Vibrational Spectroscopy for In Vitro Monitoring Stem Cell Differentiation
Abstract
1. Introduction
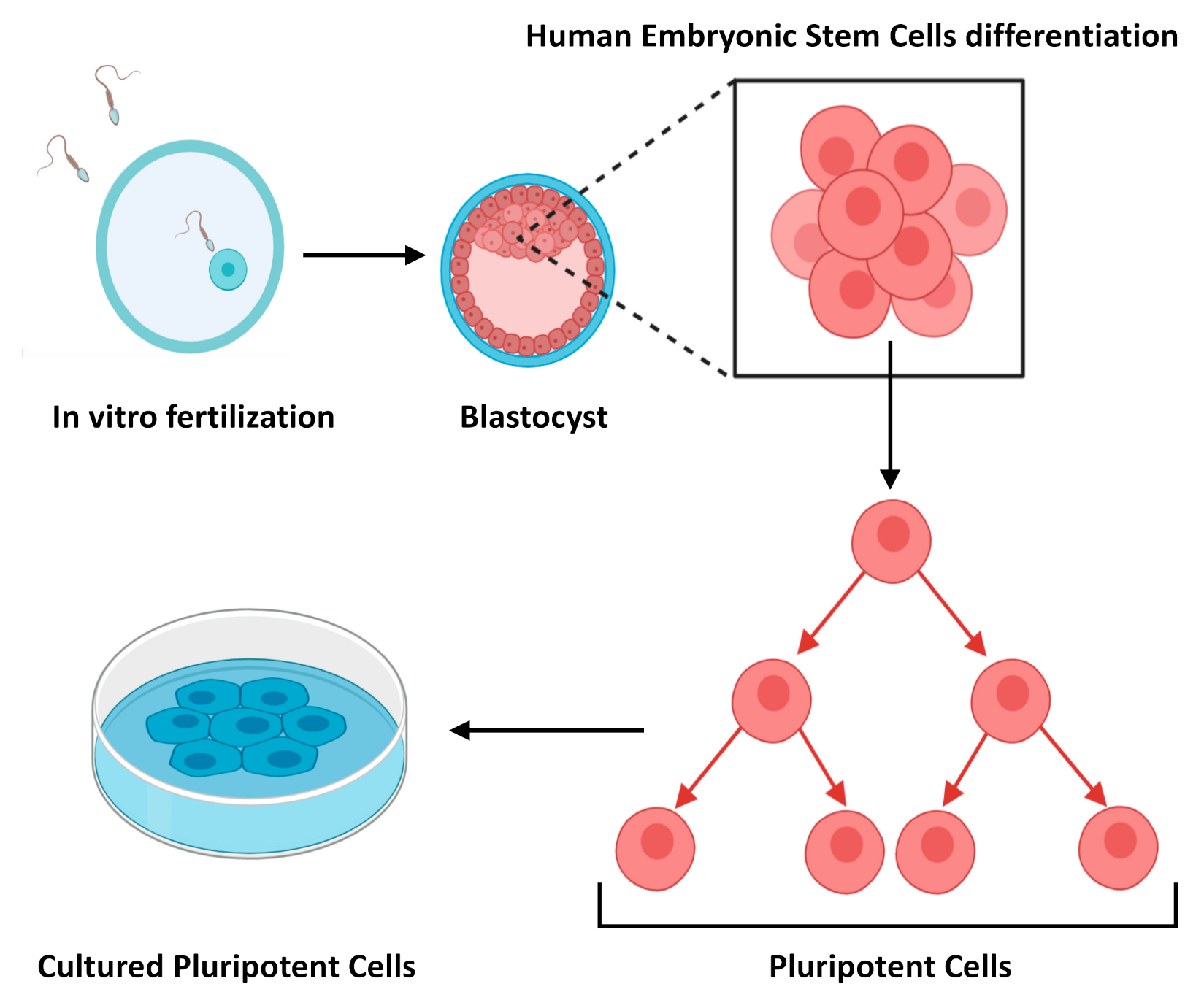
Stem Cells and Stem Cell Technology
2. Vibrational Spectroscopy for Stem Cell Characterisation and Differentiation
2.1. Applications of IR and Raman Spectroscopy to Characteristion of Stem Cell Differentiation: Cellular Studies
2.1.1. Embryonic Stem Cells
2.1.2. Mesenchymal Stem Cell
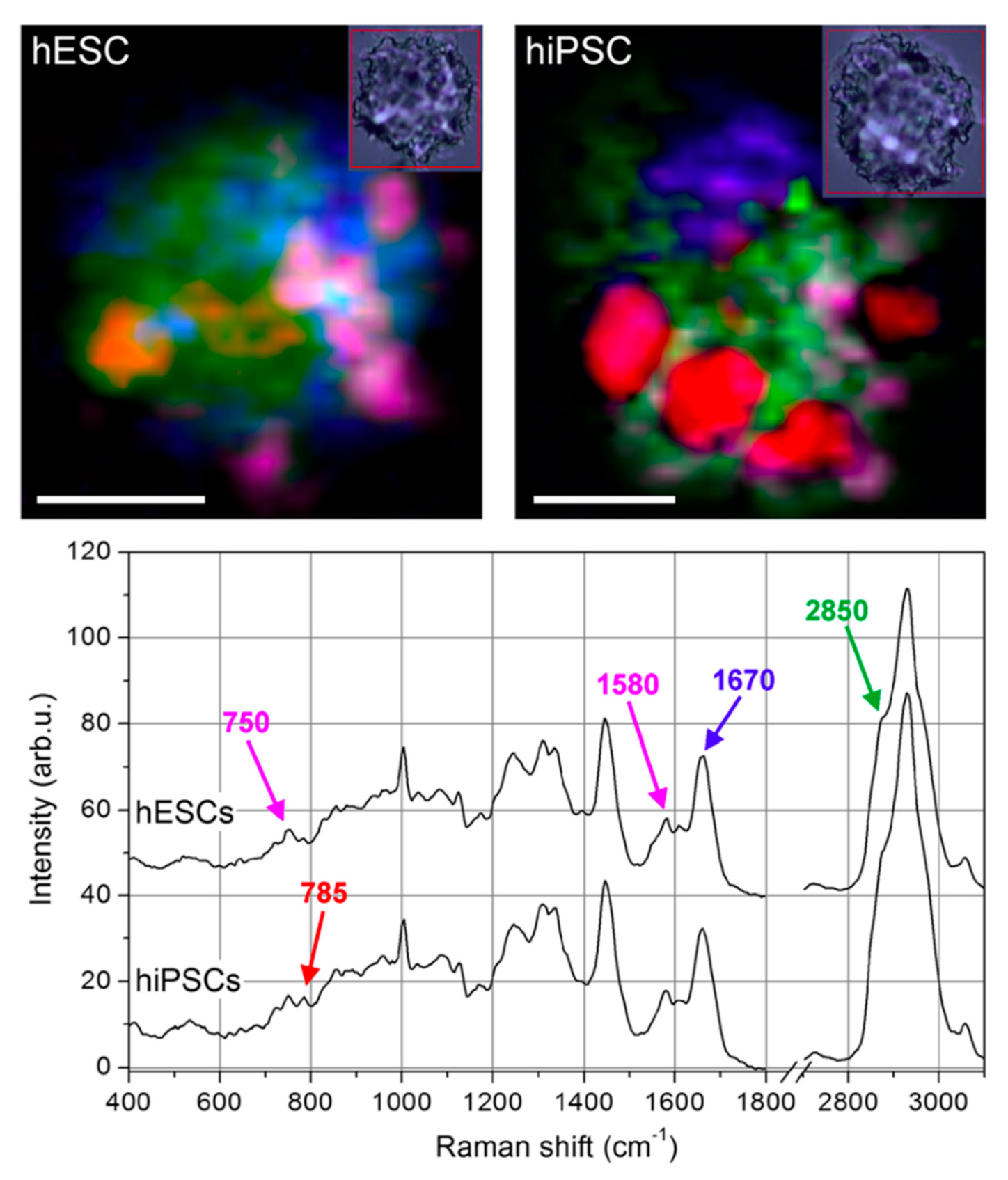
2.1.3. Induced Pluripotent Stem Cells
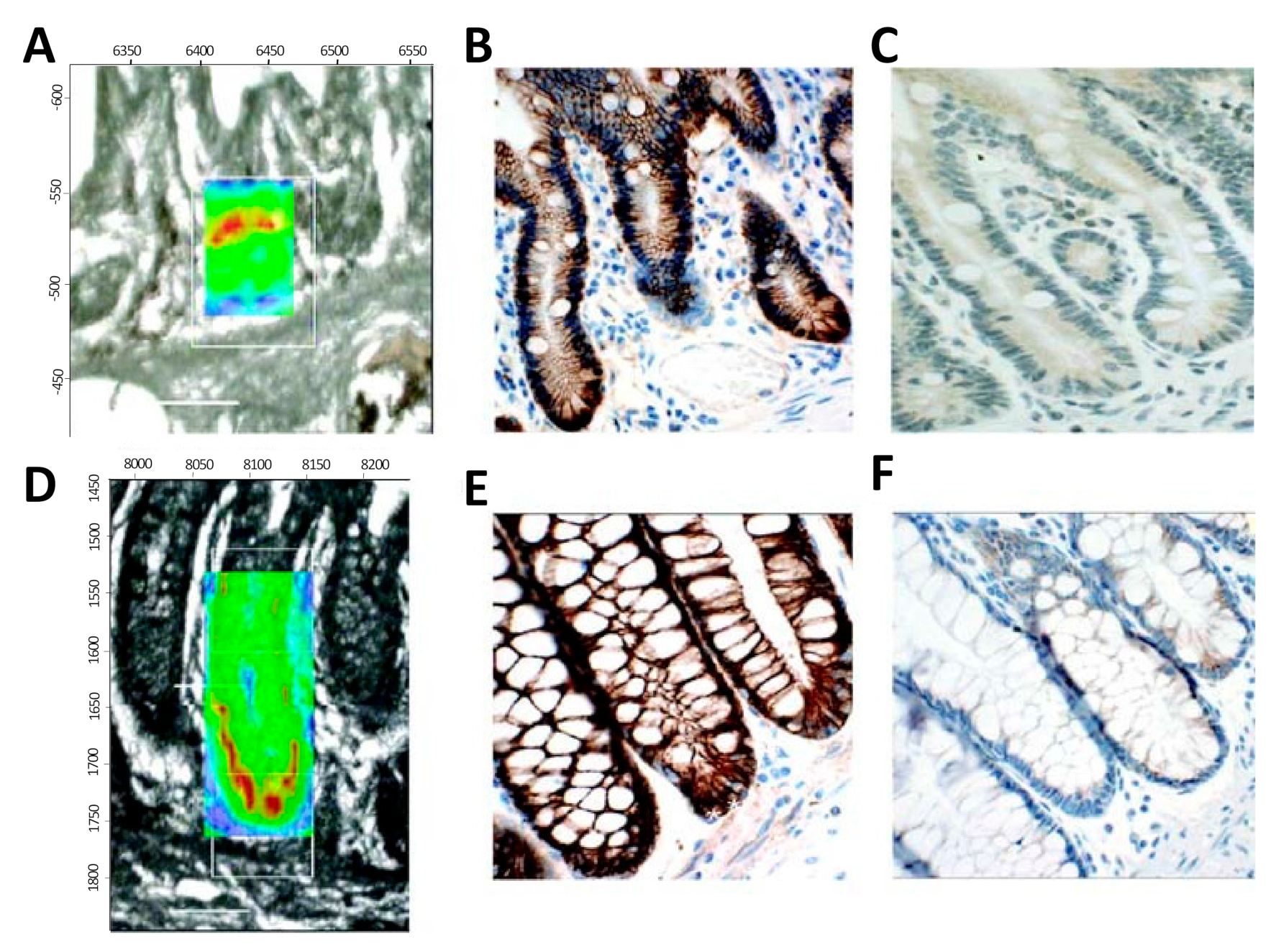
2.2. Applications of IR and Raman Spectroscopy to Characteristion of Stem Cell Differentiation: Tissue Studies
3. Future Perspectives on the Use of Vibrational Spectroscopy for Stem Cell Technology; Data Analysis and Machine Learning, 3D Cultures and Nanotechnology
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sell, S. Stem Cell Handbook, 2nd ed.; Sell, S., Ed.; Humana Press, Division of Translational Medicine, Wadsworth Center, New York State Department of Health: Albany, NY, USA, 2013. [Google Scholar] [CrossRef]
- Evans, M.J.; Kaufman, M.H. Establishment in culture of pluripotential cells from mouse embryos. Nature 1981, 292, 154–156. [Google Scholar] [CrossRef] [PubMed]
- Thomson, J.A.; Itskovitz-Eldor, J.; Shapiro, S.S.; Waknitz, M.A.; Swiergiel, J.J.; Marshall, V.S.; Jones, J.M. Embryonic stem cell lines derived from human blastocysts. Science 1998, 282, 1145–1147. [Google Scholar] [CrossRef] [PubMed]
- Pierce, G.B. Teratocarcinoma: Model for a developmental concept of cancer. Curr. Top Dev. Biol. 1967, 2, 223–246. [Google Scholar] [CrossRef] [PubMed]
- Stevens, L.C.; Hummel, K.P. A description of spontaneous congenital testicular teratomas in strain 129 mice. J. Natl. Cancer Inst. 1957, 18, 719–747. [Google Scholar] [PubMed]
- Andrews, P.W. From teratocarcinomas to embryonic stem cells. Philos. Trans. R. Soc. B Biol. Sci. 2002, 357, 405–417. [Google Scholar] [CrossRef]
- Odorico, J.S.; Kaufman, D.S.; Thomson, J.A. Multilineage differentiation from human embryonic stem cell lines. Stem Cells 2001, 19, 193–204. [Google Scholar] [CrossRef]
- Lo, B.; Parham, L. Ethical issues in Stem Cell Res.earch. Endocr. Rev. 2009, 30, 204–213. [Google Scholar] [CrossRef]
- Wang, S.; Qu, X.; Zhao, R.C. Clinical applications of mesenchymal stem cells. J. Hematol. Oncol. 2012, 5, 19. [Google Scholar] [CrossRef]
- Friedenstein, A.J.; Petrakova, K.V.; Kurolesova, A.I.; Frolova, G.P. Heterotopic of bone marrow. Analysis of precursor cells for osteogenic and hematopoietic tissues. Transplantation 1968, 6, 230–247. [Google Scholar] [CrossRef]
- Prockop, D.J. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science 1997, 276, 71–74. [Google Scholar] [CrossRef]
- Keating, A.; Singer, J.W.; Killen, P.D.; Striker, G.E.; Salo, A.C.; Sanders, J.; Thomas, E.D.; Thorning, D.; Fialkow, P.J. Donor origin of the in vitro haematopoietic microenvironment after marrow transplantation in man. Nature 1982, 298, 280–283. [Google Scholar] [CrossRef] [PubMed]
- Owen, M.; Friedenstein, A.J. Stromal stem cells: Marrow-derived osteogenic precursors. Ciba Found. Symp. 1988, 136, 42–60. [Google Scholar] [CrossRef] [PubMed]
- Schipani, E.; Kronenberg, H.M. Adult mesenchymal stem cells. In StemBook; Harvard Stem Cell Institute: Cambridge, MA, USA, 2008. [Google Scholar] [CrossRef]
- Ghita, A.; Pascut, F.C.; Sottile, V.; Denning, C.; Notingher, I. Applications of Raman micro-spectroscopy to stem cell technology: Label-free molecular discrimination and monitoring cell differentiation. EPJ Tech. Instrum. 2015, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Abdelwahid, E.; Siminiak, T.; Guarita-Souza, L.C.; Teixeira de Carvalho, K.A.; Gallo, P.; Shim, W.; Condorelli, G. Stem cell therapy in heart diseases: A review of selected new perspectives, practical considerations and clinical applications. Curr. Cardiol. Rev. 2011, 7, 201–212. [Google Scholar] [CrossRef] [PubMed]
- Yi, T.; Song, S.U. Immunomodulatory properties of mesenchymal stem cells and their therapeutic applications. Arch. Pharm. Res. 2012, 35, 213–221. [Google Scholar] [CrossRef]
- Takahashi, K.; Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 2006, 126, 663–676. [Google Scholar] [CrossRef]
- Takahashi, K.; Tanabe, K.; Ohnuki, M.; Narita, M.; Ichisaka, T.; Tomoda, K.; Yamanaka, S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 2007, 131, 861–872. [Google Scholar] [CrossRef]
- Vats, A.; Bielby, R.C.; Tolley, N.S.; Nerem, R.; Polak, J.M. Stem cells. Lancet 2005, 366, 592–602. [Google Scholar] [CrossRef]
- Ng, S.A.; Sullivan, K.M. Application of stem cell transplantation in autoimmune diseases. Curr. Opin. Hematol. 2019, 26, 392–398. [Google Scholar] [CrossRef]
- Van Bekkum, D.W. Stem cell transplantation in experimental models of autoimmune disease. J. Clin. Immunol. 2000, 20, 10–16. [Google Scholar] [CrossRef]
- Alvarez-Buylla, A.; Theelen, M.; Nottebohm, F. Proliferation "hot spots" in adult avian ventricular zone reveal radial cell division. Neuron 1990, 5, 101–109. [Google Scholar] [CrossRef]
