3D Propolis-Sodium Alginate Scaffolds: Influence on Structural Parameters, Release Mechanisms, Cell Cytotoxicity and Antibacterial Activity
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Preparation of Solutions
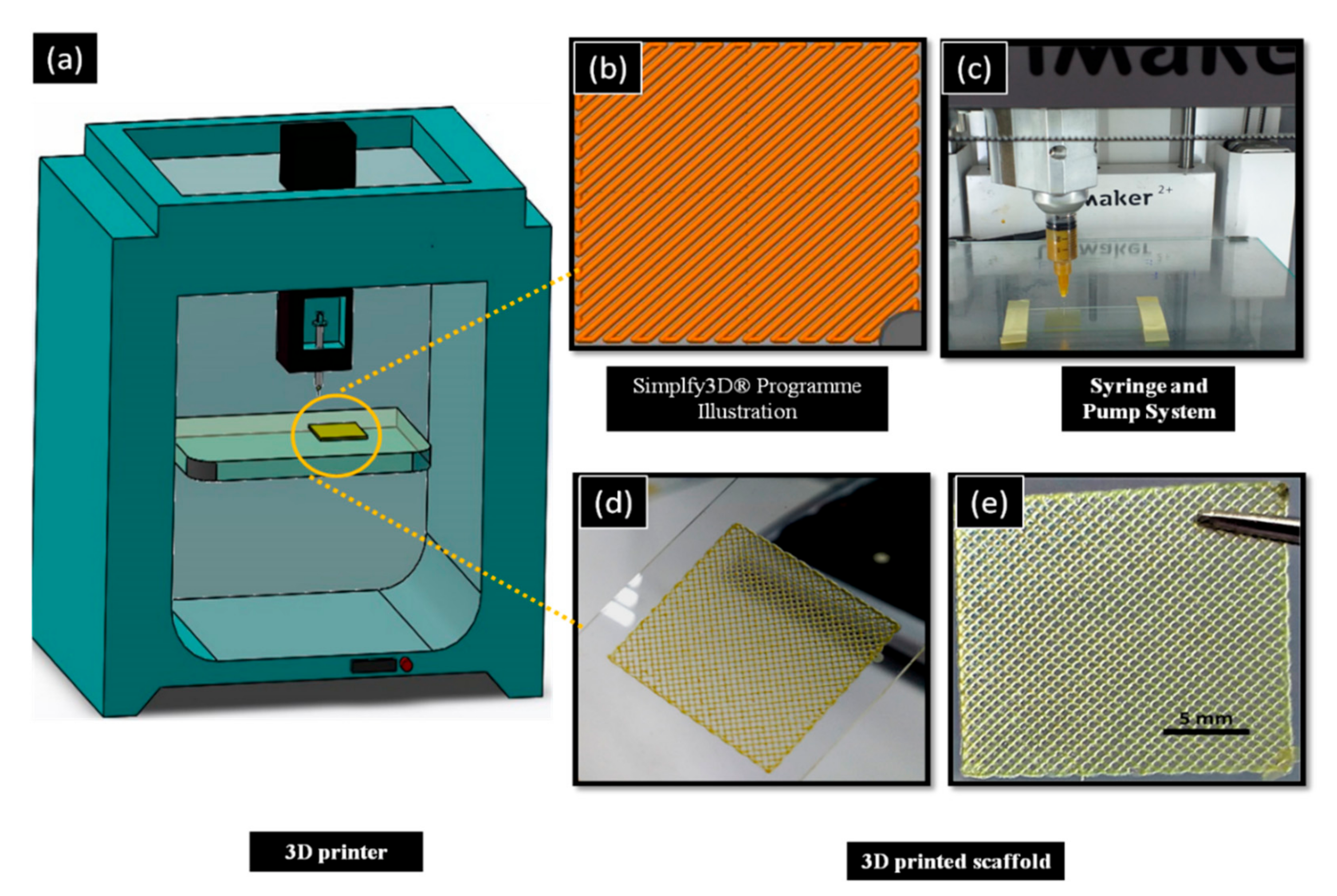
2.3. Design and 3D Printing of Scaffolds
2.4. Characterizations
2.4.1. Physical Properties Characterization
2.4.2. Fourier Transform-Infrared Spectroscopy (FT-IR)
2.4.3. Scanning Electron Microscopy (SEM)
2.4.4. Mechanical Properties of the Scaffolds
2.4.5. In Vitro Release Profile of Ps
2.4.6. Ps Release Kinetics
2.4.7. Swelling and Degradation Behaviors of Scaffolds
2.4.8. Antibacterial Assay
2.4.9. Cell Cytotoxicity
2.4.10. Statistical Analysis
3. Results and Discussion
3.1. Synthesis Results
3.2. Characterization Results
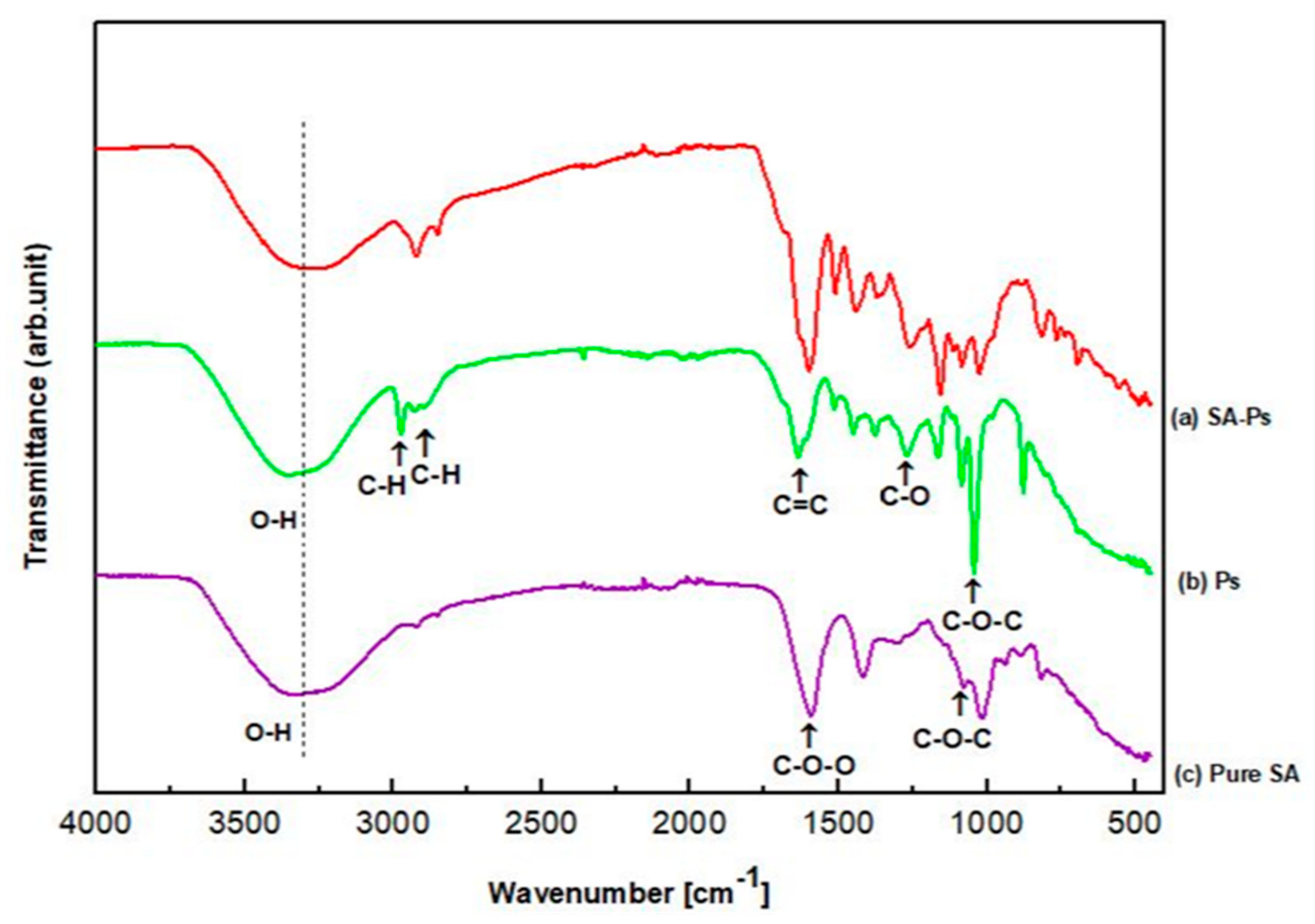
3.2.1. Fourier-Transform Infrared Spectroscopy (FT-IR)
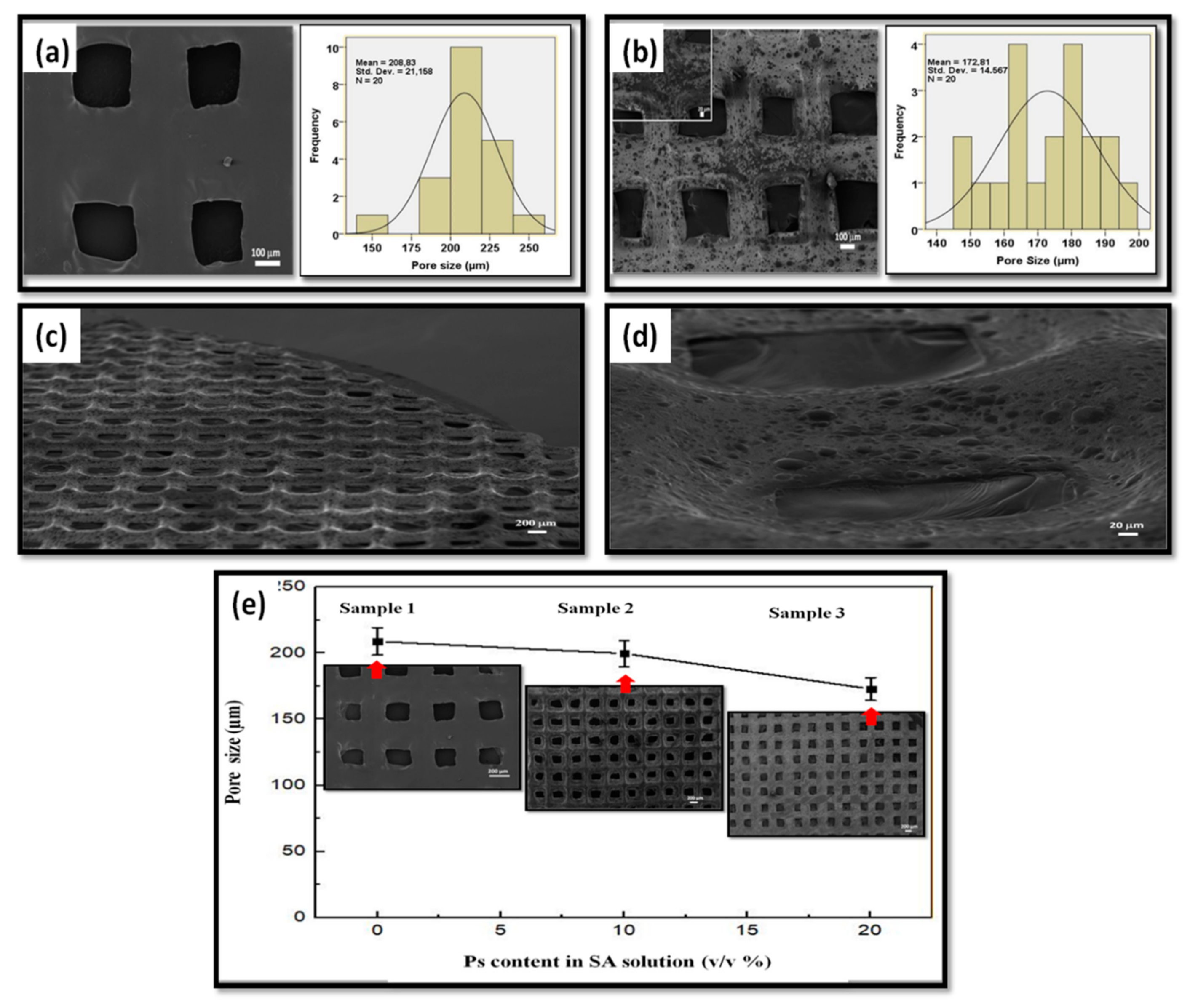
3.2.2. Scanning Electron Microscopy (SEM)
3.2.3. Tensile Test
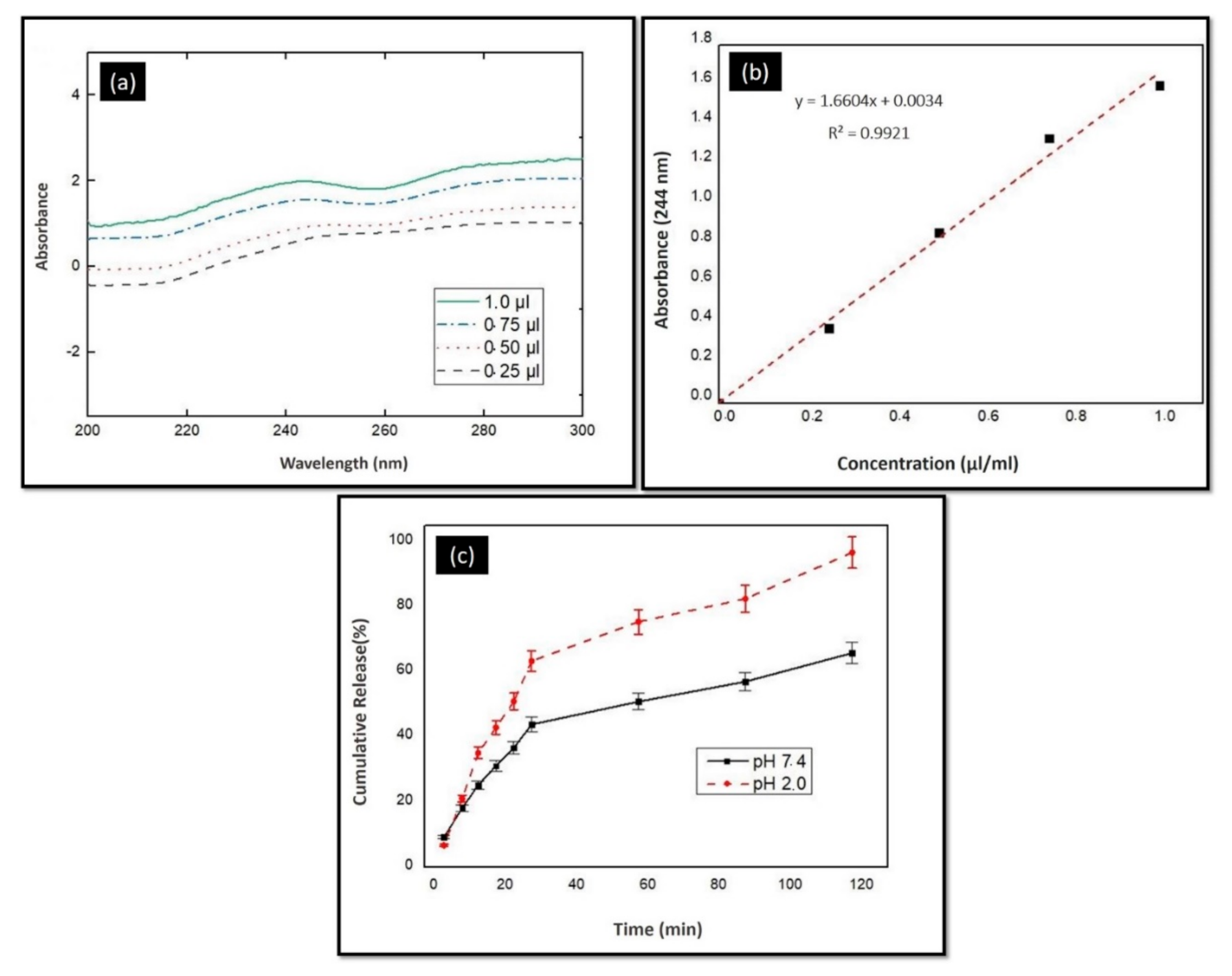
3.2.4. In Vitro Release Tests
3.2.5. Ps Release Kinetics
3.2.6. Swelling and Degradation Tests
3.2.7. Biological Assessments
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 3D | Three dimensional |
| AM | Additive Manufacturing |
| DMEM | Dulbecco’s Modified Eagle’s Medium |
| DMSO | Dimethyl sulfoxide |
| FBS | Fetal Bovine Serum |
| FT-IR | Fourier Transform-Infrared Spectroscopy |
| HFFF2 | Human Dermal Fibroblasts |
| PBS | Phosphate-Buffered Saline |
| Ps | Propolis |
| SA | Sodium Alginate |
| SBS | Scientific Bio Solutions |
| SD | Standard deviation |
| SEM | Scanning electron microscopy |
| STL | Stereolithography |
| TGF-β | Transforming Growth Factor-β |
References
- Ngo, T.D.; Kashani, A.; Imbalzano, G.; Nguyen, K.T.; Hui, D. Additive manufacturing (3D printing): A review of materials, methods, applications and challenges. Compos. Part B Eng. 2018, 143, 172–196. [Google Scholar] [CrossRef]
- Liaw, C.-Y.; Guvendiren, M. Current and emerging applications of 3D printing in medicine. Biofabrication 2017, 9, 024102. [Google Scholar] [CrossRef] [PubMed]
- Van Kogelenberg, S.; Yue, Z.; Dinoro, J.N.; Baker, C.S.; Wallace, G.G. Three-Dimensional Printing and Cell Therapy for Wound Repair. Adv. Wound Care 2018, 7, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Serra, T.; Mateos-Timoneda, M.A.; Planell, J.A.; Navarro, M. 3D printed PLA-based scaffolds: A versatile tool in regenerative medicine. Organogenesis 2013, 9, 239–244. [Google Scholar] [CrossRef]
- Subia, B.; Kundu, J.; Kundu, C.S. Biomaterial Scaffold Fabrication Techniques for Potential Tissue Engineering Applications. Tissue Eng. 2010, 141. [Google Scholar] [CrossRef]
- Eltom, A.; Zhong, G.; Muhammad, A. Scaffold Techniques and Designs in Tissue Engineering Functions and Purposes: A Review. Adv. Mater. Sci. Eng. 2019, 2019, 1–13. [Google Scholar] [CrossRef]
- An, J.; Teoh, J.E.M.; Suntornnond, R.; Chua, C.K. Design and 3D Printing of Scaffolds and Tissues. Engineerings 2015, 1, 261–268. [Google Scholar] [CrossRef]
