The Middle Lamella of Plant Fibers Used as Composite Reinforcement: Investigation by Atomic Force Microscopy
Abstract
1. Introduction
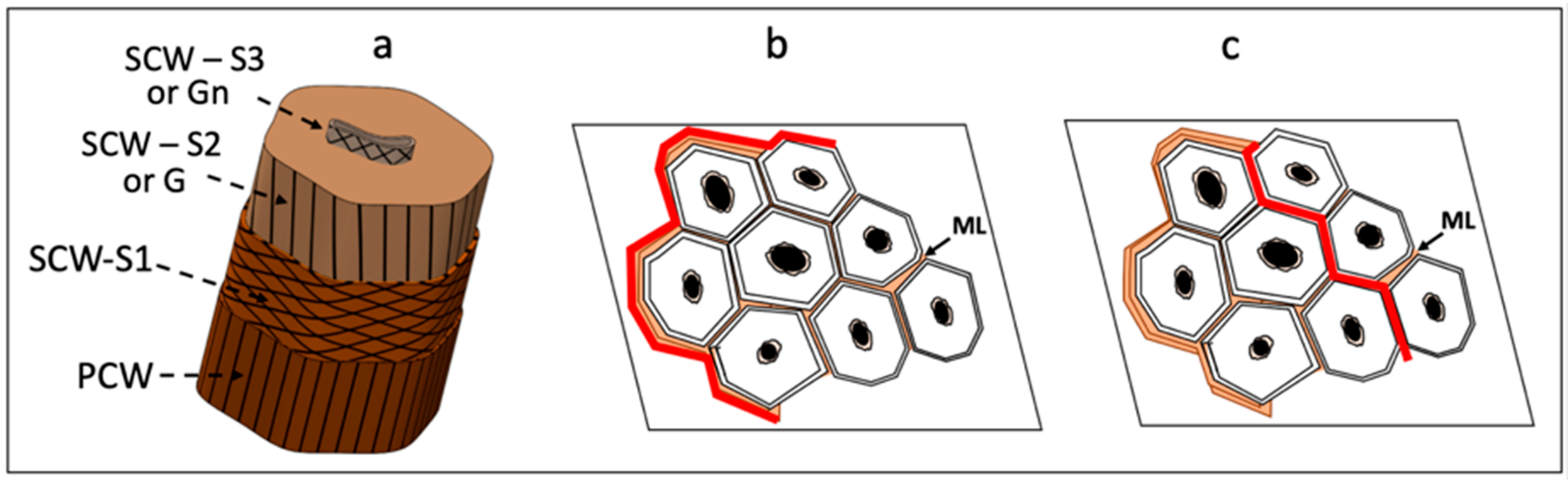
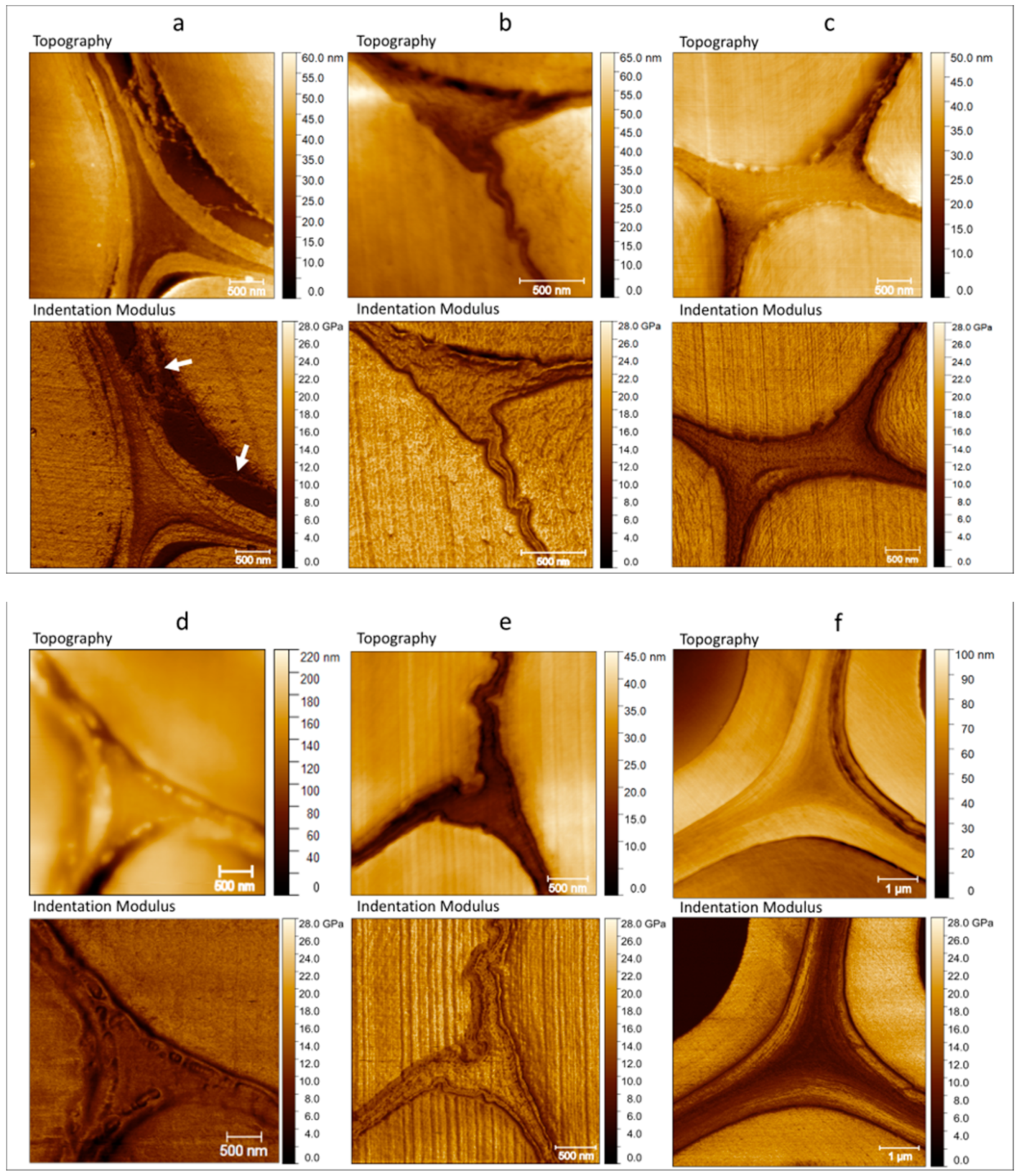
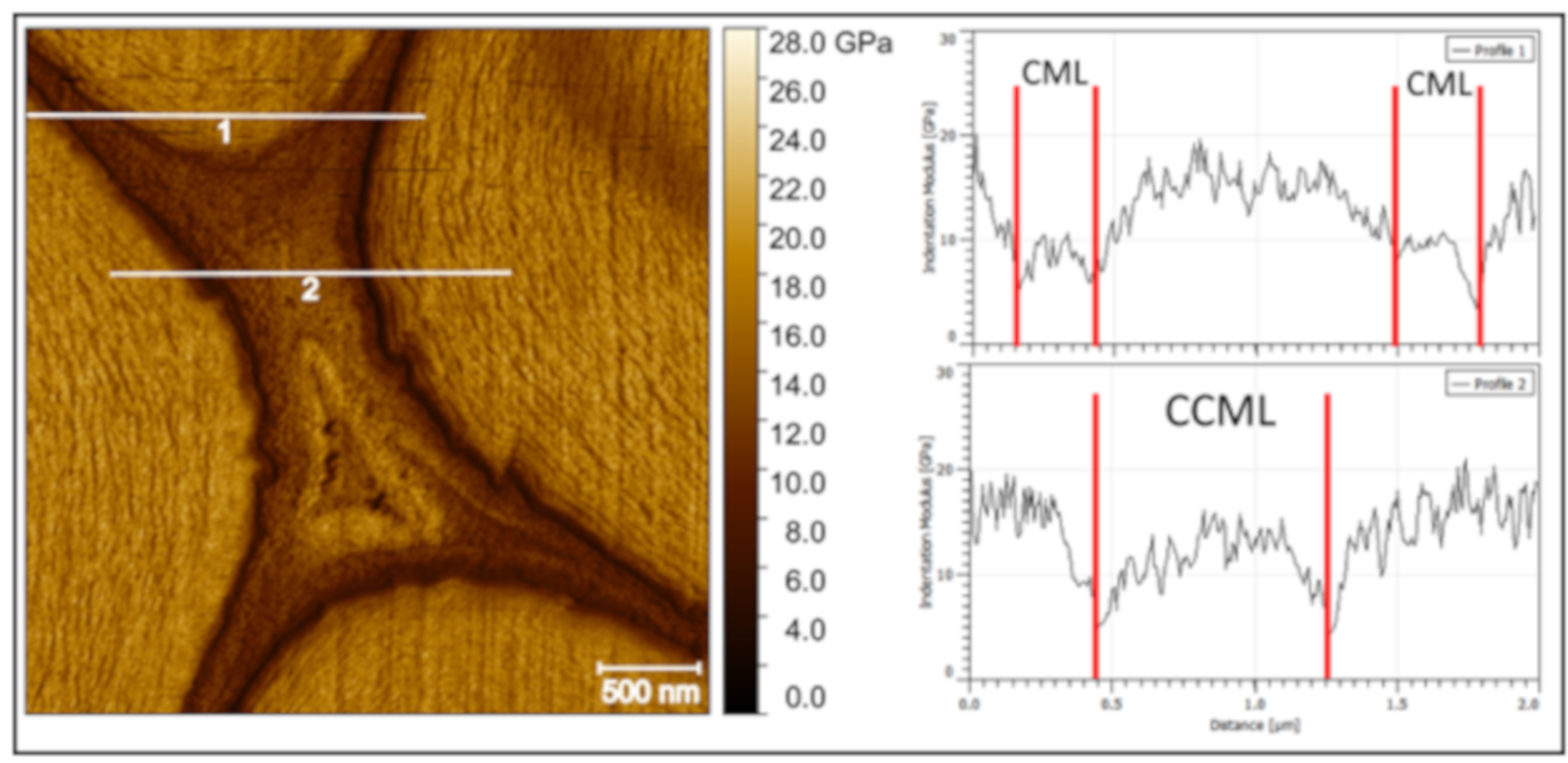
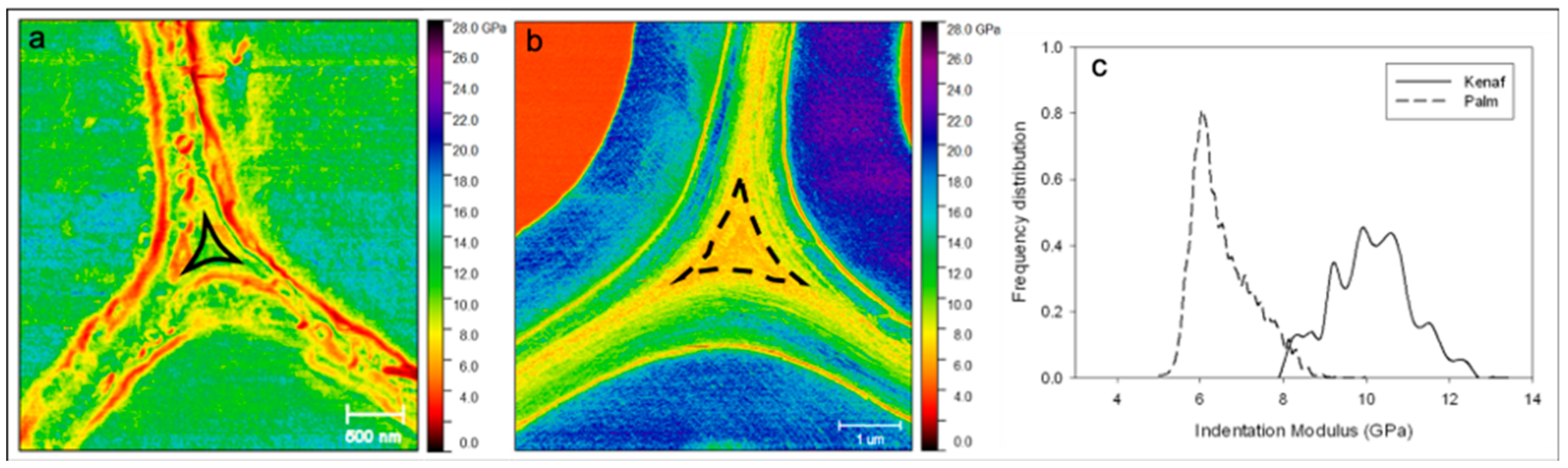
2. Results and Discussion
3. Materials and Methods
3.1. Materials
3.2. Methods
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A

References
- Merotte, J.; Le Duigou, A.; Bourmaud, A.; Behlouli, K.; Baley, C. Mechanical and acoustic behaviour of porosity controlled randomly dispersed flax/PP biocomposite. Polym. Test. 2016, 51, 174–180. [Google Scholar] [CrossRef]
- Pil, L.; Bensadoun, F.; Pariset, J.; Verpoest, I. Why are designers fascinated by flax and hemp fibre composites? Compos. Part A Appl. Sci. Manuf. 2015, 83, 193–205. [Google Scholar] [CrossRef]
- Miao, M.; Finn, N. Conversion of natural fibres into structural composites. J. Text. Eng. 2008, 54, 165–177. [Google Scholar] [CrossRef]
- Kicińska-Jakubowska, A.; Bogacz, E.; Zimniewska, M. Review of natural fibers. Part I—Vegetable fibers. J. Nat. Fibers 2012, 9, 150–167. [Google Scholar] [CrossRef]
- Duval, A.; Bourmaud, A.; Augier, L.; Baley, C. Influence of the sampling area of the stem on the mechanical properties of hemp fibers. Mater. Lett. 2011, 65, 797–800. [Google Scholar] [CrossRef]
- Ansell, M.P.; Mwaikambo, L.Y. 2 – The structure of cotton and other plant fibres. In Handbook of Textile Fibre Structure: Natural, Regenerated, Inorganic and Specialist Fibres; Woodhead Publishing Limited: Cambridge, UK, 2009; Volume 2, pp. 62–94. ISBN 9781845697303. [Google Scholar]
- Bourmaud, A.; Beaugrand, J.; Shah, D.U.; Placet, V.; Baley, C. Towards the design of high-performance plant fibre composites. Prog. Mater. Sci. 2018, 97, 347–408. [Google Scholar] [CrossRef]
- Wambua, P.; Ivens, J.; Verpoest, I. Natural fibres: can they replace glass in fibre reinforced plastics? Compos. Sci. Technol. 2003, 63, 1259–1264. [Google Scholar] [CrossRef]
- Graupner, N.; Labonte, D.; Humburg, H.; Buzkan, T.; Dörgens, A.; Kelterer, W.; Müssig, J. Functional gradients in the pericarp of the green coconut inspire asymmetric fibre-composites with improved impact strength, and preserved flexural and tensile properties. Bioinspir. Biomim. 2017, 12, 026009. [Google Scholar] [CrossRef]
