Plasma and Urinary (Poly)phenolic Profiles after 4-Week Red Raspberry (Rubus idaeus L.) Intake with or without Fructo-Oligosaccharide Supplementation
Abstract
1. Introduction
2. Results
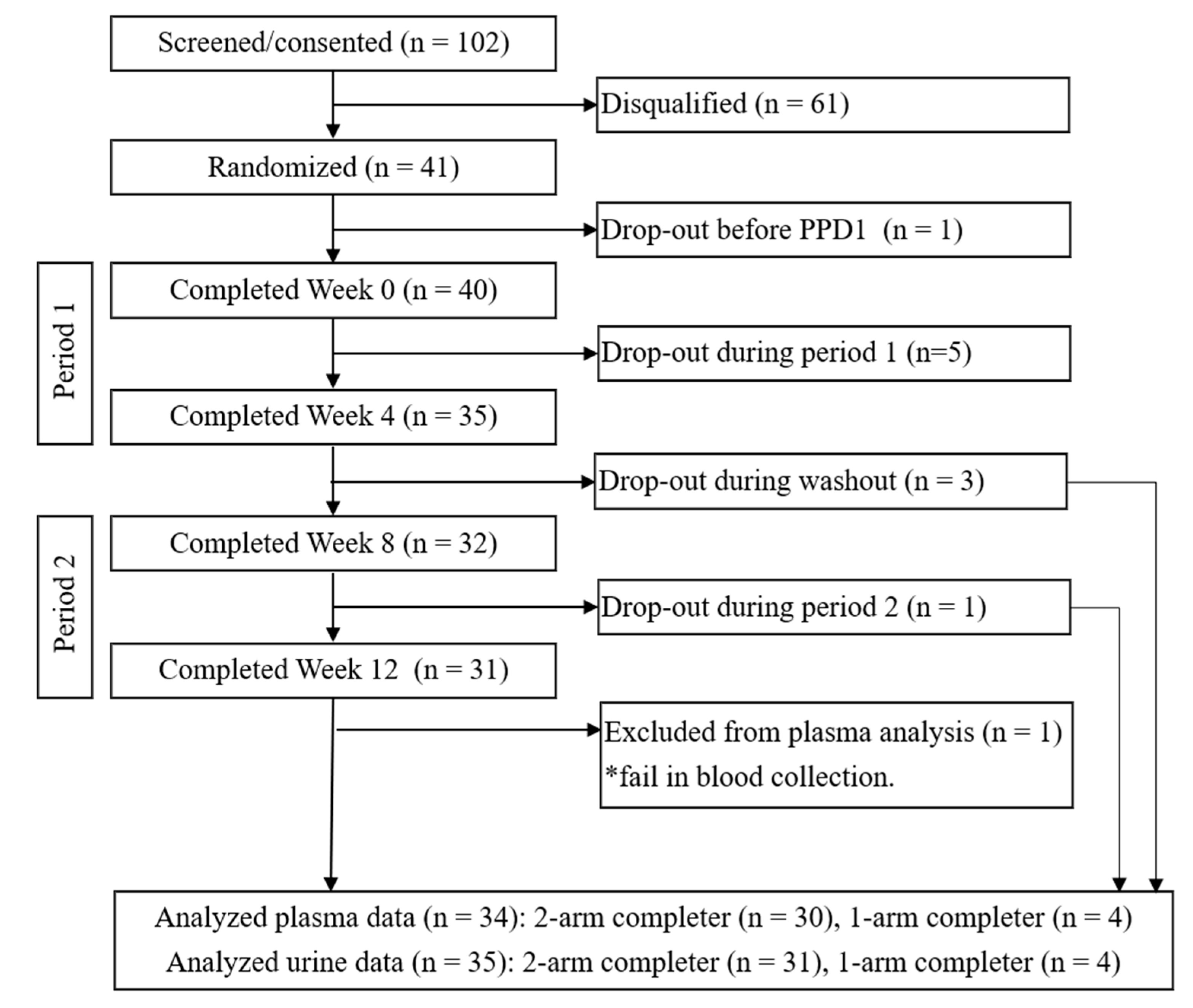
2.1. Subject Demographics and Characteristics
2.2. (Poly)phenol Content of RRB Interventions
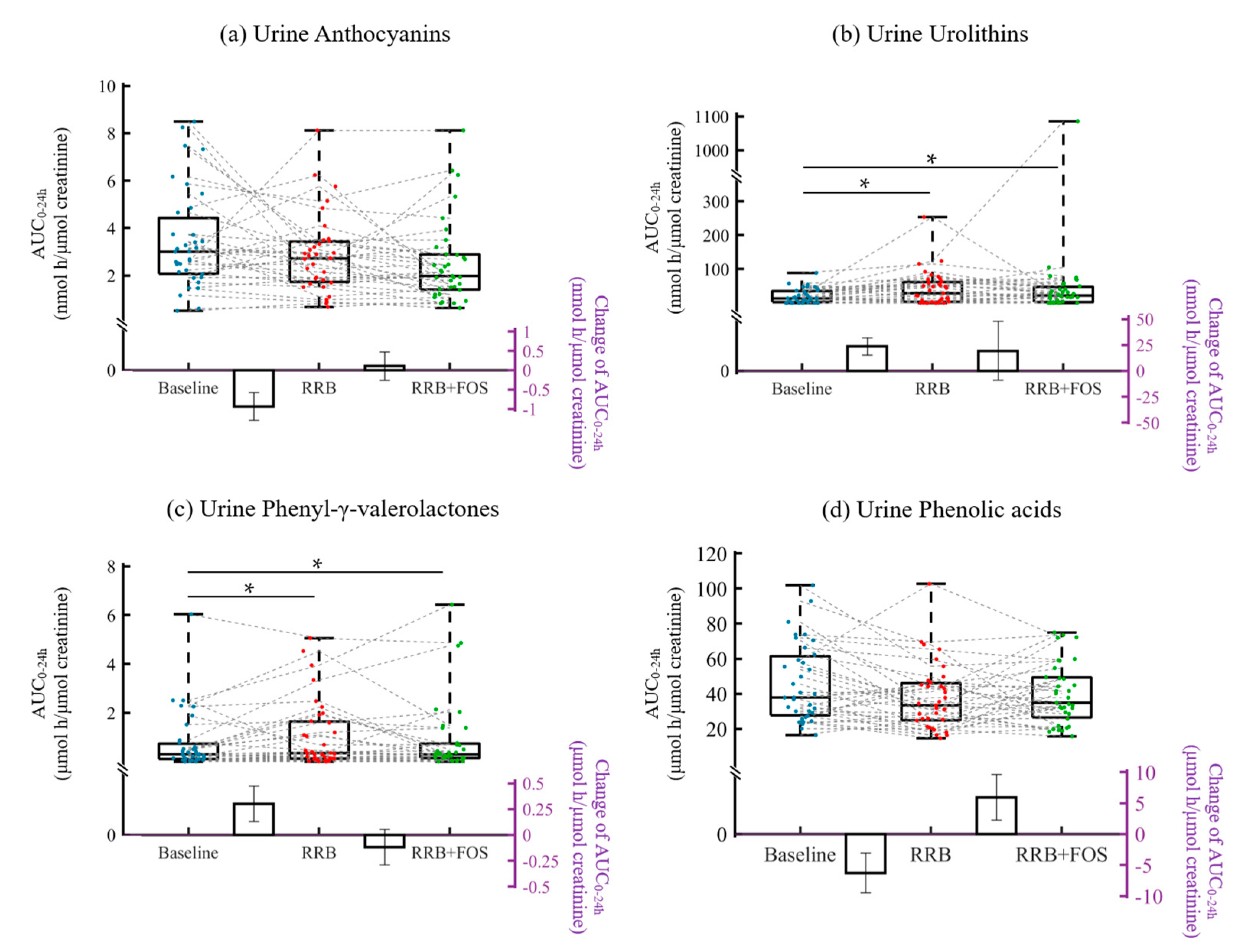
2.3. Plasma and Urine (poly)phenolic Metabolites: Effect of Chronic Exposure to RRB or RRB+FOS
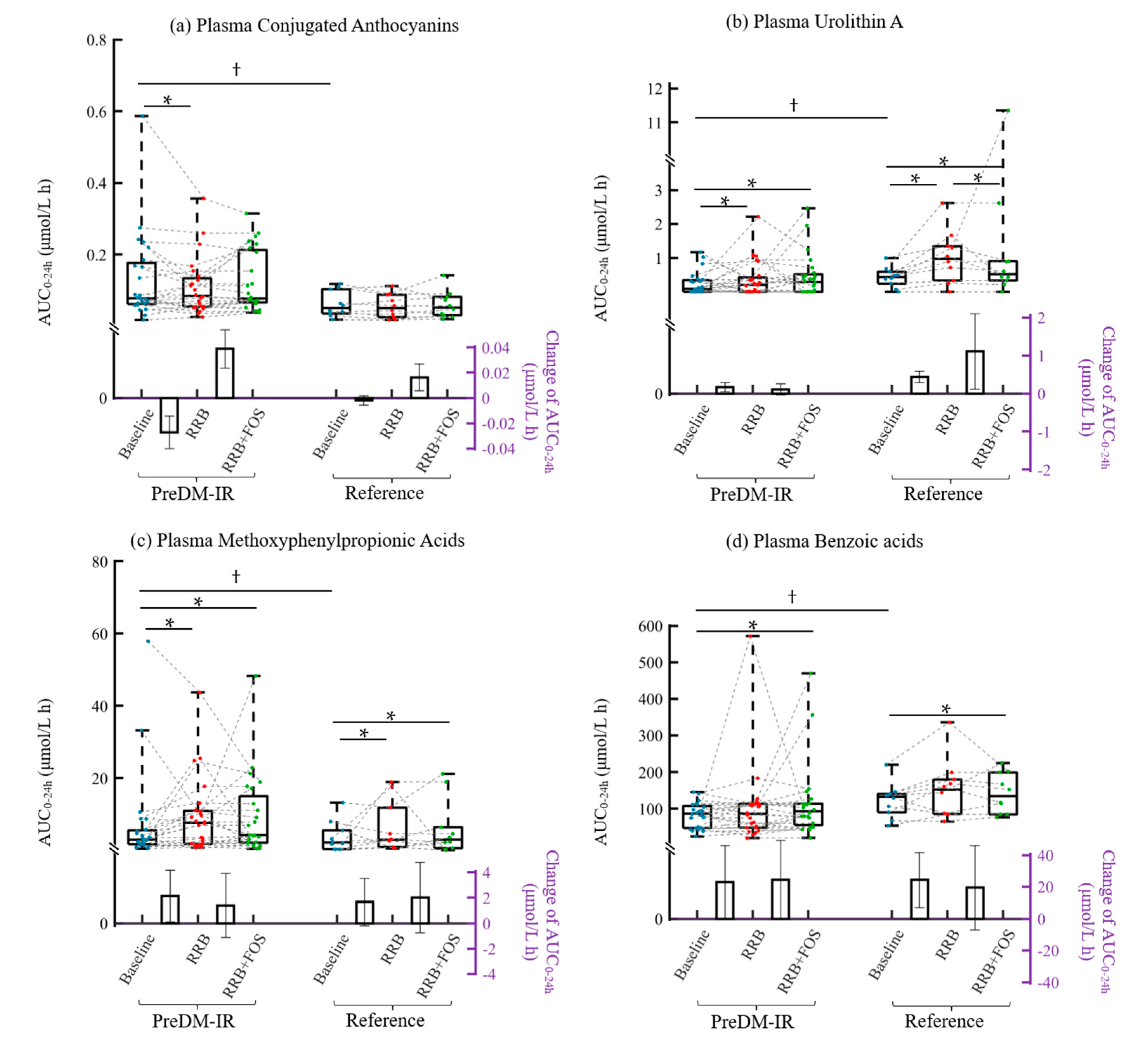
2.4. Metabolic Status and Plasma and Urine (Poly)phenolic Metabolites
3. Discussion
4. Materials and Methods
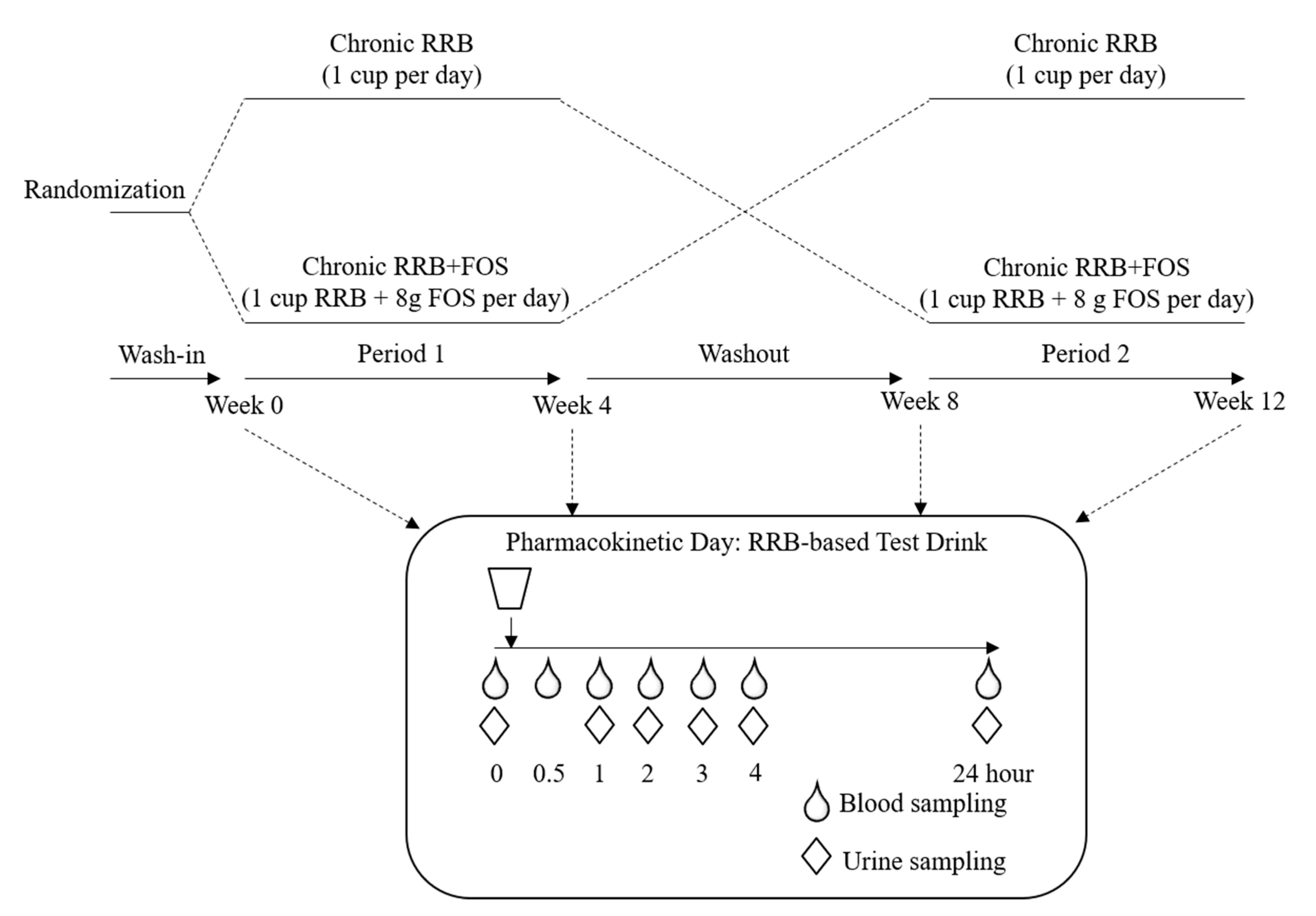
4.1. Study Design
4.2. Study Participants
4.3. Study Foods and RRB Supplements
4.4. Study Procedures
4.5. Dietary Assessment
4.6. Chemical and Reagents
4.7. Plasma and Urine Samples Processing and Analysis by UHPLC-QQQ
4.8. Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ludwig, I.A.; Mena, P.; Calani, L.; Borges, G.; Pereira-Caro, G.; Bresciani, L.; Del Rio, D.; Lean, M.E.J.; Crozier, A. New insights into the bioavailability of red raspberry anthocyanins and ellagitannins. Free Radic. Biol. Med. 2015, 89, 758–769. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Sandhu, A.; Edirisinghe, I.; Burton-Freeman, B. An exploratory study of red raspberry (Rubus idaeus L.) (poly)phenols/metabolites in human biological samples. Food Funct. 2018, 9, 806–818. [Google Scholar] [CrossRef] [PubMed]
- Burton-Freeman, B.M.; Sandhu, A.K.; Edirisinghe, I. Red Raspberries and Their Bioactive Polyphenols: Cardiometabolic and Neuronal Health Links. Adv. Nutr. 2016, 7, 44–65. [Google Scholar] [CrossRef]
- Zou, T.; Wang, B.; Yang, Q.; de Avila, J.M.; Zhu, M.-J.; You, J.; Chen, D.; Du, M. Raspberry promotes brown and beige adipocyte development in mice fed high-fat diet through activation of AMP-activated protein kinase (AMPK) α1. J. Nutr. Biochem. 2018, 55, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Lila, M.A.; Burton-Freeman, B.; Grace, M.; Kalt, W. Unraveling Anthocyanin Bioavailability for Human Health. Annu. Rev. Food Sci. Technol. 2016, 7, 375–393. [Google Scholar] [CrossRef]