- Konta, A.A.; García-Piña, M.; Serrano, D. Personalised 3D Printed Medicines: Which Techniques and Polymers Are More Successful? Bioengineering 2017, 4, 79. [Google Scholar] [CrossRef]
- Jayatissa, N.U.; Unagolla, J.; Bhaduri, S. 3D printed polymer scaffolds for bone tissue engineering. Ohio J. Sci. 2018, 118, A15–A16. [Google Scholar]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef]
- Aderibigbe, B.A.; Buyana, B. Alginate in Wound Dressings. Pharmaceutics 2018, 10, 42. [Google Scholar] [CrossRef] [PubMed]
- Straccia, M.C.; D’Ayala, G.G.; Romano, I.; Oliva, A.; Laurienzo, P. Alginate Hydrogels Coated with Chitosan for Wound Dressing. Mar. Drugs 2015, 13, 2890–2908. [Google Scholar] [CrossRef] [PubMed]
- Kamoun, E.A.; Kenawy, E.-R.S.; Tamer, T.M.; El-Meligy, M.A.; Eldin, M.S.M. Poly (vinyl alcohol)-alginate physically crosslinked hydrogel membranes for wound dressing applications: Characterization and bio-evaluation. Arab. J. Chem. 2015, 8, 38–47. [Google Scholar] [CrossRef]
- Luo, Y.; Luo, G.; Gelinsky, M.; Huang, P.; Ruan, C. 3D bioprinting scaffold using alginate/polyvinyl alcohol bioinks. Mater. Lett. 2017, 189, 295–298. [Google Scholar] [CrossRef]
- Liakos, I.; Rizzello, L.; Scurr, D.J.; Pompa, P.P.; Bayer, I.S.; Athanassiou, A. All-natural composite wound dressing films of essential oils encapsulated in sodium alginate with antimicrobial properties. Int. J. Pharm. 2014, 463, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Keskin, M.; Keskin, Ş.; Kolayli, S. Preparation of alcohol free propolis-alginate microcapsules, characterization and release property. LWT 2019, 108, 89–96. [Google Scholar] [CrossRef]
- Oryan, A.; Alemzadeh, E.; Moshiri, A. Potential role of propolis in wound healing: Biological properties and therapeutic activities. Biomed. Pharmacother. 2018, 98, 469–483. [Google Scholar] [CrossRef]
- Sharaf, S.; El-Naggar, M.E. Wound dressing properties of cationized cotton fabric treated with carrageenan/cyclodextrin hydrogel loaded with honey bee propolis extract. Int. J. Biol. Macromol. 2019, 133, 583–591. [Google Scholar] [CrossRef]
- Oliveira, R.N.; McGuinness, G.B.; Rouze, R.; Quilty, B.; Cahill, P.A.; Soares, G.D.A.; Thiré, R.M.D.S.M. PVA hydrogels loaded with a Brazilian propolis for burn wound healing applications. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Albayrak, S. Propolis: NaturaL Antimicrobial Matter. Ankara Üniversitesi Eczacılık Fakültesi Dergisi. 2008, 37, 201–215. [Google Scholar]
- Bueno-Silva, B.; Marsola, A.; Ikegaki, M.; Alencar, S.M.; Rosalen, P.L. The effect of seasons on Brazilian red propolis and its botanical source: Chemical composition and antibacterial activity. Nat. Prod. Res. 2017, 31, 1318–1324. [Google Scholar] [CrossRef]
- Nina, N.; Lima, B.; Feresin, G.E.; Gimenez, A.; Capusiri, E.S.; Schmeda-Hirschmann, G. Antibacterial and leishmanicidal activity of Bolivian propolis. Lett. Appl. Microbiol. 2016, 62, 290–296. [Google Scholar] [CrossRef]
- Oliveira, A.V.; Ferreira, A.L.; Nunes, S.; Dandlen, S.A.; Miguel, D.G.D.G.; Faleiro, M.L. Antibacterial activity of propolis extracts from the south of Portugal. Pak. J. Pharm. Sci. 2017, 30, 1–9. [Google Scholar]
