Towards the Properties of Different Biomass-Derived Proteins via Various Extraction Methods
Abstract
1. Introduction
2. Results and Discussion
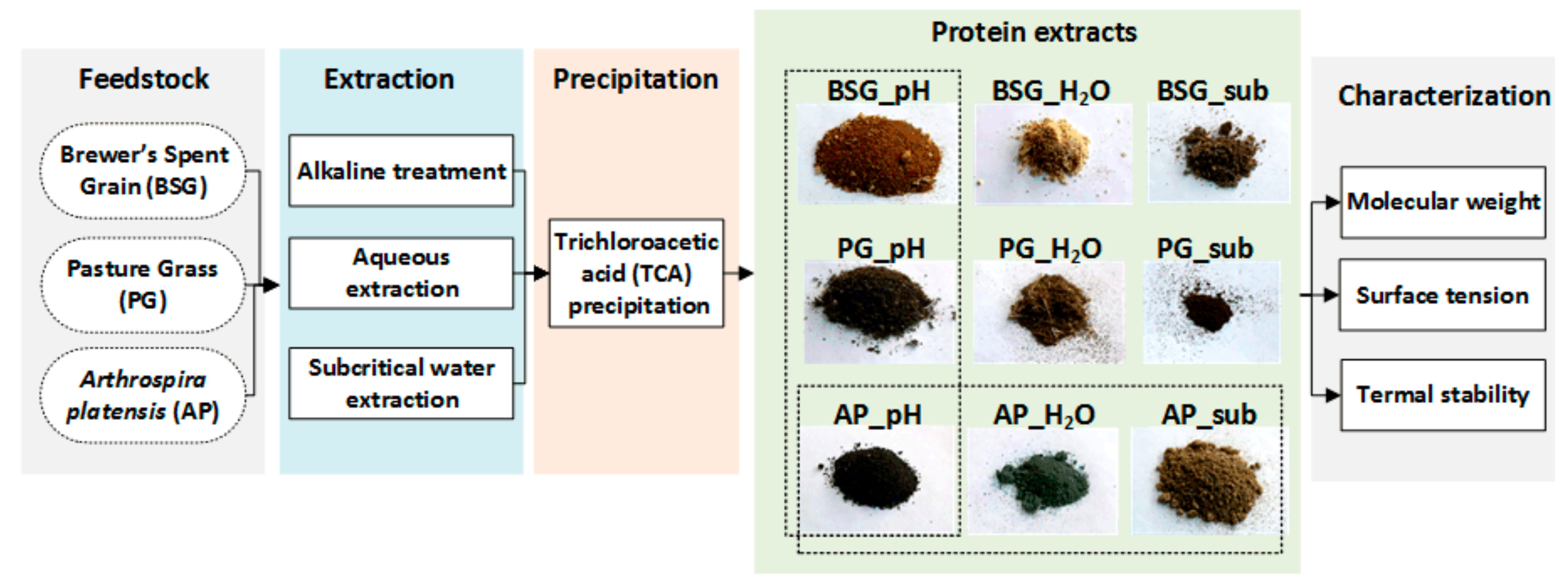
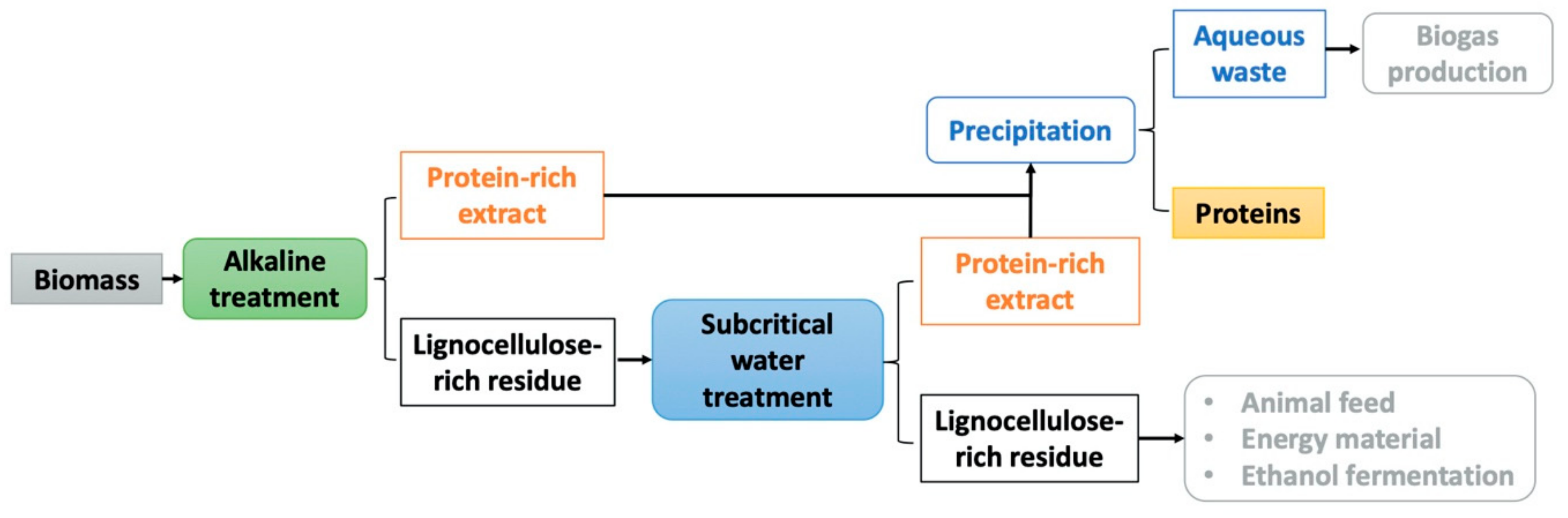
2.1. Extraction Method Comparison
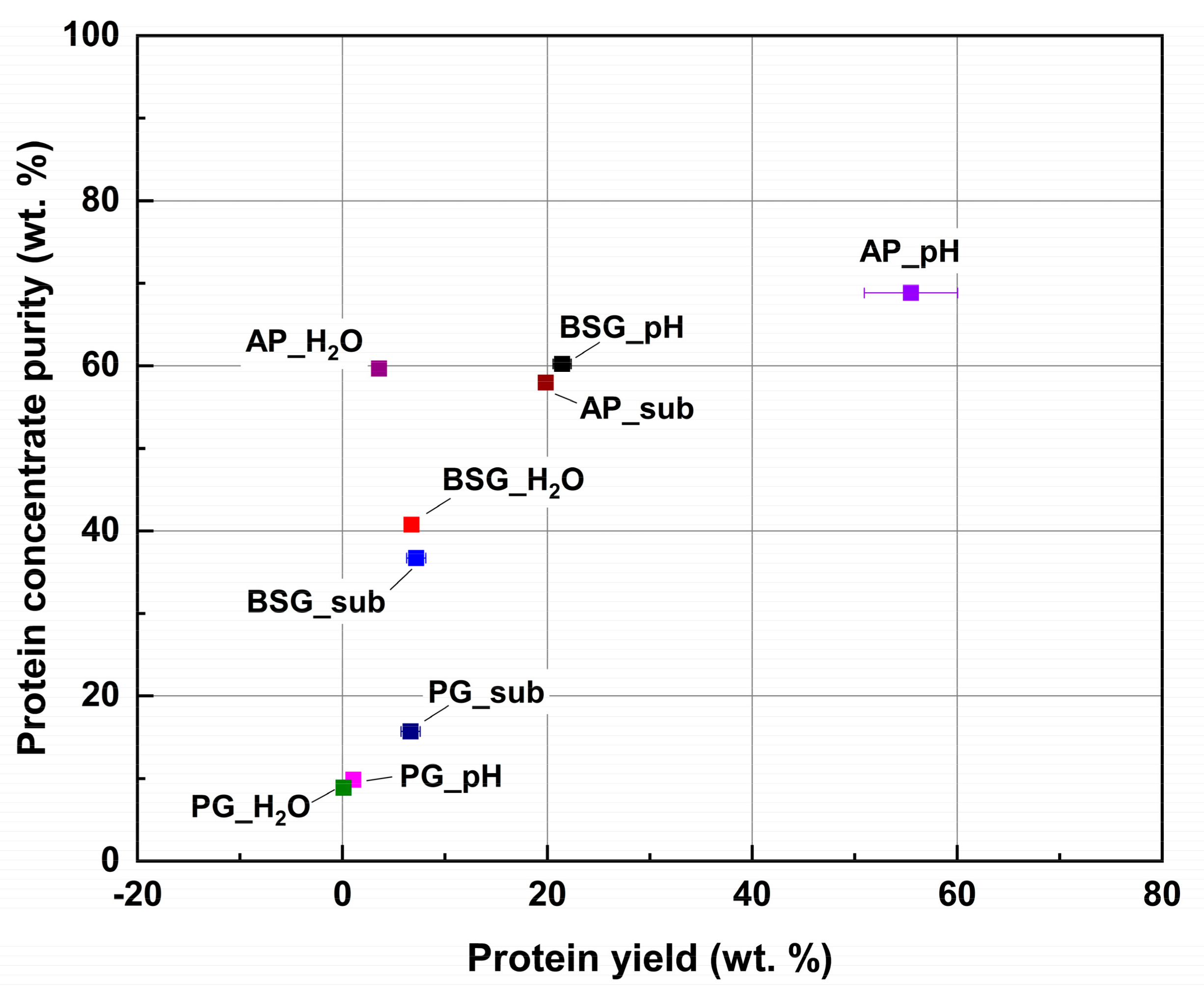
2.2. Protein Extraction Yield and Purity
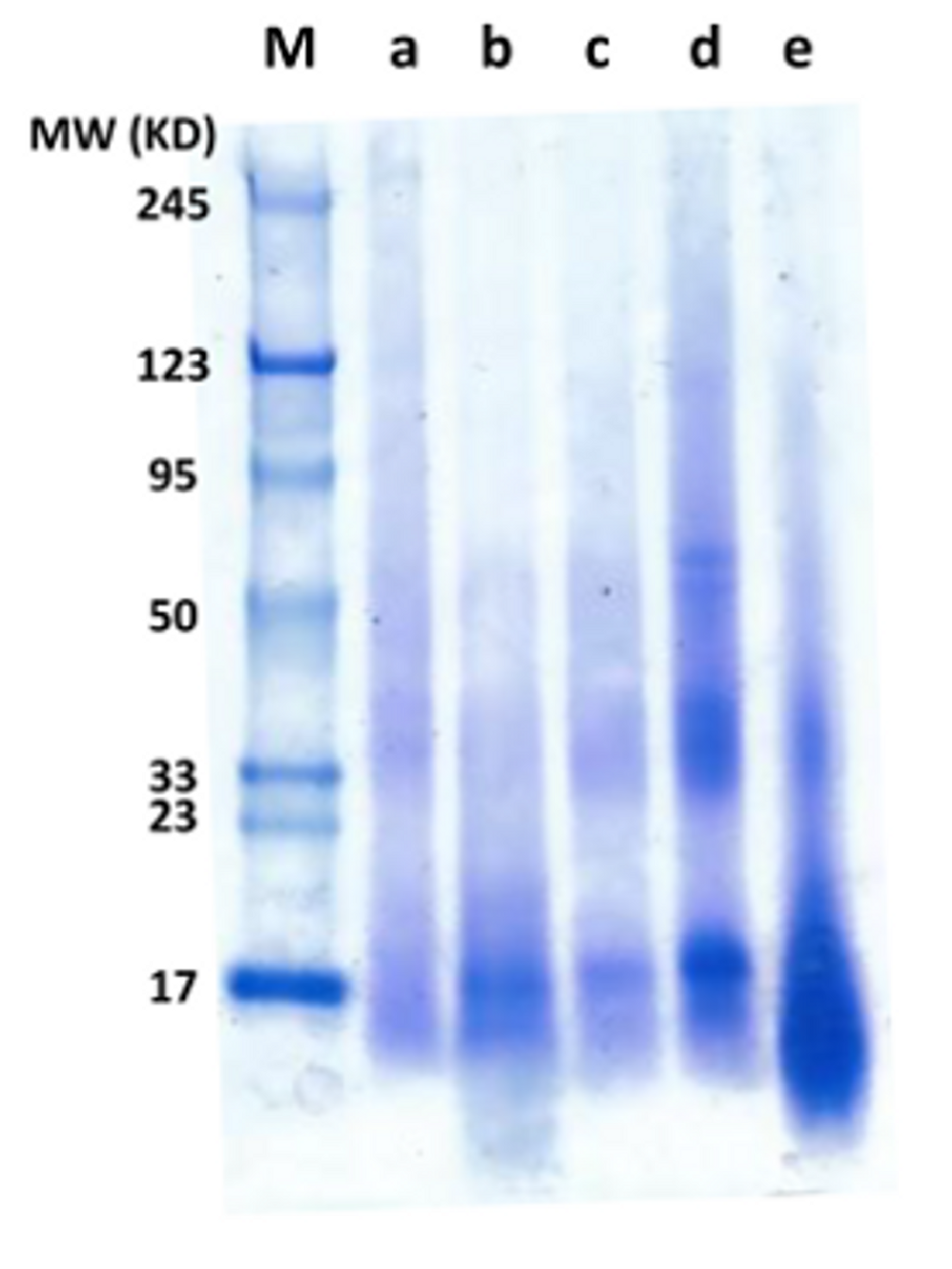
2.3. Molecular Weight
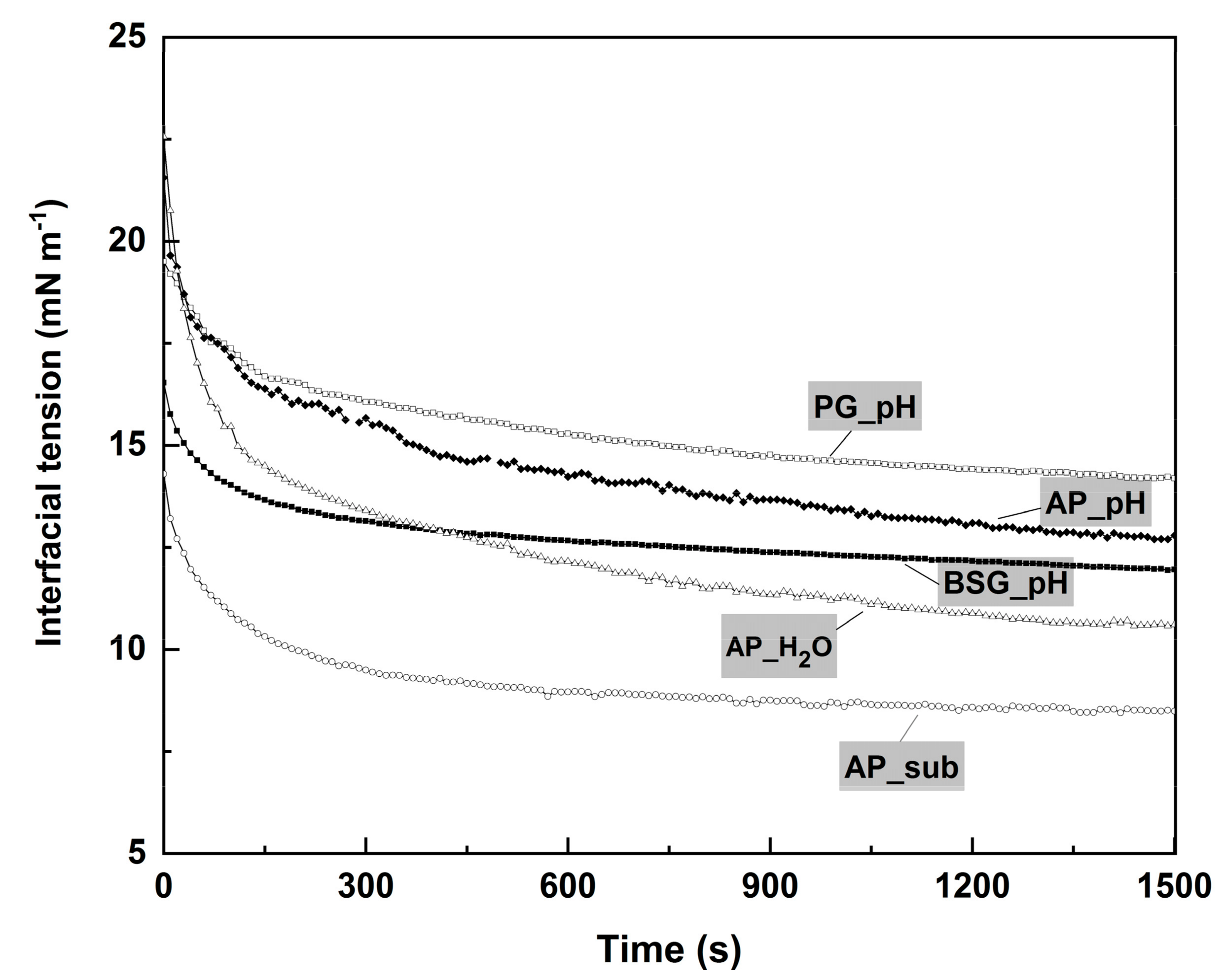
2.4. Surface Tension
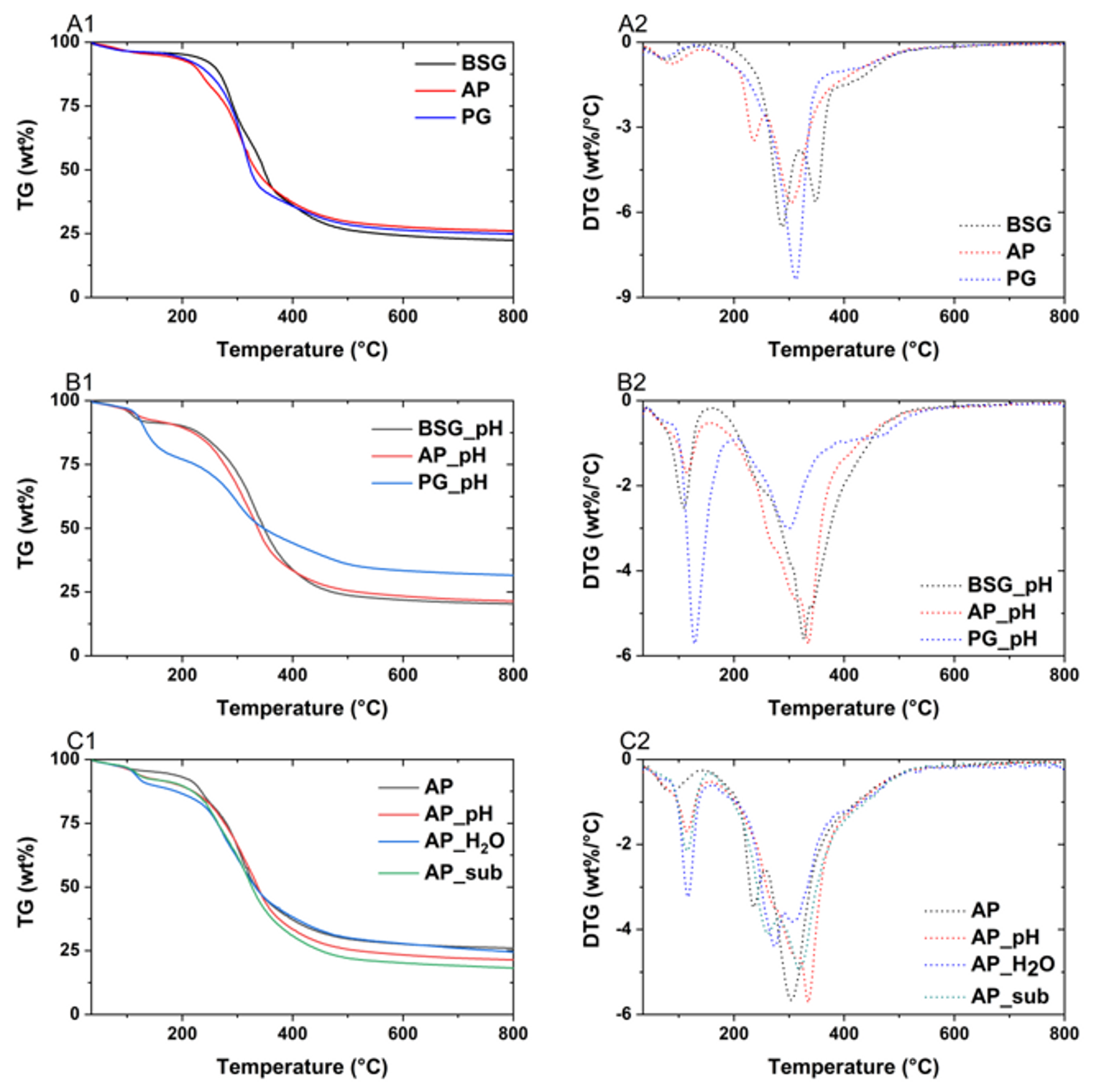
2.5. Thermal Stability
3. Materials and Methods
3.1. Materials
3.2. Protein Concentrates Preparation
3.2.1. Alkaline Treatment
3.2.2. Aqueous Extraction
3.2.3. Subcritical Water Extraction
3.2.4. Trichloroacetic Acid (TCA) Precipitation
3.3. Composition Analysis
3.4. Lowry Protein Determination
3.5. Extraction Yield and Purity Determination
3.6. Protein Functional Properties Characterization
3.6.1. SDS-Polyacrylamide Gel Electrophoresis (SDS-PAGE)
3.6.2. Interfacial Tension
3.6.3. Thermogravimetric Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food Security: The Challenge of Feeding 9 Billion People. Science 2010, 327, 812–818. [Google Scholar] [CrossRef]
- Bleakley, S.; Hayes, M. Algal Proteins: Extraction, Application, and Challenges Concerning Production. Foods 2017, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Baines, D.; Seal, R. Natural Food Additives, Ingredients and Flavourings, 1st ed.; Woodhead Publishing: Cambridge, UK, 2012. [Google Scholar]
- Lammens, T.M.; Franssen, M.C.R.; Scott, E.L.; Sanders, J.P.M. Availability of protein-derived amino acids as feedstock for the production of bio-based chemicals. Biomass Bioenerg. 2012, 44, 168–181. [Google Scholar] [CrossRef]
- Sari, Y.W. Biomass and its potential for protein and amino acids: Valorizing agricultural by-products. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, January 2015. [Google Scholar]
- Nadathur, S.R.; Wanasundara, J.P.D.; Scanlin, L. Sustainable Protein Source; Academic Press: Cambridge, MA, USA, 2016; ISBN 978-0-12-802778-3. [Google Scholar]
