Mechanistic Aspects and Therapeutic Potential of Quercetin against COVID-19-Associated Acute Kidney Injury
Abstract
1. Introduction
2. Materials and Methods
3. General Aspects of Acute Kidney Injury
4. Viral Infections and Acute Kidney Injury
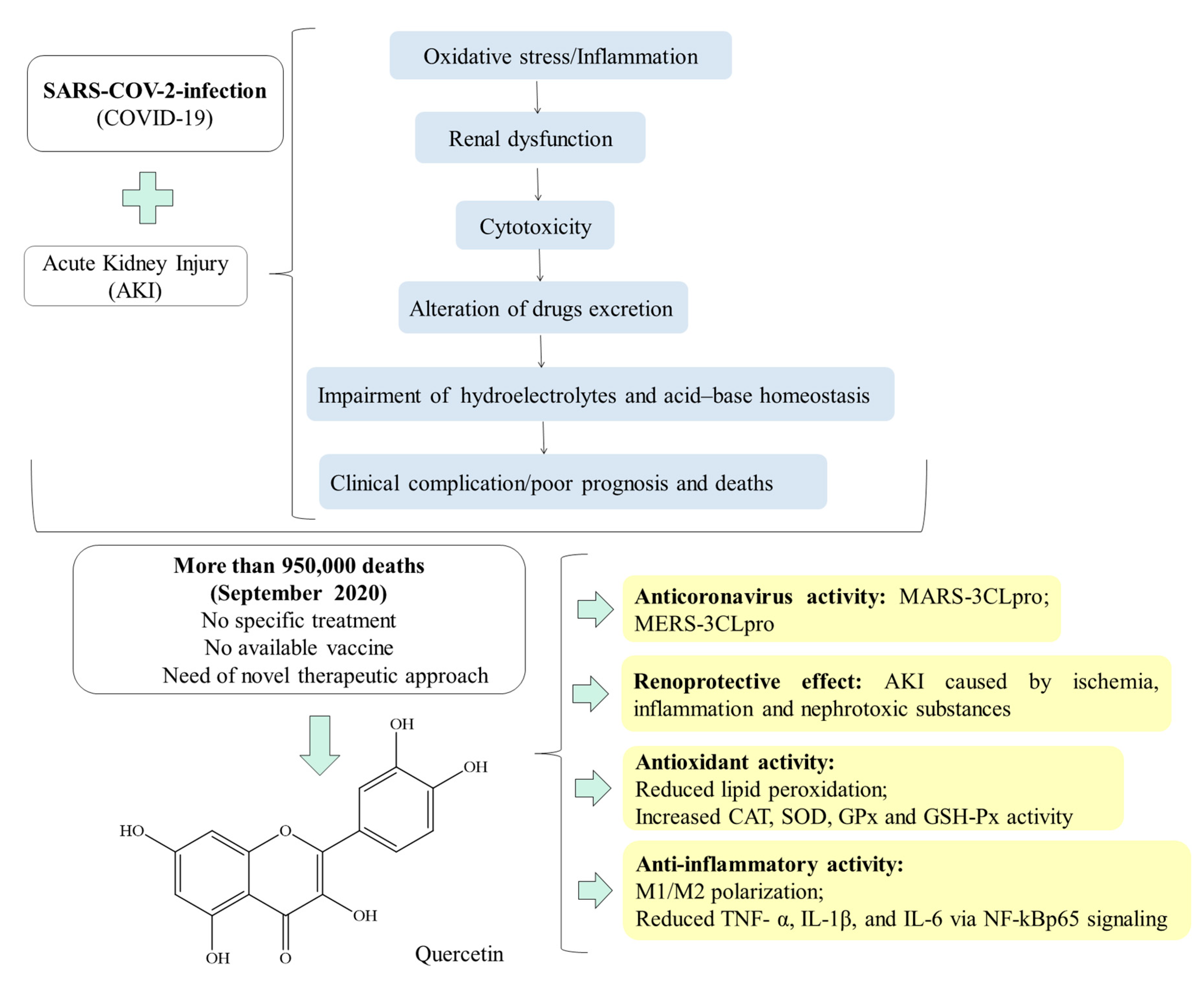
5. Coronavirus and Acute Kidney Injury
6. Quercetin: A Natural Polyphenol Compound with Pharmacological Properties for the Treatment of COVID-19-Induced Morbidities
7. Quercetin and Acute Kidney Injury
8. Quercetin: A Helpful Therapeutic Drug Against SARS-COV-2-Induced AKI?
Funding
Conflicts of Interest
Abbreviations
| 3CL(pro) | 3c-Like Protease |
| ACLF | Acute-On-Chronic Liver Failure |
| ADPR-cyclase | ADP-Ribosyl Cyclase |
| AKI | Acute Kidney Injury |
| AKIN | Acute Kidney Network |
| Akt | Protein Kinase B |
| AMPK | AMP-Activated Protein Kinase |
| ASK1 | Apoptosis Signal-Regulating Kinase 1 |
| BMDMs | Bone Marrow-Derived Macrophages |
| BUN | Blood Urea Nitrogen |
| cADPR-hydrolase | Cyclic ADP-Ribose Hydrolase |
| CoVs | Coronaviruses |
| COVID-19 | Coronavirus disease 2019 |
| DHAV | Duck Hepatitis A Virus |
| GFR | Glomerular Filtration Rate |
| GSH-Px | Glutathione Peroxidase |
| HBV | Hepatitis B Virus |
| HCV | Hepatitis C Virus |
| HIV | Human Immunodeficiency Virus |
| HIVAN | Human Immunodeficiency Virus-Associated Nephropathy |
| I/R | Ischemia and reperfusion |
| ICU | Intensive Care Unit |
| IL | Interleukin |
| IL-18 | Interleukin-18 |
| iNOS | Nitric Oxide Synthase |
| IRF5 | Interferon Regulatory Factor 5 |
| JNK3 | C-Jun N-Terminal Kinase 3 |
| KDIGO | Kidney Disease: Improving Global Outcomes |
| KIM-1 | Kidney Injury Molecule-1 |
| L-FABP | Liver-Type Fatty Acid-Binding Protein |
| LPS | Lipopolysaccharide |
| MDA | Malondialdehyde |
| MERS-CoV | Middle East Respiratory Syndrome-Related Coronavirus |
| MMP-1 | Matrix Metalloproteinase-1 |
| MyD88 | Myeloid Differentiation Protein |
| NF-κB | Nuclear Factor κB |
| NGAL | Neutrophil Gelatinase-Associated Lipocalin |
| PI3K | Phosphoinositide 3-Kinase |
| RBF | Renal Blood Flow |
| RIFLE | Risk, Injury, Failure, Loss, End-Stage Kidney Disease |
| RNS | Reactive Nitrogen Species |
| ROS | Reactive Oxygen Species |
| SARS | Severe Acute Respiratory Syndrome |
| SARS-CoV | Severe Acute Respiratory Syndrome Coronavirus |
| SCr | Serum Creatinine |
| SOD | Superoxide Dismutase |
| SYK | Tyrosine-Protein Kinase |
| TGF-β1 | Transforming Growth Factor Beta |
| TNF-α | Tumor Necrosis Factor Alpha |
| TRAF6 | Tumor Necrosis Factor Receptor-Associated Factor 6 |
References
- Beaney, T.; Clarke, J.M.; Jain, V.; Golestaneh, A.K.; Lyons, G.; Salman, D.; Majeed, A. Excess mortality: The gold standard in measuring the impact of COVID-19 worldwide? J. R. Soc. Med. 2020, 113, 329–334. [Google Scholar] [CrossRef]
- Becher, T.; Frerichs, I. Mortality in COVID-19 is not merely a question of resource availability. Lancet Respir. Med. 2020, 8, 832–833. [Google Scholar] [CrossRef]
- Immovilli, P.; Morelli, N.; Rota, E.; Guidetti, D. COVID-19 mortality and health-care resources: Organization. Med. Intensiva. 2020, 1–2. [Google Scholar] [CrossRef]
- Chen, L.; Liu, S.; Tian, J.; Pan, H.; Liu, Y.; Hu, J.; Wang, M.; Hou, X. Disease progression patterns and risk factors associated with mortality in deceased patients with COVID-19 in Hubei Province, China. Immun. Inflamm. Dis. 2020, 1–11. [Google Scholar] [CrossRef]
- Grasselli, G.; Greco, M.; Zanella, A.; Albano, G.; Antonelli, M.; Bellani, G.; Bonanomi, E.; Cabrini, L.; Carlesso, E.; Castelli, G.; et al. Risk Factors Associated with Mortality Among Patients With COVID-19 in Intensive Care Units in Lombardy, Italy. JAMA Intern. Med. 2020, 180, 1345–1355. [Google Scholar] [CrossRef]
- Lim, M.A.; Pranata, R.; Huang, I.; Yonas, E.; Soeroto, A.Y.; Supriyadi, R. Multiorgan Failure with Emphasis on Acute Kidney Injury and Severity of COVID-19: Systematic Review and Meta-Analysis. Can. J. Kidney Health Dis. 2020, 7, 2054358120938573. [Google Scholar] [CrossRef]
- Carriazo, S.; Kanbay, M.; Ortiz, A. Kidney disease and electrolytes in COVID-19: More than meets the eye. Clin. Kidney. J. 2020, 13, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.T.; Shao, S.C.; Lai, E.C.C.; Hung, M.J.; Chen, Y.C. Mortality rate of acute kidney injury in SARS, MERS, and COVID-19 infection: A systematic review and meta-analysis. Crit. Care 2020, 24, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Cheruiyot, I.; Kipkorir, V.; Ngure, B.; Misiani, M.; Munguti, J.; Henry, B.; Lippi, G. Acute Kidney Injury is Associated with Worse Prognosis in COVID-19 Patients: A systematic Review and Meta-analysis. Acta Biomed. 2020, 91, 1–19. [Google Scholar] [CrossRef]
- Durvasula, R.; Wellington, T.; McNamara, E.; Watnick, S. COVID-19 and Kidney Failure in the Acute Care Setting: Our Experience from Seattle. Am. J. Kidney Dis. 2020, 76, 4–6. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.; Mahmood, S. Kidney Injury in COVID-19: An Emerging Concern to the Clinician. SN Compr. Clin. Med. 2020, 2, 1808–1809. [Google Scholar] [CrossRef]
- Meena, P.; Bhargava, V.; Rana, D.S.; Bhalla, A.K.; Gupta, A. COVID-19 and the kidney: A matter of concern. Curr. Med. Res. Pract. 2020, 10, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Aziz, T.M.; Stockand, J.D. Recent progress and challenges in drug development against COVID-19 coronavirus (SARS-CoV-2)-an update on the status. Infect. Genet. Evol. 2020, 83, 104327. [Google Scholar] [CrossRef] [PubMed]
- Lega, S.; Naviglio, S.; Volpi, S.; Tommasini, A. Recent Insight into SARS-CoV2 Immunopathology and Rationale for Potential Treatment and Preventive Strategies in COVID-19. Vaccines 2020, 8, 224. [Google Scholar] [CrossRef] [PubMed]
