Thymol Chemotype Origanum vulgare L. Essential Oil as a Potential Selective Bio-Based Herbicide on Monocot Plant Species
Abstract
1. Introduction
2. Results
2.1. GC-MS Analyzes
2.2. Phytotoxic Activity
2.2.1. Germination
2.2.2. Root Length
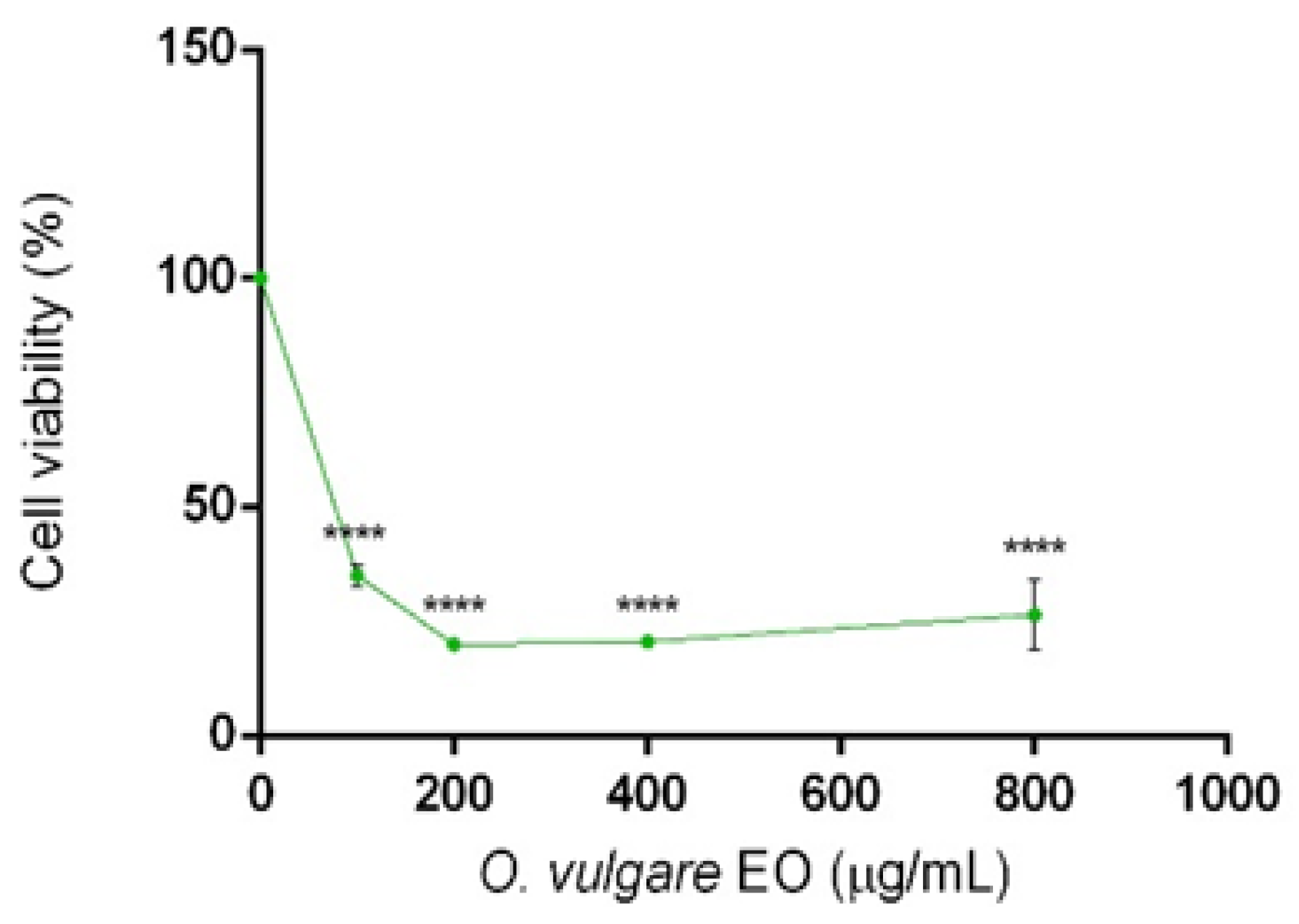
2.3. Cytotoxic Activity
2.4. In Vitro Antibacterial Effect
2.5. In Vitro Antifungal Effect
2.6. Free Radical-Scavenging Capacity
3. Discussion
4. Materials and Methods
4.1. Essential Oil
4.2. Tested Plant Seeds
4.3. Phytotoxic Assay
4.4. GC/MS Analysis
4.5. Antibacterial Activity
4.5.1. Tested Bacteria
4.5.2. Bactericidal Assay
4.6. Antifungal Activity
4.6.1. Tested Fungi
4.6.2. Fungicidal Assay
4.7. Free Radical-Scavenging Capacity
4.7.1. Ascorbic Acid
4.7.2. Statistical Analysis
4.8. Cytotoxic Activity
4.8.1. Cell Cultures
4.8.2. MTT Assay
4.9. Data Analysis
4.9.1. Germination Activity
4.9.2. Roots Length
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Duke, S.O. Why have no new herbicide modes of action appeared in recent years? Pest Manag. Sci. 2012, 68, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Korres, N.E.; Burgos, N.R.; Travlos, I.; Vurro, M.; Gitsopoulos, T.K.; Varanasi, V.K.; Duke, S.O.; Kudsk, P.; Brabham, C.; Rouse, C.E.; et al. New directions for integrated weed management: Modern technologies, tools and knowledge discovery. Adv. Agron. 2019, 155, 243–319. [Google Scholar]
- Dayan, F.E.; Owens, D.K.; Duke, S.O. Rationale for a natural products approach to herbicide discovery. Pest. Manag. Sci. 2012, 68, 519–528. [Google Scholar] [CrossRef] [PubMed]
- Gerwick, B.C.; Sparks, T.C. Natural products for pest control: An analysis of their role, value and future. Pest. Manag. Sci. 2014, 70, 1169–1185. [Google Scholar] [CrossRef] [PubMed]
- Della Pepa, T.; Elshafie, H.S.; Capasso, R.; De Feo, V.; Camele, I.; Nazzaro, F.; Scognamiglio, M.R.; Caputo, L. Antimicrobial and phytotoxic activity of Origanum heracleoticum and O. majorana essential oils growing in Cilento (Southern Italy). Molecules 2019, 24, 2576. [Google Scholar] [CrossRef] [PubMed]
- Keifer, M.C.; Firestone, J. Neurotoxicity of pesticides. J. Agromed. 2007, 12, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Jankowska, M.; Rogalska, J.; Wyszkowska, J.; Stankiewicz, M. Molecular targets for components of essential oils in the insect nervous system—A review. Molecules 2018, 23, 34. [Google Scholar] [CrossRef]
- Araniti, F.; Landi, M.; Lupinia, A.; Sunseria, F.; Guidib, L.; Abenavolia, M.R. Origanum vulgare essential oils inhibit glutamate and aspartate metabolism altering the photorespiratory pathway in Arabidopsis thaliana seedlings. J. Plant. Physiol. 2018, 231, 297–309. [Google Scholar] [CrossRef]
