Hydrocarbon Removal by Two Differently Developed Microbial Inoculants and Comparing Their Actions with Biostimulation Treatment
Abstract
1. Introduction
2. Results
2.1. Structure of Hydrocarbon-Degrading Community C1
2.2. Hydrocarbon Removal
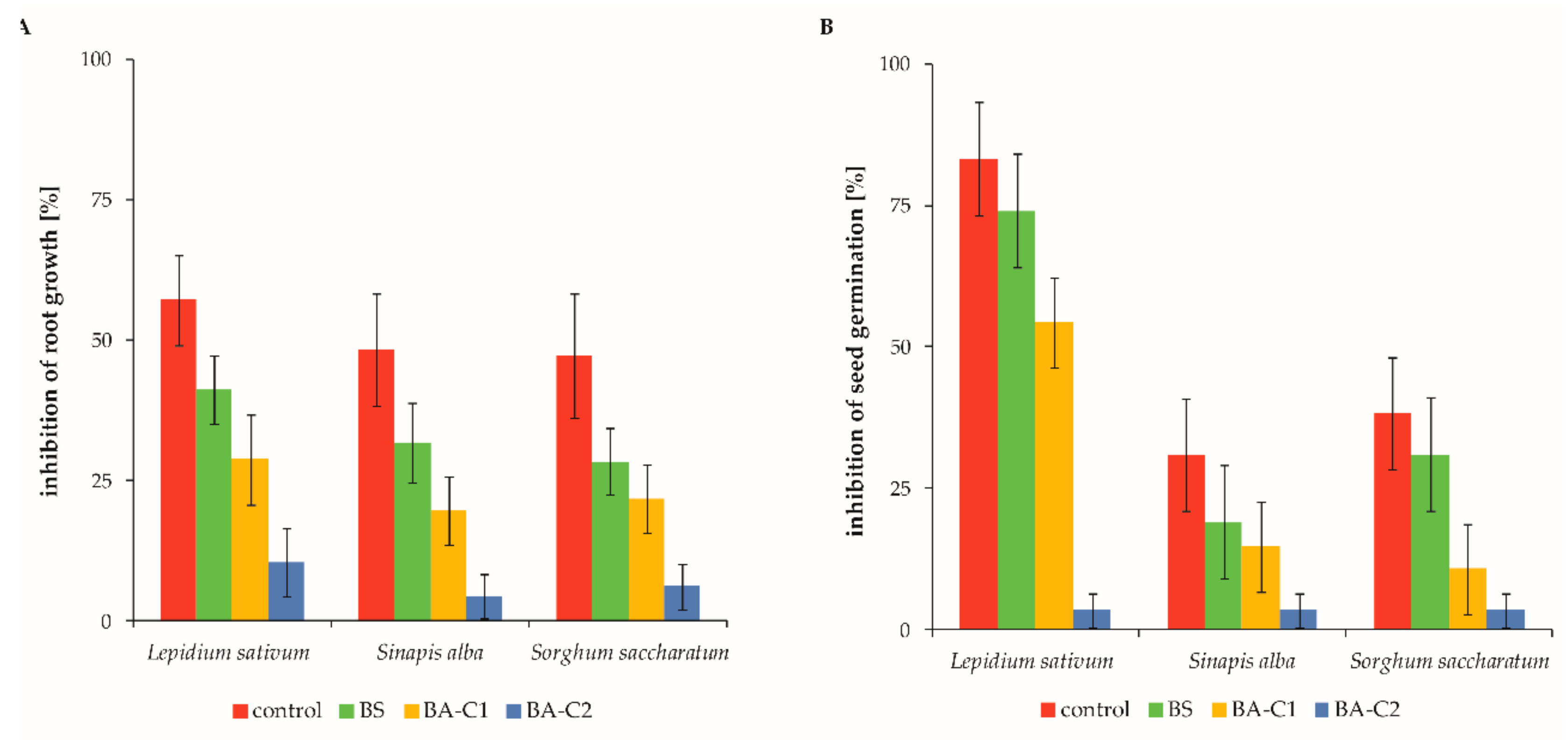
2.3. Toxicity and Mutagenicity Assessment
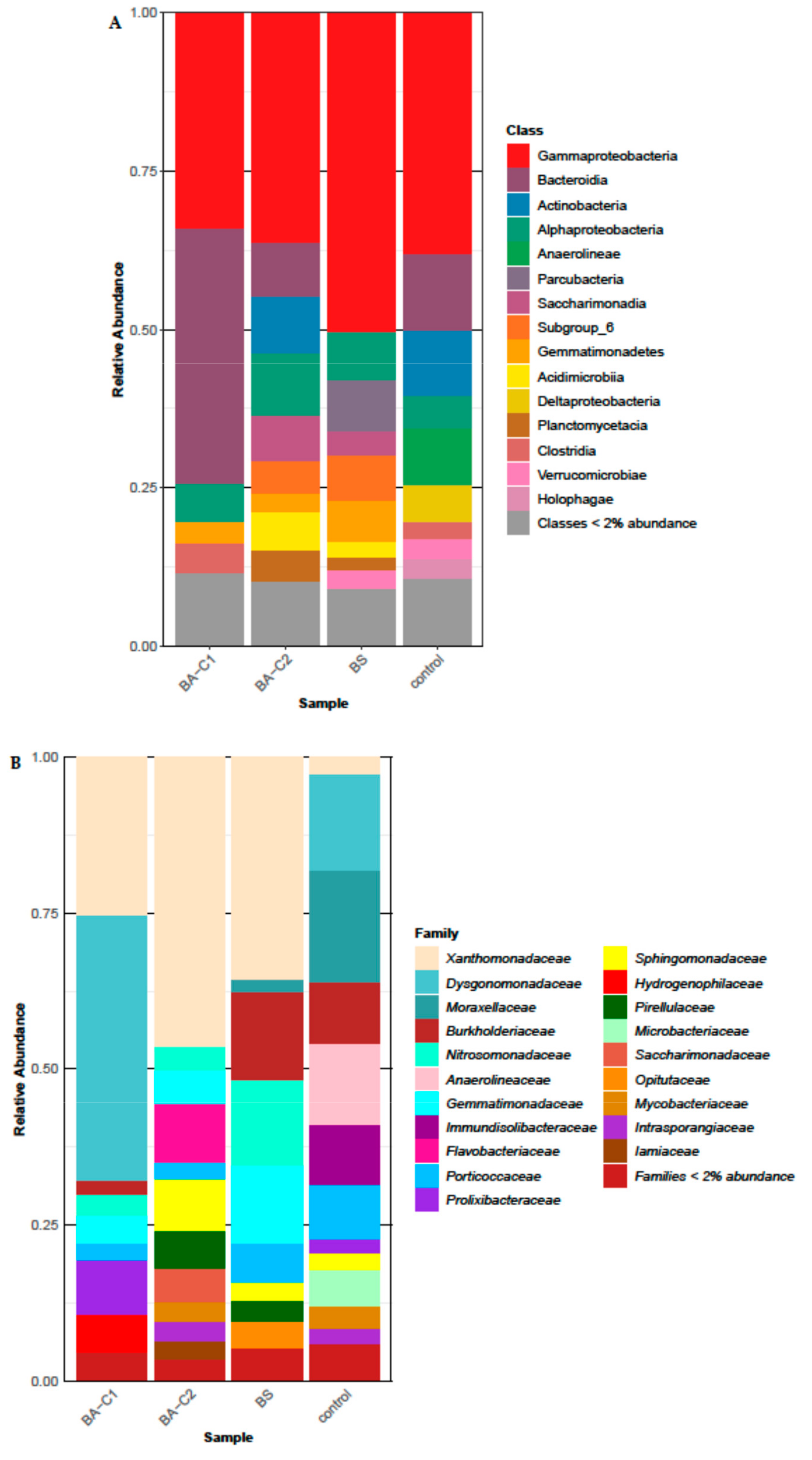
2.4. Soil Community Structure after Treatments
2.5. Gene Expression of Catabolic Genes
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Isolation of Hydrocarbon-Degrading Community C1
4.3. Construction of Mixed Culture C2
4.4. Experimental Design
4.5. DNA Extraction, PCR and Illumina MiSeq-Based Sequencing
4.6. Metatranscriptome Sequencing
4.7. Statistical Analyses
4.8. Data Availability
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Omores, R.A.; Wewers, F.; Ikhide, P.O.; Farrar, T.; Giwa, A. Spatio–temporal distribution of polycyclic aromatic hydrocarbons in urban soils in Cape Town, South Africa. Int. J. Environ. Res. 2017, 11, 189–196. [Google Scholar] [CrossRef]
- Rosell-Melé, A.; Moraleda-Cibrián, N.; Cartró-Sabaté, M.; Colomer-Ventura, F.; Mayor, P.; Orta-Martínez, M. Oil pollution in soils and sediments from the North Peruvian Amazon. Sci. Total Environ. 2018, 610-611, 1010–1019. [Google Scholar] [CrossRef] [PubMed]
- Truskewycz, A.; Gundry, T.D.; Khudur, L.S.; Kolobaric, A.; Taha, M.; Aburto-Medina, A.; Ball, A.S.; Shahsavari, E. Petroleum hydrocarbon contamination in terrestrial ecosystems—fate and microbial responses. Molecules 2019, 24, 3400. [Google Scholar] [CrossRef] [PubMed]
- Escobar, H. Mysterious oil spill threatens marine biodiversity haven in Brazil. Sciencemag.org. Available online: https://www.sciencemag.org/news/2019/11/mysterious-oil-spill-threatens-marine-biodiversity-haven-brazil (accessed on 30 November 2019).
- Juwarkar, A.A.; Singh, S.K.; Mudhoo, A. A comprehensive overview of elements in bioremediation. Rev. Environ. Sci. Biotechnol. 2010, 9, 215–288. [Google Scholar] [CrossRef]
- Siles, J.A.; García-Sánchez, M. Microbial dynamics during the bioremediation of petroleum hydrocarbon-contaminated soils through biostimulation: an overview. In Approaches in Bioremediation. Nanotechnology in the Life Sciences; Prasad, R., Aranda, E., Eds.; Springer: Cham, Switzerland, 2018; pp. 115–134. [Google Scholar]
- Al-Kindi, S.; Abed, R.M.M. Comparing oil degradation efficiency and bacterial communities in contaminated soils subjected to biostimulation using different organic wastes. Water Air Soil Pollut. 2016, 227, 36. [Google Scholar] [CrossRef]
- Silva-Castro, G.A.; Rodriguez-Calvo, A.; Laguna, J.; González-López, J.; Calvo, C. Autochthonous microbial responses and hydrocarbons degradation in polluted soil during biostimulating treatments under different soil moisture. Assay in pilot plant. Int. Biodeter. Biodegr. 2016, 108, 91–98. [Google Scholar] [CrossRef]
- Chang, W.; Akbari, A.; David, C.A.; Ghoshal, S. Selective biostimulation of cold- and salt-tolerant hydrocarbon-degrading Dietzia maris in petroleum-contaminated sub-Arctic soils with high salinity. J. Chem. Technol. Biotechnol. 2018, 93, 294–304. [Google Scholar] [CrossRef]
- Poi, G.; Aburto-Medina, A.; Moke, P.C.; Ball, A.S.; Shahsavari, E. Large scale bioaugmentation of soil contaminated with petroleum hydrocarbons using a mixed microbial consortium. Ecol. Eng. 2017, 102, 64–71. [Google Scholar] [CrossRef]
- Trögl, J.; Esuola, C.O.; Kříženecká, S.; Kuráň, P.; Seidlová, L.; Veronesi-Dáňová, P.; Popelka, J.; Babalola, O.O.; Hrabák, P.; Czinnerová, M.; et al. Biodegradation of high concentrations of aliphatic hydrocarbons in soil from a petroleum refinery: implications for applicability of new actinobacterial strains. Appl. Sci. 2018, 8, 1855. [Google Scholar] [CrossRef]
