The Underlying Mechanisms of Curcumin Inhibition of Hyperglycemia and Hyperlipidemia in Rats Fed a High-Fat Diet Combined With STZ Treatment
Abstract
1. Introduction
2. Results
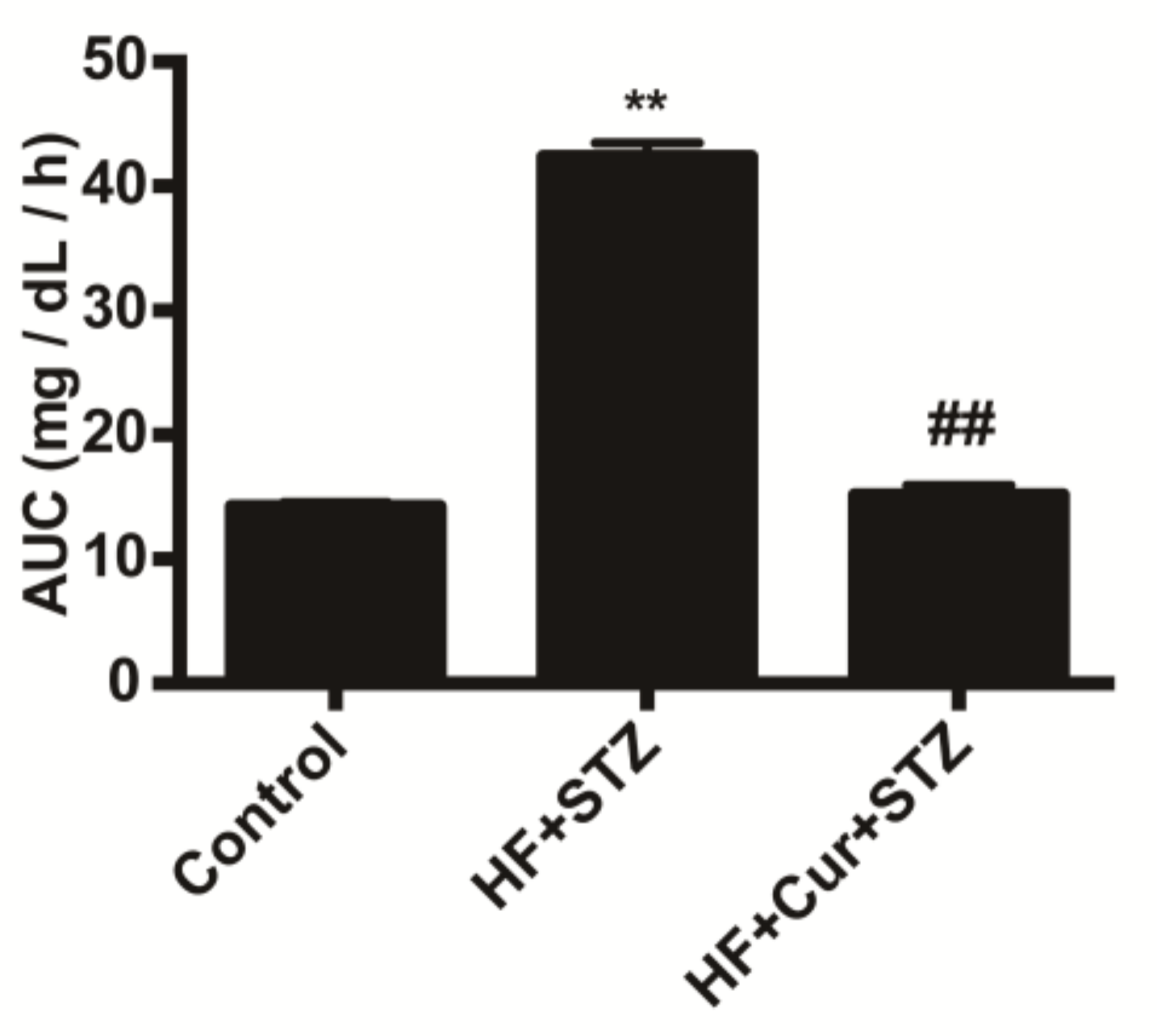
2.1. The Effect of Curcumin on Glucose Tolerance in Rats Fed A High-Fat Diet
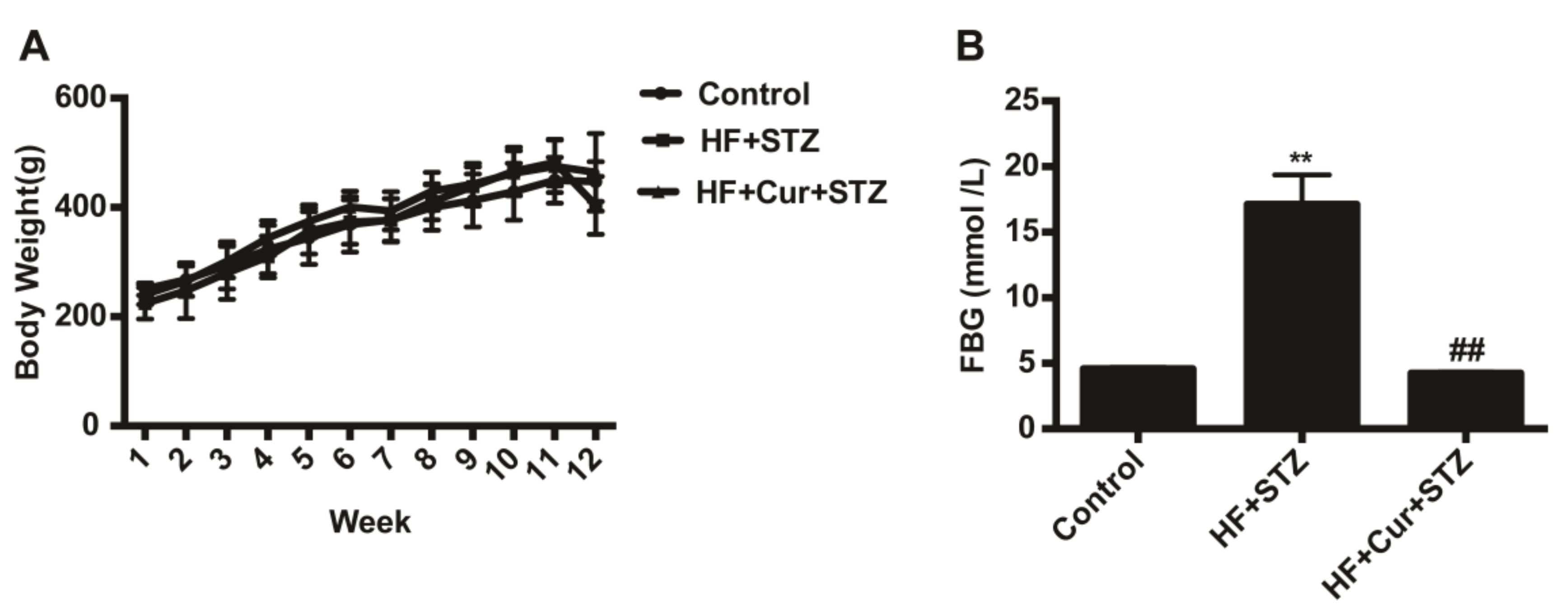
2.2. The Effect of Curcumin on Bodyweight and Fasting Blood Glucose (FBG) in Rats Fed a High-Fat Diet Combined with STZ Treatment
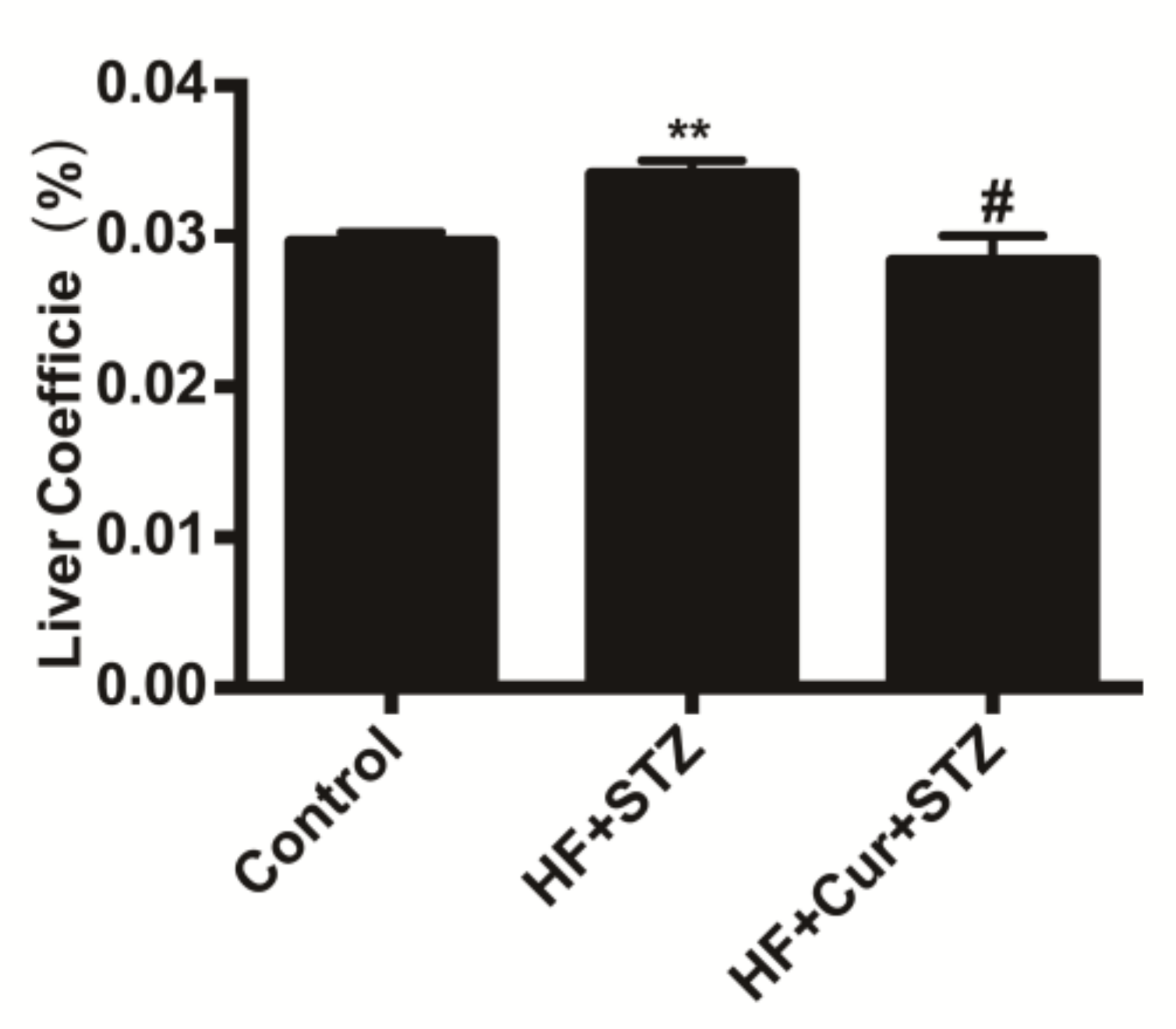
2.3. The Effect of Curcumin on the Coefficients of the Liver to Body Weight of Rats Fed A High-Fat Diet Combined with STZ Treatment
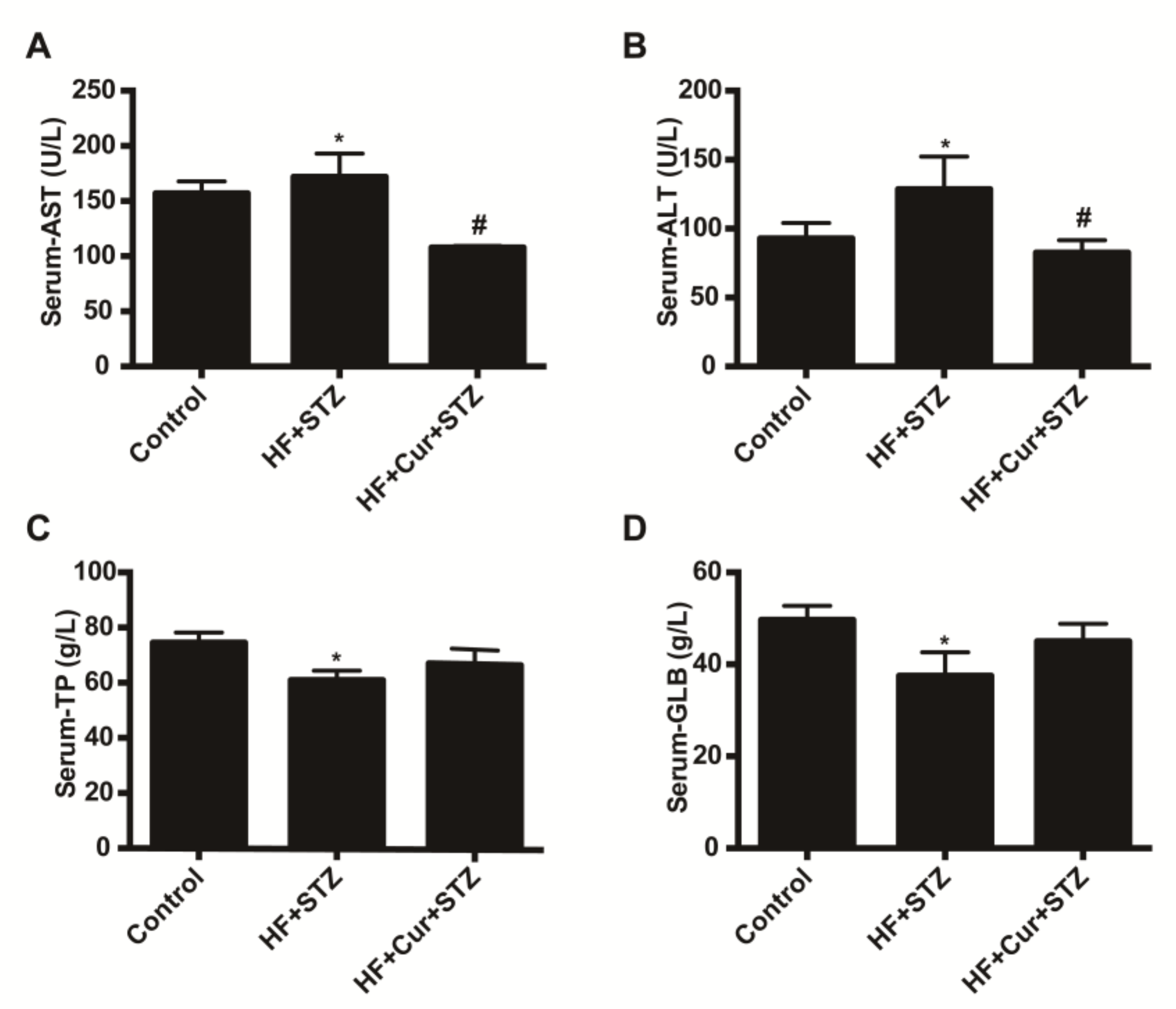
2.4. The Effect of Curcumin on Biochemical Blood Indicators in Rats Fed A High-Fat Diet Combined with STZ Treatment
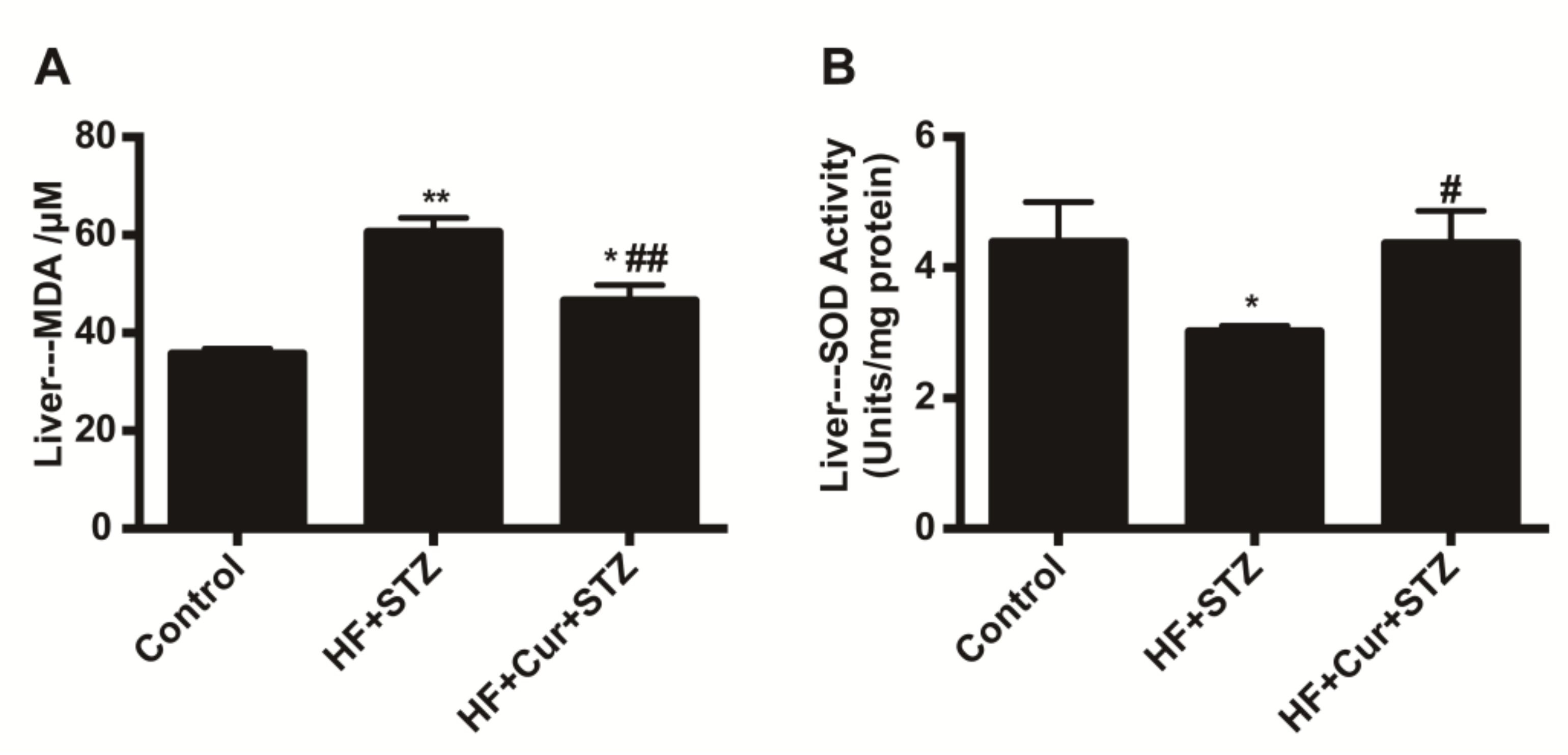
2.5. The Effect of Curcumin on Oxidative Stress Markers in Liver Tissues of Rats Fed A High-Fat Diet Combined with STZ Treatment
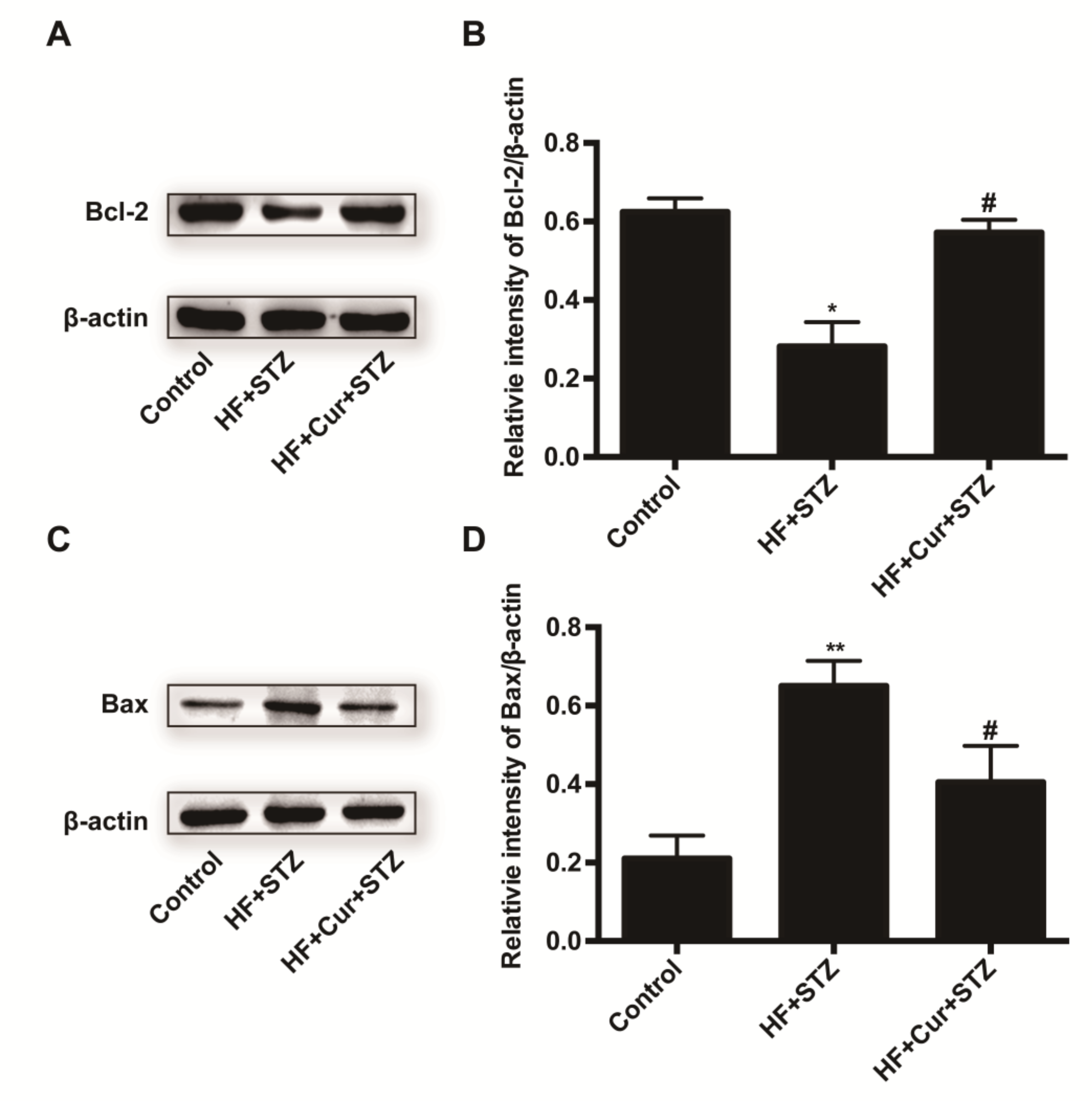
2.6. The Effect of Curcumin Intervention on Bcl-2 and Bax Expression in Rats Fed A High-Fat Diet Combined with STZ Treatment
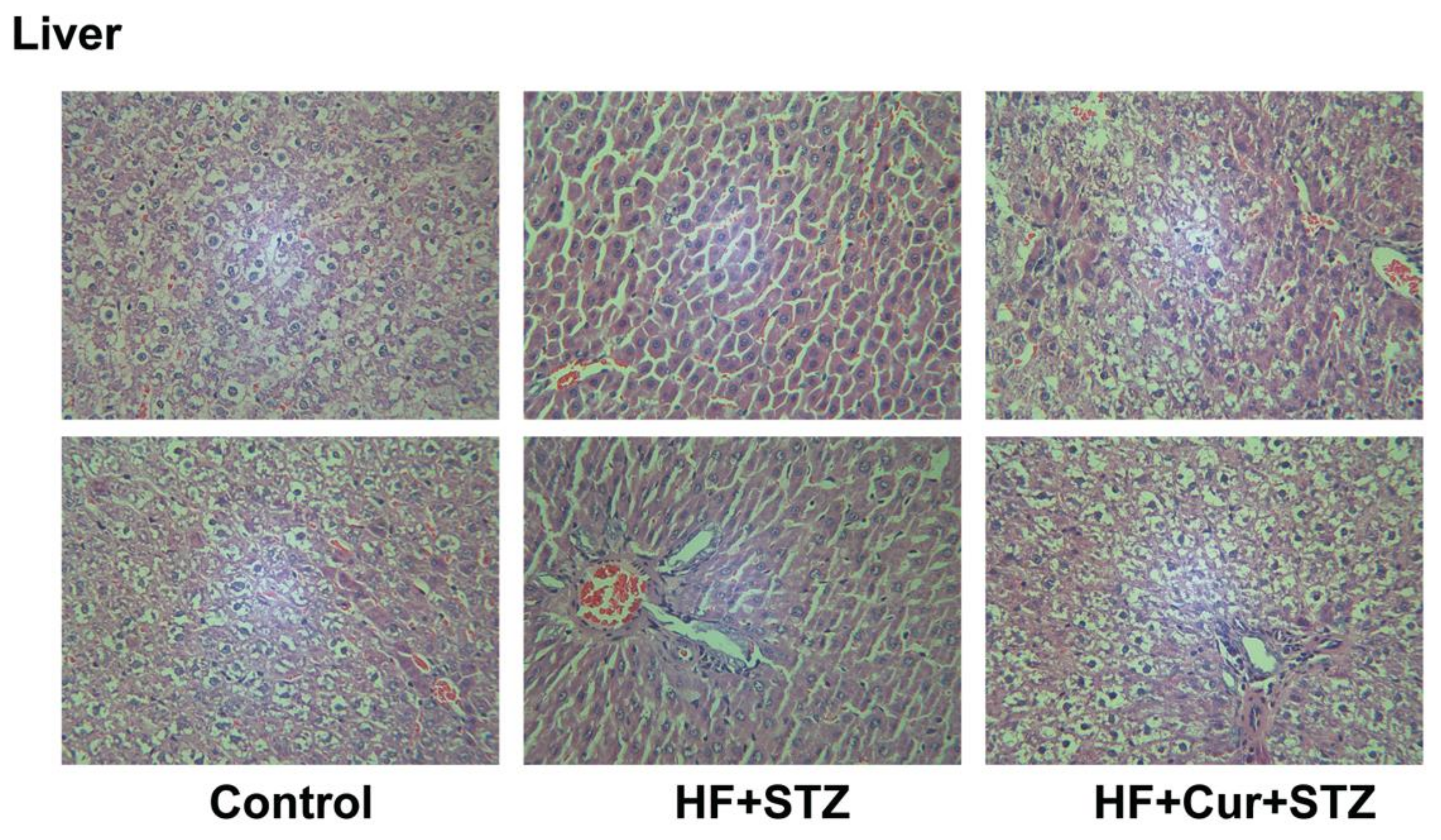
2.7. The Effect of Curcumin Treatment on Liver Histopathology in Rats Fed A High-Fat Diet Combined with STZ Treatment
3. Discussion
4. Materials and Methods
4.1. Chemicals and Reagent Kits
4.2. Experimental Animals
4.3. Experimental Design and Procedures
4.4. Determination of Glucose Tolerance in Experimented Rats
4.5. Estimation of Biochemical Parameters in the Blood×
4.6. Estimation of Oxidative Stress Markers and Apoptosis-Related Protein Expression in Liver Tissue
4.7. Histological Change Assessment in the Liver
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chaiyasut, C.; Kusirisin, W.; Lailerd, N.; Lerttrakarnnon, P.; Suttajit, M.; Srichairatanakool, S. Effects of phenolic compounds of fermented thai indigenous plants on oxidative stress in streptozotocin-induced diabetic rats. Evid. Based Complement. Alternat. Med. 2011, 2011, 749307. [Google Scholar] [CrossRef] [PubMed]