- Ahsan, W.; Alhazmi, H.A.; Patel, K.S.; Mangla, B.; Al Bratty, M.; Javed, S.; Najmi, A.; Sultan, M.H.; Makeen, H.A.; Khalid, A.; et al. Recent Advancements in the Diagnosis, Prevention, and Prospective Drug Therapy of COVID-19. Front. Public. Health 2020, 8, 384. [Google Scholar] [CrossRef]
- Patel, N.; Rein, J.L.; Sanchez-Russo, L.; Winston, J.; Uribarri, J. COVID-19-Associated Acute Kidney Injury: A Case Series. Kidney Med. 2020, 2, 668–669. [Google Scholar] [CrossRef]
- Batiha, G.E.; Beshbishy, A.M.; Ikram, M.; Mulla, Z.S.; El-Hack, M.E.A.; Taha, A.E.; Algammal, A.M.; Elewa, Y.H.A. The Pharmacological Activity, Biochemical Properties, and Pharmacokinetics of the Major Natural Polyphenolic Flavonoid: Quercetin. Foods 2020, 9, 374. [Google Scholar] [CrossRef]
- Darband, S.G.; Kaviani, M.; Yousefi, B.; Sadighparvar, S.; Pakdel, F.G.; Attari, J.A.; Mohebbi, I.; Naderi, S.; Majidinia, M. Quercetin: A functional dietary flavonoid with potential chemo-preventive properties in colorectal cancer. J. Cell. Physiol. 2018, 233, 6544–6560. [Google Scholar] [CrossRef]
- Diniz, L.R.L.; Filho, C.S.M.B.; Fielding, B.C.; de Sousa, D.P. Natural Antioxidants: A Review of Studies on Human and Animal Coronavirus. Oxidative Med. Cell. Longev. 2020, 2020, 1–14. [Google Scholar] [CrossRef]
- Yao, F.; Zhang, R.; Fu, R.; He, W. Preventive and therapeutic effects of quercetin on hyperuricemia and renal injury in rats. J. Hyg. Res. 2011, 40, 175–177. [Google Scholar]
- Shoskes, D.A. Effect of bioflavonoids quercetin and curcumin on ischemic renal injury: A new class of renoprotective agents. Transplantation 1998, 66, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Pan, Y.; Zhang, Q.Y.; Wang, F.M.; Kong, L.D. Quercetin and allopurinol ameliorate kidney injury in STZ-treated rats with regulation of renal NLRP3 inflammasome activation and lipid accumulation. PLoS ONE 2012, 7, e38285. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; He, J.; Qin, W.; Zhao, L. Quercetin alleviates lipopolysaccharide-induced acute kidney injury in mice by suppressing TLR4/NF-kappaB pathway. Nan Fang Yi Ke Da Xue Xue Bao 2019, 39, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Tan, R.Z.; Wang, C.; Deng, C.; Zhong, X.; Yan, Y.; Luo, Y.; Lan, H.Y.; He, T.; Wang, L. Quercetin protects against cisplatin-induced acute kidney injury by inhibiting Mincle/Syk/NF-kappaB signaling maintained macrophage inflammation. Phytother. Res. 2020, 34, 139–152. [Google Scholar] [CrossRef]
- Chaudhary, S.; Ganjoo, P.; Raiusddin, S.; Parvez, S. Nephroprotective activities of quercetin with potential relevance to oxidative stress induced by valproic acid. Protoplasma 2015, 252, 209–217. [Google Scholar] [CrossRef]
- Gomes, I.B.; Porto, M.L.; Santos, M.C.L.; Campagnaro, B.P.; Pereira, T.M.; Meyrelles, S.S.; Vasquez, E.C. Renoprotective, anti-oxidative and anti-apoptotic effects of oral low-dose quercetin in the C57BL/6J model of diabetic nephropathy. Lipids Health Dis. 2014, 13, 1–10. [Google Scholar] [CrossRef]
- Morales, A.I.; Vicente-Sánchez, C.; Sandoval, J.M.S.; Egido, J.; Mayoral, P.; Arévalo, M.A.; Fernández-Tagarro, M.; López-Novoa, J.M.; Pérez-Barriocanal, F. Protective effect of quercetin on experimental chronic cadmium nephrotoxicity in rats is based on its antioxidant properties. Food Chem. Toxicol. 2006, 44, 2092–2100. [Google Scholar] [CrossRef]
- Lu, H.; Wu, L.; Liu, L.; Ruan, Q.; Zhang, X.; Hong, W.; Wu, S.; Jin, G.; Bai, Y. Quercetin ameliorates kidney injury and fibrosis by modulating M1/M2 macrophage polarization. Biochem. Pharmacol. 2018, 154, 203–212. [Google Scholar] [CrossRef]
- Elbe, H.; Dogan, Z.; Taslidere, E.; Cetin, A.; Turkoz, Y. Beneficial effects of quercetin on renal injury and oxidative stress caused by ciprofloxacin in rats: A histological and biochemical study. Hum. Exp. Toxicol. 2016, 35, 276–281. [Google Scholar] [CrossRef]
- Koza, Y. Acute kidney injury: Current concepts and new insights. J. Inj. Violence Res. 2016, 8, 58–62. [Google Scholar] [CrossRef]
- Hoste, E.A.; de Corte, W. AKI patients have worse long-term outcomes, especially in the immediate post-ICU period. Crit. Care 2012, 16, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Singbartl, K.; Kellum, J.A. AKI in the ICU: Definition, epidemiology, risk stratification, and outcomes. Kidney Int. 2012, 81, 819–825. [Google Scholar] [CrossRef] [PubMed]
- Hoste, E.A.; Bagshaw, S.M.; Bellomo, R.; Cely, C.M.; Colman, R.; Cruz, D.N.; Edipidis, K.; Forni, L.G.; Gomersall, C.D.; Govil, D.; et al. Epidemiology of acute kidney injury in critically ill patients: The multinational AKI-EPI study. Intensive Care Med. 2015, 41, 1411–1423. [Google Scholar] [CrossRef] [PubMed]
- Doi, K.; Rabb, H. Impact of acute kidney injury on distant organ function: Recent findings and potential therapeutic targets. Kidney Int. 2016, 89, 555–564. [Google Scholar] [CrossRef]
- Yu, S.M.W.; Bonventre, J.V. Acute Kidney Injury and Progression of Diabetic Kidney Disease. Adv. Chronic Kidney Dis. 2018, 25, 166–180. [Google Scholar] [CrossRef]
- Truche, A.S.; Ragey, S.P.; Souweine, B.; Bailly, S.; Zafrani, L.; Bouadma, L.; Clec’h, C.; Garrouste-Orgeas, M.; Lacave, G.; Schwebel, C.; et al. ICU survival and need of renal replacement therapy with respect to AKI duration in critically ill patients. Ann. Intensive Care 2018, 8, 127. [Google Scholar] [CrossRef]
- Abdelaziz, T.S.; Fouda, R.; Hussin, W.M.; Elyamny, M.S.; Abdelhamid, Y.M. Preventing acute kidney injury and improving outcome in critically ill patients utilizing risk prediction score (PRAIOC-RISKS) study. A prospective controlled trial of AKI prevention. J. Nephrol. 2020, 33, 325–334. [Google Scholar] [CrossRef]
