Participation of MicroRNAs in the Treatment of Cancer with Phytochemicals
Abstract
1. Introduction
2. Oncogenic MiRNAs Inhibited by Phytochemicals Currently Evaluated in Preclinical Studies and Clinical Trials
2.1. MiRNAs and Nitrogen-Containing Compounds
2.1.1. Berberine and Evodiamine
2.1.2. Matrine
2.1.3. Neferine
2.1.4. Nitidine Chloride and Swainsonine
2.1.5. Piperlongumine
2.1.6. Sanguinarine
2.1.7. Sinomenine
2.1.8. Sophocarpine
2.2. MiRNAs and an Organosulfur Compound
Sulforaphane
2.3. MiRNAs and Phenolic Compounds
2.3.1. Baicalin
2.3.2. Curcumin
2.3.3. Epigallocatechin Gallate
2.3.4. Formononetin, Galangin, Gambogic Acid, Honokiol, and Puerarin
2.3.5. Genistein
2.3.6. Icariin, Isoliquiritigenin, and Luteolin
2.3.7. Physcion 8-O-β-Glucopyranoside and Procyanidin
2.3.8. Resveratrol
2.3.9. Silibinin
2.4. MiRNAs and Terpenoid Compounds
2.4.1. Brucein D
2.4.2. Celastrol
2.4.3. Ginsenosides
2.4.4. Maytenin, 22-β-Hydroxymaytenin, and Oridonin
2.4.5. Triptolide
2.4.6. α-Pinene
3. Tumor-Suppressive MiRNAs Induced by Phytochemicals Currently Tested in Preclinical Studies and Clinical Trials
3.1. MiRNAs and Nitrogen-Containing Compounds
3.1.1. 5-Aminolevulinic acid
3.1.2. Coptisine
3.1.3. Indole-3-Carbinol
3.1.4. Matrine
3.1.5. Sanguinarine
3.2. MiRNAs, Organosulfur, and Phytosterol Compounds
3.2.1. Allicin
3.2.2. Sulforaphane
3.2.3. β-Sitosterol-d-Glucoside
3.3. MiRNAs and Phenolic Compounds
3.3.1. Apigenin, Astragalin, and Baicalein
3.3.2. Brazilein
3.3.3. Chrysin, Curcumin, and Genistein
3.3.4. Delphinidin, Epigallocatechin Gallate, and Gossypol
3.3.5. Hydroxygenkwanin, Isorhapontigenin, and Kaempferol
3.3.6. Licochalcone A and Luteolin
3.3.7. Physcion 8-O-β-Glucopyranoside and Quercetin
3.3.8. Resveratrol and Trans-3,5,4′-trimethoxystilbene
3.3.9. Silymarin
3.4. MiRNAs and Terpenoid Compounds
3.4.1. Ailanthone and Andrographolide
3.4.2. Artemisinin and Artesunate
3.4.3. Astragaloside IV and Cannabidiol
3.4.4. Cucurbitacin D, Curcumol, and Lycopene
3.4.5. Oleanolic Acid and Pristimerin
3.4.6. Toosendanin and Triptolide
3.4.7. Tubeimoside-1 and Ursolic Acid
4. MiRNAs Affecting the Anti-Cancer Activity of Phytochemicals Currently Tested in Preclinical Studies and Clinical Trials
4.1. 1′S-1′-Acetoxychavicol Acetate
4.2. Apigenin
4.3. Delphinidin
4.4. Epigallocatechin Gallate
4.5. Luteolin and Silibinin
4.6. Oridonin
4.7. Physcion 8-O-β-Glucopyranoside
4.8. Shikonin
5. MiRNAs Regulating the Sensitivity of Cancer Cells to Phytochemicals Currently Used in Cancer Therapy
5.1. Etoposide
5.1.1. MiRNAs Regulating Apoptosis and Autophagy
5.1.2. MiRNAs Regulating EMT and Wnt/β-Catenin Signaling
5.1.3. MiRNAs Regulating DNA Damage Repair
5.1.4. A miRNA Regulating a Drug Transporter Level
5.2. Irinotecan
5.2.1. MiR-200
5.2.2. MiR-514b-5p
5.2.3. MiR-627
5.2.4. MiR-4454
5.3. Paclitaxel
5.3.1. MiRNAs Negatively Regulating Apoptosis
5.3.2. MiRNAs Positively Regulating Apoptosis
5.3.3. MiRNAs Inhibiting EMT- and Stemness-Related Factors
5.3.4. MiRNAs Regulating the Level of Drug Transporters
5.4. Vincristine
5.4.1. MiRNAs Regulating Apoptosis and Autophagy
5.4.2. MiRNAs Inhibiting EMT- and Stemness-Related Factors
5.4.3. MiRNAs Regulating the Intracellular Concentration of Vincristine
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Mathers, C.; Parkin, D.M.; Pineros, M.; Znaor, A.; Bray, F. Estimating the global cancer incidence and mortality in 2018: Globocan sources and methods. Int. J. Cancer 2019, 144, 1941–1953. [Google Scholar] [CrossRef]
- Saltalamacchia, G.; Frascaroli, M.; Bernardo, A.; Quaquarini, E. Renal and cardiovascular toxicities by new systemic treatments for prostate cancer. Cancers 2020, 12, 1750. [Google Scholar] [CrossRef] [PubMed]
- Nurgali, K.; Jagoe, R.T.; Abalo, R. Editorial: Adverse effects of cancer chemotherapy: Anything new to improve tolerance and reduce sequelae? Front. Pharmacol. 2018, 9, 245. [Google Scholar] [CrossRef] [PubMed]
- Seo, H.A.; Moeng, S.; Sim, S.; Kuh, H.J.; Choi, S.Y.; Park, J.K. Microrna-based combinatorial cancer therapy: Effects of micrornas on the efficacy of anti-cancer therapies. Cells 2019, 9, 29. [Google Scholar] [CrossRef] [PubMed]
- Lichota, A.; Gwozdzinski, K. Anticancer activity of natural compounds from plant and marine environment. Int. J. Mol. Sci. 2018, 19, 3533. [Google Scholar] [CrossRef]
- Starczewska, E.; Beyaert, M.; Michaux, L.; Vekemans, M.C.; Saussoy, P.; Bol, V.; Arana Echarri, A.; Smal, C.; Van Den Neste, E.; Bontemps, F. Targeting DNA repair with aphidicolin sensitizes primary chronic lymphocytic leukemia cells to purine analogs. Oncotarget 2016, 7, 38367–38379. [Google Scholar] [CrossRef]
- Montane, X.; Kowalczyk, O.; Reig-Vano, B.; Bajek, A.; Roszkowski, K.; Tomczyk, R.; Pawliszak, W.; Giamberini, M.; Mocek-Plociniak, A.; Tylkowski, B. Current perspectives of the applications of polyphenols and flavonoids in cancer therapy. Molecules 2020, 25, 3342. [Google Scholar] [CrossRef]
- Mitra, T.; Bhattacharya, R. Phytochemicals modulate cancer aggressiveness: A review depicting the anticancer efficacy of dietary polyphenols and their combinations. J. Cell. Physiol. 2020, 235, 7696–7708. [Google Scholar] [CrossRef]
- Li, F.; Jiang, T.; Li, Q.; Ling, X. Camptothecin (cpt) and its derivatives are known to target topoisomerase i (top1) as their mechanism of action: Did we miss something in cpt analogue molecular targets for treating human disease such as cancer? Am. J. Cancer Res. 2017, 7, 2350–2394. [Google Scholar]
- Choudhari, A.S.; Mandave, P.C.; Deshpande, M.; Ranjekar, P.; Prakash, O. Phytochemicals in cancer treatment: From preclinical studies to clinical practice. Front. Pharmacol. 2019, 10, 1614. [Google Scholar] [CrossRef]
- Bergman, M.E.; Davis, B.; Phillips, M.A. Medically useful plant terpenoids: Biosynthesis, occurrence, and mechanism of action. Molecules 2019, 24, 3961. [Google Scholar] [CrossRef]
- Chen, F.; Zhu, H.H.; Zhou, L.F.; Wu, S.S.; Wang, J.; Chen, Z. Inhibition of c-flip expression by mir-512-3p contributes to taxol-induced apoptosis in hepatocellular carcinoma cells. Oncol. Rep. 2010, 23, 1457–1462. [Google Scholar] [CrossRef]
- Park, J.K.; Doseff, A.I.; Schmittgen, T.D. Micrornas targeting caspase-3 and -7 in panc-1 cells. Int. J. Mol. Sci. 2018, 19, 1206. [Google Scholar] [CrossRef]
- Moeng, S.; Son, S.W.; Lee, J.S.; Lee, H.Y.; Kim, T.H.; Choi, S.Y.; Kuh, H.J.; Park, J.K. Extracellular vesicles (evs) and pancreatic cancer: From the role of evs to the interference with ev-mediated reciprocal communication. Biomedicines 2020, 8, 267. [Google Scholar] [CrossRef]
- Sun, M.; Zhang, Q.; Yang, X.; Qian, S.Y.; Guo, B. Vitamin d enhances the efficacy of irinotecan through mir-627-mediated inhibition of intratumoral drug metabolism. Mol. Cancer Ther. 2016, 15, 2086–2095. [Google Scholar] [CrossRef]
- Wu, Y.; Jin, X.; Liao, W.; Hu, L.; Dawuda, M.M.; Zhao, X.; Tang, Z.; Gong, T.; Yu, J. 5-aminolevulinic acid (ala) alleviated salinity stress in cucumber seedlings by enhancing chlorophyll synthesis pathway. Front. Plant Sci. 2018, 9, 635. [Google Scholar] [CrossRef]
- Feng, M.; Luo, X.; Gu, C.; Li, Y.; Zhu, X.; Fei, J. Systematic analysis of berberine-induced signaling pathway between mirna clusters and mrnas and identification of mir-99a approximately 125b cluster function by seed-targeting inhibitors in multiple myeloma cells. RNA Biol. 2015, 12, 82–91. [Google Scholar] [CrossRef]
- Chai, F.N.; Ma, W.Y.; Zhang, J.; Xu, H.S.; Li, Y.F.; Zhou, Q.D.; Li, X.G.; Ye, X.L. Coptisine from rhizoma coptidis exerts an anti-cancer effect on hepatocellular carcinoma by up-regulating mir-122. Biomed. Pharmacother. 2018, 103, 1002–1011. [Google Scholar] [CrossRef]
- Jiang, J.; Hu, C. Evodiamine: A novel anti-cancer alkaloid from evodia rutaecarpa. Molecules 2009, 14, 1852–1859. [Google Scholar] [CrossRef]
- Hargraves, K.G.; He, L.; Firestone, G.L. Phytochemical regulation of the tumor suppressive microrna, mir-34a, by p53-dependent and independent responses in human breast cancer cells. Mol. Carcinog. 2016, 55, 486–498. [Google Scholar] [CrossRef]
- Fu, S.; Zhao, N.; Jing, G.; Yang, X.; Liu, J.; Zhen, D.; Tang, X. Matrine induces papillary thyroid cancer cell apoptosis in vitro and suppresses tumor growth in vivo by downregulating mir-182-5p. Biomed. Pharmacother. 2020, 128, 110327. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, S.; Wang, T.; Shao, H.; Gao, J.; Wang, Y.; Ge, Y. Neferine inhibits mda-mb-231cells growth and metastasis by regulating mir-374a/fgfr-2. Chem. Biol. Interact 2019, 309, 108716. [Google Scholar] [CrossRef]
- Mao, X.; Hu, Z.; Wang, Q.; Zhang, N.; Zhou, S.; Peng, Y.; Zheng, J. Nitidine chloride is a mechanism-based inactivator of cyp2d6. Drug Metab. Dispos. 2018, 46, 1137–1145. [Google Scholar] [CrossRef]
- Karki, K.; Hedrick, E.; Kasiappan, R.; Jin, U.H.; Safe, S. Piperlongumine induces reactive oxygen species (ros)-dependent downregulation of specificity protein transcription factors. Cancer Prev. Res. (Phila) 2017, 10, 467–477. [Google Scholar] [CrossRef]
- Dong, X.Z.; Song, Y.; Lu, Y.P.; Hu, Y.; Liu, P.; Zhang, L. Sanguinarine inhibits the proliferation of bgc-823 gastric cancer cells via regulating mir-96-5p/mir-29c-3p and the mapk/jnk signaling pathway. J. Nat. Med. 2019, 73, 777–788. [Google Scholar] [CrossRef]
- Xu, F.; Li, Q.; Wang, Z.; Cao, X. Sinomenine inhibits proliferation, migration, invasion and promotes apoptosis of prostate cancer cells by regulation of mir-23a. Biomed. Pharmacother. 2019, 112, 108592. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, B.; Chen, G.; Wu, W.; Zhou, L.; Shi, Y.; Zeng, Q.; Li, Y.; Sun, Y.; Deng, X.; et al. Targeting mir-21 with sophocarpine inhibits tumor progression and reverses epithelial-mesenchymal transition in head and neck cancer. Mol. Ther. 2017, 25, 2129–2139. [Google Scholar] [CrossRef]
- Sun, L.; Jin, X.; Xie, L.; Xu, G.; Cui, Y.; Chen, Z. Swainsonine represses glioma cell proliferation, migration and invasion by reduction of mir-92a expression. BMC Cancer 2019, 19, 247. [Google Scholar] [CrossRef]
- Lv, Q.; Xia, Q.; Li, J.; Wang, Z. Allicin suppresses growth and metastasis of gastric carcinoma: The key role of microrna-383-5p-mediated inhibition of erbb4 signaling. Biosci. Biotechnol. Biochem. 2020, 84, 1997–2004. [Google Scholar] [CrossRef]
- Martin, S.L.; Kala, R.; Tollefsbol, T.O. Mechanisms for the inhibition of colon cancer cells by sulforaphane through epigenetic modulation of microrna-21 and human telomerase reverse transcriptase (htert) down-regulation. Curr. Cancer Drug Targets 2018, 18, 97–106. [Google Scholar] [CrossRef]
- Xu, H.; Li, Y.; Han, B.; Li, Z.; Wang, B.; Jiang, P.; Zhang, J.; Ma, W.; Zhou, D.; Li, X.; et al. Anti-breast-cancer activity exerted by beta-sitosterol-d-glucoside from sweet potato via upregulation of microrna-10a and via the pi3k-akt signaling pathway. J. Agric. Food Chem. 2018, 66, 9704–9718. [Google Scholar] [CrossRef]
- Sok, S.P.; Arshad, N.M.; Azmi, M.N.; Awang, K.; Ozpolat, B.; Hasima Nagoor, N. The apoptotic effect of 1’s-1’-acetoxychavicol acetate (aca) enhanced by inhibition of non-canonical autophagy in human non-small cell lung cancer cells. PLoS ONE 2017, 12, e0171329. [Google Scholar] [CrossRef]
- Gao, A.M.; Zhang, X.Y.; Ke, Z.P. Apigenin sensitizes bel-7402/adm cells to doxorubicin through inhibiting mir-101/nrf2 pathway. Oncotarget 2017, 8, 82085–82091. [Google Scholar] [CrossRef]
- Li, W.; Hao, J.; Zhang, L.; Cheng, Z.; Deng, X.; Shu, G. Astragalin reduces hexokinase 2 through increasing mir-125b to inhibit the proliferation of hepatocellular carcinoma cells in vitro and in vivo. J. Agric. Food Chem. 2017, 65, 5961–5972. [Google Scholar] [CrossRef]
- Bie, B.; Sun, J.; Li, J.; Guo, Y.; Jiang, W.; Huang, C.; Yang, J.; Li, Z. Baicalein, a natural anti-cancer compound, alters microrna expression profiles in bel-7402 human hepatocellular carcinoma cells. Cell. Physiol. Biochem. 2017, 41, 1519–1531. [Google Scholar] [CrossRef]
- Tao, Y.; Zhan, S.; Wang, Y.; Zhou, G.; Liang, H.; Chen, X.; Shen, H. Baicalin, the major component of traditional chinese medicine scutellaria baicalensis induces colon cancer cell apoptosis through inhibition of oncomirnas. Sci. Rep. 2018, 8, 14477. [Google Scholar] [CrossRef]
- Tao, L.Y.; Li, J.Y.; Zhang, J.Y. Brazilein, a compound isolated from caesalpinia sappan linn., induced growth inhibition in breast cancer cells via involvement of gsk-3beta/beta-catenin/cyclin d1 pathway. Chem. Biol. Interact. 2013, 206, 1–5. [Google Scholar] [CrossRef]
- Javan, N.; Khadem Ansari, M.H.; Dadashpour, M.; Khojastehfard, M.; Bastami, M.; Rahmati-Yamchi, M.; Zarghami, N. Synergistic antiproliferative effects of co-nanoencapsulated curcumin and chrysin on mda-mb-231 breast cancer cells through upregulating mir-132 and mir-502c. Nutr. Cancer 2019, 71, 1201–1213. [Google Scholar] [CrossRef]
- Zhou, L.; Lu, Y.; Liu, J.S.; Long, S.Z.; Liu, H.L.; Zhang, J.; Zhang, T. The role of mir-21/reck in the inhibition of osteosarcoma by curcumin. Mol. Cell. Probes 2020, 51, 101534. [Google Scholar] [CrossRef]
- Huang, C.C.; Hung, C.H.; Hung, T.W.; Lin, Y.C.; Wang, C.J.; Kao, S.H. Dietary delphinidin inhibits human colorectal cancer metastasis associating with upregulation of mir-204-3p and suppression of the integrin/fak axis. Sci. Rep. 2019, 9, 18954. [Google Scholar] [CrossRef]
- Lin, C.H.; Wang, H.H.; Chen, T.H.; Chiang, M.C.; Hung, P.H.; Chen, Y.J. Involvement of microrna-296 in the inhibitory effect of epigallocatechin gallate against the migratory properties of anoikis-resistant nasopharyngeal carcinoma cells. Cancers 2020, 12, 973. [Google Scholar] [CrossRef]
- Wu, Y.; Zhang, X.; Li, Z.; Yan, H.; Qin, J.; Li, T. Formononetin inhibits human bladder cancer cell proliferation and invasiveness via regulation of mir-21 and pten. Food Funct. 2017, 8, 1061–1066. [Google Scholar] [CrossRef]
- Zou, Y.; Li, R.; Kuang, D.; Zuo, M.; Li, W.; Tong, W.; Jiang, L.; Zhou, M.; Chen, Y.; Gong, W.; et al. Galangin inhibits cholangiocarcinoma cell growth and metastasis through downregulation of microrna-21 expression. Biomed. Res. Int. 2020, 2020, 5846938. [Google Scholar] [CrossRef]
- Gao, G.; Bian, Y.; Qian, H.; Yang, M.; Hu, J.; Li, L.; Yu, L.; Liu, B.; Qian, X. Gambogic acid regulates the migration and invasion of colorectal cancer via microrna-21-mediated activation of phosphatase and tensin homolog. Exp. Ther. Med. 2018, 16, 1758–1765. [Google Scholar] [CrossRef]