- Nandini, H.S.; Naik, P.R. Action of corilagin on hyperglycemia, hyperlipidemia and oxidative stress in streptozotocin-induced diabetic rats. Chem. Biol. Interact. 2019, 299, 186–193. [Google Scholar] [CrossRef]
- Su, L.Q.; Wang, Y.D.; Chi, H.Y. Effect of curcumin on glucose and lipid metabolism, FFAs and TNF-alpha in serum of type 2 diabetes mellitus rat models. Saudi J. Biol. Sci. 2017, 24, 1776–1780. [Google Scholar] [CrossRef]
- Van Raalte, D.H.; Verchere, C.B. Improving glycaemic control in type 2 diabetes: Stimulate insulin secretion or provide beta-cell rest? Diabetes Obes. Metab. 2017, 19, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Gebhardt, R.; Matz-Soja, M. Liver zonation: Novel aspects of its regulation and its impact on homeostasis. World J. Gastroenterol. 2014, 20, 8491–8504. [Google Scholar] [CrossRef] [PubMed]
- Trefts, E.; Gannon, M.; Wasserman, D.H. The liver. Curr. Biol. 2017, 27, R1147–R1151. [Google Scholar] [CrossRef] [PubMed]
- Staehr, P.; Hother-Nielsen, O.; Beck-Nielsen, H. The role of the liver in type 2 diabetes. Rev. Endocr. Metab. Disord. 2004, 5, 105–110. [Google Scholar] [CrossRef] [PubMed]
- McGarry, J.D. Banting lecture 2001: Dysregulation of fatty acid metabolism in the etiology of type 2 diabetes. Diabetes 2002, 51, 7–18. [Google Scholar] [CrossRef]
- Unger, R.H. Lipotoxicity in the pathogenesis of obesity-dependent NIDDM. Genetic and clinical implications. Diabetes 1995, 44, 863–870. [Google Scholar] [CrossRef]
- Prentki, M.; Corkey, B.E. Are the beta-cell signaling molecules malonyl-CoA and cystolic long-chain acyl-CoA implicated in multiple tissue defects of obesity and NIDDM? Diabetes 1996, 45, 273–283. [Google Scholar] [CrossRef]
- Firneisz, G. Non-alcoholic fatty liver disease and type 2 diabetes mellitus: The liver disease of our age? World J. Gastroenterol. 2014, 20, 9072–9089. [Google Scholar] [PubMed]
