Urinary Resveratrol Metabolites Output: Differential Associations with Cardiometabolic Markers and Liver Enzymes in House-Dwelling Subjects Featuring Metabolic Syndrome
Abstract
1. Introduction
2. Results
2.1. Participant Characteristics
2.2. Association between Total Urine Resveratrol Metabolites Concentrations, Cardiometabolic Profile, and Liver Markers
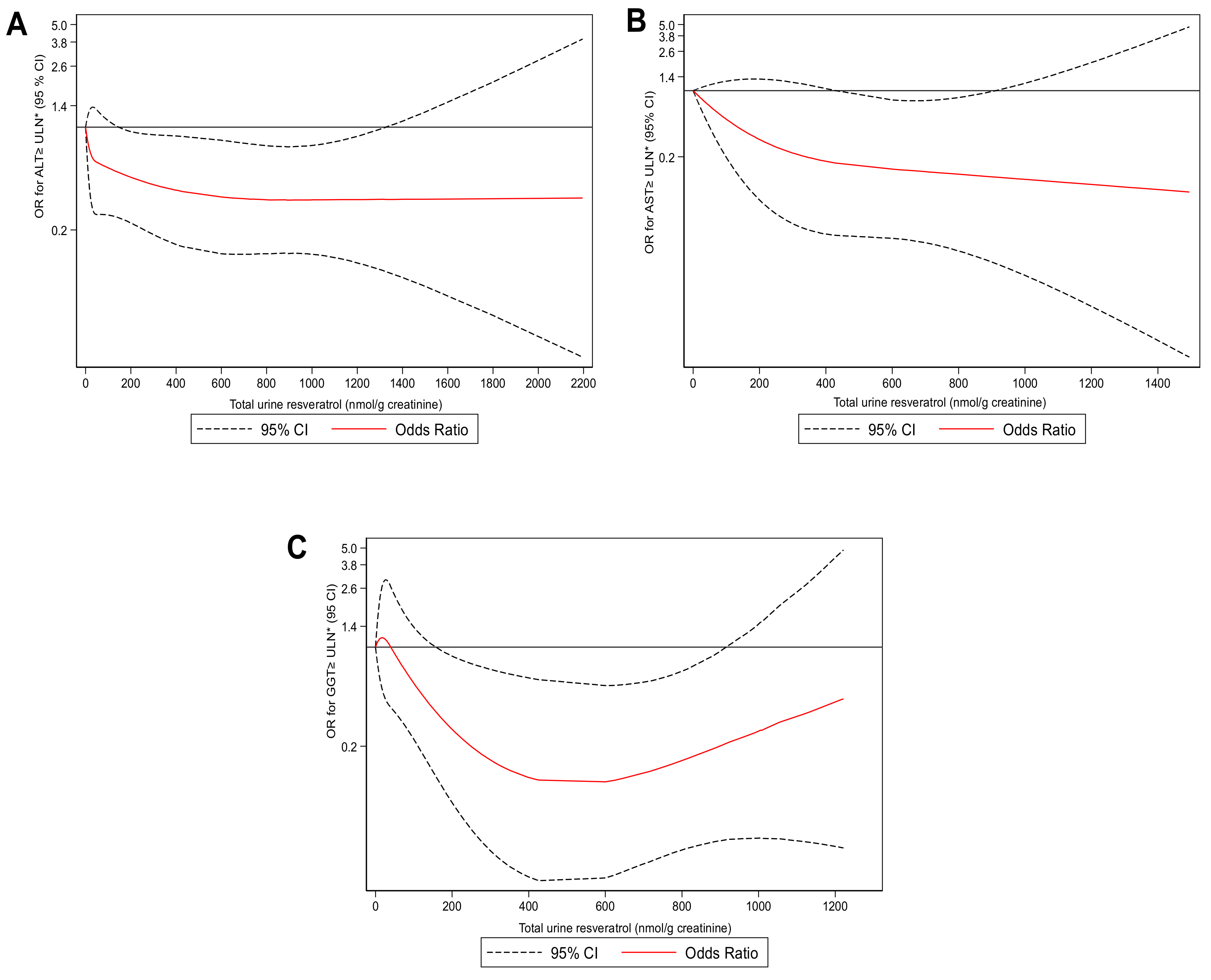
2.3. Risk of Higher Liver Enzymes and Total Urine Resveratrol Metabolites
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Sociodemographic, Clinical, Anthropometric, and Body Composition Variables
4.3. Dietary Record
4.4. Urine and Plasma Collection, and Biochemical Determinations
4.5. Urine Resveratrol Metabolites Measurements
4.6. Glucose Homeostasis and Liver Markers Measurements
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Eckel, R.H.; Alberti, K.; Grundy, S.M.; Zimmet, P.Z. The metabolic syndrome. Lancet 2010, 375, 181–183. [Google Scholar] [CrossRef]
- Younossi, Z.M. Non-alcoholic fatty liver disease—A global public health perspective. J. Hepatol. 2019, 70, 531–544. [Google Scholar] [CrossRef] [PubMed]
- Chalasani, N.; Younossi, Z.; Lavine, J.E.; Charlton, M.; Cusi, K.; Rinella, M.; Harrison, S.A.; Brunt, E.M.; Sanyal, A.J. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology 2018, 67, 328–357. [Google Scholar] [CrossRef]
- European Association for the Study of the Liver; European Association for the Study of Diabetes; European Association for the Study of Obesity. EASL–EASD–EASO Clinical Practice Guidelines for the management of non-alcoholic fatty liver disease. J. Hepatol. 2016, 64, 1388–1402. [Google Scholar] [CrossRef]
- Bullón-Vela, M.V.; Abete, I.; Martínez, J.A.; Zulet, M.A. Obesity and Nonalcoholic Fatty Liver Disease: Role of Oxidative Stress. In Obesity: Oxidative Stress and Dietary Antioxidants; Moral, A.M.d., García, M.A.C., Eds.; Academic Press: London, UK, 2018; pp. 111–133. ISBN 9780128125045. [Google Scholar]
- Khan, R.S.; Bril, F.; Cusi, K.; Newsome, P.N. Modulation of Insulin Resistance in Nonalcoholic Fatty Liver Disease. Hepatology 2019, 70. [Google Scholar] [CrossRef] [PubMed]
- Kawano, Y.; Cohen, D.E. Mechanisms of hepatic triglyceride accumulation in non-alcoholic fatty liver disease. J. Gastroenterol. 2013, 48, 434–441. [Google Scholar] [CrossRef] [PubMed]
- Tebani, A.; Bekri, S. Paving the way to precision nutrition through metabolomics. Front. Nutr. 2019, 6, 41. [Google Scholar] [CrossRef]
- Zelber-Sagi, S.; Salomone, F.; Mlynarsky, L. The Mediterranean dietary pattern as the diet of choice for non-alcoholic fatty liver disease: Evidence and plausible mechanisms. Liver Int. 2017, 37, 936–949. [Google Scholar] [CrossRef]
- Martínez-González, M.A.; Bastarrika, G. Mediterranean diet as the ideal model for preventing non-alcoholic fatty liver disease (NAFLD). Hepatobiliary Surg. Nutr. 2020, 9, 379–381. [Google Scholar] [CrossRef]
- Godos, J.; Federico, A.; Dallio, M.; Scazzina, F. Mediterranean diet and nonalcoholic fatty liver disease: Molecular mechanisms of protection. Int. J. Food Sci. Nutr. 2017, 68, 18–27. [Google Scholar] [CrossRef]
- Bullón-Vela, V.; Abete, I.; Tur, J.A.; Pintó, X.; Corbella, E.; Martínez-González, M.A.; Toledo, E.; Corella, D.; Macías, M.; Tinahones, F.; et al. Influence of lifestyle factors and staple foods from the Mediterranean diet on non-alcoholic fatty liver disease among older individuals with metabolic syndrome features. Nutrition 2020, 71, 110620. [Google Scholar] [CrossRef] [PubMed]
- Silva Figueiredo, P.; Inada, A.; Ribeiro Fernandes, M.; Granja Arakaki, D.; Freitas, K.; Avellaneda Guimarães, R.; Aragão do Nascimento, V.; Aiko Hiane, P. An Overview of Novel Dietary Supplements and Food Ingredients in Patients with Metabolic Syndrome and Non-Alcoholic Fatty Liver Disease. Molecules 2018, 23, 877. [Google Scholar] [CrossRef] [PubMed]