- Lins, L.; Dal Maso, S.; Foncoux, B.; Kamili, A.; Laurin, Y.; Genva, M.; Jijakli, M.H.; De Clerck, C.; Fauconnier, M.L.; Deleu, M. Insights into the relationships between herbicide activities, molecular structure and membrane interaction of cinnamon and citronella essential oils components. Int. J. Mol. Sci. 2019, 20, 4007. [Google Scholar] [CrossRef]
- López, M.D.; Jórdan, J.J.; Pascual-Villalobo, M.J. Toxic compounds in essential oils of coriander, caraway and basil active against stored rice pests. J. Stored Prod. Res. 2008, 44, 273–278. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Camele, I. An Overview of the biological effects of some mediterranean essential oils on human health. BioMed Res. Int. 2017, 2017, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Elshafie, H.S.; Gruľová, D.; Baranová, B.; Caputo, L.; De Martino, L.; Sedlák, V.; Camele, I.; De Feo, V. Antimicrobial activity and chemical composition of essential oil extracted from Solidago canadensis L. growing wild in Slovakia. Molecules 2019, 24, 1206. [Google Scholar] [CrossRef] [PubMed]
- Arminante, F.; De Falco, E.; De Feo, V.; De Martino, L.; Mancini, E.; Quaranta, E. Allelopathic Activity of Essential Oils from Mediterranean Labiatae. Acta Hort. 2006, 723, 347–356. [Google Scholar] [CrossRef]
- Kokkini, S.; Karousou, R.; Hanlidou, E. Herbs of the Labiatae. In Encyclopedia of Food Sciences and Nutrition, 2nd ed.; Caballero, B., Ed.; Academic Press: San Diego, CA, USA, 2003; pp. 3082–3090. [Google Scholar]
- Robu, T.; Milică, C. Plante medicinale autohtone; Institutul European: Iaşi, Romania, 2004; p. 308. ISBN 973-611-277-2. [Google Scholar]
- Fikry, S.; Khalil, N.; Salama, O. Chemical profiling, biostatic and biocidal dynamics of Origanum vulgare L. essential oil. AMB Express 2019, 9, 41. [Google Scholar] [CrossRef]
- Alagawany, M.; Abd El-Hack, M.A.; Farag, M.; Shaheen, H.; Abdel-Latif, M.; Noreldin, A.; Patra, A. The usefulness of oregano and its derivatives in poultry nutrition. World’s Poult. Sci. J. 2018, 74, 463–474. [Google Scholar] [CrossRef]
- Didry, N.; Dubreuil, L.; Pinkas, M. Antibacterial activity of thymol, carvacrol and cinnamaldehyde alone or in combination. Die Pharm. 1993, 48, 301–304. [Google Scholar]
- Lee, C.-J.; Chen, L.-W.; Chen, L.-G.; Chang, T.-L.; Huang, C.-W.; Huang, M.-C.; Wang, C.-C. Correlations of the components of tea tree oil with its antibacterial effects and skin irritation. J. Food Drug Anal. 2013, 21, 169–176. [Google Scholar] [CrossRef]
- Magi, G.; Marini, E.; Facinelli, B. Antimicrobial activity of essential oils and carvacrol, and synergy of carvacrol and erythromycin, against clinical, erythromycin-resistant Group A streptococci. Front. Microbiol. 2015, 6, 165. [Google Scholar] [CrossRef]
