Curcumin, an Active Constituent of Turmeric Spice: Implication in the Prevention of Lung Injury Induced by Benzo(a) Pyrene (BaP) in Rats
Abstract
1. Introduction
2. Results
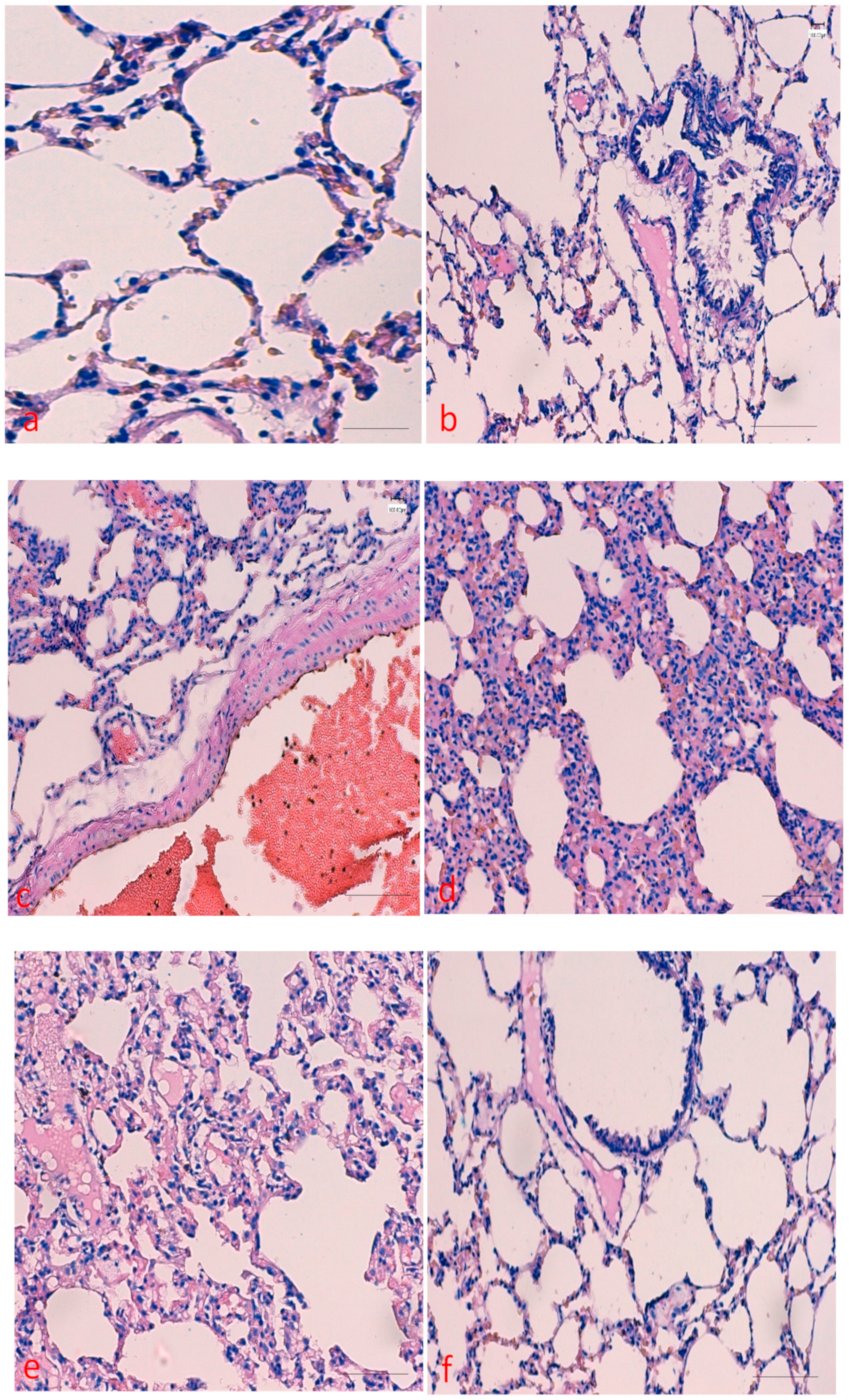
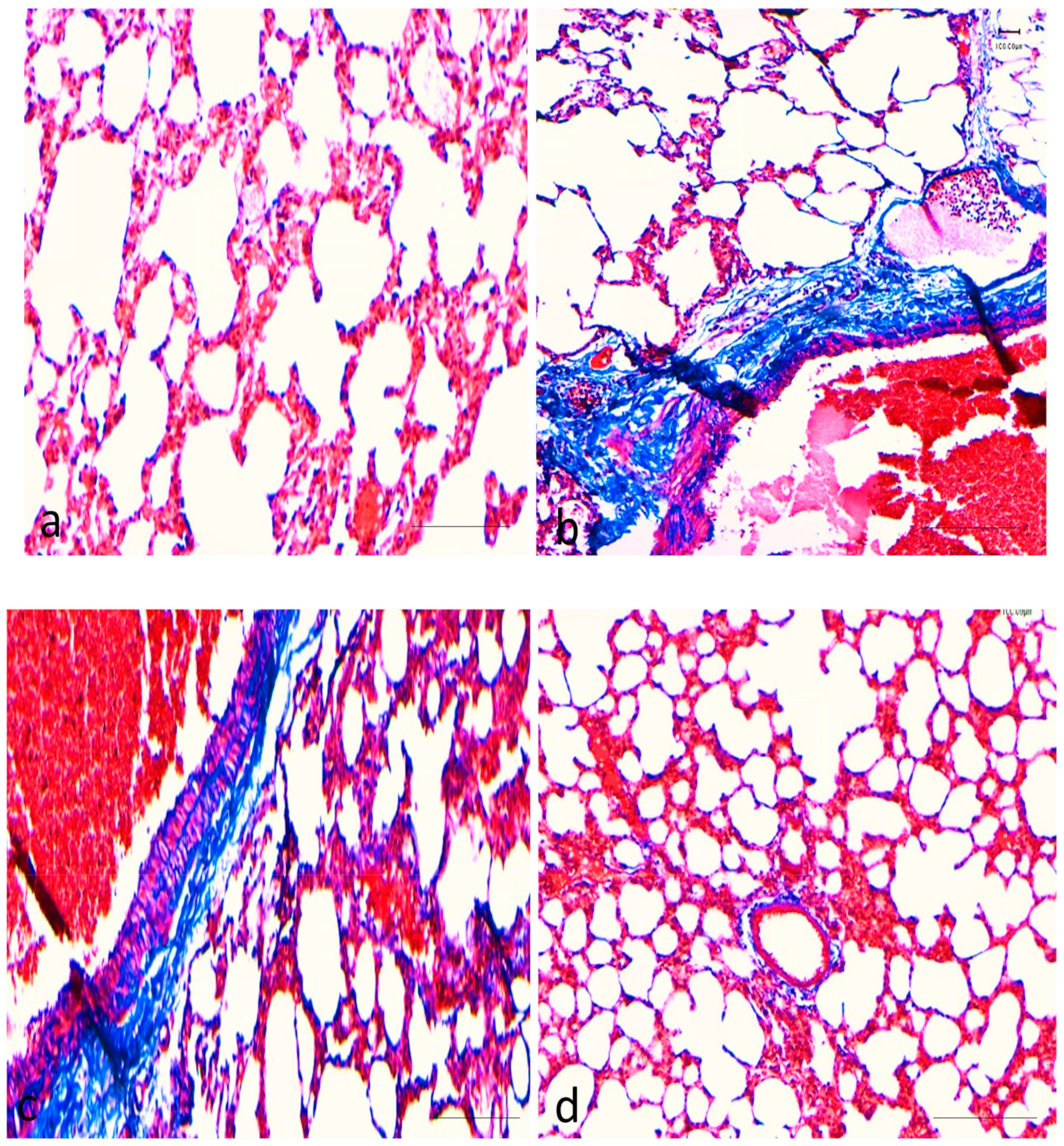
2.1. Effects of Curcumin on Benzopyrene(BaP)-Induced Lung Tissue Alterations
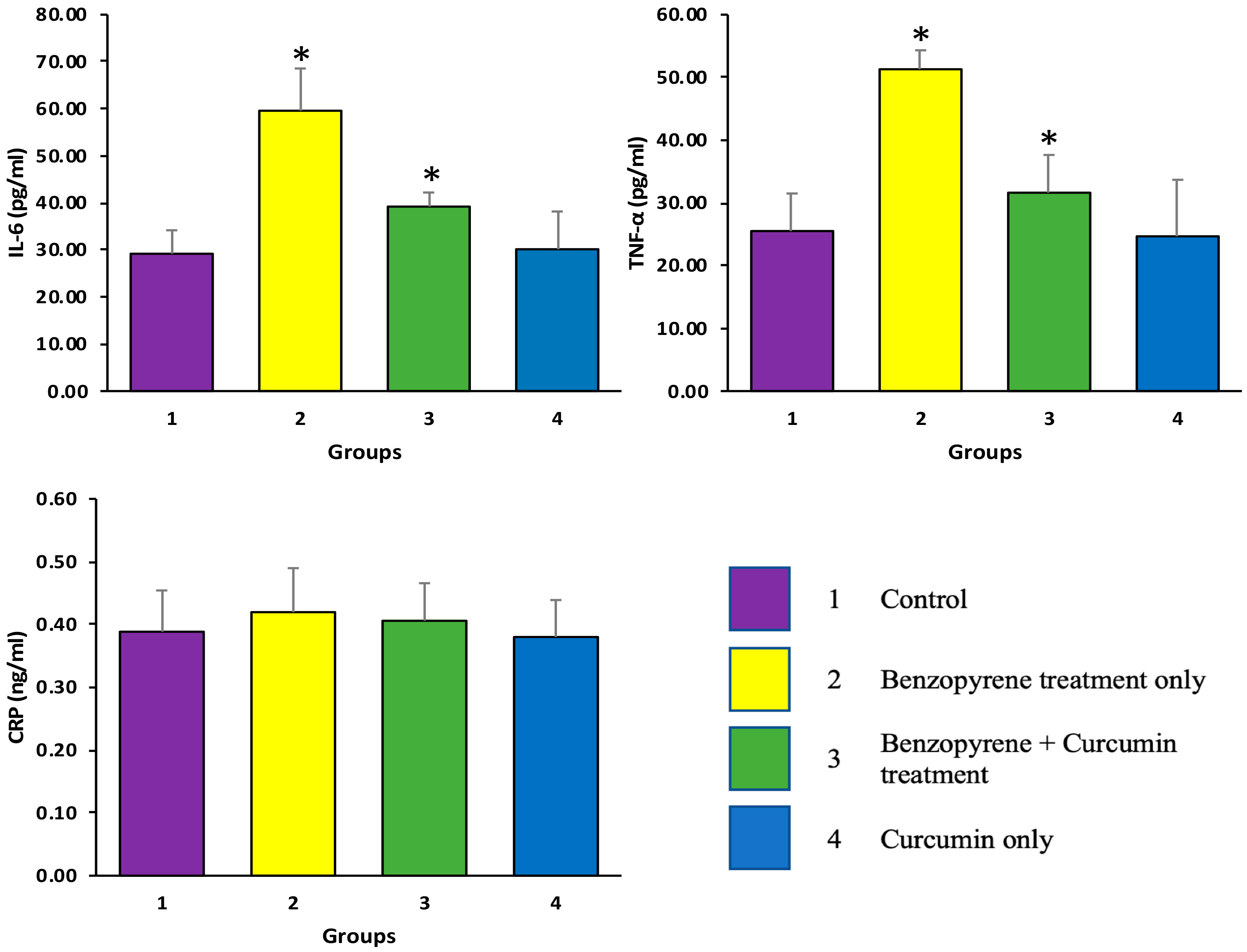
2.2. Effects of Curcumin on BaP-Induced Production of TNF-α, IL-6, and CRP
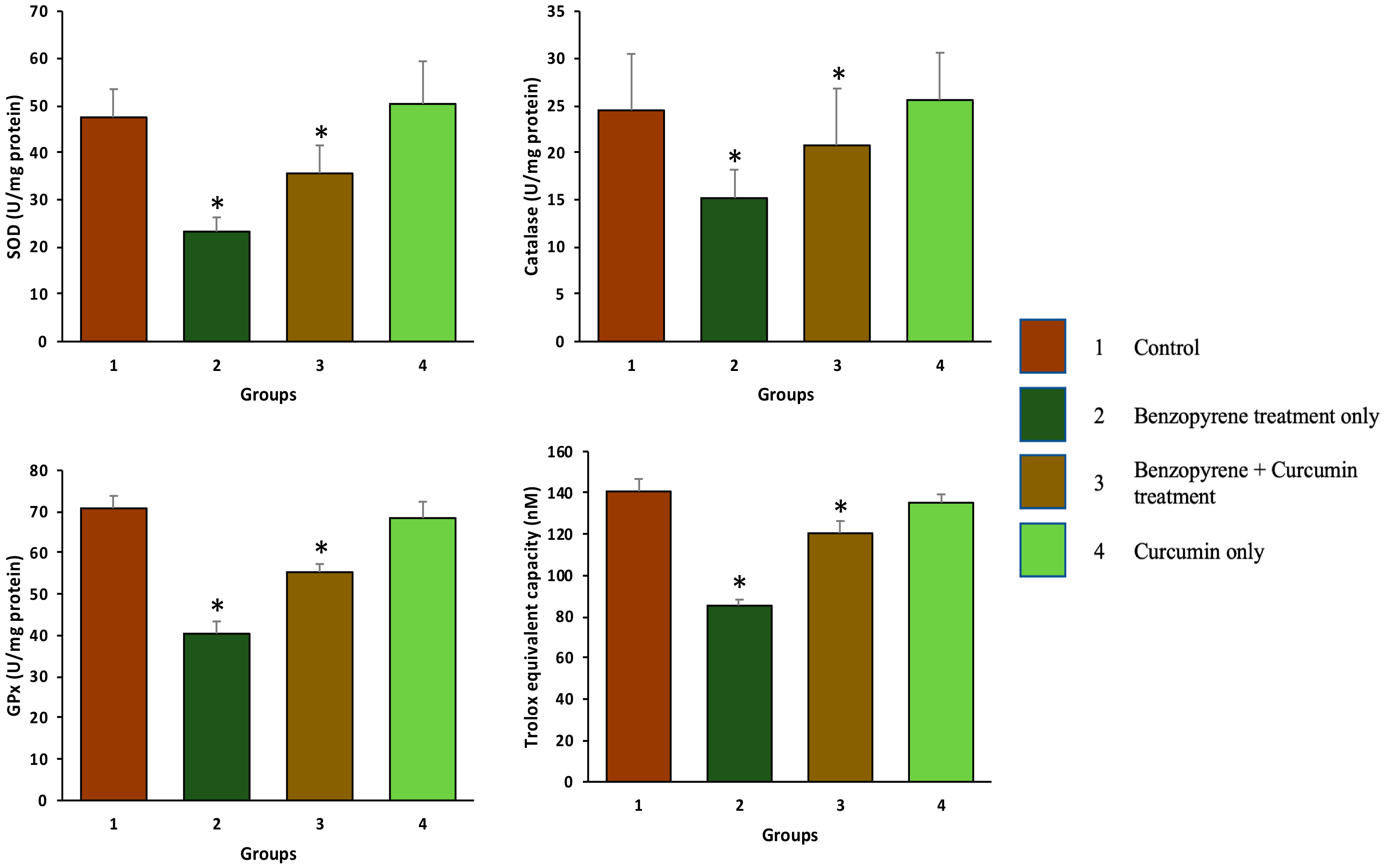
2.3. Effect of Curcumin on Antioxidant Enzymes
2.4. Effect of Curcumin on Total Antioxidant Capacity (TAC)
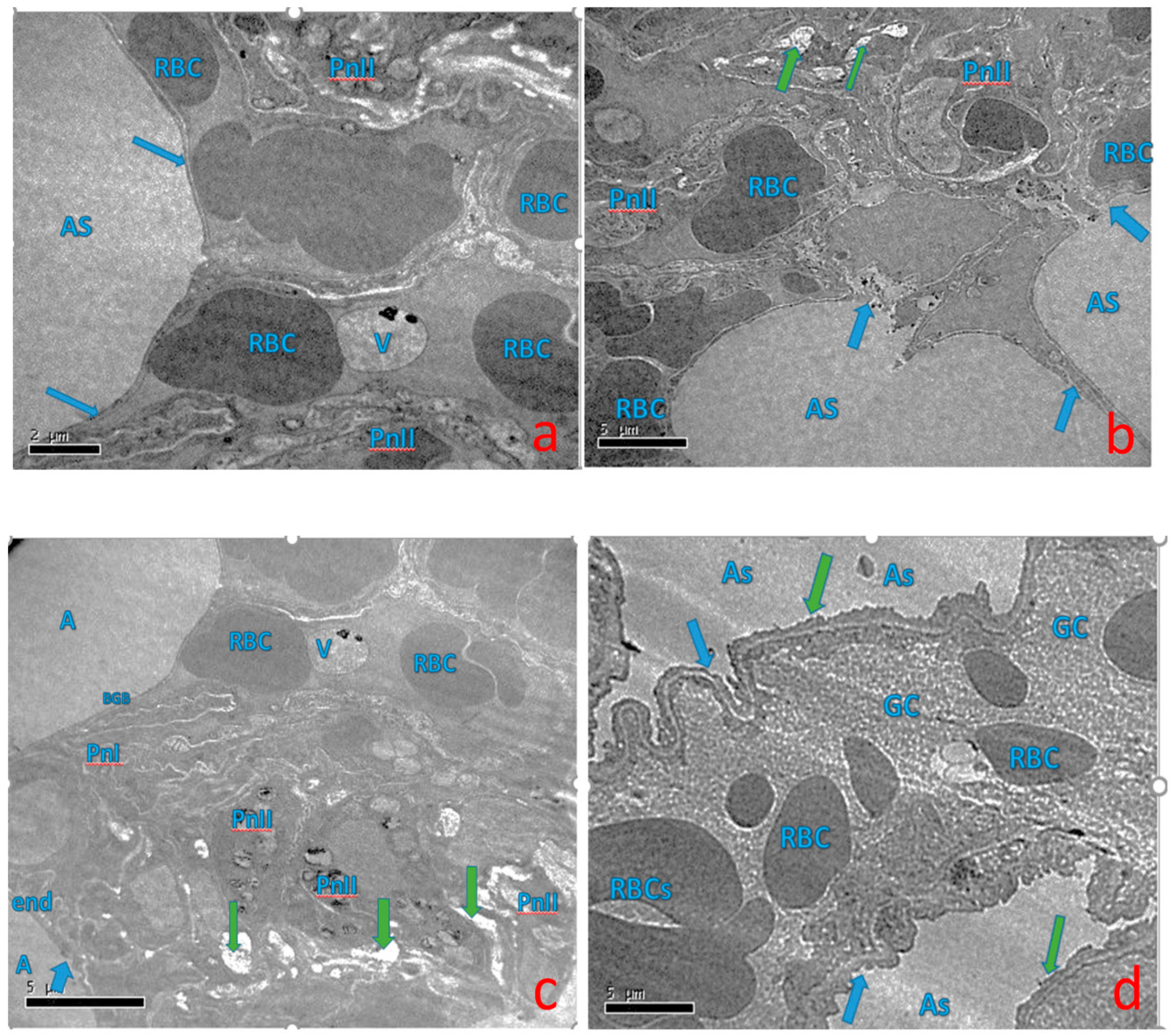
2.5. Effect of Curcumin on Ultra-Structural Changes of Lung Tissue
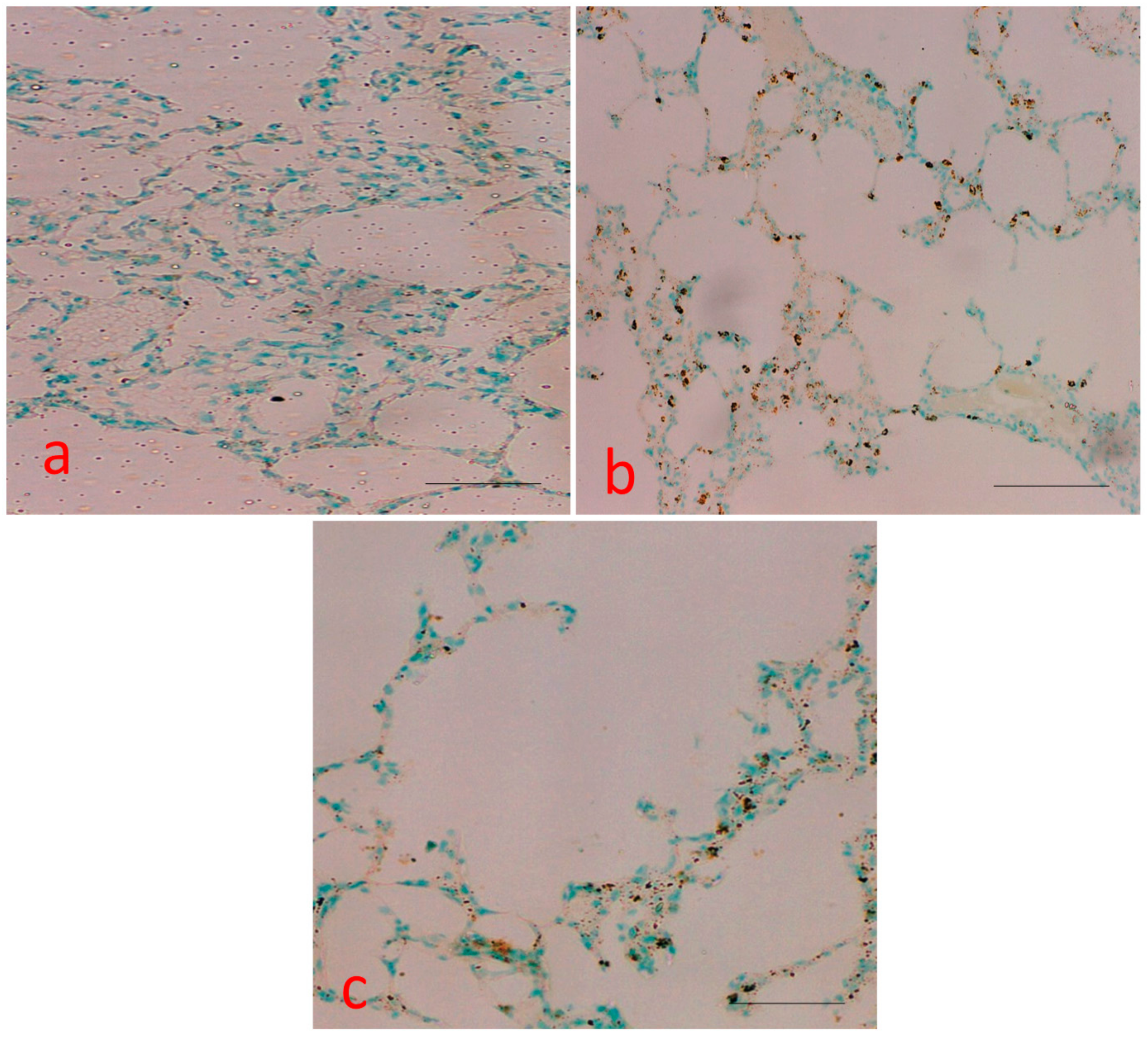
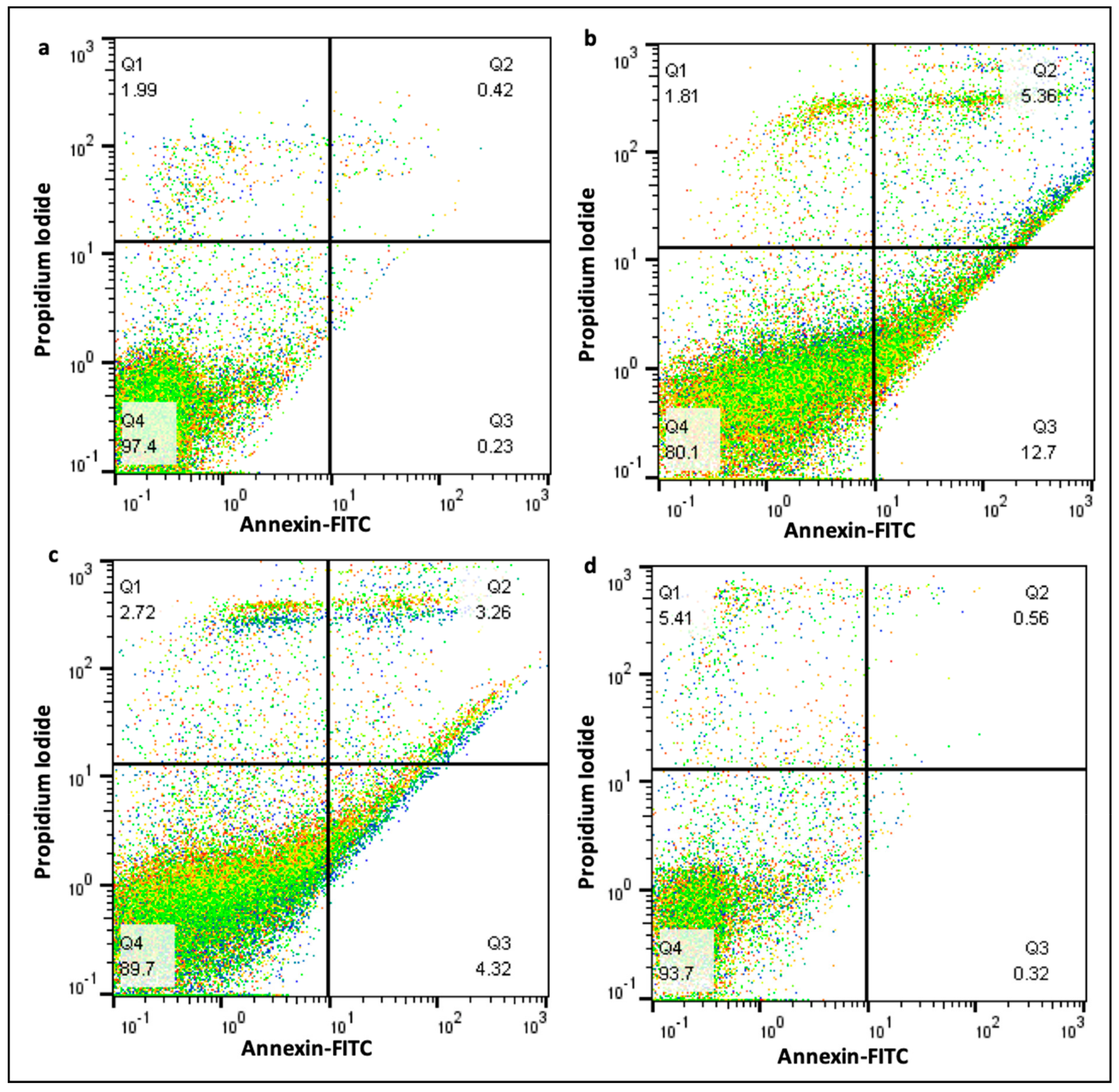
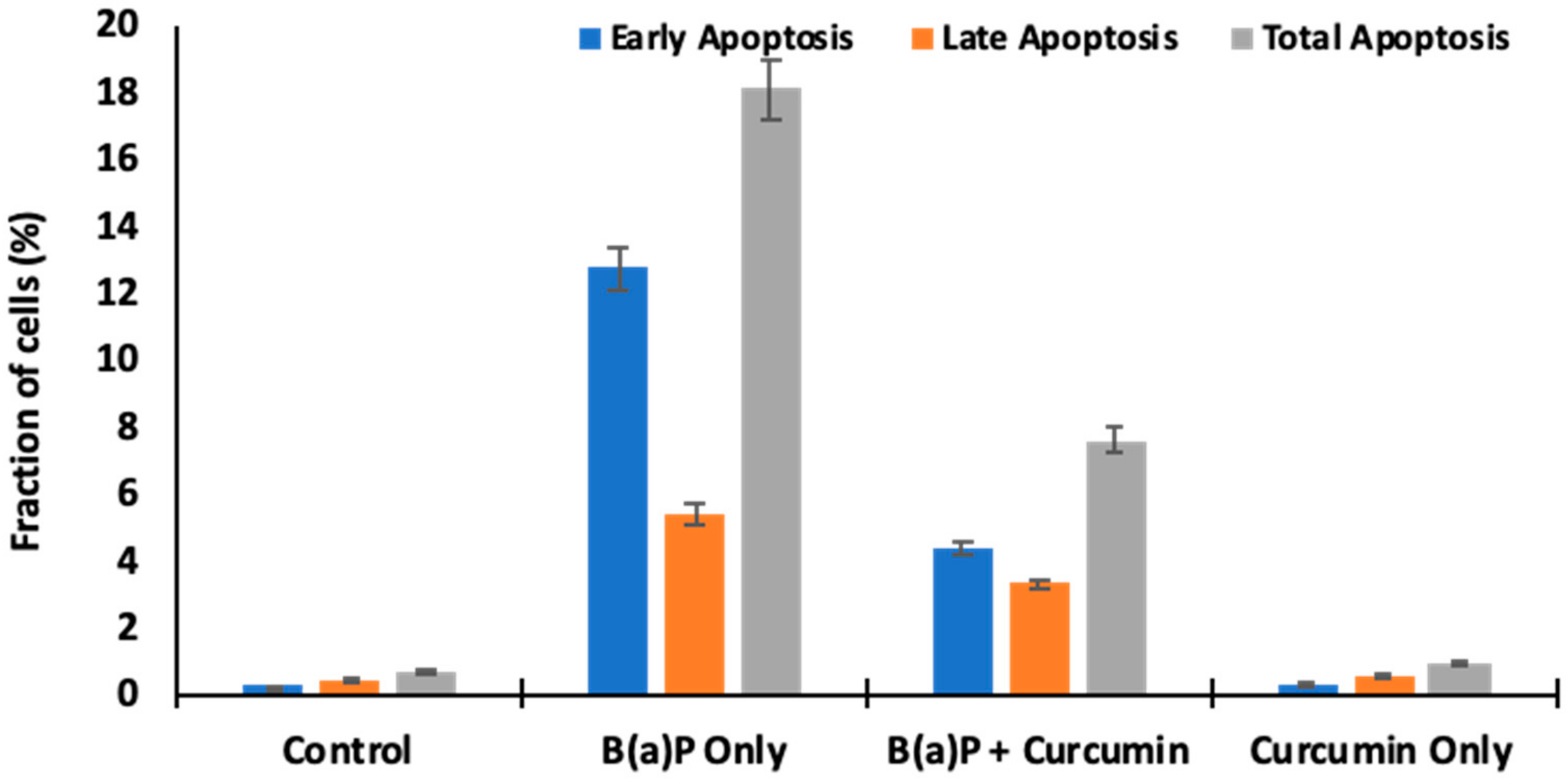
2.6. Measurement of Apoptotic Bodies
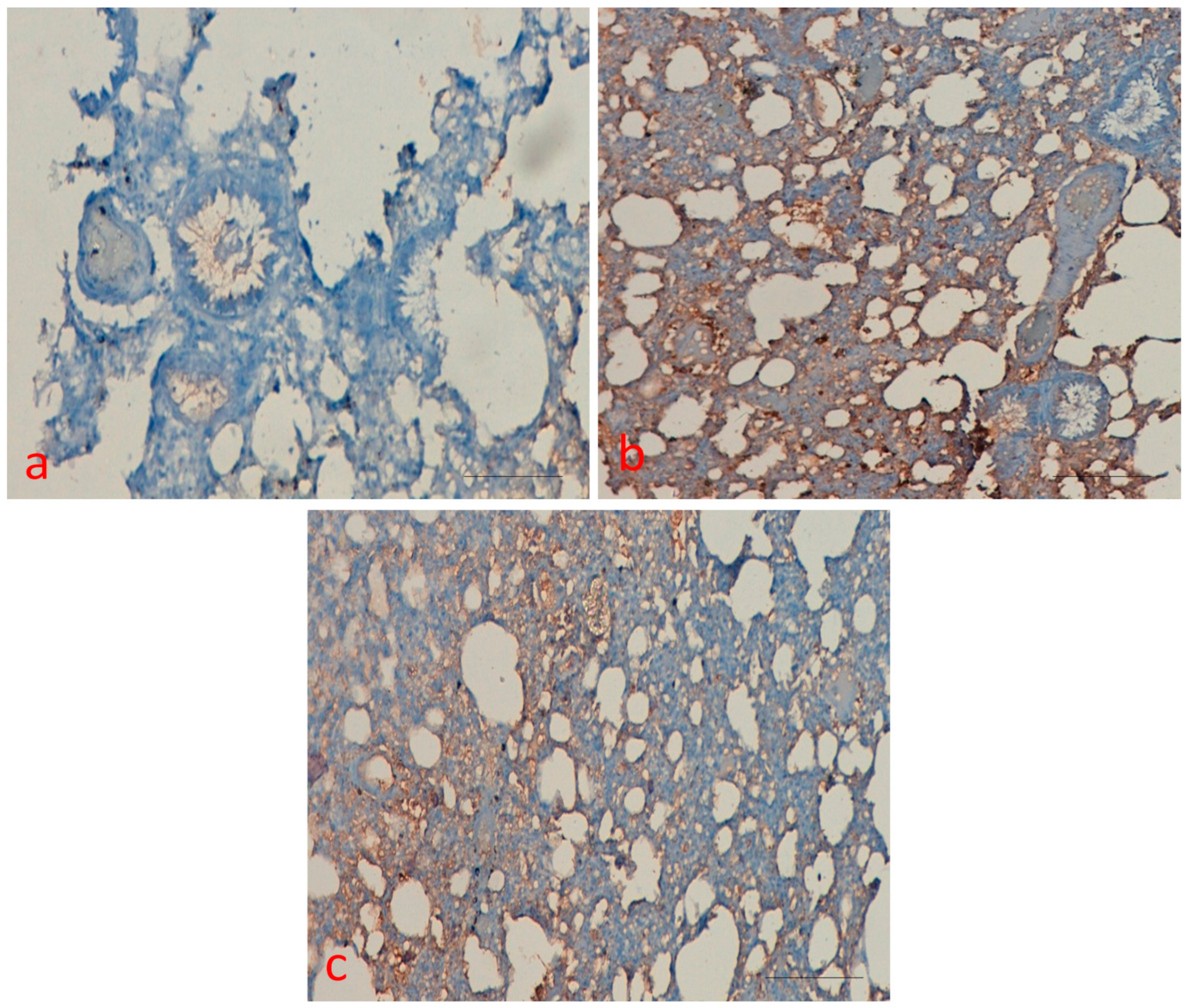
2.7. Effect of Curcumin on p53 Protein Expression
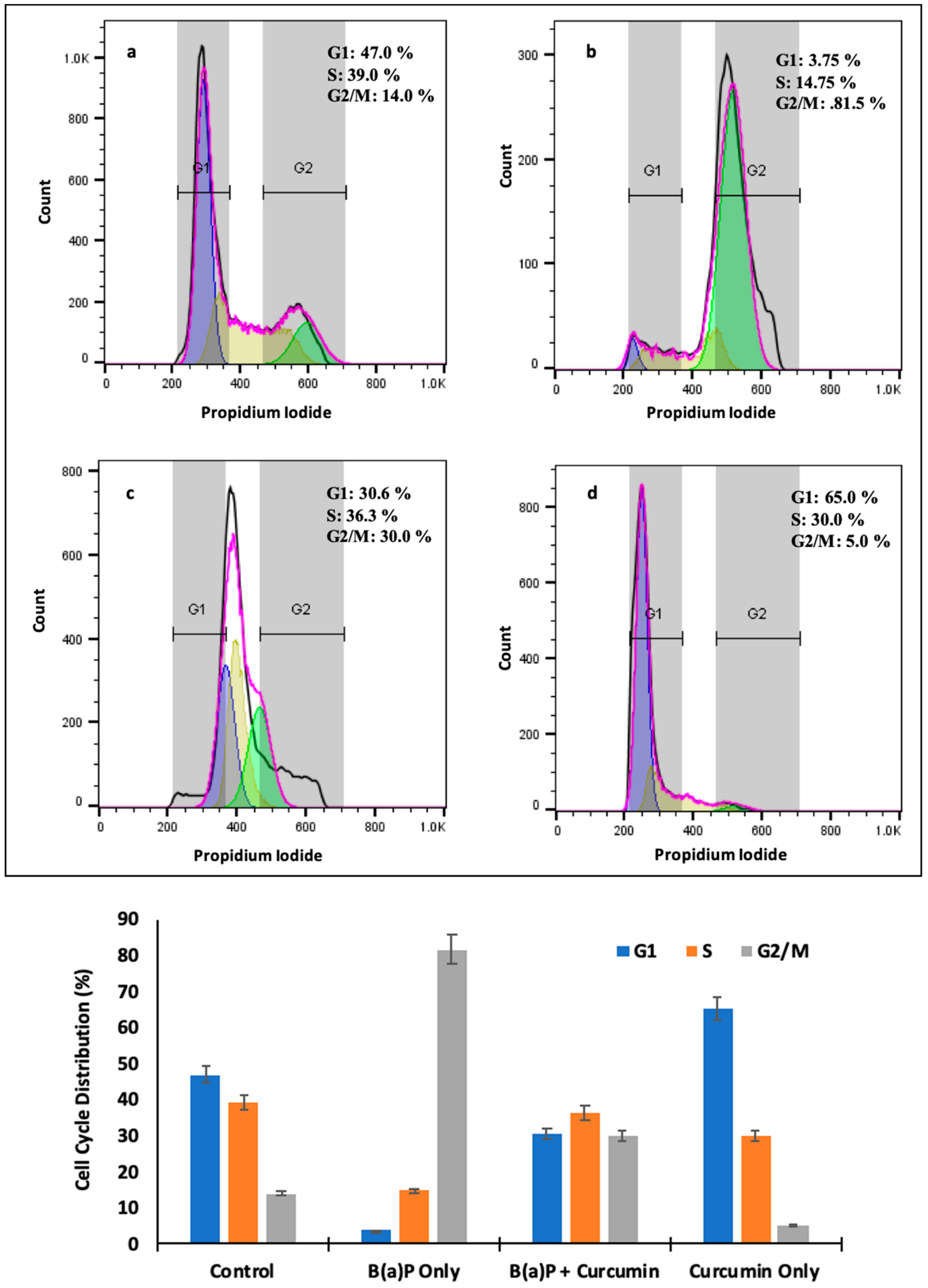
2.8. Effect of Curcumin on Cell Cycle
2.9. Effect of Curcumin on Cell Death
3. Discussion
4. Materials and Methods
4.1. Animals and Experimental Protocol
4.2. Measurement of Antioxidant Enz
Antioxidant enzymes (SOD, CAT, and GPx) and Total Antioxidant Capacity
4.3. Measurement of Inflammatory Marker (TNF-α, Interleuin-6), and CRP Level
4.4. Histopathological Analysis
4.5. Immunohistochemical Staining
4.6. Terminal Deoxynucleotidyl Transferase-Mediated dUTP-Biotin Nick-End Labeling (TUNEL) Assay
4.7. Transmission Electron Microscopy
4.8. Cell Cycle Analysis by Flow Cytometry
4.9. Apoptosis Analysis by Flow Cytometry
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Guerreiro, C.B.B.; Horálek, J.; de Leeuw, F.; Couvidat, F. Benzo(a)pyrene in Europe: Ambient Air Concentrations, Population Exposure and Health Effects. Environ Pollut. 2016, 214, 657–667. [Google Scholar] [CrossRef]