- Chiva-Blanch, G.; Arranz, S.; Lamuela-Raventos, R.M.; Estruch, R. Effects of wine, alcohol and polyphenols on cardiovascular disease risk factors: Evidences from human studies. Alcohol Alcohol. 2013, 48, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Valenti, L.; Riso, P.; Mazzocchi, A.; Porrini, M.; Fargion, S.; Agostoni, C. Dietary Anthocyanins as Nutritional Therapy for Nonalcoholic Fatty Liver Disease. Oxidative Med. Cell. Longev. 2013, 2013, 1–8. [Google Scholar] [CrossRef]
- Burns, J.; Yokota, T.; Ashihara, H.; Lean, M.E.J.; Crozier, A. Plant foods and herbal sources of resveratrol. J. Agric. Food Chem. 2002, 50, 3337–3340. [Google Scholar] [CrossRef] [PubMed]
- Jeandet, P.; Delaunois, B.; Conreux, A.; Donnez, D.; Nuzzo, V.; Cordelier, S.; Clément, C.; Courot, E. Biosynthesis, metabolism, molecular engineering, and biological functions of stilbene phytoalexins in plants. BioFactors 2010, 36, 331–341. [Google Scholar] [CrossRef]
- Noh, H.; Freisling, H.; Assi, N.; Zamora-Ros, R.; Achaintre, D.; Affret, A.; Mancini, F.; Boutron-Ruault, M.-C.; Flögel, A.; Boeing, H.; et al. Identification of Urinary Polyphenol Metabolite Patterns Associated with Polyphenol-Rich Food Intake in Adults from Four European Countries. Nutrients 2017, 9, 796. [Google Scholar] [CrossRef]
- Springer, M.; Moco, S. Resveratrol and its human metabolites—Effects on metabolic health and obesity. Nutrients 2019, 11, 143. [Google Scholar] [CrossRef]
- Rotches-Ribalta, M.; Andres-Lacueva, C.; Estruch, R.; Escribano, E.; Urpi-Sarda, M. Pharmacokinetics of resveratrol metabolic profile in healthy humans after moderate consumption of red wine and grape extract tablets. Pharmacol. Res. 2012, 66, 375–382. [Google Scholar] [CrossRef]
- Ramírez-Garza, S.L.; Laveriano-Santos, E.P.; Marhuenda-Muñoz, M.; Storniolo, C.E.; Tresserra-Rimbau, A.; Vallverdú-Queralt, A.; Lamuela-Raventós, R.M. Health effects of resveratrol: Results from human intervention trials. Nutrients 2018, 10, 1892. [Google Scholar] [CrossRef]
- Walle, T.; Hsieh, F.; DeLegge, M.H.; Oatis, J.E.; Walle, U.K. High absorption but very low bioavailability of oral resveratrol in humans. Drug Metab. Dispos. 2004, 32, 1377–1382. [Google Scholar] [CrossRef]
- Jeandet, P.; Sobarzo-Sánchez, E.; Silva, A.S.; Clément, C.; Nabavi, S.F.; Battino, M.; Rasekhian, M.; Belwal, T.; Habtemariam, S.; Koffas, M.; et al. Whole-cell biocatalytic, enzymatic and green chemistry methods for the production of resveratrol and its derivatives. Biotechnol. Adv. 2020, 39, 107461. [Google Scholar] [CrossRef]
- Gambini, J.; Inglés, M.; Olaso, G.; Lopez-Grueso, R.; Bonet-Costa, V.; Gimeno-Mallench, L.; Mas-Bargues, C.; Abdelaziz, K.M.; Gomez-Cabrera, M.C.; Vina, J.; et al. Properties of Resveratrol: In Vitro and In Vivo Studies about Metabolism, Bioavailability, and Biological Effects in Animal Models and Humans. Oxidative Med. Cell. Longev. 2015, 2015, 13. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Milenkovic, D.; Van de Wiele, T.; Rodriguez-Mateos, A.; de Roos, B.; Garcia-Conesa, M.T.; Landberg, R.; Gibney, E.R.; Heinonen, M.; Tomás-Barberán, F.; et al. Addressing the inter-individual variation in response to consumption of plant food bioactives: Towards a better understanding of their role in healthy aging and cardiometabolic risk reduction. Mol. Nutr. Food Res. 2017, 61, 1600557. [Google Scholar] [CrossRef]
- Charytoniuk, T.; Drygalski, K.; Konstantynowicz-Nowicka, K.; Berk, K.; Chabowski, A. Alternative treatment methods attenuate the development of NAFLD: A review of resveratrol molecular mechanisms and clinical trials. Nutrition 2017, 34, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Wu, C.; Qiu, S.; Yuan, X.; Li, L. Effects of resveratrol on glucose control and insulin sensitivity in subjects with type 2 diabetes: Systematic review and meta-analysis. Nutr. Metab. 2017, 14, 1–10. [Google Scholar] [CrossRef]
