Advances of Anti-Caries Nanomaterials
Abstract
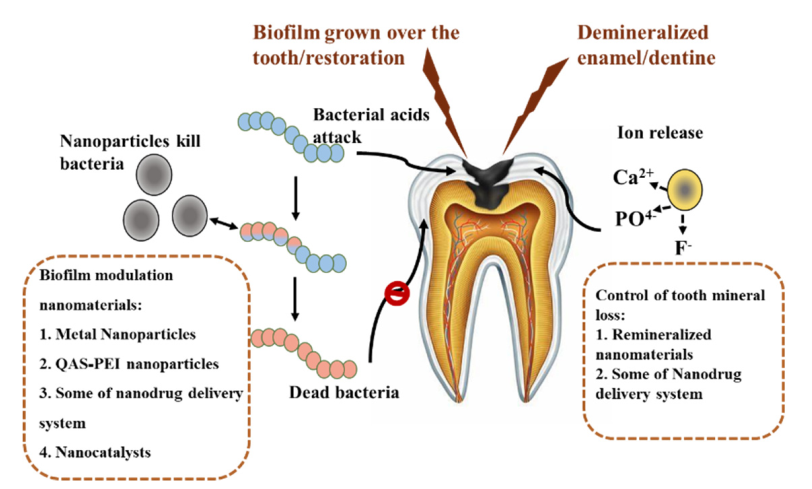
1. Introduction
2. Metal Nanoparticles Used in Caries Infections
2.1. Silver Nanoparticle (NAg)
2.2. Nano-Zinc (NZn) and Nano-Zincoxide (NZnO)
2.3. Other Metal Nanoparticles
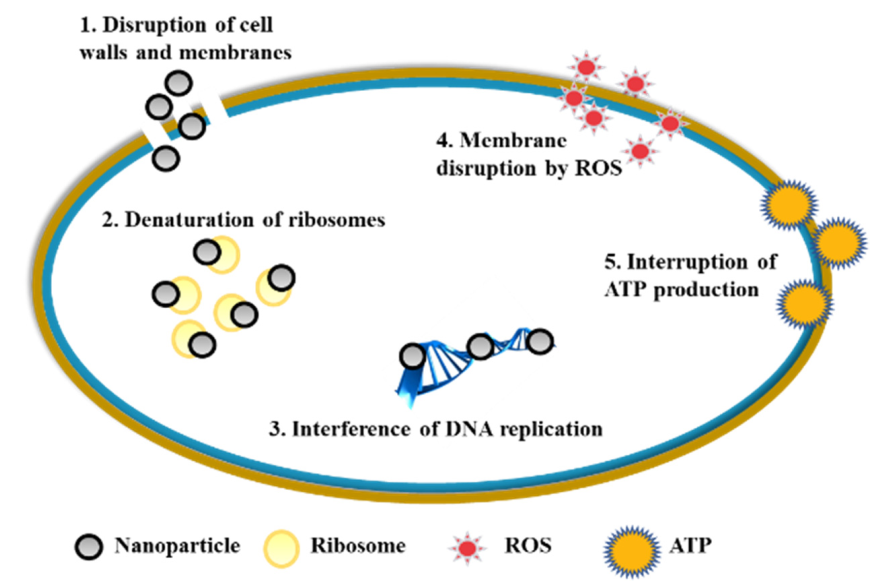
2.4. The Anti-Caries Mechanism of Mental Nanoparticles
3. Quaternary Ammonium Salt Polyethylenimine (QAS-PEI) Nanoparticles
4. Remineralized Nano Anti-Caries Materials
4.1. Nano Particulate Hydroxyapatite (NHAP)
4.2. Nanosized Calcium Fluoride (NCaF2)
4.3. Nanosized Amorphous Calciumphosphate Particle (NACP)
4.4. Bioactive Glass Nanoparticle (NBG)
4.5. Remineralization Mechanism of Nanoparticles
5. Nanodrug Delivery System
5.1. Mesoporous Silica Nanoparticle (MSN)
5.2. Liposomes
5.3. Halloysite Nano-Tube (HNT)
5.4. Polyamidoamine (PAMAM)
5.5. Dental Caries Vaccine
6. Biomimetic Nanocatalyst
7. Conclusions
| Type | Component | Modified Materials | Concentration | Model In Vitro Experiment | Mechanism of Anti-Caries | Ref. |
|---|---|---|---|---|---|---|
| Metal Nanoparticles | Silver Nanoparticle (NAg) | Resin Adhesive | Resin: 0%~0.088% Adhesive: 1–5% | Streptococcus mutans and Lactobacillus | ①Ag ions penetrate the cell membrane and enter the microbial body. ②Mechanism of reactive oxygen species (ROS). | [103] |
| NAg-NZnO | Resin | NAg, 1% NZnO, 1% | Streptococcus mutans and Lactobacillus | [104] | ||
| NAg-Laden Hydroxyapatite | Resin | 6–8 wt.% | Streptococcus mutans | ①Antibacterial mechanism of NAg. ②Hydroxyapatite can precipitate on the surface of demineralized enamel to form a new layer and promote remineralization. | [105][45] | |
| NAg- Polyamidoamine (PAMAM)-Cellulose Nanocrystals (CNCs) | Resin | 0.1 wt.% | Staphylococcus aureus and Escherichia coli | Antibacterial mechanism of NAg and remineralized mechanism of PAMAM. | [14] | |
| NAg-Quaternary Ammonium Dimethacrylate (QADM) | Adhesive | NAg, 0.05% QADM, 10% | Streptococcus mutans | ①Antibacterial mechanism of NAg. ②Mechanism of quaternary ammonium salt QAS. CHK1-mediated two-component regulatory system results in the accumulation of ROS which induces cell apoptosis. | [106] | |
| NAg- 12-methacryloxydodecylpyridium bromide (MDPB) | Adhesive | NAg, 0.1% MDPB, 2.5% | Human saliva biofilms | [107] | ||
| NAg- Dimethylaminododecyl methacrylate (DMADDM) | Adhesive | NAg, 0.1% DMADDM, 5% | Dental plaque microcosm biofilm model | [108] | ||
| Nano-Zinc (NZn) | Adhesive | 2.15 ± 0.05 µg Zn/mg NPs | Extracted unerupted human third molars | Inhibit MMP activity, reduction of the decomposition of dentin collagen bundle, protection of mineral crystal at the interface of resin-tooth formation. | [109] | |
| Nano-Zincoxide (NZnO) | Resin | 1% | Streptococcus mutans | ①NZnO has higher surface potential energy and can release more zinc ions to kill bacteria. ②NZnO can also activate the photocatalytic antibacterial mechanism and produce a large number of free radicals to interact with bacteria. | [21] | |
| TiO2 nanoparticles (NTiO2) | Glass-ionomer | 3% and 5% | Streptococcus mutans | ①Contact inhibited mechanism. ②Mechanism of reactive oxygen species (ROS). | [28] | |
| Nano-SiO2 (NSiO2) | Acrylic resin | 1% | Lactobacillus acidophilus and Streptococcus mutans | [31] | ||
| Copper Nanoparticles (NCuO) | Adhesive | 0.01, 0.5, and 1 wt.% | Streptococcus mutans | [32] | ||
| MgO Nanoparticles (NMgO) | Glass-ionomer | 1% and 2.5% | Streptococcus mutans and Streptococcus sobrinus | [35] | ||
| QAS | Quaternary Ammonium Salt Polyethylenimine (QAS-PEI) Nanoparticles | Resin | 1–2 wt.% | Streptococcus mutans and Lactobacillus | The electrostatic interaction between positively charged QAS-PEI and negatively charged bacterial cell walls. | [43] |
| Remineralized Nanopaticles | Nano Particulate Hydroxyapatite (NHAP) | Resin | 2–5–10% | Sound premolars fixed in acrylic blocks and coated with nail polish | NHAP can stably release Ca2+ and PO43− to promote remineralization. | [48] |
| Nanosized Calcium Fluoride (NCaF2) | Resin | 17% | Biofilm by Streptococcus mutans on the tooth surface | NCaF2 materials can keep fluorine release at a better level for a long time to promote tooth remineralization. | [51] | |