- Rihouey, C.; Paynel, F.; Gorshkova, T.; Morvan, C. Flax fibers: assessing the non-cellulosic polysaccharides and an approach to supramolecular design of the cell wall. Cellulose 2017, 24, 1985–2001. [Google Scholar] [CrossRef]
- Rogers, K. The Cell; Britannica Educational Publications (in Association with Rosen Educational Services): New York, NY, USA, 2011; ISBN 978-1-61530-386-1. [Google Scholar]
- Lazić, B.D.; Pejić, B.M.; Kramar, A.D.; Vukčević, M.M.; Mihajlovski, K.R.; Rusmirović, J.D.; Kostić, M.M. Influence of hemicelluloses and lignin content on structure and sorption properties of flax fibers (Linum usitatissimum L.). Cellulose 2018, 25, 697–709. [Google Scholar] [CrossRef]
- Zamil, M.S.; Geitmann, A. The middle lamella—more than a glue. Phys. Biol. 2017, 14, 015004. [Google Scholar] [CrossRef] [PubMed]
- Sfiligoj Smole, M.; Hribernik, S.; Stana Kleinschek, K.; Kreže, T. Plant Fibres for Textile and Technical Applications. In Advances in Agrophysical Research; Grundas, P.S., Ed.; IntechOpen Limited: London, UK, 2013; pp. 369–398. [Google Scholar]
- Raghavan, R.; Adusumalli, R.-B.; Buerki, G.; Hansen, S.; Zimmermann, T.; Michler, J. Deformation of the compound middle lamella in spruce latewood by micro-pillar compression of double cell walls. J. Mater. Sci. 2012, 47, 6125–6130. [Google Scholar] [CrossRef]
- Bourmaud, A.; Siniscalco, D.; Foucat, L.; Goudenhooft, C.; Falourd, X.; Pontoire, B.; Arnould, O.; Beaugrand, J.; Baley, C. Evolution of flax cell wall ultrastructure and mechanical properties during the retting step. Carbohydr. Polym. 2019, 206, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Siniscalco, D.; Arnould, O.; Bourmaud, A.; Le Duigou, A.; Baley, C. Monitoring temperature effects on flax cell-wall mechanical properties within a composite material using AFM. Polym. Test. 2018, 69, 91–99. [Google Scholar] [CrossRef]
- Doumbia, A.S.A.S.; Castro, M.; Jouannet, D.; Kervoëlen, A.; Falher, T.; Cauret, L.; Bourmaud, A. Flax/polypropylene composites for lightened structures: Multiscale analysis of process and fibre parameters. Mater. Des. 2015, 87, 331–341. [Google Scholar] [CrossRef]
- Bourmaud, A.; Ausias, G.; Lebrun, G.; Tachon, M.-L.L.; Baley, C. Observation of the structure of a composite polypropylene/flax and damage mechanisms under stress. Ind. Crops Prod. 2013, 43, 225–236. [Google Scholar] [CrossRef]
- Monti, A.; El Mahi, A.; Jendli, Z.; Guillaumat, L. Mechanical behaviour and damage mechanisms analysis of a flax-fibre reinforced composite by acoustic emission. Compos. Part A Appl. Sci. Manuf. 2016, 90, 100–110. [Google Scholar] [CrossRef]
