Characterization and Dimethyl Phthalate Flocculation Performance of the Cationic Polyacrylamide Flocculant P(AM-DMDAAC) Produced by Microwave-Assisted Synthesis
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis of P(AM–DMDAAC)
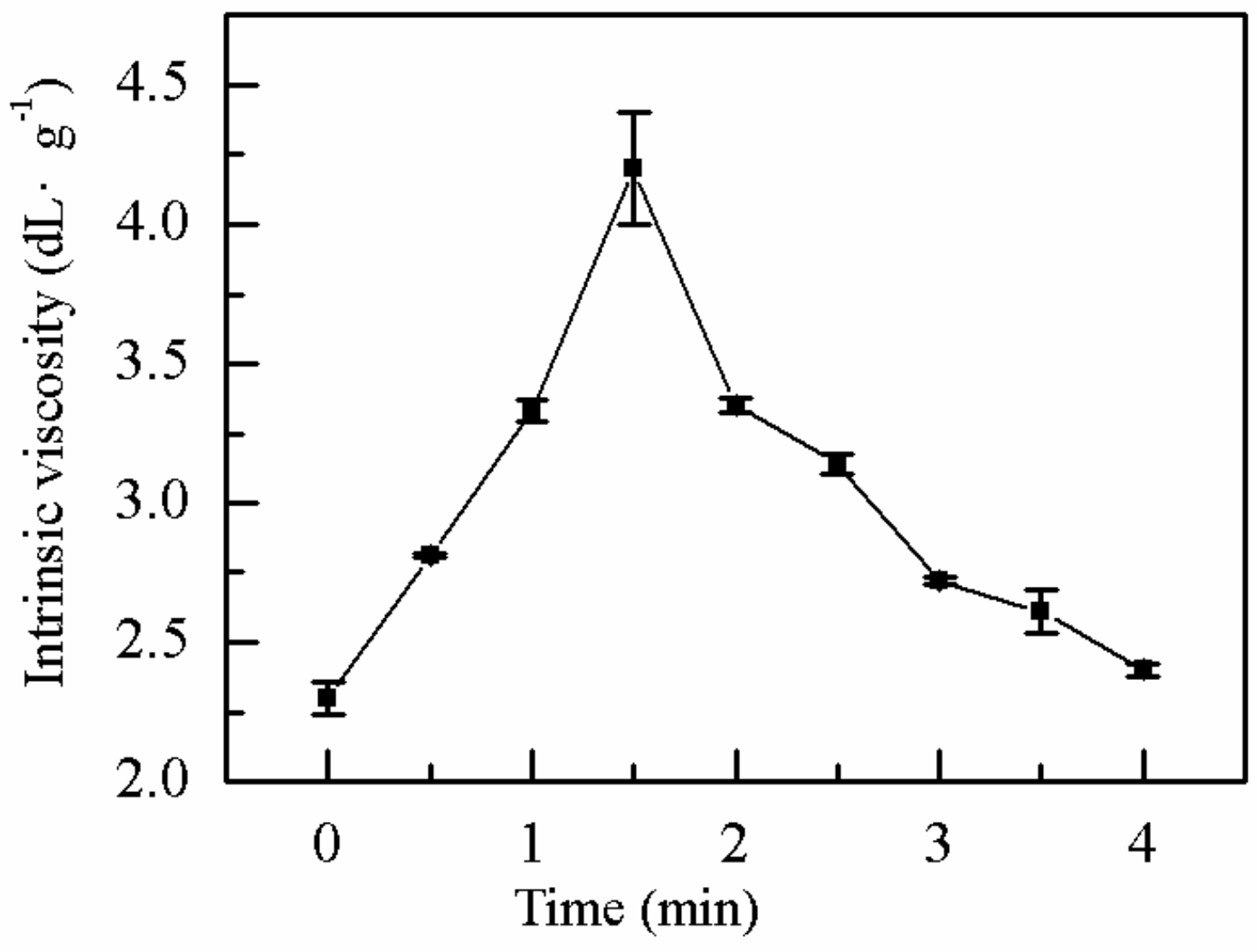
2.1.1. Effect of MV Time on η
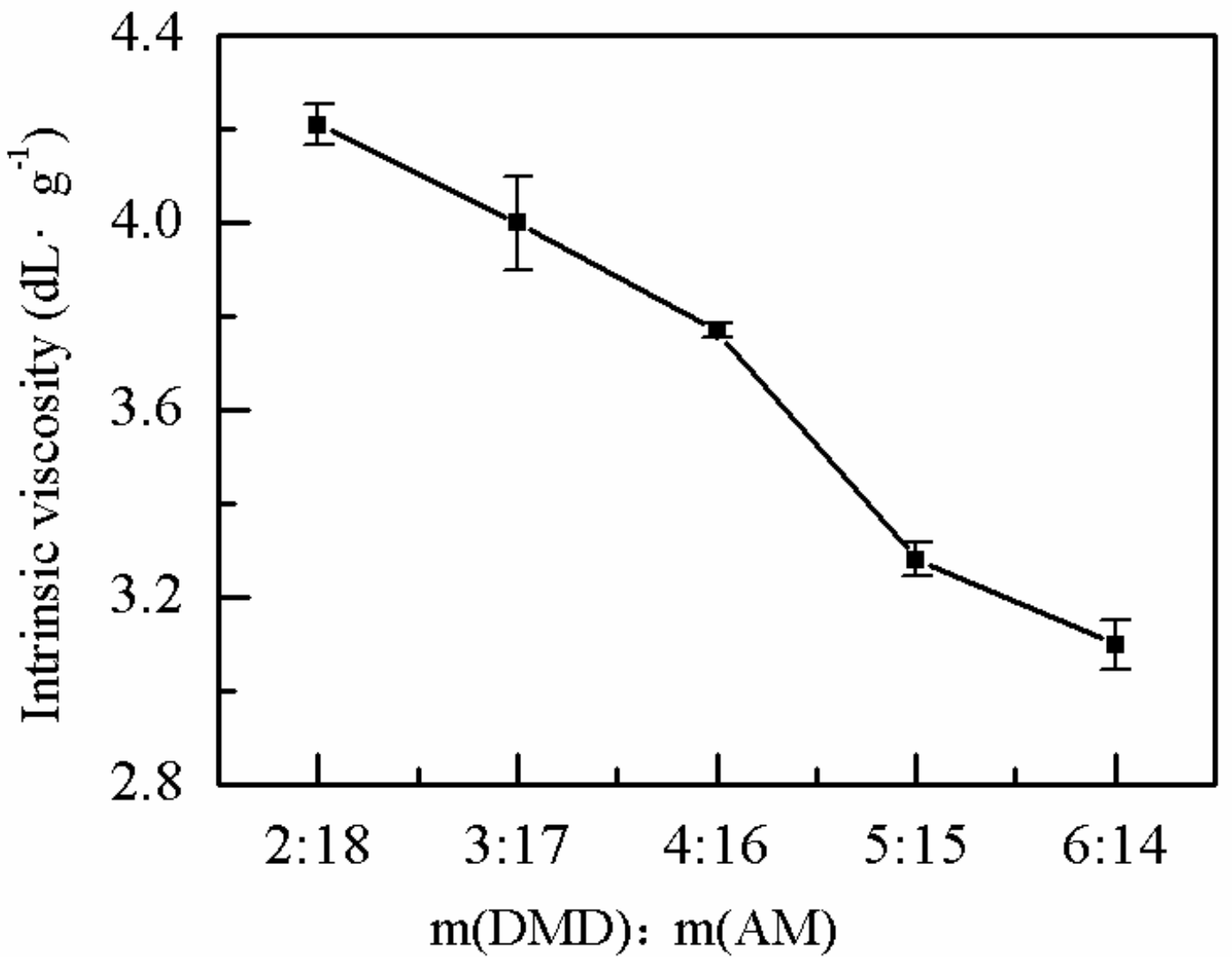
2.1.2. Effects of the Monomer Mass Ratio on η
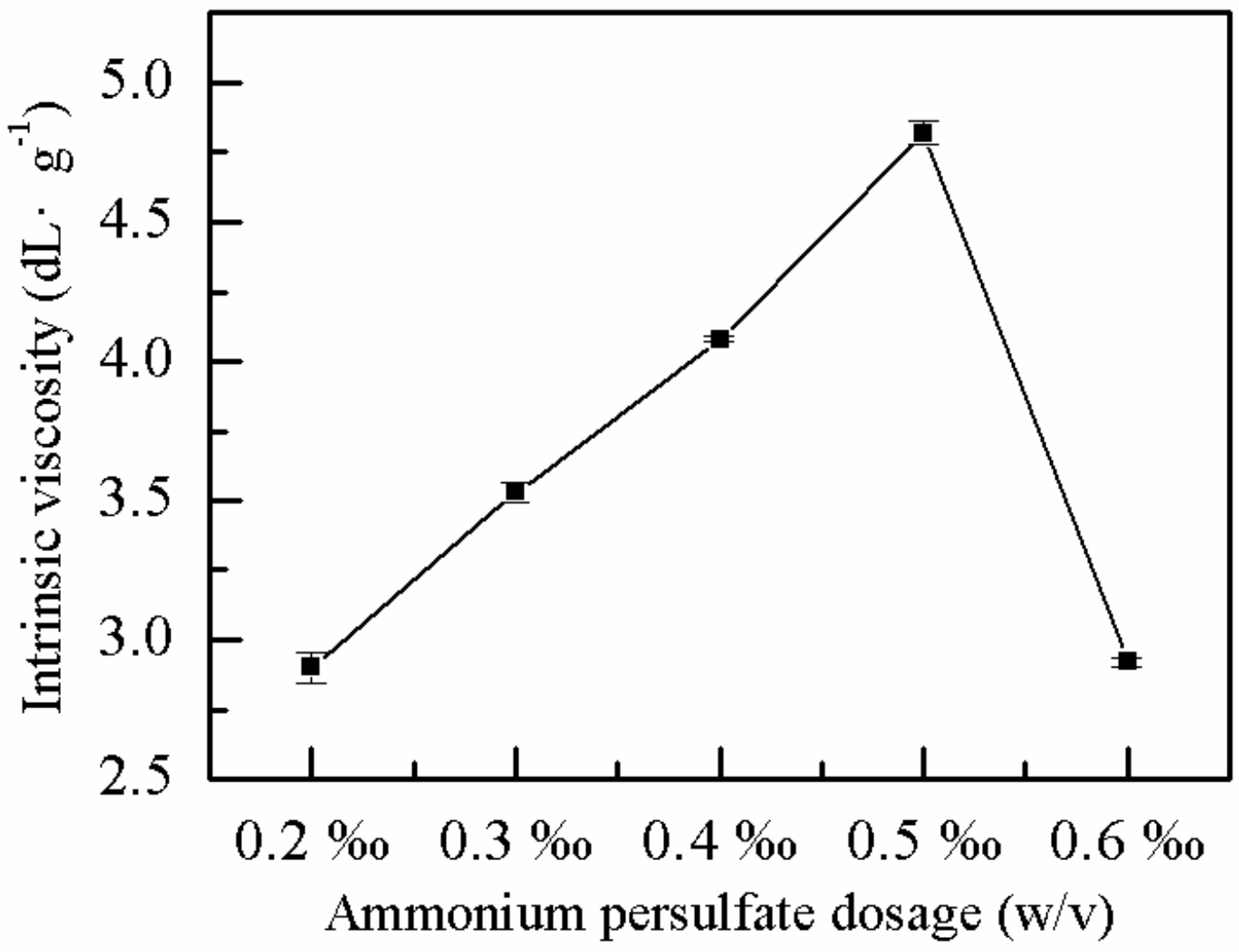
2.1.3. Effect of Ammonium Persulfate Dose on η
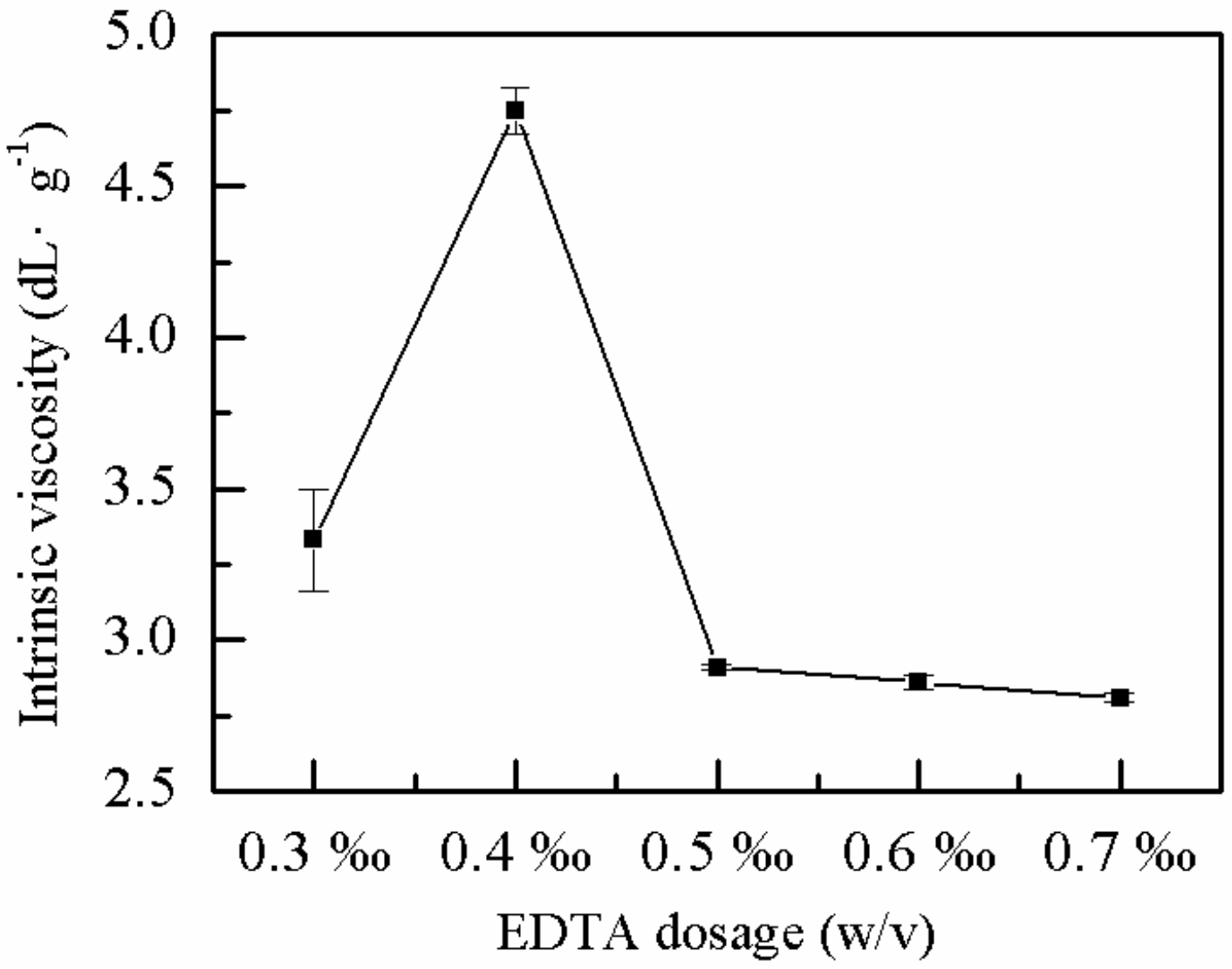
2.1.4. Effect of EDTA Dose on η
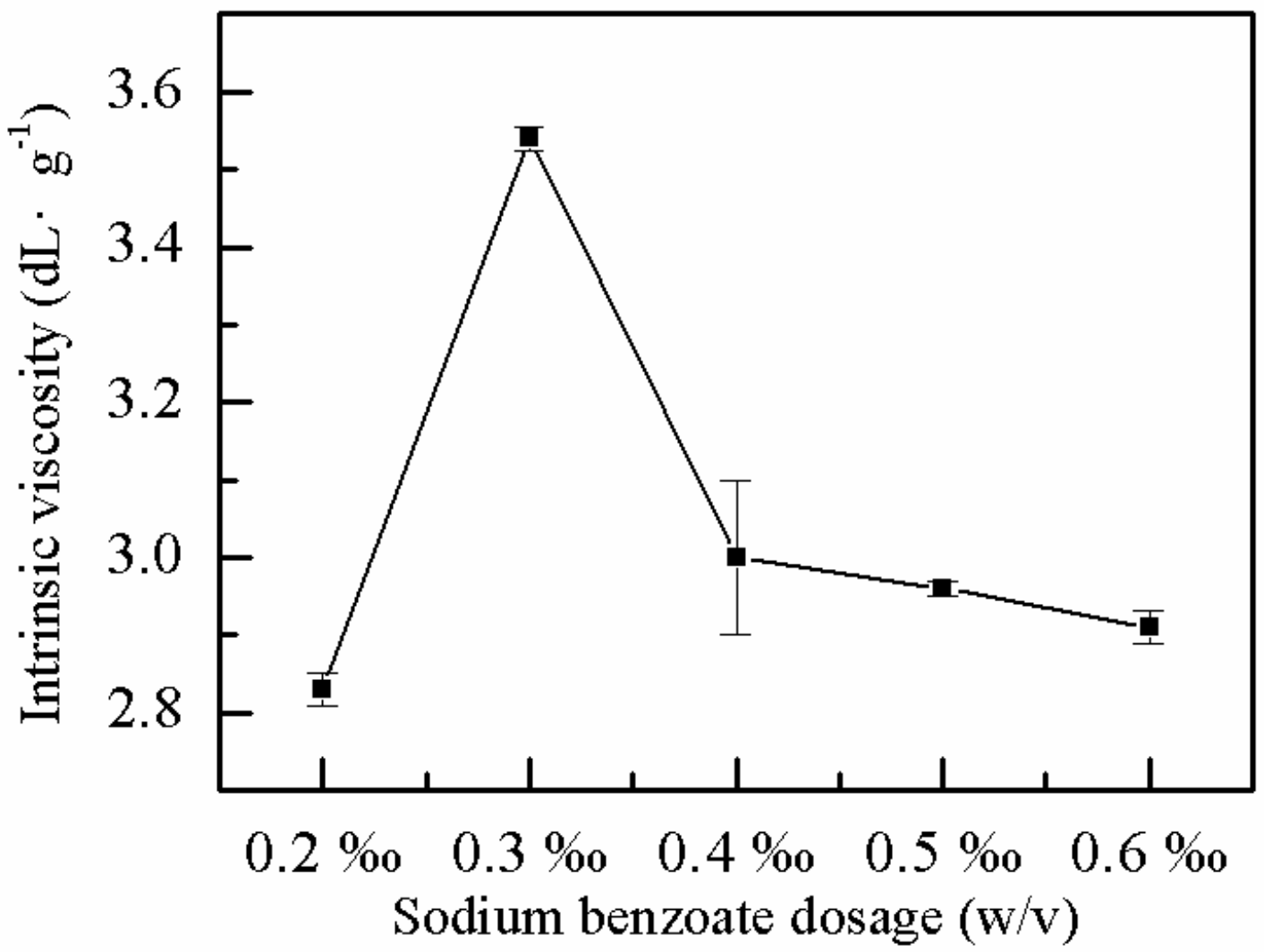
2.1.5. Effect of Sodium Benzoate on η
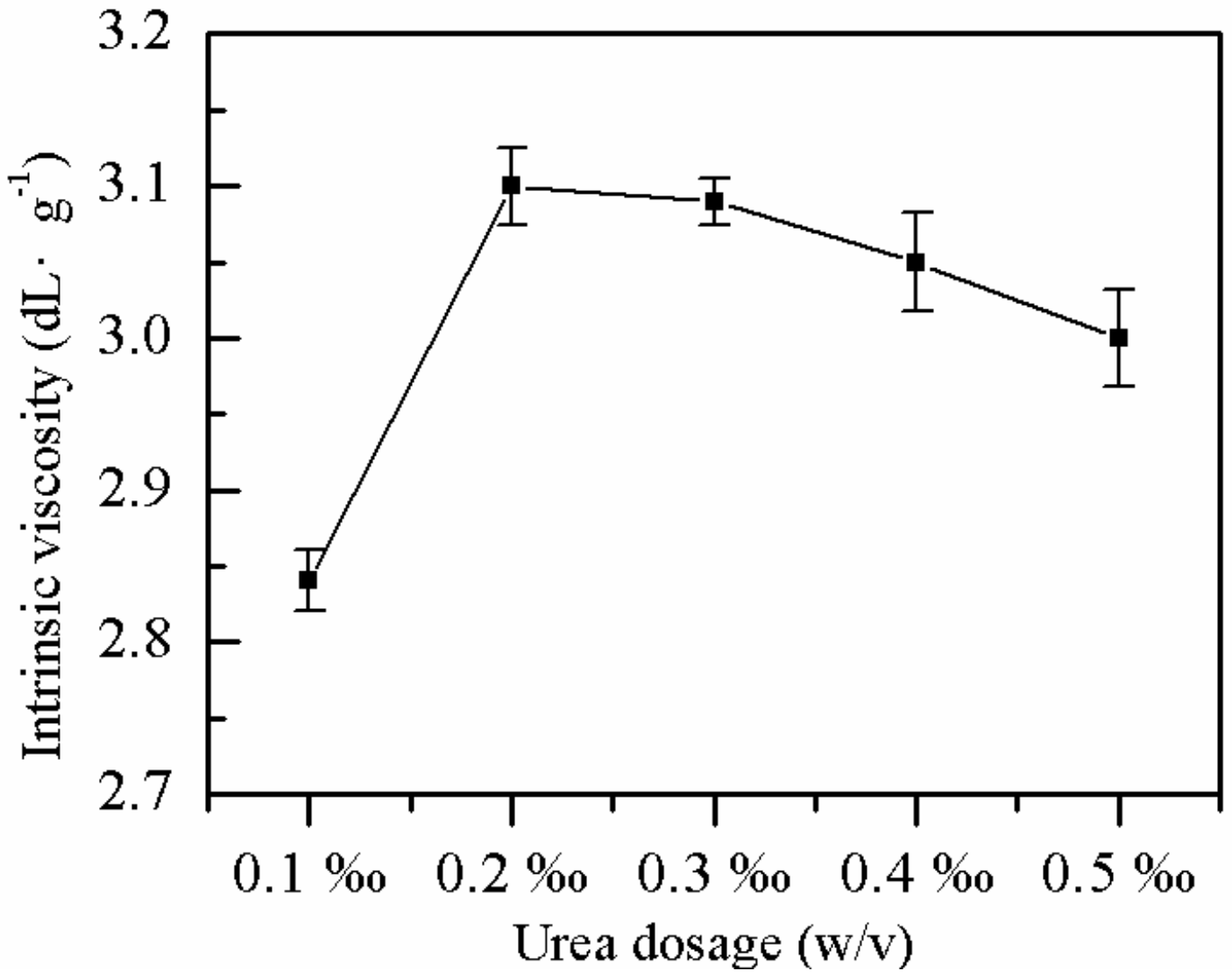
2.1.6. Effect of Urea Dose on η
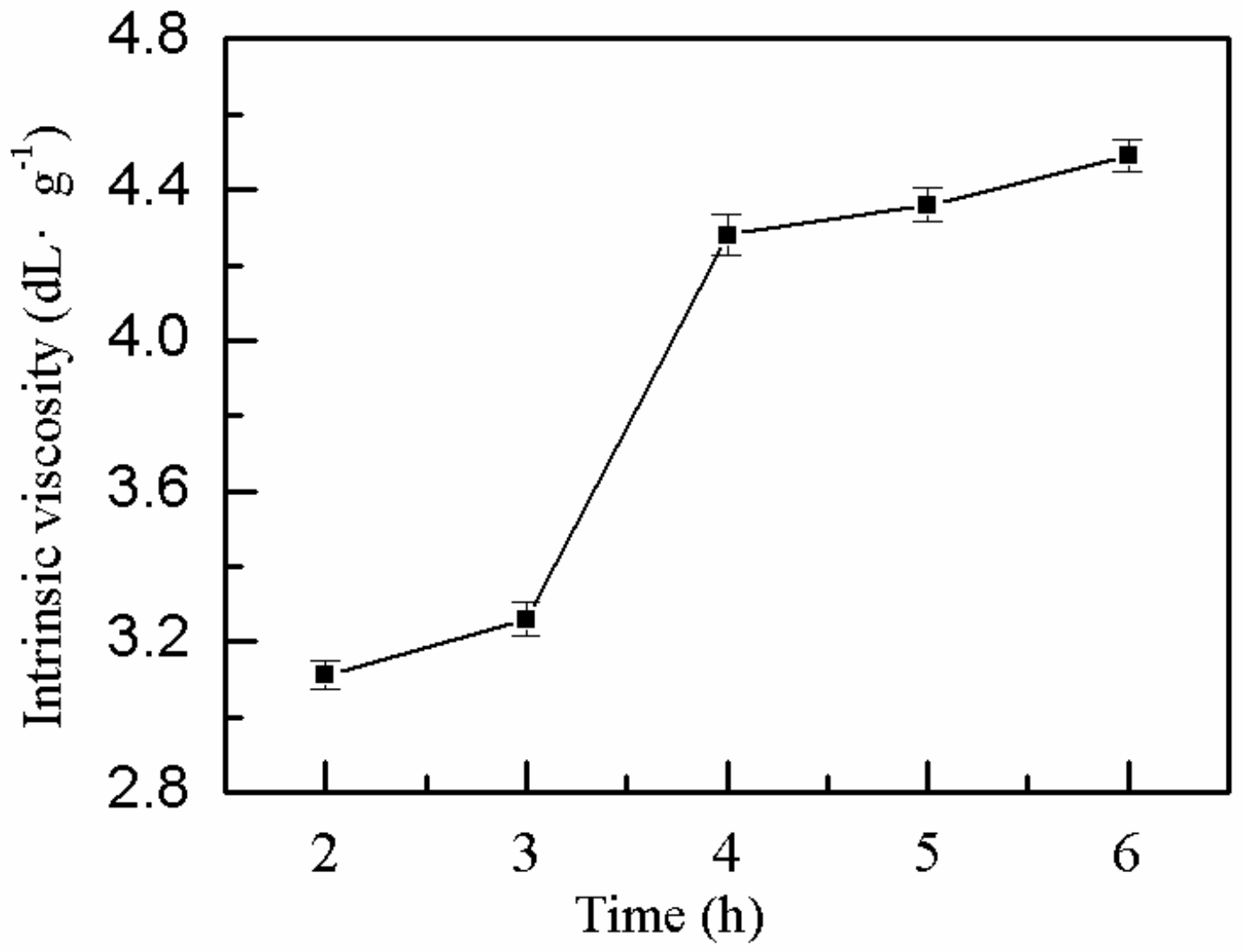
2.1.7. Effect of Bath Time on η
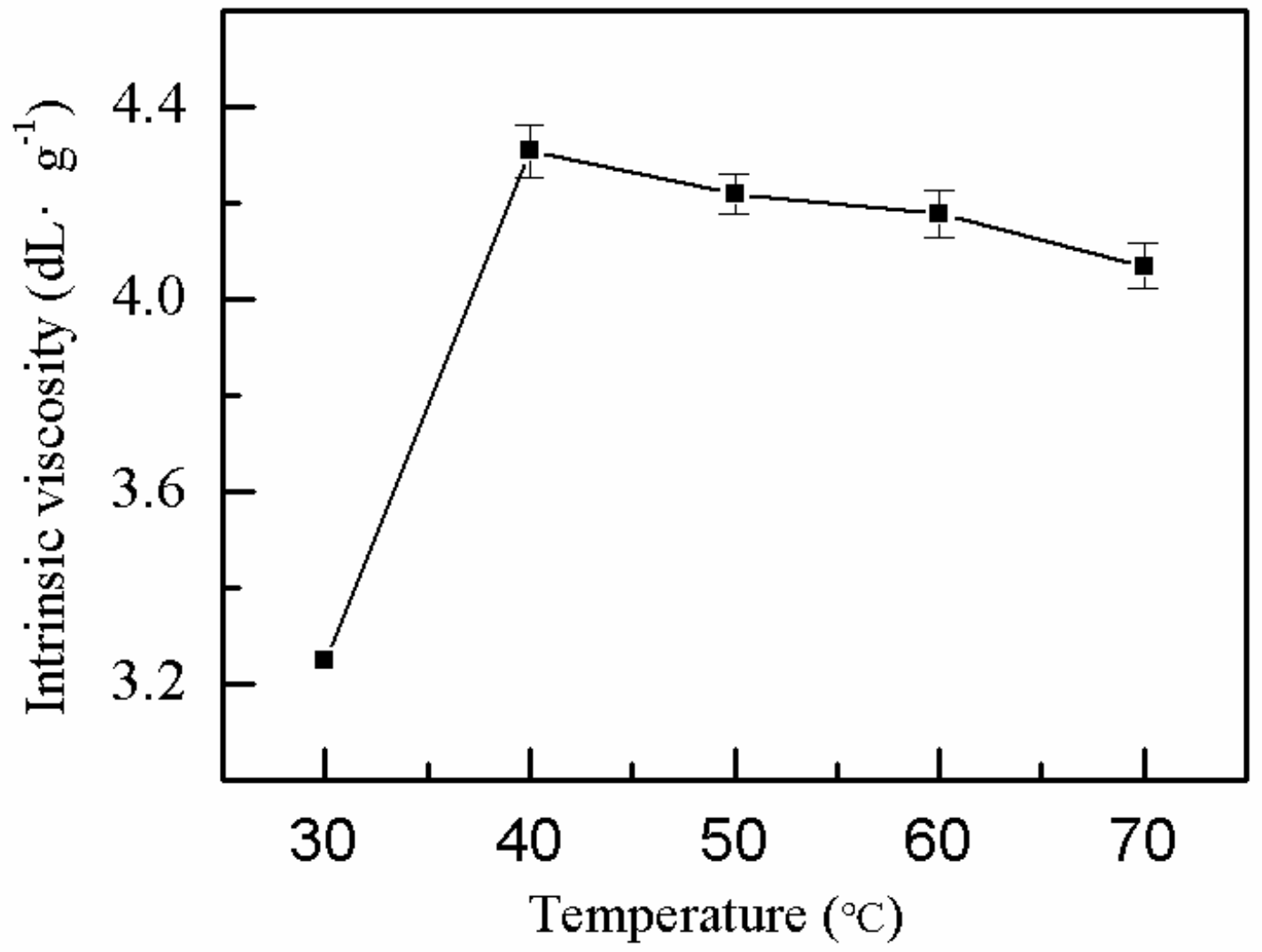
2.1.8. Effect of Reaction Temperature on η
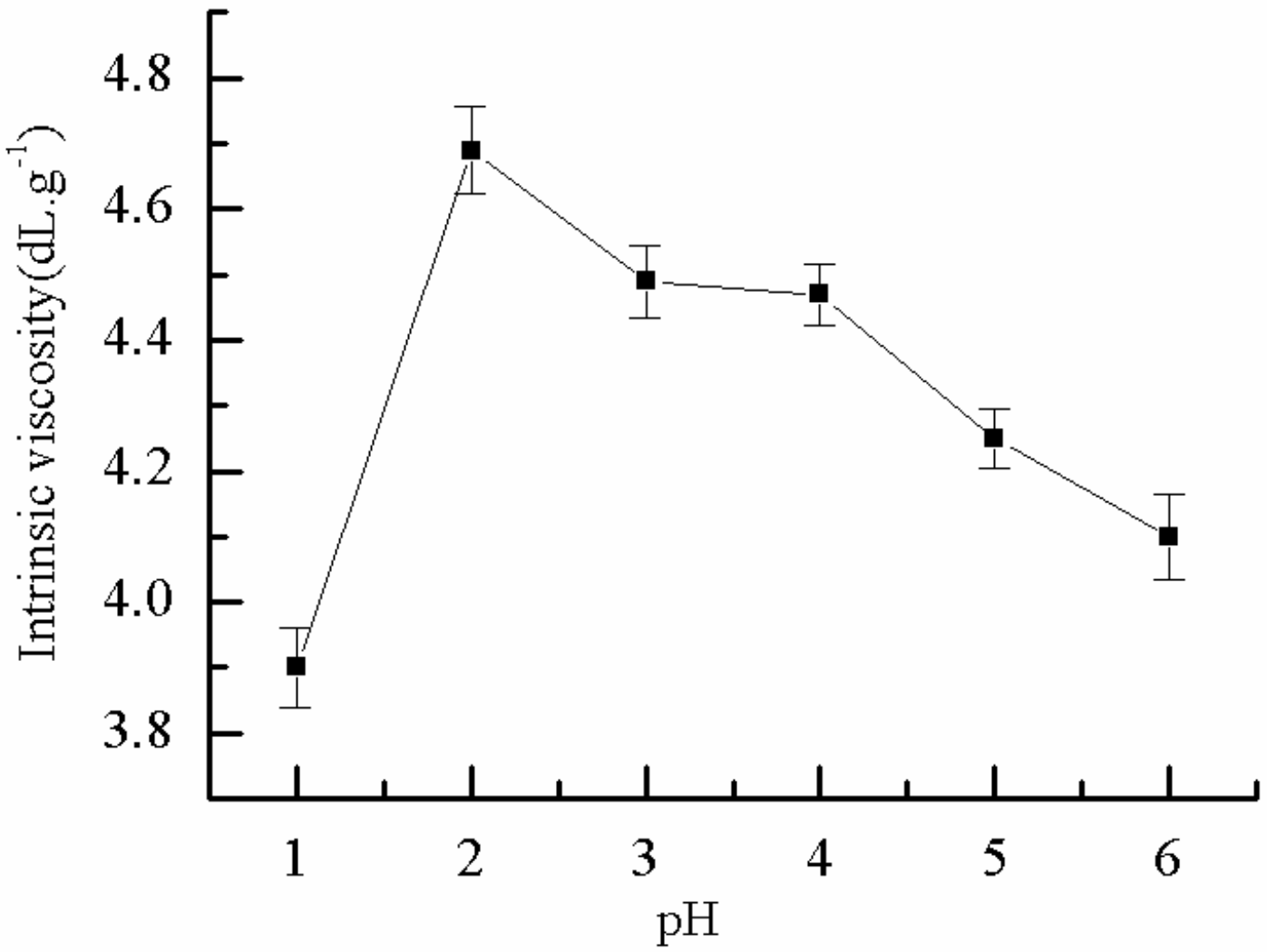
2.1.9. Effect of pH on η
2.2. Characterization
2.2.1. SEM of P(AM–DMDAAC)
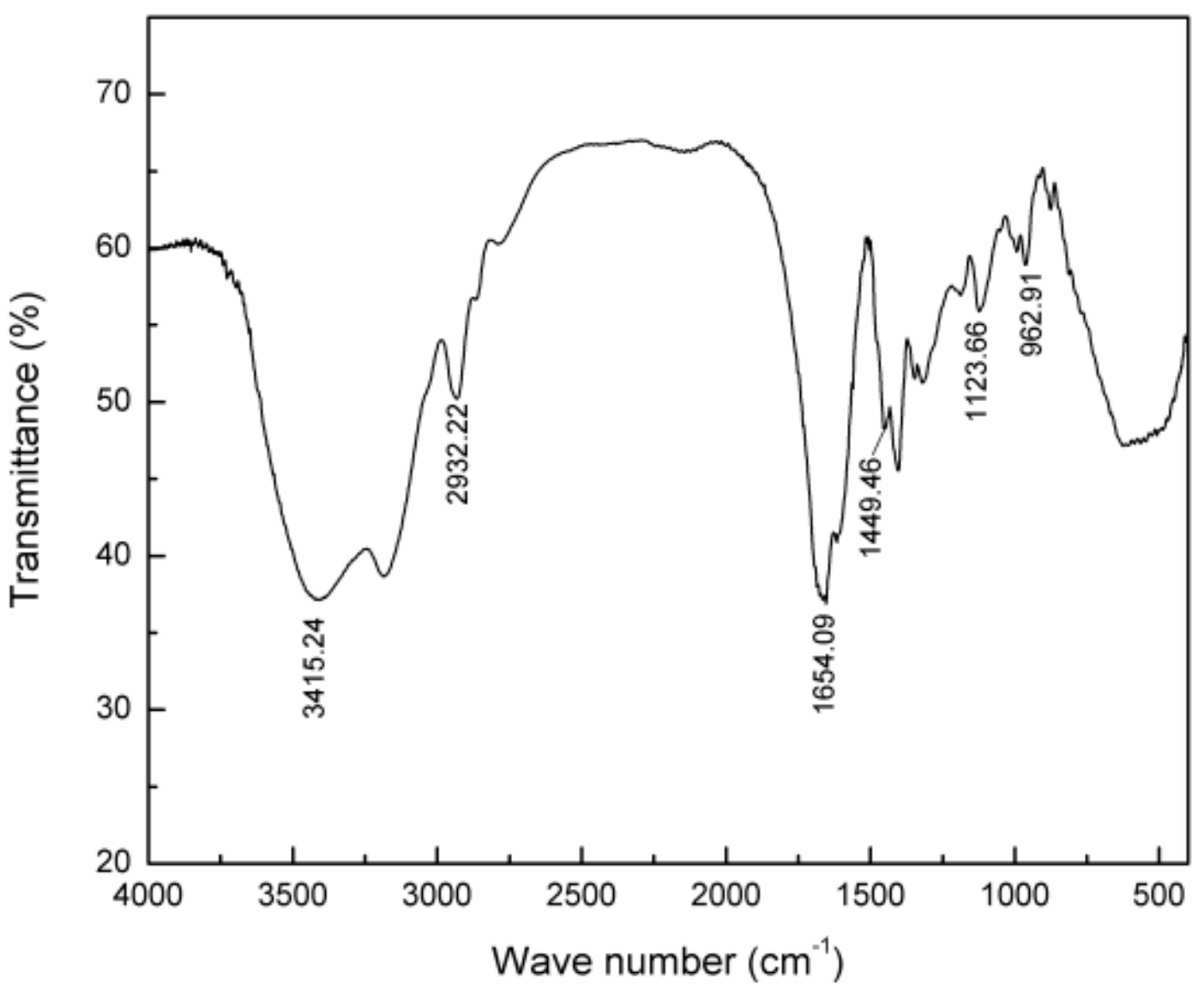
2.2.2. Infrared Spectrum of P(AM–DMDAAC)
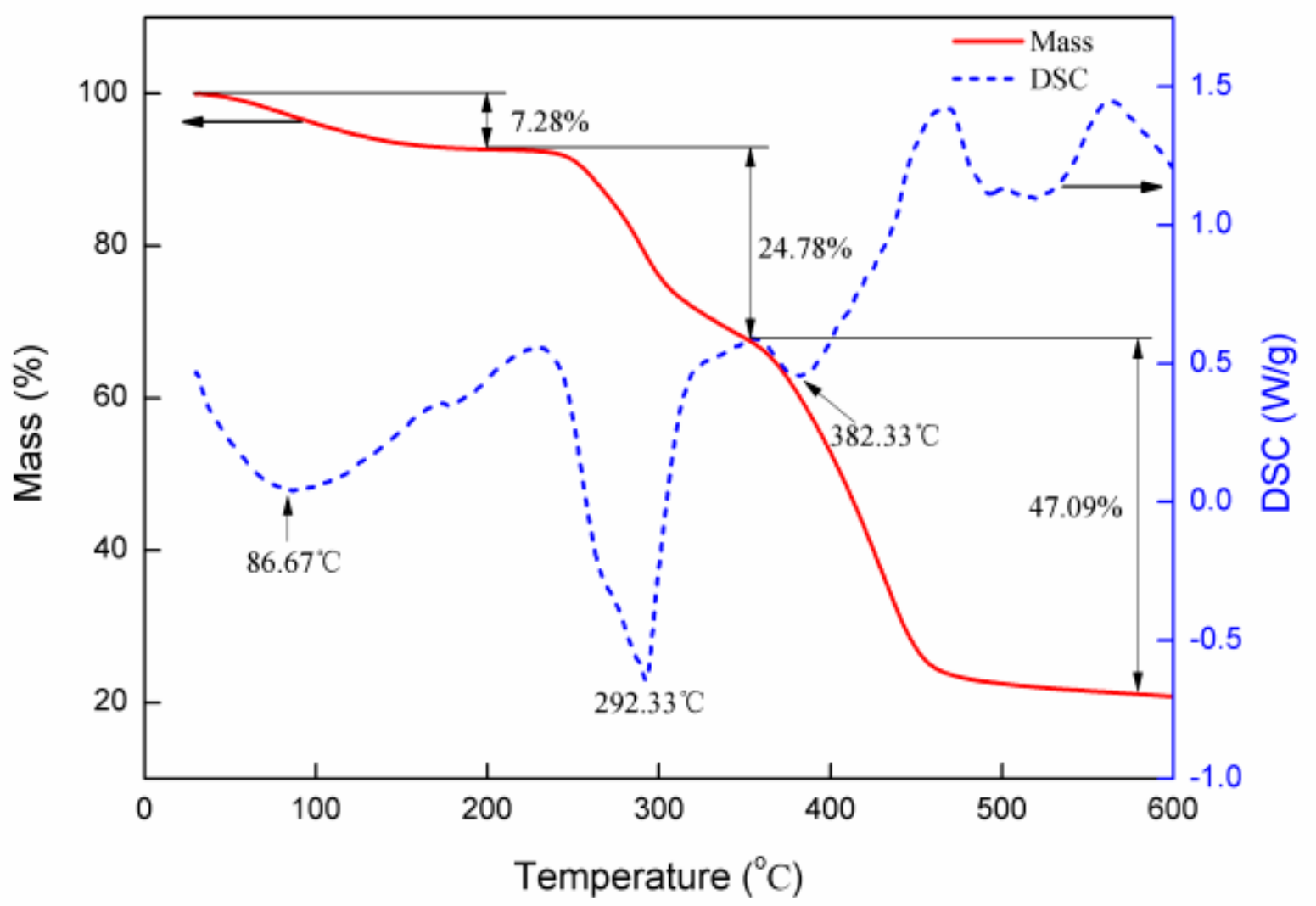
2.2.3. DTA–TGA of P(AM–DMDAAC)
2.3. Validation of the Effectiveness of P(AM–DMDAAC) in DMP Removal
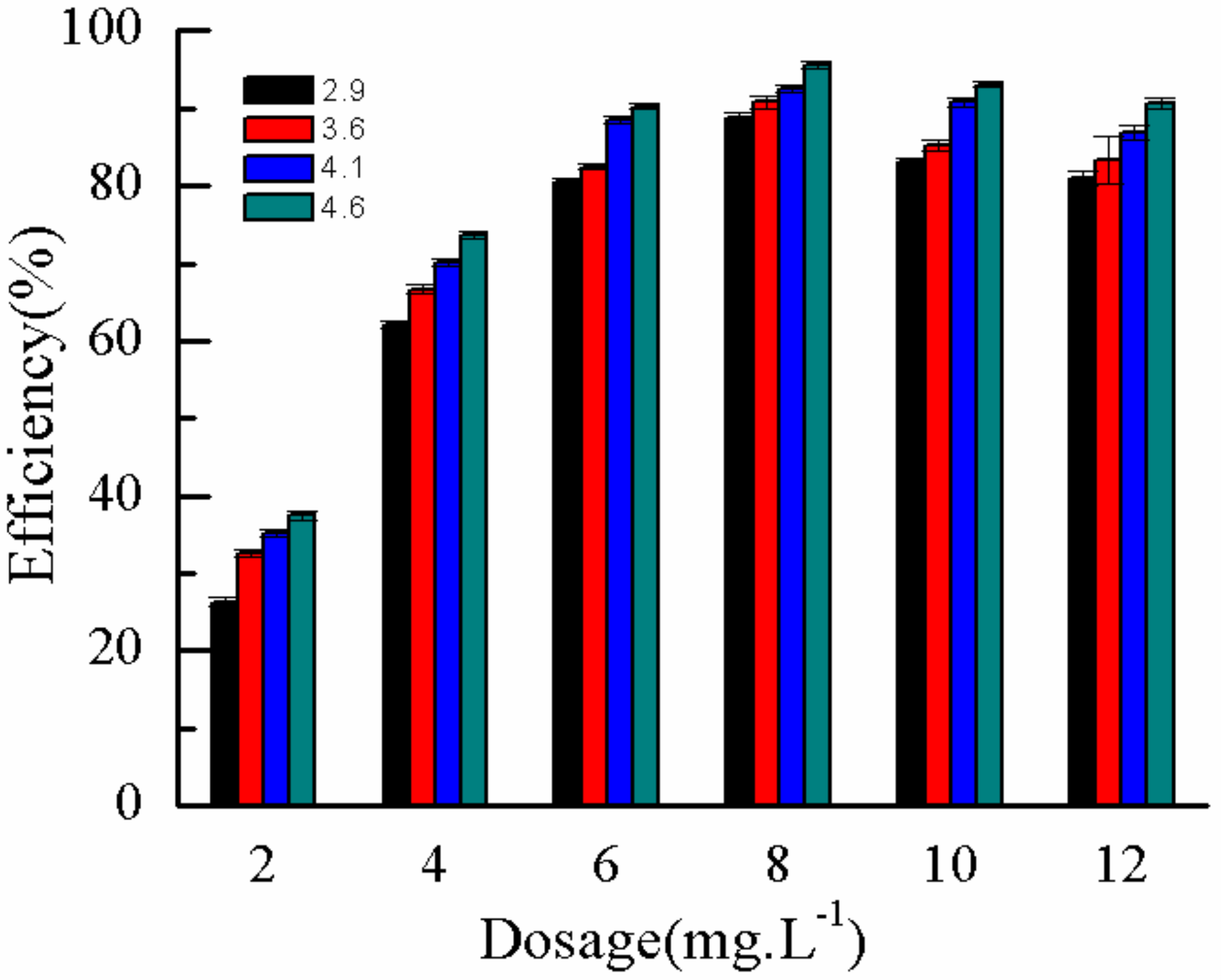
2.3.1. Effect of DMP Tnitial Concentration
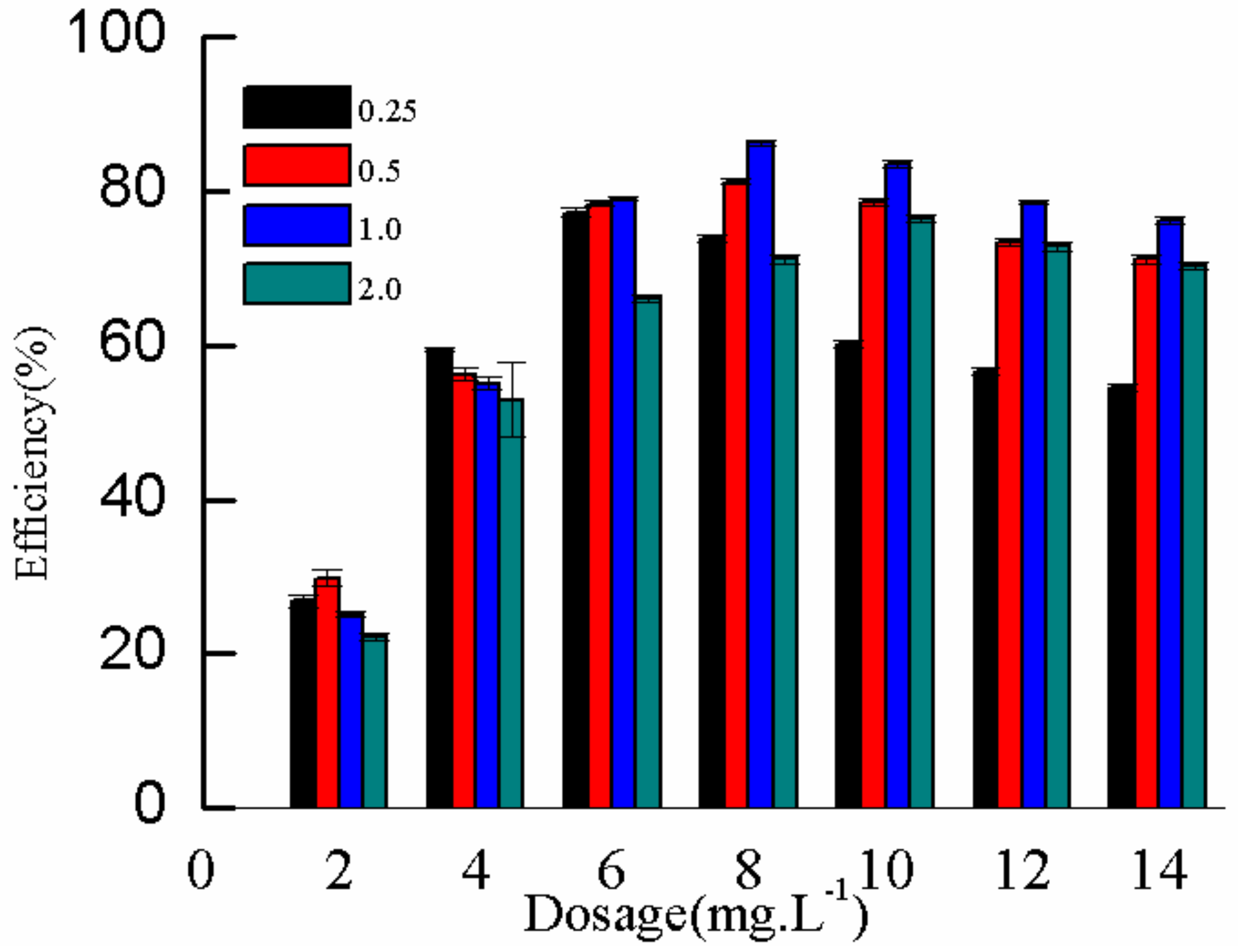
2.3.2. Effect of η
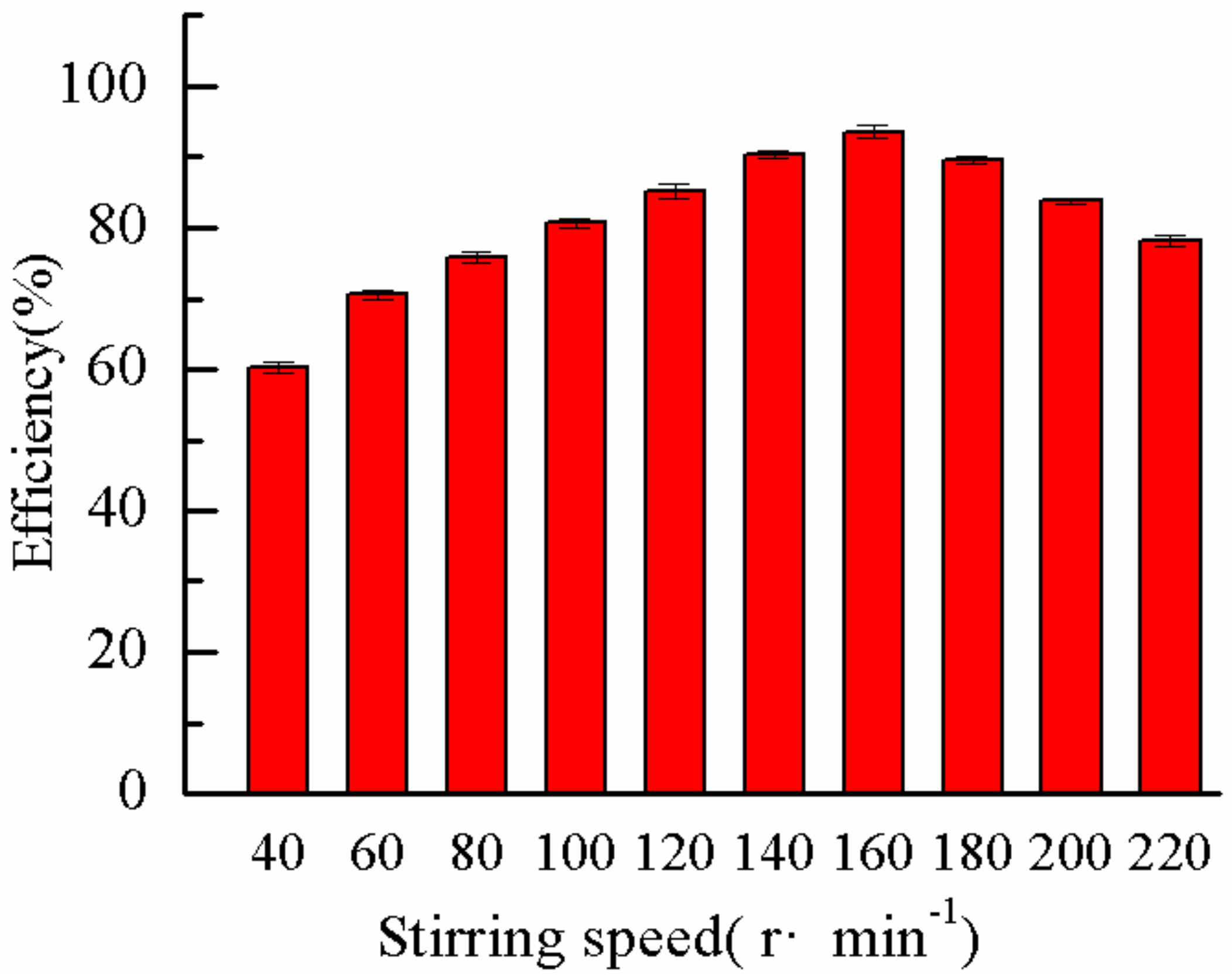
2.3.3. Effect of Stirring Speed
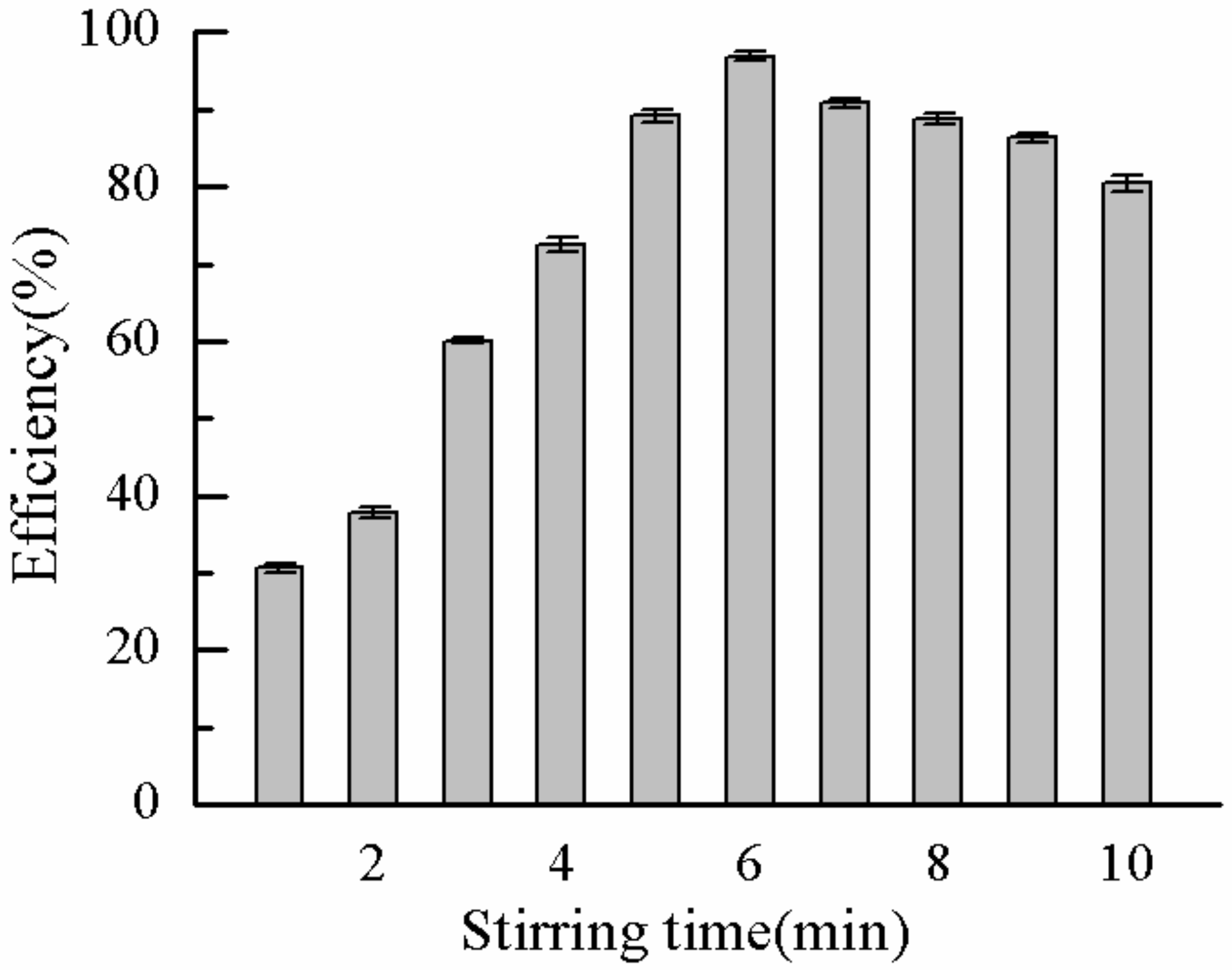
2.3.4. Effect of Stirring Time
2.3.5. Effect of Settling Time
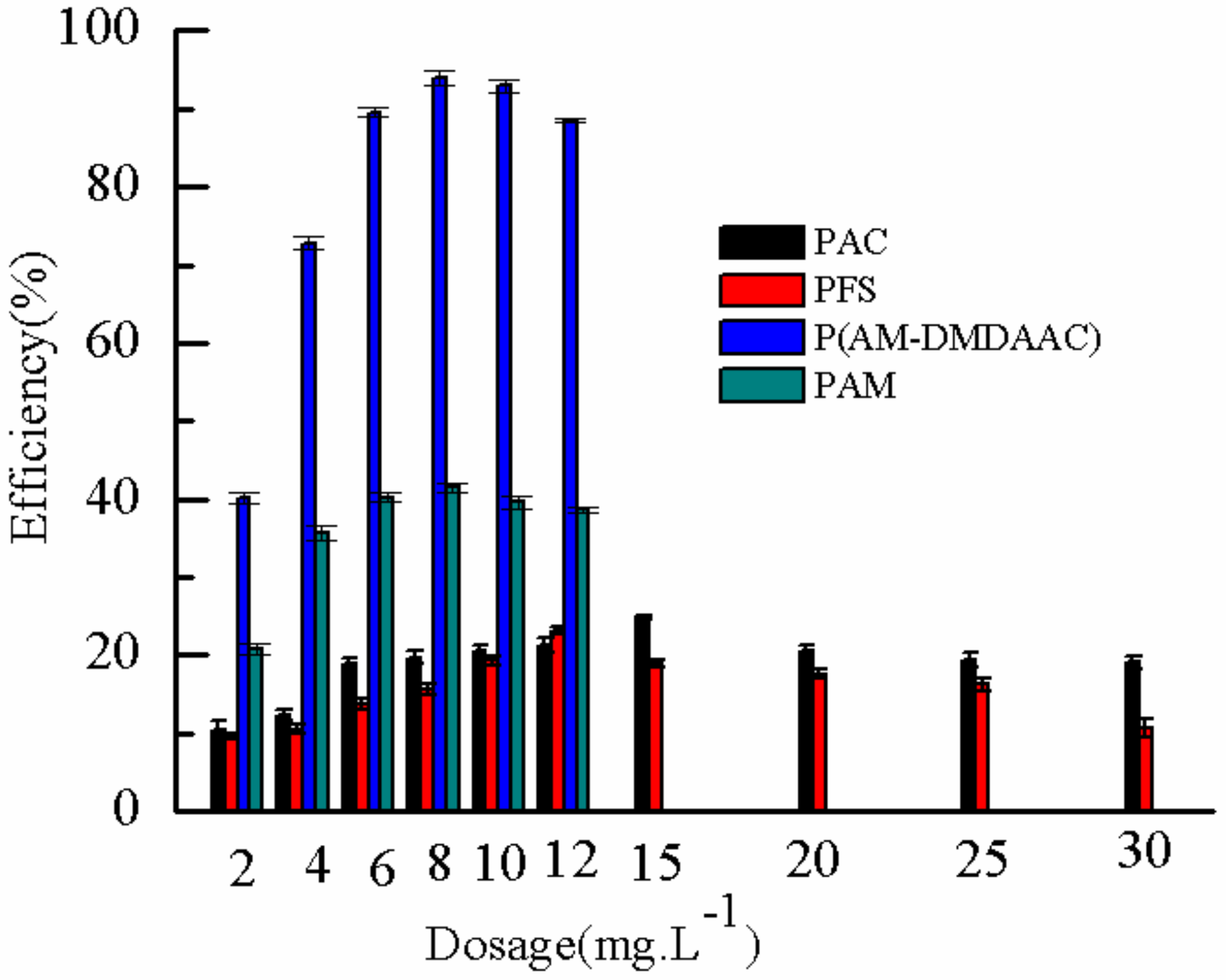
2.3.6. Comparison of Different Flocculants
3. Materials and Methods
3.1. Synthesis of P(AM–DMDAAC)
3.2. Characterization of P(AM–DMDAAC)
3.3. Flocculation Test
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Xiang, Y.; Gao, M.-L.; Ding, F.; Shen, T. The effiffifficient removal of dimethyl phthalate by three organo-vermiculites with imidazolium-based gemini surfactants in aqueous media. Colloid Surf. A 2019, 580, 123726. [Google Scholar] [CrossRef]
