Potential Beneficial Effects of Extra Virgin Olive Oils Characterized by High Content in Minor Polar Compounds in Nephropathic Patients: A Pilot Study
Abstract
1. Introduction
2. Results and Discussion
2.1. Chemical Characterization of EVOOs
2.2. Human Study
3. Materials and Methods
3.1. Reagents
3.2. EVOOs Production
3.3. Determination of Acidity, Peroxides, and Total Polyphenols
3.4. HPLC-DAD-MS Analyses of EVOOs
3.5. Patients and Methods
3.6. Questionnaires
3.7. Laboratory Parameters
3.8. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schwingshackl, L.; Morze, J.; Hoffmann, G. Mediterranean diet and health status: Active ingredients and pharmacological mechanisms. Br. J. Pharmacol. 2020, 177, 1241–1257. [Google Scholar] [CrossRef] [PubMed]
- Andreoli, A.; Lauro, S.; Di Daniele, N.; Sorge, R.; Celi, M.; Volpe, S.L. Effect of a moderately hypoenergetic Mediterranean diet and exercise program on body cell mass and cardiovascular risk factors in obese women. Eur. J. Clin. Nutr. 2008, 62, 892–897. [Google Scholar] [CrossRef] [PubMed]
- Romani, A.; Ieri, F.; Urciuoli, S.; Noce, A.; Marrone, G.; Nediani, C.; Bernini, R. Health Effects of Phenolic Compounds Found in Extra-Virgin Olive Oil, By-Products, and Leaf of Olea europaea L. Nutrients 2019, 11, 1776. [Google Scholar] [CrossRef] [PubMed]
- Franconi, F.; Campesi, I.; Romani, A. Is Extra Virgin Olive Oil an Ally for Women’s and Men’s Cardiovascular Health? Cardiovasc. Ther. 2020, 2020, 6719301. [Google Scholar] [CrossRef]
- Parkinson, L.; Cicerale, S. The Health Benefiting Mechanisms of Virgin Olive Oil Phenolic Compounds. Molecules 2016, 21, 1734. [Google Scholar] [CrossRef]
- Noce, A.; Marrone, G.; Di Daniele, F.; Ottaviani, E.; Wilson Jones, G.; Bernini, R.; Romani, A.; Rovella, V. Impact of Gut Microbiota Composition on Onset and Progression of Chronic Non-Communicable Diseases. Nutrients 2019, 11, 1073. [Google Scholar] [CrossRef]
- Campia, U.; Tesauro, M.; Di Daniele, N.; Cardillo, C. The vascular endothelin system in obesity and type 2 diabetes: Pathophysiology and therapeutic implications. Life Sci. 2014, 118, 149–155. [Google Scholar] [CrossRef]
- Romani, A.; Campo, M.; Urciuoli, S.; Marrone, G.; Noce, A.; Bernini, R. An Industrial and Sustainable Platform for the Production of Bioactive Micronized Powders and Extracts Enriched in Polyphenols From Olea europaea L. and Vitis vinifera L. Wastes. Front. Nutr. 2020, 7, 120. [Google Scholar] [CrossRef]
- Alessandri, S.; Ieri, F.; Romani, A. Minor polar compounds in extra virgin olive oil: Correlation between HPLC-DAD-MS and the Folin-Ciocalteu spectrophotometric method. J. Agric. Food Chem. 2014, 62, 826–835. [Google Scholar] [CrossRef]