- Woźniak-Karczewska, M.; Lisiecki, P.; Białas, W.; Owsianiak, M.; Piotrowska-Cyplik, A.; Wolko, Ł.; Ławniczak, Ł.; Heipieper, H.J.; Gutierrez, T.; Chrzanowski, Ł. Effect of bioaugmentation on long-term biodegradation of diesel/biodiesel blends in soil microcosms. Sci. Total Environ. 2019, 671, 948–958. [Google Scholar] [CrossRef]
- Brzeszcz, J.; Kaszycki, P. Aerobic bacteria degrading both n-alkanes and aromatic hydrocarbons: an undervalued strategy for metabolic diversity and flexibility. Biodegradation 2018, 29, 359–407. [Google Scholar] [CrossRef] [PubMed]
- Dombrowski, N.; Donaho, J.A.; Gutierrez, T.; Seitz, K.W.; Teske, A.P.; Baker, B.J. Reconstructing metabolic pathways of hydrocarbon-degrading bacteria from the Deepwater Horizon oil spill. Nat. Microbiol. 2016, 1, 16057. [Google Scholar] [CrossRef] [PubMed]
- Kurm, V.; van der Putten, W.H.; Weidner, S.; Geisen, S.; Snoek, B.L.; Bakx, T.; Gera Hol, W.H. Competition and predation as possible causes of bacterial rarity. Environ. Microbiol. 2019, 21, 1356–1368. [Google Scholar] [CrossRef] [PubMed]
- Steliga, T.; Kapusta, P.; Jakubowicz, P. Effectiveness of bioremediation processes of hydrocarbon pollutants in weathered drill wastes. Water Air Soil Pollut. 2009, 202, 211–228. [Google Scholar] [CrossRef]
- Steliga, T.; Jakubowicz, P.; Kapusta, P. Changes in toxicity during in situ bioremediation of weathered drill wastes contaminated with petroleum hydrocarbons. Bioresour. Technol. 2012, 125, 1–10. [Google Scholar] [CrossRef]
- Ali, N.; Dashti, N.; Salamah, S.; Al-Awadhi, H.; Sorkhoh, N.; Radwan, S. Autochthonous bioaugmentation with environmental samples rich in hydrocarbonoclastic bacteria for bench-scale bioremediation of oily seawater and desert soil. Environ. Sci. Pollut. Res. 2016, 23, 8686–8698. [Google Scholar] [CrossRef]
- Jacques, R.J.S.; Okeke, B.C.; Bento, F.M.; Teixeira, A.S.; Peralba, M.C.R.; Camargo, F.A.O. Microbial consortium bioaugmentation of a polycyclic aromatic hydrocarbons contaminated soil. Bioresour. Technol. 2008, 99, 2637–2643. [Google Scholar] [CrossRef]
- Sprocati, A.R.; Alisi, C.; Tasso, F.; Marconi, P.; Sciullo, A.; Pinto, V.; Chiavarini, S.; Ubaldi, C.; Cremisini, C. Effectiveness of a microbial formula, as a bioaugmentation agent, tailored for bioremediation of diesel oil and heavy metal co-contaminated soil. Process. Biochem. 2012, 47, 1649–1655. [Google Scholar] [CrossRef]
- Rivelli, W.; Franzetti, A.; Gandolfi, I.; Cordoni, S.; Bestetti, G. Persistence and degrading activity of free and immobilised allochthonous bacteria during bioremediation of hydrocarbon-contaminated soils. Biodegradation 2013, 24, 1–11. [Google Scholar] [CrossRef]
- Fodelianakis, S.; Antoniou, E.; Mapelli, F.; Magagnini, M.; Nikolopoulou, M.; Marasco, R.; Barbato, M.; Tsiola, A.; Tsikopoulou, I.; Giaccaglia, L.; et al. Allochthonous bioaugmentation in ex situ treatment of crude oil-polluted sediments in the presence of an effective degrading indigenous microbiome. J. Hazard. Mater. 2015, 287, 78–86. [Google Scholar] [CrossRef]
- Pacwa-Płociniczak, M.; Płaza, G.A.; Piotrowska-Seget, Z. Monitoring the changes in a bacterial community in petroleum-polluted soil bioaugmented with hydrocarbon-degrading strains. Appl. Soil Ecol. 2016, 105, 76–85. [Google Scholar] [CrossRef]