- Sharifi-Rad, M.; Varoni, E.M.; Iriti, M.; Martorell, M.; Setzer, W.N.; Contreras, M.D.M.; Salehi, B.; Soltani-Nejad, A.; Rajabi, S.; Tajbakhsh, M.; et al. Carvacrol and human health: A comprehensive review. Phytother. Res. 2018, 32, 1675–1687. [Google Scholar] [CrossRef]
- Kintzios, S. The Genera Origanum and Lippia, 1st ed.; Taylor & Francis: London, UK, 2004. [Google Scholar]
- Elshafie, H.S.; Armentano, M.F.; Carmosino, M.; Bufo, S.A.; De Feo, V.; Camele, I. Cytotoxic activity of Origanum vulgare L. on hepatocellular carcinoma cell line HepG2 and evaluation of its biological activity. Molecules 2017, 22, 1435. [Google Scholar] [CrossRef]
- Elshafie, H.S.; Mancini, E.; Sakr, S.; De Martino, L.; Mattia, C.A.; De Feo, V.; Camele, I. Antifungal activity of some constituents of Origanum vulgare L. essential oil against postharvest disease of peach fruit. J. Med. Food 2015, 18, 929–934. [Google Scholar] [CrossRef] [PubMed]
- Mancini, E.; Camele, I.; Elshafie, H.S.; De Martino, L.; Pellegrino, C.; Grulova, D.; De Feo, V. Chemical Composition and biological activity of the essential oil of Origanum vulgare ssp. hirtum from different areas in the Southern Apennines (Italy). Chem. Biodivers. 2014, 11, 639–651. [Google Scholar] [CrossRef] [PubMed]
- Adebayo, O.; Dang, T.; Bélanger, A.; Khanizadeh, S. Antifungal studies of selected essential oils and a commercial formulation against Botrytis Cinerea. J. Food Res. 2013, 2, 217–226. [Google Scholar] [CrossRef]
- Kordali, S.; Cakir, A.; Özer, H.; Cakmakci, R.; Kesdek, M.; Mete, E. Antifungal, phytotoxic and insecticidal properties of essential oil isolated from Turkish Origanum acutidens and its three components, carvacrol, thymol and p-cymene. Bioresour. Technol. 2008, 99, 8788–8795. [Google Scholar] [CrossRef]
- Onaran, A.; Yilar, M.; Belguzar, S.; Bayan, Y.; Aksit, H. Antifungal and bioherbicidal properties of essential oils of Thymus fallax Fish & Mey. Origanum vulgare L. and Mentha dumetorum Schult. Asian J. Chem. 2014, 26, 5159–5164. [Google Scholar]
- Fouad, R.; Bousta, D.; Lalami, A.E.O.; Chahdi, F.O.; Amri, I.; Jamoussi, B.; Greche, H. Chemical composition and herbicidal effects of essential oils of Cymbopogon citratus (DC) Stapf, Eucalyptus cladocalyx, Origanum vulgare L. and Artemisia absinthium L. cultivated in Morocco. J. Essent. Oil Bear. Plants 2015, 18, 112–123. [Google Scholar] [CrossRef]
- FAOSTAT. Food and Agriculture Organization of the United Nations, Statistics Division. Forestry Production and Trade. Available online: http://www.fao.org/faostat/en/#data/FO (accessed on 4 April 2019).