- Koeze, J.; Keus, F.; Dieperink, W.; van der Horst, I.C.C.; Zijlstra, J.G.; van Meurs, M. Incidence, timing and outcome of AKI in critically ill patients varies with the definition used and the addition of urine output criteria. BMC Nephrol. 2017, 18, 70. [Google Scholar] [CrossRef]
- Liu, K.D.; Yang, J.; Tan, T.C.; Glidden, D.V.; Zheng, S.; Pravoverov, L.; Hsu, C.; Go, A.S. Risk Factors for Recurrent Acute Kidney Injury in a Large Population-Based Cohort. Am. J. Kidney Dis. 2019, 73, 163–173. [Google Scholar] [CrossRef]
- Dylewska, M.; Chomicka, I.; Malyszko, J. Hypertension in patients with acute kidney injury. Wiad. Lek. 2019, 72, 2199–2201. [Google Scholar] [CrossRef]
- Peerapornratana, S.; Manrique-Caballero, C.L.; Gomez, H.; Kellum, J.A. Acute kidney injury from sepsis: Current concepts, epidemiology, pathophysiology, prevention and treatment. Kidney Int. 2019, 96, 1083–1099. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.; Rodrigues, N.; Godinho, I.; Gameiro, J.; Neves, M.; Gouveia, J.; Costa, E.S.Z.; Lopes, J.A. Acute kidney injury in patients with severe sepsis or septic shock: A comparison between the ‘Risk, Injury, Failure, Loss of kidney function, End-stage kidney disease’ (RIFLE), Acute Kidney Injury Network (AKIN) and Kidney Disease: Improving Global Outcomes (KDIGO) classifications. Clin. Kidney, J. 2017, 10, 332–340. [Google Scholar] [CrossRef]
- Ülger, F.; Küçük, M.P.; Küçük, A.O.; Ilkaya, N.K.; Murat, N.; Bilgiç, B.; Abanoz, H. Evaluation of acute kidney injury (AKI) with RIFLE, AKIN, CK, and KDIGO in critically ill trauma patients. Eur. J. Trauma Emerg. Surg. 2018, 44, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Teo, S.H.; Endre, Z.H. Biomarkers in acute kidney injury (AKI). Best Pract. Res. Clin. Anaesthesiol. 2017, 31, 331–344. [Google Scholar] [CrossRef] [PubMed]
- Tsai, T.Y.; Chien, H.; Tsai, F.C.; Pan, H.C.; Yang, H.Y.; Lee, S.Y.; Hsu, H.H.; Fang, J.T.; Yang, C.W.; Chen, Y.C. Comparison of RIFLE, AKIN, and KDIGO classifications for assessing prognosis of patients on extracorporeal membrane oxygenation. J. Formos. Med. Assoc. 2017, 116, 844–851. [Google Scholar] [CrossRef]
- Park, S.; Lee, H. Acute kidney injury prediction models: Current concepts and future strategies. Curr. Opin. Nephrol. Hypertens. 2019, 28, 552–559. [Google Scholar] [CrossRef]
- Grooth, H.J.; Parienti, J.J.; Schetz, M. AKI biomarkers are poor discriminants for subsequent need for renal replacement therapy, but do not disqualify them yet. Intensive Care Med. 2018, 44, 1156–1158. [Google Scholar] [CrossRef]
- Luft, F.C. Biomarkers and predicting acute kidney injury. Acta Physiol. (Oxf.) 2020, e13479. [Google Scholar] [CrossRef]
- Zdziechowska, M.; Gluba-Brzózka, A.; Poliwczak, A.R.; Franczyk, B.; Kidawa, M.; Zielinska, M.; Rysz, J. Serum NGAL, KIM-1, IL-18, L-FABP: New biomarkers in the diagnostics of acute kidney injury (AKI) following invasive cardiology procedures. Int. Urol. Nephrol. 2020, 52, 2135–2143. [Google Scholar] [CrossRef]
- Gayat, E. Biomarkers of acute kidney injury: Mixed results and huge heterogeneity of reporting. BMJ Evid. Based Med. 2019, 24, e9. [Google Scholar] [CrossRef]
- Bunz, H.; Weyrich, P.; Peter, A.; Baumann, D.; Tschritter, O.; Guthoff, M.; Beck, R.; Jahn, G.; Artunc, F.; Haring, H.U.; et al. Urinary Neutrophil Gelatinase-Associated Lipocalin (NGAL) and proteinuria predict severity of acute kidney injury in Puumala virus infection. BMC Infect. Dis. 2015, 15, 464. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.; Shao, X.; Xie, Y.; Wang, Q.; Che, X.; Zhang, M.; Xu, W.; Xu, Y.; Ni, Z.; Mou, S. Kidney Injury Molecule-1 is Elevated in Nephropathy and Mediates Macrophage Activation via the Mapk Signalling Pathway. Cell. Physiol. Biochem. 2017, 41, 769–783. [Google Scholar] [CrossRef] [PubMed]
- Han, S.J.; Lee, H.T. Mechanisms and therapeutic targets of ischemic acute kidney injury. Kidney Res. Clin. Pract. 2019, 38, 427–440. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Dong, Z.; Harris, R.; Murray, P.; Parikh, S.M.; Rosner, M.H.; Kellum, J.A.; Ronco, C. Cellular and Molecular Mechanisms of AKI. J. Am. Soc. Nephrol. 2016, 27, 1288–1299. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Zhang, Q.; Wen, J.; Chen, T.; He, L.; Wang, Y.; Yin, J.; Wu, R.; Xue, R.; Li, S.; et al. Ischemic Duration and Frequency Determines AKI-to-CKD Progression Monitored by Dynamic Changes of Tubular Biomarkers in IRI Mice. Front. Physiol. 2019, 10, 153. [Google Scholar] [CrossRef]
- Prasad, N.; Patel, M.R. Infection-Induced Kidney Diseases. Front. Med. (Lausanne) 2018, 5, 327:1–327:11. [Google Scholar] [CrossRef]
- Prakash, J.; Gupta, T.; Prakash, S.; Rathore, S.S.; Sunder, S. Acute kidney injury in patients with human immunodeficiency virus infection. Indian, J. Nephrol. 2015, 25, 86–90. [Google Scholar] [CrossRef]
- Moretti, M.; Lava, S.A.G.; Zgraggen, L.; Simonetti, G.D.; Kottanattu, L.; Bianchetti, M.G.; Milani, G.P. Acute kidney injury in symptomatic primary Epstein-Barr virus infectious mononucleosis: Systematic review. J. Clin. Virol. 2017, 91, 12–17. [Google Scholar] [CrossRef]
- Moudgil, A.; Nast, C.C.; Bagga, A.; Wei, L.; Nurmamet, A.; Cohen, A.H.; Jordan, S.C.; Toyoda, M. Association of parvovirus B19 infection with idiopathic collapsing glomerulopathy. Kidney Int. 2001, 59, 2126–2133. [Google Scholar] [CrossRef]
- Fujinaga, S.; Hara, T. Acute Kidney Injury Following Plastic Bronchitis Associated with Influenza B Virus in a Child with Nephrotic Syndrome. Indian Pediatr. 2015, 52, 523–525. [Google Scholar] [CrossRef]