- Jing, W.-W.; Li, D.-Q.; Li, J.; Li, X.-F.; Wu, Z.-H.; Liu, Y.-L. Photodegradation of dimethyl phthalate (DMP) by UV–TiO2 in aqueous solution: Operational parameters and kinetic analysis. Int. J. Environ. Sci. Technol. 2017, 15, 969–976. [Google Scholar] [CrossRef]
- Jia, H.-Z.; Cao, Y.; Qu, G.-Z.; Wang, T.-C.; Guo, X.-T.; Xia, T.-J. Dimethyl phthalate contaminated soil remediation by dielectric barrier discharge: Performance and residual toxicity. Chem. Eng. J. 2018, 351, 1076–1084. [Google Scholar] [CrossRef]
- Gu, J.-L.; Zhao, H.; Liu, L.; Yang, D.; Chen, H.; Sun, T. Investigation of the binding interactions between dimethyl phthalate and its metabolite with bovine serum albumin by multispectroscopy. Spectrochim. Acta A 2019, 117771. [Google Scholar] [CrossRef]
- Liu, J.; Wang, K.; Jia, R.-B.; Wang, Z.-S. Removal of Esters from Drinking Water Using Ozone GAC Process. Environ. Sci. 2003, 24, 77–80. [Google Scholar]
- Gao, X.; Li, H.-M.; Guo, J.-S.; Yu, Z.-X.; Wang, F.-T.; Lu, L. Removal of Phthalate Easters from Drinking Water with Zeolite Filter Column. J. Civil. Arch. Environ. Eng. 2009, 31, 128–131. [Google Scholar]
- Wang, T.-C.; Jia, H.-Z.; Guo, X.-T.; Xia, T.-J.; Qu, G.-Z.; Sun, Q.-H.; Yin, X.-Q. Evaluation of the potential of dimethyl phthalate degradation in aqueous using sodium percarbonate activated by discharge plasma. Chem. Eng. J. 2018, 346, 65–76. [Google Scholar] [CrossRef]
- Busca, G.; Berardinelli, S.; Resini, C.; Arrighi, L. Technologies for the removal of phenol from fluid streams: A short review of recent developments. J. Hazard. Mater. 2008, 160, 265–288. [Google Scholar] [CrossRef]
- Iurascu, B.; Siminiceanu, I.; Vione, D.; Vicente, M.-A.; Gil, A. Phenol degradation in water through a heterogeneous photo-fenton process catalyzed by fe-treated laponite. Water Res. 2009, 43, 1313–1322. [Google Scholar] [CrossRef]