- Gentner, N.J.; Weber, L.P. Intranasal benzo[a]pyrene alters circadian blood pressure patterns and causes lung inflammation in rats. Arch. Toxicol. 2011, 85, 337–346. [Google Scholar] [CrossRef] [PubMed]
- Staniszewska, M.; Graca, B.; Bełdowska, M.; Saniewska, D. Factors controlling benzo(a)pyrene concentration in aerosols in the urbanized coastal zone. A case study: Gdynia, Poland (Southern Baltic Sea). Environ. Sci. Pollut. Res. Int. 2013, 20, 4154–4163. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Yuan, J.; Wang, Q.; Lei, T.; Shen, X.; Cui, B.; Zhang, F.; Ding, W.; Lu, Z. Metformin protects against PM2.5-induced lung injury and cardiac dysfunction independent of AMP-activated protein kinase alpha2. Redox Biol. 2020, 28, 101345. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Xu, H.; Wang, H.; Zhang, Y.; Ya, P.; Yang, C.; Li, F. Protective effects of lemongrass essential oil against benzo(a)pyrene-induced oxidative stress and DNA damage in human embryonic lung fibroblast cells. Toxicol. Mech. Methods. 2017, 27, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Sawyer, K.; Mundandhara, S.; Ghio, A.J.; Madden, M.C. The effects of ambient particulate matter on human alveolar macrophage oxidative and inflammatory responses. J. Toxicol. Environ. Health A. 2010, 73, 41–57. [Google Scholar] [CrossRef] [PubMed]
- Granberg, A.L.; Brunstrom, B.; Brandt, I. Cytochrome P450-dependent binding of 7,12-dimethylbenz[a]anthracene (DMBA) and benzo[a]pyrene (B[a]P) in murine heart, lung, and liver endothelial cells. Arch. Toxicol. 2000, 74, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Mu, G.; Fan, L.; Zhou, Y.; Liu, Y.; Ma, J.; Yang, S.; Wang, B.; Xiao, L.; Ye, Z.; Shi, T.; et al. Personal exposure to PM2.5-bound polycyclic aromatic hydrocarbons and lung function alteration: Results of a panel study in China. Sci. Total Environ. 2019, 684, 458–465. [Google Scholar] [CrossRef]