- Szczepaniak, Z.; Czarny, J.; Staninska-Pięta, J.; Lisiecki, P.; Zgoła-Grześkowiak, A.; Cyplik, P.; Chrzanowski, Ł.; Wolko, Ł.; Marecik, R.; Juzwa, W.; et al. Influence of soil contamination with PAH on microbial community dynamics and expression level of genes responsible for biodegradation of PAH and production of rhamnolipids. Environ. Sci. Pollut. Res. 2016, 23, 23043–23056. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Wu, J.; Zhang, X.; Ye, X. Effect of bioaugmentation and biostimulation on hydrocarbon degradation and microbial community composition in petroleum-contaminated loessal soil. Chemosphere 2018, 236, 124456. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.J.; Lee, E.Y.; Kim, Y.J.; Cho, K.-S.; Ryu, H.W. Degradation of polyaromatic hydrocarbons by Burkholderia cepacia 2A-12. World J. Microbiol. Biotechnol. 2003, 19, 411–417. [Google Scholar] [CrossRef]
- Agnello, A.C.; Bagard, M.; van Hullebusch, E.D.; Esposito, G.; Huguenot, D. Comparative bioremediation of heavy metals and petroleum hydrocarbons co-contaminated soil by natural attenuation, phytoremediation, bioaugmentation and bioaugmentation-assisted phytoremediation. Sci. Total Environ. 2016, 563, 693–703. [Google Scholar] [CrossRef]
- Varjani, S.J.; Upasan, V.N. Biodegradation of petroleum hydrocarbons by oleophilic strain of Pseudomonas aeruginosa NCIM 5514. Bioresour. Technol. 2016, 222, 195–201. [Google Scholar] [CrossRef]
- Keswani, C.; Prakash, O.; Bharti, N.; Vílchez, J.I.; Sansinenea, E.; Lally, R.D.; Borriss, R.; Singh, S.P.; Gupta, V.K.; Fraceto, L.F.; et al. Re-addressing the biosafety issues of plant growth promoting rhizobacteria. Sci. Total Environ. 2019, 690, 841–852. [Google Scholar] [CrossRef]
- Bacosa, H.P.; Suto, K.; Inoue, C. Bacterial community dynamics during the preferential degradation of aromatic hydrocarbons by a microbial consortium. Int. Biodeter. Biodegr. 2012, 74, 109–115. [Google Scholar] [CrossRef]
- Masy, T.; Demanèche, S.; Tromme, O.; Thonart, P.; Jacques, P.; Hiligsmann, S.; Vogel, T.M. Hydrocarbon biostimulation and bioaugmentation in organic carbon and clay-rich soils. Soil. Biol. Biochem. 2016, 99, 66–74. [Google Scholar] [CrossRef]
- Brzeszcz, J.; Steliga, T.; Kapusta, P.; Turkiewicz, A.; Kaszycki, P. R-strategist versus K-strategist for the application in bioremediation of hydrocarbon-contaminated soils. Int. Biodeter. Biodegr. 2016, 106, 41–52. [Google Scholar] [CrossRef]
- Colombo, M.; Cavalca, L.; Bernasconi, S.; Andreoni, V. Bioremediation of polyaromatic hydrocarbon contaminated soils by native microflora and bioaugmentation with Sphingobium chlorophenolicum strain C3R: A feasibility study in solid- and slurry-phase microcosms. Int. Biodeter. Biodegr. 2011, 65, 191–197. [Google Scholar] [CrossRef]
- Innemanová, P.; Filipová, A.; Michalíková, K.; Wimmerová, L.; Cajthaml, T. Bioaugmentation of PAH-contaminated soils: A novel procedure for introduction of bacterial degraders into contaminated soil. Ecol. Eng. 2018, 118, 93–96. [Google Scholar] [CrossRef]
- Montenegro, I.P.F.M.; Mucha, A.P.; Reis, I.; Rodrigues, P.; Almeida, C.M.R. Effect of petroleum hydrocarbons in copper phytoremediation by a salt marsh plant (Juncus maritimus) and the role of autochthonous bioaugmentation. Environ. Sci. Pollut. Res. 2016, 23, 19471–19480. [Google Scholar] [CrossRef] [PubMed]
- Roy, A.; Dutta, A.; Pal, S.; Gupta, A.; Sarkar, J.; Chatterjee, A.; Saha, A.; Sarkar, P.; Sar, P.; Kazy, S.K. Biostimulation and bioaugmentation of native microbial community accelerated bioremediation of oil refinery sludge. Bioresour. Technol. 2018, 253, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Isaac, P.; Martínez, F.A.; Bourguignon, N.; Sánchez, L.A.; Ferrero, M.A. Improved PAHs removal performance by a defined bacterial consortium of indigenous Pseudomonas and actinobacteria from Patagonia, Argentina. Int. Biodeter. Biodegr. 2015, 101, 23–31. [Google Scholar] [CrossRef]