- Chandra, P.; Rathore, A.S.; Kay, K.L.; Everhart, J.L.; Curtis, P.; Burton-Freeman, B.; Cassidy, A.; Kay, C.D. Contribution of Berry Polyphenols to the Human Metabolome. Molecules 2019, 24, 4220. [Google Scholar] [CrossRef] [PubMed]
- Mena, P.; Bresciani, L.; Brindani, N.; Ludwig, I.A.; Pereira-Caro, G.; Angelino, D.; Llorach, R.; Calani, L.; Brighenti, F.; Clifford, M.N.; et al. Phenyl-γ-valerolactones and phenylvaleric acids, the main colonic metabolites of flavan-3-ols: Synthesis, analysis, bioavailability, and bioactivity. Nat. Prod. Rep. 2019, 36, 714–752. [Google Scholar] [CrossRef]
- Mena, P.; Ludwig, I.A.; Tomatis, V.B.; Acharjee, A.; Calani, L.; Rosi, A.; Brighenti, F.; Ray, S.; Griffin, J.L.; Bluck, L.J.; et al. Inter-individual variability in the production of flavan-3-ol colonic metabolites: Preliminary elucidation of urinary metabotypes. Eur. J. Nutr. 2019, 58, 1529–1543. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; García-Villalba, R.; González-Sarrías, A.; Selma, M.V.; Espín, J.C. Ellagic Acid Metabolism by Human Gut Microbiota: Consistent Observation of Three Urolithin Phenotypes in Intervention Trials, Independent of Food Source, Age, and Health Status. J. Agric. Food Chem. 2014, 62, 6535–6538. [Google Scholar] [CrossRef]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef]
- Le Bourgot, C.; Apper, E.; Blat, S.; Respondek, F. Fructo-oligosaccharides and glucose homeostasis: A systematic review and meta-analysis in animal models. Nutr. Metab. (Lond.) 2018, 15, 9. [Google Scholar] [CrossRef]
- Juśkiewicz, J.; Milala, J.; Jurgoński, A.; Król, B.; Zduńczyk, Z. Consumption of polyphenol concentrate with dietary fructo-oligosaccharides enhances cecal metabolism of quercetin glycosides in rats. Nutrition 2011, 27, 351–357. [Google Scholar] [CrossRef]