- Karkovic Markovic, A.; Toric, J.; Barbaric, M.; Jakobusic Brala, C. Hydroxytyrosol, Tyrosol and Derivatives and Their Potential Effects on Human Health. Molecules 2019, 24, 2001. [Google Scholar] [CrossRef]
- Fuccelli, R.; Fabiani, R.; Rosignoli, P. Hydroxytyrosol Exerts Anti-Inflammatory and Anti-Oxidant Activities in a Mouse Model of Systemic Inflammation. Molecules 2018, 23, 3212. [Google Scholar] [CrossRef]
- Bernini, R.; Gilardini Montani, M.S.; Merendino, N.; Romani, A.; Velotti, F. Hydroxytyrosol-Derived Compounds: A Basis for the Creation of New Pharmacological Agents for Cancer Prevention and Therapy. J. Med. Chem. 2015, 58, 9089–9107. [Google Scholar] [CrossRef] [PubMed]
- Bernini, R.; Carastro, I.; Palmini, G.; Tanini, A.; Zonefrati, R.; Pinelli, P.; Brandi, M.L.; Romani, A. Lipophilization of Hydroxytyrosol-Enriched Fractions from Olea europaea L. Byproducts and Evaluation of the in Vitro Effects on a Model of Colorectal Cancer Cells. J. Agric. Food Chem 2017, 65, 6506–6512. [Google Scholar] [CrossRef] [PubMed]
- Benincasa, C.; La Torre, C.; Plastina, P.; Fazio, A.; Perri, E.; Caroleo, M.C.; Gallelli, L.; Cannataro, R.; Cione, E. Hydroxytyrosyl Oleate: Improved Extraction Procedure from Olive Oil and By-Products, and In Vitro Antioxidant and Skin Regenerative Properties. Antioxidants 2019, 8, 233. [Google Scholar] [CrossRef]
- Ahmad Farooqi, A.; Fayyaz, S.; Silva, A.S.; Sureda, A.; Nabavi, S.F.; Mocan, A.; Nabavi, S.M.; Bishayee, A. Oleuropein and Cancer Chemoprevention: The Link is Hot. Molecules 2017, 22, 705. [Google Scholar] [CrossRef] [PubMed]
- Nediani, C.; Ruzzolini, J.; Romani, A.; Calorini, L. Oleuropein, a Bioactive Compound from Olea europaea L. as a Potential Preventive and Therapeutic Agent in Non-Communicable Diseases. Antioxidants 2019, 8, 578. [Google Scholar] [CrossRef]
- Bernini, R.; Gualandi, G.; Crestini, C.; Barontini, M.; Belfiore, M.C.; Willfor, S.; Eklund, P.; Saladino, R. A novel and efficient synthesis of highly oxidized lignans by a methyltrioxorhenium/hydrogen peroxide catalytic system. Studies on their apoptogenic and antioxidant activity. Bioorg. Med. Chem. 2009, 17, 5676–5682. [Google Scholar] [CrossRef]
- D’Andrea, G.; Ceccarelli, M.; Bernini, R.; Clemente, M.; Santi, L.; Caruso, C.; Micheli, L.; Tirone, F. Hydroxytyrosol stimulates neurogenesis in aged dentate gyrus by enhancing stem and progenitor cell proliferation and neuron survival. FASEB J. 2020, 34, 4512–4526. [Google Scholar] [CrossRef]
