Nanohydroxyapatite-Mediated Imatinib Delivery for Specific Anticancer Applications
Abstract
1. Introduction
2. Results and Discussion
2.1. Characterisation of nHAp, Imatinib and nHAp/Imatinib
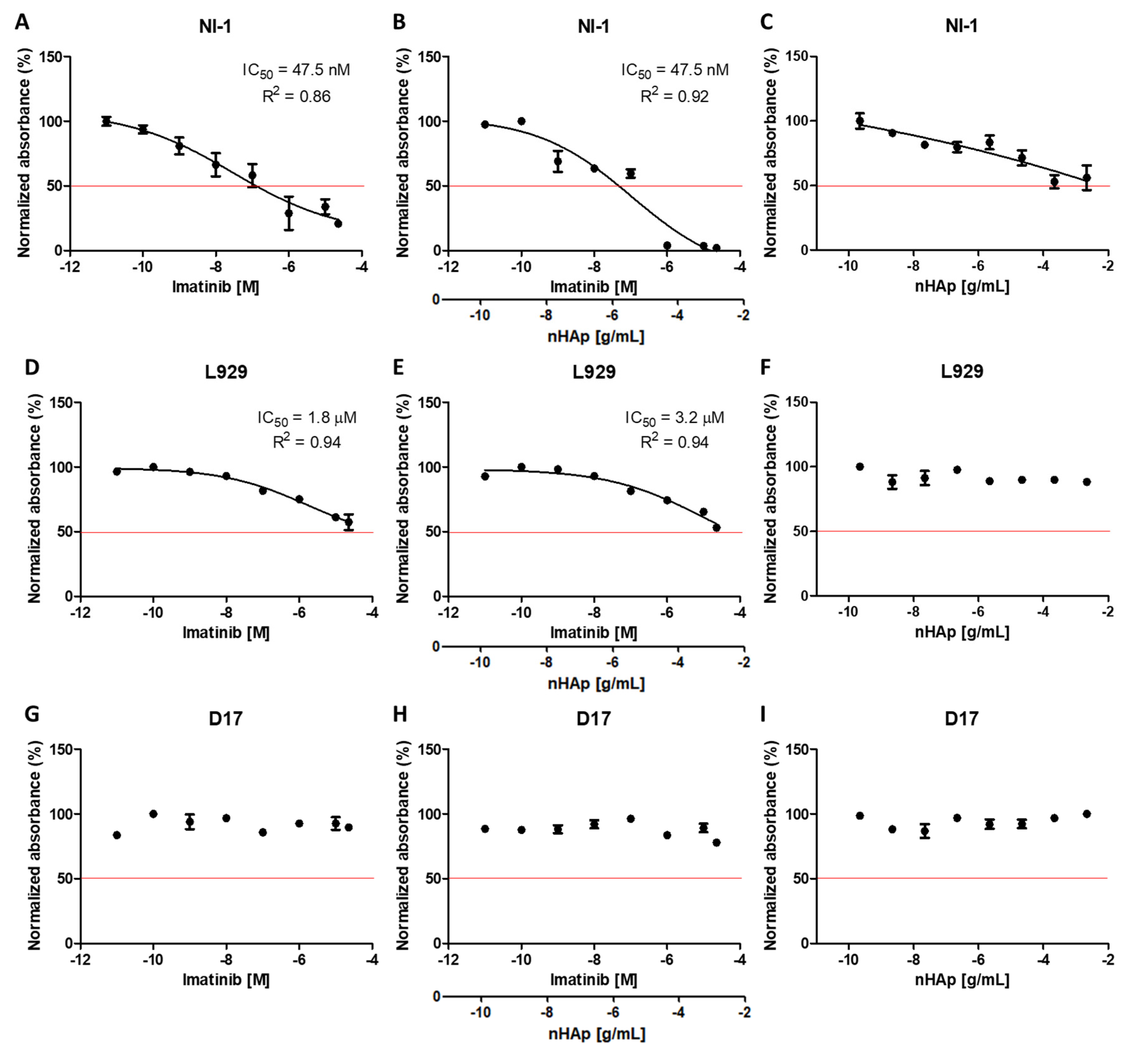
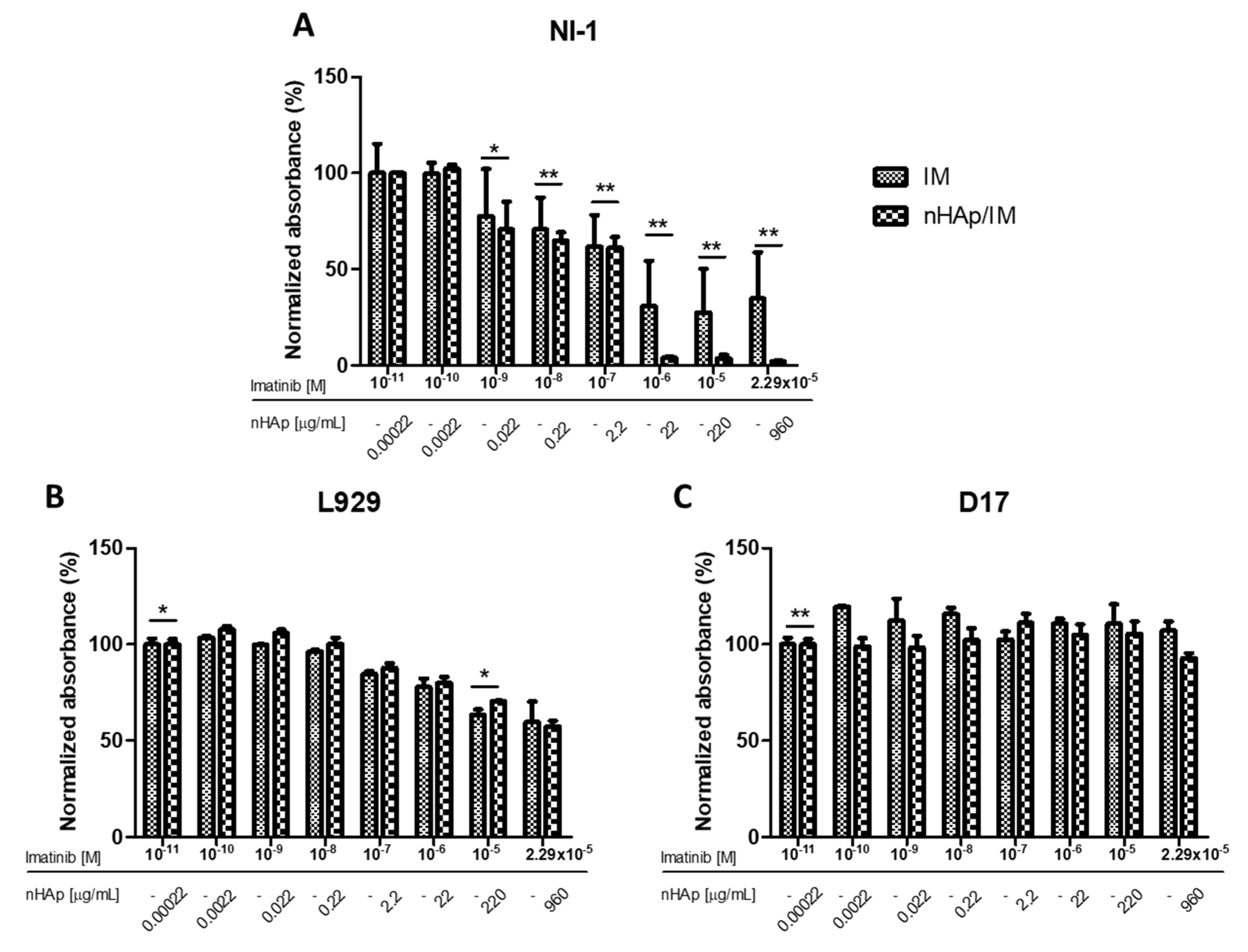
2.2. The Cytotoxicity Evaluation of the nHAp/IM
3. Materials and Methods
3.1. X-ray Powder Diffraction (XRPD)
3.2. Scanning Electron Microscopy with Energy-Dispersive X-ray Spectroscopy (SEM-EDS)
3.3. Absorption Spectroscopy
Imatinib Release
3.4. Fourier Transform Infrared Spectroscopy (FT-IR)
3.5. Dynamic Light Scattering Particle Size Analysis (DLS) and Zeta (ζ) Potential
3.6. Biological Analysis
3.6.1. Cell Cultures
3.6.2. Cytotoxicity Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Arora, A.; Scholar, E.M. Role of tyrosine kinase inhibitors in cancer therapy. J. Pharmacol. Exp. Ther. 2005, 315, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Druker, B.J.; Tamura, S.; Buchdunger, E.; Ohno, S.; Segal, G.M.; Fanning, S.; Zimmermann, J.; Lydon, N.B. Effects of a selective inhibitor of the Ab1 tyrosine kinase on the growth of Bcr-Ab1 positive cells. Nat. Med. 1996, 2, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Ohashi, K.; Maruvka, Y.E.; Michor, F.; Pao, W. Epidermal growth factor receptor tyrosine kinase inhibitor-resistant disease. J. Clin. Oncol. 2013, 31, 1070–1080. [Google Scholar] [CrossRef] [PubMed]
- Babaei, M.A.; Kamalidehghan, B.; Saleem, M.; Huri, H.Z.; Ahmadipour, F. Receptor tyrosine kinase (c-Kit) inhibitors: A potential therapeutic target in cancer cells. Drug Des. Dev. Ther. 2016, 10, 2443–2459. [Google Scholar] [CrossRef] [PubMed]
- Jiao, Q.; Bi, L.; Ren, Y.; Song, S.; Wang, Q.; Wang, Y. Advances in studies of tyrosine kinase inhibitors and their acquired resistance. Mol. Cancer 2018, 17, 1–12. [Google Scholar] [CrossRef]