| Nanosized Amorphous Calciumphosphate Particle (NACP) | Resin | Ca and P with concentrations of 8 mmol/L and 5.333 mmol/L | Dental plaque microcosm biofilm model | NACP can release higher levels of Ca2+ and PO43− at low pH, with the acid invasion neutralization, increasing the pH value from 4 to 6.5 to resist dental caries. | [61,62] | |
| Bioactive Glass Nanoparticle (NBG) | Resin | 20 wt% | Bioglass | ①It will release Ca2+ and PO43− to form a mineralized layer with a porous network. ②NBG can interfere with the degradation of collagenase, formation of high alkaline pH, resulting in antibacterial ions (such as Ag+) release to achieve an antibacterial effect. | [73] | |
| Drug Delivery System | Mesoporous Silica Nanoparticle (MSN) | Adhesive | 34 wt% | Multi-species biofilms | As a carrier, the system can slowly release antibacterial/remineralization particles. | [81] |
| Liposome | - | 0.05%, 0.2% | Dental enamel, Saliva | [83] | ||
| Halloysite Nano-Tube (HNT) | Adhesive | 20% | Streptococcus mutans | [86] | ||
| PAMAM | Adhesive/resin | 0.3% (w/v) | Dentin disks, Artificial saliva | [87] | ||
| Caries Vaccine | - | 50 mmol/L | Plasmid | [100] | ||
| Nanocatalyst | Catalytic iIon Oxide Nanoparticles (CAT-NP) | gargle | 4% | a rodent caries model | As a catalyst, it can catalyze the effect of H2O2 against cariogenic bacteria. | [101] |
| Dextran-Coated Iron Oxide Nanoparticles termed Nanozymes (Dex-NZM) | gargle | - | a rodent caries model | As a catalyst, it can catalyze the effect of Fe4O3 against cariogenic bacteria. | [102] |
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Li, X.; Kolltveit, K.M.; Tronstad, L.; Olsen, I. Systemic diseases caused by oral infection. Clin. Microbiol. Rev. 2000, 13, 547–558. [Google Scholar] [CrossRef] [PubMed]
- Vos, T.; Allen, C.; Arora, M.; Barber, R.M.; Bhutta, Z.A.; Brown, A.; Carter, A.; Casey, D.C.; Charlson, F.J.; Chen, A.Z. Global, regional, and national incidence, prevalence, and years lived with disability for 310 diseases and injuries, 1990–2015. Lancet 2016, 388, 1545–1602. [Google Scholar] [CrossRef]
- Choudhury, H.; Gorain, B.; Karmakar, S.; Biswas, E.; Dey, G.; Barik, R.; Mandal, M.; Pal, T.K. Improvement of cellular uptake, in vitro antitumor activity and sustained release profile with increased bioavailability from a nanoemulsion platform. Int. J. Pharm. 2014, 460, 131–143. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Guardiola, A.; Ortiz-Cano, R.; Sandoval-Salinas, M.E.; Fernández-Rossier, J.; Casanova, D.; Pérez-Jiménez, A.J.; Sancho-García, J.C. From cyclic nanorings to single-walled carbon nanotubes: Disclosing the evolution of their electronic structure with the help of theoretical methods. J. Phys. Chem. Chem. Phys. 2019, 21, 2547–2557. [Google Scholar] [CrossRef] [PubMed]
- Song, W.; Ge, S. Application of Antimicrobial Nanoparticles in Dentistry. Molecules 2019, 24, 1033. [Google Scholar] [CrossRef] [PubMed]
- Zakharova, O.V.; Godymchuk, A.Y.; Gusev, A.A.; Gulchenko, S.I.; Vasyukova, I.A.; Kuznetsov, D.V. Considerable Variation of Antibacterial Activity of Cu Nanoparticles Suspensions Depending on the Storage Time, Dispersive Medium, and Particle Sizes. Biomed. Res. Int. 2015, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Gurunathan, S.; Han, J.W.; Dayem, A.A.; Eppakayala, V.; Kim, J.H. Oxidative stress-mediated antibacterial activity of graphene oxide and reduced graphene oxide in Pseudomonas aeruginosa. Int. J. Nanomed. 2012, 7, 5901–5914. [Google Scholar] [CrossRef]
- Leung, Y.H.; Ng, A.M.; Xu, X.; Shen, Z.; Gethings, L.A.; Wong, M.T.; Chan, C.M.; Guo, M.Y.; Ng, Y.H.; Djurisic, A.B.; et al. Mechanisms of antibacterial activity of MgO: Non-ROS mediated toxicity of MgO nanoparticles towards Escherichia coli. Small 2014, 10, 1171–1183. [Google Scholar] [CrossRef]
- Oliveira, C.A.; Campos, R.M.; Macedo, J.P.; Silva, A.R.; Maximo, L.N.; Silva, T.M.; Franca, F.M.; Turssi, C.P.; Basting, R.T.; Goncalves, S.E.P.; et al. Incorporation of ZnCl2 into an etch-and-rinse adhesive system on flexural strength, degree of conversion and bond durability to caries-affected dentin. Am. J. Dent. 2019, 32, 299–305. [Google Scholar]
- Elkassas, D.; Arafa, A. The innovative applications of therapeutic nanostructures in dentistry. Nanomedicine 2017, 13, 1543–1562. [Google Scholar] [CrossRef]