- EFSA. Scientific Opinion on the Substantiation of Health Claims Related to Polyphenols in Olive and Protection of LDL Particles from Oxidative Damage (ID 1333, 1638, 1639, 1696, 2865), Maintenance of Normal Blood HDL Cholesterol Concentrations (ID 1639), Maintenance of Normal Blood Pressure (ID 3781), “Anti-Inflammatory Properties” (ID 1882), “Contributes to the Upper Respiratory Tract Health” (ID 3468), “Can Help to Maintain a Normal Function of Gastrointestinal Tract” (3779), and “Contributes to Body Defences against External Agents” (ID 3467) Pursuant to Article 13(1) of Regulation (EC) No 1924/2006. Available online: https://www.efsa.europa.eu/en/efsajournal/pub/2033 (accessed on 5 August 2020).
- Caporaso, N.; Savarese, M.; Paduano, A.; Guidone, G.; De Marco, E.; Sacchi, R. Nutritional quality assessment of extra virgin olive oil from the Italian retail market: Do natural antioxidants satisfy EFSA health claims? J. Food Compos. Anal. 2015, 40, 154–162. [Google Scholar] [CrossRef]
- Salvini, S.; Sera, F.; Caruso, D.; Giovannelli, L.; Visioli, F.; Saieva, C.; Masala, G.; Ceroti, M.; Giovacchini, V.; Pitozzi, V.; et al. Daily consumption of a high-phenol extra-virgin olive oil reduces oxidative DNA damage in postmenopausal women. Br. J. Nutr. 2006, 95, 742–751. [Google Scholar] [CrossRef]
- Manna, C.; D’Angelo, S.; Migliardi, V.; Loffredi, E.; Mazzoni, O.; Morrica, P.; Galletti, P.; Zappia, V. Protective effect of the phenolic fraction from virgin olive oils against oxidative stress in human cells. J. Agric. Food Chem. 2002, 50, 6521–6526. [Google Scholar] [CrossRef] [PubMed]
- Beauchamp, G.K.; Keast, R.S.; Morel, D.; Lin, J.; Pika, J.; Han, Q.; Lee, C.H.; Smith, A.B.; Breslin, P.A. Phytochemistry: Ibuprofen-like activity in extra-virgin olive oil. Nature 2005, 437, 45–46. [Google Scholar] [CrossRef] [PubMed]
- Kamvissis, V.N.; Barbounis, E.G.; Megoulas, E.G.; Koupparis, M.A. A novel photometric method for evaluation of the oxidative stability of virgin olive oils. J. AOAC Int. 2008, 91, 794–801. [Google Scholar] [CrossRef]
- Noce, A.; Fabrini, R.; Dessi, M.; Bocedi, A.; Santini, S.; Rovella, V.; Pastore, A.; Tesauro, M.; Bernardini, S.; Di Daniele, N.; et al. Erythrocyte glutathione transferase activity: A possible early biomarker for blood toxicity in uremic diabetic patients. Acta Diabetol. 2014, 51, 219–224. [Google Scholar] [CrossRef]
- Dessi, M.; Noce, A.; Bertucci, P.; Noce, G.; Rizza, S.; De Stefano, A.; Manca di Villahermosa, S.; Bernardini, S.; De Lorenzo, A.; Di Daniele, N. Plasma and erythrocyte membrane phospholipids and fatty acids in Italian general population and hemodialysis patients. Lipids Health Dis. 2014, 13, 54. [Google Scholar] [CrossRef]
- Daenen, K.; Andries, A.; Mekahli, D.; Van Schepdael, A.; Jouret, F.; Bammens, B. Oxidative stress in chronic kidney disease. Pediatr. Nephrol. 2019, 34, 975–991. [Google Scholar] [CrossRef]