- Finlay, M.R.V.; Anderton, M.; Ashton, S.; Ballard, P.; Bethel, P.A.; Box, M.R.; Bradbury, R.H.; Brown, S.J.; Butterworth, S.; Campbell, A.; et al. Discovery of a potent and selective EGFR inhibitor (AZD9291) of both sensitizing and T790M resistance mutations that spares the wild type form of the receptor. J. Med. Chem. 2014, 57, 8249–8267. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, S.; Wang, W.; Li, Q.; Yamada, T.; Kita, K.; Donev, I.S.; Nakamura, T.; Matsumoto, K.; Shimizu, E.; Nishioka, Y.; et al. Dual inhibition of met kinase and angiogenesis to overcome HGF-induced EGFR-TKI resistance in EGFR mutant lung cancer. Am. J. Pathol. 2012, 181, 1034–1043. [Google Scholar] [CrossRef]
- Sos, M.L.; Rode, H.B.; Heynck, S.; Peifer, M.; Fischer, F.; Klüter, S.; Pawar, V.G.; Reuter, C.; Heuckmann, J.M.; Weiss, J.; et al. Chemogenomic profiling provides insights into the limited activity of irreversible EGFR inhibitors in tumor cells expressing the T790M EGFR resistance mutation. Cancer Res. 2010, 70, 868–874. [Google Scholar] [CrossRef]
- Godin-Heymann, N.; Ulkus, L.; Brannigan, B.W.; McDermott, U.; Lamb, J.; Maheswaran, S.; Settleman, J.; Haber, D.A. The T790M “gatekeeper” mutation in EGFR mediates resistance to low concentrations of an irreversible EGFR inhibitor. Mol. Cancer Ther. 2008, 7, 874–879. [Google Scholar] [CrossRef]
- Burris Iii, H.A.; Taylor, C.W.; Jones, S.F.; Koch, K.M.; Versola, M.J.; Arya, N.; Fleming, R.A.; Smith, D.A.; Pandite, L.; Spector, N.; et al. A Phase I and Pharmacokinetic Study of Oral Lapatinib Administered Once or Twice Daily in Patients with Solid Malignancies. Clin. Cancer Res. 2009, 15, 6702–6708. [Google Scholar] [CrossRef]
- Roskoski, R. Small molecule inhibitors targeting the EGFR/ErbB family of protein-tyrosine kinases in human cancers. Pharmacol. Res. 2019, 139, 395–411. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Shi, K.; Hao, Y.; Yang, C.; Zha, R.; Yi, C.; Qian, Z. Advances in nanotechnology-based delivery systems for EGFR tyrosine kinases inhibitors in cancer therapy. Asian J. Pharm. Sci. 2020, 15, 26–41. [Google Scholar] [CrossRef] [PubMed]