- Zhang, S.Y.; Shao, D.; Liu, H.; Feng, J.; Feng, B.; Song, X.; Zhao, Q.; Chu, M.; Jiang, C.; Huang, W.; et al. Metabolomics analysis reveals that benzo[a]pyrene, a component of PM2.5, promotes pulmonary injury by modifying lipid metabolism in a phospholipase A2-dependent manner in vivo and in vitro. Redox Biol. 2017, 13, 459–469. [Google Scholar] [CrossRef]
- Qamar, W.; Khan, A.Q.; Khan, R.; Lateef, A.; Tahir, M.; Rehman, M.U.; Ali, F.; Sultana, S. Benzo(a)pyrene-induced pulmonary inflammation, edema, surfactant dysfunction, and injuries in rats: Alleviation by farnesol. Exp. Lung Res. 2012, 38, 19–27. [Google Scholar] [CrossRef]
- Dai, W.; Fu, Y.; Deng, Y.; Zeng, Z.; Gu, P.; Liu, H.; Liu, J.; Xu, X.; Wu, D.; Luo, X.; et al. Regulation of Wnt Singaling Pathway by Poly (ADP-Ribose) Glycohydrolase (PARG) Silencing Suppresses Lung Cancer in Mice Induced by Benzo(a)pyrene Inhalation Exposure. Front. Pharmacol. 2019, 10, 338. [Google Scholar] [CrossRef] [PubMed]
- Dhatwalia, S.K.; Kumar, M.; Bhardwaj, P.; Dhawan, D.K. White tea - A cost effective alternative to EGCG in fight against benzo(a)pyrene (BaP) induced lung toxicity in SD rats. Food Chem. Toxicol. 2019, 131, 110551. [Google Scholar] [CrossRef] [PubMed]
- Shi, Q.; Fijten, R.R.; Spina, D.; Vasquez, Y.R.; Arlt, V.M.; Godschalk, R.W.; Van Schooten, F.J. Altered gene expression profiles in the lungs of benzo[a]pyrene-exposed mice in the presence of lipopolysaccharide-induced pulmonary inflammation. Toxicol. Appl. Pharmacol. 2017, 336, 8–19. [Google Scholar] [CrossRef] [PubMed]
- Shahid, A.; Ali, R.; Ali, N.; Kazim Hasan, S.; Barnwal, P.; Mohammad Afzal, S.; Vafa, A.; Sultana, S. Methanolic bark extract of Acacia catechu ameliorates benzo(a)pyrene induced lung toxicity by abrogation of oxidative stress, inflammation, and apoptosis in mice. Environ. Toxicol. 2017, 32, 1566–1577. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, C.; Pan, Y.; Yang, X.; Huang, Y.; Feng, Z.; Li, X.; Yang, S.; Liang, G. A novel synthetic mono-carbonyl analogue of curcumin, A13, exhibits anti-inflammatory effects in vivo by inhibition of inflammatory mediators. Inflammation 2012, 35, 594–604. [Google Scholar] [CrossRef]