- Xia, M.; Fu, D.; Chakraborty, R.; Singh, R.P.; Terry, N. Enhanced crude oil depletion by constructed bacterial consortium comprising bioemulsifier producer and petroleum hydrocarbon degraders. Bioresour. Technol. 2019, 282, 456–463. [Google Scholar] [CrossRef]
- Rontani, J.-F.; Bonin, P. Utilization of n-alkyl-substituted cyclohexanes by a marine Alcaligenes. Chemosphere 1992, 24, 1441–1446. [Google Scholar] [CrossRef]
- Sun, L.; Zhu, G.; Liao, X.; Yan, X. Interactions between Pteris vittata L. genotypes and a polycyclic aromatic hydrocarbon (PAH)-degrading bacterium (Alcaligenes sp.) in arsenic uptake and PAH-dissipation. Environ. Pollut. 2017, 230, 862–870. [Google Scholar] [CrossRef]
- Wang, X.; Jin, D.; Zhou, L.; Zhang, Z. Draft genome sequence of aquamicrobium defluvii strain W13Z1, a psychrotolerant halotolerant hydrocarbon-degrading bacterium. Genome Announc. 2015, 3, e00984-15. [Google Scholar] [CrossRef]
- Tumaikina, Y.A.; Turkovskaya, O.V.; Ignatov, V.V. Degradation of hydrocarbons and their derivatives by a microbial association on the base of Canadian pondweed. Appl. Microbiol. Biotechnol. 2008, 44, 382–388. [Google Scholar] [CrossRef]
- Wang, X.; Wang, X.; Liu, M.; Zhou, L.; Gu, Z.; Zhao, J. Bioremediation of marine oil pollution by Brevundimonas diminuta: effect of salinity and nutrients. Desalin. Water Treat. 2016, 57, 19768–19775. [Google Scholar] [CrossRef]
- Sun, J.-Q.; Xu, L.; Zhang, Z.; Li, Y.; Tang, Y.-Q.; Wu, X.-L. Diverse bacteria isolated from microtherm oil-production water. Antonie van Leeuwenhoek 2014, 105, 401–411. [Google Scholar] [CrossRef]
- Schwarz, A.; Adetutu, E.M.; Juhasz, A.L.; Aburto-Medina, A.; Ball, A.S.; Shahsavari, S. Microbial degradation of phenanthrene in pristine and contaminated sandy soils. Microb. Ecol. 2018, 75, 888–902. [Google Scholar] [CrossRef]
- Blanco-Enríquez, E.G.; Zavala-Díaz de la Serna, F.J.; Peralta-Pérez, M.D.R.; Ballinas-Casarrubias, L.; Salmerón, I.; Rubio-Arias, H.; Rocha-Gutiérrez, B.A. Characterization of a Microbial Consortium for the Bioremoval of Polycyclic Aromatic Hydrocarbons (PAHs) in Water. Int. J. Environ. Res. Public Health 2018, 15, 975. [Google Scholar] [CrossRef]
- Garrido-Sanz, D.; Redondo-Nieto, M.; Guirado, M.; Jiménez, O.P.; Millán, R.; Martin, M.; Rivilla, R. Metagenomic insights into the bacterial functions of a diesel-degrading consortium for the rhizoremediation of diesel-polluted soil. Genes 2019, 10, 456. [Google Scholar] [CrossRef]
- Ryan, M.P.; Pembroke, J.T. Brevundimonas spp: Emerging global opportunistic pathogens. Virulence 2017, 9, 480–493. [Google Scholar] [CrossRef]
- Guzman Prieto, A.M.; van Schaik, W.; Rogers, M.R.C.; Coque, T.M.; Baquero, F.; Corander, J.; Willems, R.J.L. Global emergence and dissemination of enterococci as nosocomial pathogens: attack of the clones? Front. Microbiol. 2016, 7, 788. [Google Scholar] [CrossRef]
- Larsen, S.B.; Karakashev, D.; Angelidaki, I.; Schmidt, J.E. Ex-situ bioremediation of polycyclic aromatic hydrocarbons in sewage sludge. J. Hazard. Mater. 2009, 164, 1568–1572. [Google Scholar] [CrossRef]
- Hamadi, H.; Benzarti, S.; Manisadžianas, L.; Aoyama, I.; Jedidi, M. Bioaugmentation and biostimulation effects on PAH dissipation and soil ecotoxicity under controlled conditions. Soil Biol. Biochem. 2007, 39, 1926–1935. [Google Scholar] [CrossRef]
- Chibwe, L.; Geier, M.C.; Nakamura, J.; Tanguay, R.L.; Aitken, M.D.; Simonich, S.L.M. Aerobic bioremediation of PAH contaminated soils results in increased genotoxicity and developmental toxicity. Environ. Sci. Technol. 2015, 49, 13889–13898. [Google Scholar] [CrossRef]