- Yildirim, A.; Duran, G.G.; Duran, N.; Jenedi, K.; Bolgul, B.S.; Miraloglu, M.; Muz, M. Antiviral Activity of Hatay Propolis Against Replication of Herpes Simplex Virus Type 1 and Type 2. Med. Sci. Monit. 2016, 22, 422–430. [Google Scholar] [CrossRef]
- Kujumgiev, A.; Tsvetkova, I.; Serkedjieva, Y.; Bankova, V.; Christov, R.; Popov, S. Antibacterial, antifungal and antiviral activity of propolis of different geographic origin. J. Ethnopharmacol. 1999, 64, 235–240. [Google Scholar] [CrossRef]
- Mouhoubi-Tafinine, Z.; Ouchemoukh, S.; Tamendjari, A. Antioxydant activity of some algerian honey and propolis. Ind. Crop. Prod. 2016, 88, 85–90. [Google Scholar] [CrossRef]
- Valenzuela-Barra, G.; Castro, C.; Figueroa, C.; Barriga, A.; Silva, X.; Heras, B.D.L.; Hortelano, S.; Delporte, C. Anti-inflammatory activity and phenolic profile of propolis from two locations in Región Metropolitana de Santiago, Chile. J. Ethnopharmacol. 2015, 168, 37–44. [Google Scholar] [CrossRef]
- da Frozza, C.O.S.; Santos, D.A.; Rufatto, L.C.; Minetto, L.; Scariot, F.J.; Echeverrigaray, S.; Pich, C.T.; Moura, S.; Padilha, F.F.; Borsuk, S.; et al. Antitumor activity of Brazilian red propolis fractions against Hep-2 cancer cell line. Biomed. Pharmacother. 2017, 91, 951–963. [Google Scholar] [CrossRef] [PubMed]
- Martinotti, S.; Ranzato, E. Propolis: A new frontier for wound healing? Burn. Trauma 2015, 3, 9. [Google Scholar] [CrossRef]
- De Moura, S.A.L.; Negri, G.; Salatino, A.; Lima, L.D.D.C.; Dourado, L.P.A.; Mendes, J.B.; Andrade, S.P.; Ferreira, M.A.N.D.; Cara, D.C. Aqueous Extract of Brazilian Green Propolis: Primary Components, Evaluation of Inflammation and Wound Healing by Using Subcutaneous Implanted Sponges. Evidence-Based Complement. Altern. Med. 2011, 2011, 1–8. [Google Scholar] [CrossRef]
- Juliano, C.; Pala, C.; Cossu, M. Preparation and characterisation of polymeric films containing propolis. J. Drug Deliv. Sci. Technol. 2007, 17, 177–181. [Google Scholar] [CrossRef]
- Yang, F.; Zhang, M.; Bhandari, B.; Liu, Y. Investigation on lemon juice gel as food material for 3D printing and optimization of printing parameters. LWT 2018, 87, 67–76. [Google Scholar] [CrossRef]
- Xu, M.; Zhai, D.; Chang, J.; Wu, C. In vitro assessment of three-dimensionally plotted nagelschmidtite bioceramic scaffolds with varied macropore morphologies. Acta Biomater. 2014, 10, 463–476. [Google Scholar] [CrossRef] [PubMed]
- Gao, C.; Liu, M.; Chen, J.; Zhang, X. Preparation and controlled degradation of oxidized sodium alginate hydrogel. Polym. Degrad. Stab. 2009, 94, 1405–1410. [Google Scholar] [CrossRef]
- Vieira, T.; Silva, J.C.; Rego, A.M.B.D.; Borges, J.P.; Henriques, C. Electrospun biodegradable chitosan based-poly(urethane urea) scaffolds for soft tissue engineering. Mater. Sci. Eng. C 2019, 103, 109819. [Google Scholar] [CrossRef] [PubMed]
- You, F.; Wu, X.; Chen, X. 3D printing of porous alginate/gelatin hydrogel scaffolds and their mechanical property characterization. Int. J. Polym. Mater. 2016, 66, 299–306. [Google Scholar] [CrossRef]