- Beaugrand, J.; Guessasma, S.; Maigret, J.-E. Damage mechanisms in defected natural fibers. Sci. Rep. 2017, 7, 14041. [Google Scholar] [CrossRef]
- Guessasma, S.; Beaugrand, J. Damage kinetics at the sub-micrometric scale in bast fibers using finite element simulation and high-resolution X-Ray micro-tomography. Front. Plant Sci. 2019, 10, 194. [Google Scholar] [CrossRef]
- Zamil, M.S.; Yi, H.; Puri, V.M. Mechanical characterization of outer epidermal middle lamella of onion under tensile loading. Am. J. Bot. 2014, 101, 778–787. [Google Scholar] [CrossRef]
- Charlet, K.; Beakou, A. Interfaces within flax fibre bundle: Experimental characterization and numerical modelling. J. Compos. Mater. 2014, 48, 3263–3269. [Google Scholar] [CrossRef]
- Qin, L.; Lin, L.; Fu, F.; Fan, M. Micromechanical properties of wood cell wall and interface compound middle lamella using quasi-static nanoindentation and dynamic modulus mapping. J. Mater. Sci. 2018, 53, 549–558. [Google Scholar] [CrossRef]
- Jakes, J.E.; Frihart, C.R.; Beecher, J.F.; Moon, R.J.; Resto, P.J.; Melgarejo, Z.H.; Suárez, O.M.; Baumgart, H.; Elmustafa, A.A.; Stone, D.S. Nanoindentation near the edge. J. Mater. Res. 2009, 24, 1016–1031. [Google Scholar] [CrossRef]
- Gindl, W.; Gupta, H.-S.; Schöberl, T.; Lichtenegger, H.-C.; Fratzl, P. Mechanical properties of spruce wood cell walls by nanoindentation. Appl. Phys. A 2004, 79, 2069–2073. [Google Scholar] [CrossRef]
- Wimmer, R.; Lucas, B. Comparing mechanical properties of secondary and cell corner middle lamella in spruce wood. IAWA J. 1997, 18, 77–88. [Google Scholar] [CrossRef]
- Arnould, O.; Arinero, R. Towards a better understanding of wood cell wall characterisation with contact resonance atomic force microscopy. Compos. Part A-Appl. Sci 2015, 74, 69–76. [Google Scholar] [CrossRef]
- Clair, B.; Arinero, R.; Leveque, G.; Ramonda, M.; Thibaut, B. Imaging the mechanical properties of wood cell layers by atomic force modulation microscopy. IAWA J. 2003, 24, 223–230. [Google Scholar] [CrossRef]
- Muraille, L.; Aguié-Béghin, V.; Chabbert, B.; Molinari, M. Bioinspired lignocellulosic films to understand the mechanical properties of lignified plant cell walls at nanoscale. Sci. Rep. 2017, 7, 44065. [Google Scholar] [CrossRef]