- Tian, Z.; Gold, A.; Nakamura, J.; Zhang, Z.; Vila, J.; Singleton, D.; Collins, L.D.; Aitken, M.D. Non-target analysis reveals a bacterial metabolite of pyrene implicated in the genotoxicity of contaminated soil after bioremediation. Environ. Sci. Technol. 2017, 51, 7091–7100. [Google Scholar] [CrossRef] [PubMed]
- Lundstedt, S.; White, P.A.; Lemieux, C.L.; Lynes, K.D.; Lambert, I.B.; Öberg, L.; Haglund, P.; Tysklind, M. Sources, fate, and toxic hazards of oxygenated polycyclic aromatic hydrocarbons (PAHs) at PAH- contaminated sites. Ambio 2007, 36, 475–485. [Google Scholar] [CrossRef]
- Moody, J.D.; Freeman, J.P.; Doerge, D.R.; Cerniglia, C.E. Degradation of phenanthrene and anthracene by cell suspensions of Mycobacterium sp. strain PYR-1. Appl. Environ. Microbiol. 2001, 67, 1476–1483. [Google Scholar] [CrossRef] [PubMed]
- Hennessee, C.T.; Li, Q.X. Effects of polycyclic aromatic hydrocarbon mixtures on degradation, gene expression, and metabolite production in four Mycobacterium species. Appl. Environ. Microbiol. 2016, 82, 3357–3369. [Google Scholar] [CrossRef] [PubMed]
- Biache, C.; Ouali, S.; Cébron, A.; Lorgeoux, C.; Colombano, S.; Faure, P. Bioremediation of PAH-contaminated soils: consequences on formation and degradation of polar-polycyclic aromatic compounds and microbial community abundance. J. Hazard. Mater. 2017, 329, 1–10. [Google Scholar] [CrossRef]
- Płaza, G.A.; Jangid, K.; Łukasik, K.; Nałęcz-Jawecki, G.; Berry, C.J.; Brigmon, R.L. Reduction of petroleum hydrocarbons and toxicity in refinery wastewater by bioremediation. Bull. Environ. Contam. Toxicol. 2008, 81, 329–333. [Google Scholar] [CrossRef]
- Li, J.; Guo, C.; Lu, G.; Yi, X.; Dang, Z. Bioremediation of petroleum-contaminated acid soil by a constructed bacterial consortium immobilized on sawdust: influences of multiple factors. Water Air Soil Pollut. 2016, 227, 444. [Google Scholar] [CrossRef]
- Farber, R.; Rosenberg, A.; Rozenfeld, S.; Banet, G.; Cahan, R. Bioremediation of artificial diesel-contaminated soil using bacterial consortium immobilized to plasma-pretreated wood waste. Microorganisms 2019, 7, 497. [Google Scholar] [CrossRef]
- Lang, F.S.; Destain, J.; Delvigne, F.; Druart, P.; Ongena, M.; Thonart, P. Biodegradation of polycyclic aromatic hydrocarbons in mangrove sediments under different strategies: natural attenuation, biostimulation, and bioaugmentation with Rhodococcus erythropolis T902.1. Water Air Soil Pollut. 2016, 227, 297. [Google Scholar] [CrossRef]
- Pacwa-Płociniczak, M.; Czapla, J.; Płociniczak, T.; Piotrowska-Seget, Z. The effect of bioaugmentation of petroleum-contaminated soil with Rhodococcus erythropolis strains on removal of petroleum from soil. Ecotoxicol. Environ. Saf. 2019, 169, 615–622. [Google Scholar] [CrossRef]
- Kaszycki, P.; Petryszak, P.; Pawlik, M.; Kołoczek, H. Ex-situ bioremediation of soil polluted with oily waste: the use of specialized microbial consortia for process bioaugmentation. Ecol. Chem. Eng. S 2011, 18, 83–92. [Google Scholar]
- Taccari, M.; Milanovic, V.; Comtini, F.; Casucci, C.; Ciani, M. Effects of biostimulation and bioaugmentation on diesel removal and bacterial community. Int. Biodeter. Biodegr. 2012, 66, 39–46. [Google Scholar] [CrossRef]
- Bento, F.M.; Camargo, F.A.O.; Okeke, B.C.; Frankenberger, W.T. Comparative bioremediation of soils contaminated with diesel oil by natural attenuation, biostimulation and bioaugmentation. Bioresour. Technol. 2005, 96, 1049–1055. [Google Scholar] [CrossRef] [PubMed]
- Gallego, J.L.R.; Gracía-Martínez, M.J.; Llamas, J.F.; Belloch, C.; Peláez, A.I.; Sánchez, J. Biodegradation of oil tank bottom sludge using microbial consortia. Biodegradation 2007, 18, 269–281. [Google Scholar] [CrossRef] [PubMed]