- Chua, C.; Wong, C.; Yeong, W. Chapter Five-Material Characterization for Additive Manufacturing. In Standards, Quality Control, and Measurement Sciences in 3D Printing and Additive Manufacturing; London Academic Publishing: London, UK, 2017; pp. 95–137. [Google Scholar]
- Ergul, N.M.; Unal, S.; Kartal, I.; Kalkandelen, C.; Ekren, N.; Kilic, O.; Chi-Chang, L.; Gunduz, O. 3D printing of chitosan/poly(vinyl alcohol) hydrogel containing synthesized hydroxyapatite scaffolds for hard-tissue engineering. Polym. Test. 2019, 79, 106006. [Google Scholar] [CrossRef]
- Ouyang, L.; Yao, R.; Zhao, Y.; Sun, W. Effect of bioink properties on printability and cell viability for 3D bioplotting of embryonic stem cells. Biofabrication 2016, 8, 035020. [Google Scholar] [CrossRef]
- Daemi, H.; Barikani, M. Synthesis and characterization of calcium alginate nanoparticles, sodium homopolymannuronate salt and its calcium nanoparticles. Sci. Iran. 2012, 19, 2023–2028. [Google Scholar] [CrossRef]
- Pereira, R.; Tojeira, A.; Vaz, D.C.; Mendes, A.; Bártolo, P. Preparation and Characterization of Films Based on Alginate and Aloe Vera. Int. J. Polym. Anal. Charact. 2011, 16, 449–464. [Google Scholar] [CrossRef]
- Cesur, S.; Oktar, F.N.; Ekren, N.; Kilic, O.; Alkaya, D.B.; Seyhan, S.A.; Ege, Z.R.; Lin, C.-C.; Kuruca, S.E.; Erdemir, G.; et al. Preparation and characterization of electrospun polylactic acid/sodium alginate/orange oyster shell composite nanofiber for biomedical application. J. Aust. Ceram. Soc. 2019, 56, 533–543. [Google Scholar] [CrossRef]
- Zheng, J.; Yu, X.; Wang, C.; Cao, Z.; Yang, H.; Ma, D.; Xu, X. Facile synthesis of three-dimensional reinforced Sn@polyaniline/sodium alginate nanofiber hydrogel network for high performance lithium-ion battery. J. Mater. Sci. Mater. Electron. 2016, 27, 4457–4464. [Google Scholar] [CrossRef]
- Mimmo, T.; Marzadori, C.; Montecchio, D.; Gessa, C. Characterisation of Ca– and Al–pectate gels by thermal analysis and FT-IR spectroscopy. Carbohydr. Res. 2005, 340, 2510–2519. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.N.; Mancini, M.C.; De Oliveira, F.C.S.; Passos, T.M.; Quilty, B.; Thiré, R.M.D.S.M.; McGuinness, G.B. FTIR analysis and quantification of phenols and flavonoids of five commercially available plants extracts used in wound healing. Matéria (Rio de Janeiro) 2016, 21, 767–779. [Google Scholar] [CrossRef]
- Hussein, U.K.; Hassan, N.E.-H.Y.; Elhalwagy, M.E.; Zaki, A.R.; Abubakr, H.O.; Venkata, K.C.N.; Jang, K.Y.; Bishayee, A. Ginger and Propolis Exert Neuroprotective Effects against Monosodium Glutamate-Induced Neurotoxicity in Rats. Molecules 2017, 22, 1928. [Google Scholar] [CrossRef]
- Martín-Ramos, P.; Fernández-Coppel, I.A.; Ruíz-Potosme, N.M.; Martín-Gil, J. Potential of ATR-FTIR Spectroscopy for the Classification of Natural Resins. Biol. Eng. Med. Sci. Rep. 2018, 4, 03–06. [Google Scholar] [CrossRef]