- Zhao, H.; Song, A.; Zhang, Y.; Shu, L.; Song, G.; Ma, H. Effect of Resveratrol on Blood Lipid Levels in Patients with Type 2 Diabetes: A Systematic Review and Meta-Analysis. Obesity 2019, 27, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Pannu, N.; Bhatnagar, A. Resveratrol: From enhanced biosynthesis and bioavailability to multitargeting chronic diseases. Biomed. Pharmacother. 2019, 109, 2237–2251. [Google Scholar] [CrossRef]
- Suzuki, A.; Abdelmalek, M.F. Nonalcoholic Fatty Liver Disease in Women. Women’s Health 2009, 5, 191–203. [Google Scholar] [CrossRef]
- Vela, M.V.B.; Abete, I.; Zulet, M.D.L.; Tur, J.A.; Pintó, X.; Corbella, E.; González, M.A.M.; Corella, D.; González, M.M.; Ros, E.; et al. Risk factors differentially associated with non-alcoholic fatty liver disease in males and females with metabolic syndrome. Rev. Española Enferm. Dig. 2019, 112, 111–133. [Google Scholar] [CrossRef]
- Ros, E.; Martínez-González, M.A.; Estruch, R.; Salas-Salvadó, J.; Fitó, M.; Martínez, J.A.; Corella, D. Mediterranean Diet and Cardiovascular Health: Teachings of the PREDIMED Study. Adv. Nutr. 2014, 5, 330S–336S. [Google Scholar] [CrossRef] [PubMed]
- Tejada, S.; Capó, X.; Mascaró, C.M.; Monserrat-Mesquida, M.; Quetglas-Llabrés, M.M.; Pons, A.; Tur, J.A.; Sureda, A. Hepatoprotective effects of resveratrol in non-alcoholic fatty live disease. Curr. Pharm. Des. 2020, 26. [Google Scholar] [CrossRef] [PubMed]
- Theodotou, M.; Fokianos, K.; Mouzouridou, A.; Konstantinou, C.; Aristotelous, A.; Prodromou, D.; Chrysikou, A. The effect of resveratrol on hypertension: A clinical trial. Exp. Ther. Med. 2017, 13, 295–301. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Urpi-Sarda, M.; Lamuela-Raventós, R.M.; Martínez-González, M.Á.; Salas-Salvadó, J.; Arós, F.; Fitó, M.; Lapetra, J.; Estruch, R.; Andres-Lacueva, C. High urinary levels of resveratrol metabolites are associated with a reduction in the prevalence of cardiovascular risk factors in high-risk patients. Pharmacol. Res. 2012, 65, 615–620. [Google Scholar] [CrossRef] [PubMed]
- Almeida, L.; Vaz-da-Silva, M.; Falcão, A.; Soares, E.; Costa, R.; Loureiro, A.I.; Fernandes-Lopes, C.; Rocha, J.F.; Nunes, T.; Wright, L.; et al. Pharmacokinetic and safety profile of trans-resveratrol in a rising multiple-dose study in healthy volunteers. Mol. Nutr. Food Res. 2009, 53, 7–15. [Google Scholar] [CrossRef]
- Urpí-Sardà, M.; Jáuregui, O.; Lamuela-Raventós, R.M.; Jaeger, W.; Miksits, M.; Covas, M.-I.; Andres-Lacueva, C. Uptake of Diet Resveratrol into the Human Low-Density Lipoprotein. Identification and Quantification of Resveratrol Metabolites by Liquid Chromatography Coupled with Tandem Mass Spectrometry. Anal. Chem. 2005, 77, 3149–3155. [Google Scholar] [CrossRef]
- Khalil, A.; Berrougui, H. Editorial: Mechanism of action of resveratrol in lipid metabolism and atherosclerosis. Clin. Lipidol. 2009, 4, 527–531. [Google Scholar] [CrossRef]
- Semba, R.D.; Ferrucci, L.; Bartali, B.; Urpí-Sarda, M.; Zamora-Ros, R.; Sun, K.; Cherubini, A.; Bandinelli, S.; Andres-Lacueva, C. Resveratrol levels and all-cause mortality in older community-dwelling adults. JAMA Intern. Med. 2014, 174, 1077–1084. [Google Scholar] [CrossRef]
- Choi, Y.-J.; Suh, H.-R.; Yoon, Y.; Lee, K.-J.; Kim, D.G.; Kim, S.; Lee, B.-H. Protective effect of resveratrol derivatives on high-fat diet induced fatty liver by activating AMP-activated protein kinase. Arch. Pharmacal Res. 2014, 37, 1169–1176. [Google Scholar] [CrossRef]
- Gouédard, C.; Barouki, R.; Morel, Y. Induction of the paraoxonase-1 gene expression by resveratrol. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 2378–2383. [Google Scholar] [CrossRef]
- Armutcu, F.; Akyol, S.; Ucar, F.; Erdogan, S.; Akyol, O. Chapter Three—Markers in Nonalcoholic Steatohepatitis, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2013; Volume 61, ISBN 9780124076808. [Google Scholar]
- Min, H.K.; Kapoor, A.; Fuchs, M.; Mirshahi, F.; Zhou, H.; Maher, J.; Kellum, J.; Warnick, R.; Contos, M.J.; Sanyal, A.J. Increased hepatic synthesis and dysregulation of cholesterol metabolism is associated with the severity of nonalcoholic fatty liver disease. Cell Metab. 2012, 15, 665–674. [Google Scholar] [CrossRef]