- Rahmani, A.H.; Alsahli, M.A.; Aly, S.M.; Khan, M.A.; Aldebasi, Y.H. Role of curcumin in disease prevention and treatment. Adv. Biomed. Res. 2018, 7, 38. [Google Scholar] [CrossRef]
- Rahmani, A.H.; al Zohairy, M.A.; Aly, S.M.; Khan, M.A. Curcumin: A potential candidate in prevention of cancer via modulation of molecular pathways. Biomed. Res. Int. 2014, 2014, 761608. [Google Scholar] [CrossRef]
- Deng, C.; Dang, F.; Gao, J.; Zhao, H.; Qi, S.; Gaoa, M. Acute benzo[a]pyrene treatment causes different antioxidant response and DNA damage in liver, lung, brain, stomach and kidney. Heliyon. 2018, 4, e00898. [Google Scholar] [CrossRef]
- Imran, M.; Sergent, O.; Tête, A.; Gallais, I.; Chevanne, M.; Lagadic-Gossmann, D.; Podechard, N. Membrane Remodeling as a Key Player of the Hepatotoxicity Induced by Co-Exposure to Benzo[a]pyrene and Ethanol of Obese Zebrafish Larvae. Biomolecules 2018, 8. [Google Scholar] [CrossRef]
- Barnwal, P.; Vafa, A.; Afzal, S.M.; Shahid, A.; Hasan, S.K.; Alpashree; Sultana, S. Benzo(a)pyrene induces lung toxicity and inflammation in mice: Prevention by carvacrol. Hum. Exp. Toxicol. 2018, 37, 752–761. [Google Scholar] [CrossRef]
- Siddiqui, F.A.; Prakasam, G.; Chattoppadhyay, S.; Rahaman, A.U.; Padder, R.A.; Ansari, M.A.; Irshad, R.; Manghalara, K.; Bamezai, R.N.K.; Mangalhara, K.; et al. Curcumin decreases Warburg effect in cancer cells by down-regulating pyruvate kinase M2 via mTOR-HIF1alpha inhibition. Sci. Rep. 2018, 8, 8323. [Google Scholar] [CrossRef]
- Yuan, Z.; Syed, M.A.; Panchal, D.; Rogers, D.; Joo, M.; Sadikot, R.T. Curcumin mediated epigenetic modulation inhibits TREM-1 expression in response to lipopolysaccharide. Int. J. Biochem. Cell Biol. 2012, 44, 2032–2043. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Lu, Y.; Zhao, L.; Hu, L.; Qiu, Q.; Zhang, Z.; Li, M.; Hong, G.; Wu, B.; Zhao, G.; et al. Curcumin attenuates sepsis-induced acute organ dysfunction by preventing inflammation and enhancing the suppressive function of Tregs. Int. Immunopharmacol. 