- Zazo, J.-A.; Casas, J.-A.; Mohedano, A.-F. Rodriguez JJ. Semicontinuous fenton oxidation of phenol in aqueous solution. a kinetic study. Water Res. 2009, 43, 4063–4069. [Google Scholar] [CrossRef]
- Ispas, C.-R.; Ravalli, M.-T.; Steere, A.; Andreescu, S. Multifunctional biomagnetic capsules for easy removal of phenol and bisphenol a. Water Res. 2010, 44, 1961–1969. [Google Scholar] [CrossRef] [PubMed]
- Grabowska, E.; Reszczyńska, J.; Zaleska, A. Mechanism of phenol photodegradation in the presence of pure and modified-TiO2: A review. Water Res. 2012, 46, 5453–5471. [Google Scholar] [CrossRef] [PubMed]
- Almeida, F.-T.; Ferreira, B.-C.; Moreira, A.-L.; Freitas, R.-P.; Gil, L.-F.; Gurgel, L.-V. Application of a new bifunctionalized chitosan derivative with zwitterionic characteristics for the adsorption of Cu(2+), Co(2+), Ni(2+), and oxyanions of Cr(6+) from aqueous solutions: Kinetic and equilibrium aspects. J. Colloid Interface Sci. 2015, 466, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Zewail, T.-M.; Yousef, N.-S. Chromium ions (Cr6+, & Cr3+) removal from synthetic wastewater by electrocoagulation using vertical expanded fe anode. J. Electroanal. Chem. 2014, 735, 123–128. [Google Scholar]
- Ye, S.; Zhang, M.; Yang, H.; Wang, H.; Xiao, S.; Liu, Y.; Wang, J.-H. Biosorption of Cu2+, Pb2+, and Cr6+, by a novel exopolysaccharide from arthrobacter, ps-5. Carbohyd. Polym. 2014, 101, 50–56. [Google Scholar]
- Teh, C.-Y.; Budiman, P.-M.; Shak, K.-P.-Y.; Wu, T.-Y. Recent advancement of coagulation-flocculation and its application in wastewater treatment. Ind. Eng. Chem. Res. 2016, 55, 4363–4389. [Google Scholar] [CrossRef]
- Kadooka, H.; Jami, M.-S.; Tanaka, T.; Iwata, M.-J. Mechanism of clarification of colloidal suspension using composite dry powdered flocculant. J. Water Process Eng. 2016, 11, 32–38. [Google Scholar] [CrossRef]