- Ren, D.; Wang, H.; Yu, Z.; Wang, H.; Yu, Y. Mechanical imaging of bamboo fiber cell walls and their composites by means of peakforce quantitative nanomechanics (PQNM) technique. Holzforschung 2015, 69, 975–984. [Google Scholar] [CrossRef]
- Eder, M.; Arnould, O.; Dunlop, J.W.C.; Hornatowska, J.; Salmen, L.; Salmén, L.; Salmen, L. Experimental micromechanical characterisation of wood cell walls. Wood Sci. Technol. 2013, 47, 163–182. [Google Scholar] [CrossRef]
- Jäger, A.; Bader, T.; Hofstetter, K.; Eberhardsteiner, J. The relation between indentation modulus, microfibril angle, and elastic properties of wood cell walls. Compos. Part A Appl. Sci. Manuf. 2011, 42, 677–685. [Google Scholar] [CrossRef]
- Malek, S.; Gibson, L.J. Multi-scale modelling of elastic properties of balsa. Int. J. Solids Struct. 2017, 113–114, 118–131. [Google Scholar] [CrossRef]
- Bourmaud, A.; Mérotte, J.; Siniscalco, D.; Le Gall, M.; Gager, V.; Le Duigou, A.; Pierre, F.; Behlouli, K.; Arnould, O.; Beaugrand, J.; et al. Main criteria of sustainable natural fibre for efficient unidirectional biocomposites. Compos. Part A Appl. Sci. Manuf. 2019, 124, 105504. [Google Scholar] [CrossRef]
- Dhakal, H.; Bourmaud, A.; Berzin, F.; Almansour, F.; Zhang, Z.; Shah, D.U.; Beaugrand, J. Mechanical properties of leaf sheath date palm fibre waste biomass reinforced polycaprolactone (PCL) biocomposites. Ind. Crops Prod. 2018, 126, 394–402. [Google Scholar] [CrossRef]
- Goudenhooft, C.; Siniscalco, D.; Arnould, O.; Bourmaud, A.; Sire, O.; Gorshkova, T.; Baley, C. Investigation of the mechanical properties of flax cell walls during plant development: The relation between performance and cell wall structure. Fibers 2018, 6, 6. [Google Scholar] [CrossRef]
- Arnould, O.; Siniscalco, D.; Bourmaud, A.; Le Duigou, A.; Baley, C. Better insight into the nano-mechanical properties of flax fibre cell walls. Ind. Crops Prod. 2017, 97, 224–228. [Google Scholar] [CrossRef]
- Gao, J.; Kim, J.S.; Terziev, N.; Allegretti, O.; Daniel, G. Chemical and ultrastructural changes in compound middle lamella (CML) regions of softwoods thermally modified by the Termovuoto process. Holzforschung 2014, 68, 849–859. [Google Scholar] [CrossRef]
- Abdul Khalil, H.P.S.; Yusra, A.F.I.; Bhat, A.H.; Jawaid, M. Cell wall ultrastructure, anatomy, lignin distribution, and chemical composition of Malaysian cultivated kenaf fiber. Ind. Crops Prod. 2010, 31, 113–121. [Google Scholar] [CrossRef]