- López, D.N.; Galante, M.; Robson, M.; Boeris, V.; Spelzini, D. Amaranth, quinoa and chia protein isolates: Physicochemical and structural properties. Int. J. Biol. Macromol. 2018, 109, 152–159. [Google Scholar] [CrossRef]
- Aydemir, L.Y.; Yemenicioĝlu, A. Potential of Turkish Kabuli type chickpea and green and red lentil cultivars as source of soy and animal origin functional protein alternatives. LWT - Food Sci. Technol. 2013, 50, 686–694. [Google Scholar] [CrossRef]
- Pham, T.T.; Tran, T.T.T.; Ton, N.M.N.; Le, V.V.M. Effects of pH and Salt Concentration on Functional Properties of Pumpkin Seed Protein Fractions. J. Food Process. Preserv. 2017, 41. [Google Scholar] [CrossRef]
- Contreras, M.d.M.; Lama-Muñoz, A.; Manuel Gutiérrez-Pérez, J.; Espínola, F.; Moya, M.; Castro, E. Protein extraction from agri-food residues for integration in biorefinery: Potential techniques and current status. Bioresour. Technol. 2019, 280, 459–477. [Google Scholar] [CrossRef]
- Hettiarachchy, N.S.; Sato, K.; Marshall, M.R.; Kannan, A. Food Proteins and Peptides: Chemistry, Functionality, Interactions, and Commercialization; CRC Press: Boca Raton, FL, USA, 2016; ISBN 9781138199002-CAT# K31432. [Google Scholar]
- Dai, L.; Bergfreund, J.; Reichert, C.L.; Fischer, P.; Weiss, J. Shear rheological properties of acid hydrolyzed insoluble proteins from Chlorella protothecoides at the oil-water interface. J. Colloid Interf. Sci. 2019, 551, 297–304. [Google Scholar] [CrossRef]
- Romero, A.; Beaumal, V.; David-Briand, E.; Cordobés, F.; Anton, M.; Guerrero, A. Interfacial and emulsifying behaviour of crayfish protein isolate. LWT - Food Sci. Technol. 2011, 44, 1603–1610. [Google Scholar] [CrossRef]
- Kinsella, J.E. Functional properties of proteins in foods: A survey. CRC Crit. Rev. Food Sci. Nutr. 1976, 7, 219–280. [Google Scholar] [CrossRef]
- Agbor, V.B.; Cicek, N.; Sparling, R.; Berlin, A.; Levin, D.B. Biomass pretreatment: Fundamentals toward application. Biotechnol. Adv. 2011, 29, 675–685. [Google Scholar] [CrossRef] [PubMed]