- Hsieh, P.L.; Liao, Y.W.; Hsieh, C.W.; Chen, P.N.; Yu, C.C. Soy isoflavone genistein impedes cancer stemness and mesenchymal transition in head and neck cancer through activating mir-34a/rtcb axis. Nutrients 2020, 12, 1924. [Google Scholar] [CrossRef]
- Lin, J.; Wu, Y.; Yang, D.; Zhao, Y. Induction of apoptosis and antitumor effects of a small molecule inhibitor of bcl-2 and bcl-xl, gossypol acetate, in multiple myeloma in vitro and in vivo. Oncol. Rep. 2013, 30, 731–738. [Google Scholar] [CrossRef]
- Chou, L.F.; Chen, C.Y.; Yang, W.H.; Chen, C.C.; Chang, J.L.; Leu, Y.L.; Liou, M.J.; Wang, T.H. Suppression of hepatocellular carcinoma progression through foxm1 and emt inhibition via hydroxygenkwanin-induced mir-320a expression. Biomolecules 2019, 10, 20. [Google Scholar] [CrossRef]
- Yang, J.; Zou, Y.; Jiang, D. Honokiol suppresses proliferation and induces apoptosis via regulation of the mir21/pten/pi3k/akt signaling pathway in human osteosarcoma cells. Int. J. Mol. Med. 2018, 41, 1845–1854. [Google Scholar]
- Fang, L.; Xu, W.; Kong, D. Icariin inhibits cell proliferation, migration and invasion by down-regulation of microrna-625-3p in thyroid cancer cells. Biomed. Pharmacother. 2019, 109, 2456–2463. [Google Scholar] [CrossRef]
- Wang, T.T.; Chen, Z.Z.; Xie, P.; Zhang, W.J.; Du, M.Y.; Liu, Y.T.; Zhu, H.Y.; Guo, Y.S. Isoliquiritigenin suppresses the proliferation and induced apoptosis via mir-32/lats2/wnt in nasopharyngeal carcinoma. Eur. J. Pharmacol. 2019, 856, 172352. [Google Scholar] [CrossRef]
- Zeng, X.; Xu, Z.; Gu, J.; Huang, H.; Gao, G.; Zhang, X.; Li, J.; Jin, H.; Jiang, G.; Sun, H.; et al. Induction of mir-137 by isorhapontigenin (iso) directly targets sp1 protein translation and mediates its anticancer activity both in vitro and in vivo. Mol. Cancer Ther. 2016, 15, 512–522. [Google Scholar] [CrossRef]
- Han, X.; Liu, C.F.; Gao, N.; Zhao, J.; Xu, J. Kaempferol suppresses proliferation but increases apoptosis and autophagy by up-regulating microrna-340 in human lung cancer cells. Biomed. Pharmacother. 2018, 108, 809–816. [Google Scholar] [CrossRef]
- Huang, W.C.; Su, H.H.; Fang, L.W.; Wu, S.J.; Liou, C.J. Licochalcone a inhibits cellular motility by suppressing e-cadherin and mapk signaling in breast cancer. Cells 2019, 8, 218. [Google Scholar] [CrossRef]
- Jiang, Z.Q.; Li, M.H.; Qin, Y.M.; Jiang, H.Y.; Zhang, X.; Wu, M.H. Luteolin inhibits tumorigenesis and induces apoptosis of non-small cell lung cancer cells via regulation of microrna-34a-5p. Int. J. Mol. Sci. 2018, 19, 447. [Google Scholar] [CrossRef]
- Niu, Y.; Zhang, J.; Tong, Y.; Li, J.; Liu, B. Physcion 8-o-beta-glucopyranoside induced ferroptosis via regulating mir-103a-3p/gls2 axis in gastric cancer. Life Sci. 2019, 237, 116893. [Google Scholar] [CrossRef]
- Mao, J.T.; Xue, B.; Smoake, J.; Lu, Q.Y.; Park, H.; Henning, S.M.; Burns, W.; Bernabei, A.; Elashoff, D.; Serio, K.J.; et al. Microrna-19a/b mediates grape seed procyanidin extract-induced anti-neoplastic effects against lung cancer. J. Nutr. Biochem. 2016, 34, 118–125. [Google Scholar] [CrossRef]
- Zhou, Y.; Xue, R.; Wang, J.; Ren, H. Puerarin inhibits hepatocellular carcinoma invasion and metastasis through mir-21-mediated pten/akt signaling to suppress the epithelial-mesenchymal transition. Braz. J. Med. Biol. Res. 2020, 53, e8882. [Google Scholar] [CrossRef]
- Nwaeburu, C.C.; Bauer, N.; Zhao, Z.; Abukiwan, A.; Gladkich, J.; Benner, A.; Herr, I. Up-regulation of microrna let-7c by quercetin inhibits pancreatic cancer progression by activation of numbl. Oncotarget 2016, 7, 58367–58380. [Google Scholar] [CrossRef]
- Wu, F.; Cui, L. Resveratrol suppresses melanoma by inhibiting nf-kappab/mir-221 and inducing tfg expression. Arch Dermatol. Res. 2017, 309, 823–831. [Google Scholar] [CrossRef]
- Liu, J.; Qu, C.B.; Xue, Y.X.; Li, Z.; Wang, P.; Liu, Y.H. Mir-143 enhances the antitumor activity of shikonin by targeting bag3 expression in human glioblastoma stem cells. Biochem. Biophys. Res. Commun. 2015, 468, 105–112. [Google Scholar] [CrossRef]
- Zadeh, M.M.; Motamed, N.; Ranji, N.; Majidi, M.; Falahi, F. Silibinin-induced apoptosis and downregulation of microrna-21 and microrna-155 in mcf-7 human breast cancer cells. J. Breast Cancer 2016, 19, 45–52. [Google Scholar] [CrossRef]
- Federico, A.; Dallio, M.; Di Fabio, G.; Zarrelli, A.; Zappavigna, S.; Stiuso, P.; Tuccillo, C.; Caraglia, M.; Loguercio, C. Silybin-phosphatidylcholine complex protects human gastric and liver cells from oxidative stress. In Vivo 2015, 29, 569–575. [Google Scholar]
- Zarrelli, A.; Romanucci, V.; Tuccillo, C.; Federico, A.; Loguercio, C.; Gravante, R.; Di Fabio, G. New silibinin glyco-conjugates: Synthesis and evaluation of antioxidant properties. Bioorg. Med. Chem. Lett. 2014, 24, 5147–5149. [Google Scholar] [CrossRef]
- Singh, T.; Prasad, R.; Katiyar, S.K. Therapeutic intervention of silymarin on the migration of non-small cell lung cancer cells is associated with the axis of multiple molecular targets including class 1 hdacs, zeb1 expression, and restoration of mir-203 and e-cadherin expression. Am. J. Cancer Res. 2016, 6, 1287–1301. [Google Scholar]
- Lu, M.; Liu, B.; Xiong, H.; Wu, F.; Hu, C.; Liu, P. Trans-3,5,4 -trimethoxystilbene reduced gefitinib resistance in nsclcs via suppressing mapk/akt/bcl-2 pathway by upregulation of mir-345 and mir-498. J. Cell. Mol. Med. 2019, 23, 2431–2441. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, C.; Min, D. Ailanthone up-regulates mir-449a to restrain acute myeloid leukemia cells growth, migration and invasion. Exp. Mol. Pathol. 2019, 108, 114–120. [Google Scholar] [CrossRef]
- Yang, P.Y.; Hsieh, P.L.; Wang, T.H.; Yu, C.C.; Lu, M.Y.; Liao, Y.W.; Lee, T.H.; Peng, C.Y. Andrographolide impedes cancer stemness and enhances radio-sensitivity in oral carcinomas via mir-218 activation. Oncotarget 2017, 8, 4196–4207. [Google Scholar] [CrossRef]
- Konstat-Korzenny, E.; Ascencio-Aragon, J.A.; Niezen-Lugo, S.; Vazquez-Lopez, R. Artemisinin and its synthetic derivatives as a possible therapy for cancer. Med. Sci. 2018, 6, 19. [Google Scholar] [CrossRef]
- Xu, F.; Cui, W.Q.; Wei, Y.; Cui, J.; Qiu, J.; Hu, L.L.; Gong, W.Y.; Dong, J.C.; Liu, B.J. Astragaloside iv inhibits lung cancer progression and metastasis by modulating macrophage polarization through ampk signaling. J. Exp. Clin. Cancer Res. 2018, 37, 207. [Google Scholar] [CrossRef]
- Xiao, Z.; Ching Chow, S.; Han Li, C.; Chun Tang, S.; Tsui, S.K.; Lin, Z.; Chen, Y. Role of microrna-95 in the anticancer activity of brucein d in hepatocellular carcinoma. Eur. J. Pharmacol. 2014, 728, 141–150. [Google Scholar] [CrossRef]
- Alharris, E.; Singh, N.P.; Nagarkatti, P.S.; Nagarkatti, M. Role of mirna in the regulation of cannabidiol-mediated apoptosis in neuroblastoma cells. Oncotarget 2019, 10, 45–59. [Google Scholar] [CrossRef]
- Ni, H.; Han, Y.; Jin, X. Celastrol inhibits colon cancer cell proliferation by downregulating mir-21 and pi3k/akt/gsk-3beta pathway. Int. J. Clin. Exp. Pathol. 2019, 12, 808–816. [Google Scholar]
- Sikander, M.; Hafeez, B.B.; Malik, S.; Alsayari, A.; Halaweish, F.T.; Yallapu, M.M.; Chauhan, S.C.; Jaggi, M. Cucurbitacin d exhibits potent anti-cancer activity in cervical cancer. Sci. Rep. 2016, 6, 36594. [Google Scholar] [CrossRef]
- Zeng, C.; Fan, D.; Xu, Y.; Li, X.; Yuan, J.; Yang, Q.; Zhou, X.; Lu, J.; Zhang, C.; Han, J.; et al. Curcumol enhances the sensitivity of doxorubicin in triple-negative breast cancer via regulating the mir-181b-2-3p-abcc3 axis. Biochem. Pharmacol. 2020, 174, 113795. [Google Scholar] [CrossRef]
- Wang, P.; Du, X.; Xiong, M.; Cui, J.; Yang, Q.; Wang, W.; Chen, Y.; Zhang, T. Ginsenoside rd attenuates breast cancer metastasis implicating derepressing microrna-18a-regulated smad2 expression. Sci. Rep. 2016, 6, 33709. [Google Scholar] [CrossRef]
- Gao, Q.; Zheng, J. Ginsenoside rh2 inhibits prostate cancer cell growth through suppression of microrna-4295 that activates cdkn1a. Cell Prolif. 2018, 51, e12438. [Google Scholar] [CrossRef]
- Li, D.; Chen, L.; Zhao, W.; Hao, J.; An, R. Microrna-let-7f-1 is induced by lycopene and inhibits cell proliferation and triggers apoptosis in prostate cancer. Mol. Med. Rep. 2016, 13, 2708–2714. [Google Scholar] [CrossRef]
- Hernandes, C.; Miguita, L.; de Sales, R.O.; Silva, E.P.; Mendonca, P.O.R.; Lorencini da Silva, B.; Klingbeil, M.F.G.; Mathor, M.B.; Rangel, E.B.; Marti, L.C.; et al. Anticancer activities of the quinone-methide triterpenes maytenin and 22-beta-hydroxymaytenin obtained from cultivated maytenus ilicifolia roots associated with down-regulation of mirna-27a and mir-20a/mir-17-5p. Molecules 2020, 25, 760. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, M.; Li, D. Oleanolic acid suppresses the proliferation of lung carcinoma cells by mir-122/cyclin g1/mef2d axis. Mol. Cell. Biochem. 2015, 400, 1–7. [Google Scholar] [CrossRef]
- Weng, H.; Huang, H.; Dong, B.; Zhao, P.; Zhou, H.; Qu, L. Inhibition of mir-17 and mir-20a by oridonin triggers apoptosis and reverses chemoresistance by derepressing bim-s. Cancer Res. 2014, 74, 4409–4419. [Google Scholar] [CrossRef]
- Cheng, S.; Zhang, Z.; Hu, C.; Xing, N.; Xia, Y.; Pang, B. Pristimerin suppressed breast cancer progression via mir-542-5p/dub3 axis. Onco Targets Ther. 2020, 13, 6651–6660. [Google Scholar] [CrossRef]
- Wang, G.; Huang, Y.X.; Zhang, R.; Hou, L.D.; Liu, H.; Chen, X.Y.; Zhu, J.S.; Zhang, J. Toosendanin suppresses oncogenic phenotypes of human gastric carcinoma sgc7901 cells partly via mir200amediated downregulation of beta-catenin pathway. Int. J. Oncol. 2017, 51, 1563–1573. [Google Scholar] [CrossRef]
- Li, S.G.; Shi, Q.W.; Yuan, L.Y.; Qin, L.P.; Wang, Y.; Miao, Y.Q.; Chen, Z.; Ling, C.Q.; Qin, W.X. C-myc-dependent repression of two oncogenic mirna clusters contributes to triptolide-induced cell death in hepatocellular carcinoma cells. J. Exp. Clin. Cancer Res. 2018, 37, 51. [Google Scholar] [CrossRef]
- Shi, H.; Bi, H.; Sun, X.; Dong, H.; Jiang, Y.; Mu, H.; Liu, G.; Kong, W.; Gao, R.; Su, J. Antitumor effects of tubeimoside-1 in nci-h1299 cells are mediated by microrna-126-5p-induced inactivation of vegf-a/vegfr-2/erk signaling pathway. Mol. Med. Rep. 2018, 17, 4327–4336. [Google Scholar] [CrossRef]
- Xiang, F.; Fan, Y.; Ni, Z.; Liu, Q.; Zhu, Z.; Chen, Z.; Hao, W.; Yue, H.; Wu, R.; Kang, X. Ursolic acid reverses the chemoresistance of breast cancer cells to paclitaxel by targeting mirna-149-5p/myd88. Front. Oncol. 2019, 9, 501. [Google Scholar] [CrossRef]
- Xu, Q.; Li, M.; Yang, M.; Yang, J.; Xie, J.; Lu, X.; Wang, F.; Chen, W. Alpha-pinene regulates mir-221 and induces g2/m phase cell cycle arrest in human hepatocellular carcinoma cells. Biosci. Rep. 2018, 38, BSR20180980. [Google Scholar] [CrossRef]
- Long, X.; Shi, Y.; Ye, P.; Guo, J.; Zhou, Q.; Tang, Y. Microrna-99a suppresses breast cancer progression by targeting fgfr3. Front. Oncol. 2019, 9, 1473. [Google Scholar] [CrossRef]
- Murray, M.Y.; Rushworth, S.A.; Zaitseva, L.; Bowles, K.M.; Macewan, D.J. Attenuation of dexamethasone-induced cell death in multiple myeloma is mediated by mir-125b expression. Cell Cycle 2013, 12, 2144–2153. [Google Scholar] [CrossRef]
- Yue, X.; Zhang, C.; Zhao, Y.; Liu, J.; Lin, A.W.; Tan, V.M.; Drake, J.M.; Liu, L.; Boateng, M.N.; Li, J.; et al. Gain-of-function mutant p53 activates small gtpase rac1 through sumoylation to promote tumor progression. Genes Dev. 2017, 31, 1641–1654. [Google Scholar] [CrossRef]
- Liu, H.; Huang, C.; Wu, L.; Wen, B. Effect of evodiamine and berberine on mir-429 as an oncogene in human colorectal cancer. Onco Targets Ther. 2016, 9, 4121–4127. [Google Scholar]
- Liu, N.; Li, P.; Zang, S.; Liu, Q.; Ma, D.; Sun, X.; Ji, C. Novel agent nitidine chloride induces erythroid differentiation and apoptosis in cml cells through c-myc-mirnas axis. PLoS ONE 2015, 10, e0116880. [Google Scholar] [CrossRef]
- Pan, J.; Shen, J.; Si, W.; Du, C.; Chen, D.; Xu, L.; Yao, M.; Fu, P.; Fan, W. Resveratrol promotes mica/b expression and natural killer cell lysis of breast cancer cells by suppressing c-myc/mir-17 pathway. Oncotarget 2017, 8, 65743–65758. [Google Scholar] [CrossRef]
- Lan, F.; Pan, Q.; Yu, H.; Yue, X. Sulforaphane enhances temozolomide-induced apoptosis because of down-regulation of mir-21 via wnt/beta-catenin signaling in glioblastoma. J. Neurochem. 2015, 134, 811–818. [Google Scholar] [CrossRef]
- Lewinska, A.; Adamczyk-Grochala, J.; Deregowska, A.; Wnuk, M. Sulforaphane-induced cell cycle arrest and senescence are accompanied by DNA hypomethylation and changes in microrna profile in breast cancer cells. Theranostics 2017, 7, 3461–3477. [Google Scholar] [CrossRef]
- Zan, L.; Chen, Q.; Zhang, L.; Li, X. Epigallocatechin gallate (egcg) suppresses growth and tumorigenicity in breast cancer cells by downregulation of mir-25. Bioengineered 2019, 10, 374–382. [Google Scholar] [CrossRef]
- Liu, Z.M.; Yang, X.L.; Jiang, F.; Pan, Y.C.; Zhang, L. Matrine involves in the progression of gastric cancer through inhibiting mir-93-5p and upregulating the expression of target gene ahnak. J. Cell Biochem. 2020, 121, 2467–2477. [Google Scholar] [CrossRef]
- Coker-Gurkan, A.; Bulut, D.; Genc, R.; Arisan, E.D.; Obakan-Yerlikaya, P.; Palavan-Unsal, N. Curcumin prevented human autocrine growth hormone (gh) signaling mediated nf-kappab activation and mir-183-96-182 cluster stimulated epithelial mesenchymal transition in t47d breast cancer cells. Mol. Biol. Rep. 2019, 46, 355–369. [Google Scholar] [CrossRef]
- Zhou, W.; Wang, S.; Ying, Y.; Zhou, R.; Mao, P. Mir-196b/mir-1290 participate in the antitumor effect of resveratrol via regulation of igfbp3 expression in acute lymphoblastic leukemia. Oncol. Rep. 2017, 37, 1075–1083. [Google Scholar] [CrossRef]
- Jia, Y.; Chen, L.; Guo, S.; Li, Y. Baicalin induced colon cancer cells apoptosis through mir-217/dkk1-mediated inhibition of wnt signaling pathway. Mol. Biol. Rep. 2019, 46, 1693–1700. [Google Scholar] [CrossRef]
- Allegri, L.; Rosignolo, F.; Mio, C.; Filetti, S.; Baldan, F.; Damante, G. Effects of nutraceuticals on anaplastic thyroid cancer cells. J. Cancer Res. Clin. Oncol. 2018, 144, 285–294. [Google Scholar] [CrossRef]
- Ma, J.; Cheng, L.; Liu, H.; Zhang, J.; Shi, Y.; Zeng, F.; Miele, L.; Sarkar, F.H.; Xia, J.; Wang, Z. Genistein down-regulates mir-223 expression in pancreatic cancer cells. Curr. Drug Targets 2013, 14, 1150–1156. [Google Scholar] [CrossRef]
- Xiang, S.; Chen, H.; Luo, X.; An, B.; Wu, W.; Cao, S.; Ruan, S.; Wang, Z.; Weng, L.; Zhu, H.; et al. Isoliquiritigenin suppresses human melanoma growth by targeting mir-301b/lrig1 signaling. J. Exp. Clin. Cancer Res. 2018, 37, 184. [Google Scholar] [CrossRef]