- Day, A.; Ruel, K.; Neutelings, G.; Crônier, D.; David, H.; Hawkins, S.; Chabbert, B. Lignification in the flax stem: Evidence for an unusual lignin in bast fibers. Planta 2005, 222, 234–245. [Google Scholar] [CrossRef]
- Le Duigou, A.; Bourmaud, A.; Balnois, E.; Davies, P.; Baley, C. Improving the interfacial properties between flax fibres and PLLA by a water fibre treatment and drying cycle. Ind. Crops Prod. 2012, 39, 31–39. [Google Scholar] [CrossRef]
- Bourmaud, A.; Gibaud, M.; Lefeuvre, A.; Morvan, C.; Baley, C. Influence of the morphology characters of the stem on the lodging resistance of Marylin flax. Ind. Crops Prod. 2015, 66, 27–37. [Google Scholar] [CrossRef]
- Radotić, K.; Simić-Krstić, J.; Jeremić, M.; Trifunović, M. A study of lignin formation at the molecular level by scanning tunneling microscopy. Biophys. J. 1994, 66, 1763–1767. [Google Scholar] [CrossRef]
- Terashima, N.; Awano, T.; Takabe, K.; Yoshida, M. Formation of macromolecular lignin in ginkgo xylem cell walls as observed by field emission scanning electron microscopy. C. R. Biol. 2004, 327, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Stan, G.; Cook, R.F. Mapping the elastic properties of granular Au films by contact resonance atomic force microscopy. Nanotechnology 2008, 19, 235701. [Google Scholar] [CrossRef] [PubMed]
- Hanley, S.J.; Gray, D.G. Atomic force microscope images of black spruce wood sections and pulp fibres. Holzforschung 1994, 48, 29–34. [Google Scholar] [CrossRef]
- Kundu, A.; Sarkar, D.; Mandal, N.A.; Sinha, M.K.; Mahapatra, B.S. A secondary phloic (bast) fibre-shy (bfs) mutant of dark jute (Corchorus olitorius L.) develops lignified fibre cells but is defective in cambial activity. Plant Growth Regul. 2012, 67, 45–55. [Google Scholar] [CrossRef]
- Daher, F.B.; Braybrook, S.A. How to let go: pectin and plant cell adhesion. Front. Plant Sci. 2015, 6, 523. [Google Scholar] [CrossRef]
- Bourmaud, A.; Dhakal, H.; Habrant, A.; Padovani, J.; Siniscalco, D.; Ramage, M.H.; Beaugrand, J.; Shah, D.U. Exploring the potential of waste leaf sheath date palm fibres for composite reinforcement through a structural and mechanical analysis. Compos. Part A Appl. Sci. Manuf. 2017, 103. [Google Scholar] [CrossRef]
- Baley, C.; Perrot, Y.; Busnel, F.; Guezenoc, H.; Davies, P. Transverse tensile behaviour of unidirectional plies reinforced with flax fibres. Mater. Lett. 2006, 60, 2984–2987. [Google Scholar] [CrossRef]