- Lu, H.; Oh, H.H.; Kawazoe, N.; Yamagishi, K.; Chen, G. PLLA–collagen and PLLA–gelatin hybrid scaffolds with funnel-like porous structure for skin tissue engineering. Sci. Technol. Adv. Mater. 2012, 13, 064210. [Google Scholar] [CrossRef]
- Egan, P.F. Integrated Design Approaches for 3D Printed Tissue Scaffolds: Review and Outlook. Materials 2019, 12, 2355. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.H.; Park, I.K.; Kim, J.M.; Lee, J.H. In vitro and in vivo characteristics of PCL scaffolds with pore size gradient fabricated by a centrifugation method. Biomaterials 2007, 28, 1664–1671. [Google Scholar] [CrossRef]
- Yannas, I.V.; Lee, E.; Orgill, D.P.; Skrabut, E.M.; Murphy, G.F. Synthesis and characterization of a model extracellular matrix that induces partial regeneration of adult mammalian skin. Proc. Natl. Acad. Sci. USA 1989, 86, 933–937. [Google Scholar] [CrossRef]
- Bobyn, J.D.; Stackpool, G.J.; Hacking, S.; Tanzer, M.; Krygier, J.J. Characteristics of bone ingrowth and interface mechanics of a new porous tantalum biomaterial. J. Bone Jt. Surg. Br. 1999, 81, 907–914. [Google Scholar] [CrossRef]
- Zardiackas, L.D.; Parsell, D.E.; Dillon, L.D.; Mitchell, D.W.; Nunnery, L.A.; Poggie, R. Structure, metallurgy, and mechanical properties of a porous tantalum foam. J. Biomed. Mater. Res. 2011, 58, 180–187. [Google Scholar] [CrossRef]
- Salem, A.K.; Stevens, R.; Pearson, R.G.; Davies, M.; Tendler, S.J.B.; Roberts, C.J.; Williams, P.; Shakesheff, K.M. Interactions of 3T3 fibroblasts and endothelial cells with defined pore features. J. Biomed. Mater. Res. 2002, 61, 212–217. [Google Scholar] [CrossRef]
- Naseri, N.; Poirier, J.-M.; Girandon, L.; Fröhlich, M.; Oksman, K.; Mathew, A.P. 3-Dimensional porous nanocomposite scaffolds based on cellulose nanofibers for cartilage tissue engineering: Tailoring of porosity and mechanical performance. RSC Adv. 2016, 6, 5999–6007. [Google Scholar] [CrossRef]
- Sultan, S.; Mathew, A.P. 3D printed scaffolds with gradient porosity based on a cellulose nanocrystal hydrogel. Nanoscale 2018, 10, 4421–4431. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.N.; McGuinness, G.B.; Ramos, M.E.T.; Kajiyama, C.E.; Thiré, R.M.; Pinto, J.C.; Balaban, R. Properties of PVA Hydrogel Wound-Care Dressings Containing UK Propolis. Macromol. Symp. 2016, 368, 122–127. [Google Scholar] [CrossRef]
- Sutjarittangtham, K.; Sanpa, S.; Tunkasiri, T.; Rachtanapun, P.; Chantawannakul, P.; Intatha, U.; Pengpat, K.; Rujijanagul, G.; Eitssayeam, S. Preparation of Polycaprolactone/Ethanolic Extract Propolis Nanofibers Films. Adv. Mater. Res. 2012, 506, 226–229. [Google Scholar] [CrossRef]
- Yang, T.; Xie, P.; Wu, Z.; Liao, Y.; Chen, W.; Hao, Z.; Wang, Y.; Zhimin, Z.; Teng, W. The Injectable Woven Bone-Like Hydrogel to Perform Alveolar Ridge Preservation with Adapted Remodeling Performance After Tooth Extraction. Front. Bioeng. Biotechnol. 2020, 8. (In English) [Google Scholar] [CrossRef]
- Tsyganova, A.A.; Golovanova, O.A. Synthesis of a Composite Material Based on a Mixture of Calcium Phosphates and Sodium Alginate. Inorg. Mater. 2019, 55, 1156–1161. (In English) [Google Scholar] [CrossRef]