- Wang, K. Molecular mechanisms of hepatic apoptosis regulated by nuclear factors. Cell Signal. 2015, 27, 729–738. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative stress: An essential factor in the pathogenesis of gastrointestinal mucosal diseases. Physiol. Rev. 2014, 94, 329–354. [Google Scholar] [CrossRef] [PubMed]
- Turmel, H.; Hartmann, A.; Parain, K.; Douhou, A.; Srinivasan, A.; Agid, Y.; Hirsch, E.C. Caspase-3 activation in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-treated mice. Mov. Disord. 2001, 16, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Qin, J.; Zhen, X.; Yang, Q.; Huang, L. Curcumin protects against hepatic stellate cells activation and migration by inhibiting the CXCL12/CXCR4 biological axis in liver fibrosisA study in vitro and in vivo. Biomed. Pharmacother. 2018, 101, 599–607. [Google Scholar] [CrossRef]
- Wojcik, M.; Krawczyk, M.; Wojcik, P.; Cypryk, K.; Wozniak, L.A. Molecular Mechanisms Underlying Curcumin-Mediated Therapeutic Effects in Type 2 Diabetes and Cancer. Oxid. Med. Cell. Longev. 2018, 2018, 9698258. [Google Scholar] [CrossRef]
- Ha, H.; Lee, H.B. Reactive oxygen species as glucose signaling molecules in mesangial cells cultured under high glucose. Kidney Int Suppl. 2000, 77, S19–S25. [Google Scholar] [CrossRef]
- Masuda, T.; Hidaka, K.; Shinohara, A.; Maekawa, T.; Takeda, Y.; Yamaguchi, H. Chemical studies on antioxidant mechanism of curcuminoid: Analysis of radical reaction products from curcumin. J. Agric. Food Chem. 1999, 47, 71–77. [Google Scholar] [CrossRef]
- Goel, A.; Kunnumakkara, A.B.; Aggarwal, B.B. Curcumin as “Curecumin”: From kitchen to clinic. Biochem. Pharmacol. 2008, 75, 787–809. [Google Scholar] [CrossRef]
- Kuttan, R.; Bhanumathy, P.; Nirmala, K.; George, M.C. Potential anticancer activity of turmeric (Curcuma longa). Cancer Lett. 1985, 29, 197–202. [Google Scholar] [CrossRef]