- Dotsenko, G.; Lange, L. Enzyme Enhanced Protein Recovery from Green Biomass Pulp. Waste Biomass Valori. 2017, 8, 1257–1264. [Google Scholar] [CrossRef]
- Montusiewicz, A.; Pasieczna-Patkowska, S.; Lebiocka, M.; Szaja, A.; Szymańska-Chargot, M. Hydrodynamic cavitation of brewery spent grain diluted by wastewater. Chem. Eng. J. 2017, 313, 946–956. [Google Scholar] [CrossRef]
- Lynch, K.M.; Steffen, E.J.; Arendt, E.K. Brewers’ spent grain: A review with an emphasis on food and health. J. Inst. Brew. 2016, 122, 553–568. [Google Scholar] [CrossRef]
- Becker, E.W. Micro-algae as a source of protein. Biotechnol. Adv. 2007, 25, 207–210. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.R.; Markakis, P. Blue-green algae as a source of protein. Food Chem. 1981, 7, 239–247. [Google Scholar] [CrossRef]
- Celus, I.; Brijs, K.; Delcour, J.A. The effects of malting and mashing on barley protein extractability. J. Cereal Sci. 2006, 44, 203–211. [Google Scholar] [CrossRef]
- Schingoethe, D.J.; Ahrar, M. Protein Solubility, Amino Acid Composition, and Biological Value of Regular and Heat-Treated Soybean and Sunflower Meals. J. Dairy Sci. 1979, 62, 925–931. [Google Scholar] [CrossRef]
- Kinsella, J.E. Ribulose bisphosphate carboxylase/oxygenase (rubisco) from green leaves—Potential as a food protein. Food Rev. Int. 1988, 4, 93–127. [Google Scholar]
- Kärenlampi, S.O.; White, P.J. Potato Proteins, Lipids, and Minerals. In Advances in Potato Chemistry and Technology; Academic Press: Cambridge, MA, USA, 2009; pp. 99–125. [Google Scholar]
- Sindhu, R.; Binod, P.; Pandey, A. Biological pretreatment of lignocellulosic biomass - An overview. Bioresour. Technol. 2016, 199, 76–82. [Google Scholar] [CrossRef]
- Voudouris, P.; Tenorio, A.T.; Lesschen, J.P.; Mulder, W.; Kyriakopoulou, K.E.; Sanders, J.P.M.; van der Goot, A.J.; Bruins, M.E. Sustainable protein technology: An. evaluation on the STW Protein programme and an outlook for the future; Wageningen Food & Biobased Research: Wageningen, the Netherlands, 2017. [Google Scholar]