- Gao, Y.; Jin, X. Dual Crosslinked Methacrylated Alginate Hydrogel Micron Fibers and Tissue Constructs for Cell Biology. Mar. Drugs 2019, 17, 557. [Google Scholar] [CrossRef]
- Simpson, N.E.; Stabler, C.L.; Simpson, C.P.; Sambanis, A.; Constantinidis, I. The role of the CaCl2–guluronic acid interaction on alginate encapsulated βTC3 cells. Biomaterials 2004, 25, 2603–2610. [Google Scholar] [CrossRef]
- Stangl, R.; Rinne, B.; Kastl, S.; Hendrich, C. The influence of pore geometry in cp Ti-implants--a cell culture investigation. Eur. Cell Mater 2001, 2, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Straccia, M.C.; Romano, I.; Oliva, A.; Santagata, G.; Laurienzo, P. Crosslinker effects on functional properties of alginate/N-succinylchitosan based hydrogels. Carbohydr. Polym. 2014, 108, 321–330. [Google Scholar] [CrossRef]
- Bodini, R.; Sobral, P.; Favaro-Trindade, C.; Carvalho, R. Properties of gelatin-based films with added ethanol–propolis extract. LWT Food Sci. Technol. 2013, 51, 104–110. [Google Scholar] [CrossRef]
- Chang-Bravo, L.; Córdoba, A.L.; Martino, M. Biopolymeric matrices made of carrageenan and corn starch for the antioxidant extracts delivery of Cuban red propolis and yerba mate. React. Funct. Polym. 2014, 85, 11–19. [Google Scholar] [CrossRef]
- Zhang, H.; Fu, Y.; Niu, F.; Li, Z.; Ba, C.; Jin, B.; Chen, G.; Li, X. Enhanced antioxidant activity and in vitro release of propolis by acid-induced aggregation using heat-denatured zein and carboxymethyl chitosan. Food Hydrocoll. 2018, 81, 104–112. [Google Scholar] [CrossRef]
- Uzun, M.; Anand, S.; Shah, T. The effect of wound dressings on the pH stability of fuids. J. Wound Care 2012, 21, 88–95. [Google Scholar] [CrossRef]
- Jaya, S.; Durance, T.; Wang, R. Effect of alginate-pectin composition on drug release characteristics of microcapsules. J. Microencapsul. 2008, 26, 143–153. [Google Scholar] [CrossRef] [PubMed]
- Torre, M.L.; Giunchedi, P.; Maggi, L.; Stefli, R.; Machiste, E.O.; Conte, U. Formulation and Characterization of Calcium Alginate Beads Containing Ampicillin. Pharm. Dev. Technol. 1998, 3, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Maji, K.; Dasgupta, S.; Pramanik, K.; Bissoyi, A. Preparation and Evaluation of Gelatin-Chitosan-Nanobioglass 3D Porous Scaffold for Bone Tissue Engineering. Int. J. Biomater. 2016, 2016, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Boateng, J.S.; Matthews, K.H.; Stevens, H.N.; Eccleston, G.M. Wound Healing Dressings and Drug Delivery Systems: A Review. J. Pharm. Sci. 2008, 97, 2892–2923. [Google Scholar] [CrossRef] [PubMed]
- Gupta, B.; Agarwal, R.; Alam, M. Textile-Based Smart Wound Dressings. IJFTR 2010, 35, 174–187. [Google Scholar]
- Candido, J.; Conceição, N.A.; Moreira, A.P.D.; Calçada, L.A.; Araújo, L.S.; Santos, R.A.; Middea, A.; Luchese, R.; Prudencio, E.R.; Castro, R.N.; et al. Alginate hydrogels incorporating neomycin or propolis as potential dressings for diabetic ulcers: Structure, swelling, and antimicrobial barrier properties. Polym. Adv. Technol. 2019, 30, 2623–2635. [Google Scholar] [CrossRef]