- Noce, A.; Bocedi, A.; Campo, M.; Marrone, G.; Di Lauro, M.; Cattani, G.; Di Daniele, N.; Romani, A. A Pilot Study of a Natural Food Supplement as New Possible Therapeutic Approach in Chronic Kidney Disease Patients. Pharmaceuticals 2020, 13, 148. [Google Scholar] [CrossRef] [PubMed]
- Bocedi, A.; Noce, A.; Rovella, V.; Marrone, G.; Cattani, G.; Iappelli, M.; De Paolis, P.; Iaria, G.; Sforza, D.; Gallu, M.; et al. Erythrocyte glutathione transferase in kidney transplantation: A probe for kidney detoxification efficiency. Cell Death Dis. 2018, 9, 288. [Google Scholar] [CrossRef]
- Noce, A.; Marrone, G.; Di Lauro, M.; Urciuoli, S.; Pietroboni Zaitseva, A.; Wilson Jones, G.; Di Daniele, N.; Romani, A. Cardiovascular Protection of Nephropathic Male Patients by Oral Food Supplements. Cardiovasc. Ther. 2020, 2020, 1807941. [Google Scholar] [CrossRef]
- Presti, G.; Guarrasi, V.; Gulotta, E.; Provenzano, F.; Provenzano, A.; Giuliano, S.; Monfreda, M.; Mangione, M.R.; Passantino, R.; San Biagio, P.L.; et al. Bioactive compounds from extra virgin olive oils: Correlation between phenolic content and oxidative stress cell protection. Biophys. Chem. 2017, 230, 109–116. [Google Scholar] [CrossRef]
- Rossi, M.; Caruso, F.; Kwok, L.; Lee, G.; Caruso, A.; Gionfra, F.; Candelotti, E.; Belli, S.L.; Molasky, N.; Raley-Susman, K.M.; et al. Protection by extra virgin olive oil against oxidative stress in vitro and in vivo. Chemical and biological studies on the health benefits due to a major component of the Mediterranean diet. PLoS ONE 2017, 12, e0189341. [Google Scholar] [CrossRef] [PubMed]
- Bocedi, A.; Noce, A.; Marrone, G.; Noce, G.; Cattani, G.; Gambardella, G.; Di Lauro, M.; Di Daniele, N.; Ricci, G. Glutathione Transferase P1-1 an Enzyme Useful in Biomedicine and as Biomarker in Clinical Practice and in Environmental Pollution. Nutrients 2019, 11, 1741. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.W.; Lin, S.Y.; Kuo, C.C.; Huang, C.C. Serum Uric Acid and Progression of Kidney Disease: A Longitudinal Analysis and Mini-Review. PLoS ONE 2017, 12, e0170393. [Google Scholar] [CrossRef]
- Siu, Y.P.; Leung, K.T.; Tong, M.K.; Kwan, T.H. Use of allopurinol in slowing the progression of renal disease through its ability to lower serum uric acid level. Am. J. Kidney Dis. 2006, 47, 51–59. [Google Scholar] [CrossRef]
- Goicoechea, M.; Garcia de Vinuesa, S.; Verdalles, U.; Verde, E.; Macias, N.; Santos, A.; Perez de Jose, A.; Cedeno, S.; Linares, T.; Luno, J. Allopurinol and progression of CKD and cardiovascular events: Long-term follow-up of a randomized clinical trial. Am. J. Kidney Dis. 2015, 65, 543–549. [Google Scholar] [CrossRef] [PubMed]