- Moeng, S.; Son, S.W.; Seo, H.A.; Lee, J.S.; Kim, C.K.; Kuh, H.J.; Park, J.K. Luteolin-regulated microrna-301-3p targets caspase-8 and modulates trail sensitivity in panc-1 cells. Anticancer Res. 2020, 40, 723–731. [Google Scholar] [CrossRef]
- Song, L.; Liu, D.; Zhao, Y.; He, J.; Kang, H.; Dai, Z.; Wang, X.; Zhang, S.; Zan, Y. Sinomenine inhibits breast cancer cell invasion and migration by suppressing nf-kappab activation mediated by il-4/mir-324-5p/cuedc2 axis. Biochem. Biophys. Res. Commun. 2015, 464, 705–710. [Google Scholar] [CrossRef]
- Peng, F.; Tang, H.; Liu, P.; Shen, J.; Guan, X.; Xie, X.; Gao, J.; Xiong, L.; Jia, L.; Chen, J.; et al. Isoliquiritigenin modulates mir-374a/pten/akt axis to suppress breast cancer tumorigenesis and metastasis. Sci. Rep. 2017, 7, 9022. [Google Scholar] [CrossRef]
- Wang, K.; Tan, S.L.; Lu, Q.; Xu, R.; Cao, J.; Wu, S.Q.; Wang, Y.H.; Zhao, X.K.; Zhong, Z.H. Curcumin suppresses microrna-7641-mediated regulation of p16 expression in bladder cancer. Am. J. Chin. Med. 2018, 46, 1357–1368. [Google Scholar] [CrossRef]
- Jung, Y.Y.; Um, J.Y.; Narula, A.S.; Namjoshi, O.A.; Blough, B.E.; Kumar, A.P.; Ahn, K.S. Identification of matrine as a novel regulator of the cxcr4 signaling axis in tumor cells. Int. J. Mol. Sci. 2020, 21, 4731. [Google Scholar] [CrossRef]
- Shen, E.; Wang, X.; Liu, X.; Lv, M.; Zhang, L.; Zhu, G.; Sun, Z. Microrna-93-5p promotes epithelial-mesenchymal transition in gastric cancer by repressing tumor suppressor ahnak expression. Cancer Cell Int. 2020, 20, 76. [Google Scholar] [CrossRef]
- Sobhani, N.; Ianza, A.; D’Angelo, A.; Roviello, G.; Giudici, F.; Bortul, M.; Zanconati, F.; Bottin, C.; Generali, D. Current status of fibroblast growth factor receptor-targeted therapies in breast cancer. Cells 2018, 7, 76. [Google Scholar] [CrossRef]
- Sun, S.; Jiang, Y.; Zhang, G.; Song, H.; Zhang, X.; Zhang, Y.; Liang, X.; Sun, Q.; Pang, D. Increased expression of fibroblastic growth factor receptor 2 is correlated with poor prognosis in patients with breast cancer. J. Surg. Oncol. 2012, 105, 773–779. [Google Scholar] [CrossRef]
- Deng, G.; Zeng, S.; Ma, J.; Zhang, Y.; Qu, Y.; Han, Y.; Yin, L.; Cai, C.; Guo, C.; Shen, H. The anti-tumor activities of neferine on cell invasion and oxaliplatin sensitivity regulated by emt via snail signaling in hepatocellular carcinoma. Sci. Rep. 2017, 7, 41616. [Google Scholar] [CrossRef]
- Nazim, U.M.; Yin, H.; Park, S.Y. Neferine treatment enhances the trailinduced apoptosis of human prostate cancer cells via autophagic flux and the jnk pathway. Int. J. Oncol. 2020, 56, 1152–1161. [Google Scholar]
- Turczyk, L.; Kitowska, K.; Mieszkowska, M.; Mieczkowski, K.; Czaplinska, D.; Piasecka, D.; Kordek, R.; Skladanowski, A.C.; Potemski, P.; Romanska, H.M.; et al. Fgfr2-driven signaling counteracts tamoxifen effect on eralpha-positive breast cancer cells. Neoplasia 2017, 19, 791–804. [Google Scholar] [CrossRef]
- Nakamura, S.; Yokota, D.; Tan, L.; Nagata, Y.; Takemura, T.; Hirano, I.; Shigeno, K.; Shibata, K.; Fujisawa, S.; Ohnishi, K. Down-regulation of thanatos-associated protein 11 by bcr-abl promotes cml cell proliferation through c-myc expression. Int. J. Cancer 2012, 130, 1046–1059. [Google Scholar] [CrossRef]
- Sodaro, G.; Cesaro, E.; Montano, G.; Blasio, G.; Fiorentino, F.; Romano, S.; Jacquel, A.; Aurberger, P.; Costanzo, P. Role of znf224 in c-myc repression and imatinib responsiveness in chronic myeloid leukemia. Oncotarget 2018, 9, 3417–3431. [Google Scholar] [CrossRef]
- Grandjenette, C.; Schnekenburger, M.; Karius, T.; Ghelfi, J.; Gaigneaux, A.; Henry, E.; Dicato, M.; Diederich, M. 5-aza-2’-deoxycytidine-mediated c-myc down-regulation triggers telomere-dependent senescence by regulating human telomerase reverse transcriptase in chronic myeloid leukemia. Neoplasia 2014, 16, 511–528. [Google Scholar] [CrossRef]
- Li, Y.; Choi, P.S.; Casey, S.C.; Dill, D.L.; Felsher, D.W. Myc through mir-17-92 suppresses specific target genes to maintain survival, autonomous proliferation, and a neoplastic state. Cancer Cell 2014, 26, 262–272. [Google Scholar] [CrossRef]
- Niu, H.; Wang, K.; Zhang, A.; Yang, S.; Song, Z.; Wang, W.; Qian, C.; Li, X.; Zhu, Y.; Wang, Y. Mir-92a is a critical regulator of the apoptosis pathway in glioblastoma with inverse expression of bcl2l11. Oncol. Rep. 2012, 28, 1771–1777. [Google Scholar] [CrossRef]
- Song, H.; Zhang, Y.; Liu, N.; Zhao, S.; Kong, Y.; Yuan, L. Mir-92a-3p exerts various effects in glioma and glioma stem-like cells specifically targeting cdh1/beta-catenin and notch-1/akt signaling pathways. Int. J. Mol. Sci. 2016, 17, 1799. [Google Scholar] [CrossRef]
- Liu, P.J.; Ye, Y.X.; Wang, Y.X.; Du, J.X.; Pan, Y.H.; Fang, X.B. Mirna-92a promotes cell proliferation and invasion through binding to klf4 in glioma. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 6612–6620. [Google Scholar]
- Rawat, L.; Hegde, H.; Hoti, S.L.; Nayak, V. Piperlongumine induces ros mediated cell death and synergizes paclitaxel in human intestinal cancer cells. Biomed. Pharmacother. 2020, 128, 110243. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, J.; Venkat, P.S.; Gu, C.; Meng, Y. Sanguinarine exhibits potent efficacy against cervical cancer cells through inhibiting the stat3 pathway in vitro and in vivo. Cancer Manag. Res. 2019, 11, 7557–7566. [Google Scholar] [CrossRef]
- Zhu, M.; Gong, Z.; Wu, Q.; Shi, X.; Su, Q.; Zhang, Y. Sanguinarine suppresses migration and metastasis in colorectal carcinoma associated with the inversion of emt through the wnt/beta-catenin signaling. Clin. Transl. Med. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Su, Q.; Fan, M.; Wang, J.; Ullah, A.; Ghauri, M.A.; Dai, B.; Zhan, Y.; Zhang, D.; Zhang, Y. Sanguinarine inhibits epithelial-mesenchymal transition via targeting hif-1alpha/tgf-beta feed-forward loop in hepatocellular carcinoma. Cell Death Dis. 2019, 10, 939. [Google Scholar] [CrossRef]
- He, X.; Zou, K. Mirna-96-5p contributed to the proliferation of gastric cancer cells by targeting foxo3. J. Biochem. 2020, 167, 101–108. [Google Scholar] [CrossRef]
- Gao, Y.; Qi, W.; Sun, L.; Lv, J.; Qiu, W.; Liu, S. Foxo3 inhibits human gastric adenocarcinoma (ags) cell growth by promoting autophagy in an acidic microenvironment. Cell Physiol. Biochem. 2018, 49, 335–348. [Google Scholar] [CrossRef]
- Wang, L.; Yu, T.; Li, W.; Li, M.; Zuo, Q.; Zou, Q.; Xiao, B. The mir-29c-kiaa1199 axis regulates gastric cancer migration by binding with wbp11 and ptp4a3. Oncogene 2019, 38, 3134–3150. [Google Scholar] [CrossRef]
- Liu, X.; Cai, J.; Sun, Y.; Gong, R.; Sun, D.; Zhong, X.; Jiang, S.; He, X.; Bao, E.; Yang, L.; et al. Microrna-29a inhibits cell migration and invasion via targeting roundabout homolog 1 in gastric cancer cells. Mol. Med. Rep. 2015, 12, 3944–3950. [Google Scholar] [CrossRef]
- Gao, L.; Cheng, D.; Yang, J.; Wu, R.; Li, W.; Kong, A.N. Sulforaphane epigenetically demethylates the cpg sites of the mir-9-3 promoter and reactivates mir-9-3 expression in human lung cancer a549 cells. J. Nutr. Biochem. 2018, 56, 109–115. [Google Scholar] [CrossRef]
- Tang, J.; Wang, Z.; Chen, L.; Huang, G.; Hu, X. Gossypol acetate induced apoptosis of pituitary tumor cells by targeting the bcl-2 via the upregulated microrna mir-15a. Int. J. Clin. Exp. Med. 2015, 8, 9079–9085. [Google Scholar]
- Zhang, B.; Wang, X.; Deng, J.; Zheng, H.; Liu, W.; Chen, S.; Tian, J.; Wang, F. P53-dependent upregulation of mir-16-2 by sanguinarine induces cell cycle arrest and apoptosis in hepatocellular carcinoma. Cancer Lett. 2019, 459, 50–58. [Google Scholar] [CrossRef]
- Hu, Z.; Fan, H.; Lv, G.; Zhou, Q.; Yang, B.; Zheng, J.; Cao, W. 5-aminolevulinic acid-mediated sonodynamic therapy induces anti-tumor effects in malignant melanoma via p53-mir-34a-sirt1 axis. J. Dermatol. Sci. 2015, 79, 155–162. [Google Scholar] [CrossRef]
- Gallardo, M.; Kemmerling, U.; Aguayo, F.; Bleak, T.C.; Munoz, J.P.; Calaf, G.M. Curcumin rescues breast cells from epithelialmesenchymal transition and invasion induced by antimir34a. Int. J. Oncol. 2020, 56, 480–493. [Google Scholar]
- Otsuka, K.; Yamamoto, Y.; Ochiya, T. Regulatory role of resveratrol, a microrna-controlling compound, in hnrnpa1 expression, which is associated with poor prognosis in breast cancer. Oncotarget 2018, 9, 24718–24730. [Google Scholar] [CrossRef]
- Zhang, W.; Jiang, H.; Chen, Y.; Ren, F. Resveratrol chemosensitizes adriamycin-resistant breast cancer cells by modulating mir-122-5p. J. Cell Biochem. 2019, 120, 16283–16292. [Google Scholar] [CrossRef]
- Zhang, D.; Han, Y.; Xu, L. Upregulation of mir-124 by physcion 8-o-beta-glucopyranoside inhibits proliferation and invasion of malignant melanoma cells via repressing rlip76. Biomed. Pharmacother. 2016, 84, 166–176. [Google Scholar] [CrossRef]
- Beccafico, S.; Morozzi, G.; Marchetti, M.C.; Riccardi, C.; Sidoni, A.; Donato, R.; Sorci, G. Artesunate induces ros- and p38 mapk-mediated apoptosis and counteracts tumor growth in vivo in embryonal rhabdomyosarcoma cells. Carcinogenesis 2015, 36, 1071–1083. [Google Scholar] [CrossRef]
- Mou, Z.; Wang, Y.; Li, Y. Brazilein induces apoptosis and g1/g0 phase cell cycle arrest by up-regulation of mir-133a in human vestibular schwannoma cells. Exp. Mol. Pathol. 2019, 107, 95–101. [Google Scholar] [CrossRef]
- Ye, Q.; Su, L.; Chen, D.; Zheng, W.; Liu, Y. Astragaloside iv induced mir-134 expression reduces emt and increases chemotherapeutic sensitivity by suppressing creb1 signaling in colorectal cancer cell line sw-480. Cell Physiol. Biochem. 2017, 43, 1617–1626. [Google Scholar] [CrossRef]
- Guo, X.; Huang, H.; Jin, H.; Xu, J.; Risal, S.; Li, J.; Li, X.; Yan, H.; Zeng, X.; Xue, L.; et al. Iso, via upregulating mir-137 transcription, inhibits gsk3beta-hsp70-mmp-2 axis, resulting in attenuating urothelial cancer invasion. Mol. Ther. Nucleic. Acids 2018, 12, 337–349. [Google Scholar] [CrossRef]
- Xiao, X.; Zhang, Y.; Pan, W.; Chen, F. Mir-139-mediated notch1 regulation is crucial for the inhibition of osteosarcoma progression caused by resveratrol. Life Sci. 2020, 242, 117215. [Google Scholar] [CrossRef]
- Guo, Q.; Dong, B.; Nan, F.; Guan, D.; Zhang, Y. 5-aminolevulinic acid photodynamic therapy in human cervical cancer via the activation of microrna-143 and suppression of the bcl-2/bax signaling pathway. Mol. Med. Rep. 2016, 14, 544–550. [Google Scholar] [CrossRef]
- Chen, G.; Ma, Y.; Jiang, Z.; Feng, Y.; Han, Y.; Tang, Y.; Zhang, J.; Ni, H.; Li, X.; Li, N. Lico a causes er stress and apoptosis via up-regulating mir-144-3p in human lung cancer cell line h292. Front. Pharmacol. 2018, 9, 837. [Google Scholar] [CrossRef]
- Xu, Z.; Zeng, X.; Xu, J.; Xu, D.; Li, J.; Jin, H.; Jiang, G.; Han, X.; Huang, C. Isorhapontigenin suppresses growth of patient-derived glioblastoma spheres through regulating mir-145/sox2/cyclin d1 axis. Neuro. Oncol. 2016, 18, 830–839. [Google Scholar] [CrossRef]
- Gao, W.; Ge, S.; Sun, J. Ailanthone exerts anticancer effect by up-regulating mir-148a expression in mda-mb-231 breast cancer cells and inhibiting proliferation, migration and invasion. Biomed. Pharmacother. 2019, 109, 1062–1069. [Google Scholar] [CrossRef]
- Chen, Q.; Luo, J.; Wu, C.; Lu, H.; Cai, S.; Bao, C.; Liu, D.; Kong, J. The mirna-149-5p/myd88 axis is responsible for ursolic acid-mediated attenuation of the stemness and chemoresistance of non-small cell lung cancer cells. Environ. Toxicol. 2020, 35, 561–569. [Google Scholar] [CrossRef]
- Hang, S.; Wang, X.; Li, H. Triptolide inhibits viability and migration while promotes apoptosis in nephroblastoma cells by regulation of mir-193b-3p. Exp. Mol. Pathol. 2019, 108, 80–88. [Google Scholar] [CrossRef]
- Nwaeburu, C.C.; Abukiwan, A.; Zhao, Z.; Herr, I. Quercetin-induced mir-200b-3p regulates the mode of self-renewing divisions in pancreatic cancer. Mol. Cancer 2017, 16, 23. [Google Scholar] [CrossRef]
- Karimi Dermani, F.; Saidijam, M.; Amini, R.; Mahdavinezhad, A.; Heydari, K.; Najafi, R. Resveratrol inhibits proliferation, invasion, and epithelial-mesenchymal transition by increasing mir-200c expression in hct-116 colorectal cancer cells. J. Cell. Biochem 2017, 118, 1547–1555. [Google Scholar] [CrossRef]
- Yao, Y.; Rao, C.; Zheng, G.; Wang, S. Luteolin suppresses colorectal cancer cell metastasis via regulation of the mir384/pleiotrophin axis. Oncol. Rep. 2019, 42, 131–141. [Google Scholar]
- Jiang, P.; Xu, C.; Chen, L.; Chen, A.; Wu, X.; Zhou, M.; Haq, I.U.; Mariyam, Z.; Feng, Q. Egcg inhibits csc-like properties through targeting mir-485/cd44 axis in a549-cisplatin resistant cells. Mol. Carcinog. 2018, 57, 1835–1844. [Google Scholar] [CrossRef]
- Gao, A.M.; Zhang, X.Y.; Hu, J.N.; Ke, Z.P. Apigenin sensitizes hepatocellular carcinoma cells to doxorubic through regulating mir-520b/atg7 axis. Chem. Biol. Interact. 2018, 280, 45–50. [Google Scholar] [CrossRef]
- Vislovukh, A.; Kratassiouk, G.; Porto, E.; Gralievska, N.; Beldiman, C.; Pinna, G.; El’skaya, A.; Harel-Bellan, A.; Negrutskii, B.; Groisman, I. Proto-oncogenic isoform a2 of eukaryotic translation elongation factor eef1 is a target of mir-663 and mir-744. Br. J. Cancer 2013, 108, 2304–2311. [Google Scholar] [CrossRef]
- Yang, P.W.; Lu, Z.Y.; Pan, Q.; Chen, T.T.; Feng, X.J.; Wang, S.M.; Pan, Y.C.; Zhu, M.H.; Zhang, S.H. Microrna-6809-5p mediates luteolin-induced anticancer effects against hepatoma by targeting flotillin 1. Phytomedicine 2019, 57, 18–29. [Google Scholar] [CrossRef]
- Wang, H.; Tan, Z.; Hu, H.; Liu, H.; Wu, T.; Zheng, C.; Wang, X.; Luo, Z.; Wang, J.; Liu, S.; et al. Microrna-21 promotes breast cancer proliferation and metastasis by targeting lztfl1. BMC Cancer 2019, 19, 738. [Google Scholar] [CrossRef]
- Nguyen, D.D.; Chang, S. Development of novel therapeutic agents by inhibition of oncogenic micrornas. Int. J. Mol. Sci. 2017, 19, 65. [Google Scholar] [CrossRef]
- Wang, C.; Li, Q.; He, Y. Microrna215p promotes epithelial to mesenchymal transition by targeting srybox 17 in endometrial cancer. Oncol. Rep. 2020, 43, 1897–1905. [Google Scholar]
- Han, M.; Liu, M.; Wang, Y.; Chen, X.; Xu, J.; Sun, Y.; Zhao, L.; Qu, H.; Fan, Y.; Wu, C. Antagonism of mir-21 reverses epithelial-mesenchymal transition and cancer stem cell phenotype through akt/erk1/2 inactivation by targeting pten. PLoS ONE 2012, 7, e39520. [Google Scholar] [CrossRef]
- Liu, N.; Guo, X.H.; Liu, J.P.; Cong, Y.S. Role of telomerase in the tumour microenvironment. Clin. Exp. Pharmacol. Physiol. 2020, 47, 357–364. [Google Scholar] [CrossRef]