- El-Moselhy, M.A.; Taye, A.; Sharkawi, S.S.; El-Sisi, S.F.; Ahmed, A.F. The antihyperglycemic effect of curcumin in high fat diet fed rats. Role of TNF-alpha and free fatty acids. Food. Chem. Toxicol. 2011, 49, 1129–1140. [Google Scholar] [CrossRef] [PubMed]
- Suryanarayana, P.; Saraswat, M.; Mrudula, T.; Krishna, T.P.; Krishnaswamy, K.; Reddy, G.B. Curcumin and turmeric delay streptozotocin-induced diabetic cataract in rats. Investg. Ophthalmol. Vis. Sci. 2005, 46, 2092–2099. [Google Scholar] [CrossRef] [PubMed]
- Farhangkhoee, H.; Khan, Z.A.; Chen, S.; Chakrabarti, S. Differential effects of curcumin on vasoactive factors in the diabetic rat heart. Nutr. Metab. Lond. 2006, 3, 27. [Google Scholar] [CrossRef] [PubMed]
- Vivian, S.; Sari, F.R.; Veeraveedu, P.T.; Thandavarayan, R.A.; Meilei, H.; Vijayakumar, S.; Arun Prasath, L.; Kenji, S.; Hiroshi, K.; Kenichi, W.; et al. Curcumin ameliorates macrophage infiltration by inhibiting NF-κB activation and proinflammatory cytokines in streptozotocin induced-diabetic nephropathy. Nutr. Metab. Lond. 2011, 8, 35. [Google Scholar]
- Garcia Alloza, M.; Borrelli, L.A.; Rozkalne, A.; Hyman, B.T.; Bacskai, B.J. Curcumin labels amyloid pathology in vivo, disrupts existing plaques, and partially restores distorted neurites in an Alzheimer mouse model. J. Neurochem. 2007, 102, 1095–1104. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Zhang, D.; Yang, Z.; Hu, X.; Qian, S.; Liu, J.; Willson, B.; Block, M.; Hong, J.-S. Curcumin protects dopaminergic neuron against LPS induced neurotoxicity in primary rat neuron/glia culture. Neurochem. Res. 2008, 33, 2044–2053. [Google Scholar] [CrossRef] [PubMed]
- Mehla, J.; Reeta, K.H.; Gupta, P.; Gupta, Y.K. Protective effect of curcumin against seizures and cognitive impairment in a pentylenetetrazole-kindled epileptic rat model. Life Sci. 2010, 87, 596–603. [Google Scholar] [CrossRef]
- Xu, L.; Li, Y.; Dai, Y.; Peng, J. Natural products for the treatment of type 2 diabetes mellitus: Pharmacology and mechanisms. Pharmacol. Res. 2018, 130, 451–465. [Google Scholar] [CrossRef]
