42 | ||
| 1486 | 1483* | trans-β-Bergamotene | 0.13 | 0.51 | |
| 1487 | 1484 | Germacrene D | 2.93 | 0.08 | 0.58 |
| 1490 | 1489 | β-Selinene | 0.46 | ||
| 1491 | 1489 | cis-Eudesma-6,11-diene | 0.1 | ||
| 1492 | 1492 | cis-β-Guaiene | 1.21 | ||
| 1495 | 1496 | Valencene | 0.3 | ||
| 1498 | 1496 | Viridiflorene | 1.03 | 1.29 | |
| 1499 | 1500 | α-Muurolene | 0.89 | 0.8 | |
| 1508 | 1505 | Premnaspirodiene | 0.09 | ||
| 1510 | 1505 | β-Bisabolene | 0.29 | ||
| 1511 | 1509 | Guaia-1(10),11-diene | 0.8 | ||
| 1512 | 1509 | α-Bulnesene | 0.16 | ||
| 1513 | 1511 | δ-Amorphene | 0.64 | 0.05 | |
| 1514 | 1513 | ϒ-Cadinene | 0.63 | ||
| 1515 | 1514 | β-Curcumene | 0.04 | ||
| 1516 | 1514 | Cubebol | 0.21 | 1.03 | |
| 1521 | 1518 | Dodecadienal (2E,4E) | 0.09 | 0.31 | |
| 1522 | 1522 | δ-Cadinene | 2.75 | 1.1 | 2.61 |
| 1523 | 1528 | Zonarene | 0.44 | ||
| 1528 | 1531 | (Z)-Nerolidol | 0.35 | ||
| 1531 | 1533 | trans-Cadina-1,4-diene | 0.21 | ||
| 1537 | 1537 | α-Cadinene | 0.29 | ||
| 1542 | 1544 | α-Calacorene | 0.23 | 0.58 | |
| 1546 | 1547 | Italicene epoxide | 1.57 | 1.22 | 4.37 |
| 1556 | 1556 | trans-Dauca-4(11),7-diene | 0.1 | ||
| 1563 | 1561 | (E)-Nerolidol | 0.09 | 0.22 | |
| 1564 | 1562 | epi-Longipinanol | 0.86 | 0.15 | 0.71 |
| 1567 | 1567 | Palustrol | 0.31 | ||
| 1572 | 1570 | Dendrolasin | 0.21 | ||
| 1575 | 1571 | Caryolan-8-ol | 0.22 | ||
| 1576 | 1577 | Spathulenol | 2.67 | 1.5 | |
| 1581 | 1582 | Caryophyllene oxide | 10.25 | 16.3 | 22.16 |
| 1582 | 1484 | Germacrene B | 0.56 | ||
| 1590 | 1586 | Thujopsan-2α-ol | 0.69 | ||
| 1592 | 1589 | Allohedycayol | 0.53 | ||
| 1593 | 1590 | Globulol | 1.78 | ||
| 1595 | 1592 | Viridiflorol | 0.72 | 0.16 | |
| 1596 | 1595 | Cubeban-11-ol | 1.7 | ||
| 1597 | 1596 | Fokienol | 0.98 | ||
| 1605 | 1600 | Rosifoliol | 0.5 | 0.2 | |
| 1607 | 1608 | Humulene epoxide II | 1.05 | 0.36 | 1.07 |
| 1615 | 1618 | 1,10-di-epi-Cubenol | 0.74 | ||
| 1620 | 1618 | Junenol | 1.18 | ||
| 1625 | 1627 | 1-epi-Cubenol | 1.85 | 0.48 | 0.52 |
| 1627 | 1629 | Eremoligenol | 0.58 | 0.2 | |
| 1632 | 1635 | cis-Cadin-4-en-7-ol | 1.18 | ||
| 1641 | 1638 | epi-α-Cadinlol | 1.17 | ||
| 1642 | 1639 | Caryophylla-4(12),8(13)-dien-5-α-ol | 3.31 | 1.69 | 3.35 |
| 1643 | 1639 | Alloromadendrene epoxide | 1.13 | 0.49 | 1.57 |
| 1644 | 1640 | epi-α-Muurolol | 1.71 | 0.39 | |
| 1645 | 1644 | α-Muurolol | 1.21 | ||