- Coroller, G.; Lefeuvre, A.; Le Duigou, A.; Bourmaud, A.; Ausias, G.; Gaudry, T.; Baley, C. Effect of flax fibres individualisation on tensile failure of flax/epoxy unidirectional composite. Compos. Part A Appl. Sci. Manuf. 2013, 51, 62–70. [Google Scholar] [CrossRef]
- Barthelat, F.; Espinosa, H.D. An experimental investigation of deformation and fracture of nacre–Mother of pearl. Exp. Mech. 2007, 47, 311–324. [Google Scholar] [CrossRef]
- Tanguy, M.; Bourmaud, A.; Beaugrand, J.; Gaudry, T.; Baley, C. Polypropylene reinforcement with flax or jute fibre; Influence of microstructure and constituents properties on the performance of composite. Compos. Part B Eng. 2018, 139, 64–74. [Google Scholar] [CrossRef]
- Graupner, N.; Fischer, H.; Ziegmann, G.; Müssig, J. Improvement and analysis of fibre/matrix adhesion of regenerated cellulose fibre reinforced PP-, MAPP- and PLA-composites by the use of Eucalyptus globulus lignin. Compos. Part B Eng. 2014, 66, 117–125. [Google Scholar] [CrossRef]
- Padovani, J.; Legland, D.; Pernes, M.; Gallos, A.; Thomachot-Schneider, C.; Shah, D.U.; Bourmaud, A.; Beaugrand, J. Beating of hemp bast fibres: an examination of a hydro-mechanical treatment on chemical, structural, and nanomechanical property evolutions. Cellulose 2019, 26, 5665–5683. [Google Scholar] [CrossRef]
- Berzin, F.; Beaugrand, J.; Dobosz, S.; Budtova, T.; Vergnes, B. Lignocellulosic fiber breakage in a molten polymer. Part 3. Modeling of the dimensional change of the fibers during compounding by twin screw extrusion. Compos. Part A Appl. Sci. Manuf. 2017, 101, 422–431. [Google Scholar] [CrossRef]
- Le Duc, A.; Vergnes, B.; Budtova, T. Polypropylene/natural fibres composites: Analysis of fibre dimensions after compounding and observations of fibre rupture by rheo-optics. Compos. Part A Appl. Sci. Manuf. 2011, 42, 1727–1737. [Google Scholar] [CrossRef]
- Alix, S.; Lebrun, L.; Marais, S.; Philippe, E.; Bourmaud, A.; Baley, C.; Morvan, C. Pectinase treatments on technical fibres of flax: Effects on water sorption and mechanical properties. Carbohydr. Polym. 2012, 87, 177–185. [Google Scholar] [CrossRef]
- Biagiotti, J.; Puglia, D.; Kenny, J.M. A Review on natural fibre-based composites-Part I: Structure, processing and properties of vegetable fibres. J. Nat. Fibers 2004, 1, 37–68. [Google Scholar] [CrossRef]