- Brown, P.R.; Sadiq, O.; Weick, A.; Lenhart, A.; Elbatta, M.; Fernandez, C.; Kutait, A.; Pompa, R.; Jafri, S.M. Acute Kidney Injury in Patients Undergoing Chronic Hepatitis C Virus Treatment With Ledipasvir/Sofosbuvir. Hepatol. Commun. 2018, 2, 1172–1178. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Tang, L.; Tang, H.; Pu, J.; Gong, S.; Fang, D.; Zhang, H.; Li, Y.P.; Zhu, X.; Wang, W.; et al. Zika Virus Infection Induces Acute Kidney Injury Through Activating NLRP3 Inflammasome Via Suppressing Bcl-2. Front. Immunol. 2019, 10, 1925. [Google Scholar] [CrossRef]
- Khalil, M.A.; Sarwar, S.; Chaudry, M.A.; Maqbool, B.; Khalil, Z.; Tan, J.; Yaqub, S.; Hussain, S.A. Acute kidney injury in dengue virus infection. Clin. Kidney J. 2012, 5, 390–394. [Google Scholar] [CrossRef] [PubMed]
- Casas-Aparicio, G.A.; Léon-Rodriguéz, I.; Hernández-Zenteno, R.J.; Castillejos-López, M.; Alvarado-de Barrera, C.A.; Ormsby, C.E.; Reyes-Teran, G. Aggressive fluid accumulation is associated with acute kidney injury and mortality in a cohort of patients with severe pneumonia caused by influenza A H1N1 virus. PLoS ONE 2018, 13, e0192592. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Liu, Z.; Han, Q.; Li, Z.; Zhang, G.; Qiu, J.; Lou, S.; Li, N.; Wang, Y.; Li, M. Acute kidney injury in patients with hemorrhagic fever with renal syndrome caused by Hantaan virus: Comparative evaluation by RIFLE and AKIN criteria. Vector Borne Zoonotic Dis. 2011, 11, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Libraty, D.H.; Makela, S.; Vlk, J.; Hurme, M.; Vaheri, A.; Ennis, F.A.; Mustonen, J. The degree of leukocytosis and urine GATA-3 mRNA levels are risk factors for severe acute kidney injury in Puumala virus nephropathia epidemica. PLoS ONE 2012, 7, e35402. [Google Scholar] [CrossRef] [PubMed]
- Nadkarni, G.N.; Patel, A.; Simoes, P.K.; Yacoub, R.; Annapureddy, N.; Kamat, S.; Konstantinidis, I.; Perumalswami, P.; Branch, A.; Coca, S.G. Dialysis-requiring acute kidney injury among hospitalized adults with documented hepatitis C Virus infection: A nationwide inpatient sample analysis. J. Viral Hepat. 2016, 23, 32–38. [Google Scholar] [CrossRef]
- Gupta, A.; Quigg, R.J. Glomerular Diseases Associated With Hepatitis B and C. Adv. Chronic Kidney Dis. 2015, 22, 343–351. [Google Scholar] [CrossRef]
- Morisue, A.; Fukuoka, K.; Goto, R.; Ota, K.; Yamashita, H.; Shinno, Y.; Yamadori, I. Hepatitis C virus-related glomerulonephritis with acute kidney injury requiring hemodialysis that improved with virus removal and eradication using double-filtration plasmapheresis without interferon. CEN Case Rep. 2015, 4, 38–42. [Google Scholar] [CrossRef][Green Version]
- Nadasdy, T.; Hebert, L.A. Infection-related glomerulonephritis: Understanding mechanisms. Semin. Nephrol. 2011, 31, 369–375. [Google Scholar] [CrossRef]
- Godinho, I.; Nogueira, E.; Jorge, S.; Alves, A.T.; da Costa, A.G. Membranoproliferative glomerulonephritis and interstitial nephritis in the setting of Epstein-Barr virus-related hemophagocytic syndrome. Clin. Nephrol. 2018, 89, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Abdulkader, R.C.; Ho, Y.L.; Santos, S.S.; Caires, R.; Arantes, M.F.; Andrade, L. Characteristics of acute kidney injury in patients infected with the 2009 influenza A (H1N1) virus. Clin. J. Am. Soc. Nephrol. 2010, 5, 1916–1921. [Google Scholar] [CrossRef]
- Indhumathi, E.; Makkena, V.K.; Mamidi, V.; Jayaprakash, V.; Jayakumar, M. Influenza A (H1N1) Virus Infection Associated Acute Kidney Injury-A Study from a Tertiary Care Center in South India. Saudi J. Kidney Dis. Transpl. 2020, 31, 759–766. [Google Scholar] [CrossRef] [PubMed]
- Salter, T.; Burton, H.; Douthwaite, S.; Newsholme, W.; Horsfield, C.; Hilton, R. Immune Complex Mediated Glomerulonephritis with Acute Thrombotic Microangiopathy following Newly Detected Hepatitis B Virus Infection in a Kidney Transplant Recipient. Case Rep. Transplant. 2016, 2016, 1–5. [Google Scholar] [CrossRef]
- Kawakita, C.; Kinomura, M.; Otaka, N.; Kitagawa, M.; Sugiyama, H.; Kusano, N.; Mizuno, M.; Wada, J. HIV-associated Immune Complex Kidney Disease with C3-dominant Deposition Induced by HIV Infection after Treatment of IgA Nephropathy. Intern. Med. 2019, 58, 3001–3007. [Google Scholar] [CrossRef] [PubMed]
- Tajima, K.; Kohno, K.; Shiono, Y.; Suzuki, I.; Kato, Y.; Hiroshima, Y.; Yamamoto, M.; Ohtake, H.; Iwaba, A.; Yamakawa, M.; et al. Acute kidney injury and inflammatory immune reconstitution syndrome in mixed genotype (A/E) hepatitis B virus co-infection in HIV-associated lymphoma. Int. J. Clin. Exp. Pathol. 2013, 6, 536–542. [Google Scholar] [PubMed]
- Garg, S.; Hoenig, M.; Edwards, E.M.; Bliss, C.; Heeren, T.; Tumilty, S.; Walley, A.Y.; Koziel, M.J.; Skolnik, P.R.; Horsburgh, C.R.; et al. Incidence and predictors of acute kidney injury in an urban cohort of subjects with HIV and hepatitis C virus coinfection. AIDS Patient Care STDS 2011, 25, 135–141. [Google Scholar] [CrossRef]
- Cohen, S.D.; Chawla, L.S.; Kimmel, P.L. Acute kidney injury in patients with human immunodeficiency virus infection. Curr. Opin. Crit. Care 2008, 14, 647–653. [Google Scholar] [CrossRef] [PubMed]
- Michal, J.L.; Rab, S.; Patel, M.; Kyle, A.W.; Miller, L.S.; Easley, K.A.; and Kalapila, A.G. Incidence of Acute Kidney Injury in Patients Coinfected with HIV and Hepatitis C Virus Receiving Tenofovir Disoproxil Fumarate and Ledipasvir/Sofosbuvir in a Real-World, Urban, Ryan White Clinic. AIDS Res. Hum. Retroviruses 2018, 34, 690–698. [Google Scholar] [CrossRef] [PubMed]