- Cavonius, L.R.; Albers, E.; Undeland, I. pH-shift processing of Nannochloropsis oculata microalgal biomass to obtain a protein-enriched food or feed ingredient. Algal Res. 2015, 11, 95–102. [Google Scholar] [CrossRef]
- Arauzo, P.J.; Du, L.; Olszewski, M.P.; Meza Zavala, M.F.; Alhnidi, M.J.; Kruse, A. Effect of protein during hydrothermal carbonization of brewer’s spent grain. Bioresour. Technol. 2019, 293. [Google Scholar] [CrossRef] [PubMed]
- Kruse, A.; Dahmen, N. Water–A magic solvent for biomass conversion. J. Supercrit. Fluids 2014, 96, 36–45. [Google Scholar] [CrossRef]
- Alenezi, R.; Leeke, G.A.; Santos, R.C.D.; Khan, A.R. Hydrolysis kinetics of sunflower oil under subcritical water conditions. Chem. Eng. Res. Des. 2009, 87, 867–873. [Google Scholar] [CrossRef]
- Clifford, T. Fundamentals of Supercritical Fluids; Oxford University Press: Oxford, UK, 1999; ISBN 9780198501374. [Google Scholar]
- Kammes, K.L.; Bals, B.D.; Dale, B.E.; Allen, M.S. Grass leaf protein, a coproduct of cellulosic ethanol production, as a source of protein for livestock. Anim. Feed Sci. Technol. 2011, 164, 79–88. [Google Scholar] [CrossRef]
- Bals, B.; Dale, B.E. Economic comparison of multiple techniques for recovering leaf protein in biomass processing. Biotechnol. Bioeng. 2011, 108, 530–537. [Google Scholar] [CrossRef]
- Zhang, C.; Sanders, J.P.M.; Bruins, M.E. Critical parameters in cost-effective alkaline extraction for high protein yield from leaves. Biomass Bioenerg. 2014, 67, 466–472. [Google Scholar] [CrossRef]
- Ahmed, R.; Chun, B.S. Subcritical water hydrolysis for the production of bioactive peptides from tuna skin collagen. J. Supercrit. Fluids 2018, 141, 88–96. [Google Scholar] [CrossRef]
- Feyzi, S.; Milani, E.; Golimovahhed, Q.A. Grass Pea (Lathyrus sativus L.) Protein Isolate: The Effect of Extraction Optimization and Drying Methods on the Structure and Functional Properties. Food Hydrocoll. 2018, 74, 187–196. [Google Scholar] [CrossRef]