- Jurgoński, A.; Juśkiewicz, J.; Fotschki, B.; Kołodziejczyk, K.; Milala, J.; Kosmala, M.; Grzelak-Błaszczyk, K.; Markiewicz, L. Metabolism of strawberry mono- and dimeric ellagitannins in rats fed a diet containing fructo-oligosaccharides. Eur. J. Nutr. 2017, 56, 853–864. [Google Scholar] [CrossRef] [PubMed]
- Abdul-Ghani, M.A.; Matsuda, M.; Balas, B.; DeFronzo, R.A. Muscle and Liver Insulin Resistance Indexes Derived from the Oral Glucose Tolerance Test. Diabetes Care 2007, 30, 89–94. [Google Scholar] [CrossRef]
- Afshin, A.; Sur, P.J.; Fay, K.A.; Cornaby, L.; Ferrara, G.; Salama, J.S.; Mullany, E.C.; Abate, K.H.; Abbafati, C.; Abebe, Z.; et al. Health effects of dietary risks in 195 countries, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2019, 393, 1958–1972. [Google Scholar] [CrossRef]
- Allin, K.H.; Tremaroli, V.; Caesar, R.; Jensen, B.A.H.; Damgaard, M.T.F.; Bahl, M.I.; Licht, T.R.; Hansen, T.H.; Nielsen, T.; Dantoft, T.M.; et al. Aberrant intestinal microbiota in individuals with prediabetes. Diabetologia 2018, 61, 810–820. [Google Scholar] [CrossRef] [PubMed]
- Zhong, H.; Ren, H.; Lu, Y.; Fang, C.; Hou, G.; Yang, Z.; Chen, B.; Yang, F.; Zhao, Y.; Shi, Z.; et al. Distinct gut metagenomics and metaproteomics signatures in prediabetics and treatment-naïve type 2 diabetics. EBioMedicine 2019, 47, 373–383. [Google Scholar] [CrossRef]
- Stevens, J.F.; Maier, C.S. The chemistry of gut microbial metabolism of polyphenols. Phytochem. Rev. 2016, 15, 425–444. [Google Scholar] [CrossRef] [PubMed]
- de Ferrars, R.M.; Czank, C.; Zhang, Q.; Botting, N.P.; Kroon, P.A.; Cassidy, A.; Kay, C.D. The pharmacokinetics of anthocyanins and their metabolites in humans. Br. J. Pharmacol. 2014, 171, 3268–3282. [Google Scholar] [CrossRef]
- Selma, M.V.; Tomás-Barberán, F.A.; Beltrán, D.; García-Villalba, R.; Espín, J.C. Gordonibacter urolithinfaciens sp. nov., a urolithin-producing bacterium isolated from the human gut. Int. J. Syst. Evol. Microbiol. 2014, 64, 2346–2352. [Google Scholar] [CrossRef]