- Park, S.K.; Ryoo, J.H.; Oh, C.M.; Choi, J.M.; Choi, Y.J.; Lee, K.O.; Jung, J.Y. The risk of type 2 diabetes mellitus according to 2-h plasma glucose level: The Korean Genome and Epidemiology Study (KoGES). Diabetes Res. Clin. Pract. 2018, 146, 130–137. [Google Scholar] [CrossRef]
- Ferrannini, E.; Gastaldelli, A.; Miyazaki, Y.; Matsuda, M.; Pettiti, M.; Natali, A.; Mari, A.; DeFronzo, R.A. Predominant role of reduced beta-cell sensitivity to glucose over insulin resistance in impaired glucose tolerance. Diabetologia 2003, 46, 1211–1219. [Google Scholar] [CrossRef]
- De Andrade Mesquita, L.; Pavan Antoniolli, L.; Cittolin-Santos, G.F.; Gerchman, F. Distinct metabolic profile according to the shape of the oral glucose tolerance test curve is related to whole glucose excursion: A cross-sectional study. BMC Endocr. Disord. 2018, 18, 56. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Xiao, X.; Zheng, J.; Li, M.; Yu, M.; Ping, F.; Wang, T.; Wang, X. Influence of Maternal Inulin-Type Prebiotic Intervention on Glucose Metabolism and Gut Microbiota in the Offspring of C57BL Mice. Front. Endocrinol. Lausanne 2019, 10, 675. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Sun, X.; Xiao, X.; Zheng, J.; Li, M.; Yu, M.; Ping, F.; Wang, Z.; Qi, C.; Wang, T.; et al. Maternal chromium restriction induces insulin resistance in adult mice offspring through miRNA. Int. J. Mol. Med. 2018, 41, 1547–1559. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Nishikawa, S.; Ikehata, A.; Dochi, K.; Tani, T.; Takahashi, T.; Imaizumi, A.; Tsuda, T. Curcumin improves glucose tolerance via stimulation of glucagon-like peptide-1 secretion. Mol. Nutr. Food Res. 2017, 61, 1600471. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Compean, D.; Gonzalez-Gonzalez, J.A.; Lavalle-Gonzalez, F.J.; Gonzalez-Moreno, E.I.; Maldonado-Garza, H.J.; Villarreal-Perez, J.Z. Current Concepts in Diabetes Mellitus and Chronic Liver Disease: Clinical Outcomes, Hepatitis C Virus Association, and Therapy. Dig. Dis. Sci. 2016, 61, 371–380. [Google Scholar] [CrossRef]