- Park, K. Controlled drug delivery systems: Past forward and future back. J. Control. Release 2014, 190, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Tan, S.; Li, S.; Shen, Q.; Wang, K. Cancer drug delivery in the nano era: An overview and perspectives (Review). Oncol. Rep. 2017, 38, 611–624. [Google Scholar] [CrossRef] [PubMed]
- Tong, C.W.S.; Wu, W.K.K.; Loong, H.H.F.; Cho, W.C.S.; To, K.K.W. Drug combination approach to overcome resistance to EGFR tyrosine kinase inhibitors in lung cancer. Cancer Lett. 2017, 405, 100–110. [Google Scholar] [CrossRef]
- Palazzo, B.; Iafisco, M.; Laforgia, M.; Margiotta, N.; Natile, G.; Bianchi, C.L.; Walsh, D.; Mann, S.; Roveri, N. Biomimetic hydroxyapatite-drug nanocrystals as potential bone substitutes with antitumor drug delivery properties. Adv. Funct. Mater. 2007, 17, 2180–2188. [Google Scholar] [CrossRef]
- Yilmaz, B.; Alshemary, A.Z.; Evis, Z. Co-doped hydroxyapatites as potential materials for biomedical applications. Microchem. J. 2019, 144, 443–453. [Google Scholar] [CrossRef]
- Hench, L.L. Bioceramics. J. Am. Ceram. Soc. 1998, 81, 1705–1728. [Google Scholar] [CrossRef]
- Marycz, K.; Sobierajska, P.; Smieszek, A.; Maredziak, M.; Wiglusz, K.; Wiglusz, R.J. Li+ activated nanohydroxyapatite doped with Eu3+ ions enhances proliferative activity and viability of human stem progenitor cells of adipose tissue and olfactory ensheathing cells. Further perspective of nHAP:Li+, Eu3+ application in theranostics. Mater. Sci. Eng. C. 2017, 78, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Mizushima, Y.; Ikoma, T.; Tanaka, J.; Hoshi, K.; Ishihara, T.; Ogawa, Y.; Ueno, A. Injectable porous hydroxyapatite microparticles as a new carrier for protein and lipophilic drugs. J. Control. Release 2006, 110, 260–265. [Google Scholar] [CrossRef] [PubMed]