- Festa, S.; Coppotelli, B.M.; Madueño, L.; Loviso, C.L.; Macchi, M.; Neme Tauil, R.M.; Valacco, M.P.; Morelli, I.S. Assigning ecological roles to the populations belonging to a phenanthrene-degrading bacterial consortium using omic approaches. PLoS ONE 2017, 12, e0184505. [Google Scholar] [CrossRef]
- Sydow, M.; Owsianiak, M.; Szczepaniak, Z.; Framski, G.; Smets, B.F.; Ławniczak, Ł.; Lisiecki, P.; Szulc, A.; Cyplik, P.; Chrzanowski, Ł. Evaluating robustness of a diesel-degrading bacterial consortium isolated from contaminated soil. New Biotechnol. 2016, 33, 852–859. [Google Scholar] [CrossRef]
- Falkowicz, S.; Cicha-Szot, R.; Nelson, S.; Launt, P.; Rogaliński, M. Microbial assisted waterflood effectively increases production from a mature Carpathian oil field: project results and analysis of economic efficiency at eighty months. Nafta-Gaz 2019, 75, 131–138. [Google Scholar] [CrossRef]
- Yang, R.; Zhang, G.; Li, S.; Moazeni, F.; Li, Y.; Wu, Y.; Zhang, w.; Chen, T.; Liu, G.; Zhang, B.; et al. Degradation of crude oil by mixed cultures of bacteria isolated from the Qinghai-Tibet plateau and comparative analysis of metabolic mechanisms. Environ. Sci. Pollut. Res. 2016, 23, 23043–23056. [Google Scholar] [CrossRef]
- Xu, Y.; Lu, M. Bioremediation of crude oil-contaminated soil: comparison of different biostimulation and bioaugmentation treatments. J. Hazard. Mater. 2010, 183, 395–401. [Google Scholar] [CrossRef]
- Corteselli, E.M.; Aitken, M.D.; Singleton, D.R. Description of Immundisolibacter cernigliae gen. nov., sp. nov., a high-molecular-weight polycyclic aromatic hydrocarbon-degrading bacterium within the class Gammaproteobacteria, and proposal of Immundisolibacterales ord. nov. and Immundisolibacteraceae fam. nov. Int. J. Syst. Evol. Microbiol. 2017, 67, 925–931. [Google Scholar]
- Teramoto, M.; Suzuki, M.; Hatmanti, A.; Harayama, S. The potential of Cycloclasticus and Altererythrobacter strains for use in bioremediation of petroleum-aromatic-contaminated tropical marine environments. J. Biosci. Bioeng. 2010, 110, 48–52. [Google Scholar] [CrossRef]
- Cheng, H.; Wu, Y.-H.; Huo, Y.-Y.; Wang, C.-S.; Xu, X.-W. Draft genome Sequence of Altererythrobacter marensis DSM 21428T, isolated from seawater. Genome Announc. 2016, 4, e01607–e01615. [Google Scholar] [CrossRef]
- Patel, V.; Cheturvedula, S.; Madamwar, D. Phenanthrene degradation by Pseudoxanthomonas sp. DMVP2 isolated from hydrocarbon contaminated sediment of Amlakhadi canal, Gujarat, India. J. Hazard. Mater. 2011, 201–202, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Brassington, K.J.; Prpich, G.; Paton, G.I.; Semple, K.T.; Pollard, S.J.T.; Coulon, F. Insights into the biodegradation of weathered hydrocarbons in contaminated soils by bioaugmentation and nutrient stimulation. Chemosphere 2016, 161, 300–307. [Google Scholar] [CrossRef]
- Ramadass, K.; Megharaj, M.; Venkateswarlu, K.; Naidu, R. Bioavailability of weathered hydrocarbons in engine oil-contaminated soil: Impact of bioaugmentation mediated by Pseudomonas spp. on bioremediation. Sci. Total Environ. 2018, 636, 968–974. [Google Scholar] [CrossRef]
- Gielnik, A.; Pechaud, Y.; Huguenot, D.; Cébron, A.; Riom, J.-M.; Guibaud, G.; Esposito, G.; van Hullebusch, E. Effect of digestate application on microbial respiration and bacterial communities’ diversity during bioremediation of weathered petroleum hydrocarbons contaminated soils. Sci. Total Environ. 2019, 670, 271–281. [Google Scholar] [CrossRef]
- Adetutu, E.M.; Ball, A.S.; Weber, J.; Aleer, S.; Dandie, C.E.; Juhasz, A.L. Impact of bacterial and fungal processes on 14C-hexadecane mineralisation in weathered hydrocarbon contaminated soil. Sci. Total Environ. 2012, 414, 585–591. [Google Scholar] [CrossRef]