- Harb, A.A.; Bustanji, Y.K.; Almasri, I.M.; Abdalla, S.S. Eugenol Reduces LDL Cholesterol and Hepatic Steatosis in Hypercholesterolemic Rats by Modulating TRPV1 Receptor. Sci. Rep. 2019, 9, 14003. [Google Scholar] [CrossRef]
- Zhang, Y.; Wei, D.D.; Yuan, R.R.; Ge, Q.X.; Chen, F.; Yang, S.X.; Chen, B.P.; Shi, J. Effects of ApoE gene polymorphism on the efficacy of atorvastatin in the treatment of hyperlipidemia. Zhonghua Yi Xue Za Zhi 2017, 97, 291–294. [Google Scholar]
- Wilson, P.W. Assessing coronary heart disease risk with traditional and novel risk factors. Clin. Cardiol. 2004, 27, 7–11. [Google Scholar] [CrossRef]
- Jaiswal, M.; Schinske, A.; Pop-Busui, R. Lipids and lipid management in diabetes. Best Pract. Res. Clin. Endocrinol. Metab. 2014, 28, 325–338. [Google Scholar] [CrossRef]
- Pan, Y.; Zhao, D.; Yu, N.; An, T.; Miao, J.; Mo, F.; Gu, Y.; Zhang, D.; Gao, S.; Jiang, G.; et al. Curcumin improves glycolipid metabolism through regulating peroxisome proliferator activated receptor gamma signalling pathway in high-fat diet-induced obese mice and 3T3-L1 adipocytes. R. Soc. Open Sci. 2017, 4, 170917. [Google Scholar] [CrossRef]
- Xu, N.; Xu, H.; Zhao, M.; Xu, Y.; Huang, L. Associations of systemic, serum lipid and lipoprotein metabolic pathway gene variations with polypoidal choroidal vasculopathy in China. PLoS ONE 2019, 14, e0226763. [Google Scholar] [CrossRef] [PubMed]
- Mineo, C.; Deguchi, H.; Griffin, J.H.; Shaul, P.W. Endothelial and antithrombotic actions of HDL. Circ. Res. 2006, 98, 1352–1364. [Google Scholar] [CrossRef] [PubMed]
- Hamed, A.E.; Elwan, N.; Naguib, M.; Elwakil, R.; Esmat, G.; El Kassas, M.; Abd-Elsalam, S.; Moussa, S. Diabetes Association with Liver Diseases: An Overview for Clinicians. Endocr. Metab. Immune Disord. Drug Targets 2019, 19, 274–280. [Google Scholar] [CrossRef] [PubMed]