- Chen, M.; Przyborowski, M.; Berthiaume, F. Stem cells for skin tissue engineering and wound healing. Crit. Rev. Biomed. Eng. 2009, 37, 399–421. [Google Scholar] [CrossRef] [PubMed]
- Nourian Dehkordi, A.; Mirahmadi Babaheydari, F.; Chehelgerdi, M.; Raeisi Dehkordi, S. Skin tissue engineering: Wound healing based on stem-cell-based therapeutic strategies. Stem Cell Res. Ther. 2019, 10, 111. [Google Scholar] [CrossRef]
- Lapidot, T.; Sirard, C.; Vormoor, J.; Murdoch, B.; Hoang, T.; Caceres-Cortes, J.; Minden, M.; Paterson, B.; Caligiuri, M.A.; Dick, J.E. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 1994, 367, 645–648. [Google Scholar] [CrossRef] [PubMed]
- Scott, C.T.; Magnus, D. Wrongful termination: Lessons from the Geron clinical trial. Stem Cells Transl. Med. 2014, 3, 1398–1401. [Google Scholar] [CrossRef]
- Lebkowski, J. GRNOPC1: The world’s first embryonic stem cell-derived therapy. Interview with Jane Lebkowski. Regen. Med. 2011, 6, 11–13. [Google Scholar] [CrossRef]
- Sulé-Suso, J.; Forsyth, N.R.; Untereiner, V.; Sockalingum, G.D. Vibrational spectroscopy in stem cell characterisation: Is there a niche? Trends Biotechnol. 2014, 32, 254–262. [Google Scholar] [CrossRef]
- Nagano, K.; Yoshida, Y.; Isobe, T. Cell surface biomarkers of embryonic stem cells. Proteomics 2008, 8, 4025–4035. [Google Scholar] [CrossRef]
- Tacchetti, C.; Tavella, S.; Dozin, B.; Quarto, R.; Robino, G.; Cancedda, R. Cell condensation in chondrogenic differentiation. Exp. Cell Res. 1992, 200, 26–33. [Google Scholar] [CrossRef]
- Pruszak, J.; Sonntag, K.C.; Aung, M.H.; Sanchez-Pernaute, R.; Isacson, O. Markers and methods for cell sorting of human embryonic stem cell-derived neural cell populations. Stem Cells 2007, 25, 2257–2268. [Google Scholar] [CrossRef]
- Fukuda, H.; Takahashi, J.; Watanabe, K.; Hayashi, H.; Morizane, A.; Koyanagi, M.; Sasai, Y.; Hashimoto, N. Fluorescence-activated cell sorting-based purification of embryonic stem cell-derived neural precursors averts tumor formation after transplantation. Stem Cells 2006, 24, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, S.; Burridge, P.W.; Kropp, E.M.; Chuppa, S.L.; Kwok, W.M.; Wu, J.C.; Boheler, K.R.; Gundry, R.L. High efficiency differentiation of human pluripotent stem cells to cardiomyocytes and characterization by flow cytometry. J. Vis. Exp. 2014, 52010. [Google Scholar] [CrossRef] [PubMed]
- Nairn, A.V.; dela Rosa, M.; Moremen, K.W. Transcript analysis of stem cells. Methods Enzymol. 2010, 479, 73–91. [Google Scholar] [CrossRef] [PubMed]
- Noaksson, K.; Zoric, N.; Zeng, X.; Rao, M.S.; Hyllner, J.; Semb, H.; Kubista, M.; Sartipy, P. Monitoring differentiation of human embryonic stem cells using real-time PCR. Stem Cells 2005, 23, 1460–1467. [Google Scholar] [CrossRef]
- Byrne, H.J.; Baranska, M.; Puppels, G.J.; Stone, N.; Wood, B.; Gough, K.M.; Lasch, P.; Heraud, P.; Sulé-Suso, J.; Sockalingum, G.D. Spectropathology for the next generation: Quo vadis? Analyst 2015, 140, 2066–2073. [Google Scholar] [CrossRef]
- Baker, M.J.; Byrne, H.J.; Chalmers, J.; Gardner, P.; Goodacre, R.; Henderson, A.; Kazarian, S.G.; Martin, F.L.; Moger, J.; Stone, N.; et al. Clinical applications of infrared and Raman spectroscopy: State of play and future challenges. Analyst 2018, 143, 1735–1757. [Google Scholar] [CrossRef]
- Byrne, H.J.; Sockalingum, G.; Stone, N. Raman Microscopy: Complement or Competitor? In Biomedical Applications of Synchrotron Infrared Microspectroscopy: A Practical Approach; Moss, D., Ed.; Royal Society of Chemistry: London, UK, 2010; pp. 105–143. [Google Scholar] [CrossRef]
- Jackson, M.; Mantsch, H.H. The use and misuse of FTIR spectroscopy in the determination of protein structure. Crit. Rev. Biochem. Mol. Biol. 1995, 30, 95–120. [Google Scholar] [CrossRef]
- Casal, H.L.; Mantsch, H.H. Polymorphic phase behaviour of phospholipid membranes studied by infrared spectroscopy. Biochim. Biophys. Acta 1984, 779, 381–401. [Google Scholar] [CrossRef]
- Mathlouthi, M.; Koenig, J.L. Vibrational spectra of carbohydrates. Adv. Carbohydr. Chem. Biochem. 1986, 44, 7–89. [Google Scholar] [CrossRef]
- Wilkinson, G.R.; Clark, R.J.H.; Hester, R.E. (Eds.) Advances in infrared and Raman spectroscopy, volume 12. Wiley, New York. 1985. J. Raman Spectrosc. 1986, 17, 487. [Google Scholar] [CrossRef]
- Wong, P.T.T.; Lacelle, S.; Hossein, M.Y. Normal and Malignant Human Colonic Tissues Investigated by Pressure-Tuning FT-IR Spectroscopy. Appl. Spectrosc. 1993, 47, 1830–1836. [Google Scholar] [CrossRef]
- Dukor, R.K. Vibrational Spectroscopy in the Detection of Cancer. In Handbook of Vibrational Spectroscopy; Chalmers, J.M., Griffiths, P.R., Eds.; Wiley: Hoboken, NJ, USA, 2006. [Google Scholar] [CrossRef]
- Gazi, E.; Baker, M.; Dwyer, J.; Lockyer, N.P.; Gardner, P.; Shanks, J.H.; Reeve, R.S.; Hart, C.A.; Clarke, N.W.; Brown, M.D. A correlation of FTIR spectra derived from prostate cancer biopsies with gleason grade and tumour stage. Eur. Urol. 2006, 50, 750–760, discussion 760-751. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, D.C.; Bhargava, R.; Hewitt, S.M.; Levin, I.W. Infrared spectroscopic imaging for histopathologic recognition. Nature Biotechnol. 2005, 23, 469–474. [Google Scholar] [CrossRef]