- Beltrán, D.; Romo-Vaquero, M.; Espín, J.C.; Tomás-Barberán, F.A.; Selma, M.V. Ellagibacter isourolithinifaciens gen. nov., sp. nov., a new member of the family Eggerthellaceae, isolated from human gut. Int. J. Syst. Evol. Microbiol. 2018, 68, 1707–1712. [Google Scholar] [CrossRef] [PubMed]
- Romo-Vaquero, M.; Cortés-Martín, A.; Loria-Kohen, V.; Ramírez-de-Molina, A.; García-Mantrana, I.; Collado, M.C.; Espín, J.C.; Selma, M.V. Deciphering the Human Gut Microbiome of Urolithin Metabotypes: Association with Enterotypes and Potential Cardiometabolic Health Implications. Mol. Nutr. Food Res. 2019, 63, e1800958. [Google Scholar] [CrossRef] [PubMed]
- Márquez Campos, E.; Stehle, P.; Simon, M.C. Microbial Metabolites of Flavan-3-Ols and Their Biological Activity. Nutrients 2019, 11, 2260. [Google Scholar] [CrossRef] [PubMed]
- Feliciano, R.P.; Boeres, A.; Massacessi, L.; Istas, G.; Ventura, M.R.; Nunes dos Santos, C.; Heiss, C.; Rodriguez-Mateos, A. Identification and quantification of novel cranberry-derived plasma and urinary (poly)phenols. Arch. Biochem. Biophys. 2016, 599, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mateos, A.; Feliciano, R.P.; Boeres, A.; Weber, T.; dos Santos, C.N.; Ventura, M.R.; Heiss, C. Cranberry (poly)phenol metabolites correlate with improvements in vascular function: A double-blind, randomized, controlled, dose-response, crossover study. Mol. Nutr. Food Res. 2016, 60, 2130–2140. [Google Scholar] [CrossRef]
- Gómez-Juaristi, M.; Sarria, B.; Martínez-López, S.; Bravo Clemente, L.; Mateos, R. Flavanol Bioavailability in Two Cocoa Products with Different Phenolic Content. A Comparative Study in Humans. Nutrients 2019, 11, 1441. [Google Scholar] [CrossRef]
- Sun, Y.N.; Li, W.; Song, S.B.; Yan, X.T.; Zhao, Y.; Jo, A.R.; Kang, J.S.; Young Ho, K. A new phenolic derivative with soluble epoxide hydrolase and nuclear factor-kappaB inhibitory activity from the aqueous extract of Acacia catechu. Nat. Prod. Res. 2016, 30, 2085–2092. [Google Scholar] [CrossRef]