- Wong, R.H.X.; Howe, P.R.C. Resveratrol counteracts insulin resistance—Potential role of the circulation. Nutrients 2018, 10, 1160. [Google Scholar] [CrossRef] [PubMed]
- van der Made, S.M.; Plat, J.; Mensink, R.P. Resveratrol Does Not Influence Metabolic Risk Markers Related to Cardiovascular Health in Overweight and Slightly Obese Subjects: A Randomized, Placebo-Controlled Crossover Trial. PLoS ONE 2015, 10, e0118393. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yuan, W.; Fang, J.; Wang, W.; He, P.; Lei, J.; Wang, C. Efficacy of resveratrol supplementation against non-alcoholic fatty liver disease: A meta-analysis of placebo-controlled clinical trials. PLoS ONE 2016, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Elgebaly, A.; Radwan, I.A.I.; Aboelnas, M.M.; Ibrahim, H.H.; Eltoomy, M.F.M.; Atta, A.A.; Mesalam, H.A.; Sayed, A.A.; Othman, A.A. Resveratrol supplementation in patients with non-alcoholic fatty liver disease: Systematic review and meta-analysis. J. Gastrointest. Liver Dis. 2017, 26, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Faghihzadeh, F.; Adibi, P.; Rafiei, R.; Hekmatdoost, A. Resveratrol supplementation improves inflammatory biomarkers in patients with nonalcoholic fatty liver disease. Nutr. Res. 2014, 34, 837–843. [Google Scholar] [CrossRef]
- Chachay, V.S.; Macdonald, G.A.; Martin, J.H.; Whitehead, J.P.; O’Moore-Sullivan, T.M.; Lee, P.; Franklin, M.; Klein, K.; Taylor, P.J.; Ferguson, M.; et al. Resveratrol Does Not Benefit Patients With Nonalcoholic Fatty Liver Disease. Clin. Gastroenterol. Hepatol. 2014, 12, 2092–2103.e6. [Google Scholar] [CrossRef]
- Dellinger, R.W.; Garcia, A.M.G.; Meyskens, F.L. Differences in the glucuronidation of resveratrol and pterostilbene: Altered enzyme specificity and potential gender differences. Drug Metab. Pharmacokinet. 2014, 29, 112–119. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Rothwell, J.A.; Achaintre, D.; Ferrari, P.; Boutron-Ruault, M.C.; Mancini, F.R.; Affret, A.; Kühn, T.; Katzke, V.; Boeing, H.; et al. Evaluation of urinary resveratrol as a biomarker of dietary resveratrol intake in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Br. J. Nutr. 2017, 117, 1596–1602. [Google Scholar] [CrossRef]
- Zamora-Ros, R.; Urpí-Sardà, M.; Lamuela-Raventós, R.M.; Estruch, R.; Martínez-González, M.Á.; Bulló, M.; Arós, F.; Cherubini, A.; Andres-Lacueva, C. Resveratrol metabolites in urine as a biomarker of wine intake in free-living subjects: The PREDIMED Study. Free Radic. Biol. Med. 2009, 46, 1562–1566. [Google Scholar] [CrossRef] [PubMed]
- Ortuño, J.; Covas, M.-I.; Farre, M.; Pujadas, M.; Fito, M.; Khymenets, O.; Andres-Lacueva, C.; Roset, P.; Joglar, J.; Lamuela-Raventós, R.M.; et al. Matrix effects on the bioavailability of resveratrol in humans. Food Chem. 2010, 120, 1123–1130. [Google Scholar] [CrossRef]
- Bugianesi, E.; Rosso, C.; Cortez-Pinto, H. How to diagnose NAFLD in 2016. J. Hepatol. 2016, 65, 643–644. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.A.; Buil-Cosiales, P.; Corella, D.; Bulló, M.; Fitó, M.; Vioque, J.; Romaguera, D.; Martínez, J.A.; Wärnberg, J.; López-Miranda, J.; et al. Cohort Profile: Design and methods of the PREDIMED-Plus randomized trial. Int. J. Epidemiol. 2019, 48, 387–388. [Google Scholar] [CrossRef]
- Salas-Salvadó, J.; Díaz-López, A.; Ruiz-Canela, M.; Basora, J.; Fitó, M.; Corella, D.; Serra-Majem, L.; Wärnberg, J.; Romaguera, D.; Estruch, R.; et al. Effect of a Lifestyle Intervention Program With Energy-Restricted Mediterranean Diet and Exercise on Weight Loss and Cardiovascular Risk Factors: One-Year Results of the PREDIMED-Plus Trial. Diabetes Care 2018, dc180836. [Google Scholar] [CrossRef]
- Alberti, K.G.M.M.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.; James, W.P.T.; Loria, C.M.; Smith, S.C. Harmonizing the Metabolic Syndrome. Circulation 2009, 120, 1640–1645. [Google Scholar] [CrossRef]