- Lord, R.C.; Yu, N.T. Laser-excited Raman spectroscopy of biomolecules: I. Native lysozyme and its constituent amino acids. J. Mol. Biol. 1970, 50, 509–524. [Google Scholar] [CrossRef]
- Walton, A.G.; Deveney, M.J.; Koenig, J.L. Raman spectroscopy of calcified tissue. Calcif. Tissue Res. 1970, 6, 162–167. [Google Scholar] [CrossRef]
- Tobin, M.C. Raman spectra of crystalline lysozyme, pepsin, and alpha chymotrypsin. Science 1968, 161, 68–69. [Google Scholar] [CrossRef]
- Yu, N.T.; Robert, B.H.J.; Chang, C.C.; Huber, J.D. Single-crystal raman spectra of native insulin: Structures of insulin fibrils, glucagon fibrils, and intact calf lens. Arch. Biochem. Biophys. 1974, 160, 614–622. [Google Scholar] [CrossRef]
- Puppels, G.J. Whole Cell Studies and Tissue Characterization by RamanSpectroscopy. In Biomedical Applications of Spectroscopy; Clark, R.J.H., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 1996; p. 402. [Google Scholar]
- Ewen, S.; Geoffrey, D. Modern Raman Spectroscopy-A Pratical Approach; John Weley & Sons, Ltd: The Atrium, West Sussex, UK, 2005. [Google Scholar]
- Smith, J.; Kendall, C.; Sammon, A.; Christie-Brown, J.; Stone, N. Raman spectral mapping in the assessment of axillary lymph nodes in breast cancer. Technol. Cancer Res. Treat. 2003, 2, 327–332. [Google Scholar] [CrossRef]
- Molckovsky, A.; Song, L.M.; Shim, M.G.; Marcon, N.E.; Wilson, B.C. Diagnostic potential of near-infrared Raman spectroscopy in the colon: Differentiating adenomatous from hyperplastic polyps. Gastrointest. Endosc. 2003, 57, 396–402. [Google Scholar] [CrossRef]
- Gniadecka, M.; Wulf, H.C.; Nielsen, O.F.; Christensen, D.H.; Hercogova, J. Distinctive molecular abnormalities in benign and malignant skin lesions: Studies by Raman spectroscopy. Photochem. Photobiol. 1997, 66, 418–423. [Google Scholar] [CrossRef]
- Hanlon, E.B.; Manoharan, R.; Koo, T.W.; Shafer, K.E.; Motz, J.T.; Fitzmaurice, M.; Kramer, J.R.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Prospects for in vivo Raman spectroscopy. Phys. Med. Biol. 2000, 45, R1–R59. [Google Scholar] [CrossRef] [PubMed]
- Caspers, P.J.; Lucassen, G.W.; Wolthuis, R.; Bruining, H.A.; Puppels, G.J. In vitro and in vivo Raman spectroscopy of human skin. Biospectroscopy 1998, 4, S31–S39. [Google Scholar] [CrossRef]
- Boydston-White, S.; Romeo, M.; Chernenko, T.; Regina, A.; Miljkovic, M.; Diem, M. Cell-cycle-dependent variations in FTIR micro-spectra of single proliferating HeLa cells: Principal component and artificial neural network analysis. Biochim. Biophys. Acta 2006, 1758, 908–914. [Google Scholar] [CrossRef]
- Matthaus, C.; Boydston-White, S.; Miljkovic, M.; Romeo, M.; Diem, M. Raman and infrared microspectral imaging of mitotic cells. Appl. Spectrosc. 2006, 60, 1–8. [Google Scholar] [CrossRef]
- Short, K.W.; Carpenter, S.; Freyer, J.P.; Mourant, J.R. Raman spectroscopy detects biochemical changes due to proliferation in mammalian cell cultures. Biophys. J. 2005, 88, 4274–4288. [Google Scholar] [CrossRef] [PubMed]
- Ami, D.; Neri, T.; Natalello, A.; Mereghetti, P.; Doglia, S.M.; Zanoni, M.; Zuccotti, M.; Garagna, S.; Redi, C.A. Embryonic stem cell differentiation studied by FT-IR spectroscopy. Biochim. Biophys. Acta 2008, 1783, 98–106. [Google Scholar] [CrossRef]
- Notingher, I.; Hench, L.L. Raman microspectroscopy: A noninvasive tool for studies of individual living cells in vitro. Expert Rev. Med. Devices 2006, 3, 215–234. [Google Scholar] [CrossRef]
- Notingher, I.; Bisson, I.; Bishop, A.E.; Randle, W.L.; Polak, J.M.; Hench, L.L. In situ spectral monitoring of mRNA translation in embryonic stem cells during differentiation in vitro. Anal. Chem. 2004, 76, 3185–3193. [Google Scholar] [CrossRef]
- Meade, A.D.; Lyng, F.M.; Knief, P.; Byrne, H.J. Growth substrate induced functional changes elucidated by FTIR and Raman spectroscopy in in-vitro cultured human keratinocytes. Anal. Bioanal. Chem. 2007, 387, 1717–1728. [Google Scholar] [CrossRef]
- Liu, K.Z.; Jia, L.; Kelsey, S.M.; Newland, A.C.; Mantsch, H.H. Quantitative determination of apoptosis on leukemia cells by infrared spectroscopy. Apoptosis 2001, 6, 269–278. [Google Scholar] [CrossRef]
- Notingher, I.; Verrier, S.; Haque, S.; Polak, J.M.; Hench, L.L. Spectroscopic study of human lung epithelial cells (A549) in culture: Living cells versus dead cells. Biopolymers 2003, 72, 230–240. [Google Scholar] [CrossRef] [PubMed]
- Zelig, U.; Dror, Z.; Iskovich, S.; Zwielly, A.; Ben-Harush, M.; Nathan, I.; Mordechai, S.; Kapelushnik, J. Biochemical analysis and quantification of hematopoietic stem cells by infrared spectroscopy. J. Biomed. Opt. 2010, 15, 037008. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wang, Z.; Sun, S.; Liu, M.; Zhang, H. A rapid method for detecting conformational changes during differentiation and apoptosis of HL60 cells by Fourier-transform infrared spectroscopy. Biotechnol. Appl. Biochem. 2001, 33, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Sulé-Suso, J.; Sockalingum, G.D.; Kegelaer, G.; Manfait, M.; El Haj, A.J. Study of tumor cell invasion by Fourier transform infrared microspectroscopy. Biopolymers 2005, 78, 311–317. [Google Scholar] [CrossRef] [PubMed]