- Košnář, Z.; Mercl, F.; Tlustoš, P. Ability of natural attenuation and phytoremediation using maize (Zea mays L.) to decrease soil contents of polycyclic aromatic hydrocarbons (PAHs) derived from biomass fly ash in comparison with PAHs–spiked soil. Ecotoxicol. Environ. Safety 2018, 153, 16–22. [Google Scholar] [CrossRef]
- Gallego, J.R.; Sierra, C.; Villa, R.; Peláez, A.I.; Sánchez, J. Weathering processes only partially limit the potential for bioremediation of hydrocarbon-contaminated soils. Organic Geochemistry 2010, 41, 896–900. [Google Scholar] [CrossRef]
- Wrenn, B.A.; Venosa, A.D. Selective enumeration of aromatic and aliphatic hydrocarbon degrading bacteria by a most-probable-number procedure. Can. J. Microbiol. 1996, 42, 252–258. [Google Scholar] [CrossRef]
- Klindworth, A.; Pruesse, E.; Schweer, T.; Peplies, J.; Quast, C.; Horn, M.; Glöckner, F.O. Evaluation of general 16S ribosomal RNA gene PCR primers for classical and next-generation sequencing-based diversity studies. Nucleic Acid Res. 2013, 41, e1. [Google Scholar] [CrossRef] [PubMed]
- Callahan, B.J.; McMurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Prusse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glöckner, F.O. The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res. 2012, 41, D590–D596. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.S. DECIPHER: Harnessing local sequence context to improve multiple sequence alignment. BMC Bioinformatics 2015, 16, 322. [Google Scholar] [CrossRef]
- Schliep, K.P. phangorn: phylogenetic analysis in R. Bioinformatics 2011, 27, 529–593. [Google Scholar] [CrossRef]
- McMurdie, P.J.; Holmes, S. phyloseq: An R package for reproducible interactive analysis and graphics of microbiome census data. PLoS ONE 2013, 8, e61217. [Google Scholar] [CrossRef]
- Meyer, F.; Paarmann, D.; D’Souza, M.; Olson, R.; Kubal, M.; Paczian, T.; Rodriguez, A.; Stevens, R.; Wilke, A.; Wilkening, J.; et al. The metagenomics RAST server - a public resource for the automatic phylogenetic and functional analysis of metagenomes. BMC Bioinformatics 2008, 9, 386. [Google Scholar] [CrossRef]
- Huson, D.H.; Auch, A.F.; Qi, J.; Schuster, S.C. MEGAN analysis of metagenomic data. Genome Res. 2007, 17, 377–386. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.; Ryan, R. PAST: paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2008, 4, 9–17. [Google Scholar]
Sample Availability: Not available. |


| ASV-ID | Class | Genus | Relative Abundance |
|---|---|---|---|
| ASV15 | Gammaproteobacteria | Alcaligenes | 27% |
| ASV1 | Alphaproteobacteria | Pseudochrobactrum | 22% |
| ASV35 | Alphaproteobacteria | Aquamicrobium | 12% |
| ASV66 | Firmicutes | Enterococcus | 12% |
| ASV47 | Alphaproteobacteria | Brevundimonas | 9% |
| ASV366 | Firmicutes | Alkaliphilus | 4% |
| ASV475 | Firmicutes | Clostridium sensu stricto 16 | 2% |
| ASV516 | Firmicutes | Clostridioides | 2% |
| ASV673 | Firmicutes | Melissococcus | 1% |
| ASV622 | Actinobacteria | Leucobacter | 1% |
| Hydrocarbons | Content ± SD (mg/kg d.w. soil) | ||||
|---|---|---|---|---|---|
| Initial (0 Day) | After 60 Days | ||||
| Control | BS | BA-C1 | BA-C2 | ||
| Total aliphatic hydrocarbons (TAHs) | 17757.2 ± 1175.2 AB | 17467.2 ± 1049.5 CD | 11372.0 ± 699.8 E | 5290.9 ± 297.7 ACF | 2310.3 ± 126.2 BDEF |
| Unidentified aliphatic hydrocarbons | 5496.6 ± 304.5 | 5512.6 ± 286.0 | 3779.2 ± 245.4 | 1647.5 ± 108.2 | 707.3 ± 49.9 |
| ∑nC8–nC22 | 9324.1 ± 655.3 GH | 8983.8 ± 564.1 IJ | 5274.2 ± 316.9 K | 2254.5 ± 109.6 GIL | 861.2 ± 39.3 HJKL |
| ∑nC23–nC35 | 2936.5 ± 215.4 M | 2917.0 ± 199.4 N | 2283.7 ± 137.5 O | 1388.9 ± 79.9 | 741.8 ± 37.0 MNO |