- Van Hazendonk, J.M.; Reinerik, E.J.M.; de Waard, P.; van Dam, J.E.G. Structural analysis of acetylated hemicellulose polysaccharides from fibre flax (Linum usitatissimum L.). Carbohydr. Res. 1996, 291, 141–154. [Google Scholar] [CrossRef]
- Esau, K. Plant Anatomy; Wiley: New York, NY, USA, 1953. [Google Scholar]
- Crônier, D.; Monties, B.; Chabbert, B. Structure and chemical composition of bast fibers isolated from developing hemp stem. J. Agric. Food Chem. 2005, 53, 8279–8289. [Google Scholar] [CrossRef]
- Marrot, L.; Lefeuvre, A.; Pontoire, B.; Bourmaud, A.; Baley, C. Analysis of the hemp fiber mechanical properties and their scattering (Fedora 17). Ind. Crops Prod. 2013, 51. [Google Scholar] [CrossRef]
- Gümüşkaya, E.; Usta, M.; Balaban, M. Carbohydrate components and crystalline structure of organosolv hemp (Cannabis sativa L.) bast fibers pulp. Bioresour. Technol. 2007, 98, 491–497. [Google Scholar] [CrossRef] [PubMed]
- Khan, F.; Ahmad, S.R. Chemical modification and spectroscopic analysis of jute fibre. Polym. Degrad. Stab. 1996, 52, 335–340. [Google Scholar] [CrossRef]
- Roy, A.; Chakraborty, S.; Kundu, S.P.; Basak, R.K.; Majumder, S.B.; Adhikari, B. Improvement in mechanical properties of jute fibres through mild alkali treatment as demonstrated by utilisation of the Weibull distribution model. Bioresour. Technol. 2012, 107, 222–228. [Google Scholar] [CrossRef]
- Karimi, S.; Tahir, P.M.; Karimi, A.; Dufresne, A.; Abdulkhani, A. Kenaf bast cellulosic fibers hierarchy: A comprehensive approach from micro to nano. Carbohydr. Polym. 2014, 101, 878–885. [Google Scholar] [CrossRef]
- Ashori, A.; Harun, J.; Raverty, W.D.; Yusoff, M.N.M. Chemical and morphological characteristics of Malaysian cultivated kenaf (Hibiscus cannabinus) fiber. Polym. Plast. Technol. Eng. 2006, 45, 131–134. [Google Scholar] [CrossRef]
- Zhou, C.; Ohtani, Y.; Sameshima, K.; Zhen, M. Selection of plant population of kenaf (Hibiscus cannabinus L.) as a papermaking raw material on arid hillside land in China. J. Wood Sci. 1998, 44, 296–302. [Google Scholar] [CrossRef]
- Bacci, L.; Di Lonardo, S.; Albanese, L.; Mastromei, G.; Perito, B. Effect of different extraction methods on fiber quality of nettle (Urtica dioica L.). Text. Res. J. 2011, 81, 827–837. [Google Scholar] [CrossRef]
- Saadaoui, N.; Rouilly, A.; Fares, K.; Rigal, L. Characterization of date palm lignocellulosic by-products and self-bonded composite materials obtained thereof. Mater. Des. 2013, 50, 302–308. [Google Scholar] [CrossRef]