- Regulation, E.C. Regulation (EC) No 1107/2009 of the European Parliament and of the Council of 21 October 2009. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32009R1107 (accessed on 5 January 2020).
- Maes, C.; Bouquillon, S.; Fauconnier, M.-L. Encapsulation of essential oils for the development of biosourced pesticides with controlled release: A Review. Molecules 2019, 24, 2539. [Google Scholar] [CrossRef]
- Bahuguna, A.; Khan, I.; Bajpai, V.K.; Kang, S.C. MTT assay to evaluate the cytotoxic potential of a drug. Bangladesh J. Pharmacol. 2017, 12, 8. [Google Scholar] [CrossRef]
- Berridge, M.V.; Herst, P.M.; Tan, A.S. Tetrazolium dyes as tools in cell biology: New insights into their cellular reduction. Biotechnol. Annu. Rev. 2005, 11, 127–152. [Google Scholar]
- Sebaugh, J.L. Guidelines for accurate EC50/IC50 estimation. Pharm. Stat. 2011, 10, 128–134. [Google Scholar] [CrossRef]
- Argyropoulos, E.I.; Eleftherohorinos, I.G.; Vokou, D. In vitro evaluation of essential oils from Mediterranean aromatic plants of the lamiaceae for weed control in tomato and cotton crops. Allelopath. J. 2008, 22, 69–78. [Google Scholar]
- De Mastro, G.; Fracchiolla, M.; Verdini, L.; Montemurro, P. Oregano and itspotential use as bioherbicide. Acta Hortic. 2006, 723, 335–346. [Google Scholar] [CrossRef]
- De Almeida, L.F.R.; Frei, F.; Mancini, E.; De Martino, L.; De Feo, V. Phytotoxic activities of mediterranean essential oils. Molecules 2010, 15, 4309–4323. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, M.D.; Blázquez, M.A. Herbicidal value of essential oils from oregano-like flavour species. Food Agric. Immunol. 2017, 28, 1168–1180. [Google Scholar] [CrossRef]
- Szepanowski, F.; Kleinschnitz, C.; Stettner, M. Glyphosate-based herbicide: A risk factor for demyelinating conditions of the peripheral nervous system? Neural Regen. Res. 2019, 14, 2079–2080. [Google Scholar] [CrossRef]
- Al-Kalaldeh, J.Z.; Abu-Dahab, R.; Afifi, F.U. Volatile oil composition and antiproliferative activity of Laurus nobilis, Origanum syriacum, Origanum vulgare, and Salvia triloba against human breast adenocarcinoma cells. Nutr. Res. 2010, 30, 271–278. [Google Scholar] [CrossRef]
- Begnini, K.R.; Nedel, F.; Lund, R.G.; Carvalho, P.H.D.A.; Rodrigues, M.R.A.; Beira, F.T.A.; Del-Pino, F.A.B. Composition and antiproliferative effect of essential oil of Origanum vulgare against tumor cell lines. J. Med. Food 2014, 17, 1129–1133. [Google Scholar] [CrossRef]
- Geran, R.I.; Greenberg, N.H.; Macdonald, M.M.; Schumacher, A.M.; Abbott, B.J. Protocols for screening chemical agents and natural products against animal tumours and other biological systems. Cancer Chemother. Rep. 1972, 3, 59–61. [Google Scholar]
- Kaskatepe, B.; Yildiz, S.S.; Kiymaci, M.E.; Yazgan, A.N.; Cesur, S.; Erdem, S.A. Chemical composition and antimicrobial activity of the commercial Origanum onites L. oil against nosocomial carbapenem resistant extended spectrum beta lactamase producer Escherichia coli isolates. Acta Boil. Hung. 2017, 68, 466–476. [Google Scholar] [CrossRef]
