Critical Review of Existing MHC I Immunopeptidome Isolation Methods
Abstract
1. Introduction
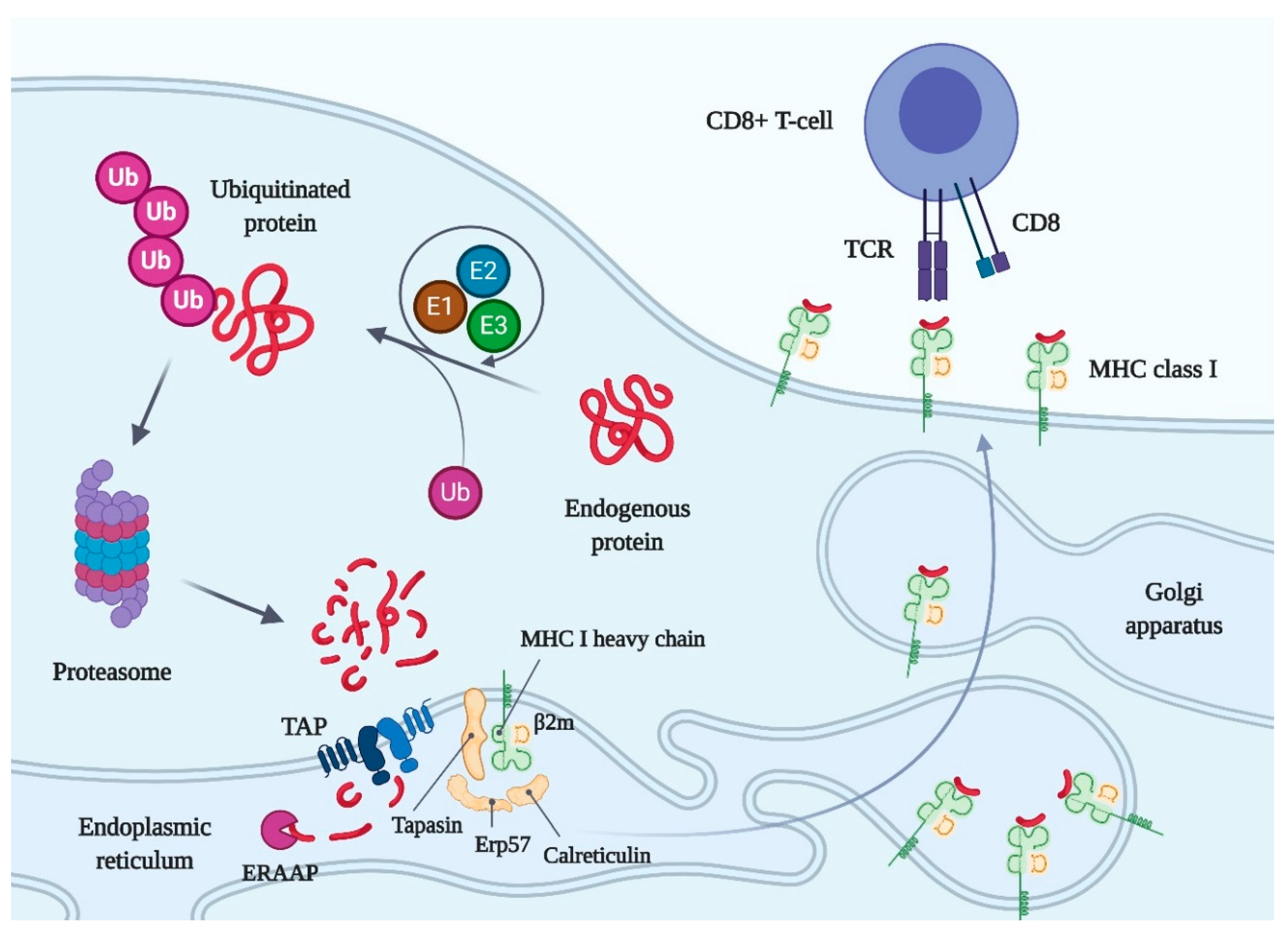
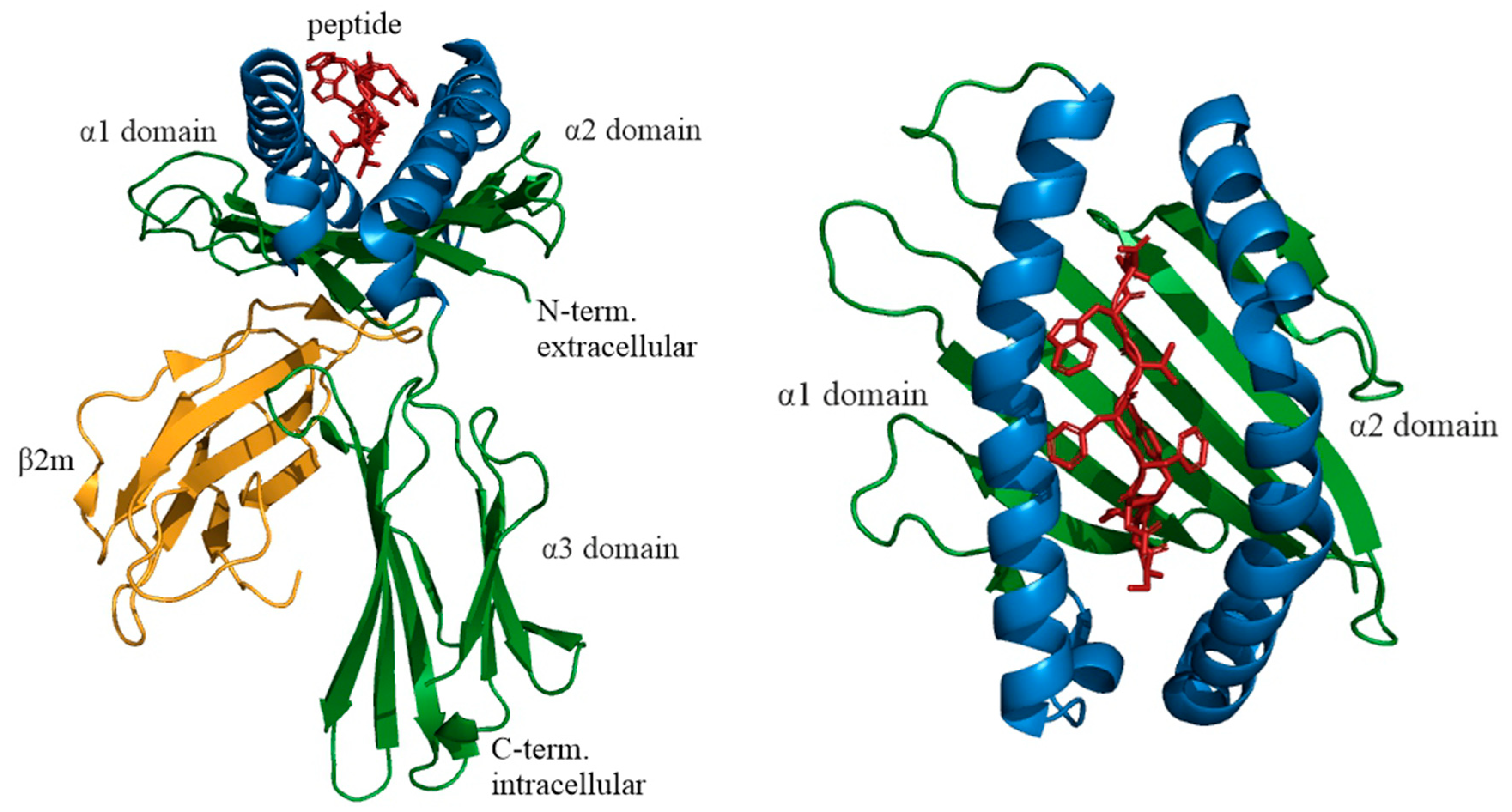
2. General Information on Immunopeptidome and HLA
3. Methods for Immunopeptidome Analysis
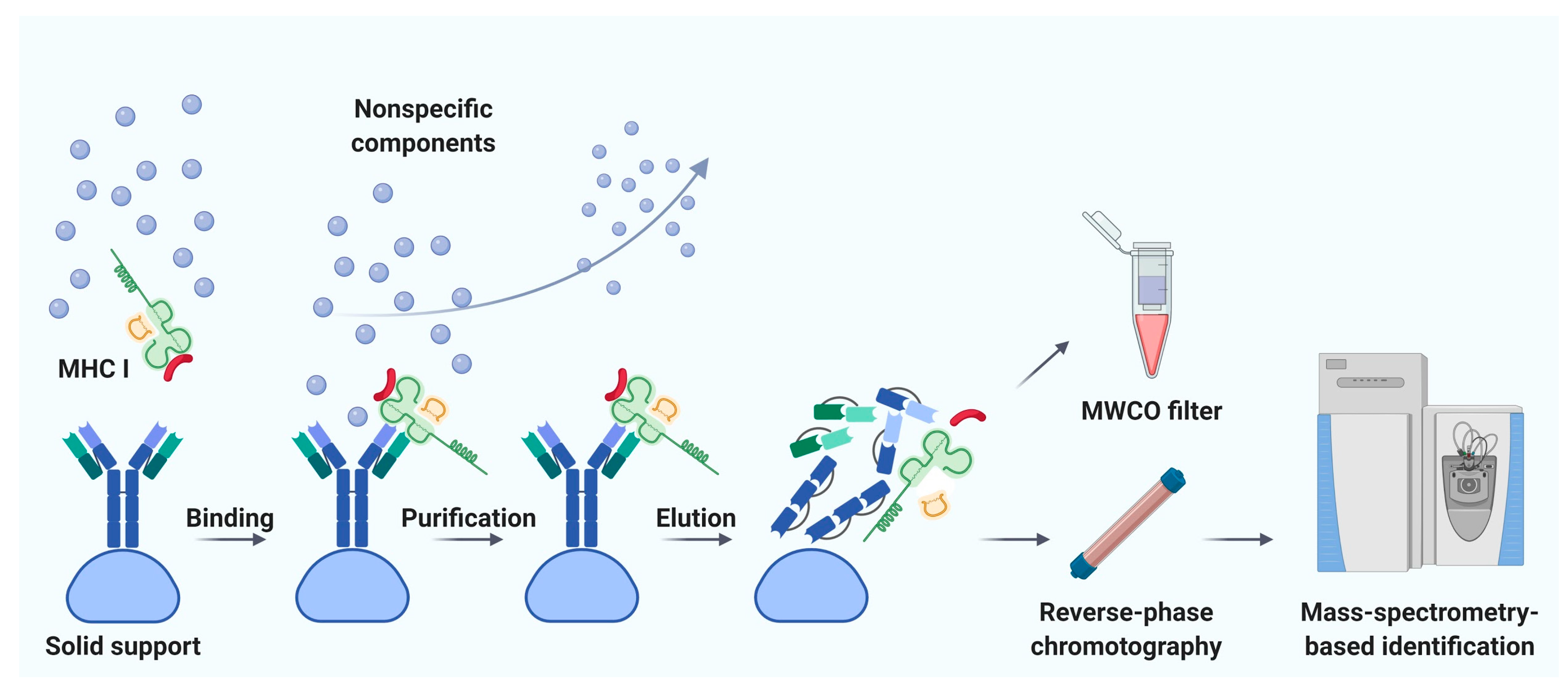
4. Current Protocols for Affinity Chromatography
4.1. The Choice of Biological Material
4.2. Various Detergents for the Isolation of Membrane-Bound MHC
4.3. Selection of Specific Antibodies
4.4. Solid Supports and Methods for Antibody Immobilization
4.5. Various Immunopeptidome Post-Fractionation and Purification Techniques
4.6. Approaches to LC-MS/MS Data Identification of Isolated MHC Ligands
4.7. Methods to Confirm Immunogenicity of the MHC I Ligands
5. Future Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gorer, P.A.; Lyman, S.; Snell, G.D.; Haldane, J.B.S. Studies on the genetic and antigenic basis of tumour transplantation Linkage between a histocompatibility gene and ’fused’ in mice. Proc. R. Soc. B Biol. Sci. 1948, 135, 499–505. [Google Scholar] [CrossRef]
- Van Rood, J.J.; Eernisse, J.G.; Van Leeuwen, A. Leucocyte Antibodies in Sera from Pregnant Women. Nature 1958, 181, 1735–1736. [Google Scholar] [CrossRef] [PubMed]
- Gruen, J.R.; Weissman, S.M. Human MHC class III and IV genes and disease associations. Front. Biosci. 2001, 6, 960–972. [Google Scholar] [CrossRef]
- Robinson, J.; Halliwell, J.A.; Hayhurst, J.D.; Flicek, P.; Parham, P.; Marsh, S.G.E. The IPD and IMGT/HLA database: Allele variant databases. Nucleic Acids Res. 2015, 43, D423–D431. [Google Scholar] [CrossRef]
- Mellins, E.D.; Stern, L.J. HLA-DM and HLA-DO, key regulators of MHC-II processing and presentation. Curr. Opin. Immunol. 2014, 26, 115–122. [Google Scholar] [CrossRef]
- Trowsdale, J.; Knight, J.C. Major Histocompatibility Complex Genomics and Human Disease. Annu. Rev. Genom. Hum. Genet. 2013, 14, 301–323. [Google Scholar] [CrossRef]
- Tian, C.; Hromatka, B.S.; Kiefer, A.K.; Eriksson, N.; Noble, S.M.; Tung, J.Y.; Hinds, D.A. Genome-wide association and HLA region fine-mapping studies identify susceptibility loci for multiple common infections. Nat. Commun. 2017, 8, 1–13. [Google Scholar] [CrossRef]
- Petersdorf, E.W.; O’Huigin, C. The MHC in the era of next-generation sequencing: Implications for bridging structure with function. Hum. Immunol. 2019, 80, 67–78. [Google Scholar] [CrossRef]
- Wissemann, W.T.; Hill-Burns, E.M.; Zabetian, C.P.; Factor, S.A.; Patsopoulos, N.; Hoglund, B.; Holcomb, C.; Donahue, R.J.; Thomson, G.; Erlich, H.; et al. Association of Parkinson Disease with Structural and Regulatory Variants in the HLA Region. Am. J. Hum. Genet. 2013, 93, 984–993. [Google Scholar] [CrossRef]
- Kuiper, J.J.; Van Setten, J.; Ripke, S.; Slot, R.V.; Mulder, F.; Missotten, T.; Baarsma, G.S.; Francioli, L.C.; Pulit, S.L.; De Kovel, C.G.; et al. A genome-wide association study identifies a functional ERAP2 haplotype associated with birdshot chorioretinopathy. Hum. Mol. Genet. 2014, 23, 6081–6087. [Google Scholar] [CrossRef]
- Sulzer, D.; Alcalay, R.N.; Garretti, F.; Cote, L.; Kanter, E.; Agin-Liebes, J.P.; Liong, C.; McMurtrey, C.; Hildebrand, W.H.; Mao, X.; et al. T cells from patients with Parkinson’s disease recognize α-synuclein peptides. Nature 2017, 546, 656–661. [Google Scholar] [CrossRef] [PubMed]
- He, D.; Wang, J.; Yi, L.; Guo, X.; Guo, S.; Guo, G.; Tu, W.; Wu, W.; Yang, L.; Xiao, R.; et al. Association of the HLA-DRB1 with Scleroderma in Chinese Population. PLoS ONE 2014, 9, e106939. [Google Scholar] [CrossRef] [PubMed]
- Wiśniewski, A.; Matusiak, Ł.; Szczerkowska-Dobosz, A.; Nowak, I.; Kuśnierczyk, P. HLA-C*06:02-independent, gender-related association of PSORS1C3 and PSORS1C1/CDSN single-nucleotide polymorphisms with risk and severity of psoriasis. Mol. Genet. Genom. 2018, 293, 957–966. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, A.; David, J.K.; Maden, S.K.; Wood, M.A.; Weeder, B.R.; Nellore, A.; Thompson, R.F. Human Leukocyte Antigen Susceptibility Map for Severe Acute Respiratory Syndrome Coronavirus 2. J. Virol. 2020, 94, 00510–00520. [Google Scholar] [CrossRef] [PubMed]
- Apps, R.; Qi, Y.; Carlson, J.M.; Chen, H.; Gao, X.; Thomas, R.; Yuki, Y.; Del Prete, G.Q.; Goulder, P.; Brumme, Z.L.; et al. Influence of HLA-C Expression Level on HIV Control. Science 2013, 340, 87–91. [Google Scholar] [CrossRef]
- Hopfensperger, K.; Richard, J.; Stürzel, C.M.; Bibollet-Ruche, F.; Apps, R.; Leoz, M.; Plantier, J.-C.; Hahn, B.H.; Finzi, A.; Kirchhoff, F.; et al. Convergent Evolution of HLA-C Downmodulation in HIV-1 and HIV-2. mBio 2020, 11, 11. [Google Scholar] [CrossRef]
- Jung, E.S.; Jang, H.W.; Chung, S.H.; Oh, H.-B.; Cheon, J.H.; Lee, J.H.; Park, S.J.; Park, M.H.; Kim, T.-G.; Yang, S.-K.; et al. HLA-C*01 is a Risk Factor for Crohn’s Disease. Inflamm. Bowel Dis. 2016, 22, 796–806. [Google Scholar] [CrossRef]
- Fitzgerald, O.; Haroon, M.; Giles, J.T.; Winchester, R. Concepts of pathogenesis in psoriatic arthritis: Genotype determines clinical phenotype. Arthritis Res. Ther. 2015, 17, 1–11. [Google Scholar] [CrossRef]
- Betz, R.C.; Petukhova, L.; Ripke, S.; Huang, H.; Menelaou, A.; Redler, S.; Becker, T.; Heilmann, S.; Yamany, T.; Duvic, M.; et al. Genome-wide meta-analysis in alopecia areata resolves HLA associations and reveals two new susceptibility loci. Nat. Commun. 2015, 6, 1–8. [Google Scholar] [CrossRef]