- Abbas, A.; Hall, J.A.; Patterson, W.L., III; Ho, E.; Hsu, A.; Al-Mulla, F.; Georgel, P.T. Sulforaphane modulates telomerase activity via epigenetic regulation in prostate cancer cell lines. Biochem. Cell Biol. 2016, 94, 71–81. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, J.J.; Tao, H.; Jin, W.S. Microrna-21 controls htert via pten in human colorectal cancer cell proliferation. J. Physiol. Biochem. 2015, 71, 59–68. [Google Scholar] [CrossRef]
- Li, Y.; Cheng, H.S.; Chng, W.J.; Tergaonkar, V. Activation of mutant tert promoter by ras-erk signaling is a key step in malignant progression of braf-mutant human melanomas. Proc. Natl. Acad. Sci. USA 2016, 113, 14402–14407. [Google Scholar] [CrossRef]
- Hannafon, B.N.; Cai, A.; Calloway, C.L.; Xu, Y.F.; Zhang, R.; Fung, K.M.; Ding, W.Q. Mir-23b and mir-27b are oncogenic micrornas in breast cancer: Evidence from a crispr/cas9 deletion study. BMC Cancer 2019, 19, 642. [Google Scholar] [CrossRef]
- Ho, J.Y.; Hsu, R.J.; Liu, J.M.; Chen, S.C.; Liao, G.S.; Gao, H.W.; Yu, C.P. Microrna-382-5p aggravates breast cancer progression by regulating the rerg/ras/erk signaling axis. Oncotarget 2017, 8, 22443–22459. [Google Scholar] [CrossRef]
- Clark, J.C.; Thomas, D.M.; Choong, P.F.; Dass, C.R. Reck—A newly discovered inhibitor of metastasis with prognostic significance in multiple forms of cancer. Cancer Metastasis Rev. 2007, 26, 675–683. [Google Scholar] [CrossRef]
- Zhang, J.; Kong, X.; Li, J.; Luo, Q.; Li, X.; Shen, L.; Chen, L.; Fang, L. Mir-96 promotes tumor proliferation and invasion by targeting reck in breast cancer. Oncol. Rep. 2014, 31, 1357–1363. [Google Scholar] [CrossRef]
- Kessenbrock, K.; Plaks, V.; Werb, Z. Matrix metalloproteinases: Regulators of the tumor microenvironment. Cell 2010, 141, 52–67. [Google Scholar] [CrossRef]
- Liao, H.; Wang, Z.; Deng, Z.; Ren, H.; Li, X. Curcumin inhibits lung cancer invasion and metastasis by attenuating glut1/mt1-mmp/mmp2 pathway. Int. J. Clin. Exp. Med. 2015, 8, 8948–8957. [Google Scholar]
- Mbese, Z.; Khwaza, V.; Aderibigbe, B.A. Curcumin and its derivatives as potential therapeutic agents in prostate, colon and breast cancers. Molecules 2019, 24, 4386. [Google Scholar] [CrossRef]
- Zhang, W.; Qian, P.; Zhang, X.; Zhang, M.; Wang, H.; Wu, M.; Kong, X.; Tan, S.; Ding, K.; Perry, J.K.; et al. Autocrine/paracrine human growth hormone-stimulated microrna 96-182-183 cluster promotes epithelial-mesenchymal transition and invasion in breast cancer. J. Biol. Chem. 2015, 290, 13812–13829. [Google Scholar] [CrossRef]
- Mukhina, S.; Mertani, H.C.; Guo, K.; Lee, K.O.; Gluckman, P.D.; Lobie, P.E. Phenotypic conversion of human mammary carcinoma cells by autocrine human growth hormone. Proc. Natl. Acad. Sci. USA 2004, 101, 15166–15171. [Google Scholar] [CrossRef]
- Zhu, T.; Starling-Emerald, B.; Zhang, X.; Lee, K.O.; Gluckman, P.D.; Mertani, H.C.; Lobie, P.E. Oncogenic transformation of human mammary epithelial cells by autocrine human growth hormone. Cancer Res. 2005, 65, 317–324. [Google Scholar]
- Li, P.; Sheng, C.; Huang, L.; Zhang, H.; Huang, L.; Cheng, Z.; Zhu, Q. Mir-183/-96/-182 cluster is up-regulated in most breast cancers and increases cell proliferation and migration. Breast Cancer Res. 2014, 16, 473. [Google Scholar] [CrossRef]
- Kim, M.; Katayose, Y.; Rojanala, L.; Shah, S.; Sgagias, M.; Jang, L.; Jung, Y.J.; Lee, S.H.; Hwang, S.G.; Cowan, K.H. Induction of apoptosis in p16ink4a mutant cell lines by adenovirus-mediated overexpression of p16ink4a protein. Cell Death Differ. 2000, 7, 706–711. [Google Scholar] [CrossRef]
- Silva, G.; Aboussekhra, A. P16(ink4a) inhibits the pro-metastatic potentials of osteosarcoma cells through targeting the erk pathway and tgf-beta1. Mol. Carcinog. 2016, 55, 525–536. [Google Scholar] [CrossRef]
- Wang, Z.; Li, T.E.; Chen, M.; Pan, J.J.; Shen, K.W. Mir-106b-5p contributes to the lung metastasis of breast cancer via targeting cnn1 and regulating rho/rock1 pathway. Aging (Albany NY) 2020, 12, 1867–1887. [Google Scholar] [CrossRef]
- Chen, H.; Pan, H.; Qian, Y.; Zhou, W.; Liu, X. Mir-25-3p promotes the proliferation of triple negative breast cancer by targeting btg2. Mol. Cancer 2018, 17, 4. [Google Scholar] [CrossRef]
- Xie, C.M.; Tan, M.; Lin, X.T.; Wu, D.; Jiang, Y.; Tan, Y.; Li, H.; Ma, Y.; Xiong, X.; Sun, Y. The fbxw7-shoc2-raptor axis controls the cross-talks between the ras-erk and mtorc1 signaling pathways. Cell Rep. 2019, 26, 3037–3050.e4. [Google Scholar] [CrossRef]
- Sun, D.; Wang, C.; Long, S.; Ma, Y.; Guo, Y.; Huang, Z.; Chen, X.; Zhang, C.; Chen, J.; Zhang, J. C/ebp-beta-activated microrna-223 promotes tumour growth through targeting rasa1 in human colorectal cancer. Br. J. Cancer 2015, 112, 1491–1500. [Google Scholar] [CrossRef]
- Harmon, A.W.; Patel, Y.M.; Harp, J.B. Genistein inhibits ccaat/enhancer-binding protein beta (c/ebpbeta) activity and 3t3-l1 adipogenesis by increasing c/ebp homologous protein expression. Biochem. J. 2002, 367, 203–208. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, H.C.; Zhang, Y.; Huang, H. Microrna-625-3p inhibits gastric cancer metastasis through modulating ezh2. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 1177–1185. [Google Scholar]
- Huang, L.; Jin, K.; Lan, H. Luteolin inhibits cell cycle progression and induces apoptosis of breast cancer cells through downregulation of human telomerase reverse transcriptase. Oncol. Lett. 2019, 17, 3842–3850. [Google Scholar] [CrossRef]
- Wang, H.; Luo, Y.; Qiao, T.; Wu, Z.; Huang, Z. Luteolin sensitizes the antitumor effect of cisplatin in drug-resistant ovarian cancer via induction of apoptosis and inhibition of cell migration and invasion. J. Ovarian Res. 2018, 11, 93. [Google Scholar] [CrossRef]
- Tu, S.H.; Ho, C.T.; Liu, M.F.; Huang, C.S.; Chang, H.W.; Chang, C.H.; Wu, C.H.; Ho, Y.S. Luteolin sensitises drug-resistant human breast cancer cells to tamoxifen via the inhibition of cyclin e2 expression. Food Chem. 2013, 141, 1553–1561. [Google Scholar] [CrossRef]
- Stockwell, B.R.; Friedmann Angeli, J.P.; Bayir, H.; Bush, A.I.; Conrad, M.; Dixon, S.J.; Fulda, S.; Gascon, S.; Hatzios, S.K.; Kagan, V.E.; et al. Ferroptosis: A regulated cell death nexus linking metabolism, redox biology, and disease. Cell 2017, 171, 273–285. [Google Scholar] [CrossRef]
- Gao, M.; Monian, P.; Quadri, N.; Ramasamy, R.; Jiang, X. Glutaminolysis and transferrin regulate ferroptosis. Mol. Cell 2015, 59, 298–308. [Google Scholar] [CrossRef]
- Hou, C.; Chen, Y.; Huang, X.; Huang, Q.; Li, M.; Tan, X. Mir-19 targets pten and mediates high mobility group protein b1(hmgb1)-induced proliferation and migration of human airway smooth muscle cells. PLoS ONE 2019, 14, e0219081. [Google Scholar] [CrossRef]
- Liu, D.T.; Yao, H.R.; Li, Y.Y.; Song, Y.Y.; Su, M.Y. Microrna-19b promotes the migration and invasion of ovarian cancer cells by inhibiting the pten/akt signaling pathway. Oncol. Lett. 2018, 16, 559–565. [Google Scholar] [CrossRef]
- Zhang, P.; Li, H.; Wu, M.L.; Chen, X.Y.; Kong, Q.Y.; Wang, X.W.; Sun, Y.; Wen, S.; Liu, J. C-myc downregulation: A critical molecular event in resveratrol-induced cell cycle arrest and apoptosis of human medulloblastoma cells. J. Neurooncol. 2006, 80, 123–131. [Google Scholar] [CrossRef]
- Li, W.; Li, C.; Ma, L.; Jin, F. Resveratrol inhibits viability and induces apoptosis in the smallcell lung cancer h446 cell line via the pi3k/akt/cmyc pathway. Oncol. Rep. 2020, 44, 1821–1830. [Google Scholar]
- Bhardwaj, A.; Sethi, G.; Vadhan-Raj, S.; Bueso-Ramos, C.; Takada, Y.; Gaur, U.; Nair, A.S.; Shishodia, S.; Aggarwal, B.B. Resveratrol inhibits proliferation, induces apoptosis, and overcomes chemoresistance through down-regulation of stat3 and nuclear factor-kappab-regulated antiapoptotic and cell survival gene products in human multiple myeloma cells. Blood 2007, 109, 2293–2302. [Google Scholar] [CrossRef]
- Ko, J.H.; Sethi, G.; Um, J.Y.; Shanmugam, M.K.; Arfuso, F.; Kumar, A.P.; Bishayee, A.; Ahn, K.S. The role of resveratrol in cancer therapy. Int. J. Mol. Sci. 2017, 18, 2589. [Google Scholar] [CrossRef]
- Dutton-Regester, K.; Aoude, L.G.; Nancarrow, D.J.; Stark, M.S.; O’Connor, L.; Lanagan, C.; Pupo, G.M.; Tembe, V.; Carter, C.D.; O’Rourke, M.; et al. Identification of tfg (trk-fused gene) as a putative metastatic melanoma tumor suppressor gene. Genes Chromosomes Cancer 2012, 51, 452–461. [Google Scholar] [CrossRef]
- Visone, R.; Russo, L.; Pallante, P.; De Martino, I.; Ferraro, A.; Leone, V.; Borbone, E.; Petrocca, F.; Alder, H.; Croce, C.M.; et al. Micrornas (mir)-221 and mir-222, both overexpressed in human thyroid papillary carcinomas, regulate p27kip1 protein levels and cell cycle. Endocr. Relat. Cancer 2007, 14, 791–798. [Google Scholar] [CrossRef]
- Zheng, G.; Li, N.; Jia, X.; Peng, C.; Luo, L.; Deng, Y.; Yin, J.; Song, Y.; Liu, H.; Lu, M.; et al. Mycn-mediated mir-21 overexpression enhances chemo-resistance via targeting cadm1 in tongue cancer. J. Mol. Med. 2016, 94, 1129–1141. [Google Scholar] [CrossRef]
- Zhang, G.; Zhong, L.; Luo, H.; Wang, S. Microrna-155-3p promotes breast cancer progression through down-regulating cadm1. Onco Targets Ther. 2019, 12, 7993–8002. [Google Scholar] [CrossRef]
- Khalighfard, S.; Alizadeh, A.M.; Irani, S.; Omranipour, R. Plasma mir-21, mir-155, mir-10b, and let-7a as the potential biomarkers for the monitoring of breast cancer patients. Sci. Rep. 2018, 8, 17981. [Google Scholar] [CrossRef]
- Natarajan, G.; Ramalingam, S.; Ramachandran, I.; May, R.; Queimado, L.; Houchen, C.W.; Anant, S. Cugbp2 downregulation by prostaglandin e2 protects colon cancer cells from radiation-induced mitotic catastrophe. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 294, G1235–G1244. [Google Scholar] [CrossRef]
- Yeung, Y.T.; Fan, S.; Lu, B.; Yin, S.; Yang, S.; Nie, W.; Wang, M.; Zhou, L.; Li, T.; Li, X.; et al. Celf2 suppresses non-small cell lung carcinoma growth by inhibiting the prex2-pten interaction. Carcinogenesis 2020, 41, 377–389. [Google Scholar] [CrossRef]
- Subramaniam, D.; Ramalingam, S.; Linehan, D.C.; Dieckgraefe, B.K.; Postier, R.G.; Houchen, C.W.; Jensen, R.A.; Anant, S. Rna binding protein cugbp2/celf2 mediates curcumin-induced mitotic catastrophe of pancreatic cancer cells. PLoS ONE 2011, 6, e16958. [Google Scholar] [CrossRef]
- Lai, Z.Q.; Ip, S.P.; Liao, H.J.; Lu, Z.; Xie, J.H.; Su, Z.R.; Chen, Y.L.; Xian, Y.F.; Leung, P.S.; Lin, Z.X. Brucein d, a naturally occurring tetracyclic triterpene quassinoid, induces apoptosis in pancreatic cancer through ros-associated pi3k/akt signaling pathway. Front. Pharmacol. 2017, 8, 936. [Google Scholar] [CrossRef]
- Xu, L.N.; Zhao, N.; Chen, J.Y.; Ye, P.P.; Nan, X.W.; Zhou, H.H.; Jiang, Q.W.; Yang, Y.; Huang, J.R.; Yuan, M.L.; et al. Celastrol inhibits the growth of ovarian cancer cells in vitro and in vivo. Front. Oncol. 2019, 9, 2. [Google Scholar] [CrossRef]
- Lee, H.W.; Jang, K.S.; Choi, H.J.; Jo, A.; Cheong, J.H.; Chun, K.H. Celastrol inhibits gastric cancer growth by induction of apoptosis and autophagy. BMB Rep. 2014, 47, 697–702. [Google Scholar] [CrossRef]
- Tang, Y.C.; Zhang, Y.; Zhou, J.; Zhi, Q.; Wu, M.Y.; Gong, F.R.; Shen, M.; Liu, L.; Tao, M.; Shen, B.; et al. Ginsenoside rg3 targets cancer stem cells and tumor angiogenesis to inhibit colorectal cancer progression in vivo. Int. J. Oncol. 2018, 52, 127–138. [Google Scholar] [CrossRef]
- Li, T.; Sun, W.; Dong, X.; Yu, W.; Cai, J.; Yuan, Q.; Shan, L.; Efferth, T. Total ginsenosides of chinese ginseng induces cell cycle arrest and apoptosis in colorectal carcinoma ht-29 cells. Oncol. Lett. 2018, 16, 4640–4648. [Google Scholar] [CrossRef]
- Chung, K.S.; Cho, S.H.; Shin, J.S.; Kim, D.H.; Choi, J.H.; Choi, S.Y.; Rhee, Y.K.; Hong, H.D.; Lee, K.T. Ginsenoside rh2 induces cell cycle arrest and differentiation in human leukemia cells by upregulating tgf-beta expression. Carcinogenesis 2013, 34, 331–340. [Google Scholar] [CrossRef]
- Singha, P.K.; Pandeswara, S.; Geng, H.; Lan, R.; Venkatachalam, M.A.; Dobi, A.; Srivastava, S.; Saikumar, P. Increased smad3 and reduced smad2 levels mediate the functional switch of tgf-beta from growth suppressor to growth and metastasis promoter through tmepai/pmepa1 in triple negative breast cancer. Genes Cancer 2019, 10, 134–149. [Google Scholar] [CrossRef]
- Petersen, M.; Pardali, E.; van der Horst, G.; Cheung, H.; van den Hoogen, C.; van der Pluijm, G.; Ten Dijke, P. Smad2 and smad3 have opposing roles in breast cancer bone metastasis by differentially affecting tumor angiogenesis. Oncogene 2010, 29, 1351–1361. [Google Scholar] [CrossRef]
- Lu, S.; Zhou, C.; Zou, B.; Zhang, H.; Feng, M. Mir-4295 facilitates cell proliferation and metastasis in head and neck squamous cell carcinoma by targeting nptx1. Genes Immun. 2020, 21, 4–12. [Google Scholar] [CrossRef]
- Yan, R.; Li, K.; Yuan, D.W.; Wang, H.N.; Zhang, Y.; Dang, C.X.; Zhu, K. Downregulation of microrna-4295 enhances cisplatin-induced gastric cancer cell apoptosis through the egfr/pi3k/akt signaling pathway by targeting lrig1. Int. J. Oncol. 2018, 53, 2566–2578. [Google Scholar] [CrossRef]
- Zeng, G.; Xun, W.; Wei, K.; Yang, Y.; Shen, H. Microrna-27a-3p regulates epithelial to mesenchymal transition via targeting yap1 in oral squamous cell carcinoma cells. Oncol. Rep. 2016, 36, 1475–1482. [Google Scholar] [CrossRef]
- Kannan, M.; Jayamohan, S.; Moorthy, R.K.; Chabattula, S.C.; Ganeshan, M.; Arockiam, A.J.V. Aeg-1/mir-221 axis cooperatively regulates the progression of hepatocellular carcinoma by targeting pten/pi3k/akt signaling pathway. Int. J. Mol. Sci. 2019, 20, 5526. [Google Scholar] [CrossRef]
- Park, J.K.; Kogure, T.; Nuovo, G.J.; Jiang, J.; He, L.; Kim, J.H.; Phelps, M.A.; Papenfuss, T.L.; Croce, C.M.; Patel, T.; et al. Mir-221 silencing blocks hepatocellular carcinoma and promotes survival. Cancer Res. 2011, 71, 7608–7616. [Google Scholar] [CrossRef]
- Suehiro, S.; Ohnishi, T.; Yamashita, D.; Kohno, S.; Inoue, A.; Nishikawa, M.; Ohue, S.; Tanaka, J.; Kunieda, T. Enhancement of antitumor activity by using 5-ala-mediated sonodynamic therapy to induce apoptosis in malignant gliomas: Significance of high-intensity focused ultrasound on 5-ala-sdt in a mouse glioma model. J. Neurosurg. 2018, 129, 1416–1428. [Google Scholar] [CrossRef]
- Austin, E.; Koo, E.; Jagdeo, J. Thermal photodynamic therapy increases apoptosis and reactive oxygen species generation in cutaneous and mucosal squamous cell carcinoma cells. Sci. Rep. 2018, 8, 12599. [Google Scholar] [CrossRef]