- Cheng, Y.J.; Zeiger, D.N.; Howarter, J.A.; Zhang, X.; Lin, N.J.; Antonucci, J.M.; Lin-Gibson, S. In situ formation of silver nanoparticles in photocrosslinking polymers. J. Biomed. Mater. Res. B Appl. Biomater. 2011, 97, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Weir, M.D.; Xu, H.H.; Antonucci, J.M.; Lin, N.J.; Lin-Gibson, S.; Xu, S.M.; Zhou, X. Effect of amorphous calcium phosphate and silver nanocomposites on dental plaque microcosm biofilms. J. Biomed. Mater. Res. B Appl. Biomater. 2012, 100, 1378–1386. [Google Scholar] [CrossRef]
- Azarsina, M.; Kasraei, S.; Yousef-Mashouf, R.; Dehghani, N.; Shirinzad, M. The antibacterial properties of composite resin containing nanosilver against Streptococcus mutans and Lactobacillus. J. Contemp. Dent. Pr. 2013, 14, 1014–1018. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Yang, J.; Jia, Y.G.; Lu, B.; Ren, L. A Study of 3D-Printable Reinforced Composite Resin: PMMA Modified with Silver Nanoparticles Loaded Cellulose Nanocrystal. Materials 2018, 11, 2444. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Zhang, K.; Zhou, C.C.; Weir, M.D.; Zhou, X.D.; Xu, H.H. One-year water-ageing of calcium phosphate composite containing nano-silver and quaternary ammonium to inhibit biofilms. Int. J. Oral Sci. 2016, 29, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Zhao, Q.; Lu, S.; Fu, Y.; Yu, D.; Zhao, W. Inhibitory effect of reduced graphene oxide-silver nanocomposite on progression of artificial enamel caries. J. Appl. Oral Sci. 2018, 27, e20180042. [Google Scholar] [CrossRef]
- Zhao, I.S.; Yin, I.X.; Mei, M.L.; Lo, E.C.M.; Tang, J.; Li, Q.; So, L.Y.; Chu, C.H. Remineralising Dentine Caries Using Sodium Fluoride with Silver Nanoparticles: An In Vitro Study. Int. J. Nanomed. 2020, 15, 2829–2839. [Google Scholar] [CrossRef]
- Metin-Gursoy, G.; Taner, L.; Akca, G. Nanosilver coated orthodontic brackets: In vivo antibacterial properties and ion release. Eur. J. Orthod. 2017, 39, 9–16. [Google Scholar] [CrossRef]
- Toledano, M.; Sauro, S.; Cabello, I.; Watson, T.; Osorio, R. A Zn-doped etch-and-rinse adhesive may improve the mechanical properties and the integrity at the bonded-dentin interface. Dent. Mater. 2013, 29, e142–e152. [Google Scholar] [CrossRef]
- Toledano, M.; Aguilera, F.S.; Osorio, E.; Cabello, I.; Toledano-Osorio, M.; Osorio, R. Self-etching zinc-doped adhesives improve the potential of caries-affected dentin to be functionally remineralized. Biointerphases 2015, 10, 031002. [Google Scholar] [CrossRef]
- Cheng, L.; Weir, M.D.; Xu, H.H.; Antonucci, J.M.; Kraigsley, A.M.; Lin, N.J.; Lin-Gibson, S.; Zhou, X. Antibacterial amorphous calcium phosphate nanocomposites with a quaternary ammonium dimethacrylate and silver nanoparticles. Dent. Mater. 2012, 28, 561–572. [Google Scholar] [CrossRef]
- Tavassoli Hojati, S.; Alaghemand, H.; Hamze, F.; Ahmadian Babaki, F.; Rajab-Nia, R.; Rezvani, M.B.; Kaviani, M.; Atai, M. Antibacterial, physical and mechanical properties of flowable resin composites containing zinc oxide nanoparticles. Dent. Mater. 2013, 29, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Al-Mosawi, R.M.; Al-Badr, R.M. The Study Effects of Dental Composite Resin as Antibacterial Agent Which Contain Nanoparticles of Zinc Oxide on the Bacteria Associated with Oral Infection. J. Dent. Med. Sci. 2017, 16, 49–55. [Google Scholar] [CrossRef]
- Yusof, N.A.A.; Zain, N.M.; Pauzi, N. Synthesis of ZnO nanoparticles with chitosan as stabilizing agent and their antibacterial properties against Gram-positive and Gram-negative bacteria. Int. J. Biol. Macromol. 2019, 124, 1132–1136. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hua, H.; Li, W.; Wang, R.; Jiang, X.; Zhu, M. Strong antibacterial dental resin composites containing cellulose nanocrystal/zinc oxide nanohybrids. J. Dent. 2019, 80, 23–29. [Google Scholar] [CrossRef]
- Gutierrez, M.F.; Bermudez, J.; Davila-Sanchez, A.; Alegria-Acevedo, L.F.; Mendez-Bauer, L.; Hernandez, M.; Astorga, J.; Reis, A.; Loguercio, A.D.; Farago, P.V.; et al. Zinc oxide and copper nanoparticles addition in universal adhesive systems improve interface stability on caries-affected dentin. J. Mech. Behav. Biomed. Mater. 2019, 100, 103366. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Contreras, R.; Scougall-Vilchis, R.J.; Contreras-Bulnes, R.; Sakagami, H.; Morales-Luckie, R.A.; Nakajima, H. Mechanical, antibacterial and bond strength properties of nano-titanium-enriched glass ionomer cement. J. Appl. Oral Sci. Rev. FOB 2015, 23, 321–328. [Google Scholar] [CrossRef]