- Atroshenko, S.A.; Korolyov, I.A.; Didenko, N. Standards of Medical Care in Diabetes—2014. Diabetes Care 2014, 37, S14–S80. [Google Scholar] [CrossRef]
- Rosique-Esteban, N.; Díaz-López, A.; Martínez-González, M.A.; Corella, D.; Goday, A.; Martínez, J.A.; Romaguera, D.; Vioque, J.; Arós, F.; Garcia-Rios, A.; et al. Leisure-time physical activity, sedentary behaviors, sleep, and cardiometabolic risk factors at baseline in the PREDIMED-PLUS intervention trial: A cross-sectional analysis. PLoS ONE 2017, 12, e0172253. [Google Scholar] [CrossRef]
- Rosique-Esteban, N.; Babio, N.; Díaz-López, A.; Romaguera, D.; Alfredo Martínez, J.; Sanchez, V.M.; Schröder, H.; Estruch, R.; Vidal, J.; Buil-Cosiales, P.; et al. Leisure-time physical activity at moderate and high intensity is associated with parameters of body composition, muscle strength and sarcopenia in aged adults with obesity and metabolic syndrome from the PREDIMED-Plus study. Clin. Nutr. 2019, 38, 1324–1331. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, M.A.; Lopez-Fontana, C.; Varo, J.J.; Sanchez-Villegas, A.; Martinez, J.A. Validation of the Spanish version of the physical activity questionnaire used in the Nurses’ Health Study and the Health Professionals’ Follow-up Study. Public Health Nutr. 2005, 8, 920–927. [Google Scholar] [CrossRef]
- Fernández-Ballart, J.D.; Piñol, J.L.; Zazpe, I.; Corella, D.; Carrasco, P.; Toledo, E.; Perez-Bauer, M.; Martínez-González, M.Á.; Salas-Salvadó, J.; Martín-Moreno, J.M. Relative validity of a semi-quantitative food-frequency questionnaire in an elderly Mediterranean population of Spain. Br. J. Nutr. 2010, 103, 1808–1816. [Google Scholar] [CrossRef] [PubMed]
- Schröder, H.; Fitó, M.; Estruch, R.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Lamuela-Raventós, R.; Ros, E.; Salaverría, I.; Fiol, M.; et al. A Short Screener Is Valid for Assessing Mediterranean Diet Adherence among Older Spanish Men and Women. J. Nutr. 2011, 141, 1140–1145. [Google Scholar] [CrossRef] [PubMed]
- Sayón-Orea, C.; Razquin, C.; Bulló, M.; Corella, L.; Fitó, M.; Romaguera, R.; Vioque, J.; Alonso-Gómez, Ángel, M.; Wärnberg, J.; Martínez, J.A.; et al. Effect of a Nutritional and Behavioral Intervention on Energy-Reduced Mediterranean Diet Adherence Among Patients With Metabolic Syndrome. JAMA 2019, 322, 1486. [Google Scholar] [CrossRef] [PubMed]
- Bairaktari, E.T.; Seferiadis, K.I.; Elisaf, M.S. Evaluation of Methods for the Measurement of Low-Density Lipoprotein Cholesterol. J. Cardiovasc. Pharmacol. Ther. 2005, 10, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Feliciano, R.P.; Mecha, E.; Bronze, M.R.; Rodriguez-Mateos, A. Development and validation of a high-throughput micro solid-phase extraction method coupled with ultra-high-performance liquid chromatography-quadrupole time-of-flight mass spectrometry for rapid identification and quantification of phenolic metabolites in human plasma and urine. J. Chromatogr. A 2016, 1464, 21–31. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Song, Y.; Manson, J.E.; Tinker, L.; Howard, B.V.; Kuller, L.H.; Nathan, L.; Rifai, N.; Liu, S. Insulin sensitivity and insulin secretion determined by homeostasis model assessment and risk of diabetes in a multiethnic cohort of women: The women’s health initiative observational study. Diabetes Care 2007, 30, 1747–1752. [Google Scholar] [CrossRef]
- Silfen, M.E.; Manibo, A.M.; McMahon, D.J.; Levine, L.S.; Murphy, A.R.; Oberfield, S.E. Comparison of simple measures of insulin sensitivity in young girls with premature adrenarche: The fasting glucose to insulin ratio may be a simple and useful measure. J. Clin. Endocrinol. Metab. 2001, 86, 2863–2868. [Google Scholar] [CrossRef]
- Lee, J.-H.; Kim, D.; Kim, H.J.; Lee, C.-H.; Yang, J.I.; Kim, W.; Kim, Y.J.; Yoon, J.-H.; Cho, S.-H.; Sung, M.-W.; et al. Hepatic steatosis index: A simple screening tool reflecting nonalcoholic fatty liver disease. Dig. Liver Dis. 2010, 42, 503–508. [Google Scholar] [CrossRef]