- van Manen, H.J.; Kraan, Y.M.; Roos, D.; Otto, C. Single-cell Raman and fluorescence microscopy reveal the association of lipid bodies with phagosomes in leukocytes. Proc. Natl. Acad. Sci. USA 2005, 102, 10159–10164. [Google Scholar] [CrossRef]
- Farhane, Z.; Nawaz, H.; Bonnier, F.; Byrne, H.J. In vitro label-free screening of chemotherapeutic drugs using Raman microspectroscopy: Towards a new paradigm of spectralomics. J. Biophotonics 2018, 11, e201700258. [Google Scholar] [CrossRef]
- Efeoglu, E.; Maher, M.A.; Casey, A.; Byrne, H.J. Toxicological assessment of nanomaterials: The role of in vitro Raman microspectroscopic analysis. Anal. Bioanal. Chem. 2018, 410, 1631–1646. [Google Scholar] [CrossRef]
- Molony, C.; McIntyre, J.; Maguire, A.; Hakimjavadi, R.; Burtenshaw, D.; Casey, G.; Di Luca, M.; Hennelly, B.; Byrne, H.J.; Cahill, P.A. Label-free discrimination analysis of de-differentiated vascular smooth muscle cells, mesenchymal stem cells and their vascular and osteogenic progeny using vibrational spectroscopy. Biochim. Biophys. Acta Mol. Cell Res. 2018, 1865, 343–353. [Google Scholar] [CrossRef]
- Kerr, L.T.; Hennelly, B.M. A multivariate statistical investigation of background subtraction algorithms for Raman spectra of cytology samples recorded on glass slides. Chemom. Intell. Lab. Syst. 2016, 158, 61–68. [Google Scholar] [CrossRef]
- Kerr, L.T.; Lynn, T.M.; Cullen, I.M.; Daly, P.J.; Shah, N.; O’Dea, S.; Malkin, A.; Hennelly, B.M. Methodologies for bladder cancer detection with Raman based urine cytology. Anal. Methods 2016, 8, 4991–5000. [Google Scholar] [CrossRef]
- Bonnier, F.; Byrne, H.J. Understanding the molecular information contained in principal component analysis of vibrational spectra of biological systems. Analyst 2012, 137, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Notingher, I. Raman Spectroscopy Cell-based Biosensors. Sensors 2007, 137, 322–332. [Google Scholar] [CrossRef]
- Movasaghi, Z.; Rehman, S.; Rehman, I.U. Raman Spectroscopy of Biological Tissues. Appl. Spectrosc. Rev. 2014, 50, 46–111. [Google Scholar] [CrossRef]
- Aksoy, C.; Severcan, F. Role of Vibrational Spectroscopy in Stem Cell Research. J. Spectrosc. 2012, 27, 167–184. [Google Scholar] [CrossRef]
- Notingher, I.; Bisson, I.; Polak, J.M.; Hench, L.L. In situ spectroscopic study of nucleic acids in differentiating embryonic stem cells. Vib. Spectrosc. 2004, 35, 199–203. [Google Scholar] [CrossRef]
- Chan, J.W.; Lieu, D.K.; Huser, T.; Li, R.A. Label-free separation of human embryonic stem cells and their cardiac derivatives using Raman spectroscopy. Anal. Chem. 2009, 81, 1324–1331. [Google Scholar] [CrossRef]
- Tanthanuch, W.; Thumanu, K.; Lorthongpanich, C.; Parnpai, R.; Heraud, P. Neural differentiation of mouse embryonic stem cells studied by FTIR spectroscopy. J. Mol. Struct. 2010, 967, 189–195. [Google Scholar] [CrossRef]
- Heraud, P.; Ng, E.S.; Caine, S.; Yu, Q.C.; Hirst, C.; Mayberry, R.; Bruce, A.; Wood, B.R.; McNaughton, D.; Stanley, E.G.; et al. Fourier transform infrared microspectroscopy identifies early lineage commitment in differentiating human embryonic stem cells. Stem Cell Res. 2010, 4, 140–147. [Google Scholar] [CrossRef]
- Thumanu, K.; Tanthanuch, W.; Ye, D.; Sangmalee, A.; Lorthongpanich, C.; Parnpai, R.; Heraud, P. Spectroscopic signature of mouse embryonic stem cell-derived hepatocytes using synchrotron Fourier transform infrared microspectroscopy. J. Biomed. Opt. 2011, 16, 057005. [Google Scholar] [CrossRef]
- Schulze, H.G.; Konorov, S.O.; Caron, N.J.; Piret, J.M.; Blades, M.W.; Turner, R.F. Assessing differentiation status of human embryonic stem cells noninvasively using Raman microspectroscopy. Anal. Chem. 2010, 82, 5020–5027. [Google Scholar] [CrossRef]
- Pascut, F.C.; Kalra, S.; George, V.; Welch, N.; Denning, C.; Notingher, I. Non-invasive label-free monitoring the cardiac differentiation of human embryonic stem cells in-vitro by Raman spectroscopy. Biochim. Biophys. Acta 2013, 1830, 3517–3524. [Google Scholar] [CrossRef] [PubMed]
- Pascut, F.C.; Goh, H.T.; Welch, N.; Buttery, L.D.; Denning, C.; Notingher, I. Noninvasive detection and imaging of molecular markers in live cardiomyocytes derived from human embryonic stem cells. Biophys. J. 2011, 100, 251–259. [Google Scholar] [CrossRef]
- Konorov, S.O.; Schulze, H.G.; Atkins, C.G.; Piret, J.M.; Aparicio, S.A.; Turner, R.F.; Blades, M.W. Absolute quantification of intracellular glycogen content in human embryonic stem cells with Raman microspectroscopy. Anal. Chem. 2011, 83, 6254–6258. [Google Scholar] [CrossRef] [PubMed]