- De Angelis, S.; Noce, A.; Di Renzo, L.; Cianci, R.; Naticchia, A.; Giarrizzo, G.F.; Giordano, F.; Tozzo, C.; Splendiani, G.; De Lorenzo, A. Is rasburicase an effective alternative to allopurinol for management of hyperuricemia in renal failure patients? A double blind-randomized study. Eur. Rev. Med. Pharmacol. Sci. 2007, 11, 179–184. [Google Scholar] [PubMed]
- Covas, M.I.; Nyyssonen, K.; Poulsen, H.E.; Kaikkonen, J.; Zunft, H.J.; Kiesewetter, H.; Gaddi, A.; de la Torre, R.; Mursu, J.; Baumler, H.; et al. The effect of polyphenols in olive oil on heart disease risk factors: A randomized trial. Ann. Intern. Med. 2006, 145, 333–341. [Google Scholar] [CrossRef]
- Blanco-Molina, A.; Castro, G.; Martin-Escalante, D.; Bravo, D.; Lopez-Miranda, J.; Castro, P.; Lopez-Segura, F.; Fruchart, J.C.; Ordovas, J.M.; Perez-Jimenez, F. Effects of different dietary cholesterol concentrations on lipoprotein plasma concentrations and on cholesterol efflux from Fu5AH cells. Am. J. Clin. Nutr. 1998, 68, 1028–1033. [Google Scholar] [CrossRef]
- Carluccio, M.A.; Massaro, M.; Scoditti, E.; De Caterina, R. Vasculoprotective potential of olive oil components. Mol. Nutr. Food Res. 2007, 51, 1225–1234. [Google Scholar] [CrossRef]
- Oliveras-Lopez, M.J.; Molina, J.J.; Mir, M.V.; Rey, E.F.; Martin, F.; de la Serrana, H.L. Extra virgin olive oil (EVOO) consumption and antioxidant status in healthy institutionalized elderly humans. Arch. Gerontol. Geriatr. 2013, 57, 234–242. [Google Scholar] [CrossRef]
- Machowetz, A.; Gruendel, S.; Garcia, A.L.; Harsch, I.; Covas, M.I.; Zunft, H.J.; Koebnick, C. Effect of olive oil consumption on serum resistin concentrations in healthy men. Horm. Metab. Res. 2008, 40, 697–701. [Google Scholar] [CrossRef]
- Bernini, R.; Mincione, E.; Barontini, M.; Crisante, F. Convenient synthesis of hydroxytyrosol and its lipophilic derivatives from tyrosol or homovanillyl alcohol. J. Agric. Food Chem. 2008, 56, 8897–8904. [Google Scholar] [CrossRef] [PubMed]
- Romani, A.; Lapucci, C.; Cantini, C.; Ieri, F.; Mulinacci, N.; Visioli, F. Evolution of minor polar compounds and antioxidant capacity during storage of bottled extra virgin olive oil. J. Agric. Food Chem. 2007, 55, 1315–1320. [Google Scholar] [CrossRef] [PubMed]
- Kopple, J.D. National kidney foundation K/DOQI clinical practice guidelines for nutrition in chronic renal failure. Am. J. Kidney Dis. 2001, 37, S66–S70. [Google Scholar] [CrossRef]