2018, 61, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ren, Y.; Yang, Z.; Sun, Z.; Zhang, W.; Chen, X.; Nie, S. Curcumin relieves paraquat-induced lung injury through inhibiting the thioredoxin interacting protein/NLR pyrin domain containing 3mediated inflammatory pathway. Mol. Med. Rep. 2019, 20, 5032–5040. [Google Scholar] [PubMed]
- Hassani, S.; Sepand, M.R.; Jafari, A.; Jaafari, J.; Rezaee, R.; Zeinali, M.; Tavakoli, F.; Razavi-Azarkhiavi, K. Protective effects of curcumin and vitamin E against chlorpyrifos-induced lung oxidative damage. Hum. Exp. Toxicol. 2015, 34, 668–676. [Google Scholar] [CrossRef]
- Huang, K.; Shi, C.; Min, J.; Li, L.; Zhu, T.; Yu, H.; Deng, H. Study on the Mechanism of Curcumin Regulating Lung Injury Induced by Outdoor Fine Particulate Matter (PM2.5). Mediators Inflamm. 2019, 2019, 8613523. [Google Scholar] [CrossRef]
- Khan, S.; Naseem, I. A comparative insight into the oxidative damage and cell death potential of photoilluminated aminophylline - riboflavin system in normal and cancer lung cells of swiss albino mice. Toxicol. In Vitro 2019, 61, 104651. [Google Scholar] [CrossRef]
- Kim, G.; Piao, C.; Oh, J.; Lee, M. Combined delivery of curcumin and the heme oxygenase-1 gene using cholesterol-conjugated polyamidoamine for anti-inflammatory therapy in acute lung injury. Phytomedicine 2019, 56, 165–174. [Google Scholar] [CrossRef]
- Liu, Y.F.; Yang, C.W.; Liu, H.; Sui, S.G.; Li, X.D. Efficacy and Therapeutic Potential of Curcumin Against Sepsis-Induced Chronic Lung Injury in Male Albino Rats. J. Nutr. Health Aging. 2017, 21, 307–313. [Google Scholar] [CrossRef]
- Nguyen, N.H.; Tran, G.B.; Nguyen, C.T. Anti-oxidative effects of superoxide dismutase 3 on inflammatory diseases. J. Mol. Med. (Berl). 2020, 98, 59–69. [Google Scholar] [CrossRef]