- Konorov, S.O.; Schulze, H.G.; Piret, J.M.; Aparicio, S.A.; Turner, R.F.; Blades, M.W. Raman microscopy-based cytochemical investigations of potential niche-forming inhomogeneities present in human embryonic stem cell colonies. Appl. Spectrosc. 2011, 65, 1009–1016. [Google Scholar] [CrossRef] [PubMed]
- Konorov, S.O.; Glover, C.H.; Piret, J.M.; Bryan, J.; Schulze, H.G.; Blades, M.W.; Turner, R.F. In situ analysis of living embryonic stem cells by coherent anti-stokes Raman microscopy. Anal. Chem. 2007, 79, 7221–7225. [Google Scholar] [CrossRef]
- Downes, A.; Mouras, R.; Bagnaninchi, P.; Elfick, A. Raman spectroscopy and CARS microscopy of stem cells and their derivatives. J. Raman Spectrosc. 2011, 42, 1864–1870. [Google Scholar] [CrossRef]
- Tsikritsis, D.; Shi, H.; Wang, Y.; Velugotla, S.; Sršeň, V.; Elfick, A.; Downes, A. Label-free biomarkers of human embryonic stem cell differentiation to hepatocytes. Cytom. A 2016, 89, 575–584. [Google Scholar] [CrossRef]
- Tan, Y.; Konorov, S.O.; Schulze, H.G.; Piret, J.M.; Blades, M.W.; Turner, R.F. Comparative study using Raman microspectroscopy reveals spectral signatures of human induced pluripotent cells more closely resemble those from human embryonic stem cells than those from differentiated cells. Analyst 2012, 137, 4509–4515. [Google Scholar] [CrossRef]
- Parrotta, E.; De Angelis, M.T.; Scalise, S.; Candeloro, P.; Santamaria, G.; Paonessa, M.; Coluccio, M.L.; Perozziello, G.; De Vitis, S.; Sgura, A.; et al. Two sides of the same coin? Unraveling subtle differences between human embryonic and induced pluripotent stem cells by Raman spectroscopy. Stem Cell Res. Ther. 2017, 8, 271. [Google Scholar] [CrossRef]
- Jiang, Y.; Jahagirdar, B.N.; Reinhardt, R.L.; Schwartz, R.E.; Keene, C.D.; Ortiz-Gonzalez, X.R.; Reyes, M.; Lenvik, T.; Lund, T.; Blackstad, M.; et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 2002, 418, 41–49. [Google Scholar] [CrossRef]
- Gentleman, E.; Swain, R.J.; Evans, N.D.; Boonrungsiman, S.; Jell, G.; Ball, M.D.; Shean, T.A.; Oyen, M.L.; Porter, A.; Stevens, M.M. Comparative materials differences revealed in engineered bone as a function of cell-specific differentiation. Nat. Mater. 2009, 8, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Ishii, K.; Kimura, A.; Kushibiki, T.; Awazu, K. Fourier transform infrared spectroscopic analysis of cell differentiation. Biomed. Opt. (BiOS) 2007, 6439, 80–86. [Google Scholar] [CrossRef]
- Azrad, E.; Zahor, D.; Vago, R.; Nevo, Z.; Doron, R.; Robinson, D.; Gheber, L.A.; Rosenwaks, S.; Bar, I. Probing the effect of an extract of elk velvet antler powder on mesenchymal stem cells using Raman microspectroscopy: Enhanced differentiation toward osteogenic fate. J. Raman Spectrosc. 2006, 37, 480–486. [Google Scholar] [CrossRef]
- McManus, L.L.; Burke, G.A.; McCafferty, M.M.; O’Hare, P.; Modreanu, M.; Boyd, A.R.; Meenan, B.J. Raman spectroscopic monitoring of the osteogenic differentiation of human mesenchymal stem cells. Analyst 2011, 136, 2471–2481. [Google Scholar] [CrossRef] [PubMed]
- McManus, L.L.; Bonnier, F.; Burke, G.A.; Meenan, B.J.; Boyd, A.R.; Byrne, H.J. Assessment of an osteoblast-like cell line as a model for human primary osteoblasts using Raman spectroscopy. Analyst 2012, 137, 1559–1569. [Google Scholar] [CrossRef]
- Krafft, C.; Salzer, R.; Seitz, S.; Ern, C.; Schieker, M. Differentiation of individual human mesenchymal stem cells probed by FTIR microscopic imaging. Analyst 2007, 132, 647–653. [Google Scholar] [CrossRef]
- Moody, B.; Haslauer, C.M.; Kirk, E.; Kannan, A.; Loboa, E.G.; McCarty, G.S. In situ monitoring of adipogenesis with human-adipose-derived stem cells using surface-enhanced Raman spectroscopy. Appl. Spectrosc. 2010, 64, 1227–1233. [Google Scholar] [CrossRef]
- Chonanant, C.; Jearanaikoon, N.; Leelayuwat, C.; Limpaiboon, T.; Tobin, M.J.; Jearanaikoon, P.; Heraud, P. Characterisation of chondrogenic differentiation of human mesenchymal stem cells using synchrotron FTIR microspectroscopy. Analyst 2011, 136, 2542–2551. [Google Scholar] [CrossRef]
- Lazarevic, J.J.; Kukolj, T.; Bugarski, D.; Lazarevic, N.; Bugarski, B.; Popovic, Z.V. Probing primary mesenchymal stem cells differentiation status by micro-Raman spectroscopy. In Spectrochim Acta A Mol Biomol Spectrosc; Elsevier B.V.: Amsterdam, The Netherlands, 2019; Volume 213, pp. 384–390. [Google Scholar]
- Pudlas, M.; Brauchle, E.; Klein, T.J.; Hutmacher, D.W.; Schenke-Layland, K. Non-invasive identification of proteoglycans and chondrocyte differentiation state by Raman microspectroscopy. J. Biophotonics 2013, 6, 205–211. [Google Scholar] [CrossRef]
- Bergholt, M.S.; St-Pierre, J.P.; Offeddu, G.S.; Parmar, P.A.; Albro, M.B.; Puetzer, J.L.; Oyen, M.L.; Stevens, M.M. Raman Spectroscopy Reveals New Insights into the Zonal Organization of Native and Tissue-Engineered Articular Cartilage. ACS Cent Sci. 2016, 2, 885–895. [Google Scholar] [CrossRef]