- Waage, J.; Standl, M.; Curtin, J.A.; Jessen, L.E.; Thorsen, J.; Tian, C.; Schoettler, N.; Flores, C.; Abdellaoui, A.; Ahluwalia, T.S.; et al. Genome-wide association and HLA fine-mapping studies identify risk loci and genetic pathways underlying allergic rhinitis. Nat. Genet. 2018, 50, 1072–1080. [Google Scholar] [CrossRef]
- Ferreiro-Iglesias, A.; Lesseur, C.; McKay, J.; Hung, R.J.; Han, Y.; Zong, X.; Christiani, D.; Johansson, M.; Xiao, X.; Li, Y.; et al. Fine mapping of MHC region in lung cancer highlights independent susceptibility loci by ethnicity. Nat. Commun. 2018, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Marsh, S.G.E.; Albert, E.D.; Bodmer, W.F.; Bontrop, R.E. Nomenclature for factors of the HLA system. Tissue 2010, 75, 291–455. [Google Scholar]
- Lin, M.; Tseng, H.-K.; Lin, R.-S.; Huang, C.-H.; Trejaut, J.A.; Lee, H.-L.; Loo, J.-H.; Chun-Hsiung, H.; Chen, P.-J.; Su, Y.-W.; et al. Association of HLA class I with severe acute respiratory syndrome coronavirus infection. BMC Med. Genet. 2003, 4, 9. [Google Scholar] [CrossRef] [PubMed]
- Brewerton, D.A.; Hart, F.D.; Nicholls, A.; Caffrey, M.; James, D.C.O.; Sturrock, R.D. ANKYLOSING SPONDYLITIS AND HL-A 27. Lancet 1973, 301, 904–907. [Google Scholar] [CrossRef]
- Cauli, A.; Dessole, G.; Fiorillo, M.T.; Vacca, A.; Mameli, A.; Bitti, P.; Passiu, G.; Sorrentino, R.; Mathieu, A. Increased level of HLA-B27 expression in ankylosing spondylitis patients compared with healthy HLA-B27-positive subjects: A possible further susceptibility factor for the development of disease. Rheumatology 2002, 41, 1375–1379. [Google Scholar] [CrossRef]
- Ohno, S.; Ohguchi, M.; Hirose, S.; Matsuda, H.; Wakisaka, A.; Aizawa, M. Close Association of HLA-Bw51 with Behcet’s Disease. Arch. Ophthalmol. 1982, 100, 1455–1458. [Google Scholar] [CrossRef]
- Wallace, G.R. HLA-B*51 the primary risk in Behcet disease. Proc. Natl. Acad. Sci. USA 2014, 111, 8706–8707. [Google Scholar] [CrossRef]
- Sveinbjornsson, G.; Gudbjartsson, G.S.D.F.; Halldorsson, B.V.; Kristinsson, K.G.; Gottfredsson, M.; Barrett, J.C.; Gudmundsson, L.J.; Blondal, K.B.T.; Gylfason, A.; Gudjonsson, S.A.; et al. HLA class II sequence variants influence tuberculosis risk in populations of European ancestry. Nat. Genet. 2016, 48, 318–322. [Google Scholar] [CrossRef]
- Howson, J.M.M.; Walker, N.M.; Clayton, D.; Todd, J.A. Type 1 Diabetes Genetics Consortium Confirmation of HLA class II independent type 1 diabetes associations in the major histocompatibility complex including HLA-B and HLA-A. Diabetes Obes. Metab. 2009, 11, 31–45. [Google Scholar] [CrossRef]
- Noble, J.A.; Valdes, A.M.; Varney, M.D.; Carlson, J.A.; Moonsamy, P.; Fear, A.L.; Lane, J.A.; Lavant, E.; Rappner, R.; Louey, A.; et al. HLA Class I and Genetic Susceptibility to Type 1 Diabetes: Results from the Type 1 Diabetes Genetics Consortium. Diabetes 2010, 59, 2972–2979. [Google Scholar] [CrossRef]
- Van Lummel, M.; Buis, D.T.P.; Ringeling, C.; De Ru, A.H.; Pool, J.; Papadopoulos, G.K.; Van Veelen, P.A.; Reijonen, H.; Drijfhout, J.W.; Roep, B.O. Epitope Stealing as a Mechanism of Dominant Protection by HLA-DQ6 in Type 1 Diabetes. Diabetes 2019, 68, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, R.C.; Pan-Hammarström, Q.; Graham, R.R.; Fontan, G.; Lee, A.T.; Ortmann, W.; Wang, N.; Urcelay, E.; Fernández-Arquero, M.; Núñez, C.; et al. High-Density SNP Mapping of the HLA Region Identifies Multiple Independent Susceptibility Loci Associated with Selective IgA Deficiency. PLoS Genet. 2012, 8, e1002476. [Google Scholar] [CrossRef] [PubMed]
- Weinstock, C.; Matheis, N.; Barkia, S.; Haager, M.-C.; Janson, A.; Marković, A.; Bux, J.; Kahaly, G.J. Autoimmune polyglandular syndrome type 2 shows the same HLA class II pattern as type 1 diabetes. Tissue Antigens 2011, 77, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Bouchie, A.; DeFrancesco, L. Erratum: Nature Biotechnology’s academic spinouts of 2015. Nat. Biotechnol. 2016, 34, 888. [Google Scholar] [CrossRef]
- Ribas, A.; Wolchok, J.D. Cancer immunotherapy using checkpoint blockade. Science 2018, 359, 1350–1355. [Google Scholar] [CrossRef]
- Miliotou, A.N.; Papadopoulou, L.C. CAR T-cell Therapy: A New Era in Cancer Immunotherapy. Curr. Pharm. Biotechnol. 2018, 19, 5–18. [Google Scholar] [CrossRef]
- Zhao, L.; Cao, Y.J. Engineered T Cell Therapy for Cancer in the Clinic. Front. Immunol. 2019, 10, 2250. [Google Scholar] [CrossRef]
- Rosenberg, S.A.; Restifo, N.P. Adoptive cell transfer as personalized immunotherapy for human cancer. Science 2015, 348, 62–68. [Google Scholar] [CrossRef]
- Murphy, J.P.; Kim, Y.; Clements, D.R.; Konda, P.; Schuster, H.; Kowalewski, D.J.; Paulo, J.A.; Cohen, A.M.; Stevanovic, S.; Gygi, S.P.; et al. Therapy-Induced MHC I Ligands Shape Neo-Antitumor CD8 T Cell Responses during Oncolytic Virus-Based Cancer Immunotherapy. J. Proteome Res. 2019, 18, 2666–2675. [Google Scholar] [CrossRef]
- Li, Q.; Ding, Z.-Y. The Ways of Isolating Neoantigen-Specific T Cells. Front. Oncol. 2020, 10, 1347. [Google Scholar] [CrossRef]
- Roerden, M.; Nelde, A.; Walz, J.S. Neoantigens in Hematological Malignancies-Ultimate Targets for Immunotherapy? Front. Immunol. 2019, 10, 3004. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Ott, P.A.; Wu, C.J. Towards personalized, tumour-specific, therapeutic vaccines for cancer. Nat. Rev. Immunol. 2018, 18, 168–182. [Google Scholar] [CrossRef] [PubMed]