- Ahmadieh, H.; Azar, S.T. Liver disease and diabetes: Association, pathophysiology, and management. Diabetes Res. Clin. Pract. 2014, 104, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Han, H.S.; Kang, G.; Kim, J.S.; Choi, B.H.; Koo, S.H. Regulation of glucose metabolism from a liver-centric perspective. Exp. Mol. Med. 2016, 48, e218. [Google Scholar] [CrossRef]
- Garcia-Compean, D.; Gonzalez-Gonzalez, J.A.; Lavalle-Gonzalez, F.J.; Gonzalez-Moreno, E.I.; Villarreal-Perez, J.Z.; Maldonado-Garza, H.J. The treatment of diabetes mellitus of patients with chronic liver disease. Ann. Hepatol. 2015, 14, 780–788. [Google Scholar] [CrossRef]
- Lucchesi, A.N.; Freitas, N.T.; Cassettari, L.L.; Marques, S.F.; Spadella, C.T. Diabetes mellitus triggers oxidative stress in the liver of alloxan-treated rats: A mechanism for diabetic chronic liver disease. Acta Cir. Bras. 2013, 28, 502–508. [Google Scholar] [CrossRef]
- Gupta, S.K.; Kumar, B.; Nag, T.C.; Agrawal, S.S.; Agrawal, R.; Agrawal, P.; Saxena, R.; Srivastava, S. Curcumin prevents experimental diabetic retinopathy in rats through its hypoglycemic, antioxidant, and anti-inflammatory mechanisms. J. Ocul. Pharmacol. Ther. 2011, 27, 123–130. [Google Scholar] [CrossRef]
- Zhong, W.; Qian, K.; Xiong, J.; Ma, K.; Wang, A.; Zou, Y. Curcumin alleviates lipopolysaccharide induced sepsis and liver failure by suppression of oxidative stress-related inflammation via PI3K/AKT and NF-kappaB related signaling. Biomed. Pharmacother. 2016, 83, 302–313. [Google Scholar] [CrossRef]
- Huang, Z.; Ye, B.; Dai, Z.; Wu, X.; Lu, Z.; Shan, P.; Huang, W. Curcumin inhibits autophagy and apoptosis in hypoxia/reoxygenation-induced myocytes. Mol. Med. Rep. 2015, 11, 4678–4684. [Google Scholar] [CrossRef]
- Daverey, A.; Agrawal, S.K. Pre and post treatment with curcumin and resveratrol protects astrocytes after oxidative stress. Brain Res. 2018, 1692, 45–55. [Google Scholar] [CrossRef] [PubMed]
- Safhi, M.M.; Alam, M.F.; Sivakumar, S.M.; Anwer, T. Hepatoprotective Potential of Sargassum muticum against STZ-Induced Diabetic Liver Damage in Wistar Rats by Inhibiting Cytokines and the Apoptosis Pathway. Anal. Cell. Pathol. Amst. 2019, 2019, 7958701. [Google Scholar] [CrossRef] [PubMed]