- Pijanka, J.K.; Kumar, D.; Dale, T.; Yousef, I.; Parkes, G.; Untereiner, V.; Yang, Y.; Dumas, P.; Collins, D.; Manfait, M.; et al. Vibrational spectroscopy differentiates between multipotent and pluripotent stem cells. Analyst 2010, 135, 3126–3132. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.W.; Lieu, D.K. Label-free biochemical characterization of stem cells using vibrational spectroscopy. J. Biophotonics 2009, 2, 656–668. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Friedrich, A.M.; Johnson, L.V.; Clegg, D.O. Memory in induced pluripotent stem cells: Reprogrammed human retinal-pigmented epithelial cells show tendency for spontaneous redifferentiation. Stem Cells 2010, 28, 1981–1991. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.; Doi, A.; Wen, B.; Ng, K.; Zhao, R.; Cahan, P.; Kim, J.; Aryee, M.J.; Ji, H.; Ehrlich, L.I.; et al. Epigenetic memory in induced pluripotent stem cells. Nature 2010, 467, 285–290. [Google Scholar] [CrossRef]
- Okano, H.; Nakamura, M.; Yoshida, K.; Okada, Y.; Tsuji, O.; Nori, S.; Ikeda, E.; Yamanaka, S.; Miura, K. Steps toward safe cell therapy using induced pluripotent stem cells. Circ. Res. 2013, 112, 523–533. [Google Scholar] [CrossRef]
- Vazquez-Zapien, G.J.; Mata-Miranda, M.M.; Sanchez-Monroy, V.; Delgado-Macuil, R.J.; Perez-Ishiwara, D.G.; Rojas-Lopez, M. FTIR Spectroscopic and Molecular Analysis during Differentiation of Pluripotent Stem Cells to Pancreatic Cells. Stem Cells Int. 2016, 2016, 6709714. [Google Scholar] [CrossRef]
- Mata-Miranda, M.M.; Vazquez-Zapien, G.J.; Rojas-Lopez, M.; Sanchez-Monroy, V.; Perez-Ishiwara, D.G.; Delgado-Macuil, R.J. Morphological, molecular and FTIR spectroscopic analysis during the differentiation of kidney cells from pluripotent stem cells. Biol. Res. 2017, 50, 14. [Google Scholar] [CrossRef]
- Chen, Y.J.; Cheng, Y.D.; Liu, H.Y.; Lin, P.Y.; Wang, C.S. Observation of biochemical imaging changes in human pancreatic cancer tissue using Fourier-transform infrared microspectroscopy. Chang. Gung Med. J. 2006, 29, 518–527. [Google Scholar]
- Hsu, C.C.; Xu, J.; Brinkhof, B.; Wang, H.; Cui, Z.; Huang, W.E.; Ye, H. A single-cell Raman-based platform to identify developmental stages of human pluripotent stem cell-derived neurons. Proc. Natl. Acad. Sci. USA 2020, 117, 18412–18423. [Google Scholar] [CrossRef]
- Wu, H.H.; Ho, J.H.; Lee, O.K. Detection of hepatic maturation by Raman spectroscopy in mesenchymal stromal cells undergoing hepatic differentiation. Stem Cell Res. Ther. 2016, 7, 6. [Google Scholar] [CrossRef]
- Uzunbajakava, N.; Lenferink, A.; Kraan, Y.; Volokhina, E.; Vrensen, G.; Greve, J.; Otto, C. Nonresonant confocal Raman imaging of DNA and protein distribution in apoptotic cells. Biophys. J. 2003, 84, 3968–3981. [Google Scholar] [CrossRef]
- Ye, D.; Heraud, P.; Parnpai, R.; Li, T. Reversal of Experimental Liver Damage after Transplantation of Stem-Derived Cells Detected by FTIR Spectroscopy. Stem Cells Int. 2017, 2017, 4585169. [Google Scholar] [CrossRef] [PubMed]
- Chung, Y.G.; Tu, Q.; Cao, D.; Harada, S.; Eisen, H.J.; Chang, C. Raman spectroscopy detects cardiac allograft rejection with molecular specificity. Clin. Transl. Sci. 2009, 2, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Umar, S. Intestinal stem cells. Curr. Gastroenterol. Rep. 2010, 12, 340–348. [Google Scholar] [CrossRef]
- Walsh, M.J.; Fellous, T.G.; Hammiche, A.; Lin, W.R.; Fullwood, N.J.; Grude, O.; Bahrami, F.; Nicholson, J.M.; Cotte, M.; Susini, J.; et al. Fourier transform infrared microspectroscopy identifies symmetric PO(2)(-) modifications as a marker of the putative stem cell region of human intestinal crypts. Stem Cells 2008, 26, 108–118. [Google Scholar] [CrossRef]
- Bentley, A.J.; Nakamura, T.; Hammiche, A.; Pollock, H.M.; Martin, F.L.; Kinoshita, S.; Fullwood, N.J. Characterization of human corneal stem cells by synchrotron infrared micro-spectroscopy. Mol. Vis. 2007, 13, 237–242. [Google Scholar]
- Kelly, J.G.; Nakamura, T.; Kinoshita, S.; Fullwood, N.J.; Martin, F.L. Evidence for a stem-cell lineage in corneal squamous cell carcinoma using synchrotron-based Fourier-transform infrared microspectroscopy and multivariate analysis. Analyst 2010, 135, 3120–3125. [Google Scholar] [CrossRef]
- Bergholt, M.S.; Serio, A.; Albro, M.B. Raman Spectroscopy: Guiding Light for the Extracellular Matrix. Front. Bioeng. Biotechnol. 2019, 7, 303. [Google Scholar] [CrossRef]
- Draper, E.R.; Morris, M.D.; Camacho, N.P.; Matousek, P.; Towrie, M.; Parker, A.W.; Goodship, A.E. Novel assessment of bone using time-resolved transcutaneous Raman spectroscopy. J. Bone Miner. Res. 2005, 20, 1968–1972. [Google Scholar] [CrossRef]
- Mandair, G.S.; Morris, M.D. Contributions of Raman spectroscopy to the understanding of bone strength. Bonekey Rep. 2015, 4, 620. [Google Scholar] [CrossRef]
- Aydin, H.M.; Hu, B.; Suso, J.S.; El Haj, A.; Yang, Y. Study of tissue engineered bone nodules by Fourier transform infrared spectroscopy. Analyst 2011, 136, 775–780. [Google Scholar] [CrossRef] [PubMed]
- Eyre, D.R. The collagens of articular cartilage. Semin. Arthritis Rheum. 1991, 21, 2–11. [Google Scholar] [CrossRef]