- Hilf, N.; Kuttruff-Coqui, S.; Frenzel, K.; Bukur, V.; Stevanović, S.; Gouttefangeas, C.; Platten, M.; Tabatabai, G.; Dutoit, V.; Van Der Burg, S.H.; et al. Actively personalized vaccination trial for newly diagnosed glioblastoma. Nature 2018, 565, 240–245. [Google Scholar] [CrossRef]
- Weinschenk, T.; Gouttefangeas, C.; Schirle, M.; Obermayr, F.; Walter, S.; Schoor, O.; Kurek, R.; Loeser, W.; Bichler, K.-H.; Wernet, D.; et al. Integrated functional genomics approach for the design of patient-individual antitumor vaccines. Cancer Res. 2002, 62, 5818–5827. [Google Scholar]
- Schuller, A.P.; Green, R. Roadblocks and resolutions in eukaryotic translation. Nat. Rev. Mol. Cell Biol. 2018, 19, 526–541. [Google Scholar] [CrossRef] [PubMed]
- Amm, I.; Sommer, T.; Wolf, D.H. Protein quality control and elimination of protein waste: The role of the ubiquitin–proteasome system. Biochim. Biophys. Acta 2014, 1843, 182–196. [Google Scholar] [CrossRef]
- Collins, G.A.; Goldberg, A.L. The Logic of the 26S Proteasome. Cell 2017, 169, 792–806. [Google Scholar] [CrossRef]
- Kudriaeva, A.A.; Belogurov, A.A. Proteasome: A Nanomachinery of Creative Destruction. Biochemistry 2019, 84, 159–192. [Google Scholar] [CrossRef]
- Dahlmann, B. Mammalian proteasome subtypes: Their diversity in structure and function. Arch. Biochem. Biophys. 2016, 591, 132–140. [Google Scholar] [CrossRef]
- Vigneron, N.; Eynde, B.J.V.D. Proteasome subtypes and the processing of tumor antigens: Increasing antigenic diversity. Curr. Opin. Immunol. 2012, 24, 84–91. [Google Scholar] [CrossRef]
- Takahama, Y.; Ohigashi, I.; Murata, S.; Tanaka, K. Thymoproteasome and peptidic self. Immunogenetics 2018, 71, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Chang, S.-C.; Momburg, F.; Bhutani, N.; Goldberg, A.L. The ER aminopeptidase, ERAP1, trims precursors to lengths of MHC class I peptides by a “molecular ruler” mechanism. Proc. Natl. Acad. Sci. USA 2005, 102, 17107–17112. [Google Scholar] [CrossRef] [PubMed]
- Blees, A.; Januliene, D.; Hofmann, T.; Koller, N.; Schmidt, C.; Trowitzsch, S.; Moeller, A.; Tampé, R. Structure of the human MHC-I peptide-loading complex. Nature 2017, 551, 525–528. [Google Scholar] [CrossRef] [PubMed]
- Hulpke, S.; Tampé, R. The MHC I loading complex: A multitasking machinery in adaptive immunity. Trends Biochem. Sci. 2013, 38, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Powis, S.J.; Antoniou, A.N. Measuring Synthesis and Degradation of MHC Class I Molecules. Methods Mol. Biol. 2019, 1988, 83–100. [Google Scholar] [PubMed]
- Antoniou, A.N.; Powis, S.J.; Elliott, T. Assembly and export of MHC class I peptide ligands. Curr. Opin. Immunol. 2003, 15, 75–81. [Google Scholar] [CrossRef]
- Viotti, C. ER to Golgi-Dependent Protein Secretion: The Conventional Pathway. Methods Mol. Biol. 2016, 1459, 3–29. [Google Scholar]
- Young, J.D.; Liu, C.C.; Leong, L.G.; Cohn, Z.A. The pore-forming protein (perforin) of cytolytic T lymphocytes is immunologically related to the components of membrane attack complex of complement through cysteine-rich domains. J. Exp. Med. 1986, 164, 2077–2082. [Google Scholar] [CrossRef]
- Prager, I.; Watzl, C. Mechanisms of natural killer cell-mediated cellular cytotoxicity. J. Leukoc. Biol. 2019, 105, 1319–1329. [Google Scholar] [CrossRef]
- Qi, Q.; Liu, Y.; Cheng, Y.; Glanville, J.; Zhang, D.; Lee, J.-Y.; Olshen, R.A.; Weyand, C.M.; Boyd, S.D.; Goronzy, J.J. Diversity and clonal selection in the human T-cell repertoire. Proc. Natl. Acad. Sci. USA 2014, 111, 13139–13144. [Google Scholar] [CrossRef]
- Messaoudi, I.; Patiño, J.A.G.; Dyall, R.; LeMaoult, J.; Nikolich-Žugich, J. Direct Link Between mhc Polymorphism, T Cell Avidity, and Diversity in Immune Defense. Science 2002, 298, 1797–1800. [Google Scholar] [CrossRef] [PubMed]
- Gascoigne, N.R.J.; Rybakin, V.; Acuto, O.; Brzostek, J. TCR Signal Strength and T Cell Development. Annu. Rev. Cell Dev. Biol. 2016, 32, 327–348. [Google Scholar] [CrossRef] [PubMed]
- Mann, D.L.; Rogentine, G.N.; Fahey, J.L.; Nathenson, S.G. Molecular Heterogeneity of Human Lymphoid (HL-A) Alloantigens. Science 1969, 163, 1460–1462. [Google Scholar] [CrossRef] [PubMed]
- Bjorkman, P.J.; Saper, M.A.; Samraoui, B.; Bennett, W.S.; Strominger, J.L.; Wiley, D.C. Structure of the human class I histocompatibility antigen, HLA-A2. Nature 1987, 329, 506–512. [Google Scholar] [CrossRef]
- Falk, K.; Rötzschke, O.; Stevanovié, S.; Jung, G.; Rammensee, H.-G. Allele-specific motifs revealed by sequencing of self-peptides eluted from MHC molecules. Nature 1991, 351, 290–296. [Google Scholar] [CrossRef]
- Engelhard, V.H. Structure of peptides associated with MHC class I molecules. Curr. Opin. Immunol. 1994, 6, 13–23. [Google Scholar] [CrossRef]
- Bell, M.J.; Burrows, J.M.; Brennan, R.; Miles, J.J.; Tellam, J.; McCluskey, J.; Rossjohn, J.; Khanna, R.; Burrows, S.R. The peptide length specificity of some HLA class I alleles is very broad and includes peptides of up to 25 amino acids in length. Mol. Immunol. 2009, 46, 1911–1917. [Google Scholar] [CrossRef]
- Sugawara, S.; Abo, T.; Kumagai, K. A simple method to eliminate the antigenicity of surface class I MHC molecules from the membrane of viable cells by acid treatment at pH 3. J. Immunol. Methods 1987, 100, 83–90. [Google Scholar] [CrossRef]
- Lee, J.M.; Watts, T.H. On the dissociation and reassociation of MHC class II-foreign peptide complexes. Evidence that brief transit through an acidic compartment is not sufficient for binding site regeneration. J. Immunol. 1990, 144, 1829–1834. [Google Scholar]