- Elsaka, S.E.; Hamouda, I.M.; Swain, M.V. Titanium dioxide nanoparticles addition to a conventional glass-ionomer restorative: Influence on physical and antibacterial properties. J. Dent. 2011, 39, 589–598. [Google Scholar] [CrossRef]
- Esteban Florez, F.L.; Hiers, R.D.; Larson, P.; Johnson, M.; O’Rear, E.; Rondinone, A.J.; Khajotia, S.S. Antibacterial dental adhesive resins containing nitrogen-doped titanium dioxide nanoparticles. Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 93, 931–943. [Google Scholar] [CrossRef]
- Esteban Florez, F.L.; Kraemer, H.; Hiers, R.D.; Sacramento, C.M.; Rondinone, A.J.; Silverio, K.G.; Khajotia, S.S. Sorption, solubility and cytotoxicity of novel antibacterial nanofilled dental adhesive resins. Sci. Rep. 2020, 10, 13503. [Google Scholar] [CrossRef]
- Sodagar, A.; Khalil, S.; Kassaee, M.Z.; Shahroudi, A.S.; Pourakbari, B.; Bahador, A. Antimicrobial properties of poly (methyl methacrylate) acrylic resins incorporated with silicon dioxide and titanium dioxide nanoparticles on cariogenic bacteria. J. Orthod. Sci. 2016, 5, 7–13. [Google Scholar] [CrossRef]
- Toodehzaeim, M.H.; Zandi, H.; Meshkani, H.; Hosseinzadeh Firouzabadi, A. The Effect of CuO Nanoparticles on Antimicrobial Effects and Shear Bond Strength of Orthodontic Adhesives. J. Dent. 2018, 19, 1–5. [Google Scholar]
- Sabatini, C.; Mennito, A.S.; Wolf, B.J.; Pashley, D.H.; Renne, W.G. Incorporation of bactericidal poly-acrylic acid modified copper iodide particles into adhesive resins. J. Dent. 2015, 43, 546–555. [Google Scholar] [CrossRef] [PubMed]
- Gutierrez, M.F.; Malaquias, P.; Matos, T.P.; Szesz, A.; Souza, S.; Bermudez, J.; Reis, A.; Loguercio, A.D.; Farago, P.V. Mechanical and microbiological properties and drug release modeling of an etch-and-rinse adhesive containing copper nanoparticles. Dent. Mater. 2017, 33, 309–320. [Google Scholar] [CrossRef] [PubMed]
- Noori, A.J.; Kareem, F.A. The effect of magnesium oxide nanoparticles on the antibacterial and antibiofilm properties of glass-ionomer cement. Heliyon 2019, 5, e02568. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Zhang, Y.; Pan, X.; Zhu, F.; Jiang, C. Antibacterial activity and mechanism of silver nanoparticles against multidrug-resistant Pseudomonas aeruginosa. Int. J. Nanomed. 2019, 14, 1469–1487. [Google Scholar] [CrossRef]
- Hosono, H.; Abe, Y. Silver ion selective porous lithium titanium phosphate glass-ceramics cation exchanger and its application to bacteriostatic materials. Mater. Res. Bull. 1994, 29, 1157–1162. [Google Scholar] [CrossRef]
- Chen, H.; Zhou, Y.; Zhou, X.; Liao, B.; Xu, H.H.K.; Chu, C.-H.; Cheng, L.; Ren, B. Dimethylaminododecyl methacrylate inhibits Candida albicans and oropharyngeal candidiasis in a pH-dependent manner. Appl. Microbiol. Biotechnol. 2020, 104, 3585–3595. [Google Scholar] [CrossRef]
- Chen, H.; Han, Q.; Zhou, X.; Zhang, K.; Wang, S.; Xu, H.H.K.; Weir, M.D.; Feng, M.; Li, M.; Peng, X.; et al. Heat-Polymerized Resin Containing Dimethylaminododecyl Methacrylate Inhibits Candida albicans Biofilm. Materials 2017, 10, 431. [Google Scholar] [CrossRef]
- Zou, Y.; Li, D.; Shen, M.; Shi, X. Polyethylenimine-Based Nanogels for Biomedical Applications. Macromol. Biosci. 2019, 19, e1900272. [Google Scholar] [CrossRef]
- Zaltsman, N.; Kesler-Shvero, D.; Weiss, E.I.; Beyth, N. Synthesis Variants of Quaternary Ammonium Polyethyleneimine Nanoparticles and Their Antibacterial Efficacy in Dental Materials. J. Appl. Biomater. Funct. Mater. 2018, 14, e205–e211. [Google Scholar] [CrossRef]
- Yudovin-Farber, I.; Beyth, N.; Weiss, E.I.; Domb, A.J. Antibacterial effect of composite resins containing quaternary ammonium polyethyleneimine nanoparticles. J. Nanoparticle Res. 2009, 12, 591–603. [Google Scholar] [CrossRef]
- Melo, M.A.; Guedes, S.F.; Xu, H.H.; Rodrigues, L.K. Nanotechnology-based restorative materials for dental caries management. Trends Biotechnol. 2013, 31, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Beyth, N.; Houri-Haddad, Y.; Baraness-Hadar, L.; Yudovin-Farber, I.; Domb, A.J.; Weiss, E.I.J.B. Surface antimicrobial activity and biocompatibility of incorporated polyethylenimine nanoparticles. Biomaterials 2008, 29, 4157–4163. [Google Scholar] [CrossRef] [PubMed]