- Safi, C.; Ursu, A.V.; Laroche, C.; Zebib, B.; Merah, O.; Pontalier, P.-Y.; Vaca-Garcia, C. Aqueous extraction of proteins from microalgae: Effect of different cell disruption methods. Algal Res. 2014, 3, 61–65. [Google Scholar] [CrossRef]
- Zhao, X.; Stökle, K.; Becker, G.C.; Zimmermann, M.; Kruse, A. Hydrothermal carbonization of Spirulina platensis and Chlorella vulgaris combined with protein isolation and struvite production. Bioresour. Technol. Reports 2019, 6, 159–167. [Google Scholar] [CrossRef]
- Celus, I.; Brijs, K.; Delcour, J.A. Enzymatic hydrolysis of Brewers’ spent grain proteins and technofunctional properties of the resulting hydrolysates. J. Agric. Food Chem. 2007, 55, 8703–8710. [Google Scholar] [CrossRef] [PubMed]
- Safi, C.; Charton, M.; Ursu, A.V.; Laroche, C.; Zebib, B.; Pontalier, P.Y.; Vaca-Garcia, C. Release of hydro-soluble microalgal proteins using mechanical and chemical treatments. Algal Res. 2014, 3. [Google Scholar] [CrossRef]
- Pirie, N.W. Leaf Protein as a Human Food. Sci. New Ser. 1966, 152, 1701–1705. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, M.; Adschiri, T.; Arai, K. Kinetics of Cellulose Conversion at 25 MPa in Sub- and Supercritical Water. AIChE J. 2004, 50, 192–202. [Google Scholar] [CrossRef]
- Gerde, J.A.; Wang, T.; Yao, L.; Jung, S.; Johnson, L.A.; Lamsal, B. Optimizing protein isolation from defatted and non-defatted Nannochloropsis microalgae biomass. Algal Res. 2013, 2, 145–153. [Google Scholar] [CrossRef]
- Carpentier, S.C.; Witters, E.; Laukens, K.; Deckers, P.; Swennen, R.; Panis, B. Preparation of protein extracts from recalcitrant plant tissues: An evaluation of different methods for two-dimensional gel electrophoresis analysis. Proteomics 2005, 5, 2497–2507. [Google Scholar] [CrossRef]
- Ward, J.J.; Sodhi, J.S.; McGuffin, L.J.; Buxton, B.F.; Jones, D.T. Prediction and Functional Analysis of Native Disorder in Proteins from the Three Kingdoms of Life. J. Mol. Biol. 2004, 337, 635–645. [Google Scholar] [CrossRef]