- Lambert, J.D.; Rice, J.E.; Hong, J.; Hou, Z.; Yang, C.S. Synthesis and biological activity of the tea catechin metabolites, M4 and M6 and their methoxy-derivatives. Bioorg. Med. Chem. Lett. 2005, 15, 873–876. [Google Scholar] [CrossRef]
- Takagaki, A.; Nanjo, F. Effects of Metabolites Produced from (−)-Epigallocatechin Gallate by Rat Intestinal Bacteria on Angiotensin I-Converting Enzyme Activity and Blood Pressure in Spontaneously Hypertensive Rats. J. Agric. Food Chem. 2015, 63, 8262–8266. [Google Scholar] [CrossRef]
- Mele, L.; Carobbio, S.; Brindani, N.; Curti, C.; Rodriguez-Cuenca, S.; Bidault, G.; Mena, P.; Zanotti, I.; Vacca, M.; Vidal-Puig, A.; et al. Phenyl-γ-valerolactones, flavan-3-ol colonic metabolites, protect brown adipocytes from oxidative stress without affecting their differentiation or function. Mol. Nutr. Food Res. 2017, 61, 1700074. [Google Scholar] [CrossRef]
- Xiao, D.; Zhu, L.; Edirisinghe, I.; Fareed, J.; Brailovsky, Y.; Burton-Freeman, B. Attenuation of Postmeal Metabolic Indices with Red Raspberries in Individuals at Risk for Diabetes: A Randomized Controlled Trial. Obesity 2019, 27, 542–550. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Ford, E.S.; McGuire, L.C.; Mokdad, A.H.; Little, R.R.; Reaven, G.M. Trends in Hyperinsulinemia among Nondiabetic Adults in the U.S. Diabetes Care 2006, 29, 2396–2402. [Google Scholar] [CrossRef] [PubMed]
- Subar, A.F.; Thompson, F.E.; Potischman, N.; Forsyth, B.H.; Buday, R.; Richards, D.; McNutt, S.; Hull, S.G.; Guenther, P.M.; Schatzkin, A.; et al. Formative Research of a Quick List for an Automated Self-Administered 24 Hour Dietary Recall. J. Am. Diet. Assoc. 2007, 107, 1002–1007. [Google Scholar] [CrossRef] [PubMed]

| Variable 2 | PreDM-IR (n = 25) | Reference (n = 10) | p Value |
|---|---|---|---|
| Age (years) | 35 ± 2 | 31 ± 3 | NS |
| Female: Male | 11:14 | 7: 3 | NS |
| CAU:AA:AS:HIS | 8:6:8:3 | 3:2:3:2 | NS |
| Fasting Glucose (mmol/L) | 5.7 ± 0.1 | 5.1 ± 0.2 | <0.0001 |