- Pezzani, R.; Vitalini, S.; Iriti, M. Bioactivities of Origanum vulgare L.: An update. Phytochem. Rev. 2017, 16, 1253–1268. [Google Scholar] [CrossRef]
- Moshayedi, S.; Shahraz, F.; Schaffner, D.W.; Khanlarkhani, A.; Shojaee-Aliabadi, S.; Shahnia, M.; Khaksar, R. In Vitro control of Enterococcus faecalis by Zataria multilfolira Boiss, Origanum vulgare L. and Mentha pulegium essential oils. J. Food Saf. 2013, 33, 327–332. [Google Scholar] [CrossRef]
- Teixeira, B.; Marques, A.; Ramos, C.; Serrano, C.; Matos, O.; Neng, N.R.; Nogueira, J.M.F.; Saraiva, J.A.; Nunes, M.L. Chemical composition and bioactivity of different oregano (Origanum vulgare) extracts and essential oil. J. Sci. Food. Agric. 2013, 93, 2707–2714. [Google Scholar] [CrossRef] [PubMed]
- Kačániová, M.; Vukovič, N.; Horská, E.; Šalamon, I.; Bobková, A.; Hleba, L.; Mellen, M.; Vatľák, A.; Petrová, J.; Bobko, M. Antibacterial activity against Clostridium genus and antiradical activity of the essential oils from different origin. J. Environ. Sci. Heal. Part B 2014, 49, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Jnaid, Y.; Yacoub, R.; Al-Biski, F. Antioxidant and antimicrobial activities of Origanum vulgare essential oil. Int. Food Res. J. 2016, 23, 1706–1710. [Google Scholar]
- Adrar, N.; Oukil, N.; Bedjou, F. Antioxidant and antibacterial activities of Thymus numidicus and Salvia officinalis essential oils alone or in combination. Ind. Crop. Prod. 2016, 88, 112–119. [Google Scholar] [CrossRef]
- Adams, R.P. Identification of essential oil components by Gas Chromatography/Mass Spectrometry; Allured: Carol Stream, IL, USA, 2007. [Google Scholar]
- Hochmuth, D. Mass Spectral Library “Terpenoids and Related Constituents of Essential oils”; Library of MassFinder 3: Hamburg, Germany, 2006. [Google Scholar]
- Bhunia, A.; Johnson, M.C.; Ray, B. Purification, characterization and antimicrobial spectrum of a bacteriocin produced by Pediococcus acidilactici. J. Appl. Bacteriol. 1988, 65, 261–268. [Google Scholar] [CrossRef]
- King, E.O.; Ward, M.K.; Raney, D.E. Two simple media for the demonstration of pyocyanin and fluorescin. J. Lab. Clin. Med. 1954, 44, 301–307. [Google Scholar]
- Soylu, E.M.; Kurt, S.; Soylu, S. In vitro and in vivo antifungal activities of the essential oils of various plants against tomato grey mould disease agent Botrytis cinerea. Int. J. Food Microbiol. 2010, 143, 183–189. [Google Scholar] [CrossRef]
- Zygadlo, J.A.; Guzmán, C.A.; Grosso, N.R. Antifungal properties of the leaf oils of Tagetes minuta L. and T. filifolia Lag. J. Essent. Oil Res. 1994, 6, 617–621. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.; Berset, C. Review: Use of a free radical method to evaluate antioxidant activity. Food Sci. Technol. 1995, 28, 25–30. [Google Scholar]