- Miao, L.; St Clair, D.K. Regulation of superoxide dismutase genes: Implications in disease. Free Radic. Biol. Med. 2009, 47, 344–356. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Yang, H.; Ramesh, A.; Goodwin, J.S.; Okoro, E.U.; Guoet, Z. Overexpression of Catalase Enhances Benzo(a)pyrene Detoxification in Endothelial Microsomes. PLoS ONE 2016, 11, e0162561. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.S.; Xiong, Y.; Ma, W.; Spector, A.; Ho, D.S. Mice lacking catalase develop normally but show differential sensitivity to oxidant tissue injury. J. Biol. Chem. 2004, 279, 32804–32812. [Google Scholar] [CrossRef] [PubMed]
- Freeman, B.A.; Turrens, J.F.; Mirza, Z.; Crapo, J.D.; Young, S.L. Modulation of oxidant lung injury by using liposome-entrapped superoxide dismutase and catalase. Fed. Proc. 1985, 44, 2591–2595. [Google Scholar] [PubMed]
- Shetty, S.K.; Tiwari, N.; Marudamuthu, A.S.; Puthusseri, B.; Bhandary, Y.P.; Fu, J.; Levin, J.; Idell, S.; Shetty, S. p53 and miR-34a Feedback Promotes Lung Epithelial Injury and Pulmonary Fibrosis. Am. J. Pathol. 2017, 187, 1016–1034. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Pu, J.; Tang, C.; Wu, Z. Curcumin Inhibits Heat-Induced Apoptosis by Suppressing NADPH Oxidase 2 and Activating the Akt/mTOR Signaling Pathway in Bronchial Epithelial Cells. Cell Physiol. Biochem. 2017, 41, 2091–2103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Xie, Y.; Yan, R.; Shan, H.; Tang, J.; Cai, Y.; Yin, J.; Chen, M.; Zhang, J.; Yang, X.; et al. Curcumin ameliorates alveolar epithelial injury in a rat model of chronic obstructive pulmonary disease. Life Sci. 2016, 164, 1–8. [Google Scholar] [CrossRef]
- Zhu, W.; Cromie, M.M.; Cai, Q.; Lv, T.; Singh, K.; Gaoet, W. Curcumin and vitamin E protect against adverse effects of benzo[a]pyrene in lung epithelial cells. PLoS ONE 2014, 9, e92992. [Google Scholar]
- Liu, G.; Park, Y.J.; Tsuruta, Y.; Lorne, E.; Abraham, E. p53 Attenuates lipopolysaccharide-induced NF-kappaB activation and acute lung injury. J. Immunol. 2009, 182, 5063–5071. [Google Scholar] [CrossRef]