- Bedogni, G.; Bellentani, S.; Miglioli, L.; Masutti, F.; Passalacqua, M.; Castiglione, A.; Tiribelli, C. The Fatty Liver Index: A simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006, 6, 33. [Google Scholar] [CrossRef]
- Prati, D.; Taioli, E.; Zanella, A.; Della Torre, E.; Butelli, S.; Del Vecchio, E.; Vianello, L.; Zanuso, F.; Mozzi, F.; Milani, S.; et al. Updated Definitions of Healthy Ranges for Serum Alanine Aminotransferase Levels. Ann. Intern. Med. 2002, 137, 1. [Google Scholar] [CrossRef] [PubMed]
- Aragon, G.; Younossi, Z.M. When and how to evaluate mildly elevated liver enzymes in apparently healthy patients. Clevel. Clin. J. Med. 2010, 77, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Nivukoski, U.; Niemelä, M.; Bloigu, A.; Bloigu, R.; Aalto, M.; Laatikainen, T.; Niemelä, O. Impacts of unfavourable lifestyle factors on biomarkers of liver function, inflammation and lipid status. PLoS ONE 2019, 14, e0218463. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
| Men | Women | p¶ | ||
|---|---|---|---|---|
| All | (n = 153) | (n = 113) | ||
| Age (years) | 65.8 (5.1) | 64.6 (5.4) | 67.5 (3.9) | <0.001 |
| BMI (kg/m2) | 32.2 (3.4) | 31.8 (3.1) | 32.8 (3.7) | 0.019 |
| Waist circumference (cm) | 107.3 (9.0) | 109.9 (8.5) | 103.9 (8.6) | <0.001 |
| VAT (g) | 2403.6 (888.9) | 2850.3 (826.2) | 1831.5 (589.7) | <0.001 |
| SBP (mmHg) | 144.8 (16.3) | 144.9 (15.8) | 144.6 (17.0) | 0.887 |
| DBP (mmHg) | 87.5 (8.5) | 87.9 (8.2) | 86.9 (8.9) | 0.338 |
| Type 2 diabetes, n (%) | 100 (37.6) | 61 (39.9) | 39(34.5) | 0.373 |
| Smoking, n (%) | <0.001 | |||
| Never | 105 (39.5) | 29 (18.9) | 76 (67.3) | |
| Former | 124 (46.6) | 97 (63.4) | 27 (23.9) | |
| Current | 37 (13.9) | 27 (17.7) | 10 (8.8) | |
| Lipid-lowering treatment | 93 (56.0) | 54 (57.5) | 39(54.2) | 0.673 |
| Any anti-diabetic treatment | 69 (25.9) | 43 (28.1) | 26 (23.0) | 0.349 |
| Glucose (mmol/L) | 6.7 (1.9) | 6.8 (2.1) | 6.5 (1.6) | 0.286 |
| HbA1c (%) | 6.1 (0.9) | 6.1 (1.0) | 6.1 (0.8) | 0.721 |
| Insulin (mU/L) | 14.0 (9.0) | 13.0 (7.1) | 15.6 (11.0) | 0.020 |
| HOMA-IR | 4.2 (3.2) | 3.9 (2.4) | 4.7 (4.2) | 0.056 |
| Total cholesterol (mg/dL) | 200.4 (36.5) | 192.2 (34.2) | 211.4 (36.9) | <0.001 |
| Triglycerides (mg/dL) | 148.3 (61.8) | 151.1 (69.0) | 144.6 (50.6) | 0.402 |
| HDL-c (mg/dL) | 45.8 (10.0) | 43.0 (8.9) | 49.5 (10.1) | <0.001 |
| LDL-c (mg/dL) | 125.8 (33.1) | 119.8 (31.3) | 133.6 (33.9) | <0.001 |
| VLDL-c (mg/dL) | 29.7 (12.4) | 30.2 (13.8) | 28.9 (10.1) | 0.402 |
| ALT (U/L) | 28.1 (20.6) | 31.2 (24.5) | 23.8 (12.3) | 0.004 |
| AST (U/L) | 23.7 (13.4) | 25.2 (16.3) | 21.8 (7.4) | 0.042 |
| GGT (U/L) | 42.1 (41.1) | 44.9 (41.5) | 38.5 (40.3) | 0.209 |
| ALT > ULN, n (%) * | 122 (46.0) | 55 (36.0) | 67 (59.8) | <0.001 |
| AST> ULN, n (%) * | 28 (10.5) | 12 (7.8) | 16 (14.2) | 0.097 |
| GGT> ULN, n (%) * | 51 (19.3) | 24 (15.9) | 27 (23.9) | 0.103 |
| FLI | 78.7 (15.1) | 80.4 (14.4) | 76.4 (15.9) | 0.035 |
| HSI | 43.0 (4.9) | 42.3 (4.7) | 44.0 (5.1) | 0.004 |
| Physical activity (MET-min/week) | 3099.9 (2757.8) | 3690 (3101.4) | 2301.0 (1954.8) | <0.001 |
| Men | Women | |||
|---|---|---|---|---|
| All | (n = 153) | (n = 114) | p¶ | |
| Total energy intake (kcal/d) | 2608.6 (539.1) | 2689.4 (534.5) | 2499.1 (528.0) | 0.004 |
| Carbohydrate intake (g/d) | 282.9 (75.1) | 287.0 (73.8) | 277.5 (76.9) | 0.310 |
| Protein intake (g/d) | 102.5 (22.9) | 101.3 (23.8) | 104.1 (21.8) | 0.330 |
| Fat intake (g/d) | 108.5 (27.1) | 110.9 (26.9) | 105.3 (27.2) | 0.097 |
| MUFAs (g/d) | 55.3 (14.3) | 56.4 (14.2) | 53.9 (14.5) | 0.171 |
| PUFAs (g/d) | 18.3 (7.2) | 19.1 (7.2) | 17.3 (7.1) | 0.049 |
| Linoleic (g/d) | 15.4 (6.9) | 16.1 (6.8) | 14.6 (7.0) | 0.075 |
| Linolenic (g/d) | 1.6 (0.8) | 1.6 (0.8) | 1.5 (0.8) | 0.317 |
| Omega-3 (g/d) | 0.9 (0.5) | 0.9 (0.5) | 0.9 (0.4) | 0.305 |