- Lee, S.K.; Zakaria, H.M.; Cheng, H.; Luyengi, L.; Gamez, E.J.; Mehta, R.G.; Kinghorn, A.D.; Pezzuto, J.M. Evaluation of the antioxidant potential of natural products. Comb. Chem. High Throughput Screen. 1998, 1, 35–46. [Google Scholar] [PubMed]
- Caputo, L.; Souza, L.F.; Alloisio, S.; Cornara, L.; De Feo, V. Coriandrum sativum and Lavandula angustifolia essential oils: Chemical composition and activity on central nervous system. Int. J. Mol. Sci. 2016, 17, 1999. [Google Scholar] [CrossRef] [PubMed]
- Caputo, L.; Nazzaro, F.; Souza, L.F.; Aliberti, L.; De Martino, L.; Fratianni, F.; Coppola, R.; De Feo, V. Laurus nobilis: Composition of essential oil and its biological activities. Molecules 2017, 22, 930. [Google Scholar] [CrossRef] [PubMed]
- Van Meerloo, J.; Kaspers, G.J.L.; Cloos, J. Cell sensitivity assays: The MTT assay. Adv. Struct. Saf. Stud. 2011, 731, 237–245. [Google Scholar]
- Hammer, O.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological statistics software package for education and data analysis. Palaeontol. Electron. 2001, 4, 1–9. [Google Scholar]



| No. | Name of Compound | KIexp | KI lit | % | Identif. | |
|---|---|---|---|---|---|---|
| 1. | α-pinene | 938 | 936 | 0.2 | KI, MS, S | M |
| 2. | camphene | 951 | 950 | 0.2 | KI, MS | M |
| 3. | sabinene | 976 | 973 | 0.1 | KI, MS | M |
| 4. | β-pinene | 980 | 978 | 0.3 | KI, MS, S | M |
| 5. | α-terpinene | 1016 | 1013 | 0.7 | KI, MS, S | M |
| 6. | p-cymene | 1020 | 1015 | 5.7 | KI, MS, S | M |
| 7. | 1,8-cineole | 1033 | 1024 | 0.6 | KI, MS, S | MO |
| 8. | (Z)-β-ocimene | 1035 | 1029 | T | KI, MS, S | M |
| 9. | γ-terpinene | 1060 | 1051 | 2.5 | KI, MS, S | M |
| 10. | terpinolene | 1088 | 1082 | T | KI, MS, S | M |
| 11. | linalool | 1098 | 1086 | 2.6 | KI, MS, S | MO |
| 12. | camphor | 1121 | 1123 | 0.7 | KI, MS, S | MO |
| 13. | L-trans-pinocarveol | 1130 | 1125 | T | KI, MS | MO |
| 14. | borneol | 1152 | 1150 | 0.8 | KI, MS, S | MO |
| 15. | terpinen-4-ol | 1160 | 1164 | 0.7 | KI, MS | MO |
| 16. | α-terpineol | 1178 | 1176 | 0.4 | KI, MS | MO |
| 17. | carvone | 1217 | 1214 | T | KI, MS, S | MO |
| 18. | carvotanacetone | 1230 | 1220 | T | KI, MS | MO |
| 19. | thymol | 1270 | 1267 | 76.0 | KI, MS, S | MO |
| 20. | carvacrol | 1282 | 1278 | 3.2 | KI, MS, S | MO |
| 21. | eugenol | 1333 | 1331 | 0.1 | KI, MS | MO |
| 22. | α-cubebene | 1354 | 1355 | 0.3 | KI, MS | S |
| 23. | α-gurjunene | 1411 | 1413 | T | KI, MS | S |
| 24. | α-himachalene | 1449 | 1450 | 0.1 | KI, MS | S |
| 25. | humulene | 1454 | 1455 | T | KI, MS | S |
| 26. | allo-aromadendrene | 1461 | 1462 | T | KI, MS | S |
| 27. | β-guaiene | 1490 | 1488 | T | KI, MS | S |
| 28. | valencene | 1494 | 1494 | 0.1 | KI, MS | S |
| 29. | α-muurolene | 1495 | 1496 | T | KI, MS | S |
| 30. | γ-cadinene | 1512 | 1507 | 0.1 | KI, MS | S |
| 31. | calamenene | 1513 | 1517 | T | KI, MS | S |
| 32. | β-cadinene | 1520 | 1526 | 0.4 | KI, MS | S |