- Tsai, W.C.; Hsu, P.W.; Lai, T.C.; Chau, G.Y.; Lin, C.W.; Chen, C.M.; Lin, C.D.; Liao, Y.L.; Wang, J.L.; Chau, Y.P.; et al. Microrna-122, a tumor suppressor microrna that regulates intrahepatic metastasis of hepatocellular carcinoma. Hepatology 2009, 49, 1571–1582. [Google Scholar] [CrossRef]
- Xu, Q.; Zhang, M.; Tu, J.; Pang, L.; Cai, W.; Liu, X. Microrna-122 affects cell aggressiveness and apoptosis by targeting pkm2 in human hepatocellular carcinoma. Oncol. Rep. 2015, 34, 2054–2064. [Google Scholar] [CrossRef]
- Slabakova, E.; Culig, Z.; Remsik, J.; Soucek, K. Alternative mechanisms of mir-34a regulation in cancer. Cell Death Dis. 2017, 8, e3100. [Google Scholar] [CrossRef]
- Katz, E.; Nisani, S.; Chamovitz, D.A. Indole-3-carbinol: A plant hormone combatting cancer. F1000Research 2018, 7, 689. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, X.; Cheng, R.; Yang, F.; Yu, M.; Wang, C.; Cui, S.; Hong, Y.; Liang, H.; Liu, M.; et al. The jun/mir-22/hur regulatory axis contributes to tumourigenesis in colorectal cancer. Mol. Cancer 2018, 17, 11. [Google Scholar] [CrossRef]
- Yamakuchi, M.; Yagi, S.; Ito, T.; Lowenstein, C.J. Microrna-22 regulates hypoxia signaling in colon cancer cells. PLoS ONE 2011, 6, e20291. [Google Scholar] [CrossRef]
- Wang, J.; Li, Y.; Ding, M.; Zhang, H.; Xu, X.; Tang, J. Molecular mechanisms and clinical applications of mir-22 in regulating malignant progression in human cancer (review). Int. J. Oncol. 2017, 50, 345–355. [Google Scholar] [CrossRef]
- Cimmino, A.; Calin, G.A.; Fabbri, M.; Iorio, M.V.; Ferracin, M.; Shimizu, M.; Wojcik, S.E.; Aqeilan, R.I.; Zupo, S.; Dono, M.; et al. Mir-15 and mir-16 induce apoptosis by targeting bcl2. Proc. Natl. Acad. Sci. USA 2005, 102, 13944–13949. [Google Scholar] [CrossRef]
- Shi, L.; Jackstadt, R.; Siemens, H.; Li, H.; Kirchner, T.; Hermeking, H. P53-induced mir-15a/16-1 and ap4 form a double-negative feedback loop to regulate epithelial-mesenchymal transition and metastasis in colorectal cancer. Cancer Res. 2014, 74, 532–542. [Google Scholar] [CrossRef]
- Cheng, B.; Ding, F.; Huang, C.Y.; Xiao, H.; Fei, F.Y.; Li, J. Role of mir-16-5p in the proliferation and metastasis of hepatocellular carcinoma. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 137–145. [Google Scholar]
- Li, W.; Ge, Z.; Liu, C.; Liu, Z.; Bjorkholm, M.; Jia, J.; Xu, D. Cip2a is overexpressed in gastric cancer and its depletion leads to impaired clonogenicity, senescence, or differentiation of tumor cells. Clin. Cancer Res. 2008, 14, 3722–3728. [Google Scholar] [CrossRef]
- Li, X.; Yuan, J.; Cao, Q.; Xie, A.; Chen, J. Microrna3835p inhibits the proliferation and promotes the apoptosis of gastric cancer cells by targeting cancerous inhibitor of pp2a. Int. J. Mol. Med. 2020, 46, 397–405. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, K. Mir-383 down-regulates the oncogene cip2a to influence glioma proliferation and invasion. Onco Targets Ther. 2020, 13, 4063–4074. [Google Scholar] [CrossRef]
- Naumann, P.; Liermann, J.; Fortunato, F.; Schmid, T.E.; Weber, K.J.; Debus, J.; Combs, S.E. Sulforaphane enhances irradiation effects in terms of perturbed cell cycle progression and increased DNA damage in pancreatic cancer cells. PLoS ONE 2017, 12, e0180940. [Google Scholar] [CrossRef]
- Xiong, K.; Shao, L.H.; Zhang, H.Q.; Jin, L.; Wei, W.; Dong, Z.; Zhu, Y.Q.; Wu, N.; Jin, S.Z.; Xue, L.X. Microrna-9 functions as a tumor suppressor and enhances radio-sensitivity in radio-resistant a549 cells by targeting neuropilin 1. Oncol. Lett. 2018, 15, 2863–2870. [Google Scholar] [CrossRef]
- Ke, K.; Lou, T. Microrna-10a suppresses breast cancer progression via pi3k/akt/mtor pathway. Oncol. Lett. 2017, 14, 5994–6000. [Google Scholar] [CrossRef]
- Ryoo, I.G.; Kim, G.; Choi, B.H.; Lee, S.H.; Kwak, M.K. Involvement of nrf2 signaling in doxorubicin resistance of cancer stem cell-enriched colonospheres. Biomol. Ther. (Seoul) 2016, 24, 482–488. [Google Scholar] [CrossRef]
- DeWaal, D.; Nogueira, V.; Terry, A.R.; Patra, K.C.; Jeon, S.M.; Guzman, G.; Au, J.; Long, C.P.; Antoniewicz, M.R.; Hay, N. Hexokinase-2 depletion inhibits glycolysis and induces oxidative phosphorylation in hepatocellular carcinoma and sensitizes to metformin. Nat. Commun. 2018, 9, 446. [Google Scholar] [CrossRef]
- Sun, Y.; Chen, C.; Zhang, P.; Xie, H.; Hou, L.; Hui, Z.; Xu, Y.; Du, Q.; Zhou, X.; Su, B.; et al. Reduced mir-3127-5p expression promotes nsclc proliferation/invasion and contributes to dasatinib sensitivity via the c-abl/ras/erk pathway. Sci. Rep. 2014, 4, 6527. [Google Scholar] [CrossRef]
- Hsieh, C.Y.; Tsai, P.C.; Chu, C.L.; Chang, F.R.; Chang, L.S.; Wu, Y.C.; Lin, S.R. Brazilein suppresses migration and invasion of mda-mb-231 breast cancer cells. Chem. Biol. Interact. 2013, 204, 105–115. [Google Scholar] [CrossRef]
- Handayani, S.; Susidarti, R.A.; Jenie, R.I.; Meiyanto, E. Two active compounds from caesalpinia sappan l. In combination with cisplatin synergistically induce apoptosis and cell cycle arrest on widr cells. Adv. Pharm. Bull. 2017, 7, 375–380. [Google Scholar] [CrossRef]
- Imani, S.; Wu, R.C.; Fu, J. Microrna-34 family in breast cancer: From research to therapeutic potential. J. Cancer 2018, 9, 3765–3775. [Google Scholar] [CrossRef]
- Wang, D.; Ren, J.; Ren, H.; Fu, J.L.; Yu, D. Microrna-132 suppresses cell proliferation in human breast cancer by directly targeting foxa1. Acta Pharmacol. Sin. 2018, 39, 124–131. [Google Scholar] [CrossRef]
- Zhang, Z.G.; Chen, W.X.; Wu, Y.H.; Liang, H.F.; Zhang, B.X. Mir-132 prohibits proliferation, invasion, migration, and metastasis in breast cancer by targeting hn1. Biochem. Biophys. Res. Commun. 2014, 454, 109–114. [Google Scholar] [CrossRef]
- Kitada, S.; Leone, M.; Sareth, S.; Zhai, D.; Reed, J.C.; Pellecchia, M. Discovery, characterization, and structure-activity relationships studies of proapoptotic polyphenols targeting b-cell lymphocyte/leukemia-2 proteins. J. Med. Chem. 2003, 46, 4259–4264. [Google Scholar] [CrossRef]
- Fei, B.Y.; He, X.; Ma, J.; Zhang, M.; Chai, R. Foxm1 is associated with metastasis in colorectal cancer through induction of the epithelial-mesenchymal transition. Oncol. Lett. 2017, 14, 6553–6561. [Google Scholar] [CrossRef][Green Version]
- Wei, P.; Zhang, N.; Wang, Y.; Li, D.; Wang, L.; Sun, X.; Shen, C.; Yang, Y.; Zhou, X.; Du, X. Foxm1 promotes lung adenocarcinoma invasion and metastasis by upregulating snail. Int. J. Biol. Sci. 2015, 11, 186–198. [Google Scholar] [CrossRef]
- Lu, C.; Liao, Z.; Cai, M.; Zhang, G. Microrna-320a downregulation mediates human liver cancer cell proliferation through the wnt/beta-catenin signaling pathway. Oncol. Lett. 2017, 13, 573–578. [Google Scholar] [CrossRef]
- Xie, F.; Yuan, Y.; Xie, L.; Ran, P.; Xiang, X.; Huang, Q.; Qi, G.; Guo, X.; Xiao, C.; Zheng, S. Mirna-320a inhibits tumor proliferation and invasion by targeting c-myc in human hepatocellular carcinoma. Onco Targets Ther. 2017, 10, 885–894. [Google Scholar] [CrossRef]
- Imran, M.; Salehi, B.; Sharifi-Rad, J.; Aslam Gondal, T.; Saeed, F.; Imran, A.; Shahbaz, M.; Tsouh Fokou, P.V.; Umair Arshad, M.; Khan, H.; et al. Kaempferol: A key emphasis to its anticancer potential. Molecules 2019, 24, 2277. [Google Scholar] [CrossRef]
- Kim, T.W.; Lee, S.Y.; Kim, M.; Cheon, C.; Ko, S.G. Kaempferol induces autophagic cell death via ire1-jnk-chop pathway and inhibition of g9a in gastric cancer cells. Cell Death Dis. 2018, 9, 875. [Google Scholar] [CrossRef]
- Wang, X.; Song, Y. Microrna-340 inhibits the growth and invasion of angiosarcoma cells by targeting sirt7. Biomed. Pharmacother. 2018, 103, 1061–1068. [Google Scholar] [CrossRef]
- Li, H.; Tian, Z.; Qu, Y.; Yang, Q.; Guan, H.; Shi, B.; Ji, M.; Hou, P. Sirt7 promotes thyroid tumorigenesis through phosphorylation and activation of akt and p70s6k1 via dbc1/sirt1 axis. Oncogene 2019, 38, 345–359. [Google Scholar] [CrossRef]
- Wang, J.; Liu, Z.; Hu, T.; Han, L.; Yu, S.; Yao, Y.; Ruan, Z.; Tian, T.; Huang, T.; Wang, M.; et al. Nrf2 promotes progression of non-small cell lung cancer through activating autophagy. Cell Cycle 2017, 16, 1053–1062. [Google Scholar] [CrossRef]
- Jiang, W.; Xu, Z.; Yu, L.; Che, J.; Zhang, J.; Yang, J. Microrna-144-3p suppressed tgf-beta1-induced lung cancer cell invasion and adhesion by regulating the src-akt-erk pathway. Cell Biol. Int. 2019, 44, 51–61. [Google Scholar] [CrossRef]
- Zhao, Y.; Xie, Z.; Lin, J.; Liu, P. Mir-144-3p inhibits cell proliferation and induces apoptosis in multiple myeloma by targeting c-met. Am. J. Transl. Res. 2017, 9, 2437–2446. [Google Scholar]
- Elahouel, R.; Blanc, C.; Carpentier, G.; Frechault, S.; Cascone, I.; Destouches, D.; Delbe, J.; Courty, J.; Hamma-Kourbali, Y. Pleiotrophin exerts its migration and invasion effect through the neuropilin-1 pathway. Neoplasia 2015, 17, 613–624. [Google Scholar] [CrossRef]
- Song, L.; Gong, H.; Lin, C.; Wang, C.; Liu, L.; Wu, J.; Li, M.; Li, J. Flotillin-1 promotes tumor necrosis factor-alpha receptor signaling and activation of nf-kappab in esophageal squamous cell carcinoma cells. Gastroenterology 2012, 143, 995–1005.e12. [Google Scholar] [CrossRef]
- Wu, Z.; Owens, C.; Chandra, N.; Popovic, K.; Conaway, M.; Theodorescu, D. Ralbp1 is necessary for metastasis of human cancer cell lines. Neoplasia 2010, 12, 1003–1012. [Google Scholar] [CrossRef]
- Lee, S.; Wurtzel, J.G.; Singhal, S.S.; Awasthi, S.; Goldfinger, L.E. Ralbp1/rlip76 depletion in mice suppresses tumor growth by inhibiting tumor neovascularization. Cancer Res. 2012, 72, 5165–5173. [Google Scholar] [CrossRef]
- Stuckler, D.; Singhal, J.; Singhal, S.S.; Yadav, S.; Awasthi, Y.C.; Awasthi, S. Rlip76 transports vinorelbine and mediates drug resistance in non-small cell lung cancer. Cancer Res. 2005, 65, 991–998. [Google Scholar]
- Liu, N.; Du, C.H. Rlip76 silencing inhibits cell proliferation and invasion in melanoma cell line a375. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 2054–2060. [Google Scholar]
- Pham, T.N.D.; Stempel, S.; Shields, M.A.; Spaulding, C.; Kumar, K.; Bentrem, D.J.; Matsangou, M.; Munshi, H.G. Quercetin enhances the anti-tumor effects of bet inhibitors by suppressing hnrnpa1. Int. J. Mol. Sci. 2019, 20, 4293. [Google Scholar] [CrossRef]
- Tummala, R.; Lou, W.; Gao, A.C.; Nadiminty, N. Quercetin targets hnrnpa1 to overcome enzalutamide resistance in prostate cancer cells. Mol. Cancer. Ther. 2017, 16, 2770–2779. [Google Scholar] [CrossRef]
- Puca, F.; Tosti, N.; Federico, A.; Kuzay, Y.; Pepe, A.; Morlando, S.; Savarese, T.; D’Alessio, F.; Colamaio, M.; Sarnataro, D.; et al. Hmga1 negatively regulates numb expression at transcriptional and post transcriptional level in glioblastoma stem cells. Cell Cycle 2019, 18, 1446–1457. [Google Scholar] [CrossRef]
- Akaboshi, S.; Watanabe, S.; Hino, Y.; Sekita, Y.; Xi, Y.; Araki, K.; Yamamura, K.; Oshima, M.; Ito, T.; Baba, H.; et al. Hmga1 is induced by wnt/beta-catenin pathway and maintains cell proliferation in gastric cancer. Am. J. Pathol. 2009, 175, 1675–1685. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Xu, C.; Tang, S.C.; Wang, J.; Wang, H.; Wang, P.; Du, N.; Qin, S.; Li, G.; Xu, S.; et al. Let-7c blocks estrogen-activated wnt signaling in induction of self-renewal of breast cancer stem cells. Cancer Gene Ther. 2016, 23, 83–89. [Google Scholar] [CrossRef]
- Nadiminty, N.; Tummala, R.; Liu, C.; Lou, W.; Evans, C.P.; Gao, A.C. Nf-kappab2/p52:C-myc:Hnrnpa1 pathway regulates expression of androgen receptor splice variants and enzalutamide sensitivity in prostate cancer. Mol. Cancer Ther. 2015, 14, 1884–1895. [Google Scholar] [CrossRef] [PubMed]
- Yamamura, S.; Saini, S.; Majid, S.; Hirata, H.; Ueno, K.; Deng, G.; Dahiya, R. Microrna-34a modulates c-myc transcriptional complexes to suppress malignancy in human prostate cancer cells. PLoS ONE 2012, 7, e29722. [Google Scholar] [CrossRef] [PubMed]
- Teng, K.Y.; Barajas, J.M.; Hu, P.; Jacob, S.T.; Ghoshal, K. Role of b cell lymphoma 2 in the regulation of liver fibrosis in mir-122 knockout mice. Biology (Basel) 2020, 9, 157. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Xu, Z.; Li, C.; Xu, C.; Lei, Z.; Zhang, H.T.; Zhao, J. Mir-145 and mir-203 represses tgf-beta-induced epithelial-mesenchymal transition and invasion by inhibiting smad3 in non-small cell lung cancer cells. Lung Cancer 2016, 97, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Liang, H.; Liao, Z.; Wang, Y.; Hu, X.; Chen, X.; Xu, L.; Hu, Z. Mir-203 enhances let-7 biogenesis by targeting lin28b to suppress tumor growth in lung cancer. Sci. Rep. 2017, 7, 42680. [Google Scholar] [CrossRef] [PubMed]
- Paranjape, A.N.; Balaji, S.A.; Mandal, T.; Krushik, E.V.; Nagaraj, P.; Mukherjee, G.; Rangarajan, A. Bmi1 regulates self-renewal and epithelial to mesenchymal transition in breast cancer cells through nanog. BMC Cancer 2014, 14, 785. [Google Scholar] [CrossRef]
- Rao, P.K.; Missiaglia, E.; Shields, L.; Hyde, G.; Yuan, B.; Shepherd, C.J.; Shipley, J.; Lodish, H.F. Distinct roles for mir-1 and mir-133a in the proliferation and differentiation of rhabdomyosarcoma cells. FASEB J. 2010, 24, 3427–3437. [Google Scholar] [CrossRef] [PubMed]
- Taulli, R.; Bersani, F.; Foglizzo, V.; Linari, A.; Vigna, E.; Ladanyi, M.; Tuschl, T.; Ponzetto, C. The muscle-specific microrna mir-206 blocks human rhabdomyosarcoma growth in xenotransplanted mice by promoting myogenic differentiation. J. Clin. Invest. 2009, 119, 2366–2378. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.Q.; Yao, C.; Bian, W.H.; Chen, X.; Xue, J.X.; Zhu, Z.Y.; Ying, Y.; Xu, Y.L.; Wang, C. Effects of astragaloside iv on treatment of breast cancer cells execute possibly through regulation of nrf2 via pi3k/akt/mtor signaling pathway. Food Sci. Nutr. 2019, 7, 3403–3413. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.X.; Shi, L.; Zhang, Y.; Zhu, Y.C.; Bai, C.X.; Wang, X.D.; Zhou, J.B. Myocyte enhancer factor 2d provides a cross-talk between chronic inflammation and lung cancer. J. Transl. Med. 2017, 15, 65. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Wang, Y.; Lin, Y.; Liu, Y.; Wang, Y.; Jia, J.; Singh, P.; Chi, Y.I.; Wang, C.; Dong, C.; et al. Dub3 inhibition suppresses breast cancer invasion and metastasis by promoting snail1 degradation. Nat. Commun. 2017, 8, 14228. [Google Scholar] [CrossRef] [PubMed]
- Tang, J.; Zhong, G.; Wu, J.; Chen, H.; Jia, Y. Sox2 recruits klf4 to regulate nasopharyngeal carcinoma proliferation via pi3k/akt signaling. Oncogenesis 2018, 7, 61. [Google Scholar] [CrossRef] [PubMed]