| Fasting Insulin (pmol/L) | 84 ± 9 | 34 ± 4 | 0.002 |
| HOMA-IR | 3.3 ± 0.4 | 1.0 ± 0.1 | 0.0005 |
| BMI (kg/m2) | 28 ± 1 | 22 ± 1 | 0.006 |
| Weight (kg) | 84 ± 4 | 64 ± 5 | 0.02 |
| RT | Compounds | MRM Transition | RRBtest Drink | 4-Week Supplementations | |
|---|---|---|---|---|---|
| Daily RRB Drink | Daily RRB+FOS Drink | ||||
| 6.5 | Cyanidin 3,5-O-diglucoside | 611+/287 | 0.8 ± 0.1 | 0.6 ± 0.0 | 0.5 ± 0.0 |
| 8.2 | Cyanidin 3-O-sophoroside | 611+/287 | 192.2 ± 2.2 | 61.0 ± 0.4 | 60.2 ± 1.1 |
| 9.1 | Cyanidin 3-O-sambubioside | 581+/287 | 10.1 ± 0.1 | 2.9 ± 0.0 | 2.4 ± 0.1 |
| 9.2 | Cyanidin 3-O-glucoside | 449+/287 | 27.4 ± 0.1 | 8.9 ± 0.0 | 9.9 ± 0.3 |
| 9.4 | Pelargonidin 3-O-sophoroside | 595+/271 | 6.3 ± 0.1 | 3.6 ± 0.1 | 3.4 ± 0.1 |
| 10.5 | Pelargonidin 3-O-glucoside | 433+/271 | 0.5 ± 0.0 | 0.3 ± 0.0 | 0.3 ± 0.0 |
| Total anthocyanins | 236.8 ± 1.9 | 77.3 ± 0.3 | 76.7 ± 1.3 | ||
| 3.5 | Pedunculagin isomer 1 | 783−/301 | 0.5 ± 0.0 | 1.2 ± 0.0 | 0.9 ± 0.0 |
| 5.2 | Pedunculagin isomer 2 | 783−/301 | 0.3 ± 0.0 | 0.6 ± 0.0 | 0.4 ± 0.0 |
| 6.8 | Sanguiin H-10 isomer 1 | 7832−/301 | 1.7 ± 0.1 | 0.7 ± 0.0 | 0.7 ± 0.0 |
| 6.8 | Sanguiin H-6 minus gallic moiety isomer 1 | 8582−/301 | 1.0 ± 0.0 | 0.5 ± 0.0 | 0.5 ± 0.0 |
| 7.4 | Corilagin | 633−/301 | 0.3 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 10.0 | Sanguiin H-10 isomer 2 | 7832−/301 | 3.6 ± 0.1 | 1.6 ± 0.0 | 1.5 ± 0.1 |
| 11.1 | Sanguiin H-6 minus gallic moiety isomer 2 | 8582−/301 | 0.3 ± 0.0 | 0.3 ± 0.0 | 0.2 ± 0.0 |
| 11.3 | Sanguiin H-10 isomer 3 | 7832−/301 | 1.1 ± 0.1 | 0.2 ± 0.1 | 0.2 ± 0.0 |
| 11.9 | Lambertianin C | 14012−/301 | 22.7 ± 0.6 | 8.0 ± 0.7 | 8.3 ± 0.3 |
| 12.3 | Sanguiin H6 | 9342−/301 | 81.4 ± 1.7 | 25.2 ± 1.3 | 25.5 ± 0.5 |
| 13.1 | Ellagic acid pentoside isomer 1 | 433−/301 | 1.7 ± 0.0 | 0.8 ± 0.0 | 0.7 ± 0.0 |
| 13.3 | Ellagic acid pentoside isomer 2 | 433−/301 | 2.2 ± 0.1 | 0.7 ± 0.0 | 0.7 ± 0.0 |
| 13.6 | Ellagic acid | 301−/301 | 5.9 ± 0.1 | 2.0 ± 0.1 | 2.3 ± 0.2 |