- Derjaguin, B.V.; Muller, V.M.; Toporov, Y.P. Effect of contact deformations on the adhesion of particles. J. Colloid Interface Sci. 1975, 53, 314–326. [Google Scholar] [CrossRef]
- Nečas, D.; Klapetek, P. Gwyddion: An open-source software for SPM data analysis. Open Phys. 2012, 10, 181–188. [Google Scholar] [CrossRef]
Sample Availability: Samples are available from the authors. |
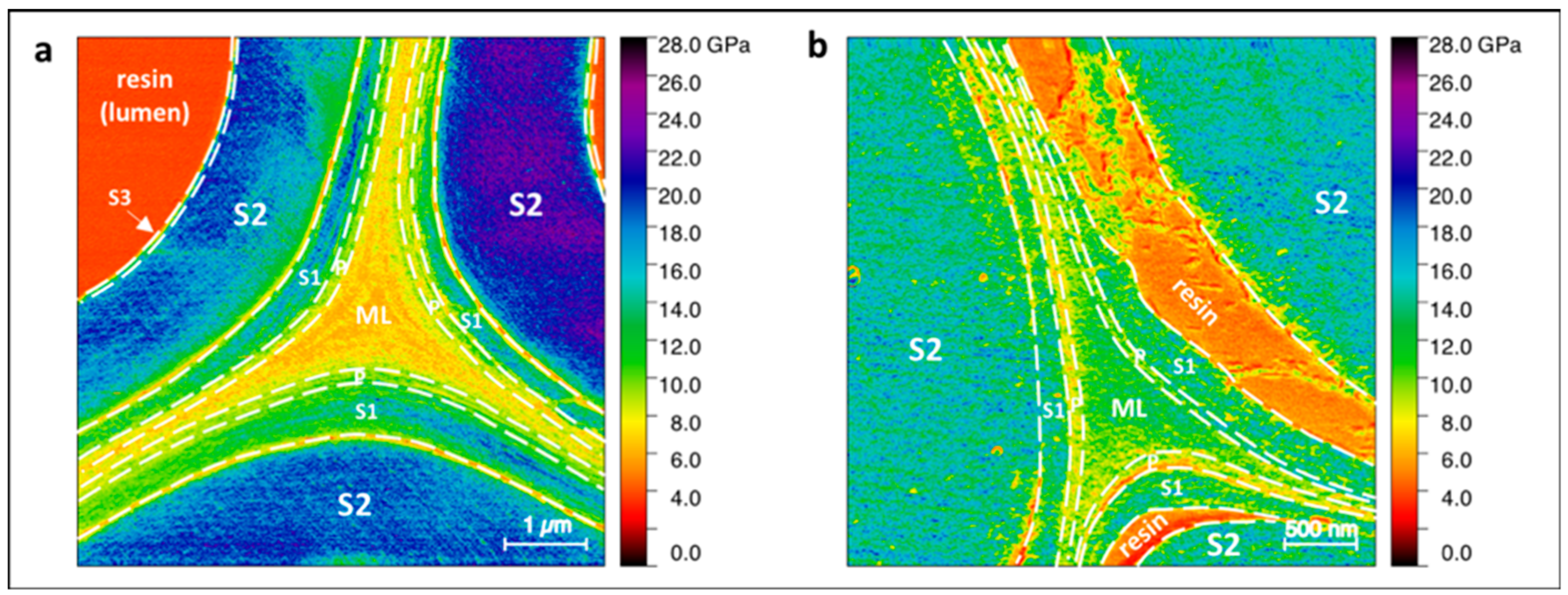
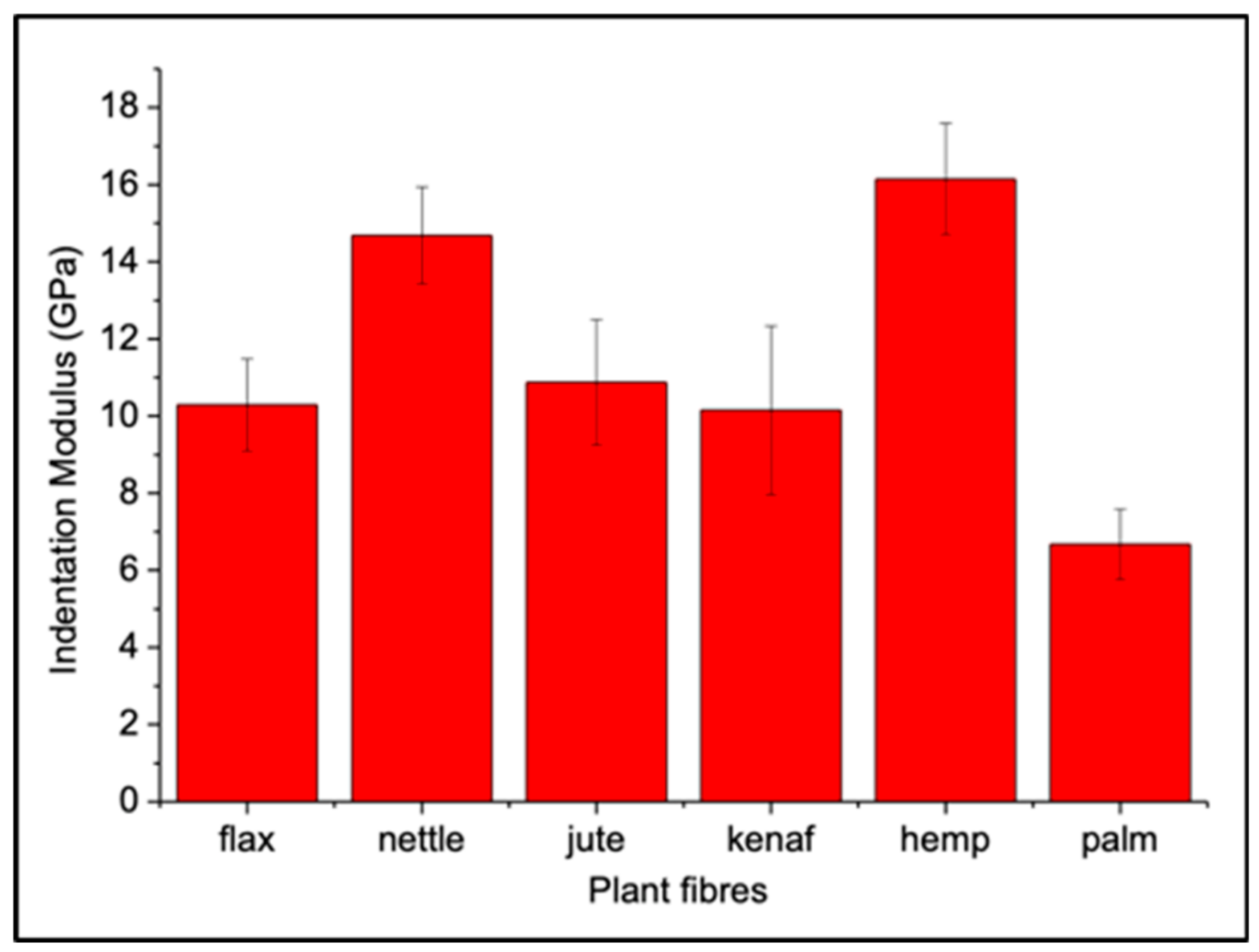
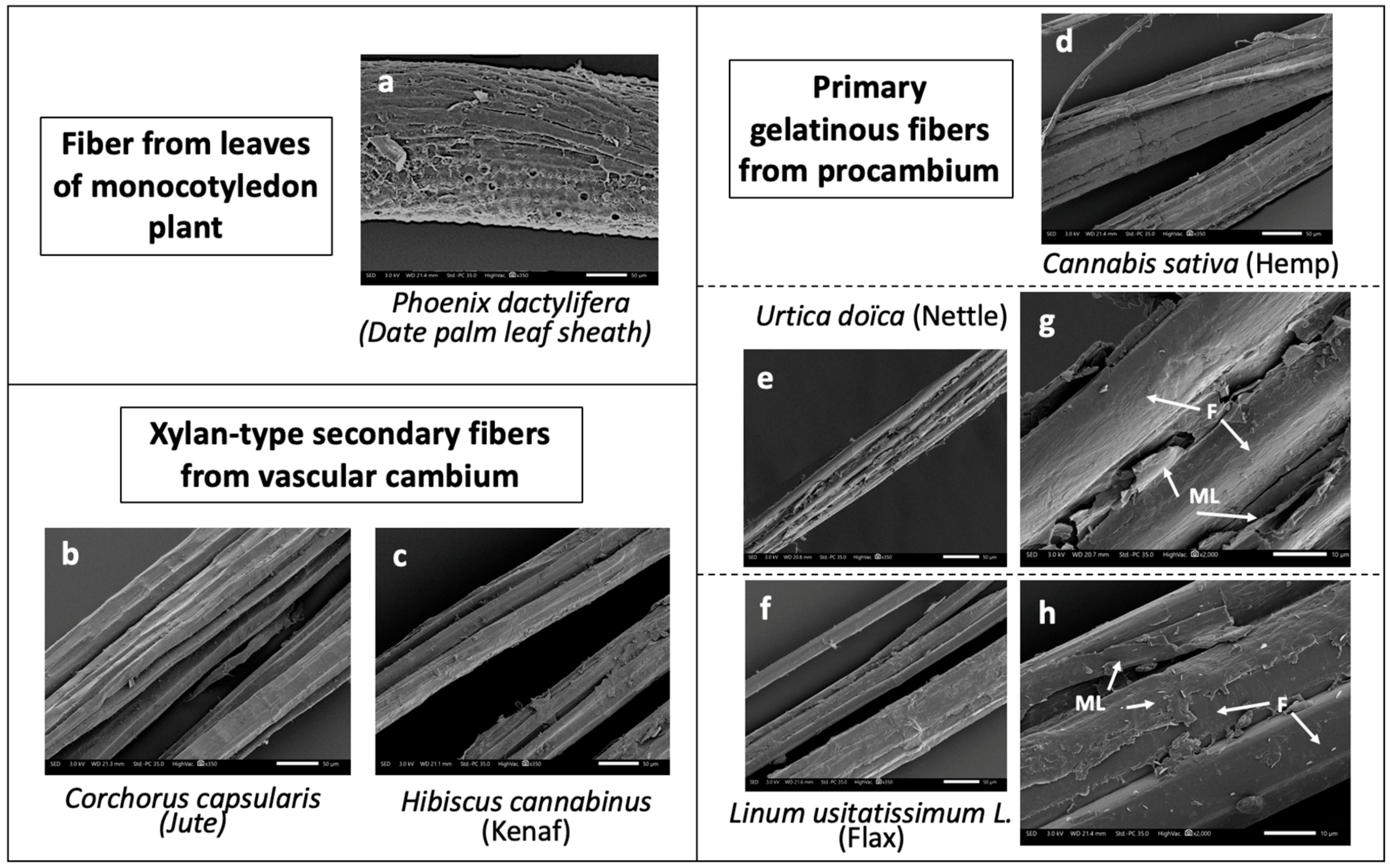

| Indentation Modulus (GPa) | |
|---|---|
| Linum Usitatissimum L. (Flax) | 10.2 (± 1.2) |
| Cannabis Sativa (Hemp) | 16.1 (± 1.4) |
| Corchorus capsularis (Jute) | 10.9 (± 1.6) |
| Hibiscus cannabinus (Kenaf) | 10.2 (± 2.2) |
| Urtica Doïca (Nettle) | 14.7 (± 1.3) |
| Phoenix dactylifera (Date palm leaf sheath) | 6.7 (± 0.9) |
| Cellulose (%) | Hemicellulose (%) | Lignin (%) | Reference | |
|---|---|---|---|---|
| Linum Usitatissimum L. (Flax) | 60–85 | 14.0–20.6 | 1–3 | [60,61,62] |
| Cannabis Sativa (Hemp) | 55–90 | 12 | 2–5 | [63,64,65,66] |
| Corchorus capsularis (Jute) | 58.0–71.5 | 13.6–24.0 | 11.8–16 | [67,68] |
| Hibiscus cannabinus (Kenaf) | 52.0–61.2 | 18.5–29.7 | 12.9–16.1 | [69,70,71] |
| Urtica Doïca (Nettle) | 65.3–86.3 | 5.2–12.5 | 1.6–3.8 | [72] |
| Phoenix dactylifera (Date palm leaf sheath) | 34–45.1 | 27.7–28.9 | 16.9–18.2 | [51,73] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melelli, A.; Arnould, O.; Beaugrand, J.; Bourmaud, A. The Middle Lamella of Plant Fibers Used as Composite Reinforcement: Investigation by Atomic Force Microscopy. Molecules 2020, 25, 632. https://doi.org/10.3390/molecules25030632
Melelli A, Arnould O, Beaugrand J, Bourmaud A. The Middle Lamella of Plant Fibers Used as Composite Reinforcement: Investigation by Atomic Force Microscopy. Molecules. 2020; 25(3):632. https://doi.org/10.3390/molecules25030632
Chicago/Turabian StyleMelelli, Alessia, Olivier Arnould, Johnny Beaugrand, and Alain Bourmaud. 2020. "The Middle Lamella of Plant Fibers Used as Composite Reinforcement: Investigation by Atomic Force Microscopy" Molecules 25, no. 3: 632. https://doi.org/10.3390/molecules25030632
APA StyleMelelli, A., Arnould, O., Beaugrand, J., & Bourmaud, A. (2020). The Middle Lamella of Plant Fibers Used as Composite Reinforcement: Investigation by Atomic Force Microscopy. Molecules, 25(3), 632. https://doi.org/10.3390/molecules25030632