- Ou, X.; Mao, S.; Jiang, Y.; Zhang, S.; Ke, C.; Ma, G.; Cheng, A.; Wang, M.; Zhu, D.; Chen, S.; et al. Viral-host interaction in kidney reveals strategies to escape host immunity and persistently shed virus to the urine. Oncotarget 2017, 8, 7336–7349. [Google Scholar] [CrossRef][Green Version]
- Chaibi, K.; Dao, M.; Pham, T.; Gumucio-Sanguino, V.D.; Di Paolo, F.A.; Pavot, A.; Cohen, Y.; Dreyfuss, D.; Perez-Fernandez, X.; Gaudry, S. Severe Acute Kidney Injury in COVID-19 Patients with Acute Respiratory Distress Syndrome. Am. J. Respir. Crit. Care Med. 2020, 1–11. [Google Scholar] [CrossRef]
- Gabarre, P.; Dumas, G.; Dupont, T.; Darmon, M.; Azoulay, E.; Zafrani, L. Acute kidney injury in critically ill patients with COVID-19. Intensive Care Med. 2015, 46, 1339–1348. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Shi, S.; Zhu, J.; Shi, J.; Dai, K.; Chen, X. Analysis of 92 deceased patients with COVID-19. J. Med. Virol. 2020, 92, 2511–2515. [Google Scholar] [CrossRef]
- Yang, X.; Jin, Y.; Li, R.; Zhang, Z.; Sun, R.; Chen, D. Prevalence and impact of acute renal impairment on COVID-19: A systematic review and meta-analysis. Crit. Care 2020, 24, 1–8. [Google Scholar] [CrossRef]
- Gagliardi, I.; Patella, G.; Michael, A.; Serra, R.; Provenzanno, M.; Andreucci, M. COVID-19 and the Kidney: From Epidemiology to Clinical Practice. J. Clin. Med. 2020, 9, 2506. [Google Scholar] [CrossRef] [PubMed]
- Hirpara, K.V.; Aggarwal, P.; Mukherjee, A.J.; Joshi, N.; Burman, A.C. Quercetin and its derivatives: Synthesis, pharmacological uses with special emphasis on anti-tumor properties and prodrug with enhanced bio-availability. Anti-Cancer Agents Med. Chem. 2009, 9, 138–161. [Google Scholar] [CrossRef]
- Li, Y.; Yao, J.; Han, C.; Yang, J.; Chaudhry, M.T.; Wang, S.; Liu, H.; Yin, Y. Quercetin, Inflammation and Immunity. Nutrients 2016, 8, 167. [Google Scholar] [CrossRef] [PubMed]
- Dunnick, J.K.; Hailey, J.R. Toxicity and carcinogenicity studies of quercetin, a natural component of foods. Fundam. Appl. Toxicol. 1992, 19, 423–431. [Google Scholar] [CrossRef]
- Li, G.; Zeng, X.; Xie, Y.; Cai, Z.; Moore, J.C.; Yuan, X.; Cheng, Z.; Ji, G. Pharmacokinetic properties of isorhamnetin, kaempferol and quercetin after oral gavage of total flavones of Hippophae rhamnoides L. in rats using a UPLC-MS method. Fitoterapia 2012, 83, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Hollman, P.C.H.; van Trijp, J.M.P.; Buysman, M.N.C.P.; vd Gaag, M.S.; Mengelers, M.J.B.; de Vries, J.H.M.; Katan, M.B. Relative bioavailability of the antioxidant flavonoid quercetin from various foods in man. FEBS Lett. 1997, 418, 152–156. [Google Scholar] [CrossRef]
- Kahraman, A.; Erkasap, N.; Koken, T.; Serteser, M.; Aktepe, F.; Erkasap, S. The antioxidative and antihistaminic properties of quercetin in ethanol-induced gastric lesions. Toxicology 2003, 183, 133–142. [Google Scholar] [CrossRef]
- Karuppagounder, V.; Arumugam, S.; Thandavarayan, R.A.; Sreedhar, R.; Giridharan, V.V.; Watanabe, K. Molecular targets of quercetin with anti-inflammatory properties in atopic dermatitis. Drug Discov. Today 2016, 21, 632–639. [Google Scholar] [CrossRef] [PubMed]
- Dabulici, C.M.; Sarbu, I.; Vamanu, E. The Bioactive Potential of Functional Products and Bioavailability of Phenolic Compounds. Foods 2020, 9, 953. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Swarts, S.G.; Yin, L.; Liu, C.; Tian, Y.; Cao, Y.; Swarts, M.; Yang, S.; Zhang, S.B.; Zhang, K.; et al. Antioxidant properties of quercetin. Adv. Exp. Med. Biol. 2011, 701, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Mamani-Matsuda, M.; Kauss, T.; Al-Kharrat, A.; Rambert, J.; Fawaz, F.; Thiolat, D.; Moynet, D.; Coves, S.; Malvy, D.; Mossalayi, M.D. Therapeutic and preventive properties of quercetin in experimental arthritis correlate with decreased macrophage inflammatory mediators. Biochem. Pharmacol. 2006, 72, 1304–1310. [Google Scholar] [CrossRef] [PubMed]
- Das, M.; Ray, P.K. Lipid antioxidant properties of quercetin in vitro. Biochem. Int. 1988, 17, 203–209. [Google Scholar]
- Park, H.R.; Yoon, H.; Kim, M.K.; Lee, S.D.; Chong, Y. Synthesis and antiviral evaluation of 7-O-arylmethylquercetin derivatives against SARS-associated coronavirus (SCV) and hepatitis C virus (HCV). Arch. Pharm. Res. 2012, 35, 77–85. [Google Scholar] [CrossRef]
- Choi, H.J.; Kim, J.H.; Lee, C.H.; Ahn, Y.J.; Song, J.H.; Baek, S.H.; Kwon, D.H. Antiviral activity of quercetin 7-rhamnoside against porcine epidemic diarrhea virus. Antivir. Res. 2009, 81, 77–81. [Google Scholar] [CrossRef]
- Kim, H.P.; Mani, I.; Iversen, L.; Ziboh, V.A. Effects of naturally-occurring flavonoids and biflavonoids on epidermal cyclooxygenase and lipoxygenase from guinea-pigs. Prostaglandins Leukot. Essent. Fatty Acids 1998, 58, 17–24. [Google Scholar] [CrossRef]
- Bureau, G.; Longpre, F.; Martinoli, M.G. Resveratrol and quercetin, two natural polyphenols, reduce apoptotic neuronal cell death induced by neuroinflammation. J. Neurosci. Res. 2008, 86, 403–410. [Google Scholar] [CrossRef]
- Lee, K.M.; Hwang, M.K.; Lee, D.E.; Lee, K.W.; Lee, H.J. Protective effect of quercetin against arsenite-induced COX-2 expression by targeting PI3K in rat liver epithelial cells. J. Agric. Food Chem. 2010, 58, 5815–5820. [Google Scholar] [CrossRef] [PubMed]
- Endale, M.; Park, S.C.; Kim, S.; Kim, S.H.; Yang, Y.; Cho, J.Y.; Rhee, M.H. Quercetin disrupts tyrosine-phosphorylated phosphatidylinositol 3-kinase and myeloid differentiation factor-88 association, and inhibits MAPK/AP-1 and IKK/NF-kappaB-induced inflammatory mediators production in RAW 264.7 cells. Immunobiology 2013, 218, 1452–1467. [Google Scholar] [CrossRef] [PubMed]