| 14.9 | Methyl ellagic acid pentoside 1 | 447−/301 | 0.6 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 15.2 | Methyl ellagic acid pentoside 2 | 447−/301 | 0.3 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 15.2 | Ellagic acid acetyl pentoside isomer 1 | 475−/301 | 0.3 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 15.5 | Ellagic acid acetyl pentoside isomer 2 | 475−/301 | 0.6 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| Total ellagic acid and ETs | 124.5 ± 2.4 | 42.6 ± 1.7 | 42.7 ± 0.5 | ||
| 6.1 | Procyanidin B EC/EC 2 dimer 1 | 577−/289 | 0.7 ± 0.1 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 6.7 | Catechin | 289−/125 | 0.6 ± 0.1 | 0.1 ± 0.0 | 0.2 ± 0.0 |
| 7.8 | Procyanidin B EC/EC 2 dimer 2 | 577−/289 | 7.9 ± 0.6 | 1.8 ± 0.0 | 2.3 ± 0.1 |
| 8.3 | Procyanidin B EC/EC 2 dimer 3 | 577−/289 | 0.4 ± 0.0 | 0.1 ± 0.0 | 0.2 ± 0.0 |
| 8.4 | Proanthocyanidin EF/EC 2 dimer 1 | 561−/289 | 0.4 ± 0.0 | 0.1 ± 0.0 | 0.2 ± 0.0 |
| 9.4 | Epicatechin | 289−/125 | 9.0 ± 0.4 | 2.8 ± 0.1 | 2.6 ± 0.2 |
| 9.6 | Proanthocyanidin EF/EC/EC 2 trimer | 849−/289 | 0.6 ± 0.0 | 0.1 ± 0.0 | 0.2 ± 0.0 |
| 9.7 | Proanthocyanidin EF/EC 2 dimer 2 | 561−/289 | 0.2 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 10.4 | Proanthocyanidin EF/EF/EC 2 trimer 1 | 833−/289 | 0.2 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 12.1 | Proanthocyanidin EF/EF/EC 2 trimer 2 | 833−/289 | 1.1 ± 0.1 | 0.3 ± 0.0 | 0.4 ± 0.0 |
| Total flavan-3-ols | 21.1 ± 1.0 | 5.7 ± 0.2 | 6.5 ± 0.2 | ||
| 11.5 | Quercetin 3-O-galactosylglucoside | 625−/301 | 1.4 ± 0.1 | 0.3 ± 0.0 | 0.3 ± 0.0 |
| 11.8 | Quercetin 3-O-sophoroside | 625−/301 | 0.6 ± 0.0 | 0.3 ± 0.0 | 0.3 ± 0.0 |
| 12.9 | Quercetin 3-O-galactosylrhamnoside | 609−/301 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 13.9 | Quercetin 3-O-galactoside | 463−/301 | 0.4 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| 14.1 | Quercetin 3-O-glucosde | 463−/301 | 0.3 ± 0.0 | 0.2 ± 0.0 | 0.2 ± 0.0 |