- Rezaie, A.; Parker, R.D.; Abdollahi, M. Oxidative stress and pathogenesis of inflammatory bowel disease: An epiphenomenon or the cause? Dig. Dis. Sci. 2007, 52, 2015–2021. [Google Scholar] [CrossRef] [PubMed]
- Janero, D.R. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic. Biol. Med. 1990, 9, 515–540. [Google Scholar] [CrossRef]
- Yang, E.; Korsmeyer, S.J. Molecular thanatopsis: A discourse on the BCL2 family and cell death. Blood 1996, 88, 386–401. [Google Scholar] [CrossRef]
- Vaux, D.L.; Strasser, A. The molecular biology of apoptosis. Pro. Natl. Acad. Sci. USA 1996, 93, 2239–2244. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xia, Z.-H.; Chen, W.-B.; Shi, L.; Jiang, X.; Li, K.; Wang, Y.-X.; Liu, Y.-Q. The Underlying Mechanisms of Curcumin Inhibition of Hyperglycemia and Hyperlipidemia in Rats Fed a High-Fat Diet Combined With STZ Treatment. Molecules 2020, 25, 271. https://doi.org/10.3390/molecules25020271
Xia Z-H, Chen W-B, Shi L, Jiang X, Li K, Wang Y-X, Liu Y-Q. The Underlying Mechanisms of Curcumin Inhibition of Hyperglycemia and Hyperlipidemia in Rats Fed a High-Fat Diet Combined With STZ Treatment. Molecules. 2020; 25(2):271. https://doi.org/10.3390/molecules25020271
Chicago/Turabian StyleXia, Zhen-Hong, Wen-Bo Chen, Li Shi, Xue Jiang, Ke Li, Yu-Xiang Wang, and Yan-Qiang Liu. 2020. "The Underlying Mechanisms of Curcumin Inhibition of Hyperglycemia and Hyperlipidemia in Rats Fed a High-Fat Diet Combined With STZ Treatment" Molecules 25, no. 2: 271. https://doi.org/10.3390/molecules25020271
APA StyleXia, Z.-H., Chen, W.-B., Shi, L., Jiang, X., Li, K., Wang, Y.-X., & Liu, Y.-Q. (2020). The Underlying Mechanisms of Curcumin Inhibition of Hyperglycemia and Hyperlipidemia in Rats Fed a High-Fat Diet Combined With STZ Treatment. Molecules, 25(2), 271. https://doi.org/10.3390/molecules25020271