- Falk, K.; Rötzschke, O.; Rammensee, H.-G. Cellular peptide composition governed by major histocompatibility complex class I molecules. Nature 1990, 348, 248–251. [Google Scholar] [CrossRef]
- Gebreselassie, D.; Spiegel, H.; Vukmanovic, S. Sampling of Major Histocompatibility Complex Class I-Associated Peptidome Suggests Relatively Looser Global Association of HLA-B*5101 With Peptides. Hum. Immunol. 2006, 67, 894–906. [Google Scholar] [CrossRef] [PubMed]
- Fortier, M.-H.; Caron, E.; Hardy, M.-P.; Voisin, G.; Lemieux, S.; Perreault, C.; Thibault, P. The MHC class I peptide repertoire is molded by the transcriptome. J. Exp. Med. 2008, 205, 595–610. [Google Scholar] [CrossRef] [PubMed]
- Lanoix, J.; Durette, C.; Courcelles, M.; Cossette, É.; Comtois-Marotte, S.; Hardy, M.-P.; Côté, C.; Perreault, C.; Thibault, P. Comparison of the MHC I Immunopeptidome Repertoire of B-Cell Lymphoblasts Using Two Isolation Methods. Proteomics 2018, 18, e1700251. [Google Scholar] [CrossRef] [PubMed]
- Nelde, A.; Kowalewski, D.J.; Stevanović, S. Purification and Identification of Naturally Presented MHC Class I and II Ligands. Methods Mol. Biol. 2019, 1988, 123–136. [Google Scholar] [CrossRef]
- Kote, S.; Pirog, A.; Bedran, G.; Alfaro, J.; Dapic, I. Mass Spectrometry-Based Identification of MHC-Associated Peptides. Cancers 2020, 12, 535. [Google Scholar] [CrossRef]
- Storkus, W.J.; Zeh, H.J.; Salter, R.D.; Lotze, M.T. Identification of T-cell epitopes: Rapid isolation of class I-presented peptides from viable cells by mild acid elution. J. Immunother. Emphas. Tumor Immunol. 1993, 14, 94–103. [Google Scholar] [CrossRef]
- Frassanito, M.A.; Mayordomo, J.I.; DeLeo, R.M.; Storkus, W.J.; Lotze, M.T.; DeLeo, A.B. Identification of Meth A sarcoma-derived class I major histocompatibility complex-associated peptides recognized by a specific CD8+ cytotoxic T lymphocyte. Cancer Res. 1995, 55, 124–128. [Google Scholar]
- Subramanian, A. Immunoaffinity Chromatography. Mol. Biotechnol. 2002, 20, 41–48. [Google Scholar] [CrossRef]
- Fitzgerald, J.; Leonard, P.; Darcy, E.; Sharma, S.; O’Kennedy, R. Immunoaffinity Chromatography: Concepts and Applications. Methods Mol. Biol. 2017, 1485, 27–51. [Google Scholar] [CrossRef]
- Hunt, D.F.; Henderson, R.A.; Shabanowitz, J.; Sakaguchi, K.; Michel, H.; Sevilir, N.; Cox, A.L.; Appella, E.; Engelhard, V.H. Characterization of peptides bound to the class I MHC molecule HLA-A2.1 by mass spectrometry. Science 1992, 255, 1261–1263. [Google Scholar] [CrossRef]
- Engelhard, V.H.; Appella, E.; Shabanowitz, J.; Sevilir, N.; Slingluff, C.L.; Hunt, D.F.; Benjamin, D.C.; Bodnar, W.M.; Cox, A.L.; Chen, Y.; et al. Mass Spectrometric Analysis of Peptides Associated with the Human Class I MHC Molecules HLA-A2.1 and HLA-B7 and Identification of Structural Features that Determine Binding. Chem. Immunol. Allergy 1993, 57, 39–62. [Google Scholar] [CrossRef]
- Kowalewski, D.J.; Stevanović, S. Biochemical Large-Scale Identification of MHC Class I Ligands. Methods Mol. Biol. 2012, 960, 145–157. [Google Scholar] [CrossRef]
- Mommen, G.P.M.; Frese, C.K.; Meiring, H.D.; Brink, J.V.G.-V.D.; De Jong, A.P.J.M.; Van Els, C.A.C.M.; Heck, A.J.R. Expanding the detectable HLA peptide repertoire using electron-transfer/higher-energy collision dissociation (EThcD). Proc. Natl. Acad. Sci. USA 2014, 111, 4507–4512. [Google Scholar] [CrossRef]
- Bassani-Sternberg, M.; Pletscher-Frankild, S.; Jensen, L.J.; Mann, M. Mass Spectrometry of Human Leukocyte Antigen Class I Peptidomes Reveals Strong Effects of Protein Abundance and Turnover on Antigen Presentation. Mol. Cell. Proteom. 2015, 14, 658–673. [Google Scholar] [CrossRef]
- Ritz, D.; Gloger, A.; Weide, B.; Garbe, C.; Neri, D.; Fugmann, T. High-sensitivity HLA class I peptidome analysis enables a precise definition of peptide motifs and the identification of peptides from cell lines and patients’ sera. Proteomics 2016, 16, 1570–1580. [Google Scholar] [CrossRef]
- Caron, E.; Kowalewski, D.J.; Chiek Koh, C.; Sturm, T.; Schuster, H.; Aebersold, R. Analysis of Major Histocompatibility Complex (MHC) Immunopeptidomes Using Mass Spectrometry. Mol. Cell. Proteom. 2015, 14, 3105–3117. [Google Scholar] [CrossRef]
- Winzeler, A.; Wang, J.T. Culturing Hybridoma Cell Lines for Monoclonal Antibody Production. Cold Spring Harb. Protoc. 2013, 2013, 640–642. [Google Scholar] [CrossRef]
- Vizcaíno, J.A.; Kubiniok, P.; Kovalchik, K.A.; Ma, Q.; Duquette, J.D.; Mongrain, I.; Deutsch, E.W.; Peters, B.; Sette, A.; Sirois, I.; et al. The Human Immunopeptidome Project: A Roadmap to Predict and Treat Immune Diseases. Mol. Cell. Proteom. 2019, 19, 31–49. [Google Scholar] [CrossRef]
- Shraibman, B.; Barnea, E.; Kadosh, D.M.; Haimovich, Y.; Slobodin, G.; Rosner, I.; López-Larrea, C.; Hilf, N.; Kuttruff, S.; Song, C.; et al. Identification of Tumor Antigens Among the HLA Peptidomes of Glioblastoma Tumors and Plasma. Mol. Cell. Proteom. 2019, 18, 1255–1268. [Google Scholar] [CrossRef]
- Hassan, C.; Kester, M.G.D.; Van Veelen, P.A.; Oudgenoeg, G.; De Ru, A.H.; Janssen, G.M.C.; Drijfhout, J.W.; Spaapen, R.M.; Jiménez, C.R.; Heemskerk, M.H.M.; et al. Accurate quantitation of MHC-bound peptides by application of isotopically labeled peptide MHC complexes. J. Proteom. 2014, 109, 240–244. [Google Scholar] [CrossRef]
- Schumacher, F.-R.; Delamarre, L.; Jhunjhunwala, S.; Modrusan, Z.; Phung, Q.T.; Elias, J.E.; Lill, J.R. Building proteomic tool boxes to monitor MHC class I and class II peptides. Proteomics 2017, 17, 1600061. [Google Scholar] [CrossRef]