- Stellas, D.; Souliotis, V.L.; Bekyrou, M.; Smirlis, D.; Kirsch-Volders, M.; Degrassi, F.; Cundari, E.; Kyrtopoulos, S.A. Benzo [a] pyrene-induced cell cycle arrest in HepG2 cells is associated with delayed induction of mitotic instability. Mutat. Res. 2014, 769, 59–68. [Google Scholar] [CrossRef]
- Kim, S.M.; Lee, H.M.; Hwang, K.A. Benzo(a)pyrene induced cell cycle arrest and apoptosis in human choriocarcinoma cancer cells through reactive oxygen species-induced endoplasmic reticulum-stress pathway. Food Chem. Toxicol. 2017, 107, 339–348. [Google Scholar] [CrossRef]
- Asweto, C.O.; Wu, J.; Hu, H.; Feng, L.; Yang, X.; Duan, J.; Sunet, Z. Combined effect of silica nanoparticles and benzo[a]pyrene on cell cycle arrest induction and apoptosis in human umbilical vein endothelial cells. Int. J. Environ. Res. Public Health. 2017, 14, 289. [Google Scholar] [CrossRef] [PubMed]
- He, Y.C.; He, L.; Khoshaba, R.; Lu, F.G.; Cai, C.; Zhou, F.L.; Liao, D.F.; Cao, D. Curcumin Nicotinate Selectively Induces Cancer Cell Apoptosis and Cycle Arrest through a P53-Mediated Mechanism. Molecules 2019, 24. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Tao, X.; Fu, Q.; Ge, C.; Li, R.; Li, Z.; Zhu, Y.; Tian, H.; Li, Q.; Liu, M.; et al. Curcumin inhibits cell proliferation and migration in NSCLC through a synergistic effect on the TLR4/MyD88 and EGFR pathways. Oncol. Rep. 2019, 42, 1843–1855. [Google Scholar] [CrossRef] [PubMed]
- Vallianou, N.G.; Evangelopoulos, A.; Schizas, N.; Kazazis, C. Potential Anticancer Properties and Mechanisms of Action of Curcumin. Anticancer Res. 2015, 35, 645–651. [Google Scholar] [PubMed]
- Xu, X.; Chen, D.; Ye, B.; Zhong, F.; Chen, G. Curcumin induces the apoptosis of non-small cell lung cancer cells through a calcium signaling pathway. Int. J. Mol. Med. 2015, 35, 1610–1616. [Google Scholar] [CrossRef]