- Nandakumar, M.P.; Shen, J.; Raman, B.; Marten, M.R. Solubilization of trichloroacetic acid (TCA) precipitated microbial proteins via NaOH for two-dimensional electrophoresis. J. Proteome Res. 2003, 2, 89–93. [Google Scholar] [CrossRef]
- Fic, E.; Kedracka-Krok, S.; Jankowska, U.; Pirog, A.; Dziedzicka-Wasylewska, M. Comparison of protein precipitation methods for various rat brain structures prior to proteomic analysis. Electrophoresis 2010, 31, 3573–3579. [Google Scholar] [CrossRef]
- Barbarino, E.; Lourenço, S.O. An evaluation of methods for extraction and quantification of protein from marine macro- and microalgae. J. Appl. Phycol. 2005, 17, 447–460. [Google Scholar] [CrossRef]
- Oliveira, R.; Marques, F.; Azeredo, J. Purification of polysaccharides from a biofilm matrix by selective precipitation of proteins. Biotechnol. Tech. 1999, 13, 391–393. [Google Scholar] [CrossRef]
- United States Environmental Protection Agency. Available online: https://www.epa.gov/greenchemistry/basics-green-chemistry (accessed on 29 December 2019).
- Vilkhu, K.; Mawson, R.; Simons, L.; Bates, D. Applications and opportunities for ultrasound assisted extraction in the food industry — A review. Innov. Food Sci. Emerg. Technol. 2008, 9, 161–169. [Google Scholar] [CrossRef]
- Hou, Y.; Wu, Z.; Dai, Z.; Wang, G.; Wu, G. Protein hydrolysates in animal nutrition: Industrial production, bioactive peptides, and functional significance. J. Anim. Sci. Biotechnol. 2017, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhao, M.; Yang, X.; Jiang, Y. Improvement on functional properties of wheat gluten by enzymatic hydrolysis and ultrafiltration. J. Cereal Sci. 2006, 44, 93–100. [Google Scholar] [CrossRef]
- Garcia, E.S.; Van Leeuwen, J.J.A.; Safi, C.; Sijtsma, L.; Van Den Broek, L.A.M.; Eppink, M.H.M.; Wijffels, R.H.; Van Den Berg, C. Techno-Functional Properties of Crude Extracts from the Green Microalga Tetraselmis suecica. J. Agric. Food Chem. 2018, 66, 7831–7838. [Google Scholar] [CrossRef]
- Waghmare, A.G.; Salve, M.K.; LeBlanc, J.G.; Arya, S.S. Concentration and characterization of microalgae proteins from Chlorella pyrenoidosa. Bioresour. Bioprocess. 2016, 3. [Google Scholar] [CrossRef]