- Maldonado, M.; Nam, J. The role of changes in extracellular matrix of cartilage in the presence of inflammation on the pathology of osteoarthritis. Biomed Res. Int. 2013, 2013, 284873. [Google Scholar] [CrossRef] [PubMed]
- Lim, N.S.; Hamed, Z.; Yeow, C.H.; Chan, C.; Huang, Z. Early detection of biomolecular changes in disrupted porcine cartilage using polarized Raman spectroscopy. J. Biomed. Opt. 2011, 16, 017003. [Google Scholar] [CrossRef] [PubMed]
- Unal, M.; Akkus, O.; Sun, J.; Cai, L.; Erol, U.L.; Sabri, L.; Neu, C.P. Raman spectroscopy-based water content is a negative predictor of articular human cartilage mechanical function. Osteoarthr. Cartil. 2019, 27, 304–313. [Google Scholar] [CrossRef]
- Domenici, F.; Capocefalo, A.; Brasili, F.; Bedini, A.; Giliberti, C.; Palomba, R.; Silvestri, I.; Scarpa, S.; Morrone, S.; Paradossi, G.; et al. Ultrasound delivery of Surface Enhanced InfraRed Absorption active gold-nanoprobes into fibroblast cells: A biological study via Synchrotron-based InfraRed microanalysis at single cell level. Sci. Rep. 2019, 9, 11845. [Google Scholar] [CrossRef]
- Bilezikian, J.P.; Raisz, L.G.; Martin, T.J. Principles of Bone Biology, 4th ed.; Academic Press: Cambridge, MA, USA, 2019. [Google Scholar]
- Bonnier, F.; Meade, A.D.; Merzha, S.; Knief, P.; Bhattacharya, K.; Lyng, F.M.; Byrne, H.J. Three dimensional collagen gels as a cell culture matrix for the study of live cells by Raman spectroscopy. Analyst 2010, 135, 1697–1703. [Google Scholar] [CrossRef]
- Guo, S.; Beleites, C.; Neugebauer, U.; Afseth, N.K.; Alsamad, F.; Anand, S.; Araujo-Andrade, C.; Aškrabić, S.; Avci, E.; Baia, M.; et al. Comparability of Raman spectroscopic configurations: A large scale cross-laboratory study. Anal. Chem. 2020, in press. [Google Scholar] [CrossRef]
- Palonpon, A.F.; Sodeoka, M.; Fujita, K. Molecular imaging of live cells by Raman microscopy. Curr. Opin. Chem. Biol. 2013, 17, 708–715. [Google Scholar] [CrossRef]
- Ghita, A.; Pascut, F.C.; Mather, M.; Sottile, V.; Notingher, I. Cytoplasmic RNA in undifferentiated neural stem cells: A potential label-free Raman spectral marker for assessing the undifferentiated status. Anal. Chem. 2012, 84, 3155–3162. [Google Scholar] [CrossRef]
- Pully, V.V.; Lenferink, A.; Otto, C. Hybrid Rayleigh, Raman and two-photon excited fluorescence spectral confocal microscopy of living cells. J. Raman Spectrosc. 2010, 41, 599–608. [Google Scholar] [CrossRef]
- Rangan, S.; Schulze, H.G.; Vardaki, M.Z.; Blades, M.W.; Piret, J.M.; Turner, R.F.B. Applications of Raman spectroscopy in the development of cell therapies: State of the art and future perspectives. Analyst 2020, 145, 2070–2105. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; McReynolds, N.; Campbell, E.C.; Mazilu, M.; Barbosa, J.; Dholakia, K.; Powis, S.J. The use of wavelength modulated Raman spectroscopy in label-free identification of T lymphocyte subsets, natural killer cells and dendritic cells. PLoS ONE 2015, 10, e0125158. [Google Scholar] [CrossRef] [PubMed]
- Schie, I.W.; Rüger, J.; Mondol, A.S.; Ramoji, A.; Neugebauer, U.; Krafft, C.; Popp, J. High-Throughput Screening Raman Spectroscopy Platform for Label-Free Cellomics. Anal. Chem. 2018, 90, 2023–2030. [Google Scholar] [CrossRef]
- Esmonde-White, K.A.; Cuellar, M.; Uerpmann, C.; Lenain, B.; Lewis, I.R. Raman spectroscopy as a process analytical technology for pharmaceutical manufacturing and bioprocessing. Anal. Bioanal. Chem. 2017, 409, 637–649. [Google Scholar] [CrossRef]
- Goodacre, R.; Baker, M.J.; Graham, D.; Schultz, Z.D.; Diem, M.; Marques, M.P.; Cinque, G.; Vernooij, R.; Sulé-Suso, J.; Byrne, H.J.; et al. Biofluids and other techniques: General discussion. Faraday Discuss. 2016, 187, 575–601. [Google Scholar] [CrossRef]
- Mitchell, A.L.; Gajjar, K.B.; Theophilou, G.; Martin, F.L.; Martin-Hirsch, P.L. Vibrational spectroscopy of biofluids for disease screening or diagnosis: Translation from the laboratory to a clinical setting. J. Biophotonics 2014, 7, 153–165. [Google Scholar] [CrossRef]
- Parachalil, D.R.; Commerford, D.; Bonnier, F.; Chourpa, I.; McIntyre, J.; Byrne, H.J. Raman spectroscopy as a potential tool for label free therapeutic drug monitoring in human serum: The case of busulfan and methotrexate. Analyst 2019, 144, 5207–5214. [Google Scholar] [CrossRef]
- Almond, L.M.; Hutchings, J.; Lloyd, G.; Barr, H.; Shepherd, N.; Day, J.; Stevens, O.; Sanders, S.; Wadley, M.; Stone, N.; et al. Endoscopic Raman spectroscopy enables objective diagnosis of dysplasia in Barrett’s esophagus. Gastrointest. Endosc. 2014, 79, 37–45. [Google Scholar] [CrossRef]
- Jermyn, M.; Desroches, J.; Aubertin, K.; St-Arnaud, K.; Madore, W.J.; De Montigny, E.; Guiot, M.C.; Trudel, D.; Wilson, B.C.; Petrecca, K.; et al. A review of Raman spectroscopy advances with an emphasis on clinical translation challenges in oncology. Phys. Med. Biol. 2016, 61, R370–R400. [Google Scholar] [CrossRef]