- Alpízar, A.; Marino, F.; Ramos-Fernández, A.; Lombardía, M.; Jeko, A.; Pazos, F.; Paradela, A.; Santiago, C.; Heck, A.J.R.; Marcilla, M. A Molecular Basis for the Presentation of Phosphorylated Peptides by HLA-B Antigens. Mol. Cell. Proteom. 2017, 16, 181–193. [Google Scholar] [CrossRef]
- Chong, C.; Marino, F.; Pak, H.; Racle, J.; Daniel, R.T.; Müller, M.; Gfeller, D.; Coukos, G.; Bassani-Sternberg, M. High-throughput and Sensitive Immunopeptidomics Platform Reveals Profound Interferonγ-Mediated Remodeling of the Human Leukocyte Antigen (HLA) Ligandome. Mol. Cell. Proteom. 2018, 17, 533–548. [Google Scholar] [CrossRef] [PubMed]
- Van Bleek, G.M.; Nathenson, S.G. Isolation of an endogenously processed immunodominant viral peptide from the class IH--2K b molecule. Nature 1990, 348, 213–216. [Google Scholar] [CrossRef] [PubMed]
- Schuster, H.; Peper, J.K.; Bösmüller, H.-C.; Röhle, K.; Backert, L.; Bilich, T.; Ney, B.; Löffler, M.W.; Kowalewski, D.J.; Trautwein, N.; et al. The immunopeptidomic landscape of ovarian carcinomas. Proc. Natl. Acad. Sci. USA 2017, 114, E9942–E9951. [Google Scholar] [CrossRef]
- Geiger, T.; Cox, J.; Mann, M. Proteomics on an Orbitrap Benchtop Mass Spectrometer Using All-ion Fragmentation. Mol. Cell. Proteom. 2010, 9, 2252–2261. [Google Scholar] [CrossRef]
- Johnson, D.R.; Pober, J.S. Tumor necrosis factor and immune interferon synergistically increase transcription of HLA class I heavy- and light-chain genes in vascular endothelium. Proc. Natl. Acad. Sci. USA 1990, 87, 5183–5187. [Google Scholar] [CrossRef]
- Keller, I.E.; Vosyka, O.; Takenaka, S.; Kloß, A.; Dahlmann, B.; Willems, L.I.; Verdoes, M.; Overkleeft, H.S.; Marcos, E.; Adnot, S.; et al. Regulation of Immunoproteasome Function in the Lung. Sci. Rep. 2015, 5, srep10230. [Google Scholar] [CrossRef]
- Javitt, A.; Barnea, E.; Kramer, M.P.; Wolf-Levy, H.; Levin, Y.; Admon, A.; Merbl, Y. Pro-inflammatory Cytokines Alter the Immunopeptidome Landscape by Modulation of HLA-B Expression. Front. Immunol. 2019, 10, 141. [Google Scholar] [CrossRef]
- Prilliman, K.; Lindsey, M.; Zuo, Y.; Jackson, K.W.; Zhang, Y.; Hildebrand, W. Large-scale production of class I bound peptides: Assigning a signature to HLA-B*1501. Immunogenetics 1997, 45, 379–385. [Google Scholar] [CrossRef]
- Hickman, H.D.; Luis, A.D.; Buchli, R.; Few, S.R.; Sathiamurthy, M.; VanGundy, R.S.; Giberson, C.F.; Hildebrand, W.H. Toward a definition of self: Proteomic evaluation of the class I peptide repertoire. J. Immunol. 2004, 172, 2944–2952. [Google Scholar] [CrossRef] [PubMed]
- Halevi, L.; Barnea, E.; Beer, I.; Mann, M.; Admon, A. The HLA-B2705 peptidome. Arthritis Rheum. 2010, 62, 420–429. [Google Scholar] [CrossRef] [PubMed]
- Lazarus, D.; Weinstein-Marom, H.; Fishman, S.; Yossef, R.; Zuri, D.; Barnea, E.; Admon, A.; Margalit, A.; Gross, G. Efficient peptide recovery from secreted recombinant MHC-I molecules expressed via mRNA transfection. Immunol. Lett. 2015, 165, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Petricoin, E.F.; Belluco, C.; Araujo, R.P.; Liotta, L.A. The blood peptidome: A higher dimension of information content for cancer biomarker discovery. Nat. Rev. Cancer 2006, 6, 961–967. [Google Scholar] [CrossRef] [PubMed]
- Puppo, F.; Scudeletti, M.; Indiveri, F.; Ferrone, S. Serum HLA class I antigens: Markers and modulators of an immune response? Immunol. Today 1995, 16, 124–127. [Google Scholar] [CrossRef]
- Bassani-Sternberg, M.; Barnea, E.; Beer, I.; Avivi, I.; Katz, T.; Admon, A. Soluble plasma HLA peptidome as a potential source for cancer biomarkers. Proc. Natl. Acad. Sci. USA 2010, 107, 18769–18776. [Google Scholar] [CrossRef] [PubMed]
- DeMaria, S.; Schwab, R.; Gottesman, S.R.; Bushkin, Y. Soluble beta 2-microglobulin-free class I heavy chains are released from the surface of activated and leukemia cells by a metalloprotease. J. Biol. Chem. 1994, 269, 6689–6694. [Google Scholar]
- Tabayoyong, W.B.; Zavazava, N. Soluble HLA revisited. Leuk. Res. 2007, 31, 121–125. [Google Scholar] [CrossRef]
- Galati, G.; Arcelloni, C.; Paroni, R.; Heltai, S.; Rovere, P.; Rugarli, C.; Manfredi, A.A. Quantitative cytometry of MHC class I digestion from living cells. Cytometry 1997, 27, 77–83. [Google Scholar] [CrossRef]
- Antwi, K.; Hanavan, P.D.; Myers, C.E.; Ruiz, Y.W.; Thompson, E.J.; Lake, D.F. Proteomic identification of an MHC-binding peptidome from pancreas and breast cancer cell lines. Mol. Immunol. 2009, 46, 2931–2937. [Google Scholar] [CrossRef]
- Bettencourt, P.; Müller, J.; Nicastri, A.; Cantillon, D.; Madhavan, M.; Charles, P.D.; Fotso, C.B.; Wittenberg, R.; Bull, N.; Pinpathomrat, N.; et al. Identification of antigens presented by MHC for vaccines against tuberculosis. NPJ Vaccines 2020, 5, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Berlin, C.; Kowalewski, D.J.; Stevanović, S.; Stickel, J.S.; Schuster, H.; Mirza, N.; Walz, S.; Handel, M.; Schmid-Horch, B.; Salih, H.R.; et al. Mapping the HLA ligandome landscape of acute myeloid leukemia: A targeted approach toward peptide-based immunotherapy. Leukemia 2014, 29, 647–659. [Google Scholar] [CrossRef] [PubMed]
- Linke, D. Detergents: An overview. Methods Enzymol. 2009, 463, 603–617. [Google Scholar] [PubMed]
- Bassani-Sternberg, M.; Bräunlein, E.; Klar, R.; Engleitner, T.; Sinitcyn, P.; Audehm, S.; Straub, M.; Weber, J.; Slotta-Huspenina, J.; Specht, K.; et al. Direct identification of clinically relevant neoepitopes presented on native human melanoma tissue by mass spectrometry. Nat. Commun. 2016, 7, 13404. [Google Scholar] [CrossRef]