- Kandere-Grzybowska, K.; Kempuraj, D.; Cao, J.; Cetrulo, C.L.; Theoharides, T.C. Regulation of IL-1-induced selective IL-6 release from human mast cells and inhibition by quercetin. Br. J. Pharmacol. 2006, 148, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Stewart, L.K.; Soileau, J.L.; Ribnicky, D.; Wang, Z.Q.; Raskin, I.; Poulev, A.; Majewski, M.; Cefalu, W.T.; Gettys, T.W. Quercetin transiently increases energy expenditure but persistently decreases circulating markers of inflammation in C57BL/6J mice fed a high-fat diet. Metabolism 2008, 57, S39–S46. [Google Scholar] [CrossRef]
- Nieman, D.C.; Henson, D.A.; Gross, S.J.; Jenkins, D.P.; Davis, J.M.; Murphy, E.A.; Carmichael, M.D.; Dumke, C.L.; Utter, A.C.; McAnulty, S.R.; et al. Quercetin reduces illness but not immune perturbations after intensive exercise. Med. Sci Sport. Exerc. 2007, 39, 1561–1569. [Google Scholar] [CrossRef]
- Qureshi, A.A.; Khan, D.A.; Mahjabeen, W.; Papasian, C.J.; Qureshi, N. Suppression of Nitric Oxide Production and Cardiovascular Risk Factors in Healthy Seniors and Hypercholesterolemic Subjects by a Combination of Polyphenols and Vitamins. J. Clin. Exp. Cardiolog. 2012, S5, 008. [Google Scholar] [CrossRef]
- Wu, S.; Zhang, Y.; Ren, F.; Qin, Y.; Liu, J.; Liu, J.; Wang, Q.; Zhang, H. Structure-affinity relationship of the interaction between phenolic acids and their derivatives and beta-lactoglobulin and effect on antioxidant activity. Food Chem. 2018, 245, 613–619. [Google Scholar] [CrossRef]
- Musonda, C.A.; Chipman, J.K. Quercetin inhibits hydrogen peroxide (H2O2)-induced NF-kappaB DNA binding activity and DNA damage in HepG2 cells. Carcinogenesis 1998, 19, 1583–1589. [Google Scholar] [CrossRef]
- Wang, D.; Jiang, Y.; Sun-Waterhouse, D.X.; Zhai, H.; Guan, H.; Rong, X.; Li, F.; Yu, J.C.; Li, D.P. MicroRNA-based regulatory mechanisms underlying the synergistic antioxidant action of quercetin and catechin in H2O2-stimulated HepG2 cells: Roles of BACH1 in Nrf2-dependent pathways. Free Radic. Biol. Med. 2020, 153, 122–131. [Google Scholar] [CrossRef]
- Chen, Z.; Yuan, Q.; Xu, G.; Chen, H.; Lei, H.; Su, J. Effects of Quercetin on Proliferation and H2O2-Induced Apoptosis of Intestinal Porcine Enterocyte Cells. Molecules 2018, 23, 2012. [Google Scholar] [CrossRef]
- Meng, L.Q.; Yang, F.Y.; Wang, M.S.; Shi, B.K.; Chen, D.X.; Chen, D.; Zhou, Q.; He, Q.B.; Ma, L.X.; Cheng, W.L.; et al. Quercetin protects against chronic prostatitis in rat model through NF-kappaB and MAPK signaling pathways. Prostate 2018, 78, 790–800. [Google Scholar] [CrossRef]
- Kim, S.G.; Kim, J.R.; Choi, H.C. Quercetin-Induced AMP-Activated Protein Kinase Activation Attenuates Vasoconstriction Through LKB1-AMPK Signaling Pathway. J. Med. Food 2018, 21, 146–153. [Google Scholar] [CrossRef]
- Marunaka, Y. Actions of quercetin, a flavonoid, on ion transporters: Its physiological roles. Ann. N. Y. Acad. Sci. 2017, 1398, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Marunaka, Y.; Niisato, N.; Miyazaki, H.; Nakajima, K.I.; Taruno, A.; Sun, H.; Marunaka, R.; Okui, M.; Yamamoto, T.; Kanamura, N. Quercetin is a Useful Medicinal Compound Showing Various Actions Including Control of Blood Pressure, Neurite Elongation and Epithelial Ion Transport. Curr. Med. Chem. 2018, 25, 4876–4887. [Google Scholar] [CrossRef] [PubMed]
- Kahraman, A.; Erkasap, N.; Serteser, M.; Koken, T. Protective effect of quercetin on renal ischemia/reperfusion injury in rats. J. Nephrol. 2003, 16, 219–224. [Google Scholar] [PubMed]
- Inal, M.; Altinisik, M.; Bilgin, M.D. The effect of quercetin on renal ischemia and reperfusion injury in the rat. Cell Biochem. Funct. 2002, 20, 291–296. [Google Scholar] [CrossRef] [PubMed]
- Sanchéz-González, P.D.; López-Hernández, F.J.; Dueñas, M.; Prieto, M.; Sánchez-López, E.; Thomale, J.; Ruiz-Ortega, M.; López-Novoa, J.M.; Morales, A.I. Differential effect of quercetin on cisplatin-induced toxicity in kidney and tumor tissues. Food Chem. Toxicol. 2017, 107, 226–236. [Google Scholar] [CrossRef]
- Yuksel, Y.; Yuksel, R.; Yagmurca, M.; Haltas, H.; Erdamar, H.; Toktas, M.; Ozcan, O. Effects of quercetin on methotrexate-induced nephrotoxicity in rats. Hum. Exp. Toxicol. 2017, 36, 51–61. [Google Scholar] [CrossRef]
- Erboga, M.; Aktas, C.; Erboga, Z.F.; Donmez, Y.B.; Gurel, A. Quercetin ameliorates methotrexate-induced renal damage, apoptosis and oxidative stress in rats. Ren. Fail. 2015, 37, 1492–1497. [Google Scholar] [CrossRef]
- Vicente-Vicente, L.; González-Calle, D.; Casanova, A.G.; Hernández-Sánchez, M.T.; Prieto, M.; Rama-Merchán, J.C.; Martín-Moreiras, J.; Martín-Herrero, F.; Sánchez, P.L.; López-Hernández, F.J.; et al. Quercetin, a Promising Clinical Candidate for The Prevention of Contrast-Induced Nephropathy. Int. J. Mol. Sci. 2019, 20, 4961. [Google Scholar] [CrossRef]
- Nabavi, S.M.; Nabavi, S.F.; Habtemariam, S.; Moghaddam, A.H.; Latifi, A.M. Ameliorative effects of quercetin on sodium fluoride-induced oxidative stress in rat’s kidney. Ren. Fail. 2012, 34, 901–906. [Google Scholar] [CrossRef]
- Shin, Y.J.; Kim, J.J.; Kim, Y.J.; Kim, W.H.; Park, E.Y.; Kim, I.Y.; Shin, H.S.; Kim, K.S.; Lee, E.K.; Chung, K.H.; et al. Protective Effects of Quercetin Against HgCl(2)-Induced Nephrotoxicity in Sprague-Dawley Rats. J. Med. Food 2015, 18, 524–534. [Google Scholar] [CrossRef] [PubMed]
- Bahar, E.; Lee, G.H.; Bhattarai, K.R.; Lee, H.Y.; Kim, H.K.; Handigund, M.; Choi, M.K.; Han, S.Y.; Chae, H.J.; Yoon, H. Protective role of quercetin against manganese-induced injury in the liver, kidney, and lung; and hematological parameters in acute and subchronic rat models. Drug Des. Dev. Ther. 2017, 11, 2605–2619. [Google Scholar] [CrossRef] [PubMed]