- Kfoury, A.; Le Corf, K.; El Sabeh, R.; Journeaux, A.; Badran, B.; Hussein, N.; Lebecque, S.; Manie, S.; Renno, T.; Coste, I. Myd88 in DNA repair and cancer cell resistance to genotoxic drugs. J. Natl. Cancer Inst. 2013, 105, 937–946. [Google Scholar] [CrossRef] [PubMed]
- Echizen, K.; Hirose, O.; Maeda, Y.; Oshima, M. Inflammation in gastric cancer: Interplay of the cox-2/prostaglandin e2 and toll-like receptor/myd88 pathways. Cancer Sci. 2016, 107, 391–397. [Google Scholar] [CrossRef]
- Phuah, N.H.; Azmi, M.N.; Awang, K.; Nagoor, N.H. Down-regulation of microrna-210 confers sensitivity towards 1’s-1’-acetoxychavicol acetate (aca) in cervical cancer cells by targeting smad4. Mol. Cells 2017, 40, 291–298. [Google Scholar] [CrossRef]
- Phuah, N.H.; Azmi, M.N.; Awang, K.; Nagoor, N.H. Suppression of microrna-629 enhances sensitivity of cervical cancer cells to 1’s-1’-acetoxychavicol acetate via regulating rsu1. Onco Targets Ther. 2017, 10, 1695–1705. [Google Scholar] [CrossRef]
- Chakrabarti, M.; Ray, S.K. Anti-tumor activities of luteolin and silibinin in glioblastoma cells: Overexpression of mir-7-1-3p augmented luteolin and silibinin to inhibit autophagy and induce apoptosis in glioblastoma in vivo. Apoptosis 2016, 21, 312–328. [Google Scholar] [CrossRef]
- Xue, C.L.; Liu, H.G.; Li, B.Y.; He, S.H.; Yue, Q.F. Physcion 8-o-beta-glucopyranoside exhibits anti-growth and anti-metastatic activities in ovarian cancer by downregulating mir-25. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 5101–5112. [Google Scholar]
- Jiang, L.; Tao, C.; He, A.; He, X. Overexpression of mir-126 sensitizes osteosarcoma cells to apoptosis induced by epigallocatechin-3-gallate. World J. Surg. Oncol. 2014, 12, 383. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, M.; Ray, S.K. Direct transfection of mir-137 mimics is more effective than DNA demethylation of mir-137 promoter to augment anti-tumor mechanisms of delphinidin in human glioblastoma u87mg and ln18 cells. Gene 2015, 573, 141–152. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, M.; Banik, N.L.; Ray, S.K. Mir-138 overexpression is more powerful than htert knockdown to potentiate apigenin for apoptosis in neuroblastoma in vitro and in vivo. Exp. Cell Res. 2013, 319, 1575–1585. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Li, H.; Long, L.; Hui, L.; Chen, H.; Wang, X.; Shen, H.; Xu, W. Mir-126 enhances the sensitivity of non-small cell lung cancer cells to anticancer agents by targeting vascular endothelial growth factor A. Acta Biochim. Biophys. Sin. (Shanghai) 2012, 44, 519–526. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Zhou, J.; Zhu, X.; Yuan, H. Mir-126 reverses drug resistance to trail through inhibiting the expression of c-flip in cervical cancer. Gene 2017, 627, 420–427. [Google Scholar] [CrossRef]
- Lechman, E.R.; Gentner, B.; Ng, S.W.; Schoof, E.M.; van Galen, P.; Kennedy, J.A.; Nucera, S.; Ciceri, F.; Kaufmann, K.B.; Takayama, N.; et al. Mir-126 regulates distinct self-renewal outcomes in normal and malignant hematopoietic stem cells. Cancer Cell 2016, 29, 214–228. [Google Scholar] [CrossRef]
- Zhang, H.; Zuo, Z.; Lu, X.; Wang, L.; Wang, H.; Zhu, Z. Mir-25 regulates apoptosis by targeting bim in human ovarian cancer. Oncol. Rep. 2012, 27, 594–598. [Google Scholar]
- Wang, F.; Mayca Pozo, F.; Tian, D.; Geng, X.; Yao, X.; Zhang, Y.; Tang, J. Shikonin inhibits cancer through p21 upregulation and apoptosis induction. Front. Pharmacol. 2020, 11, 861. [Google Scholar] [CrossRef]
- Kim, H.J.; Hwang, K.E.; Park, D.S.; Oh, S.H.; Jun, H.Y.; Yoon, K.H.; Jeong, E.T.; Kim, H.R.; Kim, Y.S. Shikonin-induced necroptosis is enhanced by the inhibition of autophagy in non-small cell lung cancer cells. J. Transl. Med. 2017, 15, 123. [Google Scholar] [CrossRef]
- Wiench, B.; Eichhorn, T.; Paulsen, M.; Efferth, T. Shikonin directly targets mitochondria and causes mitochondrial dysfunction in cancer cells. Evid. Based Complement. Alternat. Med. 2012, 2012, 726025. [Google Scholar] [CrossRef]
- Chen, J.C.; Hsieh, Y.Y.; Lo, H.L.; Li, A.; Chou, C.J.; Yang, P.M. In vitro and in silico mechanistic insights into mir-21-5p-mediated topoisomerase drug resistance in human colorectal cancer cells. Biomolecules 2019, 9, 467. [Google Scholar] [CrossRef] [PubMed]
- Ni, W.; Luo, L.; Zuo, P.; Li, R.; Xu, X.; Wen, F.; Hu, D. Mir-374a inhibitor enhances etoposide-induced cytotoxicity against glioma cells through upregulation of foxo1. Oncol. Res. 2019, 27, 703–712. [Google Scholar] [CrossRef] [PubMed]
- Kollinerova, S.; Dostal, Z.; Modriansky, M. Microrna hsa-mir-29b potentiates etoposide toxicity in hela cells via down-regulation of mcl-1. Toxicol. In Vitro 2017, 40, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhao, F.; Cai, S.; Pu, Y. Mir-193a regulates chemoresistance of human osteosarcoma cells via repression of irs2. J. Bone Oncol. 2019, 17, 100241. [Google Scholar] [CrossRef]
- Rebucci, M.; Sermeus, A.; Leonard, E.; Delaive, E.; Dieu, M.; Fransolet, M.; Arnould, T.; Michiels, C. Mirna-196b inhibits cell proliferation and induces apoptosis in hepg2 cells by targeting igf2bp1. Mol. Cancer 2015, 14, 79. [Google Scholar] [CrossRef]
- Tao, L.; Wu, Y.Q.; Zhang, S.P. Mir-21-5p enhances the progression and paclitaxel resistance in drug-resistant breast cancer cell lines by targeting pdcd4. Neoplasma 2019, 66, 746–755. [Google Scholar] [CrossRef]
- Feng, L.; Shen, F.; Zhou, J.; Li, Y.; Jiang, R.; Chen, Y. Hypoxia-induced up-regulation of mir-27a promotes paclitaxel resistance in ovarian cancer. Biosci. Rep. 2020, 40, BSR20192457. [Google Scholar] [CrossRef]
- Li, Z.; Hu, S.; Wang, J.; Cai, J.; Xiao, L.; Yu, L.; Wang, Z. Mir-27a modulates mdr1/p-glycoprotein expression by targeting hipk2 in human ovarian cancer cells. Gynecol. Oncol. 2010, 119, 125–130. [Google Scholar] [CrossRef]
- Zhou, M.; Liu, Z.; Zhao, Y.; Ding, Y.; Liu, H.; Xi, Y.; Xiong, W.; Li, G.; Lu, J.; Fodstad, O.; et al. Microrna-125b confers the resistance of breast cancer cells to paclitaxel through suppression of pro-apoptotic bcl-2 antagonist killer 1 (bak1) expression. J. Biol. Chem. 2010, 285, 21496–21507. [Google Scholar] [CrossRef]
- Zhao, K.; Li, X.; Chen, X.; Zhu, Q.; Yin, F.; Ruan, Q.; Xia, J.; Niu, Z. Inhibition of mir-140-3p or mir-155-5p by antagomir treatment sensitize chordoma cells to chemotherapy drug treatment by increasing pten expression. Eur. J. Pharmacol. 2019, 854, 298–306. [Google Scholar] [CrossRef]
- Filipska, M.; Skrzypski, M.; Czetyrbok, K.; Stokowy, T.; Stasilojc, G.; Supernat, A.; Jassem, J.; Zaczek, A.J.; Bigda, J. Mir-192 and mir-662 enhance chemoresistance and invasiveness of squamous cell lung carcinoma. Lung Cancer 2018, 118, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Ren, L.L.; Yan, T.T.; Shen, C.Q.; Tang, J.Y.; Kong, X.; Wang, Y.C.; Chen, J.; Liu, Q.; He, J.; Zhong, M.; et al. The distinct role of strand-specific mir-514b-3p and mir-514b-5p in colorectal cancer metastasis. Cell Death Dis. 2018, 9, 687. [Google Scholar] [CrossRef] [PubMed]
- Geng, W.; Song, H.; Zhao, Q.; Dong, K.; Pu, Q.; Gao, H.; Lv, Y. Mir-520h stimulates drug resistance to paclitaxel by targeting the otud3-pten axis in breast cancer. Biomed. Res. Int. 2020, 2020, 9512793. [Google Scholar] [CrossRef] [PubMed]
- Hou, X.; Niu, Z.; Liu, L.; Guo, Q.; Li, H.; Yang, X.; Zhang, X. Mir-1207-5p regulates the sensitivity of triple-negative breast cancer cells to taxol treatment via the suppression of lzts1 expression. Oncol. Lett. 2019, 17, 990–998. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Zhou, X.; Bao, Y.; Xiong, G.; Cui, Y.; Zhou, H. Involvement of mir-4262 in paclitaxel resistance through the regulation of pten in non-small cell lung cancer. Open Biol. 2019, 9, 180227. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.M.; Tan, T.; Zhang, T.Y.; Xiao, X.F.; Gu, H. Mir1 reverses multidrug resistance in gastric cancer cells via downregulation of sorcin through promoting the accumulation of intracellular drugs and apoptosis of cells. Int. J. Oncol. 2019, 55, 451–461. [Google Scholar]
- Hong, T.; Ding, J.; Li, W. Mir-7 reverses breast cancer resistance to chemotherapy by targeting mrp1 and bcl2. Onco Targets Ther. 2019, 12, 11097–11105. [Google Scholar] [CrossRef]
- Huang, L.; Hu, C.; Chao, H.; Wang, R.; Lu, H.; Li, H.; Chen, H. Mir-29c regulates resistance to paclitaxel in nasopharyngeal cancer by targeting itgb1. Exp. Cell Res. 2019, 378, 1–10. [Google Scholar] [CrossRef]
- Yang, G.; Fu, Y.; Lu, X.; Wang, M.; Dong, H.; Li, Q. Mir34a regulates the chemosensitivity of retinoblastoma cells via modulation of magea/p53 signaling. Int. J. Oncol. 2019, 54, 177–187. [Google Scholar]
- Wang, P.; Li, Z.; Liu, H.; Zhou, D.; Fu, A.; Zhang, E. Microrna-126 increases chemosensitivity in drug-resistant gastric cancer cells by targeting ezh2. Biochem. Biophys. Res. Commun. 2016, 479, 91–96. [Google Scholar] [CrossRef]
- Chen, M.; Li, D.; Gong, N.; Wu, H.; Su, C.; Xie, C.; Xiang, H.; Lin, C.; Li, X. Mir-133b down-regulates abcc1 and enhances the sensitivity of crc to anti-tumor drugs. Oncotarget 2017, 8, 52983–52994. [Google Scholar] [CrossRef]
- Guan, X.; Guan, Y. Mir-145-5p attenuates paclitaxel resistance and suppresses the progression in drug-resistant breast cancer cell lines. Neoplasma 2020, 67, 972–981. [Google Scholar] [CrossRef]
- Juang, V.; Chang, C.H.; Wang, C.S.; Wang, H.E.; Lo, Y.L. Ph-responsive peg-shedding and targeting peptide-modified nanoparticles for dual-delivery of irinotecan and microrna to enhance tumor-specific therapy. Small 2019, 15, e1903296. [Google Scholar] [CrossRef]
- Zhu, W.; Xu, H.; Zhu, D.; Zhi, H.; Wang, T.; Wang, J.; Jiang, B.; Shu, Y.; Liu, P. Mir-200bc/429 cluster modulates multidrug resistance of human cancer cell lines by targeting bcl2 and xiap. Cancer Chemother. Pharmacol. 2012, 69, 723–731. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Heng, C.; Li, Y.; Yu, L. Mir-302a sensitizes leukemia cells to etoposide by targeting rad52. Oncotarget 2017, 8, 73884–73891. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Su, K.; Zhang, Y.; Zhang, W.; Chu, D.; Zhao, Q.; Guo, R. Microrna-365 targets multiple oncogenes to inhibit proliferation, invasion, and self-renewal of aggressive endometrial cancer cells. Cancer Manag. Res. 2018, 10, 5171–5185. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Xie, C.; Liu, Y.; Shi, Q.; Chen, Y. Up-regulation of mir-383-5p suppresses proliferation and enhances chemosensitivity in ovarian cancer cells by targeting trim27. Biomed. Pharmacother. 2019, 109, 595–601. [Google Scholar] [CrossRef]
- Chen, S.; Wu, J.; Jiao, K.; Wu, Q.; Ma, J.; Chen, D.; Kang, J.; Zhao, G.; Shi, Y.; Fan, D.; et al. Microrna-495-3p inhibits multidrug resistance by modulating autophagy through grp78/mtor axis in gastric cancer. Cell Death Dis. 2018, 9, 1070. [Google Scholar] [CrossRef]
- Lyu, H.; Wang, S.; Huang, J.; Wang, B.; He, Z.; Liu, B. Survivin-targeting mir-542-3p overcomes her3 signaling-induced chemoresistance and enhances the antitumor activity of paclitaxel against her2-overexpressing breast cancer. Cancer Lett. 2018, 420, 97–108. [Google Scholar] [CrossRef]
- Abdelfattah, N.; Rajamanickam, S.; Panneerdoss, S.; Timilsina, S.; Yadav, P.; Onyeagucha, B.C.; Garcia, M.; Vadlamudi, R.; Chen, Y.; Brenner, A.; et al. Mir-584-5p potentiates vincristine and radiation response by inducing spindle defects and DNA damage in medulloblastoma. Nat. Commun. 2018, 9, 4541. [Google Scholar] [CrossRef]
- Xue, J.; Chi, Y.; Chen, Y.; Huang, S.; Ye, X.; Niu, J.; Wang, W.; Pfeffer, L.M.; Shao, Z.M.; Wu, Z.H.; et al. Mirna-621 sensitizes breast cancer to chemotherapy by suppressing fbxo11 and enhancing p53 activity. Oncogene 2016, 35, 448–458. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Tang, J.; Zhang, L.; Bu, Y.; Zhang, X. Mir-874 regulates multiple-drug resistance in gastric cancer by targeting atg16l1. Int. J. Oncol. 2018, 53, 2769–2779. [Google Scholar] [CrossRef]
- Jia, M.; Wei, Z.; Liu, P.; Zhao, X. Silencing of abcg2 by microrna-3163 inhibits multidrug resistance in retinoblastoma cancer stem cells. J. Korean Med. Sci. 2016, 31, 836–842. [Google Scholar] [CrossRef]
- Kannathasan, T.; Kuo, W.W.; Chen, M.C.; Viswanadha, V.P.; Shen, C.Y.; Tu, C.C.; Yeh, Y.L.; Bharath, M.; Shibu, M.A.; Huang, C.Y. Chemoresistance-associated silencing of mir-4454 promotes colorectal cancer aggression through the gnl3l and nf-kappab pathway. Cancers (Basel) 2020, 12, 1231. [Google Scholar] [CrossRef]
- Liu, M.; Gong, C.; Xu, R.; Chen, Y.; Wang, X. Microrna-5195-3p enhances the chemosensitivity of triple-negative breast cancer to paclitaxel by downregulating eif4a2. Cell Mol. Biol. Lett. 2019, 24, 47. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, A.; Fernandes, M.; Catarino, R.; Medeiros, R. Rad52 functions in homologous recombination and its importance on genomic integrity maintenance and cancer therapy. Cancers (Basel) 2019, 11, 1622. [Google Scholar] [CrossRef] [PubMed]
- Westover, D.; Li, F. New trends for overcoming abcg2/bcrp-mediated resistance to cancer therapies. J. Exp. Clin. Cancer Res. 2015, 34, 159. [Google Scholar] [CrossRef]
- Olszewski, U.; Liedauer, R.; Ausch, C.; Thalhammer, T.; Hamilton, G. Overexpression of cyp3a4 in a colo 205 colon cancer stem cell model in vitro. Cancers 2011, 3, 1467–1479. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; Liu, X. The tumor-suppressor gene lzts1 suppresses hepatocellular carcinoma proliferation by impairing pi3k/akt pathway. Biomed. Pharmacother. 2015, 76, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Aoudjit, F.; Vuori, K. Integrin signaling inhibits paclitaxel-induced apoptosis in breast cancer cells. Oncogene 2001, 20, 4995–5004. [Google Scholar] [CrossRef]
- Ma, L.; Yao, N.; Chen, P.; Zhuang, Z. Trim27 promotes the development of esophagus cancer via regulating pten/akt signaling pathway. Cancer Cell Int. 2019, 19, 283. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, M.E.; Moore, H.M.; Li, X.; Toy, K.A.; Huang, W.; Sabel, M.S.; Kidwell, K.M.; Kleer, C.G. Ezh2 expands breast stem cells through activation of notch1 signaling. Proc. Natl. Acad. Sci. USA 2014, 111, 3098–3103. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, N.; Bhattacharya, S.; Steele, R.; Phillips, N.; Ray, R.B. Involvement of c-fos in the promotion of cancer stem-like cell properties in head and neck squamous cell carcinoma. Clin. Cancer Res. 2017, 23, 3120–3128. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Teng, C.; Huang, W.; Peng, J.; Wang, C. Sox2 promotes the epithelial to mesenchymal transition of esophageal squamous cells by modulating slug expression through the activation of stat3/hif-alpha signaling. Int. J. Mol. Sci. 2015, 16, 21643–21657. [Google Scholar] [CrossRef]