| 14.2 | Quercetin 3-O-glucuronide | 477−/301 | 0.4 ± 0.0 | 0.5 ± 0.0 | 0.5 ± 0.0 |
| Total flavonols | 3.1 ± 0.1 | 1.5 ± 0.0 | 1.5 ± 0.0 | ||
| 2.4 | Gallic acid | 169−/125 | 0.7 ± 0.1 | 0.1 ± 0.0 | 0.2 + 0.0 |
| 4.1 | 3,4-Dihydroxybenzoic acid | 153−/109 | 0.1 ± 0.0 | 0.0 ± 0.0 | 0.1 ± 0.0 |
| 5.3 | Caffeoyl hexoside isomer 1 | 341−/179 | 1.3 ± 0.0 | 1.9 ± 0.0 | 1.7 ± 0.0 |
| 6.7 | Caffeoyl hexoside isomer 2 | 341−/179 | 0.4 ± 0.0 | 0.3 ± 0.0 | 0.3 ± 0.0 |
| 7.2 | p-Coumaryl hexoside | 325−/145 | 0.4 ± 0.0 | 0.3 ± 0.0 | 0.2 ± 0.0 |
| Total phenolic acids | 2.9 ± 0.1 | 2.6 ± 0.0 | 2.4 ± 0.0 | ||
| Total (poly)phenols | 388.4 ± 3.3 | 129.7 ± 1.9 | 129.8 ± 0.5 | ||
Sample Availability: Samples of the compounds are not available from the authors. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Sandhu, A.; Edirisinghe, I.; Burton-Freeman, B.M. Plasma and Urinary (Poly)phenolic Profiles after 4-Week Red Raspberry (Rubus idaeus L.) Intake with or without Fructo-Oligosaccharide Supplementation. Molecules 2020, 25, 4777. https://doi.org/10.3390/molecules25204777
Zhang X, Sandhu A, Edirisinghe I, Burton-Freeman BM. Plasma and Urinary (Poly)phenolic Profiles after 4-Week Red Raspberry (Rubus idaeus L.) Intake with or without Fructo-Oligosaccharide Supplementation. Molecules. 2020; 25(20):4777. https://doi.org/10.3390/molecules25204777
Chicago/Turabian StyleZhang, Xuhuiqun, Amandeep Sandhu, Indika Edirisinghe, and Britt M. Burton-Freeman. 2020. "Plasma and Urinary (Poly)phenolic Profiles after 4-Week Red Raspberry (Rubus idaeus L.) Intake with or without Fructo-Oligosaccharide Supplementation" Molecules 25, no. 20: 4777. https://doi.org/10.3390/molecules25204777
APA StyleZhang, X., Sandhu, A., Edirisinghe, I., & Burton-Freeman, B. M. (2020). Plasma and Urinary (Poly)phenolic Profiles after 4-Week Red Raspberry (Rubus idaeus L.) Intake with or without Fructo-Oligosaccharide Supplementation. Molecules, 25(20), 4777. https://doi.org/10.3390/molecules25204777