- Oliveira, R.N.; Moreira, A.P.D.; Thiré, R.M.D.S.M.; Quilty, B.; Passos, T.M.; Simon, P.; Mancini, M.C.; McGuinness, G.B. Absorbent polyvinyl alcohol-sodium carboxymethyl cellulose hydrogels for propolis delivery in wound healing applications. Polym. Eng. Sci. 2017, 57, 1224–1233. [Google Scholar] [CrossRef]
- Gupta, N.V.; Shivakumar, H. Investigation of Swelling Behavior and Mechanical Properties of a pH-Sensitive Superporous Hydrogel Composite. Iran. J. Pharm. Res. IJPR 2012, 11, 481–493. [Google Scholar] [PubMed]
- Zhang, H.; Zhou, L.; Zhang, W. Control of Scaffold Degradation in Tissue Engineering: A Review. Tissue Eng. Part B Rev. 2014, 20, 492–502. [Google Scholar] [CrossRef]
- Odelius, K.; Hoglund, A.; Kumar, S.; Hakkarainen, M.; Ghosh, A.K.; Bhatnagar, N.; Albertsson, A.-M. Porosity and Pore Size Regulate the Degradation Product Profile of Polylactide. Biomacromolecules 2011, 12, 1250–1258. [Google Scholar] [CrossRef]
- EUCAST Development Laboratory (EDL) for Bacteria. Breakpoint Tables for Interpretation of MICs and Zone Diameters, Version 9.0; EUCAST: Växjö, Sweden, 2019. [Google Scholar]
- Arıkan, H.K.; Solak, H.H. Propolis Extract-PVA Nanocomposites of Textile Design: Antimicrobial Effect on Gram Positive and Negative Bacterias. Int. J. Second. Metab. 2017, 4, 218–224. [Google Scholar] [CrossRef]
- Silici, S.; Kutluca, S. Chemical composition and antibacterial activity of propolis collected by three different races of honeybees in the same region. J. Ethnopharmacol. 2005, 99, 69–73. [Google Scholar] [CrossRef]
- Akca, A.E.; Akca, G.; Topçu, F.T.; Macit, E.; Pikdöken, L.; Özgen, I. Şerif The Comparative Evaluation of the Antimicrobial Effect of Propolis with Chlorhexidine against Oral Pathogens: An In Vitro Study. Biomed. Res. Int. 2016, 2016, 1–8. [Google Scholar] [CrossRef]
- Przybyłek, I.; Karpiński, T.M. Antibacterial Properties of Propolis. Molecules 2019, 24, 2047. [Google Scholar] [CrossRef] [PubMed]
- Bankova, V.S.; Christov, R.; Tejera, A.D. Lignans and other constituents of propolis from the canary islands. Phytochemistry 1998, 49, 1411–1415. [Google Scholar] [CrossRef]
- Sforcin, J.M. Biological Properties and Therapeutic Applications of Propolis. Phytother. Res. 2016, 30, 894–905. [Google Scholar] [CrossRef]
- Kędzia, B.; Hołderna-Kędzia, E. Aktywność antybiotyczna propolisu krajowego i europejskiego. Postępy Fitoter. 2013, 2, 97–107. [Google Scholar]
- Frozza, C.O.D.S.; Garcia, C.S.C.; Gambato, G.; De Souza, M.D.O.; Salvador, M.; Moura, S.; Padilha, F.F.; Seixas, F.K.; Collares, T.; Borsuk, S.; et al. Chemical characterization, antioxidant and cytotoxic activities of Brazilian red propolis. Food Chem. Toxicol. 2013, 52, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Silva, R.P.D.; Machado, B.A.S.; Barreto, G.D.A.; Costa, S.S.; Andrade, L.N.; Amaral, R.G.; Carvalho, A.A.; Padilha, F.F.; Barbosa, J.D.V.; Umsza-Guez, M.A. Antioxidant, antimicrobial, antiparasitic, and cytotoxic properties of various Brazilian propolis extracts. PLoS ONE 2017, 12, e0172585. [Google Scholar] [CrossRef]