- Zhu, H.; Zhang, Y.; Yang, X.; Liu, H.; Shao, L.; Zhang, X.; Yao, J. One-step green synthesis of non-hazardous dicarboxyl cellulose flocculant and its flocculation activity evaluation. J. Hazard. Mater. 2015, 296, 1–8. [Google Scholar] [CrossRef]
- Zhu, G.-C.; Bian, Y.-N.; Hursthouse, A.-S.; Xu, S.-N.; Xiong, N.-N.; Wan, P. The role of magnetic MOFs nanoparticles in enhanced iron coagulation of aquatic dissolved organic matter. Chemosphere 2020, 125921. [Google Scholar] [CrossRef]
- Sun, Y.-J.; Zhou, S.-B.; Pan, S.-Y.; Zhu, S.-C.; Yu, Y.; Zheng, H.-L. Performance Evaluation and Optimization of Flocculation Process for Removing Heavy Metal. Chem. Eng. J. 2020, 385, 123911. [Google Scholar] [CrossRef]
- Zhu, G.-C.; Wang, Q.; Yin, J.; Li, Z.-W.; Zhang, P.; Ren, B.-Z.; Wan, P. Toward a better understanding of coagulation for dissolved organic nitrogen using polymeric zinc-iron-phosphate coagulant. Water Res. 2016, 100, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Zhu, G.-C.; Liu, J.-F.; Bian, Y.-N. Evaluation of cationic polyacrylamide-based hybrid coagulation for the removal of dissolved organic nitrogen. Environ. Sci. Pollut. Res. 2018, 25, 14447–14459. [Google Scholar] [CrossRef] [PubMed]
- Fosso-Kankeu, E.; Mittal, H.; Waanders, F.; Ntwampe, I.-O.; Ray, S.-S. Preparation and characterization of gum karaya hydrogel nanocomposite flocculant for metal ions removal from mine effluents. Int. J. Environ. Sci. Technol. 2016, 13, 711–724. [Google Scholar] [CrossRef]
- Wang, T.; Yang, W.-L.; Hong, Y.; Hou, Y.-L. Magnetic nanoparticles grafted with amino-riched dendrimer as magnetic flocculant for efficient harvesting of oleaginous microalgae. Chem. Eng. J. 2016, 297, 304–314. [Google Scholar] [CrossRef]
- Weng, B.; Xu, F.; Garza, G.; Alcoutlabi, M.; Salinas, A.; Lozano, K. The production of carbon nanotube reinforced poly(vinyl) butyral nanofibers by the forcespinning method. Polym. Eng. Sci. 2015, 55, 81–87. [Google Scholar] [CrossRef]