- Cupisti, A.; Brunori, G.; Di Iorio, B.R.; D’Alessandro, C.; Pasticci, F.; Cosola, C.; Bellizzi, V.; Bolasco, P.; Capitanini, A.; Fantuzzi, A.L.; et al. Nutritional treatment of advanced CKD: Twenty consensus statements. J. Nephrol. 2018, 31, 457–473. [Google Scholar] [CrossRef]
- Noce, A.; Vidiri, M.F.; Marrone, G.; Moriconi, E.; Bocedi, A.; Capria, A.; Rovella, V.; Ricci, G.; De Lorenzo, A.; Di Daniele, N. Is low-protein diet a possible risk factor of malnutrition in chronic kidney disease patients? Cell Death Discov. 2016, 2, 16026. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, M.A.; Garcia-Arellano, A.; Toledo, E.; Salas-Salvado, J.; Buil-Cosiales, P.; Corella, D.; Covas, M.I.; Schroder, H.; Aros, F.; Gomez-Gracia, E.; et al. A 14-item Mediterranean diet assessment tool and obesity indexes among high-risk subjects: The PREDIMED trial. PLoS ONE 2012, 7, e43134. [Google Scholar] [CrossRef]
- Wanner, M.; Probst-Hensch, N.; Kriemler, S.; Meier, F.; Autenrieth, C.; Martin, B.W. Validation of the long international physical activity questionnaire: Influence of age and language region. Prev. Med. Rep. 2016, 3, 250–256. [Google Scholar] [CrossRef]
- Cesarone, M.R.; Belcaro, G.; Carratelli, M.; Cornelli, U.; De Sanctis, M.T.; Incandela, L.; Barsotti, A.; Terranova, R.; Nicolaides, A. A simple test to monitor oxidative stress. Int. Angiol. 1999, 18, 127–130. [Google Scholar] [PubMed]
- Lewis, N.A.; Newell, J.; Burden, R.; Howatson, G.; Pedlar, C.R. Critical Difference and Biological Variation in Biomarkers of Oxidative Stress and Nutritional Status in Athletes. PLoS ONE 2016, 11, e0149927. [Google Scholar] [CrossRef]
- Mihai, S.; Codrici, E.; Popescu, I.D.; Enciu, A.M.; Albulescu, L.; Necula, L.G.; Mambet, C.; Anton, G.; Tanase, C. Inflammation-Related Mechanisms in Chronic Kidney Disease Prediction, Progression, and Outcome. J. Immunol. Res. 2018, 2018, 2180373. [Google Scholar] [CrossRef] [PubMed]
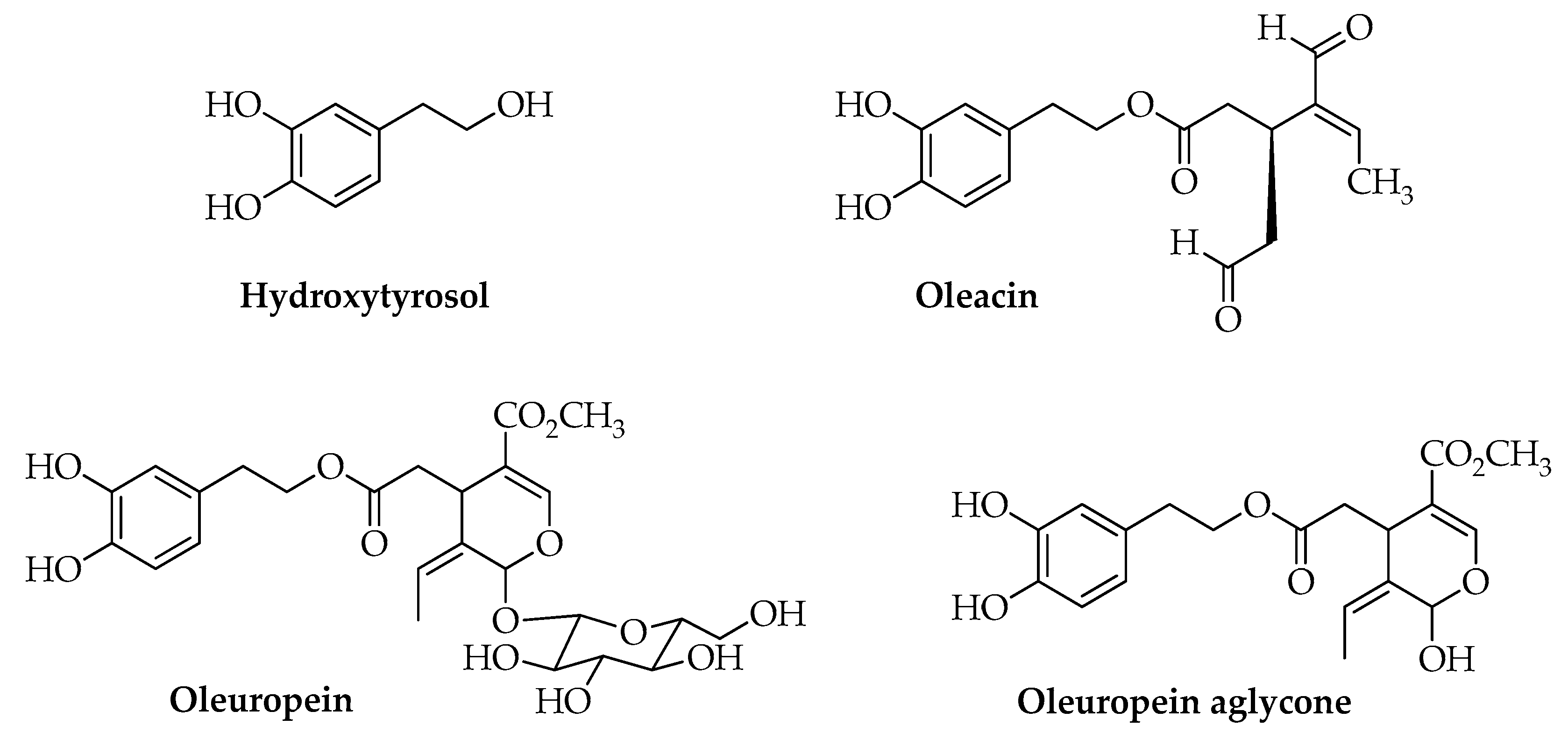
| Sample | Cultivar (%, v/v) |
|---|---|
| FR | Frantoio (100) |
| LE | Leccino (100) |
| MO | Moraiolo (100) |
| MOS | Moraiolo (100) |
| IN | Intosso (100) |
| TB | Leccino (33.3), Moraiolo (33.3), Frantoio (33.3) |
| TAB | Leccino (95), Intosso (5) |
| Acidity (% oleic acid) | Peroxides (meqO2/Kg) | Polyphenols (mg tyrosol/Kg) | |
|---|---|---|---|
| FR | 0.25 | 6.67 | 545 |
| LE | 0.15 | 5.21 | 791 |
| MO | 0.24 | 5.80 | 717 |
| MOS | 0.15 | 7.81 | 483 |
| IN | 0.15 | 4.49 | 400 |
| TB | 0.16 | 8.80 | 342 |
| TAB | 0.17 | 4.98 | 890 |
| Compound | FR | LE | MO | MOS | IN | TB | TAB |
|---|---|---|---|---|---|---|---|
| mg/L * | |||||||
| Hydroxytyrosol | 0.80 | nd | 7.04 | 1.88 | 2.06 | 1.47 | 3.10 |
| Tyrosol | 0.98 | nd | 4.00 | 1.87 | 3.76 | 1.86 | 1.02 |