| Fiber (g/d) | 30.0 (9.8) | 29.3 (10.1) | 30.9 (9.3) | 0.175 |
| Total cholesterol (g/d) | 375.7 (110.8) | 382.4 (124.4) | 366.5 (88.9) | 0.248 |
| Total vegetables (g/d) | 333.0 (124.4) | 318.2 (123.0) | 353.0 (124.0) | 0.024 |
| Total fruits (g/d) | 423.9 (220.4) | 407.3 (221.6) | 446.3 (217.8) | 0.154 |
| Grapes intake (g/d) | 9.7 (21.8) | 12.6 (26.4) | 5.8 (12.3) | 0.012 |
| Cherries and plums (g/d) | 14.4 (19.4) | 14.9 (19.8) | 13.6 (18.9) | 0.599 |
| Nuts intake (g/d) | 15.1 (18.1) | 15.6 (18.1) | 14.3 (18.2) | 0.566 |
| Homemade fruit juice (mL/d) | 4.1 (23.6) | 5.6 (29.4) | 2.0 (11.5) | 0.209 |
| Fruit juice bottle (mL/d) | 12.4 (54.8) | 12.7 (52.9) | 12.1 (57.5) | 0.931 |
| Adherence to MedDiet (0–17 points) | 8.8 (2.5) | 8.8 (2.4) | 9.0 (2.5) | 0.484 |
| Alcohol consumption (g/d) | 12.9 (17.7) | 19.7 (19.8) | 3.5 (7.2) | <0.001 |
| Total red wine (g/d) | 61.4 (105.9) | 91.3 (120.8) | 20.8 (62.1) | <0.001 |
| Young red wine (g/d) | 56.7 (105.3) | 84.1 (121.3) | 19.6 (61.8) | <0.001 |
| Aged red wine (g/d) | 4.7 (25.6) | 7.2 (32.5) | 1.2 (9.5) | 0.056 |
| Rosé wine (g/d) | 10.4 (49.8) | 16.7 (64.2) | 1.9 (11.9) | 0.017 |
| Moscatel wine (g/d) | 0.5 (7.7) | 0.8 (10.1) | 0.06 (0.7) | 0.429 |
| White wine (g/d) | 7.4 (33.6) | 10.6 (42.4) | 3.1 (14.4) | 0.073 |
| trans-resveratrol-3-O-glucuronide (nmol/g creatinine) | 0.7 (1.6) | 0.7 (0.9) | 0.7 (2.2) | 0.973 |
| trans-resveratrol-4′-O-glucuronide (nmol/g creatinine) | 171.9 (375.8) | 143.7 (314.7) | 210.1 (444.1) | 0.154 |
| trans-resveratrol-3-O-sulfate (nmol/g creatinine) | 0.2 (0.5) | 0.2 (0.6) | 0.1 (0.2) | 0.023 |
| cis-resveratrol-3-O-glucuronide and cis-resveratrol-4′-O-glucuronide (nmol/g creatinine) | 2.0 (5.3) | 2.6 (6.0) | 1.1 (4.1) | 0.023 |
| Total Urine Resveratrol Metabolites (nmol/g Creatinine) | ||||
|---|---|---|---|---|
| T1 | T2 | T3 | p-Trend | |
| (≤4.6) | (>4.6 to 58.1) | (>58.1 to 2481.2) | ||
| n | 89 | 89 | 88 | |
| β Coefficient (95% IC) | β Coefficient (95% IC) | |||
| Glucose markers | ||||
| Glucose (mmol/L) | ||||
| Crude model | 0 REF. | 0.02 (−0.55, 0.60) | 0.11 (−0.46, 0.69) | 0.677 |
| Adjusted model | 0 REF. | 0.04 (−0.56, 0.63) | 0.04 (−0.55, 0.63) | 0.933 |
| HbA1c (%) | ||||
| Crude model | 0 REF. | −0.07 (−0.35, 0.21) | −0.01 (−0.28, 0.27) | 0.842 |
| Adjusted model | 0 REF. | −0.07 (−0.36, 0.22) | −0.04 (−0.32, 0.25) | 0.982 |
| Insulin sensitivity/resistance markers | ||||
| Insulin (mU/L) | ||||
| Crude model | 0 REF. | −1.23 (−3.94, 1.48) | −1.15 (−3.86, 1.55) | 0.623 |
| Adjusted model | 0 REF. | −0.22 (−2.87, 2.43) | −0.58 (−3.20, 2.03) | 0.672 |
| HOMA-IR | ||||
| Crude model | 0 REF. | −0.45 (−1.44, 0.54) | −0.46 (−1.44, 0.52) | 0.558 |
| Adjusted model | 0 REF. | −0.11 (−1.09, 0.87) | −0.31 (−1.28, 0.65) | 0.534 |
| HOMA-%B | ||||
| Crude model | 0 REF. | −15.19 (−37.11, 6.73) | −11.00 (−32.79, 10.79) | 0.682 |
| Adjusted model | 0 REF. | −11.07 (−32.44, 10.30) | −6.32 (−27.35, 14.71) | 0.904 |
| FGIR | ||||
| Crude model | 0 REF. | −0.12 (−0.30, 0.06) | −0.07 (−0.25, 0.11) | 0.859 |
| Adjusted model | 0 REF. | −0.14 (−0.33, 0.05) | −0.07 (−0.26, 0.11) | 0.922 |
| FIRI | ||||
| Crude model | 0 REF. | −0.40 (−1.29, 0.49) | −0.42 (−1.30, 0.47) | 0.558 |
| Adjusted model | 0 REF. | −0.10 (−0.98, 0.78) | −0.28 (−1.14, 0.58) | 0.534 |
| Total Urine Resveratrol Metabolites (nmol/g Creatinine) | ||||
|---|---|---|---|---|
| T1 | T2 | T3 | p-Trend | |
| (≤4.6) | (>4.6 to 58.1) | (>58.1 to 2481.2) | ||
| n | 89 | 89 | 88 | |
| β Coefficient (95% IC) | β Coefficient (95% IC) | |||
| Blood lipids | ||||
| Total cholesterol (mg/dL) | ||||
| Crude model | 0 REF. | −13.10 (−23.77, −2.43) | −13.26 (−23.96, −2.56) | 0.132 |
| Adjusted model | 0 REF. | −8.93 (−19.54, 1.68) | −11.67 (−22.21, −1.13) | 0.108 |
| LDL-c (mg/dL) | ||||
| Crude model | 0 REF. | −11.36 (−21.15, −1.57) | −8.21 (−18.02, 1.61) | 0.502 |
| Adjusted model | 0 REF. | −9.32 (−19.13, 0.48) | −7.77 (−17.57, 2.02) | 0.435 |
| HDL-c (mg/dL) | ||||