- Tsuchida, S.; Engelberts, D.; Peltekova, V.; Hopkins, N.; Frndova, H.; Babyn, P.; McKerlie, C.; Post, M.; McLoughlin, P.; Kavanagh, B.P. Atelectasis Causes Alveolar Injury in Nonatelectatic Lung Regions. Am. J. Respir. Crit. Care Med. 2006, 174, 279–289. [Google Scholar] [CrossRef]
- Husain, N.E.; Babiker, A.Y.; Albutti, A.S.; Alsahli, M.A.; Aly, S.M.; Rahmani, A.H. Clinicopathological significance of vimentin and cytokeratin protein in the genesis of squamous cell carcinoma of cervix. Obstet. Gynecol. Int. 2016, 2016, 8790120. [Google Scholar] [CrossRef][Green Version]
- Hayat, M.A. Principles and Techniques of Electron. Microscopy: Biological Applications, 4th ed.; Cambridge University Press: Edinburg, UK, 2000; pp. 37–59. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almatroodi, S.A.; Alrumaihi, F.; Alsahli, M.A.; Alhommrani, M.F.; Khan, A.; Rahmani, A.H. Curcumin, an Active Constituent of Turmeric Spice: Implication in the Prevention of Lung Injury Induced by Benzo(a) Pyrene (BaP) in Rats. Molecules 2020, 25, 724. https://doi.org/10.3390/molecules25030724
Almatroodi SA, Alrumaihi F, Alsahli MA, Alhommrani MF, Khan A, Rahmani AH. Curcumin, an Active Constituent of Turmeric Spice: Implication in the Prevention of Lung Injury Induced by Benzo(a) Pyrene (BaP) in Rats. Molecules. 2020; 25(3):724. https://doi.org/10.3390/molecules25030724
Chicago/Turabian StyleAlmatroodi, Saleh A., Faris Alrumaihi, Mohammed A. Alsahli, Mazen Fahad Alhommrani, Arif Khan, and Arshad Husain Rahmani. 2020. "Curcumin, an Active Constituent of Turmeric Spice: Implication in the Prevention of Lung Injury Induced by Benzo(a) Pyrene (BaP) in Rats" Molecules 25, no. 3: 724. https://doi.org/10.3390/molecules25030724
APA StyleAlmatroodi, S. A., Alrumaihi, F., Alsahli, M. A., Alhommrani, M. F., Khan, A., & Rahmani, A. H. (2020). Curcumin, an Active Constituent of Turmeric Spice: Implication in the Prevention of Lung Injury Induced by Benzo(a) Pyrene (BaP) in Rats. Molecules, 25(3), 724. https://doi.org/10.3390/molecules25030724