- Craciun, G.; Ighigeanu, D.; Manaila, E.; Stelescu, M.-D. Synthesis and Characterization of Poly(Acrylamide-Co-Acrylic Acid) Flocculant Obtained by Electron Beam Irradiation. Int. J. Pharm. 2015, 365, 89–99. [Google Scholar] [CrossRef]
- Zheng, H.-L.; Sun, Y.-J.; Zhu, C.-J.; Guo, J.-S.; Zhao, C.; Liao, Y.; Guan, Q.-Q. Uv-initiated polymerization of hydrophobically associating cationic flocculants: Synthesis, characterization, and dewatering properties. Chem. Eng. J. 2013, 234, 318–326. [Google Scholar] [CrossRef]
- Shak, K.-P.-Y.; Wu, T.-Y. Synthesis and characterization of a plant-based seed gum via etherification for effective treatment of high-strength agro-industrial wastewater. Chem. Eng. J. 2017, 307, 928–938. [Google Scholar] [CrossRef]
- Zheng, H.-L.; Sun, Y.-J.; Guo, J.-S.; Li, F.; Fan, W.; Liao, Y.; Guan, Q.-Q. Characterization and evaluation of dewatering properties of padb, a highly efficient cationic flocculant. Ind. Eng. Chem. Res. 2014, 53, 2572–2582. [Google Scholar] [CrossRef]
- Zhang, P.; Ren, B.-Z.; Zhou, Y.; Liu, K.-J. Removal of humic acid from aqueous solution by dimethyl diallyl ammonium chloride and acrylamide. Asian J. Chem. 2013, 25, 4431–4433. [Google Scholar] [CrossRef]
- Li, X.; Zheng, H.-L.; Gao, B.-Y.; Sun, Y.-J.; Tang, X.-M.; Xu, B.-C. Optimized preparation of micro-block CPAM by response surface methodology and evaluation of dewatering performance. RSC Adv. 2017, 7, 208–217. [Google Scholar] [CrossRef]
- Nie, R.C.; Guo, L.-Y.; Xu, C.-Y. Study on synthesis and flocculation property of cation-polyacrylamide. Int. J. Coal Sci. Technol. 2008, 14, 143–146. [Google Scholar] [CrossRef]
- Sun, Y.-J.; Zhu, C.-Y.; Sun, W.-Q.; Xu, Y.-H.; Xiao, X.; Zheng, H.-L. Plasma-initiated polymerization of chitosan-based CS-g-P(AM-DMDAAC) flocculant for the enhanced flocculation of low-algal-turbidity water. Carbohyd. Polym. 2017, 164, 222–232. [Google Scholar] [CrossRef]
- Liu, Z.-M.; Wei, Y.-L. Synthesis and Flocculating Properties of Cationic Polyacrylamide by Aqueous Two-phase Polymerization. Chin. J. Appl. Chem. 2011, 28, 874–878. [Google Scholar]