| 33. | α-calacorene | 1534 | 1527 | 0.1 | KI, MS | S |
| 34. | elemol | 1539 | 1541 | T | KI, MS | SO |
| 35. | caryophyllene oxide | 1580 | 1578 | 0.4 | KI, MS | SO |
| 36. | globulol | 1583 | 1589 | T | KI, MS | SO |
| 37. | cedrol | 1598 | 1603 | T | KI, MS | SO |
| 38. | γ-eudesmol | 1620 | 1618 | T | KI, MS | SO |
| 39. | allo-aromadendrene epoxide | 1621 | 1623 | 0.1 | KI, MS | SO |
| 40. | tau.cadinol | 1634 | 1633 | T | KI, MS | SO |
| 41. | tau.muurolol | 1635 | 1633 | T | KI, MS | SO |
| 42. | cubenol | 1636 | 1630 | T | KI, MS | SO |
| Total Identified | 96.4 |
| Applied Doses of EO (µg/mL) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Control | 100 | 50 | 25 | 10 | 5 | 2.5 | 1.25 | 0.625 | 0.25 | 0.125 | 0.0625 | |
| Sinapis alba | ||||||||||||
| Numb. GS | 23 | 22 | 23 | 22 | 25 | 24 | 21 | 24 | 22 | 23 | 27 | 27 |
| % GS | 76.67 | 73.33 | 76.67 | 73.33 | 83.33 | 80.00 | 70.00 | 80.00 | 73.33 | 76.67 | 90.00 | 90.00 |
| Lepidium sativum | ||||||||||||
| Numb. GS | 29 | 28 | 27 | 27 | 27 | 30 | 29 | 29 | 30 | 30 | 30 | 30 |
| % GS | 96.67 | 93.33 | 90.00 | 90.00 | 90.00 | 100 | 96.67 | 96.67 | 100 | 100 | 100 | 100 |
| Hordeum vulgare | ||||||||||||
| Numb. GS | 17 | 9 | 13 | 22 | 17 | 16 | 19 | 17 | 16 | 19 | 13 | 6 |
| % GS | 56.67 | 30.00 | 43.33 | 73.33 | 56.67 | 53.33 | 63.33 | 56.67 | 53.33 | 63.33 | 43.33 | 53.33 |
| Triticum aestivum | ||||||||||||
| Numb. GS | 18 | 19 | 23 | 23 | 22 | 22 | 22 | 24 | 18 | 18 | 23 | 21 |
| % GS | 60 | 63.33 | 76.67 | 76.67 | 73.33 | 73.33 | 73.33 | 80.00 | 60.00 | 60.00 | 76.67 | 70.00 |
| Applied EO Doses [µg/mL] | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cont. | 100 | 50 | 25 | 10 | 5 | 2.5 | 1.25 | 0.625 | 0.25 | 0.125 | 0.0625 | ||
| Sinapis alba | Min | 0.3 | 0.6 | 0.5 | 0.3 | 0.5 | 0.6 | 0.3 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Max | 3.9 | 7.2 | 6.5 | 10.1 | 6.1 | 10.6 | 5.7 | 5.4 | 4.6 | 4.3 | 5.5 | 5.5 | |
| Mean | 1.5 | 3.2 | 3.1 | 3.4 | 3.2 | 2.8 | 2.3 | 1.9 | 1.8 | 1.2 | 1.8 | 2.3 | |
| SD | 0.9 | 1.9 | 1.9 | 2.8 | 1.6 | 2.3 | 1.8 | 1.6 | 1.5 | 1.1 | 1.6 | 1.6 | |
| Median | 1.3 | 3.3 | 2.8 | 2.5 | 3.1 | 2.1 | 1.6 | 1.9 | 1.2 | 0.7 | 1.2 | 2.5 | |
| Signif. | *** | *** | ** | *** | ** | * | |||||||
| Lepidium sativum | Min | 0.3 | 0.5 | 2.1 | 1.8 | 0.5 | 0.6 | 0.5 | 0.2 | 0.3 | 0.4 | 0.5 | 1.5 |
| Max | 9.6 | 5.8 | 11.2 | 13.2 | 13.1 | 12.0 | 13.1 | 12.8 | 12.5 | 13.0 | 11.0 | 12.3 | |
| Mean | 4.9 | 3.6 | 7.5 | 9.0 | 5.7 | 8.4 | 7.2 | 8.2 | 8.3 | 7.9 | 7.1 | 7.6 | |
| SD | 2.4 | 1.3 | 2.3 | 3.3 | 3.6 | 3.7 | 3.5 | 3.2 | 3.5 | 3.7 | 2.6 | 2.7 | |
| Median | 5.5 | 4.0 | 8.1 | 10.3 | 5.3 | 10.2 | 8.3 | 8.6 | 9.0 | 9.0 | 7.8 | 8.1 | |
| Signif. | * | *** | *** | *** | ** | *** | *** | *** | *** | *** | |||
| Hordeum vulgare | Min | 1.6 | 0.6 | 0.7 | 0.1 | 0.4 | 0.4 | 0.6 | 0.3 | 0.3 | 0.4 | 0.3 | 0.5 |
| Max | 4.7 | 3.1 | 6.3 | 4.6 | 6.3 | 5.3 | 5.7 | 3.5 | 4.0 | 6.8 | 4.3 | 3.4 | |