- Vyavhare, S.; Sharma, D.S.; Kulkarni, V.K. Effect of three different pastes on remineralization of initial enamel lesion: An in vitro study. J. Clin. Pediatr. Dent. 2015, 39, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Memarpour, M.; Shafiei, F.; Rafiee, A.; Soltani, M.; Dashti, M.H. Effect of hydroxyapatite nanoparticles on enamel remineralization and estimation of fissure sealant bond strength to remineralized tooth surfaces: An in vitro study. BMC Oral Health 2019, 19, 92. [Google Scholar] [CrossRef]
- Andrade Neto, D.M.; Carvalho, E.V.; Rodrigues, E.A.; Feitosa, V.P.; Sauro, S.; Mele, G.; Carbone, L.; Mazzetto, S.E.; Rodrigues, L.K.; Fechine, P.B. Novel hydroxyapatite nanorods improve anti-caries efficacy of enamel infiltrants. Dent. Mater. 2016, 32, 784–793. [Google Scholar] [CrossRef]
- Haghgoo, R.; Rezvani, M.B.; Salehi Zeinabadi, M. Comparison of nano-hydroxyapatite and sodium fluoride mouthrinse for remineralization of incipient carious lesions. J. Dent. 2014, 11, 406–410. [Google Scholar]
- Leitune, V.C.; Collares, F.M.; Trommer, R.M.; Andrioli, D.G.; Bergmann, C.P.; Samuel, S.M. The addition of nanostructured hydroxyapatite to an experimental adhesive resin. J. Dent. 2013, 41, 321–327. [Google Scholar] [CrossRef]
- Bossu, M.; Saccucci, M.; Salucci, A.; Di Giorgio, G.; Bruni, E.; Uccelletti, D.; Sarto, M.S.; Familiari, G.; Relucenti, M.; Polimeni, A. Enamel remineralization and repair results of Biomimetic Hydroxyapatite toothpaste on deciduous teeth: An effective option to fluoride toothpaste. J. Nanobiotechnol. 2019, 17, 17. [Google Scholar] [CrossRef]
- Ghafar, H.; Khan, M.I.; Sarwar, H.S.; Yaqoob, S.; Hussain, S.Z.; Tariq, I.; Madni, A.U.; Shahnaz, G.; Sohail, M.F. Development and Characterization of Bioadhesive Film Embedded with Lignocaine and Calcium Fluoride Nanoparticles. AAPS Pharmscitech. 2020, 21, 60. [Google Scholar] [CrossRef] [PubMed]
- Weir, M.D.; Moreau, J.L.; Levine, E.D.; Strassler, H.E.; Chow, L.C.; Xu, H.H. Nanocomposite containing CaF(2) nanoparticles: Thermal cycling, wear and long-term water-aging. Dent. Mater. 2012, 28, 642–652. [Google Scholar] [CrossRef]
- Yi, J.; Dai, Q.; Weir, M.D.; Melo, M.A.S.; Lynch, C.D.; Oates, T.W.; Zhang, K.; Zhao, Z.; Xu, H.H.K. A nano-CaF2-containing orthodontic cement with antibacterial and remineralization capabilities to combat enamel white spot lesions. J. Dent. 2019, 89, 103172. [Google Scholar] [CrossRef] [PubMed]
- Fei, X.; Li, Y.; Weir, M.D.; Baras, B.H.; Wang, H.; Wang, S.; Sun, J.; Melo, M.A.S.; Ruan, J.; Xu, H.H.K. Novel pit and fissure sealant containing nano-CaF2 and dimethylaminohexadecyl methacrylate with double benefits of fluoride release and antibacterial function. Dent. Mater. 2020, 36, 1241–1253. [Google Scholar] [CrossRef]
- Kulshrestha, S.; Khan, S.; Hasan, S.; Khan, M.E.; Misba, L.; Khan, A.U. Calcium fluoride nanoparticles induced suppression of Streptococcus mutans biofilm: An in vitro and in vivo approach. Appl. Microbiol. Biotechnol. 2016, 100, 1901–1914. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Seo, S.J.; Kim, H.W. Bioactive glass-based nanocomposites for personalized dental tissue regeneration. Dent. Mater. J. 2016, 35, 710–720. [Google Scholar] [CrossRef]
- Li, F.; Wang, P.; Weir, M.D.; Fouad, A.F.; Xu, H.H. Evaluation of antibacterial and remineralizing nanocomposite and adhesive in rat tooth cavity model. Acta Biomater. 2014, 10, 2804–2813. [Google Scholar] [CrossRef]
- Wu, J.; Weir, M.D.; Melo, M.A.; Xu, H.H. Development of novel self-healing and antibacterial dental composite containing calcium phosphate nanoparticles. J. Dent. 2015, 43, 317–326. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, L.; Niu, L.N.; Yu, T.; Xu, H.H.K.; Weir, M.D.; Oates, T.W.; Tay, F.R.; Chen, J.H. Antibacterial and remineralizing orthodontic adhesive containing quaternary ammonium resin monomer and amorphous calcium phosphate nanoparticles. J. Dent. 2018, 72, 53–63. [Google Scholar] [CrossRef]
- Weir, M.D.; Chow, L.C.; Xu, H.H. Remineralization of demineralized enamel via calcium phosphate nanocomposite. J. Dent. Res. 2012, 91, 979–984. [Google Scholar] [CrossRef]