- Davis, J.P.; Foegeding, E.A.; Hansen, F.K. Electrostatic effects on the yield stress of whey protein isolate foams. Colloids Surfaces B 2004, 34, 13–23. [Google Scholar] [CrossRef]
- Rangsansarid, J.; Cheetangdee, N.; Kinoshita, N.; Fukuda, K. Bovine serum albumin-sugar conjugates through the Maillard reaction: Effects on interfacial behavior and emulsifying ability. J. Oleo Sci. 2008, 57, 539–547. [Google Scholar] [CrossRef]
- Grossmann, L.; Ebert, S.; Hinrichs, J.; Weiss, J. Formation and Stability of Emulsions Prepared with a Water-Soluble Extract from the Microalga Chlorella protothecoides. J. Agric. Food Chem. 2019, 67, 6551–6558. [Google Scholar] [CrossRef]
- Grønli, M.G.; Várhegyi, G.; Di Blasi, C. Thermogravimetric analysis and devolatilization kinetics of wood. Ind. Eng. Chem. Res. 2002, 41, 4201–4208. [Google Scholar] [CrossRef]
- Rommi, K.; Niemi, P.; Kemppainen, K.; Kruus, K. Impact of thermochemical pre-treatment and carbohydrate and protein hydrolyzing enzyme treatment on fractionation of protein and lignin from brewer’s spent grain. J. Cereal Sci. 2018, 79, 168–173. [Google Scholar] [CrossRef]
- Dinwoodie, J.M. Wood: Nature’s cellular polymeric composite. Phys. Technol. 1978, 9, 185–191. [Google Scholar] [CrossRef]
- Fan, Y.; Hornung, U.; Dahmen, N.; Kruse, A. Hydrothermal liquefaction of protein-containing biomass: Study of model compounds for Maillard reactions. Biomass Convers. Bior. 2018, 8, 909–923. [Google Scholar] [CrossRef]
- Ricci, L.; Umiltà, E.; Righetti, M.C.; Messina, T.; Zurlini, C.; Montanari, A.; Bronco, S.; Bertoldo, M. On the thermal behavior of protein isolated from different legumes investigated by DSC and TGA. J. Sci. Food Agric. 2018, 98, 5368–5377. [Google Scholar] [CrossRef]
- Mariotti, F.; Tomé, D.; Mirand, P.P. Converting nitrogen into protein - Beyond 6.25 and Jones’ factors. Crit. Rev. Food Sci. Nutr. 2008, 48, 177–184. [Google Scholar] [CrossRef]
Sample Availability: Not available. |