| ∑PAHs | 2777.5 ± 211.2 a | 2785.6 ± 162.4 b | 2120.8 ± 118.6 c | 988.7 ± 54.3 | 411.6 ± 21.9 abc |
| Unidentified PAHs | 455.8 ± 32.7 | 483.2 ± 24.1 | 387.2 ± 19.8 | 229.4 ± 11.9 | 99.7 ± 4.5 |
| ∑Two- and three-ring PAHs | 1459.5 ± 207.6 de | 1413.2 ± 182.9 fg | 1000.0 ± 129.6 h | 329.6 ± 41.3 df | 112.0 ± 14.7 egh |
| ∑Four- and five- ring PAHs | 829.1 ± 79.0 i | 856.4 ± 86.5 j | 701.95 ± 62.0 k | 401.7 ± 26.6 | 177.4 ± 6.6 ijk |
| ∑Six-ring PAHs | 32.2 ± 3.9 | 32.8 ± 3.3 | 31.7 ± 3.3 | 28.1 ± 2.5 | 22.5 ± 1.4 |
| Microcosms | nC17/Pristane | nC18/Phytane |
|---|---|---|
| Control | 9.0 ± 0.6 | 8.2 ± 0.8 |
| BS | 5.2 ± 0.4 | 4.5 ± 0.5 |
| BA-C1 | 2.0 ± 0.2 | 1.6 ± 0.2 |
| BA-C2 | 0.7 ± 0.05 | 0.5 ± 0.05 |
| Microcosms | Ostracodtoxkit Test | Microtox Solid Phase Test | Ames Test | |
|---|---|---|---|---|
| Mortality (%) (Chronic Toxicity) | Growth Inhibition (%) (Chronic Toxicity) | Toxicity (TU) | Mutagenicity Ratio | |
| Control | 54.6 ± 5,1 | No data | 28.7 ± 2.9 | 14.2 |
| BS | 40.3 ± 3.2 | 44.5 ± 4.3 | 22.9 ± 2.2 | 10.5 |
| BA-C1 | 33.1 ± 3.2 | 36.5 ± 3.5 | 13.2 ± 1.3 | 7.2 |
| BA-C2 | 17.0 ± 0.9 | 20.4 ± 1.3 | 1.2 ± 0.2 | 1.2 |
| Indices | Control | BS | BA-C1 | BA-C2 |
|---|---|---|---|---|
| Observed richness | 1676 | 1335 | 1190 | 1279 |
| Shannon index | 5.08 | 4.82 | 4.73 | 5.55 |
| Simpson | 0.98 | 0.97 | 0.96 | 0.99 |
| Chao-1 | 2111.52 | 1608.19 | 1379.15 | 1481.41 |
| ACE | 2163.65 | 1605.98 | 1356.98 | 1439.74 |
| Strain | NCBI Accession Number | nC18H38 | iso-C19H40 | TOL, XYL | NAP | ANT | PHEN | FLU | FLUO | PYR |
|---|---|---|---|---|---|---|---|---|---|---|
| Rhodococcus erythropolis IN119 | KT923331 | + | + | + | + | − | − | − | − | − |
| Mycolicibacterium frederiksbergense IN53 | JN572675 | + | + | − | + | + | + | − | − | + |
| Dietzia sp. IN133 | KT923300 | + | + | + | + | − | − | − | − | − |
| Pseudomonas sp. IN132 | KT923299 | + | + | + | + | +/− | + | +/− | +/− | +/− |
| Arthrobacter sp. IN212 | KT923314 | + | + | + | +/− | +/− | +/− | − | − | − |
| Rhodococcus sp. IN136 | KT923330 | + | + | − | + | − | − | − | + | − |
| Gordonia sp. IN138 | KT923297 | + | + | + | + | +/− | − | − | − | − |
| Treatment | Treatment Details | Purpose |
|---|---|---|
| abiotic control | HgCl2-treated soil (sterilized soil) | abiotic control |
| control | soil | control (natural attenuation) |
| BS | soil + inorganic N, P | biostimulation |
| BA-C1 | soil + inorganic N, P + consortium C1 | biostimulation and bioaugmentation |
| BA-C2 | soil + inorganic N, P + mixed culture C2 | biostimulation and bioaugmentation |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brzeszcz, J.; Kapusta, P.; Steliga, T.; Turkiewicz, A. Hydrocarbon Removal by Two Differently Developed Microbial Inoculants and Comparing Their Actions with Biostimulation Treatment. Molecules 2020, 25, 661. https://doi.org/10.3390/molecules25030661
Brzeszcz J, Kapusta P, Steliga T, Turkiewicz A. Hydrocarbon Removal by Two Differently Developed Microbial Inoculants and Comparing Their Actions with Biostimulation Treatment. Molecules. 2020; 25(3):661. https://doi.org/10.3390/molecules25030661
Chicago/Turabian StyleBrzeszcz, Joanna, Piotr Kapusta, Teresa Steliga, and Anna Turkiewicz. 2020. "Hydrocarbon Removal by Two Differently Developed Microbial Inoculants and Comparing Their Actions with Biostimulation Treatment" Molecules 25, no. 3: 661. https://doi.org/10.3390/molecules25030661
APA StyleBrzeszcz, J., Kapusta, P., Steliga, T., & Turkiewicz, A. (2020). Hydrocarbon Removal by Two Differently Developed Microbial Inoculants and Comparing Their Actions with Biostimulation Treatment. Molecules, 25(3), 661. https://doi.org/10.3390/molecules25030661