| 1651 | 1645 | Cubenol | 0.11 | ||
| 1656 | 1649 | β-Eudesmol | 0.22 | 0.28 | |
| 1359 | 1652 | Himachalol | 0.15 | 0.4 | |
| 1656 | 1652 | α-Cadinol | 5.0 | 0.1 | 0.46 |
| 1660 | 1658 | Selin-11-en-4α-ol | 0.58 | ||
| 1662 | 1661 | Allohimachalol | 3.49 | ||
| 1670 | 1668 | 14-Hydroxy-9-epi-(E)-caryophyllene | 7.02 | 4.63 | 7.84 |
| 1688 | 1685 | Germacra-4(15),5,10(14)-trien-1α-ol | 0.72 | 3.7 | |
| 1696 | 1700 | Eudesm-7(11)-en-4-ol | 0.21 | ||
| 1699 | 1706 | 14-Hydroxy-4,5-dihydro-caryophyllene | 0.17 | ||
| 1711 | 1713 | 14-Hydroxy-α-humulene | 0.09 | ||
| 1712 | 1714 | Nootkatol | 0.08 | ||
| 1734 | 1740 | Mint sulfide | 0.05 | ||
| 1840 | 1841 | Phytone | 0.05 | 0.08 | |
| 1944 | 1942 | Phytol | 0.02 | 0.43 | |
| Aldehydes | 2.38 | 40.5 | 2.81 | ||
| Hydrocarbon sesquiterpenes | 36.21 | 23 | 26.74 | ||
| Oxygenated sesquiterpenes | 53.41 | 30.5 | 53.89 | ||
| Others | 0.27 | 0.02 | 0.85 | ||
| Total | 96.93 | 98.8 | 95.27 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira, O.O.; da Cruz, J.N.; Franco, C.d.J.P.; Silva, S.G.; da Costa, W.A.; de Oliveira, M.S.; Andrade, E.H.d.A. First Report on Yield and Chemical Composition of Essential Oil Extracted from Myrcia eximia DC (Myrtaceae) from the Brazilian Amazon. Molecules 2020, 25, 783. https://doi.org/10.3390/molecules25040783
Ferreira OO, da Cruz JN, Franco CdJP, Silva SG, da Costa WA, de Oliveira MS, Andrade EHdA. First Report on Yield and Chemical Composition of Essential Oil Extracted from Myrcia eximia DC (Myrtaceae) from the Brazilian Amazon. Molecules. 2020; 25(4):783. https://doi.org/10.3390/molecules25040783
Chicago/Turabian StyleFerreira, Oberdan Oliveira, Jorddy Neves da Cruz, Celeste de Jesus Pereira Franco, Sebastião Gomes Silva, Wanessa Almeida da Costa, Mozaniel Santana de Oliveira, and Eloisa Helena de Aguiar Andrade. 2020. "First Report on Yield and Chemical Composition of Essential Oil Extracted from Myrcia eximia DC (Myrtaceae) from the Brazilian Amazon" Molecules 25, no. 4: 783. https://doi.org/10.3390/molecules25040783
APA StyleFerreira, O. O., da Cruz, J. N., Franco, C. d. J. P., Silva, S. G., da Costa, W. A., de Oliveira, M. S., & Andrade, E. H. d. A. (2020). First Report on Yield and Chemical Composition of Essential Oil Extracted from Myrcia eximia DC (Myrtaceae) from the Brazilian Amazon. Molecules, 25(4), 783. https://doi.org/10.3390/molecules25040783