- Abelin, J.G.; Keskin, D.B.; Clauser, K.R.; Hacohen, N.; Rooney, M.S.; Carr, S.A.; Wu, C.J.; Sarkizova, S.; Hartigan, C.R.; Zhang, W.; et al. Mass Spectrometry Profiling of HLA-Associated Peptidomes in Mono-allelic Cells Enables More Accurate Epitope Prediction. Immunity 2017, 46, 315–326. [Google Scholar] [CrossRef]
- Blatnik, R.; Mohan, N.; Becker, S.; Nadler, W.M.; Rucevic, M.; Larsen, M.R.; Salek, M.; Riemer, A.B.; Bonsack, M.; Falkenby, L.G.; et al. A Targeted LC-MS Strategy for Low-Abundant HLA Class-I-Presented Peptide Detection Identifies Novel Human Papillomavirus T-Cell Epitopes. Proteomics 2018, 18, 1700390. [Google Scholar] [CrossRef]
- Stopfer, L.E.; Mesfin, J.M.; Joughin, B.A.; Lauffenburger, D.A.; White, F.M. Multiplexed relative and absolute quantitative immunopeptidomics reveals MHC I repertoire alterations induced by CDK4/6 inhibition. Nat. Commun. 2020, 11, 2760. [Google Scholar] [CrossRef]
- Abelin, J.G.; Trantham, P.D.; Penny, S.A.; Patterson, A.M.; Ward, S.T.; Hildebrand, W.H.; Cobbold, M.; Bai, D.L.; Shabanowitz, J.; Hunt, D.F. Complementary IMAC enrichment methods for HLA-associated phosphopeptide identification by mass spectrometry. Nat. Protoc. 2015, 10, 1308–1318. [Google Scholar] [CrossRef]
- Nicastri, A.; Liao, H.; Muller, J.; Purcell, A.W.; Ternette, N. The Choice of HLA-Associated Peptide Enrichment and Purification Strategy Affects Peptide Yields and Creates a Bias in Detected Sequence Repertoire. Proteomics 2020, 20, e1900401. [Google Scholar] [CrossRef]
- Partridge, T.; Nicastri, A.; Kliszczak, A.E.; Yindom, L.-M.; Kessler, B.M.; Ternette, N.; Borrow, P. Discrimination Between Human Leukocyte Antigen Class I-Bound and Co-Purified HIV-Derived Peptides in Immunopeptidomics Workflows. Front. Immunol. 2018, 9, 912. [Google Scholar] [CrossRef]
- Sanz-Bravo, A.; Alvarez-Navarro, C.; Martín-Esteban, A.; Barnea, E.; Admon, A.; De Castro, J.A.L. Ranking the Contribution of Ankylosing Spondylitis-associated Endoplasmic Reticulum Aminopeptidase 1 (ERAP1) Polymorphisms to Shaping the HLA-B*27 Peptidome. Mol. Cell. Proteom. 2018, 17, 1308–1323. [Google Scholar] [CrossRef] [PubMed]
- Purcell, A.W.; Ramarathinam, S.H.; Ternette, N. Mass spectrometry–based identification of MHC-bound peptides for immunopeptidomics. Nat. Protoc. 2019, 14, 1687–1707. [Google Scholar] [CrossRef]
- Brodsky, F.M.; Parham, P. Monomorphic anti-HLA-A,B,C monoclonal antibodies detecting molecular subunits and combinatorial determinants. J. Immunol. 1982, 128, 129–135. [Google Scholar] [PubMed]
- Hilton, H.G.; Parham, P. Direct binding to antigen-coated beads refines the specificity and cross-reactivity of four monoclonal antibodies that recognize polymorphic epitopes of HLA class I molecules. Tissue Antigens 2013, 81, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Pandey, K.; Mifsud, N.A.; Sian, T.C.L.K.; Ayala, R.; Ternette, N.; Ramarathinam, S.H.; Purcell, A.W. In-depth mining of the immunopeptidome of an acute myeloid leukemia cell line using complementary ligand enrichment and data acquisition strategies. Mol. Immunol. 2020, 123, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Escobar, H.; Crockett, D.K.; Reyes-Vargas, E.; Baena, A.; Rockwood, A.L.; Jensen, P.E.; Delgado, J.C. Large Scale Mass Spectrometric Profiling of Peptides Eluted from HLA Molecules Reveals N-Terminal-Extended Peptide Motifs. J. Immunol. 2008, 181, 4874–4882. [Google Scholar] [CrossRef]
- Sarkizova, S.; Klaeger, S.; Le, P.M.; Li, L.W.; Oliveira, G.; Keshishian, H.; Hartigan, C.R.; Zhang, W.; Braun, D.A.; Ligon, K.L.; et al. A large peptidome dataset improves HLA class I epitope prediction across most of the human population. Nat. Biotechnol. 2019, 38, 199–209. [Google Scholar] [CrossRef]
- Hermanson, G.T. Bioconjugate Techniques; Elsevier BV: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Fishman, J.B.; Berg, E.A. Protein A and Protein G Purification of Antibodies. Cold Spring Harb. Protoc. 2019, 2019. [Google Scholar] [CrossRef]
- Demmers, L.C.; Heck, A.J.R.; Wu, W. Pre-fractionation Extends but also Creates a Bias in the Detectable HLA Class Ι Ligandome. J. Proteome Res. 2019, 18, 1634–1643. [Google Scholar] [CrossRef] [PubMed]
- Rappsilber, J.; Mann, M.; Ishihama, Y. Protocol for micro-purification, enrichment, pre-fractionation and storage of peptides for proteomics using StageTips. Nat. Protoc. 2007, 2, 1896–1906. [Google Scholar] [CrossRef]
- Kulak, N.A.; Pichler, G.; Paron, I.; Nagaraj, N.; Mann, M. Minimal, encapsulated proteomic-sample processing applied to copy-number estimation in eukaryotic cells. Nat. Methods 2014, 11, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Cobbold, M.; De La Peña, H.; Norris, A.; Polefrone, J.M.; Qian, J.; English, A.M.; Cummings, K.L.; Penny, S.; Turner, J.E.; Cottine, J.; et al. MHC Class I-Associated Phosphopeptides Are the Targets of Memory-like Immunity in Leukemia. Sci. Transl. Med. 2013, 5, 203ra125. [Google Scholar] [CrossRef] [PubMed]
- Block, H.; Maertens, B.; Spriestersbach, A.; Brinker, N.; Kubicek, J.; Fabis, R.; Labahn, J.; Schäfer, F. Immobilized-Metal Affinity Chromatography (IMAC). Methods Enzymol. 2009, 463, 439–473. [Google Scholar] [CrossRef] [PubMed]