- Feng, L.; Zheng, H.-L.; Tang, X.-M.; Zheng, X.-Y.; Liu, S.; Sun, Q.; Wang, M.-X. The investigation of the specific behavior of a cationic block structure and its excellent flocculation performance in high-turbidity water treatment. RSC Adv. 2018, 8, 15119–15133. [Google Scholar] [CrossRef]
- Liu, Y.Z.; Zheng, H.-L.; Wang, Y.-L.; Zheng, X.-Y.; Wang, M.-X.; Ren, J.; Zhao, C.-L. Synthesis of a cationic polyacrylamide by a photocatalytic surface-initiated method and evaluation of its flocculation and dewatering performance: Nano-TiO 2 as a photo initiator. RSC Adv. 2018, 8, 28329–28340. [Google Scholar] [CrossRef]
- Xu, K.; Wang, H.-W.; Liang, X.-C.; Tan, Y.; Yao, X.-P.; Wang, P.-X. A Novel Hyperbranched Polymeric Flocculant for Waste-Water Treatment. J. Polym. Environ. 2018, 26, 2782–2792. [Google Scholar] [CrossRef]
- Pal, S.; Sen, G.; Ghosh, S.; Singh, R.-P. High performance polymeric flocculants based on modified polysaccharides—Microwave assisted synthesis. Carbohyd. Polym. 2012, 87, 336–342. [Google Scholar] [CrossRef]
- Zheng, H.-L.; Ma, J.-Y.; Zhu, C.-J.; Zhang, Z.; Liu, L.-W.; Sun, Y.-J.; Tang, X.-M. Synthesis of anion polyacrylamide under uv initiation and its application in removing dioctyl phthalate from water through flocculation process. Sep. Purif. Technol. 2014, 123, 35–44. [Google Scholar] [CrossRef]
- Hu, X.-M.; Mei, Z.-Q.; Liu, H.-Y.; Huang, Z.-T.; Wang, L.-F. Study on the microwave induced synthesis of tetraphenyl porphyrin. J. South China Univ. Technol. (Nat. Sci.) 1999, 27, 11–15. [Google Scholar]
- Hu, X.-M.; Mei, Z.-Q.; Huang, Z.-T.; Lai, S.-L. The synthesis of copper phthalocyanine and its sulfonat ion under microwave irradiat ion. J. South China Univ. Technol. (Nat. Sci.) 1999, 25, 34–38. [Google Scholar]
- Ren, E.-H.; Xiao, H.-G.; Guo, R.-H.; Lin, S.-J. Effects of cationic degree on decolorization properties of hydrophobic cationic flocculant. Textile Dye. Finish. J. 2019, 41, 21–25. [Google Scholar]