- Renugadevi, J.; Prabu, S.M. Quercetin protects against oxidative stress-related renal dysfunction by cadmium in rats. Exp. Toxicol. Pathol. 2010, 62, 471–481. [Google Scholar] [CrossRef] [PubMed]
- Gholampour, F.; Sadidi, Z. Hepatorenal protection during renal ischemia by quercetin and remote ischemic perconditioning. J. Surg. Res. 2018, 231, 224–233. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Chander, V.; Chopra, K. Quercetin, a bioflavonoid, attenuates ferric nitrilotriacetate-induced oxidative renal injury in rats. Drug Chem. Toxicol. 2004, 27, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Almaghrabi, O.A. Molecular and biochemical investigations on the effect of quercetin on oxidative stress induced by cisplatin in rat kidney. Saudi J. Biol. Sci. 2015, 22, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Kinaci, M.K.; Erkasap, N.; Kucuk, A.; Koken, T.; Tosun, M. Effects of quercetin on apoptosis, NF-kappaB and NOS gene expression in renal ischemia/reperfusion injury. Exp. Ther. Med. 2012, 3, 249–254. [Google Scholar] [CrossRef]
- Liu, J.; Li, X.; Yue, Y.; Li, J.; He, T.; He, Y. The inhibitory effect of quercetin on IL-6 production by LPS-stimulated neutrophils. Cell. Mol. Immunol. 2005, 2, 455–460. [Google Scholar]
- Shu, B.; Feng, Y.; Gui, Y.; Lu, Q.; Wei, W.; Xue, X.; Sun, X.; He, W.; Yang, J.; Dai, C. Blockade of CD38 diminishes lipopolysaccharide-induced macrophage classical activation and acute kidney injury involving NF-kappaB signaling suppression. Cell. Signal. 2018, 42, 249–258. [Google Scholar] [CrossRef]
- Chen, B.L.; Wang, L.T.; Huang, K.H.; Wang, C.C.; Chiang, C.K.; Liu, S.H. Quercetin attenuates renal ischemia/reperfusion injury via an activation of AMP-activated protein kinase-regulated autophagy pathway. J. Nutr. Biochem. 2014, 25, 1226–1234. [Google Scholar] [CrossRef] [PubMed]
- Pei, B.; Yang, M.; Qi, X.; Shen, X.; Chen, X.; Zhang, F. Quercetin ameliorates ischemia/reperfusion-induced cognitive deficits by inhibiting ASK1/JNK3/caspase-3 by enhancing the Akt signaling pathway. Biochem. Biophys. Res. Commun. 2016, 478, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Omura, S. Quercetin regulates the inhibitory effect of monoclonal non-specific suppressor factor beta on tumor necrosis factor-alpha production in LPS-stimulated macrophages. Biosci. Biotechnol. Biochem. 2008, 72, 1915–1920. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Lin, S.; Wang, M.; Huang, J.; Zhu, Y. The Risk of Acute Kidney Injury in Hepatitis B Virus-Related Acute on Chronic Liver Failure with Tenofovir Treatment. BioMed Res. Int. 2020, 2020, 1–8. [Google Scholar] [CrossRef]
- Singh, D.; Chander, V.; Chopra, K. The effect of quercetin, a bioflavonoid on ischemia/reperfusion induced renal injury in rats. Arch. Med. Res. 2004, 35, 484–494. [Google Scholar] [CrossRef] [PubMed]
| Models of Acute Kidney Injury (AKI) | Pharmacological Effect | Mechanism | Reference |
|---|---|---|---|
| Ischemia/reperfusion-induced AKI models | |||
| AKI induced by unilateral ischemia/reperfusion via left renal pedicle occlusion with simultaneous right nephrectomy | Renoprotective | Inhibited the decrease in creatinine clearance and tubular damage; | [21] |
| Anti-inflammatory | Attenuated expression of normal T-cell expressed and secretion of monocyte chemoattractant protein-1, and allograft inflammatory factor. | ||
| AKI induced by right renal and the left femoral blood vessels ischemia for 30 min followed by reperfusion in rats | Antioxidant | Reduced the renal cortex xanthine oxidase levels; Increased the renal cortex of superoxide dismutase, glutathione peroxidase, and catalase activities. | [116] |
| AKI induced by left renal pedicle occlusion for 45 min followed by 60 min of reperfusion with contralateral nephrectomy in rats | Anti-inflammatory | Reduced the TBARS, TNF-α levels, MPO activity and protein carbonyl; | [115] |
| Antioxidant | Increased the glutathione levels and the superoxide dismutase and catalase activities. | ||
| Renal pedicles occluded after nephrectomy; ischemia was given for 45 min followed by reperfusion for 24 h | Renoprotective | Attenuated the renal dysfunction via reduction in the SCr and BUN levels; Increased the creatinine and urea clearance; Reduced the morphological damage; | [135] |
| Antioxidant | Reduced the elevated lipid peroxidation; Restored the depleted renal antioxidant enzymes, such as glutathione reductase. | ||
| AKI induced by non-traumatic vascular clamp applied to the left renal pedicle for 2 h and allowed to reperfusion for 6 h | Antioxidant | Decreased the MDA levels; Increased the GSH levels; | [128] |
| Anti-inflammatory | Decreased the number of apoptotic, p53-positive cells as well as reduced the NF-κB and eNOS expressions. | ||
| AKI induced by clamping renal arteries for 45 min followed by 24 h reperfusion | Renoprotective | Sustained creatinine clearance and fractional excretion of sodium; Reduced tubular damage; | [125] |
| Antioxidant | Increased glutathione peroxidase and catalase activities; Inhibited the decrease in malondialdehyde levels. | ||
| Unilateral ureteral obstruction-induced renal fibrosis | Anti-inflammatory | Inhibited tubulointerstitial injury; Reduced the synthesis and release of inflammatory factors; Inhibited the infiltration of CD68+ macrophages in renal interstitium; Decreased levels of iNOS and IL-12, as well as the proportion of F4/80+/CD11b+/CD86+ macrophages; Inhibitory effects associated with downregulated activities of NF-κB p65 and IRF5, and thus led to the inactivation of upstream signaling TLR4/Myd88; Inhibited the polarization of F4/80+/CD11b+/CD206+ M2 macrophages; Reduced excessive accumulation of extracellular matrix and interstitial fibrosis by antagonizing the TGF-β1/Smad2/3 signaling. | [28] |