- Barroso, E.M.; Smits, R.W.; van Lanschot, C.G.; Caspers, P.J.; Ten Hove, I.; Mast, H.; Sewnaik, A.; Hardillo, J.A.; Meeuwis, C.A.; Verdijk, R.; et al. Water Concentration Analysis by Raman Spectroscopy to Determine the Location of the Tumor Border in Oral Cancer Surgery. Cancer Res. 2016, 76, 5945–5953. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Cuddihy, M.J.; Kotov, N.A. Three-dimensional cell culture matrices: State of the art. Tissue Eng. Part B Rev. 2008, 14, 61–86. [Google Scholar] [CrossRef] [PubMed]
- Duval, K.; Grover, H.; Han, L.H.; Mou, Y.; Pegoraro, A.F.; Fredberg, J.; Chen, Z. Modeling Physiological Events in 2D vs. 3D Cell Culture. Physiology 2017, 32, 266–277. [Google Scholar] [CrossRef] [PubMed]
- Gattazzo, F.; Urciuolo, A.; Bonaldo, P. Extracellular matrix: A dynamic microenvironment for stem cell niche. Biochim. Biophys. Acta 2014, 1840, 2506–2519. [Google Scholar] [CrossRef]
- Jeanes, A.I.; Maya-Mendoza, A.; Streuli, C.H. Cellular microenvironment influences the ability of mammary epithelia to undergo cell cycle. PLoS ONE 2011, 6, e18144. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, G.; Shen, C.; Uygun, K.; Yarmush, M.L.; Meng, Q. A novel 3D liver organoid system for elucidation of hepatic glucose metabolism. Biotechnol. Bioeng. 2012, 109, 595–604. [Google Scholar] [CrossRef]
- Nobile, S.; Nobile, L. Nanotechnology for biomedical applications: Recent advances in neurosciences and bone tissue engineering. Polym. Eng. Sci. 2017, 57, 644–650. [Google Scholar] [CrossRef]
- Dayem, A.A.; Choi, H.Y.; Yang, G.M.; Kim, K.; Saha, S.K.; Kim, J.H.; Cho, S.G. The potential of nanoparticles in stem cell differentiation and further therapeutic applications. Biotechnol. J. 2016, 11, 1550–1560. [Google Scholar] [CrossRef]
- Yi, C.; Liu, D.; Fong, C.C.; Zhang, J.; Yang, M. Gold nanoparticles promote osteogenic differentiation of mesenchymal stem cells through p38 MAPK pathway. ACS Nano 2010, 4, 6439–6448. [Google Scholar] [CrossRef]
- Baranes, K.; Shevach, M.; Shefi, O.; Dvir, T. Gold Nanoparticle-Decorated Scaffolds Promote Neuronal Differentiation and Maturation. Nano Lett. 2016, 16, 2916–2920. [Google Scholar] [CrossRef]
- Zhang, R.; Lee, P.; Lui, V.C.; Chen, Y.; Liu, X.; Lok, C.N.; To, M.; Yeung, K.W.; Wong, K.K. Silver nanoparticles promote osteogenesis of mesenchymal stem cells and improve bone fracture healing in osteogenesis mechanism mouse model. Nanomedicine 2015, 11, 1949–1959. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, J.M.; Sousa, R.A.; Kotobuki, N.; Tadokoro, M.; Hirose, M.; Mano, J.F.; Reis, R.L.; Ohgushi, H. The osteogenic differentiation of rat bone marrow stromal cells cultured with dexamethasone-loaded carboxymethylchitosan/poly(amidoamine) dendrimer nanoparticles. Biomaterials 2009, 30, 804–813. [Google Scholar] [CrossRef] [PubMed]


| A | Wavenumber | FTIR Peak Assignments | Association |
|---|---|---|---|
| (i) | 1036 | C-C skeletal stretching | Proteins |
| (ii) | 1072 | PO2 symmetric stretching | DNA/RNA |
| (iii) | 1152 | C-C and C-O stretching | Proteins |
| (iv) | 1220–1280 | PO2 asymmetric stretching Amide III | DNA/RNA |
| (v) | 1312 | CH2 stretching | Phospholipids |
| (vi) | 1400 | CH3 symmetric stretching | Proteins |
| (vii) | 1456 | CH3 asymmetric stretching | Proteins |
| (viii) | 1546 | Amide II | Proteins |
| (ix) | 1620–1700 | Amide I | Proteins |
| (x) | 1742 | Ester, C=O stretching | Lipids |
| (xi) | 2854 | CH2 symmetric stretching | Lipids |
| (xii) | 2926 | CH2 asymmetric stretching | Lipids |
| B | Wavenumber | Raman Peaks Assignment | Association |
| (i) | 785–788 | Stretching of DNA/RNA related | Nucleic Acid |
| (ii) | 1004 | Phenylalanine | Protein |
| (iii) | 1090 | Stretching of DNA related bonds Stretching of C-N backbone | Nucleic Acid Protein |
| (iv) | 1127 | Stretching of C-N backbone Stretching of C-C | Protein Lipid |
| (v) | 1262 | DNA/RNA breathing modes Amide III | Nucleic Acid Lipid |
| (vi) | 1319 | CH2, CH3 twisting DNA/RNA breathing modes CH deformation vibration | Lipid Nucleic Acid Protein |
| (vii) | 1341 | DNA/RNA breathing modes CH deformation vibration | Nucleic Acid Protein |
| (viii) | 1451 | CH2 deformation vibration | Protein/Lipid |
| (ix) | 1585 | DNA/RNA breathing modes | Nucleic Acid |
| (x) | 1619 | Tyrosine; tryptophan | Protein |
| (xi) | 1662 | DNA/RNA breathing modes Amide I Fatty acids | Nucleic Acid Protein Lipid |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ravera, F.; Efeoglu, E.; Byrne, H.J. Vibrational Spectroscopy for In Vitro Monitoring Stem Cell Differentiation. Molecules 2020, 25, 5554. https://doi.org/10.3390/molecules25235554
Ravera F, Efeoglu E, Byrne HJ. Vibrational Spectroscopy for In Vitro Monitoring Stem Cell Differentiation. Molecules. 2020; 25(23):5554. https://doi.org/10.3390/molecules25235554
Chicago/Turabian StyleRavera, Francesca, Esen Efeoglu, and Hugh J. Byrne. 2020. "Vibrational Spectroscopy for In Vitro Monitoring Stem Cell Differentiation" Molecules 25, no. 23: 5554. https://doi.org/10.3390/molecules25235554
APA StyleRavera, F., Efeoglu, E., & Byrne, H. J. (2020). Vibrational Spectroscopy for In Vitro Monitoring Stem Cell Differentiation. Molecules, 25(23), 5554. https://doi.org/10.3390/molecules25235554