- Kunieda, K.; Seki, T.; Nakatani, S.; Wakabayashi, M.; Shiro, T.; Inoue, K.; Sougawa, M.; Kimura, R.; Harada, K. Implantation treatment method of slow release anticancer doxorubicin containing hydroxyapatite (DOX-HAP) complex. A basic study of a new treatment for hepatic cancer. Br. J. Cancer 1993, 67, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Brigger, I.; Dubernet, C.; Couvreur, P. Nanoparticles in cancer therapy and diagnosis. Adv. Drug Deliv. Rev. 2002, 54, 631–651. [Google Scholar] [CrossRef]
- Feng, S.S.; Chien, S. Chemotherapeutic engineering: Application and further development of chemical engineering principles for chemotherapy of cancer and other diseases. Chem. Eng. Sci. 2003, 58, 4087–4114. [Google Scholar] [CrossRef]
- Salimi, M.N.; Anuar, A. Characterizations of biocompatible and bioactive hydroxyapatite particles. Procedia Eng. 2013, 53, 192–196. [Google Scholar] [CrossRef]
- Gratton, S.E.A.; Ropp, P.A.; Pohlhaus, P.D.; Luft, J.C.; Madden, V.J.; Napier, M.E.; DeSimone, J.M. The effect of particle design on cellular internalization pathways. Proc. Natl. Acad. Sci. USA 2008, 105, 11613–11618. [Google Scholar] [CrossRef]
- Chen, L.; Mccrate, J.M.; Lee, J.C.M.; Li, H. The role of surface charge on the uptake and biocompatibility of hydroxyapatite nanoparticles with osteoblast cells. Nanotechnology 2011, 22. [Google Scholar] [CrossRef]
- Motskin, M.; Wright, D.M.; Muller, K.; Kyle, N.; Gard, T.G.; Porter, A.E.; Skepper, J.N. Hydroxyapatite nano and microparticles: Correlation of particle properties with cytotoxicity and biostability. Biomaterials 2009, 30, 3307–3317. [Google Scholar] [CrossRef]
- Honary, S.; Zahir, F. Effect of Zeta Potential on the Properties of Nano-Drug Delivery Systems—A Review (Part 1). Trop. J. Pharm. Res. 2013, 12, 255–264. [Google Scholar] [CrossRef]