- Al-Dulaijan, Y.A.; Cheng, L.; Weir, M.D.; Melo, M.A.S.; Liu, H.; Oates, T.W.; Wang, L.; Xu, H.H.K. Novel rechargeable calcium phosphate nanocomposite with antibacterial activity to suppress biofilm acids and dental caries. J. Dent. 2018, 72, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Weir, M.D.; Chow, L.C.; Antonucci, J.M.; Chen, J.; Xu, H.H. Novel rechargeable calcium phosphate dental nanocomposite. Dent. Mater. 2016, 32, 285–293. [Google Scholar] [CrossRef]
- Gao, Y.; Liang, K.; Weir, M.D.; Gao, J.; Imazato, S.; Tay, F.R.; Lynch, C.D.; Oates, T.W.; Li, J.; Xu, H.H.K. Enamel remineralization via poly(amido amine) and adhesive resin containing calcium phosphate nanoparticles. J. Dent. 2020, 92, 103262. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Peng, X.; Zhou, X.; Weir, M.D.; Melo, M.A.S.; Tay, F.R.; Imazato, S.; Oates, T.W.; Cheng, L.; Xu, H.H.K. In vitro evaluation of composite containing DMAHDM and calcium phosphate nanoparticles on recurrent caries inhibition at bovine enamel-restoration margins. Dent. Mater. 2020, 36, 1343–1355. [Google Scholar] [CrossRef] [PubMed]
- Tao, S.; He, L.; Xu, H.H.K.; Weir, M.D.; Fan, M.; Yu, Z.; Zhang, M.; Zhou, X.; Liang, K.; Li, J. Dentin remineralization via adhesive containing amorphous calcium phosphate nanoparticles in a biofilm-challenged environment. J. Dent. 2019, 89, 103193. [Google Scholar] [CrossRef]
- Yue, S.; Wu, J.; Zhang, Q.; Zhang, K.; Weir, M.D.; Imazato, S.; Bai, Y.; Xu, H.H.K. Novel dental adhesive resin with crack self-healing, antimicrobial and remineralization properties. J. Dent. 2018, 75, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhou, C.; Ruan, J.; Weir, M.D.; Tay, F.; Sun, J.; Melo, M.A.S.; Oates, T.W.; Chang, X.; Xu, H.H.K. Self-healing adhesive with antibacterial activity in water-aging for 12 months. Dent. Mater. 2019, 35, 1104–1116. [Google Scholar] [CrossRef]
- Zhou, W.; Zhou, X.; Huang, X.; Zhu, C.; Weir, M.D.; Melo, M.A.S.; Bonavente, A.; Lynch, C.D.; Imazato, S.; Oates, T.W.; et al. Antibacterial and remineralizing nanocomposite inhibit root caries biofilms and protect root dentin hardness at the margins. J. Dent. 2020, 97, 103344. [Google Scholar] [CrossRef]
- Ibrahim, M.S.; Balhaddad, A.A.; Garcia, I.M.; Collares, F.M.; Weir, M.D.; Xu, H.H.K.; Melo, M.A.S. pH-responsive calcium and phosphate-ion releasing antibacterial sealants on carious enamel lesions in vitro. J. Dent. 2020, 97, 103323. [Google Scholar] [CrossRef]
- Xie, X.J.; Xing, D.; Wang, L.; Zhou, H.; Weir, M.D.; Bai, Y.X.; Xu, H.H. Novel rechargeable calcium phosphate nanoparticle-containing orthodontic cement. Int. J. Oral Sci. 2017, 9, 24–32. [Google Scholar] [CrossRef]
- Vichery, C.; Nedelec, J.M. Bioactive Glass Nanoparticles: From Synthesis to Materials Design for Biomedical Applications. Materials 2016, 9, 288. [Google Scholar] [CrossRef] [PubMed]
- Corral Nunez, C.; Covarrubias, C.; Fernandez, E.; Oliveira, O.B.J. Enhanced bioactive properties of BiodentineTM modified with bioactive glass nanoparticles. J. Appl. Oral Sci. 2017, 25, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Taubock, T.T.; Zehnder, M.; Schweizer, T.; Stark, W.J.; Attin, T.; Mohn, D. Functionalizing a dentin bonding resin to become bioactive. Dent. Mater. 2014, 30, 868–875. [Google Scholar] [CrossRef] [PubMed]
- Robinson, C.; Shore, R.C.; Brookes, S.J.; Strafford, S.; Wood, S.R.; Kirkham, J. The chemistry of enamel caries. Crit. Rev. Oral Biol. Med. 2000, 11, 481–495. [Google Scholar] [CrossRef]
- Arifa, M.K.; Ephraim, R.; Rajamani, T. Recent Advances in Dental Hard Tissue Remineralization: A Review of Literature. Int. J. Clin. Pediatr. Dent. 2019, 12, 139–144. [Google Scholar] [CrossRef]
- Philip, N. State of the Art Enamel Remineralization Systems: The Next Frontier in Caries Management. Caries Res. 2019, 53, 284–295. [Google Scholar] [CrossRef]
- Jung, J.H.; Park, S.B.; Yoo, K.H.; Yoon, S.Y.; Bae, M.K.; Lee, D.J.; Ko, C.C.; Kwon, Y.H.; Kim, Y.I. Effect of different sizes of bioactive glass-coated mesoporous silica nanoparticles on dentinal tubule occlusion and mineralization. Clin. Oral Investig. 2019, 23, 2129–2141. [Google Scholar] [CrossRef]