- Zheng, H.-L.; Tang, X.; Shen, L.-X.; Gao, X.; Wang, W.; You, Y.-F. Synthesis, characterization and application of cationic P(AM-DMC)used for sludge dewatering. J. Chongqing Univ. 2010, 30, 115–122. [Google Scholar]
- Zhang, Z.-Q. Ultrasonic Assisted Synthesis of PDA and Its Characterization and Application. Master’s Thesis, Chongqing University, Chongqing, China, 2012. [Google Scholar]
- Li, X.; Zheng, H.-L.; Gao, B.-Y.; Zhao, C.; Sun, Y.-J. UV-initiated polymerization of acid and alkali-resistant cationic flocculant P(AM-MAPTAC): Synthesis, characterization, and application in sludge dewatering. Sep. Purif. Technol. 2017, 187, 244–254. [Google Scholar] [CrossRef]
- Chen, H.-X.; Tang, H.-M.; Wu, X.-Y.; Liu, Y.-G.; Bai, J.-H.; Zhao, F. Synthesis, Characterization, and Property Evaluation of a Hydrophobically Modified Polyacrylamide as Enhanced Oil Recovery Chemica. J. Dispersion Sci. Technol. 2016, 37, 486–495. [Google Scholar] [CrossRef]
- Zhang, P.; Liao, L.-N.; Zhu, G.-C. Performance of PATC-PDMDAAC Composite Coagulants in Low-Temperature and Low-Turbidity Water Treatment. Materials 2019, 12, 2824. [Google Scholar] [CrossRef]
- Ma, J.-Y.; Shi, J.; Ding, H.-C.; Zhu, G.-C.; Fu, K.; Fu, X. Synthesis of cationic poly-acrylamide by low-pressure UV initiation for turbidity water flocculation. Chem. Eng. J. 2017, 312, 20–29. [Google Scholar]
- Feng, L.; Liu, S.; Zheng, H.-L.; Liang, J.-J.; Sun, Y.-J.; Zhang, S.-X.; Chen, X. Using ultrasonic (US)-initiated template copolymerization for preparation of an enhanced cationic polyacrylamide (CPAM) and its application in sludge dewatering. Ultrason. Sonochem. 2018, 44, 53–63. [Google Scholar] [CrossRef]
- Ma, J.-Y.; Fu, K.; Shi, J.; Sun, Y.-J.; Zhang, X.-X.; Ding, L. Ultraviolet-assisted synthesis of polyacrylamide-grafted chitosan nanoparticles and flocculation performance. Carbohy. Polym. 2016, 151, 565–575. [Google Scholar] [CrossRef]