| Elenolic acid | 198.98 | 31.54 | 129.71 | 196.78 | 101.41 | 116.73 | 150.06 |
| Elenolic acid derivatives | 31.40 | 60.80 | 22.24 | 29.32 | 5.80 | 21.20 | 9.32 |
| Oleacin | 154.81 | 361.89 | 45.51 | 123.58 | 54.06 | 67.47 | 315.46 |
| Oleocanthal | 44.37 | 192.42 | 40.77 | 44.03 | 54.91 | 94.12 | 197.84 |
| Oleuropein aglycone | 84.14 | 67.70 | 143.12 | 143.46 | 64.75 | 83.42 | 164.58 |
| Secoiridoids derivatives | 40.33 | 17.11 | 89.55 | 36.43 | 36.54 | 48.39 | 96.43 |
| Lignans | 99.09 | 160.25 | 205.02 | 129.48 | 94.67 | 62.14 | 208.17 |
| Total MCP | 654.90 | 891.71 | 686.96 | 706.83 | 417.96 | 496.80 | 1145.98 |
| Total phenolic compounds | 424.52 | 799.37 | 535.01 | 480.73 | 310.75 | 358.87 | 986.60 |
| Total hydroxytyrosol and derivatives | 325.43 | 639.12 | 329.99 | 351.25 | 216.08 | 296.73 | 778.43 |
| MOS | TB | p (ANOVA Test) | |
|---|---|---|---|
| N | 14 | 13 | |
| Gender (male/female) | 4/10 | 4/9 | ns |
| Age (years) | 70.8 ± 12.4 a | 65.9 ± 11.4 a | ns |
| Height (m) | 1.65 ± 0.11 a | 1.66 ± 0.12 a | ns |
| Weight (kg) | 78.9 ± 13.9 a | 75.6 ± 15.6 a | ns |
| BMI (kg/m2) | 28.83 ± 3.86 a | 27.43 ± 5.81 a | ns |
| MOS | TB | |||||
|---|---|---|---|---|---|---|
| T0 | T1 | T0 vs. T1 | T0 | T1 | T0 vs. T1 | |
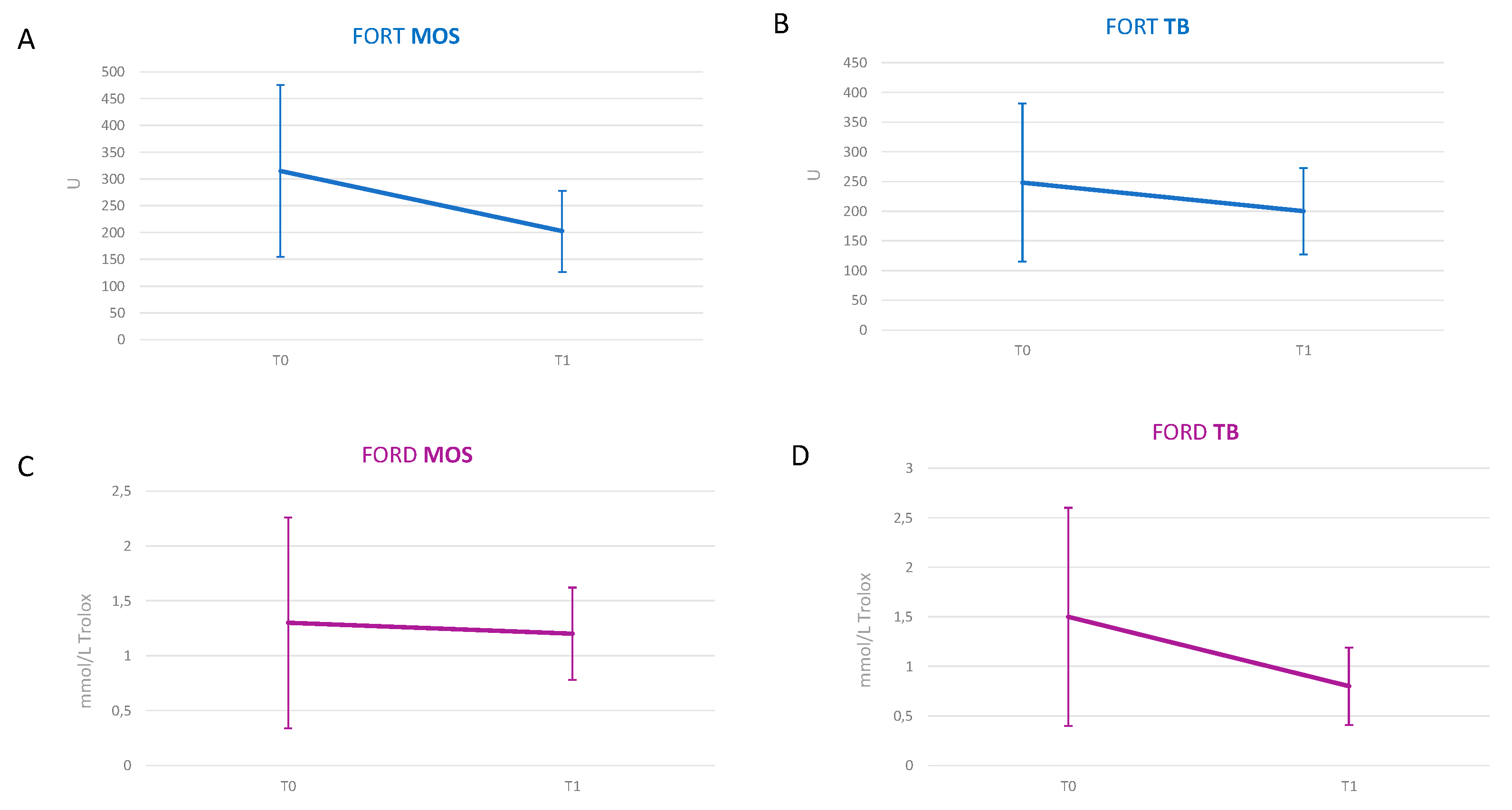
| Creatinine (mg/dL) | 2.04 ± 0.68 a | 1.9 ± 0.68 a | ns b | 2.22 ± 1.26 a | 2.17 ± 1.26 a | ns b |
| e-GFR (mL/min/1.72 m2) | 35.4 ± 16.32 a | 38.1 ± 16.69 a | 0.04 b | 37 ± 20.84 a | 39.11 ± 22.95 a | ns b |
| Albuminuria (mg/dL) | 9.2 ± 16.2 a | 20.4 ± 37.8 a | ns b | 25 ± 63.7 a | 6 ± 10.49 a | ns b |
| Albumin (g/dL) | 4.17 ± 0.26 a | 4.31 ± 0.27 a | 0.021 b | 4.12 ± 0.25 a | 4.39 ± 0.38 a | 0.032 b |
| Azotemia (mg/dL) | 69.5 ± 24.2 a | 61.8 ± 20.6 a | ns b | 61.09 ± 18.41 a | 63.9 ± 24.13 a | ns b |
| Sodium (mEq/L) | 139.8 ± 3.6 a | 139.9 ± 2.0 a | ns b | 141.64 ± 2.06 a | 140.09 ± 2.74 a | ns b |
| Potassium (mEq/L) | 4.41 ± 0.56 a | 4.35 ± 0.61 a | ns b | 4.44 ± 0.43 a | 4.31 ± 0.4 a | ns b |
| Calcium (mg/dL) | 9.92 ± 0.43 a | 9.72 ± 0.38 a | ns b | 9.85 ± 0.46 a | 9.7 ± 0.47 a | ns b |
| Phosphorus (mg/dL) | 3.39 ± 0.49 a | 3.27 ± 0.54 a | ns b | 3.61 ± 0.61 a | 3.53 ± 0.67 a | ns b |
| TC (mg/dL) | 176.58 ± 44.17 a | 171.9 ± 38.0 a | ns b | 173.64 ± 59.61 a | 180.73 ± 52.4 a | ns b |
| HDL-C (mg/dL) | 40.08 ± 9.59 a | 56.4 ± 37.1 a | ns b | 45 ± 11.79 a | 48.45 ± 12.88 a | ns b |
| LDL-C (mg/dL) | 102.25 ± 31.85 a | 94.5 ± 34.09 a | ns b | 109.9 ± 51.9 a | 100.3 ± 41.6 a | ns b |
| Triglycerides (mg/dL) | 165.75 ± 83.18 a | 145.08 ± 70.9 a | 0.016 b | 114.5 ± 69.6 a | 117.8 ± 59.8 a | ns b |
| Sideremia (μg/dL) | 89.08 ± 36.47 a | 128 ± 170.0 a | ns b | 76.27 ± 23.48 a | 71.55 ± 20.71 a | ns b |