- Yamagishi, N.; Nakao, R.; Kondo, R.; Nishitsuji, M.; Saito, Y.; Kuga, T.; Hatayama, T.; Nakayama, Y. Increased expression of sorcin is associated with multidrug resistance in leukemia cells via up-regulation of mdr1 expression through camp response element-binding protein. Biochem. Biophys. Res. Commun. 2014, 448, 430–436. [Google Scholar] [CrossRef]
- Xu, P.; Jiang, Y.F.; Wang, J.H. Shrna-mediated silencing of sorcin increases drug chemosensitivity in myeloma km3/ddp and u266/adm cell lines. Int. J. Clin. Exp. Pathol. 2015, 8, 2300–2310. [Google Scholar]
- Nam, J.S.; Sharma, A.R.; Nguyen, L.T.; Chakraborty, C.; Sharma, G.; Lee, S.S. Application of bioactive quercetin in oncotherapy: From nutrition to nanomedicine. Molecules 2016, 21, 108. [Google Scholar] [CrossRef]
- Alles, J.; Fehlmann, T.; Fischer, U.; Backes, C.; Galata, V.; Minet, M.; Hart, M.; Abu-Halima, M.; Grasser, F.A.; Lenhof, H.P.; et al. An estimate of the total number of true human mirnas. Nucleic Acids Res. 2019, 47, 3353–3364. [Google Scholar] [CrossRef]
- Matarlo, J.S.; Krumpe, L.R.H.; Heinz, W.F.; Oh, D.; Shenoy, S.R.; Thomas, C.L.; Goncharova, E.I.; Lockett, S.J.; O’Keefe, B.R. The natural product butylcycloheptyl prodiginine binds pre-mir-21, inhibits dicer-mediated processing of pre-mir-21, and blocks cellular proliferation. Cell Chem. Biol. 2019, 26, 1133–1142.e4. [Google Scholar] [CrossRef]
- Li, Z.; Rana, T.M. Therapeutic targeting of micrornas: Current status and future challenges. Nat. Rev. Drug Discov. 2014, 13, 622–638. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
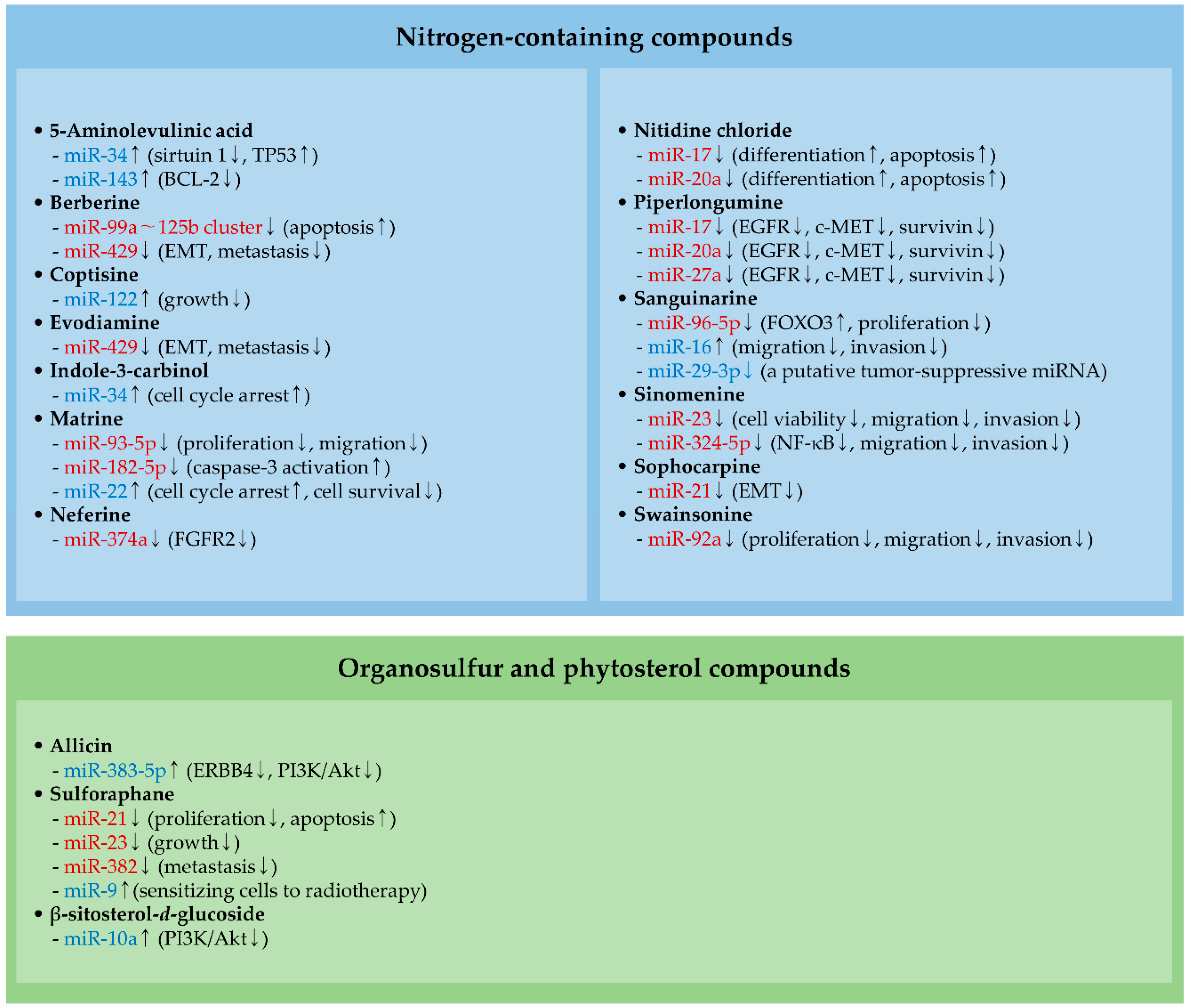


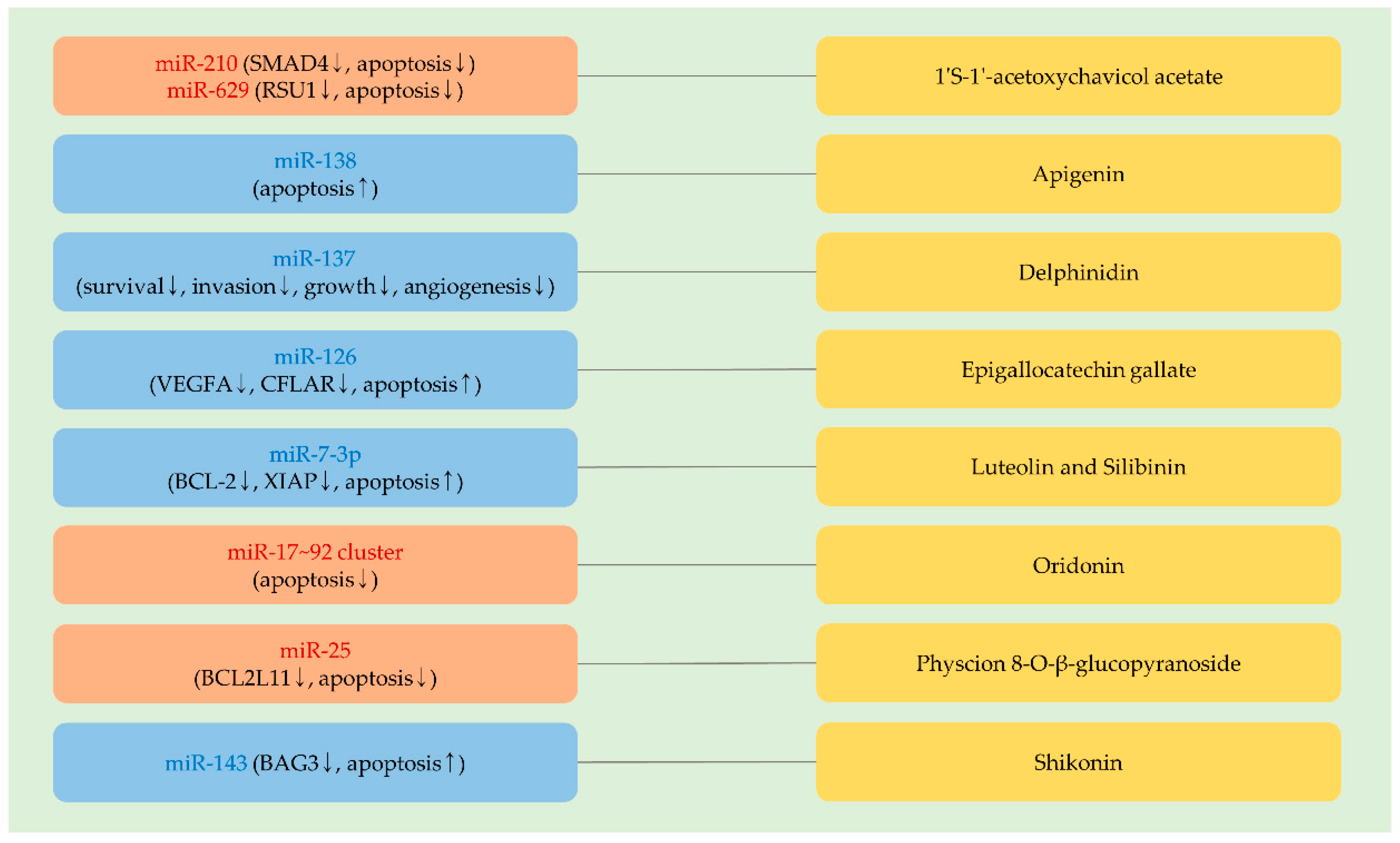

| Phytochemical | Source of Phytochemical (NCT Number, Condition/Disease, Recruitment Status if Applicable) | Ref. |
|---|---|---|
| Nitrogen-containing compounds (alkaloids and amino acids) | ||
| 5-Aminolevulinic acid * | A precursor of tetrapyrroles in higher plants (NCT02144077, basal cell carcinoma, active) | [16] |
| Berberine * | An isoquinoline alkaloid in plants, such as barberry and goldenseal (NCT03281096, colorectal adenomas, recruiting) | [17] |
| Coptisine | A protoberberine alkaloid in Rhizoma coptidis | [18] |
| Evodiamine | An indoloquinazoline alkaloid in Evodia rutaecarpa | [19] |
| Indole-3-carbinol | An indole alkaloid in vegetables such as cauliflower | [20] |
| Matrine | A quinolizidine alkaloid in the root of Sophora flavescens | [21] |
| Neferine | A bisbenzylisoquinline alkaloid in the seed embryo of Nelumbo nucifera | [22] |
| Nitidine chloride | A benzophenanthridine alkaloid in the roots of Zanthoxylum nitidum | [23] |
| Piperlongumine | An alkaloid from Piper longum Linn | [24] |
| Sanguinarine | A benzophenanthridine alkaloid in Papaveraceae plants | [25] |
| Sinomenine | An isoquinoline alkaloid in the dry roots and stems of Sinomenium acutum | [26] |
| Sophocarpine | A tetracyclic quinolizidine alkaloid in Sophora alopecuroides Linn | [27] |
| Swainsonine | An indolizidine alkaloid in Swainsona canescens | [28] |
| Organosulfur and phytosterol compounds | ||
| Allicin | An organosulfur compound from garlic (Allium sativum) | [29] |
| Sulforaphane * | An isothiocyanate abundant in cruciferous vegetables such as broccoli sprouts (NCT03182959, head and neck cancer, active) | [30] |
| β-sitosterol-d-glucoside | A phytosterol from sweet potato | [31] |
| Phenolic compounds | ||
| 1′S-1′-acetoxychavicol acetate | A phenylpropanoid from Alpinia conchigera | [32] |
| Apigenin | A flavone from fruits (e.g., oranges) and vegetables (e.g., onions) | [33] |
| Astragalin | A flavonoid broadly present in food such as lotus leaves | [34] |
| Baicalein(aglycone of baicalin) | A flavonoid from Scutellaria radix | [35] |
| Baicalin | A flavonoid from the root of Scutellaria baicalensis Georgi | [36] |
| Brazilein | A polyphenolic compound from Caesalpinia sappan | [37] |
| Chrysin | A flavone from several plants, including Oroxylum indicum | [38] |
| Curcumin * | A polyphenolic curcuminoid in Curcuma longa (turmeric plant) (NCT02064673, prostate cancer, recruiting) | [39] |
| Delphinidin | A flavonoid in fruits and vegetables such as tomatoes | [40] |
| Epigallocatechin gallate * | A polyphenol in green tea (NCT02891538, colorectal cancer, recruiting) | [41] |
| Formononetin | An isoflavone in the root of Astragalus membranaceus | [42] |
| Galangin | A flavonoid in Alpinia officinarum | [43] |
| Gambogic acid | A xanthonoid from Garcinia hanburyi trees | [44] |
| Genistein * | An isoflavone and phytoestrogen primarily in Soybeans (NCT00118040, bladder cancer, active) | [45] |
| Gossypol (AT-101) * | A polyphenol from cotton roots and seeds (NCT01633541, advanced laryngeal cancer, active) | [46] |
| Hydroxygenkwanin | A flavonoid from Daphne genkwa | [47] |
| Honokiol | A polyphenol in the genus Magnolia | [48] |
| Icariin | A flavonoid in Epimedium (Horny Goat Weed and Yin Yang Huo) | [49] |
| Isoliquiritigenin | A chalcone from Glycyrrhizae radix | [50] |
| Isorhapontigenin | A stilbene from Gnetum cleistostachyum | [51] |
| Kaempferol | A flavonoid in various plants such as Brussels sprouts | [52] |
| Licochalcone A | A chalcone from Glycyrrhiza uralensis | [53] |
| Luteolin | A flavonoid in several vegetables such as cabbage | [54] |
| Physcion 8-O-β-glucopyranoside | An anthraquinone in Rumex japonicus Houtt | [55] |
| Procyanidin | A polyphenol in dietary fruits such as grapes | [56] |
| Puerarin | An isoflavone in the root of Pueraria (Radix puerariae) | [57] |
| Quercetin * | A flavonoid in fruits and vegetables, such as onions and broccoli (NCT01912820, prostate cancer, active) | [58] |
| Resveratrol | A stilbenoid in grapes and red wine, etc | [59] |
| Shikonin | A naphthoquinone from Lithospermum erythrorhizon | [60] |
| Silibinin | A polyphenolic flavonoid in milk thistle (Silybum marianum) | [61,62,63] |
| Silymarin | A mixture of flavonolignans from Silybum marianum L. Gaertn. | [64] |
| Trans-3,5,4′-trimethoxystilbene | A derivative of resveratrol | [65] |
| Terpenoid compounds | ||
| Ailanthone | A quassinoid from Ailanthus altissima | [66] |
| Andrographolide * | A diterpene lactone from Andrographis paniculate (NCT04196075, advanced esophageal cancer, recruiting) | [67] |
| Artemisinin | A sesquiterpene lactone from Artemisia annua | [68] |
| Artesunate * | A derivative of artemisinin (NCT02633098, colorectal cancer, recruiting) | [68] |
| Astragaloside IV | A pentacyclic triterpenoid from Astragali radix | [69] |
| Brucein D | A triterpene quassinoid in Brucea javanica fruit | [70] |
| Cannabidiol * | A terpenophenolic compound from Cannabis sativa (NCT04428203, prostate cancer, recruiting) | [71] |
| Celastrol | A triterpene in Tripterygium wilfordii | [72] |
| Cucurbitacin D | A derivative of cucurbitacin that is a tetracyclic triterpene from the Cucurbitaceae family | [73] |
| Curcumol | A sesquiterpenoid from Rhizoma Curcumae | [74] |
| Ginsenoside Rd and Rh2 | Triterpene saponins in Panax genus | [75,76] |
| Lycopene * | A carotenoid from fruits such as tomatoes (NCT03167268, colorectal cancer, recruiting) | [77] |
| Maytenin and 22-β-hydroxymaytenin | Quinone-methide triterpenes in Maytenus ilicifolia | [78] |
| Oleanolic acid | A pentacyclic triterpene in herbs and vegetables | [79] |
| Oridonin | A diterpenoid in genus Isodon plants | [80] |
| Pristimerin | A triterpenoid from the Celastraceae and Hippocrateaceae families | [81] |
| Toosendanin | A triterpenoid from Melia toosendan Sieb et Zucc | [82] |
| Triptolide * | A diterpene triepoxide in Tripterygium wilfordii Hook F (NCT03129139, advanced solid tumors, recruiting) | [83] |
| Tubeimoside-1 | A triterpenoid saponin from Bolbostemma paniculatum | [84] |
| Ursolic acid * | A pentacyclic triterpene in plants such as apples (NCT04403568, prostate cancer, not yet recruiting) | [85] |
| α-pinene | A monoterpene in pine needles | [86] |
| Anti-Cancer Agent | Source | Primary Anti-Cancer Action/Application * | Ref. |
|---|---|---|---|
| Etoposide | A derivative of podophyllotoxin, a non-alkaloid lignan that is isolated from Podophyllum peltatum | Topoisomerase II inhibition/Approved for small cell lung cancer and testicular cancer | [10] |
| Irinotecan | A derivative of camptothecin that is a monoterpene indole alkaloid from Camptotheca acuminata | Topoisomerase I inhibition/Approved for colorectal cancer | [9] |
| Paclitaxel | A terpenoid isolated from the Pacific yew tree | Stabilization of microtubule polymer/Approved for AIDS-related Kaposi sarcoma, breast cancer, non-small cell lung cancer, and ovarian cancer | [11] |
| Vincristine | A vinca alkaloid from Catharanthus roseus | An inhibition of microtubule polymerization/Approved for acute leukemia. Also used to treat Hodgkin lymphoma, neuroblastoma, non-Hodgkin lymphoma, rhabdomyosarcoma, and Wilms tumor | [10] |
| miRNA | Phytochemical (A Type of Cancer) | Effective in Vitro Concentration of Phytochemical/Treatment Time | Effective in Vivo Dose of Phytochemical in Mouse Models of Cancers (A Route of Administration) | Ref. |
|---|---|---|---|---|
| miR-17–92 cluster | Ginsenoside Rd (breast cancer) | ≥50 μM/72 h | 50 mg/kg (intraperitoneal) | [75] |
| Maytenin and 22-β-hydroxymaytenin (head and neck squamous cell carcinomas) | 1.5–1.6 μM/24 h * (maytenin), 1.9–2.5 μM/24 h * (22-β-hydroxymaytenin) | 2 mg/kg (maytenin, intraperitoneal) | [78] | |
| Nitidine chloride (chronic myeloid leukemia) | ≥4 μM/72 h | - | [91] | |
| Oridonin (myelogenous leukemia) | ≥5 μM/24 h | 10–15 mg/kg (intraperitoneal) | [80] | |
| Procyanidin (lung cancer) | ≥10 μM/24 h | 56–112 mg/kg (oral) | [56] | |
| Resveratrol (breast cancer) | ≥6.25 μM/48 h | 25–100 mg/kg (intraperitoneal) | [92] | |
| Swainsonine (glioblastoma) | ≥20 μM/12 h | - | [28] | |
| Triptolide (hepatocellular carcinoma) | ≥50 nM/48 h | 0.2 mg/kg (intraperitoneal) | [83] | |
| miR-21 | Celastrol (colorectal cancer) | 3.2 μM/72 h * | - | [72] |
| Curcumin (osteosarcoma) | ≥2.5 μM/72 h | - | [39] | |
| Formononetin (bladder cancer) | ≥50 μM/24 h | - | [42] | |
| Galangin (cholangiocarcinoma) | ≥50 μM/24 h | - | [43] | |
| Gambogic acid (colorectal cancer) | ≥1 μM/48 h | - | [44] | |
| Honokiol (osteosarcoma) | ≥1 μM/24 h | - | [48] | |
| Puerarin (hepatocellular carcinoma) | ≥50 μM/48 h | 40 mg/kg (intravenous) | [57] | |
| Silibinin (breast cancer) | 200 μM/48 h * | - | [61] | |
| Sophocarpine (head and neck cancer) | 1–1.5 μM/48 h * | 5 mg/kg (intravenous) | [27] | |
| Sulforaphane (colorectal cancer) | ≥5 μM/72 h | - | [30] | |
| Sulforaphane (glioblastoma) | ≥5 μM/24 h | - | [93] | |
| miR-23 | Baicalin (colorectal cancer) | 165.5 μM/24 h * | 50–100 mg/kg (intraperitoneal) | [36] |
| Sinomenine (prostate cancer) | ≥0.25 mM/24 h | - | [26] | |
| Sulforaphane (breast cancer) | ≥2.5 μM/24 h | - | [94] | |
| miR-32 | Isoliquiritigenin (nasopharyngeal cancer) | ≥12.5 μM/48 h | 25–100 mg/kg (oral) | [50] |
| miR-95 | Brucein D (hepatocellular carcinoma) | ≥0.5 μM/72 h | 1.5 mg/kg (intraperitoneal) | [70] |
| miR-99a–125b cluster | Berberine (multiple myeloma) | 75 μM/48 h ** | - | [17] |
| miR-103-3p | Physcion 8-O-β-glucopyranoside (gastric cancer) | ≥10 μM/96 h | 30–50 mg/kg (intraperitoneal) | [55] |
| miR-106b–25 cluster | Epigallocatechin gallate (breast cancer) | ≥10 μM/24 h | 100 mg/kg (oral) | [95] |
| Matrine (gastric cancer) | ≥10 μM/24 h | - | [96] | |
| Triptolide (hepatocellular carcinoma) | ≥50 nM/48 h | 0.2 mg/kg (intraperitoneal) | [83] | |
| miR-155 | Silibinin (breast cancer) | 200 μM/48 h * | - | [61] |
| miR-183-96-182 cluster | Curcumin (breast cancer) | 20 μM/24 h * | - | [97] |
| Matrine (papillary thyroid cancer) | ≥1 mM/48 h | - | [21] | |
| Sanguinarine (gastric cancer) | 150–200 μM/48 h * | 5–10 mg/kg (intraperitoneal) | [25] | |
| miR-196b | Resveratrol (acute lymphoblastic leukemia) | ≥25 μM/72 h | - | [98] |
| miR-217 | Baicalin (colorectal cancer) | ≥10 μM/24 h | - | [99] |
| miR-221 | Resveratrol (melanoma) | 50 μM/24 h ** | 30 mg/kg (intraperitoneal) | [59] |
| α-pinene (hepatocellular carcinoma) | ≥16 μM/24 h | - | [86] | |
| miR-222 | Resveratrol (thyroid cancer) | ≥0.5 μM/72 h | - | [100] |
| miR-223 | Genistein (pancreatic cancer) | 50–60 μM/72 h * | - | [101] |
| miR-301-3p | Isoliquiritigenin (melanoma) | ≥10 μM/24 h | 20 mg/kg (intraperitoneal) | [102] |
| Luteolin (pancreatic cancer) | 23.3 μM/96 h * | - | [103] | |
| miR-324-5p | Sinomenine (breast cancer) | ≥0.25 mM/24 h | - | [104] |
| miR-374a | Isoliquiritigenin (breast cancer) | ≥6.25 μM/72 h | - | [105] |
| Neferine (breast cancer) | ≥4 μM/24 h | - | [22] | |
| miR-382 | Sulforaphane (breast cancer) | ≥2.5 μM/24 h | - | [94] |
| miR-429 | Berberine (colorectal cancer) | ≥4 μM/72 h | - | [90] |
| Evodiamine (colorectal cancer) | ≥3 μM/72 h | - | ||
| miR-625-3p | Icariin (thyroid cancer) | ≥20 μM/24 h | - | [49] |
| miR-1290 | Resveratrol (acute lymphoblastic leukemia) | ≥25 μM/72 h | - | [98] |
| miR-4295 | Ginsenoside Rh2 (prostate cancer) | ≥16.6 μM/96 h | - | [76] |
| miR-7641 | Curcumin (bladder cancer) | ≥10 μM/48 h | - | [106] |
| miRNA | Phytochemical (A Type of Cancer) | Effective in Vitro Concentration of Phytochemical/Treatment Time | Effective in Vivo Dose of Phytochemical in Mouse Models of Cancers (A Route of Administration) | Ref. |
|---|---|---|---|---|
| Let-7c | Quercetin (pancreatic cancer) | 50 μM/48 h *** | - | [58] |
| Let-7f | Lycopene (prostate cancer) | ≥10 μM/48 h | - | [77] |
| miR-9 | Sulforaphane (lung cancer) | ≥2.5 μM/120 h | - | [129] |
| miR-10a | β-sitosterol-d-glucoside (breast cancer) | 30.82–49.76 μM/24 h ** | 60 mg–120 mg/kg (intragastric) | [31] |
| miR-15a | Gossypol (pituitary cancer) | ≥5 μM/96 h | - | [130] |
| miR-16 | Sanguinarine (hepatocellular carcinoma) | ≥0.5 μM/48 h | 10 mg/kg (intraperitoneal) | [131] |
| miR-22 | Matrine (colorectal cancer) | ≥0.25 mM/72 h | - | [43] |
| miR-27a-3p * | Maytenin (head and neck cancer) | 1.5–1.6 μM/24 h ** | 2 mg/kg (intraperitoneal) | [78] |
| miR-29-3p * | Sanguinarine (gastric cancer) | 150–200 μM/48 h ** | 5–10 mg/kg (intraperitoneal) | [25] |
| miR-34 | 5-Aminolevulinic acid (melanoma) | 1 mM/4 h + ultrasound | 200 mg/kg (intraperitoneal) + ultrasound | [132] |
| Artemisinin and Artesunate (breast cancer) | 300 μM/48 h *** (artemisinin), ≥5 μM/48 h (artesunate) | - | [20] | |