- Cai, Y.; Liu, Y.; Yan, W.; Hu, Q.; Tao, J.; Zhang, M.; Shi, Z.; Tang, R. Role of hydroxyapatite nanoparticle size in bone cell proliferation. J. Mater. Chem. 2007, 17, 3780–3787. [Google Scholar] [CrossRef]
- Iqbal, N.; Iqbal, N. Imatinib: A Breakthrough of Targeted Therapy in Cancer. Chemother. Res. Pract. 2014, 2014, 1–9. [Google Scholar] [CrossRef]
- Pardanani, A.; Elliott, M.; Reeder, T.; Li, C.Y.; Baxter, E.J.; Cross, N.C.P.; Tefferi, A. Imatinib for systemic mast-cell disease. Lancet 2003, 362, 535–537. [Google Scholar] [CrossRef]
- Hadzijusufovic, E.; Peter, B.; Herrmann, H.; Rülicke, T.; Cerny-Reiterer, S.; Schuch, K.; Kenner, L.; Thaiwong, T.; Yuzbasiyan-Gurkan, V.; Pickl, W.F.; et al. NI-1: A novel canine mastocytoma model for studying drug resistance and IgER-dependent mast cell activation. Allergy Eur. J. Allergy Clin. Immunol. 2012, 67, 858–868. [Google Scholar] [CrossRef] [PubMed]
- Karolewicz, B.; Górniak, A.; Marciniak, D.M.; Mucha, I. Molecular mobility and stability studies of amorphous imatinib mesylate. Pharmaceutics 2019, 11, 304. [Google Scholar] [CrossRef] [PubMed]
- Miyamura, K.; Ohnishi, K.; Ohtake, S.; Usui, N.; Nakaseko, C.; Fujita, H.; Fujisawa, S.; Sakura, T.; Okumura, H.; Iriyama, N.; et al. Randomized study of imatinib for chronic myeloid leukemia: Comparing standard dose escalation with aggressive escalation. Blood Adv. 2019, 3, 312–319. [Google Scholar] [CrossRef]
- Ben Ami, E.; Demetri, G.D. A safety evaluation of imatinib mesylate in the treatment of gastrointestinal stromal tumor. Expert Opin. Drug Saf. 2016, 15, 571–578. [Google Scholar] [CrossRef]
- Psimadas, D.; Georgoulias, P.; Valotassiou, V.; Loudos, G. Conformational Polymorphism on Imatinib Mesylate: Grinding Effects. J. Pharm. Sci. 2012, 101, 2271–2280. [Google Scholar] [CrossRef]
- Lin, M.; Wu, Y.; Rohani, S. A kinetic study of crystallization process of imatinib mesylate with polymorphic transformation phenomenon. J. Cryst. Growth 2019, 507, 146–153. [Google Scholar] [CrossRef]