| Glycaemia (mg/dL) | 96.6 ± 25 a | 101.0 ± 36.2 a | ns b | 94.09 ± 21.54 a | 90.36 ± 13.8 a | ns b |
| Uric acid (mg/dL) | 6.36 ± 1.9 a | 5.0 ± 1.2 a | 0.049 b | 6.23 ± 1.63 a | 6.1 ± 0.86 a | ns b |
| PTH (pg/mL) | 83.7 ± 42.7 a | 82.56 ± 45.9 a | ns b | 81.08 ± 60.65 a | 72.38 ± 29.1 a | ns b |
| CRP (mg/L) | 4.48 ± 1.2 a | 3.61 + 0.8 a | ns b | 2.92 + 0.98 a | 2.77 + 1.3 a | ns b |
| ESR (mm/h) | 34.91 ± 23.65 a | 32.36 ± 27.75 a | ns b | 40.28 ± 30.1 a | 31 ± 28.65 a | ns b |
| MOS | TB | |||||
|---|---|---|---|---|---|---|
| T0 | T1 | p (McNemar’s Test) | T0 | T1 | p (McNemar’s Test) | |
| Minimal adherence (%) | 0 (0) | 2 (15.3) | ns | 1 (7.7) | 0 (0) | ns |
| Average adherence (%) | 9 (64.3) | 7 (54) | ns | 8 (61.6) | 9 (64.3) | ns |
| Maximal adherence (%) | 5 (35.7) | 4 (30.7) | ns | 4 (30.7) | 5 (35.7) | ns |
| MOS | TB | |||||
|---|---|---|---|---|---|---|
| T0 | T1 | p (McNemar’s Test) | T0 | T1 | p (McNemar’s Test) | |
| Inactive (%) | 5 (35.7) | 9 (69.4) | ns | 8 (61.5) | 5 (35.7) | ns |
| Sufficiently active (%) | 6 (35.7) | 2 (15.3) | ns | 3 (23.2) | 6 (35.7) | ns |
| Very active (%) | 3 (21.5) | 2 (15.3) | ns | 2 (15.3) | 3 (21.5) | ns |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romani, A.; Bernini, R.; Noce, A.; Urciuoli, S.; Di Lauro, M.; Pietroboni Zaitseva, A.; Marrone, G.; Di Daniele, N. Potential Beneficial Effects of Extra Virgin Olive Oils Characterized by High Content in Minor Polar Compounds in Nephropathic Patients: A Pilot Study. Molecules 2020, 25, 4757. https://doi.org/10.3390/molecules25204757
Romani A, Bernini R, Noce A, Urciuoli S, Di Lauro M, Pietroboni Zaitseva A, Marrone G, Di Daniele N. Potential Beneficial Effects of Extra Virgin Olive Oils Characterized by High Content in Minor Polar Compounds in Nephropathic Patients: A Pilot Study. Molecules. 2020; 25(20):4757. https://doi.org/10.3390/molecules25204757
Chicago/Turabian StyleRomani, Annalisa, Roberta Bernini, Annalisa Noce, Silvia Urciuoli, Manuela Di Lauro, Anna Pietroboni Zaitseva, Giulia Marrone, and Nicola Di Daniele. 2020. "Potential Beneficial Effects of Extra Virgin Olive Oils Characterized by High Content in Minor Polar Compounds in Nephropathic Patients: A Pilot Study" Molecules 25, no. 20: 4757. https://doi.org/10.3390/molecules25204757
APA StyleRomani, A., Bernini, R., Noce, A., Urciuoli, S., Di Lauro, M., Pietroboni Zaitseva, A., Marrone, G., & Di Daniele, N. (2020). Potential Beneficial Effects of Extra Virgin Olive Oils Characterized by High Content in Minor Polar Compounds in Nephropathic Patients: A Pilot Study. Molecules, 25(20), 4757. https://doi.org/10.3390/molecules25204757