| Drug- or Toxin-induced AKI models | |||
| Fe-NTA-Induced AKI | Renoprotective | Decreased the BUN and SCr; Preserved the normal renal morphology; | [126] |
| Antioxidant | Reduced the lipid peroxidation; Restored the depleted renal antioxidant enzymes, such as glutathione reductase, catalase, superoxide dismutase, and glutathione. | ||
| Cadmium-induced AKI | Renoprotective | Reduced of BUN, SCr and uric acid levels; Protected against the Cd-induced pathological condition, as tubular necrosis, degeneration, desquamation, thickening of basement membrane and luminal cast formation; | [124] |
| Antioxidant | Decreased the renal lipid peroxidation; Increased total sulfhydryl group, glutathione, vitamin C and vitamin E, and antioxidant enzymes, such as superoxide dismutase, catalase, glutathione peroxidase, glutathione-S-transferase, glutathione reductase, and glucose 6-phosphate dehydrogenase. | ||
| Ethambutol hydrochloride-induced AKI | Renoprotective | Reduced the elevated levels of serum uric acid, BUN, and SCr; | [20] |
| Antioxidant | Increased the superoxide dismutase activity. | ||
| NaF-induced AKI | Antioxidant | Increased superoxide dismutase, glutathione, and catalase levels. | [121] |
| Methotrexate-induced AKI | Renoprotective | Reduced the renal tubular degeneration and dilation; | [118,119] |
| Antioxidant | Decreased the number of apoptotic cells and caspase-3 expression; Decreased the malondialdehyde levels; Increased superoxide dismutase, glutathione peroxidase, and catalase levels; Lowered apoptotic index. | ||
| Mercury-induced AKI | Renoprotective | Decreased the renal accumulation of Hg in the kidney; Reduced urinary excretion of protein-based biomarkers, including clusterin, KIM-1, NGAL, MCP-1, TIMP- 1, and VEGF; Protected against renal proximal tubular damage; Reduced apoptotic cell death in the kidney. | [122] |
| Valproic acid-induced AKI | Antioxidant | Decreased the lipid peroxidation and protein carbonyl; Reduced glutathione and nonprotein thiol levels. | [25] |
| Ciprofloxacin-induced AKI | Renoprotective | Reduced the tubular infiltration, dilatation, and atrophy as well as the Bowman’s space, congestion, hemorrhage, and necrosis; | [29] |
| Antioxidant | Decreased the malondialdehyde levels; Increased the superoxide dismutase and catalase activities. | ||
| Manganese-induced AKI | Renoprotective | Counteracted Mn-induced morphological glomerular damage; Decreased the expression of GRP78, CHOP, and caspase-3 proteins. | [123] |
| Cisplatin-induced AKI | Renoprotective | Maintenance of renal blood flow, BUN and SCr levels, and sodium fractional excretion; Decreased the NGAL and KIM-1 excretion; | [24,117,126] |
| Antioxidant | Reduced the rise in MDA and protein carbonyl; Increased the GSH, vitamin C, vitamin E, total antioxidant capacity, and total glutathione levels in the kidney tissue; Induced the gene expression and activities of catalase, superoxide dismutase, glutathione reductase, and glutathione peroxidase enzymes in the kidney tissue; | ||
| Anti-inflammatory | Inhibited expression and secretion of IL-1β, IL-6, and TNF-α; Reduced the activity of Mincle/Syk/NF-κB signaling in vitro; Downregulated the protein levels of Mincle, phosphorylated Syk, and NF-κB in kidney macrophages; Inhibited M1, upregulated M2 macrophage activity; Reduced the early activation of stress kinases ERK, JNK and p38. Lowered NF-κB pathway and effector caspase activations. | ||
| Contrast-induced AKI | Renoprotective | Inhibited the increase in SCr and albuminuria accompanied by a lower decrease in the GFR. | [120] |
| Sepsis-induced AKI models | |||
| Lipopolysaccharide-induced AKI | Renoprotective | Relieved kidney dysfunction; Decreased the histopathological damage; Lowered the BUN and SCr levels. Reduced the inflammatory cell accumulation; | [24,130] |
| Anti-inflammatory | Inhibited the Toll-like receptor-4, MyD88, and TRAF-6 expressions and NF-κBp65 activation in the kidneys; Inhibited the TNF-α, IL-1β, and IL-6 levels; Blockaded the CD38 expression of the macrophages possessing ADP-ribosyl cyclase and cyclic ADP-ribose hydrolase; Inhibited the LPS-induced macrophage M1 polarization accompanied by diminished NF-κB signaling activation. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diniz, L.R.L.; Souza, M.T.d.S.; Duarte, A.B.S.; Sousa, D.P.d. Mechanistic Aspects and Therapeutic Potential of Quercetin against COVID-19-Associated Acute Kidney Injury. Molecules 2020, 25, 5772. https://doi.org/10.3390/molecules25235772
Diniz LRL, Souza MTdS, Duarte ABS, Sousa DPd. Mechanistic Aspects and Therapeutic Potential of Quercetin against COVID-19-Associated Acute Kidney Injury. Molecules. 2020; 25(23):5772. https://doi.org/10.3390/molecules25235772
Chicago/Turabian StyleDiniz, Lúcio Ricardo Leite, Marilia Trindade de Santana Souza, Allana Brunna Sucupira Duarte, and Damião Pergentino de Sousa. 2020. "Mechanistic Aspects and Therapeutic Potential of Quercetin against COVID-19-Associated Acute Kidney Injury" Molecules 25, no. 23: 5772. https://doi.org/10.3390/molecules25235772
APA StyleDiniz, L. R. L., Souza, M. T. d. S., Duarte, A. B. S., & Sousa, D. P. d. (2020). Mechanistic Aspects and Therapeutic Potential of Quercetin against COVID-19-Associated Acute Kidney Injury. Molecules, 25(23), 5772. https://doi.org/10.3390/molecules25235772