| Alkaline Treatment | Aqueous Extraction | Subcritical Water Extraction | ||
|---|---|---|---|---|
| Extraction time/duration (min.) | 120 | 120 | 20 | |
| Temperature (°C) | 40 | 40 | 200 | |
| pH | 11 | 7 | 7 | |
| Solvent involved | 0.1 M NaOH | Water | Water | |
| Protein yield (wt.%) | BSG | 21.4 (0.9) | 6.8 (0.1) | 7.2 (0.9) |
| PG | 1.1 (0.1) | 0.1 a | 6.7 (0.9) | |
| AP | 55.5 (4.6) | 3.6 (0.1) | 19.9 (0.4) | |
| Protein concentrate purity (wt.%) | BSG | 60.2 (0.7) | 40.7 (0.3) | 36.7 (0.1) |
| PG | 9.8 a | 8.9 (0.3) | 15.7 a | |
| AP | 68.8 (0.2) | 59.7 (0.1) | 58.0 (0.3) | |
| Composition (wt.%) | BSG | PG | AP |
|---|---|---|---|
| Protein a | 21.9 | 9.14 | 55.9 |
| C | 50.9 (0.3) | 44.8 (0.1) | 45.2 c |
| H | 7.0 (0.1) | 6.3 c | 6.9 c |
| N | 4.3 (0.2) | 1.8 c | 9.4 c |
| S | 0.2 c | 0.1 c | 0.6 c |
| O b | 33.6 (0.4) | 40.1 (0.1) | 32.4 (0.1) |
| Ash | 3.9 c | 7.0 (0.1) | 5.5 (0.1) |
| Dry Weight | 22 c | 29.7 (0.8) | 94.9 (0.2) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du, L.; Arauzo, P.J.; Meza Zavala, M.F.; Cao, Z.; Olszewski, M.P.; Kruse, A. Towards the Properties of Different Biomass-Derived Proteins via Various Extraction Methods. Molecules 2020, 25, 488. https://doi.org/10.3390/molecules25030488
Du L, Arauzo PJ, Meza Zavala MF, Cao Z, Olszewski MP, Kruse A. Towards the Properties of Different Biomass-Derived Proteins via Various Extraction Methods. Molecules. 2020; 25(3):488. https://doi.org/10.3390/molecules25030488
Chicago/Turabian StyleDu, Lin, Pablo J. Arauzo, Maria Fernanda Meza Zavala, Zebin Cao, Maciej Pawel Olszewski, and Andrea Kruse. 2020. "Towards the Properties of Different Biomass-Derived Proteins via Various Extraction Methods" Molecules 25, no. 3: 488. https://doi.org/10.3390/molecules25030488
APA StyleDu, L., Arauzo, P. J., Meza Zavala, M. F., Cao, Z., Olszewski, M. P., & Kruse, A. (2020). Towards the Properties of Different Biomass-Derived Proteins via Various Extraction Methods. Molecules, 25(3), 488. https://doi.org/10.3390/molecules25030488