- Ou, C.-Y.; Zhang, C.-H.; Li, S.-D.; Yang, L.; Dong, J.-J.; Mo, X.-L.; Zeng, M.-T. Thermal degradation kinetics of chitosan-cobalt complex as studied by thermogravimetric analysis. Carbohy. Polym. 2010, 82, 1284–1289. [Google Scholar] [CrossRef]
- Zheng, H.-L.; Ma, J.-Y.; Zhai, J.; Zhu, C.J.; Tang, X.-M.; Liao, Y.; Sun, Y.-J. Optimization of flocculation process by response surface methodology for diethyl phthalate removal using anionic polyacrylamide. Desalin. Water Treat. 2014, 52, 5390–5400. [Google Scholar] [CrossRef]
- Zhang, P.; Zheng, H.-L.; Deng, X.-L.; Jiang, S.-J.; Wang, J.-J.; Liu, L.; Yang, B. RemoVal of Dmethyl Phthalate from Water with Enhanced Coagulation Technology. J. Civil Arch. Environ. Eng. 2011, 33, 157–161. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |


© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, P.; Zhao, D.Q. Characterization and Dimethyl Phthalate Flocculation Performance of the Cationic Polyacrylamide Flocculant P(AM-DMDAAC) Produced by Microwave-Assisted Synthesis. Molecules 2020, 25, 624. https://doi.org/10.3390/molecules25030624
Zhang P, Zhao DQ. Characterization and Dimethyl Phthalate Flocculation Performance of the Cationic Polyacrylamide Flocculant P(AM-DMDAAC) Produced by Microwave-Assisted Synthesis. Molecules. 2020; 25(3):624. https://doi.org/10.3390/molecules25030624
Chicago/Turabian StyleZhang, Peng, and Dong Qin Zhao. 2020. "Characterization and Dimethyl Phthalate Flocculation Performance of the Cationic Polyacrylamide Flocculant P(AM-DMDAAC) Produced by Microwave-Assisted Synthesis" Molecules 25, no. 3: 624. https://doi.org/10.3390/molecules25030624
APA StyleZhang, P., & Zhao, D. Q. (2020). Characterization and Dimethyl Phthalate Flocculation Performance of the Cationic Polyacrylamide Flocculant P(AM-DMDAAC) Produced by Microwave-Assisted Synthesis. Molecules, 25(3), 624. https://doi.org/10.3390/molecules25030624