- Kastrup, I.B.; Stevanovic, S.; Arsequell, G.; Valencia, G.; Zeuthen, J.; Rammensee, H.-G.; Elliott, T.; Haurum, J.S. Lectin purified human class I MHC-derived peptides: Evidence for presentation of glycopeptides in vivo. Tissue Antigens 2000, 56, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Malaker, S.A.; Penny, S.A.; Steadman, L.G.; Myers, P.T.; Loke, J.C.; Raghavan, M.; Bai, D.L.; Shabanowitz, J.; Hunt, D.F.; Cobbold, M. Identification of Glycopeptides as Posttranslationally Modified Neoantigens in Leukemia. Cancer Immunol. Res. 2017, 5, 376–384. [Google Scholar] [CrossRef]
- Zhang, X.; Qi, Y.; Zhang, Q.; Liu, W. Application of mass spectrometry-based MHC immunopeptidome profiling in neoantigen identification for tumor immunotherapy. Biomed. Pharmacother. 2019, 120, 109542. [Google Scholar] [CrossRef]
- Giansanti, P.; Tsiatsiani, L.; Low, T.Y.; Heck, A.J.R. Six alternative proteases for mass spectrometry–based proteomics beyond trypsin. Nat. Protoc. 2016, 11, 993–1006. [Google Scholar] [CrossRef]
- Müller, T.; Winter, D. Systematic Evaluation of Protein Reduction and Alkylation Reveals Massive Unspecific Side Effects by Iodine-containing Reagents. Mol. Cell. Proteom. 2017, 16, 1173–1187. [Google Scholar] [CrossRef]
- Kong, A.T.; LePrevost, F.V.; Avtonomov, D.M.; Mellacheruvu, D.; Nesvizhskii, A.I. MSFragger: Ultrafast and comprehensive peptide identification in mass spectrometry–based proteomics. Nat. Methods 2017, 14, 513–520. [Google Scholar] [CrossRef]
- Liepe, J.; Marino, F.; Sidney, J.; Jeko, A.; Bunting, D.E.; Sette, A.; Kloetzel, P.M.; Stumpf, M.P.H.; Heck, A.J.R.; Mishto, M. A large fraction of HLA class I ligands are proteasome-generated spliced peptides. Science 2016, 354, 354–358. [Google Scholar] [CrossRef]
- Faridi, P.; Li, C.; Ramarathinam, S.H.; Vivian, J.P.; Illing, P.T.; Mifsud, N.A.; Ayala, R.; Song, J.; Gearing, L.J.; Hertzog, P.J.; et al. A subset of HLA-I peptides are not genomically templated: Evidence for cis- and trans-spliced peptide ligands. Sci. Immunol. 2018, 3, eaar3947. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi-Nik, H.; Michaux, J.; Corwin, W.L.; Keller, G.L.J.; Shcheglova, T.; Pak, H.S.; Coukos, G.; Baker, B.M.; Mandoiu, I.; Bassani-Sternberg, M.; et al. Mass spectrometry driven exploration reveals nuances of neoepitope-driven tumor rejection. JCI Insight 2019, 5, 5. [Google Scholar] [CrossRef] [PubMed]
- Shao, W.; Pedrioli, P.G.A.; Wolski, W.; Scurtescu, C.; Schmid, E.; Vizcaíno, J.A.; Courcelles, M.; Schuster, H.; Kowalewski, D.; Marino, F.; et al. The SysteMHC Atlas project. Nucleic Acids Res. 2018, 46, D1237–D1247. [Google Scholar] [CrossRef] [PubMed]
- Murphy, J.P.; Konda, P.; Kowalewski, D.J.; Schuster, H.; Clements, D.; Kim, Y.; Cohen, A.M.; Sharif, T.; Nielsen, M.; Stevanović, S.; et al. MHC-I Ligand Discovery Using Targeted Database Searches of Mass Spectrometry Data: Implications for T-Cell Immunotherapies. J. Proteome Res. 2017, 16, 1806–1816. [Google Scholar] [CrossRef]
- Liepe, J.; Sidney, J.; Lorenz, F.K.M.; Sette, A.; Mishto, M. Mapping the MHC Class I–Spliced Immunopeptidome of Cancer Cells. Cancer Immunol. Res. 2018, 7, 62–76. [Google Scholar] [CrossRef]
- Vigneron, N.; Ferrari, V.; Stroobant, V.; Habib, J.A.; Eynde, B.J.V.D. Peptide splicing by the proteasome. J. Biol. Chem. 2017, 292, 21170–21179. [Google Scholar] [CrossRef]
- Zhang, J.; Xin, L.; Shan, B.; Chen, W.; Xie, M.; Yuen, D.; Zhang, W.; Zhang, Z.; Lajoie, G.A.; Ma, B. PEAKS DB:De NovoSequencing Assisted Database Search for Sensitive and Accurate Peptide Identification. Mol. Cell. Proteom. 2012, 11, M111.010587. [Google Scholar] [CrossRef]
- Li, S.; DeCourcy, A.; Tang, H. Constrained De Novo Sequencing of neo-Epitope Peptides Using Tandem Mass Spectrometry. Lect. Notes Comput. Sci. 2018, 10812, 138–153. [Google Scholar] [CrossRef]
- Freudenmann, L.K.; Marcu, A.; Stevanović, S. Mapping the tumour human leukocyte antigen (HLA) ligandome by mass spectrometry. Immunology 2018, 154, 331–345. [Google Scholar] [CrossRef]
- Mellacheruvu, D.; Wright, Z.; Couzens, A.L.; Lambert, J.-P.; St-Denis, N.A.; Li, T.; Miteva, Y.V.; Hauri, S.; Sardiu, M.E.; Low, T.Y.; et al. The CRAPome: A contaminant repository for affinity purification–mass spectrometry data. Nat. Methods 2013, 10, 730–736. [Google Scholar] [CrossRef]
- Khilko, S.N.; Corr, M.; Boyd, L.F.; Lees, A.; Inman, J.K.; Margulies, D.H. Direct detection of major histocompatibility complex class I binding to antigenic peptides using surface plasmon resonance. Peptide immobilization and characterization of binding specificity. J. Biol. Chem. 1993, 268, 15425–15434. [Google Scholar] [PubMed]
- Sidney, J.; Southwood, S.; Moore, C.; Oseroff, C.; Pinilla, C.; Grey, H.M.; Sette, A. Measurement of MHC/Peptide Interactions by Gel Filtration or Monoclonal Antibody Capture. Curr. Protoc. Immunol. 2013, 100, 18.3.1–18.3.36. [Google Scholar] [CrossRef] [PubMed]
- Kessler, J.H.; Benckhuijsen, W.E.; Mutis, T.; Melief, C.J.; Van Der Burg, S.H.; Drijfhout, J.W. Competition-Based Cellular Peptide Binding Assay for HLA Class I. Curr. Protoc. Immunol. 2004, 61, 18.12.1. [Google Scholar] [CrossRef]
- Marcilla, M.; Alvarez, I.; Ramos-Fernández, A.; Lombardía, M.; Paradela, A.; Albar, J.P. Comparative Analysis of the Endogenous Peptidomes Displayed by HLA-B*27 and Mamu-B*08: Two MHC Class I Alleles Associated with Elite Control of HIV/SIV Infection. J. Proteome Res. 2016, 15, 1059–1069. [Google Scholar] [CrossRef] [PubMed]
- Slota, M.; Lim, J.-B.; Dang, Y.; Disis, M.L. ELISpot for measuring human immune responses to vaccines. Expert Rev. Vaccines 2011, 10, 299–306. [Google Scholar] [CrossRef]