- Pathi, S.L.; Puppala, R.; Kankan, R.N.; Dharmaraj, R.R. Stable Crystal Form of Imatinib Mesylate and Process for the Preparation Thereof. U.S. Patent 8,269,003, 18 September 2012. [Google Scholar]
- Jegorov, A.; Chudik, M.; Aronhime, J.; Gavenda, J.F.A. Polymorphic Forms of Imatinib Mesylate and Processes for Preparation of Novel Crystalline Forms as well as Amorphous and Form Alpha. U.S. Patent 7,977,348, 12 July 2011. [Google Scholar]
- Sudarsanan, K.; Young, R.A. Significant precision in crystal structural details. Holly Springs hydroxyapatite. Acta Crystallogr. Sect. B Struct. Crystallogr. Cryst. Chem. 1969, 25, 1534–1543. [Google Scholar] [CrossRef]
- Veverka, M.; Šimon, P.; Lokaj, J.; Veverková, E. Crystal habit modifications of imatinib mesylate under various precipitation conditions. Monatsh. Chem. 2012, 143, 65–71. [Google Scholar] [CrossRef]
- Srivastava, A.; Joshi, B.D.; Tandon, P.; Ayala, A.P.; Bansal, A.K.; Grillo, D. Study of polymorphism in imatinib mesylate: A quantum chemical approach using electronic and vibrational spectra. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2013, 103, 325–332. [Google Scholar] [CrossRef]
- Al-Hadiya, B.M.H.; Bakheit, A.H.H.; Abd-Elgalil, A.A. Imatinib Mesylate. Profiles Drug Subst. Excip. Relat. Methodol. 2014, 39, 265–297. [Google Scholar] [CrossRef]
- Sobierajska, P.; Wiglusz, R.J. Influence of Li+ ions on the physicochemical properties of nanocrystalline calcium-strontium hydroxyapatite doped with Eu3+ ions. New J. Chem. 2019, 43, 14908–14916. [Google Scholar] [CrossRef]
- Szczepek, W.; Zagrodzki, B.; Glice, M.; Maruszak, W.; Korczak, K.; Modzelewski, R.; Lawecka, M.; Cmoch, P.; Application, F.; Data, P.; et al. Crystalline Polymorphs of Methanesulfonic Acid Addition Salts of Imatinib. U.S. Patent 7,732,601, 8 June 2010. [Google Scholar]