| Crude model | 0 REF. | 0.36 (−2.60, 3.32) | −0.38 (−3.36, 2.59) | 0.674 |
| Adjusted model | 0 REF. | 1.41 (−1.51, 4.32) | 0.27 (−2.63, 3.18) | 0.761 |
| Log triglyceride (mg/dL) | ||||
| Crude model | 0 REF. | −0.05 (−0.18, 0.07) | −0.14 (−0.26, −0.02) | 0.032 |
| Adjusted model | 0 REF. | −0.06 (−0.19, 0.07) | −0.15 (−0.28, −0.02) | 0.030 |
| VLDL-c (mg/dL) | ||||
| Crude model | 0 REF. | −1.48 (−5.11, 2.15) | −3.95 (−7.60, −0.30) | 0.043 |
| Adjusted model | 0 REF. | −1.70 (−5.47, 2.08) | −4.21 (−7.97, −0.46) | 0.039 |
| Log triglyceride/HDL ratio | ||||
| Crude model | 0 REF. | −0.07 (−0.23, 0.09) | −0.14 (−0.30, 0.02) | 0.122 |
| Adjusted model | 0 REF. | −0.10 (−0.26, 0.07) | −0.16 (−0.33, 0.002) | 0.106 |
| Total cholesterol/HDL ratio | ||||
| Crude model | 0 REF. | −0.38 (−0.69, −0.08) | −0.32 (−0.62, −0.01) | 0.304 |
| Adjusted model | 0 REF. | −0.39 (−0.70, −0.07) | −0.35 (−0.66, −0.03) | 0.241 |
| Total Urine Resveratrol Metabolites (nmol/g Creatinine) | ||||
|---|---|---|---|---|
| T1 | T2 | T3 | p-Trend | |
| (≤4.6) | (>4.6 to 58.1) | (>58.1 to 2481.2) | ||
| n | 89 | 89 | 88 | |
| β Coefficient (95% IC) | β Coefficient (95% IC) | |||
| Liver markers | ||||
| Log ALT (U/L) | ||||
| Crude model | 0 REF. | 0.03 (−0.12, 0.18) | −0.10 (−0.25, 0.05) | 0.074 |
| Adjusted model | 0 REF. | 0.03 (−0.11, 0.18) | −0.12 (−0.27, 0.02) | 0.028 |
| Log AST (U/L) | ||||
| Crude model | 0 REF. | 0.003 (−0.10, 0.10) | −0.09 (−0.19, 0.01) | 0.040 |
| Adjusted model | 0 REF. | −0.01 (−0.11, 0.09) | −0.12 (−0.22, −0.02) | 0.011 |
| Log GGT (U/L) | ||||
| Crude model | 0 REF. | 0.02 (−0.15, 0.20) | −0.23 (−0.41, −0.06) | 0.002 |
| Adjusted model | 0 REF. | 0.01 (−0.17, 0.19) | −0.24 (−0.42, −0.06) | 0.002 |
| HSI * | ||||
| Crude model | 0 REF. | −0.28 (−1.74, 1.18) | −0.59 (−2.45, 1.27) | 0.893 |
| Adjusted model | 0 REF. | 0.14 (−1.35, 1.63) | 0.11 (−1.37, 1.59) | 0.948 |
| FLI ¶ | ||||
| Crude model | 0 REF. | −0.97 (−5.46, 3.52) | −2.55 (−7.07, 1.98) | 0.294 |
| Adjusted model | 0 REF. | −1.39 (−5.97, 3.18) | −2.54 (−7.10, 2.02) | 0.346 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bullón-Vela, V.; Abete, I.; Zulet, M.A.; Xu, Y.; Martínez-González, M.A.; Sayón-Orea, C.; Ruiz-Canela, M.; Toledo, E.; Sánchez, V.M.; Estruch, R.; et al. Urinary Resveratrol Metabolites Output: Differential Associations with Cardiometabolic Markers and Liver Enzymes in House-Dwelling Subjects Featuring Metabolic Syndrome. Molecules 2020, 25, 4340. https://doi.org/10.3390/molecules25184340
Bullón-Vela V, Abete I, Zulet MA, Xu Y, Martínez-González MA, Sayón-Orea C, Ruiz-Canela M, Toledo E, Sánchez VM, Estruch R, et al. Urinary Resveratrol Metabolites Output: Differential Associations with Cardiometabolic Markers and Liver Enzymes in House-Dwelling Subjects Featuring Metabolic Syndrome. Molecules. 2020; 25(18):4340. https://doi.org/10.3390/molecules25184340
Chicago/Turabian StyleBullón-Vela, Vanessa, Itziar Abete, Maria Angeles Zulet, Yifan Xu, Miguel A. Martínez-González, Carmen Sayón-Orea, Miguel Ruiz-Canela, Estefanía Toledo, Vicente Martín Sánchez, Ramon Estruch, and et al. 2020. "Urinary Resveratrol Metabolites Output: Differential Associations with Cardiometabolic Markers and Liver Enzymes in House-Dwelling Subjects Featuring Metabolic Syndrome" Molecules 25, no. 18: 4340. https://doi.org/10.3390/molecules25184340
APA StyleBullón-Vela, V., Abete, I., Zulet, M. A., Xu, Y., Martínez-González, M. A., Sayón-Orea, C., Ruiz-Canela, M., Toledo, E., Sánchez, V. M., Estruch, R., Lamuela-Raventós, R. M., Almanza-Aguilera, E., Fitó, M., Salas-Salvadó, J., Díaz-López, A., Tinahones, F. J., Tur, J. A., Romaguera, D., Konieczna, J., ... Alfredo Martínez, J. (2020). Urinary Resveratrol Metabolites Output: Differential Associations with Cardiometabolic Markers and Liver Enzymes in House-Dwelling Subjects Featuring Metabolic Syndrome. Molecules, 25(18), 4340. https://doi.org/10.3390/molecules25184340