- Tian, L.; Peng, C.; Shi, Y.; Guo, X.; Zhong, B.; Qi, J.; Wang, G.; Cai, Q.; Cui, F. Effect of mesoporous silica nanoparticles on dentinal tubule occlusion: An in vitro study using SEM and image analysis. Dent. Mater. J. 2014, 33, 125–132. [Google Scholar] [CrossRef]
- Seneviratne, C.J.; Leung, K.C.; Wong, C.H.; Lee, S.F.; Li, X.; Leung, P.C.; Lau, C.B.; Wat, E.; Jin, L. Nanoparticle-encapsulated chlorhexidine against oral bacterial biofilms. PLoS ONE 2014, 9, e103234. [Google Scholar] [CrossRef]
- Zhang, J.F.; Wu, R.; Fan, Y.; Liao, S.; Wang, Y.; Wen, Z.T.; Xu, X. Antibacterial dental composites with chlorhexidine and mesoporous silica. J. Dent. Res. 2014, 93, 1283–1289. [Google Scholar] [CrossRef]
- Stewart, C.A.; Hong, J.H.; Hatton, B.D.; Finer, Y. Responsive antimicrobial dental adhesive based on drug-silica co-assembled particles. Acta Biomater. 2018, 76, 283–294. [Google Scholar] [CrossRef]
- Gregoriadis, G.; Ryman, B.E. Liposomes as carriers of enzymes or drugs: A new approach to the treatment of storage diseases. Biochem. J. 1971, 124, 58P. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, S.; Hiorth, M.; Rykke, M.; Smistad, G. Polymer coated liposomes for dental drug delivery--interactions with parotid saliva and dental enamel. Eur. J. Pharm. Sci. 2013, 50, 78–85. [Google Scholar] [CrossRef]
- Shu, Z.; Zhang, Y.; Yang, Q.; Yang, H. Halloysite Nanotubes Supported Ag and ZnO Nanoparticles with Synergistically Enhanced Antibacterial Activity. Nanoscale Res. Lett. 2017, 12, 135. [Google Scholar] [CrossRef] [PubMed]
- Bottino, M.C.; Batarseh, G.; Palasuk, J.; Alkatheeri, M.S.; Windsor, L.J.; Platt, J.A. Nanotube-modified dentin adhesive--physicochemical and dentin bonding characterizations. Dent. Mater. 2013, 29, 1158–1165. [Google Scholar] [CrossRef] [PubMed]
- Feitosa, S.A.; Palasuk, J.; Kamocki, K.; Geraldeli, S.; Gregory, R.L.; Platt, J.A.; Windsor, L.J.; Bottino, M.C. Doxycycline-Encapsulated Nanotube-Modified Dentin Adhesives. J. Dent. Res. 2014, 93, 1270–1276. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Yang, J.; Lin, Z.; Li, J.; Liang, K.; Yuan, H.; Li, S.; Li, J. Triclosan-loaded poly(amido amine) dendrimer for simultaneous treatment and remineralization of human dentine. Colloids Surf. B Biointerfaces 2014, 115, 237–243. [Google Scholar] [CrossRef]
- Liang, K.; Gao, Y.; Xiao, S.; Tay, F.R.; Weir, M.D.; Zhou, X.; Oates, T.W.; Zhou, C.; Li, J.; Xu, H.H.K. Poly(amido amine) and rechargeable adhesive containing calcium phosphate nanoparticles for long-term dentin remineralization. J. Dent. 2019, 85, 47–56. [Google Scholar] [CrossRef]
- Liang, K.; Weir, M.D.; Xie, X.; Wang, L.; Reynolds, M.A.; Li, J.; Xu, H.H. Dentin remineralization in acid challenge environment via PAMAM and calcium phosphate composite. Dent. Mater. 2016, 32, 1429–1440. [Google Scholar] [CrossRef]
- Xiao, S.; Liang, K.; Weir, M.D.; Cheng, L.; Liu, H.; Zhou, X.; Ding, Y.; Xu, H.H.K. Combining Bioactive Multifunctional Dental Composite with PAMAM for Root Dentin Remineralization. Materials 2017, 10, 89. [Google Scholar] [CrossRef]
- Liang, K.; Weir, M.D.; Reynolds, M.A.; Zhou, X.; Li, J.; Xu, H.H.K. Poly (amido amine) and nano-calcium phosphate bonding agent to remineralize tooth dentin in cyclic artificial saliva/lactic acid. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 72, 7–17. [Google Scholar] [CrossRef]
- Lee, J.H.; El-Fiqi, A.; Jo, J.K.; Kim, D.A.; Kim, S.C.; Jun, S.K.; Kim, H.W.; Lee, H.H. Development of long-term antimicrobial poly(methyl methacrylate) by incorporating mesoporous silica nanocarriers. Dent. Mater. 2016, 32, 1564–1574. [Google Scholar] [CrossRef]
- Moyle, P.M.; Mcgeary, R.P.; Blanchfield, J.T.; Toth, I. Mucosal immunization: Adjuvants and delivery systems. Curr. Drug Deliv. 2004, 1, 385–396. [Google Scholar] [CrossRef] [PubMed]
- Jin, Z.; Gao, S.; Cui, X.; Sun, D.; Zhao, K. Adjuvants and delivery systems based on polymeric nanoparticles for mucosal vaccines. Int. J. Pharm. 2019, 572, 118731. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.H.; Jia, R.; Fan, M.W.; Bian, Z.; Chen, Z.; Peng, B. Construction and Immunogenic Characterization of a Fusion Anti-caries DNA Vaccine against PAc and Glucosyltransferase I of Streptococcus mutans. J. Dent. Res. 2004, 83, 266–270. [Google Scholar] [CrossRef] [PubMed]