- Mout, R.; Moyano, D.F.; Rana, S.; Rotello, V.M. Surface functionalization of nanoparticles for nanomedicine. Chem. Soc. Rev. 2012, 41, 2539–2544. [Google Scholar] [CrossRef] [PubMed]
- Bouladjine, A.; Al-Kattan, A.; Dufour, P.; Drouet, C. New advances in nanocrystalline apatite colloids intended for cellular drug delivery. Langmuir 2009, 25, 12256–12265. [Google Scholar] [CrossRef]
- Wiglusz, R.J.; Pozniak, B.; Zawisza, K.; Pazik, R. An up-converting HAP@β-TCP nanocomposite activated with Er3+/Yb3+ ion pairs for bio-related applications. RSC Adv. 2015, 5, 27610–27622. [Google Scholar] [CrossRef]
- Singh, R.K.; Kim, T.H.; Patel, K.D.; Kim, J.J.; Kim, H.W. Development of biocompatible apatite nanorod-based drug-delivery system with in situ fluorescence imaging capacity. J. Mater. Chem. B 2014, 2, 2039–2050. [Google Scholar] [CrossRef]
- Surmenev, R.A.; Surmeneva, M.A.; Ivanova, A.A. Significance of calcium phosphate coatings for the enhancement of new bone osteogenesis—A review. Acta Biomater. 2014, 10, 557–579. [Google Scholar] [CrossRef]
- Balan, E.; Delattre, S.; Roche, D.; Segalen, L.; Morin, G.; Guillaumet, M.; Blanchard, M.; Lazzeri, M.; Brouder, C.; Salje, E.K.H. Line-broadening effects in the powder infrared spectrum of apatite. Phys. Chem. Miner. 2011, 38, 111–122. [Google Scholar] [CrossRef]
- Rietveld, H.M. A profile refinement method for nuclear and magnetic structures. J. Appl. Cryst. 1969, 2, 65–71. [Google Scholar] [CrossRef]
- Lutterotti, L.; Matthies, S.; Wenk, H.-R. MAUD: A friendly Java program for Material Analysis Using Diffraction. IUCr Newsl. CPD 1999, 21, 14–15. [Google Scholar] [CrossRef]