- Calhelha, R.C.; Falcão, S.I.; Queiroz, M.-J.R.; Vilas-Boas, M.; Ferreira, I.C. Cytotoxicity of Portuguese Propolis: The Proximity of theIn VitroDoses for Tumor and Normal Cell Lines. Biomed. Res. Int. 2014, 2014, 1–7. [Google Scholar] [CrossRef] [PubMed]



| Prepared Solutions | SA Content (wt %) | Ps Content (v/v %) |
|---|---|---|
| Sample 1 | 4.5 | 0 |
| Sample 2 | 4.5 | 10 |
| Sample 3 | 4.5 | 20 |
| Sample 4 | 4.5 | 40 |
| Samples | Viscosity (mPa.s) | Density (kg/m3) | Surface Tension (mN.m−1) | Image of 20 Layers Printed Scaffold |
|---|---|---|---|---|
| Sample 1 | 6109 | 1.022 ± 0.02 | 39.43 |  |
| Sample 2 | 8018 | 1.023 ± 0.03 | 44.73 |  |
| Sample 3 | 8028 | 1.024 ± 0.02 | 64.78 |  |
| Sample 4 | Measurement failed | Measurement failed | Measurement failed |  |
| Scaffolds | Tensile Strength (MPa) | Elongation at Break (%) |
|---|---|---|
| Sample 1 | 3.92 ± 1.08 | 14.74 ± 1.22 |
| Sample 2 | 2.42 ± 0.62 | 20.57 ± 1.96 |
| Sample 3 | 2.33 ± 1.76 | 25.00 ± 6.68 |
| Release pH | Higuchi Model | Korsmeyer–Peppas Model | ||
|---|---|---|---|---|
| R2 | Kh | R2 | n | |
| pH 7.4 | 0.9822 | 6.025 | 0.9715 | 0.4492 |
| pH 2.0 | 0.9515 | 7.9228 | 0.8633 | 0.6153 |
| Inhibition Zone (mm) | |||||
|---|---|---|---|---|---|
| Bacteria | SA † | Ps ‡ Low | Ps High | Ampicillin 2 | Ampicillin 10 |
| S. aureus | 0 | 15.00 ± 1.1 | 13.00 ± 1.0 | 18.00 ± 1.0 | NA |
| E. coli | 0 | 0 | 0 | NA | 19.00 ± 1.1 |
Sample Availability: Samples of the compounds are not available from the authors. Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aranci, K.; Uzun, M.; Su, S.; Cesur, S.; Ulag, S.; Amin, A.; Guncu, M.M.; Aksu, B.; Kolayli, S.; Ustundag, C.B.; et al. 3D Propolis-Sodium Alginate Scaffolds: Influence on Structural Parameters, Release Mechanisms, Cell Cytotoxicity and Antibacterial Activity. Molecules 2020, 25, 5082. https://doi.org/10.3390/molecules25215082
Aranci K, Uzun M, Su S, Cesur S, Ulag S, Amin A, Guncu MM, Aksu B, Kolayli S, Ustundag CB, et al. 3D Propolis-Sodium Alginate Scaffolds: Influence on Structural Parameters, Release Mechanisms, Cell Cytotoxicity and Antibacterial Activity. Molecules. 2020; 25(21):5082. https://doi.org/10.3390/molecules25215082
Chicago/Turabian StyleAranci, Kubra, Muhammet Uzun, Sena Su, Sumeyye Cesur, Songul Ulag, Al Amin, Mehmet Mucahit Guncu, Burak Aksu, Sevgi Kolayli, Cem Bulent Ustundag, and et al. 2020. "3D Propolis-Sodium Alginate Scaffolds: Influence on Structural Parameters, Release Mechanisms, Cell Cytotoxicity and Antibacterial Activity" Molecules 25, no. 21: 5082. https://doi.org/10.3390/molecules25215082
APA StyleAranci, K., Uzun, M., Su, S., Cesur, S., Ulag, S., Amin, A., Guncu, M. M., Aksu, B., Kolayli, S., Ustundag, C. B., Silva, J. C., Ficai, D., Ficai, A., & Gunduz, O. (2020). 3D Propolis-Sodium Alginate Scaffolds: Influence on Structural Parameters, Release Mechanisms, Cell Cytotoxicity and Antibacterial Activity. Molecules, 25(21), 5082. https://doi.org/10.3390/molecules25215082