- Rong, J.; Ji, H.G.; Ming, W.F.; Bian, Z.; Zhi, C.; Peng, B.; Bing, F.J.V. Mucosal immunization against dental caries with plasmid DNA encoding pac gene of Streptococcus mutans in rats. Vaccine 2004, 22, 2511–2516. [Google Scholar] [CrossRef]
- Shrestha, A.; Fong, S.W.; Khoo, B.C.; Kishen, A. Delivery of Antibacterial Nanoparticles into Dentinal Tubules Using High-intensity Focused Ultrasound. J. Endod. 2009, 35, 1028–1033. [Google Scholar] [CrossRef]
- Calvo, P.; Remuñán-López, C.; Vila-Jato, J.L.; Alonso, M.J. Novel hydrophilic chitosan-polyethylene oxide nanoparticles as protein carriers. J. Appl. Polym. Sci. 1997, 63, 125–132. [Google Scholar] [CrossRef]
- Mcneela., E.A.; O’Connor, D.; Jabbal-Gill., I.; Mills., K.H.G. A mucosal vaccine against diphtheria: Formulation of cross reacting material (CRM(197)) of diphtheria toxin with chitosan enhances local and systemic antibody and Th2 responses following nasal delivery. Vaccine 2000, 19, 1188–1198. [Google Scholar] [CrossRef]
- Li, Y.H.; Fan, M.W.; Bian, Z.; Chen, Z.; Zhang, Q.; Yang, H.R. Chitosan-DNA microparticles as mucosal delivery system: Synthesis, characterization and release in vitro. Chin. J. Med. 2005, 118, 936–941. [Google Scholar] [PubMed]
- Gao, L.; Liu, Y.; Kim, D.; Li, Y.; Hwang, G.; Naha, P.C.; Cormode, D.P.; Koo, H. Nanocatalysts promote Streptococcus mutans biofilm matrix degradation and enhance bacterial killing to suppress dental caries in vivo. Biomaterials 2016, 101, 272–284. [Google Scholar] [CrossRef] [PubMed]
- Naha, P.C.; Liu, Y.; Hwang, G.; Huang, Y.; Gubara, S.; Jonnakuti, V.; Simon-Soro, A.; Kim, D.; Gao, L.; Koo, H.; et al. Dextran-Coated Iron Oxide Nanoparticles as Biomimetic Catalysts for Localized and pH-Activated Biofilm Disruption. ACS Nano 2019, 13, 4960–4971. [Google Scholar] [CrossRef] [PubMed]
- Noronha, V.T.; Paula, A.J.; Durán, G.; Galembeck, A.; Cogo-Müller, K.; Franz-Montan, M.; Durán, N. Silver nanoparticles in dentistry. Dent Mater. 2017, 33, 1110–1126. [Google Scholar] [CrossRef]
- Kasraei, S.; Sami, L.; Hendi, S.; Alikhani, M.Y.; Rezaei-Soufi, L.; Khamverdi, Z. Antibacterial properties of composite resins incorporating silver and zinc oxide nanoparticles on Streptococcus mutans and Lactobacillus. Restor. Dent. Endod. 2014, 39, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Ai, M.; Du, Z.; Zhu, S.; Geng, H.; Zhang, X.; Cai, Q.; Yang, X. Composite resin reinforced with silver nanoparticles-laden hydroxyapatite nanowires for dental application. Dent. Mater. 2017, 33, 12–22. [Google Scholar] [CrossRef]
- Li, F.; Weir, M.D.; Chen, J.; Xu, H.H. Comparison of quaternary ammonium-containing with nano-silver-containing adhesive in antibacterial properties and cytotoxicity. Dent. Mater. 2013, 29, 450–461. [Google Scholar] [CrossRef]
- Zhang, K.; Li, F.; Imazato, S.; Cheng, L.; Liu, H.; Arola, D.D.; Bai, Y.; Xu, H.H. Dual antibacterial agents of nano-silver and 12-methacryloyloxydodecylpyridinium bromide in dental adhesive to inhibit caries. J. Biomed. Mater. Res. B Appl. Biomater. 2013, 101, 929–938. [Google Scholar] [CrossRef]
- Zhang, K.; Cheng, L.; Wu, E.J.; Weir, M.D.; Bai, Y.; Xu, H.H. Effect of water-ageing on dentine bond strength and anti-biofilm activity of bonding agent containing new monomer dimethylaminododecyl methacrylate. J. Dent. 2013, 41, 504–513. [Google Scholar] [CrossRef]
- Toledano, M.; Osorio, R.; Osorio, E.; Medina-Castillo, A.L.; Toledano-Osorio, M.; Aguilera, F.S. Ions-modified nanoparticles affect functional remineralization and energy dissipation through the resin-dentin interface. J. Mech. Behav. Biomed. Mater. 2017, 68, 62–79. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, H.; Gu, L.; Liao, B.; Zhou, X.; Cheng, L.; Ren, B. Advances of Anti-Caries Nanomaterials. Molecules 2020, 25, 5047. https://doi.org/10.3390/molecules25215047
Chen H, Gu L, Liao B, Zhou X, Cheng L, Ren B. Advances of Anti-Caries Nanomaterials. Molecules. 2020; 25(21):5047. https://doi.org/10.3390/molecules25215047
Chicago/Turabian StyleChen, Hui, Lisha Gu, Binyou Liao, Xuedong Zhou, Lei Cheng, and Biao Ren. 2020. "Advances of Anti-Caries Nanomaterials" Molecules 25, no. 21: 5047. https://doi.org/10.3390/molecules25215047
APA StyleChen, H., Gu, L., Liao, B., Zhou, X., Cheng, L., & Ren, B. (2020). Advances of Anti-Caries Nanomaterials. Molecules, 25(21), 5047. https://doi.org/10.3390/molecules25215047