- Oh, C.Y.; Klatt, M.G.; Bourne, C.; Dao, T.; Dacek, M.M.; Brea, E.J.; Mun, S.S.; Chang, A.Y.; Korontsvit, T.; Scheinberg, D.A. ALK and RET Inhibitors Promote HLA Class I Antigen Presentation and Unmask New Antigens within the Tumor Immunopeptidome. Cancer Immunol. Res. 2019, 7, 1984–1997. [Google Scholar] [CrossRef]
- Brea, E.J.; Oh, C.Y.; Manchado, E.; Budhu, S.; Gejman, R.S.; Mo, G.; Mondello, P.; Han, J.E.; Jarvis, C.A.; Ulmert, D.; et al. Kinase Regulation of Human MHC Class I Molecule Expression on Cancer Cells. Cancer Immunol. Res. 2016, 4, 936–947. [Google Scholar] [CrossRef]
- Gupta, S.; Maecker, H. Intracellular Cytokine Staining (ICS) on Human Lymphocytes or Peripheral Blood Mononuclear Cells (PBMCs). Bio-Protoc. 2015, 5, 5. [Google Scholar] [CrossRef]
- Woods, K.; Faridi, P.; Ostrouska, S.; Deceneux, C. The diversity of the immunogenic components of the melanoma immunopeptidome. BioRxiv 2019. [Google Scholar] [CrossRef]
- Walter, S.; Herrgen, L.; Schoor, O.; Jung, G.; Wernet, D.; Bühring, H.-J.; Rammensee, H.-G.; Stevanović, S. Cutting Edge: Predetermined Avidity of Human CD8 T Cells Expanded on Calibrated MHC/Anti-CD28-Coated Microspheres. J. Immunol. 2003, 171, 4974–4978. [Google Scholar] [CrossRef]
- Ghosh, M.; Gauger, M.; Marcu, A.; Nelde, A.; Denk, M.; Schuster, H.; Rammensee, H.-G.; Stevanović, S. Guidance Document: Validation of a High-Performance Liquid Chromatography-Tandem Mass Spectrometry Immunopeptidomics Assay for the Identification of HLA Class I Ligands Suitable for Pharmaceutical Therapies. Mol. Cell. Proteom. 2020, 19, 432–443. [Google Scholar] [CrossRef] [PubMed]
- Admon, A.; Bassani-Sternberg, M. The Human Immunopeptidome Project, a Suggestion for yet another Postgenome Next Big Thing. Mol. Cell. Proteom. 2011, 10, O111.011833. [Google Scholar] [CrossRef] [PubMed]
- Vincent, B.; Buntzman, A.; Hopson, B.; McEwen, C.; Cowell, L.G.; Akoglu, A.; Zhang, H.H.; Frelinger, J.A. iWAS—A novel approach to analyzing Next Generation Sequence data for immunology. Cell. Immunol. 2015, 299, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Swaminathan, J.; Boulgakov, A.A.; Hernandez, E.T.; Bardo, A.M.; Bachman, J.L.; Marotta, J.; Johnson, A.M.; Anslyn, E.V.; Marcotte, E.M. Highly parallel single-molecule identification of proteins in zeptomole-scale mixtures. Nat. Biotechnol. 2018, 36, 1076–1082. [Google Scholar] [CrossRef] [PubMed]
| Disease | Associated Alleles of the HLA I and II Genes | Study |
|---|---|---|
| Parkinson’s disease (PD) | HLA-B*07:02, HLA-C*07:02, HLA-DRB5*01, HLA-DRB1*15:01, HLA-DQA1*01:02, HLA-DQB1*06:02 (positively associated with PD risk) HLA-C*03:04, HLA-DRB1*04:04, HLA-DQA1*03:01 (negatively associated with PD risk) | [9,11] |
| Birdshot chorioretinopathy | HLA-A*29:02 (>95% of cases carry the HLA-A*29 allele; odds ratio (OR) = 157.5, p-Value = 6.6E-74) | [10] |
| Systemic sclerosis (SSc) | HLA-DRB1*15∶02, HLA-DRB1*16∶02 (major SSc risk allele subtypes) HLA-DRB1*01:01, HLA-DRB1*04:06 (strong SSc-protective) | [12] |
| Psoriasis | HLA-B*08, HLA-C*06:02, HLA-B*27, HLA-B*38, HLA-B*39 (positively associated with Psoriasis risk) | [13,18] |
| SARS | HLA-B*46:01 (positively associated with SARS risk, p-Value = 0.0279) | [23] |
| Allergic rhinitis | HLA-B*27 (positively associated with disease risk) | [20] |
| Lung cancer | HLA-B*08:01, HLA-DQB1*06 (positively associated with lung cancer risk for Europeans) HLA-DQB1*0401, HLA-DRB1*0701 (positively associated with lung cancer risk for Asians) | [21] |
| Ankylosing spondylitis | HLA-B*27 (positively associated with disease risk) | [24,25] |
| Behçet’s disease | HLA-B*51 (positively associated with disease risk) | [26,27] |
| Tuberculosis | rs557011 and rs9271378 (located between HLA-DQA1 and HLA-DRB1) and a missense variant encoding p.Ala210Thr in HLA-DQA1 positively associated with tuberculosis risk | [28] |
| Crohn’s disease | HLA-C*01 (significant associations with Crohn’s Disease) | [17] |
| Type-1 diabetes (T1D) | HLA-B*39:06 (positively associated with T1D) HLA-B*38 (protective for T1D) Heterozygous HLA-DQ2/8 (DQA1*05:01-DQB1*02:01/DQA1*03:01-DQB1*03:02) has the highest risk in T1D. Heterozygous HLA-DQ6/8 (DQA1*02:01-DQB1*06:02/ DQA1*03:01-DQB1*03:02) is protective against T1D. | [29,30,31] |
| Immunoglobulin A deficiency (IgAD) | HLA-DQB1*02, HLA-DRB1*03 and HLA-DRB1*07 (strong IgAD risk factors) HLA-DRB1*15 (protection from IgAD) | [32] |
| Autoimmune polyglandular syndrome (APS) type 2 | HLA-DRB1*03, HLA-DRB1*04, HLA-DQA1*03, HLA-DQB1*02 (positively associated with APS type 2) | [33] |
Sample Availability: Samples of the compounds are not available from the authors. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuznetsov, A.; Voronina, A.; Govorun, V.; Arapidi, G. Critical Review of Existing MHC I Immunopeptidome Isolation Methods. Molecules 2020, 25, 5409. https://doi.org/10.3390/molecules25225409
Kuznetsov A, Voronina A, Govorun V, Arapidi G. Critical Review of Existing MHC I Immunopeptidome Isolation Methods. Molecules. 2020; 25(22):5409. https://doi.org/10.3390/molecules25225409
Chicago/Turabian StyleKuznetsov, Alexandr, Alice Voronina, Vadim Govorun, and Georgij Arapidi. 2020. "Critical Review of Existing MHC I Immunopeptidome Isolation Methods" Molecules 25, no. 22: 5409. https://doi.org/10.3390/molecules25225409
APA StyleKuznetsov, A., Voronina, A., Govorun, V., & Arapidi, G. (2020). Critical Review of Existing MHC I Immunopeptidome Isolation Methods. Molecules, 25(22), 5409. https://doi.org/10.3390/molecules25225409