| Cucurbitacin D (cervical cancer) | ≥0.05 μM/72 h | 1 mg/kg (intra-tumoral) | [73] | |
| Curcumin (breast cancer) | ≥20 μM/48 h | - | [133] | |
| Genistein (head and neck cancer) | ≥20 μM/24 h | 25 mg–50 mg/kg (oral) | [45] | |
| Indole-3-carbinol (breast cancer) | ≥200 μM/48 h | - | [20] | |
| Luteolin (lung cancer) | 40 μM/72 h ** | 50 mg–200 mg/kg (intragastric) | [54] | |
| Resveratrol (breast cancer) | ≥12.5 μM/72 h | - | [134] | |
| miR-101 | Apigenin (hepatocellular carcinoma) | 10 μM/48 h *** | - | [33] |
| miR-122 | Coptisine (hepatocellular carcinoma) | ≥3.1 μM/24 h | 150 mg/kg (oral) | [18] |
| Oleanolic acid (lung cancer) | ≥65 μM/48 h | 120 mg/kg (oral) | [79] | |
| Resveratrol (breast cancer) | ≥100 μM/24 h | - | [135] | |
| miR-124 | Physcion 8-O-β-glucopyranoside (melanoma) | ≥10 μM/24 h | - | [136] |
| miR-125b | Astragalin (hepatocellular carcinoma) | ≥11 μM/48 h | 10–20 mg/kg (oral) | [34] |
| miR-126 | Tubeimoside-1 (lung cancer) | ≥10 μM/48 h | - | [84] |
| miR-132 | Chrysin and Curcumin (breast cancer) | ≥20 μM/48 h (chrysin), ≥10 μM/48 h (curcumin) | - | [38] |
| miR-133a | Artesunate (rhabdomyosarcoma) | ≥5 μM/24 h | 25 mg/kg (intraperitoneal) | [137] |
| Brazilein (vestibular schwannoma) | ≥10 μM/96 h | - | [138] | |
| miR-134 | Astragaloside IV (colorectal cancer) | ≥6.4 μM/48 h | - | [139] |
| miR-137 | Isorhapontigenin (bladder cancer) | 10 μM/24 h *** | 150 mg/kg (intraperitoneal) | [51] |
| Isorhapontigenin (urothelial cancer) | ≥5 μM/24 h | - | [140] | |
| miR-139-5p | Resveratrol (osteosarcoma) | ≥5 μM/48 h | - | [141] |
| miR-143 | 5-Aminolevulinic acid (cervical cancer) | ≥0.25 μM + photodynamic therapy/24 h | - | [142] |
| Cucurbitacin D (cervical cancer) | ≥0.05 μM/72 h | 1 mg/kg (intra-tumoral) | [73] | |
| miR-144-3p | Licochalcone A (lung cancer) | ≥10 μM/48 h | - | [143] |
| miR-145 | Cucurbitacin D (cervical cancer) | ≥0.05 μM/72 h | 1 mg/kg (intra-tumoral) | [73] |
| Isorhapontigenin (glioblastoma) | ≥10 μM/24 h | - | [144] | |
| miR-148a | Ailanthone (breast cancer) | ≥5 μM/48 h | - | [145] |
| miR-149-5p | Ursolic acid (breast cancer) | 20 μM/48 h *** | 10 mg/kg (intraperitoneal) | [85] |
| Ursolic acid (lung cancer) | ≥5 μM/48 h | Treated cells with ursolic acid at 20 μM for 72 h before implanting in mice | [146] | |
| miR-181-3p | Curcumol (breast cancer) | ≥254 μM/48 h | 20 mg/kg of curcumol + 2.5 mg/kg of doxorubicin (intraperitoneal) | [74] |
| miR-193-3p | Triptolide (nephroblastoma) | ≥10 nM/72 h | - | [147] |
| miR-200 | Quercetin (pancreatic cancer) | 50 μM/72 h *** | - | [148] |
| Resveratrol (colorectal cancer) | ≥25 μM/48 h | - | [149] | |
| Toosendanin (gastric cancer) | ≥0.5 μM/48 h | 0.2 mg/kg (intraperitoneal) | [82] | |
| miR-203 | Silymarin (lung cancer) | ≥10 μM/24 h | - | [64] |
| miR-204-3p | Delphinidin (colorectal cancer) | ≥25 μM/24 h | Treated cells with delphinidin at 100 μM for 24 h before implanting in mice | [40] |
| miR-206 | Artesunate (rhabdomyosarcoma) | ≥5 μM/24 h | 25 mg/kg (intraperitoneal) | [137] |
| miR-218 | Andrographolide (oral cancer) | ≥6.25 μM/24 h | 10 mg/kg (oral) | [67] |
| miR-296-3p | Epigallocatechin gallate (nasopharyngeal cancer) | ≥20 μM/24 h | - | [41] |
| miR-320a | Hydroxygenkwanin (hepatocellular carcinoma) | ≥10 μM/72 h | 1 mg/kg (intraperitoneal) | [47] |
| miR-340 | Kaempferol (lung cancer) | ≥20 μM | - | [52] |
| miR-345 | Trans-3,5,4′-trimethoxystilbene (lung cancer) | ≥0.5 μM/72 h | 30 mg/kg (oral) | [65] |
| miR-383-5p | Allicin (gastric cancer) | 62 μM/48 h *** | - | [29] |
| miR-384 | Luteolin (colorectal cancer) | ≥10 μM/48 h | 100 mg/kg (intragastric) | [150] |
| miR-424 | Resveratrol (breast cancer) | ≥12.5 μM/72 h | - | [134] |
| miR-449a | Ailanthone (acute myeloid leukemia) | ≥0.5 μM/48 h | - | [66] |
| miR-485 | Epigallocatechin gallate (lung cancer) | ≥10 μM/24 h | 20 mg/kg (intraperitoneal) | [151] |
| miR-498 | Trans-3,5,4′-trimethoxystilbene (lung cancer) | ≥0.5 μM/72 h | 30 mg/kg (oral) | [65] |
| miR-503 | Resveratrol (breast cancer) | ≥12.5 μM/72 h | - | [134] |
| miR-520b | Apigenin (hepatocellular carcinoma) | ≥10 μM/48 h | 50 mg/kg (intraperitoneal) | [152] |
| miR-542-5p | Pristimerin (breast cancer) | ≥1 μM/24 h | - | [81] |
| miR-663 | Resveratrol (breast cancer) | 100 μM/24 h *** | - | [153] |
| miR-744 | Resveratrol (breast cancer) | 100 μM/24 h *** | - | [153] |
| miR-1972 | Cannabidiol (neuroblastoma) | ≥5 μM/24 h | - | [71] |
| miR-3127-5p | Baicalein (hepatocellular carcinoma) | ≥20 μM/24 h | - | [35] |
| miR-6809-5p | Luteolin (hepatocellular carcinoma) | ≥10 μM/120 h | 50 mg/kg (intraperitoneal) | [154] |
| miRNA | Phytochemical (A Type of Cancer) | In Vitro Finding | In Vivo Experiment Condition | Ref. |
|---|---|---|---|---|
| miR-7-3p | Luteolin and Silibinin (glioblastoma) | Treatment with luteolin (20 μM/24 h) or silibinin (50 μM/24 h) induces apoptosis and miR-7-3p expression | 10 mg/kg (luteolin, oral) or 200 mg/kg (silibinin, oral) with and without miR-7-3p (50 μg, intravenous) | [279] |
| miR-25 | Physcion 8-O-β-glucopyranoside (ovarian cancer) | Overexpression of miR-25 reduces the cytotoxicity of physcion 8-O-β-glucopyranoside (10 μM) | - | [280] |
| miR-17–92 cluster | Oridonin (myelogenous leukemia) | Inhibition of miR-17 or miR-20a enhances the cytotoxicity of oridonin (2.5 and 5 μM for 72 h) | 10–15 mg/kg (oridonin, intraperitoneal) | [80] |
| miR-126 | Epigallocatechin gallate (osteosarcoma) | Overexpression of miR-126 potentiates the cytotoxicity of epigallocatechin gallate (100 μM for 48 h) | - | [281] |
| miR-137 | Delphinidin (glioblastoma) | Overexpression of miR-137 enhances apoptosis induced by delphinidin (50 μM for 24 h) | - | [282] |
| miR-138 | Apigenin (neuroblastoma) | Ectopic expression of miR-138 enhances apoptosis induced by apigenin (100 μM for 24 h) | 10 μg/mouse (apigenin, intra-tumoral) + hTERT shRNA plasmid or miR-138 mimic | [283] |
| miR-143 | Shikonin (glioblastoma) | Overexpression of miR-143 enhances apoptosis induced by shikonin (2 μM for 24 h) | 2 mg/kg (shikonin, intraperitoneal) in mice bearing miR-143-overexpressing cells | [60] |
| miR-210 | 1′S-1′-acetoxychavicol acetate (cervical cancer) | Knockdown of miR-210 potentiates the cytotoxicity of 1′S-1′-acetoxychavicol acetate (5–20 μM for 48 h) | - | [277] |
| miR-629 | 1′S-1′-acetoxychavicol acetate (cervical cancer) | Knockdown of miR-629 potentiates the cytotoxicity of 1′S-1′-acetoxychavicol acetate (5–20 μM for 48 h) | - | [278] |
| miRNAs | Phytochemicals (A Type of Cancer) | In Vitro Finding | In Vivo Experiment Condition | Ref. |
|---|---|---|---|---|
| Cancer resistance-promoting miRNAs | ||||
| miR-21 | Etoposide (colorectal cancer) | 3.97 μM/72 h * (miR-control-overexpressing cells), 12.7 μM/72 h * (miR-21-overexpressing cells) | - | [291] |
| Paclitaxel (breast cancer) | ≈ 6 μM/48 h * (anti-miR-control-treated cells), ≈ 3 μM/48 h * (anti-miR-21-treated cells) | 1 mg/kg (paclitaxel, intravenous) + anti-miR-21 (intra-tumoral) | [296] | |
| miR-27a-3p | Paclitaxel (ovarian cancer) | Silencing of miR-27a-3p increases the cytotoxicity of paclitaxel (4, 8, and 12 μM for 7 days) | 5 mg/kg (paclitaxel) in mice bearing miR-27a-3p-overexpressing cells | [297] |
| Paclitaxel (ovarian cancer) | Silencing of miR-27a-3p increases the cytotoxicity of paclitaxel (0.04–23 μM for 48 h) | - | [298] | |
| miR-125b | Paclitaxel (breast cancer) | Overexpression of miR-125b reduces the cytotoxicity of paclitaxel (4, 8, and 16 nM for 48 h) | - | [299] |
| miR-140-3p | Paclitaxel (chordoma) | Silencing of miR-140-3p or miR-155-5p increases the cytotoxicity of paclitaxel (10 μM for 24–72 h) | - | [300] |
| miR-155-5p | ||||
| miR-192 | Etoposide (lung cancer) | Knockdown of miR-192 sensitizes cells to etoposide (≤100 μM for 24 h) | - | [301] |
| miR-374a | Etoposide (glioblastoma) | Knockdown of miR-374a sensitizes cells to etoposide (0.5–8 μM for 48 h) | - | [292] |
| miR-514b-5p | Irinotecan (colorectal cancer) | Overexpression of miR-514b-5p reduces the cytotoxicity of irinotecan (25, 50, and 100 μM for 48 h) | 40 mg/kg (irinotecan) in mice bearing miR-514b-5p-overexpressing cells | [302] |
| miR-520h | Paclitaxel (breast cancer) | Overexpression of miR-520h reduces the cytotoxicity of paclitaxel (1, 5, and 10 nM for 24 h) | - | [303] |
| miR-662 | Etoposide (lung cancer) | Knockdown of miR-662 sensitizes cells to etoposide (≤100 μM for 24 h) | - | [301] |
| miR-1207-5p | Paclitaxel (breast cancer) | Knockdown of miR-1207-5p sensitizes cells to paclitaxel (10 nM for 5 days) | - | [304] |
| miR-4262 | Paclitaxel (lung cancer) | Overexpression of miR-4262 reduces the cytotoxicity of paclitaxel (4 and 8 μM) | Paclitaxel + anti-miR-4262 (intravenous) | [305] |
| Cancer resistance-suppressing miRNAs | ||||
| miR-1 | Vincristine (gastric cancer) | Knockdown of miR-1 reduces the cytotoxicity of vincristine (10 and 20 μM) | - | [306] |
| miR-7-5p | Paclitaxel (breast cancer) | 25–35 μM/72 h * (miR-control-overexpressing cells), 5–15 μM/72 h * (miR-7-5p-overexpressing cells) | - | [307] |
| miR-29-3p | Etoposide (cervical cancer) | Overexpression of miR-29-3p enhances the cytotoxicity of etoposide (60 μM for 48 h) | - | [293] |
| Paclitaxel (nasopharyngeal cancer) | 6.4–7.5 nM/96 h * (miR-control-overexpressing cells), 0.7–0.8 nM/96 h * (miR-29-3p-overexpressing cells) | 5 mg/kg (paclitaxel, intraperitoneal) in mice bearing miR-29-3p inhibitor treated cells | [308] | |
| miR-34 | Etoposide (retinoblastoma) | Overexpression of miR-34 enhances the cytotoxicity of etoposide (35 μM for 48 h) | - | [309] |
| Vincristine (retinoblastoma) | Overexpression of miR-34 enhances the cytotoxicity of vincristine (130 nM for 48 h) | - | [309] | |
| miR-126 | Vincristine (gastric cancer) | Overexpression of miR-126 enhances the cytotoxicity of vincristine (1.2, 6, and 12 μM for 48 h) | - | [310] |
| miR-133b | Vincristine (colorectal cancer) | 420 μM/24 h * (miR-control-overexpressing cells), 120 μM/24 h * (miR-133b-overexpressing cells) | - | [311] |
| miR-145-5p | Paclitaxel (breast cancer) | ≈ 6 μM/24 h * (miR-control-overexpressing cells), ≤3 μM/24 h * (miR-145-5p-overexpressing cells) | miR-145-5p mimic (intra-tumoral) in mice bearing paclitaxel-resistant cells | [312] |
| miR-193-3p | Etoposide (osteosarcoma) | Overexpression of miR-193-3p enhances the cytotoxicity of etoposide at IC50/72 h | - | [294] |
| miR-196-5p | Etoposide (hepatocellular carcinoma) | Overexpression of miR-196-5p increases apoptosis induced by etoposide (50 μM for 16 h) | - | [295] |
| miR-200 | Irinotecan (colorectal cancer) | Delivery of miR-200 using nanoparticles increases apoptosis after treatment with irinotecan (incorporated in liposomes) for 48 h | 100 mg/kg (irinotecan, intravenous) + 1.25 mg/kg (miR-200, intravenous) | [313] |
| Vincristine (gastric cancer) | Overexpression of miR-200 increases apoptosis induced by vincristine (4 μM for 48 h) | - | [314] | |
| miR-302 | Etoposide (leukemia) | Overexpression of miR-302 enhances the cytotoxicity of etoposide (20, 50, 100, and 200 μM) | 20 mg/kg (etoposide, intraperitoneal) in mice bearing miR-302-overexpressing cells | [315] |
| miR-365 | Paclitaxel (endometrial cancer) | Overexpression of miR-365 enhances the cytotoxicity of paclitaxel (200 and 300 nM for 24 h) | - | [316] |
| miR-383-5p | Paclitaxel (ovarian cancer) | Overexpression of miR-383-5p enhances the cytotoxicity of paclitaxel (≤5 μM) | Subcutaneous injection of miR-383-5p-overexpressing cells | [317] |
| miR-429 | Vincristine (gastric cancer) | Overexpression of miR-429 increases apoptosis induced by vincristine (4 μM for 48 h) | - | [314] |
| miR-495-3p | Vincristine (gastric cancer) | Overexpression of miR-495-3p enhances the cytotoxicity of vincristine (≤1.65 μM for 24 h) | Subcutaneous injection of miR-495-3p-overexpressing cells | [318] |
| miR-542-3p | Paclitaxel (breast cancer) | Overexpression of miR-542-3p increases apoptosis induced by paclitaxel (3 nM for 24 h) | 6 mg/kg (paclitaxel, intraperitoneal) + 5μg (miR-542-3p, intra-tumoral) | [319] |
| miR-584-5p | Vincristine (medulloblastoma) | Overexpression of miR-584-5p enhances the cytotoxicity of vincristine (≤1 μM for 72 h) | 0.5 mg/kg (vincristine, intraperitoneal) in mice bearing miR-584-5p-overexpressing cells | [320] |
| miR-621 | Paclitaxel (breast cancer) | Overexpression of miR-621 promotes apoptosis induced by paclitaxel (20 μM for 24 h) | 15 mg/kg (paclitaxel, intraperitoneal) in mice bearing miR-621-overexpressing cells | [321] |
| miR-627 | Irinotecan (colorectal cancer) | Overexpression of miR-627 enhances the cytotoxicity of irinotecan (5, 10, and 20 μM for 48 h) | 50 mg/kg (irinotecan, intraperitoneal) + 0.4 μg (calcitriol, intraperitoneal) | [15] |
| miR-874 | Vincristine (gastric cancer) | ≈ 4 μM/48 h * (anti-miR-control treated cells), ≈ 2 μM /48 h * (anti-miR-874 treated cells) | - | [322] |
| miR-3163 | Etoposide (retinoblastoma) | 0.57 μM/72 h * (miR-control-overexpressing cells), 0.39 μM/72 h * (miR-3163-overexpressing cells) | - | [323] |
| Vincristine (retinoblastoma) | 1.27 μM/72 h * (miR-control-overexpressing cells), 0.77 μM/72 h * (miR-3163-overexpressing cells) | - | [323] | |
| miR-4454 | Irinotecan (colorectal cancer) | Knockdown of miR-4454 reduces apoptosis induced by irinotecan (10 μM for 48 h) | Subcutaneous injection of miR-4454 expressing cells | [324] |
| miR-5195-3p | Paclitaxel (breast cancer) | Overexpression of miR-5195-3p enhances the cytotoxicity of paclitaxel (2.5–10 μM) | - | [325] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Son, S.W.; Lee, H.Y.; Moeng, S.; Kuh, H.J.; Choi, S.Y.; Park, J.K. Participation of MicroRNAs in the Treatment of Cancer with Phytochemicals. Molecules 2020, 25, 4701. https://doi.org/10.3390/molecules25204701
Son SW, Lee HY, Moeng S, Kuh HJ, Choi SY, Park JK. Participation of MicroRNAs in the Treatment of Cancer with Phytochemicals. Molecules. 2020; 25(20):4701. https://doi.org/10.3390/molecules25204701
Chicago/Turabian StyleSon, Seung Wan, Han Yeoung Lee, Sokviseth Moeng, Hyo Jeong Kuh, Soo Young Choi, and Jong Kook Park. 2020. "Participation of MicroRNAs in the Treatment of Cancer with Phytochemicals" Molecules 25, no. 20: 4701. https://doi.org/10.3390/molecules25204701
APA StyleSon, S. W., Lee, H. Y., Moeng, S., Kuh, H. J., Choi, S. Y., & Park, J. K. (2020). Participation of MicroRNAs in the Treatment of Cancer with Phytochemicals. Molecules, 25(20), 4701. https://doi.org/10.3390/molecules25204701