- Kaasalainen, M.; Aseyev, V.; von Haartman, E.; Karaman, D.Ş.; Mäkilä, E.; Tenhu, H.; Rosenholm, J.; Salonen, J. Size, Stability, and Porosity of Mesoporous Nanoparticles Characterized with Light Scattering. Nanoscale Res. Lett. 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Lowry, G.V.; Hill, R.J.; Harper, S.; Rawle, A.F.; Hendren, C.O.; Klaessig, F.; Nobbmann, U.; Sayre, P.; Rumble, J. Guidance to improve the scientific value of zeta-potential measurements in nanoEHS. Environ. Sci. Nano 2016, 3, 953–965. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |
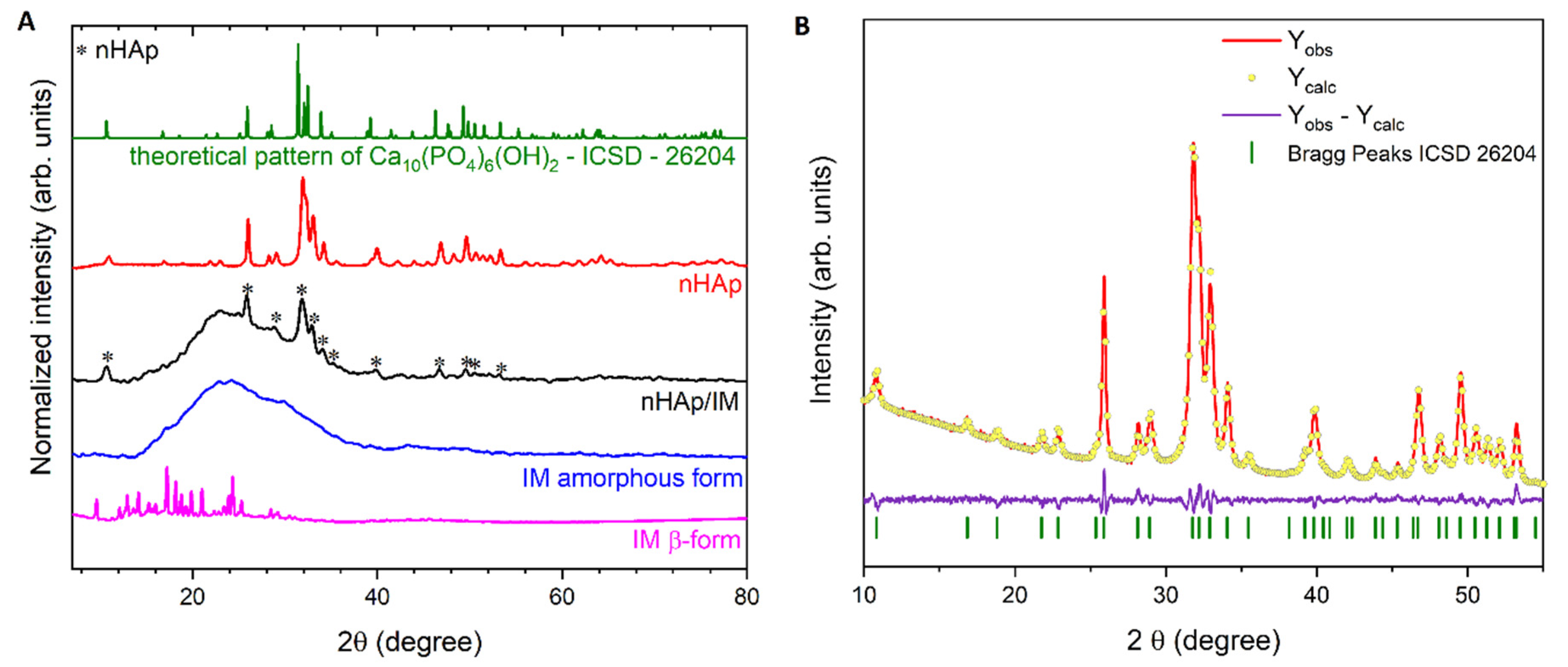
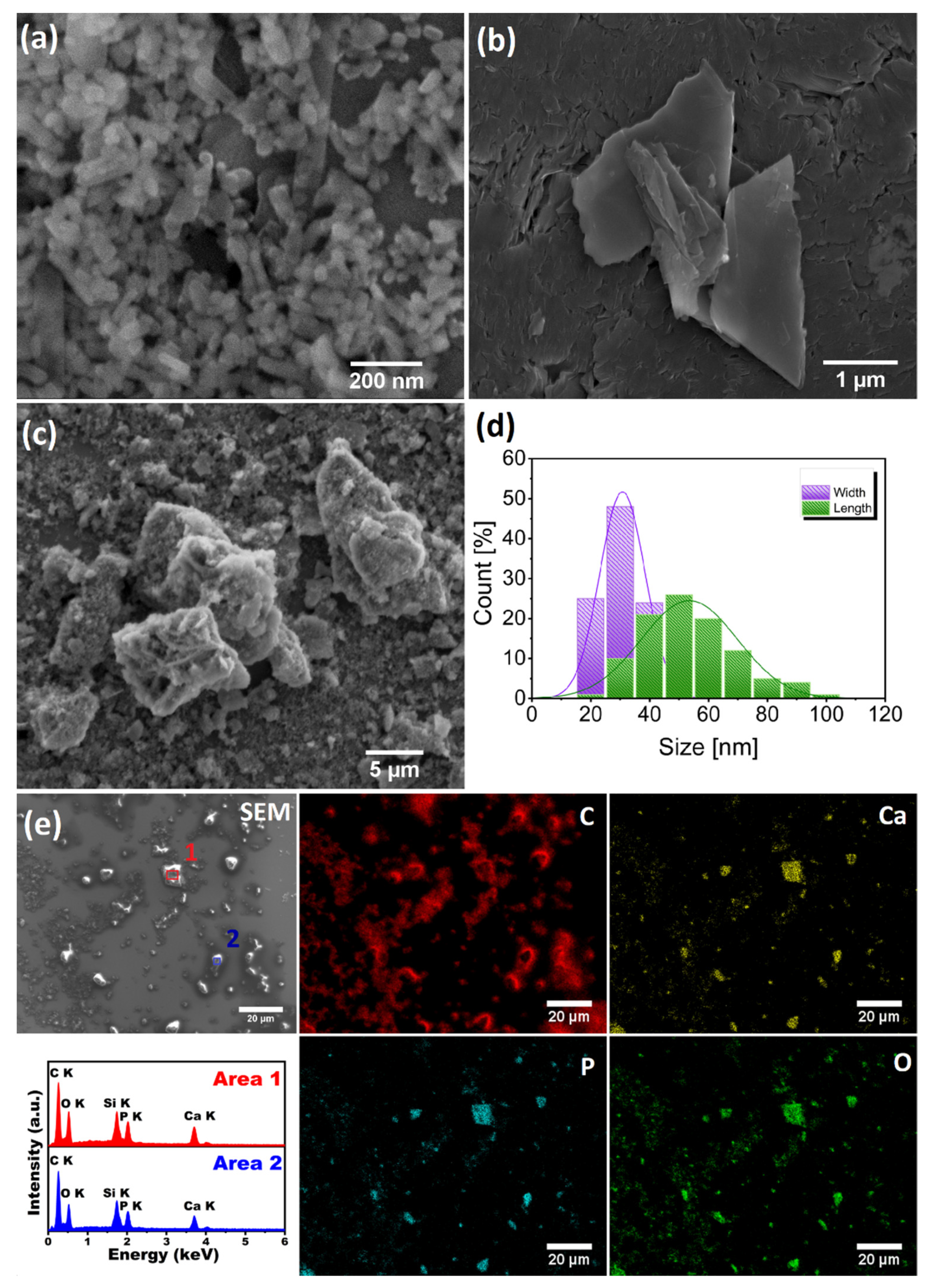
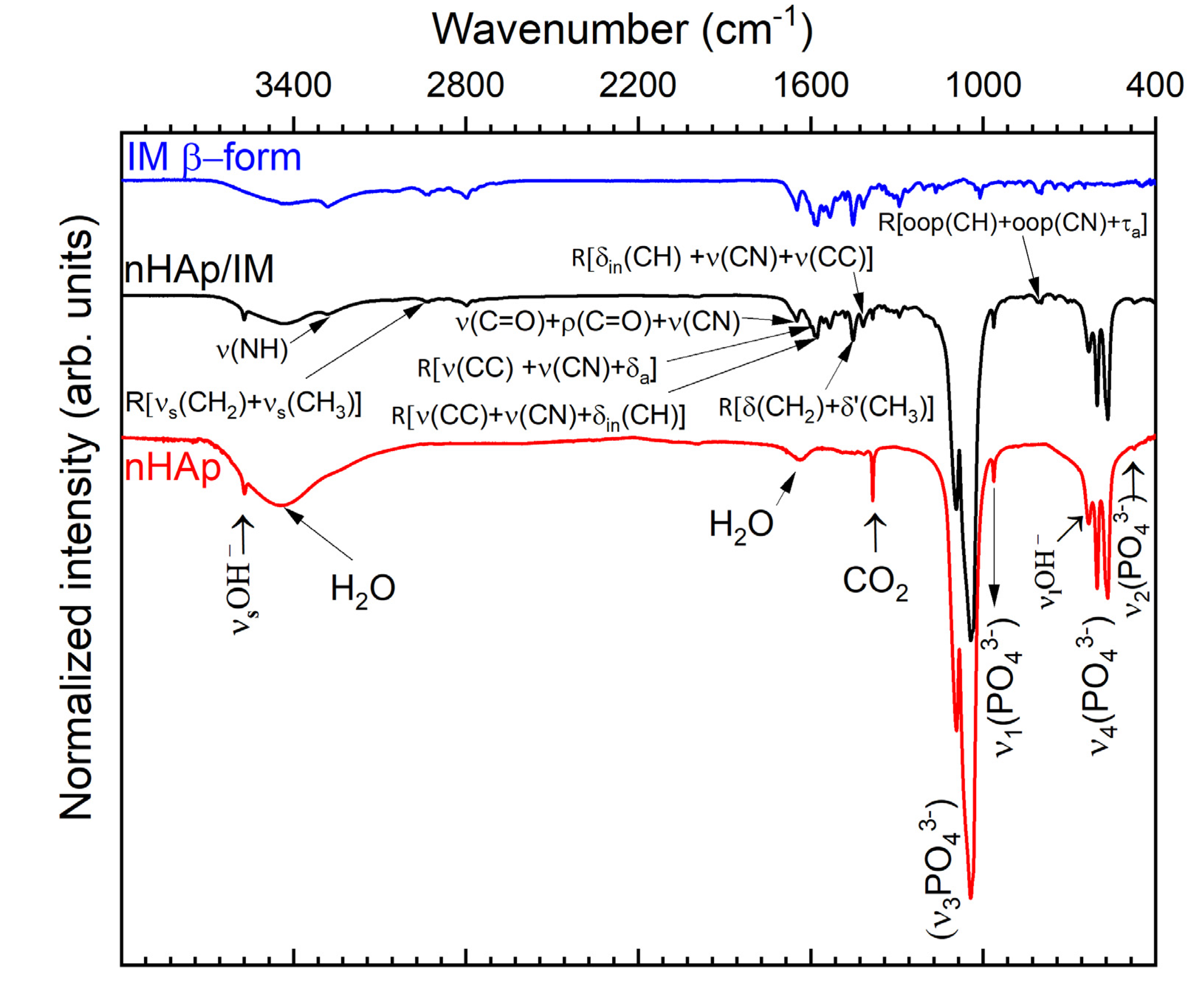

| Sample | a (Å) | c (Å) | V (Å3) | Size (nm) | Rwp (%) |
|---|---|---|---|---|---|
| single crystal [40] | 9.424(4) | 6.879(4) | 529.09(44) | – | – |
| nHAp | 9.430(4) | 6.891(4) | 530.76(02) | 45.5(5) | 3.2 |
| Sample | Zeta Potential (mV) De-Ionized Water | Zeta Potential (mV) Cell Culture Medium |
|---|---|---|
| nHAp | −18.8 ± 0.2 | −10.4 ± 0.3 |
| IM | −15.9 ± 0.7 | −8.9 ± 0.5 |
| nHAp/IM | −14.0 ± 0.4 | −10.4 ± 0.1 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sobierajska, P.; Serwotka-Suszczak, A.; Szymanski, D.; Marycz, K.; Wiglusz, R.J. Nanohydroxyapatite-Mediated Imatinib Delivery for Specific Anticancer Applications. Molecules 2020, 25, 4602. https://doi.org/10.3390/molecules25204602
Sobierajska P, Serwotka-Suszczak A, Szymanski D, Marycz K, Wiglusz RJ. Nanohydroxyapatite-Mediated Imatinib Delivery for Specific Anticancer Applications. Molecules. 2020; 25(20):4602. https://doi.org/10.3390/molecules25204602
Chicago/Turabian StyleSobierajska, Paulina, Anna Serwotka-Suszczak, Damian Szymanski, Krzysztof Marycz, and Rafal J. Wiglusz. 2020. "Nanohydroxyapatite-Mediated Imatinib Delivery for Specific Anticancer Applications" Molecules 25, no. 20: 4602. https://doi.org/10.3390/molecules25204602
APA StyleSobierajska, P., Serwotka-Suszczak, A., Szymanski, D., Marycz, K., & Wiglusz, R. J. (2020). Nanohydroxyapatite-Mediated Imatinib Delivery for Specific Anticancer Applications. Molecules, 25(20), 4602. https://doi.org/10.3390/molecules25204602