| Mean | 3.2 | 1.9 | 3.2 | 1.8 | 3.3 | 3.4 | 3.5 | 1.9 | 1.9 | 2.8 | 2.4 | 2.1 | |
| SD | 0.9 | 0.8 | 1.8 | 1.5 | 1.8 | 1.3 | 1.7 | 1.0 | 1.1 | 1.7 | 1.3 | 0.8 | |
| Median | 3.3 | 2.1 | 3.5 | 1.7 | 3.4 | 3.5 | 3.7 | 2.0 | 1.7 | 2.7 | 2.3 | 2.1 | |
| Signif. | ** | ** | *** | *** | * | *** | |||||||
| Triticum aestivum | Min | 0.4 | 0.2 | 0.5 | 0.3 | 0.2 | 0.9 | 1.1 | 0.3 | 0.3 | 1.5 | 0.3 | 0.5 |
| Max | 3.9 | 1.8 | 3.2 | 2.7 | 3.8 | 4.8 | 4.0 | 2.40 | 3.2 | 3.5 | 2.5 | 3.2 | |
| Mean | 2.6 | 1.1 | 1.9 | 1.7 | 1.9 | 2.8 | 2.5 | 1.7 | 1.9 | 2.4 | 1.5 | 1.8 | |
| SD | 0.9 | 0.5 | 0.8 | 0.6 | 1.1 | 0.8 | 0.9 | 0.5 | 0.9 | 0.6 | 0.6 | 0.7 | |
| Median | 2.8 | 1.2 | 1.8 | 1.8 | 1.9 | 2.9 | 2.8 | 1.9 | 2.0 | 2.3 | 1.5 | 1.8 | |
| Signif. | *** | * | ** | *** | *** | ** | |||||||
| Sample/Extract | (µg/mL) | Log (µg/mL) | DPPH (%) after 15 min | DPPH (%) after 30 min | DPPH (%) after 45 min |
|---|---|---|---|---|---|
| CTRL DPPH | 100 ± 2.00 | 100 ± 2.84 | 100.00 ± 3.10 | ||
| Oregano | 3 | 0.477 | 97.61 ± 0.40 | 96.75 ± 0.74 | 95.47 ± 0.92 |
| Oregano | 6 | 0.778 | 88.70 ± 1.34 | 88.20 ± 1.57 | 87.20 ± 1.69** |
| Oregano | 12.5 | 1.097 | 92.31 ± 0.54 | 89.67 ± 0.53 | 87.41 ± 0.79*** |
| Oregano | 25 | 1.398 | 86.22 ± 0.86 * | 82.42 ± 1.35** | 79.06 ± 1.66**** |
| Oregano | 50 | 1.699 | 83.52 ± 8.14**** | 72.64 ± 3.23**** | 68.00 ± 3.15**** |
| Oregano | 100 | 2.000 | 69.59 ± 4.72**** | 58.14 ± 4.46**** | 52.84 ± 4.30**** |
| Oregano | 200 | 2.301 | 46.66 ± 5.07**** | 38.89 ± 4.45**** | 33.80 ± 3.96**** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gruľová, D.; Caputo, L.; Elshafie, H.S.; Baranová, B.; De Martino, L.; Sedlák, V.; Gogaľová, Z.; Poráčová, J.; Camele, I.; De Feo, V. Thymol Chemotype Origanum vulgare L. Essential Oil as a Potential Selective Bio-Based Herbicide on Monocot Plant Species. Molecules 2020, 25, 595. https://doi.org/10.3390/molecules25030595
Gruľová D, Caputo L, Elshafie HS, Baranová B, De Martino L, Sedlák V, Gogaľová Z, Poráčová J, Camele I, De Feo V. Thymol Chemotype Origanum vulgare L. Essential Oil as a Potential Selective Bio-Based Herbicide on Monocot Plant Species. Molecules. 2020; 25(3):595. https://doi.org/10.3390/molecules25030595
Chicago/Turabian StyleGruľová, Daniela, Lucia Caputo, Hazem S. Elshafie, Beáta Baranová, Laura De Martino, Vincent Sedlák, Zuzana Gogaľová, Janka Poráčová, Ippolito Camele, and Vincenzo De Feo. 2020. "Thymol Chemotype Origanum vulgare L. Essential Oil as a Potential Selective Bio-Based Herbicide on Monocot Plant Species" Molecules 25, no. 3: 595. https://doi.org/10.3390/molecules25030595
APA StyleGruľová, D., Caputo, L., Elshafie, H. S., Baranová, B., De Martino, L., Sedlák, V., Gogaľová, Z., Poráčová, J., Camele, I., & De Feo, V. (2020). Thymol Chemotype Origanum vulgare L. Essential Oil as a Potential Selective Bio-Based Herbicide on Monocot Plant Species. Molecules, 25(3), 595. https://doi.org/10.3390/molecules25030595










